Effects of Dietary α-Linolenic Acid Treatment and the Efficiency of Its Conversion to Eicosapentaenoic and Docosahexaenoic Acids in Obesity and Related Diseases
Abstract
:1. Introduction
2. Literature Search and Study Selection
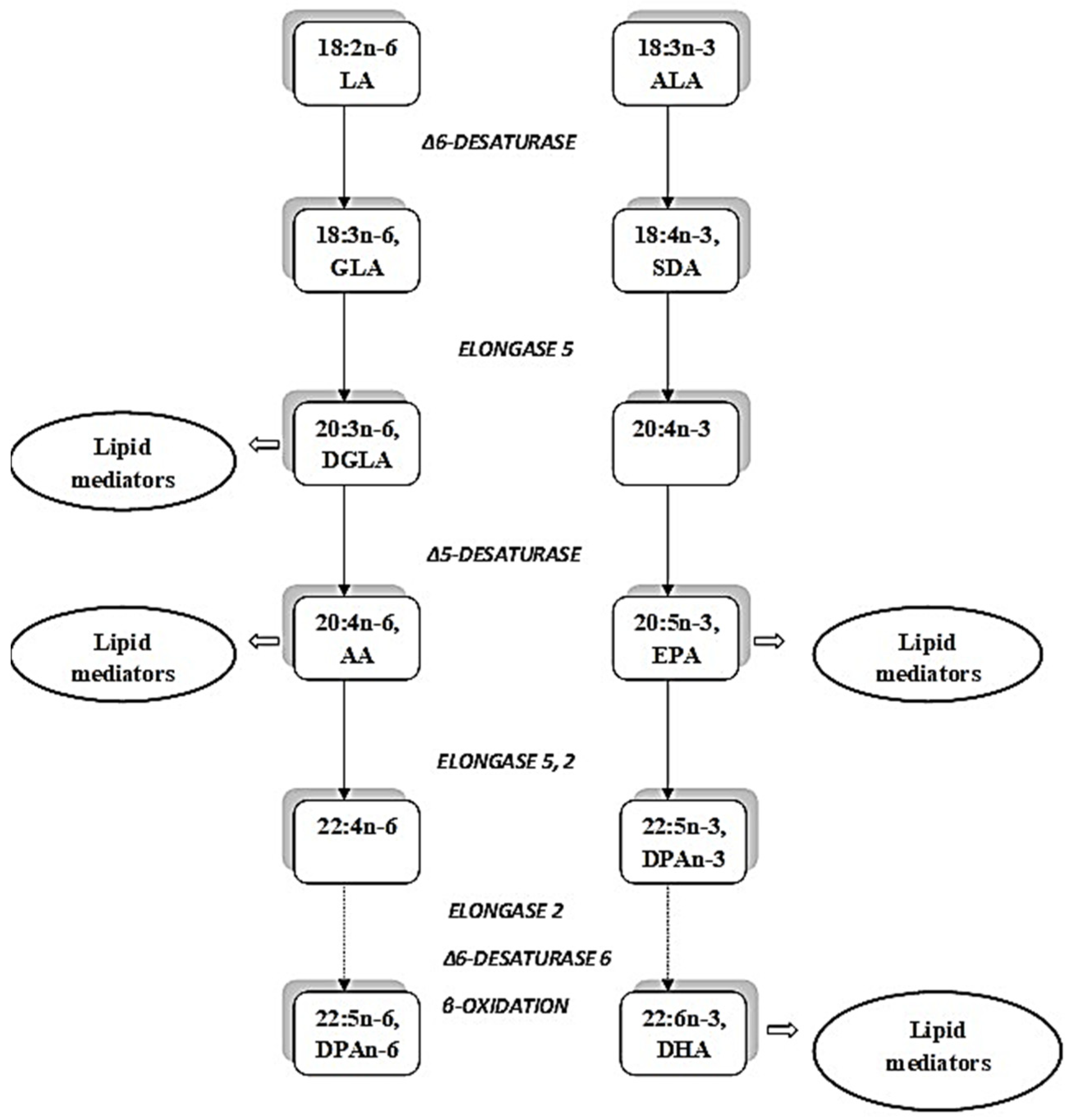
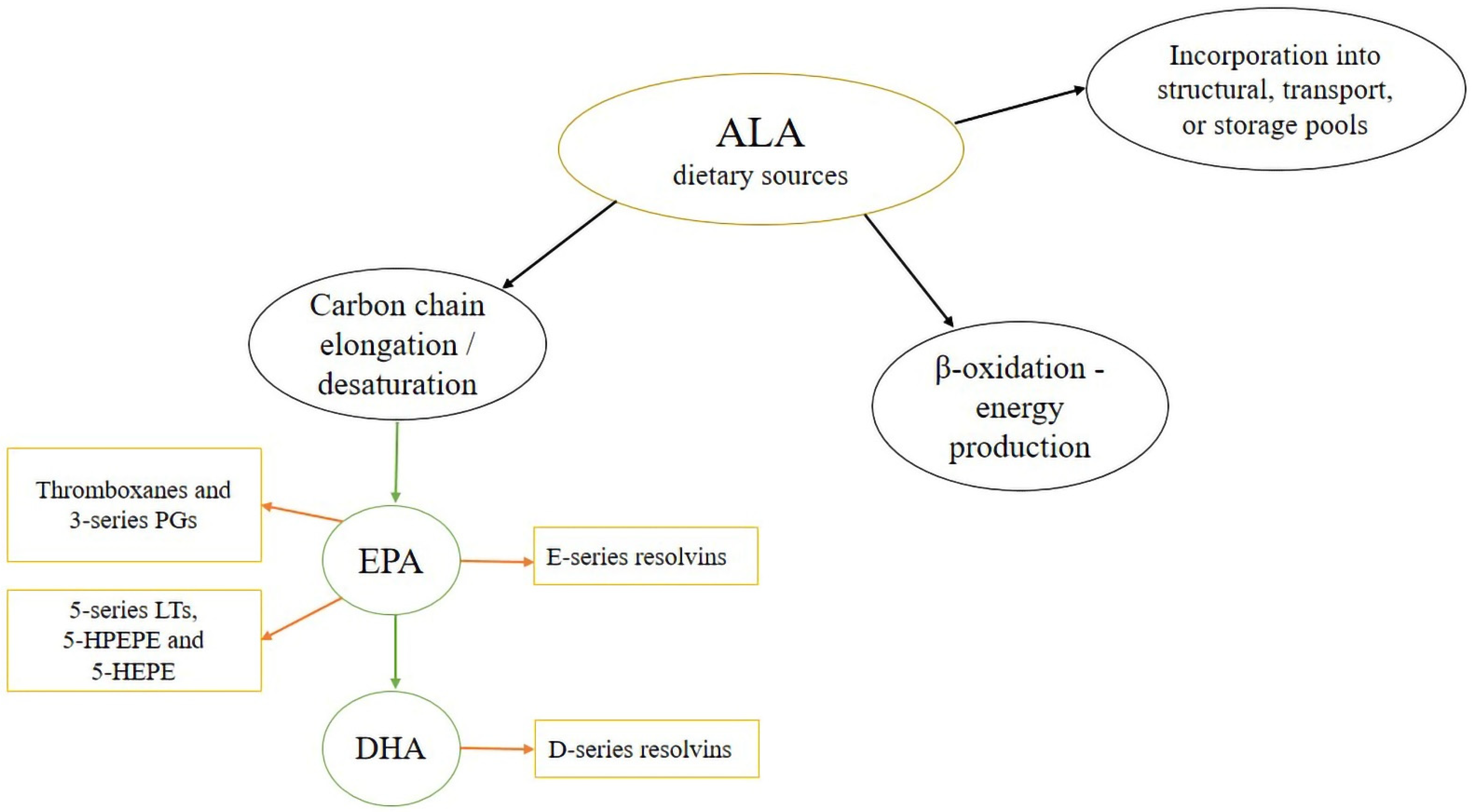
3. Bioconversion of ALA in an Elongase/Desaturase Pathway
4. Metabolic Conversion of Polyunsaturated Fatty Acids and Their Status in Obesity and Related Diseases
5. Effects of α-Linolenic Acid Treatment in Obesity, Recent Clinical Trials and Systematic Reviews
| Food Item | Reference | Study Population | Anthropometric Parameters | Blood Pressure | Serum Lipids | Insulin Sensitivity | Inflammation Parameters |
|---|---|---|---|---|---|---|---|
| Flaxseed | Yang et al., 2021 [77] | Dyslipidemic |  Body weight Body weight |  TC TC LDL-C LDL-C TG TG Apo-B Apo-B TC/HDL-C TC/HDL-C |  IL-6 IL-6 CRP CRP | ||
| Flaxseed | Masjedi et al., 2021 [79] | Dyslipidemic Overweight |  TC TC LDL-C LDL-C TG TG LDL-C LDL-C | ||||
| Flaxseed | Khalesi et al., 2015 [80] | Metabolic syndrome, Type 2 diabetes, Hyperlipidemic, Peripheral artery disease |  SBP SBP DBP DBP | ||||
| Flaxseed oil | Yang et al., 2021 [77] | Dyslipidemic |  IL-6 IL-6 | ||||
 hs-CRP hs-CRP | |||||||
| Walnut | Yang et al., 2020 [72] | Metabolic syndrome, |  Leptin levels Leptin levels | ||||
| Type 2 diabetes, |  Adiponectin levels
Adiponectin levels
| ||||||
| Overweight with hypertension and/or hypercholesterolemia, | No changes in FG, | ||||||
| Healthy | HbA1c and insulin levels | ||||||
| Walnut | Neale et al., 2020 [73] | Metabolic syndrome, Type 2 diabetes, Overweight | No changes in FG, HbA1c and insulin levels and HOMA-IR | ||||
| Walnut | Malmir et al., 2021 [74] | Abnormal glucose homoeostasis. | No changes in body weight and waist circumference |  FMD FMDNo changes in SBP and DBP |  LDL-C LDL-C | No changes in FG and HbA1c | |
| Walnut | Fang et al., 2020 [71] | Metabolic syndrome, Type 2 diabetes, Overweight and obese, Dyslipidemic, Cancer, Healthy | No changes in body weight, fat mass and waist circumference | ||||
| Walnut | Li et al., 2020 [75] | Metabolic syndrome, Chronic kidney disease, Diabetes or at high risk for diabetes, Moderate hypercholesterolemia, Healthy | No changes in SBP and DBP |
 meaning that the parameter is shown to be decreased and
meaning that the parameter is shown to be decreased and  meaning that the parameter is shown to be increased after flaxseed or walnuts dietary intake.
meaning that the parameter is shown to be increased after flaxseed or walnuts dietary intake.6. New Findings on Mechanism of ALA Action In Vivo and Its Anti-Lipolytic Effect
7. A New Approach to Weight Control Using Diacylglycerol Oils Enriched in ALA
8. The Effects of ALA Consumption on Blood Pressure and Vascular Parameters in Obesity
9. Effect of ALA on Obesity-Associated Non-Alcoholic Liver Disease
10. Comparison of Effects of ALA and DHA+EPA
11. Effect of ALA Consumption on FA Composition in Obesity
12. Effect of Stearidonic Acid Consumption on FA Composition
13. Conclusions
- Changes in fatty acid profiles in obesity
- 2.
- Effects of natural dietary sources (walnuts, flaxseed, and flaxseed oil) on features associated with obesity
- 3.
- Anti-lipolytic effect of ALA consumption
- 4.
- Role of ALA in NAFLD
- 5.
- Potential of DAG oils enriched in ALA to reduce adiposity
- 6.
- Comparison of ALA to EPA+DHA consumption effects in obesity
- 7.
- Effects of ALA treatment on n−3PUFA profiles
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, J.Y.; Kothapalli, K.S.; Brenna, J.T. Desaturase and elongase-limiting endogenous long-chain polyunsaturated fatty acid biosynthesis. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Kaur, N.; Chugh, V.; Gupta, A.K. Essential fatty acids as functional components of foods—A review. J. Food Sci. Technol. 2014, 51, 2289–2303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zatonski, W.; Campos, H.; Willett, W. Rapid declines in coronary heart disease mortality in Eastern Europe are associated with increased consumption of oils rich in alpha-linolenic acid. Eur. J. Epidemiol. 2008, 23, 3–10. [Google Scholar] [CrossRef]
- Wu, J.; Cho, E.; Giovannucci, E.L.; Rosner, B.A.; Sastry, S.M.; Schaumberg, D.A.; Willett, W.C. Dietary intake of α-linolenic acid and risk of age-related macular degeneration. Am. J. Clin. Nutr. 2017, 105, 1483–1492. [Google Scholar] [CrossRef]
- Young, I.E.; Parker, H.M.; Cook, R.L.; O’Dwyer, N.J.; Garg, M.L.; Steinbeck, K.S.; Cheng, H.L.; Donges, C.; Franklin, J.L.; O’Connor, H.T. Association between Obesity and Omega-3 Status in Healthy Young Women. Nutrients 2020, 20, 1480. [Google Scholar] [CrossRef]
- Barre, D.E. The role of consumption of alpha-linolenic, eicosapentaenoic and docosahexaenoic acids in human metabolic syndrome and type 2 diabetes–a mini-review. J. Oleo Sci. 2007, 56, 319–325. [Google Scholar] [CrossRef] [Green Version]
- Pawlosky, R.J.; Hibbeln, J.R.; Novotny, J.A.; Salem, N., Jr. Physiological compartmental analysis of alpha-linolenic acid metabolism in adult humans. J. Lipid Res. 2001, 42, 1257–1265. [Google Scholar] [CrossRef]
- Burdge, G. Alpha-linolenic acid metabolism in men and women: Nutritional and biological implications. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 137–144. [Google Scholar] [CrossRef]
- Hruby, A.; Hu, F.B. The Epidemiology of Obesity: A Big Picture. Pharmacoeconomics 2015, 33, 73–89. [Google Scholar] [CrossRef]
- D’Angelo, S.; Motti, M.L.; Meccariello, R. ω-3 and ω-6 Polyunsaturated Fatty Acids, Obesity and Cancer. Nutrients 2020, 12, 2751. [Google Scholar] [CrossRef]
- Ristic-Medic, D.; Vucic, V.; Takic, M.; Karadzic, I.; Glibetic, M. Polyunsaturated fatty acids in health and disease. J. Serb. Chem. Soc. 2013, 78, 1269–1289. [Google Scholar] [CrossRef]
- Klop, B.; Elte, J.W.; Cabezas, M.C. Dyslipidemia in obesity: Mechanisms and potential targets. Nutrients 2013, 12, 1218–1240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wondmkun, Y. Obesity, Insulin Resistance, and Type 2 Diabetes: Associations and Therapeutic Implications. Diabetes Metab. Syndr. Obes. 2020, 13, 3611–3616. [Google Scholar] [CrossRef]
- Kuzminova, N.V.; Gribenyuk, O.V.; Osovska, N.Y.; Knyazkova, I.I. Arterial hypertension, obesity and non-alcoholic fatty liver disease: Is there any connection? Arter. Hypertens. 2016, 20, 216–227. [Google Scholar] [CrossRef]
- Telle-Hansen, V.H.; Christensen, J.J.; Formo, G.A.; Holven, K.B.; Ulven, S.M. A comprehensive metabolic profiling of the metabolically healthy obesity phenotype. Lipids Health Dis. 2020, 19, 90. [Google Scholar] [CrossRef] [PubMed]
- Arsic, A.; Takic, M.; Kojadinovic, M.; Petrovic, S.; Paunovic, M.; Vucic, V.; Medic, D.R. Metabolically healthy obesity: Is there a link with polyunsaturated fatty acid intake and status? Can. J. Physiol. Pharmacol. 2021, 99, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Naghshi, S.; Aune, D.; Beyene, J.; Mobarak, S.; Asadi, M.; Sadeghi, O. Dietary intake and biomarkers of alpha linolenic acid and risk of all cause, cardiovascular, and cancer mortality: Systematic review and dose-response meta-analysis of cohort studies. BMJ 2021, 13, 2213. [Google Scholar] [CrossRef]
- Yuan, Q.; Xie, F.; Huang, W.; Hu, M.; Yan, Q.; Chen, Z.; Zheng, Y.; Liu, L. The review of alpha-linolenic acid: Sources, metabolism, and pharmacology. Phytother. Res. 2022, 36, 164–188. [Google Scholar] [CrossRef]
- Shayan, M.; Kamalian, S.; Sahebkar, A.; Tayarani-Najaran, Z. Flaxseed for Health and Disease: Review of Clinical Trials. Comb. Chem. High. Throughput. Screen 2020, 23, 699–722. [Google Scholar] [CrossRef]
- Fabbrini, E.; Sullivan, S.; Klein, S. Obesity and nonalcoholic fatty liver disease: Biochemical, metabolic, and clinical implications. Hepatology 2010, 51, 679–689. [Google Scholar] [CrossRef]
- Ahmed, I.A.; Mikail, M.A.; Mustafa, M.R.; Ibrahim, M.; Othman, R. Lifestyle interventions for non-alcoholic fatty liver disease. Saudi J. Biol. Sci. 2019, 26, 1519–1524. [Google Scholar] [CrossRef] [PubMed]
- Fekete, K.; Györei, E.; Lohner, S.; Verduci, E.; Agostoni, C.; Decsi, T. Long-chain polyunsaturated fatty acid status in obesity: A systematic review and meta-analysis. Obes. Rev. 2015, 16, 488–497. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 2016, 2, 128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calder, P.C. Omega-3 polyunsaturated fatty acids and inflammatory processes: Nutrition or pharmacology? Br. J. Clin. Pharmacol. 2013, 75, 645–662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walker, C.G.; Browning, L.M.; Mander, A.P.; Madden, J.; West, A.L.; Calder, P.C.; Jebb, S.A. Age and sex differences in the incorporation of EPA and DHA into plasma fractions, cells and adipose tissue in humans. Br. J. Nutr. 2014, 111, 679–689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greupner, T.; Kutzner, L.; Nolte, F.; Strangmann, A.; Kohrs, H.; Hahn, A.; Schebb, N.H.; Schuchardt, J.P. Effects of a 12-week high-α-linolenic acid intervention on EPA and DHA concentrations in red blood cells and plasma oxylipin pattern in subjects with a low EPA and DHA status. Food Funct. 2018, 1, 1587–1600. [Google Scholar] [CrossRef] [Green Version]
- Petrović-Oggiano, G.; Debeljak-Martačić, J.; Ranković, S.; Pokimica, B.; Mirić, A.; Glibetić, M.; Popović, T. The effect of walnut consumption on n−3 fatty acid profile of healthy people living in a non-Mediterranean West Balkan country, a small scale randomized study. Nutrients 2020, 12, 192. [Google Scholar] [CrossRef] [Green Version]
- Egert, S.; Kannenberg, F.; Somoza, V.; Erbersdobler, H.F.; Wahrburg, U. Dietary alpha-linolenic acid, EPA, and DHA have differential effects on LDL fatty acid composition but similar effects on serum lipid profiles in normolipidemic humans. J. Nutr. 2009, 139, 861–868. [Google Scholar] [CrossRef] [Green Version]
- Egert, S.; Somoza, V.; Kannenberg, F.; Fobker, M.; Krome, K.; Erbersdobler, H.F.; Wahrburg, U. Influence of three rapeseed oil-rich diets, fortified with alpha-linolenic acid, eicosapentaenoic acid or docosahexaenoic acid on the composition and oxidizability of low-density lipoproteins: Results of a controlled study in healthy volunteers. Eur. J. Clin. Nutr. 2007, 61, 314–325. [Google Scholar] [CrossRef] [Green Version]
- Brenna, J.T.; Salem, N., Jr.; Sinclair, A.J.; Cunnane, S.C. International Society for the Study of Fatty Acids and Lipids, ISSFAL. alpha-Linolenic acid supplementation and conversion to n−3 long-chain polyunsaturated fatty acids in humans. Prostaglandins Leukot Essent Fat. Acids 2009, 80, 85–91. [Google Scholar] [CrossRef]
- Silva, L.A.; Verneque, B.J.F.; Mota, A.P.L.; Duarte, C.K. Chia seed (Salvia hispanica L.) consumption and lipid profile: A systematic review and meta-analysis. Food Funct. 2021, 4, 8835–8849. [Google Scholar] [CrossRef] [PubMed]
- Serbian Food Composition Database. Available online: http://104.155.19.23/capnutra (accessed on 28 March 2022).
- Turkish Food Composition Database. Available online: http://www.turkomp.gov.tr/main (accessed on 28 March 2022).
- U.S. Department of Agriculture, Agricultural Research Service. Food Data Central. 2019. Available online: fdc.nal.usda.gov (accessed on 28 March 2022).
- Lee, J.; Rodriguez, J.P.; Kim, Y.J.; Lee, M.H.; Cho, E.J.; Lee, S. Fatty acid content in perilla cultivars and commercial oils determined by GC analysis. Nat. Prod. Sci. 2016, 22, 259–262. [Google Scholar] [CrossRef] [Green Version]
- Ando, Y.; Saito, S.; Miura, H.; Osaki, N.; Katsuragi, Y. Consumption of alpha-linolenic acid-enriched diacylglycerol induces increase in dietary fat oxidation compared with alpha-linolenic acid-enriched triacylglycerol: A randomized, double-blind trial. Nutr. Res. 2017, 48, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Trumbo, P.; Schlicker, S.; Yates, A.A.; Poos, M. Food and Nutrition Board of the Institute of Medicine, The National Academies. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein and amino acids. J. Am. Diet. Assoc. 2002, 102, 1621–1630, Erratum in J. Am. Diet. Assoc. 2003, 103, 563. [Google Scholar] [CrossRef]
- Hodson, L.; Skeaff, C.M.; Fielding, B.A. Fatty acid composition of adipose tissue and blood in humans and its use as a biomarker of dietary intake. Prog. Lipid Res. 2008, 47, 348–380. [Google Scholar] [CrossRef] [PubMed]
- Alsharari, Z.D.; Risérus, U.; Leander, K.; Sjögren, P.; Carlsson, A.C.; Vikström, M.; Laguzzi, F.; Gigante, B.; Cederholm, T.; De Faire, U.; et al. Serum Fatty Acids, Desaturase Activities and Abdominal Obesity—A Population-Based Study of 60-Year Old Men and Women. PLoS ONE 2017, 12, e0170684. [Google Scholar] [CrossRef]
- Murff, H.J.; Edwards, T.L. Endogenous Production of Long-Chain Polyunsaturated Fatty Acids and Metabolic Disease Risk. Curr. Cardiovasc. Risk Rep. 2014, 1, 418. [Google Scholar] [CrossRef] [Green Version]
- Del Pozo, M.D.P.; Lope, V.; Criado-Navarro, I.; Pastor-Barriuso, R.; Fernández de Larrea, N.; Ruiz, E.; Castelló, A.; Lucas, P.; Sierra, Á.; Romieu, I.; et al. Serum Phospholipid Fatty Acids Levels, Anthropometric Variables and Adiposity in Spanish Premenopausal Women. Nutrients 2020, 25, 1895. [Google Scholar] [CrossRef]
- Aglago, E.K.; Biessy, C.; Torres-Mejía, G.; Angeles-Llerenas, A.; Gunter, M.J.; Romieu, I.; Chajès, V. Association between serum phospholipid fatty acid levels and adiposity in Mexican women. J. Lipid Res. 2017, 58, 1462–1470. [Google Scholar] [CrossRef] [Green Version]
- Yammine, S.G.; Naja, F.; Tamim, H.; Nasrallah, M.; Biessy, C.; Aglago, E.K.; Matta, M.; Romieu, I.; Gunter, M.J.; Nasreddine, L.; et al. Association between Serum Phospholipid Fatty Acid Levels and Adiposity among Lebanese Adults: A Cross-Sectional Study. Nutrients 2018, 25, 1371. [Google Scholar] [CrossRef] [Green Version]
- Warensjö, E.; Ohrvall, M.; Vessby, B. Fatty acid composition and estimated desaturase activities are associated with obesity and lifestyle variables in men and women. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Petersson, H.; Arnlöv, J.; Zethelius, B.; Risérus, U. Serum fatty acid composition and insulin resistance are independently associated with liver fat markers in elderly men. Diabetes Res. Clin. Pract. 2010, 87, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Lee, A.; Yoo, H.J.; Kim, M.; Kim, M.; Shin, D.Y.; Lee, J.H. Association between increased visceral fat area and alterations in plasma fatty acid profile in overweight subjects: A cross-sectional study. Lipids Health Dis. 2017, 19, 248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parry, S.A.; Rosqvist, F.; Peters, S.; Young, R.K.; Cornfield, T.; Dyson, P.; Hodson, L. The influence of nutritional state on the fatty acid composition of circulating lipid fractions: Implications for their use as biomarkers of dietary fat intake. Ups. J. Med. Sci. 2021, 13, 26. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wu, L.; Chen, J.; Dong, L.; Chen, C.; Wen, Z.; Hu, J.; Fleming, I.; Wang, D.W. Metabolism pathways of arachidonic acids: Mechanisms and potential therapeutic targets. Sig. Transduct. Target Ther. 2021, 6, 94. [Google Scholar] [CrossRef]
- Frayn, K.; Karpe, F. Regulation of human subcutaneous adipose tissue blood flow. Int. J. Obes. 2014, 38, 1019–1026. [Google Scholar] [CrossRef]
- Sam, S.; Mazzone, T. Adipose tissue changes in obesity and the impact on metabolic function. Transl. Res. 2014, 164, 284–292. [Google Scholar] [CrossRef]
- Fuster, J.J.; Ouchi, N.; Gokce, N.; Walsh, K. Obesity-Induced Changes in Adipose Tissue Microenvironment and Their Impact on Cardiovascular Disease. Circ. Res. 2016, 118, 1786–1807. [Google Scholar] [CrossRef] [Green Version]
- Xiao, Y.; Liu, D.; Cline, M.A.; Gilbert, E.R. Chronic stress, epigenetics, and adipose tissue metabolism in the obese state. Nutr. Metab. 2020, 17, 88. [Google Scholar] [CrossRef]
- Chait, A.; den Hartigh, L.J. Adipose Tissue Distribution, Inflammation and Its Metabolic Consequences, Including Diabetes and Cardiovascular Disease. Front. Cardiovasc. Med. 2020, 25, 22. [Google Scholar] [CrossRef] [Green Version]
- Sjögren, P.; Sierra-Johnson, J.; Gertow, K.; Rosell, M.; Vessby, B.; de Faire, U.; Hamsten, A.; Hellenius, M.L.; Fisher, R.M. Fatty acid desaturases in human adipose tissue: Relationships between gene expression, desaturation indexes and insulin resistance. Diabetologia 2008, 51, 328–335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warensjö, E.; Rosell, M.; Hellenius, M.L.; Vessby, B.; De Faire, U.; Risérus, U. Associations between estimated fatty acid desaturase activities in serum lipids and adipose tissue in humans: Links to obesity and insulin resistance. Lipids Health Dis. 2009, 8, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Savva, S.C.; Chadjigeorgiou, C.; Hatzis, C.; Kyriakakis, M.; Tsimbinos, G.; Tornaritis, M.; Kafatos, A. Association of adipose tissue arachidonic acid content with BMI and overweight status in children from Cyprus and Crete. Br. J. Nutr. 2004, 91, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.S.; Baylin, A.; Campos, H. Adipose tissue arachidonic acid and the metabolic syndrome in Costa Rican adults. Clin. Nutr. 2007, 26, 474–482. [Google Scholar] [CrossRef] [Green Version]
- Pietiläinen, K.H.; Róg, T.; Seppänen-Laakso, T.; Virtue, S.; Gopalacharyulu, P.; Tang, J.; Rodriguez-Cuenca, S.; Maciejewski, A.; Naukkarinen, J.; Ruskeepää, A.-L.; et al. Association of lipidome remodeling in the adipocyte membrane with acquired obesity in humans. PLoS Biol. 2011, 9, e1000623. [Google Scholar] [CrossRef] [Green Version]
- Ailhaud, G.; Massiera, F.; Weill, P.; Legrand, P.; Alessandri, J.M.; Guesnet, P. Temporal changes in dietary fats: Role of n−6 polyunsaturated fatty acids in excessive adipose tissue development and relationship to obesity. Prog. Lipid Res. 2006, 45, 203–236. [Google Scholar] [CrossRef]
- Ellulu, M.S.; Patimah, I.; Khaza’ai, H.; Rahmat, A.; Abed, Y. Obesity and inflammation: The linking mechanism and the complications. Arch. Med. Sci. 2017, 13, 851–863. [Google Scholar] [CrossRef]
- Fisk, H.L.; Childs, C.E.; Miles, E.A.; Ayres, R.; Noakes, P.S.; Paras-Chavez, C.; Kuda, O.; Kopecký, J.; Antoun, E.; Lillycrop, K.A.; et al. Modification of subcutaneous white adipose tissue inflammation by omega-3 fatty acids is limited in human obesity-a double blind, randomised clinical trial. EBioMedicine 2022, 77, 103909. [Google Scholar] [CrossRef]
- Nono Nankam, P.A.; Mendham, A.E.; van Jaarsveld, P.J.; Adams, K.; Fortuin-de Smidt, M.C.; Clamp, L.; Blüher, M.; Goedecke, J.H. Exercise Training Alters Red Blood Cell Fatty Acid Desaturase Indices and Adipose Tissue Fatty Acid Profile in African Women with Obesity. Obesity 2020, 28, 1456–1466. [Google Scholar] [CrossRef]
- Huang, C.W.; Chien, Y.S.; Chen, Y.J.; Ajuwon, K.M.; Mersmann, H.M.; Ding, S.T. Role of n−3 Polyunsaturated Fatty Acids in Ameliorating the Obesity-Induced Metabolic Syndrome in Animal Models and Humans. Int. J. Mol. Sci. 2016, 17, 1689. [Google Scholar] [CrossRef] [Green Version]
- Cornier, M.A.; Dabelea, D.; Hernandez, T.L.; Lindstrom, R.C.; Steig, A.J.; Stob, N.R.; Van Pelt, R.E.; Wang, H.; Eckel, R.H. The metabolic syndrome. Endocr. Rev. 2008, 29, 777–822. [Google Scholar] [CrossRef] [PubMed]
- Wendland, E.; Farmer, A.; Glasziou, P.; Neil, A. Effect of alpha linolenic acid on cardiovascular risk markers: A systematic review. Heart 2006, 92, 166–169. [Google Scholar] [CrossRef] [Green Version]
- Su, H.; Liu, R.; Chang, M.; Huang, J.; Jin, Q.; Wang, X. Effect of dietary alpha-linolenic acid on blood inflammatory markers: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Nutr. 2018, 57, 877–891. [Google Scholar] [CrossRef] [PubMed]
- Balk, E.M.; Lichtenstein, A.H.; Chung, M.; Kupelnick, B.; Chew, P.; Lau, J. Effects of omega-3 fatty acids on serum markers of cardiovascular disease risk: A systematic review. Atherosclerosis 2006, 189, 19–30. [Google Scholar] [CrossRef] [PubMed]
- de Goede, J.; Verschuren, W.M.; Boer, J.M.; Kromhout, D.; Geleijnse, J.M. Alpha-linolenic acid intake and 10-year incidence of coronary heart disease and stro ke in 20,000 middle-aged men and women in the Netherlands. PLoS ONE 2011, 25, e17967. [Google Scholar]
- Geleijnse, J.M.; de Goede, J.; Brouwer, I.A. Alpha-linolenic acid: Is it essential to cardiovascular health? Curr. Atheroscler. Rep. 2010, 12, 359–367. [Google Scholar] [CrossRef] [Green Version]
- Egert, S.; Stehle, P. Impact of n−3 fatty acids on endothelial function: Results from human interventions studies. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 121–131. [Google Scholar] [CrossRef]
- Fang, Z.; Dang, M.; Zhang, W.; Wang, Y.; Kord-Varkaneh, H.; Nazary-Vannani, A.; O’Santos, H.; Tan, S.C.; Clark, C.; Zanghelini, F.; et al. Effects of walnut intake on anthropometric characteristics: A systematic review and dose-response meta-analysis of randomized controlled trials. Complement Ther. Med. 2020, 50, 102395. [Google Scholar] [CrossRef]
- Yang, L.; Guo, Z.; Qi, S.; Fang, T.; Zhu, H.; Santos, H.O.; Khani, V.; Wong, C.H.; Qiu, Z. Walnut intake may increase circulating adiponectin and leptin levels but does not improve glycemic biomarkers: A systematic review and meta-analysis of randomized clinical trials. Complement Ther. Med. 2020, 52, 102505. [Google Scholar] [CrossRef]
- Neale, E.P.; Guan, V.; Tapsell, L.C.; Probst, Y.C. Effect of walnut consumption on markers of blood glucose control: A systematic review and meta-analysis. Br. J. Nutr. 2020, 14, 641–653. [Google Scholar] [CrossRef]
- Malmir, H.; Larijani, B.; Esmaillzadeh, A. The effect of walnut consumption on cardiometabolic profiles of individuals with abnormal glucose homoeostasis: A systematic review and meta-analysis of clinical trials. Br. J. Nutr. 2021, 2, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Jiang, B.; Santos, H.O.; Santos, D.; Singh, A.; Wang, L. Effects of walnut intake on blood pressure: A systematic review and meta-analysis of randomized controlled trials. Phytother. Res. 2020, 34, 2921–2931. [Google Scholar] [CrossRef] [PubMed]
- Neale, E.P.; Tapsell, L.C.; Guan, V.; Batterham, M.J. The effect of nut consumption on markers of inflammation and endothelial function: A systematic review and meta-analysis of randomised controlled trials. BMJ Open 2017, 22, e016863. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Xia, H.; Wan, M.; Lu, Y.; Xu, D.; Yang, X.; Yang, L.; Sun, G. Comparisons of the effects of different flaxseed products consumption on lipid profiles, inflammatory cytokines and anthropometric indices in patients with dyslipidemia related diseases: Systematic review and a dose-response meta-analysis of randomized controlled trials. Nutr. Metab. 2021, 11, 91. [Google Scholar]
- Tamtaji, O.R.; Milajerdi, A.; Reiner, Ž.; Dadgostar, E.; Amirani, E.; Asemi, Z.; Mirsafaei, L.; Mansournia, M.A.; Dana, P.M.; Sadoughi, F.; et al. Effects of flaxseed oil supplementation on biomarkers of inflammation and oxidative stress in patients with metabolic syndrome and related disorders: A systematic review and meta-analysis of randomized controlled trials. Clin. Nutr. ESPEN 2020, 40, 27–33. [Google Scholar] [CrossRef]
- Sadat Masjedi, M.; Mohammadi Pour, P.; Shokoohinia, Y.; Asgary, S. Effects of Flaxseed on Blood Lipids in Healthy and Dyslipidemic Subjects: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Curr. Probl. Cardiol. 2021, 16, 100931. [Google Scholar] [CrossRef]
- Khalesi, S.; Irwin, C.; Schubert, M. Flaxseed consumption may reduce blood pressure: A systematic review and meta-analysis of controlled trials. J. Nutr. 2015, 145, 758–765. [Google Scholar] [CrossRef]
- Zhao, N.; Wang, L.; Guo, N. α-Linolenic acid increases the G0/G1 switch gene 2 mRNA expression in peripheral blood mononuclear cells from obese patients: A pilot study. Lipids Health Dis. 2016, 15, 36. [Google Scholar] [CrossRef] [Green Version]
- Puglisi, M.J.; Hasty, A.H.; Saraswathi, V. The role of adipose tissue in mediating the beneficial effects of dietary fish oil. J. Nutr. Biochem. 2011, 22, 101–108. [Google Scholar] [CrossRef] [Green Version]
- Zandbergen, F.; Mandard, S.; Escher, P.; Tan, N.S.; Patsouris, D.; Jatkoe, T.; Rojas-Caro, S.; Madore, S.; Wahli, W.; Tafuri, S.; et al. The G0/G1 switch gene 2 is a novel PPAR target gene. Biochem. J. 2005, 1, 313–324. [Google Scholar] [CrossRef]
- Im, D.S. Omega-3 fatty acids in anti-inflammation (pro-resolution) and GPCRs. Prog. Lipid Res. 2012, 51, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Edwards, I.J.; O’Flaherty, J.T. Omega-3 Fatty Acids and PPARgamma in Cancer. PPAR Res. 2008, 2008, 358052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vijaimohan, K.; Jainu, M.; Sabitha, K.E.; Subramaniyam, S.; Anandhan, C.; Devi, C.S. Beneficial effects of alpha linolenic acid rich flaxseed oil on growth performance and hepatic cholesterol metabolism in high fat diet fed rats. Life Sci. 2006, 79, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.J.; Jew, S.; AbuMweis, S. The effect of dietary oleic, linoleic, and linolenic acids on fat oxidation and energy expenditure in healthy men. Metabolism 2008, 57, 1198–1203. [Google Scholar] [CrossRef]
- Gillingham, L.G.; Robinson, K.S.; Jones, P.J. Effect of high-oleic canola and flaxseed oils on energy expenditure and body composition in hypercholesterolemic subjects. Metabolism 2012, 61, 1598–1605. [Google Scholar] [CrossRef]
- Tomonobu, K.; Hase, T.; Tokimitsu, I. Dietary diacylglycerol in a typical meal suppresses postprandial increases in serum lipid levels compared with dietary triacylglycerol. Nutrition 2006, 22, 128–135. [Google Scholar] [CrossRef]
- Saito, S.; Yamaguchi, T.; Shoji, K.; Hibi, M.; Sugita, T.; Takase, H. Effect of low concentration of diacylglycerol on mildly postprandial hypertriglyceridemia. Atherosclerosis 2010, 213, 539–544. [Google Scholar] [CrossRef]
- Saito, S.; Mori, A.; Osaki, N.; Katsuragi, Y. Diacylglycerol Enhances the Effects of Alpha-Linolenic Acid against Visceral Fat: A Double-Blind Randomized Controlled Trial. Obesity 2017, 25, 1667–1675. [Google Scholar] [CrossRef] [Green Version]
- Saito, S.; Fukuhara, I.; Osaki, N.; Nakamura, H.; Katsuragi, Y. Consumption of alpha-Linolenic Acid-enriched Diacylglycerol Reduces Visceral Fat Area in Overweight and Obese Subjects: A Randomized, Double-blind Controlled, Parallel-group Designed Trial. J. Oleo. Sci. 2016, 65, 603–611. [Google Scholar] [CrossRef] [Green Version]
- Pieters, D.J.; Zock, P.L.; Fuchs, D.; Mensink, R.P. Effect of α-linolenic acid on 24-h ambulatory blood pressure in untreated high-normal and stage I hypertensive subjects. Br. J. Nutr. 2019, 121, 155–163. [Google Scholar] [CrossRef]
- Joris, P.J.; Draijer, R.; Fuchs, D.; Mensink, R.P. Effect of α-linolenic acid on vascular function and metabolic risk markers during the fasting and postprandial phase: A randomized placebo-controlled trial in untreated (pre-)hypertensive individuals. Clin. Nutr. 2020, 39, 2413–2419. [Google Scholar] [CrossRef] [PubMed]
- Antraco, V.J.; Hirata, B.K.S.; de Jesus Simão, J.; Cruz, M.M.; da Silva, V.S.; da Cunha de Sá, R.D.C.; Abdala, F.M.; Armelin-Correa, L.; Alonso-Vale, M.I.C. Omega-3 Polyunsaturated Fatty Acids Prevent Nonalcoholic Steatohepatitis (NASH) and Stimulate Adipogenesis. Nutrients 2021, 15, 622. [Google Scholar] [CrossRef] [PubMed]
- Herrera Vielma, F.; Valenzuela, R.; Videla, L.A.; Zúñiga-Hernández, J. N-3 Polyunsaturated Fatty Acids and Their Lipid Mediators as A Potential Immune-Nutritional Intervention: A Molecular and Clinical View in Hepatic Disease and Other Non-Communicable Illnesses. Nutrients 2021, 26, 3384. [Google Scholar] [CrossRef]
- Hasegawa, M.; Endo, Y.; Iwamoto, Y. Intake of n−3 polyunsaturated fatty acids and non-alcoholic fatty liver disease: A cross-sectional study in Japanese men and women. Eur. J. Clin. Nutr. 2010, 64, 1179–1185. [Google Scholar]
- Ristić-Medić, D.; Takić, M.; Vučić, V.; Kandić, D.; Kostić, N.; Glibetić, M. Abnormalities in the serum phospholipids fatty acid profile in patients with alcoholic liver cirrhosis–a pilot study. J. Clin. Biochem. Nutr. 2013, 53, 49–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kruse, M.; Kemper, M.; Gancheva, S.; Osterhoff, M.; Dannenberger, D.; Markgraf, D.; Machann, J.; Hierholzer, J.; Roden, M.; Pfeiffer, A.F.H. Dietary Rapeseed Oil Supplementation Reduces Hepatic Steatosis in Obese Men-A Randomized Controlled Trial. Mol. Nutr. Food Res. 2020, 64, e2000419. [Google Scholar] [CrossRef] [PubMed]
- Hagström, H.; Stål, P. Treatment of NAFLD with intermittent calorie restriction or low-carb high-fat diet—A randomised controlled trial. JHEP Rep. 2021, 17, 100256. [Google Scholar]
- Zelber-Sagi, S.; Nitzan-Kaluski, D.; Goldsmith, R.; Webb, M.; Blendis, L.; Halpern, Z.; Oren, R. Long term nutritional intake and the risk for non-alcoholic fatty liver disease (NAFLD): A population based study. J. Hepatol. 2007, 47, 711–717. [Google Scholar] [CrossRef]
- Zhang, C.; Yang, M. Current Options and Future Directions for NAFLD and NASH Treatment. Int. J. Mol. Sci. 2021, 15, 7571. [Google Scholar] [CrossRef]
- Santurino, C.; López-Plaza, B.; Fontecha, J.; Calvo, M.V.; Bermejo, L.M.; Gómez-Andrés, D.; Gómez-Candela, C. Consumption of Goat Cheese Naturally Rich in Omega-3 and Conjugated Linoleic Acid Improves the Cardiovascular and Inflammatory Biomarkers of Overweight and Obese Subjects: A Randomized Controlled Trial. Nutrients 2020, 5, 1315. [Google Scholar] [CrossRef]
- Ristic-Medic, D.; Kovacic, M.; Takic, M.; Arsic, A.; Petrovic, S.; Paunovic, M.; Jovicic, M.; Vucic, V. Calorie-Restricted Mediterranean and Low-Fat Diets Affect Fatty Acid Status in Individuals with Nonalcoholic Fatty Liver Disease. Nutrients 2020, 23, 15. [Google Scholar] [CrossRef] [PubMed]
- Gebauer, S.K.; Psota, T.L.; Harris, W.S.; Kris-Etherton, P.M. n−3 fatty acid dietary recommendations and food sources to achieve essentiality and cardiovascular benefits. Am. J. Clin. Nutr. 2006, 83, 1526S–1535S. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Zhu, C.; Wang, Z.; Wang, S.; Yuan, P.; Song, T.; Hou, X.; Lei, Z. Effects of Fish Oil Supplementation on Cardiometabolic Risk Factors in Overweight or Obese Children and Adolescents: A Meta-Analysis of Randomized Controlled Trials. Front. Pediatr. 2021, 27, 604469. [Google Scholar] [CrossRef] [PubMed]
- Minihane, M.A. Fish oil omega-3 fatty acids and cardio-metabolic health, alone or with statins. Eur. J. Clin. Nutr. 2013, 67, 536–540. [Google Scholar] [CrossRef]
- Gao, H.; Geng, T.; Huang, T.; Zhao, Q. Fish oil supplementation and insulin sensitivity: A systematic review and meta-analysis. Lipids Health Dis. 2017, 16, 131. [Google Scholar] [CrossRef] [Green Version]
- Yang, B.; Shi, M.Q.; Li, Z.H.; Shi, L.; Wang, A.M.; Guo, X.J.; Li, D. Effects of n−3 fatty acid supplements on cardiometabolic profiles in hypertensive patients with abdominal obesity in Inner Mongolia: A randomized controlled trial. Food Funct. 2019, 20, 1661–1670. [Google Scholar] [CrossRef]
- Pauls, S.D.; Rodway, L.R.; Sidhu, K.K.; Winter, T.; Sidhu, N.; Aukema, H.M.; Zahradka, P.; Taylor, C.G. Oils Rich in α-Linolenic Acid or Docosahexaenoic Acid Have Distinct Effects on Plasma Oxylipin and Adiponectin Concentrations and on Monocyte Bioenergetics in Women with Obesity. J. Nutr. 2021, 151, 3053–3066. [Google Scholar]
- Baril-Gravel, L.; Labonté, M.E.; Couture, P.; Vohl, M.C.; Charest, A.; Guay, V.; Jenkins, D.A.; Connelly, P.W.; West, S.; Etherton, P.M.-K.; et al. Docosahexaenoic acid-enriched canola oil increases adiponectin concentrations: A randomized crossover controlled intervention trial. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 52–59. [Google Scholar] [CrossRef]
- Nieman, D.C.; Gillitt, N.; Jin, F.; Henson, D.A.; Kennerly, K.; Shanely, R.A.; Schwartz, S. Chia seed supplementation and disease risk factors in overweight women: A metabolomics investigation. J. Altern. Complement. Med. 2012, 18, 700–708. [Google Scholar] [CrossRef]
- Egert, S.; Baxheinrich, A.; Lee-Barkey, Y.H.; Tschoepe, D.; Wahrburg, U.; Stratmann, B. Effects of an energy-restricted diet rich in plant-derived α-linolenic acid on systemic inflammation and endothelial function in overweight-to-obese patients with metabolic syndrome traits. Br. J. Nutr. 2014, 28, 1315–1322. [Google Scholar] [CrossRef] [Green Version]
- Egert, S.; Baxheinrich, A.; Lee-Barkey, Y.H.; Tschoepe, D.; Stehle, P.; Stratmann, B.; Wahrburg, U. Effects of a hypoenergetic diet rich in α-linolenic acid on fatty acid composition of serum phospholipids in overweight and obese patients with metabolic syndrome. Nutrition 2018, 49, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Baxheinrich, A.; Stratmann, B.; Lee-Barkeym, Y.H.; Tschoepe, D.; Wahrburg, U. Effects of a rapeseed oil-enriched hypoenergetic diet with a high content of α-linolenic acid on body weight and cardiovascular risk profile in patients with the metabolic syndrome. Br. J. Nutr. 2012, 108, 682–691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toscano, L.T.; Toscano, L.T.; Tavares, R.L.; da Silva, C.S.O.; Silva, A.S. Chia induces clinically discrete weight loss and improves lipid profile only in altered previous values. Nutr. Hosp. 2015, 31, 1176–1182. [Google Scholar]
- Nieman, D.C.; Cayea, E.J.; Austin, M.D.; Henson, D.A.; McAnulty, S.R.; Jin, F. Chia seed does not promote weight loss or alter disease risk factors in overweight adults. Nutr. Res. 2009, 29, 414–418. [Google Scholar] [CrossRef]
- Popović, T.; Borozan, S.; Arsić, A.; Martačić, J.D.; Vučić, V.; Trbović, A.; Glibetić, M. Fish oil supplementation improved liver phospholipids fatty acid composition and parameters of oxidative stress in male Wistar rats. J. Anim. Physiol. Anim. Nutr. 2012, 96, 1020–1029. [Google Scholar] [CrossRef]
- Prasad, P.; Anjali, P.; Sreedhar, R.V. Plant-based stearidonic acid as sustainable source of omega-3 fatty acid with functional outcomes on human health. Crit. Rev. Food Sci. Nutr. 2021, 61, 1725–1737. [Google Scholar] [CrossRef]
- Pieters, D.; Mensink, R.P. Effects of stearidonic acid on serum triacylglycerol concentrations in overweight and obese subjects: A randomized controlled trial. Eur J Clin Nutr. 2015, 69, 121–126. [Google Scholar] [CrossRef]
- Lemke, S.L.; Vicini, J.L.; Su, H.; Goldstein, D.A.; Nemeth, M.A.; Krul, E.S.; Harris, W.S. Dietary intake of stearidonic acid-enriched soybean oil increases the omega-3 index: Randomized, double-blind clinical study of efficacy and safety. Am. J. Clin. Nutr. 2010, 92, 766–775. [Google Scholar] [CrossRef] [Green Version]
- Krul, E.S.; Lemke, S.L.; Mukherjea, R.; Taylor, M.L.; Goldstein, D.A.; Su, H. Effects of duration of treatment and dosage of eicosapentaenoic acid and stearidonic acid Stearidonic acid and lipid metabolism on red blood cell eicosapentaenoic acid content. Prostaglandins Leukot Essent Fat. Acids 2012, 86, 51–59. [Google Scholar] [CrossRef]
- James, M.J.; Ursin, V.M.; Cleland, L.G. Metabolism of stearidonic acid in human subjects: Comparison with the metabolism of other n−3 fatty acids. Am. J Clin. Nutr. 2003, 77, 1140–1145. [Google Scholar] [CrossRef]
- Lemke, S.L.; Maki, K.C.; Hughes, G.; Taylor, M.L.; Krul, A.S.; Goldstein, D.A.; Su, H.; Rainsl, T.M.; Mukherjea, R. Consumption of stearidonic acid-rich oil in foods increases red blood cell eicosapentaenoic acid. J. Acad Nutr. Diet. 2013, 113, 1044–1056. [Google Scholar] [CrossRef] [PubMed]
- Takic, M.; Zekovic, M.; Terzic, B.; Stojsavljevic, A.; Mijuskovic, M.; Radjen, S.; Ristic-Medic, D. Zinc Deficiency, Plasma Fatty Acid Profile and Desaturase Activities in Hemodialysis Patients: Is Supplementation Necessary? Front. Nutr. 2021, 8, 700450. [Google Scholar] [CrossRef] [PubMed]
- Knez, M.; Pantovic, A.; Zekovic, M.; Pavlovic, Z.; Glibetic, M.; Zec, M. Is There a Link between Zinc Intake and Status with Plasma Fatty Acid Profile and Desaturase Activities in Dyslipidemic Subjects? Nutrients 2019, 12, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petrović, S.; Takić, M.; Arsić, A.; Vučić, V.; Drakulić, D.; Milošević, M.; Glibetić, M. Effect of sex hormones on plasma phospholipid fatty acid composition in intact rats and rats with bilaterally occluded carotid arteries. Physiol. Res. 2014, 63, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Ranković, S.; Popović, T.; Martačić, J.D.; Petrović, S.; Tomić, M.; Ignjatović, Đ.; Tovilović-Kovačević, G.; Glibetić, M. Liver phospholipids fatty acids composition in response to different types of diets in rats of both sexes. Lipids Health Dis. 2017, 16, 94. [Google Scholar] [CrossRef] [Green Version]
 meaning that the expression or concentration of the parameter is reduced and
meaning that the expression or concentration of the parameter is reduced and  meaning that the expression or concentration of the parameter is increased after ALA treatment.
meaning that the expression or concentration of the parameter is increased after ALA treatment.
 meaning that the expression or concentration of the parameter is reduced and
meaning that the expression or concentration of the parameter is reduced and  meaning that the expression or concentration of the parameter is increased after ALA treatment.
meaning that the expression or concentration of the parameter is increased after ALA treatment.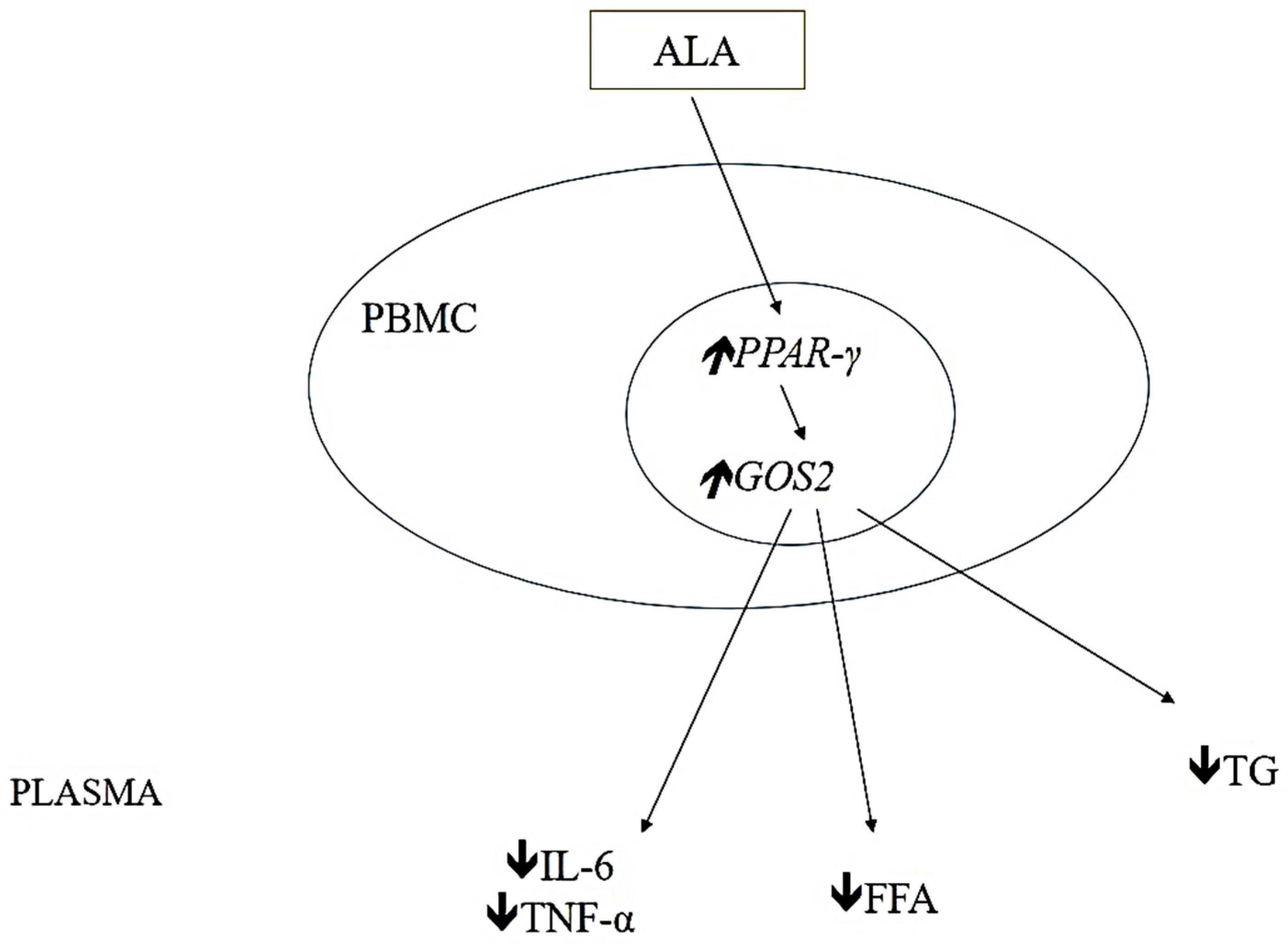
| 18:3n−3 (g/100g) | Source | Amount Needed to Meet Adequate Intake for Men (g or mL for Oils) * | Amount Needed to Meet Adequate Intake for Women (g or mL for Oils) * | |
|---|---|---|---|---|
| Seeds and legumes | ||||
| Chia seed | 17.83 | Serbian Food Composition Database [32] | 9.0 | 6.2 |
| Flaxseed | 17.09 | Turkish Food Composition Database [33] | 9.4 | 6.4 |
| Rapeseed | 2.46 | Turkish Food Composition Database [33] | 65.0 | 44.7 |
| Soybean | 1.29 | Serbian Food Composition Database [33] | 124.0 | 85.3 |
| Nut and kernel | ||||
| Walnut | 6.18 | Turkish Food Composition Database [33] | 25.9 | 17.8 |
| Pecan | 1.05 | Turkish Food Composition Database [33] | 152.4 | 104.8 |
| Fats and oils | ||||
| Flaxseed oil | 53.38 | USDA Database [34] | 3.0 | 2.1 |
| Perilla oil | 51.5 | Lee et al., 2015 [35] | 3.1 | 2.1 |
| Soybean oil | 6.78 | Serbian Food Composition Database [32] | 23.6 | 16.2 |
| Canola oil | 5.80 | Turkish Food Composition Database [33] | 27.6 | 19.0 |
| Vascular function measures |
| Carotid-to-femoral pulse wave velocity |
| Central augmentation index adjusted for heart rate |
| Central systolic blood pressure (SBP) |
| Central diastolic blood pressure (DBP) |
| Central retinal arteriolar equivalent (CRAE) |
| Central retinal venular equivalent (CRVE) |
| Retinal arteriolar-to-venular diameter ratio |
| Microvascular endothelial function parameters |
| Soluble vascular cell adhesion molecule (sVCAM) |
| Soluble intercellular adhesion molecule (sICAM) |
| Selectin |
| Metabolic risk parameters |
| Total cholesterol |
| HDL-cholesterol |
| LDL-cholesterol |
| Triglycerides |
| Free fatty acids (FFA) |
| Apolipoprotein A-1 |
| Apolipoprotein B-100 |
| Glucose |
| Insulin |
| HOMA index |
| Low-grade systematic inflammation biomarkers |
| Interleukin 6 (IL-6) |
| Interleukin 8 (IL-8) |
| Tumor necrosis factor α (TNF-α) |
| C-reactive protein (CRP) |
| Serum amyloid |
| Monocyte chemoattractant protein 1 (MCP-1) |
| Reference | Study Design | Participants Characteristics (n) | Weeks | Daily Dose | Anthropometric Parameters | Blood Pressure | Serum Lipids | Insulin Sensitivity | Inflammation Parameters | Oxidative Stress Markers |
|---|---|---|---|---|---|---|---|---|---|---|
| Toscano et al., 2015 [116] | Randomized, placebo controlled |
Overweight/obese men and women (n = 26) | 12 | 35 g | No changes in body weight and waist circumference compared to placebo | No changes in LDL-C and TG | No change in FG | |||
| Nieman et al., 2012 [112] | Randomized placebo controlled, double-blinded |
Overweight, healthy, postmenopausal women (n = 62) | 10 | 25 g | No change in body mass | No changes in SBP and AI | No change in TC | No change in FG | No changes in IL-6, IL-8, IL-10, TNF-α and CRP | |
| Nieman et al., 2009 [117] | Randomized placebo controlled, single-blinded |
Overweight/obese men and women (n = 76) | 12 | 50 g | No change in body mass | No change in SBP | No changes in TC, LDL-C and HDL-c | No change in FG | No changes in IL-6, MCP-1, TNF-α and CRP | No changes in TEAC and plasma nitrite |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takic, M.; Pokimica, B.; Petrovic-Oggiano, G.; Popovic, T. Effects of Dietary α-Linolenic Acid Treatment and the Efficiency of Its Conversion to Eicosapentaenoic and Docosahexaenoic Acids in Obesity and Related Diseases. Molecules 2022, 27, 4471. https://doi.org/10.3390/molecules27144471
Takic M, Pokimica B, Petrovic-Oggiano G, Popovic T. Effects of Dietary α-Linolenic Acid Treatment and the Efficiency of Its Conversion to Eicosapentaenoic and Docosahexaenoic Acids in Obesity and Related Diseases. Molecules. 2022; 27(14):4471. https://doi.org/10.3390/molecules27144471
Chicago/Turabian StyleTakic, Marija, Biljana Pokimica, Gordana Petrovic-Oggiano, and Tamara Popovic. 2022. "Effects of Dietary α-Linolenic Acid Treatment and the Efficiency of Its Conversion to Eicosapentaenoic and Docosahexaenoic Acids in Obesity and Related Diseases" Molecules 27, no. 14: 4471. https://doi.org/10.3390/molecules27144471
APA StyleTakic, M., Pokimica, B., Petrovic-Oggiano, G., & Popovic, T. (2022). Effects of Dietary α-Linolenic Acid Treatment and the Efficiency of Its Conversion to Eicosapentaenoic and Docosahexaenoic Acids in Obesity and Related Diseases. Molecules, 27(14), 4471. https://doi.org/10.3390/molecules27144471





