The Impact of Formulation on the Content of Phenolic Compounds in Snacks Enriched with Dracocephalum moldavica L. Seeds: Introduction to Receiving a New Functional Food Product
Abstract
1. Introduction
2. Results and Discussion
2.1. Results for Raw Extruded Pellets
2.2. Results for Fried Extruded Pellets
3. Materials and Methods
3.1. Chemicals
3.2. Production of Extruded Pellets
3.3. Preparation of Extracts
3.4. Determination of the Total Content of Polyphenolic Compounds (TPC)
3.5. Ability to Scavenge DPPH
3.6. Liquid Chromatography/Electrospray Ionization Triple Quadrupole Mass Spectrometry (LC-ESI-MS/MS)
3.7. Frying of Extruded Pellets
3.8. Statistical Analysis
4. Conclusions
5. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Sample Availability
References
- Castellanos-Gallo, L.; Galicia-García, T.; Estrada-Moreno, I.A.; Mendoza-Duarte, M.; Márquez-Meléndez, R.; Portillo-Arroyo, B.; Soto-Figueroa, C.; Leal-Ramos, M.Y.; Aldana, D.S. Development of an expanded snack of rice starch enriched with amaranth by extrusion process. Molecules 2019, 24, 2430. [Google Scholar] [CrossRef] [PubMed]
- Vallverdú-Queralt, A.; Regueiro, J.; Rinaldi De Alvarenga, J.F.; Torrado, X.; Lamuela-Raventos, R.M. Home cooking and phenolics: Effect of thermal treatment and addition of extra virgin olive oil on the phenolic profile of tomato sauces. J. Agric. Food Chem. 2014, 62, 3314–3320. [Google Scholar] [CrossRef] [PubMed]
- Brinkmann, B. Quality criteria of industrial frying oils and fats. Eur. J. Lipid Sci. Technol. 2000, 102, 539–554. [Google Scholar] [CrossRef]
- Murador, D.; Braqa, A.R.; Da Cunha, D.; De Rosso, V. Alterations in phenolic compound levels and antioxidant activity in response to cooking technique effects: A meta-analytic investigation. Crit. Rev. Food Sci. Nutr. 2018, 58, 169–177. [Google Scholar] [CrossRef]
- González-Díaz, C.; Vilaplana-Aparicio, M.J.; Iglesias-García, M. How Is Functional Food Advertising Understood? An Approximation in University Students. Nutrients 2020, 12, 3312. [Google Scholar] [CrossRef]
- Goetzke, B.; Nitzko, S.; Spiller, A. Consumption of organic and functional food. A matter of well-being and health? Appetite 2014, 77, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Li-Na, Y.; Jian-Guo, X.; Cheng-Hui, H.; Wu, T. The phenolic compounds from Dracocephalum moldavica L. Biochem. Syst. Ecol. 2014, 54, 19–22. [Google Scholar]
- Kocira, S.; Sujak, A.; Kocira, A.; Oniszczuk, A. Effect of Fylloton application on photosynthetic activity of Moldavian dragonhead (Dracocephalum moldavica L.). Agric. Agric. Sci. Proceedia 2015, 7, 108–112. [Google Scholar] [CrossRef]
- Jiang, J.; Yuan, X.; Wang, T.; Chen, H.; Zhao, H.; Yan, X.; Zheng, Q. Antioxidative and cardioprotective effects of total flavonoids extracted from Dracocephalum moldavica L. against acute ischemia/reperfusion-induced myocardial injury in isolated rat heart. Cardiovasc. Toxicol. 2014, 14, 74–82. [Google Scholar] [CrossRef]
- Dastmalchi, K.; Dorman, H.D.; Laakso, I.; Hiltunen, R. Chemical composition and antioxidative activity of Moldavian balm (Dracocephalum moldavica L.) extracts. LWT 2007, 40, 1655–1663. [Google Scholar] [CrossRef]
- Navarro, V.; Serrano, G.; Lasa, D.; Aduriz, A.L.; Ayo, J. Cooking and nutritional science: Gastronomy goes further. Int. J. Gastron. Food Sci. 2012, 1, 37–45. [Google Scholar] [CrossRef]
- Olech, M.; Pietrzak, W.; Nowak, R. Characterization of free and bound phenolic acids and flavonoid aglycones in Rosa rugosa Thunb. leaves and achenes using LC–ESI–MS/MS–MRM methods. Molecules 2020, 25, 1804. [Google Scholar] [CrossRef]
- Oniszczuk, A.; Oniszczuk, T.; Wójtowicz, A.; Wojtunik, K.; Kwaśniewska, A.; Waksmundzka-Hajnos, M. Radical scavenging activity of extruded corn gruels with addition of linden inflorescence. Open Chem. 2015, 13, 1101–1107. [Google Scholar] [CrossRef]
- Guerrero, B.G.; Montero-Montero, J.C.; Fernández-Quintero, A.; Rivera-Agredo, Y.J.; Patiño, B.O.; Gallego, S. Assessing the effect of adding maize and rice brans in the development by twin-screw extrusion of a ready-to-eat cereal formulated with flours of quality protein maize and zinc biofortified rice. DYNA 2019, 86, 298–303. [Google Scholar] [CrossRef]
- Schmid, V.; Trabert, A.; Schäfer, J.; Bunzel, M.; Karbstein, H.P.; Emin, M.A. Modification of Apple Pomace by Extrusion Processing: Studies on the Composition, Polymer Structures, and Functional Properties. Foods 2020, 9, 1385. [Google Scholar] [CrossRef] [PubMed]
- Khanal, R.C.; Howard, L.R.; Prior, R.L. Procyanidin content of grape seed and pomace, and total anthocyanin content of grape pomace as affected by extrusion processing. J. Food Sci. 2009, 74, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Alonso, R.; Grant, G.; Dewey, P.; Marzo, F. Nutritional assessment in vitro and in vivo of raw and extruded peas (Pisum sativum L.). J. Agric. Food Chem. 2000, 48, 2286–2290. [Google Scholar] [CrossRef]
- Multari, S.; Marsol-Vall, A.; Keskitalo, M.; Yang, B.; Suomela, J.P. Effects of different drying temperatures on the content of phenolic compounds and carotenoids in quinoa seeds (Chenopodium quinoa) from Finland. J. Food Compos. Anal. 2018, 72, 75–82. [Google Scholar] [CrossRef]
- Madhujith, T.; Izydorczyk, M.; Shahidi, F. Antioxidant properties of pearled barley fractions. J. Agric. Food Chem. 2006, 54, 3283–3289. [Google Scholar] [CrossRef] [PubMed]
- Patrón-Vázquez, J.; Baas-Dzul, L.; Medina-Torres, N.; Ayora-Talavera, T.; Sánchez-Contreras, Á.; García-Cruz, U.; Pacheco, N. The effect of drying temperature on the phenolic content and functional behavior of flours obtained from lemon wastes. Agronomy 2019, 9, 474. [Google Scholar] [CrossRef]
- Esparza-Martínez, F.J.; Miranda-López, R.; Guzman-Maldonado, S.H. Effect of air-drying temperature on extractable and non-extractable phenolics and antioxidant capacity of lime wastes. Ind. Crops Prod. 2016, 84, 1–6. [Google Scholar] [CrossRef]
- Korus, J.; Gumul, D.; Czechowska, K. Effect of extrusion on the phenolic composition and antioxidant activity of dry beans of Phaseolus vulgaris L. Food Technol. Biotech. 2007, 45, 139–146. [Google Scholar]
- Jung, M.Y.; Choi, D.S.; Ju, J.W. Novel technique for limitation of acrylamide formation in fried and baked corn chips and in French fries. J. Food Sci. 2003, 68, 1287–1290. [Google Scholar] [CrossRef]
- Silva, L.; Pinto, J.; Carrola, J.; Paiva-Martins, F. Oxidative stability of olive oil after food processing and comparison with other vegetable oils. Food Chem. 2010, 121, 1177–1187. [Google Scholar] [CrossRef]
- Andrikopoulos, N.K.; Kalogeropoulos, N.; Falirea, A.; Barbagianni, M.N. Performance of virgin olive oil and vegetable shortening during domestic deep-frying and pan-frying of potatoes. Inter. J. Food Sci. Technol. 2002, 37, 177–190. [Google Scholar] [CrossRef]
- Nikou, T.; Liaki, V.; Stathopoulos, P.; Skilirou, A.; Tsakiri, E.; Jakschitz, T. Comparison survey of EVOO polyphenols and exploration of healthy agingpromoting properties of oleocanthal and oleacein. Food Chem. Toxicol. 2019, 125, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, R.; Gerber, M. Food Processing and the Mediterranean Diet. Nutrients 2015, 7, 7925–7964. [Google Scholar] [CrossRef]
- Ramírez-Anaya, J.P.; Castañeda-Saucedo, M.C.; Olalla-Herrera, M.; Villalón-Mir, M.; Serrana, H.G.; Samaniego-Sánchez, C. Changes in the Antioxidant Properties of Extra Virgin Olive Oil after Cooking Typical Mediterranean Vegetables. Antioxidants 2019, 8, 246. [Google Scholar] [CrossRef]
- Molina-Garcia, L.; Santos, C.S.P.; Cunha, S.C.; Casal, S.; Fernandes, J.O. Comparative Fingerprint Changes of Toxic Volatiles in Low PUFA Vegetable Oils Under Deep-Frying. J. Am. Oil Chem. Soc. 2017, 94, 271–284. [Google Scholar] [CrossRef]
- Ramírez-Anaya, J.P.; Samaniego-Sánchez, C.; Castañeda-Saucedo, M.C.; Villalón-Mir, M.; López-García de la Serrana, H. Phenols and the antioxidant capacity of Mediterranean vegetables prepared with extra virgin olive oil using different domestic cooking techniques. Food Chem. 2015, 188, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Miglio, C.; Chiavaro, E.; Visconti, A.; Fogliano, V.; Pellegrini, N. Effects of different cooking methods on nutritional and physicochemical characteristics of selected vegetables. J. Agric. Food Chem. 2008, 56, 139–147. [Google Scholar] [CrossRef]
- De Alvarenga, J.F.R.; Tran, C.; Hurtado-Barroso, S.; Martinez-Huélamo, M.; Illan, M.; Lamuela-Raventos, R.M. Home cooking and ingredient synergism improve lycopene isomer production in Sofrito. Food Res. Int. 2017, 99, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Huélamo, M.; Vallverdú-Queralt, A.; Di Lecce, G.; Valderas-Martínez, P.; Tulipani, S.; Jáuregui, O.; Escribano-Ferrer, E.; Estruch, R.; Illan, M.; Lamuela-Raventós, R.M. Bioavailability of tomato polyphenols is enhanced by processing and fat addition: Evidence from a randomized feeding trial. Mol. Nutr. Food Res. 2016, 60, 1578–1589. [Google Scholar] [CrossRef] [PubMed]
- Bellail, A.A.; Omayma, E.S.; Mohammed, M.Y.; Ahmed, M.; El-Gamal, A. Effect of home-cooking methods on phenolic composition and antioxidant activity of sweet potato (Ipomoea batatas (L) Lam.) cultivars grown in Egypt. Food Nutr. Sci. 2012, 3, 490–499. [Google Scholar]
- Dini, I.; Tenore, G.C.; Dini, A. Effect of industrial and domestic processing on antioxidant properties of pumpkin pulp. LWT Food Sci. Technol. 2013, 53, 382–385. [Google Scholar] [CrossRef]
- Mattioli, A.V.; Farinetti, A.; Gelmini, R. The beneficial effect of Mediterranean diet on colorectal cancer. Int. J. Cancer 2019, 145, 306. [Google Scholar] [CrossRef]
- Sordini, B.; Veneziani, G.; Servili, M.; Lorefice, A.; Taticchi, A.A. Quanti-qualitative study of a phenolic extract as a natural antioxidant in the frying processes. Food Chem. 2019, 279, 426–434. [Google Scholar] [CrossRef]
- Fillion, L.; Henry, C.J.K. Nutrient losses and gains during frying: A review. Int. J. Food Sci. Nutr. 1998, 49, 157–168. [Google Scholar] [CrossRef]
- Burda, S.; Oleszek, W. Antioxidant and antiradical activities of flavonoids. J. Agric. Food Chem 2001, 49, 2774–2779. [Google Scholar] [CrossRef]
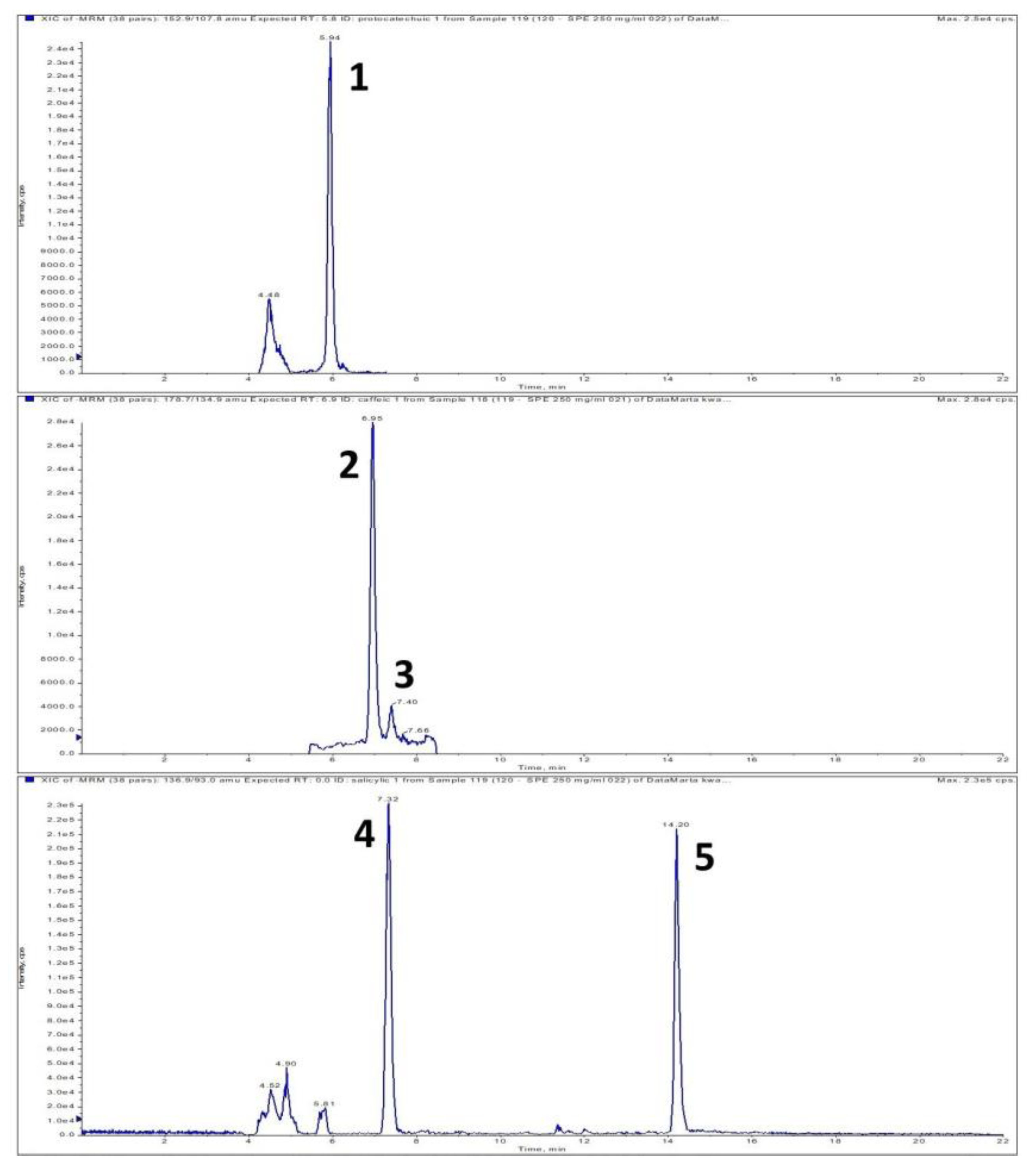
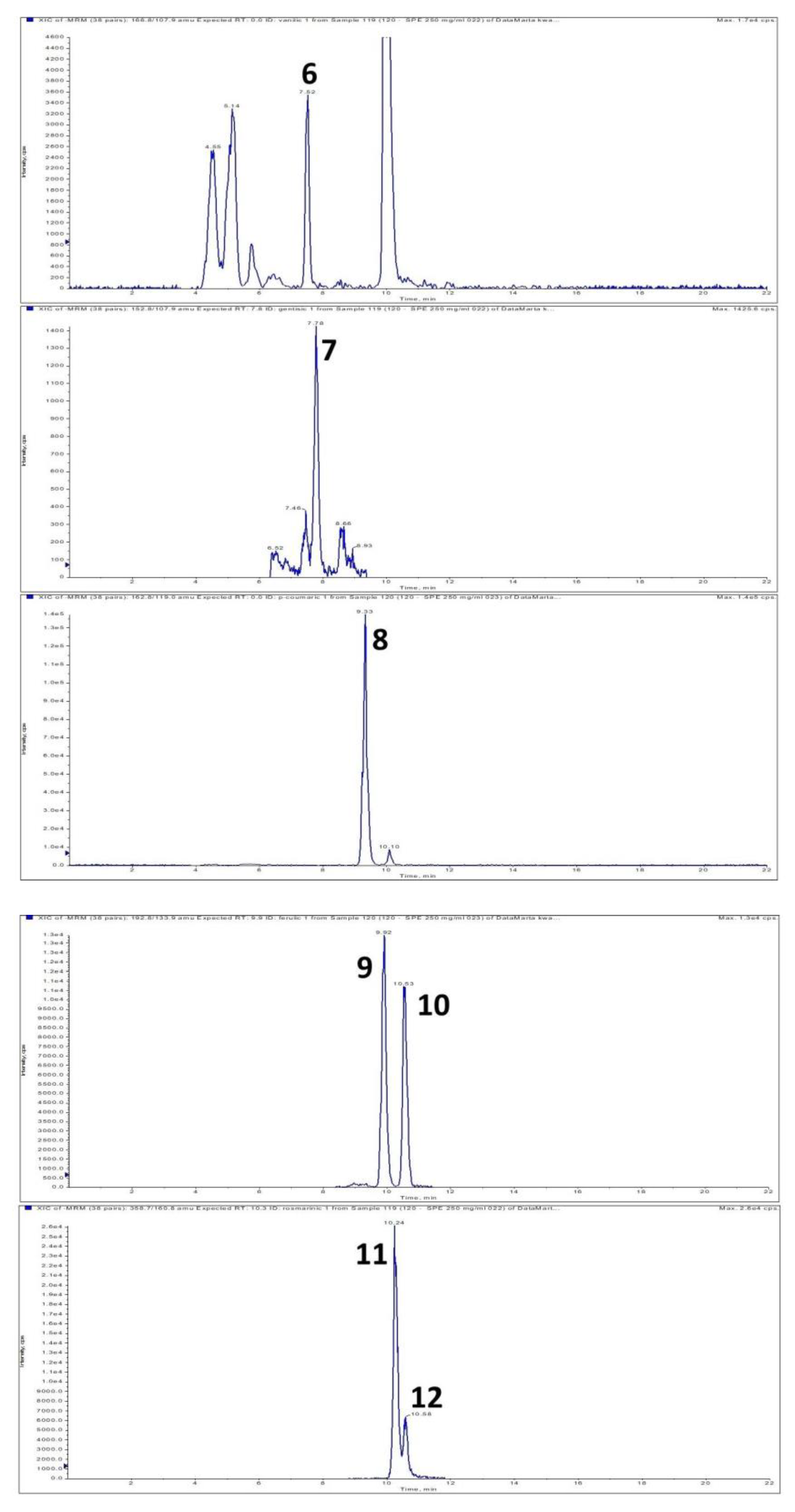
| Addition of Dragonhead Seeds (%) | Total Content of Polyphenolic Compounds TPC (mg GAE/g s.m.) | |
|---|---|---|
| Snack Pellets | Fried Snacks | |
| 0% | 0.470 a ± 0.004 | 0.239 a ± 0.002 |
| 1% | 0.546 ab ± 0.024 | 0.288 b ± 0.000 |
| 3% | 0.560 b ± 0.016 | 0.554 c ± 0.024 |
| 12% | 0.892 c ± 0.012 | 0.874 d ± 0.018 |
| 22% | 1.208 d ± 0.054 | 1.370 e ± 0.016 |
| Seeds | 2.616 ± 0.156 | |
| Addition of Dragonhead Seeds (%) | Content of Phenolic Acids (µg/g) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Protocatechuic | trans-Caffeic | 4-OH-benzoic | Vanillic | Gentisic | p-Coumaric | Ferulic | trans-Rosmarinic | cis-Rosmarinic | Isoferulic | Salicylic | Sum | ||
| Seeds | 2.141 ± 0.038 | 2.542 ± 0.105 | 6.327 ± 0.203 | 3.484 ± 0.134 | 0.026 ± 0.000 | 6.125 ± 0.325 | 3.582 ± 0.087 | 2.952 ± 0.132 | 0.956 ± 0.041 | 18.086 ± 0.248 | 4.163 ± 0.029 | 50.384 | |
| Snack pellets | 0% | 0.088 a ± 0.002 | ND | 0.322 a ± 0.005 | ND | 0.025 a ± 0.001 | 0.712 b ± 0.028 | 0.611a ± 0.007 | ND | ND | 14.200 a ± 0.407 | 0.608 a ± 0.002 | 16.566 |
| 1% | 0.139 b ± 0.005 | BQL | 0.624 b ± 0.026 | ND | 0.029 b ± 0.001 | 0.992 c ± 0.003 | 0.993 c ± 0.004 | ND | ND | 18.040 b ± 0.292 | 0.828 b ± 0.003 | 21.645 | |
| 3% | 0.168 c ± 0.0001 | BQL | 0.821 c ± 0.041 | ND | 0.025 a ± 0.000 | 0.624 a ± 0.011 | 0.648 b ± 0.005 | ND | ND | 18.162 b ± 0.327 | 1.124 c ± 0.025 | 21.572 | |
| 12% | 0.432 d ± 0.012 | 0.168a ± 0.010 | 1.543 d ± 0.073 | ND | 0.029 b ± 0.001 | 2.214 d ± 0.115 | 2.036 d ± 0.032 | 0.500 a ± 0.000 | BQL | 19.124 c ± 0.154 | 1.296 d ± 0.005 | 27.342 | |
| 22% | 0.544 e ± 0.025 | 0.524 b ± 0.026 | 2.336 e ± 0.023 | ND | 0.032 b ± 0.002 | 2.772 e ± 0.025 | 2.288 e ± 0.004 | 0.583 b ± 0.021 | BQL | 21.442 d ± 0.348 | 1.326 e ± 0.008 | 31.847 | |
| Fried snacks | 0% | 0.092 a ± 0.002 | ND | 0.528 a ± 0.000 | ND | 0.023 b ± 0.000 | 0.316 a ± 0.005 | 0.384 a ± 0.007 | ND | ND | 14.400 a ± 0.701 | 0.608 a ± 0.003 | 16.351 |
| 1% | 0.106 b ± 0.005 | ND | 0.532 a ± 0.014 | ND | 0.023 b ± 0.001 | 0.444 b ± 0.003 | 0.395 a ± 0.002 | ND | ND | 15.920 b ± 0.052 | 0.608 a ± 0.002 | 18.028 | |
| 3% | 0.113 b ± 0.000 | ND | 0.643 b ± 0.031 | ND | 0.016 a ± 0.000 | 0.484 c ± 0.003 | 0.505 b ± 0.012 | ND | ND | 19.240 c ± 0.302 | 0.620 a ± 0.021 | 21.621 | |
| 12% | 0.408 c ± 0.016 | BQL | 1.012 c ± 0.048 | 1.808 a ± 0.009 | 0.032 c ± 0.001 | 0.968 d ± 0.022 | 0.656 c ± 0.024 | 0.584 a ± 0.015 | BQL | 19.320 c ± 0.424 | 2.392 b ± 0.040 | 27.180 | |
| 22% | 0.616 d ± 0.023 | 0.760 ± 0.032 | 2.046 d ± 0.101 | 2.672 b ± 0.035 | 0.038 d ± 0.002 | 1.256 e ± 0.043 | 1.204 d ± 0.055 | 0.596 a ± 0.002 | 0.382 ± 0.013 | 35.082 d ± 0.882 | 2.400 b ± 0.111 | 47.052 | |
| Radical Scavenging Towards DPPH (%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Time (min) | Addition of Dragonhead Seeds (%) | ||||||||||
| Seeds | Snack Pellets | Fried Snacks | |||||||||
| 0% | 1% | 3% | 12% | 22% | 0% | 1% | 3% | 12% | 22% | ||
| 0 | 26.44 ± 0.151 | 18.25 b ± 0.212 | 19.57 c ± 0.051 | 19.69 c ± 0.110 | 17.92 a ± 0.062 | 22.18 d ± 0.683 | 13.25 a ± 0.212 | 13.54 a ± 0.105 | 14.19 b ± 0.120 | 15.21 c ± 0.064 | 20.17 d ± 0.163 |
| 5 | 87.23 ± 1.210 | 62.77 a ± 1.330 | 64.90 b ± 0.421 | 77.83 c ± 0.210 | 80.89 d ± 0.897 | 83.02 e ± 0.501 | 60.35 a ± 0.063 | 68.07 c ± 0.389 | 62.87 b ± 2.315 | 74.89 d ± 0.897 | 82.75 e ± 0.933 |
| 20 | 97.23 ± 0.041 | 85.13 a ± 2.198 | 88.55 b ± 0.200 | 91.35 c ± 0.801 | 92.95 c ± 2.291 | 94.55 c ± 1.302 | 83.99 a ± 0.198 | 87.19 b ± 1.983 | 90.92 c ± 0.008 | 91.01 c ± 1.930 | 96.23 d ± 1.323 |
| Total Polyphenols | Free Phenolic Acid | DPPH Radical Scavenging Activity | |
|---|---|---|---|
| Snack Pellets | |||
| Dragonhead seed content | 0.998 ** | 0.955 ** | 0.852 * |
| Total polyphenols | 0.966 ** | 0.856 * | |
| Free phenolic acids | 0.940 ** | ||
| Fried snacks | |||
| Dragonhead seed content | 0.989 ** | 0.953 ** | 0.900 ** |
| Total polyphenols | 0.933 ** | 0.993 ** | |
| Free phenolic acids | 0.767 * | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oniszczuk, T.; Kasprzak-Drozd, K.; Olech, M.; Wójtowicz, A.; Nowak, R.; Rusinek, R.; Szponar, J.; Combrzyński, M.; Oniszczuk, A. The Impact of Formulation on the Content of Phenolic Compounds in Snacks Enriched with Dracocephalum moldavica L. Seeds: Introduction to Receiving a New Functional Food Product. Molecules 2021, 26, 1245. https://doi.org/10.3390/molecules26051245
Oniszczuk T, Kasprzak-Drozd K, Olech M, Wójtowicz A, Nowak R, Rusinek R, Szponar J, Combrzyński M, Oniszczuk A. The Impact of Formulation on the Content of Phenolic Compounds in Snacks Enriched with Dracocephalum moldavica L. Seeds: Introduction to Receiving a New Functional Food Product. Molecules. 2021; 26(5):1245. https://doi.org/10.3390/molecules26051245
Chicago/Turabian StyleOniszczuk, Tomasz, Kamila Kasprzak-Drozd, Marta Olech, Agnieszka Wójtowicz, Renata Nowak, Robert Rusinek, Jarosław Szponar, Maciej Combrzyński, and Anna Oniszczuk. 2021. "The Impact of Formulation on the Content of Phenolic Compounds in Snacks Enriched with Dracocephalum moldavica L. Seeds: Introduction to Receiving a New Functional Food Product" Molecules 26, no. 5: 1245. https://doi.org/10.3390/molecules26051245
APA StyleOniszczuk, T., Kasprzak-Drozd, K., Olech, M., Wójtowicz, A., Nowak, R., Rusinek, R., Szponar, J., Combrzyński, M., & Oniszczuk, A. (2021). The Impact of Formulation on the Content of Phenolic Compounds in Snacks Enriched with Dracocephalum moldavica L. Seeds: Introduction to Receiving a New Functional Food Product. Molecules, 26(5), 1245. https://doi.org/10.3390/molecules26051245










