2.1. Fractional Factorial Design (FFD)
In the present study, the effects of four independent parameters (factors) were investigated: microwave power, Power (Watt); ratio between the volumes of the solvent and the solute (SCGs), Ratio Solv/sol (mL/g); composition of the solvent, namely ethanol-to-water ratio, Ratio Eth/water (%Ethanol), and time of microwave irradiation, Time (min). The levels choice was based on the knowledge acquired previously. For a 24−1 (two-level) FFD with four factors, 16 experimental runs were required. It should be noted that, in all the experiments performed, the maximum percentage of water was 50%, since the increase of its content in the final (extract plus solvent) solutions increased considerably the amount of energy required to obtain the final dry extract.
The maximum and minimum of the different factors and their respective coded values generated by the program Design-Expert 11 (DS-11) are shown in
Table 1.
The responses characterizing the quality of the extracts in the three response functions are: the yield (%), the total phenolic content, TPC, represented in mg of gallic acid equivalents (GAE) by g of extract (mg GAE/g), and AA as the DPPH assay in micromole of trolox equivalents (TE) by g of extract (µmol TE/g). The results of the FFD design tests are displayed in
Table 2.
2.2. Statistical Analysis of the FDD Experimental Data
The experimental design applied allowed the identification of the significant experimental factors influencing the yield, TPC, and AA of the extracts recovered. Statistical testing of the model was performed in the form of an analysis of variance (ANOVA) for each response.
The yield, TPC, and AA F-values—65.59, 146.57, and 14.40, respectively—implied that the model was significant. This was corroborated by the values of p < 0.0001 and p < 0.0006, indicating that there was only a probability of 0.01% for the two first responses and 0.06% for the AA that a higher F-value could occur due to noise. p-Values less than 0.05 indicate that the individual model terms are significant.
These results identified the independent variables X2—Ratio (Solv/sol)—and X3—Ratio (Eth/water)—as the most significant factors that influence the experimental extraction yield, TPC, and AA, with X3 being the dominant one between the two. Thus, for the yield, the contribution percentages of the ratios Solv/sol and Eth/water were 13.52 and 81.44%, respectively, and the combined contribution to the overall model of these two variables was 95%. The same trend was observed concerning TPC and AA, for which the combined contributions were 97.2 and 79.4%, respectively.
The ANOVA results on the FFD model selected are shown in
Table 3, showing the most significant terms.
The time (independent variable X
4), as well as the interaction between X
1X
2 (
Power * Ratio (Solv/sol)) and X
1X
4 (
Power*Time), have some significance on AA but with lower key values for the overall model.
p-Values greater than 0.1000 indicate that the model terms are not significant. Based on the ANOVA analysis and fitting the factors with the responses using the least squares method, the extract yield of the SCGs (Yield), as a function of the independent variables X
1–X
4, is obtained by Equation (1).
For the TPC (mg GAE/g) and AA (mg GAE/g) of SCG extracts, applying the above methodology, Equations (2) and (3) are obtained, respectively:
The ANOVA attested that a lack of fit (with a
p-value < 0.0001) was not significant for all response surface models at a 95% confidence level, which means that all models represented the data satisfactorily. Simultaneously, the R
2, adjusted R
2 (
), predicted R
2 (
), coefficient of variation (CV), and adequate precision (Ad Precision) were calculated to check the model adequacy and are presented in
Table 4.
The values of the determination coefficient (R
2) calculated (98.29, 99.23, and 92.65%) infer that the accuracy and general predictive ability of the quadratic polynomial regression models represented by Equations (1)–(3) are very good, since it is accepted that, for a good fitting model, R
2 should not be less than 80% [
19,
20].
Furthermore, the value of the adjusted R2 should be close to , suggesting that a high degree of correlation between the observed and predicted values exists. In addition, values should also be in a reasonable agreement with the (differences less than 0.2), showing that nonsignificant terms have not been included in the model.
The coefficient of variation, CV, is a measure expressing the standard deviation as a percentage of the mean. Small values of the CV represent better reproducibility. Overall, a CV higher than 10% signifies that the variation in the mean value is high and does not satisfactorily develop an adequate response model [
21].
In our case, the CVs of the yield and TPC are reasonable, while, for the AA, the CV value is the only parameter that does not satisfy the below 10% criterion. Finally, adequate precision (Ad Precision) measures the signal-to-noise ratio, and a value greater than 4 is appropriate, which is verified in all the cases examined (
Table 5).
The maximum value of a respective response can be obtained from Equations (1)–(3), respectively. Additionally, if the values corresponding to the simultaneous maximum of the three independent variables—yield, TPC, and AA—are required, those can be obtained by solving the system of Equations (1)–(3). However, as shown, the most important factor for these variations is the ethanol/water ratio, followed by the solvent/solute ratio.
To locate the ideal region of the design space for the chosen experimental factors and intended response, a FFD analysis should be used to assess the main effects and the essential interactions between them. Although Equations (1)–(3) can estimate the fitted values at the corner points of the purpose design, quadratic terms in the model to shape the curvature across the whole response surface are required. That can be achieved by applying a response surface design with axial points generated by a central composite design (CCD).
2.3. Central Composite Design (CCD)
The results of the FFD design applied at stage one indicated that the influence of power and time on the MAE was not substantial. Hence, in the CCD, the values of those independent parameters were fixed at 90 W and 4.5 min, respectively.
The other two factors—Ratio Solv/sol (10–20 mL/g) and Ratio Eth/water (50–70% Ethanol)—were used to generate the CCD test design. The ANOVA results on the CCD models selected are shown in
Table 5.
It was demonstrated that, for the responses yield and AA, the best fit equation was quadratic, while, for the TPC, a linear one. The F-values showed that the models for the yield, TPC, and AA were significant, and there was only a 0.03, 0.01, and 0.07% chance, respectively, that an F-value that high could occur due to noise. The lack of fit F-values to the three responses was nonsignificant relative to the pure error. The latter was required to have a good model fit and determine the adequacy of the model.
The final models for the responses (yield, TPC, and AA) in terms of actual factors are represented by Equations (4)–(6), respectively.
The ANOVA results on the CCD models selected are shown in
Table 6. All terms (independent variables) were significant, since the
p-values were less than 0.05 (at the 5% probability level), and only values < 0.100 were insignificant.
The ANOVA analysis demonstrated that the models were adequate and reproducible and the results were reliable. Thus, it was ascertained that the regression models obtained were suitable for the determination of significant extraction parameters values that optimized the responses—yield, TPC, and AA—of the SCG extracts recovered.
The response surface analysis was designed centered on the three-dimensional model polynomial function defined, which established the effect of the significant independent variables chosen on each observed response. The contour response plots allowed visual identification of the optimal levels of each factor and a choice of the most suitable values of the different response factors.
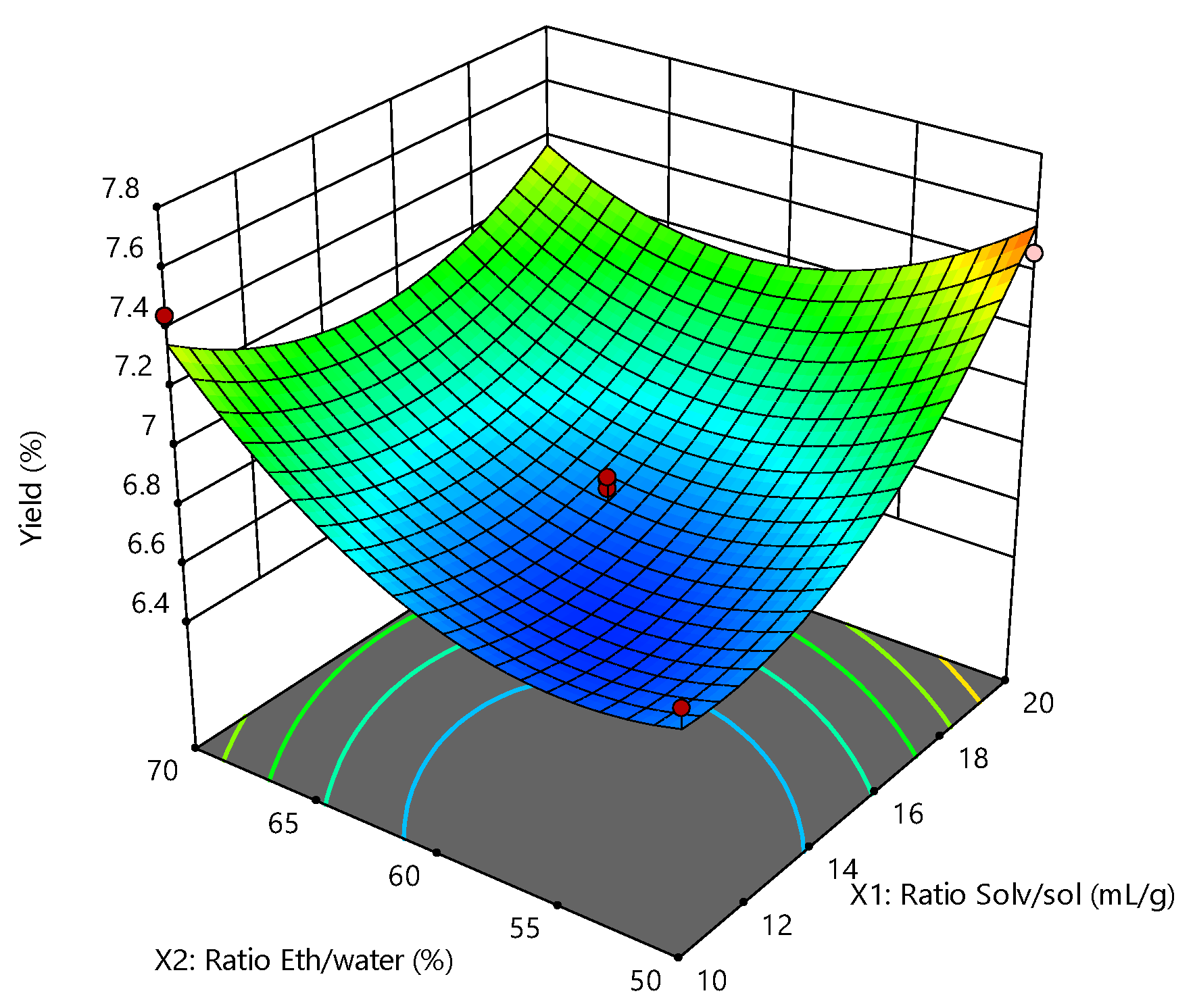
It can be deduced from
Figure 1 that, when the ratio ethanol/water is kept at a lower level, the yield increases with the growth of the ratio solvent/solute (SCGs). The extraction yield, in terms of a combination of binary factors, can change from 6.58 to 7.64%.
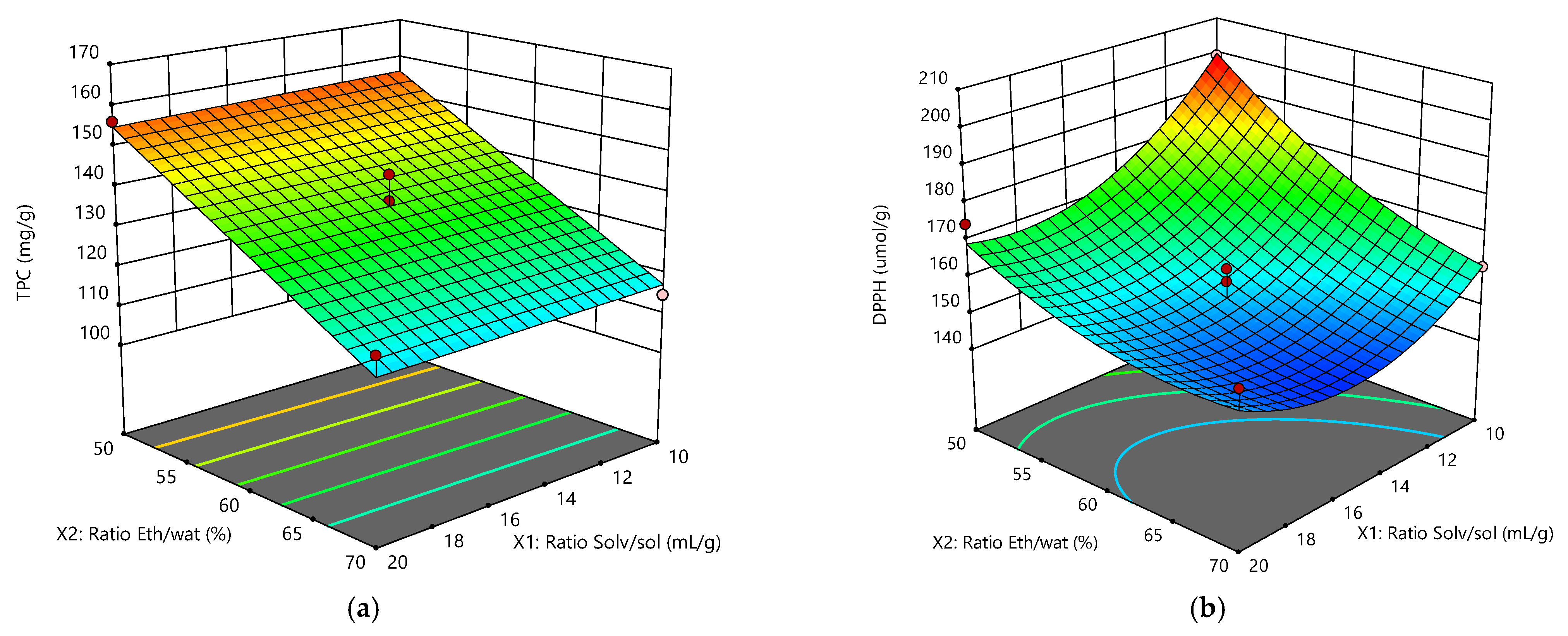
Figure 2a depicts the linear effect of the ratio ethanol/water in the solvent mixture on the TPC. It was demonstrated that, within the entire range of the ratio Solv/sol, the increase of the water percentage in the mixture raised the TPC content. This was in complete agreement with the data of
Table 5, where the parameter ratio Eth/water had a
p-value < 0.0001. On the other hand, the
p-value of the ratio Solv/sol was not significant, since it was much higher than 0.0001.
Figure 2b shows the quadratic effects of the solvent/solute and ethanol/water ratios on the AA. The maximum was achieved at the lower values of the independent parameters, a behavior pattern completely different from that depicted in
Figure 2b.
2.5. Influence of Ethanol: Water Ratio on the Extracts Composition—1H NMR Analysis
In addition to examining the influence of
Ratio (Solv/sol) and
Ratio (Eth/water) on the yield, TPC, and AA of the MAE extracts recovered, the second objective of our work was to study the impact of
Ratio (Eth/water), identified as the most significant experimental parameter, on the extract’s principal compound compositions. To realize that, an analysis of the extracts obtained in the overall range of the ethanol:water mixture composition employed was carried out. The analyses were performed by applying
1H-NMR, which has proven to be a fast and useful tool for composition identification and quantification. The influence of
Ratio (Eth/water) on the compositions of lipids and diterpenes in the extracts is presented in
Table 8 and
Table 9, respectively.
Table 8 shows that the % of molar fractions of TAGs decrease smoothly from 96.1 to 85.35%, with the decrease of the percentage of ethanol down to 50%.
The 1,2 DAGs composition, however, exhibited an opposite trend—their % molar fractions increased sharply from 1.96 to 9.04% in the range 99–60% ethanol, reached their maximum at 50% ethanol, and then decreased to 6.53% at 45.9% ethanol.
There was not a clear trend in the change of the composition of the mono- (MUFA) and di-unsaturated fatty acids (DUFA) with the change of the ethanol:water ratio. The only exception was the extract of Run 4, recovered by 99% of ethanol, for which the highest concentration of DUFA (40.4) and the lowest of MUFA (15.5) were recorded.
Table 9 shows the influence of
Ratio (Eth/water) on the compositions of the diterpenes. The reduction of ethanol % down to 60% positively affected the recovery of the diterpenes. Their % of molar fractions increased, with the highest increment registered in the range of 70–60%, the most pronounced being for cafestol. Interestingly, the diterpene % molar fractions diminished at lower than 60% ethanol in the solvent, and the most significant drop—from 11.12 to 3.17%—was observed for 16-O-Methyl-Cafestol for a relatively small increment from 50 to 45.9% in the ethanol composition. The present results are in good agreement with a previous work, where supercritical CO
2 was used to obtain the oil.
2.6. Influence of Ethanol: Water Ratio on the Eextract Compositions—Fatty Acid Methyl Esters (FAMEs) Analysis
The influence of the ethanol:water ratio on the extracts’ fatty acid compositions was also studied, and the results obtained are presented in
Table 10.
The main fatty acids identified in all the samples analyzed were palmitic (C16:0), linoleic (C18:2), oleic (C18:1), and stearic (C18:0) acids. Among those, the most abundant was linoleic acid, its highest quantity registered at 99% of ethanol.
It should be noted, that, unlike the lipids and diterpenes, the fatty acid compositions did not exhibit any clear trends regarding the ethanol:water ratio influence. Still, the highest composition of DUFA (41.8) was registered at the highest value of the ethanol:water ratio, while, for MUFA and SFA, the opposite trend was observed: the highest quantities were registered at the lowest value of that ratio.
As could be expected, the polyunsaturated/saturated ratio UI increased with the increase of the ethanol percentage, and the highest UI was calculated for Run 4, due to the highest content of linoleic acid registered in the extract. The values obtained agreed with the previous results [
4,
22,
23], where the differences in the origin and variety of the coffee and, consequently, the resulting SCGs used could explain some small variations.