Anticonvulsive Effects of Chondroitin Sulfate on Pilocarpine and Pentylenetetrazole Induced Epileptogenesis in Mice
Abstract
:1. Introduction
2. Material and Method
2.1. Animals
2.2. Drugs and Chemicals
2.3. Anti-Convulsant Activity
- A. Induction of the Pentylenetetrazole Kindled Model of Epilepsy in Mice
- B. Induction of the Status Epilepticus Provoked Spontaneous Recurrent Seizure Activity in Mice
2.4. Estimation of Oxidative Stress
2.5. Estimation of Inflammatory Mediators
2.6. Histopathological Evaluation of Brain Tissue
2.7. Experimental Protocol
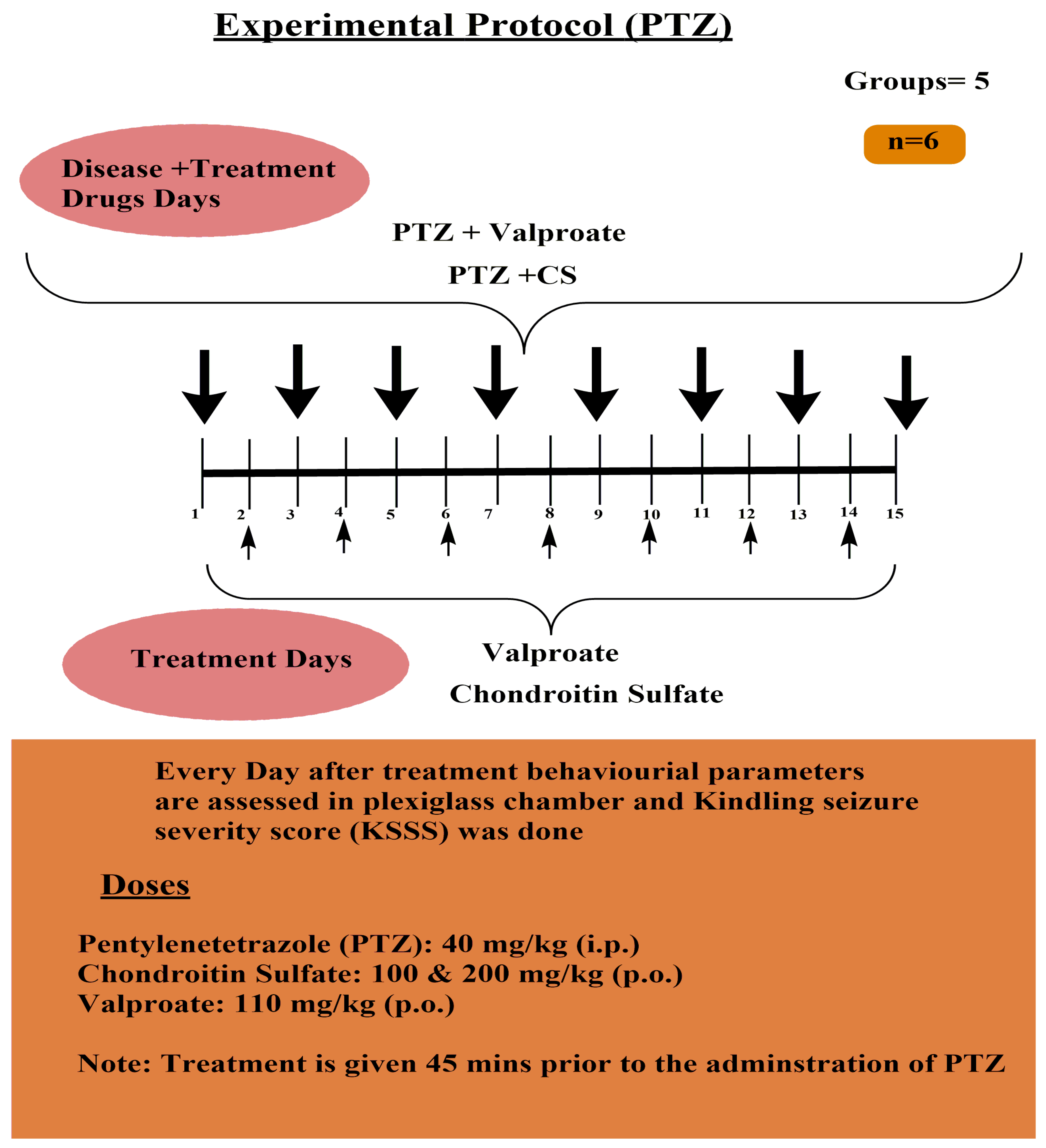
- A. Pentylenetetrazole-induced Kindled Seizure Severity Score Assessment Protocol (Figure 1)
- B. Pilocarpine-induced Spontaneous Recurrent Seizure Severity Score Assessment Protocol (Figure 2):
2.8. Molecular Docking Studies
2.9. Statistical Analysis
3. Results
3.1. Effect of Chondroitin Sulfate on PTZ-Induced Alteration in Kindling Severity Score in Mice
3.2. Effect of Chondroitin Sulfate on Pilocarpine-Induced Alteration in Spontaneous Recurrent Severity Score Mice
3.3. Effect of Chondroitin Sulfate on Pentylenetetrazole Mediated Oxidative Stress in Mice
3.4. Effect of Chondroitin Sulfate on Pilocarpine-Mediated Oxidative Stress in Mice
3.5. Effect of Chondroitin Sulfate on IL-1β, IL-6, NF-kB, and TNF-α Concentration in PTZ-Induced Kindling Seizures in Mice
3.6. Effect of Chondroitin Sulfate on IL-1β, IL-6, NF-kB, TNF-α, and Caspase-3 Concentration in Pilocarpine-Induced Spontaneous Seizures in Mice
3.7. Effect of Chondroitin Sulfate on PTZ-Ediated Caspase-3 Level in Mice
3.8. Effect of Chondroitin Sulfate on the Pilocarpine-Mediated Caspase-3 Level in Mice
3.9. Effect of Chondroitin Sulfate on PTZ- and Pilocarpine-Mediated Histopathological Changes in Mice Brains
3.10. Molecular Docking Studies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Chiavellini, P.; Canatelli-Mallat, M.; Lehmann, M.; Goya, R.G.; Morel, G.R. Therapeutic potential of glial cell line-derived neurotrophic factor and cell reprogramming for hippocampal-related neurological disorders. Neural Regen. Res. 2022, 17, 469–476. [Google Scholar]
- Striano, P.; Minassian, B.A. From Genetic Testing to Precision Medicine in Epilepsy. Neurotherapeutics 2020, 17, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Marcolini, E.; Tolchin, B. Functional Seizures. Emerg. Med. Clin. N. Am. 2021, 39, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Steriade, C.; French, J.; Devinsky, O. Epilepsy: Key experimental therapeutics in early clinical development. Expert Opin. Investig. Drugs 2020, 29, 373–383. [Google Scholar] [CrossRef]
- Colbourn, R.; Naik, A.; Hrabetova, S. ECS dynamism and its influence on neuronal excitability and seizures. Neurochem. Res. 2019, 44, 1020–1036. [Google Scholar] [CrossRef]
- Perkins, K.L.; Arranz, A.M.; Yamaguchi, Y.; Hrabetova, S. Brain extracellular space, hyaluronan, and the prevention of epileptic seizures. Rev. Neurosci. 2017, 28, 869–892. [Google Scholar] [CrossRef] [PubMed]
- Hrabetova, S.; Cognet, L.; Rusakov, D.A.; Nägerl, U.V. Unveiling the extracellular space of the brain: From super-resolved microstructure to in vivo function. J. Neurosci. 2018, 38, 9355–9363. [Google Scholar] [CrossRef] [Green Version]
- Vecino, E.; Kwok, J.C. The extracellular matrix in the nervous system. The good and the bad aspects. In Composition and Function of the Extracellular Matrix in the Human Body; InTech Open: London, UK, 2016; pp. 1–35. [Google Scholar]
- Anwar, H.; Khan, Q.U.; Nadeem, N.; Pervaiz, I.; Ali, M.; Cheema, F.F. Epileptic seizures. Discoveries 2020, 8, e110. [Google Scholar] [CrossRef]
- Papp, A.; Fehér, O.; Erdélyi, L. The ionic mechanism of the pentylenetetrazol convulsions. Acta Biol. Hung. 1987, 38, 349–361. [Google Scholar]
- Kim, D.; Kim, H.J.; Chae, H.S.; Park, N.G.; Kim, Y.B.; Jang, A. Anti-oxidation and anti-wrinkling effects of Jeju horse leg bone hydrolysates. Korean J. Food Sci. Anim. Resour. 2014, 34, 844. [Google Scholar] [CrossRef] [Green Version]
- Vázquez, J.A.; Fraguas, J.; Novoa-Carvallal, R.; Reis, R.L.; Antelo, L.T.; Pérez-Martín, R.I.; Valcarcel, J. Isolation and chemical characterization of chondroitin sulfate from cartilage by-products of blackmouth catshark (Galeus melastomus). Mar. Drugs 2018, 16, 344. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Shen, Q.; Zhang, C.; Jia, W.; Han, L.; Yu, Q. Chicken leg bone as a source of chondroitin sulfate. Carbohydr. Polym. 2019, 207, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Messina, O.D.; Wilman, M.V.; Neira, L.F.V. Nutrition, osteoarthritis and cartilage metabolism. Aging Clin. Exp. Res. 2019, 31, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Li, J.; Liu, C.; Song, C.; Li, P.; Yin, F.; Xiao, Y.; Jiang, W.; Zong, A.; Zhang, X.; et al. Protective effects of low molecular weight chondroitin sulfate on amyloid beta (Aβ)-induced damage in vitro and in vivo. Neuroscience 2015, 305, 169–182. [Google Scholar] [CrossRef] [PubMed]
- Ju, C.; Gao, J.; Hou, L.; Wang, L.; Zhang, F.; Sun, F.; Zhang, T.; Xu, P.; Shi, Z.; Hu, F.; et al. Neuroprotective effect of chondroitin sulfate on SH-SY5Y cells overexpressing wild-type or A53T mutant α-synuclein. Mol. Med. Rep. 2017, 16, 8721–8728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okamoto, M.; Mori, S.; Ichimura, M.; Endo, H. Chondroitin sulfate proteoglycans protect cultured rat’s cortical and hippocampal neurons from delayed cell death induced by excitatory amino acids. Neurosci. Lett. 1994, 172, 51–54. [Google Scholar] [CrossRef]
- Dudas, B.; Rose, M.; Cornelli, U.; Pavlovich, A.; Hanin, I. Neuroprotective properties of glycosaminoglycans: Potential treatment for neurodegenerative disorders. Neurodegener. Dis. 2008, 5, 200–205. [Google Scholar] [CrossRef] [Green Version]
- Egea, J.; García, A.G.; Verges, J.; Montell, E.; López, M.G. Antioxidant, antiinflammatory and neuroprotective actions of chondroitin sulfate and proteoglycans. Osteoarthr. Cartil. 2010, 18, S24–S27. [Google Scholar] [CrossRef] [Green Version]
- Volpi, N. Chondroitin Sulfate Safety and Quality. Molecules 2019, 24, 1447. [Google Scholar] [CrossRef] [Green Version]
- Ravizza, T.; Vezzani, A. Pharmacological targeting of brain inflammation in epilepsy: Therapeutic perspectives from experimental and clinical studies. Epilepsia Open 2018, 3, 133–142. [Google Scholar] [CrossRef] [Green Version]
- Fujimoto, T.; Kawashima, H.; Tanaka, T.; Hirose, M.; Toyama-Sorimachi, N.; Matsuzawa, Y.; Miyasaka, M. CD44 binds a chondroitin sulfate proteoglycan, aggrecan. Int. Immunol. 2001, 13, 359–366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rana, A.; Musto, A.E. The role of inflammation in the development of epilepsy. J. Neuroinflamm. 2018, 15, 1–12. [Google Scholar] [CrossRef]
- Rehni, A.K.; Singh, T.G.; Kalra, R.; Singh, N. Pharmacological inhibition of inducible nitric oxide synthase attenuates the development of seizures in mice. Nitric Oxide 2009, 21, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Beutler, E.; Kelly, B.M. The effect of sodium nitrite on red cell GSH. Experientia 1963, 19, 96–97. [Google Scholar] [CrossRef] [PubMed]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar]
- Wang, X.L.; Adachi, T.; Sim, A.S.; Wilcken, D.E.L. Plasma extracellular superoxide dismutase levels in an Australian population with coronary artery disease. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 1915–1921. [Google Scholar] [CrossRef] [Green Version]
- Dassault SystemesBiovia Corp, Discovery Studio Client v20.1.0.19295. 2019. Available online: https://discover.3ds.com/discovery-studio-visualizer (accessed on 28 October 2021).
- Wu, G.; Robertson, D.H.; Brooks, C.L., III; Vieth, M. Detailed analysis of grid-based molecular docking: A case study of CDOCKER—A CHARMm-based MD docking algorithm. J. Comput. Chem. 2003, 24, 1549–1562. [Google Scholar] [CrossRef]
- Kryger, G.; Silman, I.; Sussman, J.L. Structure of acetylcholinesterase complexed with E2020 (Aricept®): Implications for the design of new anti-Alzheimer drugs. Structure 1999, 7, 297–307. [Google Scholar] [CrossRef] [Green Version]
- Shih, K.C.; Shiau, C.W.; Chen, T.S.; Ko, C.H.; Lin, C.L.; Lin, C.Y.; Hwang, C.S.; Tang, C.Y.; Chen, W.R.; Huang, J.W. Pharmacophore modeling and virtual screening to identify potential RET kinase inhibitors. Bioorg. Med. Chem. Lett. 2011, 21, 4490–4497. [Google Scholar] [CrossRef]
- Singh, S.; Singh, T.G. Emerging perspectives on mitochondrial dysfunctioning and inflammation in epileptogenesis. Inflamm. Res. 2021, 70, 1027–1042. [Google Scholar] [CrossRef]
- Rehni, A.K.; Singh, T.G. Modulation of leukotriene D4 attenuates the development of seizures in mice. Prostaglandins Leukot. Essent. Fat. Acids 2011, 85, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Rehni, A.K.; Singh, T.G. Selenium induced anticonvulsant effect: A potential role of prostaglandin E1 receptor activation linked mechanism. J. Trace Elem. Med. Biol. 2013, 27, 31–39. [Google Scholar] [CrossRef]
- Singh, S.; Singh, T.G.; Rehni, A.K.; Sharma, V.; Singh, M.; Kaur, R. Reviving mitochondrial bioenergetics: A relevant approach in epilepsy. Mitochondrion 2021, 58, 213–226. [Google Scholar] [CrossRef]
- Schiraldi, C.; Cimini, D.; De Rosa, M. Production of chondroitin sulfate and chondroitin. Appl. Microbiol. Biotechnol. 2010, 87, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Patra, P.; Boucher, W.; Letourneau, R.; Kempuraj, D.; Chiang, G.; Athanasiou, A. Chondroitin sulphate inhibits connective tissue mast cells. Br. J. Pharmacol. 2000, 131, 1039–1049. [Google Scholar] [CrossRef] [Green Version]
- Campo, G.M.; Avenoso, A.; Campo, S.; Ferlazzo, A.M.; Calatroni, A. Chondroitin sulphate: Antioxidant properties and beneficial effects. Mini Rev. Med. Chem. 2006, 6, 1311–1320. [Google Scholar] [CrossRef]
- Zhu, X.; Dong, J.; Han, B.; Huang, R.; Zhang, A.; Xia, Z.; Chang, H.; Chao, J.; Yao, H. Neuronal Nitric Oxide Synthase Contributes to PTZ Kindling Epilepsy-Induced Hippocampal Endoplasmic Reticulum Stress and Oxidative Damage. Front. Cell. Neurosci. 2017, 11, 377. [Google Scholar] [CrossRef] [Green Version]
- Temp, F.R.; Marafiga, J.R.; Milanesi, L.H.; Duarte, T.; Rambo, L.M.; Pillat, M.M.; Mello, C.F. Cyclooxygenase-2 inhibitors differentially attenuate pentylenetetrazol-induced seizures and increase of pro-and anti-inflammatory cytokine levels in the cerebral cortex and hippocampus of mice. Eur. J. Pharmacol. 2017, 810, 15–25. [Google Scholar] [CrossRef]
- Yue, J.; Wei, Y.J.; Yang, X.L.; Liu, S.Y.; Yang, H.; Zhang, C.Q. NLRP3 inflammasome and endoplasmic reticulum stress in the epileptogenic zone in temporal lobe epilepsy: Molecular insights into their interdependence. Neuropathol. Appl. Neurobiol. 2020, 46, 770–785. [Google Scholar] [CrossRef]
- Singh, S.; Singh, T.G. Role of nuclear factor kappa B (NF-ΚB) signalling in neurodegenerative diseases: An mechanistic approach. Curr. Neuropharmacol. 2020, 18, 918–935. [Google Scholar] [CrossRef]
- Singh, S.; Singh, T.G.; Rehni, A.K. An Insight into Molecular Mechanisms and Novel Therapeutic Approaches in Epileptogenesis. CNS Neurol. Disord.-Drug Targets 2020, 19, 750–779. [Google Scholar] [CrossRef]
- Attia, G.M.; Elmansy, R.A.; Elsaed, W.M. Neuroprotective effect of nilotinib on pentylenetetrazol-induced epilepsy in adult rat hippocampus: Involvement of oxidative stress, autophagy, inflammation, and apoptosis. Folia Neuropathol. 2019, 57, 146–160. [Google Scholar] [CrossRef]
- Liu, D.H.; Agbo, E.; Zhang, S.H.; Zhu, J.L. Anticonvulsant and neuroprotective effects of paeonol in epileptic rats. Neurochem. Res. 2019, 44, 2556–2565. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Li, C.Y.; Wang, H.; Zhang, Q.M.; Han, Z.M. Therapeutic effects of scoparone on pilocarpine (Pilo)-induced seizures in mice. Biomed. Pharmacother. 2018, 97, 1501–1513. [Google Scholar] [CrossRef] [PubMed]
- Saha, L.; Chakrabarti, A.; Kumari, S.; Bhatia, A.; Banerjee, D. Antiapoptotic and neuroprotective role of Curcumin in Pentylenetetrazole (PTZ) induced kindling model in rat. Indian J. Exp. Biol. 2016, 54, 133–141. [Google Scholar] [PubMed]
- Fu, L.; Liu, K.; Wake, H.; Teshigawara, K.; Yoshino, T.; Takahashi, H.; Mori, S.; Nishibori, M. Therapeutic effects of anti-HMGB1 monoclonal antibody on pilocarpine-induced status epilepticus in mice. Science 2017, 7, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Rehni, A.K.; Singh, T.G.; Singh, N.; Arora, S. Tramadol-induced seizurogenic effect: A possible role of opioid-dependent histamine H1 receptor activation-linked mechanism. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2010, 381, 11–19. [Google Scholar] [CrossRef]





| Groups | TBARs (nmol/mg of Protein) | Catalase (µM of H2O2 Oxidised/min/mg Protein) | Glutathione (µM of Glutathione/mg of Protein) | Superoxide Dismutase (SOD) (U/mg of protein) |
|---|---|---|---|---|
| Vehicle control group | 3.4 ± 0.99 | 39.73 ± 2.15 | 48.75 ± 2.76 | 35.8 ± 2.64 |
| Pentylenetetrazole (PTZ) control | 22.1 ± 2.32 a | 14.26 ± 1.19 a | 17.01 ± 3.10 a | 12.95 ± 0.99 a |
| Chondroitin sulfate (100 mg/kg) + PTZ treatment | 11.3 ± 1.57 b | 21.2 ± 1.75 b | 26.5 ±1.47 b | 20.93 ± 1.48 b |
| Chondroitin sulfate (200 mg/kg) + PTZ treatment | 6.11 ± 0.57 b | 34.73 ± 1.60 b | 41.3 ± 1.53 b | 30.23 ± 1.24 b |
| Valproic acid (110 mg/kg)+ PTZ treatment | 4.46 ± 0.52 b | 38.48 ± 2.09 b | 45.4 ± 3.93 b | 33.85 ± 3.46 b |
| Groups | TBARs (nmol/mg of Protein) | Catalase (µM of H2O2 Oxidised/min/mg Protein) | Glutathione (µM of Glutathione/mg of Protein) | Superoxide Dismutase (SOD) (U/mg of Protein) |
|---|---|---|---|---|
| Vehicle control group | 3.4 ± 0.99 | 39.73 ± 2.15 | 48.75 ± 2.15 | 35.8 ± 2.64 |
| Pilocarpine control | 24.76 ± 3.11 a | 11.36 ± 0.93 a | 15.38 ± 0.93 a | 10.86 ± 1.23 a |
| Chondroitin sulfate (100 mg/kg) + pilocarpine treatment | 12.3 ± 1.78 b | 20.96 ± 1.53 b | 25.06 ± 1.53 b | 15.6 ± 0.81 b |
| Chondroitin sulfate (200 mg/kg) + pilocarpine treatment | 6.38 ± 0.62 b | 34.06 ± 2.01 b | 39.96 ± 2.01 b | 29.56 ± 1.24 b |
| Valproic acid (110 mg/kg) + pilocarpine treatment | 4.81 ± 0.71 b | 38.15 ± 2.67 b | 44.36 ± 2.67 b | 32.9 ± 2.02 b |
| Groups | IL-1β (pg/mg of Tissue) | IL-6 (pg/mg of Tissue) | NF-kB (pg/mg of Tissue) | TNF-α (pg/mg of Tissue) | Caspase-3 (pg/mg of Tissue) |
|---|---|---|---|---|---|
| Vehicle control group | 47.25 ± 1.61 | 39.16 ± 2.37 | 36.83 ± 1.40 | 30.43 ± 1.19 | 0.71 ± 0.147 |
| Pentylenetetrazole (PTZ) control | 155.86 ± 3.79 a | 106.83 ± 2.99 a | 83.46 ± 3.19 a | 74.8 ± 2.38 a | 8.3 ± 0.43 a |
| Chondroitin sulfate (100 mg/kg) + PTZ treatment | 110.4 ± 4.59 b | 80.5 ± 3.83 b | 66.15 ± 2.53 b | 55.68 ± 3.24 b | 6.45 ± 0.92 b |
| Chondroitin sulfate (200 mg/kg) + PTZ treatment | 62.1 ± 3.87 b | 54.3 ± 2.59 b | 48.13 ± 2.18 b | 44.5 ± 2.75 b | 4.51 ± 0.46 b |
| Valproic acid (110 mg/kg) + PTZ treatment | 54.75 ± 2.16 b | 45.16 ± 1.45 b | 41.08 ± 1.58 b | 37.56 ± 1.35 b | 3.68 ± 0.33 b |
| Groups | IL-1β (pg/mg of Tissue) | IL-6 (pg/mg of Tissue) | NF-kB (pg/mg of Tissue) | TNF-α (pg/mg of Tissue) | Caspase-3 (pg/mg of Tissue) |
|---|---|---|---|---|---|
| Vehicle control group | 47.25 ± 1.61 | 39.61± 2.37 | 36.88 ± 1.40 | 30.43± 1.19 | 0.71 ± 0.14 |
| Pilocarpine control | 175.66 ± 4.36 a | 131.83± 4.73 a | 93.18± 1.92 a | 83.46± 3.83 a | 11.01 ± 0.90 a |
| Chondroitin sulfate (100 mg/kg) + pilocarpine treatment | 120.66 ± 3.66 b | 93.51 ± 3.11 b | 68.48 ± 2.57 b | 58.55 ± 4.02 b | 6.6 ± 0.49 b |
| Chondroitin sulfate (200 mg/kg) + pilocarpine treatment | 64.13 ± 3.99 b | 59.3 ± 2.79 b | 51.98 ± 2.45 b | 46.35 ± 3.52 b | 4.8 ± 0.52 b |
| Valproic acid (110 mg/kg) + pilocarpine treatment | 56.2 ± 3.41 b | 44.9 ± 2.56 b | 43.35 ± 1.96 b | 39.31 ± 1.20 b | 3.9 ± 0.25 b |
| Protein | Binding Interaction Energy | Type of Interaction | Bonding Amino Acids |
|---|---|---|---|
| TNF-alpha | + | H-bond | Tyr 151, Ser 60, Leu120, Gly121 |
| van der Waals | Tyr59, Leu57, Gln61, Tyr119, Gly122 | ||
| IL-1β | −16.50 | H-bond | Leu134, Phe133 |
| van der Waals | Gly135, Trp120, Asp142 | ||
| IL-6 | −18.68 | H-bond | Gln175, Arg30, Asp26, Arg179, Arg182 |
| van der Waals | Leu33, Leu178 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, S.; Singh, T.G.; Singh, M.; Najda, A.; Nurzyńska-Wierdak, R.; Almeer, R.; Kamel, M.; Abdel-Daim, M.M. Anticonvulsive Effects of Chondroitin Sulfate on Pilocarpine and Pentylenetetrazole Induced Epileptogenesis in Mice. Molecules 2021, 26, 6773. https://doi.org/10.3390/molecules26226773
Singh S, Singh TG, Singh M, Najda A, Nurzyńska-Wierdak R, Almeer R, Kamel M, Abdel-Daim MM. Anticonvulsive Effects of Chondroitin Sulfate on Pilocarpine and Pentylenetetrazole Induced Epileptogenesis in Mice. Molecules. 2021; 26(22):6773. https://doi.org/10.3390/molecules26226773
Chicago/Turabian StyleSingh, Shareen, Thakur Gurjeet Singh, Manjinder Singh, Agnieszka Najda, Renata Nurzyńska-Wierdak, Rafa Almeer, Mohamed Kamel, and Mohamed M. Abdel-Daim. 2021. "Anticonvulsive Effects of Chondroitin Sulfate on Pilocarpine and Pentylenetetrazole Induced Epileptogenesis in Mice" Molecules 26, no. 22: 6773. https://doi.org/10.3390/molecules26226773
APA StyleSingh, S., Singh, T. G., Singh, M., Najda, A., Nurzyńska-Wierdak, R., Almeer, R., Kamel, M., & Abdel-Daim, M. M. (2021). Anticonvulsive Effects of Chondroitin Sulfate on Pilocarpine and Pentylenetetrazole Induced Epileptogenesis in Mice. Molecules, 26(22), 6773. https://doi.org/10.3390/molecules26226773








