Phytochemical Profile, Free Radical Scavenging and Anti-Inflammatory Properties of Acalypha Indica Root Extract: Evidence from In Vitro and In Vivo Studies
Abstract
1. Introduction
2. Results
2.1. Phytochemicals in A. indica Root Extract
2.2. Phytochemical Profile of A. indica Root Methanolic Extract
2.3. A. indica Root Extract Scavenges DPPH and Hydroxyl Radicals
2.4. A. indica Root Extract Suppress Hydrogen Peroxide and Lipid Peroxidation In Vitro
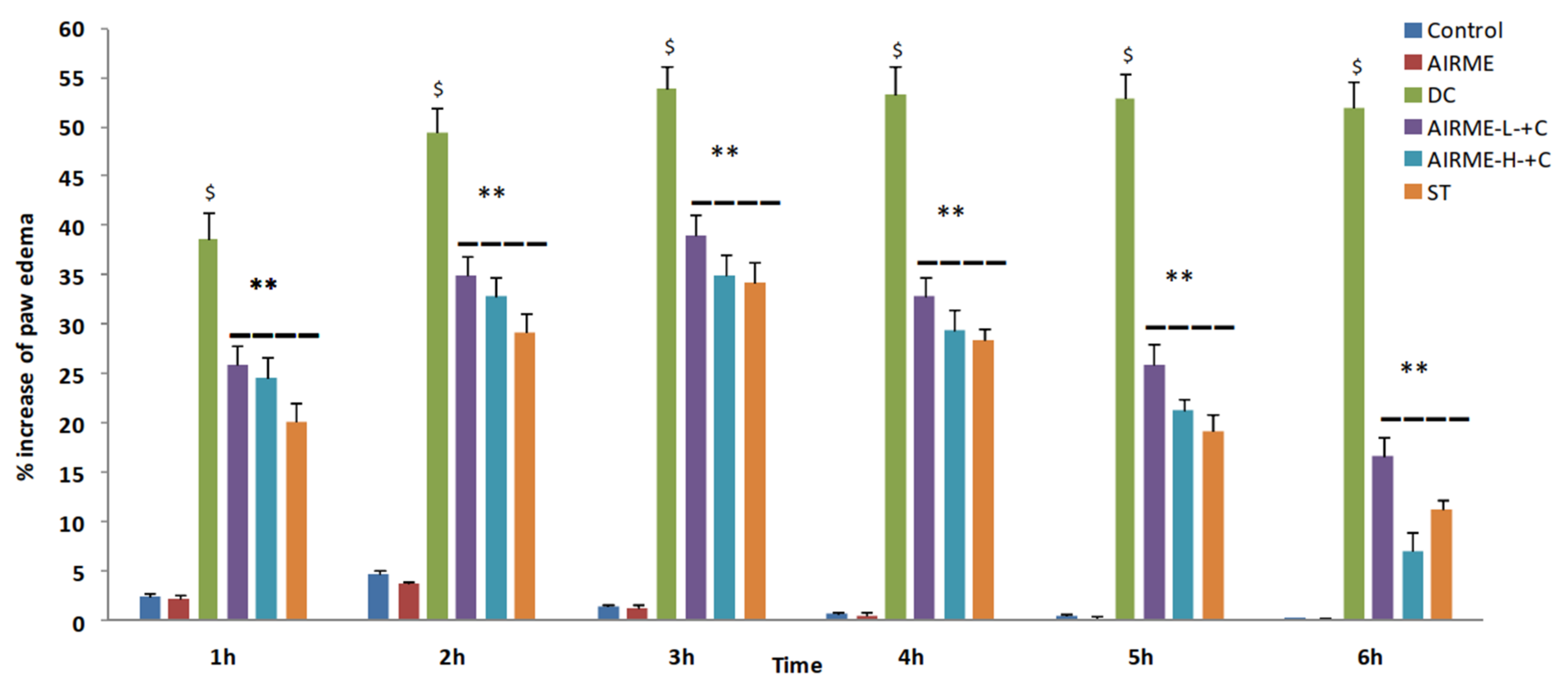
2.5. A. indica Root Extract Decreases Paw Edema Volume in Rats
2.6. A. indica Root Extract Attenuates Acute Inflammatory Response in Rats
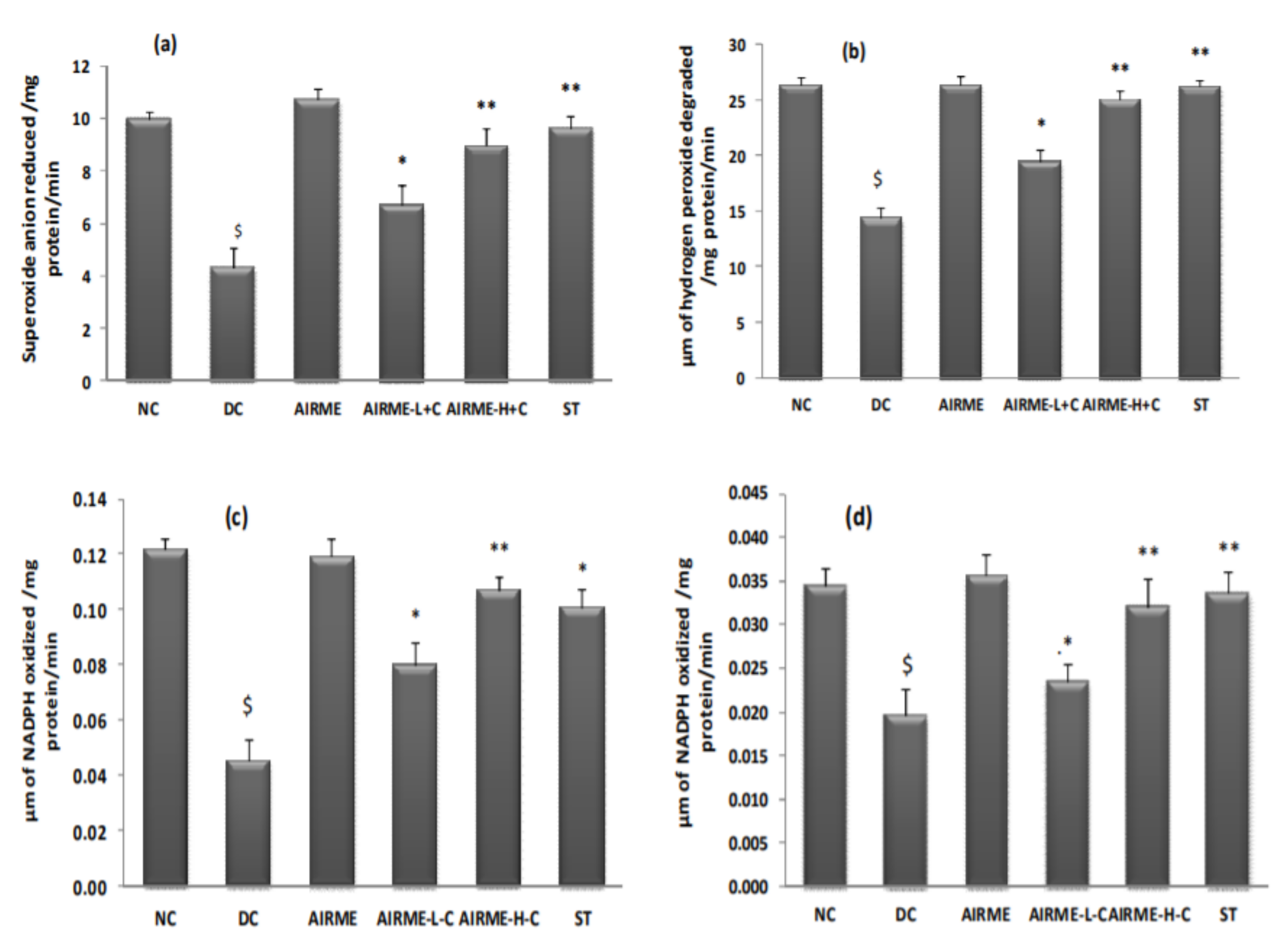
2.7. A. indica Root Extract Restores Antioxidant Enzyme Activities in Paw Tissue
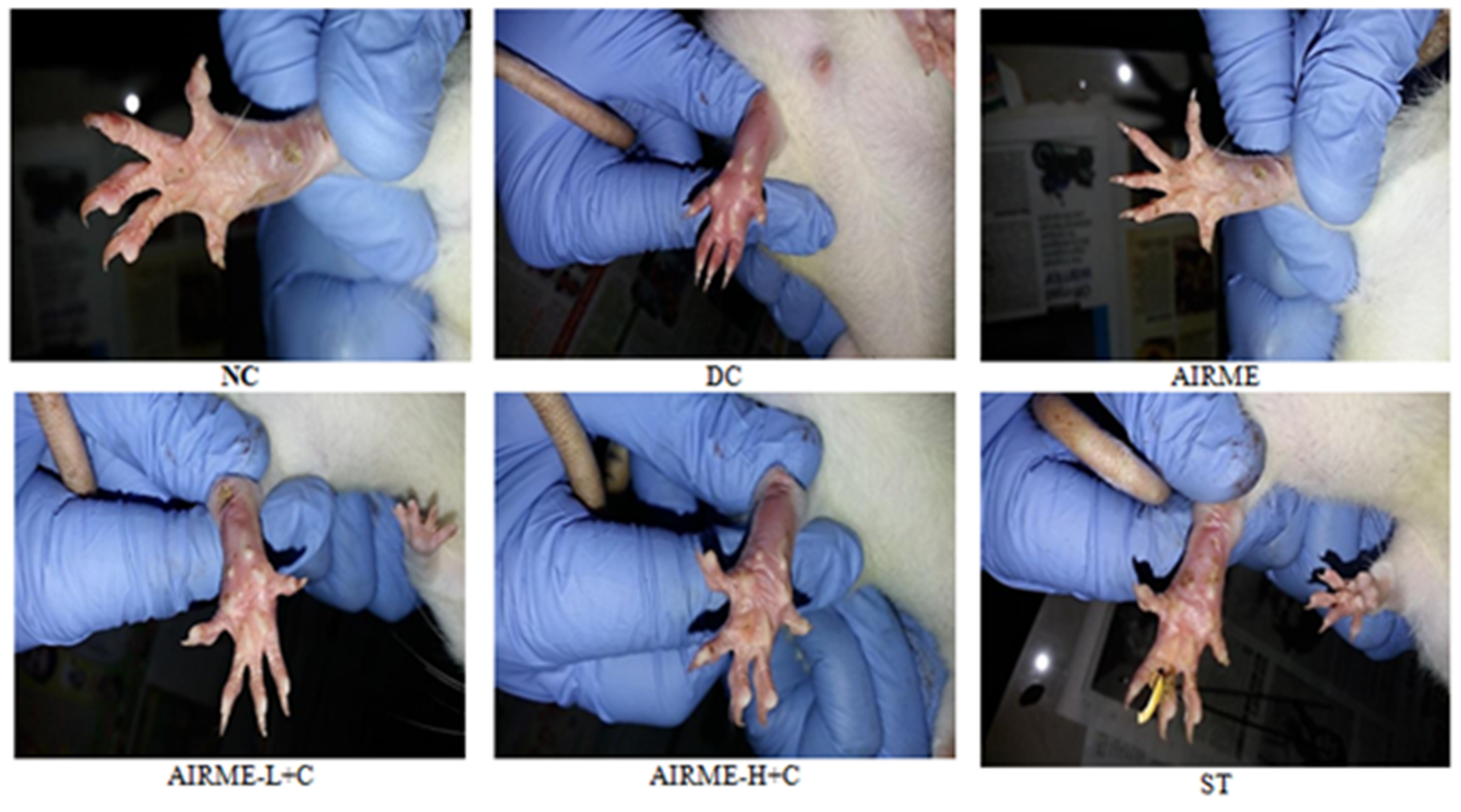
2.8. A. indica Root Extract Prevent Tissue Architectural Damage
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Collection and Preparation of A. indica Root Extract
4.3. Qualitative Estimation of Phytochemicals
4.4. Quantitative Estimation of Phytochemicals
4.4.1. Determination of Total Phenolic Content
4.4.2. Determination of Total Flavonoids Content
4.4.3. Phytochemical Profiling and Gas Chromatography-Mass Spectrometry (GC-MS) analysis of AIRME
4.4.4. Phytochemicals Profiling and HR-LC-MS Analysis of AIRME
4.4.5. Computational Calculations for the Identified Phytochemicals
4.5. In Vitro Studies
4.6. In Vivo Studies
4.6.1. Experimental Design and Treatment
- Group I—Normal control (NC): Rats in this group received 50 µL normal saline by sub-plantar injection (SPI);
- Group II—Disease control (DC): Carrageenan (1%, 100 µL) was injected (SPI) into the rat paw to induce acute localized inflammation;
- Group III—AIRME control (AIRME): Rats in this group received only AIRME (300 mg/kg bodyweight) by oral gavage;
- Group IV—AIRME low-dose plus carrageenan (AIRME-L+C): Rats were orally administered with low dose of AIRME (150 mg/kg body weight) through oral gavage 1 h prior to carrageenan injection.;
- Group V—AIRME high-dose plus carrageenan (AIRME-H+C): Rats in this group received high dose of AIRME (300 mg/kg body weight) 1 h prior to carrageenan injection, as described in group IV;
- Group VI—Standard treatment (ST): Rats in this group received diclofenac (20 mg/kg body weight) by oral gavage one hour before carrageenan injection.
4.6.2. Measurement of Paw Volume
- —thickness of paw at different time intervals (1, 2, 3, 4, 5 and 6 h) (after carrageenan induction);
- —thickness of paw at 0 h (before injection of carrageenan).
4.6.3. Blood Collection and Analyses
4.6.4. Determination of Antioxidant Enzyme Activities in Paw Tissue
4.6.5. Histopathology
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Mozafari, N.; Azadi, S.; Mehdi-Alamdarlou, S.; Ashrafi, H.; Azadi, A. Inflammation: A bridge between diabetes and COVID-19, and possible management with sitagliptin. Med. Hypotheses 2020, 143, 110111. [Google Scholar] [CrossRef]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free. Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef]
- Leick, M.; Azcutia, V.; Newton, G.; Luscinskas, F.W. Leukocyte recruitment in inflammation: Basic concepts and new mechanistic insights based on new models and microscopic imaging technologies. Cell Tissue Res. 2014, 355, 647–656. [Google Scholar] [CrossRef]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive Oxygen Species in Inflammation and Tissue Injury. Antioxidants Redox Signal. 2014, 20, 1126–1167. [Google Scholar] [CrossRef] [PubMed]
- Schieber, M.; Chandel, N.S. ROS Function in Redox Signaling and Oxidative Stress. Curr. Biol. 2014, 24, R453–R462. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-L.; Korivi, M.; Lin, M.-K.; Chang, H.C.-W.; Wu, C.-R.; Lee, M.-S.; Chen, W.T.-L.; Hseu, Y.-C. Antihemolytic and antioxidant properties of pearl powder against 2,2′-azobis(2-amidinopropane) dihydrochloride-induced hemolysis and oxidative damage to erythrocyte membrane lipids and proteins. J. Food Drug Anal. 2017, 25, 898–907. [Google Scholar] [CrossRef]
- Ali, N. Elevated level of C-reactive protein may be an early marker to predict risk for severity of COVID-19. J. Med. Virol. 2020, 92, 2409–2411. [Google Scholar] [CrossRef] [PubMed]
- Sproston, N.R.; Ashworth, J.J. Role of C-Reactive Protein at Sites of Inflammation and Infection. Front. Immunol. 2018, 9, 754. [Google Scholar] [CrossRef] [PubMed]
- Geronikaki, A.A.; Gavalas, A.M. Antioxidants and inflammatory disease: Synthetic and natural antioxidants with anti-inflammatory activity. Comb. Chem. High Throughput Screen. 2006, 9, 425–442. [Google Scholar] [CrossRef]
- Rezq, S.; Mahmoud, M.; El-Shazly, A.; El Raey, M.; Sobeh, M. Anti-Inflammatory, Antipyretic, and Analgesic Properties of Potamogeton perfoliatus Extract: In Vitro and In Vivo Study. Molecules 2021, 26, 4826. [Google Scholar] [CrossRef]
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 177. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Carvalho, A.; Heimfarth, L.; Pereira, E.; Oliveira, F.S.; Menezes, I.; Coutinho, H.D.M.; Picot, L.; Antoniolli, A.R.; Quintans, L.; Quintans-Júnior, L.J. Phytol, a Chlorophyll Component, Produces Antihyperalgesic, Anti-inflammatory, and Antiarthritic Effects: Possible NFκB Pathway Involvement and Reduced Levels of the Proinflammatory Cytokines TNF-α and IL-6. J. Nat. Prod. 2020, 83, 1107–1117. [Google Scholar] [CrossRef]
- Cavalcanti, M.R.M.; Passos, F.R.S.; Monteiro, B.S.; Gandhi, S.R.; Heimfarth, L.; Lima, B.S.; Nascimento, Y.M.; Duarte, M.C.; Araujo, A.A.S.; Menezes, I.R.A.; et al. HPLC-DAD-UV analysis, anti-inflammatory and anti-neuropathic effects of methanolic extract of Sideritis bilgeriana (lamiaceae) by NF-κB, TNF-α, IL-1β and IL-6 involvement. J. Ethnopharmacol. 2021, 265, 113338. [Google Scholar] [CrossRef]
- Van Valkenburg, J.; Bunyapraphatsara, N. Plant resources of South-East Asia No. 12 (2): Medicinal and poisonous plants 2. Backhuys Publishers. Nord. J. Bot. 2001, 21, 580. [Google Scholar]
- Zahidin, N.S.; Saidin, S.; Zulkifli, R.M.; Muhamad, I.I.; Ya’Akob, H.; Nur, H. A review of Acalypha indica L. (Euphorbiaceae) as traditional medicinal plant and its therapeutic potential. J. Ethnopharmacol. 2017, 207, 146–173. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, M.; Jebanesan, A.; Reetha, D.; Amsath, R.; Pushpanathan, T.; Samidurai, K. Antibacterial activity of Acalypha indica L. Eur. Rev. Med. Pharmacol. Sci. 2008, 12, 299–302. [Google Scholar] [PubMed]
- Kameswari, S.; Narayanan, A.L.; Rajeshkumar, S. Free radical scavenging and anti-inflammatory potential of Acalypha indica mediated selenium nanoparticles. Drug Invent. Today 2020, 13, 106–112. [Google Scholar]
- Ravi, S.; Shanmugam, B.; Subbaiah, G.V.; Prasad, S.H.; Reddy, K.S. Identification of food preservative, stress relief compounds by GC–MS and HR-LC/Q-TOF/MS; evaluation of antioxidant activity of Acalypha indica leaves methanolic extract (in vitro) and polyphenolic fraction (in vivo). J. Food Sci. Technol. 2017, 54, 1585–1596. [Google Scholar] [CrossRef]
- Rao, K.V.B.; Priya, C.L. Postprandial antihyperglycemic and antioxidant activities of Acalypha indica Linn stem extract: An in-vivo study. Pharmacogn. Mag. 2016, 12, 475–S481. [Google Scholar] [CrossRef]
- Patil, K.R.; Mahajan, U.B.; Unger, B.S.; Goyal, S.N.; Belemkar, S.; Surana, S.J.; Ojha, S.; Patil, C.R. Animal Models of Inflammation for Screening of Anti-inflammatory Drugs: Implications for the Discovery and Development of Phytopharmaceuticals. Int. J. Mol. Sci. 2019, 20, 4367. [Google Scholar] [CrossRef]
- El Jemli, M.; Kamal, R.; Marmouzi, I.; Zerrouki, A.; Cherrah, Y.; Alaoui, K. Radical-Scavenging Activity and Ferric Reducing Ability of Juniperus thurifera (L.), J. oxycedrus (L.), J. phoenicea (L.) and Tetraclinis articulata (L.). Adv. Pharmacol. Sci. 2016, 2016, 1–6. [Google Scholar] [CrossRef]
- Masella, R.; Di Benedetto, R.; Varì, R.; Filesi, C.; Giovannini, C. Novel mechanisms of natural antioxidant compounds in biological systems: Involvement of glutathione and glutathione-related enzymes. J. Nutr. Biochem. 2005, 16, 577–586. [Google Scholar] [CrossRef]
- Quan, V.V.; Nam, P.C.; Thong, N.M.; Trung, N.T.; Phan, C.-T.D.; Mechler, A. Antioxidant Motifs in Flavonoids: O–H versus C–H Bond Dissociation. ACS Omega 2019, 4, 8935–8942. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M. Free Radicals in Biology and Medicine; Oxford University Press: Oxford, UK, 2015; p. 905. [Google Scholar]
- Korivi, M.; Chen, C.-T.; Yu, S.-H.; Ye, W.; Cheng, I.-S.; Chang, J.-S.; Kuo, C.-H.; Hou, C.-W. Seaweed Supplementation Enhances Maximal Muscular Strength and Attenuates Resistance Exercise-Induced Oxidative Stress in Rats. Evid. Based Complementary Altern. Med. 2019, 2019, 3528932–3528939. [Google Scholar] [CrossRef]
- Chen, K.-N.; Peng, W.-H.; Hou, C.-W.; Chen, C.-Y.; Chen, H.-H.; Kuo, C.-H.; Korivi, M. Codonopsis javanica root extracts attenuate hyperinsulinemia and lipid peroxidation in fructose-fed insulin resistant rats. J. Food Drug Anal. 2013, 21, 347–355. [Google Scholar] [CrossRef]
- Krewski, D.; Acosta, D.; Andersen, M.; Anderson, H.; Bailar, J.C.; Boekelheide, K.; Brent, R.; Charnley, G.; Cheung, V.G.; Green, S.; et al. Toxicity Testing in the 21st Century: A Vision and a Strategy. J. Toxicol. Environ. Health Part B 2010, 13, 51–138. [Google Scholar] [CrossRef] [PubMed]
- Posadas, I.; Bucci, M.; Roviezzo, F.; Rossi, A.; Parente, L.; Sautebin, L.; Cirino, G. Carrageenan-induced mouse paw oedema is biphasic, age-weight dependent and displays differential nitric oxide cyclooxygenase-2 expression. Br. J. Pharmacol. 2004, 142, 331–338. [Google Scholar] [CrossRef]
- Sugimoto, M.A.; Sousa, L.; Pinho, V.; Perretti, M.; Teixeira, M.M. Resolution of Inflammation: What Controls Its Onset? Front. Immunol. 2016, 7, 160. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Dudeja, P.K.; Tobacman, J.K. Carrageenan-induced NFκB activation depends on distinct pathways mediated by reactive oxygen species and Hsp27 or by Bcl10. Biochim. Biophys. Acta Bioenerg. 2008, 1780, 973–982. [Google Scholar] [CrossRef]
- Winter, C.A.; Risley, E.A.; Nuss, G.W. Carrageenin-Induced Edema in Hind Paw of the Rat as an Assay for Antiinflammatory Drugs. Exp. Biol. Med. 1962, 111, 544–547. [Google Scholar] [CrossRef]
- Perianayagam, J.B.; Sharma, S.; Pillai, K. Anti-inflammatory activity of Trichodesma indicum root extract in experimental animals. J. Ethnopharmacol. 2006, 104, 410–414. [Google Scholar] [CrossRef]
- Duwiejua, M.; Woode, E.; Obiri, D. Pseudo-akuammigine, an alkaloid from Picralima nitida seeds, has anti-inflammatory and analgesic actions in rats. J. Ethnopharmacol. 2002, 81, 73–79. [Google Scholar] [CrossRef]
- Handy, R.L.C.; Moore, P.K. A comparison of the effects of L-NAME, 7-NI and L-NIL on carrageenan-induced hindpaw oedema and NOS activity. Br. J. Pharmacol. 1998, 123, 1119–1126. [Google Scholar] [CrossRef] [PubMed]
- Sonmez, O.; Sonmez, M. Role of platelets in immune system and inflammation. Porto Biomed. J. 2017, 2, 311–314. [Google Scholar] [CrossRef] [PubMed]
- Black, S.; Kushner, I.; Samols, D. C-reactive protein. J. Biol. Chem. 2004, 279, 48487–48490. [Google Scholar] [CrossRef] [PubMed]
- Sato, A.; Oe, K.; Yamanaka, H.; Yokoyama, I.; Ebina, K. C-reactive protein specifically enhances platelet-activating factor-induced inflammatory activity in vivo. Eur. J. Pharmacol. 2014, 745, 46–51. [Google Scholar] [CrossRef]
- Pan, Z.-Y.; Wang, H. Synergistic interaction between choline and aspirin against acute inflammation induced by carrageenan and lipopolysaccharide. Int. Immunopharmacol. 2014, 20, 229–237. [Google Scholar] [CrossRef]
- Willis, E.F.; Clough, G.F.; Church, M.K. Investigation into the mechanisms by which nedocromil sodium, frusemide and bumetanide inhibit the histamine-induced itch and flare response in human skin in vivo. Clin. Exp. Allergy 2004, 34, 450–455. [Google Scholar] [CrossRef]
- Belfort, R.; Berria, R.; Cornell, J.; Cusi, K. Fenofibrate Reduces Systemic Inflammation Markers Independent of Its Effects on Lipid and Glucose Metabolism in Patients with the Metabolic Syndrome. J. Clin. Endocrinol. Metab. 2010, 95, 829–836. [Google Scholar] [CrossRef]
- Mak, K.-K.; Shiming, Z.; Balijepalli, M.K.; Dinkova-Kostova, A.T.; Epemolu, O.; Mohd, Z.; Pichika, M.R. Studies on the mechanism of anti-inflammatory action of swietenine, a tetranortriterpenoid isolated from Swietenia macrophylla seeds. Phytomed. Plus 2021, 1, 100018. [Google Scholar] [CrossRef]
- Barth, C.R.; Funchal, G.A.; Luft, C.; de Oliveira, J.R.; Porto, B.N.; Donadio, M.V.F. Carrageenan-induced inflammation promotes ROS generation and neutrophil extracellular trap formation in a mouse model of peritonitis. Eur. J. Immunol. 2016, 46, 964–970. [Google Scholar] [CrossRef] [PubMed]
- Del Maestro, R. Free Radicals as Mediators of Tissue Injury. Acta Physiol Scand Suppl. 1980, 492, 43–57. [Google Scholar] [PubMed]
- Halici, Z.; Dengiz, G.O.; Odabasoglu, F.; Suleyman, H.; Cadirci, E.; Halici, M. Amiodarone has anti-inflammatory and anti-oxidative properties: An experimental study in rats with carrageenan-induced paw edema. Eur. J. Pharmacol. 2007, 566, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Ben Khedir, S.; Mzid, M.; Bardaa, S.; Moalla, D.; Sahnoun, Z.; Rebai, T. In vivo evaluation of the anti-tnflammatory effect of Pistacia lentiscus fruit oil and its effects on oxidative stress. Evid. Based Complementary Altern. Med. 2016, 2016, 6108203. [Google Scholar] [CrossRef] [PubMed]
- Cordaro, M.; Siracusa, R.; Fusco, R.; D’Amico, R.; Peritore, A.F.; Gugliandolo, E.; Genovese, T.; Scuto, M.; Crupi, R.; Mandalari, G.; et al. Cashew (Anacardium occidentale L.) Nuts Counteract Oxidative Stress and Inflammation in an Acute Experimental Model of Carrageenan-Induced Paw Edema. Antioxidants 2020, 9, 660. [Google Scholar] [CrossRef]
- Tamaddonfard, E.; Farshid, A.A.; Hosseini, L. Crocin alleviates the local paw edema induced by histamine in rats. Avicenna J. Phytomed. 2012, 2, 97. [Google Scholar]
- Naik, R.; Nemani, H.; Pothani, S.; Pothana, S.; Satyavani, M.; Qadri, S.S.; Srinivas, M.; Parim, B. Obesity-alleviating capabilities of Acalypha indica, Pergulari ademia and Tinospora cardifolia leaves methanolic extracts in WNIN/GR-Ob rats. J. Nutr. Intermed. Metab. 2019, 16, 100090. [Google Scholar] [CrossRef]
- Santos, F.; Rao, V.S. A Study of the Anti-pyretic Effect of Quinine, an Alkaloid Effective Against Cerebral Malaria, on Fever Induced by Bacterial Endotoxin and Yeast in Rats. J. Pharm. Pharmacol. 2011, 50, 225–229. [Google Scholar] [CrossRef]
- Alblihed, M.A. Astragalin attenuates oxidative stress and acute inflammatory responses in carrageenan-induced paw edema in mice. Mol. Biol. Rep. 2020, 47, 6611–6620. [Google Scholar] [CrossRef]
- Shukla, A.; Vats, S.; Shukla, R. Phytochemical Screening, proximate analysis and antioxidant activity of Dracaena reflexa lam. leaves. Indian J. Pharm. Sci. 2015, 77, 640–644. [Google Scholar] [CrossRef]
- Gutierrez, R.M.P.; Navarro, Y.T.G. Antioxidant and hepatoprotective effects of the methanol extract of the leaves ofSatureja macrostema. Pharmacogn. Mag. 2010, 6, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-C.; Yang, M.-H.; Wen, H.-M.; Chern, J.-C. Estimation of total flavonoid content in propolis by two complementary colometric methods. J. Food Drug Anal. 2002, 10, 3. [Google Scholar] [CrossRef]
- Gyamfi, M.A.; Yonamine, M.; Aniya, Y. Free-radical scavenging action of medicinal herbs from Ghana: Thonningia sanguinea on experimentally-induced liver injuries. Gen. Pharmacol. Vasc. Syst. 1999, 32, 661–667. [Google Scholar] [CrossRef]
- Nagai, T.; Myoda, T.; Nagashima, T. Antioxidative activities of water extract and ethanol extract from field horsetail (tsukushi) Equisetum arvense L. Food Chem. 2005, 91, 389–394. [Google Scholar] [CrossRef]
- Esmaeili, M.A.; Sonboli, A. Antioxidant, free radical scavenging activities of Salvia brachyantha and its protective effect against oxidative cardiac cell injury. Food Chem. Toxicol. 2010, 48, 846–853. [Google Scholar] [CrossRef]
- Upadhyay, R.; Chaurasia, J.K.; Tiwari, K.N.; Singh, K. Antioxidant Property of Aerial Parts and Root ofPhyllanthus fraternusWebster, an Important Medicinal Plant. Sci. World J. 2014, 2014, 1–7. [Google Scholar] [CrossRef]
- Adnan; Chy, N.U.; Kamal, A.M.; Barlow, J.W.; Faruque, M.O.; Yang, X.; Uddin, S.B. Evaluation of anti-nociceptive and anti-inflammatory activities of the methanol extract of Holigarna caustica (Dennst.) Oken leaves. J. Ethnopharmacol. 2019, 236, 401–411. [Google Scholar] [CrossRef]
- Sathya, M.; Kokilavani, R.; Ananta Teepa, K. Acute and subacute toxicity studies of ethanolic extract of Acalypha indica Linn in male Wistar albino rats. Asian J. Pharm. Clin. Res. 2012, 5, 97–100. [Google Scholar]
- Killari, K.N.; Prasad, K.; Talluri, M.R.; Bokam, Y.K.; Nadiminti, S.R.; Kommavari, C.S. Antiinflammatory Activity of Wheat Grass Fortified with Cow Urine Distillate. Indian J. Pharm. Sci. 2019, 81, 521–526. [Google Scholar] [CrossRef]
- Misra, H.P.; Fridovich, I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J. Biol. Chem. 1972, 247, 3170–3175. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, I.; Mannervik, B. Glutathione reductase. Methods Enzymol. 1985, 113, 484–490. [Google Scholar] [PubMed]
- Flohé, L.; Günzler, W.A. Assays of glutathione peroxidase. Methods Enzymol. 1984, 105, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Slaoui, M.; Fiette, L. Histopathology Procedures: From Tissue Sampling to Histopathological Evaluation. In Methods in Molecular Biology; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2011; Volume 691, pp. 69–82. [Google Scholar]

| No | Compounds Identified by HR-LC-MS | RT | Formula | Mass | Hbds | Hbas | Mut | Tum | Irri | RepTox | PubChem CID | Percentage |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Oxtriphylline | 0.42 | C12H22N5O3 | 284.1715 | 1 | 4 | No | No | No | No | 656652 | 0.677 |
| 2 | Choline | 0.501 | C5H14NO | 104.1074 | 1 | 2 | No | No | No | No | 6209 | 0.847 |
| 3 | Bumetanide | 0.612 | C17H20N2O5S | 364.1112 | 3 | 7 | No | No | Yes | No | 2471 | 0.847 |
| 4 | Indospicine | 0.612 | C7H15N3O2 | 173.1165 | 4 | 4 | No | No | Yes | No | 108010 | 0.776 |
| 5 | 10-Hydroxy-2E,8E-Decadiene-4,6-diynoic acid | 0.613 | C10H12O3 | 180.0772 | 2 | 2 | No | No | Yes | No | 9543616 | 0.745 |
| 6 | 2,6-Piperidinedicarboxylic acid | 0.615 | C7H11NO4 | 173.069 | 2 | 4 | No | No | No | Yes | 557515 | 0.881 |
| 7 | Ala Ser His | 0.616 | C12H19N5O5 | 313.1392 | 6 | 7 | No | No | No | No | - | 0.644 |
| 8 | Arg Pro | 0.656 | C11H21N5O3 | 271.1652 | 5 | 7 | No | No | No | No | - | 0.677 |
| 9 | His Gly Val | 0.658 | C13H21N5O4 | 311.1577 | 5 | 6 | No | No | No | No | - | 0.627 |
| 10 | Pro Ile | 0.662 | C11H20N2O3 | 228.1459 | 4 | 4 | No | No | No | No | - | 0.661 |
| 11 | Fenofibrate | 0.669 | C20H21ClO4 | 360.116 | 0 | 3 | No | No | No | No | 3339 | 0.711 |
| 12 | Mycophenolic Acid | 0.671 | C17H20O6 | 320.1239 | 2 | 5 | No | No | No | No | 446541 | 0.881 |
| 13 | Mimosine | 0.673 | C8H10N2O4 | 198.0642 | 3 | 6 | low | No | No | No | 440473 | 0.911 |
| 14 | Dimethylglycine | 0.684 | C4H9NO2 | 103.0634 | 1 | 3 | No | No | No | No | 673 | 0.640 |
| 15 | 3,7-Epoxycaryophyllan-6-one | 0.688 | C15H24O2 | 236.1759 | 0 | 2 | No | No | No | No | 4330530 | 0.745 |
| 16 | l-2-Aminoadipic acid | 0.778 | C6H11NO4 | 161.0686 | 3 | 5 | No | No | No | No | 92136 | 0.474 |
| 17 | Trans-4-Hydroxy-L-proline | 0.869 | C5H9NO3 | 131.058 | 3 | 4 | No | No | No | No | 5810 | 0.372 |
| 18 | N2-Succinylglutamic acid | 0.883 | C9H13NO7 | 247.0694 | 4 | 7 | No | No | No | No | 25244383 | 0.406 |
| 19 | Mucronulatol((+/−)) | 1.157 | C17H18O5 | 302.111 | 2 | 5 | No | No | No | Yes | 442811 | 0.372 |
| 20 | Propionylglycine methyl ester | 1.698 | C6H11NO3 | 145.0738 | 1 | 2 | No | No | No | No | - | 1.186 |
| 21 | Ile Tyr | 1.744 | C15H22N2O4 | 294.1575 | 4 | 6 | No | No | No | No | - | 0.847 |
| 22 | Phendimetrazine | 1.855 | C12H17NO | 191.131 | 0 | 2 | No | No | No | No | 30487 | 1.016 |
| 23 | 4-(2-Hydroxy-3-isopropylaminopropyl)benzoic acid | 2.163 | C13H19NO4 | 253.1313 | 3 | 5 | No | No | No | No | - | 0.406 |
| 24 | Mexiletine | 2.219 | C11H17NO | 179.1309 | 0 | 1 | No | No | Yes | No | 4178 | 0.406 |
| 25 | N-(3-Oxo-octanoyl)homoserine lactone | 2.753 | C12H19NO4 | 241.1335 | 1 | 3 | No | No | Yes | No | 127293 | 0.474 |
| 26 | Retusoquinone | 3.071 | C11H12O | 160.0872 | 0 | 1 | No | No | No | No | 6111564 | 1.694 |
| 27 | Peucenin | 3.385 | C15H16O4 | 260.103 | 2 | 4 | No | No | No | No | 68477 | 0.949 |
| 28 | Dehydrovariabilin | 3.494 | C17H14O4 | 282.0845 | 0 | 3 | No | No | No | Yes | 624785 | 1.050 |
| 29 | Normeperidine | 3.598 | C14H19NO2 | 233.1414 | 1 | 2 | No | No | No | No | 32414 | 0.881 |
| 30 | Thr Ala Ser | 3.605 | C10H19N3O6 | 277.1311 | 6 | 7 | No | No | No | No | - | 1.016 |
| 31 | Diethyl Oxalpropionate | 3.711 | C9H14O5 | 202.0842 | 0 | 3 | No | No | No | No | 97750 | 0.881 |
| 32 | 3-O-Methylrimiterol | 3.763 | C13H19NO3 | 237.1366 | 3 | 4 | No | No | No | No | - | 1.118 |
| 33 | Citrulline n-butyl ester | 4.092 | C10H21N3O3 | 231.1582 | 3 | 3 | No | No | Yes | No | 6426971 | 0.542 |
| 34 | 2(Benzylmethylamino)Ethanol N-Oxide | 4.126 | C10H15NO2 | 181.1102 | 1 | 2 | No | No | No | No | 264996 | 0.372 |
| 35 | Chloramphenicol alcohol | 4.177 | C11H14N2O6 | 270.0845 | 4 | 6 | Yes | No | No | No | 10107147 | 0.406 |
| 36 | 12-Aminododecanoic acid | 4.41 | C12H25NO2 | 215.1891 | 2 | 3 | No | No | No | No | 69661 | 0.745 |
| 37 | 3-Oxa-9azoniatricyclo[3.3.1.02,4]non ane, 9-ethyl-9-methyl-7[(phenylacetyl)oxy] | 4.522 | C18H24NO3 | 302.1757 | 1 | 4 | No | No | No | No | - | 0.542 |
| 38 | 3,11-Dihydroxy myristoic acid | 4.835 | C14H28O4 | 260.2002 | 3 | 4 | No | No | No | No | 5282923 | 0.881 |
| 39 | Avocadene acetate | 4.835 | C19H36O4 | 328.2622 | 2 | 3 | No | No | Yes | No | 3624980 | 0.881 |
| 40 | Sulindac sulfide | 4.881 | C20H17FO2S | 340.0902 | 1 | 2 | No | No | No | No | 5352624 | 1.013 |
| 41 | 6-Methoxyquinoline | 4.928 | C10H9NO | 159.0686 | 0 | 2 | Yes | No | No | No | 14860 | 1.288 |
| 42 | Edrophonium | 4.928 | C10H16NO | 166.1233 | 1 | 1 | No | No | No | No | 3202 | 1.220 |
| 43 | Quinine | 4.939 | C20H24N2O2 | 324.1837 | 1 | 4 | No | No | No | No | 3034034 | 1.152 |
| 44 | 3-[2(Dimethylamino)ethoxy]phen ylmethyl]-4-methyl-phenol | 5.11 | C18H23NO2 | 285.173 | 1 | 3 | No | No | No | No | - | 1.050 |
| 45 | Metyrapol | 5.301 | C14H16N2O | 228.1292 | 1 | 3 | No | No | No | No | 161210 | 0.949 |
| 46 | Oxprenolol | 5.322 | C15H23NO3 | 265.1682 | 2 | 7 | No | No | Yes | No | 4631 | 1.118 |
| 47 | Oxyphencyclimine | 5.541 | C20H28N2O3 | 344.2094 | 1 | 6 | No | No | No | No | 4642 | 0.986 |
| 48 | Oxycodone | 6.166 | C18H21NO4 | 315.1486 | 1 | 9 | No | No | No | No | 5284603 | 0.406 |
| 49 | Ergoline-8-methanol, 10-methoxy-1,6-dimethyl | 6.279 | C18H24N2O2 | 300.1858 | 1 | 3 | No | No | No | Yes | - | 0.847 |
| 50 | Evoxine | 6.332 | C18H21NO6 | 347.136 | 2 | 10 | No | No | No | No | 73416 | 0.542 |
| 51 | 2-Hydroxytrimipramine | 6.339 | C20H26N2O | 310.2038 | 1 | 3 | No | No | No | No | 160610 | 0.474 |
| 52 | Penbutolol | 6.575 | C18H29NO2 | 291.2196 | 2 | 5 | Yes | No | No | No | 37464 | 1.152 |
| 53 | Ala Lys Ile | 6.581 | C15H30N4O4 | 330.23 | 7 | 10 | No | No | No | No | - | 1.322 |
| 54 | Oxymorphone | 6.76 | C17H19NO4 | 301.131 | 2 | 9 | No | No | No | No | 5284604 | 0.334 |
| 55 | Diamorphine (heroin) | 6.762 | C21H23NO5 | 369.1566 | 0 | 7 | No | No | No | Yes | 5462328 | 0.406 |
| 56 | Terbinafine metabolite | 6.911 | C19H23NO3 | 313.1672 | 3 | 5 | No | No | No | No | - | 1.098 |
| 57 | Strychnine | 6.912 | C21H22N2O2 | 334.1672 | 0 | 5 | No | No | No | No | 441071 | 1.050 |
| 58 | Hydrolysis product of bussein | 7.021 | C32H40O14 | 648.241 | 8 | 26 | No | No | low | No | 54699399 | 1.145 |
| 59 | Ambelline | 7.026 | C18H21NO5 | 331.1412 | 0 | 9 | Yes | No | No | No | 25092366 | 1.321 |
| 60 | 2-Pyrrolidinone, 4-(2aminoethyl)-1-ethyl-3,3diphenyl-(AHR 5904) | 7.039 | C20H24N2O | 308.1882 | 2 | 3 | No | No | No | No | 164917 | 1.023 |
| 61 | Glu Val Asp | 7.221 | C14H23N3O8 | 361.1518 | 7 | 17 | No | No | No | No | - | 0.953 |
| 62 | 10-Nitro,9Z,12Zoctadecadienoic acid | 7.339 | C18H31NO4 | 325.2246 | 1 | 8 | No | No | No | No | 5282259 | 0.994 |
| 63 | 5β-Chol-2-en-24oic Acid | 7.397 | C24H38O2 | 358.2954 | 1 | 4 | No | No | No | No | - | 0.912 |
| 64 | 5α-Cholan-24oic Acid | 7.733 | C24H40O2 | 360.311 | 1 | 4 | No | No | No | No | 5283802 | 0.832 |
| 65 | Etioporphyrin III | 7.793 | C32H38N4 | 478.317 | 2 | 4 | No | No | No | No | 141310 | 0.814 |
| 66 | Coproporphyrin II | 8.066 | C36H38N4O8 | 654.2548 | 6 | 10 | No | No | No | No | 165072 | 0.964 |
| 67 | Arg Ala Arg | 8.473 | C15H31N9O4 | 401.2552 | 15 | 15 | No | No | No | No | - | 0.456 |
| 68 | (5Z)-4,4-difluoro1alpha,25-dihydroxyvitamin D3/(5Z)-4,4-difluoro1alpha, 25dihydroxycholecalcifer | 8.766 | C27H42F2O3 | 452.304 | 3 | 6 | No | No | No | No | - | 0.754 |
| 69 | Arg Val Pro | 8.887 | C16H30N6O4 | 370.2321 | 8 | 12 | No | No | No | No | - | 1.101 |
| 70 | Dihydrodeoxystreptomycin | 9.017 | C21H41N7O11 | 567.2862 | 16 | 29 | No | No | No | No | 11953824 | 1.021 |
| 71 | Swietenine | 9.019 | C32H40O9 | 568.2704 | 1 | 10 | No | No | No | No | 76327465 | 1.134 |
| 72 | C16 Sphinganine | 9.686 | C16H35NO2 | 273.266 | 3 | 3 | No | No | No | No | 5283572 | 0.786 |
| 73 | (Z)-N-(2hydroxyethyl)hexadec-7- | 11.085 | 297.2658 | 2 | 2 | No | No | No | No | - | 0.354 | |
| 74 | N-Hexadecyl-l-hydroxyproline | 11.684 | C21H41NO3 | 355.307 | 3 | 4 | No | No | No | No | 66690736 | 0.834 |
| 75 | Trihexyphenidyl N-oxide | 12.227 | C20H31NO2 | 317.2342 | 1 | 2 | No | No | No | No | 129318457 | 0.965 |
| 76 | Zearalenone | 12.772 | C18H22O5 | 318.1478 | 2 | 4 | No | No | No | No | 5281576 | 0.236 |
| 77 | GPEtn(10:0/11:0)[U] | 12.813 | C26H52NO8P | 537.3399 | 2 | 4 | No | No | No | No | - | 0.875 |
| 78 | 25-Hydroxycholesterol(d3) | 12.829 | C27H43D3O2 | 405.3607 | 2 | 2 | No | No | low | No | - | 0.823 |
| 79 | Disopyramide | 13.729 | C21H29N3O | 339.2395 | 1 | 3 | No | No | No | No | 3114 | 0.787 |
| 80 | 12-Hydroxy-10octadecynoic acid | 13.763 | C18H32O3 | 296.2341 | 2 | 3 | No | No | No | No | - | 0.867 |
| 81 | Anandamide (18:3, n-6) | 14.295 | C20H35NO2 | 321.2652 | 2 | 2 | No | No | No | No | 5283445 | 0.564 |
| 82 | 9-Hydroxy-12-oxo10-octadecenoic acid | 14.4 | C18H32O4 | 312.2287 | 2 | 4 | No | No | No | No | 5282968 | 0.753 |
| 83 | 13-OxoODE | 14.417 | C18H30O3 | 294.2184 | 1 | 3 | No | No | Yes | No | 5283012 | 0.700 |
| 84 | 2R-hydroxy9Z,12Z,15Z-octadecatrienoic acid | 14.648 | C18H30O3 | 294.2188 | 2 | 3 | No | No | No | No | 16061058 | 1.032 |
| 85 | Lactone of PGFMUM | 14.85 | C16H24O5 | 296.1612 | 1 | 4 | No | No | No | No | - | 0.854 |
| 86 | N Acetylsphingosine | 15.403 | C20H39NO3 | 341.2915 | 3 | 3 | No | No | No | No | 5497136 | 1.132 |
| 87 | Phytosphingosine | 16.285 | C18H39NO3 | 317.2918 | 4 | 4 | No | No | No | No | 122121 | 0.126 |
| 88 | Dihydroceramide C2 | 16.767 | C20H41NO3 | 343.3073 | 3 | 3 | No | No | No | No | 6610273 | 0.453 |
| 89 | 3-Deacetyl Khivorin | 16.926 | C30H40O9 | 544.2667 | 1 | 5 | No | No | No | No | 46878892 | 0.763 |
| 90 | Harderoporphyrin | 17.931 | C35H36N4O6 | 608.2594 | 5 | 8 | No | No | No | No | 3081462 | 0.998 |
| 91 | His Arg Val | 18.001 | C17H30N8O4 | 410.2411 | 8 | 9 | No | No | No | No | - | 0.324 |
| 92 | Harderoporphyrinogen | 18.177 | C35H42N4O6 | 614.3115 | 4 | 9 | No | No | Yes | No | 193825 | 0.231 |
| 93 | Ramipril glucuronide | 18.404 | C29H40N2O11 | 592.2643 | 5 | 10 | No | No | No | No | - | 1.563 |
| 94 | Deoxykhivorin | 18.791 | C32H42O9 | 570.282 | 0 | 4 | No | No | No | No | 6708722 | 0.232 |
| 95 | Antimycin A (A1 shown) | 19.021 | C27H38N2O9 | 534.2592 | 3 | 6 | No | No | Yes | No | 12550 | 1.324 |
| 96 | N-(2hydroxyethyl)icosanamide | 19.051 | C22H45NO2 | 355.3432 | 2 | 2 | No | No | No | No | 3787294 | 1.021 |
| 97 | 9,14,19,19,19Pentadeuterio-1α,25dihydroxyprevitamin D3 / 9,14,19,19,19-pentadeuterio1alpha,25 | 19.24 | C27H39D5O3 | 421.355 | 1 | 1 | No | No | low | No | - | 1.240 |
| 98 | 2Tricosanamidoethanesulfonic acid | 19.241 | C25H51NO4S | 461.3489 | 2 | 4 | No | No | No | No | - | 1.200 |
| 99 | Trandolapril glucuronide | 20.16 | C30H42N2O11 | 606.2793 | 5 | 10 | No | No | No | No | 92023960 | 0.932 |
| 100 | 2,4,6-Trimethyl-2,15tetracosadienoic acid | 20.184 | C27H50O2 | 406.3821 | 1 | 2 | No | No | No | No | - | 0.901 |
| 101 | 3β,6α,7αTrihydroxy-5β-cholan-24oic Acid | 20.754 | C24H40O5 | 408.2845 | 4 | 5 | No | No | No | No | - | 0.875 |
| Compounds Identified by GC-MS | ||||||||||||
| 1 | Piperidine-2,5-dione | 8.97 | C5H7NO2 | 113.047 | 1 | 2 | No | No | No | No | 533930 | 3.733 |
| 2 | Phenol, 2,4-bis(1,1-dimethylethyl)- | 12.53 | C14H22O | 206.167 | 1 | 1 | No | No | No | No | 528937 | 1.333 |
| 3 | Tridecanoic acid,12-methyl-, methyl ester | 14.92 | C15H30O2 | 242.224 | 0 | 1 | No | No | No | No | 21204 | 1.200 |
| 4 | 1-Oxacyclopentadecan-2-one,15,15-dimethyl | 15.63 | C16H30O2 | 254.224 | 0 | 1 | No | No | No | No | - | 0.800 |
| 5 | 11-Hexadecenoic acid, methyl ester | 16.88 | C17H32O2 | 268.24 | 0 | 1 | No | No | No | No | 5364696 | 1.040 |
| 6 | Pentadecanoic acid, 14-methyl-, methyl ester | 17.08 | C17H34O2 | 270.255 | 0 | 1 | No | No | No | No | 21205 | 1.493 |
| 7 | Octadecanoic acid | 17.78 | C19H38O2 | 298.287 | 0 | 1 | No | No | No | No | 5281 | 2.053 |
| 8 | 13-Hexyloxacyclotridec-10-en-2-one | 18.52 | C18H30O2 | 278.224 | 0 | 1 | No | No | No | No | 5369119 | 1.066 |
| 9 | 10-Octadecenoic acid,methyl ester | 18.82 | C19H36O2 | 296.271 | 0 | 1 | No | No | No | No | 5364425 | 1.954 |
| 10 | Octadecanoic acid,methyl ester | 19.05 | C19H38O2 | 298.287 | 0 | 1 | No | No | No | No | 8201 | 0.956 |
| 11 | Dasycarpidan-1-methanol,acetate (ester) | 20.37 | C20H26N2O2 | 326.44 | 1 | 2 | No | No | No | No | 550072 | 0.543 |
| 12 | Aspidospermidine-3-carboxylic acid, 2,3-didehydro-,methyl ester,[5a,12a,19a]- | 20.65 | C21H26N2O2 | 338.199 | 1 | 3 | No | No | No | No | - | 2.123 |
| 13 | Cephalotaxine, 3-deoxy-3,11-epoxy-, (3a,11a)- | 22.96 | C18H19NO4 | 313.131 | 0 | 5 | No | No | No | No | - | 2.666 |
| 14 | 4-H-1-Benzopyran-4-one, 7-(acetyloxy)-2-(4-(acetyloxy)phenyl)-5-methoxy- | 25.6 | C19H14O7 | 354.073 | 1 | 5 | Yes | No | No | Yes | - | 2.456 |
| Conc. (µg/mL) | Scavenging of DPPH Radical (%) | Scavenging of Hydroxyl Radical (%) | Conc. (µg/mL) | Inhibition of Hydrogen Peroxide (%) | Inhibition of Lipid Peroxidation (%) | ||||
| AIRME | Ascorbic Acid | AIRME | Ascorbic Acid | AIRME | Ascorbic Acid | AIRME | Ascorbic Acid | ||
| 10 | 41.02 ± 0.58 | 50.38 ± 0.40 | 46.17 ± 0.77 | 53.81 ± 0.85 | 20 | 22.53 ± 1.41 | 42.68 ± 0.76 | 20.68 ± 1.72 | 33.06 ± 2.85 |
| 20 | 56.92 ± 1.15 | 61.46 ± 0.66 | 53.82 ± 1.13 | 61.88 ± 1.19 | 40 | 41.73 ± 0.70 | 55.88 ± 1.13 | 31.03 ± 2.97 | 43.34 ± 2.65 |
| 30 | 62.82 ± 0.59 | 70.36 ± 0.99 | 60.74 ± 1.61 | 68.48 ± 0.86 | 60 | 51.00 ± 1.17 | 59.44 ± 0.83 | 40.80 ± 2.10 | 50.77 ± 1.98 |
| 40 | 66.02 ± 0.80 | 77.04 ± 1.21 | 64.69 ± 1.30 | 82.36 ± 1.04 | 80 | 58.31 ± 1.09 | 62.49 ± 0.80 | 52.58 ± 1,87 | 58.66 ± 2.10 |
| 50 | 68.46 ± 0.70 | 78.88 ± 1.65 | 71.48 ± 0.63 | 84.41 ± 1.61 | 100 | 63.20 ± 1.20 | 68.85 ± 0.68 | 57.75 ± 2.06 | 65.25 ± 3.06 |
| IC50 | 15.85 | 5.71 | 14.74 | 5.28 | IC50 | 65.40 | 33.31 | 79.70 | 59.45 |
| Groups | WBC (109/L) | Platelets (109/L) | CRP (ng/mL) |
|---|---|---|---|
| NC | 3.95 ± 2.06 | 320 ± 4.94 | 28.93 ± 2.95 |
| DC | 14.48 ± 4.76 $ | 1025 ± 8.67 $ | 55.94 ± 4.86 $ |
| AIRME | 4.02 ± 2.14 | 343 ± 5.07 | 30.32 ± 3.06 |
| AIRME-L+C | 9.76 ± 2.75 * | 765 ± 6.06 * | 36.98 ± 3.24 * |
| AIRME-H+C | 4.89 ± 2.43 ** | 366 ± 5.51 ** | 31.84 ± 3.095 ** |
| ST | 4.37 ± 2.14 ** | 337 ± 5.06 ** | 30.94 ± 3.065 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sahukari, R.; Punabaka, J.; Bhasha, S.; Ganjikunta, V.S.; Kondeti Ramudu, S.; Kesireddy, S.R.; Ye, W.; Korivi, M. Phytochemical Profile, Free Radical Scavenging and Anti-Inflammatory Properties of Acalypha Indica Root Extract: Evidence from In Vitro and In Vivo Studies. Molecules 2021, 26, 6251. https://doi.org/10.3390/molecules26206251
Sahukari R, Punabaka J, Bhasha S, Ganjikunta VS, Kondeti Ramudu S, Kesireddy SR, Ye W, Korivi M. Phytochemical Profile, Free Radical Scavenging and Anti-Inflammatory Properties of Acalypha Indica Root Extract: Evidence from In Vitro and In Vivo Studies. Molecules. 2021; 26(20):6251. https://doi.org/10.3390/molecules26206251
Chicago/Turabian StyleSahukari, Ravi, Jyothi Punabaka, Shanmugam Bhasha, Venkata Subbaiah Ganjikunta, Shanmugam Kondeti Ramudu, Sathyavelu Reddy Kesireddy, Weibing Ye, and Mallikarjuna Korivi. 2021. "Phytochemical Profile, Free Radical Scavenging and Anti-Inflammatory Properties of Acalypha Indica Root Extract: Evidence from In Vitro and In Vivo Studies" Molecules 26, no. 20: 6251. https://doi.org/10.3390/molecules26206251
APA StyleSahukari, R., Punabaka, J., Bhasha, S., Ganjikunta, V. S., Kondeti Ramudu, S., Kesireddy, S. R., Ye, W., & Korivi, M. (2021). Phytochemical Profile, Free Radical Scavenging and Anti-Inflammatory Properties of Acalypha Indica Root Extract: Evidence from In Vitro and In Vivo Studies. Molecules, 26(20), 6251. https://doi.org/10.3390/molecules26206251









