A Global Review on Short Peptides: Frontiers and Perspectives †
Abstract
1. Introduction
2. Brief History
3. Short Peptides: Definition
4. Frontiers and Prospects of Short Peptides
4.1. Advantages vs. Disadvantages: SWOT Analysis
4.2. To Overcome Shortcomings of Peptides: Mission (Im)possible?
4.2.1. Constrained Amino-Acids as a Molecular “Meccano”
4.2.2. Cyclic Peptides and Mimetics
4.2.3. Ultra-Short Peptides: Less Is More
4.2.4. Nanoengineering and a Supramolecular Approach
5. Synthesis
5.1. Advances in the Synthesis of Short Peptides and Modified Amino Acids
5.2. Short Difficult Peptide Synthesis
6. In Silico Studies
6.1. Geometry Optimization, Conformational Analysis
6.2. Modelling of Short Peptides
6.3. Peptide Interactions with Lipid Bilayers Using Molecular Dynamics Simulations
6.3.1. Peptide Affinity Dependency on Membrane Composition
6.3.2. Free Energy Calculations through a Peptide Reaction Path
6.3.3. Cooperative Effects
7. Peptide-Based Therapies
7.1. Monocyclic, Bicyclic and Tricyclic Cell-Penetrating Peptides as Molecular Transporters
7.2. Short Peptides in Gene Delivery
7.2.1. Targeting Peptides in GDSs
7.2.2. Cell Penetrating Peptides in GDSs
7.2.3. Endosome-Disruptive Peptides in GDSs
7.2.4. Nuclear Localization Peptides in GDSs
7.3. Taking Peptide Aptamers to a New Level
7.4. Peptide-Based Vaccines
7.5. The Role of Short Peptides in Neurodegenerative Therapy
7.6. Immune Modulation Using Altered Peptide Ligands in Autoimmune Diseases
7.7. Relevance of Short Peptides in Stem Cell Research
7.8. Short Peptide-Based Anti-Viral Agents against SARS-CoV-2
7.9. Antimicrobial Lactoferrin-Based Peptides as Anti-COVID-19
7.10. Peptides from Digestion of Proteins
7.11. Nutraceuticals
7.12. Marine Peptides
7.13. Peptide-Based Cosmeceuticals
8. Conclusions and Future Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| A | Acidic |
| ACE(I) | angiotensin-converting enzyme (inhibitors) |
| AchRs | acetylcholine receptors |
| AIDS | acquired immune deficiency syndrome |
| Alloc | allyloxycarbonyl |
| AMPs | antimicrobial peptides |
| APCs | antigen presenting cells |
| APL | altered peptide ligands |
| ARB | angiotensin receptor blockers |
| B | basic |
| BAP | bioactive peptides |
| BOC | butoxycarbonyl |
| Bzl | benzyl |
| CG | coarse grain |
| COVID-19 | coronavirus disease 2019 |
| CPPs | cell penetrating peptides |
| CSD | Cambridge Structure Database |
| CTL | cytotoxic T lymphocyte |
| 2D | two-dimensional |
| 3D | three-dimensional |
| DH | degree of hydrolysis |
| DKP | diketopiperazines |
| DNA | deoxyribonucleic acid |
| EDTA | ethylenediaminetetraacetic acid |
| FAS | fatty acid synthase |
| FDA | Food and Drug Administration |
| FMOC | fluorenylmethoxycarbonyl |
| GALA | glutamic acid–alanine–leucine–alanine |
| GAS | group A streptococcus |
| GDS | gene delivery system |
| GFR | growth factor receptors |
| HeLa | human cervical cancer cell line |
| HIV | human immunodeficiency virus |
| HMG | high motility group |
| HMGR | 3-hydroxy-3-methylglutaryl CoA reductase |
| HOAt | 1-hydroxy-7-aza-benzotriazole |
| HOBt | 1-hydroxy-benzotriazole |
| HPLC | high performance liquid chromatography |
| IFN | interferon |
| IUPAC | International Union of Pure and Applied Chemistry |
| LDL | low-density lipoprotein |
| LDLR | LDL receptor |
| LF | lactoferrin |
| LPS | lipopolysaccharide |
| LRPs | low density lipoprotein receptors |
| LRs | leptin receptors |
| MBP | myelin basic protein |
| MD | molecular dynamics |
| MHC | major histocompatibility complex |
| NF | nuclear factor |
| NLSs | nuclear localization signals |
| NPC | nuclear pore complex |
| PDC | pyruvate dehydrogenase complexes |
| PEG | polyethylene glycol |
| PES | potential energy surface |
| PPI | proton-pump inhibitors |
| RAS | renin-angiotensin system |
| RCSB PDB | Research Collaboratory for Structural Bioinformatics Protein Data Bank |
| RGD | arginine–glycine–aspartic acid |
| RNA | ribonucleic acid |
| SARS-COV-2 | Severe Acute Respiratory Syndrome Coronavirus |
| SPPS | solid-phase synthesis |
| SREBP2 | sterol regulatory element-binding protein 2 |
| SWOT | strengths, weaknesses, opportunities, and threats |
| tBu | tert-butyl |
| TFA | trifluoroacetic acid |
| TFRs | transferrin receptors |
| TMC | trimethyl chitosan |
| TNF | tumor necrosis factor alpha |
| TNFR | tumor necrosis factor receptor 1 |
References
- Muheem, A.; Shakeel, F.; Jahangir, M.A.; Anwar, M.; Mallick, N.; Jain, G.K.; Warsi, M.H.; Ahmad, F.J. A review on the strategies for oral delivery of proteins and peptides and their clinical perspectives. Saudi Pharm. J. 2016, 24, 413–428. [Google Scholar] [CrossRef]
- Fruton, J.S. Chapter 5-Emil Fischer and Franz Hofmeister. Contrasts in Scientific Style: Research Groups in the Chemical and Biochemical Sciences. Am. Philos. Soc. 1990, 191, 163–165. [Google Scholar]
- Mulder, G.J. Sur la composition de quelques substances animales. Bull. Sci. Phys. Nat. Neerl. 1838, 104, 1–192. [Google Scholar]
- Hartley, H. Origin of the world protein. Nature 1951, 168, 244. [Google Scholar] [CrossRef]
- Hamley, I.W. Introduction to Peptide Science; Wiley: Weinheim, Germany, 2020; ISBN 978-1-119-69817-3. [Google Scholar]
- Reynolds, J.A.; Tanford, C. Nature’s Robots: A History of Proteins; Oxford University Press: New York, NY, USA, 2003; p. 15. [Google Scholar]
- Vickery, H.B.; Schmidt, C.L. The history of the discovery of the amino acids. Chem. Rev. 1931, 9, 169–318. [Google Scholar] [CrossRef]
- Hansen, S. Die Entdeckung der Proteinogenen Aminosauren von 1805 in Paris bis 1935 in Illinois. 2015. Available online: https://www.arginium.de/wp-content/uploads/2015/12/Aminosäuren-Entdeckungseschichte (accessed on 15 June 2016).
- Vauquelin, L.N.; Robiquet, P.J. The discovery of a new plant principle in Asparagus sativus. Ann. Chim. 1806, 57, 88–93. [Google Scholar]
- Anfinsen, C.B.; Edsall, J.T.; Richards, F.M. The formation and stabilization of protein structure. Adv. Protein Chem. 1972, 99, 1–424. [Google Scholar] [CrossRef]
- Paal, C. Ueber die Einwirkung von phenyl-i-cyanat auf organische Aminosauren. In Berichte der Deutschen Chemischen Gesellschaft; Wiley Online Library: Berlin, Germany, 1894; pp. 974–979. [Google Scholar]
- Harper, D. Amino Online Etymology Dictionary. 2010. Available online: https://www.etymonline.com/ (accessed on 15 January 2021).
- Chandrudu, S.; Simerska, P.; Toth, I. Chemical methods for peptide and protein production. Molecules 2013, 18, 4373–4388. [Google Scholar] [CrossRef]
- Fischer, E.; Fourneau, E. Ueber einige derivate des Glykocolls. Eur. J. Inorg. Chem. 1901, 34, 2868–2877. [Google Scholar] [CrossRef]
- Wieland, T.; Bodanszky, M. The World of Peptides; Springer: Berlin/Heidelberg, Germany, 1991. [Google Scholar]
- Grant, G.A. Synthetic Peptides: A User’s Guide, 2nd ed.; Oxfprd University Press: New York, NY, USA, 2002; pp. 1–9. ISBN 9780195132618. [Google Scholar]
- Bojarska, J.; Kaczmarek, K.; Zabrocki, J.; Wolf, W.M. Amino Acids: Molecules of life. Int. J. Nutr. Sci. 2019, 4, 1035–1037. [Google Scholar]
- Frenkel-Pinter, M.; Samanta, M.; Ashenasy, G.; Leman, L.J. Prebiotic pepitdes: Molecular hibs in the origin of life. Chem. Rev. 2020, 120, 4707–4765. [Google Scholar] [CrossRef]
- Muchowska, K.; Moran, J. Peptide synthesis at the origin of life. Science 2020. [Google Scholar] [CrossRef]
- Schiller, M.R. The minimotif synthesis hypothesis for the origin of life. J. Transl. Sci. 2016, 2, 289–296. [Google Scholar] [CrossRef]
- Greenwald, J.; Kwiatkowski, W.; Riek, R. Peptide amyloids in the origin of life. J. Mol. Biol. 2018, 430, 3735–3750. [Google Scholar] [CrossRef]
- IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN) Nomenclature and Symbolism for Amino Acids and Peptides. Eur. J. Biochem. 1984, 138, 9–37. [CrossRef]
- Mirabeau, O. Searching for Novel Peptide Hormones in the Human Genome. Life Sciences [q-bio]. Ph.D. Thesis, Université Montpellier II-Sciences et Techniques du Languedoc, Montpellier, France, 2008. [Google Scholar]
- Su, M.; Ling, Y.; Yu, J.; Wu, J.; Xiao, J. Small proteins: Untapped area of potential biological importance. Front Genet. 2013, 4, 286–295. [Google Scholar] [CrossRef]
- Schmidt, A.; Teeter, M.; Weckert, E.; Lamzin, V.S. Crystal structure of small protein crambin at 0.48 A resolution. Acta Cryst. F 2011, 67, 424–428. [Google Scholar] [CrossRef]
- Patel, V.; Asatryan, B.; Siripanthong, B.; Munroe, P.B.; Tiku-Owens, A.; Lopes, L.R.; Khanji, M.Y.; Protonotarios, A.; Santangeli, P.; Muser, D.; et al. State of the art review on genetics and precision medicine in arrhythmogenic cardiomyopathy. Int. J. Mol. Sci. 2020, 21, 6615–6662. [Google Scholar] [CrossRef]
- Habault, J.; Poyet, J.L. Recent advances in cell penetrating peptide-based anticancer therapies. Molecules 2019, 24, 927–944. [Google Scholar] [CrossRef]
- Derakhshankhah, H.; Jafari, S. Cell penetrating peptides: A concise review with emphasis on biomedical applications. Biomed. Pharmacother. 2018, 108, 1090–1096. [Google Scholar] [CrossRef]
- Fisher, F.; Pavlenko, K.; Vlasov, A.; Ramenskaya, G. Peptide-based therapeutics for oncology. Pharm. Med. 2019, 33, 9–20. [Google Scholar] [CrossRef]
- Pavan, S.; Berti, F. Short peptides as biosensors transducers. Anal. Bioanal. Chem. 2012, 402, 3055–3070. [Google Scholar] [CrossRef] [PubMed]
- E-kobon, T.; Thongararm, P.; Roytrakul, S.; Meesuk, L.; Chumnanpuen, P. Prediction of anticancer peptides against MCF-7 breast cancer cells from the peptidomes of Achatina fulica mucus fractions. Comp. Struct. Biotechnol. J. 2016, 4, 49–57. [Google Scholar] [CrossRef]
- Horsley, J.R.; Jovcevski, B.; Wegener, K.L.; Yu, J.; Pukala, T.L.; Abell, A.D. Rationally designed peptide-based inhibitor of A-beta42 fibril formation and toxicity: A potential therapeutic strategy for Alzheimer’s disease. Biochem. J. 2020, 477, 2039–2054. [Google Scholar] [CrossRef]
- Baig, M.H.; Ahmad, K.; Rabbani, G.; Choi, I. Use of peptides for the management of Alzheimer’s disease: Diagnosis and inhibition. Front. Aging Neurosci. 2018, 10. [Google Scholar] [CrossRef]
- Hamley, I.W. Small bioactive peptides for biomaterials design and therapeutics. Chem. Rev. 2017, 117, 14015–14041. [Google Scholar] [CrossRef] [PubMed]
- Hauser, C.A.E.; Deng, R.; Mishra, A.; Loo, Y.; Khoe, U.; Zhuang, F.; Cheong, D.W.; Accardo, A.; Sullivan, M.B.; Riekel, C.; et al. Natural tri- to hexapeptides self-assemble in water to amyloid beta-type fiber aggregates by unexpected alpha-helical intermediate structures. Proc. Natl. Acad. Sci. USA 2011, 108, 1361–1366. [Google Scholar] [CrossRef]
- Seow, W.Y.; Hauser, C.A.E. Short to ultrashort peptide hydrogels for biomedical uses. Mater. Today 2014, 17, 381–388. [Google Scholar] [CrossRef]
- Makovitzki, A.; Avrahami, D.; Shai, Y. Ultrashort antibacterial and antifungal lipopetides. Proc. Natl. Acad. Sci. USA 2006, 103, 15997–16002. [Google Scholar] [CrossRef]
- Ni, M.; Zhuo, S. Applications of self-assembling ultrashort peptides in bionanotechnology. RSC Adv. 2019, 9, 844–852. [Google Scholar] [CrossRef]
- Fosgerau, K.; Hoffmann, T. Peptide therapeutics: Current status and future directions. Drug Discov. Today 2015, 20, 122–128. [Google Scholar] [CrossRef]
- Soudy, R.; Kimura, R.; Patel, A.; Fu, W.; Kaur, K.; Westaway, D.; Yang, J.; Jhamandas, J. Short amylin receptor antagonist peptides improve memory deficits in Alzheimer’s disease mouse model. Sci. Rep. 2019, 9, 10942–10953. [Google Scholar] [CrossRef]
- Morimoto, B.H. Therapeutic peptides for CNS indications: Progress and challenges. Bioorganic Med. Chem. 2018, 26, 2859–2862. [Google Scholar] [CrossRef]
- Haggag, Y.A.; Donia, A.A.; Osman, M.A.; El-Gizawy, S.A. Peptides as drug candidates: Limitations and recent development perspectives. Biomed. J. Sci. Tech. Res. 2018, 8, 6659–6663. [Google Scholar] [CrossRef]
- Henninot, A.; Collins, J.C.; Nuss, J.M. The current state of peptide drug discovey: Back to the future? J. Med. Chem. 2018, 61, 1382–1414. [Google Scholar] [CrossRef]
- Lee, A.C.; Harris, J.L.; Khanna, K.K.; Hong, J.H. A Comprehensive Review on Current Advances in Peptide Drug Development and Design. Int. J. Mol. Sci. 2019, 20, 2383–2404. [Google Scholar] [CrossRef]
- Qian, Z.; Liu, T.; Liu, Y.-Y.; Briesewitz, R.; Barrios, A.M.; Jhiang, S.M. Efficient delivery of cyclic peptides into mammalian cells with short sequence motifs. ACS Chem. Biol. 2013, 8, 423–431. [Google Scholar] [CrossRef]
- Taylor, R.E.; Zahid, M. Cell penetrating peptides, novel vectors for gene therapy. Pharmaceutics 2020, 12, 225–246. [Google Scholar] [CrossRef]
- Ellert-Miklaszewska, A.; Poleszak, K.; Kaminska, B. Short peptides interfering with signaling pathways as new therapeutic tools for cancer treatments. Future Med. Chem. 2017, 9, 199–221. [Google Scholar] [CrossRef]
- Lenci, E.; Trabocchi, A. Peptidomimetic toolbox for drug discovery. Chem. Soc. Rev. 2020, 49, 3262–3277. [Google Scholar] [CrossRef]
- Zhang, R.; Leeper, C.N.; Wang, X.; White, T.A.; Ulery, B.D. Immunomodulatory vasoactive intestinal peptide amphiphile micelles. Biomater. Sci. 2018, 6, 1717–1722. [Google Scholar] [CrossRef] [PubMed]
- Kalafatovic, D.; Giralt, E. Cell-penetrating peptides: Design strategies beyond primary structure and amphipathicity. Molecules 2017, 22, 1929. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.D.; Moriya, J.; Carle, V.; Pojer, F.; Abriata, L.A.; Deyle, K.; Heinis, C. De novo development of proteolytically resistant therapeutic peptides for oral administration. Nat. Biomed. Eng. 2020, 4, 560–571. [Google Scholar] [CrossRef] [PubMed]
- Brayden, D.J. Evolving peptides for oral intake. Nat. Biomed. Eng. 2020, 4, 487–488. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.D.; Whitehead, K.A.; Mitragotri, S. Materials for oral delivery of proteins and peptides. Nat. Rev. Mater. 2020, 5, 127–148. [Google Scholar] [CrossRef]
- Benedetti, E.; Pedone, C.; Saviano, M. Structure-Activity Relationships in Peptides: From Modelling to Rational Drug Design. Front. Drug Des. Discov. 2007, 3, 2007–2539. [Google Scholar]
- Di Blasio, B.; Pavone, V.; Saviano, M.; Lombardi, A.; Nastri, F.; Pedone, C.; Benedetti, E.; Crisma, M.; Anzolin, M.; Toniolo, C. Structural Characterization of the α-bend Ribbon Spiral: Crystallographic Analysis of two long (L-Pro-Aib)n Sequential Peptides. J. Am. Chem. Soc. 1992, 114, 6273–6278. [Google Scholar] [CrossRef]
- Pavone, V.; Lombardi, A.; Saviano, M.; Di Blasio, B.; Nastri, F.; Fattorusso, R.; Zaccaro, L.; Maglio, O.; Yamada, T.; Omote, Y.; et al. Mixed conformation in Cα,α-disubstituted tripeptides: X-ray Crystal structure of Z-Aib-Dph-Gly-OMe and Bz-Dph-Dph-Gly-Ome. Biopolymers 1994, 34, 1595–1604. [Google Scholar] [CrossRef]
- Moretto, V.; Formaggio, F.; Crisma, M.; Bonora, G.M.; Toniolo, C.; Benedetti, E.; Santini, A.; Saviano, M.; Di Blasio, B.; Pedone, C. Preferred Conformation of Peptides Rich in Ac8c, a Medium-ring Alicyclic Cα,α-disubstituted Glycine. J. Pep. Sci. 1996, 2, 14–27. [Google Scholar]
- Benedetti, E.; Iacovino, R.; Saviano, M. The Use of Uncoded α-Amino Acidds Residues in Drug Design in Proceedings of 24th Crystallographic Course; Codding, P.W., Ed.; ESCOM: Leiden, The Netherlands, 1998; pp. 103–112. [Google Scholar]
- Toniolo, C.; Crisma, M.; Formaggio, F.; Benedetti, E.; Santini, A.; Iacovino, R.; Saviano, M.; Di Blasio, B.; Pedone, C.; Kamphuis, J. Preferred conformation of Peptides Rich in Alicyclic Cα,α disubstituted Glycine. Biopolymers 1996, 40, 519–522. [Google Scholar] [CrossRef]
- Gatos, M.; Formaggio, F.; Crisma, M.; Valle, G.; Toniolo, C.; Bonora, G.M.; Saviano, M.; Iacovino, R.; Menchise, V.; Galdiero, S.; et al. Conformational Characterization of Peptides Rich in the Cycloaliphatic Cα,α -disubstituted Glycine 1-Amino cyclononane-1-carboxylic Acid. J. Pept. Sci. 1997, 3, 367–382. [Google Scholar] [CrossRef]
- Benedetti, E.; Di Blasio, B.; Iacovino, R.; Menchise, V.; Saviano, M.; Pedone, C.; Bonora, G.M.; Ettorre, A.; Graci, L.; Formaggio, F.; et al. Conformation Restriction through Ci↔Ci cyclication: 1-amminocycloheptane-1-carboxylic acid (Ac7c). J. Chem. Soc. Perkin 2 1997, 2, 2023–2032. [Google Scholar] [CrossRef]
- Pavone, V.; Lombardi, A.; Saviano, M.; Nastri, F.; Zaccaro, L.; Maglio, O.; Pedone, C.; Omote, Y.; Yamanaka, Y.; Yamada, T. Conformational Behaviour of C−Diphenyl Glycine. Folded Versus Extended Structures in D g Containng Tripeptides. J. Pept. Sci. 1998, 4, 21–32. [Google Scholar] [CrossRef]
- Saviano, M.; Iacovino, R.; Menchise, V.; Benedetti, E.; Bonora, G.M.; Gatos, M.; Graci, L.; Formaggio, F.; Crisma, M.; Toniolo, C. Conformational Restriction through Ci↔Ci Cyclization: Ac12c, the Largest Cycloaliphatic Cα,α-Disubstituted Glycine Known. Biopolymers 2000, 53, 200–212. [Google Scholar] [CrossRef]
- Moretto, A.; Formaggio, F.; Crisma, M.; Toniolo, C.; Saviano, M.; Iacovino, R.; Vitale, R.M.; Benedetti, E. Ac10c: A medium-ring, cycloaliphatic C−disubstituted glycine. Incorporation into model peptides and preferred conformation. J. Pept. Res. 2001, 57, 307–315. [Google Scholar] [CrossRef]
- Formaggio, F.; Crisma, M.; Toniolo, C.; Broxterman, Q.B.; Kaptein, B.; Corbier, C.; Saviano, M.; Palladino, P.; Benedetti, E. C−methyl, C-n-propylglycine homo-oligomers. Macromolecules 2003, 36, 8164–8170. [Google Scholar] [CrossRef]
- Crisma, M.; Saviano, M.; Moretto, A.; Broxterman, Q.B.; Kaptein, B.; Toniolo, C. Peptide α/310-helix dimorphism in the crystal state. J. Am. Chem. Soc. 2007, 129, 15471–15473. [Google Scholar] [CrossRef]
- Saviano, M.; Aida, M.; Corongiu, G. Molecular Dynamics Simulation in vacuo and in solution of Cyclolinopeptide A: A Conformational Study. Biopolymers 1991, 31, 1017–1024. [Google Scholar] [CrossRef]
- Di Blasio, B.; Rossi, F.; Benedetti, E.; Pavone, V.; Saviano, M.; Pedone, C.; Zanotti, G.; Tancredi, T. Bioactive Peptide: X-Ray and NMR Conformational Study of [Aib5,6-Dala8] Cyclolinopeptide A. J. Am. Chem. Soc. 1992, 114, 8277–8283. [Google Scholar] [CrossRef]
- Saviano, M.; Rossi, F.; Di Blasio, B.; Pavone, V.; Pedone, C. Molecular Dynamics Simulation in vacuo and in solution of [Aib5,6-Dala8] Cyclolinopeptide A. A Conformational and Comparative Study. J. Biomol. Struct. Dyn. 1992, 9, 1045–1060. [Google Scholar] [CrossRef]
- Rossi, F.; Saviano, M.; Di Talia, P.; Di Blasio, B.; Pedone, C.; Zanotti, G.; Mosca, M.; Saviano, G.; Tancredi, T.; Ziegler, K.; et al. Solution and Solid State Structure of an Aib-Containing Cyclodecapeptide Inhibiting the Cholate Uptake in Hepatocytes. Biopolymers 1996, 40, 465–478. [Google Scholar] [CrossRef]
- Becker, E.L.; Freer, R.J.; Toniolo, C.; Balaram, P. Membrane Receptors and Cellular Regulation; Czech, M.P., Kahn, C.R., Eds.; Liss: New York, NY, USA, 1985; pp. 129–134. [Google Scholar]
- Gatto, E.; Bocchinfuso, G.; Palleschi, A.; Oncea, S.; De Zotti, M.; Formaggio, F.; Toniolo, C.; Venanzi, M. 3D Structure, Dynamics, and Activity of Synthetic Analog of the Peptaibiotic Trichodecenin I. Chem. Biodivers. 2013, 10, 887–903. [Google Scholar] [CrossRef]
- Venanzi, M.; Gatto, E.; Formaggio, F.; Toniolo, C. The importance of being Aib. Aggregation and self-assembly studies on conformationally constrained oligopeptides. J. Pep. Sci. 2017, 23, 104–116. [Google Scholar] [CrossRef] [PubMed]
- De Zotti, M.; Muzzi, B.; Gatto, E.; Di Napoli, B.; Mazzuca, C.; Palleschi, A.; Placidi, E.; Formaggio, F.; Toniolo, C.; Venanzi, M. Tuning the Morphology of Nanostructured Peptide Films by the Introduction of a Secondary Structure Conformational Constraint: A Case Study of Hierarchical Self-Assembly. J. Phys. Chem. B. 2018, 122, 6305–6313. [Google Scholar] [CrossRef]
- Moore, G.; Smith, J.; Baylis, B.; Matsoukas, J. Design and pharmacology of peptide mimetics. Adv. Pharmacol. 1995, 33, 91–141. [Google Scholar]
- Katsara, M.; Deraos, G.; Tselios, T.; Matsoukas, M.; Friligou, I.; Matsoukas, J.; Apostolopoulos, V. Design and Synthesis of a Cyclic Double Mutant Peptide (cyclo(87−99)[A91,A96] MBP87−99) Induces Altered Responses in Mice after Conjugation to Mannan: Implications in the Immunotherapy of Multiple Sclerosis. J. Med. Chem. 2009, 52, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Katsara, M.; Tselios, T.; Deraos, S.; Deraos, G.; Matsoukas, M.; Lazoura, E.; Matsoukas, J.; Apostolopoulos, V. Round and round we go: Cyclic peptides in disease. Curr. Med. Chem. 2006, 13, 2221–2232. [Google Scholar] [PubMed]
- Matsoukas, J.; Hondrelis, J.; Agelis, G.; Barlos, K.; Gatos, D.; Ganter, R.; Moore, D.; Moore, G. Novel synthesis of cyclic amide-linked analogs of angiotensins II and III. J. Med. Chem. 1994, 37, 2958–2969. [Google Scholar] [CrossRef]
- Matsoukas, J.; Hondrelis, J.; Keramida, M.; Mavromoustakos, T.; Makriyannis, A.; Yamdagni, R.; Wu, Q.; Moore, G.J. Role of the NH2-terminal domain of angiotensin II (ANG II) and [Sar1] angiotensin II on conformation and activity. NMR evidence for aromatic ring clustering and peptide backbone folding compared with [des-1,2,3] angiotensin II. J. Med. Chem. 1994, 269, 5303–5312. [Google Scholar]
- Matsoukas, J.; Polevaya, L.; Ancans, J.; Mavromoustakos, T.; Kolocouris, A.; Roumelioti, P.; Vlahakos, D.; Yamdagni, R.; Wu, Q.; Moore, G. The design and synthesis of a potent angiotensin II cyclic analogue confirms the ring cluster receptor conformation of the hormone angiotensin II. Bioorganic Med. Chem. 2000, 8, 1–10. [Google Scholar] [CrossRef]
- Polevaya, L.; Mavromoustakos, T.; Zoumboulakis, P.; Grdadolnik, S.; Roumelioti, P.; Giatas, N.; Mutule, I.; Keivish, T.; Vlahakos, D.; Iliodromitis, E.; et al. Synthesis and study of a cyclic angiotensin II antagonist analogue reveals the role of π*–π* interactions in the C-terminal aromatic residue for agonist activity and its structure resemblance with AT1 non-peptide antagonists. Bioorganic Med. Chem. 2001, 9, 1639–1647. [Google Scholar] [CrossRef]
- Apostolopoulos, V.; Rostami, A.; Matsoukas, J. The Long Road of Immunotherapeutics against Multiple Sclerosis. Brain Sci. 2020, 10, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Keramida, M.; Tselios, T.; Mantzourani, E.; Papazisis, K.; Mavromoustakos, T.; Klaussen, C.; Agelis, G.; Deraos, S.; Friligou, I.; Habibi, H.; et al. Design, Synthesis, and Molecular Modeling of a Novel Amide-Linked Cyclic GnRH Analogue Cyclo(4−9)[Lys4,d-Trp6,Glu9] GnRH: Stimulation of Gonadotropin Gene Expression. J. Med. Chem. 2006, 49, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Alexopoulos, K.; Alexopoulos, K.; Fatseas, P.; Melissari, E.; Vlahakos, D.; Smith, J.; Mavromoustakos, T.; Saifeddine, M.; Moore, G.; Hollenberg, M.; et al. Design and synthesis of thrombin receptor-derived nonpeptide mimetics utilizing a piperazine scaffold. Bioorganic Med. Chem. 1999, 7, 1033–1041. [Google Scholar] [CrossRef]
- Tseveleki, V.; Tselios, T.; Kanistras, I.; Koutsoni, O.; Karamita, M.; Vamvakas, S.; Apostolopoulos, V.; Dotsika, E.; Matsoukas, J.; Lassmann, H.; et al. Mannan-conjugated myelin peptides prime non-pathogenic Th1 and Th17 cells and ameliorate experimental autoimmune encephalomyelitis. Exp. Neurol. 2015, 267, 254–267. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.; Matsoukas, J. Angiotensin as a model for hormone-receptor interactions. Biosci. Rep. 1985, 5, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Mavromoustakos, T.; Kolocouris, A.; Zervou, M.; Roumelioti, P.; Matsoukas, J.; Weisemann, R. An Effort to Understand the Molecular Basis of Hypertension through the Study of Conformational Analysis of Losartan and Sarmesin Using a Combination of Nuclear Magnetic Resonance Spectroscopy and Theoretical Calculations. J. Med. Chem. 1999, 42, 1714–1722. [Google Scholar] [CrossRef]
- Matsoukas, J.; Agelis, G.; Hondrelis, J.; Yamdagni, R.; Wu, Q.; Gantar, R.; Moore, D.; Moore, G.J.; Smith, J.R. Synthesis and biological activities of angiotensin II, sarilesin, and sarmesin analogs containing Aze or Pip at position 7. J. Med. Chem. 1993, 36, 904–911. [Google Scholar] [CrossRef]
- Matsoukas, J.; Agelis, G.; Wahhab, A.; Hondrelis, J.; Panagiotopoulos, D.; Yamdagni, R.; Wu, Q.; Mavromoustakos, T.; Maia, H. Differences in backbone structure between angiotensin II agonists and type I antagonists. J. Med. Chem. 1995, 38, 4660–4669. [Google Scholar] [CrossRef]
- Turner, R.; Moore, G.; Matsoukas, J. Fluorescence properties of angiotensin II analogues in receptor-simulating environments: Relationship between tyrosinate fluorescence lifetime and biological activity. Biochim. Biophys. Acta (BBA) Biomembr. 1991, 1065, 21–28. [Google Scholar] [CrossRef]
- Agelis, G.; Resvani, A.; Koukoulitsa, C.; Tumova, T.; Slaninova, J.; Kalavrizioti, D.; Spyradaki, K.; Afantitis, A.; Melagraki, G.; Siafaka, A.; et al. Rational design, efficient synthesis and biological evaluation of N,N-symmetrically bis-submituted butylimidazole analogs as a new class potent Angiotensin II receptor blockers. Eur. J. Med. Chem. 2013, 62, 352–370. [Google Scholar] [CrossRef] [PubMed]
- Agelis, G.; Kelaidonis, K.; Resvani, A.; Kalavrizioti, D.; Androutsou, M.E.; Plotas, P.; Vlahakos, D.; Koukoulitsa, C.; Tselios, T.; Mavromoustakos, T.; et al. Facile and efficient syntheses of a series of N-benzyl and N-biphenylmethyl substituted imidazole derivatives based on (E)-urocanic acid, as angiotensin II AT1 receptor blockers. Molecules 2013, 18, 7510–7532. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Zhu, L.; Cai, J.; Lei, F.; Qin, J.J.; Xie, J.; Liu, Y.M.; Zhao, Y.C.; Huang, X.; Lin, L.; et al. Association of inpatient use of angiotensin converting enzyme inhibitors and angiotensin II receptor blockers with mortality among patients with hypertension hospitalized with Covid-19. Circ. Res. 2020, 126, 1671–1681. [Google Scholar] [CrossRef] [PubMed]
- Qaradakhi, T.; Gadanec, L.; Matsoukas, J.; Apostolopoulos, V.; Zulli, A. Could DIZE be the answer to Covid 19? Maturitas 2020, 140, 83–84. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.H.; Xue, B.; Robinson, R.C.; Hauser, C.A.E. Systematic moiety variations of ultrashort peptides produce profound effects on self-assembly, nanostructure formation, hydrogelation, and phase transition. Sci. Rep. 2017, 7, 12897–12908. [Google Scholar] [CrossRef]
- Ni, M. Ultrashort peptides: Minimum number in amino acid residues, maximum number in bioapplications. Revistabionatura 2019, 4, 763–764. [Google Scholar] [CrossRef]
- Dadhwal, S.; Fairhall, J.S.; Goswami, S.K.; Hook, S.; Gamble, A.B. Alkene-azide 1,3-dipolar cycloaddition as a trigger for ultrashort peptide hydrogel dissolution. Chem. Asian J. 2019, 14, 1143–1150. [Google Scholar] [CrossRef]
- Makam, P.; Gazit, E. Minimalistic peptide supramolecular co-assembly: Expanding the conformational space for nanotechnology. Chem. Soc. Rev. 2018, 47, 3406–3420. [Google Scholar] [CrossRef]
- Kurbasic, I.; Parisi, E.; Garcia, A.M.; Marchesan, S. Self-assembling, ultrashort peptide gels as antimicrobial biomaterials. Curr. Top. Med. Chem. 2020, 20, 1300–1309. [Google Scholar] [CrossRef]
- Yadav, N.; Chauhan, M.K.; Chauhan, V.S. Short to ultrashort peptide-based hydrogels as a platform for biomedical applications. Biomater. Sci. 2020, 8, 84–100. [Google Scholar] [CrossRef]
- Ni, M.; Tresset, G.; Iliescu, C.; Hauser, A.E. Ultrashort peptide theranostic nanoparticles by microfluide-assisted rapid solvent exchange. IEE Trans. Nanobiosci. 2020, 19, 627–632. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, G.; Gabrani, R. Antiviral peptides: Identification and validation. Int. J. Pept. Res. Ther. 2020, 18, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Subbarao, N.K.; Parente, R.A.; Szoka, F.C.; Nadasdi, L.; Pongracz, K. The pH-dependent bilayer destabilization by an amphipathic peptide. Biochemistry 1987, 26, 2964–2972. [Google Scholar] [CrossRef] [PubMed]
- Nygren, P.A. Alternative binding proteins: Affibody binding proteins developed from a small three-helix bundle scaffold. FEBS J. 2008, 275, 2668–2676. [Google Scholar] [CrossRef]
- Kim, H.J.; Antel, J.P.; Duquette, P.; Alleva, D.G.; Conlon, P.J.; Bar-Or, A. Persistence of immune responses to altered and native myelin antigens in patients with multiple sclerosis treated with altered peptide ligand. Clin. Immunol. 2002, 104, 105–114. [Google Scholar] [CrossRef]
- Ji, W.; Zhang, C.; Ji, H. Purification, identification and molecular mechanism of two dipeptidyl peptidase IV (DPP-IV) inhibitory peptides from Antarctic krill (Euphausia superba) protein hydrolysate. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2017, 1064, 56–61. [Google Scholar] [CrossRef]
- Martins, M.B.; Carvalho, I. Diketopiperazines: Biological activity and synthesis. Tetrahedron 2007, 63, 9923–9932. [Google Scholar] [CrossRef]
- Feni, L.; Jutten, L.; Parente, S.; Piarulli, U.; Neundorf, I.; Dia, D. Cell-penetrating peptides containing 2,5-DKP scaffolds as shuttles for anti-cancer drugs: Conformational studies and biological activities. Chem. Commun. 2020, 56, 5685–5688. [Google Scholar] [CrossRef]
- Sun, S.J.; Liu, Y.C.; Weng, C.H.; Sun, S.W.; Li, F.; Li, H.; Zhu, H. Cyclic dipeptides mediating quorum sensing and their bilogical effects in Hypsizygus Marmoreus. Biomolecules 2020, 10, 298–311. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, P.; Ma, H.; Zhu, W. Developments around the bioactive diketopiperazines: A patent review. Expert Opin. Ther. Patents. 2013, 32, 1415–1433. [Google Scholar] [CrossRef]
- Borthwick, A.D. 2,5-Diketopiperazines: Synthesis, reactions, medicinal chemistry, and bioactive natural products. Chem. Rev. 2012, 112, 3641–3716. [Google Scholar] [CrossRef]
- Bojarska, J.; Wolf, W.M. Ultra short cyclo-peptides as bio-inspired therapeutics: Proline based 2,5-diketopiperazines (DKP). In Proceedings of the 1st International Electronic Conference on Biomolecules: Natural and Bio-Inspired Therapeutics for Human Diseases, online. 1–13 December 2020; Available online: https://www.mdpi.com/journal/biomolecules/events/12176 (accessed on 15 January 2021).
- Bojarska, J.; Maniukiewicz, W.; Sieron, L.; Remko, M. An orthorhombic polymorph of a cyclization product of perindopril. Acta Cryst. C 2013, 69, 630–633. [Google Scholar] [CrossRef] [PubMed]
- Bojarska, J.; Maniukiewicz, W.; Główka, M.L.; Siero’n, L.; Remko, M. Crystal structure of perindopril cyclization product. J. Chil. Chem. Soc. 2013, 58, 1530–1532. [Google Scholar] [CrossRef]
- Remko, M.; Bojarska, J.; Jezko, L.; Olczak, A.; Maniukiewicz, W. Molecular structure of antihypertensive drug perindopril, its active metabolite perindoprilat and impurity F. J. Mol. Struct. 2013, 1036, 292–297. [Google Scholar] [CrossRef]
- Bojarska, J.; Remko, M.; Wojciechowski, J.; Madura, I.; Kaczmarek, K.; Zabrocki, J.; Zimecki, M.; Wolf, W.M. Cyclic tetrapeptides as promising scaffold for innovative therapeutic agents: Synthesis, crystallographic, biological and in silico studies. Zeitschrift fur Kristallographie. 2020, 40. [Google Scholar]
- Bojarska, J.; Maniukiewicz, W.; Sieron, L.; Kopczacki, P.; Walczynski, K.; Remko, M. Perindoprilat monohydrate. Acta Cryst. C 2012, 68, o443–o446. [Google Scholar] [CrossRef]
- Bojarska, J.; Maniukiewicz, W.; Sieron, L.; Fruzinski, A.; Kopczacki, P.; Walczynski, K.; Remko, M. Novel pseudopolymorph of the active metabolite of perindopril. Acta Cryst. C 2012, 68, o341–o343. [Google Scholar] [CrossRef]
- Bojarska, J.; Maniukiewicz, W.; Fruziński, L.; Sieron, L.; Remko, M. Captopril and its dimer captopril disulfide: Comparative structural and conformational studies. Acta Cryst. C 2015, 71, 199–203. [Google Scholar] [CrossRef]
- Remko, M.; Bojarska, J.; Jezko, J.; Sieron, L.; Olczak, A.; Maniukiewicz, W. Crystal and molecular structure of perindopril erbumine salt. J. Mol. Struct. 2011, 997, 103–109. [Google Scholar] [CrossRef]
- Remko, M.; Bojarska, J.; Remkova, A.; Maniukiewicz, W. Molecular structure and acidity of captopril, zofenopril and their metabolites captopril disulfide and zofenoprilat. Comput. Theor. Chem. 2015, 1062, 50–55. [Google Scholar] [CrossRef]
- Zhao, K.; Xing, R.; Yan, X. Cyclic dipeptides: Biological activities and self-assembled materials. Pept. Sci. 2020, e24202. [Google Scholar] [CrossRef]
- Bojarska, J.; Remko, M.; Breza, M.; Madura, I.; Fruziński, A.; Wolf, W.M. A Proline-Based Tectons and Supramolecular Synthons for Drug Design 2.0: A Case Study of ACEI. Pharm. Des. Enzym. Inhib. Potential Drugs 2020, 13, 338. [Google Scholar] [CrossRef]
- Ying, J.; Lin, R.; Xu, P.; Wu, Y.; Liu, Y.; Zhao, Y. Prebiotic formation of cyclic dipeptides under potentially early Earth conditions. Sci. Rep. 2018, 8, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Burley, S.K.; Berman, H.M. RCSB Protein Data Bank: Biological macromolecular structures enabling researchand education in fundamental biology, biomedicine, biotechnology and energy. Nucleic Acids Res. 2019, 47, D464–D474. [Google Scholar] [CrossRef]
- Levin, A.; Hakala, T.; Schnaider, L.; Lopes Bernardes, G.; Gazit, E.; Knowles, T. Biomimetic peptide self-assembly for functional materials. Nat. Rev. Chem. 2020, 4, 615–634. [Google Scholar] [CrossRef]
- Bojarska, J.; Kaczmarek, K.; Zabrocki, J.; Wolf, W.M. Supramolecular Chemistry of short peptides and modified amino acids. In Advances in Organic Synthesis; Bentham Science Publisher, Ltd.: Sharjah, UAE, 2018; Chapter 12; Volume 11, pp. 43–107. [Google Scholar] [CrossRef]
- Bojarska, J.; Remko, M.; Wojciechowski, J.; Madura, I.; Kaczmarek, K.; Olczak, A.; Zabrocki, J.; Wolf, W.M. Supramolecular synthon polymorphism in modified amino acids. Structural, conformational and energy landscapes of N-benzoyl-2′-hydroxy-3-methylisovaline. J. Mol. Struct. 2019, 1190, 11–22. [Google Scholar] [CrossRef]
- Bojarska, J.; Kaczmarek, K.; Zabrocki, J.; Wolf, W.M. Supramolecular synthons as related to cooperativity in biocomplexes: Towards design and development of oligopeptide-based modern drugs and cosmeceuticals. Novel Approaches Drug Des. Dev. 2019, 5, 23–25. [Google Scholar] [CrossRef]
- Bojarska, J.; Zabrocki, J.; Kaczmarek, K.; Remko, M.; Wolf, W.M. New synthons in supramolecular chemistry of short biologically active peptides. Acta Cryst. A 2019, 75, e588. [Google Scholar] [CrossRef]
- Bojarska, J.; Remko, M.; Breza, M.; Madura, I.; Kaczmarek, K.; Zabrocki, J.; Wolf, W.M. A Supramolecular Approach to Structure-based Design with a Focus on Synthons Hierarchy in Ornithine-derived Ligands: Review, Synthesis, Experimental and in silico Studies. Mol. Struct. Based Des. Biol. Act. Compd. 2020, 25, 1135–1162. [Google Scholar] [CrossRef]
- Bojarska, J.; Remko, M.; Madura, I.; Kaczmarek, K.; Zabrocki, J.; Wolf, W.M. Synthesis, experimental and in silico studies of N-fluorenylmethoxycarbonyl-O-tert-butyl-N-methyltyrosine, coupled with CSD data: A survey of interactions in the crystal structures of Fmoc–amino acids. Acta Cryst. C 2020, 76, 328–345. [Google Scholar] [CrossRef]
- Zhu, J.; Cifuentes, H.; Reynolds, J.; Lamba, D.A. Immunosupression via loss of IL2r-gamma enhances long-term functional integration of hESC-derived photoreceptors in the mouse retina. Cell Stem Cell 2017, 20, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Wang, H.; Shi, B.; Shangguan, L.; Tong, W.; Yu, G. Supramolecular peptide constructed by molecular Lego allowing programmable self-assembly for photodynamic therapy. Nat. Commun. 2019, 10, 24120–24130. [Google Scholar]
- Glossop, H.D.; Heruka De Zoysa, G.; Hemar, Y.; Cardoso, P.; Wang, K.; Lu, J.; Valery, C.; Sarojini, V. Battacin-inspired ultrashort peptides: Nanostructure analysis and antimicronial activity. Biomacromolecules 2019, 20, 2515–2529. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.H.; Lee, W.H.; Ni, M.; Loo, Y.; Hauser, C.A.E. C-terminal residue of ultrashort peptides impacts on molecular self-assembly, hydrogelation, and interaction with small-molecule drugs. Sci. Rep. 2018, 8, 17127–17141. [Google Scholar] [CrossRef]
- Gessner, I.; Neundorf, I. Nanoparticles modified with cell-penetrating peptides: Conjugation mechanisms, physicochemical properties, and application in cancer diagnosis and therapy. Int. J. Mol. Sci. 2020, 21, 2536–2557. [Google Scholar] [CrossRef]
- Li, S.; Zou, Q.; Li, Y.; Yuan, C.; Xing, R.; Yan, X. Smart peptide-based supramolecular photodynamic metallo-nanodrugs designed by multicomponent coordination self-assembly. JACS 2018, 140, 10794–10802. [Google Scholar] [CrossRef]
- Li, S.; Zou, Q.; Xing, R.; Govindaraju, T.; Fakhrullin, R.; Yan, X. Peptide-modulated self-assembly as a versatile strategy for tumor supramolecular nanotheranostics. Theranostics 2019, 9, 3249–3261. [Google Scholar] [CrossRef]
- Kaczmarek, K.; Wojciechowski, J.; Wolf, W.M. (2S,4R)-4-ammonio-5-oxopyrrolidine-2-carboxylate. Acta Cryst. E 2010, 66, 831–838. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.R. Bruce Merrifield and solid-phase peptide synthesis: A historical assessment. Biopolymers 2008, 90, 175–184. [Google Scholar] [CrossRef]
- Subirós-Funosas, R.; Prohens, R.; Barbas, R.; El-Faham, A.; Albericio, F. Oxyma: An efficient additive for peptide synthesis to replace the benzotriazole-based HOBt and HOAt with a lower risk of explosion. Chemistry 2009, 15, 9394–9403. [Google Scholar] [CrossRef]
- El-Faham, A.; Albericio, F. Peptide coupling reagents, more than a letter soup. Chem. Rev. 2011, 111, 6557–6602. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.C.; White, P.D. Fmoc Solid Phase Peptide Synthesis: A Practical Approach; OUP: Oxford, UK, 2000; ISBN 978-0-19-963724-9. [Google Scholar]
- Sheppard, R. The Fluorenylmethoxycarbonyl Group in Solid Phase Synthesis. J. Pept. Sci. 2003, 9, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Haack, T.; Mutter, M. Serine derived oxazolidines as secondary structure disrupting, solubilizing building blocks in peptide synthesis. Tetrahedron Lett. 1992, 33, 1589–1592. [Google Scholar] [CrossRef]
- Sohma, Y.; Sasaki, M.; Hayashi, Y.; Kimura, T.; Kiso, Y. Novel and efficient synthesis of difficult sequence-containing peptides through O-N intramolecular acyl migration reaction of O-acyl isopeptides. Chem. Commun. 2004, 124–125. [Google Scholar] [CrossRef] [PubMed]
- Carpino, L.A.; Krause, E.; Sferdean, C.D.; Schümann, M.; Fabian, H.; Bienert, M.; Beyermann, M. Synthesis of ‘difficult’ peptide sequences: Application of a depsipeptide technique to the Jung-Redemann 10- and 26-mers and the amyloid peptide Aβ(1-42). Tetrahedron Lett. 2004, 45, 7519–7523. [Google Scholar] [CrossRef]
- Mutter, M.; Chandravarkar, A.; Boyat, C.; Lopez, J.; Santos, S.D.; Mandal, B.; Mimna, R.; Murat, K.; Patiny, L.; Saucède, L.; et al. Switch Peptides In Statu Nascendi: Induction of Conformational Transitions Relevant to Degenerative Diseases. Angew. Chem. Int. Ed. 2004, 43, 4172–4178. [Google Scholar] [CrossRef]
- Abedini, A.; Raleigh, D.P. Incorporation of Pseudoproline Derivatives Allows the Facile Synthesis of Human IAPP, a Highly Amyloidogenic and Aggregation-Prone Polypeptide. Org. Lett. 2005, 7, 693–696. [Google Scholar] [CrossRef]
- Harris, P.W.R.; Kowalczyk, R.; Hay, D.L.; Brimble, M.A. A Single Pseudoproline and Microwave Solid Phase Peptide Synthesis Facilitates an Efficient Synthesis of Human Amylin 1-37. Int. J. Pept. Res. Ther. 2013, 19, 147–155. [Google Scholar] [CrossRef]
- Wöhr, T.; Wahl, F.; Nefzi, A.; Rohwedder, B.; Sato, T.; Sun, X.C.; Mutter, M. Pseudo-prolines as a solubilizing, structure-disrupting protection technique in peptide synthesis. J. Am. Chem. Soc. 1996, 118, 9218–9227. [Google Scholar] [CrossRef]
- Tuchscherer, G.; Mutter, M. Peptidomimetics for Bridging Structure and Function: Pseudo-Prolines (ΨPro) in Peptide Synthesis, Molecular Recognition, and Drug Design. Chimia 2001, 55, 306–313. [Google Scholar]
- Skropeta, D.; Jolliffe, K.A.; Turner, P. Pseudoprolines as Removable Turn Inducers: Tools for the Cyclization of Small Peptides. J. Org. Chem. 2004, 69, 8804–8809. [Google Scholar] [CrossRef] [PubMed]
- Postma, T.M.; Albericio, F. Cysteine pseudoprolines for thiol protection and peptide macrocyclization enhancement in Fmoc-based solid-phase peptide synthesis. Org. Lett. 2014, 16, 1772–1775. [Google Scholar] [CrossRef] [PubMed]
- Sohma, Y.; Yoshiya, T.; Taniguchi, A.; Kimura, T.; Hayashi, Y.; Kiso, Y. Development of O-Acyl Isopeptide Method. Biopolymers 2007, 88, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Sohma, Y.; Taniguchi, A.; Skwarczynski, M.; Yoshiya, T.; Fukao, F.; Kimura, T.; Hayashi, Y.; Kiso, Y. ‘O-Acyl isopeptide method’ for the efficient synthesis of difficult sequence-containing peptides: Use of ‘O-acyl isodipeptide unit’. Tetrahedron Lett. 2006, 47, 3013–3017. [Google Scholar] [CrossRef]
- Yoshiya, T.; Taniguchi, A.; Sohma, Y.; Fukao, F.; Nakamura, S.; Abe, N.; Ito, N.; Skwarczynski, M.; Kimura, T.; Hayashi, Y.; et al. ‘O-Acyl isopeptide method’ for peptide synthesis: Synthesis of forty kinds of ‘O-acyl isodipeptide unit’ Boc-Ser/Thr(Fmoc-Xaa)-OH. Org. Biomol. Chem. 2007, 5, 1720–1730. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, A.; Yoshiya, T.; Abe, N.; Fukao, F.; Sohma, Y.; Kimura, T.; Hayashi, Y.; Kiso, Y. ‘O-Acyl isopeptide method’ for peptide synthesis: Solvent effects in the synthesis of Aβ1-42 isopeptide using ‘O-acyl isodipeptide unit’. J. Pept. Sci. 2007, 13, 868–874. [Google Scholar] [CrossRef]
- Yoshiya, T.; Ito, N.; Kimura, T.; Kiso, Y. Isopeptide method: Development of S-acyl isopeptide method for the synthesis of difficult sequence-containing peptides. J. Pept. Sci. 2008, 14, 1203–1208. [Google Scholar] [CrossRef]
- Sohma, Y.; Sasaki, M.; Hayashi, Y.; Kimura, T.; Kiso, Y. Design and synthesis of a novel water-soluble Aβ42 isopeptide: An efficient strategy for the preparation of Alzheimer’s disease-related peptide, Aβ1-42, via O-N intramolecular acyl migration reaction. Tetrahedron Lett. 2004, 45, 5965–5968. [Google Scholar] [CrossRef]
- Yoshiya, T.; Higa, A.; Abe, N.; Fukao, F.; Kuruma, T.; Toda, Y.; Sohma, Y.; Kiso, Y. Click peptide concept: O-acyl isopeptide of islet amyloid polypeptide as a nonaggregative precursor molecule. ChemBioChem 2011, 12, 1216–1222. [Google Scholar] [CrossRef]
- Hussein, W.M.; Liu, T.Y.; Toth, I.; Skwarczynski, M. Microwave-assisted synthesis of difficult sequence-containing peptides using the isopeptide method. Org. Biomol. Chem. 2013, 11, 2370–2376. [Google Scholar] [CrossRef]
- Wu, F.; Mayer, J.P.; Gelfanov, V.M.; Liu, F.; DiMarchi, R.D. Synthesis of Four-Disulfide Insulin Analogs via Sequential Disulfide Bond Formation. J. Org. Chem. 2017, 82, 3506–3512. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Luo, E.Y.; Flora, D.B.; Mezo, A.R. A synthetic route to human insulin using isoacyl peptides. Angew. Chem. Int. Ed. 2014, 53, 3983–3987. [Google Scholar] [CrossRef] [PubMed]
- Karas, J.A.; Scanlon, D.B.; Forbes, B.E.; Vetter, I.; Lewis, R.J.; Gardiner, J.; Separovic, F.; Wade, J.D.; Hossain, M.A. 2-Nitroveratryl as a photocleavable thiol-protecting group for directed disulfide bond formation in the chemical synthesis of insulin. Chem. Eur. J. 2014, 20, 9549–9552. [Google Scholar] [CrossRef] [PubMed]
- Arai, K.; Takei, T.; Shinozaki, R.; Noguchi, M.; Fujisawa, S.; Katayama, H.; Moroder, L.; Ando, S.; Okumura, M.; Inaba, K.; et al. Characterization and optimization of two-chain folding pathways of insulin via native chain assembly. Commun. Chem. 2018, 1, 26–37. [Google Scholar] [CrossRef]
- Kanai, S.; Machida, K.; Masuda, R.; Koide, T. Peptide precursors that acquire denatured collagen-hybridizing ability by O-to-N acyl migration at physiological pH. Org. Biomol. Chem. 2020, 18, 2823–2827. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, A.; Sohma, Y.; Hirayama, Y.; Mukai, H.; Kimura, T.; Hayashi, Y.; Matsuzaki, K.; Kiso, Y. “Click Peptide”: pH-Triggered in Situ Production and Aggregation of Monomer Aβ1-42. ChemBioChem 2009, 10, 710–715. [Google Scholar] [CrossRef]
- Yoshiya, T.; Sohma, Y.; Kimura, T.; Hayashi, Y.; Kiso, Y. ‘O-Acyl isopeptide method’: Racemization-free segment condensation in solid phase peptide synthesis. Tetrahedron Lett. 2006, 47, 7905–7909. [Google Scholar] [CrossRef]
- Coin, I.; Schmieder, P.; Bienert, M.; Beyermann, M. The depsipeptide technique applied to peptide segment condensation: Scope and limitations. J. Pept. Sci. 2008, 14, 299–306. [Google Scholar] [CrossRef]
- Yoshiya, T.; Kawashima, H.; Sohma, Y.; Kimura, T.; Kiso, Y. O-Acyl isopeptide method: Efficient synthesis of isopeptide segment and application to racemization-free segment condensation. Org. Biomol. Chem. 2009, 7, 2894–2904. [Google Scholar] [CrossRef]
- Lecaillon, J.; Gilles, P.; Subra, G.; Martinez, J.; Amblard, M. Synthesis of cyclic peptides via O-N-acyl migration. Tetrahedron Lett. 2008, 49, 4674–4676. [Google Scholar] [CrossRef]
- Yoshiya, T.; Kawashima, H.; Hasegawa, Y.; Okamoto, K.; Kimura, T.; Sohma, Y.; Kiso, Y. Epimerization-free synthesis of cyclic peptide by use of the O-acyl isopeptide method. J. Pept. Sci. 2010, 16, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Leach, A. Molecular Modelling: Principles and Applications, 2nd ed.; Prentice Hall, Inc.: Upper Saddle River, NJ, USA, 2001. [Google Scholar]
- Beachy, M.D.; Chasman, D.; Murphy, R.B.; Halgren, T.A.; Friesner, R.A. Accurate ab Initio Quantum Chemical Determination of the Relative Energetics of Peptide Conformations and Assessment of Empirical Force Fields. J. Am. Chem. Soc. 1997, 119, 5908–5920. [Google Scholar] [CrossRef]
- Sapse, A.M. Molecular Orbital Calculations for Amino Acids and Peptides; Springer Science & Business Media: Bazylei, Switzerland, 2012. [Google Scholar]
- Rossi, M.; Chutia, S.; Scheffler, M.; Blum, V. Validation Challenge of Density-Functional Theory for Peptides—Example of Ac-Phe-Ala5-LysH+. J. Phys. Chem. A 2014, 118, 7349–7359. [Google Scholar] [CrossRef] [PubMed]
- Jwad, R.; Weissberger, D.; Hunter, L. Strategies for Fine-Tuning the Conformations of Cyclic Peptides. Chem. Rev. 2020, 120, 9743–9789. [Google Scholar] [CrossRef] [PubMed]
- Mata, R.A.; Suhm, M.A. Benchmarking Quantum Chemical Methods: Are We Heading in the Right Direction? Angew. Chem. Int. Ed. 2017, 56, 11011–11018. [Google Scholar] [CrossRef]
- Lexa, K.W.; Alser, K.A.; Salisburg, A.M.; Ellens, D.J.; Hernandez, L.; Bono, S.J.; Michael, H.C.; Derby, J.R.; Skiba, J.G.; Feldgus, S.; et al. The search for low energy conformational families of small peptides: Searching for active conformations of small peptides in the absence of a known receptor. Int. J. Quantum Chem. 2007, 107, 3001–3012. [Google Scholar] [CrossRef]
- Shields, G.C. Computational approaches for the design of peptides with anti-breast cancer properties. Future Med. Chem. 2009, 1, 201–212. [Google Scholar] [CrossRef]
- Berendsen, H.J.; Hayward, S. Collective protein dynamics in relation to function. Curr. Opin. Struct. Biol. 2000, 10, 165–169. [Google Scholar] [CrossRef]
- Zhang, Z.; Shi, Y.; Liu, H. Molecular dynamics simulations of peptides and proteins with amplified collective motions. Biophys. J. 2003, 84, 3583–3593. [Google Scholar] [CrossRef]
- Copps, J.; Murphy, R.F.; Lovas, S. Molecular Dynamics Simulations of Peptides. In Peptide-Based Drug Design; Otvos, L., Ed.; Methods In Molecular Biology™; Humana Press: Totowa, NJ, USA, 2008; Volume 494. [Google Scholar] [CrossRef]
- Luzik, D.A.; Rogacheva, O.N.; Izmailov, S.A.; Indeykina, M.I.; Kononikhin, S.; Skrynnikov, R. Molecular Dynamics model of peptide-protein conjugation: Case study of covalent complex between Sos1 peptide and N-terminal SH3 domain from Grb2. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef]
- Geng, H.; Chen, F.; Ye, J.; Jiang, F. Applications of Molecular Dynamics Simulation in Structure Prediction of Peptides and Proteins. Comput. Struct. Biotechnol. J. 2019, 17, 1162–1170. [Google Scholar] [CrossRef] [PubMed]
- Georgoulia, P.S.; Glykos, N.M. Molecular simulation of peptides coming of age: Accurate prediction of folding, dynamics and structures. Arch. Biochem. Biophys. 2019, 664, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Bonomi, M.; Gervasio, F.L.; Tiana, G.; Provasi, D.; Broglia, R.A.; Parrinello, M. Insight into the folding inhibition of the HIV-1 protease by a small peptide. Biophys. J. 2007, 93, 2813–2821. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Bachiller, M.I.; Brzozowska, I.; Odolczyk, N.; Zielenkiewicz, U.; Zielenkiewicz, P.; Rademann, J. Mapping Protein-Protein Interactions of the Resistance-Related Bacterial Zeta Toxin-Epsilon Antitoxin Complex (ε₂ζ₂) with High Affinity Peptide Ligands Using Fluorescence Polarization. Toxins (Basel) 2016, 8, 222. [Google Scholar] [CrossRef]
- Kumar, R.; Chaudhary, K.; Sharma, M.; Nagpal, G.; Chauhan, J.S.; Singh, S.; Gautam, A.; Raghava, G.P.S. AHTPDB: A comprehensive platform for analysis and presentation of antihypertensive peptides. Nucleic Acids Res. 2015, 43, D956–D962. [Google Scholar] [CrossRef]
- Balatti, G.; Martini, M.F.; Pickholz, M. A Coarse Grain Approach to Study the Antimicrobial Peptides Aurein 1.2 and Maculatin 1.1 interactions with POPG/POPE Lipid Mixtures. J. Mol. Model. 2018, 24, 208–217. [Google Scholar] [CrossRef]
- Ulmschneider, J.P.; Ulmschneider, M.B. Cell penetrating peptides, novel vectors for gene therapy. Acc. Chem. Res. 2018, 51, 1106–1116. [Google Scholar] [CrossRef]
- Albano, J.M.R. Eneida de Paula and Monica Pickholz (November 5th 2018). In Molecular Dynamics Simulations to Study Drug Delivery Systems, Molecular Dynamics, Alexander Vakhrushev; IntechOpen; Available online: https://www.intechopen.com/books/molecular-dynamics/molecular-dynamics-simulations-to-study-drug-delivery-systems (accessed on 5 November 2018). [CrossRef]
- Marrink, S.J.; Risselada, H.J.; Yefimov, S.; Tieleman, D.P.; de Vries, A.H. The MARTINI force field: Coarse grained model for biomolecular simulations. J. Phys. Chem. B 2007, 111, 7812–7824. [Google Scholar] [CrossRef]
- Winger, M.; Trzesniak, D.; Baron, R.; van Gunsteren, W.F. On using a too large integration time step in molecular dynamics simulations of coarse-grained molecular models. Phys. Chem. Chem. Phys. 2009, 11, 1934–1941. [Google Scholar] [CrossRef][Green Version]
- Zhao, L.; Cao, Z.; Bian, Y.; Hu, G.; Wang, J.; Zhou, Y. Molecular Dynamics Simulations of Human Antimicrobial Peptide LL-37 in Model POPC and POPG Lipid Bilayers. Int. J. Mol. Sci. 2018, 19, 1186. [Google Scholar] [CrossRef]
- Gumbart, J.C.; Ulmschneider, M.B.; Hazel, A.; White, S.H.; Ulmschneider, J.P. Computed Free Energies of Peptide Insertion into Bilayers are Independent of Computational Method. J. Membr. Biol. 2018, 251, 345–356. [Google Scholar] [CrossRef]
- Hu, Y.; Ou, S.; Patel, S. Free energetics of arginine permeation into model DMPC lipid bilayers: Coupling of effective counterion concentration and lateral bilayer dimensions. J. Phys. Chem. B 2013, 117, 11641–11653. [Google Scholar] [CrossRef]
- Kabelka, I.; Vácha, R. Optimal Hydrophobicity and Reorientation of Amphiphilic Peptides Translocating through Membrane. Biophys. J. 2018, 115, 1045–1054. [Google Scholar] [CrossRef]
- Do, P.C.; Lee, E.H.; Le, L. Steered molecular dynamics simulation in rational drug design. Chem. Inf. Model. 2018, 58, 1473–1482. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Liu, L.; Hu, G.; Bian, Y.; Li, H.; Wang, J.; Zhou, Y. Interplay of hydrophobic and hydrophilic interactions in sequence-dependent cell penetration of spontaneous membrane-translocating peptides revealed by bias-exchange metadynamics simulations. Biochim. Biophys. Acta Biomembr. 2020, 1862, 183402–183414. [Google Scholar] [CrossRef] [PubMed]
- Balatti, G.E.; Ambroggio, E.E.; Fidelio, G.D.; Martini, M.F.; Pickholz, M. Differential Interaction of Antimicrobial Peptides with Lipid Structures Studied by Coarse-Grained Molecular Dynamics Simulations. Molecules 2017, 22, 1775–1792. [Google Scholar] [CrossRef] [PubMed]
- Balatti, G.E.; Domene, C.; Martini, M.F.; Pickholz, M.J. Differential Stability of Aurein 1.2 Pores in Model Membranes of Two Probiotic Strains. Chem. Inf. Model. 2020, 20. [Google Scholar] [CrossRef]
- Traboulsi, H.; Larkin, H.; Bonin, M.-A.; Volkov, L.; Lavoie, C.L.; Marsault, E.R. Macrocyclic cell penetrating peptides: A study of structure-penetration properties. Bioconjug. Chem. 2015, 26, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.; Martyna, A.; Hard, R.L.; Wang, J.; Appiah-Kubi, G.; Coss, C.; Phelps, M.A.; Rossman, J.S.; Pei, D. Discovery and mechanism of highly efficient cyclic cell-penetrating peptides. Biochemistry 2016, 55, 2601–2612. [Google Scholar] [CrossRef]
- Reissmann, S. Cell penetration: Scope and limitations by the application of cell-penetrating peptides. J. Pept. Sci. 2014, 20, 760–784. [Google Scholar] [CrossRef]
- Ali, M.; Amon, M.; Bender, V.; Bolte, A.; Separovic, F.; Benson, H.; Manolios, N. Cyclization enhances function of linear anti-arthritic peptides. Clin. Immunol. 2014, 150, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Mandal, D.; Nasrolahi Shirazi, A.; Parang, K. Cell-penetrating homochiral cyclic peptides as nuclear-targeting molecular transporters. Angew. Chem. Int. Ed. 2011, 50, 9633–9637. [Google Scholar] [CrossRef]
- Nasrolahi Shirazi, A.; Salem El-Sayed, N.; Kumar Tiwari, R.; Tavakoli, K.; Parang, K. Cyclic peptide containing hydrophobic and positively charged residues as a drug delivery system for curcumin. Curr. Drug Deliv. 2016, 13, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Nasrolahi Shirazi, A.; Tiwari, R.; Chhikara, B.S.; Mandal, D.; Parang, K. Design and biological evaluation of cell-penetrating peptide–doxorubicin conjugates as prodrugs. Mol. Pharm. 2013, 10, 488–499. [Google Scholar] [CrossRef]
- Nasrolahi Shirazi, A.; Tiwari, R.K.; Oh, D.; Banerjee, A.; Yadav, A.; Parang, K. Efficient delivery of cell impermeable phosphopeptides by a cyclic peptide amphiphile containing tryptophan and arginine. Mol. Pharm. 2013, 10, 2008–2020. [Google Scholar] [CrossRef]
- Nasrolahi Shirazi, A.; Tiwari, R.K.; Oh, D.; Sullivan, B.; McCaffrey, K.; Mandal, D.; Parang, K. Surface decorated gold nanoparticles by linear and cyclic peptides as molecular transporters. Mol. Pharm. 2013, 10, 3137–3151. [Google Scholar] [CrossRef]
- El-Sayed, N.; Miyake, T.; Shirazi, A.; Park, S.; Clark, J.; Buchholz, S.; Parang, K.; Tiwari, R. Design, synthesis and evaluation of homochiral peptides containing arginine and histidine as molecular transporters. Molecules 2018, 23, 1590–1605. [Google Scholar] [CrossRef]
- Oh, D.; Sun, J.; Nasrolahi Shirazi, A.; LaPlante, K.L.; Rowley, D.C.; Parang, K. Antibacterial activities of amphiphilic cyclic cell-penetrating peptides against multidrug-resistant pathogens. Mol. Pharm. 2014, 11, 3528–3536. [Google Scholar] [CrossRef]
- Darwish, S.; Sadeghiani, N.; Fong, S.; Mozaffari, S.; Hamidi, P.; Withana, T.; Yang, S.; Tiwari, R.K.; Parang, K. Synthesis and antiproliferative activities of doxorubicin thiol conjugates and doxorubicin-SS-cyclic peptide. Eur. J. Med. Chem. 2019, 161, 594–606. [Google Scholar] [CrossRef]
- Shirazi, A.; Mozaffari, S.; Sherpa, R.; Tiwari, R.; Parang, K. Efficient intracellular delivery of cell-impermeable cargo molecules by peptides containing tryptophan and histidine. Molecules 2018, 23, 1536–1548. [Google Scholar] [CrossRef]
- Shirazi, A.N.; El-Sayed, N.S.; Mandal, D.; Tiwari, R.K.; Tavakoli, K.; Etesham, M.; Parang, K. Cysteine and arginine-rich peptides as molecular carriers. Bioorg. Med. Chem. Lett. 2016, 26, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.; Darwish, S.A.; Shirazi, A.N.; Tiwari, R.K.; Parang, K. Amphiphilic bicyclic peptides as cellular delivery agents. ChemMedChem 2016, 11, 2095. [Google Scholar] [CrossRef]
- El-Sayed, N.S.; Shirazi, A.N.; Sajid, M.I.; Park, S.E.; Parang, K.; Tiwari, R.K. Synthesis and antiproliferative activities of conjugates of paclitaxel and camptothecin with a cyclic cell-penetrating peptide. Molecules 2019, 24, 1427. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Mandal, D.; El-Mowafi, S.A.; Mozaffari, S.; Tiwari, R.K.; Parang, K. Click-free synthesis of a multivalent tricyclic peptide as a molecular transporter. Pharmaceutics 2020, 12, 842–859. [Google Scholar] [CrossRef]
- Vazquez-Lombardi, R.; Phan, T.G.; Zimmermann, C.; Lowe, D.; Jermutus, L.; Christ, D. Challenges and opportunities for non-antibody scaffold drugs. Drug Discov. Today 2015, 20, 1271–1283. [Google Scholar] [CrossRef]
- Löfblom, J.; Feldwisch, J.; Tolmachev, V.; Carlsson, J.; Stahl, S.; Frejd, F.Y. Affibody molecules: Engineered proteins for therapeutic, diagnostic and biotechnological applications. FEBS Lett. 2010, 584, 2670–2680. [Google Scholar] [CrossRef]
- Parhiz, H.; Khoshnejad, M.; Myerson, J.W.; Hood, E.; Patel, P.N.; Brenner, J.S.; Muzykantov, V.R. Unintended effects of drug carriers: Big issues of small particles. Adv. Drug Deliv. Rev. 2018, 130, 90–112. [Google Scholar] [CrossRef]
- Andrieu, J.; Re, F.; Russo, L.; Nicotra, F. Phage-displayed peptides targeting specific tissues and organs. J. Drug Target. 2019, 27, 555–565. [Google Scholar] [CrossRef]
- Krumpe, L.R.; Mori, T. Potential of phage-displayed peptide library technology to identify functional targeting peptides. Expert Opin. Drug Discov. 2007, 2, 525. [Google Scholar] [CrossRef]
- Hart, S.L.; Harbottle, R.P.; Cooper, R.; Miller, A.; Williamson, R.; Coutelle, C. Gene delivery and expression mediated by an integrin-binding peptide. Gene Ther. 1995, 2, 552–554. [Google Scholar]
- Hart, S.L.; Collins, L.; Gustafsson, K.; Fabre, J.W. Integrin-mediated transfection with peptides containing arginine-glycine-aspartic acid domains. Gene Ther. 1997, 4, 1225–1230. [Google Scholar] [CrossRef] [PubMed]
- Hart, S.L. Integrin-mediated vectors for gene transfer and therapy. Curr. Opin. Mol. Ther. 1999, 1, 197–203. [Google Scholar] [PubMed]
- Avraamides, C.J.; Garmy-Susini, B.; Varner, J.A. Integrins in angiogenesis and lymphangiogenesis. Nat. Rev. Cancer 2008, 8, 604–617. [Google Scholar] [CrossRef] [PubMed]
- Brooks, P.C.; Clark, R.A.; Cheresh, D.A. Requirement of vascular integrin alpha beta 3 for angiogenesis. Science 1994, 264, 569–571. [Google Scholar] [CrossRef] [PubMed]
- Ng, Q.K.; Sutton, M.K.; Soonsawad, P.; Xing, L.; Cheng, H.; Segura, T. Engineering clustered ligand binding into nonviral vectors: Alphavbeta3 targeting as an example. Mol. Ther. 2009, 17, 828–836. [Google Scholar] [CrossRef]
- Ng, Q.K.T.; Su, H.; Armijo, A.L.; Czernin, J.; Radu, C.G.; Segura, T. Clustered Arg–Gly–Asp Peptides Enhances Tumor Targeting of Nonviral Vectors. ChemMedChem 2011, 6, 623–627. [Google Scholar] [CrossRef]
- Kang, Z.; Meng, Q.; Liu, K. Peptide-based gene delivery vectors. J. Mater. Chem. B 2019, 7, 1824–1841. [Google Scholar] [CrossRef] [PubMed]
- Han, K.; Yang, J.; Chen, S.; Chen, J.X.; Liu, C.W.; Li, C.; Cheng, H.; Zhuo, R.X.; Zhang, X.Z. Novel gene transfer vectors based on artificial recombinant multi-functional oligopeptides. Int. J. Pharm. 2012, 436, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Harisa, G.I.; Faris, T.M. Direct Drug Targeting into Intracellular Compartments: Issues, Limitations, and Future Outlook. J. Membr. Biol. 2019, 252, 527–539. [Google Scholar] [CrossRef] [PubMed]
- Pan, T.; Song, W.; Gao, H.; Li, T.; Cao, X.; Zhong, S.; Wang, Y. MiR-29b-Loaded Gold Nanoparticles Targeting to the Endoplasmic Reticulum for Synergistic Promotion of Osteogenic Differentiation. ACS Appl. Mater. Interfaces 2016, 8, 19217–19227. [Google Scholar] [CrossRef]
- Agemy, L.; Friedmann-Morvinski, D.; Kotamraju, V.K.; Roth, L.; Sugahara, K.N.; Girard, O.M.; Mattrey, R.F.; Verma, I.M.; Ruoslathi, E. Targeted nanoparticle enhanced proapoptotic peptide as potential therapy for glioblastoma. Proc. Natl. Acad. Sci. USA 2011, 108, 17450–17455. [Google Scholar] [CrossRef]
- Donahue, N.D.; Acar, H.; Wilhelm, S. Concepts of nanoparticle cellular uptake, intracellular trafficking, and kinetics in nanomedicine. Adv. Drug Deliv. Rev. 2019, 143, 68–96. [Google Scholar] [CrossRef]
- Guidotti, G.; Brambilla, L.; Rossi, D. Cell-Penetrating Peptides: From Basic Research to Clinics. Trends Pharmacol. Sci. 2017, 38, 406–424. [Google Scholar] [CrossRef]
- Vivès, E.; Brodin, P.; Lebleu, B. A truncated HIV-1 Tat protein basic domain rapidly translocates through the plasma membrane and accumulates in the cell nucleus. J. Biol. Chem. 1997, 272, 16010–16017. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.E.; Rice, K.G. Peptide-guided gene delivery. AAPS J. 2007, 9, E18–E29. [Google Scholar] [CrossRef] [PubMed]
- Lehto, T.; Simonson, O.E.; Mager, I.; Ezzat, K.; Sork, H.; Copolovici, D.M.; Viola, J.R.; Zaghloul, E.M.; Lundin, P.; Moreno, P.M.D.; et al. A peptide-based vector for efficient gene transfer in vitro and in vivo. Mol. Ther. J. Am. Soc. Gene Ther. 2011, 19, 1457–1467. Available online: http://www.nature.com/doifinder/10.1038/mt.2011.10 (accessed on 19 August 2011). [CrossRef] [PubMed]
- Wyman, T.B.; Nicol, F.; Zelphati, O.; Scaria, P.V.; Plank, C.; Szoka, F.C. Design, synthesis, and characterization of a cationic peptide that binds to nucleic acids and permeabilizes bilayers. Biochemistry 1997, 36, 3008–3017. [Google Scholar] [CrossRef] [PubMed]
- Bolhassani, A.; Ghasemi, N.; Servis, C.; Taghikhani, M.; Rafati, S. The efficiency of a novel delivery system (PEI600-Tat) in development of potent DNA vaccine using HPV16 E7 as a model antigen. Drug Deliv. 2009, 16, 196–204. [Google Scholar] [CrossRef]
- Zhang, H.; Gerson, T.; Varney, M.L.; Singh, R.K.; Vinogradov, S.V. Multifunctional Peptide-PEG Intercalating Conjugates: Programmatic of Gene Delivery to the Blood-Brain Barrier. Pharm. Res. 2010, 27, 2528–2543. [Google Scholar] [CrossRef]
- Gonçalves, E.; Kitas, E.; Seelig, J. Binding of Oligoarginine to Membrane Lipids and Heparan Sulfate: Structural and Thermodynamic Characterization of a Cell-Penetrating Peptide. Biochemistry 2005, 44, 2692–2702. [Google Scholar] [CrossRef]
- Ziegler, A.; Blatter, X.L.; Seelig, A.; Seelig, J. Protein transduction domains of HIV-1 and SIV TAT interact with charged lipid vesicles. Binding mechanism and thermodynamic analysis. Biochemistry 2003, 42, 9185–9194. [Google Scholar] [CrossRef]
- Binder, H.; Lindblom, G. Charge-Dependent Translocation of the Trojan Peptide Penetratin across Lipid Membranes. Biophys. J. 2003, 85, 982–995. [Google Scholar] [CrossRef]
- Christiaens, B.; Symoens, S.; Verheyden, S.; Engelborghs, Y.; Joliot, A.; Prochiantz, A.; Vandekerckhove; Rosseneu, M.; Vanloo, B. Tryptophan fluorescence study of the interaction of penetratin peptides with model membranes. Eur. J. Biochem. 2002, 269, 2918–2926. [Google Scholar] [CrossRef] [PubMed]
- Dom, G.; Shaw-Jackson, C.; Matis, C.; Bouffioux, O.; Picard, J.J.; Prochiantz, A.; Mingeot-Leclercq, P.; Brasseur, R.; Rezsohazy, R. Cellular uptake of Antennapedia Penetratin peptides is a two-step process in which phase transfer precedes a tryptophan-dependent translocation. Nucleic Acids Res. 2003, 31, 556–561. [Google Scholar] [CrossRef] [PubMed]
- Canine, B.F.; Wang, Y.; Hatefi, A. Evaluation of the effect of vector architecture on DNA condensation and gene transfer efficiency. J. Control. Release 2008, 129, 117–123. [Google Scholar] [CrossRef][Green Version]
- Marshall, N.B.; Oda, S.K.; London, C.A.; Moulton, H.M.; Iversen, P.L.; Kerkvliet, N.I. and Mourich, D.V. Arginine-rich cell-penetrating peptides facilitate delivery of antisense oligomers into murine leukocytes and alter pre-mRNA splicing. J. Immunol. Methods 2007, 325, 114–126. [Google Scholar] [CrossRef]
- Rothbard, J.B.; Kreider, E.; VanDeusen, C.L.; Wright, L.; Wylie, B.; Wender, P.A. Arginine-rich molecular transporters for drug delivery: Role of backbone spacing in cellular uptake. J. Med. Chem. 2002, 45, 3612–3618. [Google Scholar] [CrossRef]
- Siprashvili, Z.; Scholl, F.A.; Oliver, S.F.; Adams, A.; Contag, C.H.; Wender, P.A.; Khavari, P.A. Gene transfer via reversible plasmid condensation with cysteine-flanked, internally spaced arginine-rich peptides. Hum. Gene Ther. 2004, 14, 1225–1233. [Google Scholar] [CrossRef]
- Futaki, S.; Ohashi, W.; Suzuki, T.; Niwa, M.; Tanaka, S.; Ueda, K.; Harashima, H.; Sugiura, Y. Stearylated arginine-rich peptides: A new class of transfection systems. Bioconjug. Chem. 2001, 12, 1005–1011. [Google Scholar] [CrossRef]
- Plank, C.; Oberhauser, B.; Mechtler, K.; Koch, C.; Wagner, E. The influence of endosome-disruptive peptides on gene transfer using synthetic virus-like gene transfer systems. J. Biol. Chem. 1994, 269, 12918–12924. [Google Scholar] [CrossRef]
- Wagner, E.; Plank, C.; Zatloukal, K.; Cotten, M.; Birnstiel, M.L. Influenza virus hemagglutinin HA-2 N-terminal fusogenic peptides augment gene transfer by transferrin-polylysine-DNA complexes: Toward a synthetic virus-like gene-transfer vehicle. Proc. Natl. Acad. Sci. USA 1992, 89, 7934–7938. [Google Scholar] [CrossRef] [PubMed]
- Gupta, B.; Levchenko, T.S.; Torchilin, V.P. Intracellular delivery of large molecules and small particles by cell-penetrating proteins and peptides. Adv. Drug Deliv. Rev. 2005, 57, 637–651. [Google Scholar] [CrossRef]
- Chen, C.P.; Kim, J.S.; Steenblock, E.; Liu, D.; Rice, K.G. Gene Transfer with Poly-Melittin Peptides. Bioconjug. Chem. 2006, 17, 1057–1062. [Google Scholar] [CrossRef] [PubMed]
- Ogris, M.; Carlisle, R.C.; Bettinger, T.; Seymour, L.W. Melittin Enables Efficient Vesicular Escape and Enhanced Nuclear Access of Nonviral Gene Delivery Vectors. J. Biol. Chem. 2001, 276, 47550–47555. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.X.; Chen, C.; Wang, Y.L.; Lin, S.; Wang, Y.; Li, S.B.; Jin, X.P.; Gao, H.W.; Du, F.S.; Gong, F.; et al. Truncated peptides from melittin and its analog with high lytic activity at endosomal pH enhance branched polyethylenimine-mediated gene transfection. J. Gene Med. 2012, 14, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Abe, H. Role of histidine-related compounds as intracellular proton buffering constituents in vertebrate muscle. Biochemistry 2000, 65, 757–765. [Google Scholar]
- Abe, H.; Dobson, G.P.; Hoeger, U.; Parkhouse, W.S. Role of histidine-related compounds to intracellular buffering in fish skeletal muscle. Am. J. Physiol. 1985, 249, R449–R454. [Google Scholar] [CrossRef]
- Parhiz, H.; Hashemi, M.; Hatefi, A.; Shier, W.T.; Farzad, S.A.; Ramezani, M. Arginine-rich hydrophobic polyethylenimine: Potent agent with simple components for nucleic acid delivery. Int. J. Biol. Macromol. 2013, 60, 18–27. [Google Scholar] [CrossRef]
- Mével, M.; Neveu, C.; Goncalves, C.; Yaounanc, J.J.; Pichon, C.; Jaffre, P.A.; Midoux, P. Novel neutral imidazole-lipophosphoramides for transfection assays. Chem. Commun. 2008, 3124–3126. [Google Scholar] [CrossRef]
- Midoux, P.; Kichler, A.; Boutin, V.; Maurizot, J.C.; Monsigny, M. Membrane permeabilization and efficient gene transfer by a peptide containing several histidines. Bioconjug. Chem. 1998, 9, 260–267. [Google Scholar] [CrossRef]
- Wang, X.L.; Nguyen, T.; Gillespie, D.; Jensen, R.; Lu, Z.R. A multifunctional and reversibly polymerizable carrier for efficient siRNA delivery. Biomaterials 2008, 29, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.R.; Zhang, L.; Luther, P.W.; Mixson, A.J. Optimal transfection with the HK polymer depends on its degree of branching and the pH of endocytic vesicles. Nucleic Acids Res. 2002, 30, 1338–1345. [Google Scholar] [CrossRef] [PubMed]
- Leng, Q.; Scaria, P.; Zhu, J.; Ambulos, N.; Campbella, P.; Mixson, J. Highly branched HK peptides are effective carriers of siRNA. J. Gene Med. 2005, 7, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Dey, D.; Inayathullah, M.; :Lee, A.S.; Limiuex, M.; Zhang, X.; Wu, Y.; Nag, D.; De Almeida, P.E.; Han, L.; Rajadas, J.; et al. Efficient gene delivery of primary human cells using peptide linked polyethylenimine polymer hybrid. Biomaterials 2011, 32, 4647–4658. [Google Scholar] [CrossRef]
- Kim, T.H.; Ihm, J.E.; Choi, Y.J.; Nah, J.W.; Cho, C.S. Efficient gene delivery by urocanic acid-modified chitosan. J. Control. Release 2003, 93, 389–402. [Google Scholar] [CrossRef]
- Swami, A.; Aggarwal, A.; Pathak, A.; Patnaik, S.; Kumar, P.; Singh, Y.; Gupta, K.C. Imidazolyl-PEI modified nanoparticles for enhanced gene delivery. Int. J. Pharm. 2007, 335, 180–192. [Google Scholar] [CrossRef]
- Hébert, E. Improvement of exogenous DNA nuclear importation by nuclear localization signal-bearing vectors: A promising way for non-viral gene therapy? Biol. Cell. 2003, 95, 59–68. [Google Scholar] [CrossRef]
- Khalil, I.A.; Hayashi, Y.; Mizuno, R.; Harashima, H. Octaarginine- and pH sensitive fusogenic peptide-modified nanoparticles for liver gene delivery. J. Control Release 2011, 156, 374–380. [Google Scholar] [CrossRef]
- Robbins, J.; Dilworth, S.M.; Laskey, R.A.; Dingwall, C. Two interdependent basic domains in nucleoplasmin nuclear targeting sequence: Identification of a class of bipartite nuclear targeting sequence. Cell 1991, 64, 615–623. [Google Scholar] [CrossRef]
- Duvshani-Eshet, M.; Oz, H.K.S.; Radzishevsky, I.S.; Mor, A.; Machluf, M. Effect of peptides bearing nuclear localization signals on therapeutic ultrasound mediated gene delivery. J. Gene Med. 2008, 10, 1150–1159. [Google Scholar] [CrossRef]
- Ni, Y.H.; Hsu, H.Y.; Chen, P.J.; Chang, M.H. Protamine enhances the efficiency of liposome-mediated gene transfer in a cultured human hepatoma cell line. J. Formos. Med. Assoc. 1999, 98, 562–566. [Google Scholar]
- Vázquez, E.; Ferrer-Miralles, N.; Villaverde, A. Peptide-assisted traffic engineering for nonviral gene therapy. Drug Discov. Today 2008, 13, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Parhiz, H.; Hashemi, M.; Hatefi, A.; Shier, W.T.; Farzad, S.A.; Ramezani, M. Molecular weight-dependent genetic information transfer with disulfide-linked polyethylenimine-based nonviral vectors. J. Biomater. Appl. 2013, 28, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Parker, A.L.; Collins, L.; Zhang, X.; Fabre, J.W. Exploration of peptide motifs for potent non-viral gene delivery highly selective for dividing cells. J. Gene Med. 2005, 7, 1545–1554. [Google Scholar] [CrossRef] [PubMed]
- Parelkar, S.S.; Chan-Seng, D.; Emrick, T. Reconfiguring polylysine architectures for controlling polyplex binding and non-viral transfection. Biomaterials 2011, 32, 2432–2444. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.P.; Petrenko, V.A. Phage Display. Chem. Rev. 1997, 97, 391–410. [Google Scholar] [CrossRef]
- Lofblom, J. Bacterial display in combinatorial protein engineering. Biotechnol. J. 2011, 6, 1115–1129. [Google Scholar] [CrossRef]
- Hanes, J.; Plückthun, A. In vitro selection and evolution of functional proteins using ribosome display. Proc. Natl. Acad. Sci. USA 1997, 94, 4937–4942. [Google Scholar] [CrossRef]
- Roberts, R.W.; Szostak, J.W. RNA-peptide fusions for the in vitro selection of peptides and proteins. Proc. Natl. Acad. Sci. USA 1997, 414, 12297–12302. [Google Scholar] [CrossRef]
- Colas, P.; Cohen, B.; Jessen, T.; Grishina, I.; McCoy, J.; Brent, R. Genetic selection of peptide aptamers that recognize and inhibit cyclin-dependent kinase 2. Nature 1996, 380, 548–550. [Google Scholar] [CrossRef]
- Kolmar, H. Biological diversity and therapeutic potential of natural and engineered cystine knot miniproteins. Curr. Opin. Pharm. 2009, 9, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Byla, P.; Andersen, M.H.; Holtet, T.L.; Jacobsen, H.; Munch, M.; Gad, H.H.; Thogersen, H.C.; Hartman, R. Selection of a novel and highly specific tumor necrosis factor alpha (TNFalpha) antagonist: Insight from the crystal structure of the antagonist-TNF alpha complex. J. Biol. Chem. 2010, 285, 12096–12100. [Google Scholar] [CrossRef] [PubMed]
- Heinis, C.; Rutherford, T.; Freund, S.; Winter, G. Phage encoded combinatorial chemical libraries based on bicyclic peptides. Nat. Chem. Biol. 2009, 5, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Reverdatto, S.; Burz, D.S.; Shekhtman, A. Peptide aptamers: Development and applications. Curr. Top. Med. Chem. 2015, 15, 1082–1101. [Google Scholar] [CrossRef] [PubMed]
- Gronwall, C.; Stahl, S. Engineered affinity proteins--generation and applications. J. Biotechnol. 2009, 140, 254–269. [Google Scholar] [CrossRef]
- New, R.; Bansal, G.; Bogus, M.; Zajkowska, K.; Rickelt, S.; Toth, I. Use of Mixed Micelles for Presentation of Building Blocks in a New Combinatorial Discovery Methodology: Proof-of-Concept Studies. Molecules 2013, 18, 3427–3441. [Google Scholar] [CrossRef]
- New, R.; Bansal, G.S.; Dryjska, M.; Bogus, M.; Green, P.; Feldmann, M.; Brennan, F. esign and Optimisation of Bioactive Cyclic Peptides: Generation of a Down-Regulator of TNF Secretion. Molecules 2014, 19, 21529–21540. [Google Scholar] [CrossRef]
- Neefjes, J.; Ovaa, H. A peptide’s perspective on antigen presentation to the immune system. Nat. Chem. Biol. 2013, 9, 769–775. [Google Scholar] [CrossRef]
- Skwarczynski, M.; Toth, I. Peptide-based synthetic vaccines. Chem. Sci. 2016, 7, 842–854. [Google Scholar] [CrossRef]
- Malonis, R.J.; Lai, J.R.; Vergnolle, O. Peptide-Based Vaccines: Current Progress and Future Challenges. Chem. Rev. 2020, 120, 3210–3229. [Google Scholar] [CrossRef]
- Shi, S.; Zhu, H.; Xia, X.; Liang, Z.; Ma, X.; Sun, B. Vaccine adjuvants: Understanding the structure and mechanism of adjuvanticity. Vaccine 2019, 37, 3167–3178. [Google Scholar] [CrossRef] [PubMed]
- Azmi, F.; Ahmad Fuaad, A.A.; Skwarczynski, M.; Toth, I. Recent progress in adjuvant discovery for peptide-based subunit vaccines. Hum. Vaccin. Immunother. 2014, 10, 778–796. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Liang, Z.; Sun, B. Response to comment on: Vaccine adjuvants: Understanding the structure and mechanism of adjuvanticity. Vaccine 2020, 38, 2759. [Google Scholar] [CrossRef] [PubMed]
- Nevagi, R.J.; Skwarczynski, M.; Toth, I. Polymers for subunit vaccine delivery. Eur. Polym. J. 2019, 114, 397–410. [Google Scholar] [CrossRef]
- Marasini, N.; Ghaffar, K.A.; Skwarczynski, M.; Toth, I. Liposomes as a Vaccine Delivery System. In Micro- and Nanotechnology in Vaccine Development; Skwarczynski, M., Toth, I., Eds.; William Andrew Inc.: Norwich, UK, 2017; pp. 221–239. [Google Scholar]
- Rudra, J.S.; Mishra, S.; Chong, A.S.; Mitchell, R.A.; Nardin, E.H.; Nussenzweig, V.; Collier, J.H. Self-assembled peptide nanofibers raising durable antibody responses against a malaria epitope. Biomaterials 2012, 33, 6476–6484. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Han, H.F.; Hudalla, G.A.; Wen, Y.; Pompano, R.R.; Collier, J.H. Thermal stability of self-assembled peptide vaccine materials. Acta Biomater. 2016, 30, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Azmi, F.; Ahmad Fuaad, A.A.; Giddam, A.K.; Batzloff, M.R.; Good, M.F.; Skwarczynski, M.; Toth, I. Self-adjuvanting vaccine against group A streptococcus: Application of fibrillized peptide and immunostimulatory lipid as adjuvant. Bioorg. Med. Chem. 2014, 22, 6401–6408. [Google Scholar] [CrossRef]
- Skwarczynski, M.; Zhao, G.; Boer, J.C.; Ozberk, V.; Azuar, A.; Cruz, J.G.; Giddam, A.K.; Khalil, Z.G.; Pandey, M.; Shibu, M.A.; et al. Poly(amino acids) as a potent self-adjuvanting delivery system for peptide-based nanovaccines. Sci. Adv. 2020, 6, 2285–2296. [Google Scholar] [CrossRef]
- Bartlett, S.; Skwarczynski, M.; Xie, X.; Toth, I.; Loukas, A.; Eichenberger, R.M. Development of natural and unnatural amino acid delivery systems against hookworm infection. Prec. Nanomed. 2020, 3, 471–482. [Google Scholar] [CrossRef]
- Nevagi, R.J.; Khalil, Z.G.; Hussein, W.M.; Powell, J.; Batzloff, M.R.; Capon, R.J.; Good, M.F.; Skwarczynski, M.; Toth, I. Polyglutamic acid-trimethyl chitosan-based intranasal peptide nano-vaccine induces potent immune responses against group A streptococcus. Acta Biomater. 2018, 80, 278–287. [Google Scholar] [CrossRef]
- Nevagi, R.J.; Dai, W.; Khalil, Z.G.; Hussein, W.M.; Capon, R.J.; Skwarczynski, M.; Toth, I. Self-assembly of trimethyl chitosan and poly(anionic amino acid)-peptide antigen conjugate to produce a potent self-adjuvanting nanovaccine delivery system. Bioorg. Med. Chem. 2019, 27, 3082–3088. [Google Scholar] [CrossRef] [PubMed]
- Nevagi, R.J.; Dai, W.; Khalil, Z.G.; Hussein, W.M.; Capon, R.J.; Skwarczynski, M.; Toth, I. Structure-activity relationship of group A streptococcus lipopeptide vaccine candidates in trimethyl chitosan-based self-adjuvanting delivery system. Eur. J. Med. Chem. 2019, 179, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.R.; Luo, Y.C.; Shibu, M.A.; Toth, I.; Skwarczynski, M. Cell-Penetrating Peptides: Efficient Vectors for Vaccine Delivery. Curr. Drug Deliv. 2019, 16, 430–443. [Google Scholar] [CrossRef]
- Calzetta, L.; Pistocchini, E.; Ritondo, B.L.; Roncada, P.; Palma, E.; di Cave, D.; Mattei, M.; Britti, D. Immunoprophylaxis pharmacotherapy against canine leishmaniosis: A systematic review and meta-analysis on the efficacy of vaccines approved in European Union. Vaccine 2020, 38, 6695–6703. [Google Scholar] [CrossRef]
- COVAXX. Coronavirus Company News Summary–Pfizer/BioNTech Publishes Pre-Clinical Data for its Lead Candidate–Dasa and COVAXX Partner on Vaccine Clinical Trial in Brazil. Available online: https://www.pharmaceutical-technology.com/uncategorised/coronavirus-company-news-summary-pfizer-biontech-publishes-pre-clinical-data-for-its-lead-candidate-dasa-and-covaxx-partner-on-vaccine-clinical-trial-in-brazil/ (accessed on 11 September 2020).
- Obara, W.; Kanehira, M.; Katagiri, T.; Kato, R.; Kato, Y.; Takata, R. Present status and future perspective of peptide-based vaccine therapy for urological cancer. Cancer Sci. 2018, 109, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Joshi, M.D.; Singhania, S.; Ramsey, K.H.; Murthy, A.K. Peptide vaccine: Progress and challenges. Vaccines (Basel, Switz.) 2014, 2, 515–536. [Google Scholar] [CrossRef]
- Magzoub, M. Combating proteins with proteins: Engineering cell-penetrating peptide antagonists of amyloid-beta aggtregation and associated neurotoxicity. DNA Cell Biol. 2020, 39, 920–925. [Google Scholar] [CrossRef]
- Caputi, S.; Trubiani, O.; Sinjari, B.; Trofimova, S.; Diomede, F.; Linkova, N.; Diatlova, A.; Khavinson, V. Effect of short peptides on neuronal differentiation of stem cells. Intern. J. Immunopathol. Pharmacol. 2019, 33, 1–12. [Google Scholar] [CrossRef]
- Russo, A.F. Overview of neuropeptides: Awakening the senses? J. Head Face Pain 2017, 57, 37–46. [Google Scholar] [CrossRef]
- Katsara, M.; Minigo, G.; Plebanski, M.; Apostolopoulos, V. The good, the bad and the ugly: How altered peptide ligands modulate immunity. Expert Opin. Biol. Ther. 2008, 8, 1873–1884. [Google Scholar] [CrossRef]
- Kita, H.; Matsumura, S.; He, X.S.; Ansari, A.A.; Lian, Z.X.; Van de Water, J.; Coppel, R.L.; Kaplan, M.M.; Gershwin, M.E. Analysis of TCR antagonism and molecular mimicry of an HLA-A0201-restricted CTL epitope in primary biliary cirrhosis. Hepatology 2002, 36, 918–926. [Google Scholar] [CrossRef] [PubMed]
- Paas-Rozner, M.; Sela, M.; Mozes, E. A dual altered peptide ligand down-regulates myasthenogenic T cell responses by up-regulating CD25- and CTLA-4-expressing CD4+ T cells. Proc. Natl. Acad. Sci. USA 2003, 100, 6676–6681. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Donelan, W.; Wang, H.; Reeves, W.; Yang, L.J. Novel autoantigens in type 1 diabetes. Am. J. Transl. Res. 2013, 5, 379–392. [Google Scholar]
- Alleva, D.G.; Maki, R.A.; Putnam, A.L.; Robinson, J.M.; Kipnes, M.S.; Dandona, P.; Marks, J.B.; Simmons, D.L.; Greenbaum, C.J.; Jimenez, R.G.; et al. Immunomodulation in type 1 diabetes by NBI-6024, an altered peptide ligand of the insulin B epitope. Scand J. Immunol. 2006, 63, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.; Philotheou, A.; Bonnici, F.; Ziegler, A.G.; Jimenez, R.; Group, N.B.I.S. No effect of the altered peptide ligand NBI-6024 on beta-cell residual function and insulin needs in new-onset type 1 diabetes. Diabetes Care 2009, 32, 2036–2040. [Google Scholar] [CrossRef]
- Agnes, M.C.; Tan, A.; Jordens, R.; Geluk, A.; Roep, B.O.; Ottenhoff, T.; Drijfhout, J.W.; Koning, F. Strongly increased efficiency of altered peptide ligands by mannosylation. Int. Immunol. 1998, 10, 1299–1304. [Google Scholar] [CrossRef]
- Dargahi, N.; Katsara, M.; Tselios, T.; Androutsou, M.E.; de Courten, M.; Matsoukas, J.; Apostolopoulos, V. Multiple Sclerosis: Immunopathology and Treatment Update. Brain. Sci. 2017, 7, 78–105. [Google Scholar] [CrossRef]
- Katsara, M.; Apostolopoulos, V. Editorial: Multiple Sclerosis: Pathogenesis and Therapeutics. Med. Chem. 2018, 14, 104–105. [Google Scholar] [CrossRef]
- Katsara, M.; Matsoukas, J.; Deraos, G.; Apostolopoulos, V. Towards immunotherapeutic drugs and vaccines against multiple sclerosis. Acta Biochim. Biophys. Sin (Shanghai) 2008, 40, 636–642. [Google Scholar] [CrossRef]
- Katsara, M.; Deraos, G.; Tselios, T.; Matsoukas, J.; Apostolopoulos, V. Design of novel cyclic altered peptide ligands of myelin basic protein MBP83-99 that modulate immune responses in SJL/J mice. J. Med. Chem. 2008, 51, 3971–3978. [Google Scholar] [CrossRef]
- Katsara, M.; Deraos, S.; Tselios, T.V.; Pietersz, G.; Matsoukas, J.; Apostolopoulos, V. Immune responses of linear and cyclic PLP139-151 mutant peptides in SJL/J mice: Peptides in their free state versus mannan conjugation. Immunotherapy 2014, 6, 709–724. [Google Scholar] [CrossRef] [PubMed]
- Katsara, M.; Yuriev, E.; Ramsland, P.A.; Deraos, G.; Tselios, T.; Matsoukas, J.; Apostolopoulos, V. A double mutation of MBP(83-99) peptide induces IL-4 responses and antagonizes IFN-gamma responses. J. Neuroimmunol. 2008, 200, 77–89. [Google Scholar] [CrossRef] [PubMed]
- Katsara, M.; Yuriev, E.; Ramsland, P.A.; Deraos, G.; Tselios, T.; Matsoukas, J.; Apostolopoulos, V. Mannosylation of mutated MBP83-99 peptides diverts immune responses from Th1 to Th2. Mol. Immunol. 2008, 45, 3661–3670. [Google Scholar] [CrossRef]
- Katsara, M.; Yuriev, E.; Ramsland, P.A.; Tselios, T.; Deraos, G.; Lourbopoulos, A.; Grigoriadis, N.; Matsoukas, J.; Apostolopoulos, V. Altered peptide ligands of myelin basic protein (MBP87-99) conjugated to reduced mannan modulate immune responses in mice. Immunology 2009, 128, 521–533. [Google Scholar] [CrossRef] [PubMed]
- Candia, M.; Kratzer, B.; Pickl, W.F. On Peptides and Altered Peptide Ligands: From Origin, Mode of Action and Design to Clinical Application (Immunotherapy). Int. Arch. Allergy Immunol. 2016, 170, 211–233. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, P.J.; Garren, H.; Hirschberg, D.L.; Langer-Gould, A.M.; Levite, M.; Karpuj, M.V.; Southwood, S.; Sette, A.; Conlon, P.; Steinman, L. Microbial epitopes act as altered peptide ligands to prevent experimental autoimmune encephalomyelitis. J. Exp. Med. 1999, 189, 1275–1284. [Google Scholar] [CrossRef]
- Reynolds, N.P. Amyloid-like peptide nanofibils as scaffolds for tissue engineering: Progress and challenges. Biointerphases 2019, 14, 40801–40809. [Google Scholar] [CrossRef]
- Hellmund, K.S.; Koksch, B. Self-assembling peptides as extracellular matrix mimics to influence stem cell’s fate. Front. Chem. 2019, 7. [Google Scholar] [CrossRef]
- Sinjari, B.; Diomede, F.; Khavinson, V.; Mironova, E.; Linkova, N.; Trofimova, S.; Trubiani, O.; Caputi, S. Short peptides protect oral stem cells from ageing. Stem Cell Rev. Rep. 2020, 16, 159–166. [Google Scholar] [CrossRef]
- Tatman, P.D.; Muhonen, E.G.; Wickers, S.T.; Gee, A.O.; Kim, E.S.; Kim, D.H. Self-assembling peptides for stem cell and tissue engineering. Biomater. Sci. 2016, 4, 543–544. [Google Scholar] [CrossRef]
- Khavinson, V.; Diomede, F.; Mironova, E.; Linkova, N.; Trofimova, S.; Trubiani, O.; Caputi, S.; Sinjari, B. AEDG peptide (epitalon) stimulates gene expression and protein synthesis during neurogenesis: Possible epigenetic mechanism. Molecules 2020, 25, 609–626. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Kulkarni, C.; Kulkarni, M.M.; Ali, R.; Porwal, K.; Chattopadhyay, N. Tripeptide-induced modulation of mesenchymal stem cell biomechanics stimulates proliferation and wound healing. Chem. Commun. 2020, 56, 3043–3046. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.K.; Brindisi, M.; Shahabi, D.; Chapman, M.E.; Mesecar, A.D. Drug development and medicinal chemistry efforts toward SARS-coronavirus and covid-19 therapeutics. ChemMedChem 2020, 15, 907–932. [Google Scholar] [CrossRef] [PubMed]
- Dömling, A.; Gao, L. Chemistry and Biology of SARS-CoV-2. Chem 2020, 6, 1283–1295. [Google Scholar] [CrossRef] [PubMed]
- Cao, B.; Wang, Y.; Wen, D.; Liu, W.; Wang, J.; Fan, G.; Ruan, L.; Song, B.; Cai, Y.; Wei, M.; et al. A trial of lopinavir–ritonavir in adults hospitalized with severe Covid-19. N. Engl. J. Med. 2002, 382, 1787–1799. [Google Scholar] [CrossRef]
- Zhang, L.; Lin, D.; Sun, X.; Curth, U.; Drosten, C.; Sauerhering, L.; Becker, S.; Rox, K.; Hilgenfeld, R. Crystal structure of SARS-CoV-2 main protease provides a basis for design of improved alpha-ketoamide inhibitors. Science 2020, 368, 409–412. [Google Scholar] [CrossRef]
- Dai, W.H.; Zhang, B.; Jiang, X.M.; Su, X.H.; Li, L.; Zhao, Y.; Xie, X.; Jin, Z.M.; Peng, J.J.; Liu, F.J.; et al. Structure-based design of antiviral drug candidates targeting the SARS-CoV-2 main protease. Science 2020, 368, 1331–1335. [Google Scholar] [CrossRef]
- Jin, Z.M.; Du, X.Y.; Xu, Y.C.; Deng, Y.Q.; Liu, M.Q.; Zhao, Y.; Zhang, B.; Li, X.F.; Zhang, L.K.; Peng, C.; et al. Structure of Mpro from SARS-CoV-2 and discovery of its inhibitors. Nature 2020, 582, 289–293. [Google Scholar] [CrossRef]
- Liu, X.; Wang, X.J. Potential inhibitors against 2019-nCoV coronavirus M protease from clinically approved medicines. J. Genet. Genom. 2020, 47, 119–121. [Google Scholar] [CrossRef]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef]
- Yan, R.; Zhang, Y.; Li, Y.; Xia, L.; Guo, Y.; Zhou, Q. Structural basis for the recognition of the SARS-CoV-2 by full-length human ACE2. Science 2020, 367, 1444–1448. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Pomplun, S.; Loftis1, A.R.; Loas, A.; Pentelute, B.L. The first-in-class peptide binder to the SARS-CoV-2 spike protein. bioRxiv 2020. [Google Scholar] [CrossRef]
- Han, Y.X.; Král, P. Computational design of ACE2-based peptide inhibitors of SARS-CoV-2. ACS Nano 2020, 14, 5143–5147. [Google Scholar] [CrossRef] [PubMed]
- Baig, M.S.; Alagumuthu, M.; Rajpoot, S.; Saqib, U. Identification of a potential peptide inhibitor of SARS-CoV-2 targeting its entry into the host cells. Drugs R&D 2020, 20, 161–169. [Google Scholar] [CrossRef]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the Coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA 2020. [Google Scholar] [CrossRef]
- Zhang, J.; Zeng, H.; Gu, J.; Li, H.; Zheng, L.; Zou, Q. Progress and Prospects on Vaccine Development against SARS-CoV-2. Vaccines 2020, 8, 153–165. [Google Scholar] [CrossRef]
- Mak, T.M.; Saunders, M.E. Vaccines and Clinical Immunization. In The Immune Response; Mak, T.M., Saunders, M.E., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; pp. 695–749. [Google Scholar]
- AlKhazindar, M.; Elnagdy, S.M. Can Lactoferrin Boost Human Immunity Against COVID-19? Pathog. Glob. Health 2020. [Google Scholar] [CrossRef]
- Sherman, M.P.; Pritzl, C.J.; Xia, C.; Miller, M.M.; Zaghouani, H.; Hahm, B. Lactoferrin Acts as an Adjuvant During Influenza Vaccination of Neonatal Mice. Biochem. Biophys. Res. Commun. 2015, 467, 766–770. [Google Scholar] [CrossRef]
- Hossain, A.K.M.M. The Effect of Model Foods Rich in Bioactive Compounds on Brain-Gut Regulation and Neurodegeneration. Ph.D. Thesis, Lincoln University, Lincoln, New Zealand, 2018. [Google Scholar]
- Mada, S.B.; Ugwu, C.P.; Abarshi, M.A. Health Promoting Effects of Food-Derived Bioactive Peptides: A Review. Int. J. Pept. Res. Ther. 2020, 26, 831–848. [Google Scholar] [CrossRef]
- Jayaprakash, R.; Perera, C.O. Partial purification and characterization of bioactive peptides from cooked New Zealand green-lipped mussel (Perna canaliculus) protein hydrolyzates. Foods 2020, 9, 1–19. [Google Scholar] [CrossRef]
- Faridy, J.C.M.; Paredes-López, O. Plant Foods Hum. Nutr; Springer: Berlin, Germany, 2020; Volume 75, pp. 1573–9104. [Google Scholar]
- Minkiewicz, P.; Iwaniak, A.; Darewicz, M. BIPEP-UWM Database of bioactive peptides: Current opportunities. Int. J. Mol. Sci. 2015, 16, 20748–20773. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Wang, Q.; Qian, J.; Liang, Q.; Wang, Z.; Xu, J.; He, S.; Ma, H. Bioavailability and bioavailable forms of collagen after oral administration to rats. J. Agric. Food Chem. 2015, 63, 3752–3756. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.; Fogacci, F.; Coletti, A. Potential role of bioactive peptides in prevention and treatment of chronic diseases: A narrative review. Brit. J. Pharm. 2017, 174, 1378–1394. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.; Colletti, A. Nutraceuticals and blood pressure control: Results from clinical trials and meta-analyses. High Blood Press. Cardiovasc. Prev. 2015, 22, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Maestri, E.; Marmiroli, M.; Marmiroli, N. Bioactive peptides in plant-derived foodstuffs. J. Proteom. 2016, 147, 140–155. [Google Scholar] [CrossRef]
- Bouglé, D.; Bouhallab, S. Dietary bioactive peptides: Human studies. Crit. Rev. Food. Sci. Nutr. 2017, 57, 335–343. [Google Scholar] [CrossRef]
- Panchaud, A.M.; Affolter, M.; Kussmann, M. Mass spectrometry for nutritional peptidomics: How to analyse food bioactives and their health effects. J. Proteom. 2012, 75, 3546–3559. [Google Scholar] [CrossRef]
- Harnedy, P.A.; FitzGerald, R.J. Bioactive peptides from marine processing waste and shellfish: A review. J. Funct. Foods 2012, 4, 6–24. [Google Scholar] [CrossRef]
- Zheng, Z.; Jiang, H.; Huang, Y.; Wang, J.; Qiu, L.; Hu, Z.; Ma, X.; Lu, Y. Screening of an anti-inflammatory peptide from Hydrophis cyanocinctus and analysis of its activities and mechanism in DSS-induced acute colitis. Sci. Rep. 2016, 6, 25672–25683. [Google Scholar] [CrossRef]
- Wu, G.; Wang, J.; Luo, P.; Li, A.; Tian, S.; Jiang, H.; Xia, Z. Hydrostatin-SN1, a sea snake-derived bioactive peptide, reduces inflammation in a mouse model of acute lung injury. Front. Pharmacol. 2017, 8, 246–255. [Google Scholar] [CrossRef]
- Quah, Y.; Mohd Ismail, N.I.; Ooi, J.L.S.; Affendi, Y.A.; Abd Manan, F.; Teh, L.K.; Wong, F.C.; Chai, T.T. Purification and identification of novel cytotoxic oligopeptides from soft coral Sarcophyton glaucum. J. Zhejiang Univ. Sci. B 2019, 20, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Quah, Y.; Mohd Ismail, N.I.; Ooi, J.L.S.; Affendi, Y.A.; Abd Manan, F.; Wong, F.-C.; Chai, T.-T. Identification of novel cytotoxic peptide KENPVLSLVNGMF from marine sponge Xestospongia testudinaria, with characterization of stability in human serum. Int. J. Pept. Res. Ther. 2018, 24, 189–199. [Google Scholar] [CrossRef]
- Nikoo, M.; Xu, X.; Ahmadi Gavlighi, H. Chapter 17-Seafood waste-derived peptides: Their antioxidant activity and potential as alternative preservatives in fish products. In Protein Byproducts; Dhillon, G.S., Ed.; Academic Press: New York, NY, USA, 2016; pp. 315–332. [Google Scholar]
- Chai, T.T.; Law, Y.C.; Wong, F.C.; Kim, S.K. Enzyme-assisted discovery of antioxidant peptides from edible marine invertebrates: A review. Mar. Drugs 2017, 15, 42–68. [Google Scholar] [CrossRef]
- Zhang, L.; Falla, T.J. Cosmeceuticals and peptides. Clin. Dermatol. 2009, 27, 485–494. [Google Scholar] [CrossRef]
- Lima, T.N.; Pedriali Moraes, C.A. Bioactive peptides: Applications and relevance. Cosmetics 2018, 5, 21. [Google Scholar] [CrossRef]
- Pandey, A.; Jatana, G.K.; Sonthalia, S. “Cosmeceuticals.” StatPearls. Available online: https://www.ncbi.nlm.nih.gov/pubmed/31334943 (accessed on 10 February 2020).
- Bojarska, J. Amino Acids and Short Peptides as Anti-Aging “Superfood”. Int. J. Nutr. Sci. 2020, 5, 1039–1044. [Google Scholar]
- Negahdaripour, M.; Owji, H.; Eslami, M.; Zamani, M.; Vakili, B.; Sabetian, S. Selected application of peptide molecules as pharmaceutical agents and in cosmeceuticals. Expert Opin. Biol. Ther. 2019, 19, 1275–1287. [Google Scholar] [CrossRef]
- Errante, F.; Ledwoń, P.; Latajka, R.; Rovero, P.; Papini, A.M. Cosmeceutical peptides in the framework of sustainable wellness economy. Front. Chem. 2020, 8. [Google Scholar] [CrossRef]
- Schagen, S.K. Topical peptide treatments with effective anti-aging results. Cosmetics 2017, 4, 16–30. [Google Scholar] [CrossRef]
- Global Wellness Institute. Global Wellness Economy; Monitor: Miami, FL, USA, 2018; Available online: https://globalwellnessinstitute.org/2020 (accessed on 14 January 2021).
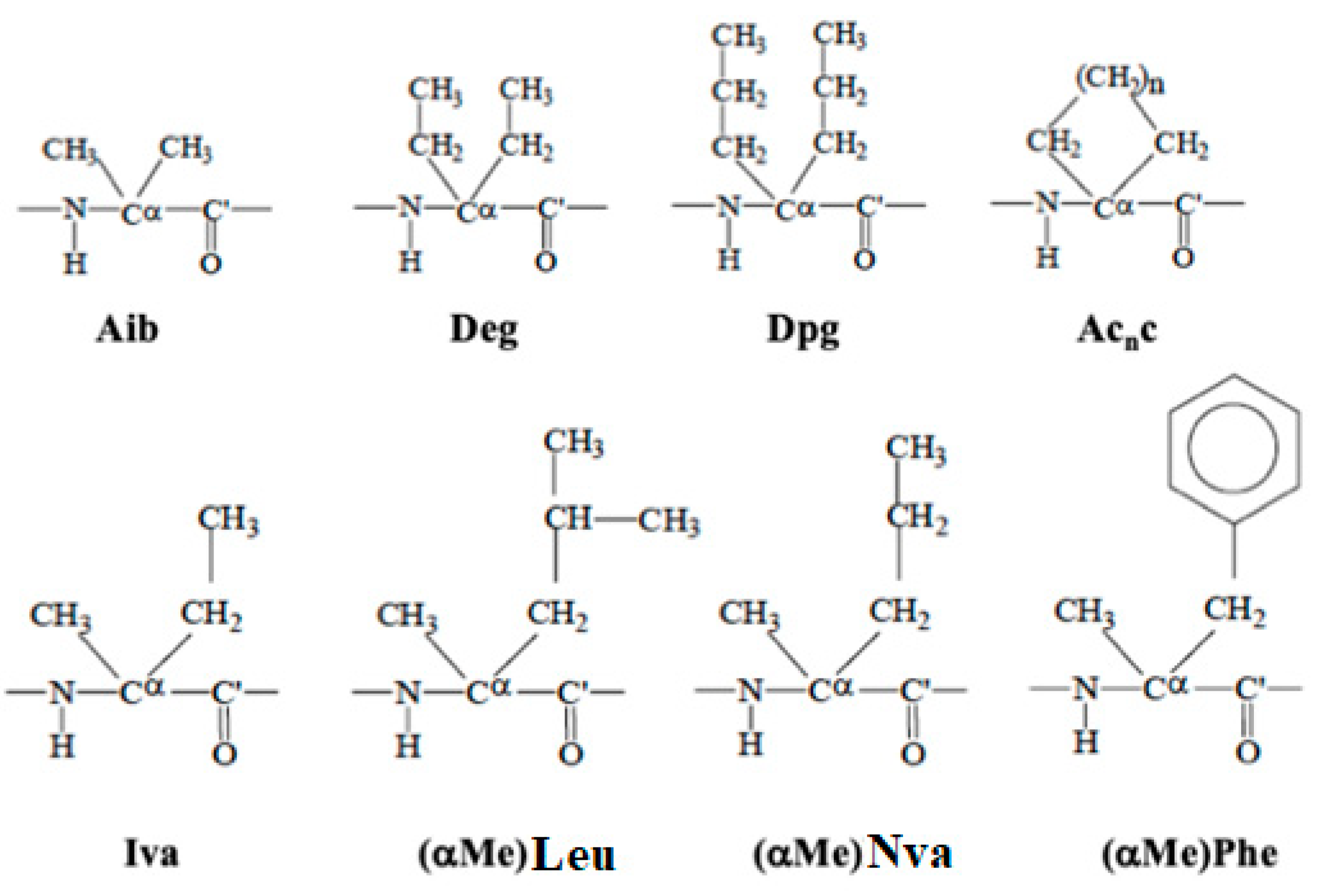
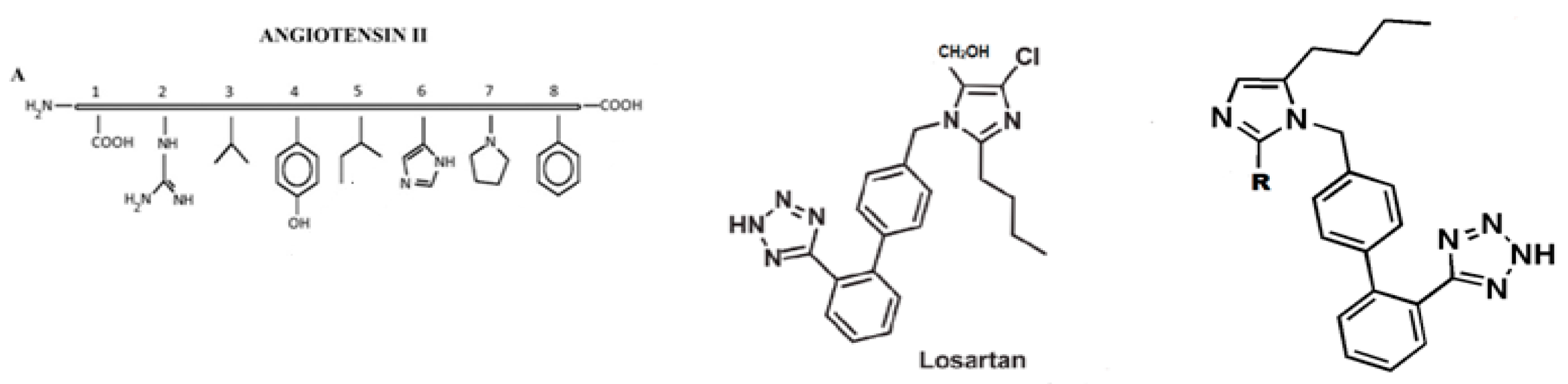
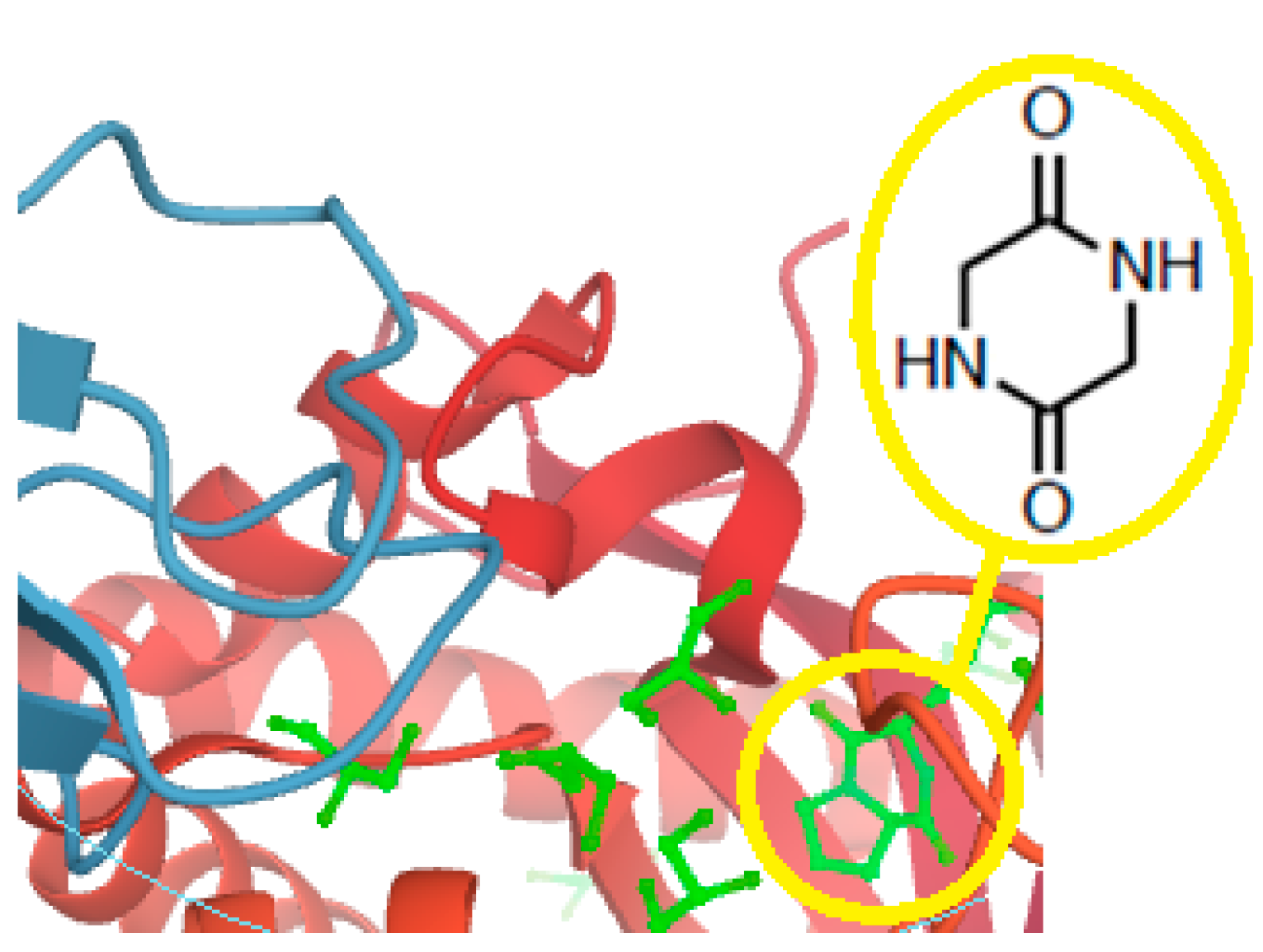
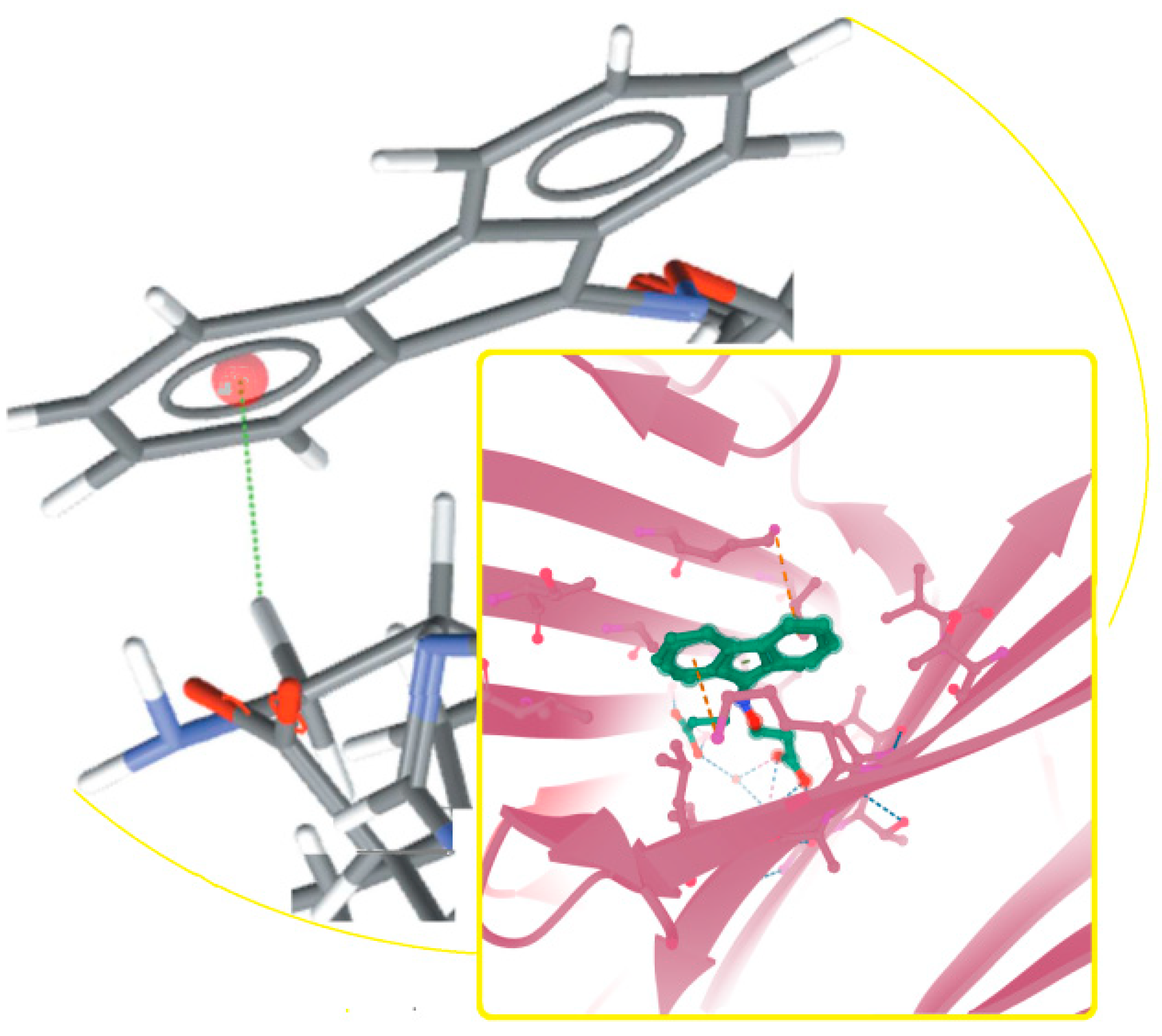
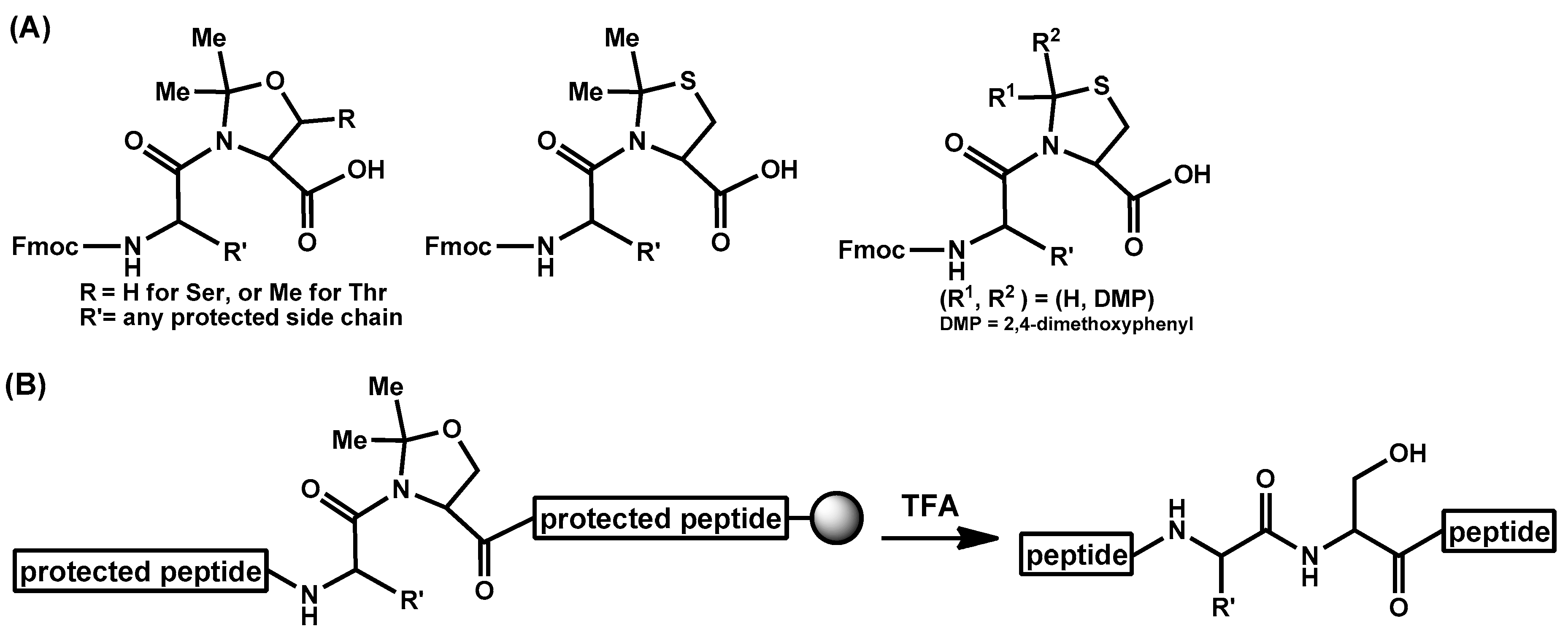
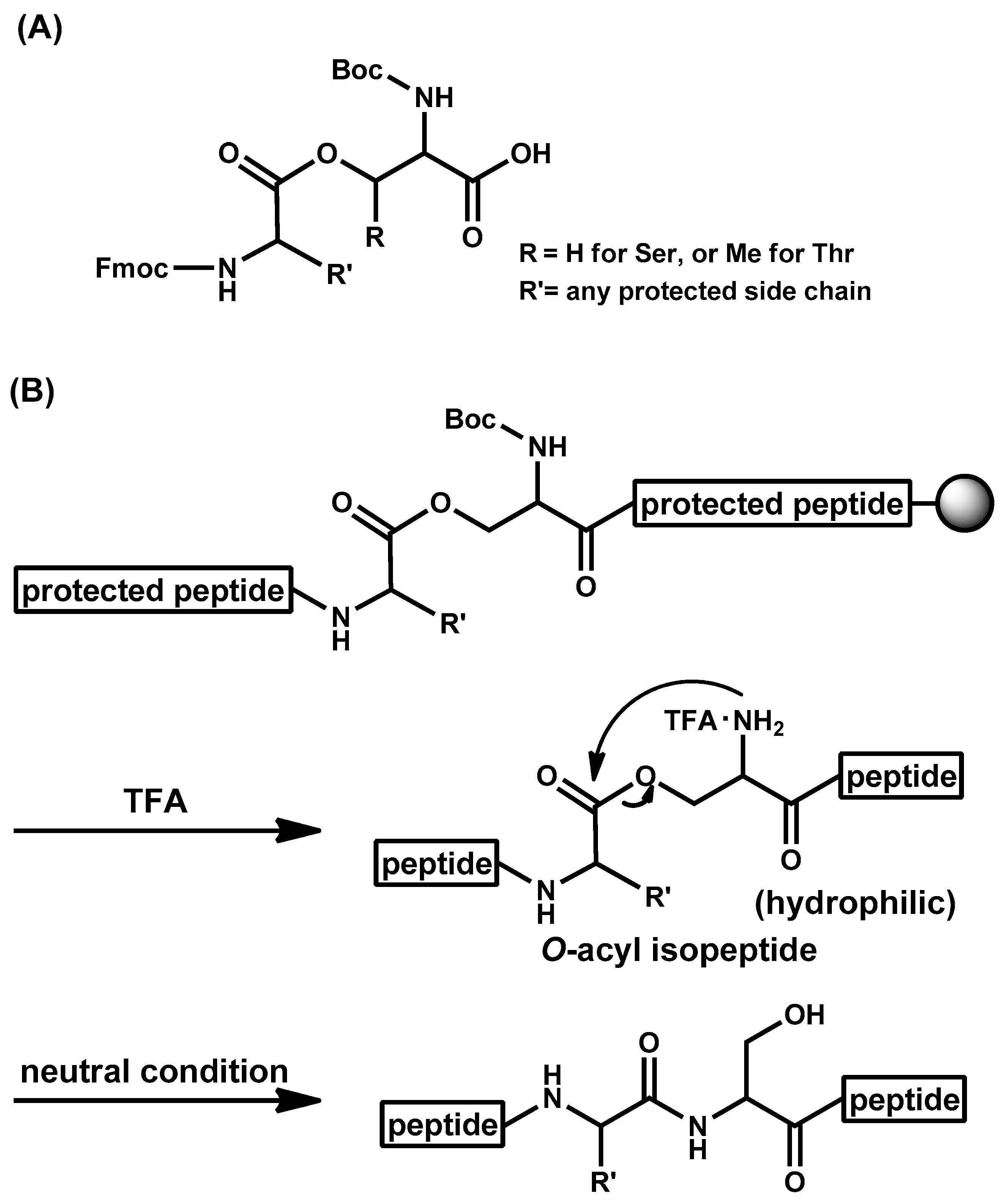

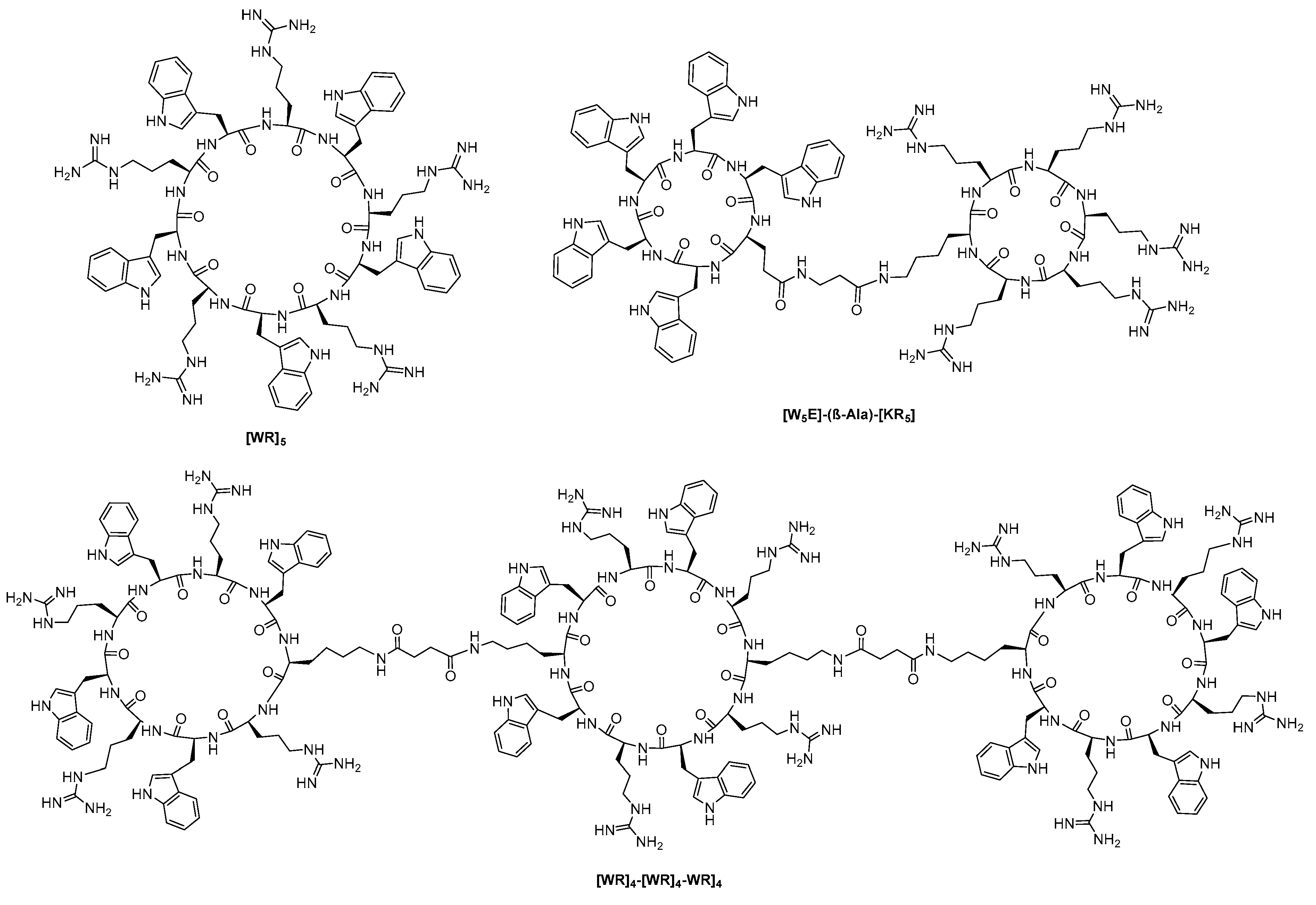
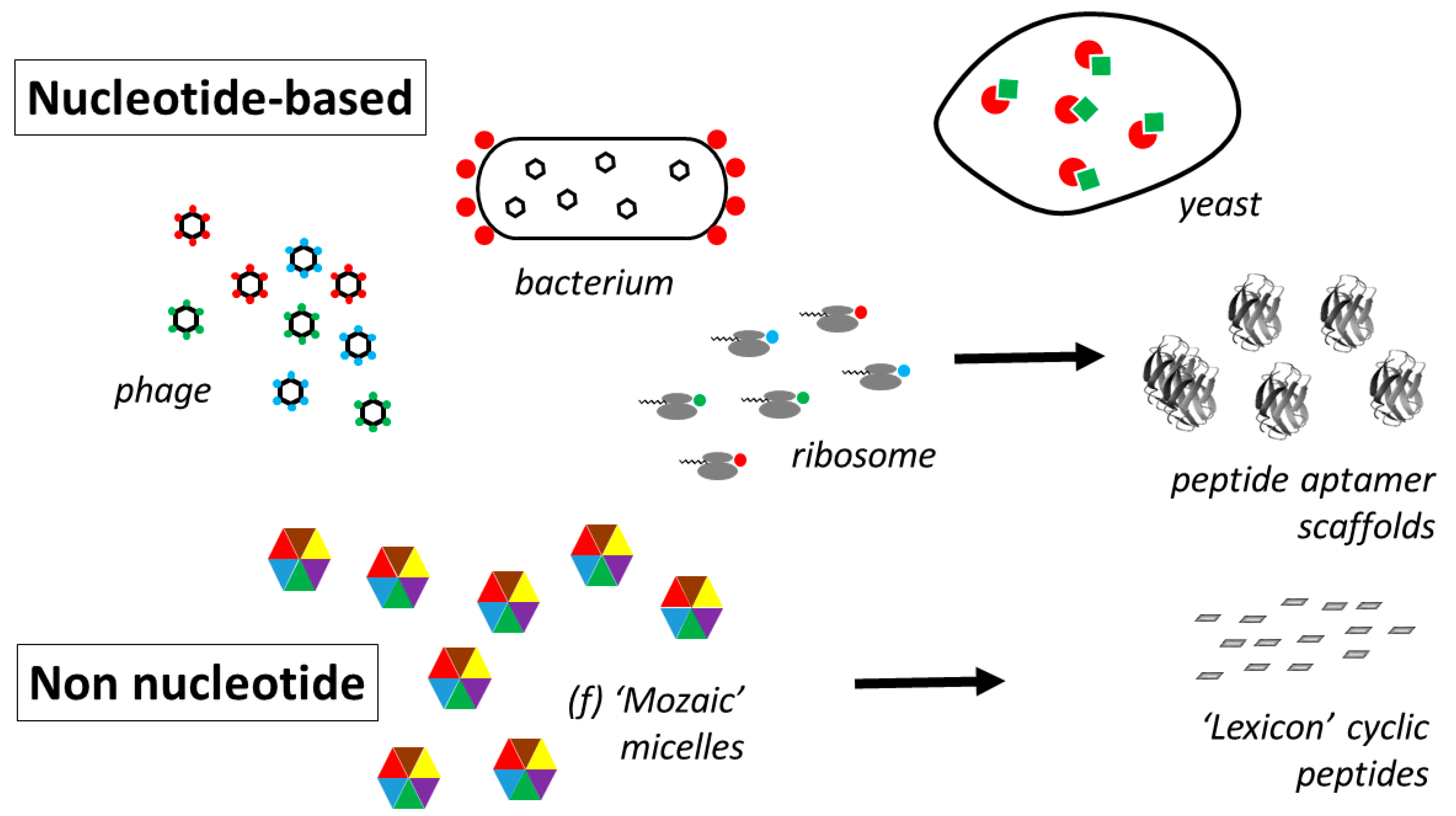
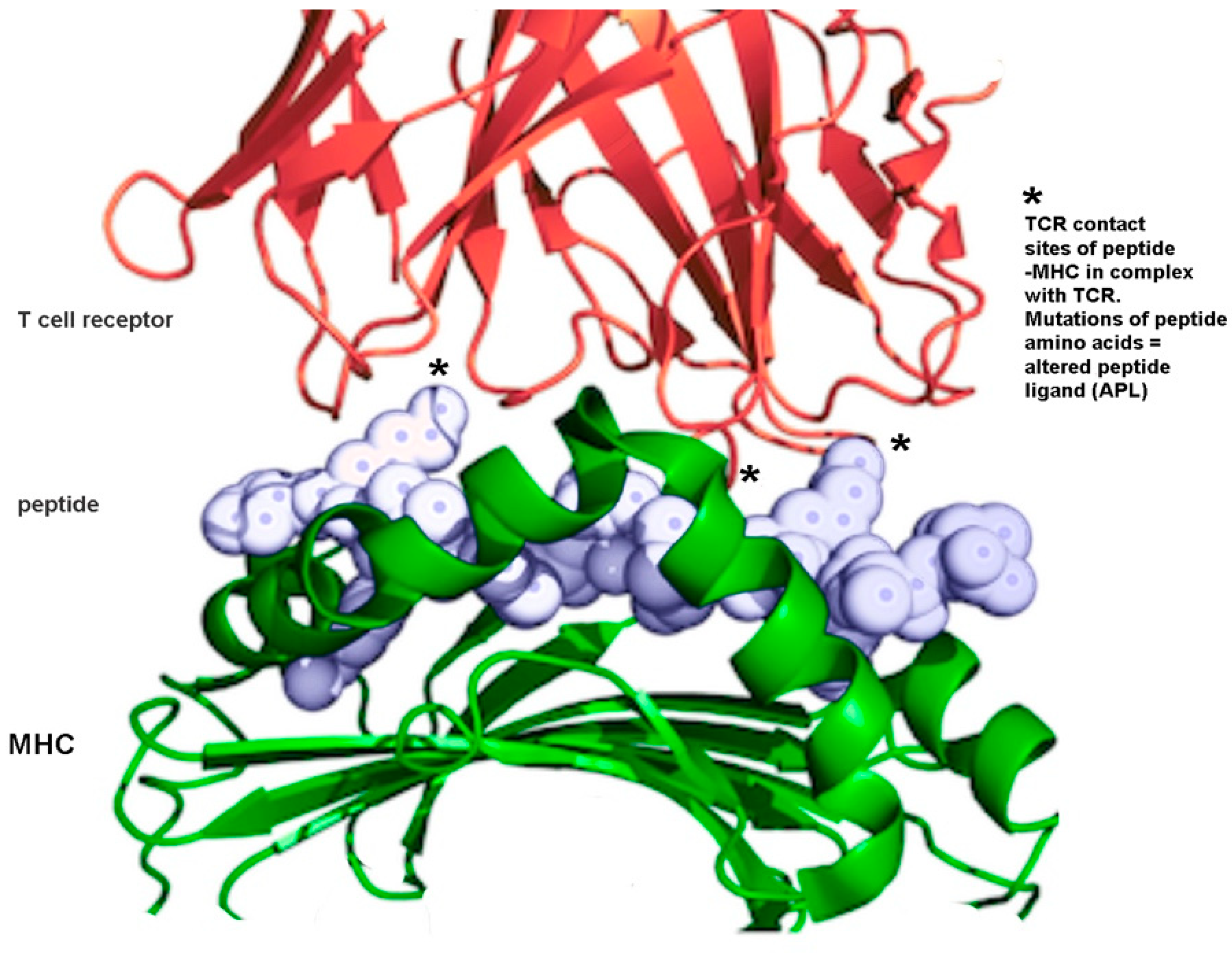
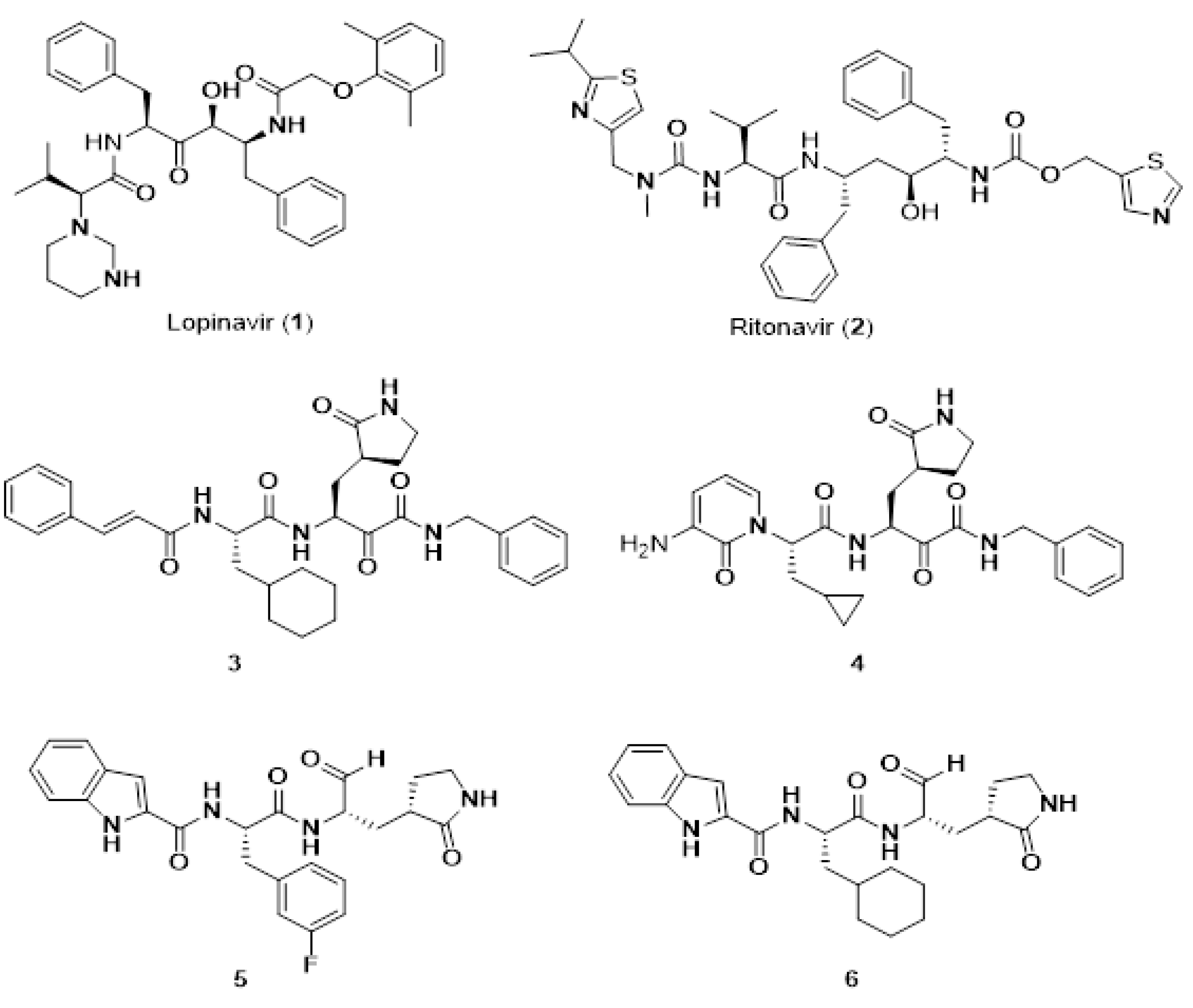
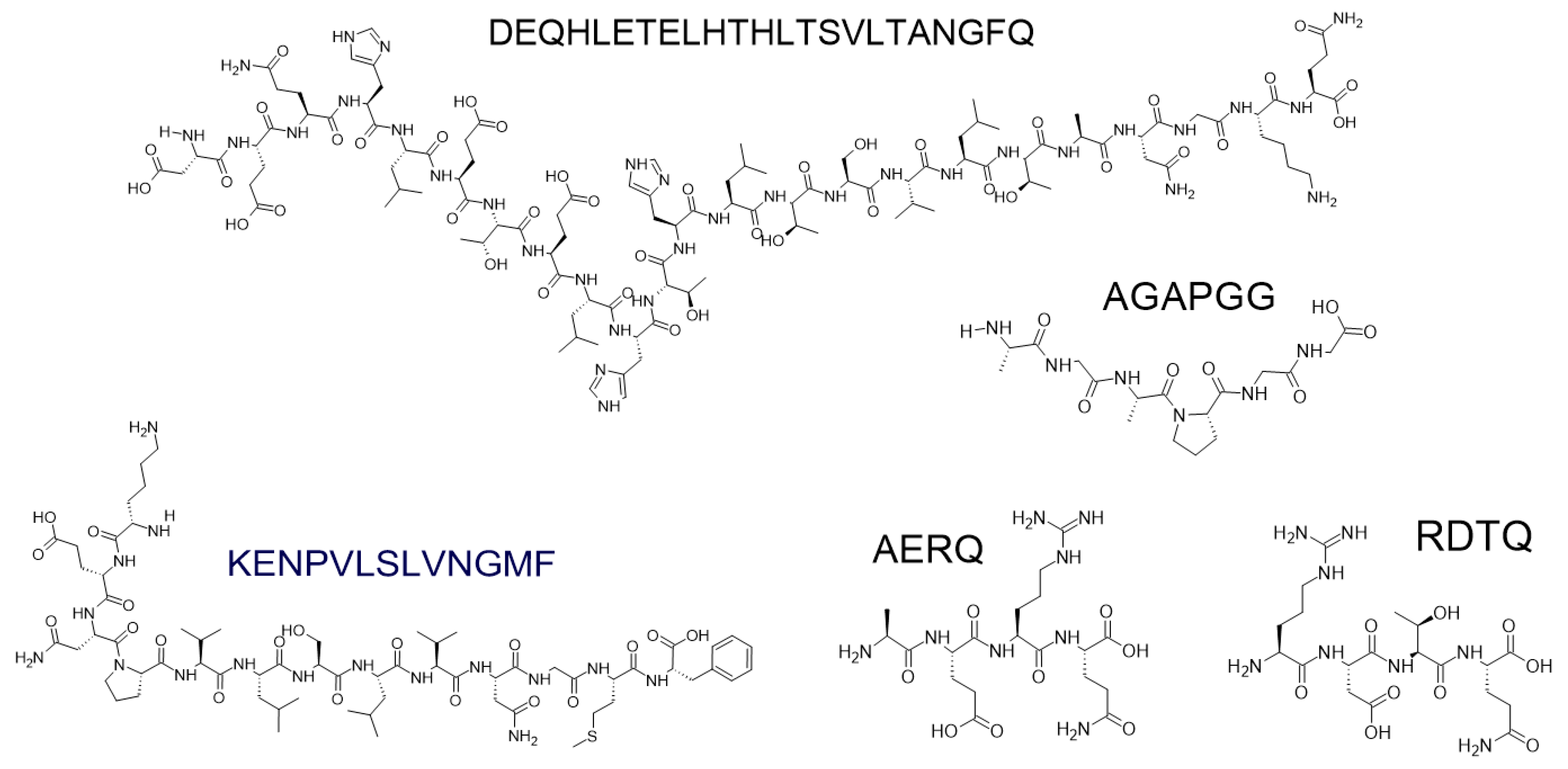
| Strengths | Weaknesses |
| essential bio-molecules with a broad range of activities & functionalities in vivo | instability in vivo (easy degradation in plasma, protease sensitivity) |
| bio-chemical diversity, easy availability | short half-life |
| structural simplicity | low (oral) bioavailability |
| easy design & cost-effective synthesis with high purity | difficult membrane permeability in the case of greater peptides * |
| easy modification, scaling up | low binding affinity * |
| mechanical stability | high conformational freedom * |
| high: modularity, flexibility *, selectivity, target specificity, affinity *, absorbability, potency, tolerability, efficacy, safety, biocompatibility, biodegradibility | |
| low toxicity, antigenicity, immunogenicity | |
| easy recognition by bio-systems | |
| ability to penetrate the cell membranes (but only very short peptides) *, high brain penetration in systematical administration | |
| versatility as both targeting moieties and therapeutic agents | |
| specific interactions with various bio-systems | |
| predictable metabolism: degradation products are amino acids (non-toxic, natural entities used as nutrients or cellular building blocks) | |
| lack or fewer secondary off-targets (side) effects (peptides do not accumulate in kidney or liver) | |
| low unspecific binding to the structures other than the desired target, minimisation of drug-drug interactions, less accumulation in tissues (low risk of complications due to intermediate metabolites) | |
| Opportunities | Threats |
| development of peptide-based delivery systems: - cell-penetrating peptides - nano-cyclic peptide-based micceles, vesicles as gene or drug carriers - conjugations with non-peptidic motifs | oncogenicity of endogenous & synthetic peptides |
| supramolecular peptide-based biofunctional materials | immunogenicity (related to greater peptides) |
| formulations development (e.g., subcutaneous injections)various forms of using (drugs, vaccines, hormones, radioisotopes) | |
| development of the peptide-based safe & effective vaccines | |
| diveristy of well-ordered, robust, long-lived self-assembled nanostructures | |
| vital tool for neurodegenerative diseases studies & various applications in anticancer therapy | |
| peptoids or peptidomimetics |
| Name of Peptide | No. of Amino Acids |
|---|---|
| diphenylalanine | 2 |
| α,β-dehydrophenylalanine | 2 |
| Fmoc-diphenylalanine-konjac glucomannan | 2 |
| Ac-EFFAAE-NH2(AIP-1/2) | 6 |
| FFKLVFF | 7 |
| P11 (QQEFQWQFRQQ) | 11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Apostolopoulos, V.; Bojarska, J.; Chai, T.-T.; Elnagdy, S.; Kaczmarek, K.; Matsoukas, J.; New, R.; Parang, K.; Lopez, O.P.; Parhiz, H.; et al. A Global Review on Short Peptides: Frontiers and Perspectives. Molecules 2021, 26, 430. https://doi.org/10.3390/molecules26020430
Apostolopoulos V, Bojarska J, Chai T-T, Elnagdy S, Kaczmarek K, Matsoukas J, New R, Parang K, Lopez OP, Parhiz H, et al. A Global Review on Short Peptides: Frontiers and Perspectives. Molecules. 2021; 26(2):430. https://doi.org/10.3390/molecules26020430
Chicago/Turabian StyleApostolopoulos, Vasso, Joanna Bojarska, Tsun-Thai Chai, Sherif Elnagdy, Krzysztof Kaczmarek, John Matsoukas, Roger New, Keykavous Parang, Octavio Paredes Lopez, Hamideh Parhiz, and et al. 2021. "A Global Review on Short Peptides: Frontiers and Perspectives" Molecules 26, no. 2: 430. https://doi.org/10.3390/molecules26020430
APA StyleApostolopoulos, V., Bojarska, J., Chai, T.-T., Elnagdy, S., Kaczmarek, K., Matsoukas, J., New, R., Parang, K., Lopez, O. P., Parhiz, H., Perera, C. O., Pickholz, M., Remko, M., Saviano, M., Skwarczynski, M., Tang, Y., Wolf, W. M., Yoshiya, T., Zabrocki, J., ... Toth, I. (2021). A Global Review on Short Peptides: Frontiers and Perspectives. Molecules, 26(2), 430. https://doi.org/10.3390/molecules26020430

















