Therapeutic Potential of Natural Products in Treating Neurodegenerative Disorders and Their Future Prospects and Challenges
Abstract
1. Introduction
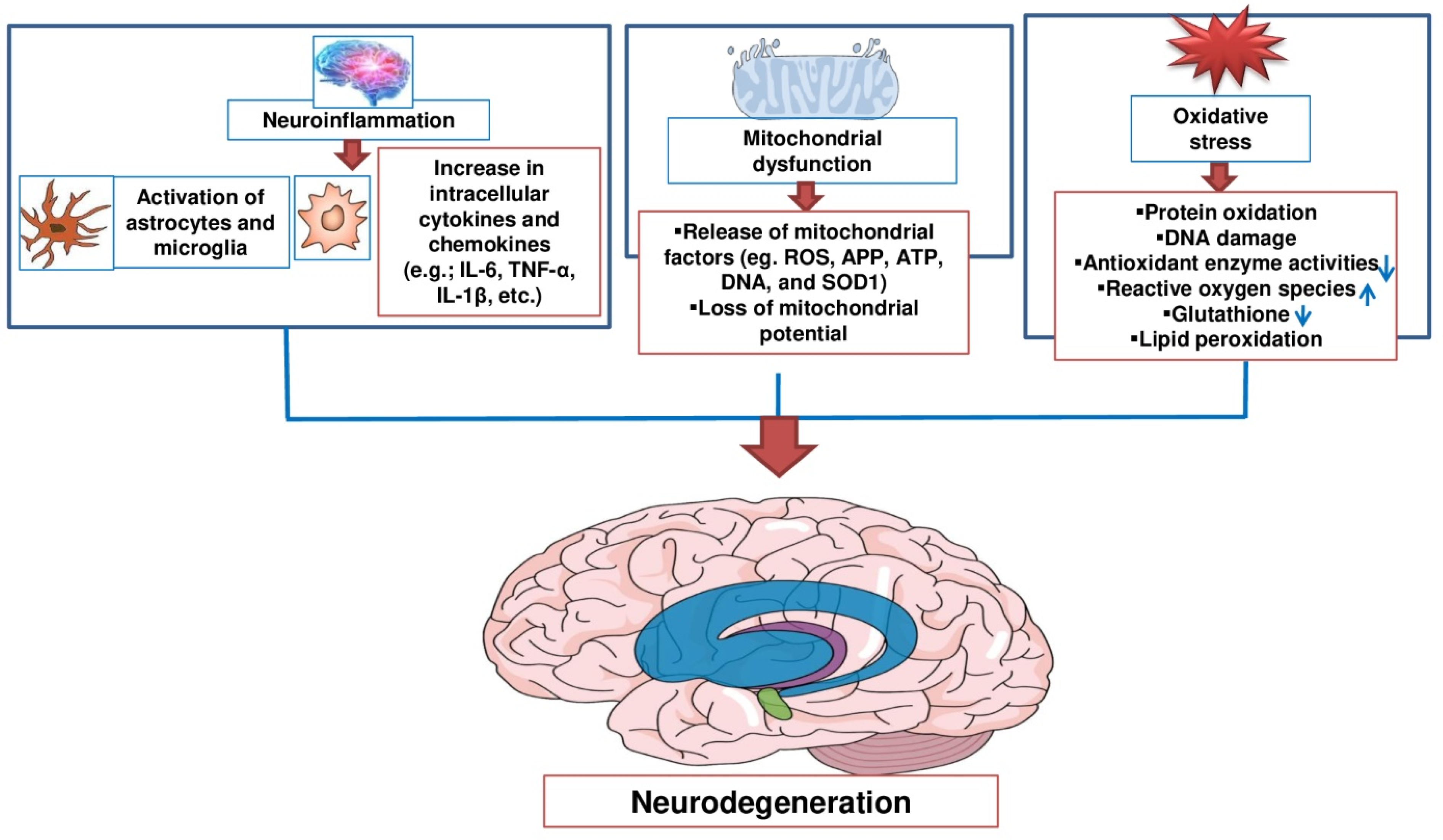
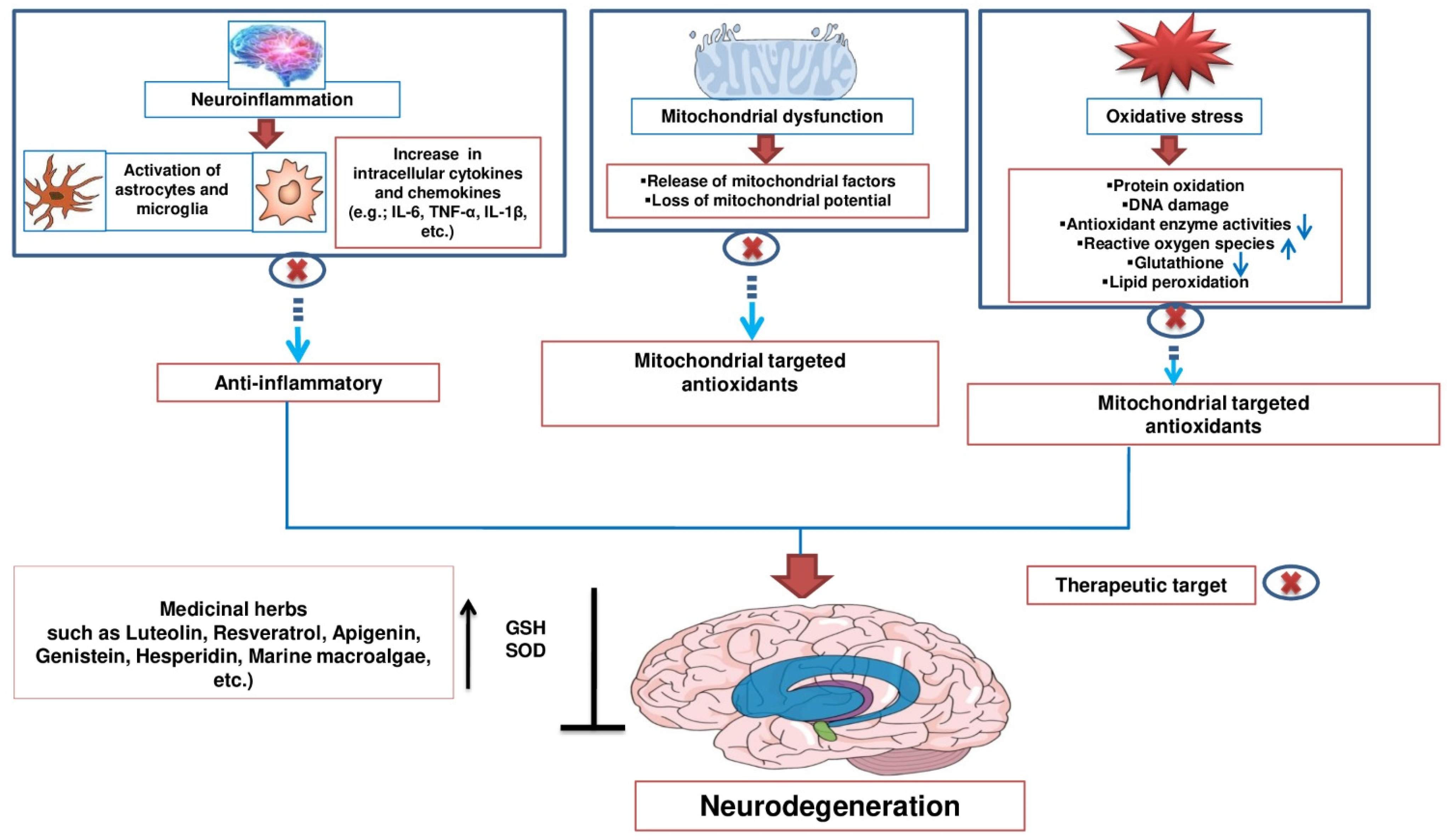
2. Potential Therapeutic Targets of Natural Products against Neurodegenerative Diseases
3. Neuroprotective Activities of Numerous Natural Products
3.1. Luteolin
3.2. Quercetin
3.3. Resveratrol
3.4. Apigenin
3.5. Genistein
3.6. Hesperidin
3.7. Uncaria Rhynchophylla
3.8. Marine Macroalgae
3.9. Cyanobacteria
4. Role of Other Natural Products in Neurodegenerative Diseases
5. Limitations, Future Prospects, and Challenges
6. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 6-OHDA | 6-hydroxydopamine |
| AD | Alzheimer’s disease |
| AlCl3 | Aluminum chloride |
| AP | Apigenin |
| APP | Amyloid precursor protein |
| Aβ | Amyloid-beta |
| Bax | Bcl-2-associated X proteins |
| BBB | Blood-brain barrier |
| Bcl2 | B-cell lymphoma 2 |
| CAG | Cytosine-adenine-guanine |
| DHM | Dihydromyricetin |
| DNA | Deoxyribonucleic acid |
| ERK | Extracellular signal-regulated kinase |
| GSH | Glutathione |
| GSK-3β | Glycogen synthase kinase-3β |
| HD | Huntington’s disease |
| IL | Interleukin |
| L-DOPA | Levodopa |
| Lu | Luteolin |
| MAPK | Mitogen-activated protein kinase |
| MPP+ | 1-methyl-4-phenylpyridinium |
| MPTP | 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| NDs | Neurodegenerative diseases |
| NF-κB | Nuclear factor-kappa B |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| PD | Parkinson’s disease |
| POMS | Profile of Mood States |
| PTK | Protein tyrosine kinase |
| QCT | Quercetin |
| ROS | Reactive oxygen species |
| RSV | Resveratrol |
| SOD-1 | Superoxide dismutase-1 |
| T-AOC | Total antioxidant ability |
| TNF-α | Tumor necrosis factor-α |
References
- Rahman, M.H.; Bajgai, J.; Fadriquela, A.; Sharma, S.; Trinh Thi, T.; Akter, R.; Goh, S.H.; Kim, C.S.; Lee, K.J. Redox effects of molecular hydrogen and its therapeutic efficacy in the treatment of neurodegenerative diseases. Processes 2021, 9, 308. [Google Scholar] [CrossRef]
- Salvadores, N.; Court, F.A. The necroptosis pathway and its role in age-related neurodegenerative diseases: Will it open up new therapeutic avenues in the next decade? Expert Opin. Ther. Targets 2020, 24, 679–693. [Google Scholar] [CrossRef]
- Yildiz-Unal, A.; Korulu, S.; Karabay, A. Neuroprotective strategies against calpain-mediated neurodegeneration. Neuropsychiatr. Dis. Treat. 2015, 11, 297–310. [Google Scholar] [CrossRef]
- Di Paolo, M.; Papi, L.; Gori, F.; Turillazzi, E. Natural products in neurodegenerative diseases: A great promise but an ethical challenge. Int. J. Mol. Sci. 2019, 20, 5170. [Google Scholar] [CrossRef]
- Thirupathi, A.; Chang, Y.Z. Brain iron metabolism and cns diseases. Adv. Exp. Med. Biol. 2019, 1173, 1–19. [Google Scholar] [CrossRef]
- Troncoso-Escudero, P.; Sepulveda, D.; Perez-Arancibia, R.; Parra, A.V.; Arcos, J.; Grunenwald, F.; Vidal, R.L. On the right track to treat movement disorders: Promising therapeutic approaches for parkinson’s and huntington’s disease. Front. Aging Neurosci. 2020, 12, 571185. [Google Scholar] [CrossRef]
- Harischandra, D.S.; Ghaisas, S.; Zenitsky, G.; Jin, H.; Kanthasamy, A.; Anantharam, V.; Kanthasamy, A.G. Manganese-induced neurotoxicity: New insights into the triad of protein misfolding, mitochondrial impairment, and neuroinflammation. Front. Neurosci. 2019, 13, 654. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.; Hardy, J.; Duff, K.E. Selective vulnerability in neurodegenerative diseases. Nat. Neurosci. 2018, 21, 1350–1358. [Google Scholar] [CrossRef] [PubMed]
- Gan, L.; Cookson, M.R.; Petrucelli, L.; La Spada, A.R. Converging pathways in neurodegeneration, from genetics to mechanisms. Nat. Neurosci. 2018, 21, 1300–1309. [Google Scholar] [CrossRef]
- Singh, A.; Kukreti, R.; Saso, L.; Kukreti, S. Oxidative stress: A key modulator in neurodegenerative diseases. Molecules 2019, 24, 1583. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Dawson, T.M.; Dawson, V.L. Cell death mechanisms of neurodegeneration. Adv. Neurobiol. 2017, 15, 403–425. [Google Scholar] [CrossRef]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The role of polyphenols in human health and food systems: A mini-review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chu, S.; Liu, Y.; Chen, N. Neuroprotective effects of anthraquinones from rhubarb in central nervous system diseases. Evid. Based Complement. Alternat. Med. 2019, 2019, 3790728. [Google Scholar] [CrossRef]
- Lutz, M.; Fuentes, E.; Avila, F.; Alarcon, M.; Palomo, I. Roles of phenolic compounds in the reduction of risk factors of cardiovascular diseases. Molecules 2019, 24, 366. [Google Scholar] [CrossRef]
- Pham, L.; Wright, D.K.; O’Brien, W.T.; Bain, J.; Huang, C.; Sun, M.; Casillas-Espinosa, P.M.; Shah, A.D.; Schittenhelm, R.B.; Sobey, C.G.; et al. Behavioral, axonal, and proteomic alterations following repeated mild traumatic brain injury: Novel insights using a clinically relevant rat model. Neurobiol. Dis. 2021, 148, 105151. [Google Scholar] [CrossRef] [PubMed]
- Bui, T.T.; Nguyen, T.H. Natural product for the treatment of alzheimer’s disease. J. Basic Clin. Physiol. Pharmacol. 2017, 28, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Babaei, F.; Mirzababaei, M.; Nassiri-Asl, M. Quercetin in food: Possible mechanisms of its effect on memory. J. Food Sci. 2018, 83, 2280–2287. [Google Scholar] [CrossRef] [PubMed]
- Obeso, J.A.; Stamelou, M.; Goetz, C.G.; Poewe, W.; Lang, A.E.; Weintraub, D.; Burn, D.; Halliday, G.M.; Bezard, E.; Przedborski, S.; et al. Past, present, and future of parkinson’s disease: A special essay on the 200th anniversary of the shaking palsy. Mov. Disord. 2017, 32, 1264–1310. [Google Scholar] [CrossRef]
- Grieco, M.; Giorgi, A.; Gentile, M.C.; d’Erme, M.; Morano, S.; Maras, B.; Filardi, T. Glucagon-like peptide-1: A focus on neurodegenerative diseases. Front. Neurosci. 2019, 13, 1112. [Google Scholar] [CrossRef]
- Shahmoradian, S.H.; Lewis, A.J.; Genoud, C.; Hench, J.; Moors, T.E.; Navarro, P.P.; Castano-Diez, D.; Schweighauser, G.; Graff-Meyer, A.; Goldie, K.N.; et al. Lewy pathology in parkinson’s disease consists of crowded organelles and lipid membranes. Nat. Neurosci. 2019, 22, 1099–1109. [Google Scholar] [CrossRef] [PubMed]
- Hunn, B.H.; Cragg, S.J.; Bolam, J.P.; Spillantini, M.G.; Wade-Martins, R. Impaired intracellular trafficking defines early parkinson’s disease. Trends Neurosci. 2015, 38, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Hilton, J.B.; White, A.R.; Crouch, P.J. Metal-deficient sod1 in amyotrophic lateral sclerosis. J. Mol. Med. 2015, 93, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Sirangelo, I.; Iannuzzi, C. The role of metal binding in the amyotrophic lateral sclerosis-related aggregation of copper-zinc superoxide dismutase. Molecules 2017, 22, 1429. [Google Scholar] [CrossRef]
- Bostan, A.C.; Strick, P.L. The basal ganglia and the cerebellum: Nodes in an integrated network. Nat. Rev. Neurosci. 2018, 19, 338–350. [Google Scholar] [CrossRef] [PubMed]
- Gil, J.M.; Rego, A.C. Mechanisms of neurodegeneration in huntington’s disease. Eur. J. Neurosci. 2008, 27, 2803–2820. [Google Scholar] [CrossRef]
- Hsu, Y.T.; Chang, Y.G.; Chern, Y. Insights into gabaaergic system alteration in huntington’s disease. Open Biol. 2018, 8, 180165. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, S.L.; van Belzen, M.J.; Boogaard, M.W.; van Roon-Mom, W.M.; Rozing, M.P.; van Hemert, A.M.; Smit, J.H.; Beekman, A.T.; van Grootheest, G.; Schoevers, R.A.; et al. Huntingtin gene repeat size variations affect risk of lifetime depression. Transl. Psychiatry 2017, 7, 1277. [Google Scholar] [CrossRef] [PubMed]
- Bhat, A.H.; Dar, K.B.; Anees, S.; Zargar, M.A.; Masood, A.; Sofi, M.A.; Ganie, S.A. Oxidative stress, mitochondrial dysfunction and neurodegenerative diseases; a mechanistic insight. Biomed. Pharmacother. 2015, 74, 101–110. [Google Scholar] [CrossRef]
- Golpich, M.; Amini, E.; Mohamed, Z.; Azman Ali, R.; Mohamed Ibrahim, N.; Ahmadiani, A. Mitochondrial dysfunction and biogenesis in neurodegenerative diseases: Pathogenesis and treatment. CNS Neurosci. Ther. 2017, 23, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Ullah, R.; Khan, M.; Shah, S.A.; Saeed, K.; Kim, M.O. Natural antioxidant anthocyanins-a hidden therapeutic candidate in metabolic disorders with major focus in neurodegeneration. Nutrients 2019, 11, 1195. [Google Scholar] [CrossRef] [PubMed]
- Nita, M.; Grzybowski, A. The role of the reactive oxygen species and oxidative stress in the pathomechanism of the age-related ocular diseases and other pathologies of the anterior and posterior eye segments in adults. Oxid. Med. Cell. Longev. 2016, 2016, 3164734. [Google Scholar] [CrossRef] [PubMed]
- Buendia, I.; Michalska, P.; Navarro, E.; Gameiro, I.; Egea, J.; Leon, R. Nrf2-are pathway: An emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol. Ther. 2016, 157, 84–104. [Google Scholar] [CrossRef]
- Gelders, G.; Baekelandt, V.; Van der Perren, A. Linking neuroinflammation and neurodegeneration in parkinson’s disease. J. Immunol. Res. 2018, 2018, 4784268. [Google Scholar] [CrossRef]
- Leszek, J.; Barreto, G.E.; Gasiorowski, K.; Koutsouraki, E.; Avila-Rodrigues, M.; Aliev, G. Inflammatory mechanisms and oxidative stress as key factors responsible for progression of neurodegeneration: Role of brain innate immune system. CNS Neurol. Disord. Drug. Targets 2016, 15, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, J.; Nutma, E.; van der Valk, P.; Amor, S. Inflammation in cns neurodegenerative diseases. Immunology 2018, 154, 204–219. [Google Scholar] [CrossRef]
- Thurgur, H.; Pinteaux, E. Microglia in the neurovascular unit: Blood-brain barrier-microglia interactions after central nervous system disorders. Neuroscience 2019, 405, 55–67. [Google Scholar] [CrossRef]
- Voet, S.; Prinz, M.; van Loo, G. Microglia in central nervous system inflammation and multiple sclerosis pathology. Trends Mol. Med. 2019, 25, 112–123. [Google Scholar] [CrossRef]
- Kabba, J.A.; Xu, Y.; Christian, H.; Ruan, W.; Chenai, K.; Xiang, Y.; Zhang, L.; Saavedra, J.M.; Pang, T. Microglia: Housekeeper of the central nervous system. Cell. Mol. Neurobiol. 2018, 38, 53–71. [Google Scholar] [CrossRef]
- Cardenas, C.; Lovy, A.; Silva-Pavez, E.; Urra, F.; Mizzoni, C.; Ahumada-Castro, U.; Bustos, G.; Jana, F.; Cruz, P.; Farias, P.; et al. Cancer cells with defective oxidative phosphorylation require endoplasmic reticulum-to-mitochondria Ca2+ transfer for survival. Sci. Signal. 2020, 13, eaay1212. [Google Scholar] [CrossRef]
- Panov, A.; Dikalov, S.; Shalbuyeva, N.; Hemendinger, R.; Greenamyre, J.T.; Rosenfeld, J. Species- and tissue-specific relationships between mitochondrial permeability transition and generation of ros in brain and liver mitochondria of rats and mice. Am. J. Physiol. Cell Physiol. 2007, 292, C708–C718. [Google Scholar] [CrossRef]
- Rao, V.K.; Carlson, E.A.; Yan, S.S. Mitochondrial permeability transition pore is a potential drug target for neurodegeneration. Biochim. Biophys. Acta Mol. Basis Dis. 2014, 1842, 1267–1272. [Google Scholar] [CrossRef]
- Gadd, M.E.; Broekemeier, K.M.; Crouser, E.D.; Kumar, J.; Graff, G.; Pfeiffer, D.R. Mitochondrial ipla2 activity modulates the release of cytochrome c from mitochondria and influences the permeability transition. J. Biol. Chem. 2006, 281, 6931–6939. [Google Scholar] [CrossRef]
- Osellame, L.D.; Rahim, A.A.; Hargreaves, I.P.; Gegg, M.E.; Richard-Londt, A.; Brandner, S.; Waddington, S.N.; Schapira, A.H.; Duchen, M.R. Mitochondria and quality control defects in a mouse model of gaucher disease-links to parkinson’s disease. Cell Metab. 2013, 17, 941–953. [Google Scholar] [CrossRef] [PubMed]
- Poewe, W.; Seppi, K.; Tanner, C.M.; Halliday, G.M.; Brundin, P.; Volkmann, J.; Schrag, A.E.; Lang, A.E. Parkinson disease. Nat. Rev. Dis. Primers 2017, 3, 17013. [Google Scholar] [CrossRef] [PubMed]
- Kent, J.A.; Patel, V.; Varela, N.A. Gender disparities in health care. Mt. Sinai J. Med. 2012, 79, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Bano, D.; Dinsdale, D.; Cabrera-Socorro, A.; Maida, S.; Lambacher, N.; McColl, B.; Ferrando-May, E.; Hengartner, M.O.; Nicotera, P. Alteration of the nuclear pore complex in Ca2+-mediated cell death. Cell. Death. Differ. 2010, 17, 119–133. [Google Scholar] [CrossRef]
- Verma, D.K.; Gupta, S.; Biswas, J.; Joshi, N.; Sivarama Raju, K.; Wahajuddin, M.; Singh, S. Metabolic enhancer piracetam attenuates the translocation of mitochondrion-specific proteins of caspase-independent pathway, poly [adp-ribose] polymerase 1 up-regulation and oxidative DNA fragmentation. Neurotox. Res. 2018, 34, 198–219. [Google Scholar] [CrossRef]
- Srivastava, P.; Yadav, R.S. Efficacy of natural compounds in neurodegenerative disorders. In The Benefits of Natural Products for Neurodegenerative Diseases; Springer: Cham, Switzerland, 2016; pp. 107–123. [Google Scholar] [CrossRef]
- Akter, R.; Chowdhury, M.; Rahman, A.; Rahman, H. Flavonoids and polyphenolic compounds as potential talented agents for the treatment of alzheimer’s disease with their antioxidant activities. Curr. Pharm. Des. 2020, 27, 345–356. [Google Scholar] [CrossRef]
- Wang, B.; Lu, Y.; Wang, R.; Liu, S.; Hu, X.; Wang, H. Transport and metabolic profiling studies of amentoflavone in caco-2 cells by uhplc-esi-ms/ms and uhplc-esi-q-tof-ms/ms. J. Pharm. Biomed. Anal. 2020, 189, 113441. [Google Scholar] [CrossRef]
- Williamson, G.; Kay, C.D.; Crozier, A. The bioavailability, transport, and bioactivity of dietary flavonoids: A review from a historical perspective. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1054–1112. [Google Scholar] [CrossRef]
- Nabavi, S.F.; Braidy, N.; Gortzi, O.; Sobarzo-Sanchez, E.; Daglia, M.; Skalicka-Wozniak, K.; Nabavi, S.M. Luteolin as an anti-inflammatory and neuroprotective agent: A brief review. Brain Res. Bull. 2015, 119, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Yulyana, Y.; Tovmasyan, A.; Ho, I.A.; Sia, K.C.; Newman, J.P.; Ng, W.H.; Guo, C.M.; Hui, K.M.; Batinic-Haberle, I.; Lam, P.Y. Redox-active Mn porphyrin-based potent SOD mimic, MnTnBuOE-2-PyP5+, enhances carbenoxolone-mediated TRAIL-induced apoptosis in glioblastoma multiforme. Stem. Cell Rev. Rep. 2016, 12, 140–155. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Koo, E.H. The amyloid precursor protein: Beyond amyloid. Mol. Neurodegener. 2006, 1, 11–12. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Uddin, M.S.; Hossain, M.F.; Mamun, A.A.; Shah, M.A.; Hasana, S.; Bulbul, I.J.; Sarwar, M.S.; Mansouri, R.A.; Ashraf, G.M.; Rauf, A.; et al. Exploring the multimodal role of phytochemicals in the modulation of cellular signaling pathways to combat age-related neurodegeneration. Sci. Total Environ. 2020, 725, 138313. [Google Scholar] [CrossRef] [PubMed]
- Calis, Z.; Mogulkoc, R.; Baltaci, A.K. The roles of flavonols/flavonoids in neurodegeneration and neuroinflammation. Mini Rev. Med. Chem. 2020, 20, 1475–1488. [Google Scholar] [CrossRef]
- Khan, H.; Ullah, H.; Aschner, M.; Cheang, W.S.; Akkol, E.K. Neuroprotective effects of quercetin in alzheimer’s disease. Biomolecules 2020, 10, 59. [Google Scholar] [CrossRef]
- Batiha, G.E.; Beshbishy, A.M.; Ikram, M.; Mulla, Z.S.; El-Hack, M.E.; Taha, A.E.; Algammal, A.M.; Elewa, Y.H. The pharmacological activity, biochemical properties, and pharmacokinetics of the major natural polyphenolic flavonoid: Quercetin. Foods 2020, 9, 374. [Google Scholar] [CrossRef]
- Schultke, E. The Flavonoid Quercetin and Its Potential As Neuroprotectant in the Therapy of Acute traumatic CNS Injury: An Experimental Study. Ph.D. Thesis, University of Saskatchewan, Saskatoon, SK, Canada, 2004. [Google Scholar]
- Jurcau, A. The role of natural antioxidants in the prevention of dementia-where do we stand and future perspectives. Nutrients 2021, 13, 282. [Google Scholar] [CrossRef]
- Kumar, G.P.; Anilakumar, K.R.; Naveen, S. Phytochemicals having neuroprotective properties from dietary sources and medicinal herbs. Pharmacogn. J. 2015, 7, 1–17. [Google Scholar] [CrossRef]
- Costa, L.G.; Garrick, J.M.; Roque, P.J.; Pellacani, C. Mechanisms of neuroprotection by quercetin: Counteracting oxidative stress and more. Oxid. Med. Cell. Longev. 2016, 2016, 2986796. [Google Scholar] [CrossRef]
- Faria, A.; Pestana, D.; Teixeira, D.; Azevedo, J.; Freitas, V.; Mateus, N.; Calhau, C. Flavonoid transport across RBE4 cells: A blood-brain barrier model. Cell. Mol. Biol. Lett. 2010, 1, 234–241. [Google Scholar] [CrossRef]
- Ishisaka, A.; Ichikawa, S.; Sakakibara, H.; Piskula, M.K.; Nakamura, T.; Kato, Y.; Ito, M.; Miyamoto, K.I.; Tsuji, A.; Kawai, Y.; et al. Accumulation of orally administered quercetin in brain tissue and its antioxidative effects in rats. Free Radic. Biol.Med. 2011, 51, 1329–1336. [Google Scholar] [CrossRef]
- Ferri, P.; Angelino, D.; Gennari, L.; Benedetti, S.; Ambrogini, P.; Del Grande, P.; Ninfali, P. Enhancement of flavonoid ability to cross the blood–brain barrier of rats by co-administration with α-tocopherol. Food Funct. 2015, 6, 394–400. [Google Scholar] [CrossRef]
- Yang, P.; Xu, F.; Li, H.F.; Wang, Y.; Li, F.C.; Shang, M.Y.; Liu, G.X.; Wang, X.; Cai, S.Q. Detection of 191 taxifolin metabolites and their distribution in rats using HPLC-ESI-IT-TOF-MS(n). Molecules 2016, 21, 1209. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Q.; Bao, X.; Ding, Y.; Shentu, J.; Cui, W. Taxifolin prevents β-amyloid-induced impairments of synaptic formation and deficits of memory via the inhibition of cytosolic phospholipase A2/prostaglandin E2 content. Metab. Brain Dis. 2018, 33, 1069–1079. [Google Scholar] [CrossRef]
- Saito, S.; Yamamoto, Y.; Maki, T.; Hattori, Y.; Ito, H.; Mizuno, K. Taxifolin inhibits amyloid-β oligomer formation and fully restores vascular integrity and memory in cerebral amyloid angiopathy. Acta. Neuropathol. Commun. 2017, 5, 26. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.H.; Akter, R.; Bhattacharya, T.; Abdel-Daim, M.M.; Alkahtani, S.; Arafah, M.W.; Al-Johani, N.S.; Alhoshani, N.M.; Alkeraishan, N.; Alhenaky, A.; et al. Resveratrol and neuroprotection: Impact and its therapeutic potential in alzheimer’s disease. Front. Pharmacol. 2020, 11, 619024. [Google Scholar] [CrossRef]
- Bastianetto, S.; Menard, C.; Quirion, R. Neuroprotective action of resveratrol. Biochim. Biophys. Acta 2015, 1852, 1195–1201. [Google Scholar] [CrossRef]
- Hou, Y.; Wang, K.; Wan, W.; Cheng, Y.; Pu, X.; Ye, X. Resveratrol provides neuroprotection by regulating the JAK2/STAT3/PI3K/AKT/mTOR pathway after stroke in rats. Genes Dis. 2018, 5, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Koushki, M.; Amiri-Dashatan, N.; Ahmadi, N.; Abbaszadeh, H.A.; Rezaei-Tavirani, M. Resveratrol: A miraculous natural compound for diseases treatment. Food Sci. Nutr. 2018, 6, 2473–2490. [Google Scholar] [CrossRef]
- Iside, C.; Scafuro, M.; Nebbioso, A.; Altucci, L. Sirt1 activation by natural phytochemicals: An overview. Front. Pharmacol. 2020, 11, 1225. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; Lucarini, M.; Souto, E.B.; Cicala, C.; Caiazzo, E.; Izzo, A.A.; Novellino, E.; Santini, A. Polyphenols: A concise overview on the chemistry, occurrence, and human health. Phytother. Res. 2019, 33, 2221–2243. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, P.E.; Beraldi, E.J.; Borges, S.C.; Natali, M.R.; Buttow, N.C. Resveratrol promotes neuroprotection and attenuates oxidative and nitrosative stress in the small intestine in diabetic rats. Biomed. Pharmacother. 2018, 105, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Tan, M.S.; Yu, J.T.; Tan, L. Resveratrol as a therapeutic agent for alzheimer’s disease. Biomed. Res. Int. 2014, 2014, 350516. [Google Scholar] [CrossRef]
- Shahidi, F.; Peng, H. Bioaccessibility and bioavailability of phenolic compounds. J. Food Bioact. 2018, 4, 11–68. [Google Scholar] [CrossRef]
- Davidov-Pardo, G.; McClements, D.J. Resveratrol encapsulation: Designing delivery systems to overcome solubility, stability and bioavailability issues. Trends Food Sci. Technol. 2014, 38, 88–103. [Google Scholar] [CrossRef]
- Sathya, M.; Moorthi, P.; Premkumar, P.; Kandasamy, M.; Jayachandran, K.S.; Anusuyadevi, M. Resveratrol intervenes cholesterol and isoprenoid-mediated amyloidogenic processing of abetapp in familial alzheimer’s disease. J. Alzheimers Dis. 2017, 60, S3–S23. [Google Scholar] [CrossRef]
- Lopez, M.S.; Dempsey, R.J.; Vemuganti, R. Resveratrol neuroprotection in stroke and traumatic cns injury. Neurochem. Int. 2015, 89, 75–82. [Google Scholar] [CrossRef]
- Zhang, G.; Liu, Y.; Xu, L.; Sha, C.; Zhang, H.; Xu, W. Resveratrol alleviates lipopolysaccharide-induced inflammation in PC-12 cells and in rat model. BMC Biotechnol. 2019, 19, 10. [Google Scholar] [CrossRef]
- Hou, Y.; Zhang, Y.; Mi, Y.; Wang, J.; Zhang, H.; Xu, J.; Yang, Y.; Liu, J.; Ding, L.; Yang, J.; et al. A novel quinolyl-substituted analogue of resveratrol inhibits LPS-induced inflammatory responses in microglial cells by blocking the NF-κB/MAPK signaling pathways. Mol. Nutr. Food Res. 2019, 63, e1801380. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Mishra, A.P.; Nigam, M.; Sener, B.; Kilic, M.; Sharifi-Rad, M.; Fokou, P.V.; Martins, N.; Sharifi-Rad, J. Resveratrol: A double-edged sword in health benefits. Biomedicines 2018, 6, 91. [Google Scholar] [CrossRef] [PubMed]
- Deane, R.; Wu, Z.; Sagare, A.; Davis, J.; Du Yan, S.; Hamm, K.; Xu, F.; Parisi, M.; LaRue, B.; Hu, H.W.; et al. LRP/amyloid β-peptide interaction mediates differential brain efflux of Aβ isoforms. Neuron 2004, 43, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Saha, A.; Sarkar, C.; Singh, S.P.; Zhang, Z.; Munasinghe, J.; Peng, S.; Chandra, G.; Kong, E.; Mukherjee, A.B. The blood-brain barrier is disrupted in a mouse model of infantile neuronal ceroid lipofuscinosis: Amelioration by resveratrol. Hum. Mol. Genet. 2012, 21, 2233–2244. [Google Scholar] [CrossRef]
- Moussa, C.; Hebron, M.; Huang, X.; Ahn, J.; Rissman, R.A.; Aisen, P.S.; Turner, R.S. Resveratrol regulates neuro-inflammation and induces adaptive immunity in Alzheimer’s disease. J. Neuroinflamm. 2017, 14, 1. [Google Scholar] [CrossRef]
- Nabavi, S.F.; Khan, H.; D’onofrio, G.; Samec, D.; Shirooie, S.; Dehpour, A.R.; Arguelles, S.; Habtemariam, S.; Sobarzo-Sanchez, E. Apigenin as neuroprotective agent: Of mice and men. Pharmacol. Res. 2018, 128, 359–365. [Google Scholar] [CrossRef]
- Yan, X.; Qi, M.; Li, P.; Zhan, Y.; Shao, H. Apigenin in cancer therapy: Anti-cancer effects and mechanisms of action. Cell Biosci. 2017, 7, 50. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.J.; Ai, C.Z.; Liu, Y.; Zhang, Y.Y.; Jiang, M.; Fan, X.R.; Lv, A.P.; Yang, L. Interactions between phytochemicals from traditional chinese medicines and human cytochrome p450 enzymes. Curr. Drug Metab. 2012, 13, 599–614. [Google Scholar] [CrossRef]
- Sanchez-Marzo, N.; Perez-Sanchez, A.; Ruiz-Torres, V.; Martinez-Tebar, A.; Castillo, J.; Herranz-Lopez, M.; Barrajon-Catalan, E. Antioxidant and photoprotective activity of apigenin and its potassium salt derivative in human keratinocytes and absorption in Caco-2 cell monolayers. Int. J. Mol. Sci. 2019, 20, 2148. [Google Scholar] [CrossRef] [PubMed]
- Siddique, Y.H.; Naz, F.; Jyoti, S.; Afzal, M. Protective effect of apigenin in transgenic drosophila melanogaster model of parkinson’s disease. Pharmacology 2011, 3, 790–795. [Google Scholar]
- Balez, R.; Steiner, N.; Engel, M.; Munoz, S.S.; Lum, J.S.; Wu, Y.; Wang, D.; Vallotton, P.; Sachdev, P.; O’Connor, M.; et al. Neuroprotective effects of apigenin against inflammation, neuronal excitability and apoptosis in an induced pluripotent stem cell model of alzheimer’s disease. Sci. Rep. 2016, 6, 31450. [Google Scholar] [CrossRef]
- Popović, M.; Caballero-Bleda, M.; Benavente-García, O.; Castillo, J. The flavonoid apigenin delays forgetting of passive avoidance conditioning in rats. J. Psychopharmacol. 2014, 28, 498–501. [Google Scholar] [CrossRef] [PubMed]
- Ganai, A.A.; Farooqi, H. Bioactivity of genistein: A review of in vitro and in vivo studies. Biomed. Pharmacother. 2015, 76, 30–38. [Google Scholar] [CrossRef]
- Rahman, M.H.; Akter, R.; Kamal, M.A. Prospective function of different antioxidant containing natural products in the treatment of neurodegenerative disease. CNS Neurol. Disord. Drug. Targets 2020. [Google Scholar] [CrossRef]
- Sheeja Malar, D.; Pandima Devi, K. Dietary polyphenols for treatment of alzheimer’s disease—Future research and development. Curr. Pharm. Biotechnol. 2014, 15, 330–342. [Google Scholar] [CrossRef]
- You, F.; Li, Q.; Jin, G.; Zheng, Y.; Chen, J.; Yang, H. Genistein protects against Aβ 25-35 induced apoptosis of PC12 cells through JNK signaling and modulation of BCL-2 family messengers. BMC Neurosci. 2017, 18, 12. [Google Scholar] [CrossRef]
- Bagheri, M.; Joghataei, M.T.; Mohseni, S.; Roghani, M. Genistein ameliorates learning and memory deficits in amyloid beta(1-40) rat model of alzheimer’s disease. Neurobiol. Learn. Mem. 2011, 95, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Dodsworth, C.; Paras, A.; Burton, B.K. High dose genistein aglycone therapy is safe in patients with mucopolysaccharidoses involving the central nervous system. Mol. Genet. Metab. 2013, 109, 382–385. [Google Scholar] [CrossRef]
- Hajialyani, M.; Farzaei, M.H.; Echeverria, J.; Nabavi, S.M.; Uriarte, E.; Sobarzo-Sanchez, E. Hesperidin as a neuroprotective agent: A review of animal and clinical evidence. Molecules 2019, 24, 648. [Google Scholar] [CrossRef] [PubMed]
- Bhadbhade, A.; Cheng, D.W. Amyloid precursor protein processing in alzheimer’s disease. Iran. J. Child Neurol. 2012, 6, 1–4. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, N.Q.; Yan, F.; Jin, H.; Zhou, S.Y.; Shi, J.S.; Jin, F. Diabetes mellitus and alzheimer’s disease: GSK-3β as a potential link. Behav. Brain Res. 2018, 339, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Jahan, S.; Imtiyaz, Z.; Alshahrani, S.; Antar Makeen, H.; Mohammed Alshehri, B.; Kumar, A.; Arafah, A.; Rehman, M.U. Neuroprotection: Targeting multiple pathways by naturally occurring phytochemicals. Biomedicines 2020, 8, 284. [Google Scholar] [CrossRef]
- Justin Thenmozhi, A.; William Raja, T.R.; Manivasagam, T.; Janakiraman, U.; Essa, M.M. Hesperidin ameliorates cognitive dysfunction, oxidative stress and apoptosis against aluminium chloride induced rat model of alzheimer’s disease. Nutr. Neurosci. 2017, 20, 360–368. [Google Scholar] [CrossRef]
- Kim, J.; Wie, M.B.; Ahn, M.; Tanaka, A.; Matsuda, H.; Shin, T. Benefits of hesperidin in central nervous system disorders: A review. Anat. Cell Biol. 2019, 52, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Wang, F.; Xiu, C.; Zhang, J.; Li, Y. Hypericum perforatum extract attenuates behavioral, biochemical, and neurochemical abnormalities in aluminum chloride-induced alzheimer’s disease rats. Biomed. Pharmacother. 2017, 91, 931–937. [Google Scholar] [CrossRef]
- Kumar, A.; Lalitha, S.; Mishra, J. Hesperidin potentiates the neuroprotective effects of diazepam and gabapentin against pentylenetetrazole-induced convulsions in mice: Possible behavioral, biochemical and mitochondrial alterations. Indian J. Pharmacol. 2014, 46, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.Y.; Lin, T.Y.; Lu, C.W.; Huang, S.K.; Wang, Y.C.; Chou, S.S.P.; Wang, S.J. Hesperidin inhibits glutamate release and exerts neuroprotection against excitotoxicity induced by kainic acid in the hippocampus of rats. Neurotoxicology 2015, 50, 157–169. [Google Scholar] [CrossRef]
- Xian, Y.F.; Lin, Z.X.; Mao, Q.Q.; Hu, Z.; Zhao, M.; Che, C.T.; Ip, S.P. Bioassay-guided isolation of neuroprotective compounds from uncaria rhynchophylla against beta-amyloid-induced neurotoxicity. Evid. Based. Complement. Alternat. Med. 2012, 2012, 802625. [Google Scholar] [CrossRef]
- Ling, L.Z.; Zhang, S.D. The complete chloroplast genome of the traditional chinese herb, uncaria rhynchophylla (rubiaceae). Mitochondrial. DNA B Resour. 2020, 5, 424–425. [Google Scholar] [CrossRef]
- Wang, Y.L.; Dong, P.P.; Liang, J.H.; Li, N.; Sun, C.P.; Tian, X.G.; Huo, X.K.; Zhang, B.J.; Ma, X.C.; Lv, C.Z. Phytochemical constituents from uncaria rhynchophylla in human carboxylesterase 2 inhibition: Kinetics and interaction mechanism merged with docking simulations. Phytomedicine 2018, 51, 120–127. [Google Scholar] [CrossRef]
- Yang, W.; Ip, S.P.; Liu, L.; Xian, Y.F.; Lin, Z.X. Uncaria rhynchophylla and its major constituents on central nervous system: A review on their pharmacological actions. Curr. Vasc. Pharmacol. 2020, 18, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.S.; Kim, H.G.; Ju, M.S.; Choi, J.G.; Jeong, S.Y.; Oh, M.S. Effects of the hook of uncaria rhynchophylla on neurotoxicity in the 6-hydroxydopamine model of parkinson’s disease. J. Ethnopharmacol. 2009, 126, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Pal, B.; Kumar, S.S. Evaluation of anti-parkinson’s activity of uncaria rhynchophylla in 6-hydroxy dopamine lesioned rat model. Int. J. Appl. Res. 2015, 1, 203–206. [Google Scholar]
- Lan, Y.L.; Zhou, J.J.; Liu, J.; Huo, X.K.; Wang, Y.L.; Liang, J.H.; Zhao, J.C.; Sun, C.P.; Yu, Z.L.; Fang, L.L.; et al. Uncaria rhynchophylla ameliorates parkinson’s disease by inhibiting HSP90 expression: Insights from quantitative proteomics. Cell. Physiol. Biochem. 2018, 47, 1453–1464. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.N.; Yang, Y.F.; Xu, W.; Yang, X.W. The blood-brain barrier permeability of six indole alkaloids from UncariaeRamulus Cum Uncis in the MDCK-pHaMDR cell monolayer model. Molecules 2017, 22, 1944. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wu, X.; Xian, Y.; Zhu, L.; Lin, G.; Lin, Z.X. Evidence on integrating pharmacokinetics to find truly therapeutic agent for Alzheimer’s disease: Comparative pharmacokinetics and disposition kinetics profiles of stereoisomers isorhynchophylline and rhynchophylline in rats. Evid. Based. Complement. Altern. Med. 2019, 2019, 4016323. [Google Scholar] [CrossRef]
- Thomas, N.V.; Kim, S.K. Beneficial effects of marine algal compounds in cosmeceuticals. Mar. Drugs 2013, 11, 146–164. [Google Scholar] [CrossRef]
- Overland, M.; Mydland, L.T.; Skrede, A. Marine macroalgae as sources of protein and bioactive compounds in feed for monogastric animals. J. Sci. Food. Agric. 2019, 99, 13–24. [Google Scholar] [CrossRef]
- Singh, R.; Parihar, P.; Singh, M.; Bajguz, A.; Kumar, J.; Singh, S.; Singh, V.P.; Prasad, S.M. Uncovering potential applications of cyanobacteria and algal metabolites in biology, agriculture and medicine: Current status and future prospects. Front. Microbiol. 2017, 8, 515. [Google Scholar] [CrossRef] [PubMed]
- Dawczynski, C.; Schafer, U.; Leiterer, M.; Jahreis, G. Nutritional and toxicological importance of macro, trace, and ultra-trace elements in algae food products. J. Agric. Food. Chem. 2007, 55, 10470–10475. [Google Scholar] [CrossRef]
- Chamorro, G.; Pérez-Albiter, M.; Serrano-García, N.; Mares-Sámano, J.J.; Rojas, P. Spirulina maxima pretreatment partially protects against 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine neurotoxicity. Nutr. Neurosci. 2006, 9, 207–212. [Google Scholar] [CrossRef]
- Silva, J.; Alves, C.; Pinteus, S.; Mendes, S.; Pedrosa, R. Neuroprotective effects of seaweeds against 6-hydroxidopamine-induced cell death on an in vitro human neuroblastoma model. BMC Complement. Altern. Med. 2018, 18, 58. [Google Scholar] [CrossRef] [PubMed]
- Cunha, L.; Grenha, A. Sulfated seaweed polysaccharides as multifunctional materials in drug delivery applications. Mar. Drugs 2016, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, A.I.; Coutinho, A.J.; Lima, S.A.; Reis, S. Marine polysaccharides in pharmaceutical applications: Fucoidan and chitosan as key players in the drug delivery match field. Mar. Drugs 2019, 17, 654. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Hao, J.; Zheng, Y.; Su, R.; Liao, Y.; Gong, X.; Liu, L.; Wang, X. Fucoidan protects dopaminergic neurons by enhancing the mitochondrial function in a rotenone-induced rat model of parkinson’s disease. Aging Dis. 2018, 9, 590–604. [Google Scholar] [CrossRef]
- Wei, H.; Gao, Z.; Zheng, L.; Zhang, C.; Liu, Z.; Yang, Y.; Teng, H.; Hou, L.; Yin, Y.; Zou, X. Protective effects of fucoidan on Aβ25-35 and d-Gal-induced neurotoxicity in PC12 Cells and d-Gal-induced cognitive dysfunction in mice. Mar. Drugs 2017, 15, 77. [Google Scholar] [CrossRef]
- Mohd Sairazi, N.S.; Sirajudeen, K.N. Natural products and their bioactive compounds: Neuroprotective potentials against neurodegenerative diseases. Evid. Based Complement. Alternat. Med. 2020, 2020, 6565396. [Google Scholar] [CrossRef]
- Pal, S.; Paul, S. Conformational deviation of thrombin binding G-quadruplex aptamer (TBA) in presence of divalent cation Sr2+: A classical molecular dynamics simulation study. Int. J. Biol. Macromol. 2019, 121, 350–363. [Google Scholar] [CrossRef]
- Hannan, M.A.; Dash, R.; Haque, M.N.; Mohibbullah, M.; Sohag, A.A.; Rahman, M.A.; Uddin, M.J.; Alam, M.; Moon, I. Neuroprotective potentials of marine algae and their bioactive metabolites: Pharmacological insights and therapeutic advances. Mar. Drugs. 2020, 18, 347. [Google Scholar] [CrossRef] [PubMed]
- Kiuru, P.; D’Auria, M.V.; Muller, C.D.; Tammela, P.; Vuorela, H.; Yli-Kauhaluoma, J. Exploring marine resources for bioactive compounds. Planta. Med. 2014, 80, 1234–1246. [Google Scholar] [CrossRef]
- Moradi-Kor, N.; Ghanbari, A.; Rashidipour, H.; Bandegi, A.R.; Youse fi, B.; Barati, M.; Kokhaei, P.; Rashidy-Pour, A. Therapeutic effects of spirulina platensis against adolescent stress-induced oxidative stress, brain-derived neurotrophic factor alterations and morphological remodeling in the amygdala of adult female rats. J. Exp. Pharmacol. 2020, 12, 75–85. [Google Scholar] [CrossRef]
- Zhang, F.; Lu, J.; Zhang, J.G.; Xie, J.X. Protective effects of a polysaccharide from spirulina platensis on dopaminergic neurons in an MPTP-induced parkinson’s disease model in C57BL/6J mice. Neural. Regen. Res. 2015, 10, 308–313. [Google Scholar] [CrossRef]
- Huang, C.; Zhang, Z.; Cui, W. Marine-derived natural compounds for the treatment of parkinson’s disease. Mar. Drugs 2019, 17, 221. [Google Scholar] [CrossRef] [PubMed]
- Koh, E.J.; Kim, K.J.; Song, J.H.; Choi, J.; Lee, H.Y.; Kang, D.H.; Heo, H.J.; Lee, B.Y. Spirulina maxima extract ameliorates learning and memory impairments via inhibiting GSK-3β phosphorylation induced by intracerebroventricular injection of amyloid-β 1–42 in mice. Int. J. Mol. Sci. 2017, 18, 2401. [Google Scholar] [CrossRef] [PubMed]
- Buono, S.; Langellotti, A.L.; Martello, A.; Rinna, F.; Fogliano, V. Functional ingredients from microalgae. Food. Funct. 2014, 5, 1669–1685. [Google Scholar] [CrossRef]
- Choi, W.Y.; Kang, D.H.; Lee, H.Y. Effect of fermented spirulina maxima extract on cognitive-enhancing activities in mice with scopolamine-induced dementia. Evid. Based. Complement. Alternat. Med. 2018, 2018, 7218504. [Google Scholar] [CrossRef] [PubMed]
- Rimbau, V.; Camins, A.; Romay, C.; González, R.; Pallàs, M. Protective effects of C-phycocyanin against kainic acid-induced neuronal damage in rat hippocampus. Neurosci. Lett. 1999, 276, 75–78. [Google Scholar] [CrossRef]
- Koh, E.J.; Seo, Y.J.; Choi, J.; Lee, H.Y.; Kang, D.H.; Kim, K.J.; Lee, B.Y. Spirulina maxima extract prevents neurotoxicity via promoting activation of BDNF/CREB signaling pathways in neuronal cells and mice. Molecules 2017, 22, 1363. [Google Scholar] [CrossRef] [PubMed]
- Angeloni, C.; Vauzour, D. Natural products and neuroprotection. Int. J. Mol. Sci. 2019, 20, 5570. [Google Scholar] [CrossRef] [PubMed]
- Durrenberger, P.F.; Fernando, F.S.; Kashefi, S.N.; Bonnert, T.P.; Seilhean, D.; Nait-Oumesmar, B.; Schmitt, A.; Gebicke-Haerter, P.J.; Falkai, P.; Grunblatt, E.; et al. Common mechanisms in neurodegeneration and neuroinflammation: A brainnet europe gene expression microarray study. J. Neural Transm. 2015, 122, 1055–1068. [Google Scholar] [CrossRef]
- Schetters, S.T.; Gomez-Nicola, D.; Garcia-Vallejo, J.J.; Van Kooyk, Y. Neuroinflammation: Microglia and T cells get ready to tango. Front. Immunol. 2017, 8, 1905. [Google Scholar] [CrossRef]
- Shan, C.S.; Zhang, H.F.; Xu, Q.Q.; Shi, Y.H.; Wang, Y.; Li, Y.; Lin, Y.; Zheng, G.Q. Herbal medicine formulas for parkinson’s disease: A systematic review and meta-analysis of randomized double-blind placebo-controlled clinical trials. Front. Aging Neurosci. 2018, 10, 349. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.S.; Jhanji, N. Natural products as potential anti-alzheimer agents. Curr. Med. Chem. 2020, 27, 5887–5917. [Google Scholar] [CrossRef] [PubMed]
- Leonoudakis, D.; Rane, A.; Angeli, S.; Lithgow, G.J.; Andersen, J.K.; Chinta, S.J. Anti-inflammatory and neuroprotective role of natural product securinine in activated glial cells: Implications for parkinson’s disease. Mediat. Inflamm. 2017, 2017, 8302636. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Huelamo, M.; Rodriguez-Morato, J.; Boronat, A.; de la Torre, R. Modulation of NRF2 by olive oil and wine polyphenols and neuroprotection. Antioxidants 2017, 6, 73. [Google Scholar] [CrossRef] [PubMed]
- Rehman, M.U.; Wali, A.F.; Ahmad, A.; Shakeel, S.; Rasool, S.; Ali, R.; Rashid, S.M.; Madkhali, H.; Ganaie, M.A.; Khan, R. Neuroprotective strategies for neurological disorders by natural products: An update. Curr. Neuropharmacol 2019, 17, 247–267. [Google Scholar] [CrossRef] [PubMed]
- Braak, H.; Del Tredici, K. The preclinical phase of the pathological process underlying sporadic alzheimer’s disease. Brain 2015, 138, 2814–2833. [Google Scholar] [CrossRef]
- Deshpande, P.; Gogia, N.; Singh, A. Exploring the efficacy of natural products in alleviating alzheimer’s disease. Neural Regen. Res. 2019, 14, 1321–1329. [Google Scholar] [CrossRef]
- Castelli, V.; Grassi, D.; Bocale, R.; d’Angelo, M.; Antonosante, A.; Cimini, A.; Ferri, C.; Desideri, G. Diet and brain health: Which role for polyphenols? Curr. Pharm. Des. 2018, 24, 227–238. [Google Scholar] [CrossRef]
- Bagli, E.; Goussia, A.; Moschos, M.M.; Agnantis, N.; Kitsos, G. Natural compounds and neuroprotection: Mechanisms of action and novel delivery systems. In Vivo 2016, 30, 535–547. [Google Scholar]
- Lan, J.; Liu, Z.; Liao, C.; Merkler, D.J.; Han, Q.; Li, J. A study for therapeutic treatment against parkinson’s disease via Chou’s 5-steps rule. Curr. Top. Med. Chem. 2019, 19, 2318–2333. [Google Scholar] [CrossRef]
- Haddad, F.; Sawalha, M.; Khawaja, Y.; Najjar, A.; Karaman, R. Dopamine and levodopa prodrugs for the treatment of parkinson’s disease. Molecules 2018, 23, 40. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Davis, R.L.; Sue, C.M. Mitochondrial Dysfunction in Parkinson’s Disease: New Mechanistic Insights and Therapeutic Perspectives. Curr. Neurol. Neurosci. Rep. 2018, 18, 21. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.E.; Paek, S.H. Mitochondrial Dysfunction in Parkinson’s Disease. Exp. Neurobiol. 2015, 24, 103. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, S.; Cuervo, A.M. Proteostasis and aging. Nat. Med. 2015, 21, 1406–1415. [Google Scholar] [CrossRef] [PubMed]
- Wales, P.; Pinho, R.; Lázaro, D.F.; Outeiro, T.F. Limelight on alpha-synuclein: Pathological and mechanistic implications in neurodegeneration. J. Parkinsons. Dis. 2013, 3, 415–459. [Google Scholar] [CrossRef]
- Ciulla, M.; Marinelli, L.; Cacciatore, I.; Stefano, A.D. Role of Dietary Supplements in the Management of Parkinson’s Disease. Biomolecules 2019, 9, 271. [Google Scholar] [CrossRef]
- Manyam, B.V.; Dhanasekaran, M.; Hare, T.A. Neuroprotective effects of the antiparkinson drug Mucunapruriens. Phytother. Res. 2004, 18, 706–712. [Google Scholar] [CrossRef]
- Brichta, L.; Greengard, P.; Flajolet, M. Advances in the pharmacological treatment of parkinson’s disease: Targeting neurotransmitter systems. Trends Neurosci. 2013, 36, 543–554. [Google Scholar] [CrossRef]
- Manoharan, S.; Essa, M.M.; Vinoth, A.; Kowsalya, R.; Manimaran, A.; Selvasundaram, R. Alzheimer’s disease and medicinal plants: An overview. Adv. Neurobiol. 2016, 12, 95–105. [Google Scholar] [CrossRef]
- Martinez-Oliveira, P.; de Oliveira, M.F.; Alves, N.; Coelho, R.P.; Pilar, B.C.; Güllich, A.A.; Ströher, D.J.; Boligon, A.; Piccoli, J.D.; Mello-Carpes, P.B. Yacon leaf extract supplementation demonstrates neuroprotective effect against memory deficit related to β-amyloid-induced neurotoxicity. J. Funct. Foods 2018, 48, 665–675. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, Z.; Zhai, W.; Pang, J.; Mo, Y.; Yang, G.; Qu, Z.; Hu, Y. Safflower yellow attenuates learning and memory deficits in amyloid β-induced alzheimer’s disease rats by inhibiting neuroglia cell activation and inflammatory signaling pathways. Metab. Brain Dis. 2019, 34, 927–939. [Google Scholar] [CrossRef]
- Ionita, R.; Postu, P.A.; Mihasan, M.; Gorgan, D.L.; Hancianu, M.; Cioanca, O.; Hritcu, L. Ameliorative effects of Matricaria chamomilla L. hydroalcoholic extract on scopolamine-induced memory impairment in rats: A behavioral and molecular study. Phytomedicine 2018, 47, 113–120. [Google Scholar] [CrossRef]
- Hishikawa, N.; Takahashi, Y.; Amakusa, Y.; Tanno, Y.; Tuji, Y.; Niwa, H.; Murakami, N.; Krishna, U.K. Effects of turmeric on alzheimer’s disease with behavioral and psychological symptoms of dementia. Ayu 2012, 33, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Khongsombat, O.; Nakdook, W.; Ingkaninan, K. Inhibitory effects of tabernaemontana divaricata root extract on oxidative stress and neuronal loss induced by amyloid β25–35 peptide in mice. J. Tradit. Complement. Med. 2018, 8, 184–189. [Google Scholar] [CrossRef] [PubMed]
- de la Rubia Orti, J.E.; Garcia-Pardo, M.P.; Drehmer, E.; Sancho Cantus, D.; Julian Rochina, M.; Aguilar, M.A.; Hu Yang, I. Improvement of main cognitive functions in patients with alzheimer’s disease after treatment with coconut oil enriched mediterranean diet: A pilot study. J. Alzheimers Dis. 2018, 65, 577–587. [Google Scholar] [CrossRef] [PubMed]
- Ali, T.; Yoon, G.H.; Shah, S.A.; Lee, H.Y.; Kim, M.O. Osmotin attenuates amyloid β-induced memory impairment, tau phosphorylation and neurodegeneration in the mouse hippocampus. Sci. Rep. 2015, 5, 11708. [Google Scholar] [CrossRef]
- Azmi, N.H.; Ismail, M.; Ismail, N.; Imam, M.U.; Alitheen, N.B.; Abdullah, M.A. Germinated brown rice alters Aβ (1-42) aggregation and modulates alzheimer’s disease-related genes in differentiated human SH-SY5Y cells. Evid. Based Complement. Alternat. Med. 2015, 2015, 153684. [Google Scholar] [CrossRef]
- Xu, S.S.; Gao, Z.X.; Weng, Z.; Du, Z.M.; Xu, W.A.; Yang, J.S.; Zhang, M.L.; Tong, Z.H.; Fang, Y.S.; Chai, X.S.; et al. Efficacy of tablet huperzine-a on memory, cognition, and behavior in alzheimer’s disease. Zhongguo Yao Li Xue Bao 1995, 16, 391–395. [Google Scholar] [PubMed]
- Colovic, M.B.; Krstic, D.Z.; Lazarevic-Pasti, T.D.; Bondzic, A.M.; Vasic, V.M. Acetylcholinesterase inhibitors: Pharmacology and toxicology. Curr. Neuropharmacol. 2013, 11, 315–335. [Google Scholar] [CrossRef]
- Ibrahim, F.W.; Zainudin, U.N.; Latif, M.A.; Hamid, A. Neuroprotective effects of ethyl acetate extract of Zingiber zerumbet (L.) smith against oxidative stress on paraquat-induced parkinsonism in rats. Sains Malays. 2018, 47, 2337–2347. [Google Scholar] [CrossRef]
- Bisht, R.; Joshi, B.C.; Kalia, A.N.; Prakash, A. Antioxidant-rich fraction of urtica dioica mediated rescue of striatal mito-oxidative damage in MPTP-induced behavioral, cellular, and neurochemical alterations in rats. Mol. Neurobiol. 2017, 54, 5632–5645. [Google Scholar] [CrossRef]
- Chonpathompikunlert, P.; Boonruamkaew, P.; Sukketsiri, W.; Hutamekalin, P.; Sroyraya, M. The antioxidant and neurochemical activity of apium graveolens L. and its ameliorative effect on MPTP-induced parkinson-like symptoms in mice. BMC Complement. Altern. Med. 2018, 18, 103. [Google Scholar] [CrossRef] [PubMed]
- Karbarz, M.; Mytych, J.; Solek, P.; Stawarczyk, K.; Tabecka-Lonczynska, A.; Koziorowski, M.; Luczaj, L. Cereal grass juice in wound healing: Hormesis and cell-survival in normal fibroblasts, in contrast to toxic events in cancer cells. J. Physiol. Pharmacol. 2019, 70, 595–604. [Google Scholar] [CrossRef]
- Kosaraju, J.; Chinni, S.; Roy, P.D.; Kannan, E.; Antony, A.S.; Kumar, M.N. Neuroprotective effect of tinospora cordifolia ethanol extract on 6-hydroxy dopamine induced parkinsonism. Indian J. Pharmacol. 2014, 46, 176–180. [Google Scholar] [CrossRef]
- Ren, Z.X.; Zhao, Y.F.; Cao, T.; Zhen, X.C. Dihydromyricetin protects neurons in an MPTP-induced model of parkinson’s disease by suppressing glycogen synthase kinase-3 beta activity. Acta Pharmacol. Sin. 2016, 37, 1315–1324. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Wang, W.; Hao, C.; Mao, X. Agaropentaose protects SH-SY5Y cells against 6-hydroxydopamine-induced neurotoxicity through modulating NF-κB and p38MAPK signaling pathways. J. Funct. Foods 2019, 57, 222–232. [Google Scholar] [CrossRef]
- Abdel-Salam, O.M.; Sleem, A.A.; Youness, E.R.; Yassen, N.N.; Shaffie, N.; El-Toumy, S.A. Capsicum protects against rotenone-induced toxicity in mice brain via reduced oxidative stress and 5-lipoxygenase activation. J. Pharm. Pharmacol. Res. 2018, 2, 60–77. [Google Scholar] [CrossRef]
- Ojha, S.; Javed, H.; Azimullah, S.; Haque, M.E. Beta-caryophyllene, a phytocannabinoid attenuates oxidative stress, neuroinflammation, glial activation, and salvages dopaminergic neurons in a rat model of parkinson disease. Mol. Cell. Biochem. 2016, 418, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.Y.; Qin, X.Y.; Yuan, M.M.; Lu, G.J.; Cheng, Y. 2,3,5,4′-tetrahydroxystilbene-2-o-beta-d-glucoside reverses stress-induced depression via inflammatory and oxidative stress pathways. Oxid. Med. Cell. Longev. 2018, 2018, 9501427. [Google Scholar] [CrossRef]
- Roy, N.K.; Parama, D.; Banik, K.; Bordoloi, D.; Devi, A.K.; Thakur, K.K.; Padmavathi, G.; Shakibaei, M.; Fan, L.; Sethi, G.; et al. An update on pharmacological potential of boswellic acids against chronic diseases. Int. J. Mol. Sci. 2019, 20, 4101. [Google Scholar] [CrossRef]
- Bhullar, K.S.; Rupasinghe, H.P. Polyphenols: Multipotent therapeutic agents in neurodegenerative diseases. Oxid. Med. Cell. Longev. 2013, 2013, 891748. [Google Scholar] [CrossRef]
- Sarbishegi, M.; Charkhat Gorgich, E.A.; Khajavi, O.; Komeili, G.; Salimi, S. The neuroprotective effects of hydro-alcoholic extract of olive (Olea europaea L.) leaf on rotenone-induced parkinson’s disease in rat. Metab. Brain Dis. 2018, 33, 79–88. [Google Scholar] [CrossRef]
- Tomani, J.C.; Gainkam, L.O.; Nshutiyayesu, S.; Mukazayire, M.J.; Ribeiro, S.O.; Stevigny, C.; Frederich, M.; Muganga, R.; Souopgui, J. An ethnobotanical survey and inhibitory effects on NLRP3 inflammasomes/caspase-1 of herbal recipes’ extracts traditionally used in rwanda for asthma treatment. J. Ethnopharmacol. 2018, 227, 29–40. [Google Scholar] [CrossRef]
- Wang, Q.; Kuang, H.; Su, Y.; Sun, Y.; Feng, J.; Guo, R.; Chan, K. Naturally derived anti-inflammatory compounds from chinese medicinal plants. J. Ethnopharmacol. 2013, 146, 9–39. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.A.; Delattre, A.M.; Carabelli, B.; Pudell, C.; Bortolanza, M.; Staziaki, P.V.; Visentainer, J.V.; Montanher, P.F.; Del Bel, E.A.; Ferraz, A.C. Neuroprotective effect of omega-3 polyunsaturated fatty acids in the 6-OHDA model of parkinson’s disease is mediated by a reduction of inducible nitric oxide synthase. Nutr. Neurosci. 2018, 21, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Saleh, A.S.; Wang, P.; Wang, N.; Yang, L.; Xiao, Z. Brown rice versus white rice: Nutritional quality, potential health benefits, development of food products, and preservation technologies. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1070–1096. [Google Scholar] [CrossRef]
- Akhondzadeh, S.; Noroozian, M.; Mohammadi, M.; Ohadinia, S.; Jamshidi, A.H.; Khani, M. Salvia officinalis extract in the treatment of patients with mild to moderate Alzheimer’s disease: A double blind, randomized and placebo-controlled trial. J. Clin. Pharm. Ther. 2003, 28, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.S.; Thomas, R.G.; Craft, S.; Van Dyck, C.H.; Mintzer, J.; Reynolds, B.A.; Brewer, J.B.; Rissman, R.A.; Raman, R.; Aisen, P.S. A randomized, double-blind, placebo-controlled trial of resveratrol for Alzheimer disease. Neurology 2015, 85, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, D.; Falsini, B.; Piccardi, M.; Ambrosio, L.; Minnella, A.M.; Savastano, M.C.; Bisti, S.; Maccarone, R.; Fadda, A.; Mello, E.; et al. Functional effect of Saffron supplementation and risk genotypes in early age-related macular degeneration: A preliminary report. J. Transl. Med. 2013, 11, 228. [Google Scholar] [CrossRef]
- Di Marco, F.; Romeo, S.; Nandasena, C.; Purushothuman, S.; Adams, C.; Bisti, S.; Stone, J. The time course of action of two neuroprotectants, dietary saffron and photobiomodulation, assessed in the rat retina. Am. J. Neurodegene. Dis. 2013, 2, 208–220. [Google Scholar]
- Chen, M.; Du, Z.Y.; Zheng, X.; Li, D.L.; Zhou, R.P.; Zhang, K. Use of curcumin in diagnosis, prevention, and treatment of Alzheimer’s disease. Neural Regen. Res. 2018, 13, 742. [Google Scholar]
- Akhondzadeh, S.; Fallah-Pour, H.; Afkham, K.; Jamshidi, A.H.; Khalighi-Cigaroudi, F. Comparison of Crocus sativus L. and imipramine in the treatment of mild to moderate depression: A pilot double-blind randomized trial [ISRCTN45683816]. BMC Complement. Altern. Med. 2004, 4, 12. [Google Scholar] [CrossRef]
- Boskabady, M.H.; Javan, H.; Sajady, M.; Rakhshandeh, H. The possible prophylactic effect of Nigella sativa seed extract in asthmatic patients. Fund. Clin. Pharmacol. 2007, 21, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Wattanathorn, J.; Mator, L.; Muchimapura, S.; Tongun, T.; Pasuriwong, O.; Piyawatkul, N.; Yimtae, K.; Sripanidkulchai, B.; Singkhoraard, J. Positive modulation of cognition and mood in the healthy elderly volunteer following the administration of Centellaasiatica. J. Ethnopharmacol. 2008, 116, 325–332. [Google Scholar] [CrossRef]
- Nathan, P.J.; Clarke, J.; Lloyd, J.; Hutchison, C.W.; Downey, L.; Stough, C. The acute effects of an extract of Bacopa monniera (Brahmi) on cognitive function in healthy normal subjects. Hum. Psychopharmacol. Clin. Exp. 2001, 16, 345–351. [Google Scholar] [CrossRef]
- Choudhary, D.; Bhattacharyya, S.; Bose, S. Efficacy and Safety of Ashwagandha (Withaniasomnifera (L.) Dunal) Root Extract in Improving Memory and Cognitive Functions. J. Diet. Suppl. 2017, 14, 599–612. [Google Scholar] [CrossRef] [PubMed]
- Conte, R.; Calarco, A.; Napoletano, A.; Valentino, A.; Margarucci, S.; Di Cristo, F.; Di Salle, A.; Peluso, G. Polyphenols nanoencapsulation for therapeutic applications. J. Biomol. Res. Ther. 2016, 5, 1000139. [Google Scholar] [CrossRef]
- Rigacci, S.; Stefani, M. Nutraceuticals and amyloid neurodegenerative diseases: A focus on natural phenols. Expert Rev. Neurother. 2015, 15, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Simon, J.E.; Wu, Q. A critical review on grape polyphenols for neuroprotection: Strategies to enhance bioefficacy. Crit. Rev. Food Sci. Nutr. 2020, 60, 597–625. [Google Scholar] [CrossRef]
- Renaud, J.; Martinoli, M.G. Considerations for the use of polyphenols as therapies in neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 1883. [Google Scholar] [CrossRef]
- Hu, S.; Maiti, P.; Ma, Q.; Zuo, X.; Jones, M.R.; Cole, G.M.; Frautschy, S.A. Clinical development of curcumin in neurodegenerative disease. Expert Rev. Neurother. 2015, 15, 629–637. [Google Scholar] [CrossRef] [PubMed]
- Rakotoarisoa, M.; Angelova, A. Amphiphilic nanocarrier systems for curcumin delivery in neurodegenerative disorders. Medicines 2018, 5, 126. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Kim, S.; Park, K. In vitro-in vivo correlation: Perspectives on model development. Int. J. Pharm. 2011, 418, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, S. Dls and zeta potential–what they are and what they are not? J. Control. Release 2016, 235, 337–351. [Google Scholar] [CrossRef] [PubMed]
- Ovais, M.; Zia, N.; Ahmad, I.; Khalil, A.T.; Raza, A.; Ayaz, M.; Sadiq, A.; Ullah, F.; Shinwari, Z.K. Phyto-therapeutic and nanomedicinal approaches to cure alzheimer’s disease: Present status and future opportunities. Front. Aging Neurosci. 2018, 10, 284. [Google Scholar] [CrossRef]
- Niu, X.Q.; Chen, J.J.; Gao, J.Q. Nanocarriers as a powerful vehicle to overcome blood-brain barrier in treating neurodegenerative diseases: Focus on recent advances. Asian J. Pharm. Sci. 2019, 14, 480–496. [Google Scholar] [CrossRef]
- Varma, L.T.; Singh, N.; Gorain, B.; Choudhury, H.; Tambuwala, M.M.; Kesharwani, P.; Shukla, R. Recent advances in self-assembled nanoparticles for drug delivery. Curr. Drug Deliv. 2020, 17, 279–291. [Google Scholar] [CrossRef]
- Kalepu, S.; Nekkanti, V. Improved delivery of poorly soluble compounds using nanoparticle technology: A review. Drug Deliv. Transl. Res. 2016, 6, 319–332. [Google Scholar] [CrossRef]
- Smith, A.; Giunta, B.; Bickford, P.C.; Fountain, M.; Tan, J.; Shytle, R.D. Nanolipidic particles improve the bioavailability and alpha-secretase inducing ability of epigallocatechin-3-gallate (EGCG) for the treatment of alzheimer’s disease. Int. J. Pharm. 2010, 389, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Lv, L.; Yang, F.; Li, H.; Yuan, J. Brain-targeted co-delivery of β-amyloid converting enzyme 1 shRNA and epigallocatechin-3-gallate by multifunctional nanocarriers for Alzheimer’s disease treatment. IUBMB Life 2020, 72, 1819–1829. [Google Scholar] [CrossRef]
- Bhatt, R.; Singh, D.; Prakash, A.; Mishra, N. Development, characterization and nasal delivery of rosmarinic acid-loaded solid lipid nanoparticles for the effective management of huntington’s disease. Drug Deliv. 2015, 22, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Del Prado-Audelo, M.L.; Caballero-Floran, I.H.; Meza-Toledo, J.A.; Mendoza-Munoz, N.; Gonzalez-Torres, M.; Floran, B.; Cortes, H.; Leyva-Gomez, G. Formulations of curcumin nanoparticles for brain diseases. Biomolecules 2019, 9, 56. [Google Scholar] [CrossRef] [PubMed]
| Name of Plant Part | Name of Model | Neuroprotective Mechanisms | References |
|---|---|---|---|
| Yacon (Poepp. and endl.) (Smallanthus sonchifolius) extract of the leaf | Rat | Memory deficits prevented | [162] |
| Natural safflower aqueous extract | Rat | Short and long-term memory improved | [163] |
| Methanolic extract of Lactucacapensis thunb. leaves | Rat | Lowering the degree of lipid peroxidation and protein oxidation | [164] |
| Turmeric powder | Human | Improvement in the quality of life and behavioral symptoms | [165] |
| Tabernaemontana divaricata root extract | Mouse | Prevented memory loss | [166] |
| Coconut oil enriched Mediterranean diet | Human | Enhanced cognitive features | [167] |
| Osmotin, a protein derived from Nicotiana tabacum | Mouse | Increased conduct of random alternation | [168] |
| Germinated brown rice | SH-SY5Y cells | Reduced production of intracellular ROS | [169] |
| Isolated from Huperzia serrata is Huperzine A | Human | Improvement in functions of memory, cognition, and actions | [170] |
| Huperzine A isolated from Huperzia serrata | Rat | Reduce oxidative damage | [171] |
| Name of Plant Part | Name of Model | Neuroprotective Mechanisms | References |
|---|---|---|---|
| Smith ethyl acetate extract Zingiber zerumbet (L.) | Rat | Prevention of neuronal damage | [172] |
| Urticadioica Linn. ethyl acetate fraction. | Rat | Enhanced motor control and alteration in oxidative protection | [173] |
| Apium graveolens L. | Mouse | Improved behavioral disorder caused by MPTP | [174] |
| Tribulus terrestris extract | Mouse | Improved the proportion of viable neurons | [175] |
| Ethanol extract of Tinospora cordifolia | Rat | Restored locomotive operation behavioral changes caused by 6-OHDA | [176] |
| Dihydromyricetin (DHM) (Ampelopsis grossedentata) | Mouse | Mitigated the deficit in the balance of movement caused by the MPTP | [177] |
| Agaropentaose, an agaro-oligosaccharide monomer that is isolated from red algae hydrolysates of agarose | SH-SY5Y cells | Inhibited potential loss of mitochondrial membrane | [178] |
| Capsicum annuum L. extract | Mouse | Restored development of cholinesterase in the brain | [179] |
| β-Caryophyllene, a cannabinoid compound originating from a plant known as phytocannabinoids | Rat | Lipid peroxidation inhibited | [180] |
| Viride var. of Coeloglossum. Extract from bracteatum | Mouse | Prevented neuronal dopaminergic loss | [181] |
| Boswellic acids | Rat | Motor functions improved | [182] |
| Rosmarinic acid isolated from callus of Perilla frutescens | Rat | Increased tyrosine hydroxylase numbers | [183] |
| Olive leaf extracts (Olea europaea L.) | Rat | Inhibited tyrosine hydroxylase-positive neuron depletion | [184] |
| Oxalis corniculata extract | Mouse | Improved preservation and retrieval of memory | [185] |
| Curcuminoids (Curcuma longa (L.) rhizomes) | Mouse | In the striatum, decreased proinflammatory cytokine and complete nitrite production | [186] |
| Supplementation of fish oil (rich in omega-3 polyunsaturated fatty acids) | Rat | Reduced loss of substantia nigra neurons and nerve terminals in the striatum) | [187] |
| Germinated brown rice | Rat | Improved the number of dopaminergic neurons that survive | [188] |
| Plant Species | Type of Clinical Study | Clinical Outcomes | Reference |
|---|---|---|---|
| Salvia officinalis | Randomized, double-blind | Significantly improved cognitive function | [189] |
| Resveratrol | Randomized, placebo-controlled, double-blind, multicenter 52-wk phase 2 trial | Resveratrol was safe and well-tolerated. Resveratrol and its major metabolites penetrated the blood–brain barrier to have CNS effects | [190] |
| Ginkgo biloba L. | Longitudinal, 3 monthly follow-ups over a 12-month period | Focal electroretinograph↑ amplitude and sensitivity amplitude that stabilized after 3 months independent of genotype | [191] |
| Crocus sativus L. | Longitudinal, open-label study, 8 monthly follow-ups over a 29 (±5)-month period | Focal electroretinograph saffron treated age-related macular degeneration patients: Visual function remained stable | [192] |
| Curcumin longa | 24 older adults with physical or cognitive impairment | Improve physical function and cognitive function | [193] |
| Crocus sativus | Depressant patients | The effect of C. Sativus similar to imipramine in the treatment of mild to moderate depression | [194] |
| Nigella sativa | Asthmaticpatients | Improvement of all asthmatic symptoms, chest wheeze and pulmonary function test values | [195] |
| Centella asiatica | Randomized, double-blind placebo-controlled trial | Improved memory function | [196] |
| Bacopa monnieri | Double-blind, placebo-controlled trial in 38 healthy volunteers (aged 18–60 years) | Significantly improved cognitive function | [197] |
| Withania somnifera | Prospective, randomized, double-blind, placebo-controlled | Significantly improved executive functions in adults with mild cognitive impairment | [198] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahman, M.H.; Bajgai, J.; Fadriquela, A.; Sharma, S.; Trinh, T.T.; Akter, R.; Jeong, Y.J.; Goh, S.H.; Kim, C.-S.; Lee, K.-J. Therapeutic Potential of Natural Products in Treating Neurodegenerative Disorders and Their Future Prospects and Challenges. Molecules 2021, 26, 5327. https://doi.org/10.3390/molecules26175327
Rahman MH, Bajgai J, Fadriquela A, Sharma S, Trinh TT, Akter R, Jeong YJ, Goh SH, Kim C-S, Lee K-J. Therapeutic Potential of Natural Products in Treating Neurodegenerative Disorders and Their Future Prospects and Challenges. Molecules. 2021; 26(17):5327. https://doi.org/10.3390/molecules26175327
Chicago/Turabian StyleRahman, Md. Habibur, Johny Bajgai, Ailyn Fadriquela, Subham Sharma, Thuy Thi Trinh, Rokeya Akter, Yun Ju Jeong, Seong Hoon Goh, Cheol-Su Kim, and Kyu-Jae Lee. 2021. "Therapeutic Potential of Natural Products in Treating Neurodegenerative Disorders and Their Future Prospects and Challenges" Molecules 26, no. 17: 5327. https://doi.org/10.3390/molecules26175327
APA StyleRahman, M. H., Bajgai, J., Fadriquela, A., Sharma, S., Trinh, T. T., Akter, R., Jeong, Y. J., Goh, S. H., Kim, C.-S., & Lee, K.-J. (2021). Therapeutic Potential of Natural Products in Treating Neurodegenerative Disorders and Their Future Prospects and Challenges. Molecules, 26(17), 5327. https://doi.org/10.3390/molecules26175327








