Can Eucalyptol Replace Antibiotics?
Abstract
1. Introduction
2. Antimicrobial Activity
3. Mechanism of Antimicrobial Activity
4. Anti-Inflammatory Activity
5. Therapeutic Uses
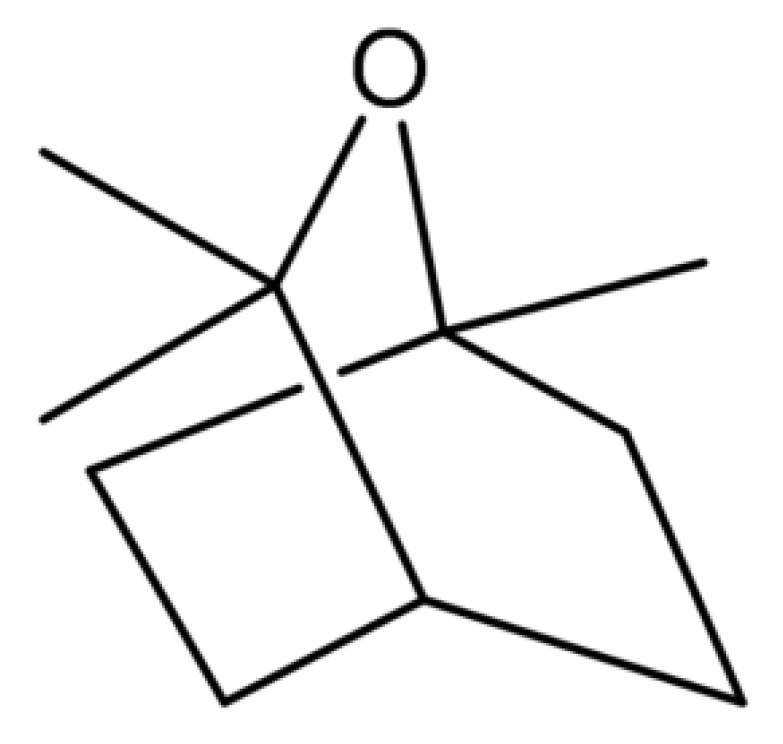
6. Eucalyptol Metabolism
7. Eucalyptol Biotransformation
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Lorenzo-Leal, A.C.; Palou, E.; López-Malo, A.; Bach, H. Antimicrobial, Cytotoxic, and Anti-Inflammatory Activities of Pimenta dioica and Rosmarinus officinalis Essential Oils. BioMed Res. Int. 2019, 2019, 1639726. [Google Scholar] [CrossRef]
- Mahmood, T.; Akhtar, N.; Khan, B.A. Herbs as alternate in treating acne. Bratisl. Med. J. 2012, 13, 25. [Google Scholar] [CrossRef]
- Wyszkowska-Kolatko, M.; Koczurkiewicz, P.; Wójcik, K.; Pękala, E. Rośliny lecznicze w terapii chorób skóry. Postępy Fitoter. 2015, 3, 184–192. [Google Scholar]
- Dhakad, A.K.; Pandey, V.V.; Beg, S.; Rawat, J.M.; Singh, A. Biological, medicinal and toxicological significance of Eucalyptus leaf essential oil: A review. J. Sci. Food Agric. 2018, 98, 833–848. [Google Scholar] [CrossRef]
- Darshan, S.; Doreswamy, R. Patented anti-inflammatory plant drug development from traditional medicine. Phytother. Res. 2004, 18, 343–357. [Google Scholar] [CrossRef]
- Góra, J.; Lis, A. Najcenniejsze Olejki Eteryczne; Part I and II; Wydawnictwo Politechniki Łódzkiej: Łódź, Polska, 2012. [Google Scholar]
- Wińska, K.; Mączka, W. Farmakologiczne właściwości 1,8-cyneolu (eukaliptolu) w chorobach układu oddechowego—Część 1. Med. Trib. 2020, 07–08, 1–4. [Google Scholar]
- Wińska, K.; Mączka, W. Farmakologiczne właściwości 1,8-cyneolu (eukaliptolu) w chorobach układu oddechowego—Część 2. Med. Trib. 2020, 09–10, 47–49. [Google Scholar]
- Aldoghaim, F.S.; Flematti, G.R.; Hammer, K.A. Antimicrobial Activity of Several Cineole-Rich Western Australian Eucalyptus Essential Oils. Microorganisms 2018, 6, 122. [Google Scholar] [CrossRef] [PubMed]
- Liangfeng, Z.; Yonghua, L.; Baoling, L.; Biyao, L.; Nianhe, X. Aromatic Plants and Essential Constituents; South China Institute of Botany, Chinese Academy of Sciences, Hai Feng Publishing Co., Chinese National Node for APINMAP: Hong Kong, China, 1993. [Google Scholar]
- Wildy, D.T.; Pate, J.S.; Bartle, J.R. Variations in composition and yield of leaf oils from alley-farmed oil mallees (Eucalyptus spp.) at a range of contrasting sites in the Western Australian wheatbelt. For. Ecol. Manag. 2000, 134, 205–217. [Google Scholar] [CrossRef]
- Daferera, D.J.; Ziogas, B.N.; Polission, M.G. GC-MS analysis of essential oils from some Greek aromatic plants and their fungitoxicity on Penicillium digitatum. J. Agric. Food Chem. 2000, 48, 2576–2581. [Google Scholar] [CrossRef] [PubMed]
- Yoro, T.; Alioune, D.; Abdoulaye, D.; Jean, C.; Saad Bouh, C.B.; Alassane, W.; Julien, P. Essential oil of Eucalyptus alba L. Growing on the Salt Zone of Fatick (Senegal) as a Source of 1,8-Cineole and Their Antibacterial Activity. J. Drug Deliv. Ther. 2020, 10, 140–143. [Google Scholar]
- Reichling, J.; Harkenthal, M.; Saller, R. In-vitro-Untersuchungen zur antimikrobiellen: Wirkung ausgewaehlter aetherischer Oele. Erfahrungsheilkunde 1999, 48, 357–366. [Google Scholar]
- Zrira, S.; Bessiere, J.M.; Menut, C.; Elamrani, A.; Benjilali, B. Chemical composition of the essential oil of nine Eucalyptus species growing in Morocco. Flavour Fragr. J. 2004, 19, 172–177. [Google Scholar] [CrossRef]
- Chisowa, E.H. Chemical composition of essential oils of three Eucalyptus species grown in Zambia. J. Essent. Oil Res. 1997, 9, 653–655. [Google Scholar] [CrossRef]
- Sefidkon, F.; Assareh, M.H.; Abravesh, Z.; Mirza, M. Chemical composition of the essential oils of five cultivated Eucalyptus species in Iran: E. intertexta, E. platypus, E. leucoxylon, E. sergentii and E. comaldulensis. J. Essent. Oil Bear. Plan. 2006, 9, 245–250. [Google Scholar] [CrossRef]
- Silva, S.M.; Abe, S.Y.; Murakami, F.S.; Frensch, G.; Marques, F.A.; Nakashima, T. Essential oils from different plant parts of Eucalyptus cinerea F. Muell. ex Benth.(Myrtaceae) as a source of 1, 8-cineole and their bioactivities. Pharmaceuticals 2011, 4, 1535–1550. [Google Scholar] [CrossRef] [PubMed]
- Babu, G.D.K.; Singh, B. Simulation of Eucalyptus cinerea oil distillation: A study on optimization of 1, 8-cineole production. Biochem. Eng. J. 2009, 44, 226–231. [Google Scholar] [CrossRef]
- Chalchat, J.C.; Muhayimana, A.; Habimana, J.B.; Chabard, J.L. Aromatic plants of Rwanda. II. Chemical composition of essential oils of ten Eucalyptus growing in Ruhande arboretum, Butare, Rwanda. J. Essent. Oil Res. 1997, 9, 159–165. [Google Scholar] [CrossRef]
- Zgheib, R.; Yassine, C.; Azzi-Achkhouty, S.; Beyrouthy, M.E. Investigation of essential oil chemical polymorphism of Salvia fruticosa naturally growing in Lebanon. J. Essent. Oil Bear. Plan. 2019, 22, 408–430. [Google Scholar] [CrossRef]
- Kökdil, G.; Kurucu, S.; Topçu, G. Chemical constituents of the essential oils of Nepeta italica L. and Nepeta sulfuriflora PH Davis. Flavour Fragr. J. 1997, 12, 33–35. [Google Scholar] [CrossRef]
- Milhau, G.; Valentin, A.; Benoit, F.; Mallié, M.; Bastide, J.M.; Pélissier, Y.; Bessière, J.M. In vitro antimalarial activity of eight essential oils. J. Essent. Oil Res. 1997, 9, 329–333. [Google Scholar] [CrossRef]
- Zollo, P.A.; Biyiti, L.; Tchoumbougnang, F.; Menut, C.; Lamaty, G.; Bouchet, P.H. Aromatic plants of tropical Central Africa. Part XXXII. Chemical composition and antifungal activity of thirteen essential oils from aromatic plants of Cameroon. Flavour Fragr. J. 1998, 13, 107–114. [Google Scholar] [CrossRef]
- Fiorini, C.; Fourast, I.; David, B.; Bessišre, J.M. Composition of the Flower, Leaf and Stem Essential Oils from Laurus nobilis L. Flavour Fragr. J. 1997, 12, 91–93. [Google Scholar] [CrossRef]
- Zrira, S.S.; Benjilali, B.B.; Fechtal, M.M.; Richard, H.H. Essential oils of twenty-seven Eucalyptus species grown in Morocco. J. Essent. Oil Res. 1992, 4, 259–264. [Google Scholar] [CrossRef]
- Baslas, R.K.; Saxena, S. Chemical Examination of Essential Oil from the Fruits of Eucalyptus globulus Labill. Herba Hung. 1984, 23, 21–23. [Google Scholar]
- Sefidkon, F.; Assareh, M.H.; Abravesh, Z.; Barazandeh, M.M. Chemical composition of the essential oils of four cultivated Eucalyptus species in Iran as medicinal plants (E. microtheca, E. spathulata, E. largiflorens and E. torquata). Iran. J. Pharm. Res. 2007, 6, 135–140. [Google Scholar]
- Benayache, S.; Benayache, F.; Benyahia, S.; Chalchat, J.C.; Garry, R.P. Leaf oils of some Eucalyptus species growing in Algeria. J. Essent. Oil Res. 2001, 13, 210–213. [Google Scholar] [CrossRef]
- Kaur, S.; Naik, S.N.; Maheshwari, B.C. Composition of the essential oil of Amomum subulatum (Roxb.) (Cardamom large) isolated by liquid CO2 extraction and steam distillation. Indian Perfum. 1993, 37, 249–252. [Google Scholar]
- Elaissi, A.; Sala, K.H.; Mabrouk, S.; Larbi, K.M.; Chemli, R.; Harzallah-Skhiri, F. Antibacterial activity and chemical composition of 20 Eucalyptus species’essential oils. Food Chem. 2011, 129, 1427–1434. [Google Scholar] [CrossRef]
- Araujo, A.R.T.S.; Périno, S.; Fernandez, X.; Cunha, C.; Rodrigues, M.; Ribeiro, M.P.; Jordao, L.; Silva, L.A.; Rodilla, J.; Coutinho, P.; et al. Solvent-Free Microwave Extraction of Thymus mastichina Essential Oil: Influence on Their Chemical Composition and on the Antioxidant and Antimicrobial Activities. Pharmaceuticals 2021, 14, 709. [Google Scholar] [CrossRef]
- Elaissi, A.; Rouis, Z.; Salem, N.A.B.; Mabrouk, S.; Salem, Y.B.; Salah, K.B.H.; Aouni, M.; Farhat, F.; Chemli, R.; Harzallah-Skhiri, F.; et al. Chemical composition of 8 eucalyptus species’ essential oils and the evaluation of their antibacterial, antifungal and antiviral activities. BMC Complement. Altern. Med. 2012, 12, 81. [Google Scholar] [CrossRef] [PubMed]
- Oyedeji, A.O.; Ekundayo, O.; Olawore, O.N.; Koenig, W.A. Essential oil composition of two varieties of Eucalyptus camaldulensis Dehn. from Nigeria. J. Essent. Oil Res. 2000, 12, 102–104. [Google Scholar] [CrossRef]
- De Medici, D.; Pieretti, S.; Salvatore, G.; Nicoletti, M.; Rasoanaivo, P. Chemical analysis of essential oils of Malagasy medicinal plants by gas chromatography and NMR spectroscopy. Flavour Fragr. J. 1992, 7, 275–281. [Google Scholar] [CrossRef]
- Zhu, L.-F.; Li, Y.-H.; Li, B.-L.; Lu, B.-Y.; Zhang, W.-L. Aromatic Plants and Essential Constituents; Supplement 1; South China Institute Botany, Chinese Academy of Science, Hai Feng Publ. Co., Peace Book Co.: Hong Kong, China, 1995; p. 160. [Google Scholar]
- Database of Compounds in Plants. Available online: https://essentialoils.org/ (accessed on 8 August 2021).
- Pino, J.A.; Marbot, R.; Quert, R.; Garcia, H. Study of Essential oils of Eucalyptus resinifera Smith, E. tereticornis Smith and Corymbia maculata (Hook.) K.D. Hill & L.A.S. Johnson, Grown in Cuba. Flavour Fragr. J. 2002, 17, 1–4. [Google Scholar]
- Mwangi, J.W.; Achola, K.J.; Sinei, K.A.; Lwande, W.; Laurent, R. Essential Oil Constituents of Artemisia afra Willd. J. Essent. Oil Res. 1995, 7, 97–99. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Ahmad, A.; Jain, N.; Aggarwal, K.K.; Syamasunder, K.V. Essential oil composition of Callistemon citrinus leaves from the lower region of Himalayas. J. Essent. Oil Res. 2001, 13, 359–361. [Google Scholar] [CrossRef]
- Boelens, M.H. Essential oils and aroma chemicals from Eucalyptus globulus Labill. Perfum. Flav. 1984, 9, 1–14. [Google Scholar]
- Taha, A.M.; Eldahshan, O.A. Chemical characteristics, antimicrobial, and cytotoxic activities of the essential oil of Egyptian Cinnamomum glanduliferum bark. Chem. Biodiver. 2017, 14, e1600443. [Google Scholar] [CrossRef]
- Dellacassa, E.; Menendez, P.; Moyna, P.; Soler, E. Chemical Composition of Eucalyptus Essential Oils Grown in Uruguay (Eucalyptus globulus). Flavour Fragr. J. 1990, 5, 91–95. [Google Scholar] [CrossRef]
- Williams, L.R.; Home, V.N.; Zhang, X.-L. The Composition and Bactericidal Activity of Oil of Melaleuca alternifolia (Tea Tree Oil) (high 1,8-cineole content). J. Aromatherap. 1988, 1, 15–17. [Google Scholar]
- Dethier, M.; Nduwimana, A.; Cordier, Y.; Menut, C.; Lamaty, G. Aromatic Plants of Tropical Central Africa. XVI Studies on Essential Oils of Five Eucalyptus Species Grown in Burundi. J. Essent. Oil Res. 1994, 6, 469–473. [Google Scholar] [CrossRef]
- Silvestre, A.J.; Cavaleiro, J.S.; Delmond, B.; Filliatre, C.; Bourgeois, G. Analysis of the variation of the essential oil composition of Eucalyptus globulus Labill. from Portugal using multivariate statistical analysis. Ind. Crops Prod. 1997, 6, 27–33. [Google Scholar] [CrossRef]
- Silvestre, A.J.; Cavaleiro, J.A.; Delmond, B.; Filliatre, C.; Bourgeois, G. The essential oil of Eucalyptus globulus Labill. from Portugal. Flavour Fragr. J. 1994, 9, 51–53. [Google Scholar] [CrossRef]
- Boelens, M.H.; Jimenez, R. The Chemical Composition of Spanish Myrtle Oils. Part II (oil of ripe fruits), J. Essent. Oil Res. 1992, 4, 349–353. [Google Scholar] [CrossRef]
- Boukhatem, M.N.; Sudha, T.; Darwish, N.H.; Chader, H.; Belkadi, A.; Rajabi, M.; Houche, A.; Benkebailli, F.; Oudjida, F.; Mousa, S.A. A new eucalyptol-rich lavender (Lavandula stoechas L.) essential oil: Emerging potential for therapy against inflammation and cancer. Molecules 2020, 25, 3671. [Google Scholar] [CrossRef]
- Gurudut, K.N.; Naik, J.P.; Srinivas, P.; Ravindranath, B. Volatile Constituents of Large Cardamom (Amomum subulatum Roxb.). Flavour Fragr. J. 1996, 11, 7–9. [Google Scholar] [CrossRef]
- Mohan Roa, L.J. Quality of essential oils and processed materials of selected spices and herbs (0.05% = trace). J. Med. Arom. Plant. Sci. 2000, 22, 808–816. [Google Scholar]
- Ahmadouch, A.; Bellakdar, J.; Berrada, M.; Denier, C.; Pinel, R. Analyse chimique des huiles essentielles de cinq especes d’Eucalyptus acclimatees au Maroc. Fitotherapia 1985, 56, 210–220. [Google Scholar]
- Bets, T.J. Solid phase microextraction of volatile constituents of fresh Eucalyptus leaves of three species. Planta Med. 2000, 66, 193–195. [Google Scholar] [CrossRef]
- Simsek, M.; Duman, R. Investigation of effect of 1,8-cineole on antimicrobial activity of chlorhexidine gluconate. Pharmacogn. Res. 2017, 9, 234–237. [Google Scholar] [CrossRef]
- Hriouech, S.; Akhmouch, A.A.; Mzabi, A.; Chefchaou, H.; Tanghort, M.; Oumokhtar, B.; Chami, N.; Remmal, A. The antistaphylococcal activity of amoxicillin/clavulanic acid, gentamicin, and 1, 8-cineole alone or in combination and their efficacy through a rabbit model of methicillin-resistant Staphylococcus aureus osteomyelitis. Evid. Based Complement. Altern. Med. 2020, 2020, 1–9. [Google Scholar] [CrossRef]
- Merghni, A.; Noumi, E.; Hadded, O.; Dridi, N.; Panwar, H.; Ceylan, O.; Mastouri, M.; Snoussi, M. Assessment of the antibiofilm and antiquorum sensing activities of Eucalyptus globulus essential oil and its main component 1,8-cineole against methicillin-resistant Staphylococcus aureus strains. Microb. Pathogen. 2018, 118, 74–80. [Google Scholar] [CrossRef]
- Yu, D.; Wang, J.; Shao, X.; Xu, F.; Wang, H. Antifungal modes of action of tea tree oil and its two characteristic components against Botrytis cinerea. J. Appl. Microbiol. 2015, 119, 1253–1262. [Google Scholar] [CrossRef] [PubMed]
- Pedrotti, C.; Marcon, Â.R.; Longaray Delamare, A.P.; Echeverrigaray, S.; da Silva Ribeiro, R.T.; Schwambach, J. Alternative control of grape rots by Essentials oils of two Eucalyptus species. J. Sci. Food Agric. 2019, 99, 6552–6561. [Google Scholar] [CrossRef]
- Kim, H.-M.; Kwon, H.; Kim, K.; Lee, S.-E. Antifungal and Antiaflatoxigenic Activities of 1,8-Cineole and t-Cinnamaldehyde on Aspergillus flavus. Appl. Sci. 2018, 8, 1655. [Google Scholar] [CrossRef]
- Packiavathy, I.A.S.V.; Agilandeswari, P.; Mustafa, K.S.; Pandian, S.K.; Ravi, A.V. Antibiofilm and quorum sensing inhibitory potential of Cuminum cyminum and its secondary metabolite methyl eugenol against gram negative bacterial pathogens. Food Res. Int. 2012, 45, 85–92. [Google Scholar] [CrossRef]
- LaSarre, B.; Federle, M.J. Exploiting quorum sensing to confuse bacterial pathogens. Microbiol. Mol. Biol. Rev. 2013, 77, 73–111. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, Z.W.; Yin, Z.Q.; Wei, Q.; Jia, R.Y.; Zhou, L.J.; Xu, J.; Song, X.; Zhou, Y.; Du, Y.H.; et al. Antibacterial activity of leaf essential oils and its constituents from Cinnamomum longepaniculatum. Int. J. Clin. Exp. Med. 2014, 7, 1721–1727. [Google Scholar]
- Nedeva, C.; Menassa, J.; Puthalakath, H. Sepsis: Inflammation is a necessary evil. Front. Cell Dev. Biol. 2019, 7, 108. [Google Scholar] [CrossRef] [PubMed]
- Santos, F.A.; Rao, V.S.N. Antiinflammatory and Antinociceptive Effects of 1,8-Cineole a Terpenoid Oxide Present In many Plant Essential Oils. Phytother. Res. 2000, 14, 240–244. [Google Scholar] [CrossRef]
- Kehrl, W.; Sonnemann, U.; Dethlefsen, U. Therapy for Acute Nonpurulent Rhinosinusitis with Cineole: Results of a Double-Blind, Randomized, Placebo-Controlled Trial. Laryngoscope 2004, 114, 738–742. [Google Scholar] [CrossRef]
- Fischer, J.; Dethlefsen, U. Efficacy of cineole in patients suffering from acute bronchitis: A placebo-controlled double-blind trial. Fischer Dethlefsen Cough 2013, 9, 25. [Google Scholar] [CrossRef]
- Juergens, U.R.; Stober, M.; Schmidt-Schilling, L.; Kleuver, T.; Vetter, H. Antiinflammatory effects of eucalyptol (1,8-cineole) in bronchial asthma: Inhibition of arachidonic acid metabolism in human blood monocytes ex vivo. Eur. J. Med. Res. 1998, 3, 407–412. [Google Scholar]
- Juergens, U.R.; Stober, M.; Vetter, H. Inhibition of cytokine production and arachidonic acid metabolism by eucalyptol (1,8-cineole) in human blood monocytes in vitro. Eur. J. Med. Res. 1998, 3, 508–510. [Google Scholar] [PubMed]
- Juergens, L.J.; Racké, K.; Tuleta, I.; Stoeber, M.; Juergens, U.R. Anti-inflammatory effects of 1,8-cineole (eucalyptol) improve glucocorticoid effects in vitro: A novel approach of steroid-sparing add-on therapy for COPD and asthma? Synergy 2017, 5, 1–8. [Google Scholar] [CrossRef]
- Juergens, L.J.; Worth, H.; Juergens, U.R. New perspectives for mucolytic, anti-inflammatory and adjunctive therapy with 1,8-cineole in COPD and asthma: Review on the new therapeutic approach. Adv. Ther. 2020, 37, 1737–1753. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Greiner, J.F.W.; Müller, J.; Zeuner, F.; Li, M.T.; Hauser, S.; Seidel, T.; Klenke, C.; Grunwald, L.M.; Schomann, T.; Widera, D.; et al. 1, 8-Cineol inhibits nuclear translocation of NF-κB p65 and NF-κB-dependent transcriptional activity. Biochim. Biophys. Acta BBA Mol. Cell Res. 2013, 1833, 2866–2878. [Google Scholar] [CrossRef] [PubMed]
- Yadav, N.; Chandra, H. Suppression of inflammatory and infection responses in lung macrophages by eucalyptus oil and its constituent 1,8-cineole: Role of pattern recognition receptors TREM-1 and NLRP3, the MAP kinase regulator MKP-1, and NFκB. PLoS ONE 2017, 12, e0188232. [Google Scholar] [CrossRef]
- Worth, H.; Schacher, C.; Dethlefsen, U. Concomitant therapy with Cineole (Eucalyptole) reduces exacerbations in COPD: A placebo-controlled double-blind trial. Respir Res. 2009, 10, 69. [Google Scholar] [CrossRef] [PubMed]
- Jager, W.; Našel, B.; Našel, C.; Binder, R.; Stimpfl, T.; Vycudilik, W.; Buchbauer, G. Pharmacokinetic studies of the fragrance compound 1,8-cyneol in humans during inhalation. Chem. Senses 1996, 21, 477–480. [Google Scholar] [CrossRef] [PubMed]
- Juergens, U.R. Anti-inflammatory properties of the monoterpene 1.8-cyneole: Current evidence for co-medication in inflammatory airway diseases. Drug Res. 2014, 64, 638–646. [Google Scholar]
- Galan, D.M.; Ezeudu, N.E.; Garcia, J.; Geronimo, C.A.; Berry, N.M.; Malcolm, B.J. Eucalyptol (1,8-cyneole): An underutilized ally in respiratory disorders? J. Essent. Oil Res. 2020, 32, 103–110. [Google Scholar] [CrossRef]
- Duisken, M.; Sandner, F.; Blfmeke, B.; Hollender, J. Metabolism of 1,8-cyneole by human cytochrome P450 enzymes: Identification of a new hydroxylated metabolite. Biochim. Biophys. Acta 2005, 1722, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Azerad, R. 1,8-Cineole: Chemical and biological oxidation reactions and products. ChemPlusChem 2014, 79, 634. [Google Scholar] [CrossRef]
- Miyazawa, M.; Shindo, M.; Shimada, T. Oxidation of 1, 8-cineole, the monoterpene cyclic ether originated from Eucalyptus polybractea, by cytochrome P450 3A enzymes in rat and human liver microsomes. Drug Metab. Disp. 2001, 29, 200–205. [Google Scholar]
- Jori, A.; Bianchetti, A.; Prestini, P.E.; Garattini, S. Effect of eucalyptol (1,8-cyneole) on the metabolism of other drugs in rats and in man. Eur. J. Pharm. 1970, 9, 362–366. [Google Scholar] [CrossRef]
- De-Oliveira, A.C.A.X.; Fidalgo-Neto, A.A.; Paumgartten, F.J.R. In vitro inhibition of liver monooxygenases by β-ionone, 1,8-cineole, (−)-menthol and terpineol. Toxicology 1999, 135, 33–41. [Google Scholar] [CrossRef]
- Hedlund, E.; Gustafsson, J.A.; Warner, M. Cytochrome P450 in the brain; a review. Curr. Drug Metab. 2001, 2, 245–263. [Google Scholar] [CrossRef]
- Wang, H.; Tompkins, L.M. CYP2B6: New insights into a historically overlooked cytochrome P450 isozyme. Curr. Drug Metab. 2008, 9, 598–610. [Google Scholar] [CrossRef]
- Unger, M.; Frank, A. Simultaneous determination of the inhibitory potency of herbal extracts on the activity of six major cytochrome P450 enzymes using liquid chromatography/mass spectrometry and automated online extraction. Rapid Commun. Mass Spectrom. RCM 2004, 18, 2273–2281. [Google Scholar] [CrossRef] [PubMed]
- Horst, K.; Rychlik, M. Quantification of 1,8-cyneole and of its metabolites in humans using stable isotope dilution assays. Mol. Nutr. Food Res. 2010, 54, 1515–1529. [Google Scholar] [CrossRef] [PubMed]
- Kirsch, F.; Buettner, A. Characterisation of the metabolites of 1, 8-cineole transferred into human milk: Concentrations and ratio of enantiomers. Metabolites 2013, 3, 47–71. [Google Scholar] [CrossRef]
- Kirsch, F.; Horst, K.; Röhrig, W.; Rychlik, M.; Buettner, A. Tracing metabolite profiles in human milk: Studies on the odorant 1, 8-cineole transferred into breast milk after oral intake. Metabolomics 2013, 9, 483–496. [Google Scholar] [CrossRef]
- MacRae, I.C.; Alberts, V.; Carman, R.M.; Shaw, I.M. Products of 1, 8-cineole oxidation by a pseudomonad. Austr. J. Chem. 1979, 32, 917–922. [Google Scholar] [CrossRef]
- Wei, G.L.; Rosazza, J.P. Stereospecific hydroxylation of 1, 8-cineole using a microbial biocatalyst. Tetrahedron Lett. 1990, 31, 2833–2836. [Google Scholar] [CrossRef]
- Williams, D.R.; Trudgill, P.W.; Taylor, D.G. Metabolism of 1, 8-cineole by a Rhodococcus species: Ring cleavage reactions. Microbiology 1989, 135, 1957–1967. [Google Scholar] [CrossRef][Green Version]
- Rasmussen, J.A.M.; Henderson, K.A.; Straffon, M.J.; Dumsday, G.J.; Coulton, J.; Zachariou, M. Two new biocatalysts for improved biological oxidation of 1, 8-cineole. Austr. J. Chem. 2006, 58, 912–916. [Google Scholar] [CrossRef]
- de Ramos, A.S.; Ribeiro, J.B.; Teixeira, B.G.; Ferreira, J.L.P.; de Silva, J.R.A.; do Ferreira, A.A.; de Souza, R.O.M.A.; Amaral, A.C.F. Hydroxylation of 1, 8-cineole by Mucor ramannianus and Aspergillus niger. Braz. J. Microbiol. 2015, 46, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Omarini, A.; Dambolena, J.S.; Lucini, E.; Mejía, S.J.; Albertó, E.; Zygadlo, J.A. Biotransformation of 1,8-cineole by solid-state fermentation of Eucalyptus waste from the essential oil industry using Pleurotus ostreatus and Favolus tenuiculus. Folia Microbiol. 2016, 61, 149–157. [Google Scholar] [CrossRef]
- Abraham, W.R. Phylogenetic influences in microbial hydroxylation of terpenoids. World J. Microbiol. Biotech. 1994, 10, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, D.B.; Adams, G.W.; Burlingame, A.L.; de Montellano, P.R.O.; De Voss, J.J. Cytochrome P450cin (CYP176A), isolation, expression, and characterization. J. Biol. Chem. 2002, 277, 27725–27732. [Google Scholar] [CrossRef]
- Balaraman, P.; Plettner, E. Chemotaxis by Pseudomonas putida (ATCC 17453) towards camphor involves cytochrome P450cam (CYP101A1). Biochim. Biophys. Acta BBA Gen. Subj. 2019, 1863, 304–312. [Google Scholar] [CrossRef] [PubMed]
- De Montellano, P.R.O. (Ed.) Cytochrome P450: Structure, Mechanism, and Biochemistry, 4th ed.; Springer: Berlin/Heidelberg, Germany, 2015. [Google Scholar]
- Stok, J.E.; Giang, P.D.; Wong, S.H.; De Voss, J.J. Exploring the substrate specificity of cytochrome P450cin. Arch. Biochem. Biophys. 2019, 672, 108060. [Google Scholar] [CrossRef] [PubMed]
- Slessor, K.E.; Hawkes, D.B.; Farlow, A.; Pearson, A.G.; Stok, J.E.; De Voss, J.J. An in vivo cytochrome P450cin (CYP176A1) catalytic system for metabolite production. J. Mol. Catal. B Enzym. 2012, 79, 15–20. [Google Scholar] [CrossRef][Green Version]
- Slessor, K.E.; Farlow, A.J.; Cavaignac, S.M.; Stok, J.E.; De Voss, J.J. Oxygen activation by P450cin: Protein and substrate mutagenesis. Arch. Biochem. Biophys. 2011, 507, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Meharenna, Y.T.; Slessor, K.E.; Cavaignac, S.M.; Poulos, T.L.; De Voss, J.J. The critical role of substrate-protein hydrogen bonding in the control of regioselective hydroxylation in P450cin. J. Biol. Chem. 2008, 283, 10804–10812. [Google Scholar] [CrossRef]
- Slessor, K.E.; Stok, J.E.; Chow, S.; De Voss, J.J. Significance of protein–substrate hydrogen bonding for the selectivity of P450-catalysed oxidations. Chem. Eur. J. 2019, 25, 4149–4155. [Google Scholar] [CrossRef]
- Lee, J.H.; Wong, S.H.; Stok, J.E.; Bagster, S.A.; Beckett, J.; Clegg, J.K.; Brock, A.J.; De Voss, J.J.; Bell, S.G. Selective hydroxylation of 1, 8-and 1, 4-cineole using bacterial P450 variants. Arch. Biochem. Biophys. 2019, 663, 54–63. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information (NCBI). National Library of Medicine (US), National Center for Biotechnology Information: Bethesda, MD, USA. 1988. Available online: https://www.ncbi.nlm.nih.gov/ (accessed on 8 August 2021).
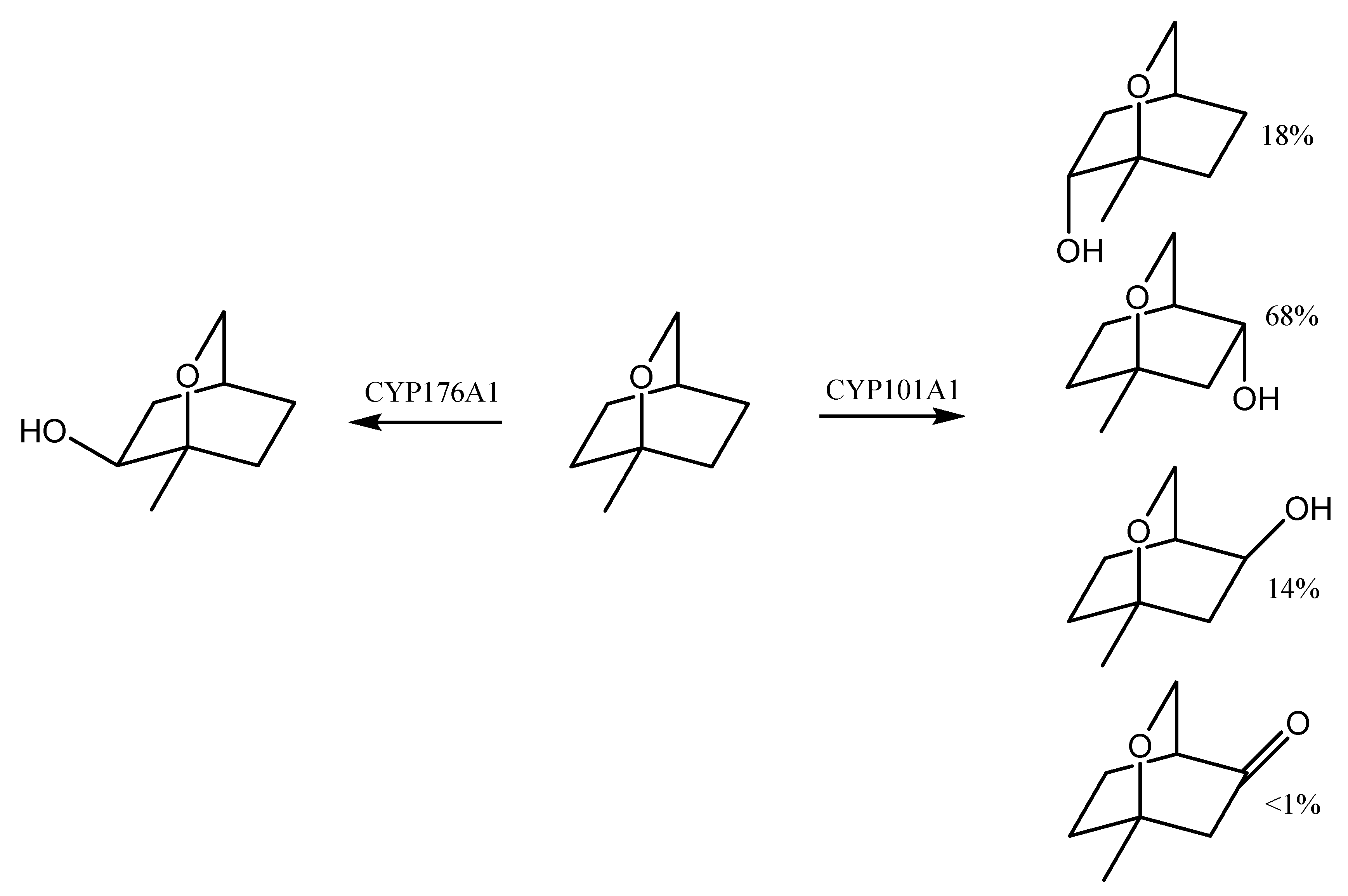

| Plant | Percentage [%] | Ref. |
|---|---|---|
| Eucalyptuskochii subsp. borealis | 97.32 | [10] |
| Eucalyptus kochii subsp. plenissima | 96.55 | [10] |
| Eucalyptus globulus Labill. | 95.13 | [11] |
| Eucalyptuskochii subsp. plenissima | 92.31 | [12] |
| Eucalyptushoristes | 90.17 | [12] |
| Rosmarinus officinalis L. | 88.9 | [13] |
| Eucalyptus alba | 88.1–76.5 | [14] |
| Eucalyptus globulus Labill. | 87.82 | [15] |
| Eucalyptus cinerea | 87.3 | [16] |
| Eucalyptus baueriana F. Muell | 87.1 | [16] |
| Eucalyptuspolybractea | 87.32 | [12] |
| Eucalyptus smithii R. Bake | 86.4 | [17] |
| Eucalyptus leucoxylon var. rostellata Miq | 85.5 | [18] |
| Eucalyptus cinere | 85.32 | [19] |
| Eucalyptus cinere | 84.4 | [20] |
| Eucalyptus smithii R. Bake | 84.27 | [21] |
| Salvia fruticosa | 83.7 | [22] |
| Eucalyptus smithii R. Baker | 83,2 | [16] |
| Eucalyptus polybractea | 82.95 | [10] |
| Eucalyptus bridgesiana R. T. Baker | 82.6 | [16] |
| Eucalyptus radiata Sieb. ex DC ssp radiata | 80.8 | [17] |
| Nepeta italica L | 80.8 | [23] |
| Eucalyptus intertexta var. intertexta R.T. Baker | 80,6 | [18] |
| Eucalyptus microtheca F. Muell. | 80.3 | [16] |
| Eucalyptus globulus Labill. | 78.9 | [24] |
| Eucalyptusloxophleba | 78.78 | [10] |
| Ocimum canum Sims. | 78.3 | [25] |
| Eucalyptus globulus | 77.02 | [10] |
| Eucalyptus foecunda Schau. | 75.5 | [16] |
| Eucalyptus sargentii subsp. Sargantii Maiden | 75.5 | [18] |
| Eucalyptus pulverulenta Sims; | 75.1 | [16] |
| Laurus nobilis L. | 73 | [26] |
| Eucalyptus globulus Labill. | 72.8 | [27] |
| Eucalyptus globulus Labill. | 72.5 | [28] |
| Eucalyptus spathulata | 72.5 | [29] |
| Eucalyptus globulus Labill. | 71.3 | [30] |
| Amomum subulatum Roxb. | 71.27 | [31] |
| Eucalyptus globulus Labill. | 71.2 | [21] |
| Eucalyptuscinerea | 70.7 | [32] |
| Thymus mastichina | 70.60–52.01 | [33] |
| Eucalyptus cinere | 70.4 | [34] |
| Eucalyptus camaldulensis Dehn., var. mysore, | 70.4 | [35] |
| Eucalyptus globulus Labill., | 70.1 | [17] |
| Melaleuca viridiflora Soland. ex Gaertn., | 70 | [36] |
| Rosmarinus officinalis L. | 69.33 | [37,38] |
| Eucalyptus sideroxylon | 69.2 | [32,34] |
| Eucalyptus resinifera Smith | 68 | [39] |
| Eucalyptus bicostata | 68.0 | [32,34] |
| Eucalyptusloxophleba | 66.93 | [10] |
| Eucalyptus propinqua Deane and Maiden | 67.5 | [16] |
| Artemisia afra Willd. | 67.37 | [40] |
| Eucalyptusloxophleba | 66.93 | [10] |
| Eucalyptus torquata | 66.9 | [29] |
| Callistemon citrinus (Curtis) Sheels (C. lanceolatus DC.) | 66.3 | [41] |
| Eucalyptus globulus Labill. | 66.1 | [42] |
| Cinnamomum glanduliferum Bark | 65.87 | [43] |
| Eucalyptus globulus Labill. | 64.5 | [44] |
| Melaleuca alternifolia (Maiden et Betche) Cheel | 64.1 | [45] |
| Eucalyptus globulus Labill. | 63.8 | [46,47] |
| Eucalyptus longifolia Link & Otto | 63.3 | [48] |
| Eucalyptus globulus Labill. | 62.5 | [49] |
| Myrtus communis L. | 61.5 | [50] |
| Nepeta sulfurifloral P.H. David | 61.5 | [23] |
| Lavandula stoechas L. | 61.36 | [50] |
| Amomum subulatum Roxb. | 61.3 | [51] |
| Cinnamomum camphora (L.) Nees et Ebermaier | 60.7 | [36] |
| Amomum subulatum Roxb. | 60 | [52] |
| Eucalyptus maidenii F. Muell. | 60 | [46] |
| Eucalyptus globulus Labill. | 60 | [53,54] |
| Essential Oil | Inhibition of Bacterial Growth (the Largest > the Smallest) | Reference |
|---|---|---|
| Eucalyptus alba(1,8-cineole: 76.5–88.1%) | S. aureus ATCC 29213 > E. coli ATCC 25922 = E. faecalis ATCC 29212 > P. aeruginosa ATCC 27853 | [14] |
| Eucalyptus cinerea(1,8-cineole: 70.7%) | S. aureus ATCC 25932 > E. coli ATCC 25922 > P. aeruginosa ATCC 227853 > E. faecalis ATCC 292112 | [32] |
| Eucalyptus sideroxylon1,8-cineole: 69.3%) | S. aureus ATCC 25932 > E. coli ATCC 25922 > P. aeruginosa ATCC 227853 > E. faecalis ATCC 292112 | [32] |
| Eucalyptus bicostata(1,8-cineole: 68%) | S. aureus ATCC 25932 > E. faecalis ATCC 292112 > E. coli ATCC 25922 > P. aeruginosa ATCC 227853 | [32] |
| Eucalyptusfasciculosa(1,8-cineole: 55%) | S. aureus ATCC 25932 > E. coli ATCC 25922 > E. faecalis ATCC 292112 | [32] |
| Eucalyptusmacarthurii (1,8-cineole: 55%) | S. aureus ATCC 25932 > E. faecalis ATCC 292112 > E. coli ATCC 25922 = P. aeruginosa ATCC 227853 | [32] |
| Eucalyptuscitriodora (1,8-cineole: 54%) | E. coli ATCC 25922 > P. aeruginosa ATCC 227853 > E. faecalis ATCC 292112 = S. aureus ATCC 25932 | [32] |
| Eucalyptus kochii subsp. borealis(1,8-cineole: 97.32%) | E. coli ATCC 25922 = S. aureus ATCC 29213 > S. aureus MRSA NCTC 10442 = E. faecalis ATCC VRE 51299 = S. Typhimurium ATCC 13311 = A. baumannii NCTC 7844 > S. epidermidis NCTC 11047 = C. albicans ATCC 90028 > E. faecalis ATCC 29212 > P. aeruginosa ATCC 27853 | [10] |
| Eucalyptus kochii subsp. plenissima plenissima(1,8-cineole: 96.55%) | S. aureus ATCC 29213 = S. Typhimurium ATCC 13311 = E. coli ATCC 25922 = A. baumannii NCTC 7844 > S. aureus MRSA NCTC 10442 > C. albicans ATCC 90028 > E. faecalis ATCC 29212 = E. faecalis VRE ATCC 51299 = S. epidermidis NCTC 11047 = P. aeruginosa ATCC 27853 | [10] |
| Eucalyptus polybractea(1,8-cineole: 82.95%) | E. faecalis VRE ATCC 51299 = S. epidermidis NCTC 11047 = A. baumannii NCTC 7844 > S. aureus MRSA NCTC 10422 = E. coli ATCC 25922 = P. aeruginosa ATCC 27853 > S. aureus ATCC 29213 = E. faecalis ATCC 29212 = C. albicans ATCC 90028 = S. Typhimurium ATCC 13311 | [10] |
| Eucalyptus loxophleba 2(1,8-cineole: 78.78%) | A. baumannii NCTC 7844 > S. Typhimurium ATCC 13311 > S. aureus ATCC 29213 = S. aureus MRSA NCTC 10442 = E. faecalis VRE ATCC 51299 > S. epidermidis NCTC 11047 = C. albicans ATCC 90028 = E. coli ATCC 25922 > E. faecalis ATCC 29212 > P. aeruginosa ATCC 27853 | [10] |
| Eucalyptus globulus(1,8-cineole: 77.02%) | S. Typhimurium ATCC 13311 > S. aureus MRSA NCTC 10442 = A. baumannii NCTC 7844 > S. aureus ATCC 29213 = E. faecalis VRE ATCC 51299 = S. epidermidis NCTC 11047 > E. coli ATCC 25922 > P. aeruginosa ATCC 27853 > E. faecalis ATCC 29212 = C. albicans ATCC 90028 | [10] |
| Eucalyptus loxophleba 1(1,8-cineole: 66.93%) | E. faecalis VRE ATCC 51299 = A. baumannii NCTC 7844 > P.aeruginosa ATCC 27853 > S. aureus MRSA NCTC 10442 = S. Typhimurium ATCC 13311 = E. coli ATCC 25922 > S. aureus ATCC 29213 = E. faecalis ATCC 29212 = S. epidermidis NCTC 11047 = C. albicans ATCC 90028 | [10] |
| Product (Polish Name) | 1,8-Cineole Content | Application |
|---|---|---|
| SOLEDUM FORTE | pure 1,8-cineole (100%) | non-suppurative sinusitis |
| ROWATINEX | 3 mg/capsule | antibacterial (stuffy nose) |
| ROWACHOL | 2 mg/capsule | gallstones, diseases of the biliary tract |
| TERPICHOL PLUS | 1.9 mg/capsule | faciliting digestion (proper production of bile) |
| SALVIASEPT | 0.6 g/100 g | inflammation of the oral cavity |
| OTRIVIN MENTHOL 0.1% | auxiliary substance | rhinitis |
| AFRIN ND MENTHOL | auxiliary substance | rhinitis |
| VICKS SINEX ALOE AND EUCALYPTUS | auxiliary substance | rhinitis |
| RINOZINE | auxiliary substance | rhinitis, sinusitis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mączka, W.; Duda-Madej, A.; Górny, A.; Grabarczyk, M.; Wińska, K. Can Eucalyptol Replace Antibiotics? Molecules 2021, 26, 4933. https://doi.org/10.3390/molecules26164933
Mączka W, Duda-Madej A, Górny A, Grabarczyk M, Wińska K. Can Eucalyptol Replace Antibiotics? Molecules. 2021; 26(16):4933. https://doi.org/10.3390/molecules26164933
Chicago/Turabian StyleMączka, Wanda, Anna Duda-Madej, Aleksandra Górny, Małgorzata Grabarczyk, and Katarzyna Wińska. 2021. "Can Eucalyptol Replace Antibiotics?" Molecules 26, no. 16: 4933. https://doi.org/10.3390/molecules26164933
APA StyleMączka, W., Duda-Madej, A., Górny, A., Grabarczyk, M., & Wińska, K. (2021). Can Eucalyptol Replace Antibiotics? Molecules, 26(16), 4933. https://doi.org/10.3390/molecules26164933







