The Therapeutic Potential of Cannabis in Counteracting Oxidative Stress and Inflammation
Abstract
1. Introduction
2. Endocannabinoid System (ECS) and Cannabinoids
3. Cannabinoids in the Inflammatory Bowel Diseases
4. Cannabinoids in Inflammatory Skin Diseases
5. Cannabinoids in the Inflammatory Respiratory System Diseases
6. Cannabinoids in the Neurological Disorders
7. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Flores-Sanchez, I.J.; Verpoorte, R. Secondary metabolism in cannabis. Phytochem. Rev. 2008, 7, 615–639. [Google Scholar] [CrossRef]
- Schofs, L.; Sparo, M.D.; Bruni, S.F.S. The antimicrobial effect behind Cannabis sativa. Pharmacol. Res. Perspect. 2021, 9, e00761. [Google Scholar] [CrossRef]
- Gieringer, D.H.; Rosenthal, E.; Carter, G.T. Marijuana Medical Handbook: Practical Guide to the Therapeutic Uses of Marijuana. 2008. Available online: https://www.worldcat.org/title/marijuana-medical-handbook-practical-guide-to-the-therapeutic-uses-of-marijuana/oclc/717176198#.YMxk0rhJ48w.mendeley (accessed on 18 June 2021).
- Blake, D.R.; Robson, P.; Ho, M.; Jubb, R.W.; McCabe, C. Preliminary assessment of the efficacy, tolerability and safety of a cannabis-based medicine (Sativex) in the treatment of pain caused by rheumatoid arthritis. Rheumatology 2005, 45, 50–52. [Google Scholar] [CrossRef] [PubMed]
- Burstein, S.H.; Zurier, R.B. Cannabinoids, Endocannabinoids, and Related Analogs in Inflammation. AAPS J. 2009, 11, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Gertsch, J.; Leonti, M.; Raduner, S.; Racz, I.; Chen, J.-Z.; Xie, X.-Q.; Altmann, K.-H.; Karsak, M.; Zimmer, A. Beta-caryophyllene is a dietary cannabinoid. Proc. Natl. Acad. Sci. USA 2008, 105, 9099–9104. [Google Scholar] [CrossRef] [PubMed]
- Sharma, C.; Al Kaabi, J.M.; Nurulain, S.M.; Goyal, S.N.; Kamal, M.A.; Ojha, S. Polypharmacological Properties and Therapeutic Potential of β-Caryophyllene: A Dietary Phytocannabinoid of Pharmaceutical Promise. Curr. Pharm. Des. 2016, 22, 3237–3264. [Google Scholar] [CrossRef] [PubMed]
- Rea, K.A.; Casaretto, J.A.; Al-Abdul-Wahid, M.S.; Sukumaran, A.; Geddes-McAlister, J.; Rothstein, S.J.; Akhtar, T.A. Biosynthesis of cannflavins A and B from Cannabis sativa L. Phytochemistry 2019, 164, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Scheau, C.; Badarau, I.A.; Mihai, L.-G.; Scheau, A.-E.; Costache, D.O.; Constantin, C.; Calina, D.; Caruntu, C.; Costache, R.S.; Caruntu, A. Cannabinoids in the Pathophysiology of Skin Inflammation. Molecules 2020, 25, 652. [Google Scholar] [CrossRef]
- Márquez, L.; Suárez, J.; Iglesias, M.; Bermudez-Silva, F.J.; De Fonseca, F.R.; Andreu, M. Ulcerative Colitis Induces Changes on the Expression of the Endocannabinoid System in the Human Colonic Tissue. PLoS ONE 2009, 4, e6893. [Google Scholar] [CrossRef]
- Lee, Y.; Jo, J.; Chung, H.Y.; Pothoulakis, C.; Im, E. Endocannabinoids in the gastrointestinal tract. Am. J. Physiol. Liver Physiol. 2016, 311, G655–G666. [Google Scholar] [CrossRef]
- Lima, M.G.; Tardelli, V.S.; Brietzke, E.; Fidalgo, T.M. Cannabis and Inflammatory Mediators. Eur. Addict. Res. 2021, 27, 16–24. [Google Scholar] [CrossRef]
- Larsen, C.; Shahinas, J. Dosage, Efficacy and Safety of Cannabidiol Administration in Adults: A Systematic Review of Human Trials. J. Clin. Med. Res. 2020, 12, 129–141. [Google Scholar] [CrossRef]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and Anti-Inflammatory Properties of Cannabidiol. Antioxidants 2019, 9, 21. [Google Scholar] [CrossRef]
- Stančić, A.; Jandl, K.; Hasenöhrl, C.; Reichmann, F.; Marsche, G.; Schuligoi, R.; Heinemann, A.; Storr, M.; Schicho, R. The GPR55 antagonist CID16020046 protects against intestinal inflammation. Neurogastroenterol. Motil. 2015, 27, 1432–1445. [Google Scholar] [CrossRef] [PubMed]
- Batalla, A.; Bos, J.; Postma, A.; Bossong, M.G. The Impact of Cannabidiol on Human Brain Function: A Systematic Review. Front. Pharmacol. 2021, 11, 618184. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Katz, S. Therapeutic Use of Cannabis in Inflammatory Bowel Disease. Gastroenterol. Hepatol. 2016, 12, 668–679. [Google Scholar]
- Di Sabatino, A.; Battista, N.; Biancheri, P.; Rapino, C.; Rovedatti, L.; Astarita, G.; Vanoli, A.; Dainese, E.; Guerci, M.; Piomelli, D.; et al. The endogenous cannabinoid system in the gut of patients with inflammatory bowel disease. Mucosal Immunol. 2011, 4, 574–583. [Google Scholar] [CrossRef]
- Katchan, V.; David, P.; Shoenfeld, Y. Cannabinoids and autoimmune diseases: A systematic review. Autoimmun. Rev. 2016, 15, 513–528. [Google Scholar] [CrossRef] [PubMed]
- Millar, S.A.; Stone, N.L.; Yates, A.S.; O’Sullivan, S.E. A Systematic Review on the Pharmacokinetics of Cannabidiol in Humans. Front. Pharmacol. 2018, 9, 1365. [Google Scholar] [CrossRef]
- Almog, S.; Aharon-Peretz, J.; Vulfsons, S.; Ogintz, M.; Abalia, H.; Lupo, T.; Hayon, Y.; Eisenberg, E. The pharmacokinetics, efficacy, and safety of a novel selective-dose cannabis inhaler in patients with chronic pain: A randomized, double-blinded, placebo-controlled trial. Eur. J. Pain 2020, 24, 1505–1516. [Google Scholar] [CrossRef]
- Swaminath, A.; Berlin, E.P.; Cheifetz, A.; Hoffenberg, E.; Kinnucan, J.; Wingate, L.; Buchanan, S.; Zmeter, N.; Rubin, D.T. The Role of Cannabis in the Management of Inflammatory Bowel Disease: A Review of Clinical, Scientific, and Regulatory Information. Inflamm. Bowel Dis. 2019, 25, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Ryberg, E.; Larsson, N.; Sjögren, S.; Hjorth, S.; Hermansson, N.-O.; Leonova, J.; Elebring, T.; Nilsson, K.; Drmota, T.; Greasley, P.J. The orphan receptor GPR55 is a novel cannabinoid receptor. Br. J. Pharmacol. 2007, 152, 1092–1101. [Google Scholar] [CrossRef] [PubMed]
- Esposito, G.; Pesce, M.; Seguella, L.; Sanseverino, W.; Lu, J.; Corpetti, C.; Sarnelli, G. The potential of cannabidiol in the COVID-19 pandemic. Br. J. Pharmacol. 2020, 177, 4967–4970. [Google Scholar] [CrossRef] [PubMed]
- Becker, W.; Alrafas, H.R.; Wilson, K.; Miranda, K.; Culpepper, C.; Chatzistamou, I.; Cai, G.; Nagarkatti, M.; Nagarkatti, P.S. Activation of Cannabinoid Receptor 2 Prevents Colitis-Associated Colon Cancer through Myeloid Cell De-activation Upstream of IL-22 Production. iScience 2020, 23, 101504. [Google Scholar] [CrossRef] [PubMed]
- Gaffal, E.; Cron, M.; Glodde, N.; Tüting, T. Anti-inflammatory activity of topical THC in DNFB-mediated mouse allergic contact dermatitis independent of CB1 and CB2 receptors. Allergy 2013, 68, 994–1000. [Google Scholar] [CrossRef]
- Granja, A.G.; Carrillo-Salinas, F.J.; Pagani, A.; Gómez-Cañas, M.; Negri, R.; Navarrete, C.M.; Mecha, M.; Mestre, L.; Fiebich, B.L.; Cantarero, I.; et al. A Cannabigerol Quinone Alleviates Neuroinflammation in a Chronic Model of Multiple Sclerosis. J. Neuroimmune Pharmacol. 2012, 7, 1002–1016. [Google Scholar] [CrossRef]
- Nam, G.; Jeong, S.K.; Park, B.M.; Lee, S.H.; Kim, H.J.; Hong, S.-P.; Kim, B.; Kim, B.-W. Selective Cannabinoid Receptor-1 Agonists Regulate Mast Cell Activation in an Oxazolone-Induced Atopic Dermatitis Model. Ann. Dermatol. 2016, 28, 22–29. [Google Scholar] [CrossRef]
- Sugawara, K.; Bíró, T.; Tsuruta, D.; Tóth, B.I.; Kromminga, A.; Zákány, N.; Zimmer, A.; Funk, W.; Gibbs, B.F.; Zimmer, A.; et al. Endocannabinoids limit excessive mast cell maturation and activation in human skin. J. Allergy Clin. Immunol. 2012, 129, 726–738. [Google Scholar] [CrossRef]
- Chiurchiù, V.; Rapino, C.; Talamonti, E.; Leuti, A.; Lanuti, M.; Gueniche, A.; Jourdain, R.; Breton, L.; Maccarrone, M. Anandamide Suppresses Proinflammatory T Cell Responses In Vitro through Type-1 Cannabinoid Receptor–Mediated mTOR Inhibition in Human Keratinocytes. J. Immunol. 2016, 197, 3545–3553. [Google Scholar] [CrossRef]
- Ihenetu, K.; Molleman, A.; Parsons, M.E.; Whelan, C.J. Inhibition of interleukin-8 release in the human colonic epithelial cell line HT-29 by cannabinoids. Eur. J. Pharmacol. 2003, 458, 207–215. [Google Scholar] [CrossRef]
- Du, Y.; Ren, P.; Wang, Q.; Jiang, S.-K.; Zhang, M.; Li, J.-Y.; Wang, L.-L.; Guan, D.-W. Cannabinoid 2 receptor attenuates inflammation during skin wound healing by inhibiting M1 macrophages rather than activating M2 macrophages. J. Inflamm. 2018, 15, 25. [Google Scholar] [CrossRef]
- Rossi, F.; Tortora, C.; Argenziano, M.; Di Paola, A.; Punzo, F. Cannabinoid Receptor Type 2: A Possible Target in SARS-CoV-2 (CoV-19) Infection? Int. J. Mol. Sci. 2020, 21, 3809. [Google Scholar] [CrossRef]
- Burstein, S.H. Ajulemic acid: Potential treatment for chronic inflammation. Pharmacol. Res. Perspect. 2018, 6, e00394. [Google Scholar] [CrossRef] [PubMed]
- Desmarais, A. Evidence supporting the benefits of marijuana for Crohn’s disease and ulcerative colitis is extremely limited: A meta-analysis of the literature. Ann. Gastroenterol. 2020, 33, 495. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Sharkey, K. Cannabinoids and the gut: New developments and emerging concepts. Pharmacol. Ther. 2010, 126, 21–38. [Google Scholar] [CrossRef]
- Wright, K.; Rooney, N.; Feeney, M.; Tate, J.; Robertson, D.; Welham, M.; Ward, S. Differential Expression of Cannabinoid Receptors in the Human Colon: Cannabinoids Promote Epithelial Wound Healing. Gastroenterology 2005, 129, 437–453. [Google Scholar] [CrossRef] [PubMed]
- Galiazzo, G.; Giancola, F.; Stanzani, A.; Fracassi, F.; Bernardini, C.; Forni, M.; Pietra, M.; Chiocchetti, R. Localization of cannabinoid receptors CB1, CB2, GPR55, and PPARα in the canine gastrointestinal tract. Histochem. Cell Biol. 2018, 150, 187–205. [Google Scholar] [CrossRef]
- Massa, F.; Marsicano, G.; Hermann, H.; Cannich, A.; Monory, K.; Cravatt, B.F.; Ferri, G.-L.; Sibaev, A.; Storr, M.; Lutz, B. The endogenous cannabinoid system protects against colonic inflammation. J. Clin. Investig. 2004, 113, 1202–1209. [Google Scholar] [CrossRef] [PubMed]
- Engel, M.A.; Kellermann, C.A.; Burnat, G.; Hahn, E.G.; Rau, T.; Konturek, P.C. Mice lacking cannabinoid CB1-, CB2-receptors or both receptors show increased susceptibility to trinitrobenzene sulfonic acid (TNBS)-induced colitis. J. Physiol. Pharmacol. 2010, 61, 89. [Google Scholar]
- Kafil, T.S.; Nguyen, T.M.; Macdonald, J.K.; Chande, N. Cannabis for the treatment of Crohn’s disease. Cochrane Database Syst. Rev. 2018, 11, CD012853. [Google Scholar] [CrossRef]
- Mulvihill, M.M.; Nomura, D.K. Therapeutic potential of monoacylglycerol lipase inhibitors. Life Sci. 2013, 92, 492–497. [Google Scholar] [CrossRef]
- Alhouayek, M.; Lambert, D.M.; Delzenne, N.M.; Cani, P.D.; Muccioli, G.G. Increasing endogenous 2-arachidonoylglycerol levels counteracts colitis and related systemic inflammation. FASEB J. 2011, 25, 2711–2721. [Google Scholar] [CrossRef] [PubMed]
- Lal, S.; Prasad, N.; Ryan, M.; Tangri, S.; Silverberg, M.S.; Gordon, A.; Steinhart, H. Cannabis use amongst patients with inflammatory bowel disease. Eur. J. Gastroenterol. Hepatol. 2011, 23, 891–896. [Google Scholar] [CrossRef]
- Storr, M.; Devlin, S.; Kaplan, G.; Panaccione, R.; Andrews, C.N. Cannabis Use Provides Symptom Relief in Patients with Inflammatory Bowel Disease but Is Associated with Worse Disease Prognosis in Patients with Crohn’s Disease. Inflamm. Bowel Dis. 2014, 20, 472–480. [Google Scholar] [CrossRef]
- Lahat, A.; Lang, A.; Ben-Horin, S. Impact of Cannabis Treatment on the Quality of Life, Weight and Clinical Disease Activity in Inflammatory Bowel Disease Patients: A Pilot Prospective Study. Digestion 2012, 85, 1–8. [Google Scholar] [CrossRef]
- Caterina, M.J. TRP Channel Cannabinoid Receptors in Skin Sensation, Homeostasis, and Inflammation. ACS Chem. Neurosci. 2014, 5, 1107–1116. [Google Scholar] [CrossRef] [PubMed]
- Wohlman, I.M.; Composto, G.M.; Heck, D.E.; Heindel, N.D.; Lacey, C.J.; Guillon, C.D.; Casillas, R.P.; Croutch, C.R.; Gerecke, N.R.; Laskin, D.L.; et al. Mustard vesicants alter expression of the endocannabinoid system in mouse skin. Toxicol. Appl. Pharmacol. 2016, 303, 30–44. [Google Scholar] [CrossRef]
- Tóth, K.F.; Ádám, D.; Bíró, T.; Oláh, A. Cannabinoid Signaling in the Skin: Therapeutic Potential of the “C(ut)annabinoid” System. Molecules 2019, 24, 918. [Google Scholar] [CrossRef] [PubMed]
- Proksch, E.; Soeberdt, M.; Neumann, C.; Kilic, A.; Abels, C. Modulators of the endocannabinoid system influence skin barrier repair, epidermal proliferation, differentiation and inflammation in a mouse model. Exp. Dermatol. 2019, 28, 1058–1065. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.M.; Porreca, F.; Lai, J.; Albrecht, P.J.; Rice, F.L.; Khodorova, A.; Davar, G.; Makriyannis, A.; Vanderah, T.W.; Mata, H.P.; et al. CB2 cannabinoid receptor activation produces antinociception by stimulating peripheral release of endogenous opioids. Proc. Natl. Acad. Sci. USA 2005, 102, 3093–3098. [Google Scholar] [CrossRef] [PubMed]
- Cintosun, A.; Lara-Corrales, I.; Pope, E. Mechanisms of Cannabinoids and Potential Applicability to Skin Diseases. Clin. Drug Investig. 2020, 40, 293–304. [Google Scholar] [CrossRef]
- Oláh, A.; Szekanecz, Z.; Bíró, T. Targeting Cannabinoid Signaling in the Immune System: “High”-ly Exciting Questions, Possibilities, and Challenges. Front. Immunol. 2017, 8, 1487. [Google Scholar] [CrossRef]
- Jeong, S.; Kim, M.S.; Lee, S.H.; Park, B.D. Epidermal Endocannabinoid System (EES) and its Cosmetic Application. Cosmetics 2019, 6, 33. [Google Scholar] [CrossRef]
- Norooznezhad, A.H.; Norooznezhad, F. Cannabinoids: Possible agents for treatment of psoriasis via suppression of angiogenesis and inflammation. Med. Hypotheses 2017, 99, 15–18. [Google Scholar] [CrossRef]
- Avila, C.; Massick, S.; Kaffenberger, B.H.; Kwatra, S.G.; Bechtel, M. Cannabinoids for the treatment of chronic pruritus: A review. J. Am. Acad. Dermatol. 2020, 82, 1205–1212. [Google Scholar] [CrossRef] [PubMed]
- Oláh, A.; Tóth, B.I.; Borbíró, I.; Sugawara, K.; Szöllõsi, A.G.; Czifra, G.; Pál, B.; Ambrus, L.; Kloepper, J.; Camera, E.; et al. Cannabidiol exerts sebostatic and antiinflammatory effects on human sebocytes. J. Clin. Investig. 2014, 124, 3713–3724. [Google Scholar] [CrossRef]
- Spleman, L.; Sinclair, R.; Freeman, M.; Davis, M.; Gebauer, K. 1061 The safety of topical cannabidiol (CBD) for the treatment of acne. J. Investig. Dermatol. 2018, 138, S180. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, B.; Park, B.M.; Jeon, J.E.; Lee, S.H.; Mann, S.; Ahn, S.K.; Hong, S.-P.; Jeong, S.K. Topical cannabinoid receptor 1 agonist attenuates the cutaneous inflammatory responses in oxazolone-induced atopic dermatitis model. Int. J. Dermatol. 2015, 54, e401–e408. [Google Scholar] [CrossRef]
- Wei, J.; Bhattacharyya, S.; Varga, J. Peroxisome proliferator-activated receptor γ: Innate protection from excessive fibrogenesis and potential therapeutic target in systemic sclerosis. Curr. Opin. Rheumatol. 2010, 22, 671–676. [Google Scholar] [CrossRef]
- Bort, A.; Alvarado-Vazquez, P.A.; Moracho-Vilrriales, C.; Virga, K.G.; Gumina, G.; Romero-Sandoval, A.; Asbill, S. Effects of JWH015 in cytokine secretion in primary human keratinocytes and fibroblasts and its suitability for topical/transdermal delivery. Mol. Pain 2017, 13, 1744806916688220. [Google Scholar] [CrossRef] [PubMed]
- Koyama, S.; Purk, A.; Kaur, M.; Soini, H.A.; Novotny, M.V.; Davis, K.; Kao, C.C.; Matsunami, H.; Mescher, A. Beta-caryophyllene enhances wound healing through multiple routes. PLoS ONE 2019, 14, e0216104. [Google Scholar] [CrossRef]
- Karsak, M.; Gaffal, E.; Date, R.; Wang-Eckhardt, L.; Rehnelt, J.; Petrosino, S.; Starowicz, K.; Steuder, R.; Schlicker, E.; Cravatt, B.; et al. Attenuation of Allergic Contact Dermatitis Through the Endocannabinoid System. Science 2007, 316, 1494–1497. [Google Scholar] [CrossRef] [PubMed]
- Haruna, T.; Soga, M.; Morioka, Y.; Imura, K.; Furue, Y.; Yamamoto, M.; Hayakawa, J.; Deguchi, M.; Arimura, A.; Yasui, K. The Inhibitory Effect of S-777469, a Cannabinoid Type 2 Receptor Agonist, on Skin Inflammation in Mice. Pharmacology 2017, 99, 259–267. [Google Scholar] [CrossRef]
- Marsella, R.; Ahrens, K.; Sanford, R.; Trujillo, A.; Massre, D.; Soeberdt, M.; Abels, C. Double blinded, vehicle controlled, crossover study on the efficacy of a topical endocannabinoid membrane transporter inhibitor in atopic Beagles. Arch. Dermatol. Res. 2019, 311, 795–800. [Google Scholar] [CrossRef]
- Tóth, B.I.; Dobrosi, N.; Dajnoki, A.; Czifra, G.; Oláh, A.; Szöllősi, A.G.; Juhász, I.; Sugawara, K.; Paus, R.; Bíró, T. Endocannabinoids Modulate Human Epidermal Keratinocyte Proliferation and Survival via the Sequential Engagement of Cannabinoid Receptor-1 and Transient Receptor Potential Vanilloid-1. J. Investig. Dermatol. 2011, 131, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Bujak, J.; Kosmala, D.; Szopa, I.M.; Majchrzak, K.; Bednarczyk, P. Inflammation, Cancer and Immunity—Implication of TRPV1 Channel. Front. Oncol. 2019, 9, 1087. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Yang, P.; Mack, M.R.; Dryn, D.; Luo, J.; Gong, X.; Liu, S.; Oetjen, L.K.; Zholos, A.V.; Mei, Z.; et al. Sensory TRP channels contribute differentially to skin inflammation and persistent itch. Nat. Commun. 2017, 8, 980. [Google Scholar] [CrossRef] [PubMed]
- Petrosino, S.; Cristino, L.; Karsak, M.; Gaffal, E.; Ueda, N.; Tüting, T.; Bisogno, T.; De Filippis, D.; D’Amico, A.; Saturnino, C.; et al. Protective role of palmitoylethanolamide in contact allergic dermatitis. Allergy 2009, 65, 698–711. [Google Scholar] [CrossRef]
- Malamas, M.S.; Farah, S.I.; Lamani, M.; Pelekoudas, D.N.; Perry, N.T.; Rajarshi, G.; Miyabe, C.Y.; Chandrashekhar, H.; West, J.; Pavlopoulos, S.; et al. Design and synthesis of cyanamides as potent and selective N-acylethanolamine acid amidase inhibitors. Bioorg. Med. Chem. 2020, 28, 115195. [Google Scholar] [CrossRef]
- Dubrac, S.; Schmuth, M. PPAR-alpha in cutaneous inflammation. Derm. Endocrinol. 2011, 3, 23–26. [Google Scholar] [CrossRef]
- Vaia, M.; Petrosino, S.; De Filippis, D.; Negro, L.; Guarino, A.; Carnuccio, R.; Di Marzo, V.; Iuvone, T. Palmitoylethanolamide reduces inflammation and itch in a mouse model of contact allergic dermatitis. Eur. J. Pharmacol. 2016, 791, 669–674. [Google Scholar] [CrossRef]
- Nakajima, J.; Nakae, D.; Yasukawa, K. Structure-dependent inhibitory effects of synthetic cannabinoids against 12-O-tetradecanoylphorbol-13-acetate-induced inflammation and skin tumour promotion in mice. J. Pharm. Pharmacol. 2013, 65, 1223–1230. [Google Scholar] [CrossRef] [PubMed]
- Glodde, N.; Jakobs, M.; Bald, T.; Tüting, T.; Gaffal, E. Differential role of cannabinoids in the pathogenesis of skin cancer. Life Sci. 2015, 138, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Bode, A.M.; Zhao, Q.; Cho, Y.-Y.; Zhu, F.; Ma, W.-Y.; Dong, Z. The Cannabinoid Receptors Are Required for Ultraviolet-Induced Inflammation and Skin Cancer Development. Cancer Res. 2008, 68, 3992–3998. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, T.E. Endocannabinoid System in the Airways. Molecules 2019, 24, 4626. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, T.E.; Larsson, O.; Adner, M. Stimulation of cannabinoid CB1 receptors prevents nerve-mediated airway hyperreactivity in NGF-induced inflammation in mouse airways. Eur. J. Pharmacol. 2016, 776, 132–138. [Google Scholar] [CrossRef]
- Bozkurt, T.E.; Kaya, Y.; Durlu-Kandilci, N.T.; Onder, S.; Sahin-Erdemli, I. The effect of cannabinoids on dinitrofluorobenzene-induced experimental asthma in mice. Respir. Physiol. Neurobiol. 2016, 231, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Pini, A.; Mannaioni, G.; Pellegrini-Giampietro, D.; Passani, M.B.; Mastroianni, R.; Bani, D.; Masini, E. The role of cannabinoids in inflammatory modulation of allergic respiratory disorders, inflammatory pain and ischemic stroke. Curr. Drug Targets 2012, 13, 984–993. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.-Y.; D’Agostino, B.; Risse, P.-A.; Marrocco, G.; Naline, E.; Zhang, Y.; Chen, H.-Z.; Finance, O.; Rinaldi-Carmona, M.; Rossi, F.; et al. Cannabinoid CB2 receptor activation prevents bronchoconstriction and airway oedema in a model of gastro-oesophageal reflux. Eur. J. Pharmacol. 2007, 573, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.J.; Birrell, M.A.; Crispino, N.; Hele, D.J.; Venkatesan, P.; Barnes, P.J.; Yacoub, M.H.; Belvisi, M.G. Inhibition of guinea-pig and human sensory nerve activity and the cough reflex in guinea-pigs by cannabinoid (CB2) receptor activation. Br. J. Pharmacol. 2003, 140, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Tashkin, D.P.; Roth, M.D. Pulmonary effects of inhaled cannabis smoke. Am. J. Drug Alcohol Abus. 2019, 45, 596–609. [Google Scholar] [CrossRef]
- Giannini, L.; Nistri, S.; Mastroianni, R.; Cinci, L.; Vannacci, A.; Mariottini, C.; Passani, M.B.; Mannaioni, P.F.; Bani, D.; Masini, E. Activation of cannabinoid receptors prevents antigen-induced asthma-like reaction in guinea pigs. J. Cell. Mol. Med. 2008, 12, 2381–2394. [Google Scholar] [CrossRef]
- Costola-De-Souza, C.; Ribeiro, A.; Ferraz-De-Paula, V.; Calefi, A.S.; Aloia, T.P.A.; Junior, J.A.G.; De Almeida, V.I.; Pinheiro, M.L.; Palermo-Neto, J. Monoacylglycerol Lipase (MAGL) Inhibition Attenuates Acute Lung Injury in Mice. PLoS ONE 2013, 8, e77706. [Google Scholar] [CrossRef]
- Novgorodtseva, T.P.; Gvozdenko, T.A.; Vitkina, T.I.; Denisenko, Y.K.; Antonyuk, M.V.; Knyshova, V.V. Regulatory signal mechanisms of systemic inflammation in respiratory pathology. Russ. Open Med. J. 2019, 8, e0106. [Google Scholar] [CrossRef]
- Tahamtan, A.; Samieipoor, Y.; Nayeri, F.S.; Rahbarimanesh, A.A.; Izadi, A.; Rashidi-Nezhad, A.; Tavakoli-Yaraki, M.; Farahmand, M.; Bont, L.; Shokri, F.; et al. Effects of cannabinoid receptor type 2 in respiratory syncytial virus infection in human subjects and mice. Virulence 2018, 9, 217–230. [Google Scholar] [CrossRef]
- Tahamtan, A.; Tavakoli-Yaraki, M.; Shadab, A.; Rezaei, F.; Marashi, S.M.; Shokri, F.; Mokhatri-Azad, T.; Salimi, V. The Role of Cannabinoid Receptor 1 in the Immunopathology of Respiratory Syncytial Virus. Viral Immunol. 2018, 31, 292–298. [Google Scholar] [CrossRef]
- Ferrini, M.E.; Hong, S.; Stierle, A.; Stella, N.; Roberts, K.; Jaffar, Z. CB2 receptors regulate natural killer cells that limit allergic airway inflammation in a murine model of asthma. Allergy 2017, 72, 937–947. [Google Scholar] [CrossRef] [PubMed]
- Frei, R.B.; Luschnig, P.; Parzmair, G.P.; Peinhaupt, M.; Schranz, S.; Fauland, A.; Wheelock, C.E.; Heinemann, A.; Sturm, E.M. Cannabinoid receptor 2 augments eosinophil responsiveness and aggravates allergen-induced pulmonary inflammation in mice. Allergy 2016, 71, 944–956. [Google Scholar] [CrossRef]
- Vuolo, F.; Abreu, S.C.; Michels, M.; Xisto, D.G.; Blanco, N.G.; Hallak, J.E.; Zuardi, A.W.; Crippa, J.A.; Reis, C.; Bahl, M.; et al. Cannabidiol reduces airway inflammation and fibrosis in experimental allergic asthma. Eur. J. Pharmacol. 2019, 843, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.; Almeida, V.I.; Costola-De-Souza, C.; Ferraz-De-Paula, V.; Pinheiro, M.L.; Vitoretti, L.B.; Junior, J.A.G.; Akamine, A.T.; Crippa, J.A.; Tavares-De-Lima, W.; et al. Cannabidiol improves lung function and inflammation in mice submitted to LPS-induced acute lung injury. Immunopharmacol. Immunotoxicol. 2014, 37, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Khodadadi, H.; Salles, É.L.; Jarrahi, A.; Chibane, F.; Costigliola, V.; Yu, J.C.; Vaibhav, K.; Hess, D.C.; Dhandapani, K.M.; Baban, B. Cannabidiol Modulates Cytokine Storm in Acute Respiratory Distress Syndrome Induced by Simulated Viral Infection Using Synthetic RNA. Cannabis Cannabinoid Res. 2020, 5, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Salles, É.L.; Khodadadi, H.; Jarrahi, A.; Ahluwalia, M.; Paffaro, V.A.; Costigliola, V.; Yu, J.C.; Hess, D.C.; Dhandapani, K.M.; Baban, B. Cannabidiol (CBD) modulation of apelin in acute respiratory distress syndrome. J. Cell. Mol. Med. 2020, 24, 12869–12872. [Google Scholar]
- Braun, A.; Engel, T.; Aguilar-Pimentel, J.A.; Zimmer, A.; Jakob, T.; Behrendt, H.; Mempel, M. Beneficial effects of cannabinoids (CB) in a murine model of allergen-induced airway inflammation: Role of CB1/CB2 receptors. Immunobiology 2011, 216, 466–476. [Google Scholar] [CrossRef]
- Rao, R.; Nagarkatti, P.S.; Nagarkatti, M. Δ9Tetrahydrocannabinol attenuates Staphylococcal enterotoxin B-induced inflammatory lung injury and prevents mortality in mice by modulation of miR-17-92 cluster and induction of T-regulatory cells. Br. J. Pharmacol. 2015, 172, 1792–1806. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.; Alghetaa, H.F.K.F.K.; Miranda, K.; Wilson, K.; Singh, N.P.P.; Cai, G.; Putluri, N.; Nagarkatti, P.; Nagarkatti, M. Δ9-Tetrahydrocannabinol Prevents Mortality from Acute Respiratory Distress Syndrome through the Induction of Apoptosis in Immune Cells, Leading to Cytokine Storm Suppression. Int. J. Mol. Sci. 2020, 21, 6244. [Google Scholar] [CrossRef]
- Mohammed, A.; Alghetaa, H.K.; Zhou, J.; Chatterjee, S.; Nagarkatti, P.; Nagarkatti, M. Protective effects of Δ 9 -tetrahydrocannabinol against enterotoxin-induced acute respiratory distress syndrome are mediated by modulation of microbiota. Br. J. Pharmacol. 2020, 177, 5078–5095. [Google Scholar] [CrossRef] [PubMed]
- Nagarkatti, P.; Miranda, K.; Nagarkatti, M. Use of Cannabinoids to Treat Acute Respiratory Distress Syndrome and Cytokine Storm Associated with Coronavirus Disease-2019. Front. Pharmacol. 2020, 11, 589438. [Google Scholar] [CrossRef] [PubMed]
- Zawatsky, C.; Abdalla, J.; Cinar, R. Synthetic cannabinoids induce acute lung inflammation via cannabinoid receptor 1 activation. ERJ Open Res. 2020, 6, 00121-2020. [Google Scholar] [CrossRef]
- Cinar, R.; Gochuico, B.R.; Iyer, M.R.; Jourdan, T.; Yokoyama, T.; Park, J.K.; Coffey, N.; Pri-Chen, H.; Szanda, G.; Liu, Z.; et al. Cannabinoid CB1 receptor overactivity contributes to the pathogenesis of idiopathic pulmonary fibrosis. JCI Insight 2017, 2, e92281. [Google Scholar] [CrossRef]
- Zou, S.; Kumar, U. Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. Int. J. Mol. Sci. 2018, 19, 833. [Google Scholar] [CrossRef] [PubMed]
- Polini, B.; Cervetto, C.; Carpi, S.; Pelassa, S.; Gado, F.; Ferrisi, R.; Bertini, S.; Nieri, P.; Marcoli, M.; Manera, C. Positive Allosteric Modulation of CB1 and CB2 Cannabinoid Receptors Enhances the Neuroprotective Activity of a Dual CB1R/CB2R Orthosteric Agonist. Life 2020, 10, 333. [Google Scholar] [CrossRef] [PubMed]
- Murikinati, S.; Jüttler, E.; Keinert, T.; Ridder, D.A.; Muhammad, S.; Waibler, Z.; Ledent, C.; Zimmer, A.; Kalinke, U.; Schwaninger, M. Activation of cannabinoid 2 receptors protects against cerebral ischemia by inhibiting neutrophil recruitment. FASEB J. 2010, 24, 788–798. [Google Scholar] [CrossRef] [PubMed]
- Cassano, T.; Calcagnini, S.; Pace, L.; De Marco, F.; Romano, A.; Gaetani, S. Cannabinoid Receptor 2 Signaling in Neurodegenerative Disorders: From Pathogenesis to a Promising Therapeutic Target. Front. Neurosci. 2017, 11, 30. [Google Scholar] [CrossRef]
- Musella, A.; Sepman, H.; Mandolesi, G.; Gentile, A.; Fresegna, D.; Haji, N.; Conrad, A.; Lutz, B.; Maccarrone, M.; Centonze, D. Pre- and postsynaptic type-1 cannabinoid receptors control the alterations of glutamate transmission in experimental autoimmune encephalomyelitis. Neuropharmacology 2014, 79, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Turner, H.; Chueh, D.; Ortiz, T.; Stokes, A.J.; Small-Howard, A.L. Cannabinoid Therapeutics in Parkinson’s Disease: Promise and Paradox. J. Herbs Spices Med. Plants 2017, 23, 231–248. [Google Scholar] [CrossRef][Green Version]
- Kendall, D.A.; Yudowski, G.A. Cannabinoid Receptors in the Central Nervous System: Their Signaling and Roles in Disease. Front. Cell. Neurosci. 2017, 10, 294. [Google Scholar] [CrossRef]
- Carrera, I. Current Drugs and Potential Future Neuroprotective Compounds for Parkinson’s Disease. Curr. Neuropharmacol. 2019, 17, 295–306. [Google Scholar] [CrossRef]
- Walter, L.; Stella, N. Cannabinoids and neuroinflammation. Br. J. Pharmacol. 2004, 141, 775–785. [Google Scholar] [CrossRef]
- Chagas, M.H.N.; Zuardi, A.W.; Tumas, V.; Pena-Pereira, M.A.; Sobreira, E.T.; Bergamaschi, M.M.; Dos Santos, A.C.; Teixeira, A.L.; Hallak, J.E.C.; Crippa, J.A.S. Effects of cannabidiol in the treatment of patients with Parkinson’s disease: An exploratory double-blind trial. J. Psychopharmacol. 2014, 28, 1088–1098. [Google Scholar] [CrossRef]
- Muller-Vahl, K.R.; Schneider, U.; Prevedel, H.; Theloe, K.; Kolbe, H.; Daldrup, T.; Emrich, H.M. Delta 9-Tetrahydrocannabinol (THC) is Effective in the Treatment of Tics in Tourette Syndrome. J. Clin. Psychiatry 2003, 64, 459–465. [Google Scholar] [CrossRef]
- Blázquez, C.; Chiarlone, A.; Sagredo, O.; Aguado, T.; Pazos, M.R.; Resel, E.; Palazuelos, J.; Julien, B.; Salazar, M.; Börner, C.; et al. Loss of striatal type 1 cannabinoid receptors is a key pathogenic factor in Huntington’s disease. Brain 2010, 134, 119–136. [Google Scholar] [CrossRef]
- Whiting, P.; Wolff, R.F.; Deshpande, S.; Di Nisio, M.; Duffy, S.; Hernandez, A.V.; Keurentjes, J.C.; Lang, S.; Misso, K.; Ryder, S.; et al. Cannabinoids for Medical Use. JAMA 2015, 313, 2456–2473. [Google Scholar] [CrossRef]
- Garcia-Arencibia, M.; González, S.; DE Lago, E.; Ramos, J.A.; Mechoulam, R.; Fernández-Ruiz, J. Evaluation of the neuroprotective effect of cannabinoids in a rat model of Parkinson’s disease: Importance of antioxidant and cannabinoid receptor-independent properties. Brain Res. 2007, 1134, 162–170. [Google Scholar] [CrossRef]
- Stella, N. Endocannabinoid signaling in microglial cells. Neuropharmacology 2009, 56, 244–253. [Google Scholar] [CrossRef]
- Sagredo, O.; Pazos, M.R.; Valdeolivas, S.; Fernandez-Ruiz, J. Cannabinoids: Novel Medicines for the Treatment of Huntingtons Disease. Recent Pat. CNS Drug Discov. 2012, 7, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Glass, M.; Dragunow, M.; Faull, R. The pattern of neurodegeneration in Huntington’s disease: A comparative study of cannabinoid, dopamine, adenosine and GABAA receptor alterations in the human basal ganglia in Huntington’s disease. Neuroscience 2000, 97, 505–519. [Google Scholar] [CrossRef]
- Palazuelos, J.; Aguado, T.; Pazos, M.R.; Julien, B.; Carrasco, C.; Resel, E.; Sagredo, O.; Benito, C.; Romero, J.; Azcoitia, I.; et al. Microglial CB2 cannabinoid receptors are neuroprotective in Huntington’s disease excitotoxicity. Brain 2009, 132, 3152–3164. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Salinas, F.J.; Navarrete, C.M.; Mecha, M.; Feliu, A.; Collado, J.A.; Cantarero, I.; Bellido, M.L.; Muñoz, E.; Guaza, C. A Cannabigerol Derivative Suppresses Immune Responses and Protects Mice from Experimental Autoimmune Encephalomyelitis. PLoS ONE 2014, 9, e94733. [Google Scholar] [CrossRef]
- Díaz-Alonso, J.; Paraíso-Luna, J.; Navarrete, C.; Del Río, C.; Cantarero, I.; Palomares, B.; Aguareles, J.; Fernández-Ruiz, J.; Bellido, M.L.; Pollastro, F.; et al. VCE-003.2, a novel cannabigerol derivative, enhances neuronal progenitor cell survival and alleviates symptomatology in murine models of Huntington’s disease. Sci. Rep. 2016, 6, 29789. [Google Scholar] [CrossRef]
- Mammana, S.; Cavalli, E.; Gugliandolo, A.; Silvestro, S.; Pollastro, F.; Bramanti, P.; Mazzon, E. Could the Combination of Two Non-Psychotropic Cannabinoids Counteract Neuroinflammation? Effectiveness of Cannabidiol Associated with Cannabigerol. Medicina 2019, 55, 747. [Google Scholar] [CrossRef] [PubMed]
- Nachnani, R.; Raup-Konsavage, W.M.; Vrana, K.E. The Pharmacological Case for Cannabigerol. J. Pharmacol. Exp. Ther. 2021, 376, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Lah, T.T.; Novak, M.; Pena Almidon, M.A.; Marinelli, O.; Žvar Baškovič, B.; Majc, B.; Mlinar, M.; Bošnjak, R.; Breznik, B.; Zomer, R.; et al. Cannabigerol Is a Potential Therapeutic Agent in a Novel Combined Therapy for Glioblastoma. Cells 2021, 10, 340. [Google Scholar] [CrossRef] [PubMed]
- Bifulco, M.; Malfitano, A.M.; Proto, M.C. Cannabinoids in the management of spasticity associated with multiple sclerosis. Neuropsychiatr. Dis. Treat. 2008, 4, 847–853. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pryce, G.; Ahmed, Z.; Hankey, D.J.R.; Jackson, S.J.; Croxford, J.L.; Pocock, J.M.; Ledent, C.; Petzold, A.; Thompson, A.; Giovannoni, G.; et al. Cannabinoids inhibit neurodegeneration in models of multiple sclerosis. Brain 2003, 126, 2191–2202. [Google Scholar] [CrossRef] [PubMed]
- Furgiuele, A.; Cosentino, M.; Ferrari, M.; Marino, F. Immunomodulatory Potential of Cannabidiol in Multiple Sclerosis: A Systematic Review. J. Neuroimmune Pharmacol. 2021, 16, 251–269. [Google Scholar] [CrossRef]
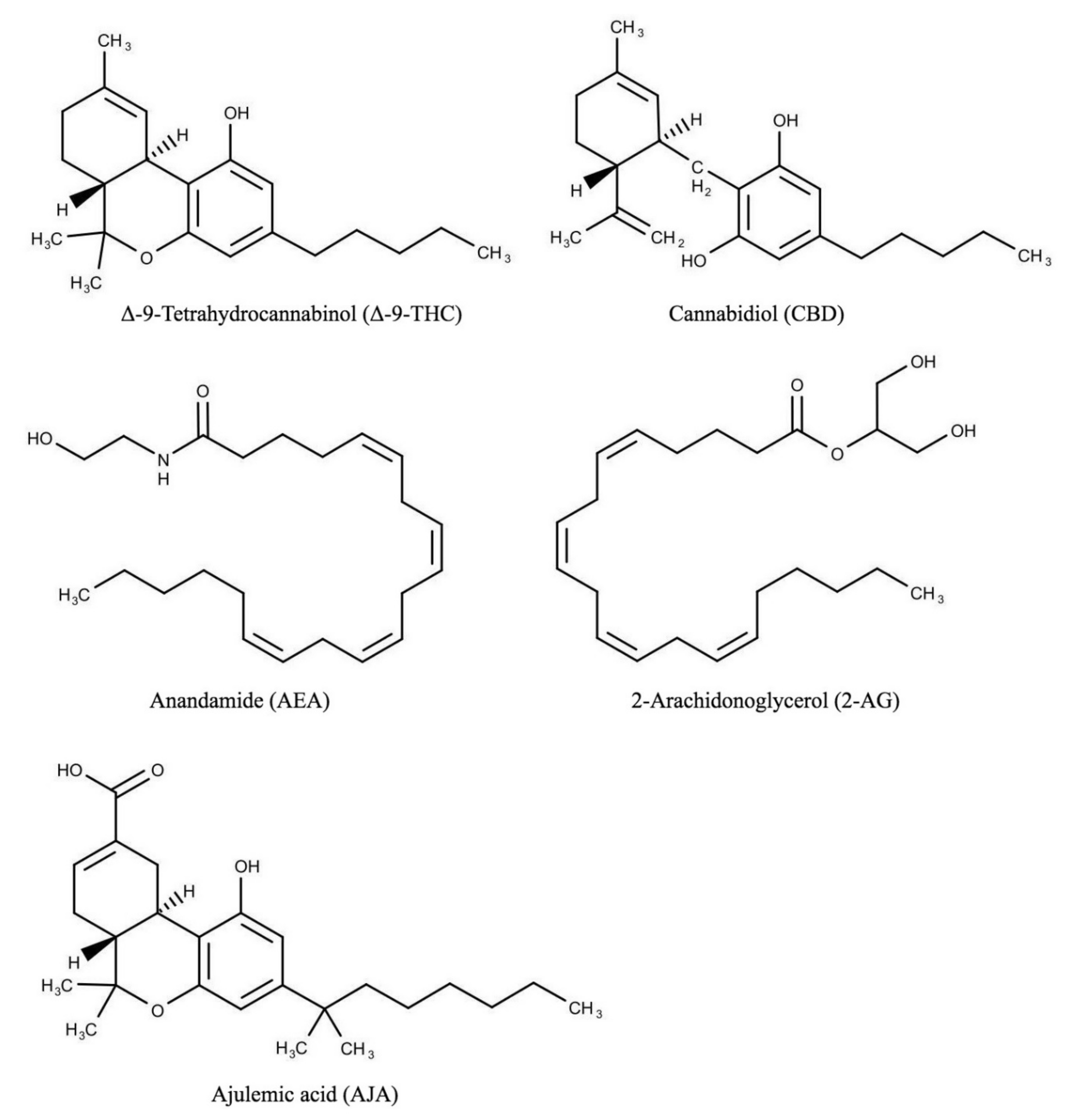
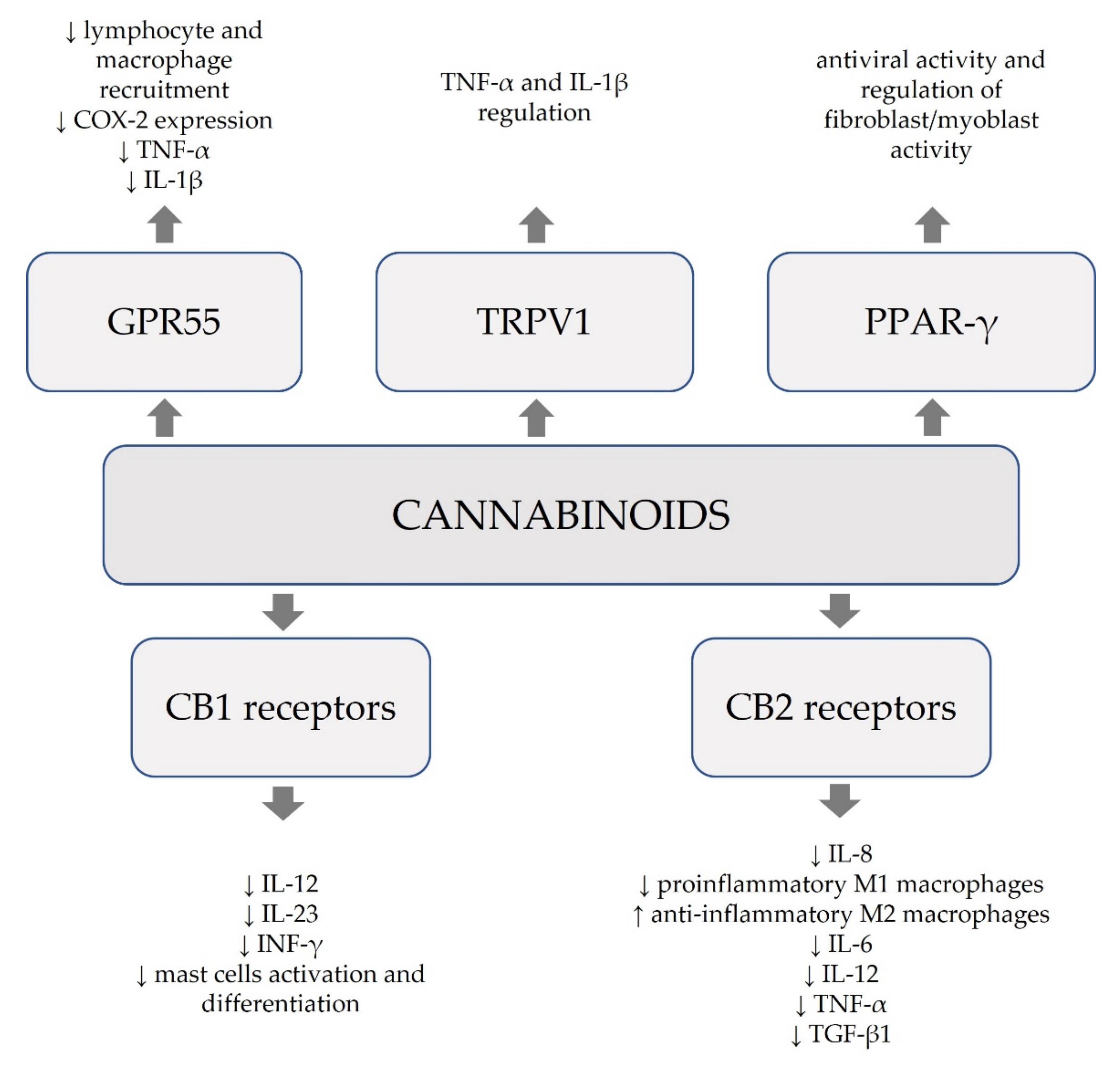
| Type of Cannabinoids | Mode of Action | Effect/Process | Reference |
|---|---|---|---|
| Cannabinoids (generally) | inhibition of leukocyte proliferation, induction of apoptosis of T lymphocytes and macrophages, decrease in the excretion of pro-inflammatory cytokines | mitigation of the inflammatory response dependent on the specific kind of the applied cannabinoid, dosage, the frequency of administration, and the cells they mediate | [17,19] |
| increased production of anti-inflammatory eicosanoids | difference between cannabinoids and cyclooxygenase-2 inhibitors that suppress the synthesis of the pro-inflammatory eicosanoids | [5] | |
| omnidirectional influence of cannabinoids | possible benefits associated with the anti-inflammatory effects, along with the positive response to attempts to relieve other symptoms | [22] | |
| Cannabinoids (natural and synthetic) | modulation of releasing pro-inflammatory mediators (IL-1β, TNF-α, and COX-2) and leucocyte recruitment mediated by G protein-coupled receptor 55 (GPR55) | involvement in development of neuropathic and inflammatory pain | [15,23] |
| CBD -phytocannabinoid of Cannabis sativa L. | affinity to PPAR-γ, 5-HT1A, adenosine A2A, and TRP | antioxidant, immunomodulatory and anti-inflammatory effects, mitigation of uncontrolled cytokine production, antiviral activity, regulation of fibroblast/myoblast activation, amelioration of lung function | [14,24] |
| THC- phytocannabinoid of Cannabis sativa L. | inhibition of the release of proinflammatory cytokines-IFNγ, TNF-α, IL-17A, and IL-22 | possible protective capacity of cannabinoids against carcinogenesis in the colon | [25] |
| functions independent of CB1 and CB2 receptors | inhibition of the production of IFN-γ by T lymphocytes and decrease in the release of pro-inflammatory chemokines and cytokines induced by IFN-γ | [26] | |
| CBG- phytocannabinoid of Cannabis sativa L. | reduction of the inflammatory molecules -TNF-α, IL-1β, IL-6, PGE2 MIP-1α in microglial cells and glutamate-induced oxidative cell death in hippocampal cells | anti-inflammatory and antioxidative properties, neuroprotective potential to reduce the severity of neurologic illnesses | [27] |
| ECS | decrease in MHC class II on the surface of dendritic cells | modulation of immunological processes, antigen presentation and inhibition of peripheral T-cell activation | [18] |
| CB1 receptor agonists | limitation of the activation and differentiation of mast cells | anti-inflammatory effect possibly beneficial in treating chronic inflammatory skin disorders | [28,29] |
| inhibition of the release of pro-inflammatory cytokines IL-12, IL-23, and INF-γ by T lymphocytes | [30] | ||
| CB2 receptor agonists | stimulation of the receptors expressed in B lymphocytes, NK cells, monocytes, neutrophils, and leucocytes CD8 and CD4 | mitigation of inflammatory response, immunomodulatory effects | [17] |
| inhibition of the release of IL-8 induced by TNF-α in colon epithelial cells | influence on immunological homeostasis of the intestine | [31] | |
| significant decrease in pro-inflammatory M1 macrophages, increase in anti-inflammatory M2 macrophages; inhibition of the release of cytokines IL-6, IL-12, CD86, iNOS; decrease in MCP-1, SDF-1, IL-6, IL-1β, TNF-α, TGF-β1, VEGF | reduced infiltration of neutrophils and macrophages, increased keratinocyte proliferation, and faster wound healing | [32] | |
| decrease in the production of pro-inflammatory cytokines and immune cell proliferation | possible benefits in limiting inflammatory response in SARS-CoV-2 infection | [33] | |
| AJA | preferentially binding to CB2 receptors; inhibition of IL-1β release | potential role in inhibiting tumor progression by reducing inflammation in the microenvironment of the tumor | [34] |
| non-cannabinoid compounds of cannabis- olivetol, cannflavin, and BCP | mechanisms mediated by CB2 receptors; decrease in the production of pro-inflammatory mediators | anti-inflammatory effects mediated by CB2 receptor-an important therapeutic target in many diseases | [6,7] |
| Study | Study Design | Number of Participants | Drug/Substance | Dosage | Condition | Treatment Duration | Endpoints/ Measures | Outcomes | Limitations |
|---|---|---|---|---|---|---|---|---|---|
| Lal 2011 [44] | questionnaire based survey | UC 100; CD 191 | self-administration of cannabis | UC/CD | quality of life improvement | symptom relief and improved quality of life | patient-reported survery; high bias | ||
| Kafil 2018 [41] | randomized controlled trial | 21 | cannabis cigarettes containing THC | 115 mg | active CD | eight weeks | clinical response and remission | positive response to the treatment and clinical remission, mild side effects | high bias |
| Kafil 2018 [41] | randomized controlled trial | 50 | cannabis oil consisting of 15% CBD and 4% THC | active CD | eight weeks | improvement in mean quality of life score and CDAI score | no clinical remission, improved quality of life, lowered CDAI score | low certainty evidence | |
| Storr 2014 [45] | questionnaire based survey | 313 | self-administration of cannabis | IBD | relief of abdominal pain, abdominal contractions, joint pain and diarrhea; higher risk of surgery in patients with Crohn’s disease | patient-reported study; high bias | |||
| Lahat 2012 [46] | questionnaire based survey | 13 | cannabis | IBD | three months | measurement of quality of life, disease activity and weight gain | improved quality of life, clinical disease activity and increased body mass index | high bias |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Graczyk, M.; Lewandowska, A.A.; Dzierżanowski, T. The Therapeutic Potential of Cannabis in Counteracting Oxidative Stress and Inflammation. Molecules 2021, 26, 4551. https://doi.org/10.3390/molecules26154551
Graczyk M, Lewandowska AA, Dzierżanowski T. The Therapeutic Potential of Cannabis in Counteracting Oxidative Stress and Inflammation. Molecules. 2021; 26(15):4551. https://doi.org/10.3390/molecules26154551
Chicago/Turabian StyleGraczyk, Michał, Agata Anna Lewandowska, and Tomasz Dzierżanowski. 2021. "The Therapeutic Potential of Cannabis in Counteracting Oxidative Stress and Inflammation" Molecules 26, no. 15: 4551. https://doi.org/10.3390/molecules26154551
APA StyleGraczyk, M., Lewandowska, A. A., & Dzierżanowski, T. (2021). The Therapeutic Potential of Cannabis in Counteracting Oxidative Stress and Inflammation. Molecules, 26(15), 4551. https://doi.org/10.3390/molecules26154551







