Antiproliferative Efficacy of N-(3-chloro-4-fluorophenyl)-6,7-dimethoxyquinazolin-4-amine, DW-8, in Colon Cancer Cells Is Mediated by Intrinsic Apoptosis
Abstract
:1. Introduction
2. Results
2.1. Chemistry
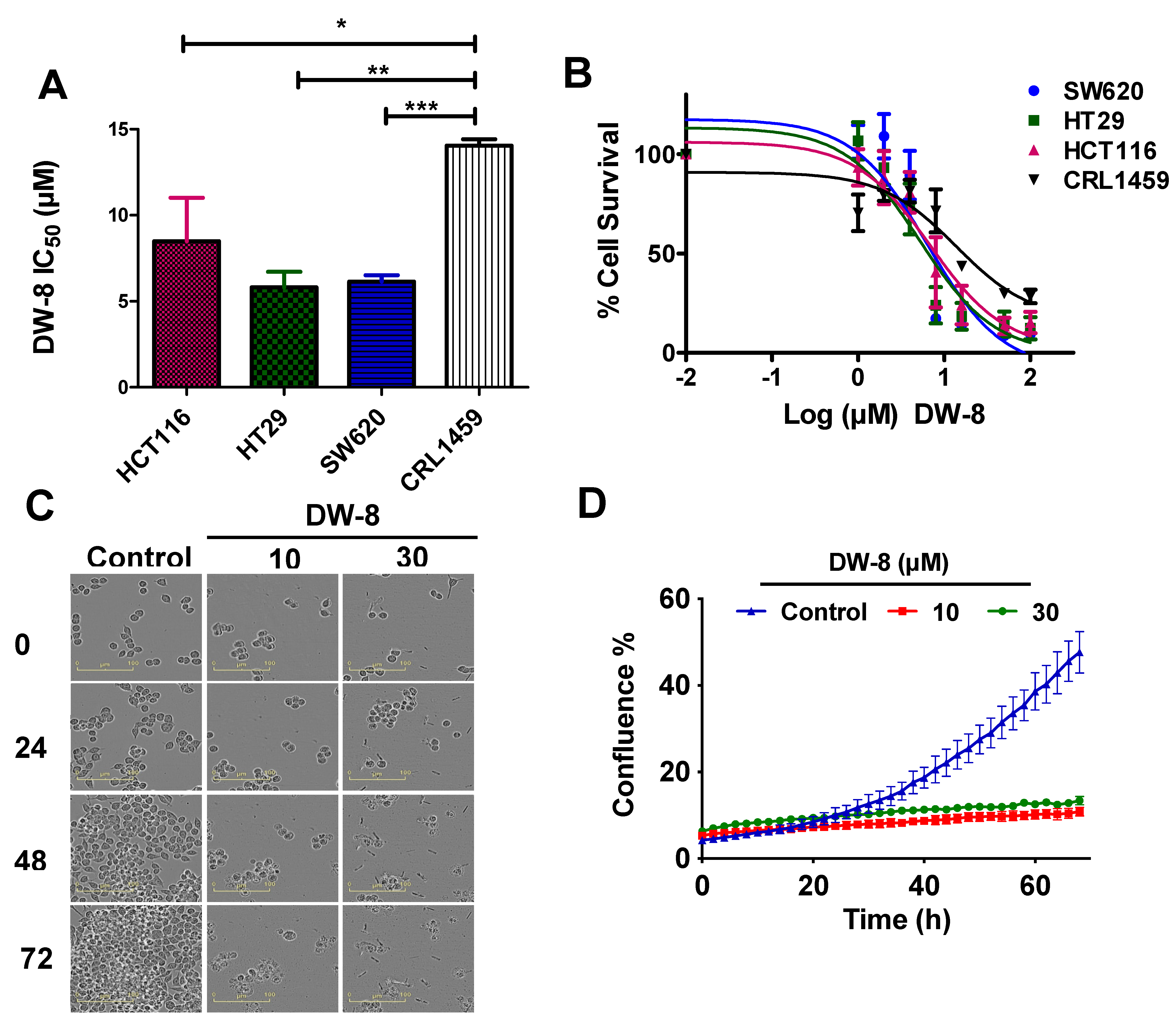
2.2. 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide-Based Cytotoxicity Assay and Morphological Changes Induced by DW-8 in SW620 Cells
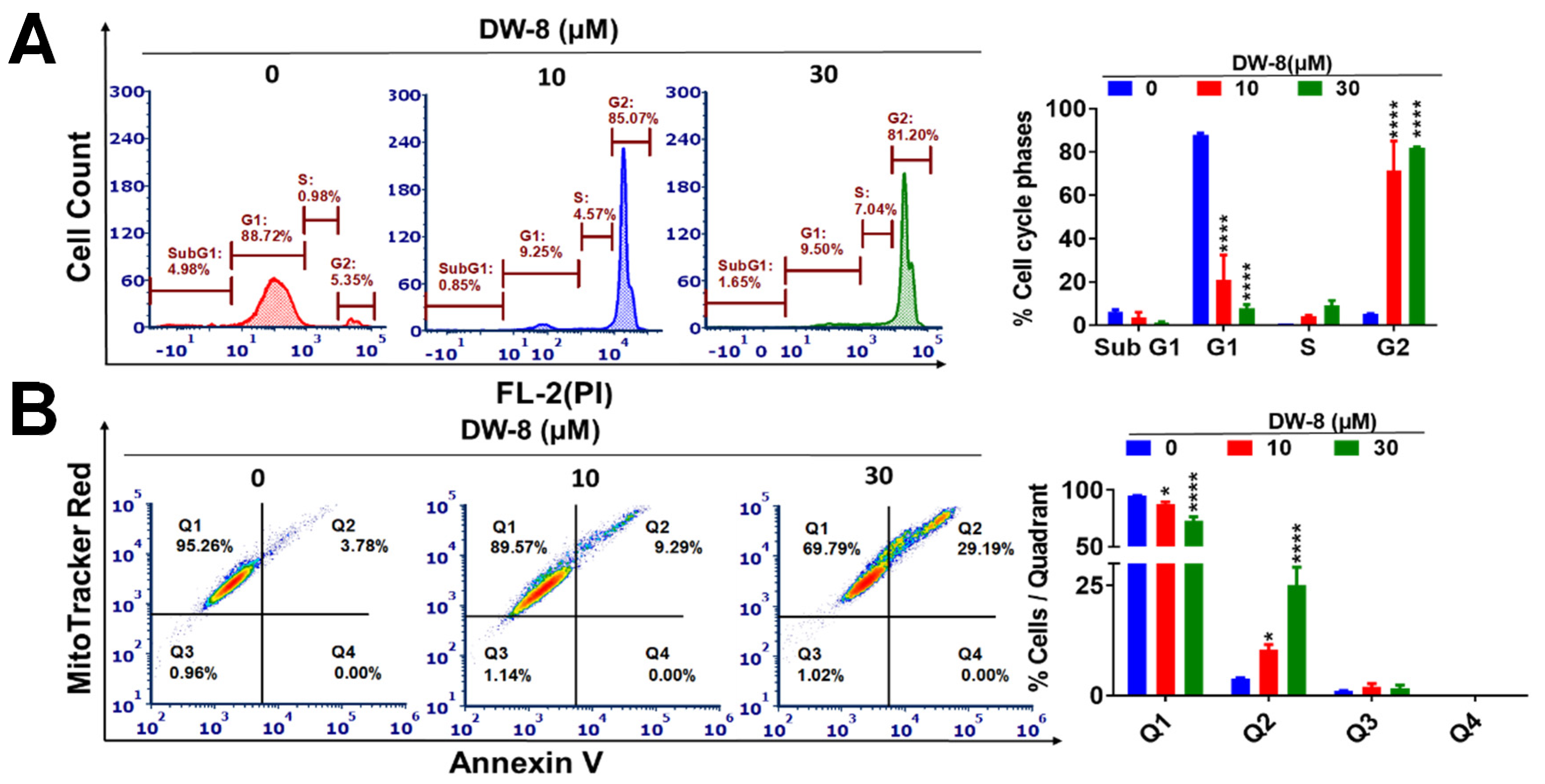
2.3. The Effect DW-8 on the Cell Cycle and Mitochondrial Membrane Potential of SW620 Cells
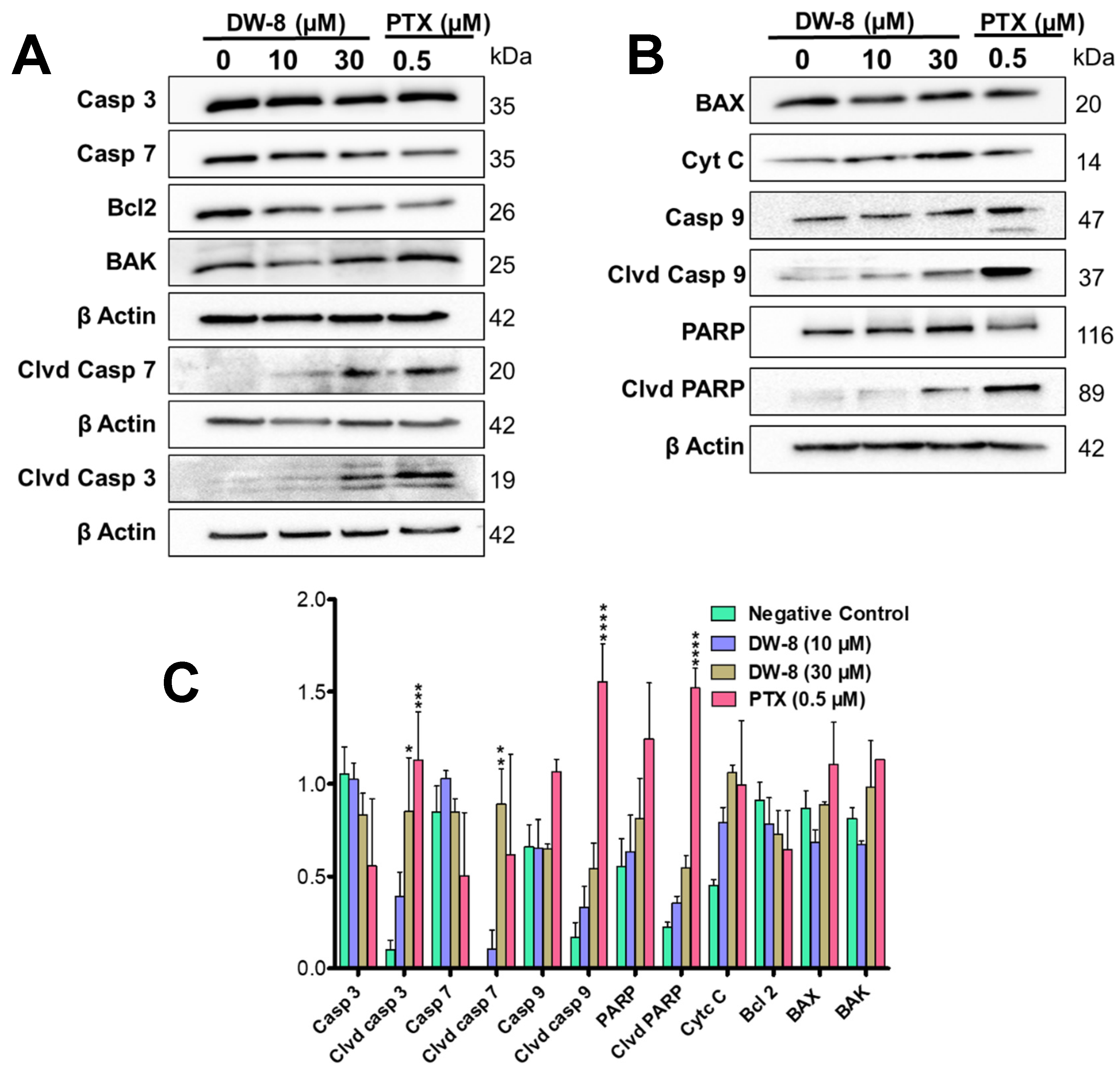
2.4. DW-8 Increases the Induction of Apoptosis by Activating the Intrinsic Apoptotic Pathway
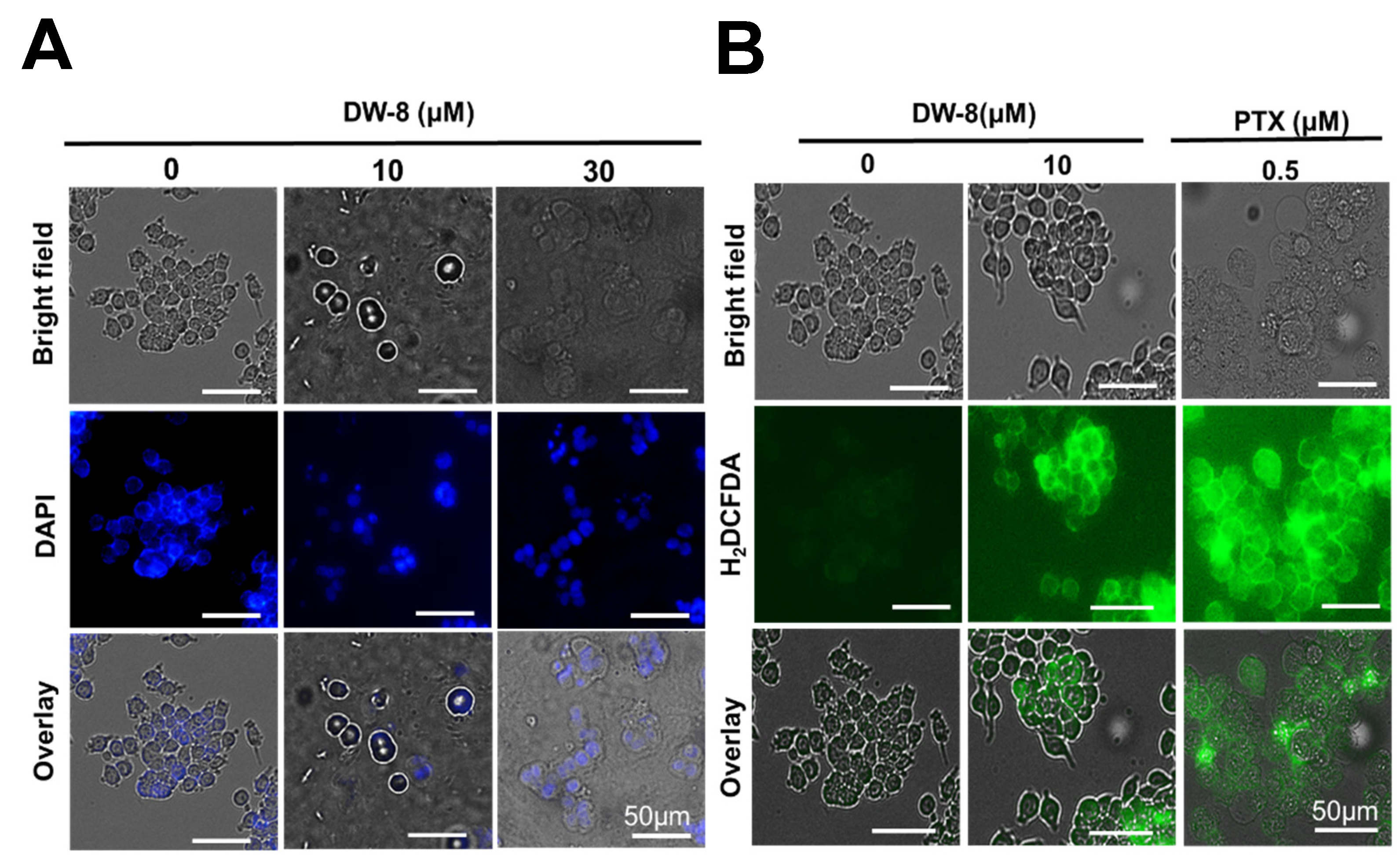
2.5. Nuclear Morphology and Reactive Oxygen Species (ROS) Assay
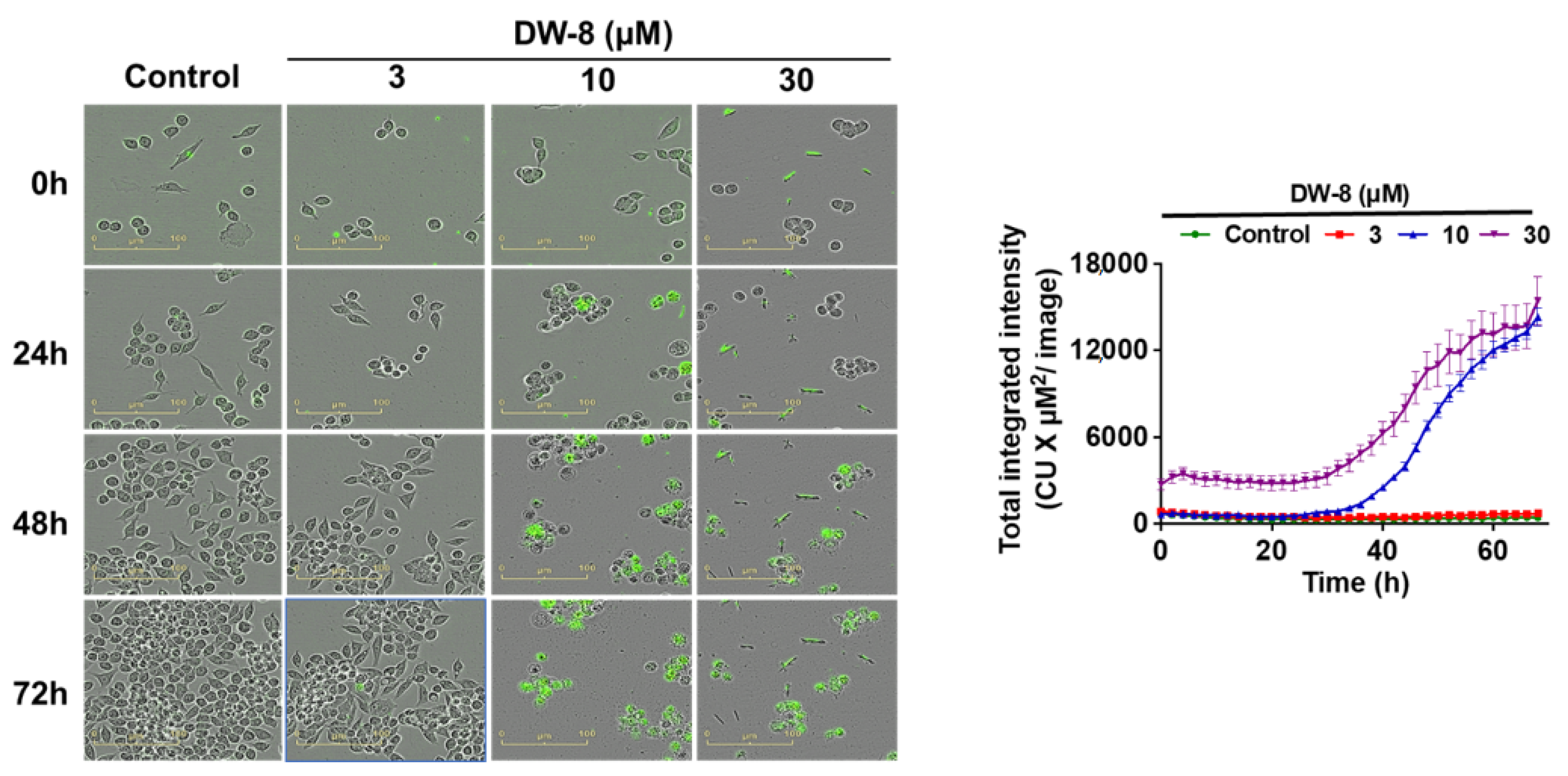
2.6. The Effect of DW-8 on Cell Viability Using the Incucyte™ Cytotox Green Assay
3. Discussion and Conclusions
4. Material and Methods
4.1. Chemistry
4.2. Biological Studies
4.2.1. Reagents
4.2.2. Cell Culture
4.2.3. MTT Assay
4.2.4. Cell Cycle Analysis
4.2.5. Detection of Reactive Oxygen Species (ROS)
4.2.6. Nuclear Staining
4.2.7. Determination of the Mitochondrial Membrane Potential and the Detection of Apoptosis
4.2.8. Determination of Cell Viability Using the IncuCyte™ Cytotox Green Assay
4.2.9. Western Blot
4.2.10. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Globocan. All Cancers Fact Sheets. 2020. Available online: https://gco.iarc.fr/today/data/factsheets/cancers/39-All-cancers-fact-sheet.pdf (accessed on 21 January 2021).
- Lee, S.-Y.; Oh, S.C. Advances of Targeted Therapy in Treatment of Unresectable Metastatic Colorectal Cancer. BioMed Res. Int. 2016, 2016, 7590245. [Google Scholar] [CrossRef] [Green Version]
- Chemotherapy of metastatic colorectal cancer: Fluorouracil plus folinic acid and irinotecan or oxaliplatin. Prescrire Int. 2005, 14, 230–233.
- Van der Jeught, K.; Xu, H.-C.; Li, Y.-J.; Lu, X.-B.; Ji, G. Drug resistance and new therapies in colorectal cancer. World J. Gastroenterol. 2018, 24, 3834–3848. [Google Scholar] [CrossRef] [PubMed]
- Shagufta, S.; Ahmad, I. An insight into the therapeutic potential of quinazoline derivatives as anticancer agents. MedChemComm 2017, 8, 871–885. [Google Scholar] [CrossRef]
- Ravez, S.; Castillo-Aguilera, O.; Depreux, P.; Goossens, L. Quinazoline derivatives as anticancer drugs: A patent review (2011–present). Expert Opin. Ther. Pat. 2015, 25, 789–804. [Google Scholar] [CrossRef]
- Mehta, D.K.; Dhanawat, M. Bestowal of Quinazoline Scaffold in Anticancer Drug Discovery. Anti-Cancer Agents Med. Chem. 2021, 21, 1350–1368. [Google Scholar] [CrossRef]
- Bansal, R.; Malhotra, A. Therapeutic progression of quinazolines as targeted chemotherapeutic agents. Eur. J. Med. Chem. 2021, 211, 113016. [Google Scholar] [CrossRef] [PubMed]
- Roskoski, R., Jr. The ErbB/HER receptor protein-tyrosine kinases and cancer. Biochem. Biophys. Res. Commun. 2004, 319, 1–11. [Google Scholar] [CrossRef]
- Fry, D.W. Mechanism of action of erbB tyrosine kinase inhibitors. EGF Recept. Fam. 2003, 137–145. [Google Scholar] [CrossRef]
- Wu, P.; Nielsen, T.E.; Clausen, M.H. FDA-approved small-molecule kinase inhibitors. Trends Pharmacol. Sci. 2015, 36, 422–439. [Google Scholar] [CrossRef] [Green Version]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Adams, J.M.; Cory, S. Bcl-2-regulated apoptosis: Mechanism and therapeutic potential. Curr. Opin. Immunol. 2007, 19, 488–496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leibowitz, B.; Yu, J. Mitochondrial signaling in cell death via the Bcl-2 family. Cancer Biol. Ther. 2010, 9, 417–422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lakhani, S.; Masud, A.; Kuida, K.; Porter, G.A., Jr.; Booth, C.J.; Mehal, W.Z.; Inayat, I.; Flavell, R.A. Caspases 3 and 7: Key Mediators of Mitochondrial Events of Apoptosis. Science 2006, 311, 847–851. [Google Scholar] [CrossRef] [Green Version]
- Yakovlev, A.G.; Wang, G.; Stoica, B.; Boulares, H.A.; Spoonde, A.Y.; Yoshihara, K.; Smulson, M.E. A Role of the Ca2+/Mg2+-dependent Endonuclease in Apoptosis and Its Inhibition by Poly(ADP-ribose) Polymerase. J. Biol. Chem. 2000, 275, 21302–21308. [Google Scholar] [CrossRef] [Green Version]
- Waiker, D.K.; Karthikeyan, C.; Poongavanam, V.; Kongsted, J.; Lozach, O.; Meijer, L.; Trivedi, P. Synthesis, biological evaluation and molecular modelling studies of 4-anilinoquinazoline derivatives as protein kinase inhibitors. Bioorg. Med. Chem. 2014, 22, 1909–1915. [Google Scholar] [CrossRef]
- Häcker, G. The morphology of apoptosis. Cell Tissue Res. 2000, 301, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Kang, J.; Fu, C. The independence of and associations among apoptosis, autophagy, and necrosis. Signal. Transduct. Target. Ther. 2018, 3, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ricci, M.S.; Zong, W. Chemotherapeutic Approaches for Targeting Cell Death Pathways. Oncologist 2006, 11, 342–357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stewart, Z.A.; Westfall, M.D.; A Pietenpol, J. Cell-cycle dysregulation and anticancer therapy. Trends Pharmacol. Sci. 2003, 24, 139–145. [Google Scholar] [CrossRef]
- Vermes, I.; Haanen, C.; Steffens-Nakken, H.; Reutelingsperger, C. A novel assay for apoptosis flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled annexin V. J. Immunol. Methods 1995, 184, 39–51. [Google Scholar] [CrossRef] [Green Version]
- Koopman, G.; Reutelingsperger, C.P.; Kuijten, G.A.; Keehnen, R.M.; Pals, S.T.; van Oers, M.H. Annexin V for flow cytometric detection of phosphatidylserine expression on B cells undergoing apoptosis. Blood 1994, 84, 1415–1420. [Google Scholar] [CrossRef] [Green Version]
- Zhang, G.; Gurtu, V.; Kain, S.R.; Yan, G. Early Detection of Apoptosis Using a Fluorescent Conjugate of Annexin V. Biotechiques 1997, 23, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Kholmukhamedov, A.; Schwartz, J.M.; Lemasters, J.J. Mitotracker probes and mitochondrial membrane potential. Shock 2013, 39, 543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larsen, T.A.; Goodsell, D.S.; Cascio, D.; Grzeskowiak, K.; Dickerson, R.E. The Structure of DAPI Bound to DNA. J. Biomol. Struct. Dyn. 1989, 7, 477–491. [Google Scholar] [CrossRef] [PubMed]
- Axel, D.I.; Kunert, W.; Göggelmann, C.; Oberhoff, M.; Herdeg, C.; Küttner, A.; Wild, D.H.; Brehm, B.R.; Riessen, R.; Köveker, G.; et al. Paclitaxel Inhibits Arterial Smooth Muscle Cell Proliferation and Migration In Vitro and In Vivo Using Local Drug Delivery. Circulation 1997, 96, 636–645. [Google Scholar] [CrossRef]
- Yoshida, K.; Miki, Y. The cell death machinery governed by the p53 tumor suppressor in response to DNA damage. Cancer Sci. 2010, 101, 831–835. [Google Scholar] [CrossRef] [PubMed]
- Dashzeveg, N.; Yoshida, K. Cell death decision by p53 via control of the mitochondrial membrane. Cancer Lett. 2015, 367, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Yotnda, P. Production and detection of reactive oxygen species (ROS) in cancers. J. Vis. Exp. 2011, 57, 3357. [Google Scholar] [CrossRef] [PubMed]
- Alexandre, J.; Batteux, F.; Nicco, C.; Chéreau, C.; Laurent, A.; Guillevin, L.; Weill, B.; Goldwasser, F. Accumulation of hydrogen peroxide is an early and crucial step for paclitaxel-induced cancer cell death both in vitro andin vivo. Int. J. Cancer 2006, 119, 41–48. [Google Scholar] [CrossRef]
- Xu, Y.; Xin, Y.; Diao, Y.; Lu, C.; Fu, J.; Luo, L.; Yin, Z. Synergistic Effects of Apigenin and Paclitaxel on Apoptosis of Cancer Cells. PLoS ONE 2011, 6, e29169. [Google Scholar] [CrossRef] [Green Version]
- Amawi, H.; Hussein, N.A.; Ashby, C.R.J.; Alnafisah, R.; Sanglard, L.M.; Manivannan, E.; Karthikeyan, C.; Trivedi, P.; Eisenmann, K.M.; Robey, R.; et al. Bax/Tubulin/Epithelial-Mesenchymal Pathways Determine the Efficacy of Silybin Analog HM015k in Colorectal Cancer Cell Growth and Metastasis. Front. Pharmacol. 2018, 9, 520. [Google Scholar] [CrossRef] [PubMed]
- Balaji, S.; Terrero, D.; Tiwari, A.K.; Ashby, C.R.; Raman, D. Alternative approaches to overcome chemoresistance to apoptosis in cancer. DNA Repair 2021, 126, 91–122. [Google Scholar] [CrossRef]
- Kamal, A.; Faazil, S.; Malik, M.S. Apoptosis-inducing agents: A patent review (2010–2013). Expert Opin. Ther. Pat. 2014, 24, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Li, Y.; Zhang, G. Cell cycle regulation and anticancer drug discovery. Cancer Biol. Med. 2017, 14, 348. [Google Scholar] [PubMed]
- Simon, H.-U.; Haj-Yehia, A.; Levi-Schaffer, F. Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis 2000, 5, 415–418. [Google Scholar] [CrossRef]
- Tukaramrao, D.; Malla, S.; Saraiya, S.; Hanely, R.; Ray, A.; Kumari, S.; Raman, D.; Tiwari, A. A Novel Thienopyrimidine Analog, TPH104, Mediates Immunogenic Cell Death in Triple-Negative Breast Cancer Cells. Cancers 2021, 13, 1954. [Google Scholar] [CrossRef]
- Amawi, H.; Karthikeyan, C.; Pathak, R.; Hussein, N.; Christman, R.; Robey, R.; Ashby, C.R.; Trivedi, P.; Malhotra, A.; Tiwari, A.K. Thienopyrimidine derivatives exert their anticancer efficacy via apoptosis induction, oxidative stress and mitotic catastrophe. Eur. J. Med. Chem. 2017, 138, 1053–1065. [Google Scholar] [CrossRef]
- Al-Oudat, B.A.; Ramapuram, H.; Malla, S.; Audat, S.A.; Hussein, N.; Len, J.M.; Kumari, S.; Bedi, M.F.; Ashby, J.C.R.; Tiwari, A.K. Novel Chrysin-De-Allyl PAC-1 Hybrid Analogues as Anticancer Compounds: Design, Synthesis, and Biological Evaluation. Molecules 2020, 25, 3063. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.M. The Bicinchoninic Acid (BCA) Assay for Protein Quantitation. Methods Mol. Biol. 2009, 11–15. [Google Scholar] [CrossRef]
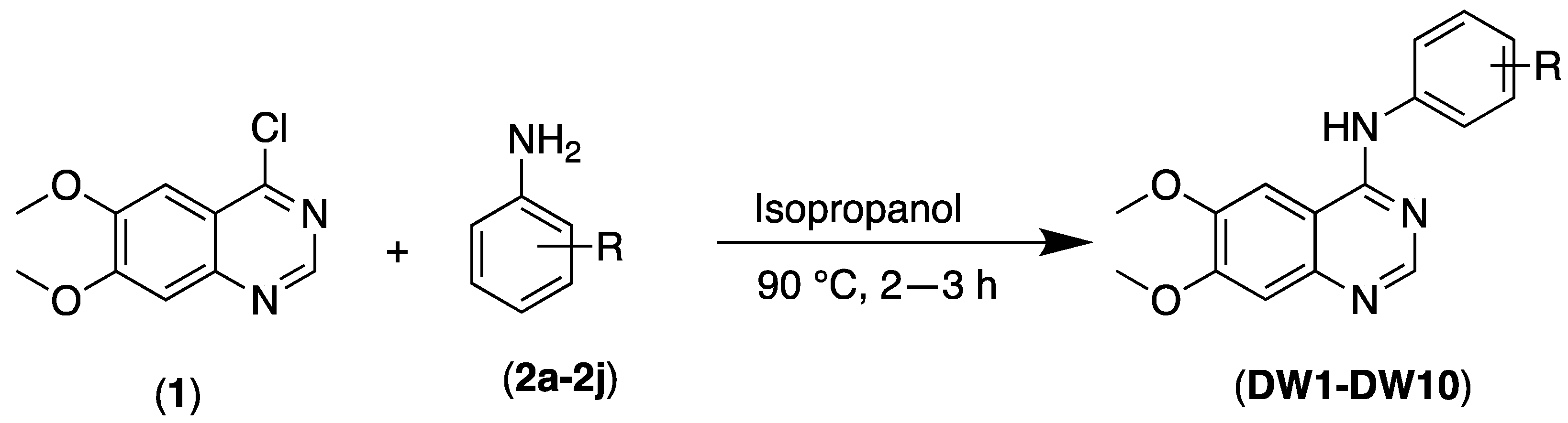
| DW Compounds | R | IC50 (µM) | |
|---|---|---|---|
| HCT116 | BT-20 | ||
| 1 | H | 64.44 ± 0.90 | 65.94 ± 7.89 |
| 2 | 3-CF3 | 40.05 ± 4.65 | 30.06 ± 7.84 |
| 3 | 3,4-dimethyl | 75.77 ± 10.98 | 88.50 ± 30.65 |
| 4 | 4-COCH3 | >100 | >100 |
| 5 | 3,4-dimethoxy | >100 | >100 |
| 6 | 4-OCH3 | >100 | >100 |
| 7 | 4-NHCOCH3 | >100 | >100 |
| 8 | 3-Cl, 4-F | 8.50 ± 2.53 | 11.69 ± 3.73 |
| 9 | 4-CF3 | 44.70 ± 5.63 | 39.46 ± 3.74 |
| 10 | 4-OCF3 | 49.20 ± 7.27 | 45.61 ± 3.31 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neupane, R.; Malla, S.; Abou-Dahech, M.S.; Balaji, S.; Kumari, S.; Waiker, D.K.; Moorthy, N.S.H.N.; Trivedi, P.; Ashby, C.R., Jr.; Karthikeyan, C.; et al. Antiproliferative Efficacy of N-(3-chloro-4-fluorophenyl)-6,7-dimethoxyquinazolin-4-amine, DW-8, in Colon Cancer Cells Is Mediated by Intrinsic Apoptosis. Molecules 2021, 26, 4417. https://doi.org/10.3390/molecules26154417
Neupane R, Malla S, Abou-Dahech MS, Balaji S, Kumari S, Waiker DK, Moorthy NSHN, Trivedi P, Ashby CR Jr., Karthikeyan C, et al. Antiproliferative Efficacy of N-(3-chloro-4-fluorophenyl)-6,7-dimethoxyquinazolin-4-amine, DW-8, in Colon Cancer Cells Is Mediated by Intrinsic Apoptosis. Molecules. 2021; 26(15):4417. https://doi.org/10.3390/molecules26154417
Chicago/Turabian StyleNeupane, Rabin, Saloni Malla, Mariam Sami Abou-Dahech, Swapnaa Balaji, Shikha Kumari, Digambar Kumar Waiker, N. S. Hari Narayana Moorthy, Piyush Trivedi, Charles R. Ashby, Jr., Chandrabose Karthikeyan, and et al. 2021. "Antiproliferative Efficacy of N-(3-chloro-4-fluorophenyl)-6,7-dimethoxyquinazolin-4-amine, DW-8, in Colon Cancer Cells Is Mediated by Intrinsic Apoptosis" Molecules 26, no. 15: 4417. https://doi.org/10.3390/molecules26154417
APA StyleNeupane, R., Malla, S., Abou-Dahech, M. S., Balaji, S., Kumari, S., Waiker, D. K., Moorthy, N. S. H. N., Trivedi, P., Ashby, C. R., Jr., Karthikeyan, C., & Tiwari, A. K. (2021). Antiproliferative Efficacy of N-(3-chloro-4-fluorophenyl)-6,7-dimethoxyquinazolin-4-amine, DW-8, in Colon Cancer Cells Is Mediated by Intrinsic Apoptosis. Molecules, 26(15), 4417. https://doi.org/10.3390/molecules26154417







