The DEPTQ+ Experiment: Leveling the DEPT Signal Intensities and Clean Spectral Editing for Determining CHn Multiplicities
Abstract
1. Introduction
2. Results and Discussion
2.1. Theory
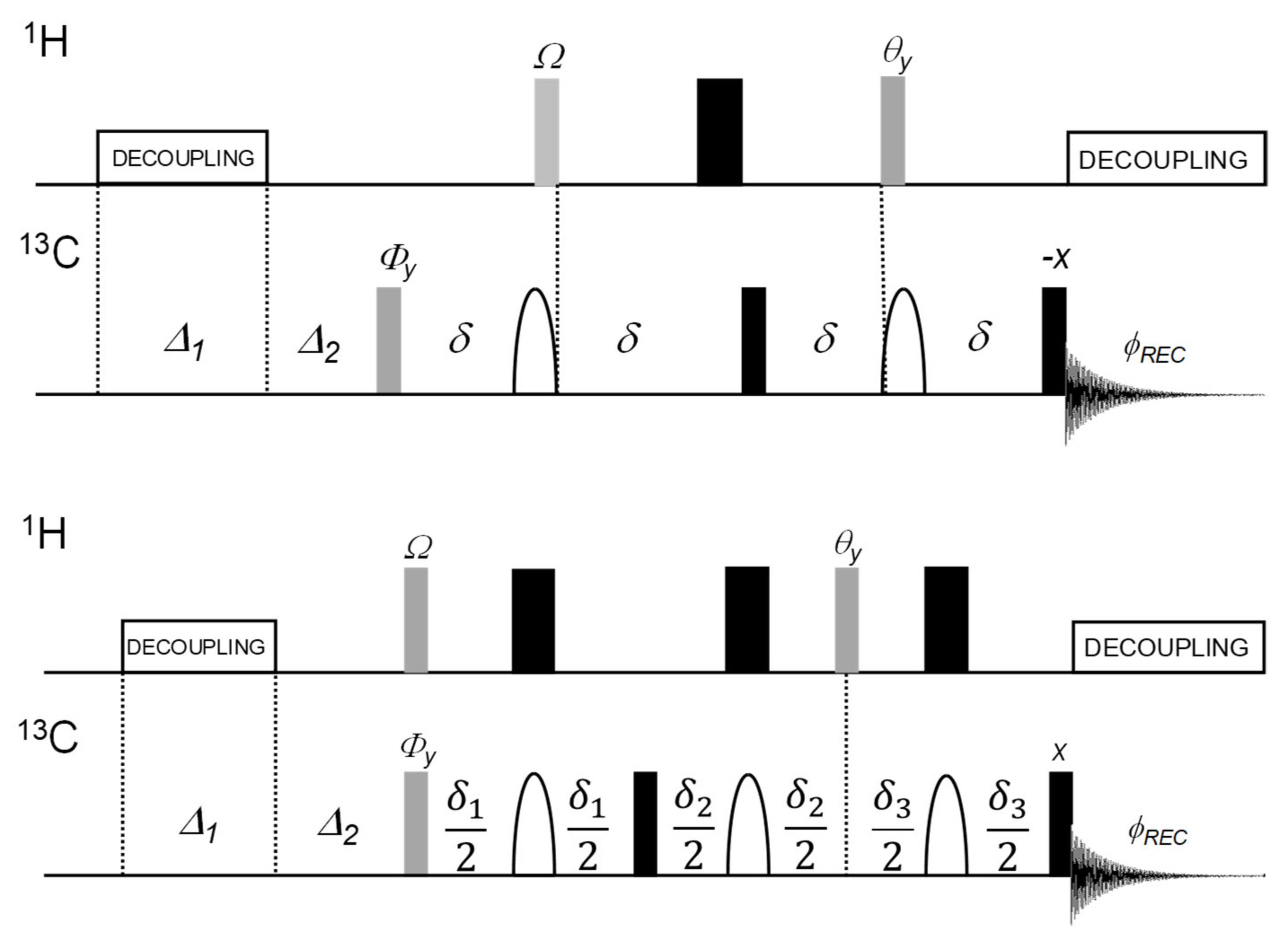
2.1.1. Original DEPTQ Pulse Sequence
Quaternary Carbons
ICH2 = Cx [0.35 sin2(2πJCHδ) − 1.00 sin4(πJCHδ)]
ICH3 = Cx [0.53 sin2(2πJCHδ) cos2(πJCHδ) − 0.75 sin2(2πJCHδ) sin2(πJCHδ) + 1.06
sin6(πJCHδ)]
ICH2 = −1.00 Cx
ICH3 = +1.06 Cx
ICH2 = Cx [0.35 sin2(2πJCHδ)]
ICH3 = Cx [2.11 sin2(πJCHδ) cos4(πJCHδ)]
ICH2 = 0
ICH3 = 0
ICH2 = Cx [0.35 sin2(2πJCHδ) − 1.00 sin4(πJCHδ)] − Cx [0.35 sin2(2πJCHδ)] = −Cx
sin4(πJCHδ)
ICH3 = Cx [0.53 sin2(2πJCHδ) cos2(πJCHδ) − 0.75 sin2(2πJCHδ) sin2(πJCHδ) + 1.06
sin6(πJCHδ)] − Cx [2.11 sin2(πJCHδ) cos4(πJCHδ)]
ICH3 = +1.06 Cx
2.1.2. The DEPTQ+ Pulse Sequence
Quaternary Carbons
ICH2 = Cx [0.71 sin(πJCHδ1) cos(πJCHδ2) sin(2πJCHδ3) − 1.00 sin(πJCHδ1) sin(πJCHδ2)
sin2(πJCHδ3)]
ICH3 = Cx [−2.11 sin(πJCHδ1) cos2(πJCHδ2) sin(πJCHδ3) cos2(πJCHδ3) + 3 sin(πJCHδ1)
cos(πJCHδ2) sin(πJCHδ2) cos(πJCHδ3) sin2(πJCHδ3) − 1.06 sin(πJCHδ1) sin2(πJCHδ2)
sin3(πJCHδ3)]
ICH2 = −Cx [0.71 sin (πJCHδ1) cos (πJCHδ2) sin (2πJCHδ3)]
ICH3 = −Cx [2.11 sin (πJCHδ1) cos2(πJCHδ2) sin (πJCHδ3) cos2(πJCHδ3)]
2.2. Experimental Results
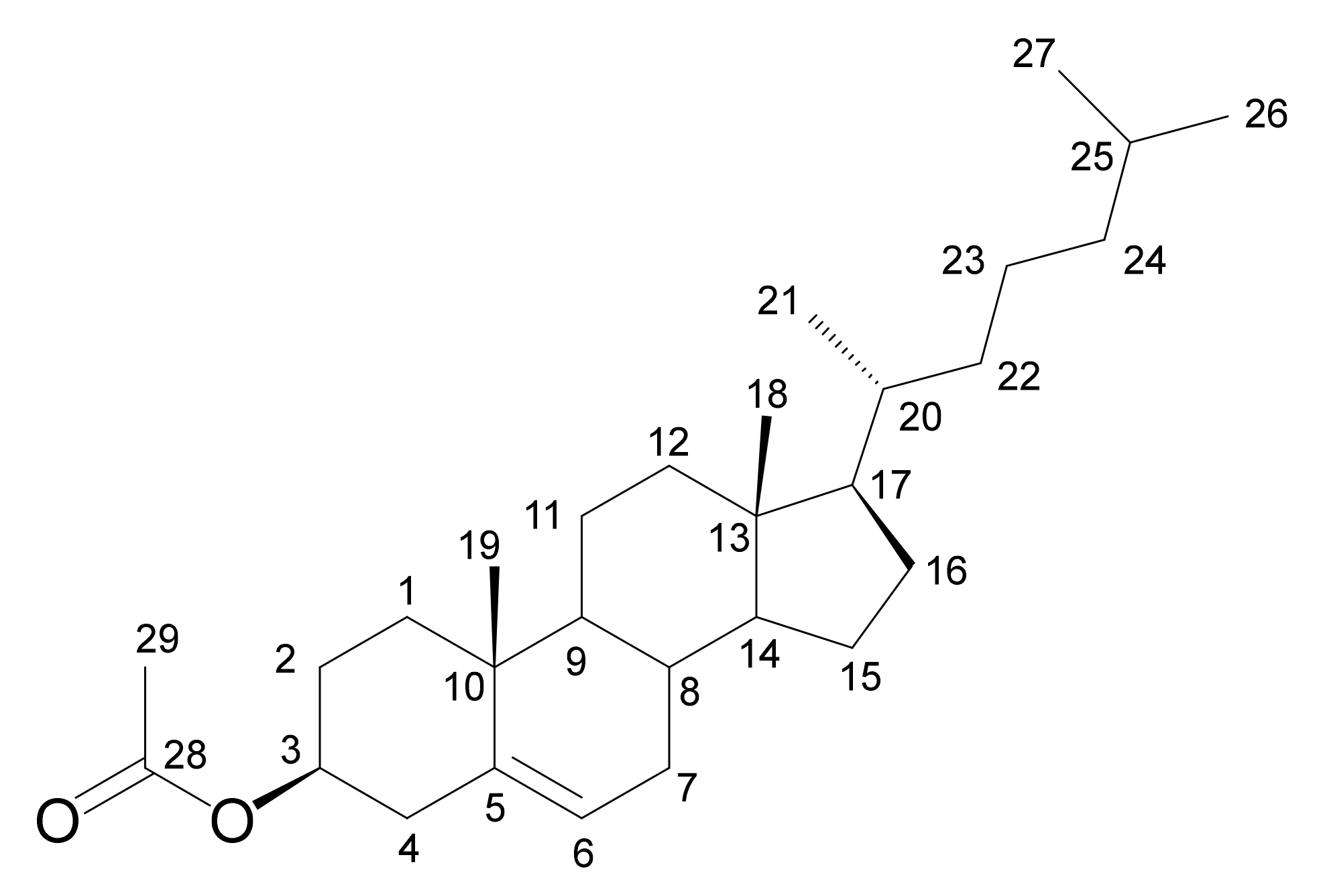
2.2.1. Cholesteryl Acetate
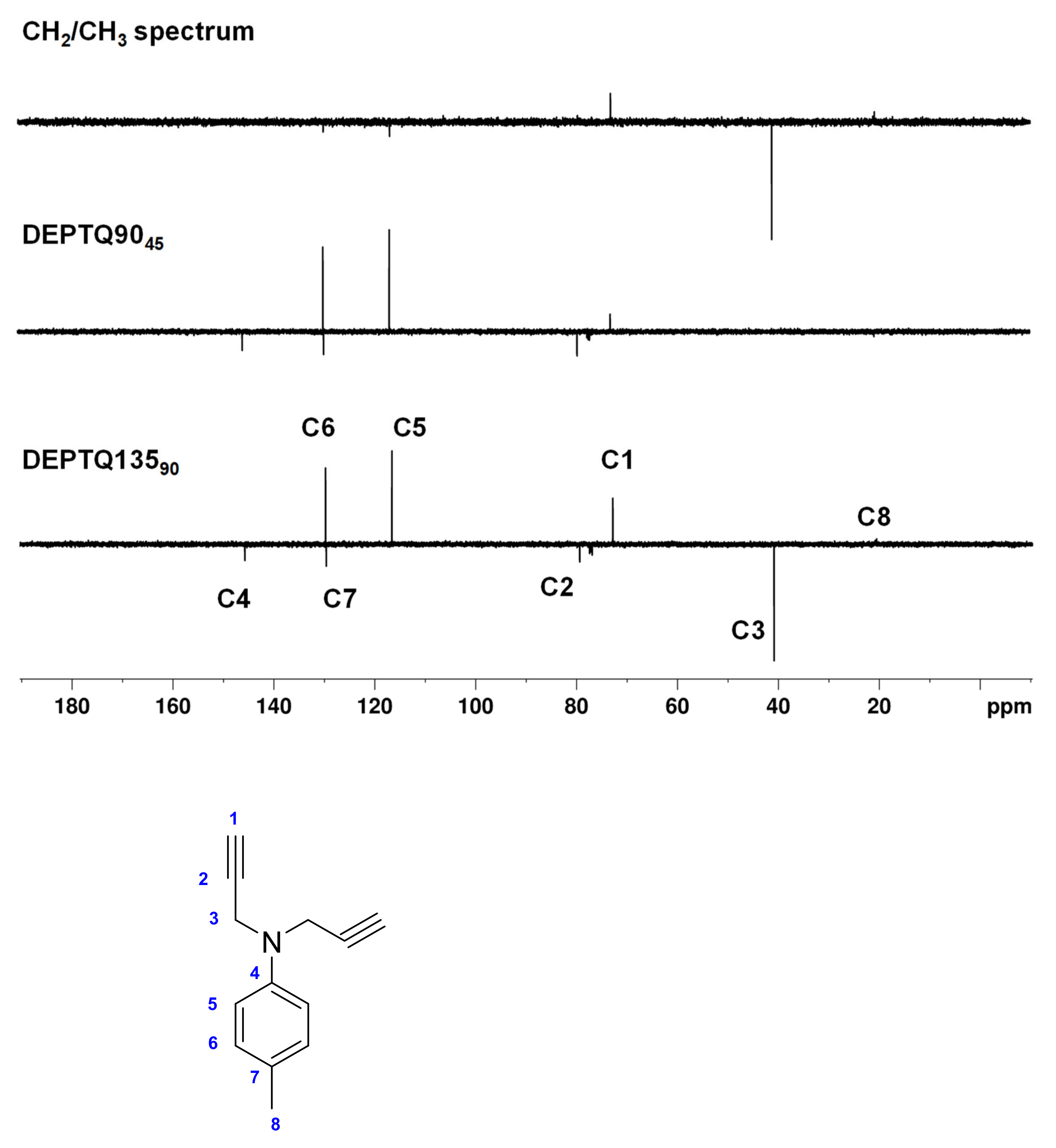
2.2.2. 4-Methyl-N,N-di(prop-2-yn-1-yl)aniline
2.3. Practical Aspects
3. Materials and Methods
3.1. Synthesis and Characterization of 4-Methyl-N,N-di(prop-2-yn-1-yl)aniline
3.2. NMR Measurements
3.3. Numerical Simulations
3.4. NMR Simulations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Sample Availability
References
- Saurí, J.; Liu, Y.; Parella, T.; Williamson, R.T.; Martin, G.E. Selecting the Most Appropriate NMR Experiment to Access Weak and/or Very Long-Range Heteronuclear Correlations. J. Nat. Prod. 2016, 79, 1400–1406. [Google Scholar] [CrossRef]
- Le Cocq, C.; Lallemand, J.-Y. Precise carbon-13 n.m.r. multiplicity determination. J. Chem. Soc. Chem. Commun. 1981, 150–152. [Google Scholar] [CrossRef]
- Patt, S.L.; Shoorley, J.N. Attached Proton Test for Carbon-13 NMR. J. Magn. Reson. 1982, 46, 535–539. [Google Scholar] [CrossRef]
- Homer, J.; Perry, M.C. New method for NMR signal enhancement by polarization transfer, and attached nucleus testing. J. Chem. Soc. Chem. Commun. 1994, 373–374. [Google Scholar] [CrossRef]
- Homer, J.; Perry, M.C. Enhancement of the NMR spectra of insensitive nuclei using PENDANT with long-range coupling constants. J. Chem. Soc. Perkin Trans. 2 1995, 533–536. [Google Scholar] [CrossRef]
- Burum, D.; Ernst, R. Net polarization transfer via a J-ordered state for signal enhancement of low-sensitivity nuclei. J. Magn. Reson. 1980, 39, 163–168. [Google Scholar] [CrossRef]
- Dodrell, D.M.; Pegg, D.T.; Bendall, M.R. Distortionless enhancement of NMR signals by polarization transfer. J. Magn. Reson. 1982, 48, 323–327. [Google Scholar] [CrossRef]
- Primasova, H.; Bigler, P.; Furrer, J. Chapter One—The DEPT Experiment and Some of Its Useful Variants. In Annual Reports on NMR Spectroscopy; Webb, G.A., Ed.; Academic Press: Cambridge, MA, USA, 2017; Volume 92, pp. 1–82. [Google Scholar]
- Burger, R.; Bigler, P. DEPTQ: Distortionless enhancement by polarization transfer including the detection of quaternary nuclei. J. Magn. Reson. 1998, 135, 529–534. [Google Scholar] [CrossRef]
- Bigler, P.; Kümmerle, R.; Bermel, W. Multiplicity editing including quaternary carbons: Improved performance for the13C-DEPTQ pulse sequence. Magn. Reson. Chem. 2007, 45, 469–472. [Google Scholar] [CrossRef]
- Bigler, P. Fast 13 C-NMR Spectral Editing for Determining CH n Multiplicities. Spectrosc. Lett. 2008, 41, 162–165. [Google Scholar] [CrossRef]
- Bildsøe, H.; Dønstrup, S.; Jakobsen, H.; Sørensen, O. Subspectral editing using a multiple quantum trap: Analysis of J cross-talk. J. Magn. Reson. 1983, 53, 154–162. [Google Scholar] [CrossRef]
- Furrer, J. Old and new experiments for obtaining quaternary-carbon-only NMR spectra. Appl. Spectrosc. Rev. 2021, 56, 128–142. [Google Scholar] [CrossRef]
- Bodenhausen, G.; Ruben, D.J. Natural abundance nitrogen-15 NMR by enhanced heteronuclear spectroscopy. Chem. Phys. Lett. 1980, 69, 185–189. [Google Scholar] [CrossRef]
- Bax, A.; Subramanian, S. Sensitivity-enhanced two-dimensional heteronuclear shift correla-tion NMR spectroscopy. J. Magn. Reson. 1986, 67, 565–569. [Google Scholar]
- Furrer, J. A comprehensive discussion of hmbc pulse sequences, part 1: The classical HMBC. Concepts Magn. Reson. Part. A 2012, 40, 101–127. [Google Scholar] [CrossRef]
- Reynolds, W.F.; Burns, D.C. Getting the Most Out of HSQC and HMBC Spectra. In Annual Reports on NMR Spectroscopy; Webb, G.A., Ed.; Elsevier BV: Amsterdam, The Netherlands, 2012; Volume 76, pp. 1–21. [Google Scholar]
- Williamson, R.T.; Buevich, A.V.; Martin, G.E.; Parella, T. LR-HSQMBC: A Sensitive NMR Technique to Probe Very Long-Range Heteronuclear Coupling Pathways. J. Org. Chem. 2014, 79, 3887–3894. [Google Scholar] [CrossRef]
- Furrer, J. A comprehensive discussion of HMBC pulse sequences. 2. Some useful variants. Concepts Magn. Reson. Part. A 2012, 40, 146–169. [Google Scholar] [CrossRef]
- Saurí, J.; Frédérich, M.; Tchinda, A.T.; Parella, T.; Williamson, R.T.; Martin, G.E. Carbon Multiplicity Editing in Long-Range Heteronuclear Correlation NMR Experiments: A Valuable Tool for the Structure Elucidation of Natural Products. J. Nat. Prod. 2015, 78, 2236–2241. [Google Scholar] [CrossRef]
- Sørensen, O.W.; Dønstrup, S.; Bildsøe, H.; Jakobsen, H.J. Suppression of J cross-talk in subspectral editing. The SEMUT GL pulse sequence. J. Magn. Reson. 1983, 55, 347–354. [Google Scholar] [CrossRef]
- Elyashberg, M.; Argyropoulos, D. Computer Assisted Structure Elucidation (CASE): Current and future perspectives. Magn. Reson. Chem. 2020. [Google Scholar] [CrossRef]
- Elyashberg, M. Identification and structure elucidation by NMR spectroscopy. TrAC Trends Anal. Chem. 2015, 69, 88–97. [Google Scholar] [CrossRef]
- Buevich, A.V.; Elyashberg, M.E. Synergistic Combination of CASE Algorithms and DFT Chemical Shift Predictions: A Powerful Approach for Structure Elucidation, Verification, and Revision. J. Nat. Prod. 2016, 79, 3105–3116. [Google Scholar] [CrossRef] [PubMed]
- Pupier, M.; Nuzillard, J.-M.; Wist, J.; Schlörer, N.E.; Kuhn, S.; Erdelyi, M.; Steinbeck, C.; Williams, A.J.; Butts, C.; Claridge, T.D.; et al. NMReDATA, a standard to report the NMR assignment and parameters of organic compounds. Magn. Reson. Chem. 2018, 56, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Elyashberg, M.; Argyropoulos, D. NMR-based Computer-assisted Structure Elucidation (CASE) of Small Organic Molecules in Solution: Recent Advances. Emagres 2019, 8, 239–253. [Google Scholar] [CrossRef]
- Buevich, A.V.; Elyashberg, M.E. Enhancing computer-assisted structure elucidation with DFT analysis of J-couplings. Magn. Reson. Chem. 2020, 58, 594–606. [Google Scholar] [CrossRef]
- Mateescu, G.D.; Valeriu, A. 2D NMR Density Matrix and Product Operator Treatment; Prentice Hall: Hoboken, NJ, USA, 1993. [Google Scholar]
- Bendall, M.R.; Pegg, D.T. Complete accurate editing of decoupled 13C spectra using DEPT and a quaternary-only sequence. J. Magn. Reson. 1983, 53, 272–296. [Google Scholar] [CrossRef]
- Kögler, H.; Sørensen, O.; Bodenhausen, G.; Ernst, R. Low-pass J filters. Suppression of neighbor peaks in heteronuclear relayed correlation spectra. J. Magn. Reson. 1983, 55, 157–163. [Google Scholar] [CrossRef]
- Furrer, J.; Guerra, S.; Deschenaux, R. Accordion-optimized DEPT experiments. Magn. Reson. Chem. 2011, 49, 16–22. [Google Scholar] [CrossRef]
- Holman, M.A.; Williamson, N.M.; Ward, A.D. Preparation and Cyclization of Some N-(2,2-Dimethylpropargyl) Homo- and Heteroaromatic Amines and the Synthesis of Some Pyrido[2,3-d]pyrimidines. Aust. J. Chem. 2005, 58, 368–374. [Google Scholar] [CrossRef]
- Majumdar, K.C.; Ganai, S. An expedient approach to substituted triazolo[1,5-a][1,4]benzodiazepines via Cu-catalyzed tandem Ullmann C–N coupling/azide-alkyne cycloaddition. Tetrahedron Lett. 2013, 54, 6192–6195. [Google Scholar] [CrossRef]




| DEPTQ | DEPTQ+ | |
|---|---|---|
| CH groups | sin2(πJCHδ) | sin(πJCHδ1) sin(πJCHδ3) |
| CH2 groups | sin4(πJCHδ) | sin(πJCHδ1) sin(πJCHδ2) sin2(πJCHδ3) |
| CH3 groups | sin6(πJCHδ) | sin(πJCHδ1) sin2(πJCHδ2) sin3(πJCHδ3) |
| Experiment | Angle θ | Amplitude of CH | Amplitude of CH2 | Amplitude of CH3 |
|---|---|---|---|---|
| DEPTQ+ | 45° | −0.71 | 1 | −1.06 |
| DEPTQ | 0.71 | 1 | 1.06 | |
| DEPTQ+ | 90° | −1 | 0 | 0 |
| DEPTQ | 1 | 0 | 0 | |
| DEPTQ+ | 135° | −0.71 | −1 | −1.06 |
| DEPTQ | 0.71 | −1 | 1.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bigler, P.; Melendez, C.; Furrer, J. The DEPTQ+ Experiment: Leveling the DEPT Signal Intensities and Clean Spectral Editing for Determining CHn Multiplicities. Molecules 2021, 26, 3490. https://doi.org/10.3390/molecules26123490
Bigler P, Melendez C, Furrer J. The DEPTQ+ Experiment: Leveling the DEPT Signal Intensities and Clean Spectral Editing for Determining CHn Multiplicities. Molecules. 2021; 26(12):3490. https://doi.org/10.3390/molecules26123490
Chicago/Turabian StyleBigler, Peter, Camilo Melendez, and Julien Furrer. 2021. "The DEPTQ+ Experiment: Leveling the DEPT Signal Intensities and Clean Spectral Editing for Determining CHn Multiplicities" Molecules 26, no. 12: 3490. https://doi.org/10.3390/molecules26123490
APA StyleBigler, P., Melendez, C., & Furrer, J. (2021). The DEPTQ+ Experiment: Leveling the DEPT Signal Intensities and Clean Spectral Editing for Determining CHn Multiplicities. Molecules, 26(12), 3490. https://doi.org/10.3390/molecules26123490







