Compositional Changes in Grapes and Leaves as a Consequence of Smoke Exposure of Vineyards from Multiple Bushfires across a Ripening Season
Abstract
1. Introduction
2. Results and Discussion
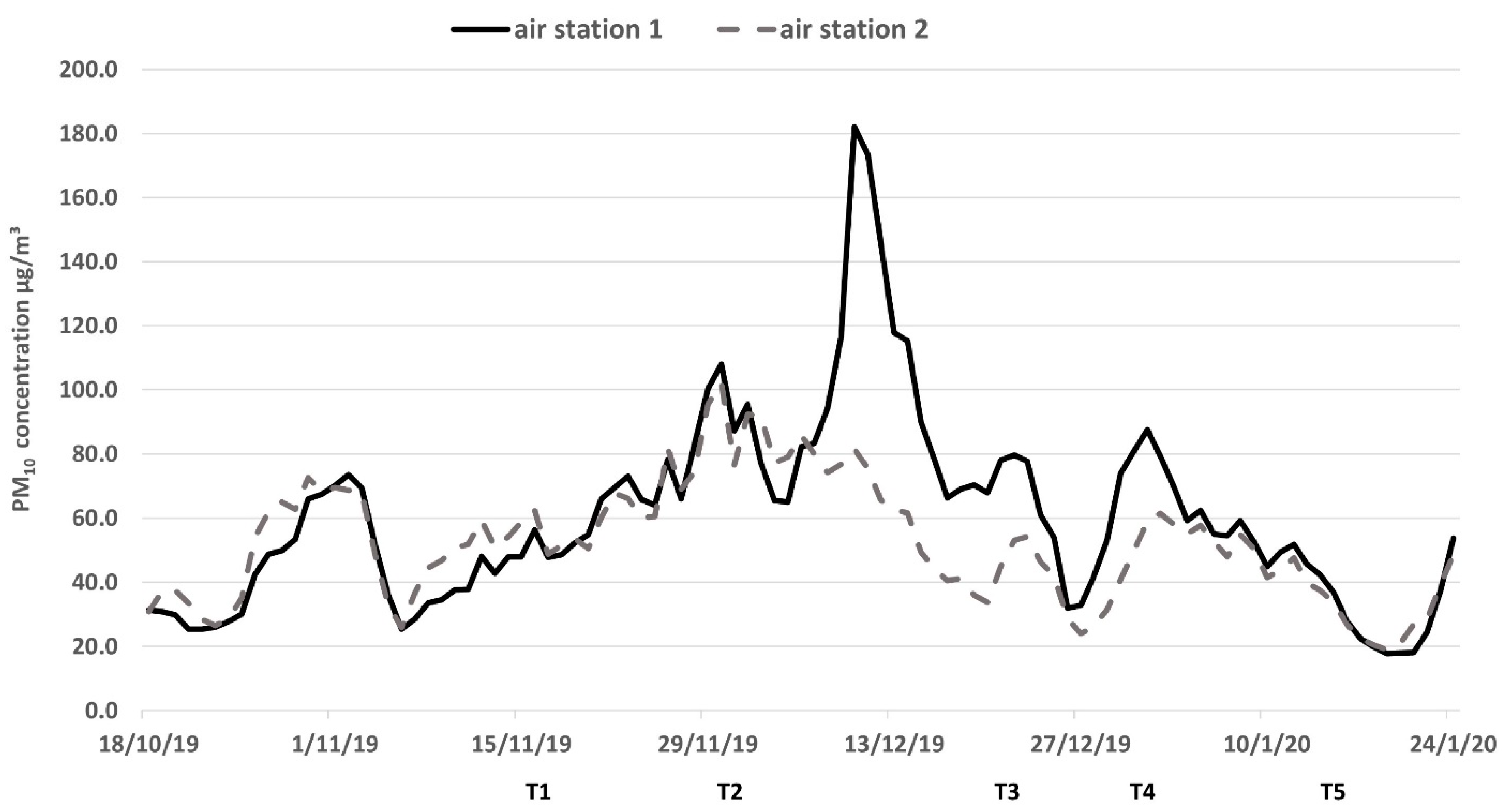
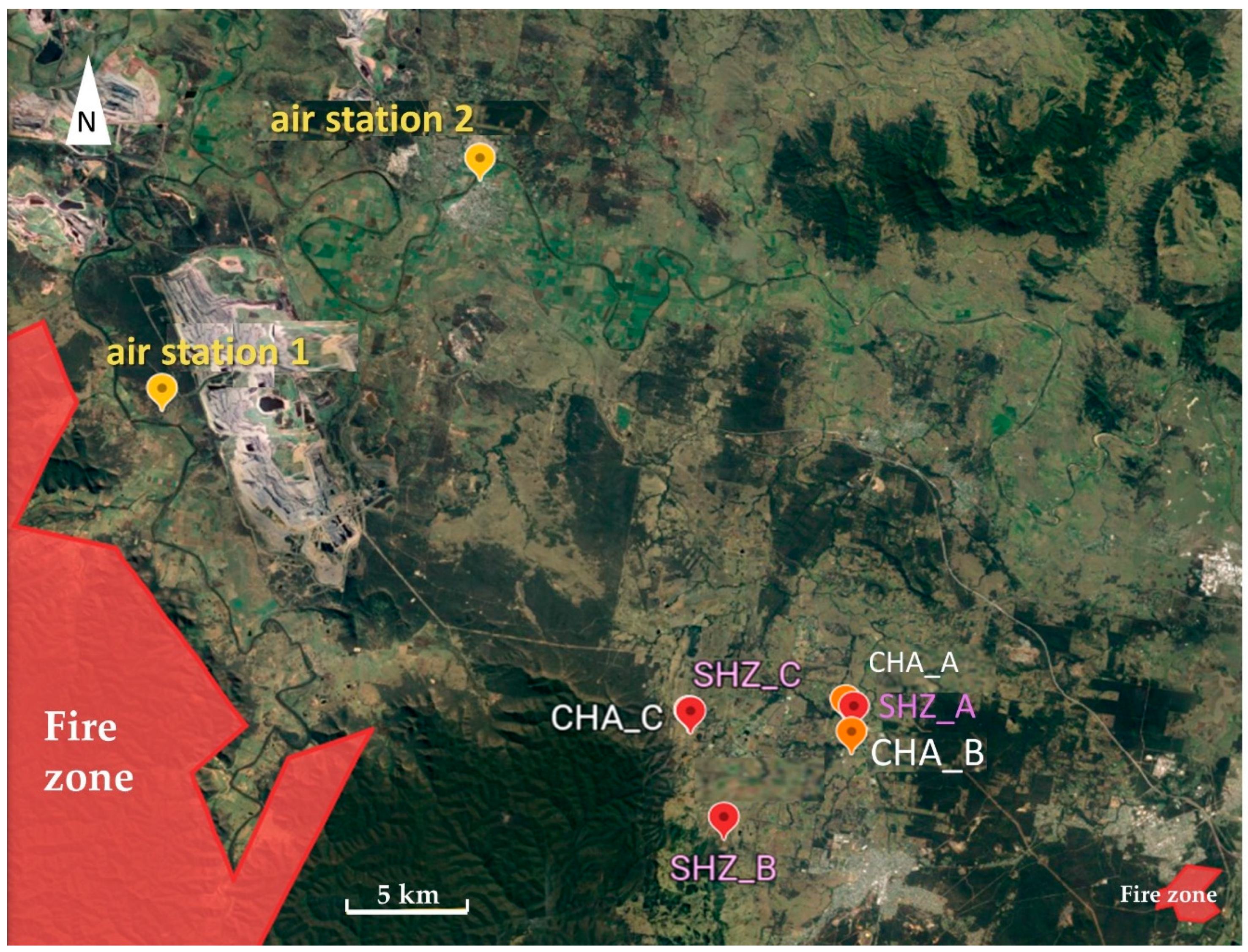
2.1. Smoke Exposure of Vineyards
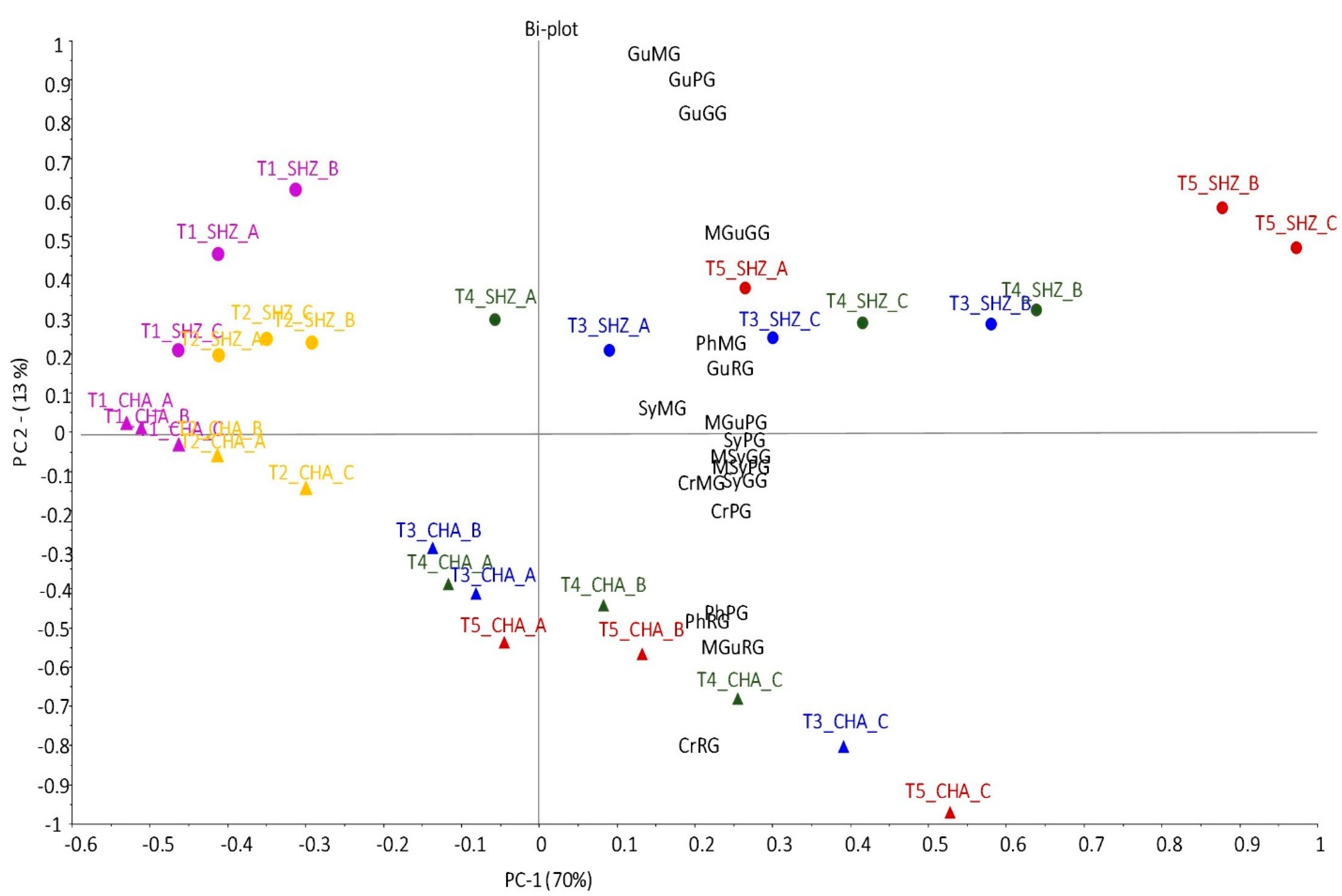
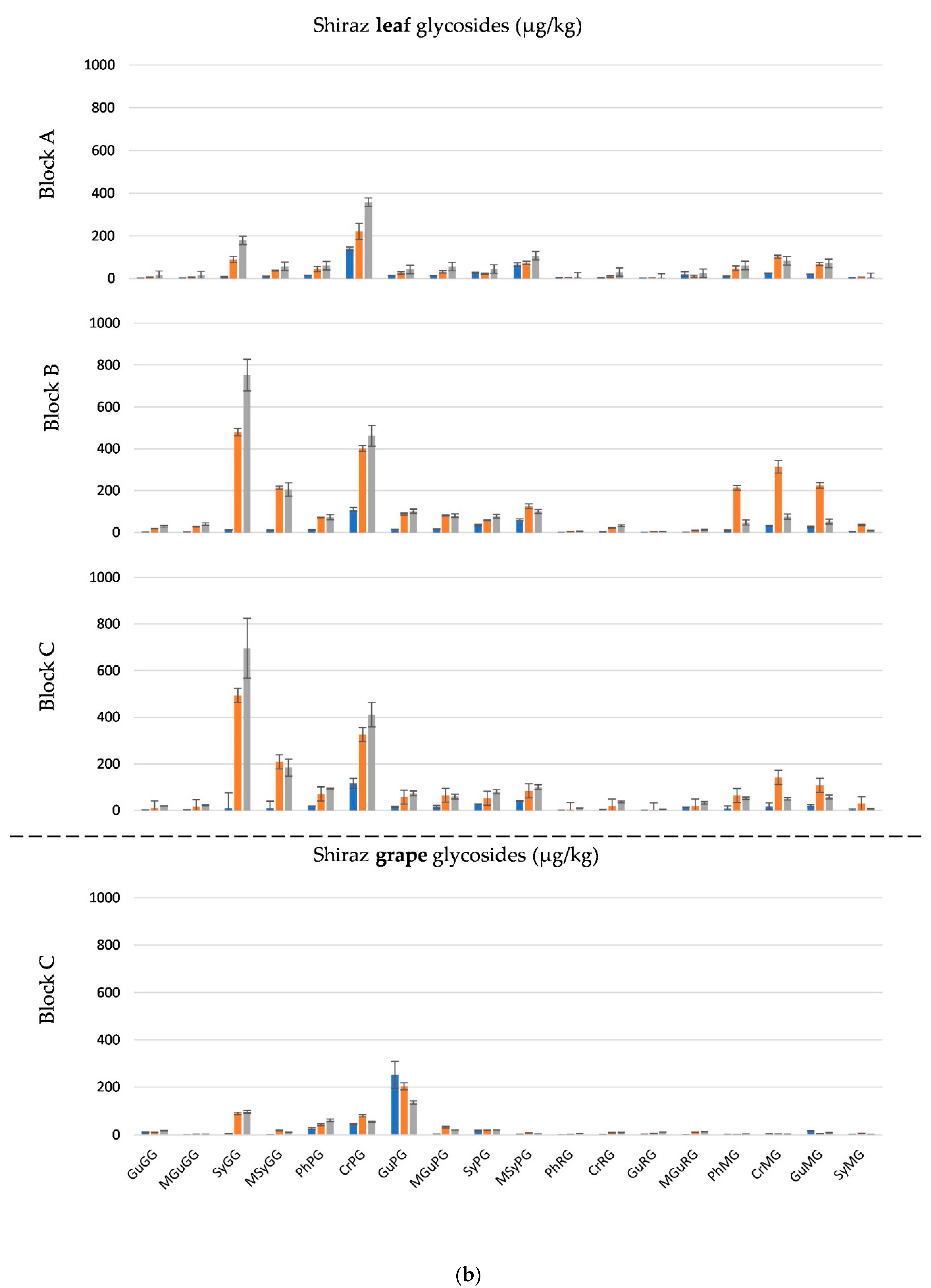
2.2. Smoke Exposure Markers in Grapes
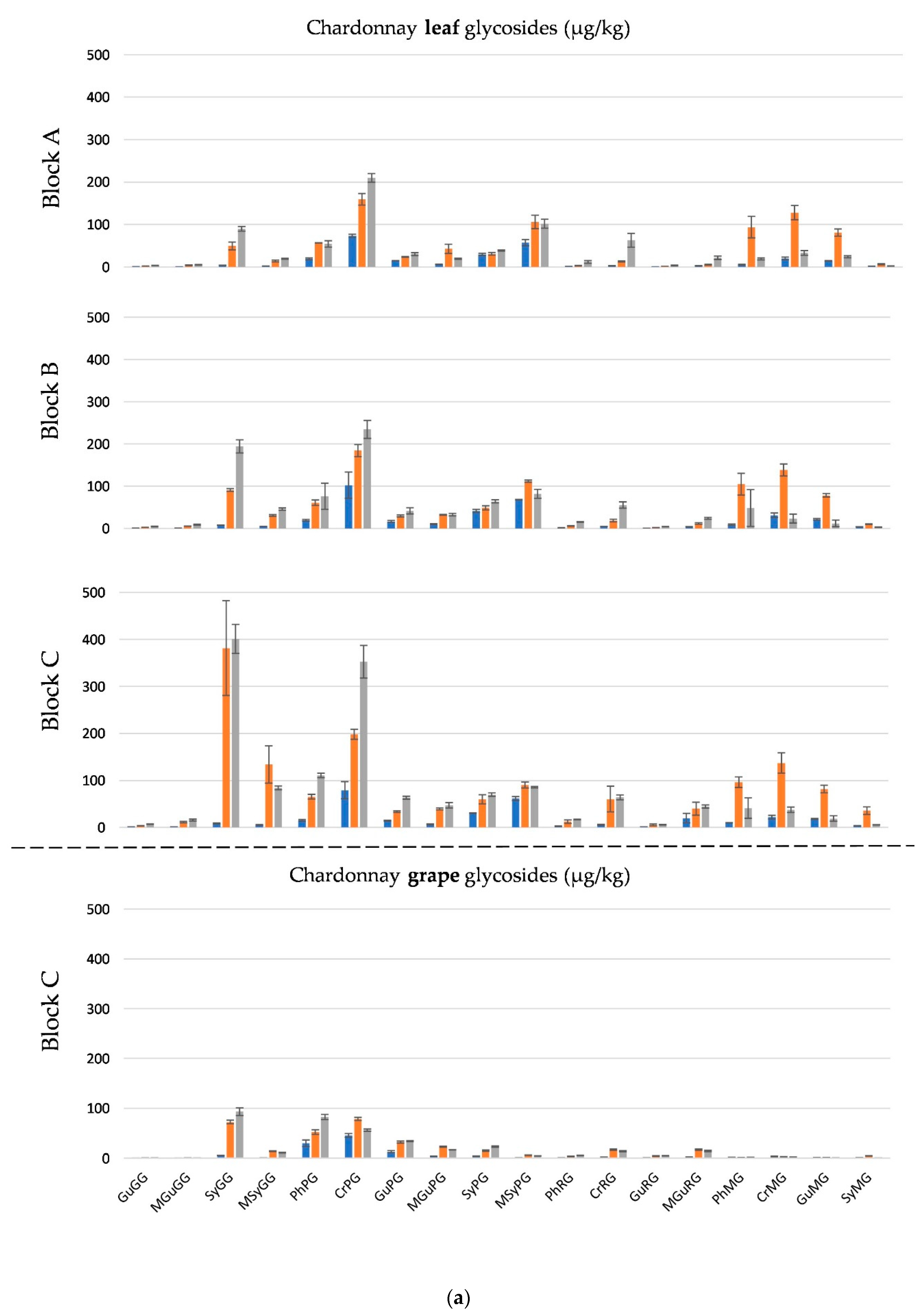
2.3. Smoke Exposure Markers in Leaves
3. Materials and Methods
3.1. Grape Berry and Leaf Sampling
3.2. Sample Preparation
3.3. Chemical Analysis
3.4. Data Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Krstic, M.P.; Johnson, D.L.; Herderich, M.J. Review of smoke taint in wine: Smoke-derived volatile phenols and their glycosidic metabolites in grapes and vines as biomarkers for smoke exposure and their role in the sensory perception of smoke taint. Aust. J. Grape Wine Res. 2015, 21, 537–553. [Google Scholar] [CrossRef]
- Høj, P.; Pretorius, I.; Blair, R.E. The Australian Wine Research Institute, Annual Report; The Australian Wine Research Institute: Adelaide, Australia, 2003. [Google Scholar]
- McArthur, A.G. Weather and Grassland Fire Behaviour; Forestry and Timber Bureau: Canberra, Australia, 1966; Volume 100, p. 23. [Google Scholar]
- Dowdy, A.J.; Ye, H.; Pepler, A.; Thatcher, M.; Osbrough, S.L.; Evans, J.P.; Di Virgilio, G.; McCarthy, N. Future changes in extreme weather and pyroconvection risk factors for Australian wildfires. Sci. Rep. 2019, 9, 10073. [Google Scholar] [CrossRef]
- Hennessy, K.; Lucas, C.; Nicholls, N.; Bathols, J.; Suppiah, R.; Ricketts, J. Climate Change Impacts on Fire-Weather in South-East Australia; CSIRO Marine and Atmospheric Research: Aspendale, Australia, 2006. [Google Scholar]
- Dowdy, A.J. Climatological variability of fire weather in Australia. J. Appl. Meteorol. Clim. 2018, 57, 221–234. [Google Scholar] [CrossRef]
- Bureau of Meteorology. Special Climate Statement 72—Dangerous Bushfire Weather in Spring 2019; Australian Government Bureau of Meteorology: Melbourn, Australia, 2019. [Google Scholar]
- Kennison, K.R.; Wilkinson, K.L.; Williams, H.G.; Smith, J.H.; Gibberd, M.R. Smoke-derived taint in wine: Effect of postharvest smoke exposure of grapes on the chemical composition and sensory characteristics of wine. J. Agric. Food Chem. 2007, 55, 10897–10901. [Google Scholar] [CrossRef] [PubMed]
- Parker, M.; Osidacz, P.; Baldock, G.A.; Hayasaka, Y.; Black, C.A.; Pardon, K.H.; Jeffery, D.W.; Geue, J.P.; Herderich, M.J.; Francis, I.L. Contribution of several volatile phenols and their glycoconjugates to smoke-related sensory properties of red wine. J. Agric. Food Chem. 2012, 60, 2629–2637. [Google Scholar] [CrossRef] [PubMed]
- Hayasaka, Y.; Baldock, G.A.; Pardon, K.H.; Jeffery, D.W.; Herderich, M.J. Investigation into the formation of guaiacol conjugates in berries and leaves of grapevine Vitis vinifera L. Cv. Cabernet Sauvignon using stable isotope tracers combined with HPLC-MS and MS/MS analysis. J. Agric. Food Chem. 2010, 58, 2076–2081. [Google Scholar] [CrossRef]
- Hayasaka, Y.; Parker, M.; Baldock, G.A.; Pardon, K.H.; Black, C.A.; Jeffery, D.W.; Herderich, M.J. Assessing the impact of smoke exposure in grapes: Development and validation of a HPLC-MS/MS method for the quantitative analysis of smoke-derived phenolic glycosides in grapes and wine. J. Agric. Food Chem. 2013, 61, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Kennison, K.R.; Wilkinson, K.L.; Pollnitz, A.P.; Williams, H.G.; Gibberd, M.R. Effect of timing and duration of grapevine exposure to smoke on the composition and sensory properties of wine. Aust. J. Grape Wine Res. 2009, 15, 228–237. [Google Scholar] [CrossRef]
- Kelly, D.; Zerihun, A.; Hayasaka, Y.; Gibberd, M. Winemaking practice affects the extraction of smoke-borne phenols from grapes into wines. Aust. J. Grape Wine Res. 2014, 20, 386–393. [Google Scholar] [CrossRef]
- Ristic, R.; Fudge, A.L.; Pinchbeck, K.A.; De Bei, R.; Fuentes, S.; Hayasaka, Y.; Tyerman, S.D.; Wilkinson, K.L. Impact of grapevine exposure to smoke on vine physiology and the composition and sensory properties of wine. Theor. Exp. Plant Phys. 2016, 28, 67–83. [Google Scholar] [CrossRef]
- Szeto, C.; Ristic, R.; Capone, D.; Puglisi, C.; Pagay, V.; Culbert, J.; Jiang, W.; Herderich, M.; Tuke, J.; Wilkinson, K. Uptake and glycosylation of smoke-derived volatile phenols by Cabernet Sauvignon grapes and their subsequent fate during winemaking. Molecules 2020, 25, 3720. [Google Scholar] [CrossRef]
- Hayasaka, Y.; Baldock, G.A.; Parker, M.; Pardon, K.H.; Black, C.A.; Herderich, M.J.; Jeffery, D.W. Glycosylation of smoke-derived volatile phenols in grapes as a consequence of grapevine exposure to bushfire smoke. J. Agric. Food Chem. 2010, 58, 10989–10998. [Google Scholar] [CrossRef]
- Hayasaka, Y.; Dungey, K.A.; Baldock, G.A.; Kennison, K.R.; Wilkinson, K.L. Identification of a β-D-glucopyranoside precursor to guaiacol in grape juice following grapevine exposure to smoke. Anal. Chim. Acta 2010, 660, 143–148. [Google Scholar] [CrossRef]
- Caffrey, A.; Lerno, L.; Rumbaugh, A.; Girardello, R.; Zweigenbaum, J.; Oberholster, A.; Ebeler, S.E. Changes in smoke-taint volatile-phenol glycosides in wildfire smoke-exposed Cabernet Sauvignon grapes throughout winemaking. Am. J. Enol. Vitic. 2019, 70, 373–381. [Google Scholar] [CrossRef]
- Noestheden, M.; Dennis, E.G.; Romero-Montalvo, E.; DiLabio, G.A.; Zandberg, W.F. Detailed characterization of glycosylated sensory-active volatile phenols in smoke-exposed grapes and wine. Food Chem. 2018, 259, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Coombe, B.G. Growth stages of the grapevine: Adoption of a system for identifying grapevine growth stages. Aust. J. Grape Wine Res. 1995, 1, 100–110. [Google Scholar] [CrossRef]
- Kennison, K.R.; Wilkinson, K.L.; Pollnitz, A.P.; Williams, H.G.; Gibberd, M.R. Effect of smoke application to field-grown Merlot grapevines at key phenological growth stages on wine sensory and chemical properties. Aust. J. Grape Wine Res. 2011, 17, S5–S12. [Google Scholar] [CrossRef]
- Sheppard, S.I.; Dhesi, M.K.; Eggers, N.J. Effect of pre- and postveraison smoke exposure on guaiacol and 4-methylguaiacol concentration in mature grapes. Am. J. Enol. Vitic. 2009, 60, 98–103. [Google Scholar]
- Härtl, K.; Huang, F.-C.; Giri, A.P.; Franz-Oberdorf, K.; Frotscher, J.; Shao, Y.; Hoffmann, T.; Schwab, W. Glucosylation of smoke-derived volatiles in grapevine (Vitis vinifera) is catalyzed by a promiscuous resveratrol/guaiacol glucosyltransferase. J. Agric. Food Chem. 2017, 65, 5681–5689. [Google Scholar] [CrossRef] [PubMed]
- Kennison, K.R. Smoke Derived Taint in Grapes and Wine. Ph.D. Thesis, Curtin University, Perth, Australia, 2011. [Google Scholar]
- Brodison, K. Bulletin 4847: Effect of Smoke in Grape and Wine Production; Department of Agriculture and Food Western Australia: Perth, Australia, 2013. [Google Scholar]
- Pardo-Garcia, A.I.; Wilkinson, K.L.; Culbert, J.A.; Lloyd, N.D.R.; Alonso, G.L.; Salinas, M.R. Accumulation of guaiacol glycoconjugates in fruit, leaves and shoots of Vitis vinifera cv. Monastrell following foliar applications of guaiacol or oak extract to grapevines. Food Chem. 2017, 217, 782–789. [Google Scholar] [CrossRef]
- How the ABC Located The Tree Which Started the Gospers Mountain Bushfire and the Sydney ‘Mega-Blaze’. Available online: https://www.abc.net.au/news/2020-08-02/how-we-found-tree-which-sparked-the-sydney-mega-blaze-bushfire/12510960 (accessed on 26 March 2021).
- Cessnock City Council. Hunter Valley’s Bushfire Devastated Communityes; Cessnock City Council: Cessnock, Australia, 2020; pp. 4–5. [Google Scholar]
- Upper Hunter Air Quality Maps. Available online: https://www.dpie.nsw.gov.au/air-quality/air-quality-maps/live-air-quality-data-upper-hunter (accessed on 6 January 2021).
- National Environment Protection (Ambient Air Quality) Measure. Available online: https://www.environment.gov.au/protection/air-quality/ambient-air-quality-nepm (accessed on 15 January 2021).
- Google Earth. Available online: https://www.google.com/earth/ (accessed on 4 January 2021).
- Kennison, K.R.; Gibberd, M.R.; Pollnitz, A.P.; Wilkinson, K.L. Smoke-derived taint in wine: The release of smoke-derived volatile phenols during fermentation of Merlot juice following grapevine exposure to smoke. J. Agric. Food Chem. 2008, 56, 7379–7383. [Google Scholar] [CrossRef]
- Van der Hulst, L.; Munguia, P.; Culbert, J.A.; Ford, C.M.; Burton, R.A.; Wilkinson, K.L. Accumulation of volatile phenol glycoconjugates in grapes following grapevine exposure to smoke and potential mitigation of smoke taint by foliar application of kaolin. Planta 2019, 249, 941–952. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.; Zerihun, A.; Singh, D.P.; Vitzthum von Eckstaedt, C.; Gibberd, M.; Grice, K.; Downey, M. Exposure of grapes to smoke of vegetation with varying lignin composition and accretion of lignin derived putative smoke taint compounds in wine. Food Chem. 2012, 135, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Bönisch, F.; Frotscher, J.; Stanitzek, S.; Rühl, E.; Wüst, M.; Bitz, O.; Schwab, W. A UDP-glucose: Monoterpenol glucosyltransferase adds to the chemical diversity of the grapevine metabolome. Plant Physiol. 2014, 165, 561–581. [Google Scholar] [CrossRef] [PubMed]
- Pollnitz, A.P.; Pardon, K.H.; Sykes, M.; Sefton, M.A. The effects of sample preparation and gas chromatograph injection techniques on the accuracy of measuring guaiacol, 4-methylguaiacol and other volatile oak compounds in oak extracts by stable isotope dilution analyses. J. Agric. Food Chem. 2004, 52, 3244–3252. [Google Scholar] [CrossRef] [PubMed]
| Time Point | Sampling Date | E-L Stage | |||||
|---|---|---|---|---|---|---|---|
| CHA_A | CHA_B | CHA_C | SHZ_A | SHZ_B | SHZ_C | ||
| T1 | 15/11/2019 | 30–31 | 27–32 | 31 | 27–29 | 27–29 | 27–29 |
| T2 | 29/11/2019 | 32 | 32 | 32 | 32 | 31 | 32 |
| T3 | 16/12/2019 | 33 | 33 | 33 | 33 | 33 | 33 |
| T4 | 28/12/2019 | 35 | 35 | 35 | 35 | 35 | 35 |
| T5 | 10/01/2020 | 38 | 38 | 38 | 37–38 | 37–38 | 37–38 |
| Variety | Block | Time Point | ||||
|---|---|---|---|---|---|---|
| T1 | T2 | T3 | T4 | T5 | ||
| Chardonnay | A | 0.23 | 0.42 | 0.51 | 0.84 | 1.03 |
| B | 0.20 | 0.35 | 0.35 | 0.64 | 0.73 | |
| C | 0.30 | 0.40 | 0.66 | 0.78 | 0.79 | |
| Shiraz | A | 0.12 | 0.40 | 0.69 | 0.82 | 1.14 |
| B | 0.16 | 0.32 | 0.48 | 0.64 | 0.82 | |
| C | 0.17 | 0.32 | 0.45 | 0.72 | 0.80 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, W.; Parker, M.; Hayasaka, Y.; Simos, C.; Herderich, M. Compositional Changes in Grapes and Leaves as a Consequence of Smoke Exposure of Vineyards from Multiple Bushfires across a Ripening Season. Molecules 2021, 26, 3187. https://doi.org/10.3390/molecules26113187
Jiang W, Parker M, Hayasaka Y, Simos C, Herderich M. Compositional Changes in Grapes and Leaves as a Consequence of Smoke Exposure of Vineyards from Multiple Bushfires across a Ripening Season. Molecules. 2021; 26(11):3187. https://doi.org/10.3390/molecules26113187
Chicago/Turabian StyleJiang, WenWen, Mango Parker, Yoji Hayasaka, Con Simos, and Markus Herderich. 2021. "Compositional Changes in Grapes and Leaves as a Consequence of Smoke Exposure of Vineyards from Multiple Bushfires across a Ripening Season" Molecules 26, no. 11: 3187. https://doi.org/10.3390/molecules26113187
APA StyleJiang, W., Parker, M., Hayasaka, Y., Simos, C., & Herderich, M. (2021). Compositional Changes in Grapes and Leaves as a Consequence of Smoke Exposure of Vineyards from Multiple Bushfires across a Ripening Season. Molecules, 26(11), 3187. https://doi.org/10.3390/molecules26113187






