Camu-Camu Fruit Extract Inhibits Oxidative Stress and Inflammatory Responses by Regulating NFAT and Nrf2 Signaling Pathways in High Glucose-Induced Human Keratinocytes
Abstract
1. Introduction
2. Results
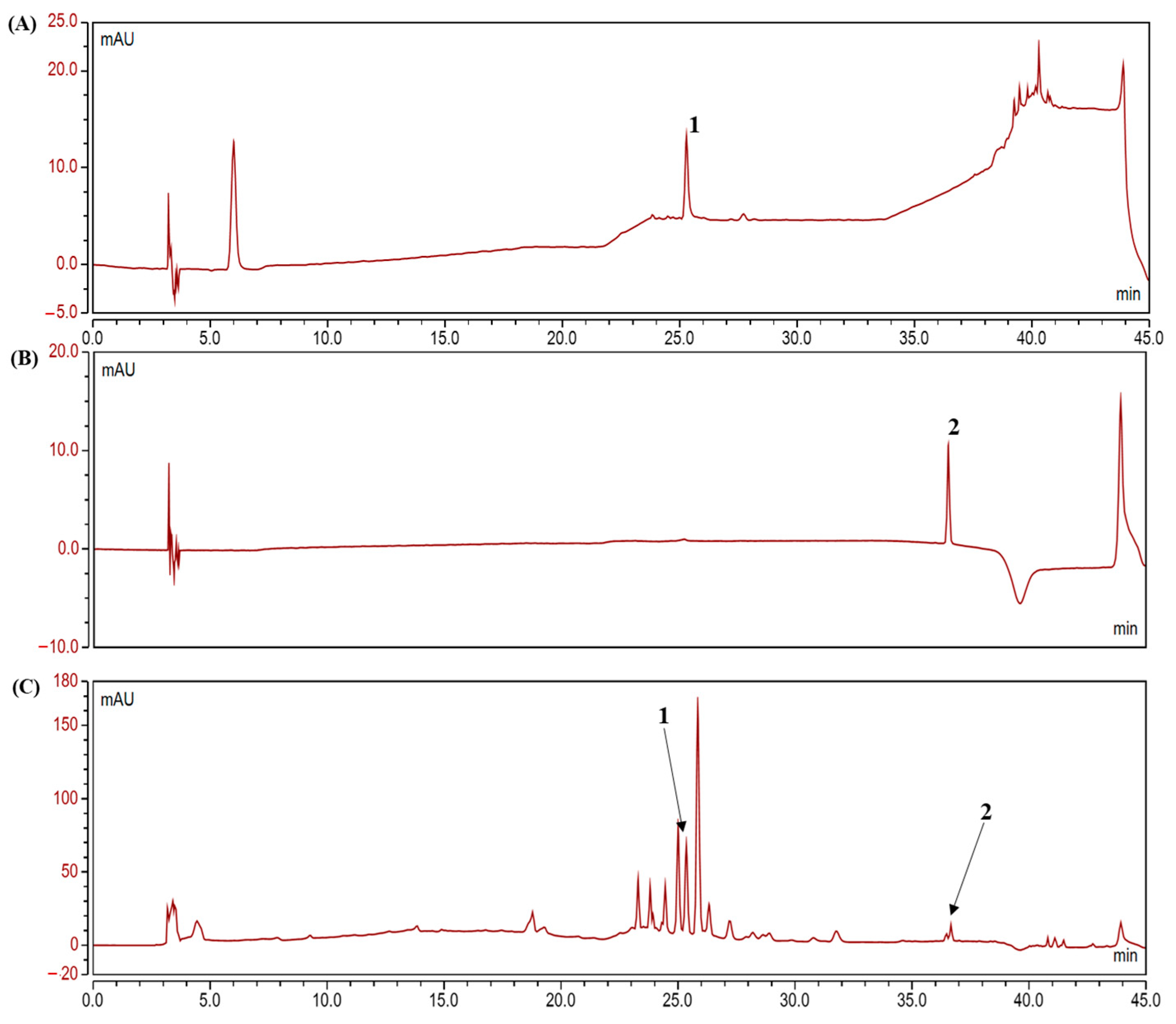
2.1. Analysis of Polyphenolic Compounds in the Camu-Camu Fruit Extract
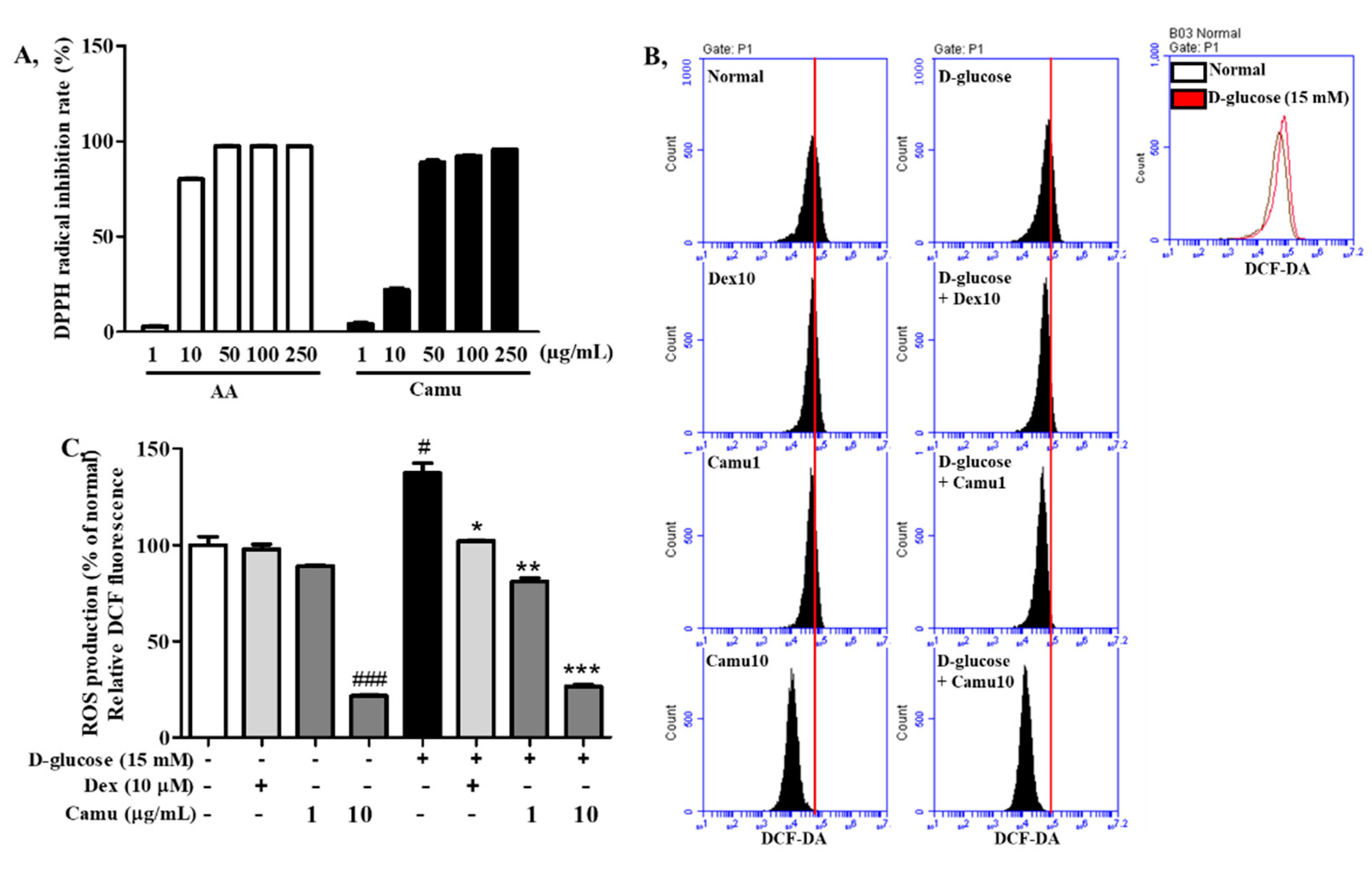
2.2. Antioxidative Activities of the Camu-Camu Fruit Extract
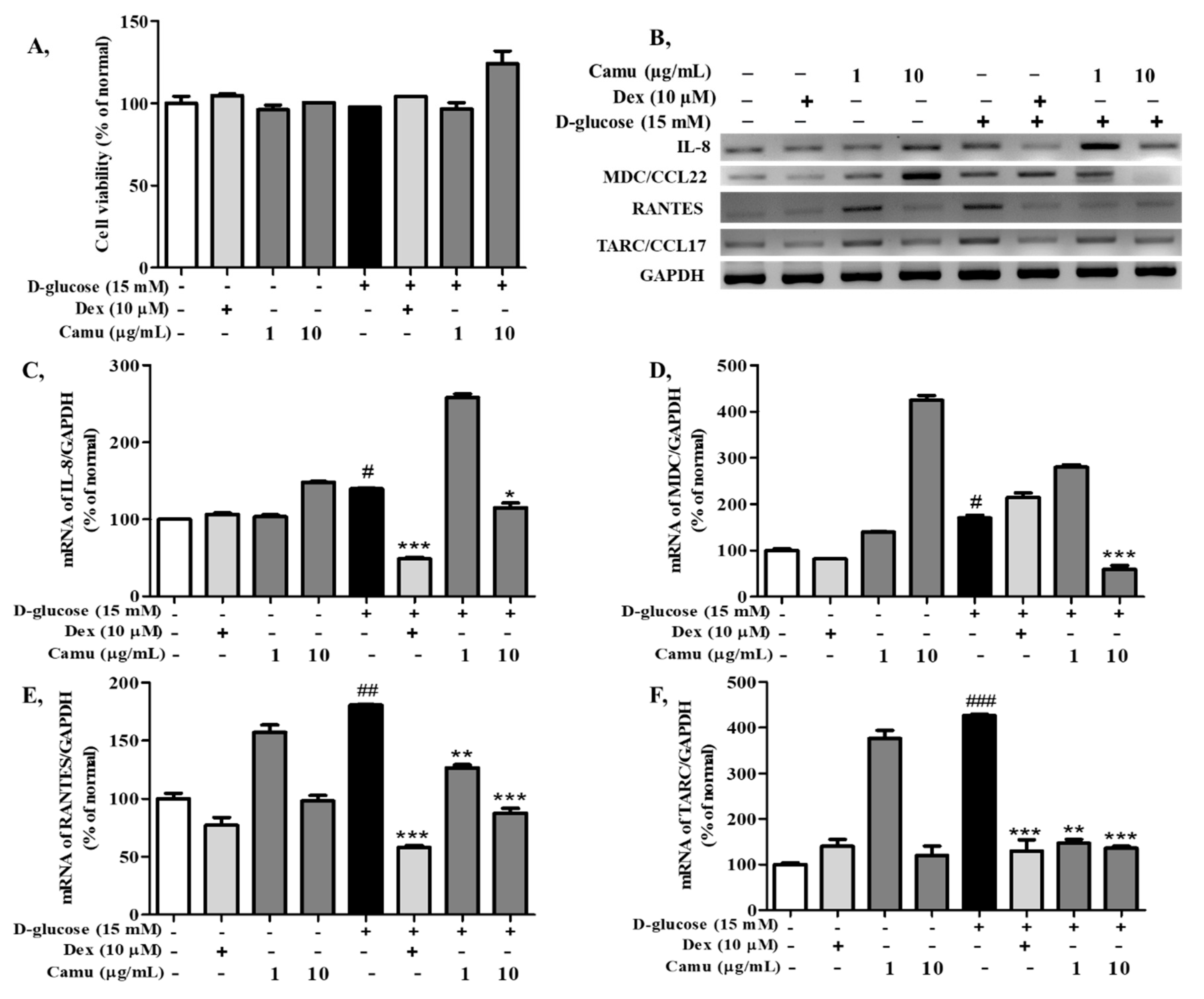
2.3. Effects of the Camu-Camu Fruit Extract on Cell Viability and mRNA Expression of Proinflammatory Cytokines and Chemokines in High Glucose-Induced HaCaT Cells
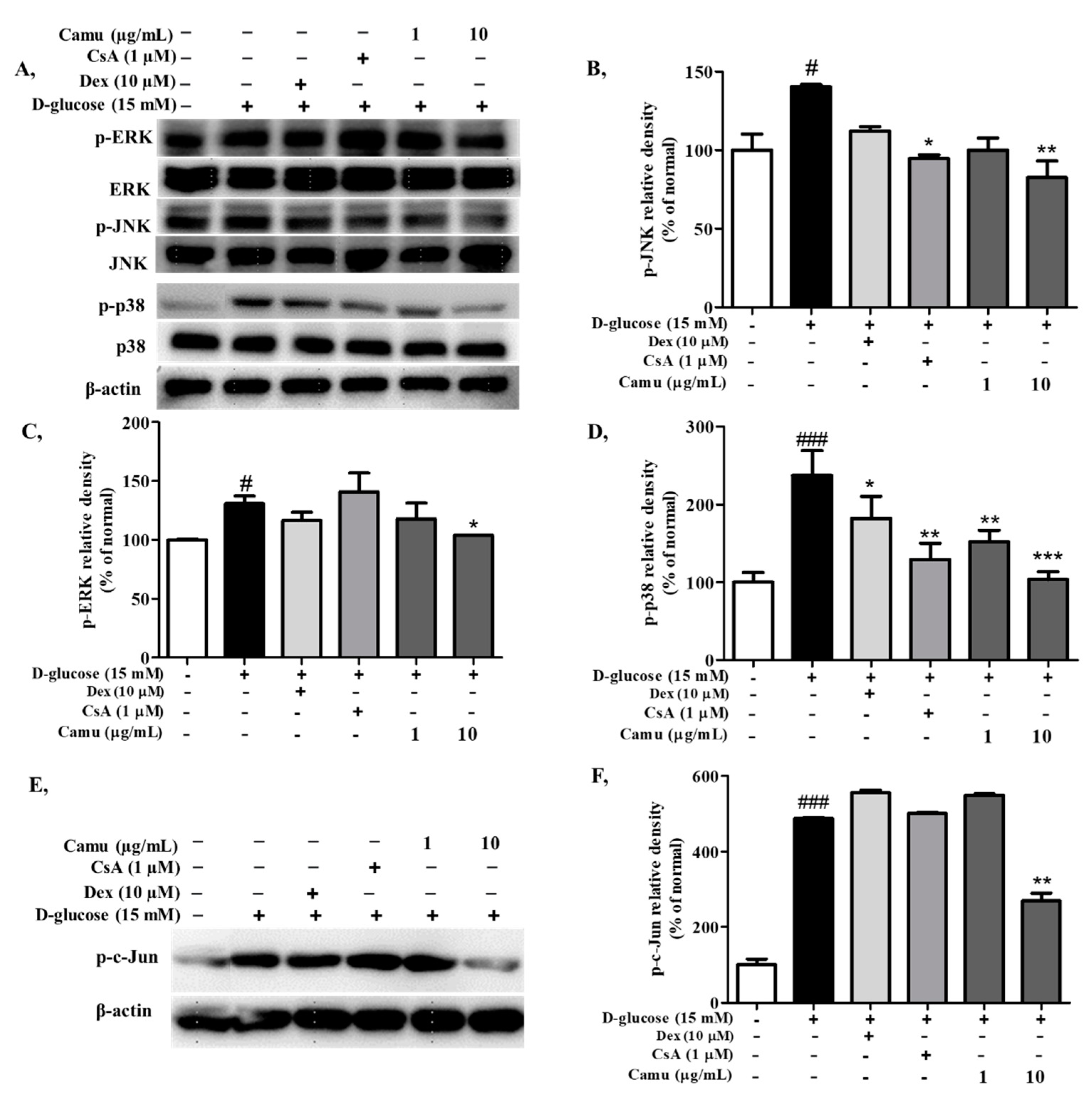
2.4. Inhibitory Effects of the Camu-Camu Fruit Extract on MAPKs and AP-1 Activation in High-Glucose-Induced HaCaT Cells
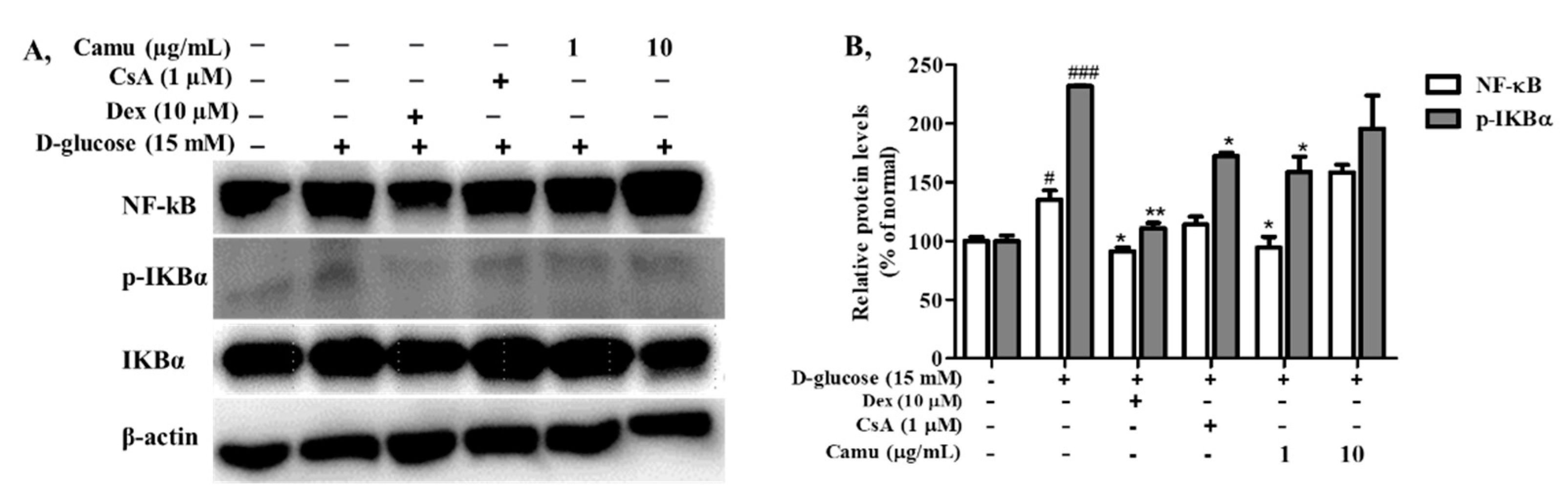
2.5. Inhibitory Effects of the Camu-Camu Fruit Extract on NF-κB in High Glucose-Induced HaCaT Cells
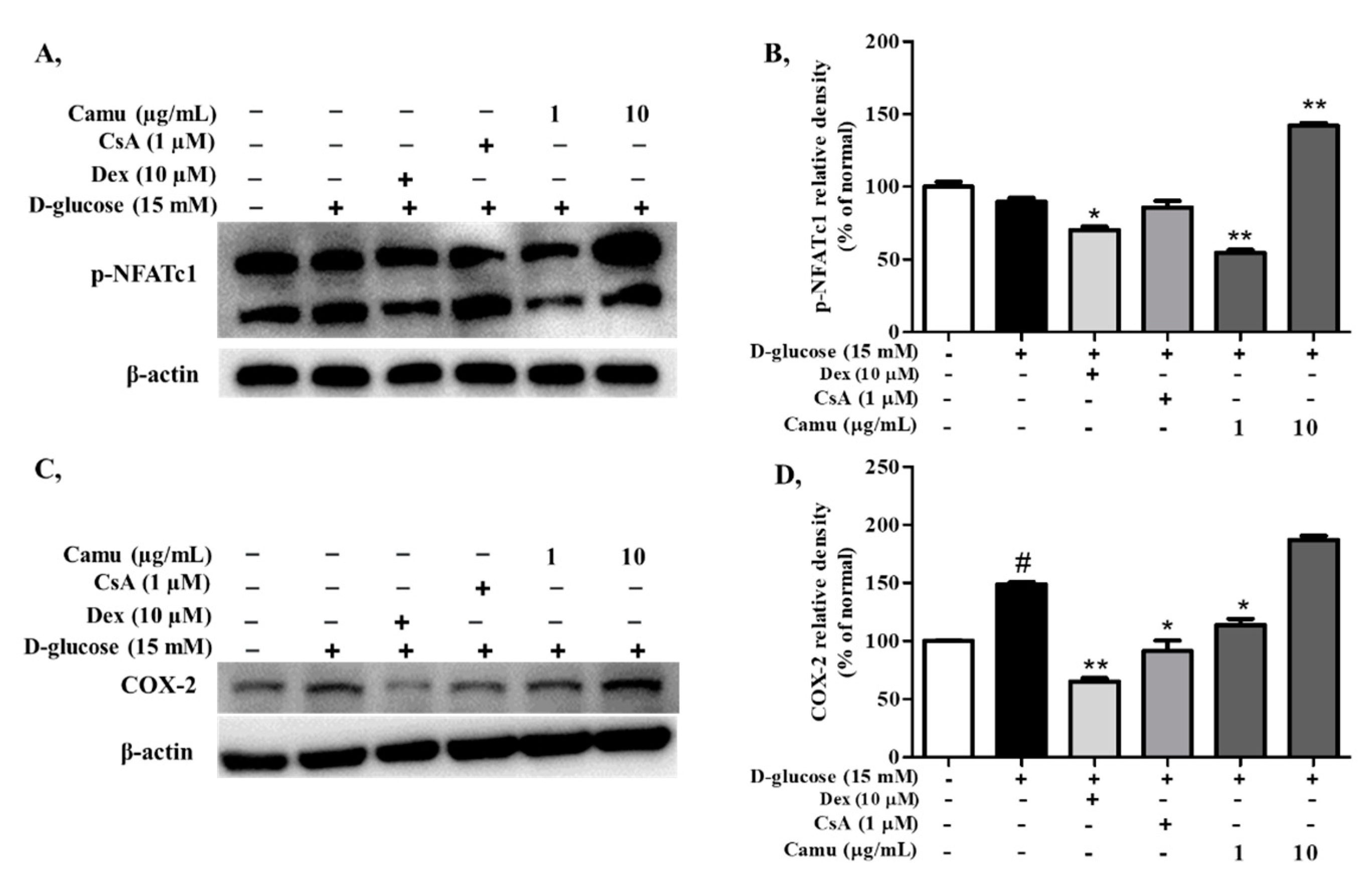
2.6. Inhibitory Effects of the Camu-Camu Fruit Extract on p-NFATc1 in High Glucose-Induced HaCaT Cells
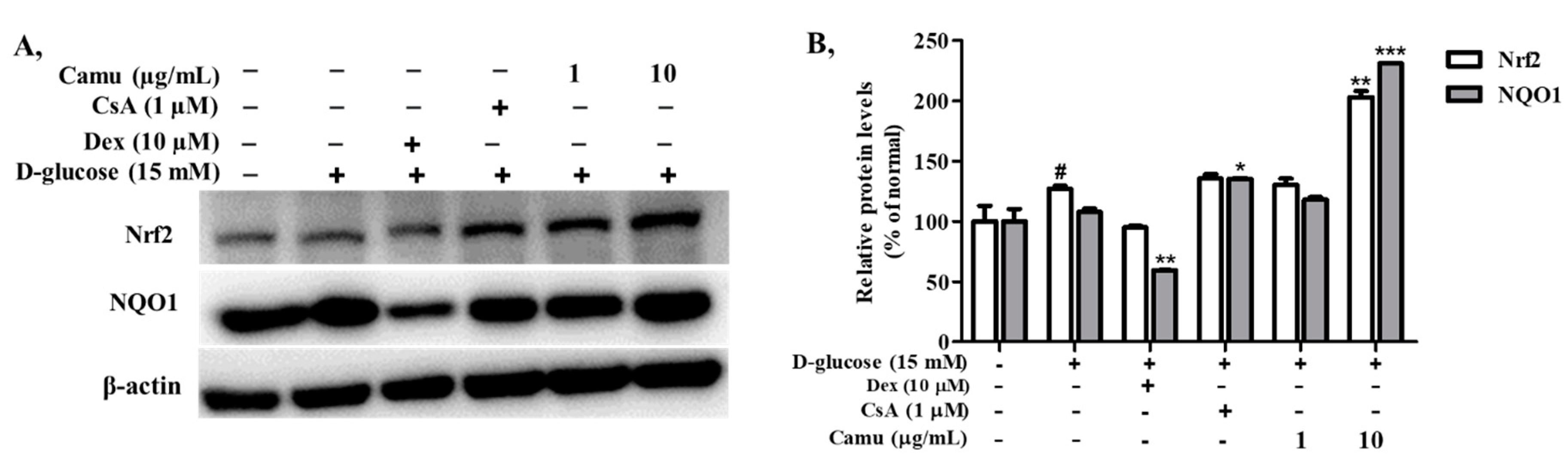
2.7. Inhibitory Effects of the Camu-Camu Fruit Extract in Nrf2 in High Glucose-Induced HaCaT Cells
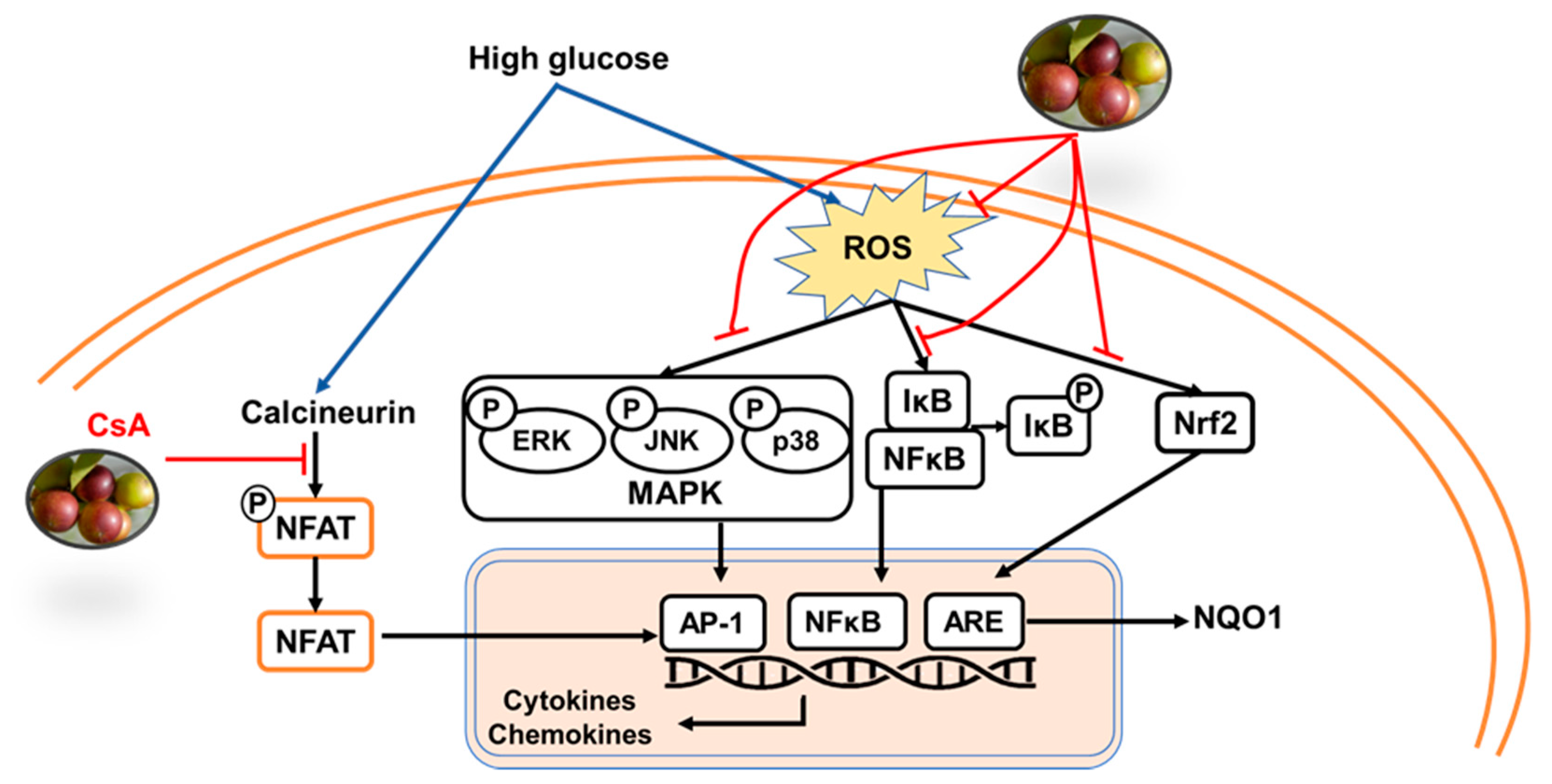
3. Discussion
4. Materials and Methods
4.1. Sample Preparation
4.2. High-Performance Liquid Chromatography Analysis
4.3. DPPH Scavenging Activity
4.4. Cell Culture, Camu-Camu Treatment, and Stimulation
4.5. Cell Viability Assay
4.6. Measurement of ROS Generation
4.7. Reverse Transcription-Polymerase Chain Reaction
4.8. Western Blot Analysis
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Abbreviations
| AP-1 | activator protein-1 |
| ARE | antioxidant responsive element |
| DCFH-DA | 2′,7′-dichlorofluorescin diacetate |
| DPPH | 1,1-diphenyl-2-picrylhydrazyl |
| ERK | extracellular signal-regulated kinase |
| HaCaT | human skin keratinocyte cell line |
| HO-1 | heme oxygenase 1 |
| IL-8 | interleukin 8 |
| JNK | c-Jun N-terminal kinase |
| MAPK | mitogen-activated protein kinase |
| MDC | human macrophage-derived chemokine |
| NFAT | nuclear factor of activated T cells |
| NF-κB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| NQO1 | NAD(P)H: quinone oxidoreductase1 |
| Nrf2 | nuclear factor E2-related factor 2 |
| RANTES | regulated upon activation, normal T cell expressed, and secreted |
| ROS | reactive oxygen species |
| TARC | thymus- and activation-regulated chemokine |
References
- Rufino, M.; Socorro, M.D.; Alves, R.E.; de Brito, E.S.; Pérez-Jiménez, J.; Saura-Calixto, F.; Mancini-Filho, J. Bioactive Compounds and Antioxidant Capacities of 18 Non-Traditional Tropical Fruits from Brazil. Food Chem. 2010, 121, 996–1002. [Google Scholar] [CrossRef]
- Castro, J.C.; Maddox, J.D.; Imán, S.A. Camu-camu—Myrciaria dubia (Kunth) McVaugh. In Exotic Fruits; Elsevier: Amsterdam, The Netherlands, 2018; pp. 97–105. [Google Scholar]
- Langley, P.C.; Pergolizzi, J.V.; Taylor, R.; Ridgway, C. Antioxidant and Associated Capacities of Camu Camu (Myrciaria dubia): A Systematic Review. J. Altern. Complement. Med. 2015, 21, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Azevêdo, J.C.S.; Borges, K.C.; Genovese, M.I.; Correia, R.T.P.; Vattem, D.A. Neuroprotective Effects of Dried Camu-Camu (Myrciaria dubia HBK McVaugh) Residue in C. Elegans. Food Res. Int. 2015, 73, 135–141. [Google Scholar] [CrossRef]
- Fujita, A.; Sarkar, D.; Wu, S.; Kennelly, E.; Shetty, K.; Genovese, M.I. Evaluation of Phenolic-Linked Bioactives of Camu-Camu (Myrciaria dubia Mc. Vaugh) for Antihyperglycemia, Antihypertension, Antimicrobial Properties and Cellular Rejuvenation. Food Res. Int. 2015, 77, 194–203. [Google Scholar] [CrossRef]
- Fidelis, M.; do Carmo, M.A.V.; da Cruz, T.M.; Azevedo, L.; Myoda, T.; Miranda Furtado, M.; Boscacci Marques, M.; Sant’Ana, A.S.; Inês Genovese, M.; Young Oh, W.; et al. Camu-Camu Seed (Myrciaria dubia)—From Side Stream to Anantioxidant, Antihyperglycemic, Antiproliferative, Antimicrobial, Antihemolytic, Anti-Inflammatory, and Antihypertensive Ingredient. Food Chem. 2020, 310, 125909. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, F.C.; Arruda, A.; Ledel, A.; Dauth, C.; Romão, N.F.; Viana, R.N.; de Barros Falcão Ferraz, A.; Picada, J.N.; Pereira, P. Antigenotoxic Effect of Acute, Subacute and Chronic Treatments with Amazonian Camu-Camu (Myrciaria dubia) Juice on Mice Blood Cells. Food Chem. Toxicol. 2012, 50, 2275–2281. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S. Oxidative Stress, Inflammation, and Disease. In Oxidative Stress and Biomaterials; Elsevier: Amsterdam, The Netherlands, 2016; pp. 35–58. [Google Scholar]
- Lin, X.; Huang, T. Oxidative Stress in Psoriasis and Potential Therapeutic Use of Antioxidants. Free. Radic. Res. 2016, 50, 585–595. [Google Scholar] [CrossRef]
- Ji, H.; Li, X.K. Oxidative Stress in Atopic Dermatitis. Oxid. Med. Cell. Longev. 2016, 2016, 2721469. [Google Scholar] [CrossRef]
- Umpierrez, G.E.; Hellman, R.; Korytkowski, M.T.; Kosiborod, M.; Maynard, G.A.; Montori, V.M.; Seley, J.J.; Van den Berghe, G. Endocrine Society Management of Hyperglycemia in Hospitalized Patients in Non-Critical Care Setting: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2012, 97, 16–38. [Google Scholar] [CrossRef] [PubMed]
- Mouri, M.; Badireddy, M. Hyperglycemia. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Shah, G.N.; Morofuji, Y.; Banks, W.A.; Price, T.O. High Glucose-Induced Mitochondrial Respiration and Reactive Oxygen Species in Mouse Cerebral Pericytes Is Reversed by Pharmacological Inhibition of Mitochondrial Carbonic Anhydrases: Implications for Cerebral Microvascular Disease in Diabetes. Biochem. Biophys. Res. Commun. 2013, 440, 354–358. [Google Scholar] [CrossRef]
- Dangwal, S.; Stratmann, B.; Bang, C.; Lorenzen, J.M.; Kumarswamy, R.; Fiedler, J.; Falk, C.S.; Scholz, C.J.; Thum, T.; Tschoepe, D. Impairment of Wound Healing in Patients with Type 2 Diabetes Mellitus Influences Circulating MicroRNA Patterns via Inflammatory Cytokines. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 1480–1488. [Google Scholar] [CrossRef]
- Yu, T.; Jhun, B.S.; Yoon, Y. High-Glucose Stimulation Increases Reactive Oxygen Species Production through the Calcium and Mitogen-Activated Protein Kinase-Mediated Activation of Mitochondrial Fission. Antioxid. Redox. Signal. 2011, 14, 425–437. [Google Scholar] [CrossRef]
- Macian, F. NFAT Proteins: Key Regulators of T-Cell Development and Function. Nat. Rev. Immunol. 2005, 5, 472–484. [Google Scholar] [CrossRef] [PubMed]
- Al-Daraji, W.I.; Grant, K.R.; Ryan, K.; Saxton, A.; Reynolds, N.J. Localization of Calcineurin/NFAT in Human Skin and Psoriasis and Inhibition of Calcineurin/NFAT Activation in Human Keratinocytes by Cyclosporin, A.J. Investig. Dermatol. 2002, 118, 779–788. [Google Scholar] [CrossRef]
- Zhou, P.; Sun, L.J.; Dötsch, V.; Wagner, G.; Verdine, G.L. Solution Structure of the Core NFATC1/DNA Complex. Cell 1998, 92, 687–696. [Google Scholar] [CrossRef]
- Khalaf, H.; Jass, J.; Olsson, P.-E. The Role of Calcium, NF-κB and NFAT in the Regulation of CXCL8 and IL-6 Expression in Jurkat T-Cells. Int. J. Biochem. Mol. Biol. 2013, 4, 150–156. [Google Scholar]
- Lee, S.I.; Yu, J.S. NFATc Mediates Lipopolysaccharide and Nicotine-Induced Expression of INOS and COX-2 in Human Periodontal Ligament Cells. J. Dent. Hyg. Sci. 2015, 15, 753–760. [Google Scholar] [CrossRef]
- Ma, Q. Role of Nrf2 in Oxidative Stress and Toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Jian, T.; Wu, Y.; Zuo, Y.; Li, J.; Lv, H.; Ma, L.; Ren, B.; Zhao, L.; Li, W.; et al. Ellagic Acid Ameliorates Oxidative Stress and Insulin Resistance in High Glucose-Treated HepG2 Cells via MiR-223/Keap1-Nrf2 Pathway. Biomed. Pharmacother. 2019, 110, 85–94. [Google Scholar] [CrossRef]
- Liao, H.; Zhang, N.; Meng, Y.; Feng, H.; Yang, J.; Li, W.; Chen, S.; Wu, H.; Deng, W.; Tang, Q. Myricetin Alleviates Pathological Cardiac Hypertrophy via TRAF6/TAK1/MAPK and Nrf2 Signaling Pathway. Oxid. Med. Cell. Longev. 2019, 2019, 1–14. [Google Scholar] [CrossRef]
- Schäfer, M.; Werner, S. Nrf2—A Regulator of Keratinocyte Redox Signaling. Free Radic. Biol. Med. 2015, 88, 243–252. [Google Scholar] [CrossRef]
- Aslan, A.; Gok, O.; Beyaz, S.; Ağca, C.A.; Erman, O.; Zerek, A. Ellagic Acid Prevents Kidney Injury and Oxidative Damage via Regulation of Nrf-2/NF-κB Signaling in Carbon Tetrachloride Induced Rats. Mol. Biol. Rep. 2020, 47, 7959–7970. [Google Scholar] [CrossRef]
- Rosillo, M.A.; Sánchez-Hidalgo, M.; Cárdeno, A.; Aparicio-Soto, M.; Sánchez-Fidalgo, S.; Villegas, I.; de la Lastra, C.A. Dietary Supplementation of an Ellagic Acid-Enriched Pomegranate Extract Attenuates Chronic Colonic Inflammation in Rats. Pharmacol. Res. 2012, 66, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Seung, N.K.; Kim, Y.J.; Kim, T.S.; Sohn, E.H. Study on the Immunomodulatory Effects of Ellagic Acid and Their Mechanisms Related to Toll-like Receptor 4 in Macrophages. Korean J. Plant Res. 2012, 25, 561–567. [Google Scholar]
- Choi, Y.H.; Yan, G.H. Ellagic Acid Attenuates Immunoglobulin E-Mediated Allergic Response in Mast Cells. Biol. Pharm. Bull. 2009, 32, 1118–1121. [Google Scholar] [CrossRef] [PubMed]
- Umesalma, S.; Sudhandiran, G. Ellagic Acid Prevents Rat Colon Carcinogenesis Induced by 1, 2 Dimethyl Hydrazine through Inhibition of AKT-Phosphoinositide-3 Kinase Pathway. Eur. J. Pharmacol. 2011, 660, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Devipriya, N.; Sudheer, A.R.; Menon, V.P. Dose-Response Effect of Ellagic Acid on Circulatory Antioxidants and Lipids during Alcohol-Induced Toxicity in Experimental Rats. Fundam. Clin. Pharmacol. 2007, 21, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.Y.; Choi, J.S.; Kang, S.W.; Lee, Y.J.; Park, J.; Kang, Y.H. Dietary Compound Ellagic Acid Alleviates Skin Wrinkle and Inflammation Induced by UV-B Irradiation. Exp. Dermatol. 2010, 19, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Rozentsvit, A.; Vinokur, K.; Samuel, S.; Li, Y.; Gerdes, A.M.; Carrillo-Sepulveda, M.A. Ellagic Acid Reduces High Glucose-Induced Vascular Oxidative Stress Through ERK1/2/NOX4 Signaling Pathway. Cell. Physiol. Biochem. 2017, 44, 1174–1187. [Google Scholar] [CrossRef]
- Mendes, L.F.; Gaspar, V.M.; Conde, T.A.; Mano, J.F.; Duarte, I.F. Flavonoid-Mediated Immunomodulation of Human Macrophages Involves Key Metabolites and Metabolic Pathways. Sci. Rep. 2019, 9, 14906. [Google Scholar] [CrossRef]
- Beken, B.; Serttas, R.; Yazicioglu, M.; Turkekul, K.; Erdogan, S. Quercetin Improves Inflammation, Oxidative Stress, and Impaired Wound Healing in Atopic Dermatitis Model of Human Keratinocytes. Pediatr. Allergy Immunol. Pulmonol. 2020, 33, 69–79. [Google Scholar] [CrossRef]
- Xu, D.; Hu, M.J.; Wang, Y.Q.; Cui, Y.L. Antioxidant Activities of Quercetin and Its Complexes for Medicinal Application. Molecules 2019, 24, 1123. [Google Scholar] [CrossRef] [PubMed]
- Shin, E.J.; Lee, J.S.; Hong, S.; Lim, T.G.; Byun, S. Quercetin Directly Targets JAK2 and PKCδ and Prevents UV-Induced Photoaging in Human Skin. Int. J. Mol. Sci. 2019, 20, 5262. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.J.; Kim, O.H.; Kim, Y.H.; Lim, J.H.; Kim, S.; Park, J.W.; Kwon, T.K. Quercetin Arrests G2/M Phase and Induces Caspase-Dependent Cell Death in U937 Cells. Cancer Lett. 2006, 240, 234–242. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB Signaling in Inflammation. Signal Transduct. Target. Ther. 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Oeckinghaus, A.; Ghosh, S. The NF-KappaB Family of Transcription Factors and Its Regulation. Cold Spring Harb. Perspect. Biol. 2009, 1, a000034. [Google Scholar] [CrossRef]
- Flockhart, R.J.; Diffey, B.L.; Farr, P.M.; Lloyd, J.; Reynolds, N.J. NFAT Regulates Induction of COX-2 and Apoptosis of Keratinocytes in Response to Ultraviolet Radiation Exposure. FASEB J. 2008, 22, 4218–4227. [Google Scholar] [CrossRef] [PubMed]
- Jaime, P.L.A.; do Amaral Souza, F.D.C. Camu-Camu Super Fruit (Myrciaria dubia (H.B.K) Mc Vaugh) at Different Maturity Stages. Afr. J. Agric. Res. 2016, 11, 2519–2523. [Google Scholar] [CrossRef]
- Furukawa, M.; Yamada, K.; Kurosawa, M.; Shikama, Y.; Wang, J.; Watanabe, M.; Kanekura, T.; Matsushita, K. High Concentration of Glucose Induces Filaggrin-1 Expression through AP-1 in Skin Keratinocytes. J. Dermatol. Sci. 2020, 98, 137–140. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, J.; Zhang, L.; Gao, P.; Wu, X. Adiponectin Attenuates High Glucose-Induced Apoptosis through the AMPK/P38 MAPK Signaling Pathway in NRK-52E Cells. PLoS ONE 2017, 12, e0178215. [Google Scholar]
- Atsaves, V.; Leventaki, V.; Rassidakis, G.Z.; Claret, F.X. AP-1 Transcription Factors as Regulators of Immune Responses in Cancer. Cancers 2019, 11, 1037. [Google Scholar] [CrossRef] [PubMed]
- Asadi, F.; Razmi, A.; Dehpour, A.R.; Shafiei, M. Tropisetron Inhibits High Glucose-Induced Calcineurin/NFAT Hypertrophic Pathway in H9c2 Myocardial Cells. J. Pharm. Pharmacol. 2016, 68, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Vaz, E.; McNeilly, A.D.; Berglund, L.M.; Ahmad, A.; Gallagher, J.R.; Dutius Andersson, A.M.; McCrimmon, R.J.; Zetterqvist, A.V.; Gomez, M.F.; Khan, F. Inhibition of NFAT Signaling Restores Microvascular Endothelial Function in Diabetic Mice. Diabetes 2020, 69, 424–435. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Martínez, S.; Redondo, J.M. Inhibitors of the Calcineurin/NFAT Pathway. Curr. Med. Chem. 2004, 11, 997–1007. [Google Scholar] [CrossRef]
- Lee, S.S.; Tan, A.W.H.; Giam, Y.C. Cyclosporin in the Treatment of Severe Atopic Dermatitis: A Retrospective Study. Ann. Acad. Med. Singap. 2004, 33, 311–313. [Google Scholar] [PubMed]
- Polce, S.A.; Burke, C.; França, L.M.; Kramer, B.; de Andrade Paes, A.M.; Carrillo-Sepulveda, M.A. Ellagic Acid Alleviates Hepatic Oxidative Stress and Insulin Resistance in Diabetic Female Rats. Nutrients 2018, 10, 531. [Google Scholar] [CrossRef]
- Ngo, H.T.T.; Hwang, E.; Kang, H.; Park, B.; Seo, S.A.; Yi, T.H. Anti-Inflammatory Effects of Achillea Millefolium on Atopic Dermatitis-Like Skin Lesions in NC/Nga Mice. Am. J. Chin. Med. 2020, 48, 1121–1140. [Google Scholar] [CrossRef]
| Active Compound | Structure | Pharmacological Effect | Reference |
|---|---|---|---|
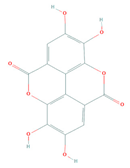 | Anti-inflammatory | [25,26] | |
| Immunomodulatory | [27] | ||
| Antiallergic | [28] | ||
| Ellagic acid (PubChem: 5281855) | Anticancer | [29] | |
| Antioxidative | [30] | ||
| Antiaging | [31] | ||
| Anti-diabetes | [32] | ||
| Quercetin (PubChem: 5280343) | 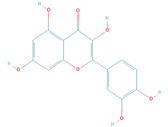 | Immunomodulatory | [33] |
| Anti-inflammatory | [34] | ||
| Antioxidative | [35] | ||
| Antiaging | [36] | ||
| Anticancer | [37] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Do, N.Q.; Zheng, S.; Park, B.; Nguyen, Q.T.N.; Choi, B.-R.; Fang, M.; Kim, M.; Jeong, J.; Choi, J.; Yang, S.-J.; et al. Camu-Camu Fruit Extract Inhibits Oxidative Stress and Inflammatory Responses by Regulating NFAT and Nrf2 Signaling Pathways in High Glucose-Induced Human Keratinocytes. Molecules 2021, 26, 3174. https://doi.org/10.3390/molecules26113174
Do NQ, Zheng S, Park B, Nguyen QTN, Choi B-R, Fang M, Kim M, Jeong J, Choi J, Yang S-J, et al. Camu-Camu Fruit Extract Inhibits Oxidative Stress and Inflammatory Responses by Regulating NFAT and Nrf2 Signaling Pathways in High Glucose-Induced Human Keratinocytes. Molecules. 2021; 26(11):3174. https://doi.org/10.3390/molecules26113174
Chicago/Turabian StyleDo, Nhung Quynh, Shengdao Zheng, Bom Park, Quynh T. N. Nguyen, Bo-Ram Choi, Minzhe Fang, Minseon Kim, Jeehaeng Jeong, Junhui Choi, Su-Jin Yang, and et al. 2021. "Camu-Camu Fruit Extract Inhibits Oxidative Stress and Inflammatory Responses by Regulating NFAT and Nrf2 Signaling Pathways in High Glucose-Induced Human Keratinocytes" Molecules 26, no. 11: 3174. https://doi.org/10.3390/molecules26113174
APA StyleDo, N. Q., Zheng, S., Park, B., Nguyen, Q. T. N., Choi, B.-R., Fang, M., Kim, M., Jeong, J., Choi, J., Yang, S.-J., & Yi, T.-H. (2021). Camu-Camu Fruit Extract Inhibits Oxidative Stress and Inflammatory Responses by Regulating NFAT and Nrf2 Signaling Pathways in High Glucose-Induced Human Keratinocytes. Molecules, 26(11), 3174. https://doi.org/10.3390/molecules26113174







