Synthesis of EPA- and DHA-Enriched Structured Acylglycerols at the sn-2 Position Starting from Commercial Salmon Oil by Enzymatic Lipase Catalysis under Supercritical Conditions
Abstract
1. Introduction
2. Results
2.1. Physical and Chemical Characterization of Deodorized Refined Commercial Salmon Oil (DRCSO)
2.2. Fatty Acid Composition and Quantification of DRCSO
2.3. Thin-Layer Chromatography (TLC) of DRCSO
2.4. Position of EPA and DHA in TG of DRCSO Identified in Different Matrices Using Mass Spectrometry (MALDI-TOF)
2.5. Influence of Fractionation under CO2SC Conditions on FA Composition of Structured Acylglycerols (EPA/DHA sAG) during Enzymatic Intra–Interesterification Process of DRCSO
2.6. Influence of Fractionation under CO2SC Conditions on MG, DG and TG of Purified EPA/DHA sAG Starting from DRCSO during Enzymatic Intra–Interesterification Process of DRCSO
2.7. Influence of Fractionation under CO2SC Conditions on the sn-2 Position of EPA and DHA in EPA/DHA sAG during Enzymatic Intra–Interesterification Process of DRSCO
3. Discussion
4. Materials and Methods
4.1. DRCSO Characterization
4.2. DRCSO Fatty Acid Composition
4.3. Preparation of EPA/DHA sAG by Enzymatic Intra–Interesterification Process under CO2-Supercritical Conditions (CO2SC) Using Different Extraction Times
4.4. Purification of EPA/DHA sAG by Neutralization with NaOH
4.5. Fatty Acid Composition and Quantification of Fractionated EPA/DHA sAG
4.6. Identification of EPA/DHA sAG and DRCSO by TLC
4.7. Positional Analysis of EPA and DHA by Mass Spectrometry (MALDI-TOF)
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Schunck, W.-H.; Konkel, A.; Fischer, R.; Weylandt, K.-H. Therapeutic potential of omega-3 fatty acid-derived epoxyeicosanoids in cardiovascular and inflammatory diseases. Pharmacol. Ther. 2018, 183, 177–204. [Google Scholar] [CrossRef]
- Minihane, A.M.; Armah, C.K.; Miles, E.A.; Madden, J.M.; Clark, A.B.; Caslake, M.J.; Packard, C.J.; Kofler, B.M.; Lietz, G.; Curtis, P.J.; et al. Consumption of Fish Oil Providing Amounts of Eicosapentaenoic Acid and Docosahexaenoic Acid That Can Be Obtained from the Diet Reduces Blood Pressure in Adults with Systolic Hypertension: A Retrospective Analysis. J. Nutr. 2016, 146, 516–523. [Google Scholar] [CrossRef]
- Ofosu, F.K.; Daliri, E.B.M.; Lee, B.H.; Yu, X. Current trends and future perspectives on omega-3 fatty acids. RRJOB 2017, 5, 11–20. [Google Scholar]
- Takahashi, M.; Tsuboyama-Kasaoka, N.; Nakatani, T.; Ishii, M.; Tsutsumi, S.; Aburatani, H.; Ezaki, O. Fish oil feeding alters liver gene expressions to defend against PPARα activation and ROS production. Am. J. Physiol. Liver Physiol. 2002, 282, G338–G348. [Google Scholar] [CrossRef] [PubMed]
- Siscovick, D.S.; Barringer, T.A.; Fretts, A.M.; Wu, J.H.Y.; Lichtenstein, A.H.; Costello, R.B.; Kris-Etherton, P.M.; Jacobson, T.A.; Engler, M.B.; Alger, H.M.; et al. Omega-3 Polyunsaturated Fatty Acid (Fish Oil) Supplementation and the Prevention of Clinical Cardiovascular Disease. Circulation 2017, 135, e867–e884. [Google Scholar] [CrossRef] [PubMed]
- Thammapat, P.; Siriamornpun, S.; Raviyan, P. Concentration of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) of Asian catfish oil by urea complexation: Optimization of reaction conditions. SJST 2016, 38, 163–170. [Google Scholar] [CrossRef]
- Food and Agriculture Organization. Fats and fatty acids in human nutrition: Report of an expert consultation. Food Nutr. 2010, 91, 1–166. [Google Scholar]
- Dominiczak, M.H.; Priest, M.; Kulkarni, U.V.; Broon, J.I. Digestion and absorption of nutrients: The gastrointesti-nal tract. In Medical Biochemistry, 4th ed.; Baynes, J.W., Dominiczak, M.H., Eds.; Saunders Elsevier: London, UK, 2014; Chapter 10. [Google Scholar]
- Bracco, U. Effect of triglyceride structure on fat absorption. Am. J. Clin. Nutr. 1994, 60, 1002S–1009S. [Google Scholar] [CrossRef]
- Moreno, N.; Perea, A. Producción de lípidos estructurados por transesterificación enzimática del aceite de soja y aceite de palmiste en reactor de lecho empacado. Grasas Aceites 2008, 59, 337–345. [Google Scholar]
- Hita, E.; Robles, A.; Camacho, B.; Ramírez, A.; Esteban, L.; Jiménez, M.J.; Muñío, M.M.; González, P.A.; Molina, E. Production of structured triacylglycerols (STAG) rich in docosahexaenoic acid (DHA) in position 2 by acidolysis of tuna oil catalyzed by lipases. Process. Biochem. 2007, 42, 415–422. [Google Scholar] [CrossRef]
- Nunes, P.A.; Cabral, P.P.; Guillén, M.; Valero, F.P.; Luna, D.; Ferreiradias, S. Production of MLM-Type Structured Lipids Catalyzed by Immobilized Heterologous Rhizopus oryzae Lipase. J. Am. Oil Chem. Soc. 2011, 88, 473–480. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Han, J.J.; Narita, M.; Rosu, R.; Yamane, T. Enzymatic synthesis of structured lipids from single cell oil of high docosahexaenoic acid content. J. Am. Oil Chem. Soc. 1999, 76, 563–569. [Google Scholar] [CrossRef]
- Pando, M.E.; Rodríguez, A.; Valenzuela, M.A.; Berríos, M.M.; Rivera, M.; Romero, N.; Barriga, A.; Aubourg, S.P. Acylglycerol synthesis including EPA and DHA from rainbow trout (Oncorhynchus mykiss) belly flap oil and caprylic acid catalyzed by Thermomyces lanuginosus lipase under supercritical carbon dioxide. Eur. Food Res. Technol. 2021, 247, 499–511. [Google Scholar] [CrossRef]
- Quirasco, M.; López-Munguía, A. Enzimas. In Química de los Alimentos, 4th ed.; Badui, S., Ed.; Pearson Addison Wesley: Mexico City, Mexico, 2006; pp. 245–297. [Google Scholar]
- Shahidi, F. Bailey’s Industrial Oil and Fat Products. In Edible Oil and Fat Products: Processing Technologies, 6th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2004. [Google Scholar]
- Shimada, Y. Enzymatic modification of lipids for functional foods and nutraceuticals. In Handbook of Functional Lipids; Akoh, C., Ed.; CRC Press Taylor & Francis Group: Vancouver, BC, Canada, 2006; pp. 77–88. [Google Scholar]
- Lyberg, A.-M.; Adlercreutz, P. Lipase specificity towards eicosapentaenoic acid and docosahexaenoic acid depends on substrate structure. Biochim. Biophys. Acta Proteins Proteom. 2008, 1784, 343–350. [Google Scholar] [CrossRef] [PubMed]
- King, J.W. Critical fluid technology for the processing of lipid-related natural products. C. R. Chim. 2004, 7, 647–659. [Google Scholar] [CrossRef]
- Rubio-Rodríguez, N.; Beltrán, S.; Jaime, I.; De Diego, S.M.; Sanz, M.T.; Carballido, J.R. Production of omega-3 polyunsaturated fatty acid concentrates: A review. Innov. Food Sci. Emerg. Technol. 2010, 11, 1–12. [Google Scholar] [CrossRef]
- Budisa, N.; Schulze-Makuch, D. Supercritical Carbon Dioxide and Its Potential as a Life-Sustaining Solvent in a Planetary Environment. Life 2014, 4, 331–340. [Google Scholar] [CrossRef]
- Lin, T.-J.; Chen, S.-W. Enrichment of n-3 polyunsaturated fatty acids into acylglycerols of borage oil via lipase-catalyzed reactions under supercritical conditions. Chem. Eng. J. 2008, 141, 318–326. [Google Scholar] [CrossRef]
- Food and Drug Administration (FDA). Code of Federal Regulations. Listing of Specific Substances Affirmed as GRAS; Sec. 184.1240 Carbon dioxide; Title 21; Food and Drug Administration (FDA): Silver Spring, MD, USA, 2020. [Google Scholar]
- Shekarchizadeh, H.; Kadivar, M.; Ghaziaskar, H.S.; Rezayat, M. Optimization of enzymatic synthesis of cocoa butter analog from camel hump fat in supercritical carbon dioxide by response surface method (RSM). J. Supercrit. Fluids 2009, 49, 209–215. [Google Scholar] [CrossRef]
- Gunnlaugsdottir, H.; Järemo, M.; Sivik, B. Process parameters influencing ethanolysis of cod liver oil in supercritical carbon dioxide. J. Supercrit. Fluids 1998, 12, 85–93. [Google Scholar] [CrossRef]
- Strohalm, M.; Hassman, M.; Košata, B.; Kodíček, M. mMass data miner: An open source alternative for mass spectrometric data analysis. Rapid Commun. Mass Spectrom. 2008, 22, 905–908. [Google Scholar] [CrossRef]
- Strohalm, M.; Kavan, D.; Novák, P.; Volný, M.; Havlicek, V. mMass 3: A Cross-Platform Software Environment for Precise Analysis of Mass Spectrometric Data. Anal. Chem. 2010, 82, 4648–4651. [Google Scholar] [CrossRef]
- Niedermeyer, T.H.J.; Strohalm, M. mMass as a Software Tool for the Annotation of Cyclic Peptide Tandem Mass Spectra. PLoS ONE 2012, 7, e44913. [Google Scholar] [CrossRef]
- Pando, M.E.; Bravo, B.; Berríos, M.; Galdames, A.; Rojas, C.; Romero, N.; Camilo, C.; Encina, C.; Rivera, M.; Rodríguez, A.; et al. Concentrating n-3 fatty acids from crude and refined commercial salmon oil. Czech J. Food Sci. 2014, 32, 169–176. [Google Scholar] [CrossRef]
- Berríos, M.M.; Rodriguez, A.; Rivera, M.; Pando, M.E.; Valenzuela, M.A.; Aubourg, S.P. Optimisation of rancidity stability in long-chain PUFA concentrates obtained from a rainbow trout (Oncorhynchus mykiss) by-product. Int. J. Food Sci. Technol. 2017, 52, 1463–1472. [Google Scholar] [CrossRef]
- Espinosa, A.; Ross, A.; Dovale-Rosabal, G.; La Fuente, F.P.-D.; Uribe-Oporto, E.; Sacristán, C.; Ruiz, P.; Valenzuela, R.; Romero, N.; Aubourg, S.P.; et al. EPA/DHA Concentrate by Urea Complexation Decreases Hyperinsulinemia and Increases Plin5 in the Liver of Mice Fed a High-Fat Diet. Molecules 2020, 25, 3289. [Google Scholar] [CrossRef] [PubMed]
- Codex Alimentarius. Standard for Fish Oils; Facultad de Ciencias Químicas y Farmacéuticas CODEX STAN 329: Italy, Rome, 2017. [Google Scholar]
- Global Organization for EPA and DHA Omega-3 Voluntary Monograph; GOED: Salt Lake City, UT, USA, 2016; Available online: http://www.goedomega3.com/images/stories/files/goedmonograph.pdf./ (accessed on 16 June 2016).
- European Pharmacopeia (Ph Eur), Compositional Guideline for Fish Oil-Natural Type II, Farmed Salmon Oil. Mono-graphs 1910 and 1912; EDQM, Council of Europe: Strasbourg, France, 2015.
- British Pharmacopoeia (BP), Compositional Guideline for Fish Oil-Natural Type II, Farmed Salmon Oil; Medicine of Healthcare Products Regulatory Agency (MHRA), TSO (The Stationery Office): London, UK, 2015; Volume 2, pp. 787–789.
- Masson, L.; Robert, P.; Romero, N.; Izaurieta, M.; Valenzuela, S.; Ortiz, J.; Dobarganes, M.C. Comportamiento de aceites poliinsaturados en la preparación de patatas fritas para consumo inmediato: Formación de nuevos com-puestos y comparación de métodos analíticos. Grasas Aceites 1997, 48, 273–281. [Google Scholar] [CrossRef][Green Version]
- Méndez, C.; Masson, L.; Jiménez, P. Estabilización de aceite de pescado por medio de antioxidantes naturales. Grasas Aceites 2010, 20, 492–500. [Google Scholar]
- Wanasundara, U.N.; Shahidi, F.; Jablonski, C.R. Comparison of standard and NMR methodologies for assessment of oxidative stability of canola and soybean oils. Food Chem. 1995, 52, 249–253. [Google Scholar] [CrossRef]
- Dovale-Rosabal, G.; Rodríguez, A.; Contreras, E.; Ortiz-Viedma, J.; Muñoz, M.; Trigo, M.; Aubourg, S.P.; Espinosa, A. Concentration of EPA and DHA from Refined Salmon Oil by Optimizing the Urea–Fatty Acid Adduction Reaction Conditions Using Response Surface Methodology. Molecules 2019, 24, 1642. [Google Scholar] [CrossRef]
- De Boer, A.A.; Ismail, A.; Marshall, K.; Bannenberg, G.; Yan, K.L.; Rowe, W.J. Examination of marine and vegetable oil oxidation data from a multi-year, third-party database. Food Chem. 2018, 254, 249–255. [Google Scholar] [CrossRef]
- Codony, R.; Guardiola, F.; Bou, R.; Tres, A. Valoración analítica y nutricional de las grasas. XXVI Curso de Especialización, Fundación Española para el Desarrollo de la Nutricional Animal. FEDNA 2010, 7, 175–206. [Google Scholar]
- Ratnayake, W.N.; Galli, C. Fat and Fatty Acid Terminology, Methods of Analysis and Fat Digestion and Metabolism: A Background Review Paper. Ann. Nutr. Metab. 2009, 55, 8–43. [Google Scholar] [CrossRef] [PubMed]
- Pando, M.E.; Rodríguez, A.; Galdames, A.; Berríos, M.M.; Rivera, M.; Romero, N.; Valenzuela, M.A.; Ortiz, J.; Aubourg, S.P. Maximization of the docosahexaenoic and eicosapentaenoic acids content in concentrates obtained from a by-product of rainbow trout (Oncorhynchus mykiss) processing. Eur. Food Res. Technol. 2017, 244, 937–948. [Google Scholar] [CrossRef]
- Tocher, D.R.; Betancor, M.B.; Sprague, M.; E Olsen, R.; A Napier, J. Omega-3 Long-Chain Polyunsaturated Fatty Acids, EPA and DHA: Bridging the Gap between Supply and Demand. Nutrients 2019, 11, 89. [Google Scholar] [CrossRef]
- Nanton, D.A.; Vegusdal, A.; Rørå, A.M.B.; Ruyter, B.; Baeverfjord, G.; Torstensen, B.E. Muscle lipid storage pattern, composition, and adipocyte distribution in different parts of Atlantic salmon (Salmo salar) fed fish oil and vegetable oil. Aquaculture 2007, 265, 230–243. [Google Scholar] [CrossRef]
- Ng, T.-T.; Li, S.; Ng, C.C.A.; So, P.-K.; Wong, T.-F.; Li, Z.-Y.; Chan, S.T.; Yao, Z.-P. Establishment of a spectral database for classification of edible oils using matrix-assisted laser desorption/ionization mass spectrometry. Food Chem. 2018, 252, 335–342. [Google Scholar] [CrossRef]
- Shinn, S.E.; Liyanage, R.; Lay, J.O.; Proctor, A. Using MALDI MS for rapid analysis of food lipids. Lipid Technol. 2015, 27, 255–257. [Google Scholar] [CrossRef]
- Badu, M.; Awudza, A.M.J. Determination of the triacylglycerol content for the identification and assessment of purity of shea butter fat, peanut oil, and palm kernel oil using maldi-tof/tof mass spectroscopic technique. Int. J. Food Prop. 2016, 20, 271–280. [Google Scholar] [CrossRef]
- AOCS. Official Methods and Recommended Practices of the American Oil Chemists Society, 4th ed.; AOCS: Champaign, IL, USA, 1993. [Google Scholar]
- Kim, R.S.; LaBella, F.S. Comparison of analytical methods for monitoring autoxidation profiles of authentic lipids. J. Lipid Res. 1987, 28, 1110–1117. [Google Scholar] [CrossRef]
- European Pharmacopoeia. Composition of Fatty Acids in Oils Rich in Omega-3-Acid, 5th ed.; 01/2005:20429; EDQM, Council of Europe: Strasbourg, France, 2005. [Google Scholar]
- AOCS. Determination of cis-, trans-, saturated, monounsaturated, and polyunsaturated fatty acids by capillary gas liquid chromatography (GLC). Sampling and analysis of commercial fats and oils. In Official Method Ce 1j-7. Official Methods and Recommended Practices of the American Oil Chemists Society (edited by AOCS); AOCS: Champaign, IL, USA, 2009. [Google Scholar]
- Cerdán, L.; Medina, A.; Giménez, A.; Ibáñez, M.; Molina, E. Synthesis of polyunsaturated fatty acid-enriched tri-glycerides by lipase-catalyzed esterification. J. Am. Oil. Chem. Soc. 1998, 75, 1329–1337. [Google Scholar] [CrossRef]
- Lin, T.-J.; Chen, S.-W.; Chang, A.-C. Enrichment of n-3 PUFA contents on triglycerides of fish oil by lipase-catalyzed trans-esterification under supercritical conditions. Biochem. Eng. J. 2006, 29, 27–34. [Google Scholar] [CrossRef]
- Wanasundara, U.N.; Shahidi, F. Positional Distribution of Fatty Acids in Triacylglycerols of Seal Blubber Oil. J. Food Lipids 1997, 4, 51–64. [Google Scholar] [CrossRef]
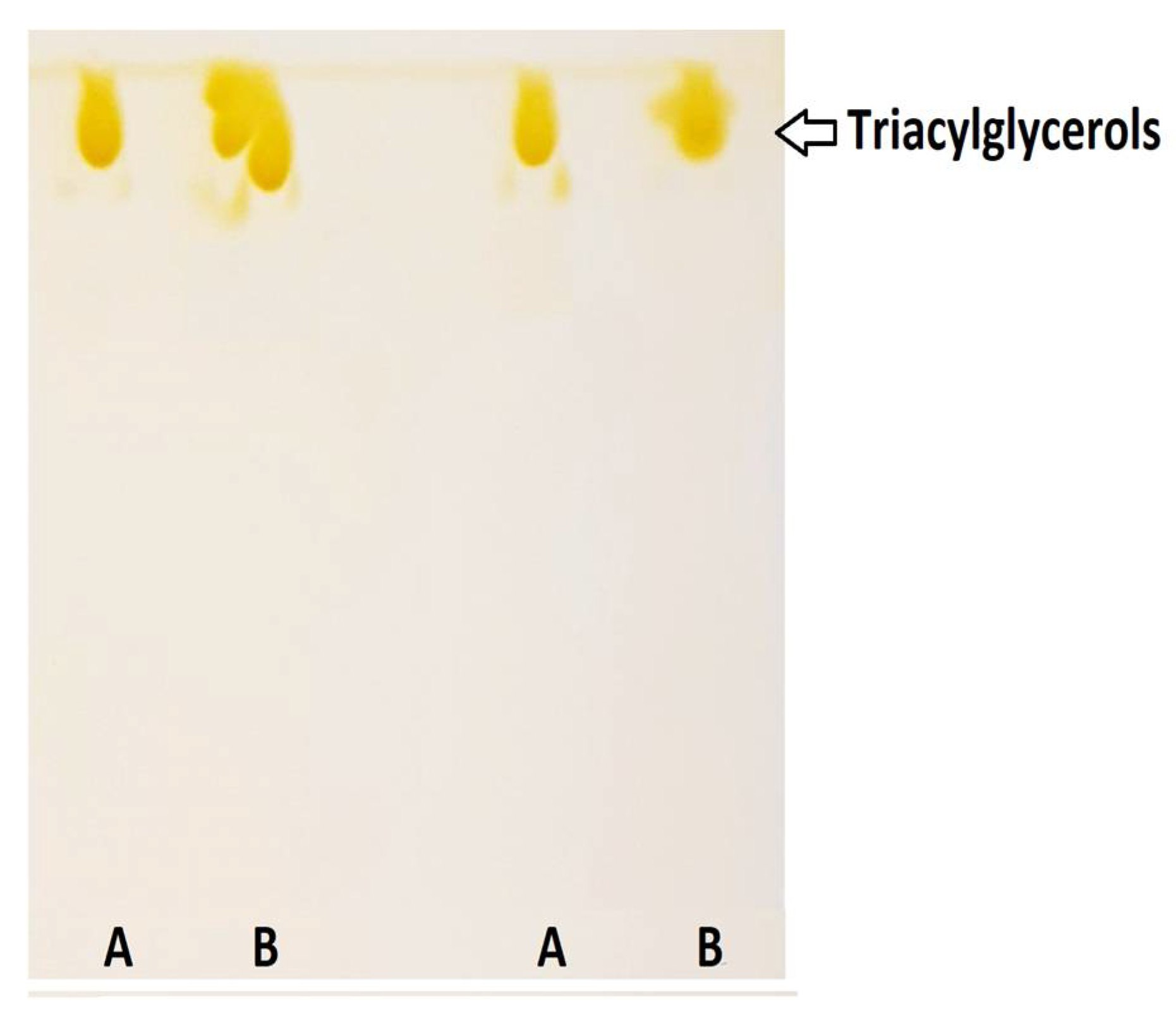
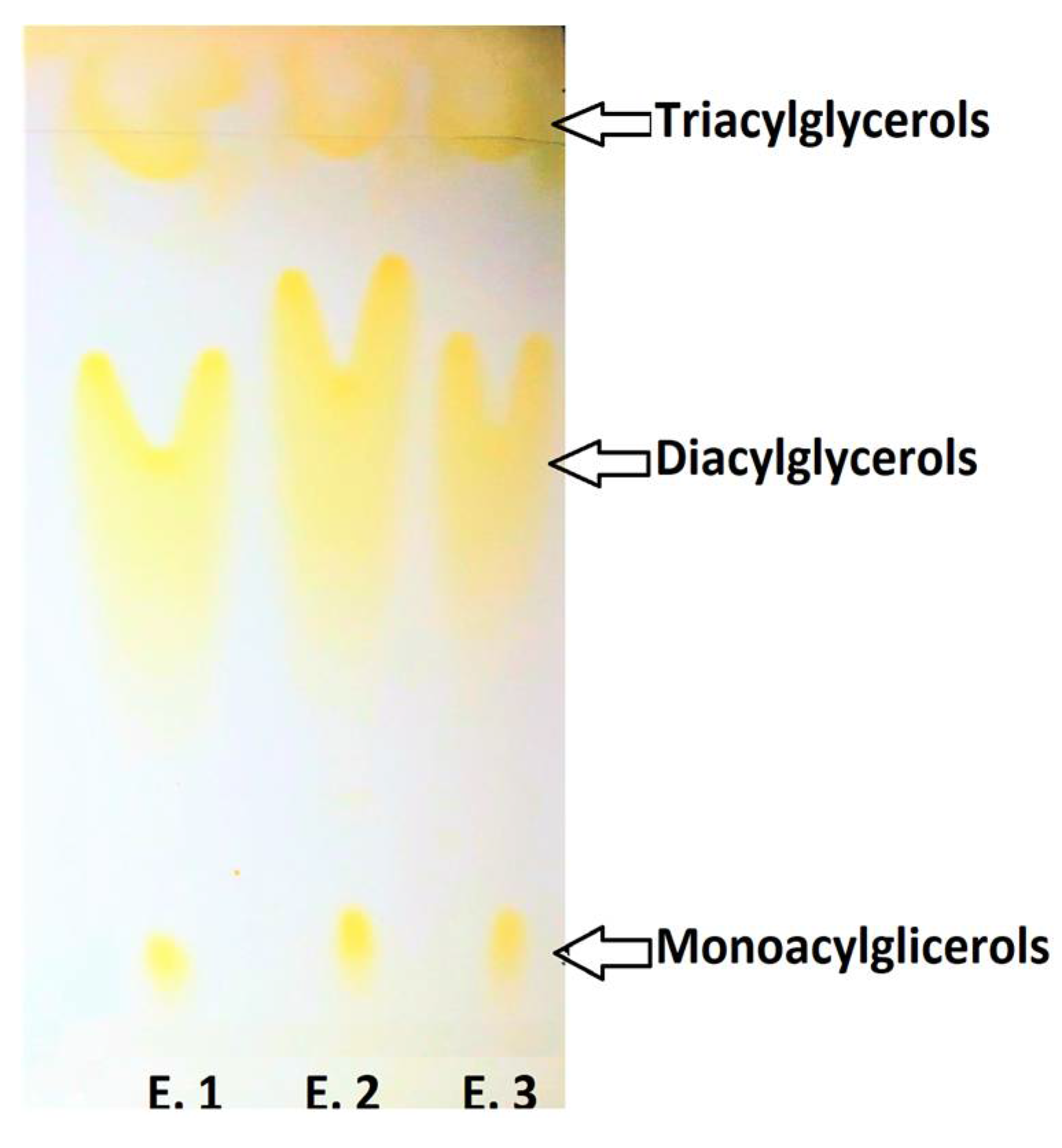
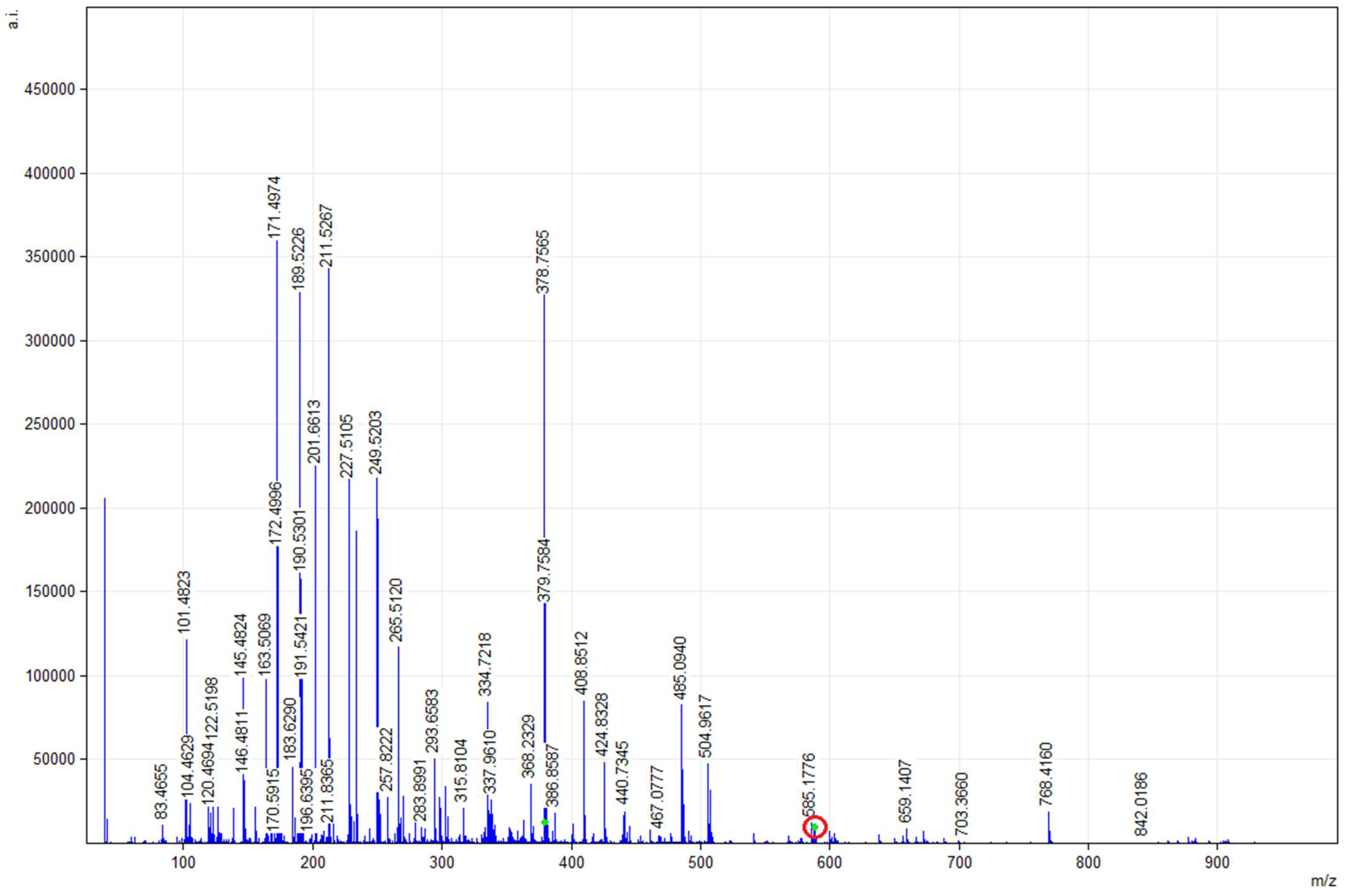

| Analyses | Value |
|---|---|
| Free fatty acids (FFA) content (g oleic acid/100 g oil) | 0.20 ± 0.00 |
| Peroxide value (PV) (meq active oxygen/kg oil) | 0.40 ± 0.00 |
| p-anisidine value (AV) | 1.13 ± 0.02 |
| TOTOX value | 1.93 ± 0.02 |
| Insoluble impurities content (%) | 0.00 ± 0.00 |
| Moisture and volatile matter content (%) | 0.04 ± 0.00 |
| Systematic Name | Systematic Abbreviation * | DRCSO (g/100 g TFA) |
|---|---|---|
| Lauric acid | 12:0 | 0.06 ± 0.00 |
| Myristic acid | 14:0 | 2.90 ± 0.01 |
| Palmitic acid | 16:0 | 12.76 ± 0.03 |
| trans-Palmitoleic acid | 9t-16:1 | 0.07± 0.00 |
| Palmitoleic acid | 9c-16:1 | 3.74 ± 0.01 |
| Heptadecanoic acid | 17:0 | 0.22 ± 0.00 |
| Heptadecenoic acid | 10c-17:1 | 0.13 ± 0.00 |
| Stearic acid | 18:0 | 3.64 ± 0.01 |
| Oleic acid | 9c-18:1 | 36.95 ± 0.08 |
| cis-Vaccenic acid | 7c-18:1 | 3.32 ± 0.00 |
| trans-Linoelaidic acid | 9t, 12t-18:2 | 0.06 ± 0.00 |
| Linoleic acid | 9c, 12c-18:2 | 15.77 ± 0.07 |
| Arachidic acid | 20:0 | 0.32 ± 0.00 |
| Gamma linolenic acid | 6c, 9c, 12c-18:3 | 0.22 ± 0.00 |
| 8-Eicosanoic acid | 8c-20:1 | 0.44 ± 0.01 |
| 11-Eicosenoic acid | 11c-20:1 | 1.84 ± 0.04 |
| α-Linolenic acid | 9c, 12c, 15c-18:3 | 4.91 ± 0.00 |
| Eicosadienoic acid | 11c, 14c-20:2 | 1.34 ± 0.01 |
| Behenoic acid | 22:0 | 0.36 ± 0.00 |
| Eicosatrienoic acid | 11c,14c,17c-20:3 | 0.33 ± 0.00 |
| Erucic acid | 13c-22:1 | 0.29 ± 0.01 |
| Arachidonic acid | 5c, 8c, 11c, 14c-20:4 | 0.35 ± 0.04 |
| Docosadienoic acid | 13c, 16c-22:2 | 0.12 ± 0.01 |
| Eicosapentaenoic acid | 5c, 8c, 11c, 14c, 17c-20:5 | 3.92 ± 0.04 |
| Nervonic acid | 9c-24:1 | 0.27 ± 0.03 |
| Docosatetraenoic acid | 7c, 10c, 13c, 16c-22:4 | 0.15 ± 0.03 |
| Docosapentaenoic acid | 7c, 10c, 13c, 16c, 19c-22:5 | 1.68 ± 0.05 |
| Docosahexaenoic acid | 4c, 7c, 10c, 13c, 16c, 19c-22:6 | 3.83 ± 0.04 |
| (a) | ||||
| m/z | sn-2 Position of EPA and/or DHA Identified in CHCA1 Matrix * | |||
| 601.4827 | DG (15:0/20:5/‒) | |||
| 877.7280 | TG (12:0/20:5/22:2), TG (16:0/16:1/22:6) | |||
| 877.6892 | TG (12:0/20:5/22:5), TG (14:0/20:5/20:5), TG (14:1/18:3/22:6) | |||
| 879.7436 | TG (12:0/20:5/22:1), TG (16:0/16:0/22:6) | |||
| 879.6473 | TG (13:0/20:5/20:5) | |||
| 879.7048 | TG (12:0/20:5/22:4), TG (14:0/18:3/22:6) | |||
| 881.7205 | TG (12:0/20:5/22:3), TG (14:0/18:2/22:6) | |||
| 881.6056 | TG (12:0/20:5/20:5) | |||
| 881.7593 | TG (12:0/20:5/22:0), TG (13:0/20:5/21:0) | |||
| 901.7831 | TG (14:0/20:5/21:0), TG (13:0/20:5/22:0) | |||
| 901.6892 | TG (12:0/22:6/22:6), TG (14:1/20:5/22:6) | |||
| 901.728 | TG (14:0/20:5/22:4), TG (14:1/20:5/22:3) | |||
| 901.7256 | TG (12:0/20:5/22:1), TG (14:0/18:0/22:6) | |||
| 903.7412 | TG (12:0/20:5/22:0), TG (13:0/20:5/21:0) | |||
| 903.7436 | TG (14:0/20:5/22:3), TG (14:1/20:5/22:2) | |||
| 903.7048 | TG (14:1/20:5/22:5), TG (16:1/20:5/20:5), TG (14:0/20:5/22:6) | |||
| 905.7205 | TG (14:0/20:5/22:5), TG (16:0/20:5/20:5), TG (14:1/20:5/22:4) | |||
| 905.7593 | TG (14:0/20:5/22:2), TG (14:1/20:5/22:1) | |||
| 905.6995 | TG (12:0/20:5/21:0) | |||
| 905.663 | TG (13:0/20:5/22:6), TG (15:1/20:5/20:5) | |||
| 907.7749 | TG (15:1/20:5/21:0), TG (14:0/20:5/22:1), TG (14:1/20:5/22:0) | |||
| 907.6786 | TG (15:0/20:5/20:5), TG (13:0/20:5/22:5) | |||
| 907.7361 | TG (14:0/20:5/22:4), TG (14:1/20:5/22:3) | |||
| 907.6212 | TG (14:1/20:5/20:5), TG (12:0/20:5/22:6) | |||
| 927.7987 | TG (15:0/20:5/22:1), TG (15:1/20:5/22:0), TG (16:1/20:5/21:0) | |||
| 927.7412 | TG (14:0/20:5/22:2), TG (14:1/20:5/22:1) | |||
| 927.6838 | TG (13:0/20:5/22:3) | |||
| 927.7436 | TG (18:0/20:5/20:5), TG (16:0/20:5/22:5), TG (16:1/20:5/22:4) | |||
| 927.7048 | TG (18:3/20:5/20:5), TG (18:3/20:5/20:5), TG (14:1/22:6/22:6) | |||
| (b) | ||||
| m/z | FAs Identified in CMBT1 Matrix * | |||
| 369.2402 | MG (16:0/‒/‒) | |||
| 381.2975 | MG (18:0/‒/‒) | |||
| 401.2662 | MG (‒/20:4/‒) | |||
| (c) | ||||
| m/z | DG Identified in DHB1 Matrix * | |||
| 603.4288 | DG (14:0/20:0/‒), DG (17:0/17:0/‒), DG (18:0/16:0/‒), DG (16:0/18:0/‒), DG (12:0/22:0/‒) | |||
| 603.4288 | DG (‒/16:0/18:1) | |||
| 603.4288 | DG (14:0/18:2/‒), DG (16:1/16:1/‒), DG (14:1/18:1/‒), DG (12:0/20:2/0:0) | |||
| (d) | ||||
| m/z | sn-2 Position of EPA and/or DHA Identified in DHB2 Matrix * | |||
| 601.4827 | DG (15:0/20:5/‒) | |||
| 905.7593 | TG (14:0/20:5/22:2), TG (14:1/20:5/22:1), TG (14:0/20:1/22:6), TG (14:1/20:0/22:6) | |||
| 905.7593 | TG (16:0/18:1/22:6), TG (16:1/18:0/22:6) | |||
| 905.6995 | TG (12:0/20:5/21:0) | |||
| 905.7205 | TG (14:0/20:5/22:5), TG (16:0/20:5/20:5), TG (14:1/20:5/22:4), TG (16:1/18:3/22:6) | |||
| 905.7205 | TG (16:0/18:4/22:6), TG (16:1/18:3/22:6), TG (14:1/20:3/22:6), TG (14:0/20:4/22:6) | |||
| 907.7361 | TG (14:0/20:5/22:4), TG (14:1/20:5/22:3), TG (16:0/18:3/22:6), TG (16:1/18:2/22:6) | |||
| 907.7749 | TG (15:1/20:5/21:0), TG (14:0/20:5/22:1), TG (14:1/20:5/22:0), TG (14:0/20:0/22:6) | |||
| (e) | ||||
| Matrix * | EPA (sn-2) | DHA (sn-2) | DHA (sn-3) | EPA (sn-2)/DHA (sn-3) |
|---|---|---|---|---|
| CHCA1 | 52 | 2 | 12 | 4 |
| CMBT1 | 0 | 0 | 0 | 0 |
| DHB1 | 0 | 0 | 0 | 0 |
| DHB2 | 12 | 0 | 12 | 0 |
| Fatty Acid | Extraction 1 (30 min) | Extraction 2 (60 min) | Extraction 3 (120 min) |
|---|---|---|---|
| 12:0 | 0.07 ± 0.00 | 0.11 ± 0.00 | 0.13 ± 0.00 |
| 14:0 | 2.88 ± 0.01 | 3.66 ± 0.03 | 4.48 ± 0.02 |
| 16:0 | 12.65 ± 0.05 | 14.12 ± 0.03 | 14.82 ± 0.04 |
| 9t-16:1 | 0.18 ± 0.00 | 0.07 ± 0.00 | 0.10 ± 0.00 |
| 9c-16:1 | 3.82 ± 0.02 | 4.25 ± 0.04 | 4.61 ± 0.06 |
| 17:0 | 0.28 ± 0.00 | 0.22 ± 0.00 | 0.23 ± 0.00 |
| 10c-17:1 | 0.13 ± 0.00 | 0.14 ± 0.00 | 0.15 ± 0.00 |
| 18:0 | 3.59 ± 0.01 | 3.50 ± 0.01 | 3.21 ± 0.02 |
| 9c-18:1 | 35.95 ± 0.10 | 36.38 ± 0.06 | 35.80 ± 0.01 |
| 7c-18:1 | 4.28 ± 0.02 | 3.30 ± 0.07 | 3.40 ± 0.01 |
| 9c, 12c-18:2 | 15.80 ± 0.02 | 15.84 ± 0.00 | 16.13 ± 0.08 |
| 20:0 | 0.29 ± 0.00 | 0.27 ± 0.00 | 0.22 ± 0.00 |
| 6c, 9c, 12c-18:3 | 0.21 ± 0.00 | 0.23 ± 0.00 | 0.24 ± 0.00 |
| 8c-20:1 | 0.45 ± 0.01 | 0.45 ± 0.00 | 0.41 ± 0.00 |
| 11c-20:1 | 1.63 ± 0.00 | 1.63 ± 0.01 | 1.15 ± 0.01 |
| 9c, 12c, 15c-18:3 | 5.16 ± 0.03 | 4.84 ± 0.04 | 5.07 ± 0.03 |
| 11c, 14c-20:2 | 1.29 ± 0.01 | 1.27 ± 0.02 | 1.17 ± 0.00 |
| 22:0 | 0.32 ± 0.00 | 0.33 ± 0.00 | 0.31 ± 0.00 |
| 11c, 14c, 17c-20:3 | 0.60 ± 0.01 | 0.28 ± 0.00 | 0.27 ± 0.00 |
| 13c-22:1 | 0.30 ± 0.00 | 0.22 ± 0.00 | 0.17 ± 0.00 |
| 5c, 8c, 11c, 14c-20:4 | 0.33 ± 0.00 | 0.32 ± 0.00 | 0.31 ± 0.00 |
| 13c, 16c-22:2 | 0.11 ± 0.00 | 0.09 ± 0.00 | 0.07 ± 0.00 |
| 5c, 8c, 11c, 14c, 17c-20:5 * | 3.82 ± 0.01 a | 3.68 ± 0.02 b | 3.57 ± 0.01 c |
| 9c-24:1 | 0.24 ± 0.00 | 0.22 ± 0.00 | 0.20 ± 0.00 |
| 7c, 11c, 13c, 16c-22:4 | 0.12 ± 0.00 | 0.11 ± 0.00 | 0.09 ± 0.00 |
| 7c, 10c, 13c, 16c, 19c-22:5 | 1.70 ± 0.00 | 1.36 ± 0.02 | 1.11 ± 0.00 |
| 4c, 7c, 10c, 13c, 16c, 19c-22:6 * | 3.79 ± 0.00a | 3.11 ± 0.01b | 2.60 ± 0.00c |
| m/z | MG and DG Identified in Extraction 1 |
|---|---|
| 379.2819 | MG (18:1/–/–) MG (18:1/–/–), MG (–/18:1/–) |
| 379.2843 | MG (–/20:4/–) |
| 587.5585 | DG (–/16:0/18:1) |
| 587.4646 | DG (14:0/18:2/–), DG (16:1/16:1/–), DG (14:1/18:1/–), DG (12:0/20:2/–) |
| 587.4670 | DG (14:1/20:4/–), DG (12:0/22:5/–), DG (14:0/20:5/–), DG (18:4/16:1/–) |
| 587.5221 | DG (16:1/17:0/–), DG (14:1/19:0/–) |
| m/z | MG and DG Identified in Extraction 2 |
|---|---|
| 379.2819 | MG (18:1/–/–), MG (18:1/–/–), MG (–/18:1/–) |
| 379.2843 | MG (–/20:4/–) |
| 603.4385 | DG (14:0/18:2/–), DG (16:1/16:1/–), DG (15:1/17:1/–), DG (15:0/17:2/–), DG (14:1/18:1/–) |
| 603.4385 | DG (12:0/20:2/–) |
| m/z | MG and DG Identified in Extraction 3 |
|---|---|
| 377.2686 | MG (–/20:5/–) |
| 409.2925 | MG (–/22:6/–) |
| 441.2402 | MG (–/22:6/–) |
| 585.4514 | DG (14:1/20:5/–), DG (12:0/22:6/–) |
| 599.467 | DG, 13:0/22:6/–), DG (15:1/20:5/–) |
| 601.4827 | DG (15:0/20:5/–) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dovale-Rosabal, G.; Rodríguez, A.; Espinosa, A.; Barriga, A.; Aubourg, S.P. Synthesis of EPA- and DHA-Enriched Structured Acylglycerols at the sn-2 Position Starting from Commercial Salmon Oil by Enzymatic Lipase Catalysis under Supercritical Conditions. Molecules 2021, 26, 3094. https://doi.org/10.3390/molecules26113094
Dovale-Rosabal G, Rodríguez A, Espinosa A, Barriga A, Aubourg SP. Synthesis of EPA- and DHA-Enriched Structured Acylglycerols at the sn-2 Position Starting from Commercial Salmon Oil by Enzymatic Lipase Catalysis under Supercritical Conditions. Molecules. 2021; 26(11):3094. https://doi.org/10.3390/molecules26113094
Chicago/Turabian StyleDovale-Rosabal, Gretel, Alicia Rodríguez, Alejandra Espinosa, Andrés Barriga, and Santiago P. Aubourg. 2021. "Synthesis of EPA- and DHA-Enriched Structured Acylglycerols at the sn-2 Position Starting from Commercial Salmon Oil by Enzymatic Lipase Catalysis under Supercritical Conditions" Molecules 26, no. 11: 3094. https://doi.org/10.3390/molecules26113094
APA StyleDovale-Rosabal, G., Rodríguez, A., Espinosa, A., Barriga, A., & Aubourg, S. P. (2021). Synthesis of EPA- and DHA-Enriched Structured Acylglycerols at the sn-2 Position Starting from Commercial Salmon Oil by Enzymatic Lipase Catalysis under Supercritical Conditions. Molecules, 26(11), 3094. https://doi.org/10.3390/molecules26113094








