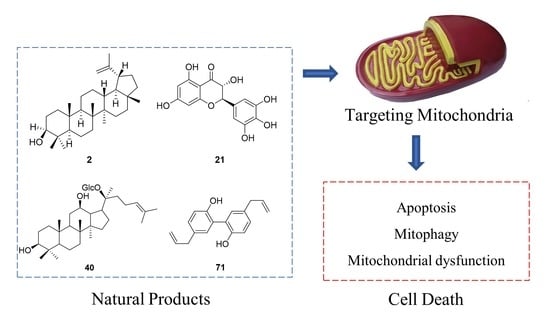
Natural Products Targeting the Mitochondria in Cancers
Abstract
1. Introduction
2. The Role of Mitochondria in Cancer Cells
3. Mitochondrial Control of Apoptosis
4. Mitochondrial Control of Energy Metabolism
5. Natural Products Induce Cancer Cell Death through a Mitochondrial Pathway
5.1. Terpenoids
5.2. Flavonoids
5.3. Saponins
5.4. Alkaloids
5.5. Coumarins
5.6. Quinones
5.7. Miscellanea
6. Natural Products and Anticancer Agents in Combination
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- The Lancet. GLOBOCAN 2018: Counting the toll of cancer. Lancet 2018, 392, 985. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef]
- Yang, Y.; He, P.; Li, N. The Antitumor Potential of Extract of the Oak Bracket Medicinal Mushroom Inonotus baumii in SMMC-7721 Tumor Cells. Evid. Based Complement. Alternat. Med. 2019, 2019, 1242784. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhang, L.; Chen, Q.; Lu, W.L.; Li, N. Antitumor Effects of Extract of the Oak Bracket Medicinal Mushroom, Phellinus baumii (Agaricomycetes), on Human Melanoma Cells A375 In Vitro and In Vivo. Int. J. Med. Mushrooms 2020, 22, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. Natural products as sources of new drugs over the period 1981–2002. J. Nat. Prod. 2003, 66, 1022–1037. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, K.C.; Yang, Z.; Liu, J.J.; Ueno, H.; Nantermet, P.G.; Guy, R.K.; Claiborne, C.F.; Renaud, J.; Couladouros, E.A.; Paulvannan, K.; et al. Total synthesis of taxol. Nature 1994, 367, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Wall, M.E.; Wani, M.C. Camptothecin and taxol: Discovery to clinic—Thirteenth Bruce F. Cain Memorial Award Lecture. Cancer Res. 1995, 55, 753–760. [Google Scholar]
- Vilpo, J.; Vilpo, L. Selective toxicity of vincristine against chronic lymphocytic leukaemia in vitro. The Tampere CLL Group. Lancet 1996, 347, 1491–1492. [Google Scholar] [CrossRef]
- Kano, Y.; Ohnuma, T.; Okano, T.; Holland, J.F. Effects of vincristine in combination with methotrexate and other antitumor agents in human acute lymphoblastic leukemia cells in culture. Cancer Res. 1988, 48, 351–356. [Google Scholar]
- Bhandary, B.; Marahatta, A.; Kim, H.R.; Chae, H.J. Mitochondria in relation to cancer metastasis. J. Bioenerg. Biomembr. 2012, 44, 623–627. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, P.M.; Ryu, S.Y.; Kinnally, K.W. Mitochondrial ion channels as therapeutic targets. FEBS Lett. 2010, 584, 2142–2152. [Google Scholar] [CrossRef] [PubMed]
- Grunewald, A.; Kumar, K.R.; Sue, C.M. New insights into the complex role of mitochondria in Parkinson’s disease. Prog. Neurobiol. 2019, 177, 73–93. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Han, Y.; Liu, J.; Song, P.; Xu, X.; Zhao, L.; Hu, C.; Xiao, L.; Liu, F.; Zhang, H.; et al. Mitochondria: A Novel Therapeutic Target in Diabetic Nephropathy. Curr. Med. Chem. 2017, 24, 3185–3202. [Google Scholar] [CrossRef] [PubMed]
- Ishimoto, Y.; Inagi, R. Mitochondria: A therapeutic target in acute kidney injury. Nephrol. Dial. Transplant. 2016, 31, 1062–1069. [Google Scholar] [CrossRef]
- Valenti, D.; Braidy, N.; De Rasmo, D.; Signorile, A.; Rossi, L.; Atanasov, A.G.; Volpicella, M.; Henrion-Caude, A.; Nabavi, S.M.; Vacca, R.A. Mitochondria as pharmacological targets in Down syndrome. Free Radic. Biol. Med. 2018, 114, 69–83. [Google Scholar] [CrossRef]
- Grad, J.M.; Cepero, E.; Boise, L.H. Mitochondria as targets for established and novel anti-cancer agents. Drug Resist. Updat. 2001, 4, 85–91. [Google Scholar] [CrossRef]
- Costantini, P.; Jacotot, E.; Decaudin, D.; Kroemer, G. Mitochondrion as a novel target of anticancer chemotherapy. J. Natl. Cancer Inst. 2000, 92, 1042–1053. [Google Scholar] [CrossRef]
- Wen, S.; Zhu, D.; Huang, P. Targeting cancer cell mitochondria as a therapeutic approach. Future Med. Chem. 2013, 5, 53–67. [Google Scholar] [CrossRef]
- Wu, D.; Wang, X.; Sun, H. The role of mitochondria in cellular toxicity as a potential drug target. Cell Biol. Toxicol. 2018, 34, 87–91. [Google Scholar] [CrossRef]
- Bhat, T.A.; Kumar, S.; Chaudhary, A.K.; Yadav, N.; Chandra, D. Restoration of mitochondria function as a target for cancer therapy. Drug Discov. Today 2015, 20, 635–643. [Google Scholar] [CrossRef]
- Brenner, D.; Mak, T.W. Mitochondrial cell death effectors. Curr. Opin. Cell Biol. 2009, 21, 871–877. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, J.; Esposti, M.D.; Gilmore, A.P. Bcl-2 proteins and mitochondria—Specificity in membrane targeting for death. Biochim. Biophys. Acta 2011, 1813, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Chalah, A.; Khosravi-Far, R. The mitochondrial death pathway. Adv. Exp. Med. Biol. 2008, 615, 25–45. [Google Scholar] [PubMed]
- Karbowski, M.; Youle, R.J. Dynamics of mitochondrial morphology in healthy cells and during apoptosis. Cell Death Differ. 2003, 10, 870–880. [Google Scholar] [CrossRef]
- Mancini, M.; Anderson, B.O.; Caldwell, E.; Sedghinasab, M.; Paty, P.B.; Hockenbery, D.M. Mitochondrial proliferation and paradoxical membrane depolarization during terminal differentiation and apoptosis in a human colon carcinoma cell line. J. Cell Biol. 1997, 138, 449–469. [Google Scholar] [CrossRef]
- Vyas, S.; Zaganjor, E.; Haigis, M.C. Mitochondria and Cancer. Cell 2016, 166, 555–566. [Google Scholar] [CrossRef]
- Ernster, L.; Schatz, G. Mitochondria: A historical review. J. Cell Biol. 1981, 91, 227s–255s. [Google Scholar] [CrossRef]
- Ren, M.; Phoon, C.K.; Schlame, M. Metabolism and function of mitochondrial cardiolipin. Prog. Lipid Res. 2014, 55, 1–16. [Google Scholar] [CrossRef]
- Ikon, N.; Ryan, R.O. Cardiolipin and mitochondrial cristae organization. Biochim. Biophys. Acta Biomembr. 2017, 1859, 1156–1163. [Google Scholar] [CrossRef]
- Dudek, J. Role of Cardiolipin in Mitochondrial Signaling Pathways. Front. Cell Dev. Biol. 2017, 5, 90. [Google Scholar] [CrossRef] [PubMed]
- Kameoka, S.; Adachi, Y.; Okamoto, K.; Iijima, M.; Sesaki, H. Phosphatidic Acid and Cardiolipin Coordinate Mitochondrial Dynamics. Trends Cell Biol. 2018, 28, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Schlattner, U.; Tokarska-Schlattner, M.; Epand, R.M.; Boissan, M.; Lacombe, M.L.; Kagan, V.E. NME4/nucleoside diphosphate kinase D in cardiolipin signaling and mitophagy. Lab. Investig. 2018, 98, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Ott, M.; Zhivotovsky, B.; Orrenius, S. Role of cardiolipin in cytochrome c release from mitochondria. Cell Death Differ. 2007, 14, 1243–1247. [Google Scholar] [CrossRef] [PubMed]
- Lucken-Ardjomande, S.; Montessuit, S.; Martinou, J.C. Contributions to Bax insertion and oligomerization of lipids of the mitochondrial outer membrane. Cell Death Differ. 2008, 15, 929–937. [Google Scholar] [CrossRef] [PubMed]
- Paradies, G.; Paradies, V.; Ruggiero, F.M.; Petrosillo, G. Role of Cardiolipin in Mitochondrial Function and Dynamics in Health and Disease: Molecular and Pharmacological Aspects. Cells 2019, 8, 728. [Google Scholar] [CrossRef]
- Kong, L.Y.; Tan, R.X. Artemisinin, a miracle of traditional Chinese medicine. Nat. Prod. Rep. 2015, 32, 1617–1621. [Google Scholar] [CrossRef]
- Ding, G.S. Important Chinese herbal remedies. Clin. Ther. 1987, 9, 345–357. [Google Scholar]
- Benelli, G.; Pavela, R.; Cianfaglione, K.; Sender, J.; Danuta, U.; Maslanko, W.; Canale, A.; Barboni, L.; Petrelli, R.; Zeppa, L.; et al. Ascaridole-rich essential oil from marsh rosemary (Ledum palustre) growing in Poland exerts insecticidal activity on mosquitoes, moths and flies without serious effects on non-target organisms and human cells. Food Chem. Toxicol. 2020, 138, 111184. [Google Scholar] [CrossRef]
- Liu, G.; Wang, K.; Kuang, S.; Cao, R.; Bao, L.; Liu, R.; Liu, H.; Sun, C. The natural compound GL22, isolated from Ganoderma mushrooms, suppresses tumor growth by altering lipid metabolism and triggering cell death. Cell Death Dis. 2018, 9, 689. [Google Scholar] [CrossRef]
- Sinha, K.; Chowdhury, S.; Banerjee, S.; Mandal, B.; Mandal, M.; Majhi, S.; Brahmachari, G.; Ghosh, J.; Sil, P.C. Lupeol alters viability of SK-RC-45 (Renal cell carcinoma cell line) by modulating its mitochondrial dynamics. Heliyon 2019, 5, e02107. [Google Scholar] [CrossRef] [PubMed]
- Potze, L.; Di Franco, S.; Grandela, C.; Pras-Raves, M.L.; Picavet, D.I.; van Veen, H.A.; van Lenthe, H.; Mullauer, F.B.; van der Wel, N.N.; Luyf, A.; et al. Betulinic acid induces a novel cell death pathway that depends on cardiolipin modification. Oncogene 2016, 35, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Pang, Q.; Wang, Y.; Yan, X. Betulinic acid induces apoptosis by regulating PI3K/Akt signaling and mitochondrial pathways in human cervical cancer cells. Int. J. Mol. Med. 2017, 40, 1669–1678. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, H.; Wang, X.; Shen, T.; Wang, S.; Ren, D. Alisol B-23-acetate, a tetracyclic triterpenoid isolated from Alisma orientale, induces apoptosis in human lung cancer cells via the mitochondrial pathway. Biochem. Biophys. Res. Commun. 2018, 505, 1015–1021. [Google Scholar] [CrossRef]
- Wang, X.; Lu, X.; Zhu, R.; Zhang, K.; Li, S.; Chen, Z.; Li, L. Betulinic Acid Induces Apoptosis in Differentiated PC12 Cells Via ROS-Mediated Mitochondrial Pathway. Neurochem. Res. 2017, 42, 1130–1140. [Google Scholar] [CrossRef]
- Yang, C.; Li, Y.; Fu, L.; Jiang, T.; Meng, F. Betulinic acid induces apoptosis and inhibits metastasis of human renal carcinoma cells in vitro and in vivo. J. Cell. Biochem. 2018, 119, 8611–8622. [Google Scholar] [CrossRef]
- Kreiter, J.; Rupprecht, A.; Zimmermann, L.; Moschinger, M.; Rokitskaya, T.I.; Antonenko, Y.N.; Gille, L.; Fedorova, M.; Pohl, E.E. Molecular Mechanisms Responsible for Pharmacological Effects of Genipin on Mitochondrial Proteins. Biophys. J. 2019, 117, 1845–1857. [Google Scholar] [CrossRef]
- Liu, X.; Wang, J.; Sun, B.; Zhang, Y.; Zhu, J.; Li, C. Cell growth inhibition, G2M cell cycle arrest, and apoptosis induced by the novel compound Alternol in human gastric carcinoma cell line MGC803. Investig. New Drugs 2007, 25, 505–517. [Google Scholar] [CrossRef]
- Li, C.; He, C.; Xu, Y.; Xu, H.; Tang, Y.; Chavan, H.; Duan, S.; Artigues, A.; Forrest, M.L.; Krishnamurthy, P.; et al. Alternol eliminates excessive ATP production by disturbing Krebs cycle in prostate cancer. Prostate 2019, 79, 628–639. [Google Scholar] [CrossRef]
- He, L.; Han, J.; Li, B.; Huang, L.; Ma, K.; Chen, Q.; Liu, X.; Bao, L.; Liu, H. Identification of a new cyathane diterpene that induces mitochondrial and autophagy-dependent apoptosis and shows a potent in vivo anti-colorectal cancer activity. Eur. J. Med. Chem. 2016, 111, 183–192. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, C.; Huang, R.Z.; Chen, H.F.; Liao, Z.X.; Sun, J.Y.; Xia, X.K.; Wang, F.X. Three new C-27-carboxylated-lupane-triterpenoid derivatives from Potentilla discolor Bunge and their in vitro antitumor activities. PLoS ONE 2017, 12, e0175502. [Google Scholar] [CrossRef] [PubMed]
- Bispo de Jesus, M.; Zambuzzi, W.F.; Ruela de Sousa, R.R.; Areche, C.; Santos de Souza, A.C.; Aoyama, H.; Schmeda-Hirschmann, G.; Rodriguez, J.A.; Monteiro de Souza Brito, A.R.; Peppelenbosch, M.P.; et al. Ferruginol suppresses survival signaling pathways in androgen-independent human prostate cancer cells. Biochimie 2008, 90, 843–854. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.X.; Lin, S.H.; Li, Y.R.; Chao, Y.H.; Lin, C.H.; Su, J.H.; Lin, C.C. Lobocrassin B Induces Apoptosis of Human Lung Cancer and Inhibits Tumor Xenograft Growth. Mar. Drugs 2017, 15, 378. [Google Scholar] [CrossRef] [PubMed]
- Nabatchian, F.; Moradi, A.; Aghaei, M.; Ghanadian, M.; Jafari, S.M.; Tabesh, S. New 6(17)-epoxylathyrane diterpene: Aellinane from Euphorbia aellenii induces apoptosis via mitochondrial pathway in ovarian cancer cell line. Toxicol. Mech. Methods 2017, 27, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Cevatemre, B.; Botta, B.; Mori, M.; Berardozzi, S.; Ingallina, C.; Ulukaya, E. The plant-derived triterpenoid tingenin B is a potent anticancer agent due to its cytotoxic activity on cancer stem cells of breast cancer in vitro. Chem. Biol. Interact. 2016, 260, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.; Lee, B.M.; Kim, O.; Tran, H.N.K.; Lee, S.; Hwangbo, C.; Min, B.S.; Lee, J.H. Triterpenoids from Ziziphus jujuba induce apoptotic cell death in human cancer cells through mitochondrial reactive oxygen species production. Food Funct. 2018, 9, 3895–3905. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.L.; Liang, J.W.; Hsu, L.C.; Chang, W.L.; Lee, S.S.; Guh, J.H. Zerumbone, a ginger sesquiterpene, induces apoptosis and autophagy in human hormone-refractory prostate cancers through tubulin binding and crosstalk between endoplasmic reticulum stress and mitochondrial insult. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2015, 388, 1223–1236. [Google Scholar] [CrossRef]
- Prasannan, R.; Kalesh, K.A.; Shanmugam, M.K.; Nachiyappan, A.; Ramachandran, L.; Nguyen, A.H.; Kumar, A.P.; Lakshmanan, M.; Ahn, K.S.; Sethi, G. Key cell signaling pathways modulated by zerumbone: Role in the prevention and treatment of cancer. Biochem. Pharmacol. 2012, 84, 1268–1276. [Google Scholar] [CrossRef]
- Assini, J.M.; Mulvihill, E.E.; Huff, M.W. Citrus flavonoids and lipid metabolism. Curr. Opin. Lipidol. 2013, 24, 34–40. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Pei, R.; Liu, X.; Bolling, B. Flavonoids and gut health. Curr. Opin. Biotechnol. 2020, 61, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, Y.; Mai, Y.; Li, H.; Wang, Z.; Xu, J.; He, X. Health Benefits of the Flavonoids from Onion: Constituents and Their Pronounced Antioxidant and Anti-Neuroinflammatory Capacities. J. Agric. Food Chem. 2020, 68, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Androutsopoulos, V.P.; Papakyriakou, A.; Vourloumis, D.; Tsatsakis, A.M.; Spandidos, D.A. Dietary flavonoids in cancer therapy and prevention: Substrates and inhibitors of cytochrome P450 CYP1 enzymes. Pharmacol. Ther. 2010, 126, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Duteil, D.; Metzger, E.; Willmann, D.; Karagianni, P.; Friedrichs, N.; Greschik, H.; Gunther, T.; Buettner, R.; Talianidis, I.; Metzger, D.; et al. LSD1 promotes oxidative metabolism of white adipose tissue. Nat. Commun. 2014, 5, 4093. [Google Scholar] [CrossRef]
- Hino, S.; Sakamoto, A.; Nagaoka, K.; Anan, K.; Wang, Y.; Mimasu, S.; Umehara, T.; Yokoyama, S.; Kosai, K.; Nakao, M. FAD-dependent lysine-specific demethylase-1 regulates cellular energy expenditure. Nat. Commun. 2012, 3, 758. [Google Scholar] [CrossRef]
- Sakamoto, A.; Hino, S.; Nagaoka, K.; Anan, K.; Takase, R.; Matsumori, H.; Ojima, H.; Kanai, Y.; Arita, K.; Nakao, M. Lysine Demethylase LSD1 Coordinates Glycolytic and Mitochondrial Metabolism in Hepatocellular Carcinoma Cells. Cancer Res. 2015, 75, 1445–1456. [Google Scholar] [CrossRef]
- Xu, X.; Peng, W.; Liu, C.; Li, S.; Lei, J.; Wang, Z.; Kong, L.; Han, C. Flavone-based natural product agents as new lysine-specific demethylase 1 inhibitors exhibiting cytotoxicity against breast cancer cells in vitro. Bioorg. Med. Chem. 2019, 27, 370–374. [Google Scholar] [CrossRef]
- Valentova, K.; Vrba, J.; Bancirova, M.; Ulrichova, J.; Kren, V. Isoquercitrin: Pharmacology, toxicology, and metabolism. Food Chem. Toxicol. 2014, 68, 267–282. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, Y.; Chen, L.; Li, H. The dietary compound luteolin inhibits pancreatic cancer growth by targeting BCL-2. Food Funct. 2018, 9, 3018–3027. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, H.; Chen, S.; Xu, Y.; Yao, A.; Liao, Q.; Han, L.; Zou, Z.; Zhang, X. Dihydromyricetin induces mitochondria-mediated apoptosis in HepG2 cells through down-regulation of the Akt/Bad pathway. Nutr. Res. 2017, 38, 27–33. [Google Scholar] [CrossRef]
- Hou, X.L.; Tong, Q.; Wang, W.Q.; Shi, C.Y.; Xiong, W.; Chen, J.; Liu, X.; Fang, J.G. Suppression of Inflammatory Responses by Dihydromyricetin, a Flavonoid from Ampelopsis grossedentata, via Inhibiting the Activation of NF-κB and MAPK Signaling Pathways. J. Nat. Prod. 2015, 78, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Ramli, F.; Karimian, H.; Dehghan, F.; Nordin, N.; Ali, H.M.; Mohan, S.; Hashim, N.M. Artonin E Induces Apoptosis via Mitochondrial Dysregulation in SKOV-3 Ovarian Cancer Cells. PLoS ONE 2016, 11, e0151466. [Google Scholar] [CrossRef] [PubMed]
- Jo, S.; Ha, T.K.; Han, S.H.; Kim, M.E.; Jung, I.; Lee, H.W.; Bae, S.K.; Lee, J.S. Myricetin Induces Apoptosis of Human Anaplastic Thyroid Cancer Cells via Mitochondria Dysfunction. Anticancer Res. 2017, 37, 1705–1710. [Google Scholar] [PubMed]
- Gao, S.; Sun, D.; Wang, G.; Zhang, J.; Jiang, Y.; Li, G.; Zhang, K.; Wang, L.; Huang, J.; Chen, L. Growth inhibitory effect of paratocarpin E, a prenylated chalcone isolated from Euphorbia humifusa Wild. by induction of autophagy and apoptosis in human breast cancer cells. Bioorg. Chem. 2016, 69, 121–128. [Google Scholar] [CrossRef]
- Zhang, X.L.; Wang, B.B.; Mo, J.S. Puerarin 6″-O-xyloside possesses significant antitumor activities on colon cancer through inducing apoptosis. Oncol. Lett. 2018, 16, 5557–5564. [Google Scholar] [CrossRef]
- El Habbash, A.I.; Mohd Hashim, N.; Ibrahim, M.Y.; Yahayu, M.; Omer, F.A.E.; Abd Rahman, M.; Nordin, N.; Lian, G.E.C. In vitro assessment of anti-proliferative effect induced by alpha-mangostin from Cratoxylum arborescens on HeLa cells. PeerJ 2017, 5, e3460. [Google Scholar] [CrossRef]
- Seydi, E.; Rahimpour, Z.; Salimi, A.; Pourahmad, J. Selective toxicity of chrysin on mitochondria isolated from liver of a HCC rat model. Bioorg. Med. Chem. 2019, 27, 115163. [Google Scholar] [CrossRef]
- Khoo, B.Y.; Chua, S.L.; Balaram, P. Apoptotic effects of chrysin in human cancer cell lines. Int. J. Mol. Sci. 2010, 11, 2188–2199. [Google Scholar] [CrossRef]
- Su, C.H.; Kuo, C.L.; Lu, K.W.; Yu, F.S.; Ma, Y.S.; Yang, J.L.; Chu, Y.L.; Chueh, F.S.; Liu, K.C.; Chung, J.G. Fisetin-induced apoptosis of human oral cancer SCC-4 cells through reactive oxygen species production, endoplasmic reticulum stress, caspase-, and mitochondria-dependent signaling pathways. Environ. Toxicol. 2017, 32, 1725–1741. [Google Scholar] [CrossRef]
- Murtaza, I.; Adhami, V.M.; Hafeez, B.B.; Saleem, M.; Mukhtar, H. Fisetin, a natural flavonoid, targets chemoresistant human pancreatic cancer AsPC-1 cells through DR3-mediated inhibition of NF-κB. Int. J. Cancer 2009, 125, 2465–2473. [Google Scholar] [CrossRef]
- Pan, Q.; Xue, M.; Xiao, S.S.; Wan, Y.J.; Xu, D.B. A Combination Therapy with Baicalein and Taxol Promotes Mitochondria-Mediated Cell Apoptosis: Involving in Akt/beta-Catenin Signaling Pathway. DNA Cell Biol. 2016, 35, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Kong, E.K.; Yu, S.; Sanderson, J.E.; Chen, K.B.; Huang, Y.; Yu, C.M. A novel anti-fibrotic agent, baicalein, for the treatment of myocardial fibrosis in spontaneously hypertensive rats. Eur. J. Pharmacol. 2011, 658, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Yang, W.; Zhang, S.N.; Lu, J.B. Alpinetin inhibits lung cancer progression and elevates sensitization drug-resistant lung cancer cells to cis-diammined dichloridoplatium. Drug Des. Dev. Ther. 2015, 9, 6119–6127. [Google Scholar]
- Umehara, K.; Nemoto, K.; Kimijima, K.; Matsushita, A.; Terada, E.; Monthakantirat, O.; De-Eknamkul, W.; Miyase, T.; Warashina, T.; Degawa, M.; et al. Estrogenic constituents of the heartwood of Dalbergia parviflora. Phytochemistry 2008, 69, 546–552. [Google Scholar] [CrossRef] [PubMed]
- Makkar, H.P.S.; Siddhuraju, P.; Becker, K. Plant Secondary Metabolites; Walker, J.M., Ed.; Publisher: Stuttgart, Germany, 2007; Volume 393, pp. 1–122. [Google Scholar]
- Wang, Y.J.; Li, Q.; Xiao, H.B.; Li, Y.J.; Yang, Q.; Kan, X.X.; Chen, Y.; Liu, X.N.; Weng, X.G.; Chen, X.; et al. Chamaejasmin B exerts anti-MDR effect in vitro and in vivo via initiating mitochondria-dependant intrinsic apoptosis pathway. Drug Des. Dev. Ther. 2015, 9, 5301–5313. [Google Scholar] [CrossRef] [PubMed]
- Plitzko, B.; Kaweesa, E.N.; Loesgen, S. The natural product mensacarcin induces mitochondrial toxicity and apoptosis in melanoma cells. J. Biol. Chem. 2017, 292, 21102–21116. [Google Scholar] [CrossRef]
- Del Hierro, J.N.; Cueva, C.; Tamargo, A.; Nunez-Gomez, E.; Moreno-Arribas, M.V.; Reglero, G.; Martin, D. In Vitro Colonic Fermentation of Saponin-Rich Extracts from Quinoa, Lentil, and Fenugreek. Effect on Sapogenins Yield and Human Gut Microbiota. J. Agric. Food Chem. 2020, 68, 106–116. [Google Scholar] [CrossRef]
- Min, H.Y.; Jang, H.J.; Park, K.H.; Hyun, S.Y.; Park, S.J.; Kim, J.H.; Son, J.; Kang, S.S.; Lee, H.Y. The natural compound gracillin exerts potent antitumor activity by targeting mitochondrial complex II. Cell Death Dis. 2019, 10, 810. [Google Scholar] [CrossRef]
- Li, G.B.; Fu, R.Q.; Shen, H.M.; Zhou, J.; Hu, X.Y.; Liu, Y.X.; Li, Y.N.; Zhang, H.W.; Liu, X.; Zhang, Y.H.; et al. Polyphyllin I induces mitophagic and apoptotic cell death in human breast cancer cells by increasing mitochondrial PINK1 levels. Oncotarget 2017, 8, 10359–10374. [Google Scholar] [CrossRef]
- Chang, J.; Wang, H.; Wang, X.; Zhao, Y.; Zhao, D.; Wang, C.; Li, Y.; Yang, Z.; Lu, S.; Zeng, Q.; et al. Molecular mechanisms of Polyphyllin I-induced apoptosis and reversal of the epithelial-mesenchymal transition in human osteosarcoma cells. J. Ethnopharmacol. 2015, 170, 117–127. [Google Scholar] [CrossRef]
- Dyshlovoy, S.A.; Rast, S.; Hauschild, J.; Otte, K.; Alsdorf, W.H.; Madanchi, R.; Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; Dierlamm, J.; et al. Frondoside A induces AIF-associated caspase-independent apoptosis in Burkitt lymphoma cells. Leuk. Lymphoma 2017, 58, 2905–2915. [Google Scholar] [CrossRef]
- Cheng, L.; Shi, L.; Wu, J.; Zhou, X.; Li, X.; Sun, X.; Zhu, L.; Xia, T.S.; Ding, Q. A hederagenin saponin isolated from Clematis ganpiniana induces apoptosis in breast cancer cells via the mitochondrial pathway. Oncol. Lett. 2018, 15, 1737–1743. [Google Scholar] [CrossRef] [PubMed]
- Song, I.S.; Jeong, Y.J.; Kim, J.; Seo, K.H.; Baek, N.I.; Kim, Y.; Kim, C.S.; Jang, S.W. Pharmacological inhibition of androgen receptor expression induces cell death in prostate cancer cells. Cell. Mol. Life Sci. 2020, 77, 4663–4673. [Google Scholar] [CrossRef]
- Vinh, L.B.; Nguyet, N.T.M.; Yang, S.Y.; Kim, J.H.; Thanh, N.V.; Cuong, N.X.; Nam, N.H.; Minh, C.V.; Hwang, I.; Kim, Y.H. Cytotoxic triterpene saponins from the mangrove Aegiceras corniculatum. Nat. Prod. Res. 2019, 33, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.M.; Kim, E.; Chun, S. Ginsenoside Compound K Induces Ros-Mediated Apoptosis and Autophagic Inhibition in Human Neuroblastoma Cells In Vitro and In Vivo. Int. J. Mol. Sci. 2019, 20, 4279. [Google Scholar] [CrossRef] [PubMed]
- Zou, K.; Li, Z.; Zhang, Y.; Zhang, H.Y.; Li, B.; Zhu, W.L.; Shi, J.Y.; Jia, Q.; Li, Y.M. Advances in the study of berberine and its derivatives: A focus on anti-inflammatory and anti-tumor effects in the digestive system. Acta Pharmacol. Sin. 2017, 38, 157–167. [Google Scholar] [CrossRef]
- Sun, Y.; Yu, J.; Liu, X.; Zhang, C.; Cao, J.; Li, G.; Liu, X.; Chen, Y.; Huang, H. Oncosis-like cell death is induced by berberine through ERK1/2-mediated impairment of mitochondrial aerobic respiration in gliomas. Biomed. Pharmacother. 2018, 102, 699–710. [Google Scholar] [CrossRef]
- Min, H.Y.; Jung, Y.; Park, K.H.; Lee, H.Y. Papuamine Inhibits Viability of Non-small Cell Lung Cancer Cells by Inducing Mitochondrial Dysfunction. Anticancer Res. 2020, 40, 323–333. [Google Scholar] [CrossRef]
- Wang, X.D.; Li, C.Y.; Jiang, M.M.; Li, D.; Wen, P.; Song, X.; Chen, J.D.; Guo, L.X.; Hu, X.P.; Li, G.Q.; et al. Induction of apoptosis in human leukemia cells through an intrinsic pathway by cathachunine, a unique alkaloid isolated from Catharanthus roseus. Phytomedicine 2016, 23, 641–653. [Google Scholar] [CrossRef]
- Rashmi, K.C.; Harsha Raj, M.; Paul, M.; Girish, K.S.; Salimath, B.P.; Aparna, H.S. A new pyrrole based small molecule from Tinospora cordifolia induces apoptosis in MDA-MB-231 breast cancer cells via ROS mediated mitochondrial damage and restoration of p53 activity. Chem. Biol. Interact. 2019, 299, 120–130. [Google Scholar] [CrossRef]
- Futamura, Y.; Muroi, M.; Aono, H.; Kawatani, M.; Hayashida, M.; Sekine, T.; Nogawa, T.; Osada, H. Bioenergetic and proteomic profiling to screen small molecule inhibitors that target cancer metabolisms. Biochim. Biophys. Acta Proteins Proteom. 2019, 1867, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.P.; Lin, Z.; Fenton, D.S.; Leavitt, L.U.; Niu, C.; Lam, P.Y.; Robes, J.M.; Peterson, R.T.; Concepcion, G.P.; Haygood, M.G.; et al. Boholamide A, an APD-Class, Hypoxia-Selective Cyclodepsipeptide. J. Nat. Prod. 2020, 83, 1249–1257. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.A.; Mondal, A.; Sachdeva, M.; Cabral, H.; Neto, Y.; Khan, I.; Groppo, M.; McChesney, J.D.; Bastos, J.K. Chemosensitizing Effect of Cernumidine Extracted from Solanum cernuum on Bladder Cancer Cells In Vitro. Chem. Biodivers. 2019, 16, e1900334. [Google Scholar] [CrossRef]
- Liu, W.Y.; Tang, Q.; Zhang, Q.; Hu, C.P.; Huang, J.B.; Sheng, F.F.; Liu, Y.L.; Zhou, M.; Lai, W.J.; Li, G.B.; et al. Lycorine Induces Mitochondria-Dependent Apoptosis in Hepatoblastoma HepG2 Cells Through ROCK1 Activation. Front. Pharmacol. 2019, 10, 651. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Huang, W.; Li, L.; Sun, X.; Song, S.; Xu, Q.; Zhang, L.; Wei, B.G.; Deng, X. Structure Determinants of Lagunamide A for Anticancer Activity and Its Molecular Mechanism of Mitochondrial Apoptosis. Mol. Pharm. 2016, 13, 3756–3763. [Google Scholar] [CrossRef]
- Wang, C.W.; Hsu, W.H.; Tai, C.J. Antimetastatic effects of cordycepin mediated by the inhibition of mitochondrial activity and estrogen-related receptor alpha in human ovarian carcinoma cells. Oncotarget 2017, 8, 3049–3058. [Google Scholar] [CrossRef]
- Kostova, I. Studying plant-derived coumarins for their pharmacological and therapeutic properties as potential anticancer drugs. Expert Opin. Drug Discov. 2007, 2, 1605–1618. [Google Scholar] [CrossRef]
- Venugopala, K.N.; Rashmi, V.; Odhav, B. Review on natural coumarin lead compounds for their pharmacological activity. Biomed. Res. Int. 2013, 2013, 963248. [Google Scholar] [CrossRef]
- Peng, X.M.; Damu, G.L.; Zhou, C. Current developments of coumarin compounds in medicinal chemistry. Curr. Pharm. Des. 2013, 19, 3884–3930. [Google Scholar] [CrossRef]
- Zhang, L.; Tong, X.; Zhang, J.; Huang, J.; Wang, J. DAW22, a natural sesquiterpene coumarin isolated from Ferula ferulaeoides (Steud.) Korov. that induces C6 glioma cell apoptosis and endoplasmic reticulum (ER) stress. Fitoterapia 2015, 103, 46–54. [Google Scholar] [CrossRef]
- Andas, A.R.; Abdul, A.B.; Rahman, H.S.; Sukari, M.A.; Abdelwahab, S.I.; Samad, N.A.; Anasamy, T.; Arbab, I.A. Dentatin from Clausena excavata Induces Apoptosis in HepG2 Cells via Mitochondrial Mediated Signaling. Asian Pac. J. Cancer Prev. 2015, 16, 4311–4316. [Google Scholar] [CrossRef]
- Arbab, I.A.; Abdul, A.B.; Sukari, M.A.; Abdullah, R.; Syam, S.; Kamalidehghan, B.; Ibrahim, M.Y.; Taha, M.M.; Abdelwahab, S.I.; Ali, H.M.; et al. Dentatin isolated from Clausena excavata induces apoptosis in MCF-7 cells through the intrinsic pathway with involvement of NF-κB signaling and G0/G1 cell cycle arrest: A bioassay-guided approach. J. Ethnopharmacol. 2013, 145, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Zhang, W.G.; Feng, X.F.; Shao, M.J.; Xing, C. Aesculetin (6,7-dihydroxycoumarin) exhibits potent and selective antitumor activity in human acute myeloid leukemia cells (THP-1) via induction of mitochondrial mediated apoptosis and cancer cell migration inhibition. J. BUON 2017, 22, 1563–1569. [Google Scholar] [PubMed]
- Grobarova, V.; Valis, K.; Talacko, P.; Pavlu, B.; Hernychova, L.; Novakova, J.; Stodulkova, E.; Flieger, M.; Novak, P.; Cerny, J. Quambalarine B, a Secondary Metabolite from Quambalaria cyanescens with Potential Anticancer Properties. J. Nat. Prod. 2016, 79, 2304–2314. [Google Scholar] [CrossRef]
- Valis, K.; Grobarova, V.; Hernychova, L.; Buganova, M.; Kavan, D.; Kalous, M.; Cerny, J.; Stodulkova, E.; Kuzma, M.; Flieger, M.; et al. Reprogramming of leukemic cell metabolism through the naphthoquinonic compound Quambalarine B. Oncotarget 2017, 8, 103137–103153. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.C.; Hou, S.M.; Huang, C.C.; Hou, C.H.; Chen, P.C.; Liu, J.F. Plumbagin induces apoptosis in human osteosarcoma through ROS generation, endoplasmic reticulum stress and mitochondrial apoptosis pathway. Mol. Med. Rep. 2017, 16, 5480–5488. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Xu, J.; Liu, X.; Xia, X.; Li, N.; Bi, X. Shikonin induces apoptosis in the human gastric cancer cells HGC-27 through mitochondria-mediated pathway. Pharmacogn. Mag. 2015, 11, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Zheng, Z.; Zhang, J.; Liu, X.; Liu, Y.; Yang, W.; Liu, Y.; Zhang, T.; Zhao, Y.; Liu, Y.; et al. Anticancer effect of 2,7-dihydroxy-3-methylanthraquinone on human gastric cancer SGC-7901 cells in vitro and in vivo. Pharm. Biol. 2016, 54, 285–292. [Google Scholar] [CrossRef]
- Feng, S.; Wang, Z.; Zhang, M.; Zhu, X.; Ren, Z. HG30, a tetrahydroanthraquinone compound isolated from the roots of Prismatomeris connate, induces apoptosis in human non-small cell lung cancer cells. Biomed. Pharmacother. 2018, 100, 124–131. [Google Scholar] [CrossRef]
- Zhang, M.; Du, H.; Huang, Z.; Zhang, P.; Yue, Y.; Wang, W.; Liu, W.; Zeng, J.; Ma, J.; Chen, G.; et al. Thymoquinone induces apoptosis in bladder cancer cell via endoplasmic reticulum stress-dependent mitochondrial pathway. Chem. Biol. Interact. 2018, 292, 65–75. [Google Scholar] [CrossRef]
- Jacobsen, K.M.; Villadsen, N.L.; Torring, T.; Nielsen, C.B.; Salomon, T.; Nielsen, M.M.; Tsakos, M.; Sibbersen, C.; Scavenius, C.; Nielsen, R.; et al. APD-Containing Cyclolipodepsipeptides Target Mitochondrial Function in Hypoxic Cancer Cells. Cell Chem. Biol. 2018, 25, 1337–1349.e12. [Google Scholar] [CrossRef] [PubMed]
- Nipin, S.P.; Kang, D.Y.; Kim, B.J.; Joung, Y.H.; Darvin, P.; Byun, H.J.; Kim, J.G.; Park, J.U.; Yang, Y.M. Methylsulfonylmethane Induces G1 Arrest and Mitochondrial Apoptosis in YD-38 Gingival Cancer Cells. Anticancer Res. 2017, 37, 1637–1646. [Google Scholar]
- Wang, Z.; Li, S.; Ren, R.; Li, J.; Cui, X. Recombinant Buckwheat Trypsin Inhibitor Induces Mitophagy by Directly Targeting Mitochondria and Causes Mitochondrial Dysfunction in Hep G2 Cells. J. Agric. Food Chem. 2015, 63, 7795–7804. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Amin, A.; Cheung, W.Y.; Xu, R.; Yu, R.; Tang, J.; Yao, X.; Liang, C. Parameritannin A-2 from Urceola huaitingii enhances doxorubicin-induced mitochondria-dependent apoptosis by inhibiting the PI3K/Akt, ERK1/2 and p38 pathways in gastric cancer cells. Chem. Biol. Interact. 2020, 316, 108924. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Li, W.; Wang, R.; Nan, Y.; Wang, Q.; Liu, W.; Jin, F. Resveratrol enhanced anticancer effects of cisplatin on non-small cell lung cancer cell lines by inducing mitochondrial dysfunction and cell apoptosis. Int. J. Oncol. 2015, 47, 1460–1468. [Google Scholar] [CrossRef]
- Wang, B.; Liu, J.; Gong, Z. Resveratrol induces apoptosis in K562 cells via the regulation of mitochondrial signaling pathways. Int. J. Clin. Exp. Med. 2015, 8, 16926–16933. [Google Scholar]
- Wang, W.; Wu, J.; Zhang, Q.; Li, X.; Zhu, X.; Wang, Q.; Cao, S.; Du, L. Mitochondria-mediated apoptosis was induced by oleuropein in H1299 cells involving activation of p38 MAP kinase. J. Cell. Biochem. 2019, 120, 5480–5494. [Google Scholar] [CrossRef]
- Shamshoum, H.; Vlavcheski, F.; Tsiani, E. Anticancer effects of oleuropein. Biofactors 2017, 43, 517–528. [Google Scholar] [CrossRef]
- Ning, D.; Jin, M.; Xv, T.; Sun, J.; Li, M. Homoisoflavanone-1 isolated from Polygonatum odoratum arrests the cell cycle and induces apoptosis in A549 cells. Oncol. Lett. 2018, 16, 3545–3554. [Google Scholar] [CrossRef]
- Wang, R.; Ma, L.; Weng, D.; Yao, J.; Liu, X.; Jin, F. Gallic acid induces apoptosis and enhances the anticancer effects of cisplatin in human small cell lung cancer H446 cell line via the ROS-dependent mitochondrial apoptotic pathway. Oncol. Rep. 2016, 35, 3075–3083. [Google Scholar] [CrossRef]
- Freitas, S.; Martins, R.; Costa, M.; Leao, P.N.; Vitorino, R.; Vasconcelos, V.; Urbatzka, R. Hierridin B Isolated from a Marine Cyanobacterium Alters VDAC1, Mitochondrial Activity, and Cell Cycle Genes on HT-29 Colon Adenocarcinoma Cells. Mar. Drugs 2016, 14, 158. [Google Scholar] [CrossRef] [PubMed]
- Kitai, Y.; Zhang, X.; Hayashida, Y.; Kakehi, Y.; Tamura, H. Induction of G2/M arrest and apoptosis through mitochondria pathway by a dimer sesquiterpene lactone from Smallanthus sonchifolius in HeLa cells. J. Food Drug Anal. 2017, 25, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Lu, M.C.; El-Shazly, M.; Lai, K.H.; Wu, T.Y.; Hsu, Y.M.; Lee, Y.L.; Liu, Y.C. Breaking down Leukemia Walls: Heteronemin, a Sesterterpene Derivative, Induces Apoptosis in Leukemia Molt4 Cells through Oxidative Stress, Mitochondrial Dysfunction and Induction of Talin Expression. Mar. Drugs 2018, 16, 212. [Google Scholar] [CrossRef] [PubMed]
- Ling, T.; Lang, W.H.; Craig, J.; Potts, M.B.; Budhraja, A.; Opferman, J.; Bollinger, J.; Maier, J.; Marsico, T.D.; Rivas, F. Studies of Jatrogossone A as a Reactive Oxygen Species Inducer in Cancer Cellular Models. J. Nat. Prod. 2019, 82, 1301–1311. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.D.; Zhang, C.; Lei, J.L.; Yu, P.; Xia, Y.Z.; Zhang, H.; Yang, L.; Kong, L.Y. Walsuronoid B induces mitochondrial and lysosomal dysfunction leading to apoptotic rather than autophagic cell death via ROS/p53 signaling pathways in liver cancer. Biochem. Pharmacol. 2017, 142, 71–86. [Google Scholar] [CrossRef]
- Luo, G.; Zhou, J.; Li, G.; Hu, N.; Xia, X.; Zhou, H. Ferruginol Diterpenoid Selectively Inhibits Human Thyroid Cancer Growth by Inducing Mitochondrial Dependent Apoptosis, Endogenous Reactive Oxygen Species (ROS) Production, Mitochondrial Membrane Potential Loss and Suppression of Mitogen-Activated Protein Kinase (MAPK) and PI3K/AKT Signaling Pathways. Med. Sci. Monit. 2019, 25, 2935–2942. [Google Scholar]
- Jin, S.; Shi, K.; Liu, L.; Chen, Y.; Yang, G. Xanthones from the Bark of Garcinia xanthochymus and the Mechanism of Induced Apoptosis in Human Hepatocellular Carcinoma HepG2 Cells via the Mitochondrial Pathway. Int. J. Mol. Sci. 2019, 20, 4803. [Google Scholar] [CrossRef]
- Losuwannarak, N.; Sritularak, B.; Chanvorachote, P. Cycloartobiloxanthone Induces Human Lung Cancer Cell Apoptosis via Mitochondria-dependent Apoptotic Pathway. In Vivo 2018, 32, 71–78. [Google Scholar]
- Yuan, S.Y.; Cheng, C.L.; Wang, S.S.; Ho, H.C.; Chiu, K.Y.; Chen, C.S.; Chen, C.C.; Shiau, M.Y.; Ou, Y.C. Escin induces apoptosis in human renal cancer cells through G2/M arrest and reactive oxygen species-modulated mitochondrial pathways. Oncol. Rep. 2017, 37, 1002–1010. [Google Scholar] [CrossRef]
- Sun, D.; Shen, W.; Zhang, F.; Fan, H.; Tan, J.; Li, L.; Xu, C.; Zhang, H.; Yang, Y.; Cheng, H. α-Hederin Arrests Cell Cycle at G2/M Checkpoint and Promotes Mitochondrial Apoptosis by Blocking Nuclear Factor-κB Signaling in Colon Cancer Cells. Biomed. Res. Int. 2018, 2018, 2548378. [Google Scholar] [CrossRef] [PubMed]
- Schulte-Michels, J.; Wolf, A.; Aatz, S.; Engelhard, K.; Sieben, A.; Martinez-Osuna, M.; Haberlein, F.; Haberlein, H. α-Hederin inhibits G protein-coupled receptor kinase 2-mediated phosphorylation of β2-adrenergic receptors. Phytomedicine 2016, 23, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.A.; Xiang, S.S.; Li, H.F.; Wu, X.S.; Li, M.L.; Shu, Y.J.; Zhang, F.; Cao, Y.; Ye, Y.Y.; Bao, R.F.; et al. Cordycepin induces S phase arrest and apoptosis in human gallbladder cancer cells. Molecules 2014, 19, 11350–11365. [Google Scholar] [CrossRef] [PubMed]
- Salvador-Reyes, L.A.; Luesch, H. Biological targets and mechanisms of action of natural products from marine cyanobacteria. Nat. Prod. Rep. 2015, 32, 478–503. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Xu, Y.; Wei, Z.; Xin, G.; Xing, Z.; Niu, H.; Huang, W. Deoxyarbutin displays antitumour activity against melanoma in vitro and in vivo through a p38-mediated mitochondria associated apoptotic pathway. Sci. Rep. 2017, 7, 7197. [Google Scholar] [CrossRef] [PubMed]
- Wen, H.; Zhou, S.; Song, J. Induction of apoptosis by magnolol via the mitochondrial pathway and cell cycle arrest in renal carcinoma cells. Biochem. Biophys. Res. Commun. 2019, 508, 1271–1278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; He, W.; Shi, H.; Huang, X.; Ji, G. Natural compound oblongifolin C inhibits autophagic flux, and induces apoptosis and mitochondrial dysfunction in human cholangiocarcinoma QBC939 cells. Mol. Med. Rep. 2016, 14, 3179–3183. [Google Scholar] [CrossRef] [PubMed]
- Weidner, C.; Rousseau, M.; Micikas, R.J.; Fischer, C.; Plauth, A.; Wowro, S.J.; Siems, K.; Hetterling, G.; Kliem, M.; Schroeder, F.C.; et al. Amorfrutin C Induces Apoptosis and Inhibits Proliferation in Colon Cancer Cells through Targeting Mitochondria. J. Nat. Prod. 2016, 79, 2–12. [Google Scholar] [CrossRef]
- Bo, P.; Lien, J.C.; Chen, Y.Y.; Yu, F.S.; Lu, H.F.; Yu, C.S.; Chou, Y.C.; Yu, C.C.; Chung, J.G. Allyl Isothiocyanate Induces Cell Toxicity by Multiple Pathways in Human Breast Cancer Cells. Am. J. Chin. Med. 2016, 44, 415–437. [Google Scholar] [CrossRef]
- Zhang, Y. Allyl isothiocyanate as a cancer chemopreventive phytochemical. Mol. Nutr. Food Res. 2010, 54, 127–135. [Google Scholar] [CrossRef]
- Hafezi, K.; Hemmati, A.A.; Abbaszadeh, H.; Valizadeh, A.; Makvandi, M. Anticancer activity and molecular mechanisms of α-conidendrin, a polyphenolic compound present in Taxus yunnanensis, on human breast cancer cell lines. Phytother. Res. 2020, 34, 1397–1408. [Google Scholar] [CrossRef]
- Zhao, L.; Wen, Q.; Yang, G.; Huang, Z.; Shen, T.; Li, H.; Ren, D. Apoptosis induction of dehydrobruceine B on two kinds of human lung cancer cell lines through mitochondrial-dependent pathway. Phytomedicine 2016, 23, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Song, I.S.; Jeong, Y.J.; Kim, J.E.; Shin, J.; Jang, S.W. Frugoside Induces Mitochondria-Mediated Apoptotic Cell Death through Inhibition of Sulfiredoxin Expression in Melanoma Cells. Cancers 2019, 11, 854. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, S.R.; Mohamed, G.A.; Shaala, L.A.; Moreno, L.; Banuls, Y.; Kiss, R.; Youssef, D.T. Proceraside A, a new cardiac glycoside from the root barks of Calotropis procera with in vitro anticancer effects. Nat. Prod. Res. 2014, 28, 1322–1327. [Google Scholar] [CrossRef] [PubMed]
- Balachandran, C.; Emi, N.; Arun, Y.; Yamamoto, Y.; Ahilan, B.; Sangeetha, B.; Duraipandiyan, V.; Inaguma, Y.; Okamoto, A.; Ignacimuthu, S.; et al. In vitro anticancer activity of methyl caffeate isolated from Solanum torvum Swartz. fruit. Chem. Biol. Interact. 2015, 242, 81–90. [Google Scholar] [CrossRef]
- Han, X.; Deng, S.; Wang, N.; Liu, Y.; Yang, X. Inhibitory effects and molecular mechanisms of tetrahydrocurcumin against human breast cancer MCF-7 cells. Food Nutr. Res. 2016, 60, 30616. [Google Scholar] [CrossRef]
- Duan, H.; Wang, R.; Yan, X.; Liu, H.; Zhang, Y.; Mu, D.; Han, J.; Li, X. Phloretin induces apoptosis of human esophageal cancer via a mitochondria-dependent pathway. Oncol. Lett. 2017, 14, 6763–6768. [Google Scholar] [CrossRef]
- Liu, Z.; Ren, B.; Wang, Y.; Zou, C.; Qiao, Q.; Diao, Z.; Mi, Y.; Zhu, D.; Liu, X. Sesamol Induces Human Hepatocellular Carcinoma Cells Apoptosis by Impairing Mitochondrial Function and Suppressing Autophagy. Sci. Rep. 2017, 7, 45728. [Google Scholar] [CrossRef]
- Xia, T.; Zhang, J.; Han, L.; Jin, Z.; Wang, J.; Li, X.; Man, S.; Liu, C.; Gao, W. Protective effect of magnolol on oxaliplatin-induced intestinal injury in mice. Phytother. Res. 2019, 33, 1161–1172. [Google Scholar] [CrossRef]
- Li, X.; Huang, J.M.; Wang, J.N.; Xiong, X.K.; Yang, X.F.; Zou, F. Combination of chrysin and cisplatin promotes the apoptosis of Hep G2 cells by up-regulating p53. Chem. Biol. Interact. 2015, 232, 12–20. [Google Scholar] [CrossRef]
- Feng, F.; Cheng, P.; Wang, C.; Wang, Y.; Wang, W. Polyphyllin I and VII potentiate the chemosensitivity of A549/DDP cells to cisplatin by enhancing apoptosis, reversing EMT and suppressing the CIP2A/AKT/mTOR signaling axis. Oncol. Lett. 2019, 18, 5428–5436. [Google Scholar] [CrossRef]
- Zhang, K.; Li, Y. Effects of ginsenoside compound K combined with cisplatin on the proliferation, apoptosis and epithelial mesenchymal transition in MCF-7 cells of human breast cancer. Pharm. Biol. 2016, 54, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Ma, J.; Liu, Y.; He, P.; Dong, W. Combining α-Hederin with cisplatin increases the apoptosis of gastric cancer in vivo and in vitro via mitochondrial related apoptosis pathway. Biomed. Pharmacother. 2019, 120, 109477. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Fan, J.; Ai, G.; Liu, J.; Luo, N.; Li, C.; Cheng, Z. Berberine in combination with cisplatin induces necroptosis and apoptosis in ovarian cancer cells. Biol. Res. 2019, 52, 37. [Google Scholar] [CrossRef] [PubMed]
- He, G.; He, G.; Zhou, R.; Pi, Z.; Zhu, T.; Jiang, L.; Xie, Y. Enhancement of cisplatin-induced colon cancer cells apoptosis by shikonin, a natural inducer of ROS in vitro and in vivo. Biochem. Biophys. Res. Commun. 2016, 469, 1075–1082. [Google Scholar] [CrossRef]
- Zhang, T.; Ma, L.; Wu, P.; Li, W.; Li, T.; Gu, R.; Dan, X.; Li, Z.; Fan, X.; Xiao, Z. Gallic acid has anticancer activity and enhances the anticancer effects of cisplatin in nonsmall cell lung cancer A549 cells via the JAK/STAT3 signaling pathway. Oncol. Rep. 2019, 41, 1779–1788. [Google Scholar]
- Huang, Z.; Yang, G.; Shen, T.; Wang, X.; Li, H.; Ren, D. Dehydrobruceine B enhances the cisplatin-induced cytotoxicity through regulation of the mitochondrial apoptotic pathway in lung cancer A549 cells. Biomed. Pharmacother. 2017, 89, 623–631. [Google Scholar] [CrossRef]
- Feng, X.Q.; Rong, L.W.; Wang, R.X.; Zheng, X.L.; Zhang, L.; Zhang, L.; Lin, Y.; Wang, X.; Li, Z.P. Luteolin and sorafenib combination kills human hepatocellular carcinoma cells through apoptosis potentiation and JNK activation. Oncol. Lett. 2018, 16, 648–653. [Google Scholar] [CrossRef]
- Kutkowska, J.; Strzadala, L.; Rapak, A. Sorafenib in Combination with Betulinic Acid Synergistically Induces Cell Cycle Arrest and Inhibits Clonogenic Activity in Pancreatic Ductal Adenocarcinoma Cells. Int. J. Mol. Sci. 2018, 19, 3234. [Google Scholar] [CrossRef]
- Xia, Y.; Li, Y.; Westover, K.D.; Sun, J.; Chen, H.; Zhang, J.; Fisher, D.E. Inhibition of Cell Proliferation in an NRAS Mutant Melanoma Cell Line by Combining Sorafenib and alpha-Mangostin. PLoS ONE 2016, 11, e0155217. [Google Scholar] [CrossRef]
- Liu, Y.; Bi, T.; Dai, W.; Wang, G.; Qian, L.; Shen, G.; Gao, Q. Lupeol enhances inhibitory effect of 5-fluorouracil on human gastric carcinoma cells. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2016, 389, 477–484. [Google Scholar] [CrossRef]
- Khan, N.; Jajeh, F.; Eberhardt, E.L.; Miller, D.D.; Albrecht, D.M.; Van Doorn, R.; Hruby, M.D.; Maresh, M.E.; Clipson, L.; Mukhtar, H.; et al. Fisetin and 5-fluorouracil: Effective combination for PIK3CA-mutant colorectal cancer. Int. J. Cancer 2019, 145, 3022–3032. [Google Scholar] [CrossRef] [PubMed]
- Attoub, S.; Arafat, K.; Khalaf, T.; Sulaiman, S.; Iratni, R. Frondoside A Enhances the Anti-Cancer Effects of Oxaliplatin and 5-Fluorouracil on Colon Cancer Cells. Nutrients 2018, 10, 560. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Yu, H.H.; Liu, Y.S.; Wang, Y.S.; Zhao, W.H. Esculetin enhances the inhibitory effect of 5-Fluorouracil on the proliferation, migration and epithelial-mesenchymal transition of colorectal cancer. Cancer Biomark. 2019, 24, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Bashmail, H.A.; Alamoudi, A.A.; Noorwali, A.; Hegazy, G.A.; AJabnoor, G.; Choudhry, H.; Al-Abd, A.M. Thymoquinone synergizes gemcitabine anti-breast cancer activity via modulating its apoptotic and autophagic activities. Sci. Rep. 2018, 8, 11674. [Google Scholar] [CrossRef]
- Aborehab, N.M.; Osama, N. Effect of Gallic acid in potentiating chemotherapeutic effect of Paclitaxel in HeLa cervical cancer cells. Cancer Cell Int. 2019, 19, 154. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Li, Y.; Shi, Y.; Zhang, Y. Enhanced inhibition of urinary bladder cancer growth and muscle invasion by allyl isothiocyanate and celecoxib in combination. Carcinogenesis 2013, 34, 2593–2599. [Google Scholar] [CrossRef]
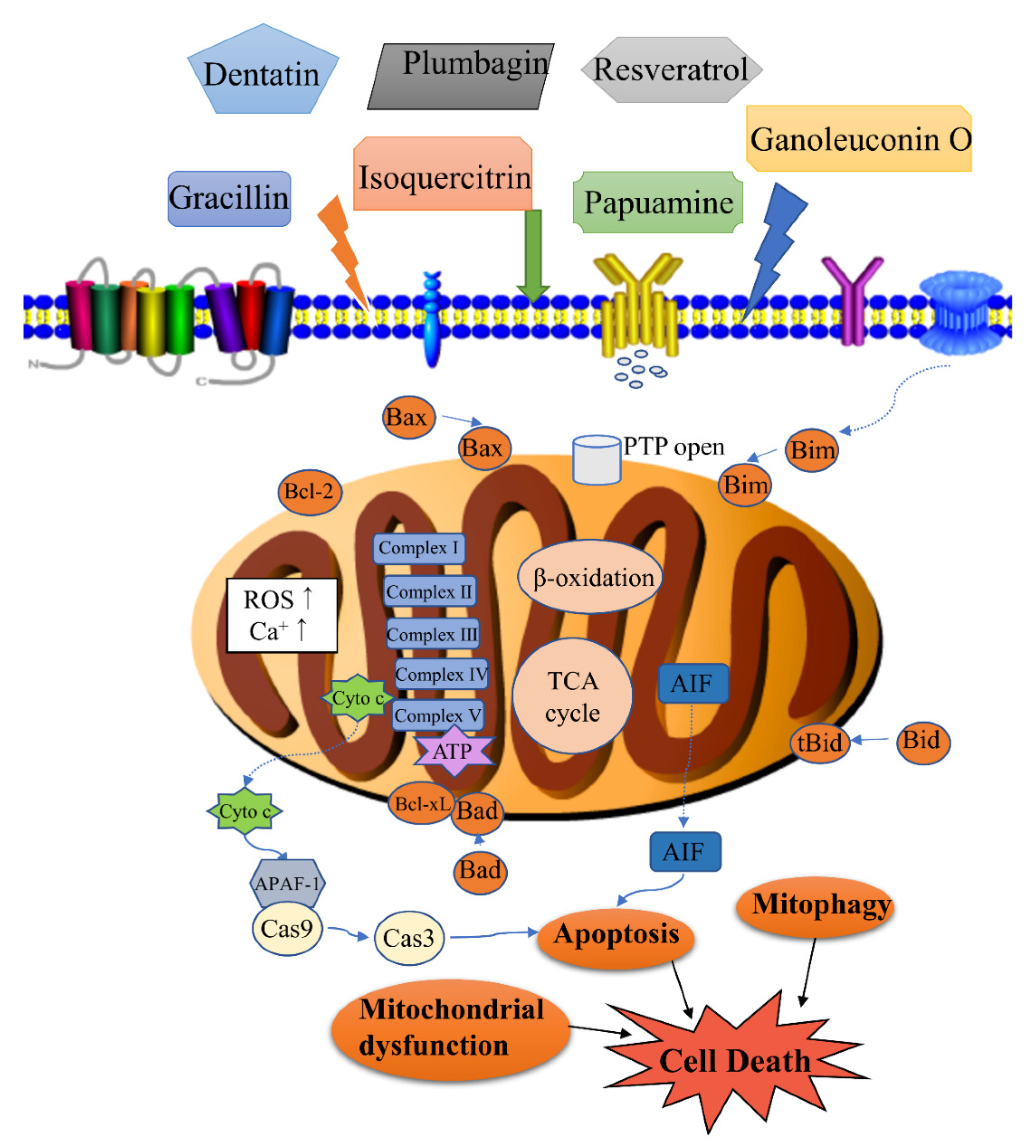
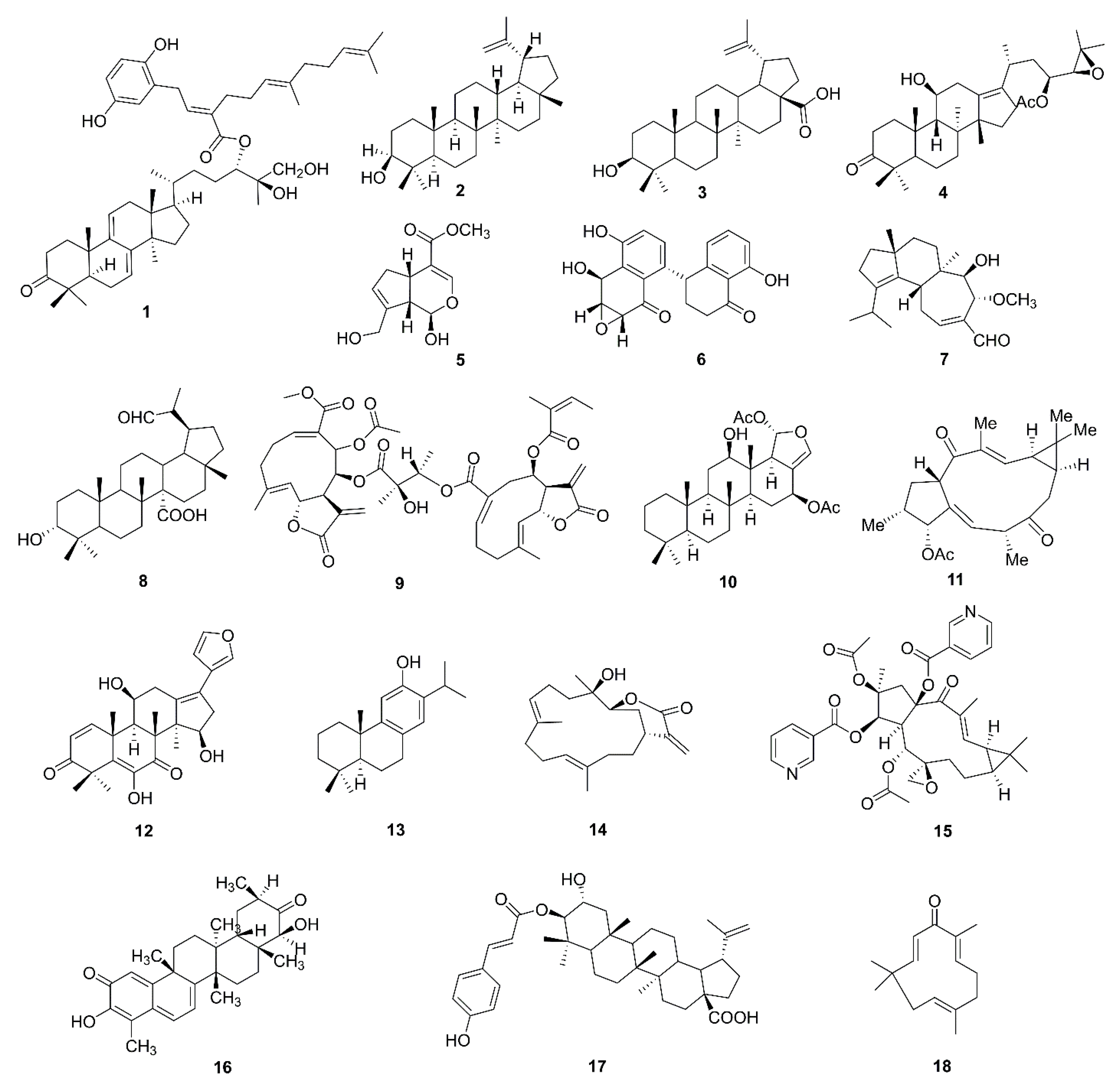
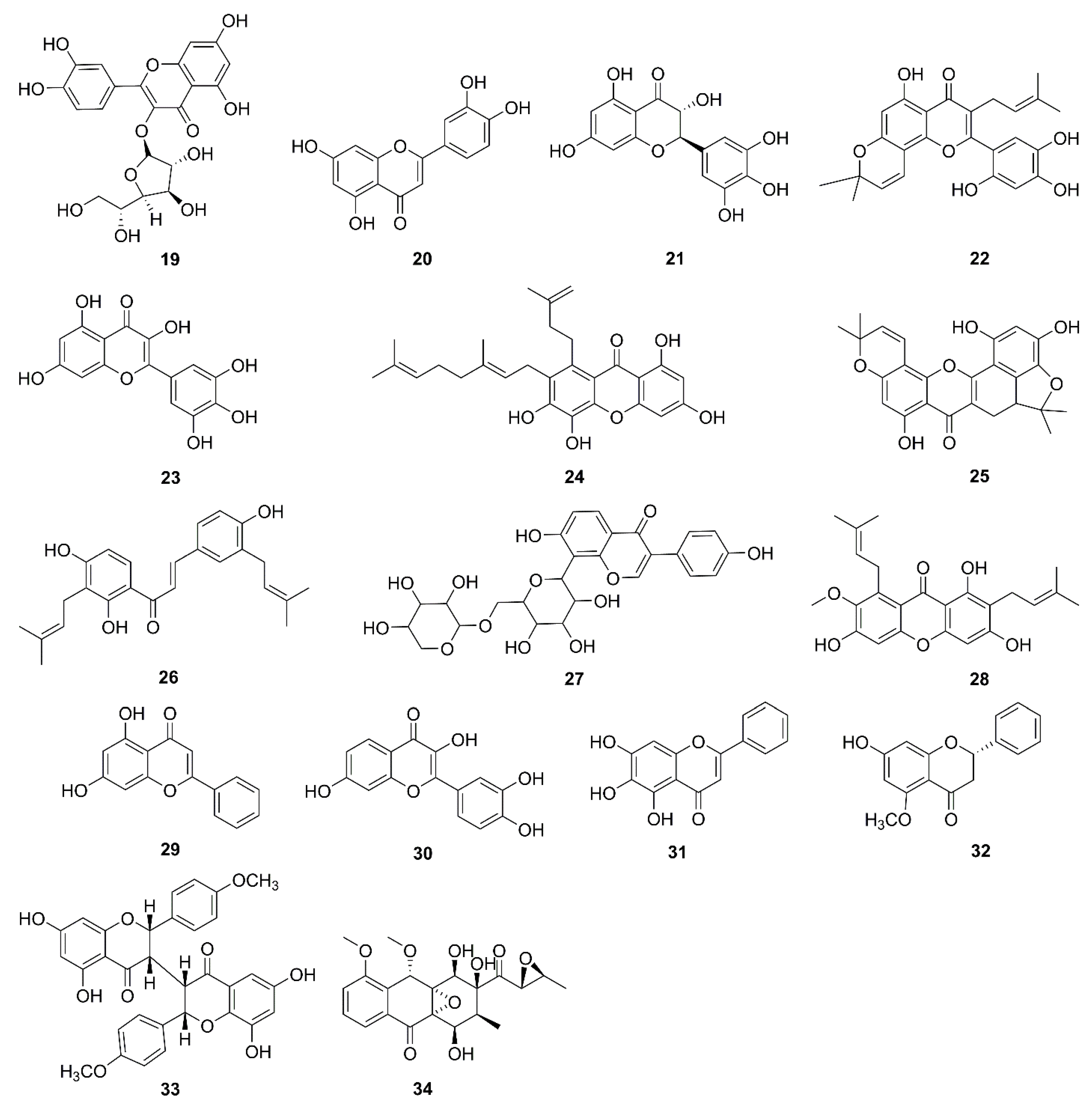
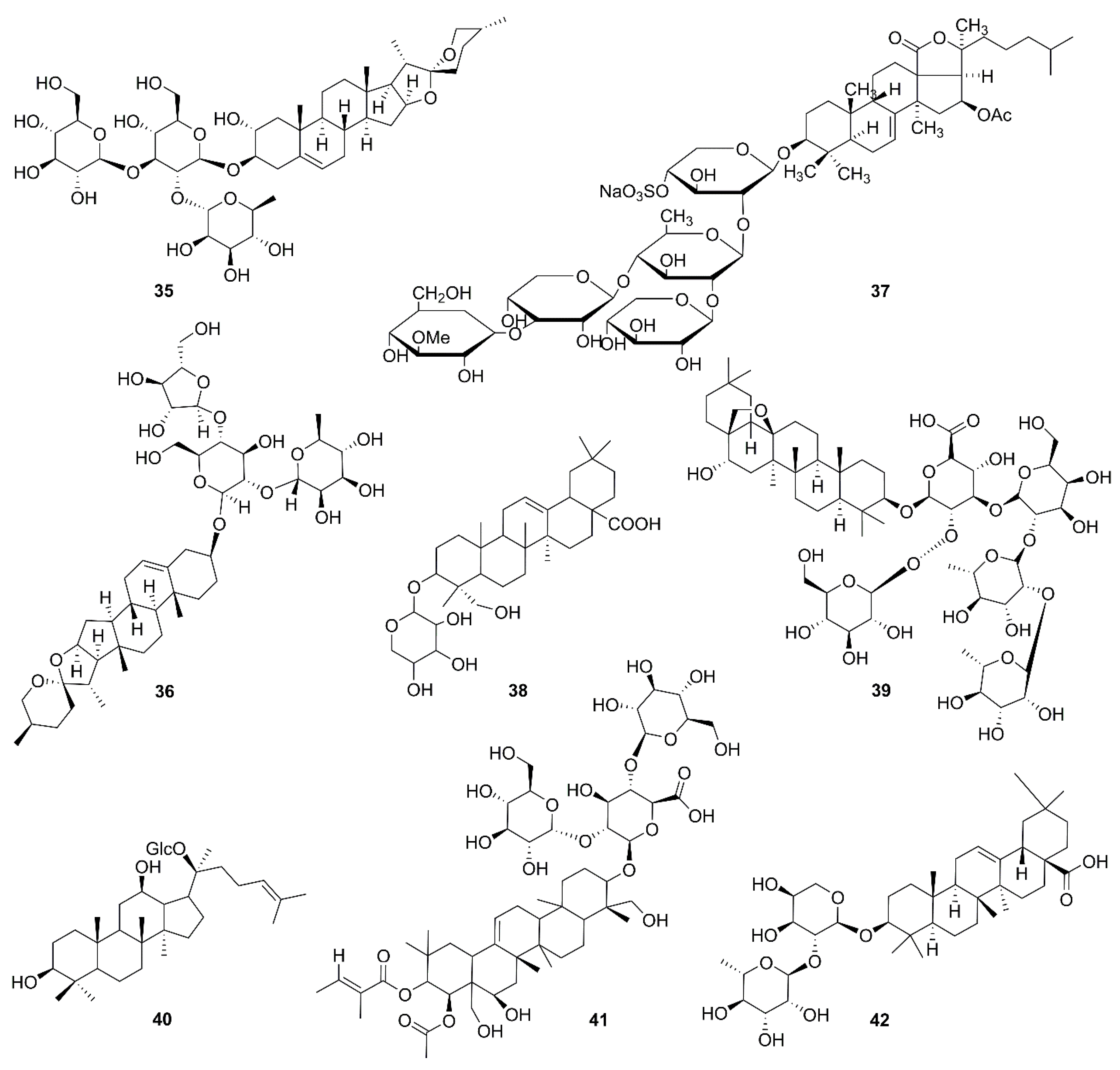
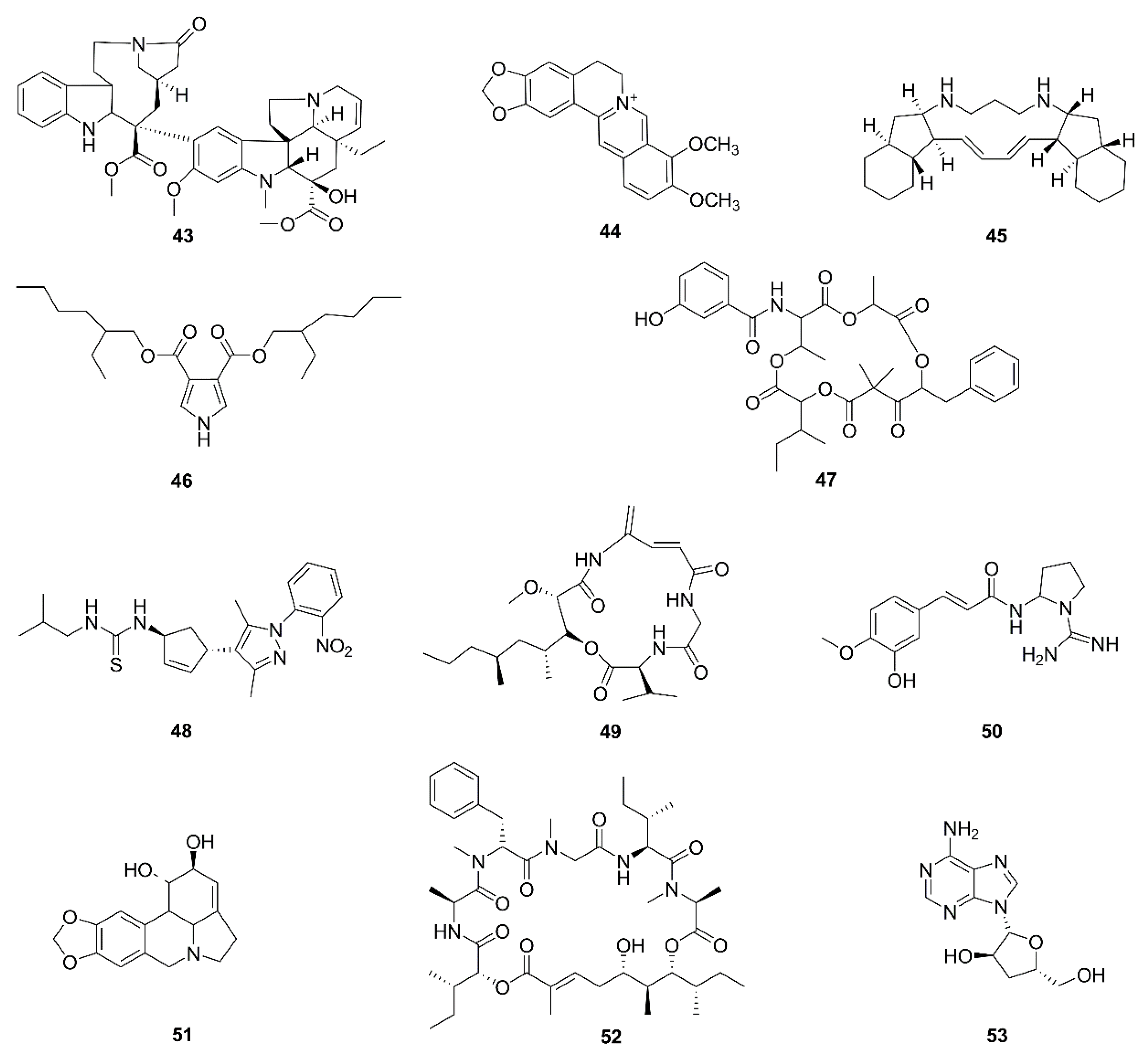

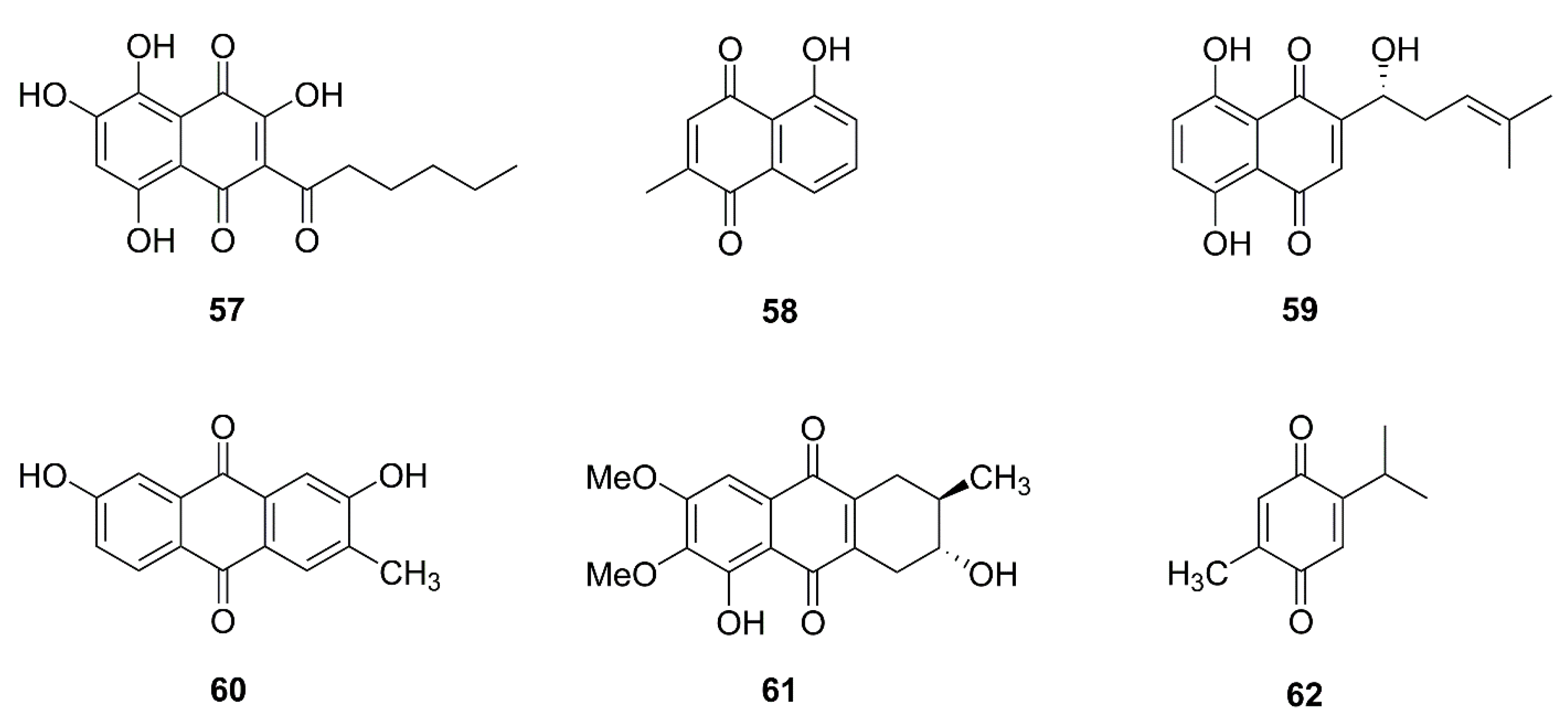
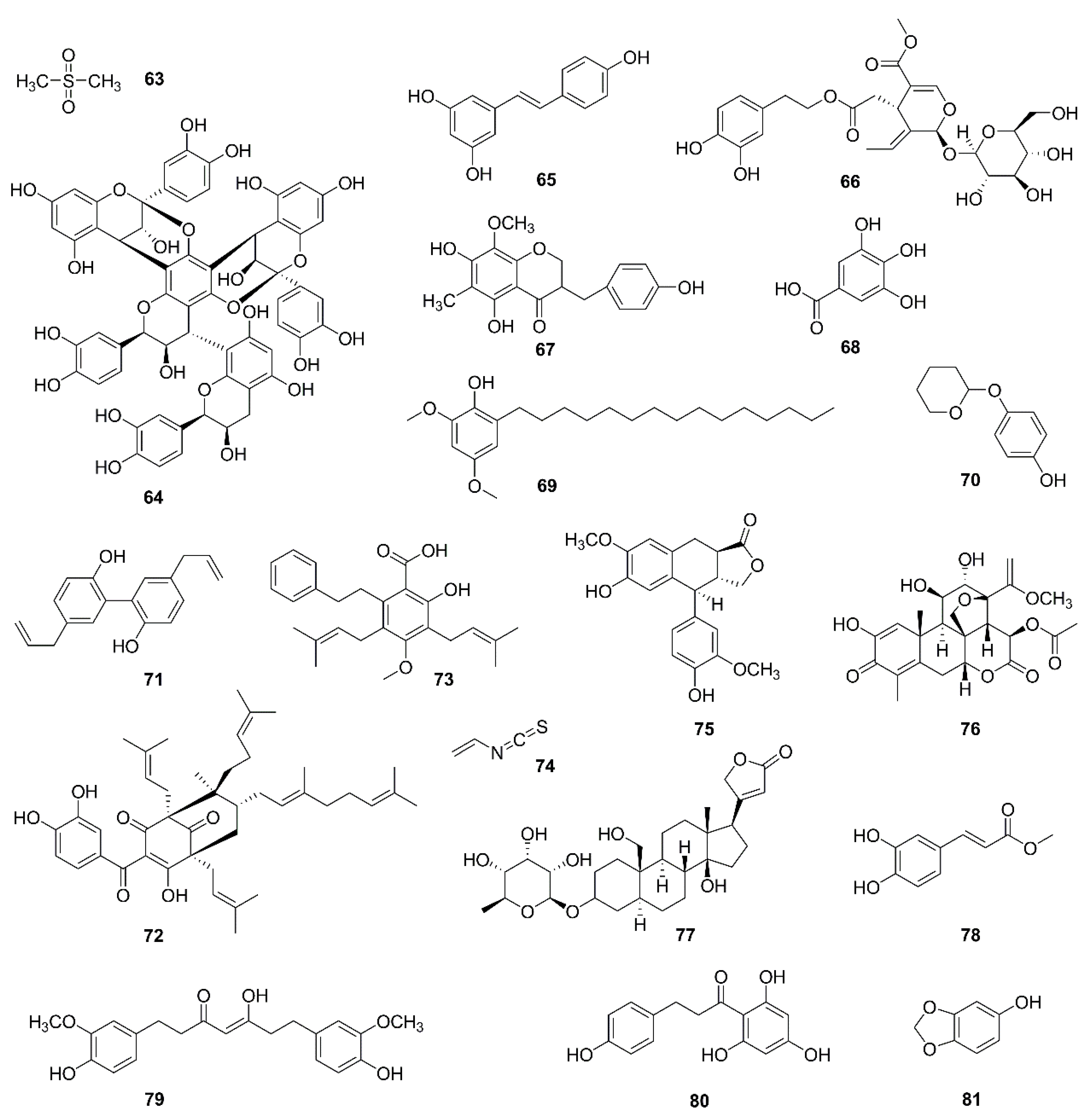
| No. | Isolated Compound | Origin | Cell Line | Mechanism | Reference |
|---|---|---|---|---|---|
| Terpenoids | |||||
| 1 | Ganoleuconin O | Ganoderma leucocontextum | Huh7.5 | Fatty acid immobilization, loss of the mitochondrial lipid cardiolipin | [30] |
| 2 | Lupeol | Bombax ceiba | SK-RC-45 | Mitochondrial hyper fission | [31] |
| 3 | Betulinic acid | Betula alba | HeLa | Cardiolipin modification, ROS generation, Bad, caspase 9 | [32,33] |
| 4 | Alisol B-23-acetate | Alisma orientale | A549, NCI-H292 | ROS generation, Bcl-2↓, Bax↑, activation of caspase-3, -9, release of cytochrome c/AIF | [34] |
| 5 | Genipin | Gardenia jasminoides | N18TG2 | Activation of dicarboxylate carrier, decreased activity of UCP1, UCP3, and complex III of the respiratory chain, UCP2 inhibition | [35] |
| 6 | Alternol | Yew tree | PC-3 | Decrease of mitochondrial respiration, isocitric acid, fumaric acid and malic acid, ATP production | [36,37] |
| 7 | Cyathin Q | Cyathus africanus | HCT116 | Bcl-2↓, Bax↑, Bcl-xL↓, ROS generation, release of cytochrome c | [38] |
| 8 | 3α-hydroxy-19α-hydrogen-29-aldehyde-27-lupanoic acid | Potentilla discolor | HepG2 | Bcl-2↓, Bax↑, release of cytochrome c | [39] |
| 9 | Uvedafolin | Smallanthus sonchifolius | HeLa | MMP loss, release of cytochrome c | [40] |
| 10 | Heteronemin | Hippospongia sp. | Molt4 | ROS generation | [41] |
| 11 | Jatrogossone A | Jatropha gossypiifolia | KOPN-8 | MMP loss, ROS generation | [42] |
| 12 | Walsuronoid B | Walsura robusta | Bel-7402, HepG2 | ROS generation, mitochondrial and lysosomal dysfunction | [43] |
| 13 | Ferruginol | Podocarpus ferruginea | MDA-T32 | ROS generation, MMP loss, Bcl-2↓ | [44,45] |
| 14 | Lobocrassin B | Lobophytum crassum | CL1-5, H520, BEAS-2B | Bcl-2↓, Bax↑, ROS generation, MMP loss, release of cytochrome c, activation of caspase-3 | [46] |
| 15 | Aellinane | Euphorbia aellenii | Caov-4 | Bcl-2↓, Bax↑, ROS generation, MMP loss | [47] |
| 16 | Tingenin B | Maytenus sp. | MCF-7s | Bcl-2↓, Bax↑, MMP loss | [48] |
| 17 | 3-O-trans-p-coumaroyl alphitolic acid | Ziziphus jujuba | PC-3 | ROS generation | [49] |
| 18 | Zerumbone | Zingiber zerumbet | PC-3, DU-145 | Tubulin binding and crosstalk between endoplasmic reticulum stress and mitochondrial insult | [50,51] |
| Flavonoids | |||||
| 19 | Isoquercitrin | Hibiscus cannabinus | MDA-MB-231 | LSD1-induced mitochondrial-mediated apoptosis pathway | [52,53] |
| 20 | Luteolin | Cauliflower, peanut, and carrot | SW1990 | Inhibitor of Bcl-2, mitochondrial permeabilization | [54] |
| 21 | Dihydromyricetin | Ampelopsis grossedentata | HepG2 | Akt/Bad signal pathway, mitochondrial apoptotic pathway, Bax↑, Bad↑, inhibition of the phosphorylation of Bad at Ser136 and Ser112 | [55,56] |
| 22 | Artonin E | Artocarpus elasticus | SKOV-3 | Release of cytochrome c, Activation of caspases-3, -8, and -9, Bax↑, Bcl-2↓, HSP70↓, survivin↓ | [57] |
| 23 | Myricetin | Fruits and vegetables | SNU-80 | Bax/Bcl-2↑, release of AIF | [58] |
| 24 | Xanthones | Garcinia xanthochymus | HepG2 | Bax↑, Bcl-2↓, Bcl-xL↓, Mcl-1↓, and survivin↓ | [59] |
| 25 | Cycloartobiloxanthone | Artocarpus gomezianus | H460 | Bax↑, Bcl-2↓, Mcl-1↓ | [60] |
| 26 | Paratocarpin E | Euphorbia humifusa | MCF-7 | Bax↑, Bcl-2↓, release of cytochrome c | [61] |
| 27 | Puerarin 6′’-O-xyloside | Pueraria lobata | SW480 | Bax↑, Bad↑, Bcl-2↓, caspase-3 and -9 activation | [62] |
| 28 | α-mangostin | Cratoxylum arborescens | HeLa | ROS generation, MMP loss, release of cytochrome c | [63] |
| 29 | Chrysin | Honey and propolis | Mitochondria isolated from hepatocytes of HCC rats | ROS generation, MMP loss, release of cytochrome c, swelling in mitochondria | [64,65] |
| 30 | Fisetin | Strawberries, apples, grapes, onions, and cucumbers | SCC-4 | ROS generation, Ca2+ production, MMP loss, Bcl-2↓, Bax↑, Bid↑, release of cytochrome c, AIF, and Endo G | [66,67] |
| 31 | Baicalein | Scutellaria baicalensis, Scutellaria radix | A2780 | Combination therapy with baicalein and taxol had much higher antitumor effects compared with the monotherapy. Release of cytochrome c, and caspase-3 and -9 activation | [68,69] |
| 32 | Alpinetin | Zingiberaceous plants | A549 | Bcl-2↓, Bax↑, Bcl-xL↓, XIAP↓, PI3K/Akt signaling pathway, sensitized drug-resistant lung cancer cells | [70,71] |
| 33 | Chamaejasmin B | Stellerachamaejasme | KB, KBV200 | Bcl-2↓, Bax↑, MMP loss, release of cytochrome c and AIF | [72] |
| 34 | Mensacarcin | Streptomyces bacteria | SK-Mel-28, SK-Mel-5, HCT-116 | Release of cytochrome c, energy production and mitochondrial function rapidly disturbed | [73] |
| Saponins | |||||
| 35 | Gracillin | Dioscorea gracillima | H226B, H460 | Targeting mitochondrial complex II, suppressing ATP synthesis, ROS generation | [74] |
| 36 | Polyphyllin I | Paris polyphylla | MDA-MB-231 | Mitochondrial translocation of DRP1, mitochondrial fission, release of cytochrome c, mitochondrial PTEN-induced kinase 1↑ | [75,76] |
| 37 | Frondoside A | Cucumaria frondosa | CA46 | Bcl-2↓, survivin↓, release of HtrA2/Omi and cytochrome c, ROS generation | [77] |
| 38 | 3β-O-α-l- arabinopyranoside | Clematis ganpiniana | MCF-7, MDA-MB-231 | Release of cytochrome c and Apaf-1, upregulation of caspase-9 and caspase-3 | [78] |
| 39 | Sakuraso-saponin | Aegiceras corniculatum | LNcaP, 22RV-1, C4-2 | Bcl-xL↓ | [79,80] |
| 40 | Ginsenoside compound K | Panax ginseng | SK-N-BE(2), SH-SY5Y | Bcl-2↓, Bcl-xL↓ | [81] |
| 41 | Escin | Aesculus hippocastanum | 786-O, Caki-1 | G2/M arrest and ROS-modulated mitochondrial pathways | [82] |
| 42 | α-Hederin | Hedera helix | SW620 | NF-κB signaling pathway, Bcl-2↓, Bax↑, release of cytochrome c | [83,84] |
| Alkaloids | |||||
| 43 | Cathachunine | Catharanthus roseus | HL60 | ROS-dependent mitochondria-mediated intrinsic pathway, Bcl-2/Bax↓, ROS generation, MMP loss, release of cytochrome c | [85] |
| 44 | Berberine | Rhizoma coptidis | T98G, LN18 | ERK1/2-mediated impairment of mitochondrial aerobic respiration | [86,87] |
| 45 | Papuamine | Haliclona sp. | H1299 | Intracellular ATP depleted by causing mitochondrial dysfunction, mitochondrial superoxide production | [88] |
| 46 | Bis (2-ethyl hexyl) 1H-pyrrole-3, 4-dicarboxylate | Tinospora cordifolia | MDA-MB-231 | ROS generation, increase in intracellular calcium, phosphorylation of p53, mitochondrial membrane depolarization, MPTP, and cardiolipin peroxidation, Bcl-2↓, Bax↑, release of cytochrome c, caspase activation, DNA fragmentation | [89] |
| 47 | Unantimycin A | Found in the fraction library of microbial metabolites | Semi-intact cells with specific substrates for each complex of the mitochondrial electron transport chain | Targeted inhibition of mitochondrial complex I | [90] |
| 48 | NPL40330 | Found in chemical library | Semi-intact cells with specific substrates for each complex of the mitochondrial electron transport chain | Targeted inhibition of mitochondrial complex III | [90] |
| 49 | Boholamide A | Marine mollusks | U87MG | Influx of Ca2+ | [91] |
| 50 | Cernumidine | Solanum cernuum | T24 | Cytotoxicity and chemosensitizing effect of cernumidine to cisplatin. Bcl-2↓, Bax↑, MMP loss | [92] |
| 51 | Lycorine | Amaryllidaceae plant family | HepG2 | mPTP opening, MMP loss, ATP depletion, release of Ca2+ and cytochrome c, caspase activation | [93] |
| 52 | Lagunamides A | Lyngbya majuscule | A549 | MMP loss, ROS generation | [94] |
| 53 | Cordycepin | Cordyceps | OVCAR-3 | Downregulation of mitochondrial function and limitation of energy production; metastasis and migration suppressed | [95,96] |
| Coumarins | |||||
| 54 | 2,3-Dihydro-7- hydroxy-2R*,3R*- dimethyl-2-[4,8-dimethyl-3(E),7- nonadienyl]-furo[3,2-c]coumarin | Ferula ferulaeoides | C6 | MMP loss, Bcl-xL↓, Bcl-2↓, Bax↑, cleavage of Bid, FAS↑, FADD↑ | [97] |
| 55 | Dentatin | Clausena excavate | HepG2 | Bcl-xL↓, Bcl-2↓, Bax↑, release of cytochrome c | [98,99] |
| 56 | Aesculetin | Cortex Fraxini | THP-1 | Bcl-2↓, Bax↑ | [100] |
| Quinones | |||||
| 57 | Quambalarine B | Quambalaria cyanescens | Jurkat E6.1 | Inhibition of mitochondrial complex I and II, inhibition of mitochondrial respiration, metabolism reprogramming | [101,102] |
| 58 | Plumbagin | Plumbago zeylanica | MG63 | ROS generation, Bcl-2↓, Bax↑, Bcl-xL↓, and Bak↓, endoplasmic reticulum stress | [103] |
| 59 | Shikonin | Lithospermum erythrorhizon | HGC-27 | Bcl-2↓, Bax↑, survivin↓ | [104] |
| 60 | 2,7-dihydroxy-3-methylanthraquinone | Hedyotis diffusa | SGC-7901 | Bcl-xl↓, Bcl-2↓, Bax↑, Bad↑, release of cytochrome c | [105] |
| 61 | 3-hydroxy-1,5,6-trimethoxy-2-methyl-9,10-anthraquinone | Prismatomeris connate | A549, H1299 | Bcl-2↓, Mcl-1↓, Bax↑ | [106] |
| 62 | Thymoquinone | Nigella sativa | T24, 253J | Bcl-2↓, Bax↑, release of cytochrome c and AIF | [107] |
| Miscellanea | |||||
| 63 | Methylsulfonylmethane | Fruits and vegetables | YD-38 | Bcl-xL↓, Bcl-2↓, Bax↑, release of cytochrome c, MMP loss | [108,109] |
| 64 | Parameritannin A-2 | Urceola huaitingii | HGC27 | Enhanced doxorubicin-induced mitochondria-dependent apoptosis, inhibition of the PI3K/Akt, ERK1/2 and p38 pathways, Bcl-2↓, Bcl-xl↓, Bax↑, Bid↑, release of cytochrome c, caspase activation | [110] |
| 65 | Resveratrol | Polygonum cuspidatum, Veratrum nigrum, Cassia obtusifolia | H838, H520; K562 | Enhanced antitumor activities of cisplatin; Induced apoptosis | [111,112] |
| 66 | Oleuropein | Olea europaea | H1299 | Bcl-2/Bax↓, release of cytochrome c, activation of caspase-3 | [113,114] |
| 67 | Homoisoflavanone-1 | Polygonatum odoratum | A549 | Mitochondria-caspase-dependent and ER stress pathways, Bcl-2/ Bak↓ | [115] |
| 68 | Gallic acid | Green tea, grapes, red wine | H446 | ROS-dependent mitochondrial apoptotic pathway | [116] |
| 69 | Hierridin b | Cyanobium sp. | HT-29 | Proteomics identified 21 differentially expressed proteins belonging to the categories protein folding/synthesis and cell structure and reduced mitochondrial activity and as confirmed by morphological analysis of mitochondrial parameters | [117,118] |
| 70 | Deoxyarbutin | Ecklonia cava | B16F10 | MMP loss, ATP depletion and ROS overload generation | [119] |
| 71 | Magnolol | Magnolia officinalis | OS-RC-2, 786-O | P53, Bcl-2/Bax↓, release of cytochrome c, caspase activation, ROS generation | [120] |
| 72 | Oblongifolin C | Garcinia yunnanensis | QBC939 | Mitochondrial dysfunction | [121] |
| 73 | Amorfrutin C | Glycyrrhiza foetida | HT-29 | mPTP opening, mitochondrial oxygen consumption and extracellular acidification increased | [122] |
| 74 | Allyl isothiocyanate | Cruciferous vegetables | MCF-7, MDA-MB-231 | ROS and Ca2+ production, MMP loss, release of cytochrome c, AIF, and Endo G, Bcl-2↓, Bax↑ | [123,124] |
| 75 | α-conidendrin | Taxus yunnanensis | MCF-7 and MDA-MB-231 | ROS generation, p53↑, Bax↑, Bcl-2↓, MMP loss, release of cytochrome c, activation of caspases-3 and -9 | [125] |
| 76 | Dehydrobruceine B | Brucea javanica | A549, NCI-H292 | MMP loss, release of cytochrome c, cleavage of caspase-9, caspase-3, and poly (ADP-ribose) polymerase (PARP) | [126] |
| 77 | Frugoside | Calotropis procera | M14, A375 | ROS generation | [127,128] |
| 78 | Methyl caffeate | Solanum torvum | MCF-7 | Bcl-2↓, Bax↑, Bid↑, p53↑, cleavage of caspase-3 and PARP, release of cytochrome c | [129] |
| 79 | Tetrahydrocurcumin | Curcuma longa | MCF-7 | ROS generation, Bcl-2↓, PARP↓, Bax↑, release of cytochrome c, MMP loss | [130] |
| 80 | Phloretin | Apple tree leaves and Manchurian apricot | EC-109 | Bcl-2↓, Bax↑ | [131] |
| 81 | Sesamol | Sesame seeds | HepG2 | Bcl-2↓, Bax↑, MMP loss, H2O2 production, PI3K Class III/Belin-1 pathway | [132] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.; He, P.-Y.; Zhang, Y.; Li, N. Natural Products Targeting the Mitochondria in Cancers. Molecules 2021, 26, 92. https://doi.org/10.3390/molecules26010092
Yang Y, He P-Y, Zhang Y, Li N. Natural Products Targeting the Mitochondria in Cancers. Molecules. 2021; 26(1):92. https://doi.org/10.3390/molecules26010092
Chicago/Turabian StyleYang, Yue, Ping-Ya He, Yi Zhang, and Ning Li. 2021. "Natural Products Targeting the Mitochondria in Cancers" Molecules 26, no. 1: 92. https://doi.org/10.3390/molecules26010092
APA StyleYang, Y., He, P.-Y., Zhang, Y., & Li, N. (2021). Natural Products Targeting the Mitochondria in Cancers. Molecules, 26(1), 92. https://doi.org/10.3390/molecules26010092