Cannabis sativa L. Inflorescences from Monoecious Cultivars Grown in Central Italy: An Untargeted Chemical Characterization from Early Flowering to Ripening
Abstract
1. Introduction
2. Results
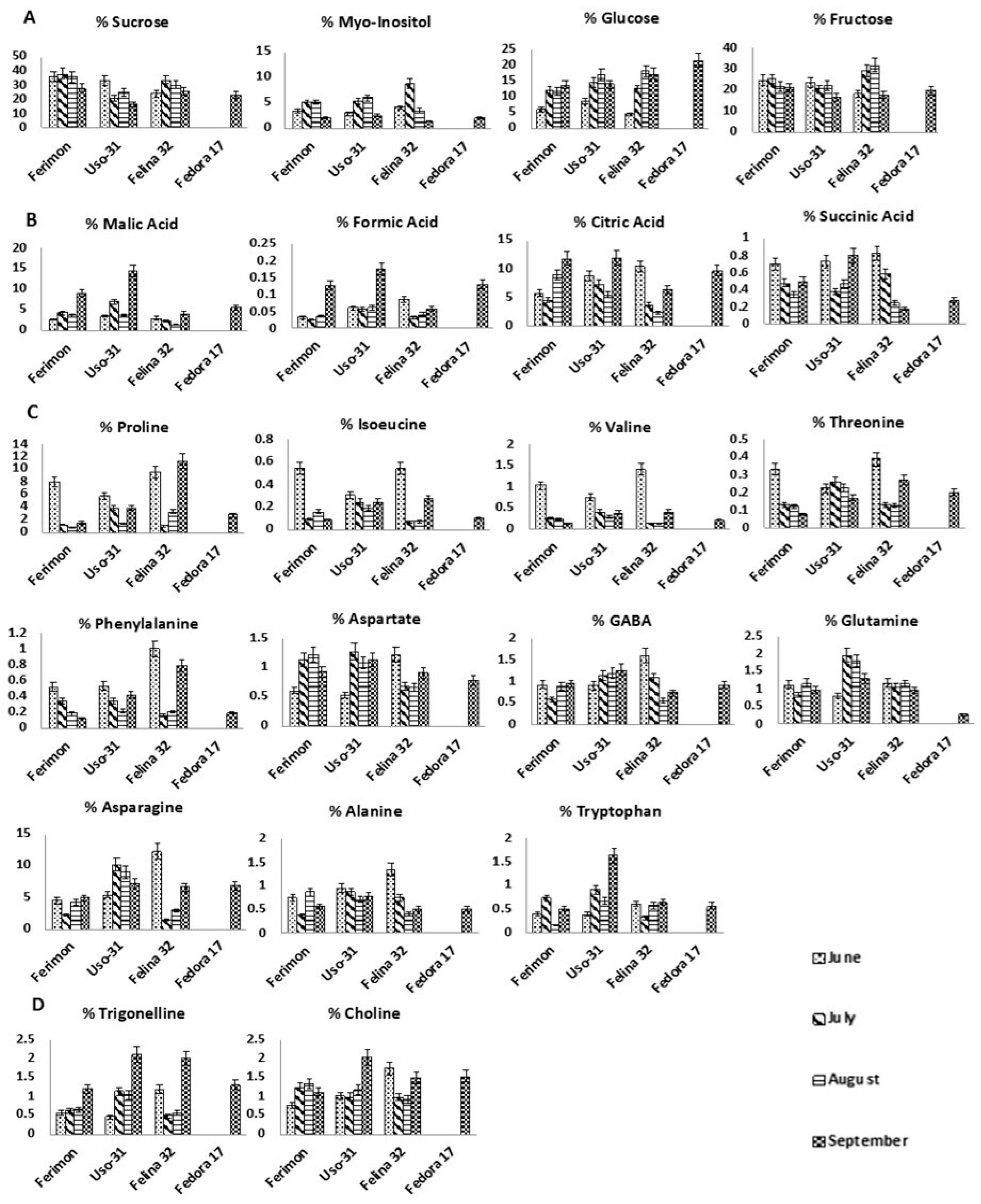
2.1. Chemical Profile of Hemp Inflorescences
2.2. Metabolic Profile over the Season of Ferimon, Uso-31, Felina 32 and Fedora 17 Inflorescences
2.2.1. Ferimon Cultivar
2.2.2. Uso-31 Cultivar
2.2.3. Felina 32 Cultivar
2.2.4. Fedora 17 Cultivar
3. Discussion
4. Materials and Methods
4.1. Chemicals and Solvents
4.2. Hemp Plant Material
4.3. Sample Preparation for NMR, GC/MS, Spectrophotometric and HPLC-PDA Analyses
4.4. Metabolic Profile by NMR Analysis
4.5. Cannabinoids Contents by UHPLC Analysis
4.6. Terpenoids Content by Gas Chromatography/Mass Spectrometry (GC/MS)
4.7. Total Phenolics, Tannins and Flavonoids by Spectrophotometric Methods
4.8. Phenolic Content by HPLC-PDA
4.9. Biogenic Amines (BAs) by HPLC-FD
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- EUR-Lex. Access to European Union Law. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:01999R1251-20040701 (accessed on 3 March 2020).
- Ingrao, C.; Lo Giudice, A.; Bacenetti, J.; Tricase, C.; Dotelli, G.; Fiala, M.; Siracusa, V.; Mbohwa, C. Energy and environmental assessment of industrial hemp for building applications: A review. Renew. Sustain. Energy Rev. 2015, 51, 29–42. [Google Scholar]
- Kreuger, E.; Prade, T.; Escobar, F.; Svensson, S.E.; Englund, J.E.; Björnsson, L. Anaerobic digestion of industrial hemp-Effect of harvest time on methane energy yield per hectare. Biomass Bioenergy 2011, 35, 893–900. [Google Scholar]
- Lühr, C.; Pecenka, R.; Budde, J.; Hoffmann, T.; Gusovius, H.J. Comparative investigations of fibreboards resulting from selected hemp varieties. Ind. Crop. Prod. 2018, 118, 81–94. [Google Scholar]
- Croxford, J.L.; Pryce, G.; Jackson, S.J.; Ledent, C.; Giovannoni, G.; Pertwee, R.G.; Yamamura, T.; Baker, D. Cannabinoid-mediated neuroprotection, not immunosuppression, may be more relevant to multiple sclerosis. J. Neuroimmunol. 2008, 193, 120–129. [Google Scholar] [PubMed]
- Appendino, G.; Gibbons, S.; Giana, A.; Pagani, A.; Grassi, G.; Stavri, M.; Smith, E.; Rahman, M.M. Antibacterial cannabinoids from Cannabis sativa: A structure-activity study. J. Nat. Prod. 2008, 71, 1427–1430. [Google Scholar]
- Burstein, S. Cannabidiol (CBD) and its analogs: A review of their effects on inflammation. Bioorganic Med. Chem. 2015, 23, 1377–1385. [Google Scholar]
- Vogl, C.R.; Mölleken, H.; Lissek-Wolf, G.; Surböck, A.; Kobert, J. Hemp (Cannabis sativa L.) as a resource for green cosmetics: Yield of seed and fatty acid compositions of 20 varieties under the growing conditions of organic farming in Austria. J. Ind. Hemp 2004, 9, 51–68. [Google Scholar]
- Callaway, J.C. Hempseed as a nutritional resource: An overview. Euphytica 2004, 140, 65–72. [Google Scholar]
- Stott, C.G.; Guy, G.W. Cannabinoids for the pharmaceutical industry. Euphytica 2004, 140, 83–93. [Google Scholar]
- Verma, R.S.; Padalia, R.C.; Verma, S.K.; Chauhan, A.; Darokar, M.P. The essential oil of “bhang” (Cannabis sativa L.) for non-narcotic applications. Curr. Sci. 2014, 107, 645–650. [Google Scholar]
- Thomas, B.F.; ElSohly, M.A. The Analytical Chemistry of Cannabis: Quality Assessment, Assurance, and Regulation of Medicinal Marijuana and Cannabinoid Preparations; Elsevier: Amsterdam, The Netherlands, 2015; ISBN 9780128046708. [Google Scholar]
- Campiglia, E.; Radicetti, E.; Mancinelli, R. Plant density and nitrogen fertilization affect agronomic performance of industrial hemp (Cannabis sativa L.) in Mediterranean environment. Ind. Crop. Prod. 2017, 100, 246–254. [Google Scholar]
- Bertoli, A.; Tozzi, S.; Pistelli, L.; Angelini, L.G. Fibre hemp inflorescences: From crop-residues to essential oil production. Ind. Crop. Prod. 2010, 32, 329–337. [Google Scholar] [CrossRef]
- Ascrizzi, R.; Ceccarini, L.; Tavarini, S.; Flamini, G.; Angelini, L.G. Valorisation of hemp inflorescence after seed harvest: Cultivation site and harvest time influence agronomic characteristics and essential oil yield and composition. Ind. Crop. Prod. 2019, 139. [Google Scholar] [CrossRef]
- Consiglio Regionale del Lazio. Interventi per promuovere la coltivazione della canapa (Cannabis Sativa) per scopi produttivi, alimentari ed ambientali e relative filiere. Available online: http://www.consiglio.regione.lazio.it/consiglio-regionale/?vw=leggiregionalidettaglio&id=9307&sv=vigente (accessed on 3 March 2020).
- Ingallina, C.; Capitani, D.; Mannina, L.; Carradori, S.; Locatelli, M.; Di Sotto, A.; Di Giacomo, S.; Toniolo, C.; Pasqua, G.; Valletta, A.; et al. Phytochemical and biological characterization of Italian “sedano bianco di Sperlonga” Protected Geographical Indication celery ecotype: A multimethodological approach. Food Chem. 2020, 309. [Google Scholar] [CrossRef]
- Sobolev, A.P.; Mannina, L.; Capitani, D.; Sanzò, G.; Ingallina, C.; Botta, B.; Fornarini, S.; Crestoni, M.E.; Chiavarino, B.; Carradori, S.; et al. A multi-methodological approach in the study of Italian PDO “Cornetto di Pontecorvo” red sweet pepper. Food Chem. 2018, 255, 120–131. [Google Scholar] [CrossRef]
- Sobolev, A.P.; Thomas, F.; Donarski, J.; Ingallina, C.; Circi, S.; Cesare Marincola, F.; Capitani, D.; Mannina, L. Use of NMR applications to tackle future food fraud issues. Trends Food Sci. Technol. 2019, 91, 347–353. [Google Scholar] [CrossRef]
- Sobolev, A.P.; Circi, S.; Capitani, D.; Ingallina, C.; Mannina, L. Molecular fingerprinting of food authenticity. Curr. Opin. Food Sci. 2017, 16, 59–66. [Google Scholar] [CrossRef]
- Happyana, N.; Kayser, O. Monitoring Metabolite Profiles of Cannabis sativa LTrichomes during Flowering Period Using 1H-NMR-Based Metabolomics and Real-Time PCR. Planta Med. 2016, 82, 1217–1223. [Google Scholar]
- Flores-Sanchez, I.J.; Peč, J.; Fei, J.; Choi, Y.H.; Dušek, J.; Verpoorte, R. Elicitation studies in cell suspension cultures of Cannabis sativa L. J. Biotechnol. 2009, 143, 157–168. [Google Scholar] [CrossRef]
- Choi, Y.H.; Kim, H.K.; Hazekamp, A.; Erkelens, C.; Lefeber, A.W.M.; Verpoorte, R. Metabolomic differentiation of Cannabis sativa cultivars using 1H-NMR spectroscopy and principal component analysis. J. Nat. Prod. 2004, 67, 953–957. [Google Scholar] [CrossRef]
- Nagy, D.U.; Cianfaglione, K.; Maggi, F.; Sut, S.; Dall’Acqua, S. Chemical Characterization of Leaves, Male and Female Flowers from Spontaneous Cannabis (Cannabis sativa L.) Growing in Hungary. Chem. Biodivers. 2019, 16. [Google Scholar] [CrossRef]
- Mazzoccanti, G.; Ismail, O.H.; D’Acquarica, I.; Villani, C.; Manzo, C.; Wilcox, M.; Cavazzini, A.; Gasparrini, F. Cannabis through the looking glass: Chemo- and enantio-selective separation of phytocannabinoids by enantioselective ultra high performance supercritical fluid chromatography. Chem. Commun. 2017, 53, 12262–12265. [Google Scholar] [CrossRef]
- Glória, M.B.A.; Tavares-Neto, J.; Labanca, R.A.; Carvalho, M.S. Influence of cultivar and germination on bioactive amines in soybeans (Glycine max L. Merril). J. Agric. Food Chem. 2005, 53, 7480–7485. [Google Scholar]
- Sikora, V.; Berenji, J.; Latković, D. Influence of agroclimatic conditions on content of main cannabinoids in industrial hemp (Cannabis sativa L.). Genetika 2011, 43, 449–456. [Google Scholar] [CrossRef]
- Brighenti, V.; Pellati, F.; Steinbach, M.; Maran, D.; Benvenuti, S. Development of a new extraction technique and HPLC method for the analysis of non-psychoactive cannabinoids in fibre-type Cannabis sativa L. (hemp). J. Pharm. Biomed. Anal. 2017, 143, 228–236. [Google Scholar] [CrossRef]
- Mandrioli, M.; Tura, M.; Scotti, S.; Toschi, T.G. Fast Detection of 10 Cannabinoids by RP-HPLC-UV Method in Cannabis sativa L. Molecules 2019, 24, 2113. [Google Scholar] [CrossRef]
- Pollastro, F.; Minassi, A.; Fresu, L.G. Cannabis Phenolics and their Bioactivities. Curr. Med. Chem. 2017, 25, 1160–1185. [Google Scholar] [CrossRef]
- Flores-Sanchez, I.J.; Verpoorte, R. Secondary metabolism in cannabis. Phytochem. Rev. 2008, 7, 615–639. [Google Scholar] [CrossRef]
- Zengin, G.; Menghini, L.; Sotto, A.D.; Mancinelli, R.; Sisto, F.; Carradori, S.; Cesa, S.; Fraschetti, C.; Filippi, A.; Angiolella, L.; et al. Chromatographic analyses, in vitro biological activities, and cytotoxicity of cannabis sativa l. Essential oil: A multidisciplinary study. Molecules 2018, 23, 3266. [Google Scholar] [CrossRef]
- Ferrante, C.; Recinella, L.; Ronci, M.; Menghini, L.; Brunetti, L.; Chiavaroli, A.; Leone, S.; Di Iorio, L.; Carradori, S.; Tirillini, B.; et al. Multiple pharmacognostic characterization on hemp commercial cultivars: Focus on inflorescence water extract activity. Food Chem. Toxicol. 2019, 125, 452–461. [Google Scholar] [CrossRef]
- Martínez-Villaluenga, C.; Gulewicz, P.; Pérez, A.; Frías, J.; Vidal-Valverde, C. Influence of lupin (Lupinus luteus L. cv. 4492 and Lupinus angustifolius L. var. zapaton) and fenugreek (Trigonella foenum-graecum L.) germination on microbial population and biogenic amines. J. Agric. Food Chem. 2006, 54, 7391–7398. [Google Scholar]
- Bartkiene, E.; Juodeikiene, G.; Vidmantiene, D. Nutritional quality of fermented defatted soya and flaxseed flours and their effect on texture and sensory characteristics of wheat sourdough bread. Int. J. Food Sci. Nutr. 2012, 63, 722–729. [Google Scholar] [CrossRef]
- Musarra, M.; Jirillo, R.; Rapa, M.; Vinci, G. Canapa sativa L. and Moringa oleifera as Naturally Functional Beverages: Innovative Trends. In Natural Beverages; Academic Press: Cambridge, MA, USA, 2019; pp. 243–265. [Google Scholar]
- Sánchez-Pérez, S.; Comas-Basté, O.; Rabell-González, J.; Veciana-Nogués, M.T.; Latorre-Moratalla, M.L.; Vidal-Carou, M.C. Biogenic amines in plant-origin foods: Are they frequently underestimated in low-histamine diets? Foods 2018, 7, 205. [Google Scholar] [CrossRef]
- Chandra, S.; Lata, H.; ElSohly, M.A.; Walker, L.A.; Potter, D. Cannabis cultivation: Methodological issues for obtaining medical-grade product. Epilepsy Behav. 2017, 70, 302–312. [Google Scholar] [CrossRef]
- BLIGH, E.G.; DYER, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Capitani, D.; Mannina, L.; Proietti, N.; Sobolev, A.P.; Tomassini, A.; Miccheli, A.; Di Cocco, M.E.; Capuani, G.; De Salvador, R.; Delfini, M. Monitoring of metabolic profiling and water status of Hayward kiwifruits by nuclear magnetic resonance. Talanta 2010, 82, 1826–1838. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are still available from the authors. |
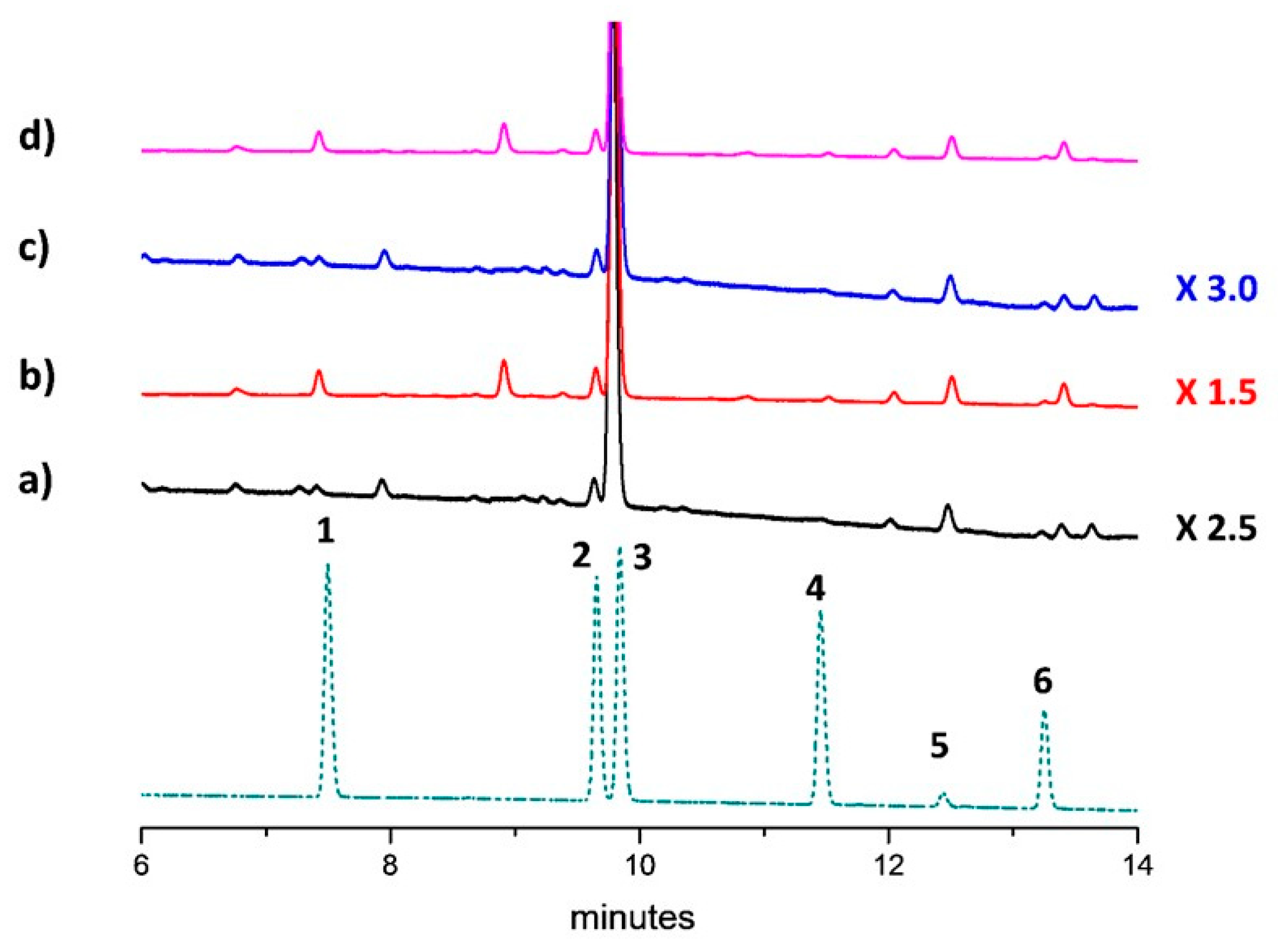
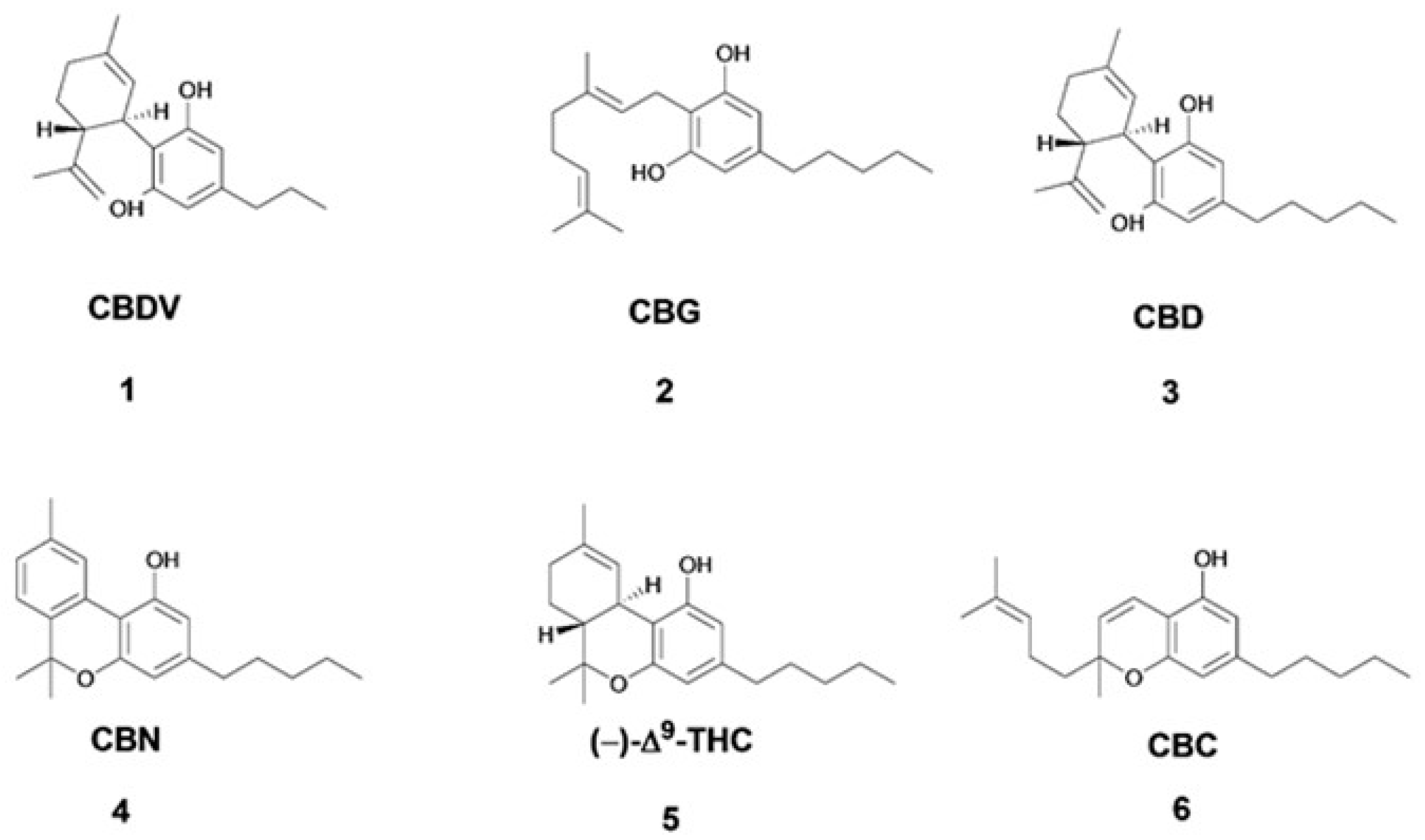
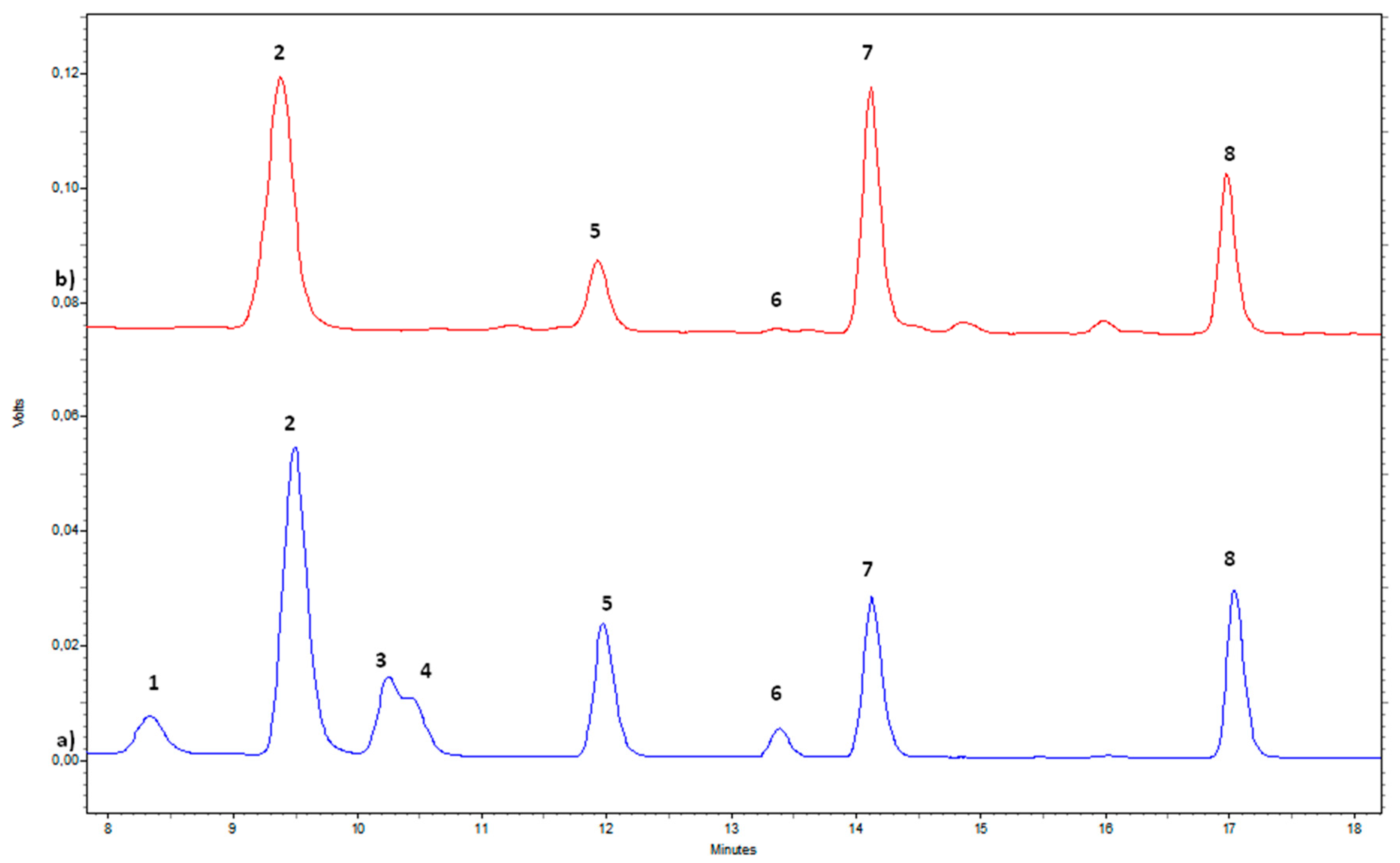
| Compound | Assignment | 1H (ppm) | Multiplicity [J(Hz)] | 13C (ppm) |
|---|---|---|---|---|
| Sugars | ||||
| α-d-Fructofuranose | CH-3 | 4.14 | 83.1 | |
| CH-5 | 4.07 * | 82.6 | ||
| β-d-Fructofuranose | CH-3 | 4.12 | 76.9 | |
| CH-4 | 4.12 | 75.9 | ||
| CH-5 | 3.85 | 81.7 | ||
| β-d-Fructopyranose | CH-3 | 3.81 | 67.1 | |
| CH-5 | 4.05 * | 66.8 | ||
| CH2-6,6′ | 3.71; 4.03 | 64.4 | ||
| α-Galactose | CH-1 | 5.28 | d [3.8] | 90.2 |
| CH-2 | 3.78 | |||
| CH-3 | 3.83 | |||
| CH-4 | 3.87 | |||
| CH-5 | 4.08 | |||
| β-Galactose | CH-1 | 4.60 | d [8.0] | 97.4 |
| CH-2 | 3.51 | |||
| CH-3 | 3.67 | |||
| CH-4 | 3.95 | |||
| CH-5 | 4.05 | |||
| CH-6 | 3.78 | |||
| α-Glucose | CH-1 | 5.25 * | d [3.8] | 93.1 |
| CH-2 | 3.56 | 72.2 | ||
| CH-3 | 3.74 | 73.8 | ||
| CH-4 | 3.45 | 70.7 | ||
| CH-5 | 3.84 | 72.5 | ||
| CH2-6,6′ | 3.86; 3.79 | 60.1 | ||
| β-Glucose | CH-1 | 4.66 * | d [8.0] | 97.0 |
| CH-2 | 3.27 | 75.2 | ||
| CH-3 | 3.51 | 76.8 | ||
| CH-4 | 3.43 | 70.7 | ||
| CH-5 | 3.48 | 75.1 | ||
| CH2-6,6′ | 3.90; 3.74 | 61.9 | ||
| Myo-Inositol | CH-2,5 | 3.56 | ||
| CH-3,6 | 3.65 | |||
| CH-4 | 3.30 * | 74.2 | ||
| Sucrose | CH-1 (Glucose) | 5.41 * | d [3.8] | 93.3 |
| CH-2 | 3.57 | 71.8 | ||
| CH-3 | 3.78 | 73.6 | ||
| CH-4 | 3.49 | 70.2 | ||
| CH-5 | 3.85 | 73.5 | ||
| CH2-6 | 3.83 | 63.5 | ||
| CH2-1′ (Fructose) | 3.69 | d [3.3] | 60.6 | |
| CH-3′ | 4.23 | d [8.7] | 77.5 | |
| CH-4′ | 4.06 | t [8.7] | 75.1 | |
| CH-5′ | 3.9 | 82.4 | ||
| CH2-6′ | 3.82 | 61.2 | ||
| Raffinose | CH-1 (Galactose) | 5.01 | d [3.8] | 99.4 |
| CH-2 | 3.85 | |||
| CH-3 | 3.91 | |||
| CH-4 | 4.03 | |||
| CH-1 (Glucose) | 5.44 | d [3.9] | ||
| CH-2 | 3.59 | |||
| CH-3 | 3.78 | |||
| CH-5 | 4.08 | |||
| CH-3 (Fructose) | 4.24 | d [8.7] | ||
| Organic acids | ||||
| Acetic acid | CH3 | 1.93 | s | 24.7 |
| COOH | 180.3 | |||
| Citric acid | α,γ-CH | 2.56 * | d [15.9] | 46.2 |
| α,γ′-CH | 2.69 | 46.2 | ||
| β-C | 74.2 | |||
| 1,5-COOH | 177.7 | |||
| 6-COOH | 180.2 | |||
| Formic acid | HCOOH | 8.47 * | s | |
| Fumaric Acid | α,β-CH=CH | 6.53 | s | |
| Malic acid | α-CH | 4.31 * | dd [9.8; 3.2] | 71.4 |
| β-CH | 2.70 | dd [15.6; 3.2] | 43.9 | |
| β′-CH | 2.39 | dd [15.6; 9.8] | 43.9 | |
| Succinic acid | α,β-CH2 | 2.42 * | s | 35.2 |
| Amino acids | ||||
| Alanine | α-CH | 3.81 | 51.6 | |
| β-CH3 | 1.49 * | d [7.3] | 17.2 | |
| COOH | 174.5 | |||
| Asparagine | α-CH | 4.02 | 52.3 | |
| β,β′-CH2 | 2.89; 2.97 * | 35.8 | ||
| Aspartate | α-CH | 3.91 | 52.3 | |
| β,β′-CH2 | 2.72; 2.82 * | dd [3.9; 17.4] | 37.5 | |
| γ-Aminobutyrate | α-CH2 | 2.31 * | t [7.4] | 37.2 |
| β-CH2 | 1.92 | 24.6 | ||
| γ-CH2 | 3.04 | t [7.6] | 40.2 | |
| Glutamine | α-CH | 3.78 | 55.9 | |
| β,β′-CH2 | 2.18 | m | 27.3 | |
| γ-CH | 2.46 * | m | 31.8 | |
| Isoleucine | α-CH | 3.69 | ||
| β-CH | 1.98 | |||
| γ-CH3 | 1.02 * | d [7.0] | 15.8 | |
| δ-CH3 | 0.94 | |||
| Leucine | α-CH | 3.77 | ||
| β-CH2 | 1.74 | |||
| γ-CH | 1.71 | |||
| δ-CH3 | 0.97 | 23.1 | ||
| δ′-CH3 | 0.96 | 22.0 | ||
| Phenylalanine | CH-2,6 | 7.34 | 130.5 | |
| CH-4 | 7.38 | 128.7 | ||
| CH-3,5 | 7.43 * | m | 130.2 | |
| Proline | α-CH | 4.14 | 62.4 | |
| γ-CH2 | 2.01 * | m | 24.9 | |
| Threonine | α-CH | 3.62 | 61.4 | |
| β-CH | 4.28 | 68.1 | ||
| γ-CH3 | 1.34 * | d [6.6] | 18.9 | |
| Tryptophan | CH-4 | 7.71 | d [7.8] | 119.6 |
| CH-7 | 7.52 * | d [7.8] | 113.0 | |
| Tyrosine | CH-3,5 | 7.19 | 131.7 | |
| CH-2,6 | 6.90 | 116.9 | ||
| Valine | α-CH | 3.63 | ||
| β-CH | 2.28 | 30.1 | ||
| γ-CH3 | 1.00 | d [7.03] | 17.8 | |
| γ′-CH3 | 1.05 * | d [7.03] | 19.1 | |
| Miscellaneous metabolites | ||||
| Choline | +N(CH3)3 | 3.21 * | s | 54.8 |
| Trigonelline | CH-1 | 9.11 * | s | |
| CH-3,5 | 8.84 | |||
| CH-4 | 8.11 |
| Cultivar | Harvesting Period | CBDV | CBG | CBD | CBN | THC | CBC |
|---|---|---|---|---|---|---|---|
| Ferimon | June | 0.0100 ± 0.0004 | 0.0210 ± 0.0008 | 0.3800 ± 0.0097 | 0.0030 ± 0.0001 | 0.0220 ± 0.0006 | 0.0400 ± 0.0014 |
| July | - | 0.0310 ± 0.0008 | 0.4420 ± 0.0103 | - | 0.0300 ± 0.0012 | - | |
| August | 0.0200 ± 0.0008 | 0.0410 ± 0.0013 | 0.5010 ± 0.0131 | - | 0.0320 ± 0.0011 | - | |
| September | 0.0300 ± 0.0007 | 0.0410 ± 0.0015 | 0.7010 ± 0.0185 | 0.0030 ± 0.0001 | 0.0410 ± 0.0014 | 0.0500 ± 0.0020 | |
| Uso-31 | June | 0.0030 ± 0.0001 | 0.0410 ± 0.0009 | 0.2700 ± 0.0057 | 0.0040 ± 0.0001 | 0.0200 ± 0.0004 | 0.0400 ± 0.0013 |
| July | 0.0080 ± 0.0002 | 0.0200 ± 0.0007 | 0.4610 ± 0.0093 | - | 0.0800 ± 0.0021 | 0.1200 ± 0.0024 | |
| August | 0.0210 ± 0.0005 | 0.0400 ± 0.0009 | 0.6500 ± 0.0130 | - | 0.0800 ± 0.0020 | - | |
| September | 0.0320 ± 0.0008 | 0.0430 ± 0.0010 | 0.8400 ± 0.0169 | - | 0.0910 ± 0.0021 | - | |
| Felina 32 | June | 0.0500 ± 0.0010 | 0.0300 ± 0.0009 | 0.8120 ± 0.0171 | - | 0.0600 ± 0.0019 | 0.0700 ± 0.0023 |
| July | 0.2800 ± 0.0084 | 0.0610 ± 0.0018 | 1.1300 ± 0.0285 | - | 0.0830 ± 0.0028 | - | |
| August | 0.5000 ± 0.0101 | 0.3100 ± 0.0093 | 1.4100 ± 0.0284 | 0.0310 ± 0.0010 | 0.0800 ± 0.0019 | 0.0500 ± 0.0018 | |
| September | 0.0810 ± 0.0026 | 0.2200 ± 0.0065 | 1.1400 ± 0.0295 | 0.0400 ± 0.0093 | 0.0730 ± 0.0024 | - | |
| Fedora 17 | September | 0.1200 ± 0.0032 | 0.0410 ± 0.0013 | 2.0200 ± 0.0405 | - | 0.0700 ± 0.0019 | - |
| Terpenoids | Ferimon | Uso-31 | Felina 32 | Fedora 17 | |||
|---|---|---|---|---|---|---|---|
| June | September | June | September | June | September | September | |
| Caryophyllene E | 15.2 ± 0.49 | 28.0 ± 2.00 | 6.3 ± 0.48 | 11.0 ± 0.49 | 16.4 ± 0.45 | 25.0 ± 0.49 | 20.0 ± 0.49 |
| Trans-α-Bergamotene | - | - | - | - | 1.9 ± 0.05 | 4.2 ± 0.25 | - |
| Humulene | 7.5 ± 0.32 | 9.0 ± 0.47 | 1.3 ± 0.04 | 4.7 ± 0.23 | 10.9 ± 0.45 | 16.7 ± 0.50 | 5.0 ± 0.25 |
| Alloaromadendrene | - | - | 3.2 ± 0.14 | - | - | - | |
| γ-Muurolene | - | - | 1.6 ± 0.46 | - | - | - | |
| β-Selinene | - | - | - | 3.9 ± 0.14 | 3.6 ± 0.15 | 8.3 ± 0.50 | 5.0 ± 0.23 |
| α-Selinene | - | - | - | 3.2 ± 0.15 | 1.9 ± 0.06 | 4.2 ± 0.20 | 5.0 ± 0.23 |
| Nerolidol | - | - | - | - | 3.6 ± 0.14 | - | - |
| Caryophyllene oxide | 50.0 ± 2.48 | 49.0 ± 2.52 | 72.2 ± 3.48 | 46.4 ± 2.30 | 10.9 ± 0.47 | 25.0 ± 1.20 | 45.0 ± 1.60 |
| Humulene epoxide | 10.6 ± 0.49 | 14.0 ± 0.39 | - | 11.0 ± 0.45 | 5.4 ± 0.40 | 8.3 ± 0.45 | 10.0 ± 0.35 |
| α-Caryophylladienol | 6.1 ± 0.25 | - | - | 6.3 ± 0.42 | - | 8.3 ± 0.44 | 10.0 ± 0.38 |
| Clovanediol | 4.5 ± 0.28 | - | - | 5.5 ± 0.26 | - | - | - |
| Neophytadiene | - | - | - | - | 5.4 ± 0.38 | - | - |
| Phytol | 6.1 ± 0.26 | - | 20.2 ± 1.00 | 3.2 ± 0.13 | 40 ± 1.90 | - | - |
| Cultivar Harvesting Period | Total Polyphenols | Total Tannins | Total Flavonoids | |||
|---|---|---|---|---|---|---|
| [mg TAE/g] | [mg TAE/g] | [mg QE/g] | ||||
| HA | O | HA | O | HA | O | |
| Ferimon | ||||||
| June | 1.75 ± 0.01 §,c | 0.95 ± 0.03 §,b | 0.90 ± 0.02 §,c | 0.32 ± 0.02 §,b | 3.02 ± 0.03 §,c | 0.47 ± 0.01 §,b |
| September | 0.78 ± 0.06 *,§,b,c,d | 0.71 ± 0.02 | 0.39 ± 0.02 * | 0.18 ± 0.01 * | 1.03 ± 0.03 * | 0.67 ± 0.01 * |
| Uso-31 | ||||||
| June | 2.11 ± 0.03 §,a,c,d | 0.52 ± 0.05 | 0.89 ± 0.03 §,c | 0.25 ± 0.01 | 4.07 ± 0.03 §,a,c | 0.30 ± 0.03 |
| September | 1.78 ± 0.02 * | 1.11 ± 0.02 *,§,a | 1.00 ± 0.01 §,a | 0.68 ± 0.04 *,§,a | 2.44 ± 0.01 *,§,a,c | 0.99 ± 0.01 *,§,a,d |
| Felina 32 | ||||||
| June | 1.51 ± 0.03 | 1.69 ± 0.05 §,a,b | 0.71 ± 0.01 | 0.97 ± 0.03 §,a,b | 1.63 ± 0.03 | 6.27 ± 0.05 §,a,b |
| September | 4.00 ± 0.01 * | 4.67 ± 0.03 *,§,a,b,d | 4.00 ± 0.03 *,§,a,b,d | 2.87 ± 0.03 *,§,a,b,d | 1.16 ± 0.01 * | 8.72 ± 0.05 *,§,a,b,d |
| Fedora 17 | ||||||
| September | 1.86 ± 0.04 §,c | 1.54 ± 0.02 §,a,b | 0.94 ± 0.01 §,a | 0.58 ± 0.02 §,a,b | 3.82 ± 0.02 §,a,b,c | 0.57 ± 0.03 |
| Compound | Harvesting | Ferimon | Uso-31 | Felina 32 | Fedora 17 | ||||
|---|---|---|---|---|---|---|---|---|---|
| HA | O | HA | O | HA | O | HA | O | ||
| Carvacrol | June | 0.044 ± 0.004 | 0.025 ± 0.002 | 0.123 ± 0.010 | 0.036 ± 0.003 | - | 0.031 ± 0.003 | - | - |
| September | 0.018 ± 0.002 | 0.028 ± 0.002 | 0.059 ± 0.005 | 0.050 ± 0.005 | 0.138 ± 0.015 | 0.127 ± 0.012 | 0.062 ± 0.006 | 0.055 ± 0.004 | |
| Catechin | June | 0.450 ± 0.044 | 0.021 ± 0.002 | - | - | 0.047 ± 0.003 | - | - | - |
| September | 0.194 ± 0.015 | - | 0.782 ± 0.078 | - | 3.723 ± 0.357 | 0.107 ± 0.009 | 0.657 ± 0.064 | - | |
| Rutin | June | 0.872 ± 0.094 | 0.008 ± 0.001 | 0.666 ± 0.068 | - | 0.716 ± 0.074 | 0.026 ± 0.002 | - | - |
| September | 0.436 ± 0.038 | - | 0.598 ± 0.049 | - | 3.787 ± 0.280 | - | 0.660 ± 0.066 | 0.018 ± 0.002 | |
| Quercetin | June | 0.047 ± 0.005 | 0.007 ± 0.001 | 0.069 ± 0.007 | - | 0.046 ± 0.004 | - | - | - |
| September | 0.028 ± 0.002 | - | 0.048 ± 0.005 | - | 0.125 ± 0.013 | - | 0.033 ± 0.003 | - | |
| Naringenin | June | - | - | - | - | - | - | - | - |
| September | - | - | - | - | - | - | - | 0.011 ± 0.001 | |
| Naringin | June | - | 0.007 ± 0.001 | - | - | - | - | - | - |
| September | - | - | - | - | - | - | - | - | |
| o-Coumaric acid | June | - | - | 0.469 ± 0.043 | - | 0.081 ± 0.009 | - | - | - |
| September | - | - | - | - | 0.425 ± 0.038 | 0.015 ± 0.001 | 0.055 ± 0.005 | - | |
| p-Coumaric acid | June | - | - | - | - | - | - | - | - |
| September | - | - | - | - | - | - | 0.098 ± 0.008 | - | |
| Syringic acid | June | - | - | - | - | - | - | - | - |
| September | - | - | - | - | 0.110 ± 0.010 | - | 0.020 ± 0.001 | - | |
| trans-Cinnamic acid | June | - | - | 0.038 ± 0.003 | - | - | - | - | - |
| September | - | - | - | - | - | - | - | - | |
| Chlorogenic acid | June | - | - | - | - | 0.320 ± 0.032 | - | - | - |
| September | - | - | - | - | - | - | - | - | |
| trans-Ferulic acid | June | - | - | - | - | 0.023 ± 0.001 | - | - | - |
| September | 0.004 ± 0.001 | - | - | - | 0.092 ± 0.008 | 0.029 ± 0.003 | 0.015 ± 0.001 | - | |
| 3-OH-benzoic acid | June | - | - | - | - | - | - | - | - |
| September | - | - | - | - | 0.072 ± 0.007 | - | 0.044 ± 0.004 | - | |
| 3-OH-4-MeO-benzaldehyde | June | - | - | - | - | - | - | - | - |
| September | - | - | 0.026 ± 0.002 | - | 0.565 ± 0.043 | - | 0.060 ± 0.006 | - | |
| Cultivar Harvesting Period | PUT | CAD | TYM | SPD | SPM | Total BAs |
|---|---|---|---|---|---|---|
| Ferimon | ||||||
| June | 27.97 ± 4.34 | - | - | 35.53 ± 4.08 | 14.44 ± 2.01 | 77.94 ± 9.83 |
| July | 81.33 ± 7.95 | - | 13.20 ± 1.91 | 55.48 ± 1.00 | 59.68 ± 5.19 | 209.69 ± 15.84 |
| August | 102.72 ± 5.92 | - | 2.19 ± 1.41 | 65.71 ± 5.48 | 40.93 ± 2.36 | 211.56 ± 14.86 |
| September | 12.79 ± 1.27 | 5.54 ± 0.05 | 9.10 ± 0.87 | 38.65 ± 0.94 | 27.93 ± 1.29 | 94.00 ± 1.14 |
| Uso-31 | ||||||
| June | 55.93 ± 4.27 | - | 6.35 ± 3.16 | 41.53 ± 0.41 | 15.13 ± 0.86 | 118.94 ± 8.06 |
| July | 61.64 ± 7.61 | - | 15.53 ± 1.79 | 43.80 ± 3.48 | 54.82 ± 5.84 | 175.78 ± 17.63 |
| August | 69.90 ± 6.35 | - | 19.22 ± 1.62 | 31.23 ± 0.08 | 27.86 ± 0.73 | 148.22 ± 5.06 |
| September | 10.51 ± 0.11 | 5.86 ± 0.05 | 0.44 ± 0.04 | 4.45 ± 0.02 | 4.41 ± 0.10 | 25.67 ± 0.23 |
| Felina 32 | ||||||
| June | 53.79 ± 7.79 | - | 1.32 ± 1.28 | 51.33 ± 4.74 | 18.36 ± 0.99 | 124.79 ± 14.59 |
| July | 16.06 ± 1.75 | - | 8.08 ± 1.04 | 35.53 ± 1.66 | 30.55 ± 1.34 | 90.22 ± 4.32 |
| August | 49.72 ± 2.69 | - | 1.00 ± 1.31 | 26.91 ± 2.35 | 40.38 ± 2.67 | 118.01 ± 6.93 |
| September | 75.53 ± 4.85 | 6.64 ± 0.40 | 13.71 ± 0.33 | 31.77 ± 1.99 | 33.63 ± 1.96 | 161.28 ± 8.67 |
| Fedora 17 | ||||||
| September | 18.46 ± 0.77 | 5.98 ± 0.07 | 7.05 ± 1.52 | 42.09 ± 5.59 | 34.82 ± 4.83 | 108.40 ± 12.51 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ingallina, C.; Sobolev, A.P.; Circi, S.; Spano, M.; Fraschetti, C.; Filippi, A.; Di Sotto, A.; Di Giacomo, S.; Mazzoccanti, G.; Gasparrini, F.; et al. Cannabis sativa L. Inflorescences from Monoecious Cultivars Grown in Central Italy: An Untargeted Chemical Characterization from Early Flowering to Ripening. Molecules 2020, 25, 1908. https://doi.org/10.3390/molecules25081908
Ingallina C, Sobolev AP, Circi S, Spano M, Fraschetti C, Filippi A, Di Sotto A, Di Giacomo S, Mazzoccanti G, Gasparrini F, et al. Cannabis sativa L. Inflorescences from Monoecious Cultivars Grown in Central Italy: An Untargeted Chemical Characterization from Early Flowering to Ripening. Molecules. 2020; 25(8):1908. https://doi.org/10.3390/molecules25081908
Chicago/Turabian StyleIngallina, Cinzia, Anatoly P. Sobolev, Simone Circi, Mattia Spano, Caterina Fraschetti, Antonello Filippi, Antonella Di Sotto, Silvia Di Giacomo, Giulia Mazzoccanti, Francesco Gasparrini, and et al. 2020. "Cannabis sativa L. Inflorescences from Monoecious Cultivars Grown in Central Italy: An Untargeted Chemical Characterization from Early Flowering to Ripening" Molecules 25, no. 8: 1908. https://doi.org/10.3390/molecules25081908
APA StyleIngallina, C., Sobolev, A. P., Circi, S., Spano, M., Fraschetti, C., Filippi, A., Di Sotto, A., Di Giacomo, S., Mazzoccanti, G., Gasparrini, F., Quaglio, D., Campiglia, E., Carradori, S., Locatelli, M., Vinci, G., Rapa, M., Ciano, S., Giusti, A. M., Botta, B., ... Mannina, L. (2020). Cannabis sativa L. Inflorescences from Monoecious Cultivars Grown in Central Italy: An Untargeted Chemical Characterization from Early Flowering to Ripening. Molecules, 25(8), 1908. https://doi.org/10.3390/molecules25081908




















