Use of Fluorescence In Situ Hybridization (FISH) in Diagnosis and Tailored Therapies in Solid Tumors
Abstract
1. Introduction of In Situ hybridization
2. Fluorescence In Situ Hybridization In Solid Tumors
3. Types of Probes Available in FISH Technique
4. Validation of FISH Method
5. FISH Analysis in Solid Tumors
5.1. Lung Cancer
5.2. ALK Gene
5.3. ROS1 Gene
5.4. c-MET Gene
5.5. Gliomas
5.6. Breast Cancer
5.7. Ovarian Cancer
5.8. Soft Tissues Sarcomas
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACMG | the American College of Medical Genetics and Genomics |
| ASCO-CAP | the American Society of Clinical Oncology and the College of American Pathologists |
| BC | breast cancer |
| Cdk2 | cyclin-dependent kinase 2 |
| CGH | comparative genome hybridization |
| CISH | chromogenic in situ hybridization |
| CTC | circulating tumor cells |
| DAB | 3,3′-diaminobenzidine; |
| DFSP | dermatofibrosarcoma protuberans |
| DSBs | double-stranded breaks |
| EGFR | epidermal growth factor receptor |
| ES | Ewing sarcoma |
| EZR | Ezrin |
| FDA | U.S. Food and Drugs Administration |
| FFPE | formalin-fixed paraffin-embedded |
| FISH | fluorescence in situ hybridization |
| G-banding | Giemsa-banding |
| H&E | Hematoxylin and Eosin |
| HCl | hydrochloric acid |
| HGF | hepatocyte growth factor |
| HGSOC | high-grade serous ovarian carcinoma |
| HRP | horseradish peroxidase |
| HR | homologous recombination |
| hsr | homogeneously staining region |
| IHC | Immunohistochemistry |
| KLC1 | kinesin light chain 1 |
| LOH | loss of heterozygosity |
| MAPK | mitogen-activated protein kinases |
| MLPA | multiplex ligation probe amplification |
| mRNA | messenger ribonucleic acid |
| NCNN | the National Comprehensive Cancer Network; |
| NGS | next-generation sequencing |
| NSCLC | non-small cell lung cancer |
| PDGFB | platelet-derived growth factor beta |
| RT-PCR | reverse transcriptase-polymerase chain reaction |
| SDC4 | syndecan 4 |
| SIOP | the International Society of Pediatric Oncology |
| SS | synovial sarcoma |
| SSC | saline-sodium citrate |
| STS | soft tissue sarcomas |
| TKI | tyrosine kinase inhibitors |
| TP53 | tumor protein 53 |
| TPM3 | tropomyosin 3 |
References
- Summersgil, B.; Clarc, J.; Shipley, J. Fluorescence and Chromogenic in Situ Hybridization to Detect Genetic Aberrations in Formalin-Fixed Paraffin Embedded Material, Including Tissue Microarrays. Nat. Protoc. 2008, 3, 220–234. [Google Scholar] [CrossRef] [PubMed]
- Rosa, F.E.; Santos, R.M.; Rogatto, S.R.; Domingues, M.A.C. Chromogenic in Situ Hybridization Compared with Other Approaches to Evaluate HER2/Neu Status in Breast Carcinomas. Braz. J. Med. Biol. Res. 2013, 46, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Furrer, D.; Sanschagrin, F.; Jacob, S.; Diorio, C. Advantages and Disadvantages of Technologies for HER2 Testing in Breast Cancer Specimens: Table 1. Am. J. Clin. Pathol. 2015, 144, 686–703. [Google Scholar] [CrossRef] [PubMed]
- Sáez, A.; Andreu, F.J.; Baré, M.L.; Fernández, S.; Dinarés, C.; Rey, M.J. HER-2 Gene Amplification by Chromogenic in Situ Hybridisation(CISH) Compared with Fluorescence in Situ Hybridisation(FISH) in Breast Cancer—A Study of Two Hundred Cases. Breast 2006, 15, 519–527. [Google Scholar] [CrossRef]
- Kim, H.; Yoo, S.; Paik, J.; Xu, X.; Nitta, H.; Zhang, W.; Grogan, T.M.; Choon-Taek, L.; Sangonhoon, J. Detection of ALK Gene Rearrangement in Non-Small Cell Lung Cancer: A Comparison of Fluorescence in Situ Hybridization and Chromogenic in Situ Hybridization with Correlation of ALK Protein Expression. J. Thorac. Oncol. 2011, 6, 1359–1366. [Google Scholar] [CrossRef]
- Wang, F.; Flanagan, J.; Su, N.; Wang, L.-C.; Bui, S.; Nielson, A.; Wu, X.; Vo, H.-T.; Ma, X.-J.; Luo, Y. RNAscope. J. Mol. Diagn. JMD 2012, 14, 22–29. [Google Scholar] [CrossRef]
- Rogers, T.-M.; Russell, P.A.; Wright, G.; Wainer, Z.; Pang, J.-M.; Henricksen, L.A. Comparison of Methods in the Detection of ALK and ROS1 Rearrangements in Lung Cancer. J. Thorac. Oncol. Off. Publ. Int. Assoc. Study Lung Cancer 2015, 10, 611–618. [Google Scholar] [CrossRef]
- Broholm, H.; Born, P.W.; Guterbaum, D.; Dyrbye, H.; Laursμen, H. Detecting Chromosomal Alterations at 1p and 19q by FISH and DNA Fragment Analysis--a Comparative Study in Human Gliomas. Clin. Neuropathol. 2008, 27, 378–387. [Google Scholar] [CrossRef]
- Hicks, D.G.; Kulkarni, S. HER2+ Breast Cancer: Review of Biologic Relevance and Optimal Use of Diagnostic Tools. Am. J. Clin. Pathol. 2008, 129, 263–273. [Google Scholar] [CrossRef]
- Thunnissen, E.; Bubendorf, L.; Dietel, M.; Elmberger, G.; Kerr, K.; Lopez-Rios, F.; Moch, H.; Olszewski, W.; Pauwels, P.; Penault-Llorca, F.; et al. EML4-ALK Testing in Non-Small Cell Carcinomas of the Lung: A Review with Recommendations. Virchows Arch. 2012, 461, 245–257. [Google Scholar] [CrossRef]
- Poulsen, T.S.; Espersen, M.L.M.; Kofoed, V.; Dabetic, T.; Høgdall, E.; Balslev, E. Comparison of Fluorescence In Situ Hybridization and Chromogenic In Situ Hybridization for Low and High Throughput HER2 Genetic Testing. Int. J. Breast Cancer 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Sugita, S.; Hasegawa, T. Practical Use and Utility of Fluorescence in Situ Hybridization in the Pathological Diagnosis of Soft Tissue and Bone Tumors. J. Orthop. Sci. Off. J. Jpn. Orthop. Assoc. 2017, 22, 601–612. [Google Scholar] [CrossRef]
- Jenkins, R.B.; Blair, H.; Ballman, K.V.; Giannini, C.; Arusell, R.M.; Law, M. A t(1;19)(Q10;P10) Mediates the Combined Deletions of 1p and 19q and Predicts a Better Prognosis of Patients with Oligodendroglioma. Cancer Res. 2006, 66, 9852–9861. [Google Scholar] [CrossRef] [PubMed]
- Asif, A.; Mushtaq, S.; Hassan, U.; Akhtar, N.; Hussain, M.; Azam, M.; Qazi, R. Fluorescence in Situ Hybridization(FISH) for Differential Diagnosis of Soft Tissue Sarcomas. Asian Pac. J. Cancer Prev. APJCP 2018, 19, 655–660. [Google Scholar] [PubMed]
- Polivka, J.; Polivka, J.; Repik, T.; Rohan, V.; Hes, O.; Topolcan, O. Co-Deletion of 1p/19q as Prognostic and Predictive Biomarker for Patients in West Bohemia with Anaplastic Oligodendroglioma. Anticancer Res. 2016, 36, 471–476. [Google Scholar] [PubMed]
- Salido, M.; Pijuan, L.; Martínez-Avilés, L.; Galván, A.B.; Cañadas, I.; Rovira, A.; Zanui, M.; Martínez, A. Increased ALK Gene Copy Number and Amplification Are Frequent in Non-Small Cell Lung Cancer. J. Thorac. Oncol. Off. Publ. Int. Assoc. Study Lung Cancer 2011, 6, 21–27. [Google Scholar] [CrossRef]
- Wesoła, M.; Jeleń, M. A Comparison of IHC and FISH Cytogenetic Methods in the Evaluation of HER2 Status in Breast Cancer. Adv. Clin. Exp. Med. 2015, 24, 899–904. [Google Scholar] [CrossRef]
- Duchemann, B.; Friboulet, L.; Besse, B. Therapeutic Management of ALK+ Nonsmall Cell Lung Cancer Patients. Eur. Respir. J. 2015, 46, 230–242. [Google Scholar] [CrossRef]
- Agersborg, S.; Mixon, C.; Nguyen, T.; Aithal, S.; Sudarsanam, S.; Blocker, F.; Weiss, L.; Gasparini, R. Immunohistochemistry and Alternative FISH Testing in Breast Cancer with HER2 Equivocal Amplification. Breast Cancer Res. Treat. 2018, 170, 321–328. [Google Scholar] [CrossRef]
- Halon, A.; Donizy, P.; Biecek, P.; Rudno-Rudzinska, J.; Kielan, W.; Matkowski, R. HER-2 Expression in Immunohistochemistry Has No Prognostic Significance in Gastric Cancer Patients. Sci. World J. 2012, 2012, 1–6. [Google Scholar] [CrossRef]
- Gelpi, E.; Ambros, I.M.; Birner, P.; Luegmayr, A.; Drlicek, M.; Fischer, I.; Kleinert, R.; Maier, H. Fluorescent in Situ Hybridization on Isolated Tumor Cell Nuclei: A Sensitive Method for 1p and 19q Deletion Analysis in Paraffin-Embedded Oligodendroglial Tumor Specimens. Mod. Pathol. Off. J. U. S. Can. Acad. Pathol. Inc. 2003, 16, 708–715. [Google Scholar] [CrossRef]
- Rodriguez, F.J.; Mota, R.A.; Scheithauer, B.W.; Giannini, C.; Blair, H.; New, K.C.; Wu, K.J.; Dickson, D.W. Interphase Cytogenetics for 1p19q and t(1;19)(Q10;P10) May Distinguish Prognostically Relevant Subgroups in Extraventricular Neurocytoma. Brain Pathol. Zurich Switz. 2009, 19, 623–629. [Google Scholar] [CrossRef]
- Ayhan, A.; Kuhn, E.; Wu, R.-C.; Ogawa, H.; Bahadirli-Talbott, A.; Mao, T.-L. CCNE1 Copy-Number Gain and Overexpression Identify Ovarian Clear Cell Carcinoma with a Poor Prognosis. Mod. Pathol. Off. J. U. S. Can. Acad. Pathol. Inc. 2017, 30, 297–303. [Google Scholar] [CrossRef]
- Noske, A.; Brandt, S.; Valtcheva, N.; Wagner, U.; Zhong, Q.; Bellini, E.; Fink, D.; Obermann, E.C.; Moch, H.; Wild, P.J. Detection of CCNE1/URI (19q12) Amplification by in Situ Hybridisation Is Common in High Grade and Type II Endometrial Cancer. Oncotarget 2017, 8, 14794–14805. [Google Scholar] [CrossRef]
- Hornick, J.L. Novel Uses of Immunohistochemistry in the Diagnosis and Classification of Soft Tissue Tumors. Mod. Pathol. 2014, 27, S47–S63. [Google Scholar] [CrossRef]
- Karanian, M.; Pérot, G.; Coindre, J.-M.; Chibon, F.; Pedeutour, F.; Neuville, A. Fluorescence in Situ Hybridization Analysis Is a Helpful Test for the Diagnosis of Dermatofibrosarcoma Protuberans. Mod. Pathol. Off. J. U. S. Can. Acad. Pathol. Inc. 2015, 28, 230–237. [Google Scholar] [CrossRef]
- Schildhaus, H.-U.; Deml, K.-F.; Schmitz, K.; Meiboom, M.; Binot, E.; Hauke, S.; Merkelbach-Bruse, S.; Büttner, R. Chromogenic in Situ Hybridization Is a Reliable Assay for Detection of ALK Rearrangements in Adenocarcinomas of the Lung. Mod. Pathol. Off. J. U. S. Can. Acad. Pathol. Inc. 2013, 26, 1468–1477. [Google Scholar] [CrossRef]
- Shia, J.; Klimstra, D.S.; Li, A.R.; Qin, J.; Saltz, L.; Teruya-Feldstein, J.; Akram, M.; Chung, K.Y.; Yao, D.; Paty, P.B.; et al. Epidermal Growth Factor Receptor Expression and Gene Amplification in Colorectal Carcinoma: An Immunohistochemical and Chromogenic in Situ Hybridization Study. Mod. Pathol. 2005, 18, 1350–1356. [Google Scholar] [CrossRef]
- Kumagai, A.; Motoi, T.; Tsuji, K.; Imamura, T.; Fukusato, T. Detection of SYT and EWS Gene Rearrangements by Dual-Color Break-Apart CISH in Liquid-Based Cytology Samples of Synovial Sarcoma and Ewing Sarcoma/Primitive Neuroectodermal Tumor. Am. J. Clin. Pathol. 2010, 134, 323–331. [Google Scholar] [CrossRef]
- Ciesielski, M.; Szajewski, M.; Pęksa, R.; Lewandowska, M.A.; Zieliński, J.; Walczak, J.; Szefel, J.; Kruszewski, W.J. The Relationship between HER2 Overexpression and Angiogenesis in Gastric Cancer. Medicine (Baltimore) 2018, 97. [Google Scholar] [CrossRef]
- Shackelford, R.E.; Vora, M.; Mayhall, K.; Cotelingam, J. ALK-Rearrangements and Testing Methods in Non-Small Cell Lung Cancer: A Review. Genes Cancer 2014, 5, 1–14. [Google Scholar]
- Gamberi, G.; Cocchi, S.; Benini, S.; Magagnoli, G.; Morandi, L.; Kreshak, J. Molecular Diagnosis in Ewing Family Tumors. J. Mol. Diagn. JMD 2011, 13, 313–324. [Google Scholar] [CrossRef]
- Patel, K.U.; Szabo, S.S.; Hernandez, V.S.; Prieto, V.G.; Abruzzo, L.V.; Lazar, A.J.F.; López-Terrada, D. Dermatofibrosarcoma Protuberans COL1A1-PDGFB Fusion Is Identified in Virtually All Dermatofibrosarcoma Protuberans Cases When Investigated by Newly Developed Multiplex Reverse Transcription Polymerase Chain Reaction and Fluorescence in Situ Hybridization Assays. Hum. Pathol. 2008, 39, 184–193. [Google Scholar]
- Wan, T.S.K. Cancer Cytogenetics: Methodology Revisited. Ann. Lab. Med. 2014, 34, 413. [Google Scholar] [CrossRef]
- Huber, D.; Voith von Voithenberg, L.; Kaigala, G.V. Fluorescence in Situ Hybridization (FISH): History, Limitations and What to Expect from Micro-Scale FISH? Micro Nano Eng. 2018, 1, 15–24. [Google Scholar] [CrossRef]
- Chen, A.Y.-Y.; Chen, A. Fluorescence In Situ Hybridization. J. Investig. Dermatol. 2013, 133, 1–4. [Google Scholar] [CrossRef]
- Bogdanovska-Todorovska, M.; Petrushevska, G.; Janevska, V.; Spasevska, L.; Kostadinova-Kunovska, S. Standardization and Optimization of Fluorescence in Situ Hybridization(FISH) for HER-2 Assessment in Breast Cancer: A Single Center Experience. Bosn. J. Basic Med. Sci. 2018, 18, 132–140. [Google Scholar] [CrossRef]
- Hastings, R.J.; Bown, N.; Tibiletti, M.G.; Debiec-Rychter, M.; Vanni, R.; Espinet, B. Guidelines for Cytogenetic Investigations in Tumours. Eur. J. Hum. Genet. 2016, 24, 6–13. [Google Scholar] [CrossRef]
- Sato, K. Microdevice in Cellular Pathology: Microfluidic Platforms for Fluorescence in Situ Hybridization and Analysis of Circulating Tumor Cells. Anal. Sci. Int. J. Jpn. Soc. Anal. Chem. 2015, 31, 867–873. [Google Scholar] [CrossRef]
- Wojas-Krawczyk, K.; Krawczyk, P.A.; Ramlau, R.A.; Szumiło, J.; Kozielski, J.; Kalinka-Warzocha, E. The Analysis of ALK Gene Rearrangement by Fluorescence in Situ Hybridization in Non-Small Cell Lung Cancer Patients. Contemp. Oncol. Poznan Pol. 2013, 17, 484–492. [Google Scholar]
- Kim, H.R.; Lim, S.M.; Kim, H.J.; Hwang, S.K.; Park, J.K.; Shin, E. The Frequency and Impact of ROS1 Rearrangement on Clinical Outcomes in Never Smokers with Lung Adenocarcinoma. Ann. Oncol. 2013, 24, 2364–2370. [Google Scholar] [CrossRef]
- Walluks, K.; Chen, Y.; Woelfel, C.; Yang, L.; Cui, T.; Seliger, C. Molecular and Clinicopathological Analysis of Dermatofibrosarcoma Protuberans. Pathol. Res. Pract. 2013, 209, 30–35. [Google Scholar] [CrossRef]
- Cooley, L.D.; Morton, C.C.; Sanger, W.G.; Saxe, D.F.; Mikhail, F.M. Section E6.5–6.8 of the ACMG Technical Standards and Guidelines: Chromosome Studies of Lymph Node and Solid Tumor–Acquired Chromosomal Abnormalities. Genet. Med. 2016, 18, 643–648. [Google Scholar] [CrossRef][Green Version]
- Hastings, R.; Howell, R.; Bricarelli, F.D.; Kristoffersson, U.; Cavani, S. European Cytogeneticists Association (E.C.A.). A Common European Framework for Quality Assessment for Constitutional, Acquired and Molecular Cytogenetic Investigations. General Guidelines and Quality Assurance for Cytogenetics. Newsletter 2012, 29, 7–25. [Google Scholar]
- Midha, A.; Dearden, S.; McCormack, R. EGFR Mutation Incidence in Non-Small-Cell Lung Cancer of Adenocarcinoma Histology: A Systematic Review and Global Map by Ethnicity (MutMapII). Am. J. Cancer Res. 2015, 5, 2892–2911. [Google Scholar]
- Krawczyk, P.; Ramlau, R.; Chorostowska-Wynimko, J.; Powrózek, T.; Lewandowska, M.A.; Limon, J.; Wasąg, B.; Pankowski, J. The Efficacy of EGFR Gene Mutation Testing in Various Samples from Non-Small Cell Lung Cancer Patients: A Multicenter Retrospective Study. J. Cancer Res. Clin. Oncol. 2015, 141, 61–68. [Google Scholar] [CrossRef]
- Lewandowska, M.A.; Jóźwicki, W.; Jochymski, C.; Kowalewski, J. Application of PCR Methods to Evaluate EGFR, KRAS and BRAF Mutations in a Small Number of Tumor Cells in Cytological Material from Lung Cancer Patients. Oncol. Rep. 2013, 30, 1045–1052. [Google Scholar] [CrossRef]
- Szumera-Ciećkiewicz, A.; Olszewski, W.T.; Tysarowski, A.; Kowalski, D.M.; Głogowski, M.; Krzakowski, M. EGFR Mutation Testing on Cytological and Histological Samples in Non-Small Cell Lung Cancer: A Polish, Single Institution Study and Systematic Review of European Incidence. Int. J. Clin. Exp. Pathol. 2013, 6, 2800–2812. [Google Scholar]
- Perner, S.; Wagner, P.L.; Demichelis, F.; Mehra, R.; Lafargue, C.J.; Moss, B.J. EML4-ALK Fusion Lung Cancer: A Rare Acquired Event. Neoplasia N. Y. 2008, 10, 298–302. [Google Scholar] [CrossRef]
- Soda, M.; Choi, Y.L.; Enomoto, M.; Takada, S.; Yamashita, Y.; Ishikawa, S.; Fujiwara, S.; Watanabe, H. Identification of the Transforming EML4-ALK Fusion Gene in Non-Small-Cell Lung Cancer. Nature 2007, 448, 561–566. [Google Scholar] [CrossRef]
- Martelli, M.P.; Sozzi, G.; Hernandez, L.; Pettirossi, V.; Navarro, A.; Conte, D. EML4-ALK Rearrangement in Non-Small Cell Lung Cancer and Non-Tumor Lung Tissues. Am. J. Pathol. 2009, 174, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Grenda, A.; Jarosz, B.; Krawczyk, P.; Kucharczyk, T.; Wojas-Krawczyk, K.; Reszka, K. Discrepancies between ALK Protein Disruption and Occurrence of ALK Gene Rearrangement in Polish NSCLC Patients. J. Thorac. Dis. 2018, 10, 4994–5009. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, B.; Palmer, R.H. The Role of the ALK Receptor in Cancer Biology. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2016, 27 (Suppl 3), iii4–iii15. [Google Scholar] [CrossRef] [PubMed]
- Togashi, Y.; Soda, M.; Sakata, S.; Sugawara, E.; Hatano, S.; Asaka, R. KLC1-ALK: A Novel Fusion in Lung Cancer Identified Using a Formalin-Fixed Paraffin-Embedded Tissue Only. PLoS ONE 2012, 7, e31323. [Google Scholar] [CrossRef]
- Takeuchi, K.; Choi, Y.L.; Togashi, Y.; Soda, M.; Hatano, S.; Inamura, K.; Takada, S.; Ueno, T. KIF5B-ALK, a Novel Fusion Oncokinase Identified by an Immunohistochemistry-Based Diagnostic System for ALK-Positive Lung Cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2009, 15, 3143–3149. [Google Scholar] [CrossRef]
- Rikova, K.; Guo, A.; Zeng, Q.; Possemato, A.; Yu, J.; Haack, H.; Nardone, J.; Lee, K. Global Survey of Phosphotyrosine Signaling Identifies Oncogenic Kinases in Lung Cancer. Cell 2007, 131, 1190–1203. [Google Scholar] [CrossRef]
- Birchmeier, C.; Sharma, S.; Wigler, M. Expression and Rearrangement of the ROS1 Gene in Human Glioblastoma Cells. Proc. Natl. Acad. Sci. USA 1987, 84, 9270–9274. [Google Scholar] [CrossRef]
- Birchmeier, C.; O’Neill, K.; Riggs, M.; Wigler, M. Characterization of ROS1 CDNA from a Human Glioblastoma Cell Line. Proc. Natl. Acad. Sci. USA 1990, 87, 4799–4803. [Google Scholar] [CrossRef]
- Sharma, S.; Birchmeier, C.; Nikawa, J.; O’Neill, K.; Rodgers, L.; Wigler, M. Characterization of the Ros1-Gene Products Expressed in Human Glioblastoma Cell Lines. Oncogene Res. 1989, 5, 91–100. [Google Scholar]
- Bergethon, K.; Shaw, A.T.; Ou, S.-H.I.; Katayama, R.; Lovly, C.M.; McDonald, N.T.; Massion, P.P.; Siwak-Tapp, C. ROS1 Rearrangements Define a Unique Molecular Class of Lung Cancers. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2012, 30, 863–870. [Google Scholar] [CrossRef]
- Arai, Y.; Totoki, Y.; Takahashi, H.; Nakamura, H.; Hama, N.; Kohno, T.; Tsuta, K.; Yoshida, A. Mouse Model for ROS1-Rearranged Lung Cancer. PLoS ONE 2013, 8, e56010. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, K.; Soda, M.; Togashi, Y.; Suzuki, R.; Sakata, S.; Hatano, S.; Asaka, R.; Hamanaka, W. RET, ROS1 and ALK Fusions in Lung Cancer. Nat. Med. 2012, 18, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, A.; Kohno, T.; Tsuta, K.; Wakai, S.; Arai, Y.; Shimada, Y.; Asamura, H.; Furuta, K. ROS1-Rearranged Lung Cancer: A Clinicopathologic and Molecular Study of 15 Surgical Cases. Am. J. Surg. Pathol. 2013, 37, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Rimkunas, V.M.; Crosby, K.E.; Li, D.; Hu, Y.; Kelly, M.E.; Gu, T.-L.; Mack, J.S.; Silver, M.R. Analysis of Receptor Tyrosine Kinase ROS1-Positive Tumors in Non-Small Cell Lung Cancer: Identification of a FIG-ROS1 Fusion. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 4449–4457. [Google Scholar] [CrossRef] [PubMed]
- Govindan, R.; Ding, L.; Griffith, M.; Subramanian, J.; Dees, N.D.; Kanchi, K.L.; Maher, C.A. Genomic Landscape of Non-Small Cell Lung Cancer in Smokers and Never-Smokers. Cell 2012, 150, 1121–1134. [Google Scholar] [CrossRef] [PubMed]
- Davies, K.D.; Le, A.T.; Theodoro, M.F.; Skokan, M.C.; Aisner, D.L.; Berge, E.M. Identifying and Targeting ROS1 Gene Fusions in Non-Small Cell Lung Cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 4570–4579. [Google Scholar] [CrossRef] [PubMed]
- Sehgal, K.; Patell, R.; Rangachari, D.; Costa, D.B. Targeting ROS1 Rearrangements in Non-Small Cell Lung Cancer with Crizotinib and Other Kinase Inhibitors. Transl. Cancer Res. 2018, 7 (Suppl 7), S779–S786. [Google Scholar] [CrossRef]
- Ettinger, D.S.; Aisner, D.L.; Wood, D.E.; Akerley, W.; Bauman, J.; Chang, J.Y.; Chirieac, L.R.; D’Amico, T.A. NCCN Guidelines Insights: Non–Small Cell Lung Cancer, Version 5.2018. J. Natl. Compr. Canc. Netw. 2018, 16, 807–821. [Google Scholar] [CrossRef] [PubMed]
- Gainor, J.F.; Shaw, A.T. Novel Targets in Non-Small Cell Lung Cancer: ROS1 and RET Fusions. Oncologist 2013, 18, 865–875. [Google Scholar] [CrossRef]
- Bottaro, D.P.; Rubin, J.S.; Faletto, D.L.; Chan, A.M.; Kmiecik, T.E.; Vande Woude, G.F.; Aaronson, S.A. Identification of the Hepatocyte Growth Factor Receptor as the C-Met Proto-Oncogene Product. Science 1991, 251, 802–804. [Google Scholar] [CrossRef]
- Virzì, A.R.; Gentile, A.; Benvenuti, S.; Comoglio, P.M. Reviving Oncogenic Addiction to MET Bypassed by BRAF(G469A) Mutation. Proc. Natl. Acad. Sci. USA 2018, 115, 10058–10063. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Liang, L.; Lei, X.; Multani, A.; Meric-Bernstam, F.; Tripathy, D.; Wu, Y.; Chen, H. Evaluation of CMET Aberration by Immunohistochemistry and Fluorescence in Situ Hybridization (FISH) in Triple Negative Breast Cancers. Ann. Diagn. Pathol. 2018, 35, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Dziadziuszko, R.; Wynes, M.W.; Singh, S.; Asuncion, B.R.; Ranger-Moore, J.; Konopa, K.; Rzyman, W.; Szostakiewicz, B. Correlation between MET Gene Copy Number by Silver in Situ Hybridization and Protein Expression by Immunohistochemistry in Non-Small Cell Lung Cancer. J. Thorac. Oncol. Off. Publ. Int. Assoc. Study Lung Cancer 2012, 7, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Chen, H.; Tang, Z.; Kalhor, N.; Liu, C.-H.; Yao, H.; Hu, S.; Lin, P. MET Amplification Assessed Using Optimized FISH Reporting Criteria Predicts Early Distant Metastasis in Patients with Non-Small Cell Lung Cancer. Oncotarget 2018, 9, 12959–12970. [Google Scholar] [CrossRef][Green Version]
- Lewandowska, M.A.; Czubak, K.; Klonowska, K.; Jozwicki, W.; Kowalewski, J.; Kozlowski, P. The Use of a Two-Tiered Testing Strategy for the Simultaneous Detection of Small EGFR Mutations and EGFR Amplification in Lung Cancer. PLoS ONE 2015, 10, e0117983. [Google Scholar] [CrossRef] [PubMed]
- Rochigneux, P.; Thomassin-Piana, J.; Laibe, S.; Brunelle, S.; Salem, N.; Escudier, B.; Vassal, G.; Gravis, G. Long-Term Efficacy of Crizotinib in a Metastatic Papillary Renal Carcinoma with MET Amplification: A Case Report and Literature Review. BMC Cancer 2018, 18, 1159. [Google Scholar] [CrossRef]
- Engelman, J.A.; Zejnullahu, K.; Mitsudomi, T.; Song, Y.; Hyland, C.; Park, J.O.; Lindeman, N.; Gale, C.-M. MET Amplification Leads to Gefitinib Resistance in Lung Cancer by Activating ERBB3 Signaling. Science 2007, 316, 1039–1043. [Google Scholar] [CrossRef]
- Paxton, C.N.; Rowe, L.R.; South, S.T. Observations of the Genomic Landscape beyond 1p19q Deletions and EGFR Amplification in Glioma. Mol. Cytogenet. 2015, 8, 60. [Google Scholar] [CrossRef][Green Version]
- Park, S.-H.; Won, J.; Kim, S.-I.; Lee, Y.; Park, C.-K.; Kim, S.-K.; Choi, S.-H. Molecular Testing of Brain Tumor. J. Pathol. Transl. Med. 2017, 51, 205–223. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A Summary. Acta Neuropathol.(Berl.) 2016, 131, 803–820. [Google Scholar] [CrossRef]
- Reifenberger, J.; Reifenberger, G.; Liu, L.; James, C.D.; Wechsler, W.; Collins, V.P. Molecular Genetic Analysis of Oligodendroglial Tumors Shows Preferential Allelic Deletions on 19q and 1p. Am. J. Pathol. 1994, 145, 1175–1190. [Google Scholar] [PubMed]
- Iwadate, Y.; Matsutani, T.; Shinozaki, N.; Saeki, N. Anaplastic Oligodendroglial Tumors Harboring 1p/19q Deletion Can Be Successfully Treated without Radiotherapy. Anticancer Res. 2011, 31, 4475–4479. [Google Scholar] [PubMed]
- Shah, R.; Rosso, K.; Nathanson, S.D. Pathogenesis, Prevention, Diagnosis and Treatment of Breast Cancer. World J. Clin. Oncol. 2014, 5, 283–298. [Google Scholar] [CrossRef] [PubMed]
- Coleman, M.P.; Quaresma, M.; Berrino, F.; Lutz, J.-M.; De Angelis, R.; Capocaccia, R.; Baili, P.; Rachet, B. Cancer Survival in Five Continents: A Worldwide Population-Based Study(CONCORD). Lancet Oncol. 2008, 9, 730–756. [Google Scholar] [CrossRef]
- Wojciechowska, U.; Czaderny, K.; Ciuba, A.; OLasek, P.; Didkowska, J. Nowotwory Złośliwe w Polsce w 2016 Roku; Centrum Onkologii Instytut Im. Marii Skłodowskiej-Curie: Warszawa, Poland, 2018. [Google Scholar]
- Slamon, D.J.; Clark, G.M.; Wong, S.G.; Levin, W.J.; Ullrich, A.; McGuire, W.L. Human Breast Cancer: Correlation of Relapse and Survival with Amplification of the HER-2/Neu Oncogene. Science 1987, 235, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Bofin, A.M.; Ytterhus, B.; Martin, C.; O’Leary, J.J.; Hagmar, B.M. Detection and Quantitation of HER-2 Gene Amplification and Protein Expression in Breast Carcinoma. Am. J. Clin. Pathol. 2004, 122, 110–119. [Google Scholar] [CrossRef]
- Slamon, D.J.; Godolphin, W.; Jones, L.A.; Holt, J.A.; Wong, S.G.; Keith, D.E.; Levin, W.J.; Stuart, S.G. Studies of the HER-2/Neu Proto-Oncogene in Human Breast and Ovarian Cancer. Science 1989, 244, 707–712. [Google Scholar] [CrossRef]
- Owens, M.A.; Horten, B.C.; Da Silva, M.M. HER2 Amplification Ratios by Fluorescence in Situ Hybridization and Correlation with Immunohistochemistry in a Cohort of 6556 Breast Cancer Tissues. Clin. Breast Cancer 2004, 5, 63–69. [Google Scholar] [CrossRef]
- Barros-Silva, J.D.; Leitão, D.; Afonso, L.; Vieira, J.; Dinis-Ribeiro, M.; Fragoso, M.; Bento, M.J.; Santos, L. Association of ERBB2 Gene Status with Histopathological Parameters and Disease-Specific Survival in Gastric Carcinoma Patients. Br. J. Cancer 2009, 100, 487–493. [Google Scholar] [CrossRef]
- Abrahao-Machado, L.F.; Scapulatempo-Neto, C. HER2 Testing in Gastric Cancer: An Update. World J. Gastroenterol. 2016, 22, 4619–4625. [Google Scholar] [CrossRef]
- Wolff, A.C.; Hammond, M.E.H.; Schwartz, J.N.; Hagerty, K.L.; Allred, D.C.; Cote, R.J.; Dowsett, M.; Fitzgibbons, P.L. American Society of Clinical Oncology/College of American Pathologists Guideline Recommendations for Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2007, 25, 118–145. [Google Scholar] [CrossRef] [PubMed]
- Wolff, A.C.; Hammond, M.E.H.; Allison, K.H.; Harvey, B.E.; Mangu, P.B.; Bartlett, J.M.S.; Bilous, M.; Ellis, I.O. Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update. Arch. Pathol. Lab. Med. 2018, 142, 1364–1382. [Google Scholar] [CrossRef] [PubMed]
- Vranic, S.; Teruya, B.; Repertinger, S.; Ulmer, P.; Hagenkord, J.; Gatalica, Z. Assessment of HER2 Gene Status in Breast Carcinomas with Polysomy of Chromosome 17. Cancer 2011, 117, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Vanden Bempt, I.; Van Loo, P.; Drijkoningen, M.; Neven, P.; Smeets, A.; Christiaens, M.-R.; Paridaens, R.; De Wolf-Peeters, C. Polysomy 17 in Breast Cancer: Clinicopathologic Significance and Impact on HER-2 Testing. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2008, 26, 4869–4874. [Google Scholar] [CrossRef]
- Dietel, M. Molecular Pathology: A Requirement for Precision Medicine in Cancer. Oncol. Res. Treat. 2016, 39, 804–810. [Google Scholar] [CrossRef]
- Rakha, E.A.; Pinder, S.E.; Bartlett, J.M.S.; Ibrahim, M.; Starczynski, J.; Carder, P.J.; Provenzano, E.; Hanby, A. Updated UK Recommendations for HER2 Assessment in Breast Cancer. J. Clin. Pathol. 2015, 68, 93–99. [Google Scholar] [CrossRef]
- Kroeger, P.T.; Drapkin, R. Pathogenesis and Heterogeneity of Ovarian Cancer. Curr. Opin. Obstet. Gynecol. 2017, 29, 26–34. [Google Scholar] [CrossRef]
- Etemadmoghadam, D.; Weir, B.A.; Au-Yeung, G.; Alsop, K.; Mitchell, G.; George, J.; Australian Ovarian Cancer Study Group; Davis, S.; D’Andrea, A.D.; Simpson, K.; et al. Synthetic Lethality between CCNE1 Amplification and Loss of BRCA1. Proc. Natl. Acad. Sci. USA 2013, 110, 19489–19494. [Google Scholar] [CrossRef]
- Saltus, C.W.; Calingaert, B.; Candrilli, S.; Lorenzo, M.; D’yachkova, Y.; Otto, T.; Wagner, U.; Kaye, J.A. Epidemiology of Adult Soft-Tissue Sarcomas in Germany. Sarcoma 2018, 2018, 5671926. [Google Scholar] [CrossRef]
- Mastrangelo, G.; Coindre, J.-M.; Ducimetière, F.; Dei Tos, A.P.; Fadda, E.; Blay, J.-Y.; Buja, A.; Fedeli, U. Incidence of Soft Tissue Sarcoma and beyond: A Population-Based Prospective Study in 3 European Regions. Cancer 2012, 118, 5339–5348. [Google Scholar] [CrossRef]
- Ewing, J. Diffuse Endothelioma of Bone. CA. Cancer J. Clin. 1972, 22, 95–98. [Google Scholar] [CrossRef] [PubMed]
- Zucman, J.; Delattre, O.; Desmaze, C.; Plougastel, B.; Joubert, I.; Melot, T. Cloning and Characterization of the Ewing’s Sarcoma and Peripheral Neuroepithelioma t(11;22) Translocation Breakpoints. Genes. Chromosomes Cancer 1992, 5, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Dunn, T.; Praissman, L.; Hagag, N.; Viola, M.V. ERG Gene Is Translocated in an Ewing’s Sarcoma Cell Line. Cancer Genet. Cytogenet. 1994, 76, 19–22. [Google Scholar] [CrossRef]
- Hoang, N.T.; Acevedo, L.A.; Mann, M.J.; Tolani, B. A Review of Soft-Tissue Sarcomas: Translation of Biological Advances into Treatment Measures. Available online: https://www.dovepress.com/a-review-of-soft-tissue-sarcomas-translation-of-biological-advances-in-peer-reviewed-fulltext-article-CMAR (accessed on 8 February 2020). [CrossRef]
- Limon, J.; Dal Cin, P.; Sandberg, A.A. Translocations Involving the X Chromosome in Solid Tumors: Presentation of Two Sarcomas with t(X;18)(Q13;P11). Cancer Genet. Cytogenet. 1986, 23, 87–91. [Google Scholar] [CrossRef]
- Turc-Carel, C.; Dal Cin, P.; Limon, J.; Li, F.; Sandberg, A.A. Translocation X;18 in Synovial Sarcoma. Cancer Genet. Cytogenet. 1986, 23, 93. [Google Scholar] [CrossRef]
- Turc-Carel, C.; Dal Cin, P.; Limon, J.; Rao, U.; Li, F.P.; Corson, J.M.; Zimmerman, R.; Parry, D.M. Involvement of Chromosome X in Primary Cytogenetic Change in Human Neoplasia: Nonrandom Translocation in Synovial Sarcoma. Proc. Natl. Acad. Sci. USA 1987, 84, 1981–1985. [Google Scholar] [CrossRef]
- Iliszko, M.; Rys, J.; Wozniak, A.; Chosia, M.; Sciot, R.; Debiec-Rychter, M.; Limon, J. Complex Tumor-Specific t(X;18) in Seven Synovial Sarcoma Tumors. Cancer Genet. Cytogenet. 2009, 189, 118–121. [Google Scholar] [CrossRef]
- Fletcher, C.D.; Bridge, J.A.; Hogendoorn, P.C.; Mertens, F. WHO Classification of Tumours of Soft Tissue and Bone, 4th ed.; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Takahira, T.; Oda, Y.; Tamiya, S.; Higaki, K.; Yamamoto, H.; Kobayashi, C.; Izumi, T.; Tateishi, N.; at, el. Detection of COL1A1-PDGFB Fusion Transcripts and PDGFB/PDGFRB MRNA Expression in Dermatofibrosarcoma Protuberans. Mod. Pathol. Off. J. U. S. Can. Acad. Pathol. Inc. 2007, 20, 668–675. [Google Scholar] [CrossRef]
- Segura, S.; Salgado, R.; Toll, A.; Martín-Ezquerra, G.; Yébenes, M.; Sáez, A.; Solé, F.; Barranco, C. Identification of t(17;22)(Q22;Q13) (COL1A1/PDGFB) in Dermatofibrosarcoma Protuberans by Fluorescence in Situ Hybridization in Paraffin-Embedded Tissue Microarrays. Hum. Pathol. 2011, 42, 176–184. [Google Scholar] [CrossRef]
- Salgado, R.; Llombart, B.; M Pujol, R.; Fernández-Serra, A.; Sanmartín, O.; Toll, A.; Rubio, L.; Segura, S.; López-Guerrero, J.A. Molecular Diagnosis of Dermatofibrosarcoma Protuberans: A Comparison between Reverse Transcriptase-Polymerase Chain Reaction and Fluorescence in Situ Hybridization Methodologies. Genes. Chromosomes Cancer 2011, 50, 510–517. [Google Scholar] [CrossRef]
- Simon, M.P.; Pedeutour, F.; Sirvent, N.; Grosgeorge, J.; Minoletti, F.; Coindre, J.M.; Terrier-Lacombe, M.J. Deregulation of the Platelet-Derived Growth Factor B-Chain Gene via Fusion with Collagen Gene COL1A1 in Dermatofibrosarcoma Protuberans and Giant-Cell Fibroblastoma. Nat. Genet. 1997, 15, 95–98. [Google Scholar] [CrossRef] [PubMed]
- Noujaim, J.; Thway, K.; Fisher, C.; Jones, R.L. Dermatofibrosarcoma Protuberans: From Translocation to Targeted Therapy. Cancer Biol. Med. 2015, 12, 375–384. [Google Scholar] [PubMed]
- Rutkowski, P.; Wozniak, A.; Switaj, T. Advances in Molecular Characterization and Targeted Therapy in Dermatofibrosarcoma Protuberans. Sarcoma 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Kalemkerian, G.P.; Narula, N.; Kennedy, E.B.; Biermann, W.A.; Donington, J.; Leighl, N.B.; Lew, M.; Pantelas, J. Molecular Testing Guideline for the Selection of Patients With Lung Cancer for Treatment With Targeted Tyrosine Kinase Inhibitors: American Society of Clinical Oncology Endorsement of the College of American Pathologists/International Association for the Study of Lung Cancer/Association for Molecular Pathology Clinical Practice Guideline Update. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2018, 36, 911–919. [Google Scholar]
- Bubendorf, L.; Büttner, R.; Al-Dayel, F.; Dietel, M.; Elmberger, G.; Kerr, K. Testing for ROS1 in Non-Small Cell Lung Cancer: A Review with Recommendations. Virchows Arch. Int. J. Pathol. 2016, 469, 489–503. [Google Scholar] [CrossRef]
- Dagogo-Jack, I.; Shaw, A.T. Screening for ALK Rearrangements in Lung Cancer: Time for a New Generation of Diagnostics? Oncologist 2016, 21, 662–663. [Google Scholar] [CrossRef]
| IHC | CISH | FISH | |
|---|---|---|---|
| Concept of the Method | |||
| assessment of protein expression using antigen-specific antibodies | assessment of chromogenic effect in an enzymatic reaction | assessment of chromosomal aberration using a fluorescent probe | |
| Advantages | |||
| preparation |
| ||
| analysis |
|
| |
| other |
|
| |
| Disadvantages | |||
| preparation |
|
| |
| analysis |
|
| |
| other | - | - |
|
| Examples of solid tumors with use of the method | |||
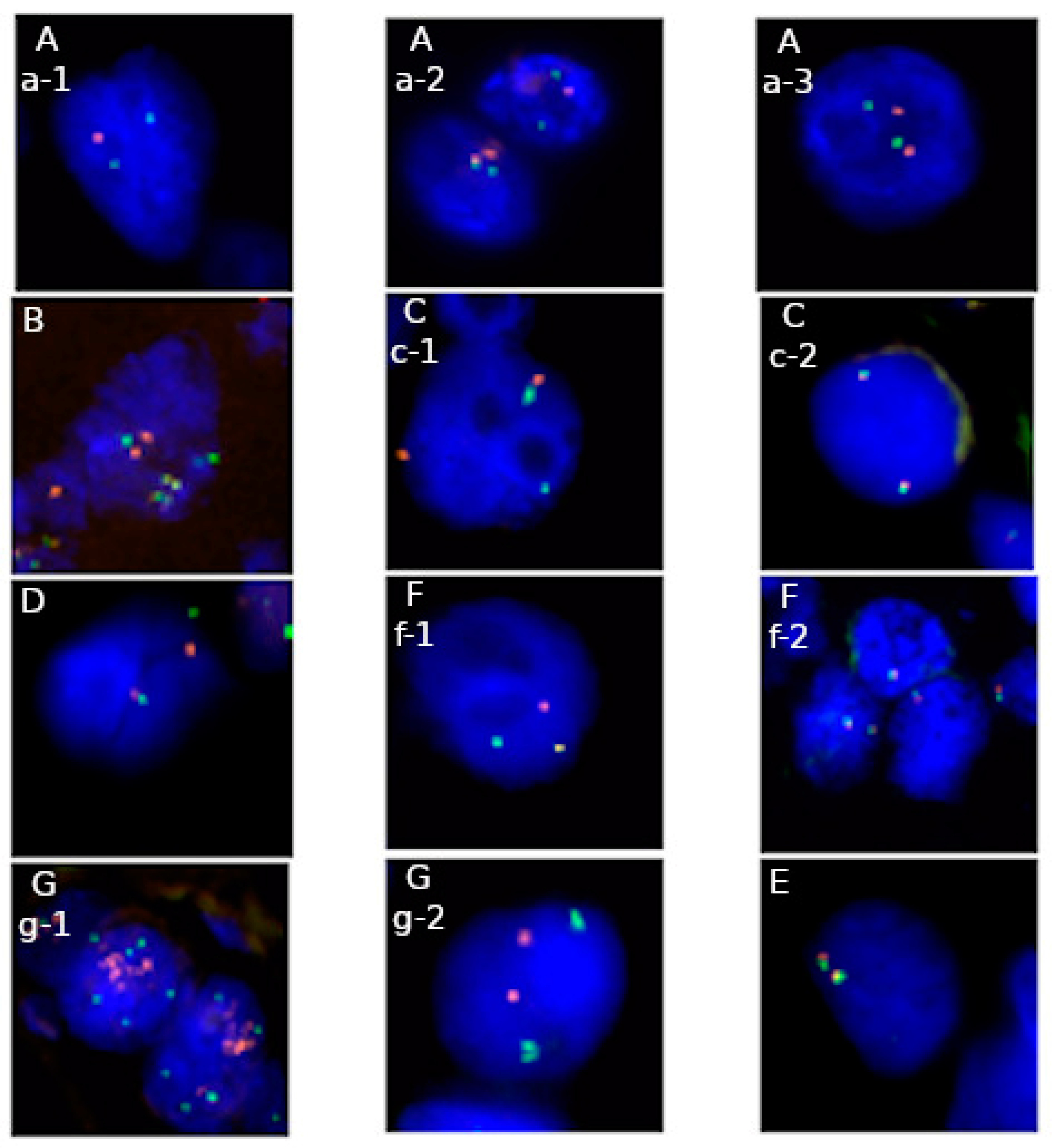
| Type of Tumor | Diagnostic Value | Prognostic Value | Predictive Value | Available FISH Probe |
|---|---|---|---|---|
| Lung cancer ALK, ROS1 | no | yes | yes crizotinib | ▪ Vysis ALK Break Apart FISH Probe Kit (Abbott Molecular) [7,40] ▪ Vysis 6q22 ROS1 Break Apart FISH Probe (Abbott Molecular) [41] ▪ ROS1 Break Apart FISH Probe (Empire Genomics) |
| Glioma co-deletion 1p19q | yes | yes | No | ▪ Vysis LSI 1p36 SpectrumOrange/1q25 SpectrumGreen Probes and Vysis LSI 19q13 SpectrumOrange/19p13 SpectrumGreen Probes (Abbott Molecular) [13] |
| Breast cancer HER2 | no | yes | yes trastuzumab, lapatinib | ▪ PathVysion HER-2 DNA Probe Kit (Abbott Molecular) [37] |
| Ovarian cancer CCNE1 | no | yes | yes platinum-based agents | ▪ CCNE1/CEN19p FISH Probe (Abnova) [23] |
| Ewing sarcoma EWSR1 | yes | yes | No | ▪ Vysis EWSR1 Break Apart FISH Probe Kit (Abbott Molecular) [14] |
| Synovial sarcoma SS18 | yes | yes | No | ▪ Vysis SS18 Break Apart FISH Probe Kit (Abbot Molecular) [14] |
| Dermatofibrosarcoma protuberans COL1A1-PDGFB | no | yes | yes TKI (imatinib) | ▪ SPEC COL1A1-PDGFB Dual Color Dual Fusion (ZytoLight) [42] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chrzanowska, N.M.; Kowalewski, J.; Lewandowska, M.A. Use of Fluorescence In Situ Hybridization (FISH) in Diagnosis and Tailored Therapies in Solid Tumors. Molecules 2020, 25, 1864. https://doi.org/10.3390/molecules25081864
Chrzanowska NM, Kowalewski J, Lewandowska MA. Use of Fluorescence In Situ Hybridization (FISH) in Diagnosis and Tailored Therapies in Solid Tumors. Molecules. 2020; 25(8):1864. https://doi.org/10.3390/molecules25081864
Chicago/Turabian StyleChrzanowska, Natalia Magdalena, Janusz Kowalewski, and Marzena Anna Lewandowska. 2020. "Use of Fluorescence In Situ Hybridization (FISH) in Diagnosis and Tailored Therapies in Solid Tumors" Molecules 25, no. 8: 1864. https://doi.org/10.3390/molecules25081864
APA StyleChrzanowska, N. M., Kowalewski, J., & Lewandowska, M. A. (2020). Use of Fluorescence In Situ Hybridization (FISH) in Diagnosis and Tailored Therapies in Solid Tumors. Molecules, 25(8), 1864. https://doi.org/10.3390/molecules25081864






