Chemistry, Biological Activities and In Silico Bioprospection of Sterols and Triterpenes from Mexican Columnar Cactaceae
Abstract
1. Introduction
2. Chemistry of Cactaceae
2.1. Sterols from Mexican Columnar Cactaceae
2.2. Triterpenes from Mexican Columnar Cactaceae
3. In Silico Bioprospection of Bioactive Compounds
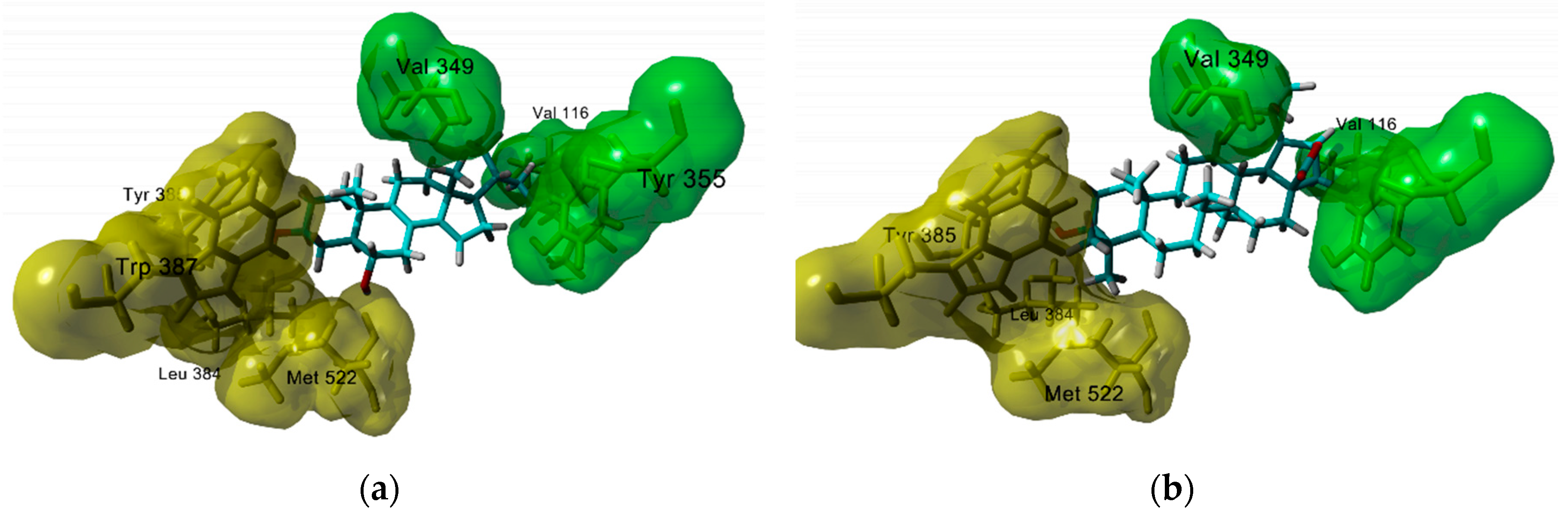
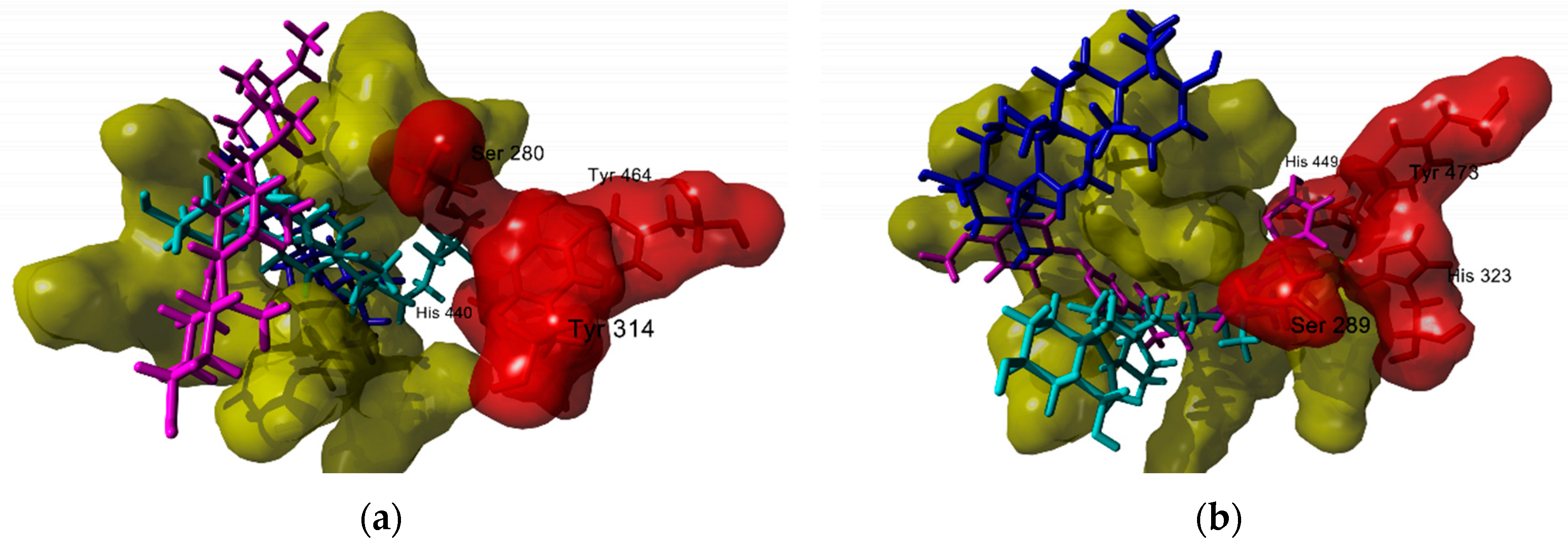
3.1. Antiinflammatory Molecular Targets
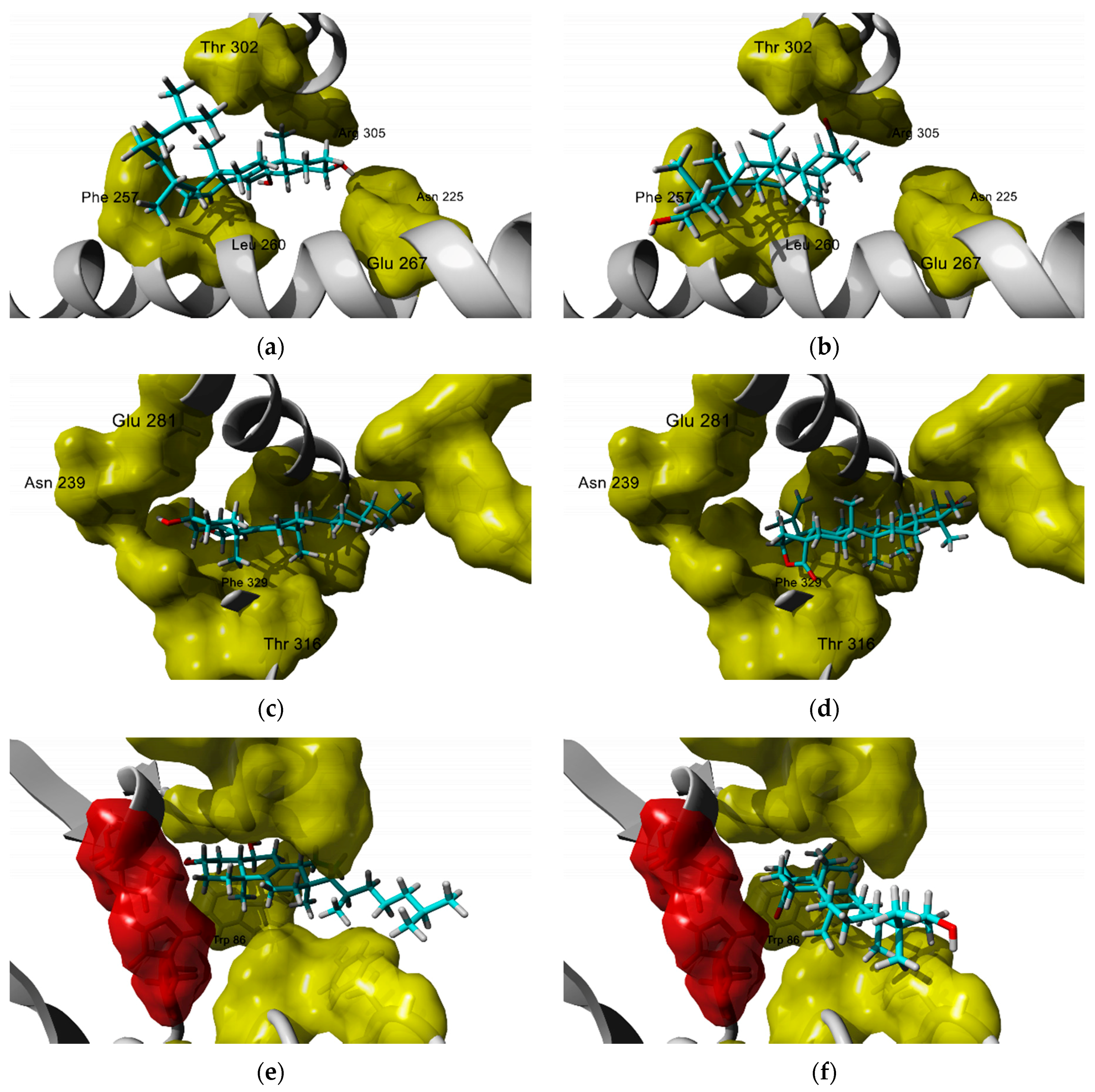
3.2. Antidiabetic and Metabolic Activities
3.3. Neuroprotective Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Reyes-Santiago, J. Conservación y restauración de cactáceas y otras plantas suculentas mexicanas. Manual Práctico. CONAFOR y SEMARNAT, México. Google: Mexico, 2009. Available online: https://www.conafor.gob.mx/biblioteca/Manual_Practico-Conservacionyrestauracion-cactaceas_suculentas.pdf (accessed on 15 December 2019).
- Bravo-Hollis, H. Las Cactáceas de México; Universidad Nacional Autónoma de México: Ciudad de México, México, 1978. [Google Scholar]
- Anderson, E.F. The Cactus Family; Timber Press: Portland, OR, USA, 2001; ISBN 0881924989. [Google Scholar]
- Guzmán, U.; Arias, S.; Dávila, P. Catálogo de autoridades taxonómicas de las cactáceas (Cactaceae: Magnoliopsida) de México. Mexico, 2007. Available online: http://www.snib.mx/descargasSNIBmx/SNIBTaxonomia_20200329_142957.zip (accessed on 29 March 2020).
- Batis, A.; Rojas, M. El Peyote Y Otros Cactos Alucinógenos De Mexico. Biodoversitas 2002, 40, 12–17. [Google Scholar]
- De Wit, M.; du Toit, A.; Osthoff, G.; Hugo, A. Cactus pear antioxidants: A comparison between fruit pulp, fruit peel, fruit seeds and cladodes of eight different cactus pear cultivars (Opuntia ficus-indica and Opuntia robusta). J. Food Meas. Charact. 2019, 13, 2347–2356. [Google Scholar] [CrossRef]
- Berrabah, H.; Taïbi, K.; Ait Abderrahim, L.; Boussaid, M. Phytochemical composition and antioxidant properties of prickly pear (Opuntia ficus-indica L.) flowers from the Algerian germplasm. J. Food Meas. Charact. 2019, 13, 1166–1174. [Google Scholar] [CrossRef]
- Harrat, N.e.l.; Louala, S.; Bensalah, F.; Affane, F.; Chekkal, H.; Lamri-Senhadji, M. Anti-hypertensive, anti-diabetic, hypocholesterolemic and antioxidant properties of prickly pear nopalitos in type 2 diabetic rats fed a high-fat diet. Nutr. Food Sci. 2019, 49, 476–490. [Google Scholar] [CrossRef]
- Aruwa, C.E.; Amoo, S.O.; Kudanga, T. Opuntia (Cactaceae) plant compounds, biological activities and prospects—A comprehensive review. Food Res. Int. 2018, 112, 328–344. [Google Scholar] [CrossRef]
- Aragona, M.; Lauriano, E.R.; Pergolizzi, S.; Faggio, C. Opuntia ficus - indica (L.) Miller as a source of bioactivity compounds for health and nutrition. Nat. Prod. Res. 2018, 32, 2037–2049. [Google Scholar] [CrossRef]
- Feugang, J.M.; Konarski, P.; Zou, D.; Stintzing, F.C.; Zou, C. Nutritional and medicinal use of Cactus pear (Opuntia spp.) cladodes and fruits. Front. Biosci. 2006, 11, 2574. [Google Scholar] [CrossRef]
- Santos-Díaz, M.S.; Camarena-Rangel, N.G. Cacti for production of metabolites: Current state and perspectives. Appl. Microbiol. Biotechnol. 2019, 103, 8657–8667. [Google Scholar] [CrossRef]
- Akihisa, T.; Yasukawa, K. Antitumor-promoting and anti-inflammatory activities of triterpenoids and sterols from plants and fungi. Stud. Nat. Prod. Chem. 2001, 25, 43–87. [Google Scholar]
- Harlev, E.; Nevo, E.; Solowey, E.; Bishayee, A. Cancer preventive and curative attributes of plants of the Cactaceae family: A review. Planta Med. 2013, 79, 713–722. [Google Scholar]
- Kontoyianni, M. Docking and Virtual Screening in Drug Discovery. Methods Mol. Biol. 2017, 1647, 255–266. [Google Scholar] [PubMed]
- Yusuf, M.; Hardianto, A.; Muchtaridi, M.; Nuwarda, R.F.; Subroto, T. Introduction of Docking-Based Virtual Screening Workflow Using Desktop Personal Computer. Encycl. Bioinforma. Comput. Biol. 2019, 688–699. [Google Scholar]
- Loza-Mejía, M.A.; Salazar, J.R. Sterols and triterpenoids as potential anti-inflammatories: Molecular docking studies for binding to some enzymes involved in inflammatory pathways. J. Mol. Graph. Model. 2015, 62, 18–25. [Google Scholar] [CrossRef]
- Loza-Mejía, M.; Salazar, J.; Sánchez-Tejeda, J. In Silico Studies on Compounds Derived from Calceolaria: Phenylethanoid Glycosides as Potential Multitarget Inhibitors for the Development of Pesticides. Biomolecules 2018, 8, 121. [Google Scholar] [CrossRef] [PubMed]
- Thao, N.P.; Kim, J.H.; Thuy Luyen, B.T.; Dat, N.T.; Kim, Y.H. In silico investigation of cycloartane triterpene derivatives from Cimicifuga dahurica (Turcz.) Maxim. roots for the development of potent soluble epoxide hydrolase inhibitors. Int. J. Biol. Macromol. 2017, 98, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Ochoa, R.; García, E.; Robledo, S.M.; Cardona G, W. Virtual and experimental screening of phenylfuranchalcones as potential anti-Leishmania candidates. J. Mol. Graph. Model. 2019, 91, 164–171. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, Y.; Wang, Y.; Ju, Y.; Meng, D. Virtual screening of active compounds from Artemisia argyi and potential targets against gastric ulcer based on Network pharmacology. Bioorg. Chem. 2019, 88, 102924. [Google Scholar] [CrossRef]
- Dawood, H.M.; Ibrahim, R.S.; Shawky, E.; Hammoda, H.M.; Metwally, A.M. Integrated in silico-in vitro strategy for screening of some traditional Egyptian plants for human aromatase inhibitors. J. Ethnopharmacol. 2018, 224, 359–372. [Google Scholar] [CrossRef]
- Huang, H.; Chu, C.-L.; Chen, L.; Shui, D. Evaluation of potential inhibitors of squalene synthase based on virtual screening and in vitro studies. Comput. Biol. Chem. 2019, 80, 390–397. [Google Scholar] [CrossRef]
- Medina-Franco, J.L. New Approaches for the Discovery of Pharmacologically-Active Natural Compounds. Biomolecules 2019, 9, 115. [Google Scholar] [CrossRef]
- Prieto-Martínez, F.; Medina-Franco, J.; Prieto-Martínez, F.D.; Medina-Franco, J.L. Flavonoids as Putative Epi-Modulators: Insight into Their Binding Mode with BRD4 Bromodomains Using Molecular Docking and Dynamics. Biomolecules 2018, 8, 61. [Google Scholar] [CrossRef] [PubMed]
- Salazar, J.R.; Martinez-Vazquez, M.; Ramirez-Apan, T.; Nieto-Camacho, A.; Cespedes, C.L.; Rodrfguez-Silverio, J.; Flores-Murrieta, F. Anti-Inflammatory and Cytotoxic Activities of Chichipegenin, Peniocerol, and Macdougallin Isolated from Myrtillocactus geometrizans (Mart. ex Pfeiff.) Con. Z. Fur Nat. Sect. C J. Biosci. 2011, 66. [Google Scholar]
- Salazar, J.R.; Céspedes, C.L. Phytoecdysteroids and related sterols isolated from mexican cacti: Their potential use as natural insecticides. In Natural Antioxidants and Biocides from Wild Medicinal Plants; Cabi Publishing: Wallingford, UK, 2013; ISBN 9781780642338. [Google Scholar]
- Torres-Olvera, M.; Salazar, J.R.; Soto-Cabrera, D.; Cerón-Nava, A.; Rosales-Guevara, J. Evaluation of the antimicrobial activity of extracts and compounds isolated from Hylocereus sp. Vitae 2014, 21. [Google Scholar]
- Uribe-Chiquete, R.F.; Salazar, J.R.; Ariza-Castolo, A.; Ramos-Gonzales, V.H. Antimicrobial activity of methanolic extract, peniocerol and longispinogenin extracted from Myrtillocactus geometrizans. Vitae 2014, 21, S71–S72. [Google Scholar]
- Nogueda-Gutiérrez, I.B.; Salazar, J.R.; Cerón-Nava, A.; Ramírez-Ponce, A.L.; Torres-Olvera, M.; Soto-Cabrera, D.; Ciprés-Meixueiro, A. Quantification of flavonoids and antioxidant and antimicrobial activities of the extract of Peniocereus maculatus. Vitae 2014, 21. [Google Scholar]
- Ramírez-Ponce, A.L.; Salazar, J.R.; Cerón-Nava, A.; Torres-Olvera, M.; Soto-Cabrera, D.; Nogueda-Gutiérrez, I.B. Quantification of total polyphenols, flavonoids and evaluation of the antioxidant and antimicrobial activities of Opuntia tomentosa extract. Vitae 2014, 21, S110–S111. [Google Scholar]
- Soto-Cabrera, D.; Salazar, J.R.; Nogueda-Gutiérrez, I.; Torres-Olvera, M.; Cerón-Nava, A.; Rosales-Guevara, J.; Terrazas, T.; Rosas-Acevedo, H. Quantification of polyphenols and flavonoid content and evaluation of anti-inflammatory and antimicrobial activities of Stenocereus stellatus extracts. Nat. Prod. Res. 2016, 30, 1885–1889. [Google Scholar] [CrossRef]
- Céspedes, C.L.; Salazar, J.R.; Martínez, M.; Aranda, E. Insect growth regulatory effects of some extracts and sterols from Myrtillocactus geometrizans (Cactaceae) against Spodoptera frugiperda and Tenebrio molitor. Phytochemistry 2005, 66, 2481–2493. [Google Scholar] [CrossRef]
- Pattee, A.F. Cereus Grandiflora, Cactus Grandiflora (Linn.), Night-Blooming cereus, Sweet-Scented Cactus, &c. Bost. Med. Surg. J. 1867, 75, 537–539. [Google Scholar]
- Britton, N.L.; Rose, J.N. The Cactaceae: Descriptions and Illustrations of Plants of the Cactus Family; Carnegie Institution of Washington: Washington, DC, USA, 1919. [Google Scholar]
- Heyl, G. Ueber das Vorkommen von Alkaloiden und Saponinen in Cacteen. Arch. Pharm. (Weinh.) 1901, 239, 451–473. [Google Scholar] [CrossRef]
- Ewell, E.E. THE CHEMISTRY OF THE CACTACEAE. J. Am. Chem. Soc. 1896, 18, 624–643. [Google Scholar] [CrossRef][Green Version]
- Nyffeler, R.; Eggli, U. A farewell to dated ideas and concepts: Molecular phylogenetics and a revised suprageneric classification of the family Cactaceae - Zurich Open Repository and Archive. Schumannia 2010, 6, 109–149. [Google Scholar]
- Fogleman, J.C.; Duperret, S.M.; Kircher, H.W. The role of phytosterols in host plant utilization by cactophilicDrosophila. Lipids 1986, 21, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Fogleman, J.C.; Armstrong, L. Ecological aspects of cactus triterpene glycosides I. Their effect on fitness components ofDrosophila mojavensis. J. Chem. Ecol. 1989, 15, 663–676. [Google Scholar] [CrossRef]
- Fogleman, J.C.; Danielson, P.B. Chemical Interactions in the Cactus-Microorganism-Drosophila Model System of the Sonoran Desert. American Zoologist 2001, 41, 877–889. [Google Scholar]
- Djerassi, C.; Krakower, G.W.; Lemin, A.J.; Liu, L.H.; Mills, J.S.; Villotti, R. The Neutral Constituents of the Cactus Lophocereus schottii. The Structure of Lophenol 4α-Methyl-Δ7-cholesten-3β-ol, A Link in Sterol Biogenesis. J. Am. Chem. Soc. 1958, XII, 6284–6292. [Google Scholar] [CrossRef]
- Campbell, C.E.; Kircher, H.W. Senita cactus: A plant with interrupted sterol biosynthetic pathways. Phytochemistry 1980, 19, 2777–2779. [Google Scholar] [CrossRef]
- Djerassi, C.; Knight, J.C.; Brockmann, H. Neue Sterine aus dem KaktusWilcoxia viperina. Chem. Ber. 1964, 97, 3118–3130. [Google Scholar] [CrossRef]
- Knight, J.C.; Wilkinson, D.I.; Djerassi, C. The Structure of the Cactus Sterol Macdougallin (14α-Methyl-Δ8 -cholestene-3β,6α-diol). A Novel Link in Sterol Biogenesis 1,2. J. Am. Chem. Soc. 1966, 88, 790–798. [Google Scholar] [CrossRef]
- Arias, S.; Terrazas, T.; Arreola-Nava, H.J.; Vázquez-Sánchez, M.; Cameron, K.M. Phylogenetic relationships in Peniocereus (Cactaceae) inferred from plastid DNA sequence data. J. Plant. Res. 2005, 118, 317–328. [Google Scholar] [CrossRef]
- Djerassi, C.; Murray, R.D.H.; Villotti, R. The Structure of the Cactus Sterol, Peniocerol (Cholest-8ene-3~,6a-diol). Proc. Chem. Soc. 1961, 450. [Google Scholar]
- Djerassi, C.; Knight, J.C.; Brockmann, H. The Structure of the Cactus Sterol Macdougallin (14α-Methyl-Δ8-Cholestene-3β,6α-Diol).A Novel Link in Sterol Biogenesis. J. Am. Chem. Soc. 1963, 85, 835. [Google Scholar] [CrossRef]
- Knight, J.C.; Pettit, G.R. Arizona flora: The sterols of Peniocereus greggii. Phytochemistry 1969, 8, 477–482. [Google Scholar] [CrossRef]
- Kircher, H.W. Triterpenes in organ pipe cactus. Phytochemistry 1980, 19, 2707–2712. [Google Scholar] [CrossRef]
- Kircher, H.W.; Bird, H.L. Five 3β, 6α-dihydroxysterols in organ-pipe cactus. Phytochemistry 1982, 21, 1705–1710. [Google Scholar] [CrossRef]
- Jiang, J.; Li, Y.; Chen, Z.; Min, Z.; Lou, F. Two novel C29-5beta-sterols from the stems of Opuntia dillenii. Steroids 2006, 71, 1073–1077. [Google Scholar] [CrossRef]
- Dinan, L. Phytoecdysteroids: Biological aspects. Phytochemistry 2001, 57, 325–339. [Google Scholar] [CrossRef]
- Dinan, L.; Savchenko, T.; Whiting, P. On the distribution of phytoecdysteroids in plants. Cell. Mol. Life Sci. 2001, 58, 1121–1132. [Google Scholar] [CrossRef]
- Salt, T.A.; Tocker, J.E.; Adler, J.H. Dominance of Δ5-sterols in eight species of the cactaceae. Phytochemistry 1987, 26, 731–733. [Google Scholar] [CrossRef]
- Schaller, H. The role of sterols in plant growth and development. Prog. Lipid Res. 2003, 42, 163–175. [Google Scholar] [CrossRef]
- Jayasuriya, H.; Herath, K.B.; Ondeyka, J.G.; Guan, Z.; Borris, R.P.; Tiwari, S.; De Jong, W.; Chavez, F.; Moss, J.; Stevenson, D.W.; et al. Diterpenoid, steroid, and triterpenoid agonists of liver X receptors from diversified terrestrial plants and marine sources. J. Nat. Prod. 2005, 68, 1247–1252. [Google Scholar] [CrossRef] [PubMed]
- Lusby, W.R.; Buchmann, S.L.; Feldlaufer, M.F. Pollen sterols from three species of sonoran cacti. Lipids 1993, 28, 469–470. [Google Scholar] [CrossRef]
- Standifer, L.N.; Devys, M.; Barbier, M. Pollen sterols—A mass spectrographic survey. Phytochemistry 1968, 7, 1361–1365. [Google Scholar] [CrossRef]
- Nes, W.D.; Schmidt, J.O. Isolation of 25(27)-dehydrolanost-8-enol from Cereus giganteus and its biosynthetic implications. Phytochemistry 1988, 27, 1705–1708. [Google Scholar] [CrossRef]
- Nomaguchi, K.; Tanaka, M.; Misawa, E.; Yamada, M.; Toida, T.; Iwatsuki, K.; Goto, T.; Kawada, T. Aloe vera phytosterols act as ligands for PPAR and improve the expression levels of PPAR target genes in the livers of mice with diet-induced obesity. Obes. Res. Clin. Pr. 2011, 5, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Misawa, E.; Tanaka, M.; Nomaguchi, K.; Nabeshima, K.; Yamada, M.; Toida, T.; Iwatsuki, K. Oral Ingestion of Aloe vera Phytosterols Alters Hepatic Gene. J. Agric. Food Chem. 2012. [Google Scholar] [CrossRef]
- Gomez-Flores, R.; Quintanilla-Licea, R.; Hernández-Martínez, H.C.; Samaniego-Escamilla, M.; Tamez-Guerra, P.; Monreal-Cuevas, E.; Tamez-Guerra, R.; Rodriguez-Padilla, C. Survival of Lymphoma-Bearing Mice by Pachycereus marginatus Cactus Extracts and Elucidation of Bioactive Compounds. Nat. Prod. Commun. 2019, 14. [Google Scholar] [CrossRef]
- El Kharrassi, Y.; Samadi, M.; Lopez, T.; Nury, T.; El Kebbaj, R.; Andreoletti, P.; El Hajj, H.I.; Vamecq, J.; Moustaid, K.; Latruffe, N.; et al. Biological activities of Schottenol and Spinasterol, two natural phytosterols present in argan oil and in cactus pear seed oil, on murine miroglial BV2 cells. Biochem. Biophys. Res. Commun. 2014, 446, 798–804. [Google Scholar] [CrossRef]
- Han, Y.H.; Ham, J.H.; Lee, N.J.; Park, C.H.; Shin, Y.H.; Lee, D.U. Antimutagenic activity of 5α-cholest-7-en-3β-ol, a new component from the starfish Asterina pectinifera. Biol. Pharm. Bull. 2000, 23, 1247–1249. [Google Scholar] [CrossRef]
- Raslan, A.E.; Radwan, M.M.; Ahmed, S.A.; Nafady, A.M.; Zaki, M.A.; Wanas, A.S.; Abou-Karam, M.; Shier, T.W.; Hassanean, H.A.; ElSohly, M.A. Monanchoramides A–D, ceramides from the marine sponge Monanchora clathrata with cytotoxic activity. Phytochem. Lett. 2018, 23, 83–89. [Google Scholar] [CrossRef]
- Socała, K.; Wlaź, P. Evaluation of the antidepressant- and anxiolytic-like activity of α-spinasterol, a plant derivative with TRPV1 antagonistic effects, in mice. Behav. Brain Res. 2016, 303, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Socała, K.; Nieoczym, D.; Pieróg, M.; Wlaź, P. α-Spinasterol, a TRPV1 receptor antagonist, elevates the seizure threshold in three acute seizure tests in mice. J. Neural Transm. 2015, 122, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Meneses-Sagrero, S.E.; Navarro-Navarro, M.; Ruiz-Bustos, E.; Del-Toro-Sánchez, C.L.; Jiménez-Estrada, M.; Robles-Zepeda, R.E. Antiproliferative activity of spinasterol isolated of Stegnosperma halimifolium (Benth, 1844). Saudi Pharm. J. 2017, 25, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Brusco, I.; Camponogara, C.; Carvalho, F.B.; Schetinger, M.R.C.; Oliveira, M.S.; Trevisan, G.; Ferreira, J.; Oliveira, S.M. α-Spinasterol: A COX inhibitor and a transient receptor potential vanilloid 1 antagonist presents an antinociceptive effect in clinically relevant models of pain in mice. Br. J. Pharm. 2017, 174, 4247–4262. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Li, W.Y.; Wu, D.C.; Wang, J.J.; Wu, C.H.; Liao, J.J.; Lin, C.K. In vitro activity of 2-methoxy-1,4-naphthoquinone and stigmasta-7,22-diene- 3β-ol from Impatiens balsamina L. against multiple antibiotic-resistant Helicobacter pylori. Evid. Based Complement. Altern. Med. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Egas, V.; Salazar-Cervantes, G.; Romero, I.; Méndez-Cuesta, C.A.; Rodríguez-Chávez, J.L.; Delgado, G. Anti-Helicobacter pylori metabolites from Heterotheca inuloides (Mexican arnica). Fitoterapia 2018, 127, 314–321. [Google Scholar] [CrossRef]
- Sedky, N.K.; El Gammal, Z.H.; Wahba, A.E.; Mosad, E.; Waly, Z.Y.; El-Fallal, A.A.; Arafa, R.K.; El-Badri, N. The molecular basis of cytotoxicity of α-spinasterol from Ganoderma resinaceum: Induction of apoptosis and overexpression of p53 in breast and ovarian cancer cell lines. J. Cell. Biochem. 2018, 119, 3892–3902. [Google Scholar] [CrossRef]
- Bolaños-Carrillo, M.A.; Ventura-Gallegos, J.L.; Saldivar-Jiménez, A.D.; Zentella-Dehesa, A.; Martínez-Vázquez, M. Effect of Sterols Isolated from Myrtillocactus geometrizans on Growth Inhibition of Colon and Breast Cancer Cells. Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar]
- Via, L.; Garcia-Argaeza, A.; Martínez-Vázquez, M.; Grancara, S.; Martinis, P.; Toninello, A. Mitochondrial Permeability Transition as Target of Anticancer Drugs. Curr. Pharm. Des. 2014, 20, 223–244. [Google Scholar] [CrossRef]
- Lajter, I.; Pan, S.P.; Nikles, S.; Ortmann, S.; Vasas, A.; Csupor-Löffler, B.; Forgó, P.; Hohmann, J.; Bauer, R. Inhibition of COX-2 and NF- κ B1 Gene Expression, NO Production, 5-LOX, and COX-1 and COX-2 Enzymes by Extracts and Constituents of Onopordum acanthium. Planta Med. 2015, 81, 1270–1276. [Google Scholar]
- Li, Y.-H.; Yang, Y.-F.; Li, K.; Jin, L.-L.; Yang, N.-Y.; Kong, D.-Y. 5 Alpha-Reductase and Aromatase Inhibitory Constituents from Brassica rapa L. Pollen. Chem. Pharm. Bull. (Tokyo) 2009, 57, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Viegelmann, C.; Parker, J.; Ooi, T.; Clements, C.; Abbott, G.; Young, L.; Kennedy, J.; Dobson, A.D.W.; Edrada-Ebel, R.A. Isolation and identification of antitrypanosomal and antimycobacterial active steroids from the sponge Haliclona simulans. Mar. Drugs 2014, 12, 2937–2952. [Google Scholar] [CrossRef] [PubMed]
- Zhen, X.H.; Quan, Y.C.; Jiang, H.Y.; Wen, Z.S.; Qu, Y.L.; Guan, L.P. Fucosterol, a sterol extracted from Sargassum fusiforme, shows antidepressant and anticonvulsant effects. Eur. J. Pharm. 2015, 768, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Abdul, Q.A.; Choi, R.J.; Jung, H.A.; Choi, J.S. Health benefit of fucosterol from marine algae: A review. J. Sci. Food Agric. 2016, 96, 1856–1866. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.A.; Bhakta, H.K.; Min, B.S.; Choi, J.S. Fucosterol activates the insulin signaling pathway in insulin resistant HepG2 cells via inhibiting PTP1B. Arch. Pharm. Res. 2016, 39, 1454–1464. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.A.; Tan, L.; Yang, H.; Im, Y.-G.; Im, Y.J. Structures of PPARγ complexed with lobeglitazone and pioglitazone reveal key determinants for the recognition of antidiabetic drugs. Sci. Rep. 2017, 7, 16837. [Google Scholar] [CrossRef]
- Jiang, H.; Li, J.; Chen, A.; Li, Y.; Xia, M.; Guo, P.; Yao, S.; Chen, S. Fucosterol exhibits selective antitumor anticancer activity against hela human cervical cell line by inducing mitochondrial mediated apoptosis, cell cycle migration inhibition and downregulation of m-TOR/PI3K/Akt signalling pathway. Oncol. Lett. 2018, 15, 3458–3463. [Google Scholar] [CrossRef]
- Perumal, P.; Sowmiya, R.; Prasanna kumar, S.; Ravikumar, S.; Deepak, P.; Balasubramani, G. Isolation, structural elucidation and antiplasmodial activity of fucosterol compound from brown seaweed, Sargassum linearifolium against malarial parasite Plasmodium falciparum. Nat. Prod. Res. 2018, 32, 1316–1319. [Google Scholar] [CrossRef]
- Wong, C.H.; Gan, S.Y.; Tan, S.C.; Gany, S.A.; Ying, T.; Gray, A.I.; Igoli, J.; Wan, E.; Pang, S.M.; Gany, S.A.; et al. Fucosterol inhibits the cholinesterase activities and reduces the release of pro-inflammatory mediators in lipopolysaccharide and amyloid-induced microglial cells. J. Appl. Phycol. 2018, 30, 3261–3270. [Google Scholar] [CrossRef]
- Le, C.F.; Kailaivasan, T.H.; Chow, S.C.; Abdullah, Z.; Ling, S.K.; Fang, C.M. Phytosterols isolated from Clinacanthus nutans induce immunosuppressive activity in murine cells. Int. Immunopharmacol. 2017, 44, 203–210. [Google Scholar] [CrossRef]
- Banzouzi, J.T.; Soh, P.N.; Ramos, S.; Hemez, J.; Soh, P.N.; Ramos, S.; Toto, P.; Hemez, J. Samvisterin, a new natural antiplasmodial betulin derivative From Uapaca paludosa (Euphorbiaceae). J. Ethnopharmacol. 2015, 173, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-L.; Lunga, P.-K.; Zhao, Y.-L.; Qin, X.-J.; Yang, X.-W.; Liu, Y.-P.; Luo, X.-D. Antibacterial constituents from Melodinus suaveolens. Chin. J. Nat. Med. 2015, 13, 307–310. [Google Scholar] [PubMed]
- Rajavel, T.; Packiyaraj, P.; Suryanarayanan, V.; Singh, S.K.; Ruckmani, K.; Pandima Devi, K. β-Sitosterol targets Trx/Trx1 reductase to induce apoptosis in A549 cells via ROS mediated mitochondrial dysregulation and p53 activation. Sci. Rep. 2018, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- GUPTA, R.; SHARMA, A.K.; DOBHAL, M.P.; SHARMA, M.C.; GUPTA, R.S. Antidiabetic and antioxidant potential of β-sitosterol in streptozotocin-induced experimental hyperglycemia. J. Diabetes 2011, 3, 29–37. [Google Scholar] [CrossRef]
- Chithambo, B.; Noundou, X.S.; Krause, R.W.M. Anti-malarial synergy of secondary metabolites from Morinda lucida Benth. J. Ethnopharmacol. 2017, 199, 91–96. [Google Scholar] [CrossRef]
- Chen, J.; Jiao, R.; Jiang, Y.; Bi, Y.; Chen, Z.-Y. Algal Sterols are as Effective as β-Sitosterol in Reducing Plasma Cholesterol Concentration. J. Agric. Food Chem. 2014, 62, 675–681. [Google Scholar] [CrossRef]
- Loizou, S.; Lekakis, I.; Chrousos, G.P.; Moutsatsou, P. β-Sitosterol exhibits anti-inflammatory activity in human aortic endothelial cells. Mol. Nutr. Food Res. 2010, 54, 551–558. [Google Scholar] [CrossRef]
- Gibson, A.C.; Horak, K.E. Systematic Anatomy and Phylogeny of Mexican Columnar Cacti. Ann. Mo. Bot. Gard. 1978, 65, 999. [Google Scholar] [CrossRef]
- Kinoshita, K.; Akiba, M.; Saitoh, M.; Ye, Y.; Koyama, K.; Takahashi, K.; Kondo, N.; Yuasa, H. Antinociceptive effect of triterpenes from cacti. Pharm. Biol. 1998, 36, 50–55. [Google Scholar] [CrossRef]
- Takizawa, T.; Kenoshita, K.; Koyama, K.; Takahashi, K.; Kondo, N.; Yuasa, H.; Kawai, K.I. A new type of triterpene from trichocereus pachanoi. J. Nat. Prod. 1993, 56, 2183–2185. [Google Scholar] [CrossRef]
- Kinoshita, K.; Takizawa, T.; Koyama, K.; Takahashi, K.; Kondo, N.; Yuasa, H.; Kawai, K.-I. New Triterpenes from Trichocereus pachanoi. J. Nat. Prod. 1995, 58, 1739–1744. [Google Scholar] [CrossRef]
- Djerassi, C.; Bowers, A.; Burstein, S.; Estrada, H.; Grossman, J.; Herrán, J.; Lemin, A.J.; Manjarrez, A.; Pakrashi, S.C. Terpenoids. XXII. 1 Triterpenes from Some Mexican and South American Plants2. J. Am. Chem. Soc. 1956, 78, 2312–2315. [Google Scholar] [CrossRef]
- Djerassi, C.; Farkas, E.; Lemin, A.J.; Collins, J.C.; Walls, F. Terpenoids. VI. Dumortierigenin, a New Triterpene Lactone from the Cactus Lemaireocereus dumortieri. J. Am. Chem. Soc. 1954, 76, 2969–2973. [Google Scholar] [CrossRef]
- Djerassi, C.; Robinson, C.H.; Thomas, D.B. Terpenoids. XXV. 1 The Structure of the Cactus Triterpene Dumortierigenin 2,3. J. Am. Chem. Soc. 1956, 78, 5685–5691. [Google Scholar] [CrossRef]
- Kinoshita, K.; Koyama, K.; Takahashi, K.; Kondo, N.; Yuasa, H. A new triterpenoid saponin from Isolatocereus dumortieri. J. Nat. Prod. 2000, 63, 701–703. [Google Scholar] [CrossRef]
- Djerassi, C.; Burstein, S.; Estrada, H.; Lemin, A.J.; Lippman, A.E.; Manjarrez, A.; Monsimer, H.G. Terpenoids. XXVIII. 1 The Triterpene Composition of the Genus Myrtillocactus2. J. Am. Chem. Soc. 1957, 79, 3525–3528. [Google Scholar] [CrossRef]
- Sandoval, A.; Manjarrez, A.; Leeming, P.R.; Thomas, G.H.; Djerassi, C. Terpenoids. XXX. The Structure of the Cactus Triterpene Chichipegenin. J. Am. Chem. Soc. 1957, 79, 4468–4472. [Google Scholar] [CrossRef]
- Djerassi, C.; Monsimer, H.G. Terpenoids. XXVII. 1 The Structure of the Cactus Triterpene Myrtillogenic Acid2. J. Am. Chem. Soc. 1957, 79, 2901–2905. [Google Scholar] [CrossRef]
- Djerassi, C.; Thomas, G.H.; Monsimer, H. Terpenoids. XVI. 1 The Constitution of the Cactus Triterpene Cochalic Acid. Partial Reductions of Methyl Diketoechinocystate2. J. Am. Chem. Soc. 1955, 77, 3579–3582. [Google Scholar] [CrossRef]
- Djerassi, C.; Thomas, G.H. Terpenoids. XII. Constitution of the cactus triterpene cochalic acid. Chem. Ind. 1954, 1354. [Google Scholar]
- Shamma, M.; Rosenstock, P. The Triterpenes of Heliabravoa chende. J. Org. Chem. 1959, 24, 726–728. [Google Scholar] [CrossRef]
- Khong, P.W.; Lewis, K.G. New triterpenoid extractives from Lemaireocereus chichipe. Aust. J. Chem. 1975, 28, 165–172. [Google Scholar] [CrossRef]
- Djerassi, C.; Geller, L.E.; Lemin, A.J. Terpenoids. VIII 1. The Structures of the Cactus Triterpenes Gummosogenin and longispinogenin. J. Am. Chem. Soc. 1954, 76, 4089–4091. [Google Scholar] [CrossRef]
- Takizawa, T.; Kinoshita, K.; Koyama, K.; Takahashi, K.; Kondo, N.; Yuasa, H. A NEW TRITERPENE FROM RATHBUNIA ALAMOSENSZS skeleton named pachanane), from isolation of a new triterpene, alamoseno- glycosides of the aerial parts of Rathbunia on the molecular ion at mlz 472 EM ]’ ring C of 1. The ’, C-nrnr values of the the methyls. J. Nat. Prod. 1995, 58, 1913–1914. [Google Scholar] [CrossRef]
- Djerassi, C.; Liu, L.H.; Farkas, E.; Lippman, A.E.; Lemin, A.J.; Geller, L.E.; McDonald, R.N.; Taylor, B.J. Terpenoids. XI.1 investigation of nine cactus species. Isolation of two new triterpenes, stellatogenin and machaeric acid2. J. Am. Chem. Soc. 1955, 77, 1200–1203. [Google Scholar] [CrossRef]
- Wollenweber, E.; Dörr, M. Wax composition of the two cacti Hylocereus purpusii and Stenocereus beneckii. Biochem. Syst. Ecol. 1995, 23, 577. [Google Scholar] [CrossRef]
- Djerassi, C.; Geller, L.E.; Lemin, A.J. Terpenoids. I. The Triterpenes of the Cactus. J. Am. Chem. Soc. 1953, 75, 2254–2256. [Google Scholar] [CrossRef]
- Djerassi, C.; Henry, J.A.; Lemin, A.J.; Rios, T.; Thomas, G.H. Terpenoids. XXIV. The structure of the Cactustriterpene Queretaroic Acid. J. Am. Chem. Soc. 1956, 78, 3783–3787. [Google Scholar] [CrossRef]
- Djerassi, C.; McDonald, R.M.; Lemin, A.J. Terpenoids. III. The Isolation of Erythrodiol, Oleanolic Acid and a New Triterpene Triol. Loneispinoeenin, from the Cactus Lemaireocereus lonsispinus. J. Am. Chem. Soc. 1953, 75, 5940–5942. [Google Scholar] [CrossRef]
- Koyama, K.; Yama, T.; Kinoshita, K.; Takahashi, K.; Kondo, N.; Yuasa, H. New triterpenes from cactaceous plants. J. Nat. Prod. 1993, 56, 2201–2203. [Google Scholar] [CrossRef]
- Okazaki, S.; Kinoshita, K.; Koyama, K.; Takahashi, K.; Yuasa, H. New triterpene saponins from Stenocereus eruca (Cactaceae). J. Nat. Med. 2007, 61, 24–29. [Google Scholar] [CrossRef]
- Ye, Y.; Kinoshita, K.; Koyama, K.; Takahashi, K.; Kondo, N.; Yuasa, H. New triterpenes from Machaerocereus eruca. J. Nat. Prod. 1998, 61, 456–460. [Google Scholar] [CrossRef]
- Djerassi, C.; Lippman, A.E. Terpenoids. X. The Triterpenes of the Cactus Lemaireocereus hystrix. J. Am. Chem. Soc. 1954, 76, 5780–5781. [Google Scholar] [CrossRef]
- Djerassi, C.D.; Lippman, A.E. Terpenoids. XIII.1 the structures of the cactus triterpenes machaeric acid and machaerinic acid. J. Am. Chem. Soc. 1955, 77, 1825–1828. [Google Scholar] [CrossRef]
- Imai, T.; Okazaki, S.; Kinoshita, K.; Koyama, K.; Takahashi, K.; Yuasa, H. Triterpenoid saponins from cultural plants of Stenocereus stellatus (Cactaceae). J. Nat. Med. 2006, 60, 49–53. [Google Scholar] [CrossRef]
- Marx, M.; Leclercq, J.; Tursch, B.; Djerassi, C. Terpenoids. LX. Revised structures of the cactus triterpene lactones stellatogenin and thurberogenin. J. Org. Chem. 1967, 32, 3150–3155. [Google Scholar] [CrossRef]
- Jolad, S.; Steelink, C. Thurberin, a new pentacyclic triterpene from organ-pipe cactus. J. Org. Chem. 1969, 34, 1367–1369. [Google Scholar] [CrossRef]
- Kaspryzyk, Z.; Pyrek, J.; Jolad, S.D.; Steelink, C. The identity of calenduladiol and thurberin: A lupenediol found in marigold flowers and organ pipe cactus. Phytochemistry 1970, 9, 2065–2066. [Google Scholar] [CrossRef]
- Kircher, H.W. Triterpene glycosides and queretaroic acid in organ pipe cactus. Phytochemistry 1977, 16, 1078–1080. [Google Scholar] [CrossRef]
- Djerassi, C.; Mills, J.S. Terpenoids. XXXII. The Structure of the Cactus Triterpene Treleasegenic Acid. Ring Conformational Alterations in a Pentacyclic Triterpene. J. Am. Chem. Soc. 1958, 80, 1236–1243. [Google Scholar] [CrossRef]
- Fomogne-fodjo, M.C.Y.; Ndinteh, D.T.; Olivier, D.K.; Kempgens, P.; Vuuren, S. Van Secondary metabolites from Tetracera potatoria stem bark with anti- mycobacterial activity. J. Ethnopharmacol. 2017, 195, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Seong, S.H.; Roy, A.; Jung, H.A.; Jung, H.J.; Choi, J.S. Protein tyrosine phosphatase 1B and α-glucosidase inhibitory activities of Pueraria lobata root and its constituents. J. Ethnopharmacol. 2016. [Google Scholar] [CrossRef]
- Kakarla, L.; Katragadda, S.B.; Tiwari, A.K.; Kotamraju, K.S.; Madhusudana, K.; Kumar, D.A.; Botlagunta, M. Free radical scavenging, α-glucosidase inhibitory and anti-inflammatory constituents from Indian sedges, Cyperus scariosus R.Br and Cyperus rotundus L. Pharm. Mag. 2016, 12, S488–S496. [Google Scholar]
- Moridi Farimani, M.; Nazarianpoor, E.; Rustaie, A.; Akhbari, M. Phytochemical constituents and biological activities of Cleome iberica DC. Nat. Prod. Res. 2017, 31, 1329–1332. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.K.; Mishra, S.; Ali, W.; Shukla, Y. Protective effects of lupeol against mancozeb-induced genotoxicity in cultured human lymphocytes. Phytomedicine 2016, 23, 714–724. [Google Scholar] [CrossRef] [PubMed]
- Rauth, S.; Ray, S.; Bhattacharyya, S.; Mehrotra, D.G.; Alam, N.; Mondal, G.; Nath, P.; Roy, A.; Biswas, J.; Murmu, N. Lupeol evokes anticancer effects in oral squamous cell carcinoma by inhibiting oncogenic EGFR pathway. Mol. Cell. Biochem. 2016, 417, 97–110. [Google Scholar] [CrossRef]
- Jin, T.; Yu, H.; Huang, X.-F. Selective binding modes and allosteric inhibitory effects of lupane triterpenes on protein tyrosine phosphatase 1B. Sci. Rep. 2016, 6, 20766. [Google Scholar] [CrossRef]
- Gajos-Michniewicz, A.; Czyz, M. Modulation of WNT/β-catenin pathway in melanoma by biologically active components derived from plants. Fitoterapia 2016, 109, 283–292. [Google Scholar] [CrossRef]
- Lee, S.; Jung, K.; Lee, D.; Rak, S.; Ro, K.; Sung, K.; Hyun, K. Protective effect and mechanism of action of lupane triterpenes from Cornus walteri in cisplatin-induced nephrotoxicity. Bioorg. Med. Chem. Lett. 2015, 25, 5613–5618. [Google Scholar] [CrossRef]
- Wang, C.M.; Chen, H.T.; Wu, Z.Y.; Jhan, Y.L.; Shyu, C.L.; Chou, C.H. Antibacterial and synergistic activity of pentacyclic triterpenoids isolated from Alstonia scholaris. Molecules 2016, 21, 139. [Google Scholar] [CrossRef]
- Do Carmo, D.F.M.; Amaral, A.C.F.; Machado, M.; Lopes, D.; Echevarria, A.; Rosário, V.E.; Silva, J.R.d.A. Evaluation of Antiplasmodial activity of extracts and constituents from Ampelozizyphus amazonicus. Pharm. Mag. 2015, 11, S244–S250. [Google Scholar]
- Navid, M.H.; Laszczyk-lauer, M.N.; Reichling, J.; Schnitzler, P. Phytomedicine Pentacyclic triterpenes in birch bark extract inhibit early step of herpes simplex virus type 1 replication. Eur. J. Integr. Med. 2014, 21, 1273–1280. [Google Scholar]
- Xu, G.-m.; Zan, T.; Li, H.-y.; Han, J.-f.; Liu, Z.-m.; Huang, J.; Dong, L.-h.; Zhang, H.-n. Betulin inhibits lipopolysaccharide/D-galactosamine-induced acute liver injury in mice through activating PPAR-γ. Biomed. Pharm. 2018, 106, 941–945. [Google Scholar] [CrossRef] [PubMed]
- Nobsathian, S.; Bullangpoti, V.; Kumrungsee, N.; Wongsa, N.; Ruttanakum, D. Larvicidal effect of compounds isolated from Maerua siamensis (Capparidaceae) against Aedes aegypti (Diptera: Culicidae) larvae. Chem. Biol. Technol. Agric. 2018, 5, 4–11. [Google Scholar] [CrossRef]
- Prasad, N.; Sabarwal, A.; Yadav, U.C.S.; Singh, R.P. Lupeol induces S-phase arrest and mitochondria-mediated apoptosis in cervical cancer cells. J. Biosci. 2018, 43, 249–261. [Google Scholar] [CrossRef]
- He, W.; Li, X.; Xia, S. Lupeol triterpene exhibits potent antitumor effects in A427 human lung carcinoma cells via mitochondrial mediated apoptosis, ROS generation, loss of mitochondrial membrane potential and downregulation of m-TOR/PI3Ksol;AKT signalling pathway. J. Buon. 2018, 23, 635–640. [Google Scholar]
- Somtimuang, C.; Olatunji, O.J.; Ovatlarnporn, C. Evaluation of In Vitro α-Amylase and α-Glucosidase Inhibitory Potentials of 14 Medicinal Plants Constituted in Thai Folk Antidiabetic Formularies. Chem. Biodivers. 2018, 15. [Google Scholar] [CrossRef]
- Kaur, G.; Chauhan, K.; Kaur, S. Lupeol induces immunity and protective efficacy in a murine model against visceral leishmaniasis. Parasitology 2019. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Jiang, M.; Xie, X.; Yang, H.; Wang, X.; Xiao, L.; Wang, N. Oleanolic acid ameliorates high glucose-induced endothelial dysfunction via PPARδ activation. Sci. Rep. 2017, 7, 3–10. [Google Scholar] [CrossRef]
- Nguyen, H.T.; Ho, D.V.; Vo, H.Q.; Le, A.T.; Nguyen, H.M.; Kodama, T.; Ito, T.; Morita, H.; Raal, A. Antibacterial activities of chemical constituents from the aerial parts of Hedyotis pilulifera. Pharm. Biol. 2017, 55, 787–791. [Google Scholar] [CrossRef]
- Mishra, T.; Arya, R.K.; Meena, S.; Joshi, P.; Pal, M.; Meena, B.; Upreti, D.K.; Rana, T.S.; Datta, D. Isolation, characterization and anticancer potential of cytotoxic triterpenes from Betula utilis bark. PLoS ONE 2016, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Sibiya, H.P.; Mabandla, M.V.; Musabayane, C.T. The effects of transdermally delivered oleanolic acid on malaria parasites and blood glucose homeostasis in P. berghei-infected male Sprague-Dawley rats. PLoS ONE 2016, 11, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Wen, X.; Zhang, W.; Wang, C.; Liu, J.; Liu, C. Hypolipidemic effect of oleanolic acid is mediated by the miR-98-5p/PGC-1β axis in high-fat diet-induced hyperlipidemic mice. FASEB J. 2017, 31, 1085–1096. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.J.; Lee, H.J.; Lee, H.E.; Park, S.J.; Gwon, Y.; Kim, H.; Zhang, J.; Shin, C.Y.; Kim, D.H.; Ryu, J.H. Oleanolic acid ameliorates cognitive dysfunction caused by cholinergic blockade via TrkB-dependent BDNF signaling. Neuropharmacology 2017, 113, 100–109. [Google Scholar] [CrossRef]
- Giner-Larza, E.M.; Máez, S.; Recio, M.C.; Giner, R.M.; Prieto, J.M.; Cerdá-Nicolás, M.; Ríos, J.L. Oleanonic acid, a 3-oxotriterpene from Pistacia, inhibits leukotriene synthesis and has anti-inflammatory activity. Eur. J. Pharm. 2001, 428, 137–143. [Google Scholar] [CrossRef]
- Cao, S.; Dong, X.L.; Ho, M.X.; Yu, W.X.; Wong, K.C.; Yao, X.S.; Wong, M.S. Oleanolic acid exerts osteoprotective effects and modulates vitamin D metabolism. Nutrients 2018, 10, 247. [Google Scholar] [CrossRef]
- Xu, G.; Xiao, Y.; Zhang, Q.; Zhou, M. Hepatoprotective natural triterpenoids. Eur. J. Med. Chem. 2018, 145, 691–716. [Google Scholar] [CrossRef]
- Montesino, N.L.; Schmidt, T.J. Salvia species as sources of natural products with antiprotozoal activity. Int. J. Mol. Sci. 2018, 19. [Google Scholar]
- Iranshahy, M.; Iranshahi, M.; Abtahi, S.R.; Karimi, G. The role of nuclear factor erythroid 2-related factor 2 in hepatoprotective activity of natural products: A review. Food Chem. Toxicol. 2018, 120, 261–276. [Google Scholar] [CrossRef]
- Chunhua, M.; Hongyan, L. NeuroToxicology Protective effect of betulin on cognitive decline in streptozotocin (STZ) -induced diabetic rats. Neurotoxicology 2016, 57, 104–111. [Google Scholar]
- Muceniece, R.; Saleniece, K.; Rumaks, J.; Krigere, L.; Dzirkale, Z.; Mezhapuke, R.; Zharkova, O.; Klusa, V. Betulin binds to γ-aminobutyric acid receptors and exerts anticonvulsant action in mice. Pharm. Biochem. Behav. 2008, 90, 712–716. [Google Scholar] [CrossRef] [PubMed]
- Król, S.K.; Kielbus, M.; Rivero-Müller, A.; Stepulak, A. Comprehensive review on betulin as a potent anticancer agent. Biomed. Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Verma, V.; Tripathi, A.C.; Saraf, S.K. Bioactive non-sterol triterpenoid from Streblus asper: Microwave-assisted extraction, HPTLC profiling, computational studies and neuro-pharmacological evaluation in BALB/c mice. Pharm. Biol. 2016, 54, 2454–2464. [Google Scholar] [CrossRef] [PubMed]
- Laavola, M.; Haavikko, R.; Hämäläinen, M.; Leppänen, T.; Nieminen, R.; Alakurtti, S.; Moreira, V.M.; Yli-Kauhaluoma, J.; Moilanen, E. Betulin Derivatives Effectively Suppress Inflammation in Vitro and in Vivo. J. Nat. Prod. 2016, 79, 274–280. [Google Scholar] [CrossRef]
- Ratna Wulan, D.; Priyo Utomo, E.; Mahdi, C. Antidiabetic Activity of Ruellia tuberosa L., Role of α-Amylase Inhibitor: In Silico, in Vitro, and in Vivo Approaches. Biochem. Res. Int. 2015, 2015. [Google Scholar] [CrossRef]
- Zhang, S.-Y.; Zhao, Q.-F.; Fang, N.-N.; Yu, J.-G. Betulin inhibits pro-inflammatory cytokines expression through activation STAT3 signaling pathway in human cardiac cells. Eur. Rev. Med. Pharm. Sci. 2015, 19, 455–460. [Google Scholar]
- Jamila, N.; Khairuddean, M.; Yeong, K.K.; Osman, H.; Murugaiyah, V. Cholinesterase inhibitory triterpenoids from the bark of Garcinia hombroniana. J. Enzym. Inhib. Med. Chem. 2015, 30, 133–139. [Google Scholar] [CrossRef]
- Akihisa, T.; Ogihara, J.; Kato, J.; Yasukawa, K.; Ukiya, M.; Yamanouchi, S.; Oishi, K. Inhibitory effects of triterpenoids and sterols on human immunodeficiency virus-1 reverse transcriptase. Lipids 2001, 36, 507–512. [Google Scholar] [CrossRef]
- Tsai, C.W.; Tsai, R.T.; Liu, S.P.; Chen, C.S.; Tsai, M.C.; Chien, S.H.; Hung, H.S.; Lin, S.Z.; Shyu, W.C.; Fu, R.H. Neuroprotective Effects of Betulin in Pharmacological and Transgenic Caenorhabditis elegans Models of Parkinson’s Disease. Cell Transpl. 2017, 26, 1903–1918. [Google Scholar] [CrossRef]
- Zhou, Z.; Zhu, C.; Cai, Z.; He, L.; Lou, X.; Qi, X. Betulin induces cytochrome c release and apoptosis in colon cancer cells via NOXA. Oncol. Lett. 2018, 7319–7327. [Google Scholar] [CrossRef]
- Ullah, F.; Hussain, H.; Hussain, J.; Bukhari, I.A.; Khan, M.T.H.; Choudhary, M.I.; Gilani, A.H.; Ahmad, V.U. Tyrosinase inhibitory pentacyclic triterpenes and analgesic and spasmolytic activities of methanol extracts of Rhododendron collettianum. Phyther. Res. 2007, 21, 1076–1081. [Google Scholar] [CrossRef] [PubMed]
- Goswami, P.; Paul, S.; Banerjee, R.; Kundu, R.; Mukherjee, A. Betulinic acid induces DNA damage and apoptosis in SiHa cells. Mutat. Res. Toxicol. Env.. Mutagen. 2018, 828, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zhan, X.K.A.I.; Li, J.U.N.L.; Zhang, S.E.N.; Xing, P.U.Y.; Xia, M.F.A.N. Betulinic acid exerts potent antitumor effects on paclitaxel - resistant human lung carcinoma cells ( H460 ) via G2 / M phase cell cycle arrest and induction of mitochondrial apoptosis. Oncol. Lett. 2018, 16, 3628–3634. [Google Scholar] [CrossRef] [PubMed]
- Gomes Castro, A.J.; Cazarolli, L.H.; Bretanha, L.C.; Sulis, P.M.; Rey Padilla, D.P.; Aragón Novoa, D.M.; Dambrós, B.F.; Pizzolatti, M.G.; Mena Barreto Silva, F.R. The potent insulin secretagogue effect of betulinic acid is mediated by potassium and chloride channels. Arch. Biochem. Biophys. 2018, 648, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Bossolani, G.D.P.; Ueda-Nakamura, T.; Silva, S.O.; Dias Filho, B.P.; Costa, T.O.G.; Quintanilla, R.H.R.; Martinez, S.T.; Veiga, V.F.; Pinto, A.C.; Nakamura, C.V. Anti-trypanosoma activity and synergistic effects of natural and semi-synthetic triterpenes and predominant cell death through autophagy in amastigote forms. J. Braz. Chem. Soc. 2017, 28, 2473–2489. [Google Scholar] [CrossRef]
- De Almeida, P.D.O.; Boleti, A.P.d.A.; Rüdiger, A.L.; Lourenço, G.A.; da Veiga Junior, V.F.; Lima, E.S. Anti-Inflammatory Activity of Triterpenes Isolated from Protium paniculatum Oil-Resins. Evid. Based Complement. Altern. Med. 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Akihisa, T.; Franzblau, S.G.; Ukiya, M.; Okuda, H.; Zhang, F.; Yasukawa, K.; Suzuki, T.; Kimura, Y. Antitubercular Activity of Triterpenoids from Asteraceae Flowers. Biol. Pharm. Bull. 2005, 28, 158–160. [Google Scholar] [CrossRef]
- Liu, K.; Qin, Y.H.; Yu, J.Y.; Ma, H.; Song, X.L. 3-rythrodiol isolated from Conyza canadensis inhibits MKN-45 human gastric cancer cell proliferation by inducing apoptosis, cell cycle arrest, DNA fragmentation, ROS generation and reduces tumor weight and volume in mouse xenograft model. Oncol. Rep. 2016, 35, 2328–2338. [Google Scholar] [CrossRef]
- Manayi, A.; Hadjiakhoondi, A.; Ardekani, M.R.S.; Vazirian, M.; Khanavi, M.; Saeidnia, S.; Ostad, S.N.; Akhtar, Y. Chemical Constituents and Cytotoxic Effect of the Main Compounds of Lythrum salicaria L. Z. Fur Nat. Sect. C J. Biosci. 2013, 68, 367–375. [Google Scholar]
- Martín, R.; Miana, M.; Jurado-López, R.; Martínez-Martínez, E.; Gómez-Hurtado, N.; Delgado, C.; Bartolomé, M.; Román, J.A.; Cordova, C.; Lahera, V.; et al. Diol triterpenes block profibrotic effects of angiotensin II and protect from cardiac hypertrophy. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Martín, R.; Hernández, M.; Córdova, C.; Nieto, M.L. Natural triterpenes modulate immune-inflammatory markers of experimental autoimmune encephalomyelitis: Therapeutic implications for multiple sclerosis. Br. J. Pharm. 2012, 166, 1708–1723. [Google Scholar] [CrossRef] [PubMed]
- Wansi, J.D.; Chiozem, D.D.; Tcho, A.T.; Toze, F.A.A.; Devkota, K.P.; Ndjakou, B.L.; Wandji, J.; Sewald, N. Antimicrobial and antioxidant effects of phenolic constituents from Klainedoxa gabonensis. Pharm. Biol. 2010, 48, 1124–1129. [Google Scholar] [CrossRef] [PubMed]
- Máñez, S.; Recio, M.C.; Giner, R.M.; Ríos, J.L. Effect of selected triterpenoids on chronic dermal inflammation. Eur. J. Pharm. 1997, 334, 103–105. [Google Scholar] [CrossRef]
- Kinoshita, K.; Yang, Y.; Koyama, K.; Takahashi, K.; Nishino, H. Inhibitory effect of some triterpenes from cacti on 32Pi-incorporation into phospholipids of HeLa cells promoted by 12-0-tetradecanoylphorbol-13-acetate. Phytomedicine 1999, 6, 73–77. [Google Scholar] [CrossRef]
- Yasukawa, K.; Akihisa, T.; Oinuma, H.; Kasahara, Y.; Kimura, Y.; Yamanouchi, S.; Kumaki, K.; Tamura, T.; Takido, M. Inhibitory effect of di- and trihydroxy triterpenes from the flowers of compositae on 12-O-tetradecanoylphorbol-13-acetate-induced inflammation in mice. Biol. Pharm. Bull. 1996, 19, 1329–1331. [Google Scholar] [CrossRef]
- Aragão, G.F.; Carneiro, L.M.V.; Junior, A.P.F.; Vieira, L.C.; Bandeira, P.N.; Lemos, T.L.G.; Viana, G.S.d.B. A possible mechanism for anxiolytic and antidepressant effects of alpha- and beta-amyrin from Protium heptaphyllum (Aubl.) March. Pharm. Biochem. Behav. 2006, 85, 827–834. [Google Scholar]
- Xu, F.; Huang, X.; Wu, H.; Wang, X. Beneficial health effects of lupenone triterpene: A review. Biomed. Pharm. 2018, 103, 198–203. [Google Scholar] [CrossRef]
- Çevik, D.; Burçin Yılmazgöz, Ş.; Kan, Y.; Akhan Güzelcan, E.; Durmaz, I.; Çetin-Atalay, R.; Kırmızıbekmez, H. Bioactivity-guided isolation of cytotoxic secondary metabolites from the roots of Glycyrrhiza glabra and elucidation of their mechanisms of action. Ind. Crop. Prod. 2018, 124, 389–396. [Google Scholar] [CrossRef]
- Subba Rao, G.; Sinsheimer, J.E.; Cochran, K.W. Antiviral activity of triterpenoid saponins containing acylated β-amyrin aglycones. J. Pharm. Sci. 1974, 63, 471–473. [Google Scholar] [CrossRef]
- Wei, C.C.; Chang, C.H.; Liao, V.H.C. Anti-Parkinsonian effects of β-amyrin are regulated via LGG-1 involved autophagy pathway in Caenorhabditis elegans. Phytomedicine 2017, 36, 118–125. [Google Scholar] [CrossRef]
- Zhang, Y.; Jayaprakasam, B.; Seeram, N.P.; Olson, L.K.; DeWitt, D.; Nair, M.G. Insulin Secretion and Cyclooxygenase Enzyme Inhibition by Cabernet Sauvignon Grape Skin Compounds. J. Agric. Food Chem. 2004, 52, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Rivero-Cruz, J.F.; Zhu, M.; Kinghorn, A.D.; Wu, C.D. Antimicrobial constituents of Thompson seedless raisins (Vitis vinifera) against selected oral pathogens. Phytochem. Lett. 2008, 1, 151–154. [Google Scholar] [CrossRef]
- Thao, N.P.; Luyen, B.T.T.; Koo, J.E.; Kim, S.; Koh, Y.S.; Van Thanh, N.; Cuong, N.X.; Van Kiem, P.; Van Minh, C.; Kim, Y.H. In vitro anti-inflammatory components isolated from the carnivorous plant Nepenthes mirabilis (Lour.) Rafarin. Pharm. Biol. 2016, 54, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Espinosa, J.J.; García-Jiménez, S.; Rios, M.Y.; Medina-Franco, J.L.; López-Vallejo, F.; Webster, S.P.; Binnie, M.; Ibarra-Barajas, M.; Ortiz-Andrade, R.; Estrada-Soto, S. Antihyperglycemic and sub-chronic antidiabetic actions of morolic and moronic acids, in vitro and in silico inhibition of 11β-HSD 1. Phytomedicine 2013, 20, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Giner-Larza, E.M.; Máñez, S.; Giner, R.M.; Recio, M.C.; Prieto, J.M.; Cerdá-Nicolás, M.; Ríos, J.L. Anti-inflammatory triterpenes from Pistacia terebinthus galls. Planta Med. 2002, 68, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Gurovic, M.S.V.; Castro, M.J.; Richmond, V.; Faraoni, M.B.; Maier, M.S.; Murray, A.P. Triterpenoids with acetylcholinesterase inhibition from chuquiraga erinacea D. Don. subsp. erinacea (Asteraceae). Planta Med. 2010, 76, 607–610. [Google Scholar] [CrossRef]
- Castro, M.J.; Richmond, V.; Romero, C.; Maier, M.S.; Estévez-Braun, A.; Ravelo, Á.G.; Faraoni, M.B.; Murray, A.P. Preparation, anticholinesterase activity and molecular docking of new lupane derivatives. Bioorganic Med. Chem. 2014, 22, 3341–3350. [Google Scholar] [CrossRef]
- Okahara, F.; Suzuki, J.; Hashizume, K.; Osaki, N.; Shimotoyodome, A. Triterpene alcohols and sterols from rice bran reduce postprandial hyperglycemia in rodents and humans. Mol. Nutr. Food Res. 2016, 60, 1521–1531. [Google Scholar] [CrossRef]
- Radha, M.; Laxmipriya, N. Evaluation of biological properties and clinical effectiveness of Aloe vera: A systematic review. J. Tradit. Complement. Med. 2015, 5, 21–26. [Google Scholar] [CrossRef]
- Ikeda, I.; Nakashima-Yoshida, K.; Sugano, M. Effects of Cycloartenol on Absorption and Serum Levels of Cholesterol in Rats. J. Nutr. Sci. Vitam. 1985, 31, 375–384. [Google Scholar] [CrossRef]
- Momo, I.; Kuete, V.; Dufat, H.; Michel, S.; Wandji, J. Antimicrobial Activity of the Methanolic Extract and Compounds From the Stem Bark of Garcinia Lucida Vesque (Clusiaceae). Int. J. Pharm. Pharm. Sci. 2011, 3, 215–217. [Google Scholar]
- Benmerache, A.; Alabdul Magid, A.; Labed, A.; Kabouche, A.; Voutquenne-Nazabadioko, L.; Hubert, J.; Morjani, H.; Kabouche, Z. Isolation and characterisation of cytotoxic compounds from Euphorbia clementei Boiss. Nat. Prod. Res. 2017, 31, 2091–2098. [Google Scholar] [CrossRef] [PubMed]
- Pujirahayu, N.; Bhattacharjya, D.K.; Suzuki, T.; Katayama, T. α-Glucosidase Inhibitory Activity of Cycloartane-Type Triterpenes Isolated from Indonesian Stingless Bee Propolis and Their Structure–Activity Relationship. Pharmaceuticals 2019, 12, 102. [Google Scholar] [CrossRef]
- Niu, H.; Li, X.; Yang, A.; Jin, Z.; Wang, X.; Wang, Q.; Yu, C.; Wei, Z.; Dou, C. Cycloartenol exerts anti-proliferative effects on Glioma U87 cells via induction of cell cycle arrest and p38 MAPK-mediated apoptosis. JBUON 2018, 23, 1840–1845. [Google Scholar]
- Tung, N.H.; Kwon, H.J.; Kim, J.H.; Ra, J.C.; Kim, J.A.; Kim, Y.H. An anti-influenza component of the bark of alnus japonica. Arch. Pharm. Res. 2010, 33, 363–367. [Google Scholar] [CrossRef]
- ACD/ChemSketch, version 12; Advanced Chemistry Development, Inc.: Toronto, ON, Canada, 2019.
- Thomsen, R.; Christensen, M.H. MolDock: A New Technique for High-Accuracy Molecular Docking. J. Med. Chem. 2006, 49, 3315–3321. [Google Scholar] [CrossRef]
- Gupta, K.; Selinsky, B.S.; Kaub, C.J.; Katz, A.K.; Loll, P.J. The 2.0 A resolution crystal structure of prostaglandin H2 synthase-1: Structural insights into an unusual peroxidase. J. Mol. Biol. 2004, 335, 503–518. [Google Scholar] [CrossRef]
- Duggan, K.C.; Walters, M.J.; Musee, J.; Harp, J.M.; Kiefer, J.R.; Oates, J.A.; Marnett, L.J. Molecular basis for cyclooxygenase inhibition by the non-steroidal anti-inflammatory drug naproxen. J. Biol. Chem. 2010, 285, 34950–34959. [Google Scholar] [CrossRef]
- Oyama, T.; Toyota, K.; Waku, T.; Hirakawa, Y.; Nagasawa, N.; Kasuga, J.; Hashimoto, Y.; Miyachi, H.; Morikawa, K. IUCr Adaptability and selectivity of human peroxisome proliferator-activated receptor (PPAR) pan agonists revealed from crystal structures. Acta Cryst. Sect. D Biol. Cryst. 2009, 65, 786–795. [Google Scholar] [CrossRef]
- Andersen, H.S.; Iversen, L.F.; Jeppesen, C.B.; Branner, S.; Norris, K.; Rasmussen, H.B.; Møller, K.B.; Møller, N.P. 2-(oxalylamino)-benzoic acid is a general, competitive inhibitor of protein-tyrosine phosphatases. J. Biol. Chem. 2000, 275, 7101–7108. [Google Scholar] [CrossRef]
- Fradera, X.; Vu, D.; Nimz, O.; Skene, R.; Hosfield, D.; Wynands, R.; Cooke, A.J.; Haunso, A.; King, A.; Bennett, D.J.; et al. X-ray structures of the LXRalpha LBD in its homodimeric form and implications for heterodimer signaling. J. Mol. Biol. 2010, 399, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.; Bledsoe, R.K.; Collins, J.L.; Boggs, S.; Lambert, M.H.; Miller, A.B.; Moore, J.; McKee, D.D.; Moore, L.; Nichols, J.; et al. X-ray crystal structure of the liver X receptor beta ligand binding domain: Regulation by a histidine-tryptophan switch. J. Biol. Chem. 2003, 278, 27138–27143. [Google Scholar] [CrossRef] [PubMed]
- Cheung, J.; Rudolph, M.J.; Burshteyn, F.; Cassidy, M.S.; Gary, E.N.; Love, J.; Franklin, M.C.; Height, J.J. Structures of human acetylcholinesterase in complex with pharmacologically important ligands. J. Med. Chem. 2012, 55, 10282–10286. [Google Scholar] [CrossRef] [PubMed]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The Protein Data Bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Espinosa, J.J.; Rios, M.Y.; Paoli, P.; Flores-Morales, V.; Camici, G.; Rosa-Lugo, V.d.l.; Hidalgo-Figueroa, S.; Navarrete-Vázquez, G.; Estrada-Soto, S. Synthesis of oleanolic acid derivatives: In vitro, in vivo and in silico studies for PTP-1B inhibition. Eur. J. Med. Chem. 2014, 87, 316–327. [Google Scholar] [CrossRef] [PubMed]
- Tarr, G.S.; Reuter, H. Review of the safety of nonsteroidal anti-inflammatory drugs and selective cyclo-oxygenase-2 inhibitors. South. Afr. Fam. Pr. 2015, 57, 18–22. [Google Scholar]
- Hitender, S.; Pushpander, K.; Deshmukh, R.; Anupam, B.; Sunil, K. Pentacyclic triterpenes: New tools to fight metabolic syndrome. Phytomedicine 2018, 50, 166–177. [Google Scholar]
- Yang, F.; Liang-Wei, Z.; Mei-Tang, F.; Ai-Hong, L.; Jia, L.; Tian-Sheng, Z.; Xiao-Ping, L.; Bin, W.; Yue-Wei, G.; Shui-Chun, M. Dictyoptesterols A–C, ∆22-24-oxo cholestane-type sterols with potent PTP1B inhibitory activity from the brown alga Dictyopteris undulata Holmes. Fitoterapia 2018, 130, 241–246. [Google Scholar] [CrossRef]
- Na, B.; Nguyen, P.-H.; Zhao, B.-T.; Vo, Q.-H.; Min, B.S.; Woo, M.H. Protein tyrosine phosphatase 1B (PTP1B) inhibitory activity and glucosidase inhibitory activity of compounds isolated from Agrimonia pilosa. Pharm. Biol. 2016, 54, 474–480. [Google Scholar] [CrossRef]
- Moustafa, E.M.; Thabet, N.M. Beta-sitosterol upregulated paraoxonase-1 via peroxisome proliferator-activated receptor-γ in irradiated rats. Can. J. Physiol. Pharm. 2017, 95, 661–666. [Google Scholar] [CrossRef]
- Puius, Y.A.; Zhao, Y.; Sullivan, M.; Lawrence, D.S.; Almo, S.C.; Zhang, Z.Y. Identification of a second aryl phosphate-binding site in protein-tyrosine phosphatase 1B: A paradigm for inhibitor design. Proc. Natl. Acad. Sci. USA 1997, 94, 13420–13425. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.R.; Ishtiaq; Hizbullah, S.M.; Habtemariam, S.; Zarrelli, A.; Muhammad, A.; Collina, S.; Khan, I. Protein tyrosine phosphatase 1B inhibitors isolated from Artemisia roxburghiana. J. Enzym. Inhib. Med. Chem. 2016, 31, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Rigano, D.; Sirignano, C.; Taglialatela-Scafati, O. The potential of natural products for targeting PPARα. Acta Pharm. Sin. B 2017, 7, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Hiebl, V.; Ladurner, A.; Latkolik, S.; Dirsch, V.M. Natural products as modulators of the nuclear receptors and metabolic sensors LXR, FXR and RXR. Biotechnol. Adv. 2018, 36, 1657–1698. [Google Scholar] [CrossRef] [PubMed]

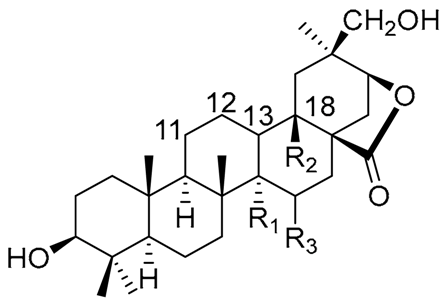
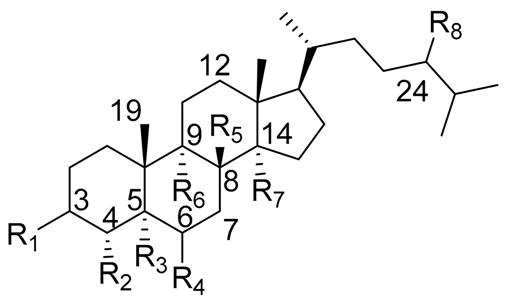 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sterol Name | R1 | R2 | R3 | R4 | R5 | R6 | R7 | R8 | △1 |
| Cholesterol | β-OH | H | - | - | H | H | H | H | 5 |
| Lophenol | β-OH | CH3 | H | H | - | H | H | H | 7 |
| Schottenol | β-OH | H | H | H | - | H | H | β-CH2CH3 | 7 |
| 24-methylenelophenol | β-OH | CH3 | H | H | - | H | H | =CH2 | 7 |
| Lathosterol | β-OH | H | H | H | - | H | H | H | 7 |
| 5α-campest-7-en-3 β-ol | β-OH | H | H | H | - | H | H | β-CH3 | 7 |
| Spinasterol | β-OH | H | H | H | - | H | H | β-CH2CH3 | 7 |
| Locereol | β-OH | CH3 | H | H | - | - | - | H | 8, 14 |
| 5α-cholesta-8,14-dien-3β-ol | β-OH | H | H | H | - | - | - | H | 8, 14 |
| Viperidinone | β-OH | H | H | =O | - | α-OH | OH | H | 7 |
| Viperidone | β-OH | H | H | =O | - | α-OH | H | H | 7 |
| Deoxiviperidone | β-OH | H | H | =O | - | H | H | H | 7 |
| Peniocerol | β-OH | H | H | α-OH | - | - | H | H | 8 |
| Macdougallin | β-OH | H | H | α-OH | - | - | CH3 | H | 8 |
| 5β-deoxiviperidone | β-OH | H | β-H * | =O | - | H | H | H | 7 |
| Cyclostenol ** | β-OH | H | H | α-OH | H | - | CH3 | H | - |
| Stenocereol | β-OH | H | H | α-OH | - | - | CH3 | H | 8,24(25) |
| Thurberol | β-OH | H | H | α-OH | - | - | - | H | 8, 14 |
| Opuntisterol *** | H | H | β-H * | β-OH | H | - | H | β-CH2CH3 | 9 |
| 24-methylenecholesterol | β-OH | H | - | - | H | H | H | =CH2 | 5,24(28) |
| 24-dehydropollinasterol ** | β-OH | H | H | H | H | - | CH3 | H | 24(25) |
| Fucosterol | β-OH | H | - | - | H | H | H | =CH-CH3 | 5,24(28) |
| β-Sitosterol | β-OH | H | - | - | H | H | H | β-CH2CH3 | 5 |
| Genus | Specie | Compound | Isoltated from 1 | Reference |
|---|---|---|---|---|
| Lophocereus | L. schotti (Engelm.) Britton & Rose | Lophenol | AP | [42,43] |
| Schottenol | ||||
| 24-methylenelophenol | ||||
| Lathosterol | ||||
| 5α-campest-7-ene-3β-ol | ||||
| Spinasterol | ||||
| Locereol | ||||
| 5α-cholesta-7,14-dien-3β-ol | ||||
| Leptocereus | L. quadricostatus (Bello) Britton & Rose | Viperidone | AP | [57] |
| Peniocereus | P. viperinus (F.A.C. Weber) Buxb. | Viperidinone | R | [44] |
| Viperidone | ||||
| Deoxiviperidone | ||||
| P. fosterianus (Cutak) Lodé | Peniocerol | R | [47] | |
| P. macdugalli Cutak | Lophenol | R | [45] | |
| Peniocerol | ||||
| Macdougallin | ||||
| 5β-desoxyviperidone | ||||
| P. greggi (Engelm.) Britton & Rose | 5β-desoxyviperidone | R | [49] | |
| Myrtillocactus | M. geometrizans (Mart. ex Pfeiff.) Console | Peniocerol | R | [26,33] |
| Macdougallin | ||||
| Stenocereus | S. thurberi (Engelm.) Buxb. | Cyclostenol | AP | [51] |
| Stenocereol | ||||
| Thurberol | ||||
| S. stellatus (Pfeiff.) Riccob. | β-sitosterol | AP | [32] | |
| Opuntia * | O. dillenii (Ker Gawl.) Haw. | Opuntisterol | AP | [52] |
| O. phaeacantha Engelm. | 24-methylenecholesterol | P | [58] | |
| O. versicolor ** | Pollinastanol | P | ||
| 24-methylenecholesterol | ||||
| Fucosterol | ||||
| Carnegiea | C. gigantea (Engelm.) Britton & Rose | 24-methylenecholesterol 24-dehydropollinastanol | P | [59,60] |
| Pachycereus | P. pringlei (Watson) Britton & Rose | 24-methylenecholesterol | P | [58] |
| Compound | Activity | Description | Reference |
|---|---|---|---|
| Lophenol | AD, CT | AD = Agonist of PPARα and PPARγ, changing the expression of genes involved in fatty acid transport, binding and oxidation in mouse liver. CT = Moderate cytotoxic effect against the L5178Y-R cell line. | [61,62,63] |
| Schottenol | MM | LXR agonists modulating gene expression of LXRα and LXRβ liven nuclear receptors. | [64] |
| Lathosterol | AM and CT | AM = Anti-mutagenic activity against MNNG and NQO. CT = Moderate cytotoxic effect was shown by the compound against MES-SA, MCF-7, and HK-2 cell lines. | [65,66] |
| Spinasterol | AI, AN, CR, CT and MM | AI = Inhibitory activity against COX-1 and COX-2 enzymes and antagonistic effect on the TRPV1 receptor. AN = Inhibitory activity against Helicobacter pyloriCR = Antidepressant-like effect due to the regulation of the TRPV1 receptor. CT = Moderate cytotoxic effect against HeLa, MCF-7, MDA-MB-231, and SKOV-3 cell lines by inducing G0/G1 arrest stimulating the expression of p53 and Bax genes and lower expression of cdk4/6 genes. MM = LXR agonists modulating gene expression of LXRα and LXRβ liven nuclear receptors. | [64,67,68,69,70,71,72,73] |
| Viperidone | MM | Strong inhibition binding to LXRα with an IC50 value of 0.10 μM. | [57] |
| Peniocerol | AI, CT and IN | AI = Potent edema inhibition in TPA induced edema assay. CT = inhibition of breast and colon carcinoma MCF-7 and HCT-15 cell lines proliferation through cell cycle arrest and apoptosis in both cell lines. Also, peniocerol causes Mitochondrial permeability transition (MPT) induction. IN = insect growth regulatory activity against Spodoptera frugiperda and Tenebrio molitor. | [26,33,74,75] |
| Macdougallin | AI, CT and IN | AI = moderate edema inhibition in TPA induced edema assay. CT = inhibition of leukemia K-562 cell line proliferation. IN = insect growth regulatory activity against Spodoptera frugiperda and Tenebrio molitor. | |
| 24-dehydropollinastanol | AD | Agonist of PPAR-α and PPAR-γ, changing the expression of genes involved in fatty acid transport, binding and oxidation in mouse liver. | [61,62] |
| 24-methylenecholesterol | AI, AN, and CT | AI = Low inhibition of key inflammatory enzymes like COX and NF-κB1. AN = Inhibitory effect for Trypanosoma brucei brucei and Mycobacterium marinum. CT = Potent inhibition of aromatase which is a therapeutic target for breast cancer treatment and has a cytotoxic effect on HS27 cell line. | [76,77,78] |
| Pollinastanol | CT | Inhibition of aromatase which is a therapeutic target for breast cancer treatment. | [77] |
| Fucosterol | AD, AO, AN, CR, AI, CT and HT | AD = Inhibition of sorbitol accumulation and diabetic key enzymes like RLAR, HRAR, PTP1B, and α-glucosidase. It also has a downregulation effect of PPAR-γ, C/EBPα, and SREBP1. AO = Regulate transaminase activity (sGOT, sGPT) and enhances the antioxidant activity of SOD and GSH-px. AN = High inhibitory effect against the parasite P. falciparum. CR = Increases serotonin and noradrenaline in the central nervous system, and it also increases central BDNF levels. Also, it showed cholinesterase inhibitory activity and neuroprotective effects.AI = Represses iNOS, TNF-α, and IL-6 binding to NF-κB and inhibits COX-2. CT = Induced HL-60 and HeLa cell line apoptosis through a mitochondrial pathway. HT = Inhibits the synthesis of glucocorticoid receptors involved in the regulation of ACE decreasing its levels. | [79,80,81,82,83,84,85] |
| β-sitosterol | AI, AD, AN, CT, MM, and IM | AI = Inhibits TNF-α, and NF-κB AD = Decreases glycated hemoglobin, serum glucose, and nitric oxide and increases insulin levels slightly. All this is a result of its potent antioxidant activity in the pancreas. AN = Growth inhibitory activity against bacteria P. smartii and the parasite P. falciparum. CT = Strong cytotoxic effect against A549 cell line by inducing apoptosis via ROS-mediated mitochondrial dysregulation. MM = Mediates cholesterol metabolism by increasing sterol excretion and decreasing cholesterol absorption and synthesis. IM = Inhibits T cell proliferation and blocks the secretion of Th2 and cytokines IL-4 and IL-10. | [86,87,88,89,90,91,92,93] |
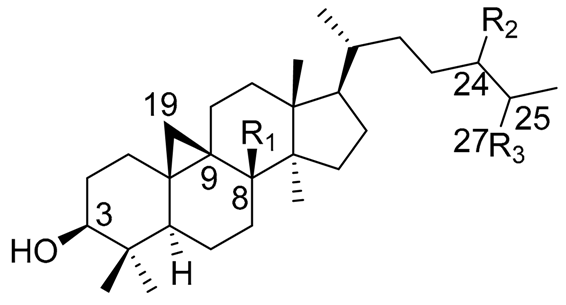 | ||||
|---|---|---|---|---|
| Triterpene Name | R1 | R2 | R3 | △ |
| Cycloartenol | H | H | CH3 | 24(25) |
| 24-methylenecycloartenol | H | =CH2 | CH3 | 24(31) |
| 25(27)-dehydrolanos-8-enol * | - | H | =CH2 | 8(9), 25(27) |
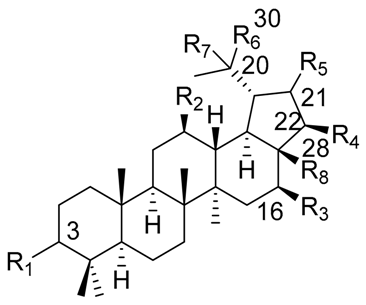 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Triterpene Name | R1 | R2 | R3 | R4 | R5 | R6 | R7 | R8 | △ |
| Lupeol | β-OH | H | H | H | H | =CH2 | - | CH3 | 20(30) |
| Betulin | β-OH | H | H | H | H | =CH2 | - | CH2OH | 20(30) |
| Betulinic acid | β-OH | H | H | H | H | =CH2 | - | COOH | 20(30) |
| Stellatogenin * | β-OH | H | H | H | β-O- | CH3 | OH | CO- | - |
| Lupenone | =O | H | H | H | H | =CH2 | - | CH3 | 20(30) |
| Thurberogenin * | β-OH | H | H | H | β-O- | =CH2 | - | CO- | 20(30) |
| 21-ketobetulinic acid | β-OH | H | H | H | =O | =CH2 | - | COOH | 20(30) |
| 16β-hydroxybetulinic acid | β-OH | H | OH | H | H | =CH2 | - | COOH | 20(30) |
| 22β-hydroxystellatogenin * | β-OH | H | H | OH | β-O- | CH3 | OH | CO- | - |
| 16β-hydroxystellatogenin * | β-OH | H | OH | H | β-O- | CH3 | OH | CO- | - |
| Calenduladiol | β-OH | OH | H | H | H | =CH2 | - | CH3 | 20(30) |
| Betulinic aldehyde | β-OH | H | H | H | H | =CH2 | - | COH | 20(30) |
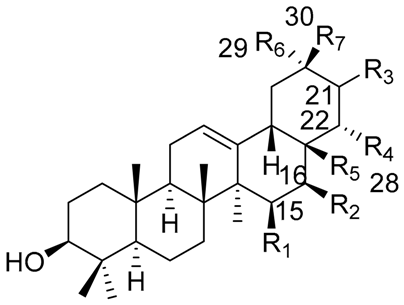 | |||||||
|---|---|---|---|---|---|---|---|
| Triterpene Name | R1 | R2 | R3 | R4 | R5 | R6 | R7 |
| β-amyrin | H | H | H | H | CH3 | CH3 | CH3 |
| Oleanolic acid | H | H | H | H | COOH | CH3 | CH3 |
| Oleanolic aldehyde | H | H | H | H | COH | CH3 | CH3 |
| Erytrodiol | H | H | H | H | CH2OH | CH3 | CH3 |
| Maniladiol | H | OH | H | H | CH3 | CH3 | CH3 |
| Longispinogenin | H | OH | H | H | CH2OH | CH3 | CH3 |
| Dumortierigenin * | -O- | H | H | OH | CO- | CH3 | CH3 |
| Cochalic acid | H | OH | H | H | COOH | CH3 | CH3 |
| Myrtillogenic acid | H | OH | H | H | CH2OH | COOH | CH3 |
| Chichipegenin | H | OH | H | OH | CH2OH | CH3 | CH3 |
| Olean-12-ene-3β,16β,22α-triol | H | OH | H | OH | CH3 | CH3 | CH3 |
| Alamosogenin | H | OH | H | H | COH | CH3 | CH2OH |
| Gummosogenin | H | OH | H | H | COH | CH3 | CH3 |
| Machaerogenin * | H | H | β-O- | H | CO- | CH3 | CH2OH |
| Machaeric acid | H | H | =O | H | COOH | CH3 | CH3 |
| Queretaroic acid | H | H | H | H | COOH | CH3 | CH2OH |
| Treleasegenic acid | H | H | β-OH | H | COOH | CH3 | CH2OH |
| Machaerinic acid | H | H | β-OH | H | COOH | CH3 | CH3 |
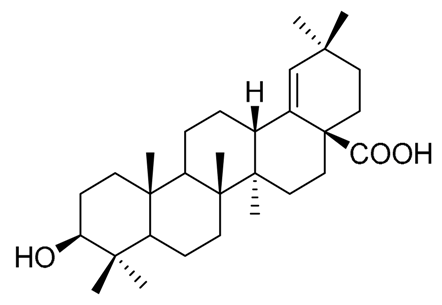
 | 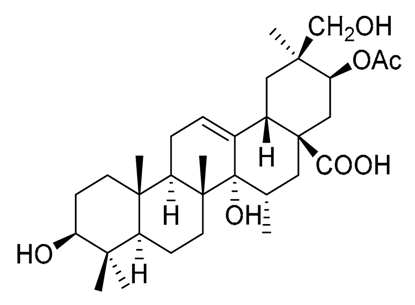 | 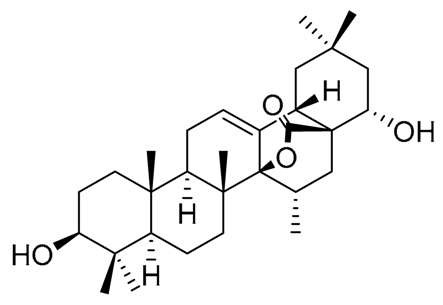 |
| Pachanol A R1 = -, R2 = H, R3 = CH3; △12(13), 14(15) Pachanol B R1 = H, R2 = -, R3 = αCH3; △11(12), 13(18) | Pachanol C | Pachanol D |
 | 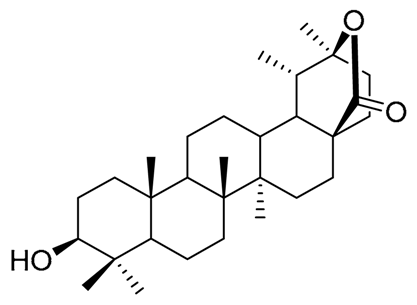 | 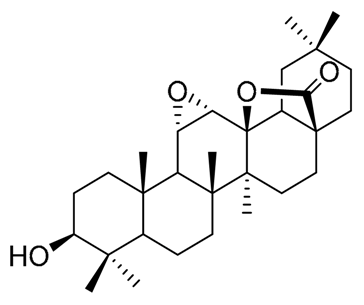 |
| Morolic acid | 27-desoxiphillirigenin | 3β-hydroxy-11α,12α-epoxyolean-28,13β-olide |
| Genus | Specie | Compound | Isolated from 1 | Reference |
|---|---|---|---|---|
| Carnegiea | C. gigantea (Engelm.) Britton & Rose | Cycloartenol, 24-methylenecycloartenol 25(27)-dehydrolanost-8-enol Lupeol | P | [60] |
| Escontria | E. chiotilla (Weber) Rose | Oleanolic acid | AP | [98] |
| Betulin | ||||
| Betulinic acid | ||||
| Maniladiol | ||||
| Erythrodiol | ||||
| Longispinogenin | ||||
| Isolatocereus | I. dumortieri (Scheidweiler) Backerberg | Dumortierigenin | AP | [99,100,101] |
| Pachanol D | ||||
| Lophocereus | L. schotti (Engelmann) Britton & Rose | Lupeol | AP | [42,43] |
| Myrtillocactus | M. cochal (Ocutt) Britton & Rose | Longispinogenin | AP | [102,103,104,105,106] |
| Cochalic acid | ||||
| Myrtillogenic acid | ||||
| Chichipegenin | ||||
| M. geometrizans (Mart. ex Pfeiff.) Console | Longispinogenin | AP | ||
| Cochalic acid | ||||
| Myrtillogenic acid | ||||
| Chichipegenin | ||||
| M. eichlamii Britton & Rose | Oleanolic acid | AP | ||
| Maniladiol | ||||
| Longispinogenin | ||||
| Cochalic acid | ||||
| Myrtillogenic acid | ||||
| Chichipegenin | ||||
| M. schenkii (Purpus) Britton & Rose | Oleanolic acid | AP | ||
| Stellatogenin | ||||
| Pachycereus | P. pringlei (Watson) Britton & Rose | Lupeol | P | [58] |
| 25(27)-dehydrolanost-8-enol | ||||
| P. weberi (Coulter) Britton & Rose | Cochalic acid | AP | [95] | |
| Peniocereus | P. fosterianus (Cutak) Lodé | Chichipegenin | R | [47] |
| P. macdougalli Cutak | β-amyrin | R | [45] | |
| Polaskia | P. chende Gibson & Horak | Oleanolic acid | AP | [107] |
| Erythrodiol | ||||
| Oleanolic aldehyde | ||||
| P. chichipe (Gosselin) Backeberg | Oleanolic acid | AP | [103,108] | |
| Longispinogenin | ||||
| Chichipegenin | ||||
| Olean-12-ene-3β,16β,22α-triol | ||||
| Stenocereus | S. alamosensis (Coulter) Gibson & Horak[Rathbunia alamosensis] | Alamosogenin | AP | [109,110] |
| Gummosogenin | ||||
| S. aragonii (Weber) Buxbaum | β-amyrin | AP | [111] | |
| S. benekei (Ehrenberg) Buxbaum | Lupeol | AP | [112,113,114] | |
| Oleanolic acid | ||||
| β-amyrin | ||||
| Lupeone | ||||
| Queretaroic acid | ||||
| S. eichlamii (Britton & Rose) Buxbaum | Oleanolic acid | AP | [115] | |
| Erythrodiol | ||||
| Longispinogenin | ||||
| S. euruca (Brandegee) Gibson & Horak | Oleanolic acid | AP | [111,116,117,118] | |
| Betulinic acid | ||||
| Stellatogenin | ||||
| Turberogenin | ||||
| Machaerogenin | ||||
| Machaeric acid | ||||
| 21-ketobetulinic acid | ||||
| 16β-hydroxybetulinic acid | ||||
| 22β-hydroxistellatogenin | ||||
| Morolic acid | ||||
| Queretaroic acid | ||||
| 27-desoxyfillirigenin | ||||
| Treleasegenic acid | ||||
| S. fimbriatus (Lamark) Lourteig | Oleanolic acid | AP | [119] | |
| Betulinic acid | ||||
| Erythrodiol | ||||
| Longispinogenin | ||||
| S. griseus (Haworth) Buxbaum | Oleanolic acid | AP | [98] | |
| Betulin | ||||
| Betulinic acid | ||||
| Erythrodiol | ||||
| Longispinogenin | ||||
| S. gummosus (Brandegee) Gibson & Horak | Gummosogenin | AP | [109,111,120] | |
| Machaeric acid | ||||
| Macherinic acid | ||||
| S. pruinosus (Otto) Buxbaum | Oleanolic acid | AP | [32,95,111] | |
| Erithrodiol | ||||
| Longispinogenin | ||||
| 3β-hydroxi-11α,12α-epoxyolean-28,13β-olide | ||||
| S. queretaroensis (Weber) Buxbaum | Oleanolic acid | AP | [114] | |
| Queretaroic acid | ||||
| S. quevedonis (Ortega) Bubaum | Oleanolic acid | AP | [98] | |
| Betulinic acid | ||||
| Longispinogenin | ||||
| S. stellatus (Pfeiffer) Riccobono | Oleanolic acid | AP | [98,116,121] | |
| Betulinic acid | ||||
| Stellatogenin | ||||
| Turberogenin | ||||
| Machaerogenin | ||||
| Queretaroic acid | ||||
| 16β-hydroxistellatogenin | ||||
| S. thurberi (Engelmann) Buxbaum | Lupeol | AP | [50,111,113,114,122,123,124,125] | |
| Oleanolic acid | ||||
| Betulin | ||||
| Maniladiol | ||||
| Erithrodiol | ||||
| Longispinogenin | ||||
| β-amyrin | ||||
| Oleanolic aldehyde | ||||
| Turberogenin | ||||
| Queretaroic acid | ||||
| Calenduladiol | ||||
| Betulinic aldehyde | ||||
| S. treleasei (Britton & Rose) Backeberg | Oleanolic acid | AP | [111,126] | |
| Longispinogenin | ||||
| Treleasegenic acid |
| Compound | Activity | Description | Reference |
|---|---|---|---|
| Lupeol | AD, AO, AN, AI, CT, RN, HP | AD = moderate inhibition of α-Glucosidase and α-Amylase and selective allosteric inhibition of PTP1B. AO = decreases ROS and LPO generation. AN = growth inhibitory activity against several bacteria as M. smegmatis, M. aurum, and E. faecalis, the parasites P. falciparum and L. donovani, and the virus HSV-1 and the reverse transcriptase of HIV-1. AI = inhibition of IL-1β and NF-κB produced an inhibitory effect on the carrageenan-induced edema assay. CT = inhibits cell growth by several mechanisms: inhibition of the phosphorylation of ECFR, Topoisomerase and WNT/β-catenin regulation, induction of cell cycle arrest and mitochondria-mediated apoptosis. IN = moderate larvicidal activity against A. aegypti. RN = strong protective effect of cisplatin-induced nephrotoxicity by upregulating the phosphorylation of MAPKs. HP = alleviate liver injury by GalN/LPS through suppression of the IRAK-mediated TLR4 signal pathway. | [13,87,127,128,129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144] |
| Oleanolic acid | AD, AN, AI, CR, CT, IM, HP | AD = strong regulation of PPARγ and miR-98-5p/PGC-1b axis causing a hypolipidemic effect. AN = growth inhibitory activity against several bacteria as S. aureus, M. smegmatis, E. faecalis, L. monocytogenes, B. cereus, and P. berghei and the parasite Leishmania spp. and inhibitory activity against the reverse transcriptase of HIV-1. AI = inhibition of Lipoxygenase and phospholipase A2 activity. CR = modulation of the BDNF-ERK1/2-CREB pathway through TrkB activation. CT = inhibition of cervical (HeLa), ovary (SK-OV-3), breast (MCF-7), colon (DLD-1) cancer cell lines proliferation. IM = reduce the synthesis of pro-inflammatory mediators, auto-antibody production, suppression of endogenous leptin production, and inhibits migration of leukocytes in the CNS. OT = increase bone mineral density. Its effect is associated with effects on Ca and vitamin D metabolism. HP = showed moderate activity towards in vitro immunological liver injury and low hepatotoxicity. | [13,136,145,146,147,148,149,150,151,152,153,154,155] |
| Betulin | AD, AN, CR, AI, CT | AD = selective allosteric inhibition of PTP1B, noncompetitive inhibitor of α-amylase and inhibition of α-glucosidase. AN = growth inhibitory activity against several bacteria as M. smegmatis and M. aurum, the parasite P. falciparum, and the virus HSV-1 and the reverse transcriptase of HIV-1. CR = protective effect on cognition inhibiting the NF-κB pathway, by regulation of the GABAA receptor, and by moderate cholinesterase inhibition. It also reduced 6-hydroxydopamine-induced dopaminergic neuron degeneration. AI = decreases NO production, iNOS expression in vitro, and NF-κB activity. It also decreases the levels of MPO, IL-1β, and TNF-α in liver tissue. CT = strong inhibitory effect on the proliferation of several cell lines triggering apoptosis by mitochondrial pathway and NOXA induction. | [87,127,133,137,138,139,143,156,157,158,159,160,161,162,163,164,165,166] |
| Betulinic acid | AD, AN, IN, AI, CT, RN, HP | AD = selective allosteric inhibition of PTP1B, noncompetitive inhibitor of α-amylase and inhibition of α-glucosidase. It also stimulates insulin secretion by the mediation of potassium and chloride channels. AN = growth inhibitory activity against several bacteria as M. smegmatis, M. aurum, S. aureus, B subtilis, E. faecalis, and B. cereus, the parasites P. falciparum and T. cruzi, and the virus HSV-1 and the reverse transcriptase inhibitor of HIV-1. IN = inhibitor of tyrosinase and could be used as an insecticidal agent. CR = neuroprotective effect on cognition by moderate cholinesterase inhibition. It also improves cAMP, cGMP and BDNF levels. AI = decreases NO production and iNOS and IL-6 expression in vitro. CT = inhibition of cervical (HeLa), ovary (SK-OV-3), breast (MCF-7), colon (DLD-1) cancer cell lines proliferation. It has also shown to induce apoptosis by DNA damage, G2/M cell cycle arrest, and Bcl-2/Bax signaling regulation. RN = strong protective effect of cisplatin-induced nephrotoxicity by upregulating the phosphorylation of MAPKs. HP = inhibition of liver oxidative stress in the iron/ascorbate system and showed hepatoprotective effects against D-GalN/TNF-α induced cell dead. | [13,19,87,127,133,135,136,137,138,143,147,160,161,163,167,168,169,170,171] |
| Maniladiol | AN, AI | AN = growth inhibitory activity against M. tuberculosis and the reverse transcriptase inhibition of HIV-1. AI = decreases the production of pro-inflammatory cytokines such as TNF-α, IL-1, and IL-6 and inhibits NO production. | [164,172,173] |
| Erythrodiol | AN, CT, HT, IM, HP | AN = growth inhibitory activity against several bacteria as B. subtilis, E. coli, and C. albicans and the reverse transcriptase inhibitor of HIV-1. Inhibitor of tyrosinase and could be used as an insecticidal agent AI = edema inhibition in the TPA-induced inflammation assay. CT = potent anti-proliferative effect inducing apoptosis, cell cycle arrest, and ROS generation. HT = reduce cardiac remodeling by inhibiting angiotensin II-induced proliferation via PPAR-γ. IM = reduce the synthesis of pro-inflammatory mediators, auto-antibody production, suppression of endogenous leptin production, and inhibits migration of leukocytes in the CNS. HP = high protection of human hepatoma cells against CCl4-induced injury with ALT level decreased. | [13,153,164,167,174,175,176,177,178,179] |
| Longispinogenin | AN, AI, CT | AN = growth inhibitory activity against M. tuberculosis. AI = edema inhibition in the TPA-induced inflammation assay. CT = inhibition of cervical (HeLa) cancer cell line proliferation. | [173,180,181] |
| Pachanol D | AC | AC = strong inhibitory effect on the acetic acid-induced writhing test. | [95] |
| Cochalic acid | CT | CT = potent inhibition of cervical (HeLa) cancer cell line proliferation. | [180] |
| Chichipegenin | AI, CT | AI = edema inhibition in the TPA-induced inflammation assay. CT = inhibition of breast and colon carcinoma MCF-7 and HCT-15 cell lines proliferation. | [26] |
| Stellatogenin | AC | AC = strong inhibitory effect on the acetic acid-induced writhing test. | [95] |
| β-amyrin | AD, AN, AI, CR, CT, HP | AD = moderate inhibition of α-Glucosidase and α-Amylase. AN = growth inhibitory activity against several bacteria as B. subtilis, S. aureus, and C. albicans, the parasite T. cruzi, and antiviral inhibitory activity against the reverse transcriptase of HIV-1 and IAV. AI = decreases the production of pro-inflammatory cytokines such as TNF-α, IL-1, and IL-6 and inhibits NO production CR = interaction with the GABAA receptor and produce sedative and hypnotic, increasing noradrenergic activity. It also showed neuroprotective activity reducing α-synuclein aggregation upregulating LGG-1 expression. CT = inhibition of proliferation in the cervical (HeLa), ovary (SK-OV-3), breast (MCF-7), colon (DLD-1), and other cancer cell lines. HP = hepatoprotective effect against acetaminophen-induced hepatotoxicity. | [143,147,164,171,172,182,183,184,185,186] |
| Oleanolic aldehyde | AD, AN | AD = dose-dependent enhancement of insulin secretion by INS-1 cells. AN = growth inhibitory activity against S. mutans and P. gingivalis. | [187,188] |
| Gummosogenin | AC, CT | AC = strong inhibitory effect on the acetic acid-induced writhing test. CT = inhibition of cervical (HeLa) cancer cell line proliferation. | [95,180] |
| Lupeone | AN, AD, AI, RN | AN = reverse transcriptase inhibitor of HIV-1. AD = moderate inhibition of α-Glucosidase and selective allosteric inhibition of PTP1B. AI = decreases the production of pro-inflammatory cytokines such as IL-12 and IL-6. RN = strong protective effect of cisplatin-induced nephrotoxicity. | [128,133,135,164,183,189] |
| Turberogenin | AC, CT | AC = strong inhibitory effect on the acetic acid-induced writhing test. CT = low inhibition of cervical (HeLa) cancer cell line proliferation. | [95,180] |
| Morolic acid | AD, AI | AD = induced a significant reduction of blood glucose levels by inhibition of 11β-HSD1. AI = inhibition of the leukocyte dermal infiltration and inhibition of key inflammatory enzymes as PLA2 and 5-LOX. | [190,191] |
| Queretaroic acid | CT | CT = inhibition of cervical (HeLa) cancer cell line proliferation. | [180] |
| Calenduladiol | CR, CT | CR = inhibition of acetylcholinesterase and butyrylcholinesterase in vitro. CT = inhibition of leukemia (NB4 and K562) cancer cell lines proliferation. | [192,193] |
| Cycloartenol | AD, AN, and CT | AD = Decrease glucose intestinal absorption that could be associated with SGLT1 regulation as well as α-glucosidase inhibition. Downregulation of fatty acid synthesis and interferes with the absorption of cholesterol. AN = Inhibitory effect against bacteria E. coli and P. aeruginosa and low inhibition of the parasite P. falciparum. CT = Weak cytotoxic activity against HL60 cell line and p38MAPK-mediated apoptosis in the U87 cell line. | [91,194,195,196,197,198,199,200] |
| Betulinic aldehyde | AN, CT | AN = growth inhibitory activity against bacteria P. smartii, E. faecalis, S. aureus, and E. coli and antiviral activity against Influenza virus (KBNP-0028) and the reverse transcriptase of HIV-1. CT = inhibition of cervical (HeLa), ovary (SK-OV-3), breast (MCF-7), colon (HCT-116) and melanoma (SK-MEL-5) cancer cell lines proliferation. | [88,135,164,201] |
| Ligand | COX-1 | COX-2 |
|---|---|---|
| Thurberol | −132.1 | −144.1 |
| Locereol | −133.1 | −141.1 |
| Fucosterol | −130.6 | −141.7 |
| 5α-cholesta-8,14-dien-3 β-ol | −130.4 | −141.5 |
| Spinasterol | −130.6 | −138.5 |
| 24-methylenecolesterol | −127.3 | −139.9 |
| β-sitosterol | −124.3 | −136.9 |
| Peniocerol | −124.3 | −134.1 |
| 24-Methylenelophenol | −127.3 | −130.7 |
| Lophenol | −123.7 | −131.2 |
| Ligand | COX-1 | COX-2 |
|---|---|---|
| Lupeone | −104.0 | −97.1 |
| Thurberogenin | −92.3 | −93.6 |
| Lupeol | −97.7 | −87.3 |
| Betulinic aldehyde | −94.9 | −88.6 |
| 16β-hydroxybetulinic acid | −82.9 | −95.1 |
| Calenduladiol | −92.8 | −85.1 |
| 16β-hydroxystellatogenin | −93.8 | −83.0 |
| 22β-hydroxystellatogenin | −94.0 | −79.3 |
| 21-ketobetulinic acid | −81.6 | −87.1 |
| Machaerogenin | −87.0 | −79.4 |
| LIGAND | PTP1B | PPAR-α | PPAR-γ |
|---|---|---|---|
| Fucosterol | −141.7 | −135.7 | −138.9 |
| β-sitosterol | −131.6 | −141.0 | −142.4 |
| Schottenol | −132.9 | −139.0 | −138.9 |
| Spinasterol | −133.1 | −140.8 | −131.2 |
| Thurberol | −135.4 | −129.3 | −140.4 |
| Cyclostenol | −140.4 | −137.3 | −126.4 |
| 24-Methylenecholesterol | −132.7 | −133.5 | −136.0 |
| Peniocerol | −132.4 | −127.9 | −140.5 |
| Opuntisterol | −126.0 | −141.5 | −132.7 |
| Steneocerol | −133.0 | −136.2 | −126.2 |
| LIGAND | PTP1B | PPAR-α | PPAR-γ |
|---|---|---|---|
| 16β-hydroxystellatogenin | −98.5 | −128.7 | −119.5 |
| Thurberogenin | −104.8 | −123.4 | −114.8 |
| Stellatogenin | −99.4 | −124.8 | −116.9 |
| Myrtillogenic acid | −95.4 | −123.8 | −119.3 |
| Alamosogenin | −98.1 | −120.5 | −118.2 |
| 22β-hydroxystellatogenin | −93.4 | −130.0 | −112.6 |
| Oleanolic acid | −101.6 | −118.9 | −114.2 |
| Machaeric acid | −105.2 | −112.3 | −115.6 |
| Oleanolic aldehyde | −100.4 | −116.8 | −115.6 |
| Machaerinic acid | −103.8 | −114.0 | −114.8 |
| LIGAND | LXR-α | LXR-β | AChE |
|---|---|---|---|
| Fucosterol | −167.1 | −167.7 | −152.9 |
| β-sitosterol | −165.2 | −164.3 | −151.7 |
| Methylenecolesterol | −163.1 | −157.8 | −148.6 |
| Thurberol | −161.9 | −158.7 | −147.5 |
| Spinasterol | −157.0 | −158.2 | −152.6 |
| Opuntisterol | −155.9 | −157.9 | −152.1 |
| Cyclostenol | −155.4 | −158.2 | −147.7 |
| Peniocerol | −158.6 | −155.8 | −143.4 |
| Schottenol | −155.9 | −153.6 | −147.4 |
| 24-Methylenelophenol | −155.7 | −154.6 | −146.3 |
| LIGAND | LXR-α | LXR-β | AChE |
|---|---|---|---|
| Thurberogenin | −153.915 | −155.492 | −143.2 |
| 16β-hydroxystellatogenin | −149.154 | −152.308 | −134.8 |
| Betulinic acid | −141.254 | −155.181 | −137.3 |
| Stellatogenin | −142.879 | −150.498 | −139.4 |
| 16β-hydroxybetulinic acid | −145.372 | −156.064 | −127.6 |
| Calenduladiol | −147.818 | −152.858 | −127.8 |
| Betulin | −141.428 | −153.007 | −133.0 |
| Lupenone | −145.652 | −146.987 | −134.2 |
| Lupeol | −146.667 | −148.674 | −129.8 |
| Alamosogenin | −141.635 | −144.049 | −138.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salazar, J.R.; Loza-Mejía, M.A.; Soto-Cabrera, D. Chemistry, Biological Activities and In Silico Bioprospection of Sterols and Triterpenes from Mexican Columnar Cactaceae. Molecules 2020, 25, 1649. https://doi.org/10.3390/molecules25071649
Salazar JR, Loza-Mejía MA, Soto-Cabrera D. Chemistry, Biological Activities and In Silico Bioprospection of Sterols and Triterpenes from Mexican Columnar Cactaceae. Molecules. 2020; 25(7):1649. https://doi.org/10.3390/molecules25071649
Chicago/Turabian StyleSalazar, Juan Rodrigo, Marco A. Loza-Mejía, and Diego Soto-Cabrera. 2020. "Chemistry, Biological Activities and In Silico Bioprospection of Sterols and Triterpenes from Mexican Columnar Cactaceae" Molecules 25, no. 7: 1649. https://doi.org/10.3390/molecules25071649
APA StyleSalazar, J. R., Loza-Mejía, M. A., & Soto-Cabrera, D. (2020). Chemistry, Biological Activities and In Silico Bioprospection of Sterols and Triterpenes from Mexican Columnar Cactaceae. Molecules, 25(7), 1649. https://doi.org/10.3390/molecules25071649