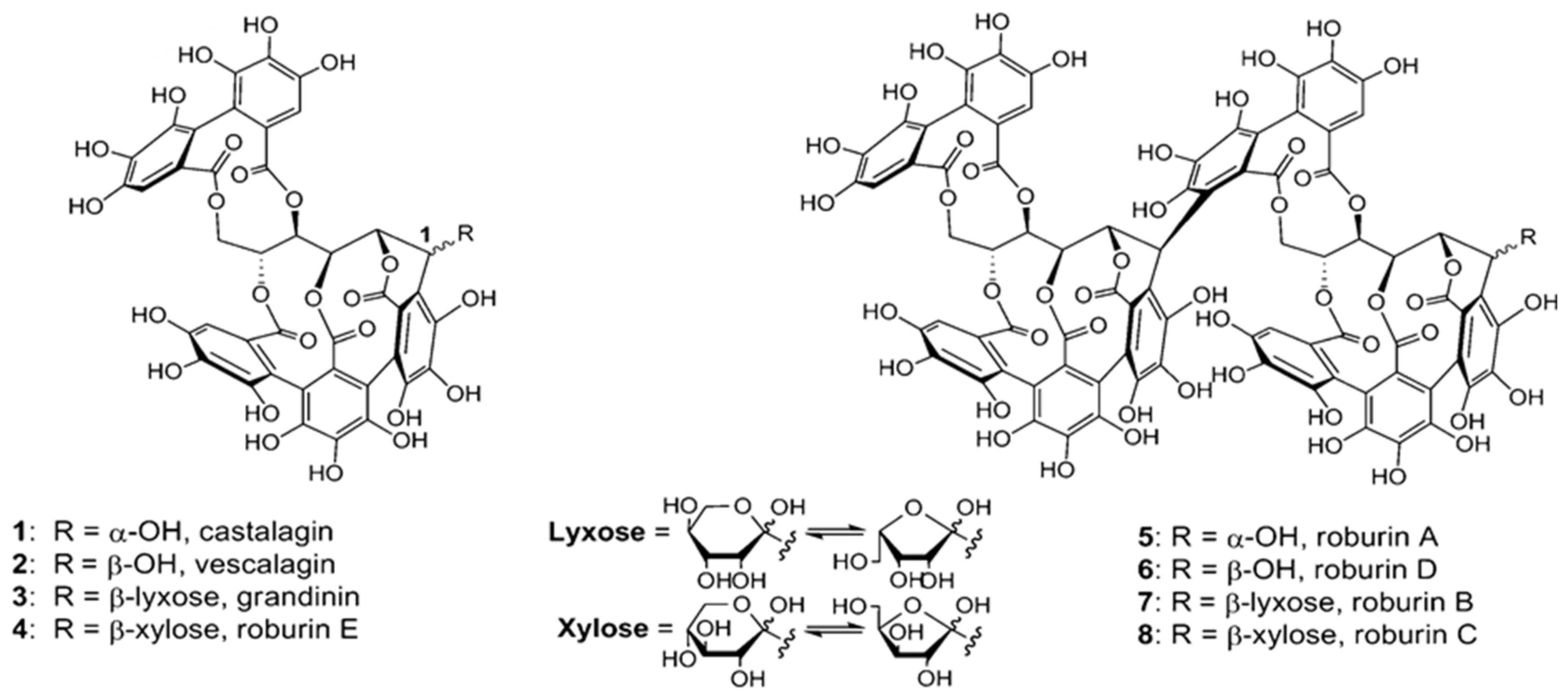
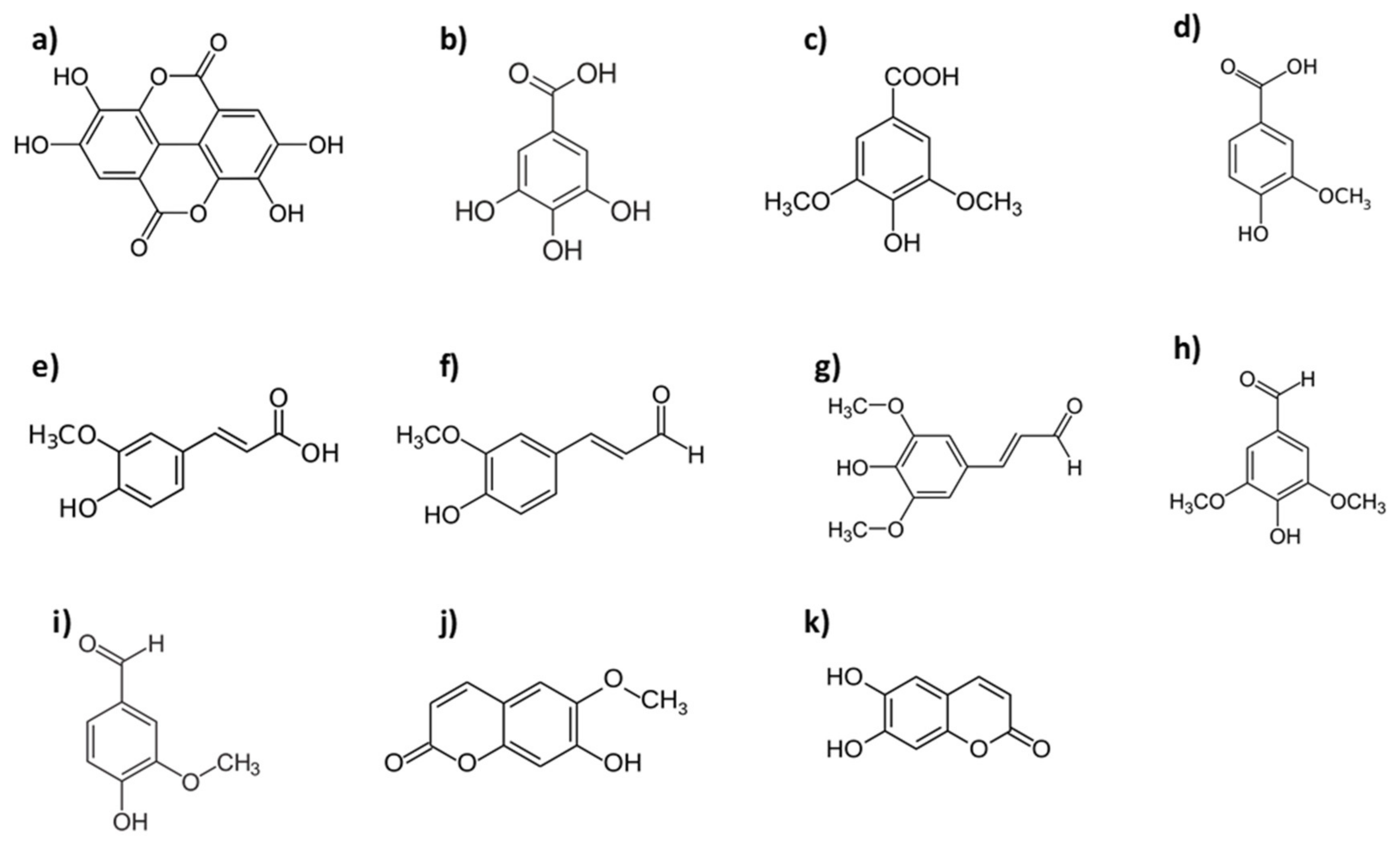
2.2.1. Alternative Wood Species from Quercus Genus
In green wood, LMWP have been studied in
Q. pyrenaica, Q. faginea and
Q. humboldtii. Seven different habitats of
Q. pyrenaica have been studied from two Spanish regions: six from Castile and León (Gata/Peña de Francia, Guadarrama, Cantabrian mountains, Iberian mountain range, Alitse-Maragatería and Gredos/Ávila mountains) [
11,
14]; and one from the Basque country (Álava) [
20,
33]. In all of these species, the main component is ellagic followed by gallic acid (
Table 5), as usually happens in traditional oaks. In general, the acid concentration in the three species is higher than that of aldehydes, following the same pattern as in traditional species. However, vanillin and syringaldehyde concentrations are higher than their corresponding acids except for vanillin in
Q. pyrenaica from the Iberian mountain range [
11] and
Q. faginea from Álava [
20,
33]. Total aldehyde concentration in the alternative woods is as in traditional ones, the two species from Álava being the ones with the lowest concentrations, especially in aldehydes with the most influence on the sensory characteristics of the wines: syringaldehyde and vanillin. However, the total acid concentration in
Q. pyrenaica is higher than in traditional woods, except for those from Gredos and Álava (
Table 5). Green wood of
Q. pyrenaica from Castile and León has a higher ellagic acid concentration than traditional woods (
Table 5). However, the same species from Álava presents concentrations similar to the woods normally used in enology. Moreover, this wood from Álava has the lowest concentrations of syringic acid, syringaldehyde, sinapaldehyde and vanillin in comparison with the woods from Castile and León. Therefore, the
Q. pyrenaica species wood from Castile and León is richer in LMWP, concentrations of 1061 to 697 µg/g, than that of Álava with 262 µg/g. Moreover, the LMWP concentration of the
Q. pyrenaica wood from Castile and León is also higher than that of the traditional ones, except for that from Gredos (Ávila). The
Q. faginea green wood from Álava presents LMWP quantities similar to
Q. pyrenaica from Álava (
Table 5) and lower quantities of syringic, vanillic and ferulic acids and of sinapaldehyde and vanillin, but higher ones of gallic acid than in traditional oaks. The green wood of
Q. humboldtii shows concentrations of aldehydes and acids similar to those of
Q. petraea,
Q. robur and
Q. alba.
Fernández de Simón et al. [
20,
33] described another two types of low molecular weight compounds, called A and B, in some of the alternative species (
Q. pyrenaica and
Q. faginea) and two of the traditional ones (
Q. petraea and
Q. robur). The A compounds, whose UV spectra are similar to those of ellagic acid, are present in all the species and in similar quantities. However, some of the B compounds, whose UV spectra are similar to those of gallic acid, are found in traditional woods but not in all the alternative species: B2, B3 and B4 are not present in
Q. pyrenaica nor are B1 and B2 found in
Q. faginea. The B compounds found in all the species are generally more abundant in the alternatives (
Q. pyrenaica and
Q. faginea) than in the traditional oaks (
Q. petraea and
Q. robur). These authors suggest that the forests of these two new species could be distinguished via the ratio of these two types of compound. In addition to green wood Fernández de Simón et al. [
33] studied wood dried for 1 year in the open, observing that the behavior of compounds A and B after drying is not clear as they increase in some cases, even detecting compounds not present in the green wood after drying (like B14, 15, 16, 17), and in other cases decrease to the point of non-detection. Compounds B2 and B4 are not detected in dry
Q. pyrenaica but all the other A and B compounds increase their concentration after this treatment except B9 which decreases to non-detectable concentrations [
33]. In addition, the seasoned
Q. pyrenaica wood presents higher quantities of all the compounds in comparison with
Q. robur,
Q. petreae and
Q. faginea, except for A3 in
Q. petraea and Q. robur and B16 in
Q. faginea. The dry wood of this species presents compound B1, which is not detected in the dry wood of any other species. B6, B7, B13, B14 and B17 are only detected in the dry wood of the new species (
Q. pyrenaica and
Q. faginea) but not in the traditional ones [
33]. Drying decreases 13 of the 27 A and B compounds analyzed in
Q. robur, with 10 of them not being detected. In
Q. petraea this treatment decreases 11 of them with 6 not being detected; and the concentration of 7 decreases in
Q. faginea with 4 being undetectable [
33].
Table 6 shows that the acid concentration in seasoned
Q. pyrenaica,
Q. faginea and
Q. humboldtii woods is still higher than the aldehydes with ellagic and gallic acids being the principal ones. Acid and aldehyde concentration in
Q. pyrenaica and
Q. faginea from Álava is higher with a longer seasoning time (
Table 6). As the data are not taken from the same study it cannot be predicted with any certainty that longer drying means a higher concentration, but everything seems to indicate that it is probably a decisive factor. Ellagic acid concentration in
Q. pyrenaica is 137 µg/g when oven dried; 1254.8 to 2679 µg/g when dried in the open without specifying time (although it is probably a long period given the rather high concentrations); and 299, 297 and 735 µg/g after 12, 24 and 36 months´ drying in open air, respectively; time again seems to be a very significant factor in LMWP concentration. On comparing
Q. pyrenaica and
Q. faginea from Álava after 12 and 36 months drying,
Q. pyrenaica wood presents higher concentrations of all the LMWP except ellagic and ferulic acids, sinapaldehyde after 12 months and ellagic, gallic and ferulic acids after 36 months. As regards
Q. humboldtii wood, Martínez-Gil et al. [
60] observe that LMWP concentration is similar to that found in
Q. petraea and
Q. alba, though with higher concentrations of gallic acid and sinapaldehyde, and lower ones of syringic acid and coniferaldehyde.
Fernández de Simón et al. [
33] studied the effect of drying on the composition of
Q. pyrenaica and
Q. faginea woods showing that LMWP concentrations increased considerably (
Table 4 and
Table 5). This study observed that gallic and ellagic acid content more than double their concentration due to drying in the open for one year, and even multiply gallic acid concentration by 7 in
Q. pyrenaica (
Table 4 and
Table 5). The only LMWP which decreases on drying is coniferaldehyde in
Q. faginea wood. However, the concentration of five LMWP: gallic and ferulic acids, sinapaldehyde, scopoletin and aesculetin, decreases in
Q. petraea wood during the same type of drying [
33].
Vivas et al. [
24] studied the composition of different
Quercus (
Q. frainetto, Q. stellata and
Q. oocarpa) woods after drying in open air for 24 months and stated that the ellagic acid concentration is greater than that of gallic acid in
Q. stellata and
Q. oocarpa, as usually occurs in traditional oaks, but is the opposite in
Q. frainetto [
24]. These authors indicated higher ellagic acid concentrations (11.2 mg/g) in
Q. stellata than in the other two species (4.2 and 0.6 mg/g in
Q. oocarpa and
Q. frainetto, respectively), but lower than
Q. alba (18.4 mg/g) and much higher than traditional European oaks (1.9 and 3.1 mg/g in
Q. robur and
Q. petraea, respectively).
Q. frainetto wood shows the highest concentration (3.2 mg/g) of gallic acid, more than double that of traditional oaks (1.4, 1.1 and 1.2 mg/g in Q. robur, Q. petraea and Q. alba, respectively) and also higher in Q. stellata, but with a smaller difference (1.8 mg/g). However, concentration in Q. oocarpa is similar to that of traditional woods with 1.3 mg/g.
LMWP concentration of the woods after toasting has only been studied in detail in
Q. pyrenaica,
Q. faginea and
Q. humboldtii (
Table 7). It has been stated that acid concentration remains higher than that of aldehydes in
Q. pyrenaica and
Q. faginea with the main ones being ellagic and gallic acids. Nevertheless, the opposite occurs in toasted
Q. humboldtii wood, as the aldehyde concentration is greater, the main one being sinapaldehyde (
Table 7). This behavior has also been observed in traditional woods, especially
Q. alba and/or
Q.
robur [
3,
13,
30,
32] (
Table 7), although the most common profile is that observed in
Q. pyrenaica and
Q. faginea. The total LMWP concentration interval in
Q. pyrenaica wood is from 607 to 20500 µg/g, while total LMWP concentrations of 2132 µg/g and 2464 µg/g, respectively, are found in
Q. faginea and
Q. humboldtii (
Table 7). As regards how toasting affects the LMWP of these alternative species, the studies of interest are those which show results before and after this treatment [
3,
13,
25,
27], one even showing results after two toasting intensities (100 °C or 150 °C for 45 min) [
27]. Gallic acid concentration decreases in the woods of all the species (
Table 5 and
Table 6): between 6 and 25% in
Q. pyrenaica, 25% in
Q. faginea and 68% in
Q. humboldtii. Moreover, gallic acid degradation in
Q. pyrenaica wood is greater with a higher toasting temperature (
Table 7). However, ellagic acid increases in all these except
Q. humboldtii, which maintains practically the same concentration (
Table 5 and
Table 6). Vanillic and syringic acid concentrations in woods also increase in the three species during toasting, except in the Castro-Vázquez [
25] study, where these two acids decrease in
Q. pyrenaica wood. Ferulic acid has only been studied in
Q. pyrenaica [
25,
27], and a significant decrease (15 to 44% of its initial concentration) is observed in both articles, this being greater as the temperature rises [
27]. Aesculetin concentration diminishes in
Q. pyrenaica and increases in
Q. faginea (
Table 5 and
Table 6). However, the compounds really affected by toasting are the aldehydes, as very significant increases are observed:
Q. humboldtii goes from 18 to 1778 µg/g,
Q. faginea from 26 to 670 µg/g and
Q. pyrenaica from 73 to 544 µg/g according to Canas et al. [
27], from 46 to 909 µg/g according to Cadahía et al. [
3] and from 43 to 91 µg/g according to Castro-Vázquez et al. [
25]. The same occurs with the traditional species since acid concentration increases slightly during toasting, but the aldehydes present a really considerable increase [
3,
13].
2.2.2. Alternative Wood Species from Different Genus to Quercus
Castanea sativa Mill.:
Seasoned chestnut wood is the richest in LMWP of the non-
Quercus species, both in acids and in aldehydes (
Table 6). The acid concentration is higher than that of aldehydes, as observed in
Quercus woods. Ellagic and gallic acids are the most abundant compounds within this group, the gallic acid concentration generally being higher than that ellagic acid, in contrast to what occurs in
Quercus species. The lowest gallic acid concentrations in dry wood have been found by Alañon et al. [
21] in woods from Lugo (Spain), dried in an oven, and by Castro-Vazquez et al. [
25] in woods from Gerês forest (Portugal) dried in open air for 24 months, with concentrations of 1155 and 1172 µg/g, respectively. Alañon et al. [
21] found significant protocatechuic acid, 4-hydroxybenzoic acid and coumaric acid concentrations in dry wood, and even quantities higher than that of ellagic acid. However, other authors who also analyzed protocatechuic acid in this wood record much lower concentrations than the 113 µg/g found by Alañon et al. [
21] and the 5 µg/g quantified by Sanz et al. [
15]. Canas et al. [
28,
29] did not observe any furanic derivatives in seasoned chestnut wood or in the traditional woods. Nevertheless, Soares et al. [
31] found 5-methylfurfural in seasoned chestnut wood and 5-methylfurfural and furfural in toasted wood, but at lower concentrations than oak. These authors [
31] did not find 5-hydroxymethylfurfural but the toasted chestnut woods studied by Canas et al. [
30] present 5-methylfurfural, furfural and 5-hydroxymethylfurfural, even at concentrations higher than oak. The highest total LMWP concentrations in dry wood are 14730 µg/g (18 months natural seasoning) [
26] and in toasted wood 35282 µg/g (185 °C for 60 min) [
32].
Toasting considerably decreases the concentration of gallic acid in chestnut wood. Sanz et al. [
15] observed an increase of this compound in wood after toasting at 160-170 °C for 35 min, though the wood after toasting at 185 °C for 45 min presents a diminished concentration, also observed by the rest of the authors. However, the ellagic acid concentration in the wood increases with toasting [
15,
25,
31,
32], and is greater after more toasting [
15,
31]. In addition, syringic and vanillic acids increase during toasting, although Castro-Vázquez et al. [
25] observed the opposite. The aldehydes are the compounds with the greatest increase during toasting, as also occurred in the
Quercus species. It is also observed that the most important aldehyde from an organoleptic viewpoint, vanillin, due to the vanilla notes it contributes, increases considerably on toasting chestnut wood. In the studies where chestnut is compared to
Quercus woods, it has been shown that vanillin concentration in chestnut is higher than in
Quercus oak (
Table 7). Similarly, it has been found in the literature that when the same liquid (wine, vinegar or brandy) is aged in chestnut and oak barrels the levels of vanillin detected in those from chestnut are higher than in those aged in traditional oak [
61,
62,
63].
Robinia pseudoacacia L.:
The total LMWP percentage in comparison with the rest of the components found in acacia wood is low since the main compounds of this wood are flavonoids followed by condensed tannins [
17] (described in
Section 2.3). The two main compounds in dry acacia wood are hydroxycinnamic derivative compounds [
17], with concentrations of 2506 and 437 µg/g. Magel et al. [
64] also observed a hydroxycinnamic derivative as the main LMWP. These hydroxycinnamic derivatives decrease after light toasting (165° for 20 min), although they continue to be the main LMWP quantitatively. However, after medium toasting (165 ° for 30 min) there is only one of them and after medium plus toasting (185° for 45 min) both are completely degraded [
17]. The following most important component quantitatively in dry acacia wood is gallic aldehyde with 108 µg/g, which increases on light and medium toasting to 137 and 245 µg/g, respectively, but is degraded on more intense toasting: 21 µg/g in medium plus toasting [
17]. Significant concentrations of β-resorcilyc aldehyde are also found in dry wood (48 µg/g), increasing as a result of the toasting process [
17]. So, after medium toasting (165 °C for 35 min), significant concentrations of coniferaldehyde and sinapaldehyde (276 and 239 µg/g, respectively) are maintained [
17]. They increase with stronger toasting (165 °C for 35 min), especially the sinapaldehyde, going from 300 to 1666 µg/g (
Table 7). This was also described by Soares et al. [
31] with more intense toasting (240 °C for 120 min) (
Table 7). However, Jordao et al. [
7] did not detect coniferaldehyde in this wood after medium toasting. Another major compound found after a medium plus toasting treatment (185° for 45 min) is syringaldehyde, with concentrations of 326 µg/g [
17], though this compound is not detected in dry or toasted wood in the other studies (
Table 7). Vanillic acid has not been detected in any of the works in which dry or toasted acacia wood composition is analyzed by HPLC (
Table 5 and
Table 6) [
6,
7,
17,
31]; however, it is a characteristic acid in traditional oak. Protocatechuic aldehyde is found in both dry and toasted acacia wood [
7,
17,
31]. Yet there is no clear behavior of this compound with the degree of toasting applied to the wood, since Soares et al. [
31] observed a slight decrease with an increased toasting level while Sanz et al. [
17] observed an increase with medium toasting and a decrease with light and medium plus toasting. As regards the furfurals, Soares et al. [
31] reported very low concentrations of furfural and of 5-hydroxymethylfurfural in acacia wood in comparison with oak. Moreover no 5-methylfurfural is found in dry or toasted wood. Nor is
p-coumaric acid detected in toasted acacia wood [
7].
Prunus avium L. and Prunus cereaus L.:
The LMWP in cherry wood have been studied in two subspecies,
P. avium and
P. cereaus. The ellagic acid in the dry wood of
P. avium is detected at very low concentrations (15.80 µg/g) in comparison with traditional oak [
21] or even not detected [
8]. The dry wood of
P. cereaus presents a higher concentration of ellagic acid (194 µg/g) than those of
P. avium, but lower than those found in naturally dried traditional wood (
Table 6). This also decreases with toasting, as Soares et al. [
31] observed that concentration in
P. cereaus goes from 194 µg/g to 89 µg/g after toasting at 160 °C for 20 min, degrading completely when thermal treatment increases (200 °C for 120 min or 240 °C for 120 min) (
Table 5 and
Table 6). Ellagic acid is only detected in toasted
P. avium wood [
7] and, moreover, at higher concentrations than those found in dry wood (
Table 5 and
Table 6), so this subspecies of the central zone of France is probably richer in this compound. Gallic acid is only detected in the dry wood of the subspecies
P. avium and at insignificant concentrations in comparison with oak (31.11 and 1.22 µg/g
Table 6). The major LMWP in dry
P. avium wood, according to Sanz et al. [
8] are methyl syringate and 3,4,5-trimethoxyphenol, followed by
p-coumaric acid and protocatechuic acid. According to Alañon et al. [
21] they are: coniferaldehyde with 332.59 µg/g and sinapic acid with 106.8 µg/g, followed by sinapaldehyde with 78.72 µg/g and syringaldehyde with 42.01 µg/g as opposed to 36.06 µg/g of protocatechuic acid or 7.11 µg/g of coumaric acid.
On the other hand, the main compound in toasted cherry wood is sinapaldehyde, with concentrations of 553 up to 1637 µg/g in
P. avium and from 156 to 619 µg/g in
P. cereaus (
Table 7). Syringaldehyde and coniferaldehyde are the following compounds in abundance in toasted
P. avium wood [
8,
32]; however, these were not detected by Jordao et al. [
7] who describes
p-coumaric acid as the main LMWP. The next major component in toasted
P. cereaus wood is protocatechuic aldehyde, and in those woods subjected to high level toasting it is also syringaldehyde [
31]. Cherry wood also presents appreciable quantities of benzoic acid [
8], which may explain the high ethyl benzoate concentration found in the vinegars obtained by acidification in cherry wood barrels in comparison with those found when using other woods [
61]. The quantities of vanillin found in toasted
P. avium wood vary from 41.9 to 313.33 µg/g and in
P. cereaus from 56.67 to 117.71 µg/g, in general being higher than those found in acacia, and somewhat lower than those recorded in ash, chestnut and oak (
Table 7).
Fraxinus americana L. and Fraxinus excelsior L.:
Gallic acid and ellagic acid are not found in ash, the major compound in the dry wood being tyrosol, with 139 and 100 µg/g in
F. americana and
F. excelsior, respectively. However, this compound has not been previously detected in oak [
18]. Tyrosol decreases during toasting, while coniferaldehyde and sinapaldehyde increase and are the major compounds in toasted
F. Americana wood followed by vanillin and syringaldehyde [
18]. This was also described for
F. excelsior by Rodríguez Madrera et al. [
32], while Sanz et al. [
18] indicated that sinapaldehyde and syringaldehyde, followed by coniferaldehyde and vanillin, are the main compounds. The dry wood of
F. excelsior has been described as being poorer in all the LMWP than
F. americana (
Table 6), although
F. excelsior is richer in syringaldehyde, sinapaldehyde, vanillin, syringic acid and ferulic acid than
F. americana after the same toasting, and
F. americana presents greater concentrations of coniferaldehyde and vanillic acid (
Table 7). Vanillin concentrations in toasted wood varies between 245 and 329 µg/g in
F. americana and 222 and 404 µg/g in
F. excelsior, concentrations generally similar or even higher than those found in traditional oaks (
Table 7). Vanillic acid and protocatechuic acid are not detected in
F. excelsior from Galicia, Spain [
32], but are by Sanz et al. [
18] although the origin of this wood is not stated. Finally, it should be mentioned that hydrotyrosol is detected in dry
F. excelsior wood but not in any other (oak, cherry, acacia or chestnut) [
6,
18].
Alnus glutinosa L. and Fagus sylvatica L.:
Detailed LMWP composition in these two species was only found in one study [
32], which looked at the wood with two toasting intensities. The major compounds described are coniferaldehyde and sinapaldehyde in both species and after both toastings, although protocatechuic acid is also quantified in
F. sylvatica. These authors did not find protocatechuic acid in any of the oaks studied [
32], nor did they find 4-hydroxybenzaldehyde, which is quantified in
A. glutinosa and
F. sylvatica. Coniferaldehyde, sinapaldehyde, syringaldehyde and vanillin concentrations are higher in oaks than in
A. glutinosa and
F. sylvatica (
Table 7).