Abstract
The number of positron-emission tomography (PET) tracers used to evaluate patients with brain tumors has increased substantially over the last years. For the management of patients with brain tumors, the most important indications are the delineation of tumor extent (e.g., for planning of resection or radiotherapy), the assessment of treatment response to systemic treatment options such as alkylating chemotherapy, and the differentiation of treatment-related changes (e.g., pseudoprogression or radiation necrosis) from tumor progression. Furthermore, newer PET imaging approaches aim to address the need for noninvasive assessment of tumoral immune cell infiltration and response to immunotherapies (e.g., T-cell imaging). This review summarizes the clinical value of the landscape of tracers that have been used in recent years for the above-mentioned indications and also provides an overview of promising newer tracers for this group of patients.
1. Introduction
For the management of patients with brain tumors, clinicians frequently need to rely on imaging information obtained from anatomical magnetic resonance imaging (MRI) before, during, and after the treatment. While contrast-enhanced MRI is of paramount value in neuro-oncology, its specificity for neoplastic tissue is low, and changes of the blood-brain barrier permeability as indicated by contrast enhancement are not limited to tumor tissue [1,2,3,4,5,6,7]. Nevertheless, precise delineation of tumor extent, including non-enhancing tumor subregions, is decisive for several diagnostic and therapeutic steps (e.g., planning of biopsy, surgery, or radiotherapy) [7,8]. Following radio- and/or chemotherapy, neurooncologists often encounter treatment-related changes. Some of these, e.g., pseudoprogression, are difficult to differentiate from actual tumor progression with conventional MRI alone [6,7,8,9,10,11,12]. Pseudoprogression describes a phenomenon characterized by an increase of contrast enhancement without clinical deterioration, and which disappears again over time without any treatment change [3,9,13,14,15,16]. Such treatment-related changes may occur early (in the case of pseudoprogression typically within the first 12 weeks after chemoradiation completion) or late (several months or even years after radiotherapy in the case 31 of radiation necrosis) [7,8,17].
Furthermore, surrogates of treatment response or progression obtained from MRI (e.g., a decrease of contrast enhancement or the fluid-attenuated inversion recovery (FLAIR) signal hyperintensity) may be unspecific. They can be influenced by inflammation, infarction, and reactive changes after surgery [2,6,12,18]. If treatment-related changes remain unidentified, an effective treatment may be erroneously terminated prematurely. The latter may also harm survival and mislead study results evaluating novel treatment approaches for tumor relapse [19].
To overcome these diagnostic challenges, imaging techniques with higher diagnostic accuracy than conventional MRI offering more than just anatomical information are needed. Apart from advanced MRI techniques, positron-emission tomography (PET) imaging has been evaluated over the past decades. It has been shown that PET imaging offers additional value in neuro-oncology since it enables the non-invasive evaluation of molecular and metabolic features of brain tumors. PET, therefore, is of great value for the indications mentioned above, which are of particular clinical interest [7,8,10,20]. Consequently, the PET task force of the Response Assessment in Neuro-Oncology (RANO) working group highlighted the additional clinical value of PET imaging using amino acid tracers compared to anatomical MRI. Accordingly, its widespread clinical use was recommended in patients with glioma and brain metastases [17,21].
The continuously growing landscape of PET tracers enables the evaluation of many biochemical processes in patients with brain tumors. With the advent of newer treatment options in neuro-oncology, in particular, targeted therapy and various immunotherapy options, the needs for additional information derived from neuroimaging in terms of characterization of the tumor environment, the evaluation of tumoral drug accumulation, immune cell infiltration, and the diagnosis of treatment-related changes following these newer treatment options are steadily increasing. Some of these requirements may be met by the existing landscape of well-established PET tracers, while others can be addressed by newer ones [22,23].
This review summarizes the value of PET tracers that have been used in brain tumors in recent years for the most relevant clinical indications. Furthermore, more unique but promising PET tracers are summarized and discussed.
2. Methods
A PubMed search using the terms “PET”, “positron”, “tracer”, “glioma”, “brain metastases”, “FDG”, “amino acid”, “methionine”, “FET”, “FDOPA”, “FACBC”, “AMT”, “TSPO”, “GE-180”, “FLT”, “FAZA”, “EGFR”, “VEGF”, “immunoPET”, “isocitrate dehydrogenase”, “radiotherapy”, “T-cell imaging”, “reporter gene”, “radiation necrosis”, “pseudoprogression”, “tumor extent”, “response assessment”, “treatment-related changes”, and combinations thereof was performed until January 2020. The PET tracers were evaluated regarding their clinical value for the delineation of tumor extent, diagnosis of treatment-related changes, the assessment of treatment response (Table 1), and according to the information provided by newer PET probes (Table 2).

Table 1.
Frequently used PET tracers for the delineation of tumor extent, diagnosis of treatment-related changes, and the assessment of treatment response in brain tumor imaging.

Table 2.
Promising PET tracers for the evaluation of newer treatment options.
3. Current Landscape of PET Imaging
PET allows targeting metabolic and molecular processes in patients with brain tumors relevant to diagnosis, treatment, and prognosis that cannot be assessed with anatomic computed tomography (CT) or MR imaging. A variety of PET tracers have been evaluated predominantly in glioma patients or patients with brain metastases with the main focus on glucose metabolism, amino acid transport, proliferation, hypoxia, blood flow, or angiogenesis. This section will provide an overview of PET tracers for brain tumors that have been evaluated in human subjects in the last years, especially for those above mentioned highly relevant indications in clinical routine. An overview is presented in Table 1.
3.1. PET Imaging of Glucose Metabolism
[18F]-2-Fluoro-2-deoxy-D-glucose ([18F]FDG) is the most widespread PET tracer in nuclear medicine. In neoplastic tissue, the uptake of [18F]FDG reflects the increased expression of glucose transporters and hexokinase. The latter enzyme phosphorylates glucose and [18F]FDG. In the central nervous system, the physiologically high and varying uptake of [18F]FDG in healthy brain parenchyma hampers the accurate delineation of the brain tumor. This limits the diagnostic accuracy for the correct identification of treatment-related changes and assessment of treatment response in gliomas and brain metastases [17,21]. It has repeatedly been shown that the diagnostic accuracy of [18F]FDG regarding the differentiation of radiation-induced changes from glioma and brain metastases recurrence is inferior to other imaging modalities, including advanced MRI and amino acid PET [17,24,25,26]. However, [18F]FDG PET seems to be of value for the delineation of tumor extent and assessment of treatment response in patients with primary central nervous system (CNS) lymphoma [27,28,29,30].
3.2. PET Using Amino Acid PET Tracers
Radiolabeled amino acid tracers (Figure 1) are of great interest in brain tumor imaging because of the high tumor-to-brain contrast based on the relatively high specificity for neoplastic tissue and the low uptake in healthy brain tissue [7,8,21,31,32,33].
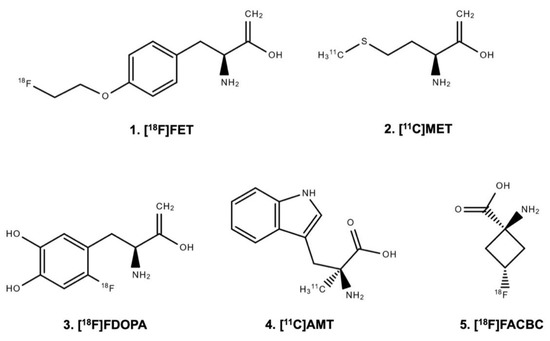
Figure 1.
Chemical structure of radiolabeled amino acids.
3.2.1. Uptake Mechanisms of Amino Acid PET Tracers
The uptake of the amino acid tracers O-(2-[18F]fluoroethyl)-L-tyrosine ([18F]FET), [11C]methyl-L-methionine ([11C]MET), and 3,4-dihydroxy-6-[18F]fluoro-L-phenylalanine ([18F]FDOPA) is mainly based on the increased expression of large neutral amino acid transporters of the l-type (LAT) in gliomas and brain metastases (i.e., subtypes LAT1 and LAT2) [7,34,35,36,37]. Moreover, LAT1 overexpression correlates with malignant phenotypes and proliferation of gliomas. It is associated with glioma angiogenesis [38,39]. A critical consideration for the practical application of [11C]MET compared to [18F]FET or [18F]FDOPA is the half-life of the [11C]-isotope ([18F] 110 vs. [11C] 20 min) [40,41], which allows the transport of [18F]FET and [18F]FDOPA to PET facilities. In contrast, the use of [11C]MET necessitates an on-site cyclotron. In many European centers, this logistical disadvantage has led to the replacement of [11C]MET predominantly by [18F]FET [7]. When using [18F]FDOPA, the physiological uptake in the striatum may hamper the evaluation of tumor extent [7,42].
The L-tryptophan analogue α-[11C]-methyl-L-tryptophan ([11C]AMT) is another radiolabeled amino acid with uptake via the LAT system. Additionally, [11C]AMT uptake is mediated via the kynurenine pathway and has a rate-limiting enzyme indoleamine 2,3-dioxygenase [43]. Indoleamine 2,3-dioxygenase is upregulated in various cancers including gliomas [44], which prompted the use of [11C]AMT PET in patients with brain tumors [45].
Other tracers such as the synthetic amino acid analog anti-1-amino-3-[18F]fluorocyclobutane-1-carboxylic acid ([18F]FACBC or [18F]fluciclovine) are also LAT-mediated but use additionally the alanine, serine, and cysteine transporter 2, which is upregulated in many human cancers [46,47,48,49].
3.2.2. Value of Amino Acid PET Tracers for Brain Tumor Patients
For the planning of diagnostic and therapeutic procedures, the precise delineation of tumor spread is essential. For example, the tumor extent as assessed by amino acid PET provides valuable information for planning stereotactic biopsies, resection, and radiotherapy [1,50,51,52]. For [11C]MET and [18F]FET, it has been shown that the delineation of tumor extent, particularly in non-enhancing gliomas, can be assessed with high accuracy using amino acid PET [51,53]. Preliminary data suggest that newer tracers such as [18F]FACBC PET are also helpful in identifying metabolically active and non-contrast enhancing tumor regions in glioma patients [54,55,56]. Moreover, it has been shown that in the majority of cases, the metabolically active tumor burden as assessed by amino acid PET extends considerably beyond the volume of MRI contrast enhancement, which is of significant relevance for subsequent treatment planning [1,7,53,57]. Regarding the comparability of PET tracers for this indication, [11C]MET, [18F]FET, and [18F]FDOPA seem to be equally informative [58,59,60,61]. Nevertheless, it has to be pointed out that 20–30% of grade II gliomas, according to the World Health Organization (WHO) classification of tumors of the central nervous system [62,63], show no amino acid uptake [64,65,66]. A negative amino acid PET, therefore, does not exclude glioma [8].
For the differentiation of treatment-related changes from tumor relapse, amino acid PET also provides valuable diagnostic information. Using [18F]FET or [18F]FDOPA, especially the differentiation of radiation injury from tumor relapse in glioma patients, as well as in patients with brain metastases, can be obtained with a relatively high diagnostic accuracy between 80–90% (Figure 2) [11,26,67,68,69,70,71,72,73,74,75,76,77,78,79,80]. Importantly, in glioma patients, parameters derived from dynamic [18F]FET PET acquisition may further increase the diagnostic accuracy [67,74,75,76,78]. This has also been demonstrated in patients with brain metastases who underwent radiosurgery for brain metastases treatment [80,81]. The diagnostic accuracy of [11C]MET PET regarding this clinical question is slightly lower (approximately 75%) [8,82,83], which is most probably related to the higher affinity of [11C]MET for inflammation [84]. First PET studies using [11C]AMT or [18F]FACBC suggest that these tracers may also be of value for the differentiation of radiation injury from glioma progression [85,86].
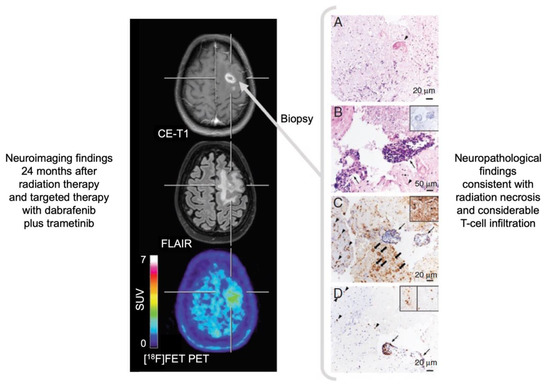
Figure 2.
Radiation necrosis and chronic inflammation in a patient with brain metastases of a B-Raf proto-oncogene (BRAF)-mutated malignant melanoma who had been treated with whole-brain radiation therapy combined with concurrent dabrafenib plus trametinib. Twenty-four months later, the contrast-enhanced magnetic resonance imaging (MRI) indicates a recurrence of the brain metastases (left panel), whereas the O-(2-[18F]fluoroethyl)-L-tyrosine ([18F]FET) positron-emission tomography (PET) shows only insignificant metabolic activity and is consistent with the findings of treatment-related MRI changes. Neuropathological findings (right panel) after stereotactic biopsy show signs of radiation necrosis as well as considerable T-cell infiltration. (A) Hyaline, eosinophilic necrosis with evidence of a necrotic vessel wall (arrowhead). (B) Vital brain parenchyma besides necrosis with activated microglia cells (arrowhead), and blood vessels with lymphocyte infiltrates (arrows) without evidence of tumor cells (inserted box). (C) Adjacent to inflamed blood vessels (arrows), a resorption of necroses by macrophages (block arrows) as well as activated microglia cells (arrowheads) and astrocytes in the brain parenchyma (inserted box). (D) The main population of intra- and perivascular T-cell infiltrates are CD3+ (arrow), but also CD4+ (inserted box left) and CD8+ (inserted box right) T-cells contribute to the infiltrates (modified from Galldiks et al. [10], with permission from Oxford University Press).
The advent of immunotherapy using immune checkpoint inhibitors and targeted therapy has improved the survival of cancer patients, particularly in melanoma and lung cancer. Recent trials suggest that patients with brain metastases from these tumor entities may also benefit from these agents alone or in combination [87]. Regarding patients with brain metastases treated with checkpoint inhibitors or targeted therapy (frequently combined with radiotherapy), initial data indicate that amino acid PET may provide valuable information for differentiating relapse from equivocal MRI findings related to immunotherapy-induced inflammation [10,88].
Recently, a variety of experimental treatment options has been introduced for treating patients with high-grade glioma. [18F]FET PET was shown to differentiate benign MRI findings related to these experimental therapies, e.g., immunotherapy with dendritic cell vaccination or targeted therapy with regorafenib, from tumor relapse [89,90]. However, the number of patients treated with these therapies and monitored with [18F]FET PET is still small, and the results should be interpreted with caution.
The assessment of the response to a particular neurooncological treatment is of clinical relevance since treatment decisions can be negatively affected by treatment-related changes. The accurate assessment of response helps both to discontinue an ineffective treatment option as early as possible and to prevent an effective treatment from being erroneously terminated prematurely with a potentially harmful influence on survival. Furthermore, the evaluation of response also helps to avoid possible treatment side effects, e.g., bone marrow depression or fatigue, and, therefore, to maintain or even improve life-quality. It has been shown in glioma patients that the assessment of response to alkylating chemotherapy (i.e., temozolomide or lomustine) using [11C]MET or [18F]FET PET provides valuable additional information compared to contrast-enhanced MRI. Importantly, metabolic PET responders (i.e., patients with a decrease of tumor-to-brain ratios or metabolically active tumor volumes at follow-up relative to baseline imaging) had a significantly longer survival than metabolic non-responders [91,92,93,94,95,96,97,98].
Following antiangiogenic therapy using bevacizumab, the use of reduced contrast enhancement as a surrogate marker for treatment response is not optimal due to a phenomenon called pseudoresponse [9]. Pseudoresponse describes a decrease of contrast enhancement related to a rapid restoration of the blood-brain barrier by antiangiogenic drugs [9]. However, a clinical benefit is not infrequently lacking in patients with an impressive radiological response (pseudoresponse). [18F]FET and [18F]FDOPA PET may provide valuable information regarding the identification of pseudoresponse [99,100,101,102]. Moreover, [18F]FDOPA and [18F]FET PET were also able to predict a favorable clinical outcome in bevacizumab responders [101,102,103,104].
After chemoradiation completion, newly diagnosed glioblastoma patients can be treated with tumor-treating fields therapy in addition to adjuvant temozolomide chemotherapy [105]. Initial studies suggest that amino acid PET might identify responding patients undergoing tumor-treating fields therapy, but it has to be considered that the response can also be related to the concurrently applied chemotherapy or delayed chemoradiation effects [106,107].
3.3. PET Imaging of the Mitochondrial Translocator Protein
PET ligands targeting the 18 kDa mitochondrial translocator protein (TSPO), located at the outer mitochondrial membrane and formerly known as the peripheral benzodiazepine receptor, are also of interest in neuro-oncology [108,109]. TSPO is associated with neuroinflammation due to its expression in activated microglia, endothelial cells, and infiltrating macrophages [109]. The PET ligand [11C]PK11195 was one of the first ligands evaluated for TSPO expression in glioma patients [110,111,112].
The recently introduced TSPO ligand GE-180 labeled with [18F] offers an increased binding specificity and was tested in patients with gliomas [113] and neuroinflammatory diseases such as multiple sclerosis [114,115,116]. Regarding the delineation of glioma extent, it has been demonstrated that the [18F]GE-180 uptake volume is significantly larger than the volume of contrast enhancement [113,117]. However, when comparing [18F]FET with [18F]GE-180 uptake volumes intraindividually in terms of spatial distribution, the overlap is only moderate (Dice similarity coefficient, 0.55) despite comparable tumor volumes [117]. These differences might help to characterize glioma heterogeneity and warrant further studies with spatial correlation of imaging findings of [18F]FET uptake to [18F]GE-180 uptake with neuropathology.
3.4. PET Imaging of Cellular Proliferation
The radiolabeled nucleoside 3′-deoxy-3′-[18F]fluorothymidine ([18F]FLT) is a pyrimidine analogue. It is used to evaluate cellular proliferation because of its rapid incorporation into newly synthesized DNA [118]. [18F]FLT is trapped intracellularly after phosphorylation by the thymidine kinase-1, a cytoplasmatic enzyme expressed during cell proliferation [46,119]. However, the requirement of a disrupted blood-brain barrier for [18F]FLT uptake may limit its diagnostic use [2,120]. For example, in terms of tumor detection and delineation, [18F]FLT PET was less sensitive than [11C]MET PET to detect WHO grade II gliomas, which usually show no contrast enhancement [121]. Furthermore, a meta-analysis evaluating the value of [18F]FLT PET for the diagnosis of glioma recurrence based on approximately 800 patients showed no superiority of [18F]FLT (pooled sensitivity, 82%; pooled specificity, 76%) compared to [18F]FDG (pooled sensitivity, 78%; pooled specificity, 77%) [122].
On the other hand, [18F]FLT PET seems to be useful for the assessment of response to antiangiogenic therapy with bevacizumab in patients with recurrent malignant glioma. [18F]FLT PET was able to identify a reduction of proliferative activity in responding patients with favorable outcome as an indicator for response compared to metabolic non-responders [123,124,125]. Furthermore, [18F]FLT PET was used in patients with malignant melanoma brain metastases treated with targeted therapy or immunotherapy using checkpoint inhibitors [126]. In that study, responding patients showed a clearer reduction of proliferative activity as assessed by [18F]FLT PET than the decrease of contrast enhancement on standard MRI.
3.5. PET Imaging of Tumor Hypoxia
Hypoxia is a key factor in treatment outcome in various cancers, including glioma. It has been shown that hypoxia is associated with tumor persistence and resistance to cancer treatment [127]. To further evaluate this phenomenon using PET, the tracer [18F]fluoromisonidazole ([18F]FMISO) has been developed, which is trapped in hypoxic but viable cells [46,128,129]. It has been demonstrated in glioblastoma patients that [18F]FMISO PET delineates additional hypoxic tumor subregions which exceed the contrast-enhancing tumor parts, indicating that hypoxia may induce peripheral tumor growth [130]. A subsequent study showed that the metabolically active tumor volume in [11C]MET PET strongly correlated with the hypoxic volume defined by [18F]FMISO [131]. Importantly, the tumor area on [11C]MET PET exceeded the area of the contrast enhancement on MRI in the range of 20–30%.
More recently, [18F]FMISO PET has been used for monitoring the effects of antiangiogenic therapy with bevacizumab in patients with recurrent malignant glioma [132,133]. It was shown that patients who had a response in both contrast-enhanced MRI and [18F]FMISO PET had significantly longer survival than patients who responded on MRI only [133].
[18F]-labeled flouroazomycin arabinoside ([18F]FAZA) may be a promising alternative to [18F]FMISO offering an improved tumor-to-background ratio due to faster blood clearance [134,135]. Preliminary data in glioblastoma patients suggest that [18F]FAZA PET might be of value for radiotherapy response assessment [136].
3.6. PET Imaging of Tumor Perfusion
The evaluation of regional cerebral blood flow (rCBF) allows identifying brain tumors with high vascularization. rCBF can be measured by PET using [15O]-labeled water. However, [15O]H2O requires an on-site cyclotron because of its very short half-life (2 min) [137]. The development and easy accessibility of perfusion-weighted CT and MRI has led to various studies evaluating brain tumors outnumbering [15O]H2O PET studies by far. In direct comparisons, it has been shown that rCBF values differ considerably between CT- and MRI-based perfusion measurements and [15O]H2O PET [138,139].
In patients with malignant gliomas undergoing chemoradiation with nitrosoureas, a reduction of rCBF was evaluated using [15O]H2O PET, but data on subsequent survival as an indicator for treatment response are lacking [140,141].
3.7. PET Imaging of Angiogenesis
The vascular endothelial growth factor (VEGF) is overexpressed by most tumors, including brain tumors, and an important trigger for neovascularization [142]. Bevacizumab is a recombinant humanized monoclonal antibody against VEGF and appears to prolong progression-free survival and decrease steroid usage in patients with malignant glioma [142,143]. Based on these properties, [89Zr]-labeled bevacizumab PET imaging has been evaluated for tumoral drug accumulation in pediatric patients with diffuse intrinsic pontine glioma [144]. That study demonstrated intertumoral heterogeneity of drug accumulation and may aid in selecting those patients with the greatest chance of benefit from bevacizumab [144,145]. Other studies have used [64Cu]-labeled conjugates of VEGF with DOTA (1,4,7,10-tetra-azacylododecane N,N′,N′′,N′′′-tetraacetic acid) for PET imaging in animal models [146,147,148]. Similarly, these approaches suggest the clinical value for the identification of patients who will benefit from anti-VEGF therapy.
In adult glioblastoma patients, it has been demonstrated that accumulation of [123I]VEGF in the tumor region of glioblastomas can be assessed using single photon emission computed tomography (SPECT) imaging [149]. Importantly, high uptake of [123I]VEGF was able to identify glioblastoma patients with a poor clinical outcome.
4. Emerging Fields of PET Imaging
Currently, blockade of immune checkpoints and other immunotherapy options (i.e., vaccination strategies, oncolytic virus approaches, cell-based immunotherapy such as chimeric antigen receptor T-cells (CAR-T cells) are under evaluation in patients with brain cancer including glioma and brain metastases. Furthermore, recent study results suggest that newer-generation targeted therapies are a promising treatment option, especially in a subset of patients with brain metastases [150]. As a new treatment option, mutations of the isocitrate dehydrogenase (IDH) gene, which frequently occur in WHO grade II and III gliomas, have also gained interest as a potential treatment target [151,152]. All these promising treatment options impose new demands on brain imaging (e.g., imaging of immune reactions in the brain). The current body of literature suggests that PET has the potential to adapt to these needs. An overview is presented in Table 2.
4.1. PET Imaging of the Epidermal Growth Factor Receptor Family
Both the epidermal growth factor receptor (EGFR) and the human epidermal growth factor receptor 2 (HER2) are transmembrane protein receptors and belong to the EGFR family. These receptors are targets for various growth factors that mediate various cellular processes such as differentiation or proliferation. In clinical oncology, various gene mutations may lead to overexpression of these proteins and are associated with the development of a variety of cancers. Importantly, these mutations also play a significant role in various treatment options, including tyrosine kinase inhibitors and monoclonal antibodies targeting EGFR, HER2, or both [153], as well as for imaging. Especially the evaluation of a response to these targeted therapy options in patients with brain metastases (from melanoma, lung, or breast cancer) as well as in glioma patients is the indication with the highest clinical potential for EGFR- and HER2-targeted PET [154,155]. In recent years, radiolabeled EGFR and HER2 antibodies, as well as tyrosine kinase inhibitors, have been used as PET imaging agents.
For identifying EGFR overexpression, PET ligands such as [11C]erlotinib, [11C]PD153035, and [89Zr]Zr-DFO-nimotuzumab have been used [156,157,158]. The most relevant PET tracers for imaging of HER2 overexpression are [64Cu]DOTA-trastuzumab and [89Zr]pertuzumab (Figure 3) [159,160].
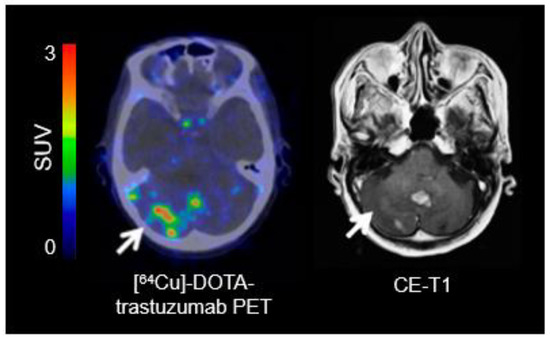
Figure 3.
[64Cu]-DOTA-trastuzumab positron-emission tomography (PET) and contrast-enhanced magnetic resonance imaging (MRI) performed one day after initiation of treatment with trastuzumab in a patient with a human epidermal growth factor receptor 2 (HER2)-positive breast cancer with brain metastases. In single brain metastases, [64Cu]-DOTA-trastuzumab PET helps to improve lesion detection (arrow) (modified from Tamura et al. [160], with permission from the Society of Nuclear Medicine and Molecular Imaging).
4.2. Immuno-Imaging: Immuno-PET and Imaging of T-Cells
In recent years, immunotherapy with antibodies directed against immune checkpoints such as the cytotoxic T-lymphocyte antigen-4 (CTLA-4; e.g., ipilimumab), the programmed cell death receptor-1 (PD-1; e.g., pembrolizumab, nivolumab), or the programmed cell death protein ligand 1 (PD-L1; e.g., atezolizumab) have gained paramount importance in clinical oncology and neuro-oncology. However, the efficacy and responsiveness of these agents may vary considerably among different cancer types and across individuals. Biomarkers obtained from tumor tissue, such as PD-1 and PD-L1 expression, can help to select patients. However, these tissue biomarkers are limited, and some patients show no response even if the target is present [161]. Therefore, the significance of PET for predicting response to immunotherapy and patient selection increases. Among other methods, immuno-PET combines antibodies or antibody fragments with a radionuclide and takes advantage of the specificity and affinity of antibodies and the sensitivity of PET [162]. Generally, targets for immuno-PET can be T-cell markers (e.g., CD4+, CD8+), immune checkpoints (e.g., CTLA-4, PD-1, PD-L1), or biomarkers of the immune response (e.g., interferon-γ, interleukin-2) [23].
First-in-human studies suggest that targeting PD-1 with [89Zr]nivolumab [163] or PD-L1 with [89Zr]atezolizumab [164] is useful as imaging biomarkers to non-invasively evaluate the expression of these immune checkpoints in patients with extra- and intracranial cancer [164,165]. Engineered target-binding proteins (adnectins) for PD-L1 ligands such as [18F]BMS-986192 are currently under evaluation [166].
Tumor-infiltrating T-cells (such as CD8+) play an essential role in the activation of immune cells in response to checkpoint inhibition [167]. Recently, it has been shown that a radiolabeled [89Zr]IAB22M2C has the potential to visualize CD8+ T-cell-enriched tumor tissue [168]. The assessment of immune cells infiltrating tumors has also been investigated with PET using radiolabeled clofarabine (2-chloro-2′-deoxy-2′-[18F]fluoro-9-b-D-arabinofuranosyl-adenine; [18F]CFA) [169]. [18F]CFA is a substrate for the enzyme deoxy-cytidine kinase, which is overexpressed in immune cells such as CD8+ T-cells [169]. Importantly, [18F]CFA PET has shown a great clinical potential to localize and quantify immune responses in glioblastoma patients undergoing dendritic cell vaccination treatment combined with immune checkpoint blockade (Figure 4) [169].
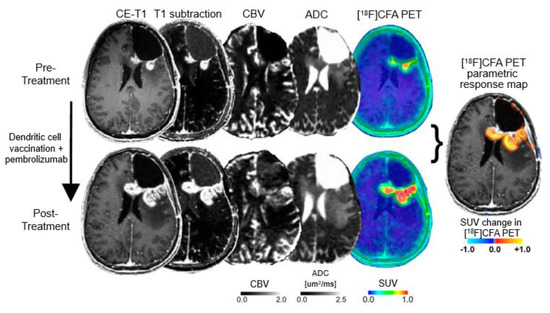
Figure 4.
Detection of immune response in a patient with recurrent glioblastoma using 2-chloro-2′-deoxy-2′-[18F]fluoro-9-b-D-arabinofuranosyl-adenine ([18F]CFA) positron-emission tomography (PET) and advanced magnetic resonance imaging (MRI) before (upper panel) and after treatment with dendritic cell vaccination and programmed cell death receptor-1 (PD-1) blockade using pembrolizumab (lower panel). Following treatment, [18F]CFA uptake is considerably increased, indicating an immune-cell infiltration, and helps distinguishing tumor progression from inflammation (modified from Antonios et al. [169], with permission from the National Academy of Sciences).
Another interesting approach is the transfection of immune cells with a reporter gene that encodes a protein that can be specifically targeted by a radiolabeled reporter probe [170]. Imaging of reporter gene expression of cells transfected with the herpes simplex virus type 1 thymidine kinase reporter gene has been demonstrated using 9-[4-[18F]fluoro-3-(hydroxymethyl)butyl]guanine ([18F]FHBG) [171]. Moreover, this technique has the potential to image recently introduced cell-based therapies with CAR-T cells [23]. Accordingly, a study with recurrent high-grade glioma patients suggested that [18F]FHBG PET can detect reporter gene expression in CAR-engineered cytotoxic T-lymphocytes [172].
4.3. PET Imaging of Isocitrate Dehydrogenase Mutations
In patients with malignant glioma, the modest outcome improvement following both standard therapy (i.e., chemoradiation with temozolomide) and newer treatment options (e.g., tumor-treating fields) has prompted various efforts to identify molecules that are fundamental to regulate tumor progression and provide additional options for personalized therapy in this group of patients.
Accordingly, the enzyme isocitrate dehydrogenase (IDH) has gained interest as a potential target. IDH is an enzyme of the Krebs cycle, catalyzing the oxidative decarboxylation of isocitrate to alpha-ketoglutarate. Mutations in the IDH1 and IDH2 gene, frequently occurring in WHO grade II or III astrocytomas and oligodendrogliomas, result in a significant increase of the oncometabolite 2-hydroxyglutarate (2-HG) [173,174]. In cells with IDH mutant enzymes, the accumulation of 2-HG alters several downstream cellular activities, causing epigenetic dysregulation and, consequently, a block in cellular differentiation, leading to oncogenesis [175].
Therefore, mutant IDH proteins are highly attractive targets for inhibitory drugs. In glioma patients, selective oral IDH inhibitors of IDH1 (i.e., ivosidenib, BAY-1436032), pan-IDH1/2 (i.e., AG-881), and vaccination strategies targeting the IDH1R132H mutation are currently under clinical evaluation. Initial results predominantly from phase-1 studies are promising and suggest that these inhibitors are safe and have antitumoral activity [151,152,176]. Although immunohistochemistry and genomic sequencing are the methods of choice for the detection of an IDH mutation, these techniques are invasive and are not appropriate for treatment monitoring, which requires continual assessment. Furthermore, the use of magnetic resonance spectroscopy (MRS) to non-invasively evaluate 2-HG is technically challenging, and may be false-positive in 20% of cases [177,178].
Newer PET probes for imaging mutant IDH expression in gliomas may be an alternative imaging method. Recent radiochemical developments suggest that triazinediamine or butyl-phenyl sulfonamide analogs labeled with [18F] are promising candidate radiotracers for noninvasive PET imaging of IDH mutations in gliomas [179,180]. Furthermore, a [18F]-labeled IDH1 inhibitor (AGI-5198) has also been investigated [181]. Interestingly, Koyaso and colleagues demonstrated that [11C]acetate uptake in IDH mutant cells is significantly higher than in IDH wild-type cells because of metabolic trapping [182]. Taken together, further efforts to translate these promising approaches for IDH imaging into clinical use are warranted.
4.4. PET-Based Theranostics
The combination of therapeutics and diagnostics, also termed as theranostics, supports the concept of precision oncology. One PET-based theranostic approach is the peptide receptor radionuclide therapy (PRRT), in which overexpressed tumor-specific receptors are used as a therapeutic target. By exchanging the radionuclide used for diagnostic PET such as [68Ga] with a radiation source, typically ß-emitters like [177Lu] or [90Y], the same PET tracer can be used for therapy. Although there are currently no theranostic approaches clinically established for gliomas or brain metastases, there are promising concepts. For example in patients with glioblastoma, a potential target is the overexpressed chemokine receptor-4 (CXCR4) which is associated with a poor clinical outcome [183,184,185]. Visualization of CXCR4 expression using diagnostic PET with CXCR4-directed [68Ga]Pentixafor® has been demonstrated in glioblastoma patients [186].
Another potential target is the prostate-specific membrane antigen (PSMA) which may be overexpressed in prostate cancer and also in predominantly malignant gliomas. Diagnostic PET imaging of PSMA expression in patients with malignant glioma can be obtained using [68Ga]-labeled PSMA ligands [187,188,189]. The use of the theranostic agent [177Lu]-PSMA-617 has demonstrated favorable safety and efficacy in patients with advanced prostate cancer, indicating the potential to be also of value for patients with malignant glioma [190]. Nevertheless, the potential of theranostics needs to be further evaluated in patients with malignant gliomas.
5. Discussion
Anatomical MRI is currently the method of choice for neuroimaging of brain tumors, but PET complements this technique and provides important biological information that cannot be obtained from anatomical MRI alone. Currently, best-established PET tracers in neuro-oncology are radiolabeled amino acids targeting L-system transporters. However, a considerable number of other PET tracers have been developed for brain tumor patients and allow the evaluation of a wide range of biochemical processes. A variety of PET biomarkers offers the potential to play a clinically significant role for the monitoring of newer treatment options such as targeted therapy and immunotherapy, e.g., by providing an early assessment of response to these options, and for a more accurate differentiation of viable tumor from treatment-related changes. A major current shortcoming is the lack of large, prospective clinical trials in patients with both glioma and brain metastases for many of these PET tracers. Despite encouraging early study results in the field, it has to be further demonstrated that these tracers improve considerably patient management and outcome.
Author Contributions
Study design, writing of manuscript drafts: J.W. and N.G. Revising manuscript, approving final content of the manuscript: All. All authors have read and agreed to the published version of the manuscript.
Funding
The Cologne Clinician Scientist-Program (CCSP) of the Deutsche Forschungsgemeinschaft (DFG, FI 773/15-1), Germany, supported this work.
Conflicts of Interest
Related to the present work, the authors disclosed no potential conflicts of interest.
References
- Lohmann, P.; Stavrinou, P.; Lipke, K.; Bauer, E.K.; Ceccon, G.; Werner, J.M.; Neumaier, B.; Fink, G.R.; Shah, N.J.; Langen, K.J.; et al. FET PET reveals considerable spatial differences in tumour burden compared to conventional MRI in newly diagnosed glioblastoma. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 591–602. [Google Scholar] [CrossRef]
- Dhermain, F.G.; Hau, P.; Lanfermann, H.; Jacobs, A.H.; van den Bent, M.J. Advanced MRI and PET imaging for assessment of treatment response in patients with gliomas. Lancet Neurol. 2010, 9, 906–920. [Google Scholar] [CrossRef]
- Brandsma, D.; Stalpers, L.; Taal, W.; Sminia, P.; van den Bent, M.J. Clinical features, mechanisms, and management of pseudoprogression in malignant gliomas. Lancet Oncol. 2008, 9, 453–461. [Google Scholar] [CrossRef]
- Hygino da Cruz, L.C., Jr.; Rodriguez, I.; Domingues, R.C.; Gasparetto, E.L.; Sorensen, A.G. Pseudoprogression and pseudoresponse: Imaging challenges in the assessment of posttreatment glioma. AJNR Am. J. Neuroradiol. 2011, 32, 1978–1985. [Google Scholar] [CrossRef]
- Yang, I.; Aghi, M.K. New advances that enable identification of glioblastoma recurrence. Nat. Rev. Clin. Oncol. 2009, 6, 648–657. [Google Scholar] [CrossRef]
- Kumar, A.J.; Leeds, N.E.; Fuller, G.N.; Van Tassel, P.; Maor, M.H.; Sawaya, R.E.; Levin, V.A. Malignant gliomas: MR imaging spectrum of radiation therapy- and chemotherapy-induced necrosis of the brain after treatment. Radiology 2000, 217, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Langen, K.J.; Galldiks, N.; Hattingen, E.; Shah, N.J. Advances in neuro-oncology imaging. Nat. Rev. Neurol. 2017, 13, 279–289. [Google Scholar] [CrossRef]
- Galldiks, N.; Lohmann, P.; Albert, N.L.; Tonn, J.C.; Langen, K.-J. Current status of PET imaging in neuro-oncology. Neurooncol. Adv. 2019, 1. [Google Scholar] [CrossRef]
- Brandsma, D.; van den Bent, M.J. Pseudoprogression and pseudoresponse in the treatment of gliomas. Curr. Opin. Neurol. 2009, 22, 633–638. [Google Scholar] [CrossRef]
- Galldiks, N.; Kocher, M.; Ceccon, G.; Werner, J.M.; Brunn, A.; Deckert, M.; Pope, W.B.; Soffietti, R.; Le Rhun, E.; Weller, M.; et al. Imaging challenges of immunotherapy and targeted therapy in patients with brain metastases: Response, progression, and pseudoprogression. Neuro Oncol. 2020, 22, 17–30. [Google Scholar] [CrossRef]
- Galldiks, N.; Dunkl, V.; Stoffels, G.; Hutterer, M.; Rapp, M.; Sabel, M.; Reifenberger, G.; Kebir, S.; Dorn, F.; Blau, T.; et al. Diagnosis of pseudoprogression in patients with glioblastoma using O-(2-[18F]fluoroethyl)-L-tyrosine PET. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 685–695. [Google Scholar] [CrossRef] [PubMed]
- Ahluwalia, M.S.; Wen, P.Y. Antiangiogenic therapy for patients with glioblastoma: Current challenges in imaging and future directions. Expert Rev. Anticancer Ther. 2011, 11, 653–656. [Google Scholar] [CrossRef]
- Taal, W.; Brandsma, D.; de Bruin, H.G.; Bromberg, J.E.; Swaak-Kragten, A.T.; Smitt, P.A.; van Es, C.A.; van den Bent, M.J. Incidence of early pseudo-progression in a cohort of malignant glioma patients treated with chemoirradiation with temozolomide. Cancer 2008, 113, 405–410. [Google Scholar] [CrossRef]
- Radbruch, A.; Fladt, J.; Kickingereder, P.; Wiestler, B.; Nowosielski, M.; Baumer, P.; Schlemmer, H.P.; Wick, A.; Heiland, S.; Wick, W.; et al. Pseudoprogression in patients with glioblastoma: Clinical relevance despite low incidence. Neuro Oncol. 2015, 17, 151–159. [Google Scholar] [CrossRef]
- Galldiks, N.; Kocher, M.; Langen, K.J. Pseudoprogression after glioma therapy: An update. Expert Rev. Neurother. 2017, 17, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Young, R.J.; Gupta, A.; Shah, A.D.; Graber, J.J.; Zhang, Z.; Shi, W.; Holodny, A.I.; Omuro, A.M. Potential utility of conventional MRI signs in diagnosing pseudoprogression in glioblastoma. Neurology 2011, 76, 1918–1924. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Langen, K.J.; Albert, N.L.; Chamberlain, M.; Soffietti, R.; Kim, M.M.; Law, I.; Le Rhun, E.; Chang, S.; Schwarting, J.; et al. PET imaging in patients with brain metastasis-report of the RANO/PET group. Neuro Oncol. 2019, 21, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Wen, P.Y.; Macdonald, D.R.; Reardon, D.A.; Cloughesy, T.F.; Sorensen, A.G.; Galanis, E.; Degroot, J.; Wick, W.; Gilbert, M.R.; Lassman, A.B.; et al. Updated response assessment criteria for high-grade gliomas: Response assessment in neuro-oncology working group. J. Clin. Oncol. 2010, 28, 1963–1972. [Google Scholar] [CrossRef]
- Reardon, D.A.; Weller, M. Pseudoprogression: Fact or wishful thinking in neuro-oncology? Lancet Oncol. 2018, 19, 1561–1563. [Google Scholar] [CrossRef]
- Nandu, H.; Wen, P.Y.; Huang, R.Y. Imaging in neuro-oncology. Ther. Adv. Neurol. Disord. 2018, 11. [Google Scholar] [CrossRef]
- Albert, N.L.; Weller, M.; Suchorska, B.; Galldiks, N.; Soffietti, R.; Kim, M.M.; la Fougere, C.; Pope, W.; Law, I.; Arbizu, J.; et al. Response Assessment in Neuro-Oncology working group and European Association for Neuro-Oncology recommendations for the clinical use of PET imaging in gliomas. Neuro Oncol. 2016, 18, 1199–1208. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Ni, D.; Ehlerding, E.B.; Luo, Q.Y.; Cai, W. PET Imaging of Receptor Tyrosine Kinases in Cancer. Mol. Cancer Ther. 2018, 17, 1625–1636. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Jiang, D.; Ehlerding, E.B.; Luo, Q.; Cai, W. Noninvasive PET Imaging of T cells. Trends Cancer 2018, 4, 359–373. [Google Scholar] [CrossRef] [PubMed]
- Chernov, M.; Hayashi, M.; Izawa, M.; Ochiai, T.; Usukura, M.; Abe, K.; Ono, Y.; Muragaki, Y.; Kubo, O.; Hori, T.; et al. Differentiation of the radiation-induced necrosis and tumor recurrence after gamma knife radiosurgery for brain metastases: Importance of multi-voxel proton MRS. Min-Minim. Invasive Neurosurg. 2005, 48, 228–234. [Google Scholar] [CrossRef]
- Hatzoglou, V.; Yang, T.J.; Omuro, A.; Gavrilovic, I.; Ulaner, G.; Rubel, J.; Schneider, T.; Woo, K.M.; Zhang, Z.; Peck, K.K.; et al. A prospective trial of dynamic contrast-enhanced MRI perfusion and fluorine-18 FDG PET-CT in differentiating brain tumor progression from radiation injury after cranial irradiation. Neuro Oncol. 2016, 18, 873–880. [Google Scholar] [CrossRef]
- Tomura, N.; Kokubun, M.; Saginoya, T.; Mizuno, Y.; Kikuchi, Y. Differentiation between Treatment-Induced Necrosis and Recurrent Tumors in Patients with Metastatic Brain Tumors: Comparison among (11)C-Methionine-PET, FDG-PET, MR Permeability Imaging, and MRI-ADC-Preliminary Results. AJNR Am. J. Neuroradiol. 2017, 38, 1520–1527. [Google Scholar] [CrossRef]
- Palmedo, H.; Urbach, H.; Bender, H.; Schlegel, U.; Schmidt-Wolf, I.G.; Matthies, A.; Linnebank, M.; Joe, A.; Bucerius, J.; Biersack, H.J.; et al. FDG-PET in immunocompetent patients with primary central nervous system lymphoma: Correlation with MRI and clinical follow-up. Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 164–168. [Google Scholar] [CrossRef]
- Birsen, R.; Blanc, E.; Willems, L.; Burroni, B.; Legoff, M.; Le Ray, E.; Pilorge, S.; Salah, S.; Quentin, A.; Deau, B.; et al. Prognostic value of early 18F-FDG PET scanning evaluation in immunocompetent primary CNS lymphoma patients. Oncotarget 2018, 9, 16822–16831. [Google Scholar] [CrossRef]
- Chiavazza, C.; Pellerino, A.; Ferrio, F.; Cistaro, A.; Soffietti, R.; Ruda, R. Primary CNS Lymphomas: Challenges in Diagnosis and Monitoring. Biomed. Res. Int. 2018, 2018, 3606970. [Google Scholar] [CrossRef]
- Herholz, K.; Langen, K.J.; Schiepers, C.; Mountz, J.M. Brain tumors. Semin. Nucl. Med. 2012, 42, 356–370. [Google Scholar] [CrossRef]
- Langen, K.J.; Watts, C. Neuro-oncology: Amino acid PET for brain tumours—Ready for the clinic? Nat. Rev. Neurol. 2016, 12, 375–376. [Google Scholar] [CrossRef] [PubMed]
- Law, I.; Albert, N.L.; Arbizu, J.; Boellaard, R.; Drzezga, A.; Galldiks, N.; la Fougere, C.; Langen, K.J.; Lopci, E.; Lowe, V.; et al. Joint EANM/EANO/RANO practice guidelines/SNMMI procedure standards for imaging of gliomas using PET with radiolabelled amino acids and [(18)F]FDG: Version 1.0. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 540–557. [Google Scholar] [CrossRef] [PubMed]
- Drake, L.R.; Hillmer, A.T.; Cai, Z. Approaches to PET Imaging of Glioblastoma. Molecules 2020, 25, 568. [Google Scholar] [CrossRef]
- Youland, R.S.; Kitange, G.J.; Peterson, T.E.; Pafundi, D.H.; Ramiscal, J.A.; Pokorny, J.L.; Giannini, C.; Laack, N.N.; Parney, I.F.; Lowe, V.J.; et al. The role of LAT1 in (18)F-DOPA uptake in malignant gliomas. J. Neurooncol. 2013, 111, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Papin-Michault, C.; Bonnetaud, C.; Dufour, M.; Almairac, F.; Coutts, M.; Patouraux, S.; Virolle, T.; Darcourt, J.; Burel-Vandenbos, F. Study of LAT1 Expression in Brain Metastases: Towards a Better Understanding of the Results of Positron Emission Tomography Using Amino Acid Tracers. PLoS ONE 2016, 11, e0157139. [Google Scholar] [CrossRef] [PubMed]
- Wiriyasermkul, P.; Nagamori, S.; Tominaga, H.; Oriuchi, N.; Kaira, K.; Nakao, H.; Kitashoji, T.; Ohgaki, R.; Tanaka, H.; Endou, H.; et al. Transport of 3-fluoro-L-alpha-methyl-tyrosine by tumor-upregulated L-type amino acid transporter 1: A cause of the tumor uptake in PET. J. Nucl. Med. 2012, 53, 1253–1261. [Google Scholar] [CrossRef]
- Okubo, S.; Zhen, H.N.; Kawai, N.; Nishiyama, Y.; Haba, R.; Tamiya, T. Correlation of L-methyl-11C-methionine (MET) uptake with L-type amino acid transporter 1 in human gliomas. J. Neurooncol. 2010, 99, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Haining, Z.; Kawai, N.; Miyake, K.; Okada, M.; Okubo, S.; Zhang, X.; Fei, Z.; Tamiya, T. Relation of LAT1/4F2hc expression with pathological grade, proliferation and angiogenesis in human gliomas. BMC Clin. Pathol. 2012, 12, 4. [Google Scholar] [CrossRef]
- Kracht, L.W.; Friese, M.; Herholz, K.; Schroeder, R.; Bauer, B.; Jacobs, A.; Heiss, W.D. Methyl-[11C]- l-methionine uptake as measured by positron emission tomography correlates to microvessel density in patients with glioma. Eur. J. Nucl. Med. Mol. Imaging 2003, 30, 868–873. [Google Scholar] [CrossRef]
- Langen, K.J.; Hamacher, K.; Weckesser, M.; Floeth, F.; Stoffels, G.; Bauer, D.; Coenen, H.H.; Pauleit, D. O-(2-[18F]fluoroethyl)-L-tyrosine: Uptake mechanisms and clinical applications. Nucl. Med. Biol. 2006, 33, 287–294. [Google Scholar] [CrossRef]
- Galldiks, N.; Law, I.; Pope, W.B.; Arbizu, J.; Langen, K.J. The use of amino acid PET and conventional MRI for monitoring of brain tumor therapy. Neuroimage Clin. 2017, 13, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Cicone, F.; Filss, C.P.; Minniti, G.; Rossi-Espagnet, C.; Papa, A.; Scaringi, C.; Galldiks, N.; Bozzao, A.; Shah, N.J.; Scopinaro, F.; et al. Volumetric assessment of recurrent or progressive gliomas: Comparison between F-DOPA PET and perfusion-weighted MRI. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 905–915. [Google Scholar] [CrossRef]
- Juhasz, C.; Dwivedi, S.; Kamson, D.O.; Michelhaugh, S.K.; Mittal, S. Comparison of amino acid positron emission tomographic radiotracers for molecular imaging of primary and metastatic brain tumors. Mol. Imaging 2014, 13. [Google Scholar] [CrossRef]
- Uyttenhove, C.; Pilotte, L.; Theate, I.; Stroobant, V.; Colau, D.; Parmentier, N.; Boon, T.; Van den Eynde, B.J. Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nat. Med. 2003, 9, 1269–1274. [Google Scholar] [CrossRef] [PubMed]
- Batista, C.E.; Juhasz, C.; Muzik, O.; Kupsky, W.J.; Barger, G.; Chugani, H.T.; Mittal, S.; Sood, S.; Chakraborty, P.K.; Chugani, D.C. Imaging correlates of differential expression of indoleamine 2,3-dioxygenase in human brain tumors. Mol. Imaging Biol. 2009, 11, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, G.; Langen, K.J.; Galldiks, N.; McConathy, J. Investigational PET tracers for high-grade gliomas. Q. J. Nucl. Med. Mol. Imaging 2018, 62, 281–294. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, B.C.; Bode, B.P. Amino acid transporters ASCT2 and LAT1 in cancer: Partners in crime? Semin. Cancer Biol. 2005, 15, 254–266. [Google Scholar] [CrossRef] [PubMed]
- Scalise, M.; Pochini, L.; Console, L.; Losso, M.A.; Indiveri, C. The Human SLC1A5 (ASCT2) Amino Acid Transporter: From Function to Structure and Role in Cell Biology. Front. Cell Dev. Biol. 2018, 6, 96. [Google Scholar] [CrossRef]
- Schuster, D.M.; Nanni, C.; Fanti, S.; Oka, S.; Okudaira, H.; Inoue, Y.; Sorensen, J.; Owenius, R.; Choyke, P.; Turkbey, B.; et al. Anti-1-amino-3-18F-fluorocyclobutane-1-carboxylic acid: Physiologic uptake patterns, incidental findings, and variants that may simulate disease. J. Nucl. Med. 2014, 55, 1986–1992. [Google Scholar] [CrossRef]
- Hayes, A.R.; Jayamanne, D.; Hsiao, E.; Schembri, G.P.; Bailey, D.L.; Roach, P.J.; Khasraw, M.; Newey, A.; Wheeler, H.R.; Back, M. Utilizing 18F-fluoroethyltyrosine (FET) positron emission tomography (PET) to define suspected nonenhancing tumor for radiation therapy planning of glioblastoma. Pract. Radiat. Oncol. 2018, 8, 230–238. [Google Scholar] [CrossRef]
- Kracht, L.W.; Miletic, H.; Busch, S.; Jacobs, A.H.; Voges, J.; Hoevels, M.; Klein, J.C.; Herholz, K.; Heiss, W.D. Delineation of brain tumor extent with [11C]L-methionine positron emission tomography: Local comparison with stereotactic histopathology. Clin. Cancer Res. 2004, 10, 7163–7170. [Google Scholar] [CrossRef] [PubMed]
- Lopez, W.O.; Cordeiro, J.G.; Albicker, U.; Doostkam, S.; Nikkhah, G.; Kirch, R.D.; Trippel, M.; Reithmeier, T. Correlation of (18)F-fluoroethyl tyrosine positron-emission tomography uptake values and histomorphological findings by stereotactic serial biopsy in newly diagnosed brain tumors using a refined software tool. Onco Targets Ther. 2015, 8, 3803–3815. [Google Scholar] [CrossRef] [PubMed]
- Pauleit, D.; Floeth, F.; Hamacher, K.; Riemenschneider, M.J.; Reifenberger, G.; Muller, H.W.; Zilles, K.; Coenen, H.H.; Langen, K.J. O-(2-[18F]fluoroethyl)-L-tyrosine PET combined with MRI improves the diagnostic assessment of cerebral gliomas. Brain 2005, 128, 678–687. [Google Scholar] [CrossRef] [PubMed]
- Bogsrud, T.V.; Londalen, A.; Brandal, P.; Leske, H.; Panagopoulos, I.; Borghammer, P.; Bach-Gansmo, T. 18F-Fluciclovine PET/CT in Suspected Residual or Recurrent High-Grade Glioma. Clin. Nucl. Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Michaud, L.; Beattie, B.J.; Akhurst, T.; Dunphy, M.; Zanzonico, P.; Finn, R.; Mauguen, A.; Schoder, H.; Weber, W.A.; Lassman, A.B.; et al. (18)F-Fluciclovine ((18)F-FACBC) PET imaging of recurrent brain tumors. Eur. J. Nucl. Med. Mol. Imaging 2019. [Google Scholar] [CrossRef] [PubMed]
- Tsuyuguchi, N.; Terakawa, Y.; Uda, T.; Nakajo, K.; Kanemura, Y. Diagnosis of Brain Tumors Using Amino Acid Transport PET Imaging with (18)F-fluciclovine: A Comparative Study with L-methyl-(11)C-methionine PET Imaging. Asia Ocean. J. Nucl. Med. Biol. 2017, 5, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Ullrich, R.; Schroeter, M.; Fink, G.R.; Jacobs, A.H.; Kracht, L.W. Volumetry of [(11)C]-methionine PET uptake and MRI contrast enhancement in patients with recurrent glioblastoma multiforme. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 84–92. [Google Scholar] [CrossRef]
- Grosu, A.L.; Astner, S.T.; Riedel, E.; Nieder, C.; Wiedenmann, N.; Heinemann, F.; Schwaiger, M.; Molls, M.; Wester, H.J.; Weber, W.A. An interindividual comparison of O-(2-[18F]fluoroethyl)-L-tyrosine (FET)- and L-[methyl-11C]methionine (MET)-PET in patients with brain gliomas and metastases. Int. J. Radiat. Oncol. Biol. Phys. 2011, 81, 1049–1058. [Google Scholar] [CrossRef]
- Becherer, A.; Karanikas, G.; Szabo, M.; Zettinig, G.; Asenbaum, S.; Marosi, C.; Henk, C.; Wunderbaldinger, P.; Czech, T.; Wadsak, W.; et al. Brain tumour imaging with PET: A comparison between [18F]fluorodopa and [11C]methionine. Eur. J. Nucl. Med. Mol. Imaging 2003, 30, 1561–1567. [Google Scholar] [CrossRef]
- Kratochwil, C.; Combs, S.E.; Leotta, K.; Afshar-Oromieh, A.; Rieken, S.; Debus, J.; Haberkorn, U.; Giesel, F.L. Intra-individual comparison of (1)(8)F-FET and (1)(8)F-DOPA in PET imaging of recurrent brain tumors. Neuro Oncol. 2014, 16, 434–440. [Google Scholar] [CrossRef]
- Lapa, C.; Linsenmann, T.; Monoranu, C.M.; Samnick, S.; Buck, A.K.; Bluemel, C.; Czernin, J.; Kessler, A.F.; Homola, G.A.; Ernestus, R.I.; et al. Comparison of the amino acid tracers 18F-FET and 18F-DOPA in high-grade glioma patients. J. Nucl. Med. 2014, 55, 1611–1616. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Ohgaki, H.; Wiestler, O.D.; Cavenee, W.K.; Burger, P.C.; Jouvet, A.; Scheithauer, B.W.; Kleihues, P. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007, 114, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Pichler, R.; Dunzinger, A.; Wurm, G.; Pichler, J.; Weis, S.; Nussbaumer, K.; Topakian, R.; Aigner, R.M. Is there a place for FET PET in the initial evaluation of brain lesions with unknown significance? Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 1521–1528. [Google Scholar] [CrossRef] [PubMed]
- Hutterer, M.; Nowosielski, M.; Putzer, D.; Jansen, N.L.; Seiz, M.; Schocke, M.; McCoy, M.; Gobel, G.; la Fougere, C.; Virgolini, I.J.; et al. [18F]-fluoro-ethyl-L-tyrosine PET: A valuable diagnostic tool in neuro-oncology, but not all that glitters is glioma. Neuro Oncol. 2013, 15, 341–351. [Google Scholar] [CrossRef]
- Jansen, N.L.; Graute, V.; Armbruster, L.; Suchorska, B.; Lutz, J.; Eigenbrod, S.; Cumming, P.; Bartenstein, P.; Tonn, J.C.; Kreth, F.W.; et al. MRI-suspected low-grade glioma: Is there a need to perform dynamic FET PET? Eur. J. Nucl. Med. Mol. Imaging 2012, 39, 1021–1029. [Google Scholar] [CrossRef]
- Werner, J.M.; Stoffels, G.; Lichtenstein, T.; Borggrefe, J.; Lohmann, P.; Ceccon, G.; Shah, N.J.; Fink, G.R.; Langen, K.J.; Kabbasch, C.; et al. Differentiation of treatment-related changes from tumour progression: A direct comparison between dynamic FET PET and ADC values obtained from DWI MRI. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1889–1901. [Google Scholar] [CrossRef]
- Kebir, S.; Fimmers, R.; Galldiks, N.; Schafer, N.; Mack, F.; Schaub, C.; Stuplich, M.; Niessen, M.; Tzaridis, T.; Simon, M.; et al. Late Pseudoprogression in Glioblastoma: Diagnostic Value of Dynamic O-(2-[18F]fluoroethyl)-L-Tyrosine PET. Clin. Cancer Res. 2016, 22, 2190–2196. [Google Scholar] [CrossRef]
- Popperl, G.; Gotz, C.; Rachinger, W.; Gildehaus, F.J.; Tonn, J.C.; Tatsch, K. Value of O-(2-[18F]fluoroethyl)- L-tyrosine PET for the diagnosis of recurrent glioma. Eur. J. Nucl. Med. Mol. Imaging 2004, 31, 1464–1470. [Google Scholar] [CrossRef]
- Rachinger, W.; Goetz, C.; Popperl, G.; Gildehaus, F.J.; Kreth, F.W.; Holtmannspotter, M.; Herms, J.; Koch, W.; Tatsch, K.; Tonn, J.C. Positron emission tomography with O-(2-[18F]fluoroethyl)-l-tyrosine versus magnetic resonance imaging in the diagnosis of recurrent gliomas. Neurosurgery 2005, 57, 505–511. [Google Scholar] [CrossRef]
- Mihovilovic, M.I.; Kertels, O.; Hanscheid, H.; Lohr, M.; Monoranu, C.M.; Kleinlein, I.; Samnick, S.; Kessler, A.F.; Linsenmann, T.; Ernestus, R.I.; et al. O-(2-((18)F)fluoroethyl)-L-tyrosine PET for the differentiation of tumour recurrence from late pseudoprogression in glioblastoma. J. Neurol. Neurosurg. Psychiatry 2019, 90, 238–239. [Google Scholar] [CrossRef] [PubMed]
- Mehrkens, J.H.; Popperl, G.; Rachinger, W.; Herms, J.; Seelos, K.; Tatsch, K.; Tonn, J.C.; Kreth, F.W. The positive predictive value of O-(2-[18F]fluoroethyl)-L-tyrosine (FET) PET in the diagnosis of a glioma recurrence after multimodal treatment. J. Neurooncol. 2008, 88, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Jena, A.; Taneja, S.; Gambhir, A.; Mishra, A.K.; D’Souza, M.M.; Verma, S.M.; Hazari, P.P.; Negi, P.; Jhadav, G.K.; Sogani, S.K. Glioma Recurrence Versus Radiation Necrosis: Single-Session Multiparametric Approach Using Simultaneous O-(2-18F-Fluoroethyl)-L-Tyrosine PET/MRI. Clin. Nucl. Med. 2016, 41, e228–e236. [Google Scholar] [CrossRef] [PubMed]
- Pyka, T.; Hiob, D.; Preibisch, C.; Gempt, J.; Wiestler, B.; Schlegel, J.; Straube, C.; Zimmer, C. Diagnosis of glioma recurrence using multiparametric dynamic 18F-fluoroethyl-tyrosine PET-MRI. Eur. J. Radiol. 2018, 103, 32–37. [Google Scholar] [CrossRef]
- Galldiks, N.; Stoffels, G.; Filss, C.; Rapp, M.; Blau, T.; Tscherpel, C.; Ceccon, G.; Dunkl, V.; Weinzierl, M.; Stoffel, M.; et al. The use of dynamic O-(2-18F-fluoroethyl)-l-tyrosine PET in the diagnosis of patients with progressive and recurrent glioma. Neuro Oncol. 2015, 17, 1293–1300. [Google Scholar] [CrossRef]
- Ceccon, G.; Lohmann, P.; Stoffels, G.; Judov, N.; Filss, C.P.; Rapp, M.; Bauer, E.; Hamisch, C.; Ruge, M.I.; Kocher, M.; et al. Dynamic O-(2-18F-fluoroethyl)-L-tyrosine positron emission tomography differentiates brain metastasis recurrence from radiation injury after radiotherapy. Neuro Oncol. 2017, 19, 281–288. [Google Scholar] [CrossRef]
- Tsuyuguchi, N.; Sunada, I.; Iwai, Y.; Yamanaka, K.; Tanaka, K.; Takami, T.; Otsuka, Y.; Sakamoto, S.; Ohata, K.; Goto, T.; et al. Methionine positron emission tomography of recurrent metastatic brain tumor and radiation necrosis after stereotactic radiosurgery: Is a differential diagnosis possible? J. Neurosurg. 2003, 98, 1056–1064. [Google Scholar] [CrossRef]
- Galldiks, N.; Stoffels, G.; Filss, C.P.; Piroth, M.D.; Sabel, M.; Ruge, M.I.; Herzog, H.; Shah, N.J.; Fink, G.R.; Coenen, H.H.; et al. Role of O-(2-(18)F-fluoroethyl)-L-tyrosine PET for differentiation of local recurrent brain metastasis from radiation necrosis. J. Nucl. Med. 2012, 53, 1367–1374. [Google Scholar] [CrossRef]
- Lizarraga, K.J.; Allen-Auerbach, M.; Czernin, J.; DeSalles, A.A.; Yong, W.H.; Phelps, M.E.; Chen, W. (18)F-FDOPA PET for differentiating recurrent or progressive brain metastatic tumors from late or delayed radiation injury after radiation treatment. J. Nucl. Med. 2014, 55, 30–36. [Google Scholar] [CrossRef]
- Cicone, F.; Minniti, G.; Romano, A.; Papa, A.; Scaringi, C.; Tavanti, F.; Bozzao, A.; Maurizi Enrici, R.; Scopinaro, F. Accuracy of F-DOPA PET and perfusion-MRI for differentiating radionecrotic from progressive brain metastases after radiosurgery. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 103–111. [Google Scholar] [CrossRef]
- Terakawa, Y.; Tsuyuguchi, N.; Iwai, Y.; Yamanaka, K.; Higashiyama, S.; Takami, T.; Ohata, K. Diagnostic accuracy of 11C-methionine PET for differentiation of recurrent brain tumors from radiation necrosis after radiotherapy. J. Nucl. Med. 2008, 49, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Nihashi, T.; Dahabreh, I.J.; Terasawa, T. Diagnostic accuracy of PET for recurrent glioma diagnosis: A meta-analysis. AJNR Am. J. Neuroradiol. 2013, 34, 944–950. [Google Scholar] [CrossRef] [PubMed]
- Minamimoto, R.; Saginoya, T.; Kondo, C.; Tomura, N.; Ito, K.; Matsuo, Y.; Matsunaga, S.; Shuto, T.; Akabane, A.; Miyata, Y.; et al. Differentiation of Brain Tumor Recurrence from Post-Radiotherapy Necrosis with 11C-Methionine PET: Visual Assessment versus Quantitative Assessment. PLoS ONE 2015, 10, e0132515. [Google Scholar] [CrossRef]
- Salber, D.; Stoffels, G.; Pauleit, D.; Oros-Peusquens, A.M.; Shah, N.J.; Klauth, P.; Hamacher, K.; Coenen, H.H.; Langen, K.J. Differential uptake of O-(2-18F-fluoroethyl)-L-tyrosine, L-3H-methionine, and 3H-deoxyglucose in brain abscesses. J. Nucl. Med. 2007, 48, 2056–2062. [Google Scholar] [CrossRef] [PubMed]
- Alkonyi, B.; Barger, G.R.; Mittal, S.; Muzik, O.; Chugani, D.C.; Bahl, G.; Robinette, N.L.; Kupsky, W.J.; Chakraborty, P.K.; Juhasz, C. Accurate differentiation of recurrent gliomas from radiation injury by kinetic analysis of alpha-11C-methyl-L-tryptophan PET. J. Nucl. Med. 2012, 53, 1058–1064. [Google Scholar] [CrossRef] [PubMed]
- Henderson, F.; Brem, S.; O’Rourke, D.M.; Nasrallah, M.; Buch, V.P.; Young, A.J.; Doot, R.K.; Pantel, A.; Desai, A.; Bagley, S.J.; et al. 18F-Fluciclovine PET to distinguish treatment-related effects from disease progression in recurrent glioblastoma: PET fusion with MRI guides neurosurgical sampling. Neurooncol. Pract. 2019. [Google Scholar] [CrossRef]
- Long, G.V.; Atkinson, V.; Lo, S.; Sandhu, S.; Guminski, A.D.; Brown, M.P.; Wilmott, J.S.; Edwards, J.; Gonzalez, M.; Scolyer, R.A.; et al. Combination nivolumab and ipilimumab or nivolumab alone in melanoma brain metastases: A multicentre randomised phase 2 study. Lancet Oncol. 2018, 19, 672–681. [Google Scholar] [CrossRef]
- Galldiks, N.; Abdulla, D.S.Y.; Scheffler, M.; Schweinsberg, V.; Schlaak, M.; Kreuzberg, N.; Landsberg, J.; Lohmann, P.; Ceccon, G.; Werner, J.M.; et al. Treatment monitoring of immunotherapy and targeted therapy using FET PET in patients with melanoma and lung cancer brain metastases: Initial experiences. J. Clin. Oncol. 2019, 37, e13525. [Google Scholar] [CrossRef]
- Kristin Schmitz, A.; Sorg, R.V.; Stoffels, G.; Grauer, O.M.; Galldiks, N.; Steiger, H.J.; Kamp, M.A.; Langen, K.J.; Sabel, M.; Rapp, M. Diagnostic impact of additional O-(2-[18F]fluoroethyl)-L-tyrosine ((18)F-FET) PET following immunotherapy with dendritic cell vaccination in glioblastoma patients. Br. J. Neurosurg. 2019. [Google Scholar] [CrossRef]
- Galldiks, N.; Werner, J.M.; Tscherpel, C.; Fink, G.R.; Langen, K.J. Imaging findings following regorafenib in malignant gliomas: FET PET adds valuable information to anatomical MRI. Neurooncol. Adv. 2019, 1. [Google Scholar] [CrossRef]
- Galldiks, N.; Kracht, L.W.; Burghaus, L.; Ullrich, R.T.; Backes, H.; Brunn, A.; Heiss, W.D.; Jacobs, A.H. Patient-tailored, imaging-guided, long-term temozolomide chemotherapy in patients with glioblastoma. Mol. Imaging 2010, 9, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Kracht, L.W.; Burghaus, L.; Thomas, A.; Jacobs, A.H.; Heiss, W.D.; Herholz, K. Use of 11C-methionine PET to monitor the effects of temozolomide chemotherapy in malignant gliomas. Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Herholz, K.; Kracht, L.W.; Heiss, W.D. Monitoring the effect of chemotherapy in a mixed glioma by C-11-methionine PET. J. Neuroimaging 2003, 13, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Langen, K.J.; Holy, R.; Pinkawa, M.; Stoffels, G.; Nolte, K.W.; Kaiser, H.J.; Filss, C.P.; Fink, G.R.; Coenen, H.H.; et al. Assessment of treatment response in patients with glioblastoma using O-(2-18F-fluoroethyl)-L-tyrosine PET in comparison to MRI. J. Nucl. Med. 2012, 53, 1048–1057. [Google Scholar] [CrossRef] [PubMed]
- Piroth, M.D.; Pinkawa, M.; Holy, R.; Klotz, J.; Nussen, S.; Stoffels, G.; Coenen, H.H.; Kaiser, H.J.; Langen, K.J.; Eble, M.J. Prognostic value of early [18F]fluoroethyltyrosine positron emission tomography after radiochemotherapy in glioblastoma multiforme. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Wyss, M.; Hofer, S.; Bruehlmeier, M.; Hefti, M.; Uhlmann, C.; Bartschi, E.; Buettner, U.W.; Roelcke, U. Early metabolic responses in temozolomide treated low-grade glioma patients. J. Neurooncol. 2009, 95, 87–93. [Google Scholar] [CrossRef]
- Roelcke, U.; Wyss, M.T.; Nowosielski, M.; Ruda, R.; Roth, P.; Hofer, S.; Galldiks, N.; Crippa, F.; Weller, M.; Soffietti, R. Amino acid positron emission tomography to monitor chemotherapy response and predict seizure control and progression-free survival in WHO grade II gliomas. Neuro Oncol. 2016, 18, 744–751. [Google Scholar] [CrossRef]
- Suchorska, B.; Unterrainer, M.; Biczok, A.; Sosnova, M.; Forbrig, R.; Bartenstein, P.; Tonn, J.C.; Albert, N.L.; Kreth, F.W. (18)F-FET-PET as a biomarker for therapy response in non-contrast enhancing glioma following chemotherapy. J. Neurooncol. 2018, 139, 721–730. [Google Scholar] [CrossRef]
- Galldiks, N.; Rapp, M.; Stoffels, G.; Dunkl, V.; Sabel, M.; Langen, K.J. Earlier diagnosis of progressive disease during bevacizumab treatment using O-(2-18F-fluorethyl)-L-tyrosine positron emission tomography in comparison with magnetic resonance imaging. Mol. Imaging 2013, 12, 273–276. [Google Scholar] [CrossRef]
- Morana, G.; Piccardo, A.; Garre, M.L.; Nozza, P.; Consales, A.; Rossi, A. Multimodal magnetic resonance imaging and 18F-L-dihydroxyphenylalanine positron emission tomography in early characterization of pseudoresponse and nonenhancing tumor progression in a pediatric patient with malignant transformation of ganglioglioma treated with bevacizumab. J. Clin. Oncol. 2013, 31, e1. [Google Scholar] [CrossRef]
- Hutterer, M.; Nowosielski, M.; Putzer, D.; Waitz, D.; Tinkhauser, G.; Kostron, H.; Muigg, A.; Virgolini, I.J.; Staffen, W.; Trinka, E.; et al. O-(2-18F-fluoroethyl)-L-tyrosine PET predicts failure of antiangiogenic treatment in patients with recurrent high-grade glioma. J. Nucl. Med. 2011, 52, 856–864. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Rapp, M.; Stoffels, G.; Fink, G.R.; Shah, N.J.; Coenen, H.H.; Sabel, M.; Langen, K.-J. Response assessment of bevacizumab in patients with recurrent malignant glioma using [18F]Fluoroethyl-l-tyrosine PET in comparison to MRI. Eur. J. Nucl. Med. Mol. Imaging 2012, 40, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Dunkl, V.; Ceccon, G.; Tscherpel, C.; Stoffels, G.; Law, I.; Henriksen, O.M.; Muhic, A.; Poulsen, H.S.; Steger, J.; et al. Early treatment response evaluation using FET PET compared to MRI in glioblastoma patients at first progression treated with bevacizumab plus lomustine. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 2377–2386. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenberg, J.; Czernin, J.; Cloughesy, T.F.; Ellingson, B.M.; Pope, W.B.; Grogan, T.; Elashoff, D.; Geist, C.; Silverman, D.H.; Phelps, M.E.; et al. Treatment response evaluation using 18F-FDOPA PET in patients with recurrent malignant glioma on bevacizumab therapy. Clin. Cancer Res. 2014, 20, 3550–3559. [Google Scholar] [CrossRef]
- Stupp, R.; Taillibert, S.; Kanner, A.; Read, W.; Steinberg, D.; Lhermitte, B.; Toms, S.; Idbaih, A.; Ahluwalia, M.S.; Fink, K.; et al. Effect of Tumor-Treating Fields Plus Maintenance Temozolomide vs Maintenance Temozolomide Alone on Survival in Patients With Glioblastoma: A Randomized Clinical Trial. JAMA 2017, 318, 2306–2316. [Google Scholar] [CrossRef]
- Bosnyak, E.; Barger, G.R.; Michelhaugh, S.K.; Robinette, N.L.; Amit-Yousif, A.; Mittal, S.; Juhasz, C. Amino Acid PET Imaging of the Early Metabolic Response During Tumor-Treating Fields (TTFields) Therapy in Recurrent Glioblastoma. Clin. Nucl. Med. 2018, 43, 176–179. [Google Scholar] [CrossRef]
- Ceccon, G.; Lazaridis, L.; Stoffels, G.; Rapp, M.; Weber, M.; Blau, T.; Lohmann, P.; Kebir, S.; Herrmann, K.; Fink, G.R.; et al. Use of FET PET in glioblastoma patients undergoing neurooncological treatment including tumour-treating fields: Initial experience. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1626–1635. [Google Scholar] [CrossRef]
- Chen, M.K.; Baidoo, K.; Verina, T.; Guilarte, T.R. Peripheral benzodiazepine receptor imaging in CNS demyelination: Functional implications of anatomical and cellular localization. Brain 2004, 127, 1379–1392. [Google Scholar] [CrossRef]
- Papadopoulos, V.; Baraldi, M.; Guilarte, T.R.; Knudsen, T.B.; Lacapere, J.J.; Lindemann, P.; Norenberg, M.D.; Nutt, D.; Weizman, A.; Zhang, M.R.; et al. Translocator protein (18kDa): New nomenclature for the peripheral-type benzodiazepine receptor based on its structure and molecular function. Trends Pharmacol. Sci. 2006, 27, 402–409. [Google Scholar] [CrossRef]
- Schweitzer, P.J.; Fallon, B.A.; Mann, J.J.; Kumar, J.S. PET tracers for the peripheral benzodiazepine receptor and uses thereof. Drug Discov. Today 2010, 15, 933–942. [Google Scholar] [CrossRef]
- Su, Z.; Roncaroli, F.; Durrenberger, P.F.; Coope, D.J.; Karabatsou, K.; Hinz, R.; Thompson, G.; Turkheimer, F.E.; Janczar, K.; Du Plessis, D.; et al. The 18-kDa mitochondrial translocator protein in human gliomas: An 11C-(R)PK11195 PET imaging and neuropathology study. J. Nucl. Med. 2015, 56, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; Herholz, K.; Gerhard, A.; Roncaroli, F.; Du Plessis, D.; Jackson, A.; Turkheimer, F.; Hinz, R. [(1)(1)C]-(R)PK11195 tracer kinetics in the brain of glioma patients and a comparison of two referencing approaches. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 1406–1419. [Google Scholar] [CrossRef] [PubMed]
- Albert, N.L.; Unterrainer, M.; Fleischmann, D.F.; Lindner, S.; Vettermann, F.; Brunegraf, A.; Vomacka, L.; Brendel, M.; Wenter, V.; Wetzel, C.; et al. TSPO PET for glioma imaging using the novel ligand (18)F-GE-180: First results in patients with glioblastoma. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 2230–2238. [Google Scholar] [CrossRef] [PubMed]
- Vomacka, L.; Albert, N.L.; Lindner, S.; Unterrainer, M.; Mahler, C.; Brendel, M.; Ermoschkin, L.; Gosewisch, A.; Brunegraf, A.; Buckley, C.; et al. TSPO imaging using the novel PET ligand [(18)F]GE-180: Quantification approaches in patients with multiple sclerosis. EJNMMI Res. 2017, 7, 89. [Google Scholar] [CrossRef] [PubMed]
- Unterrainer, M.; Mahler, C.; Vomacka, L.; Lindner, S.; Havla, J.; Brendel, M.; Boning, G.; Ertl-Wagner, B.; Kumpfel, T.; Milenkovic, V.M.; et al. TSPO PET with [(18)F]GE-180 sensitively detects focal neuroinflammation in patients with relapsing-remitting multiple sclerosis. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1423–1431. [Google Scholar] [CrossRef] [PubMed]
- Sridharan, S.; Raffel, J.; Nandoskar, A.; Record, C.; Brooks, D.J.; Owen, D.; Sharp, D.; Muraro, P.A.; Gunn, R.; Nicholas, R. Confirmation of Specific Binding of the 18-kDa Translocator Protein (TSPO) Radioligand [(18)F]GE-180: A Blocking Study Using XBD173 in Multiple Sclerosis Normal Appearing White and Grey Matter. Mol. Imaging Biol. 2019, 21, 935–944. [Google Scholar] [CrossRef]
- Unterrainer, M.; Fleischmann, D.F.; Diekmann, C.; Vomacka, L.; Lindner, S.; Vettermann, F.; Brendel, M.; Wenter, V.; Ertl-Wagner, B.; Herms, J.; et al. Comparison of (18)F-GE-180 and dynamic (18)F-FET PET in high grade glioma: A double-tracer pilot study. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 580–590. [Google Scholar] [CrossRef]
- Shields, A.F.; Grierson, J.R.; Dohmen, B.M.; Machulla, H.J.; Stayanoff, J.C.; Lawhorn-Crews, J.M.; Obradovich, J.E.; Muzik, O.; Mangner, T.J. Imaging proliferation in vivo with [F-18]FLT and positron emission tomography. Nat. Med. 1998, 4, 1334–1336. [Google Scholar] [CrossRef]
- van Waarde, A.; Elsinga, P.H. Proliferation markers for the differential diagnosis of tumor and inflammation. Curr. Pharm. Des. 2008, 14, 3326–3339. [Google Scholar] [CrossRef]
- Saga, T.; Kawashima, H.; Araki, N.; Takahashi, J.A.; Nakashima, Y.; Higashi, T.; Oya, N.; Mukai, T.; Hojo, M.; Hashimoto, N.; et al. Evaluation of primary brain tumors with FLT-PET: Usefulness and limitations. Clin. Nucl. Med. 2006, 31, 774–780. [Google Scholar] [CrossRef]
- Jacobs, A.H.; Thomas, A.; Kracht, L.W.; Li, H.; Dittmar, C.; Garlip, G.; Galldiks, N.; Klein, J.C.; Sobesky, J.; Hilker, R.; et al. 18F-fluoro-L-thymidine and 11C-methylmethionine as markers of increased transport and proliferation in brain tumors. J. Nucl. Med. 2005, 46, 1948–1958. [Google Scholar] [PubMed]
- Li, Z.; Yu, Y.; Zhang, H.; Xu, G.; Chen, L. A meta-analysis comparing 18F-FLT PET with 18F-FDG PET for assessment of brain tumor recurrence. Nucl. Med. Commun. 2015, 36, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Delaloye, S.; Silverman, D.H.; Geist, C.; Czernin, J.; Sayre, J.; Satyamurthy, N.; Pope, W.; Lai, A.; Phelps, M.E.; et al. Predicting treatment response of malignant gliomas to bevacizumab and irinotecan by imaging proliferation with [18F] fluorothymidine positron emission tomography: A pilot study. J. Clin. Oncol. 2007, 25, 4714–4721. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenberg, J.; Czernin, J.; Cloughesy, T.F.; Ellingson, B.M.; Pope, W.B.; Geist, C.; Dahlbom, M.; Silverman, D.H.; Satyamurthy, N.; Phelps, M.E.; et al. 3′-deoxy-3′-18F-fluorothymidine PET and MRI for early survival predictions in patients with recurrent malignant glioma treated with bevacizumab. J. Nucl. Med. 2012, 53, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.J.; Cloughesy, T.F.; Pope, W.B.; Nghiemphu, P.L.; Lai, A.; Zaw, T.; Czernin, J.; Phelps, M.E.; Chen, W.; Ellingson, B.M. 18F-FDOPA and 18F-FLT positron emission tomography parametric response maps predict response in recurrent malignant gliomas treated with bevacizumab. Neuro Oncol. 2012, 14, 1079–1089. [Google Scholar] [CrossRef][Green Version]
- Nguyen, N.C.; Yee, M.K.; Tuchayi, A.M.; Kirkwood, J.M.; Tawbi, H.; Mountz, J.M. Targeted Therapy and Immunotherapy Response Assessment with F-18 Fluorothymidine Positron-Emission Tomography/Magnetic Resonance Imaging in Melanoma Brain Metastasis: A Pilot Study. Front. Oncol. 2018, 8, 18. [Google Scholar] [CrossRef]
- Bell, C.; Dowson, N.; Fay, M.; Thomas, P.; Puttick, S.; Gal, Y.; Rose, S. Hypoxia imaging in gliomas with 18F-fluoromisonidazole PET: Toward clinical translation. Semin. Nucl. Med. 2015, 45, 136–150. [Google Scholar] [CrossRef]
- Rasey, J.S.; Koh, W.J.; Evans, M.L.; Peterson, L.M.; Lewellen, T.K.; Graham, M.M.; Krohn, K.A. Quantifying regional hypoxia in human tumors with positron emission tomography of [18F]fluoromisonidazole: A pretherapy study of 37 patients. Int. J. Radiat. Oncol. Biol. Phys. 1996, 36, 417–428. [Google Scholar] [CrossRef]
- Rajendran, J.G.; Krohn, K.A. F-18 fluoromisonidazole for imaging tumor hypoxia: Imaging the microenvironment for personalized cancer therapy. Semin. Nucl. Med. 2015, 45, 151–162. [Google Scholar] [CrossRef]
- Swanson, K.R.; Chakraborty, G.; Wang, C.H.; Rockne, R.; Harpold, H.L.; Muzi, M.; Adamsen, T.C.; Krohn, K.A.; Spence, A.M. Complementary but distinct roles for MRI and 18F-fluoromisonidazole PET in the assessment of human glioblastomas. J. Nucl. Med. 2009, 50, 36–44. [Google Scholar] [CrossRef]
- Kawai, N.; Maeda, Y.; Kudomi, N.; Miyake, K.; Okada, M.; Yamamoto, Y.; Nishiyama, Y.; Tamiya, T. Correlation of biological aggressiveness assessed by 11C-methionine PET and hypoxic burden assessed by 18F-fluoromisonidazole PET in newly diagnosed glioblastoma. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Barajas, R.F.; Krohn, K.A.; Link, J.M.; Hawkins, R.A.; Clarke, J.L.; Pampaloni, M.H.; Cha, S. Glioma FMISO PET/MR Imaging Concurrent with Antiangiogenic Therapy: Molecular Imaging as a Clinical Tool in the Burgeoning Era of Personalized Medicine. Biomedicines 2016, 4, 24. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, S.; Hirata, K.; Toyonaga, T.; Kobayashi, K.; Ishi, Y.; Motegi, H.; Kobayashi, H.; Shiga, T.; Tamaki, N.; Terasaka, S.; et al. Change in 18F-Fluoromisonidazole PET Is an Early Predictor of the Prognosis in the Patients with Recurrent High-Grade Glioma Receiving Bevacizumab Treatment. PLoS ONE 2016, 11, e0167917. [Google Scholar] [CrossRef] [PubMed]
- Piert, M.; Machulla, H.J.; Picchio, M.; Reischl, G.; Ziegler, S.; Kumar, P.; Wester, H.J.; Beck, R.; McEwan, A.J.; Wiebe, L.I.; et al. Hypoxia-specific tumor imaging with 18F-fluoroazomycin arabinoside. J. Nucl. Med. 2005, 46, 106–113. [Google Scholar]
- Postema, E.J.; McEwan, A.J.; Riauka, T.A.; Kumar, P.; Richmond, D.A.; Abrams, D.N.; Wiebe, L.I. Initial results of hypoxia imaging using 1-alpha-D: -(5-deoxy-5-[18F]-fluoroarabinofuranosyl)-2-nitroimidazole (18F-FAZA). Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 1565–1573. [Google Scholar] [CrossRef]
- Mapelli, P.; Zerbetto, F.; Incerti, E.; Conte, G.M.; Bettinardi, V.; Fallanca, F.; Anzalone, N.; Di Muzio, N.; Gianolli, L.; Picchio, M. 18F-FAZA PET/CT Hypoxia Imaging of High-Grade Glioma Before and After Radiotherapy. Clin. Nucl. Med. 2017, 42, e525–e526. [Google Scholar] [CrossRef]
- Ter-Pogossian, M.M.; Eichling, J.O.; Davis, D.O.; Welch, M.J. The measure in vivo of regional cerebral oxygen utilization by means of oxyhemoglobin labeled with radioactive oxygen-15. J. Clin. Investig. 1970, 49, 381–391. [Google Scholar] [CrossRef]
- Ludemann, L.; Warmuth, C.; Plotkin, M.; Forschler, A.; Gutberlet, M.; Wust, P.; Amthauer, H. Brain tumor perfusion: Comparison of dynamic contrast enhanced magnetic resonance imaging using T1, T2, and T2* contrast, pulsed arterial spin labeling, and H2(15)O positron emission tomography. Eur. J. Radiol. 2009, 70, 465–474. [Google Scholar] [CrossRef]
- Gruner, J.M.; Paamand, R.; Kosteljanetz, M.; Broholm, H.; Hojgaard, L.; Law, I. Brain perfusion CT compared with (1)(5)O-H(2)O PET in patients with primary brain tumours. Eur. J. Nucl. Med. Mol. Imaging 2012, 39, 1691–1701. [Google Scholar] [CrossRef]
- Ogawa, T.; Uemura, K.; Shishido, F.; Yamaguchi, T.; Murakami, M.; Inugami, A.; Kanno, I.; Sasaki, H.; Kato, T.; Hirata, K.; et al. Changes of cerebral blood flow, and oxygen and glucose metabolism following radiochemotherapy of gliomas: A PET study. J. Comput. Assist. Tomogr. 1988, 12, 290–297. [Google Scholar] [CrossRef]
- Mineura, K.; Yasuda, T.; Kowada, M.; Ogawa, T.; Shishido, F.; Uemura, K. Positron emission tomographic evaluation of radiochemotherapeutic effect on regional cerebral hemocirculation and metabolism in patients with gliomas. J. Neurooncol. 1987, 5, 277–285. [Google Scholar] [CrossRef]
- Jain, R.K.; di Tomaso, E.; Duda, D.G.; Loeffler, J.S.; Sorensen, A.G.; Batchelor, T.T. Angiogenesis in brain tumours. Nat. Rev. Neurosci. 2007, 8, 610–622. [Google Scholar] [CrossRef] [PubMed]
- Levin, V.A.; Bidaut, L.; Hou, P.; Kumar, A.J.; Wefel, J.S.; Bekele, B.N.; Grewal, J.; Prabhu, S.; Loghin, M.; Gilbert, M.R.; et al. Randomized double-blind placebo-controlled trial of bevacizumab therapy for radiation necrosis of the central nervous system. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 1487–1495. [Google Scholar] [CrossRef] [PubMed]
- Jansen, M.H.; Veldhuijzen van Zanten, S.E.M.; van Vuurden, D.G.; Huisman, M.C.; Vugts, D.J.; Hoekstra, O.S.; van Dongen, G.A.; Kaspers, G.L. Molecular Drug Imaging: (89)Zr-Bevacizumab PET in Children with Diffuse Intrinsic Pontine Glioma. J. Nucl. Med. 2017, 58, 711–716. [Google Scholar] [CrossRef] [PubMed]
- Veldhuijzen van Zanten, S.E.M.; Sewing, A.C.P.; van Lingen, A.; Hoekstra, O.S.; Wesseling, P.; Meel, M.H.; van Vuurden, D.G.; Kaspers, G.J.L.; Hulleman, E.; Bugiani, M. Multiregional Tumor Drug-Uptake Imaging by PET and Microvascular Morphology in End-Stage Diffuse Intrinsic Pontine Glioma. J. Nucl. Med. 2018, 59, 612–615. [Google Scholar] [CrossRef]
- Hsu, A.R.; Cai, W.; Veeravagu, A.; Mohamedali, K.A.; Chen, K.; Kim, S.; Vogel, H.; Hou, L.C.; Tse, V.; Rosenblum, M.G.; et al. Multimodality molecular imaging of glioblastoma growth inhibition with vasculature-targeting fusion toxin VEGF121/rGel. J. Nucl. Med. 2007, 48, 445–454. [Google Scholar]
- Chen, K.; Cai, W.; Li, Z.B.; Wang, H.; Chen, X. Quantitative PET imaging of VEGF receptor expression. Mol. Imaging Biol. 2009, 11, 15–22. [Google Scholar] [CrossRef]
- Cai, W.; Chen, K.; Mohamedali, K.A.; Cao, Q.; Gambhir, S.S.; Rosenblum, M.G.; Chen, X. PET of vascular endothelial growth factor receptor expression. J. Nucl. Med. 2006, 47, 2048–2056. [Google Scholar]
- Rainer, E.; Wang, H.; Traub-Weidinger, T.; Widhalm, G.; Fueger, B.; Chang, J.; Zhu, Z.; Marosi, C.; Haug, A.; Hacker, M.; et al. The prognostic value of [(123)I]-vascular endothelial growth factor ([(123)I]-VEGF) in glioma. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 2396–2403. [Google Scholar] [CrossRef]
- Wu, Y.L.; Ahn, M.J.; Garassino, M.C.; Han, J.Y.; Katakami, N.; Kim, H.R.; Hodge, R.; Kaur, P.; Brown, A.P.; Ghiorghiu, D.; et al. CNS Efficacy of Osimertinib in Patients With T790M-Positive Advanced Non-Small-Cell Lung Cancer: Data From a Randomized Phase III Trial (AURA3). J. Clin. Oncol. 2018, 36, 2702–2709. [Google Scholar] [CrossRef]
- Pusch, S.; Krausert, S.; Fischer, V.; Balss, J.; Ott, M.; Schrimpf, D.; Capper, D.; Sahm, F.; Eisel, J.; Beck, A.C.; et al. Pan-mutant IDH1 inhibitor BAY 1436032 for effective treatment of IDH1 mutant astrocytoma in vivo. Acta Neuropathol. 2017, 133, 629–644. [Google Scholar] [CrossRef] [PubMed]
- Mellinghoff, I.K.; Penas-Prado, M.; Peters, K.B.; Cloughesy, T.F.; Burris, H.A.; Maher, E.A.; Janku, F.; Cote, G.M.; Fuente, M.I.D.L.; Clarke, J.; et al. Phase 1 study of AG-881, an inhibitor of mutant IDH1/IDH2, in patients with advanced IDH-mutant solid tumors, including glioma. J. Clin. Oncol. 2018, 36, 2002. [Google Scholar] [CrossRef]
- Bublil, E.M.; Yarden, Y. The EGF receptor family: Spearheading a merger of signaling and therapeutics. Curr. Opin. Cell Biol. 2007, 19, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Hatanpaa, K.J.; Burma, S.; Zhao, D.; Habib, A.A. Epidermal growth factor receptor in glioma: Signal transduction, neuropathology, imaging, and radioresistance. Neoplasia 2010, 12, 675–684. [Google Scholar] [CrossRef]
- Kelly, W.J.; Shah, N.J.; Subramaniam, D.S. Management of Brain Metastases in Epidermal Growth Factor Receptor Mutant Non-Small-Cell Lung Cancer. Front. Oncol. 2018, 8, 208. [Google Scholar] [CrossRef]
- Weber, B.; Winterdahl, M.; Memon, A.; Sorensen, B.S.; Keiding, S.; Sorensen, L.; Nexo, E.; Meldgaard, P. Erlotinib accumulation in brain metastases from non-small cell lung cancer: Visualization by positron emission tomography in a patient harboring a mutation in the epidermal growth factor receptor. J. Thorac. Oncol. 2011, 6, 1287–1289. [Google Scholar] [CrossRef]
- Tang, Y.; Hu, Y.; Liu, W.; Chen, L.; Zhao, Y.; Ma, H.; Yang, J.; Yang, Y.; Liao, J.; Cai, J.; et al. A radiopharmaceutical [(89)Zr]Zr-DFO-nimotuzumab for immunoPET with epidermal growth factor receptor expression in vivo. Nucl. Med. Biol. 2019, 70, 23–31. [Google Scholar] [CrossRef]
- Sun, J.; Cai, L.; Zhang, K.; Zhang, A.; Pu, P.; Yang, W.; Gao, S. A pilot study on EGFR-targeted molecular imaging of PET/CT With 11C-PD153035 in human gliomas. Clin. Nucl. Med. 2014, 39, e20–e26. [Google Scholar] [CrossRef]
- Ulaner, G.A.; Lyashchenko, S.K.; Riedl, C.; Ruan, S.; Zanzonico, P.B.; Lake, D.; Jhaveri, K.; Zeglis, B.; Lewis, J.S.; O’Donoghue, J.A. First-in-Human Human Epidermal Growth Factor Receptor 2-Targeted Imaging Using (89)Zr-Pertuzumab PET/CT: Dosimetry and Clinical Application in Patients with Breast Cancer. J. Nucl. Med. 2018, 59, 900–906. [Google Scholar] [CrossRef]
- Tamura, K.; Kurihara, H.; Yonemori, K.; Tsuda, H.; Suzuki, J.; Kono, Y.; Honda, N.; Kodaira, M.; Yamamoto, H.; Yunokawa, M.; et al. 64Cu-DOTA-trastuzumab PET imaging in patients with HER2-positive breast cancer. J. Nucl. Med. 2013, 54, 1869–1875. [Google Scholar] [CrossRef]
- van der Veen, E.L.; Bensch, F.; Glaudemans, A.; Lub-de Hooge, M.N.; de Vries, E.G.E. Molecular imaging to enlighten cancer immunotherapies and underlying involved processes. Cancer Treat. Rev. 2018, 70, 232–244. [Google Scholar] [CrossRef] [PubMed]
- Decazes, P.; Bohn, P. Immunotherapy by Immune Checkpoint Inhibitors and Nuclear Medicine Imaging: Current and Future Applications. Cancers 2020, 12, 371. [Google Scholar] [CrossRef] [PubMed]
- Cole, E.L.; Kim, J.; Donnelly, D.J.; Smith, R.A.; Cohen, D.; Lafont, V.; Morin, P.E.; Huang, R.Y.; Chow, P.L.; Hayes, W.; et al. Radiosynthesis and preclinical PET evaluation of (89)Zr-nivolumab (BMS-936558) in healthy non-human primates. Bioorg. Med. Chem. 2017, 25, 5407–5414. [Google Scholar] [CrossRef]
- Bensch, F.; van der Veen, E.L.; Lub-de Hooge, M.N.; Jorritsma-Smit, A.; Boellaard, R.; Kok, I.C.; Oosting, S.F.; Schroder, C.P.; Hiltermann, T.J.N.; van der Wekken, A.J.; et al. (89)Zr-atezolizumab imaging as a non-invasive approach to assess clinical response to PD-L1 blockade in cancer. Nat. Med. 2018, 24, 1852–1858. [Google Scholar] [CrossRef] [PubMed]
- Niemeijer, A.N.; Leung, D.; Huisman, M.C.; Bahce, I.; Hoekstra, O.S.; van Dongen, G.; Boellaard, R.; Du, S.; Hayes, W.; Smith, R.; et al. Whole body PD-1 and PD-L1 positron emission tomography in patients with non-small-cell lung cancer. Nat. Commun. 2018, 9, 4664. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, D.J.; Smith, R.A.; Morin, P.; Lipovsek, D.; Gokemeijer, J.; Cohen, D.; Lafont, V.; Tran, T.; Cole, E.L.; Wright, M.; et al. Synthesis and Biologic Evaluation of a Novel (18)F-Labeled Adnectin as a PET Radioligand for Imaging PD-L1 Expression. J. Nucl. Med. 2018, 59, 529–535. [Google Scholar] [CrossRef]
- Tumeh, P.C.; Harview, C.L.; Yearley, J.H.; Shintaku, I.P.; Taylor, E.J.; Robert, L.; Chmielowski, B.; Spasic, M.; Henry, G.; Ciobanu, V.; et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 2014, 515, 568–571. [Google Scholar] [CrossRef]
- Pandit-Taskar, N.; Postow, M.; Hellmann, M.; Harding, J.; Barker, C.; O’Donoghue, J.; Ziolkowska, M.; Ruan, S.; Lyashchenko, S.; Tsai, F.; et al. First-in-human imaging with (89)Zr-Df-IAB22M2C anti-CD8 minibody in patients with solid malignancies: Preliminary pharmacokinetics, biodistribution, and lesion targeting. J. Nucl. Med. 2019. [Google Scholar] [CrossRef]
- Antonios, J.P.; Soto, H.; Everson, R.G.; Moughon, D.L.; Wang, A.C.; Orpilla, J.; Radu, C.; Ellingson, B.M.; Lee, J.T.; Cloughesy, T.; et al. Detection of immune responses after immunotherapy in glioblastoma using PET and MRI. Proc. Natl. Acad. Sci. USA 2017, 114, 10220–10225. [Google Scholar] [CrossRef]
- Nair-Gill, E.D.; Shu, C.J.; Radu, C.G.; Witte, O.N. Non-invasive imaging of adaptive immunity using positron emission tomography. Immunol. Rev. 2008, 221, 214–228. [Google Scholar] [CrossRef]
- Min, J.J.; Iyer, M.; Gambhir, S.S. Comparison of [18F]FHBG and [14C]FIAU for imaging of HSV1-tk reporter gene expression: Adenoviral infection vs stable transfection. Eur. J. Nucl. Med. Mol. Imaging 2003, 30, 1547–1560. [Google Scholar] [CrossRef] [PubMed]
- Keu, K.V.; Witney, T.H.; Yaghoubi, S.; Rosenberg, J.; Kurien, A.; Magnusson, R.; Williams, J.; Habte, F.; Wagner, J.R.; Forman, S.; et al. Reporter gene imaging of targeted T cell immunotherapy in recurrent glioma. Sci. Transl. Med. 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Parsons, D.W.; Jin, G.; McLendon, R.; Rasheed, B.A.; Yuan, W.; Kos, I.; Batinic-Haberle, I.; Jones, S.; Riggins, G.J.; et al. IDH1 and IDH2 mutations in gliomas. N. Engl. J. Med. 2009, 360, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Dang, L.; White, D.W.; Gross, S.; Bennett, B.D.; Bittinger, M.A.; Driggers, E.M.; Fantin, V.R.; Jang, H.G.; Jin, S.; Keenan, M.C.; et al. Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 2009, 462, 739–744. [Google Scholar] [CrossRef]
- Lu, C.; Ward, P.S.; Kapoor, G.S.; Rohle, D.; Turcan, S.; Abdel-Wahab, O.; Edwards, C.R.; Khanin, R.; Figueroa, M.E.; Melnick, A.; et al. IDH mutation impairs histone demethylation and results in a block to cell differentiation. Nature 2012, 483, 474–478. [Google Scholar] [CrossRef]
- Platten, M.; Schilling, D.; Bunse, L.; Wick, A.; Bunse, T.; Riehl, D.; Karapanagiotou-Schenkel, I.; Harting, I.; Sahm, F.; Schmitt, A.; et al. A mutation-specific peptide vaccine targeting IDH1R132H in patients with newly diagnosed malignant astrocytomas: A first-in-man multicenter phase I clinical trial of the German Neurooncology Working Group (NOA-16). J. Clin. Oncol. 2018, 36, 2001. [Google Scholar] [CrossRef]
- Suh, C.H.; Kim, H.S.; Paik, W.; Choi, C.; Ryu, K.H.; Kim, D.; Woo, D.C.; Park, J.E.; Jung, S.C.; Choi, C.G.; et al. False-Positive Measurement at 2-Hydroxyglutarate MR Spectroscopy in Isocitrate Dehydrogenase Wild-Type Glioblastoma: A Multifactorial Analysis. Radiology 2019, 291, 752–762. [Google Scholar] [CrossRef]
- Choi, C.; Ganji, S.K.; DeBerardinis, R.J.; Hatanpaa, K.J.; Rakheja, D.; Kovacs, Z.; Yang, X.L.; Mashimo, T.; Raisanen, J.M.; Marin-Valencia, I.; et al. 2-hydroxyglutarate detection by magnetic resonance spectroscopy in IDH-mutated patients with gliomas. Nat. Med. 2012, 18, 624–629. [Google Scholar] [CrossRef]
- Chitneni, S.K.; Reitman, Z.J.; Gooden, D.M.; Yan, H.; Zalutsky, M.R. Radiolabeled inhibitors as probes for imaging mutant IDH1 expression in gliomas: Synthesis and preliminary evaluation of labeled butyl-phenyl sulfonamide analogs. Eur. J. Med. Chem. 2016, 119, 218–230. [Google Scholar] [CrossRef]
- Chitneni, S.K.; Yan, H.; Zalutsky, M.R. Synthesis and Evaluation of a (18)F-Labeled Triazinediamine Analogue for Imaging Mutant IDH1 Expression in Gliomas by PET. ACS Med. Chem. Lett. 2018, 9, 606–611. [Google Scholar] [CrossRef]
- Chitneni, S.K.; Reitman, Z.J.; Spicehandler, R.; Gooden, D.M.; Yan, H.; Zalutsky, M.R. Synthesis and evaluation of radiolabeled AGI-5198 analogues as candidate radiotracers for imaging mutant IDH1 expression in tumors. Bioorg. Med. Chem. Lett. 2018, 28, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Koyasu, S.; Shimizu, Y.; Morinibu, A.; Saga, T.; Nakamoto, Y.; Togashi, K.; Harada, H. Increased (14)C-acetate accumulation in IDH-mutated human glioblastoma: Implications for detecting IDH-mutated glioblastoma with (11)C-acetate PET imaging. J. Neurooncol. 2019, 145, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Burger, J.A.; Kipps, T.J. CXCR4: A key receptor in the crosstalk between tumor cells and their microenvironment. Blood 2006, 107, 1761–1767. [Google Scholar] [CrossRef]
- Bian, X.W.; Yang, S.X.; Chen, J.H.; Ping, Y.F.; Zhou, X.D.; Wang, Q.L.; Jiang, X.F.; Gong, W.; Xiao, H.L.; Du, L.L.; et al. Preferential expression of chemokine receptor CXCR4 by highly malignant human gliomas and its association with poor patient survival. Neurosurgery 2007, 61, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C.; Vidal, A.; Bonnemaire, C.; Kraeber-Bodere, F.; Cherel, M.; Pallardy, A.; Rousseau, C.; Garcion, E.; Lacoeuille, F.; Hindre, F.; et al. Potential for Nuclear Medicine Therapy for Glioblastoma Treatment. Front. Pharmacol. 2019, 10, 772. [Google Scholar] [CrossRef]
- Lapa, C.; Luckerath, K.; Kleinlein, I.; Monoranu, C.M.; Linsenmann, T.; Kessler, A.F.; Rudelius, M.; Kropf, S.; Buck, A.K.; Ernestus, R.I.; et al. (68)Ga-Pentixafor-PET/CT for Imaging of Chemokine Receptor 4 Expression in Glioblastoma. Theranostics 2016, 6, 428–434. [Google Scholar] [CrossRef]
- Wernicke, A.G.; Edgar, M.A.; Lavi, E.; Liu, H.; Salerno, P.; Bander, N.H.; Gutin, P.H. Prostate-specific membrane antigen as a potential novel vascular target for treatment of glioblastoma multiforme. Arch. Pathol. Lab. Med. 2011, 135, 1486–1489. [Google Scholar] [CrossRef]
- Schwenck, J.; Tabatabai, G.; Skardelly, M.; Reischl, G.; Beschorner, R.; Pichler, B.; la Fougere, C. In vivo visualization of prostate-specific membrane antigen in glioblastoma. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 170–171. [Google Scholar] [CrossRef]
- Unterrainer, M.; Niyazi, M.; Ruf, V.; Bartenstein, P.; Albert, N.L. The endothelial prostate-specific membrane antigen is highly expressed in gliosarcoma and visualized by [68Ga]-PSMA-11 PET: A theranostic outlook for brain tumor patients? Neuro Oncol. 2017, 19, 1698–1699. [Google Scholar] [CrossRef]
- Rahbar, K.; Ahmadzadehfar, H.; Kratochwil, C.; Haberkorn, U.; Schafers, M.; Essler, M.; Baum, R.P.; Kulkarni, H.R.; Schmidt, M.; Drzezga, A.; et al. German Multicenter Study Investigating 177Lu-PSMA-617 Radioligand Therapy in Advanced Prostate Cancer Patients. J. Nucl. Med. 2017, 58, 85–90. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).