Synthesis, Characterization, and Biological Evaluation of Novel 7-Oxo-7H-thiazolo[3,2-b]-1,2,4-triazine-2-carboxylic Acid Derivatives
Abstract
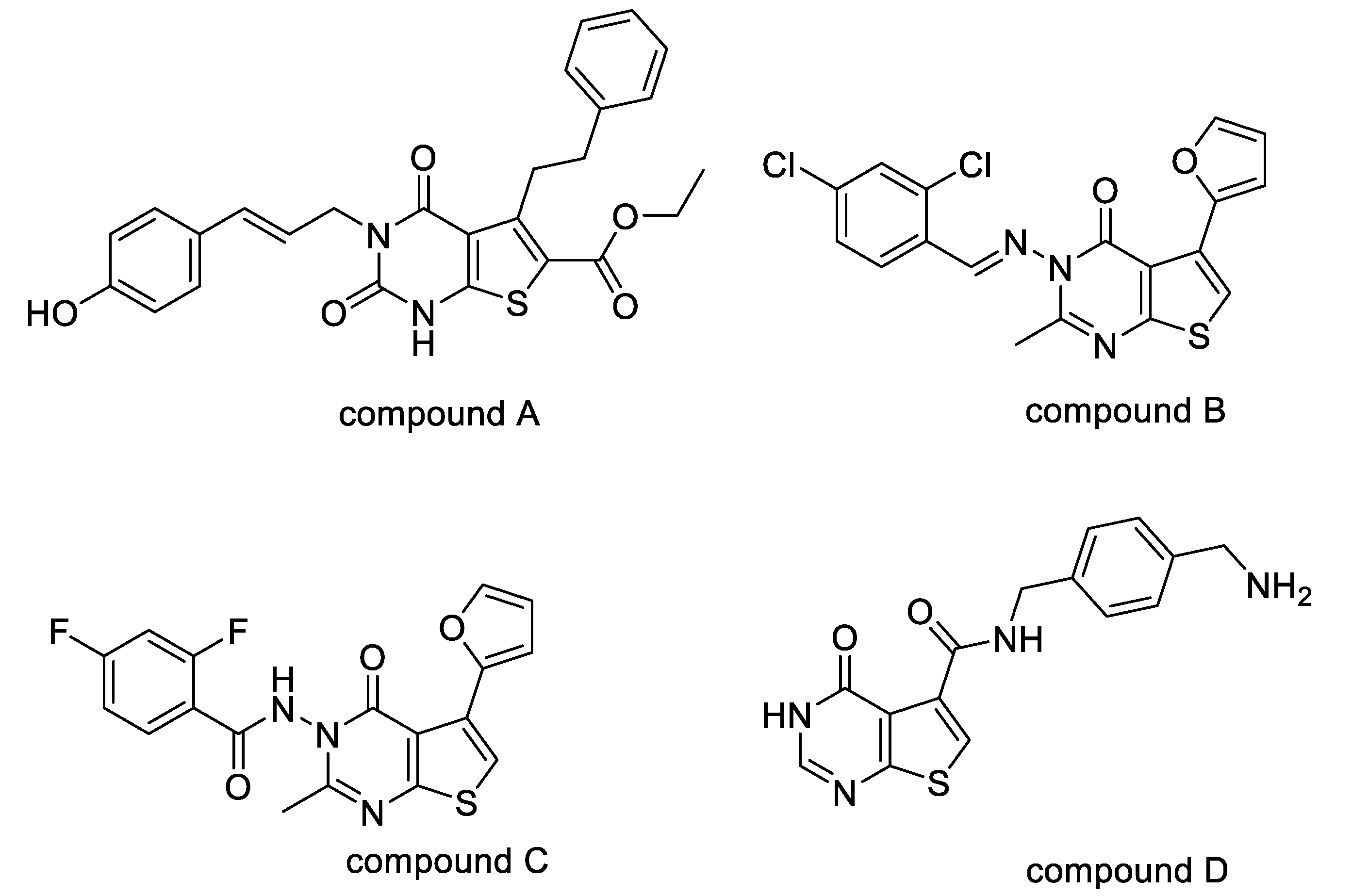
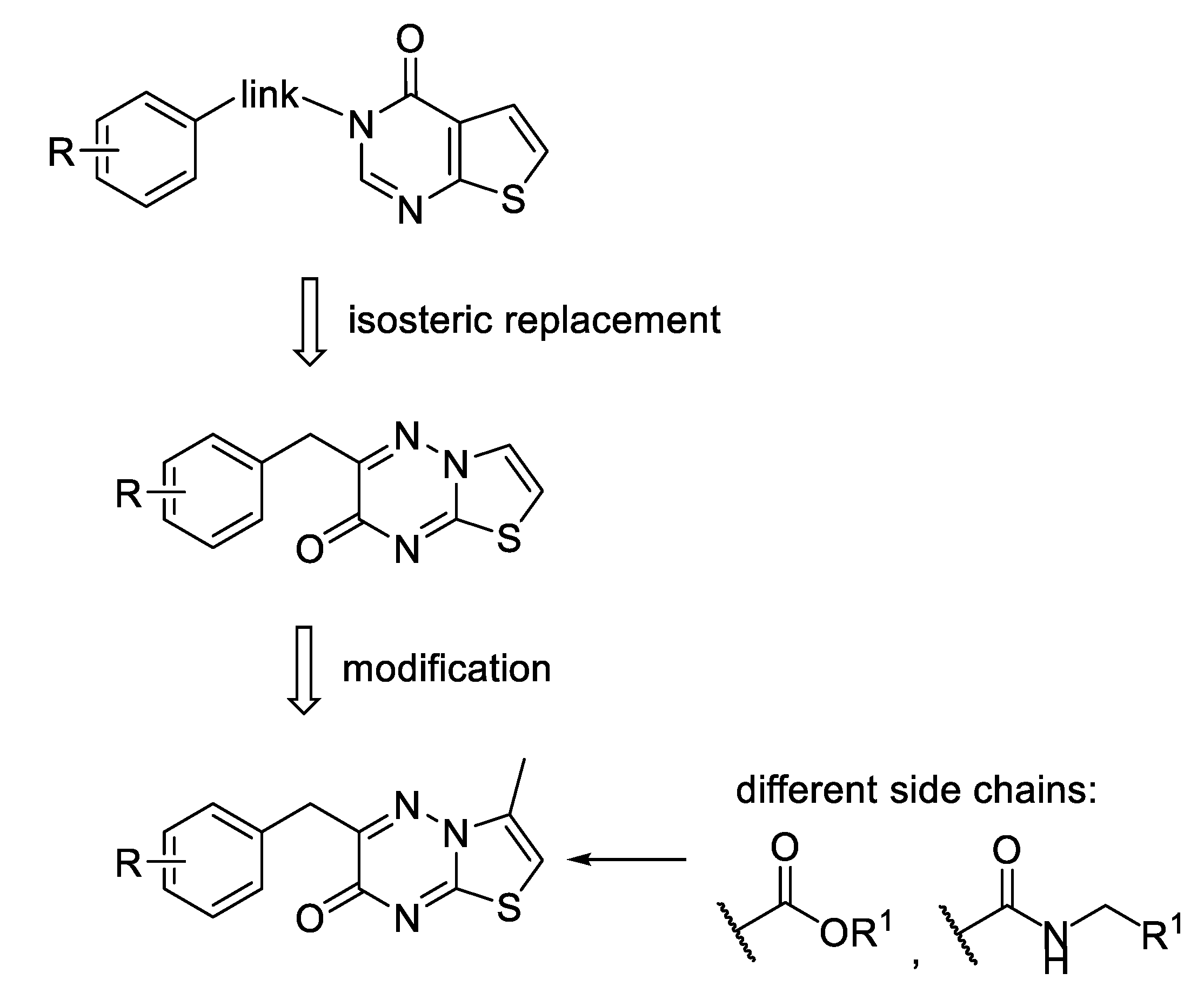
1. Introduction
2. Results and Discussion
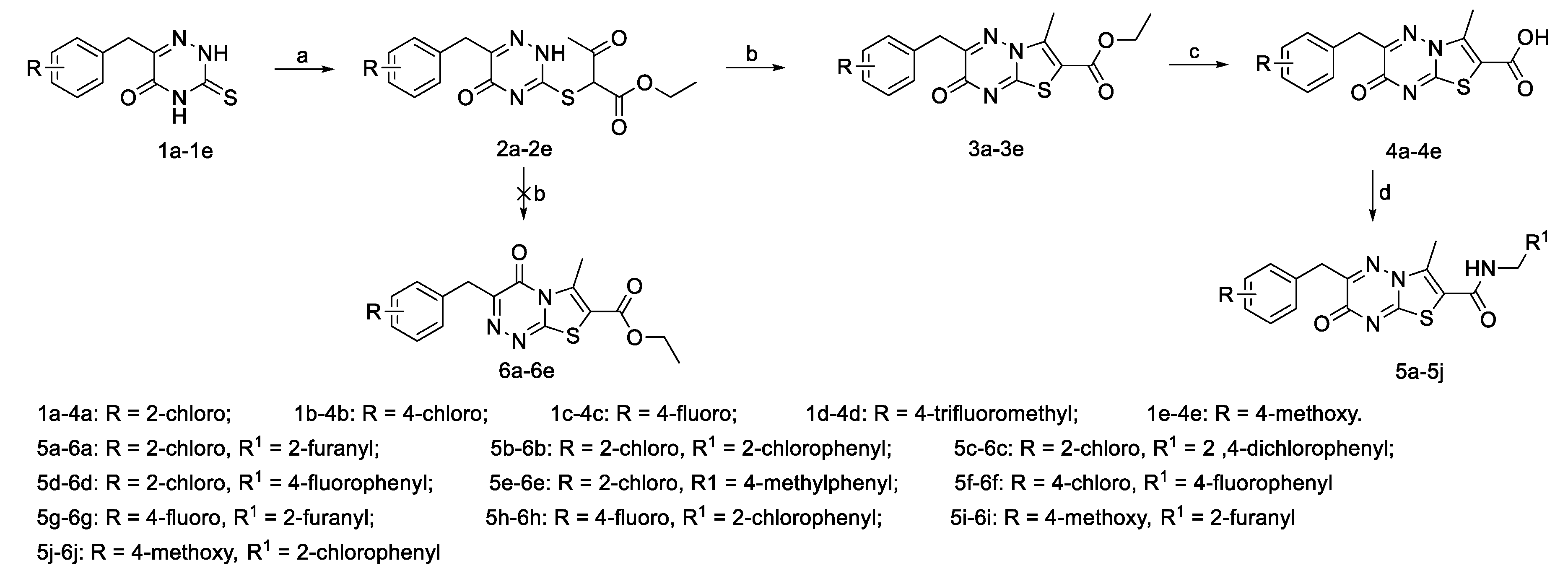
2.1. Chemistry
2.2. Biological Assays
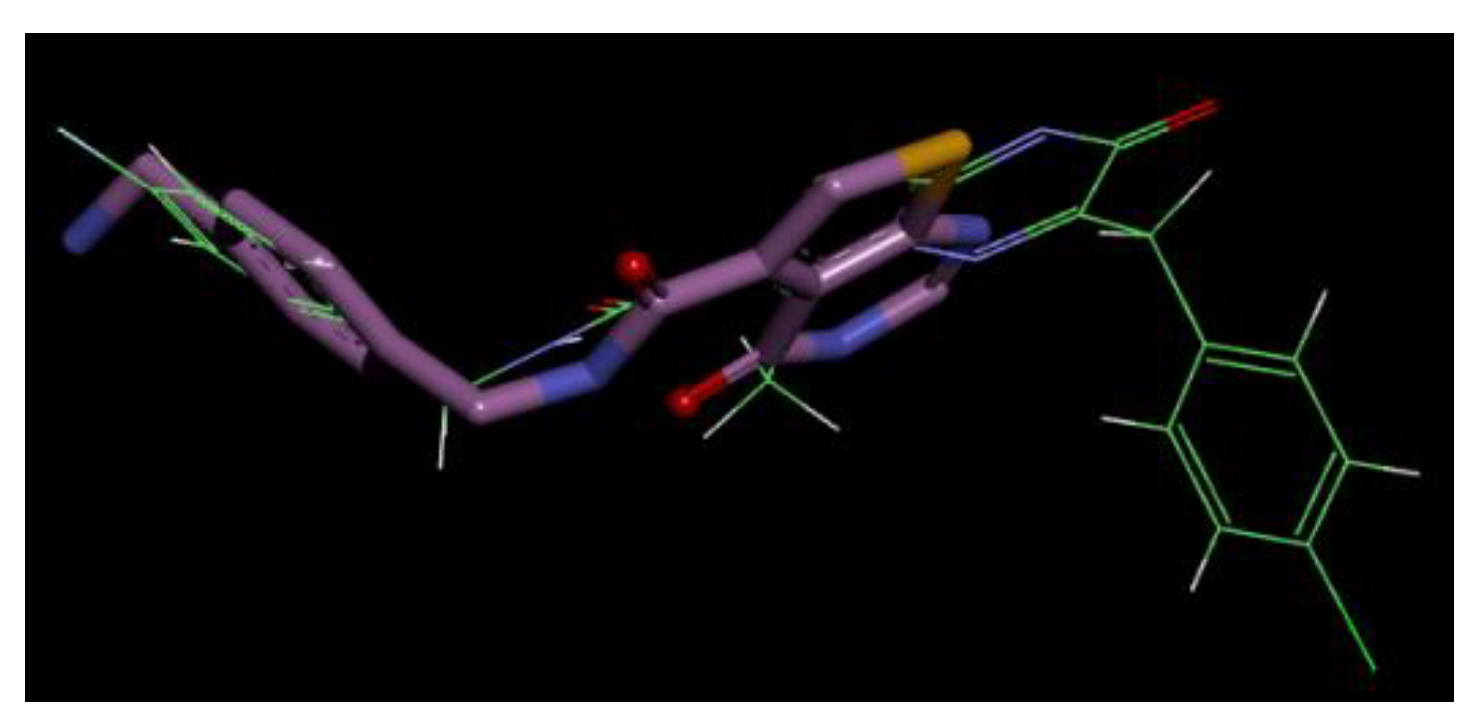
2.3. Target Identification by Pharmacophore
2.4. Leucyl-tRNA Synthetase Activity
3. Materials and Methods
3.1. General Information
3.2. Synthesis
3.2.1. General Procedure for the Synthesis of the β-Keto Esters (2a–2e)
3.2.2. General Procedure for the Synthesis of Ethyl 6-Aryl-3-methyl-7-oxo-7H-thiazolo[3,2-b]-1,2,4-triazine-2-carboxylates (3a–3e)
3.2.3. General Procedure for the Synthesis of 6-Aryl-3-methyl-7-oxo-7H-thiazolo[3,2-b]-1,2,4-triazine-2-carboxylic acids (4a–4e)
3.2.4. General Procedure for the Synthesis of the Target Compounds 5a–5j
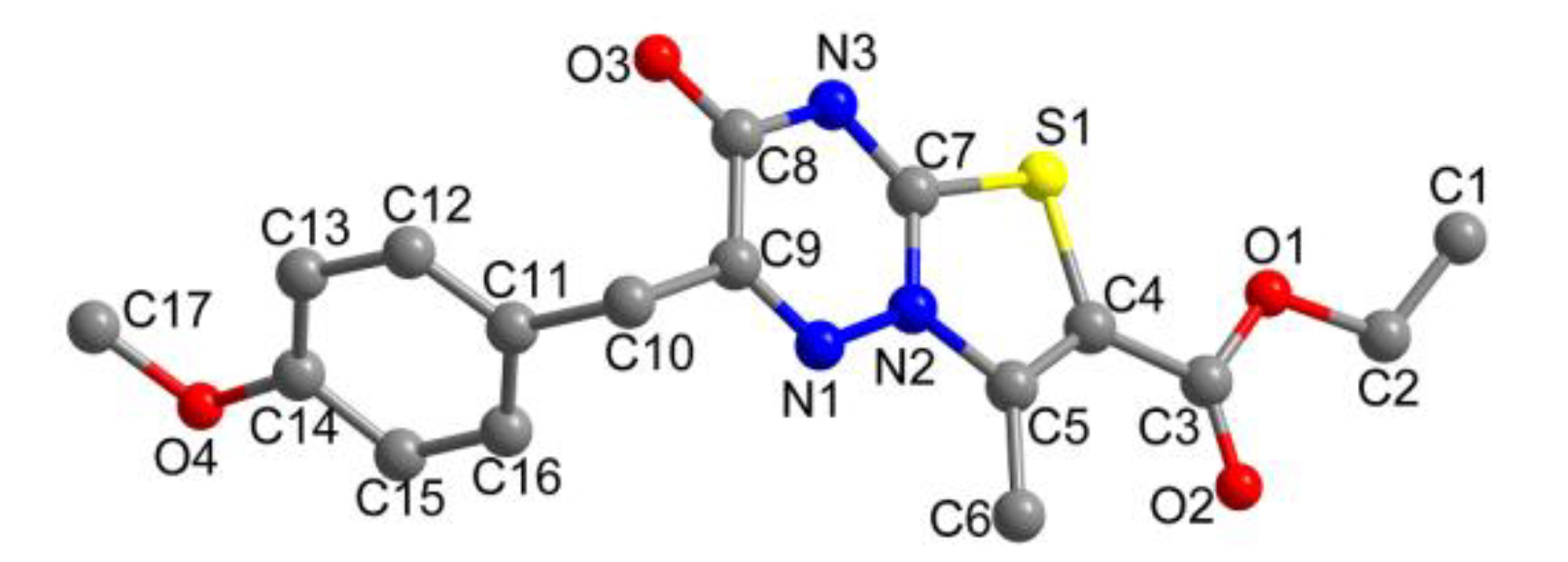
3.3. Crystal Structure Determination
3.4. Determination of MIC for Bacterial Strains
3.5. Pharmacological Target Profiling
3.6. Aminoacylation Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 1H-NMR | proton nuclear magnetic resonance spectra |
| 13C-NMR | carbon nuclear magnetic resonance spectra |
| AMP | adenosine monophosphate |
| ATP | adenosine triphosphate |
| CCDC | Cambridge Crystallographic Data Centre |
| DCM | dichloromethane |
| DMF | N,N-Dimethylformamide |
| DTT | dithiothreitol |
| EDCl | N′-(3-dimethylaminopropyl)-N-ethylcarbodiimide hydrochloride |
| HEPES | 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid |
| HOBt | 1-hydroxybenzotriazole |
| HRMS | high-resolution mass spectra |
| IC50 | half maximal inhibitory concentration |
| IR | infrared spectra |
| KOH | potassium hydroxide |
| MIC | minimum inhibitory concentration |
| MS | mass spectra |
| ORTEP | Oak ridge thermal-ellipsoid plot program |
| PDB | protein data bank |
| PPA | polyphosphoric acid |
| SHELXS | the crystallographic programs for crystal structure solution and refinement written by G. M. Sheldrick |
| TCA | trichloroacetic acid |
| TLC | thin layer chromatography |
| TMS | tetramethylsilane |
References
- Stein, K.; Farmer, J.; Singhal, S.; Marra, F.; Sutherland, S.; Quiñonez, C. The use and misuse of antibiotics in dentistry: A scoping review. J. Am. Dent. Assoc. 2018, 149, 869–884. [Google Scholar] [CrossRef]
- Chokshi, A.; Sifri, Z.; Cennimo, D.; Horng, H. Global contributors to antibiotic resistance. J. Glob. Infect. Dis. 2019, 11, 36–42. [Google Scholar]
- Vandevelde, N.M.; Tulkens, P.M.; Van Bambeke, F. Modulating antibiotic activity towards respiratory bacterial pathogens by co-medications: A multi-target approach. Drug Discov. Today 2016, 21, 1114–1129. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Viana, J.; Ishiki, H.M.; Scotti, M.T.; Scotti, L. Multi-target antitubercular drugs. Curr. Top. Med. Chem. 2018, 18, 750–758. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Sharma, P.K.; Verma, P.K.; Tomer, I.; Mathur, G.; Dhakad, P.K. Biological potential of thiazole derivatives of synthetic origin. J. Heterocycl. Chem. 2017, 54, 2103–2116. [Google Scholar] [CrossRef]
- Kashyap, A.; Adhikari, N.; Das, A.; Shakya, A.; Ghosh, S.K.; Singh, U.P.; Bhat, H.R. Review on synthetic chemistry and antibacterial importance of thiazole derivatives. Curr. Drug Disc. Technol. 2018, 15, 214–228. [Google Scholar] [CrossRef]
- Mishra, R.; Sachan, N.; Kumar, N.; Mishra, I.; Chand, P. Thiophene scaffold as prospective antimicrobial agent: A Review. J. Heterocycl. Chem. 2018, 55, 2019–2034. [Google Scholar] [CrossRef]
- Ali, E.M.H.; Abdel-Maksoud, M.S.; Oh, C.H. Thieno[2,3-d]pyrimidine as a promising scaffold in medicinal chemistry: Recent advances. Biorg. Med. Chem. 2019, 27, 1159–1194. [Google Scholar] [CrossRef]
- Dewal, M.B.; Wani, A.S.; Vidaillac, C.; Oupický, D.; Rybak, M.J.; Firestine, S.M. Thieno[2,3-d]pyrimidinedione derivatives as antibacterial agents. Eur. J. Med. Chem. 2012, 51, 145–153. [Google Scholar] [CrossRef]
- Chambhare, R.V.; Khadse, B.G.; Bobde, A.S.; Bahekar, R.H. Synthesis and preliminary evaluation of some N-[5-(2-furanyl)-2-methyl-4-oxo-4H-thieno[2,3-d]pyrimidin-3-yl]-carboxamide and 3-substituted-5-(2-furanyl)-2-methyl-3H-thieno[2,3-d]pyrimidin-4-ones as antimicrobial agents. Eur. J. Med. Chem. 2003, 38, 89–100. [Google Scholar] [CrossRef]
- Hill, P.J.; Abibi, A.; Albert, R.; Andrews, B.; Gagnon, M.M.; Gao, N.; Grebe, T.; Hajec, L.I.; Huang, J.; Livchak, S.; et al. Selective inhibitors of bacterial t-RNA-(N1G37) Methyltransferase (TrmD) that demonstrate novel ordering of the lid domain. J. Med. Chem. 2013, 56, 7278–7288. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, E.A.; Dine, S.; Soliman, F.; Labouta, I.M. Potential antineoplastics I: Substituted 3,5-dioxo-and 3-thioxo-5-oxo-2,3,4,5-tetrahydro-1,2,4-triazines. J. Pharm. Sci. 1979, 68, 243–245. [Google Scholar] [CrossRef] [PubMed]
- Forlani, L.; Tocke, A.L.; Del Vecchio, E.; Lakhdar, S.; Goumont, R.; Terrier, F. Assessing the nitrogen and carbon nucleophilicities of 2-aminothiazoles through coupling with superelectrophilic 4,6-dinitrobenzofuroxan. J. Org. Chem. 2006, 71, 5527–5537. [Google Scholar] [CrossRef] [PubMed]
- Musser, J.H.; Kreft, A.F. 5-Lipoxygenase: Properties, pharmacology, and the quinolinyl (bridged) aryl class of inhibitors. J. Med. Chem. 1992, 35, 2501–2524. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Shang, R.; Shi, L.; Wan, D.C.C.; Lin, H. Synthesis and biological evaluation of 7H-thiazolo [3,2-b]-1,2,4-triazin-7-one derivatives as dual binding site acetylcholinesterase inhibitors. Eur. J. Med. Chem. 2014, 81, 237–244. [Google Scholar] [CrossRef]
- Zhi, H.; Zhang, C.; Cheng, Z.; Jin, Z.; Huang, E.; Li, S.; Lin, H.; Wan, D.C.; Hu, C. 6-Acetyl-5H-thiazolo 3,2-a pyrimidine derivatives as the novel acetylcholinesterase inhibitors: Design, synthesis, and biological activity. Med. Chem. 2013, 9, 703–709. [Google Scholar] [CrossRef]
- Jin, Z.; Yang, L.; Liu, S.J.; Wang, J.; Li, S.; Lin, H.Q.; Wan, D.C.C.; Hu, C. Synthesis and biological evaluation of 3,6-diaryl-7H-thiazolo [3,2-b][1,2,4] triazin-7-one derivatives as acetylcholinesterase inhibitors. Arch. Pharmacal Res. 2010, 33, 1641–1649. [Google Scholar] [CrossRef]
- Heilman, W.P.; Heilman, R.D.; Scozzie, J.A.; Wayner, R.J.; Gullo, J.M.; Ariyan, Z.S. Synthesis and antihypertensive activity of novel 3-hydrazino-5-phenyl-1,2,4-triazines. J. Med. Chem. 1979, 22, 671–677. [Google Scholar] [CrossRef]
- Heravi, M.M.; Aghapoor, K.; Nooshabadi, M.A. Regioselective acid-catalyzed cyclization reaction: Unique synthesis of condensed thiazoles and selenazole. Synth. Commun. 1998, 28, 233–237. [Google Scholar] [CrossRef]
- Stanovnik, B.; Tišler, M.; Katritzky, A.R.; Denisko, O.V. The tautomerism of heterocycles: Substituent tautomerism of six-membered ring heterocycles. Adv. Heterocycl. Chem. 2006, 91, 1–134. [Google Scholar]
- Liu, S.; Shang, R.; Shi, L.; Zhou, R.; He, J.; Wan, D.C.C. Design, Synthesis, and evaluation of 7H-thiazolo-[3,2-b]-1,2,4-triazin-7-one derivatives as dual binding site acetylcholinesterase inhibitors. Chem. Biol. Drug Des. 2014, 84, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, Y.; Jiang, X. Sulfur-center-involved photocatalyzed reactions. Chem.-Asian J. 2018, 13, 2208–2242. [Google Scholar] [CrossRef] [PubMed]
- Paronikyan, E.G.; Dashyan, S.S.; Minasyan, N.S.; Stepanyan, G.M.; Babaev, E.V. Synthesis of 10-alkylsulfanyl-substituted pyrido-[4′,3′:4,5] thieno [3,2-d] pyrimidines annulated with pyran, cyclohexane, and cyclopentane rings. Chem. Heterocycl. Com. 2016, 52, 337–345. [Google Scholar] [CrossRef]
- Dave, C.G.; Patel, K.J. Pyridopyrimidines. X. Synthesis of 3-substituted 2-thioxo-5, 7-dimethylpyrido [2,3-d] pyrimidin-4 (3H)-ones and their S-alkylation under phase transfer conditions. J. Heterocycl. Chem. 2001, 38, 457–461. [Google Scholar] [CrossRef]
- Karthikeyan, M.S. Synthesis and antimicrobial studies of thiazolotriazinones. Eur. J. Med. Chem. 2010, 45, 5039–5043. [Google Scholar] [CrossRef] [PubMed]
- Kinnings, S.L.; Jackson, R.M. ReverseScreen3D: A structure-based ligand matching method to identify protein targets. J. Chem. Inf. Model. 2011, 51, 624–634. [Google Scholar] [CrossRef]
- Jiang, J.; Zhou, H.; Jiang, Q.; Sun, L.; Deng, P. Novel transforming growth factor-β receptor 1 antagonists through a pharmacophore-based virtual screening approach. Molecules 2018, 23, 2824. [Google Scholar] [CrossRef]
- Meslamani, J.; Li, J.; Sutter, J.; Stevens, A.; Bertrand, H.-O.; Rognan, D. Protein–ligand-based pharmacophores: Generation and utility assessment in computational ligand profiling. J. Chem. Inf. Model. 2012, 52, 943–955. [Google Scholar] [CrossRef]
- Stout, T.J.; Tondi, D.; Rinaldi, M.; Barlocco, D.; Pecorari, P.; Santi, D.V.; Kuntz, I.D.; Stroud, R.M.; Shoichet, B.K.; Costi, M.P. Structure-based design of inhibitors specific for bacterial Thymidylate Synthase. Biochemistry 1999, 38, 1607–1617. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A: Found. Crystallogr. 2007, 64, 112–122. [Google Scholar] [CrossRef]
- Zhang, R.; Chen, Y.; Liu, H.; Wei, X.U. Cloning and expression of Leucyl-tRNA Synthetase from Mycobacterium phlei. Chin. J. Pharm. 2016, 47, 1374–1378. [Google Scholar]
- Li, X.; Hernandez, V.; Rock, F.L.; Choi, W.; Mak, Y.S.; Mohan, M.; Mao, W.; Zhou, Y.; Easom, E.E.; Plattner, J.J. Discovery of a potent and specific M. tuberculosis Leucyl-tRNA Synthetase inhibitor:(S)-3-(Aminomethyl)-4-chloro-7-(2-hydroxyethoxy) benzo [c][1,2] oxaborol-1 (3H)-ol (GSK656). J. Med. Chem. 2017, 60, 8011–8026. [Google Scholar] [CrossRef] [PubMed]
- Palencia, A.; Li, X.; Bu, W.; Choi, W.; Ding, C.Z.; Easom, E.E.; Feng, L.; Hernandez, V.; Houston, P.; Liu, L. Discovery of novel oral protein synthesis inhibitors of Mycobacterium tuberculosis that target leucyl-tRNA synthetase. Antimicrob. Agents Chemother. 2016, 60, 6271–6280. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: All synthesized compounds are available from the authors. |
| Entry | Reaction Condition | |||
|---|---|---|---|---|
| Conventional Heating | MW-Irradiation | |||
| Time/min | Yield */% | Time/min | Yield */% | |
| 3a | 60 | 68.86 | 20 | 81.24 |
| 3b | 60 | 70.41 | 20 | 85.12 |
| 3c | 60 | 71.31 | 20 | 83.78 |
| 3d | 60 | 66.28 | 20 | 80.12 |
| 3e | 60 | 75.26 | 20 | 87.66 |
| CCDC number | 1435646 |
| Formula | (C17H17N3O4S)4·CH3OH |
| Formula weight | 1468.62 |
| Crystal color, shape | Colorless, block |
| Crystal system | Triclinic |
| Space group | P–1 |
| a/Å | 11.7136 (8) |
| b/Å | 12.5137 (9) |
| c/Å | 13.8247 (9) |
| α (°) | 104.2510 (10) |
| β (°) | 98.4010 (10) |
| γ (°) | 110.7180 (10) |
| Volume/Å3 | 1775.2 (2) |
| Temperature/K | 296 (2) |
| Z | 1 |
| Density (calculated)/g·cm−3 | 1.378 |
| F (000) | 769 |
| Reflections collected/unique | 10269/6238 [R(int) = 0.0197] |
| Goodness-of-fit of F2 | 1.137 |
| Final R indices [I > 2σ(I)] | R1 = 0.0547, wR2 = 0.1655 |
| R indices (all data) | R1 = 0.0821, wR2 = 0.1780 |
| Largest diff. peak and hole/e.Å−3 | 0.448 and −0.401 |
| Entry | Antibacterial Activity MIC (μg/mL) | Antitubercular Activity MIC (μg/mL) | |||
|---|---|---|---|---|---|
| S. aureus | B. subtilis | E. coli | P. aeruginosa | M. smegmatis | |
| 3a | >800 | >800 | 400 | >800 | >800 |
| 3b | 100 | >800 | 400 | 800 | >800 |
| 3c | 400 | 400 | 200 | 200 | >800 |
| 3d | >800 | 800 | 400 | 400 | >800 |
| 3e | >800 | >800 | >800 | >800 | >800 |
| 4a | 200 | >800 | 100 | 800 | >800 |
| 4b | 400 | 400 | 100 | 400 | >800 |
| 4c | 200 | 400 | 50 | 200 | >800 |
| 4d | >800 | >800 | 400 | >800 | >800 |
| 4e | >800 | >800 | 800 | >800 | >800 |
| 5a | 100 | 50 | 100 | 400 | >800 |
| 5b | 200 | 100 | 200 | 100 | >800 |
| 5c | 50 | 100 | 400 | >800 | >800 |
| 5d | 50 | 200 | 50 | 100 | >800 |
| 5e | 100 | 50 | 400 | 400 | >800 |
| 5f | 200 | 200 | 100 | 200 | 50 |
| 5g | 100 | 50 | 100 | 50 | >800 |
| 5h | 100 | 200 | 200 | 400 | 400 |
| 5i | 200 | 400 | 200 | 100 | >800 |
| 5j | 200 | 200 | 400 | 400 | >800 |
| Ciprofloxacin | 25 | 100 | 25 | 50 | NT |
| Rifampicin | NT * | NT | NT | NT | 25 |
| PDB Id | FitValue | Pharmacophore/Metadata/Target-Class C | Target |
|---|---|---|---|
| 1TSL | 0.774082 | Methyltransferases | Bacterial thymidylate synthase |
| 1OBH | 0.347426 | Aminoacyl-tRNA synthetases (AARSs) | Leucyl-tRNA synthetase |
| 2HGZ | 0.310616 | Aminoacyl-tRNA synthetases (AARSs) | p-benzoyl-l-phenylalanyl-tRNA synthetase |
| 1I2Z | 0.0866648 | Enoyl-[acyl-carrier-protein] reductase [NADH] | Bacterial enoyl acyl carrier protein reductase (FabI) |
| 1LX6 | 0.0729051 | Enoyl-[acyl-carrier-protein] reductase [NADH] | Bacterial enoyl-ACP reductase (FabI) |
| 1MFP | 0.058642 | Enoyl-[acyl-carrier-protein] reductase [NADH] | Bacterial Enoyl-ACP reductases FabI and FabK |
| 2B35 | 7.90584 × 10−5 | Enoyl-[acyl-carrier-protein] reductase [NADH] | Mycobacterium tuberculosis enoyl reductase (InhA) |
| Compound | Percent Inhibition (%) |
|---|---|
| 4b | 12.89 ± 2.31 |
| 5f | 78.24 ± 4.05 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, D.; Li, T.; Xie, Q.; Yu, X.; Xu, W.; Chen, Y.; Jin, Z.; Hu, C. Synthesis, Characterization, and Biological Evaluation of Novel 7-Oxo-7H-thiazolo[3,2-b]-1,2,4-triazine-2-carboxylic Acid Derivatives. Molecules 2020, 25, 1307. https://doi.org/10.3390/molecules25061307
Cai D, Li T, Xie Q, Yu X, Xu W, Chen Y, Jin Z, Hu C. Synthesis, Characterization, and Biological Evaluation of Novel 7-Oxo-7H-thiazolo[3,2-b]-1,2,4-triazine-2-carboxylic Acid Derivatives. Molecules. 2020; 25(6):1307. https://doi.org/10.3390/molecules25061307
Chicago/Turabian StyleCai, Dong, Tai Li, Qian Xie, Xiaofei Yu, Wei Xu, Yu Chen, Zhe Jin, and Chun Hu. 2020. "Synthesis, Characterization, and Biological Evaluation of Novel 7-Oxo-7H-thiazolo[3,2-b]-1,2,4-triazine-2-carboxylic Acid Derivatives" Molecules 25, no. 6: 1307. https://doi.org/10.3390/molecules25061307
APA StyleCai, D., Li, T., Xie, Q., Yu, X., Xu, W., Chen, Y., Jin, Z., & Hu, C. (2020). Synthesis, Characterization, and Biological Evaluation of Novel 7-Oxo-7H-thiazolo[3,2-b]-1,2,4-triazine-2-carboxylic Acid Derivatives. Molecules, 25(6), 1307. https://doi.org/10.3390/molecules25061307






