Isothiazolinone Biocides: Chemistry, Biological, and Toxicity Profiles
Abstract
1. Introduction
2. Synthesis of Isothiazolinones
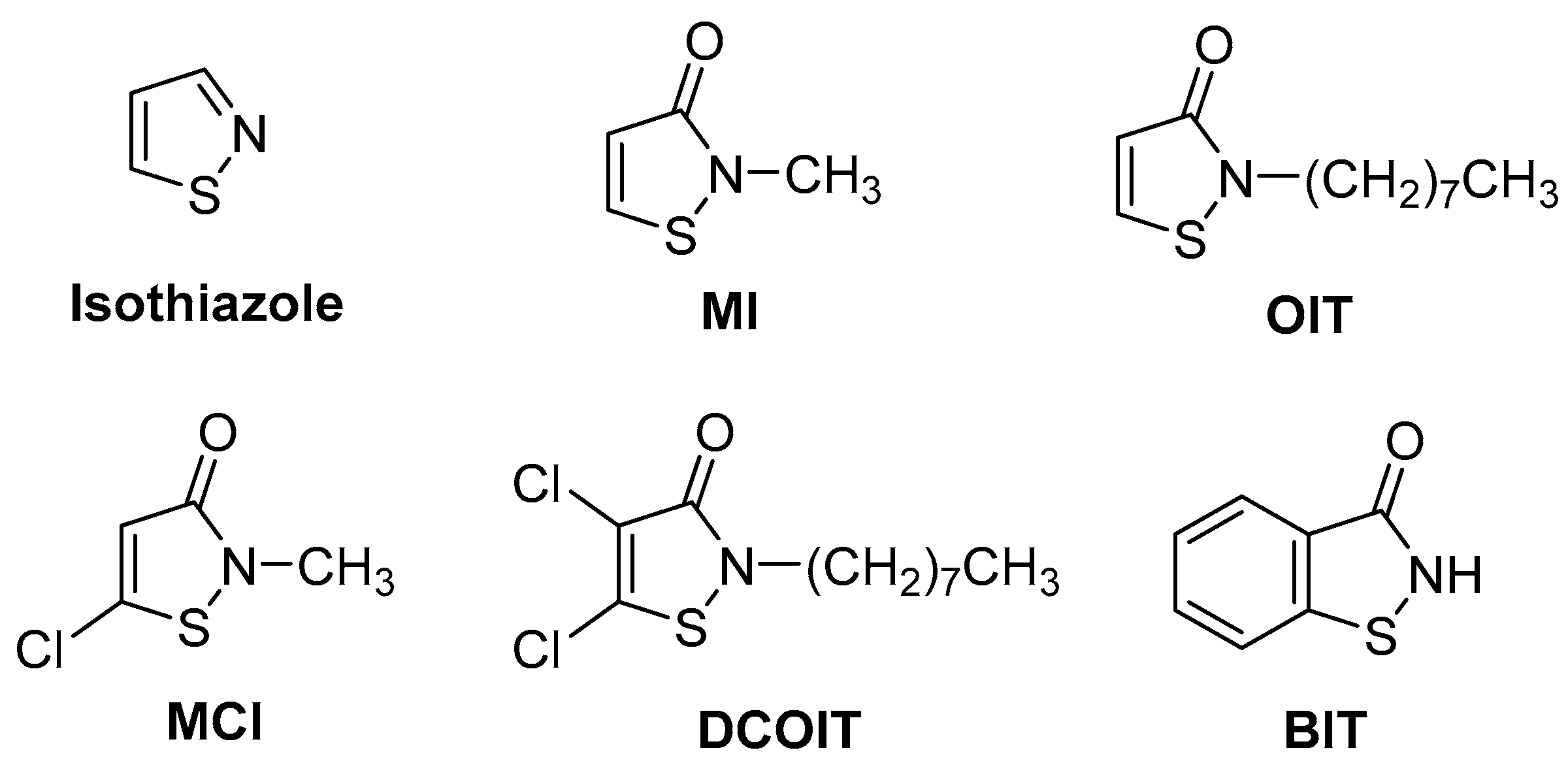
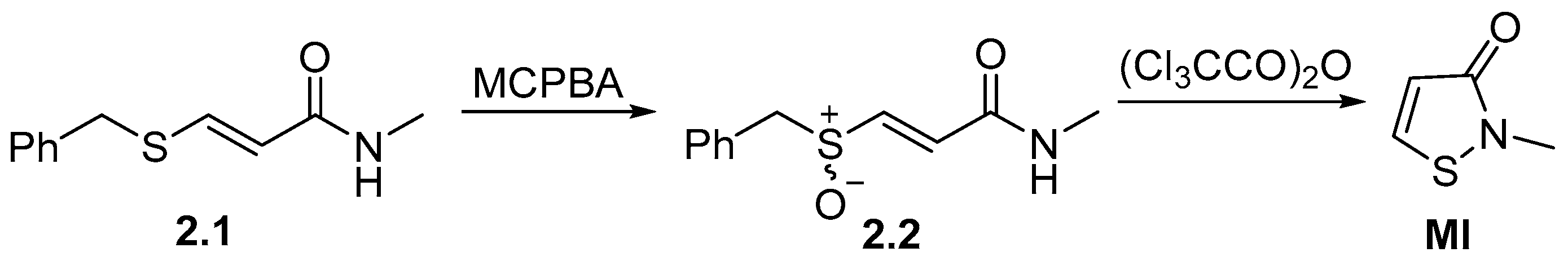
2.1. Synthesis of the Isothiazolinone Derivatives MI, MCI, OIT, and DCOIT
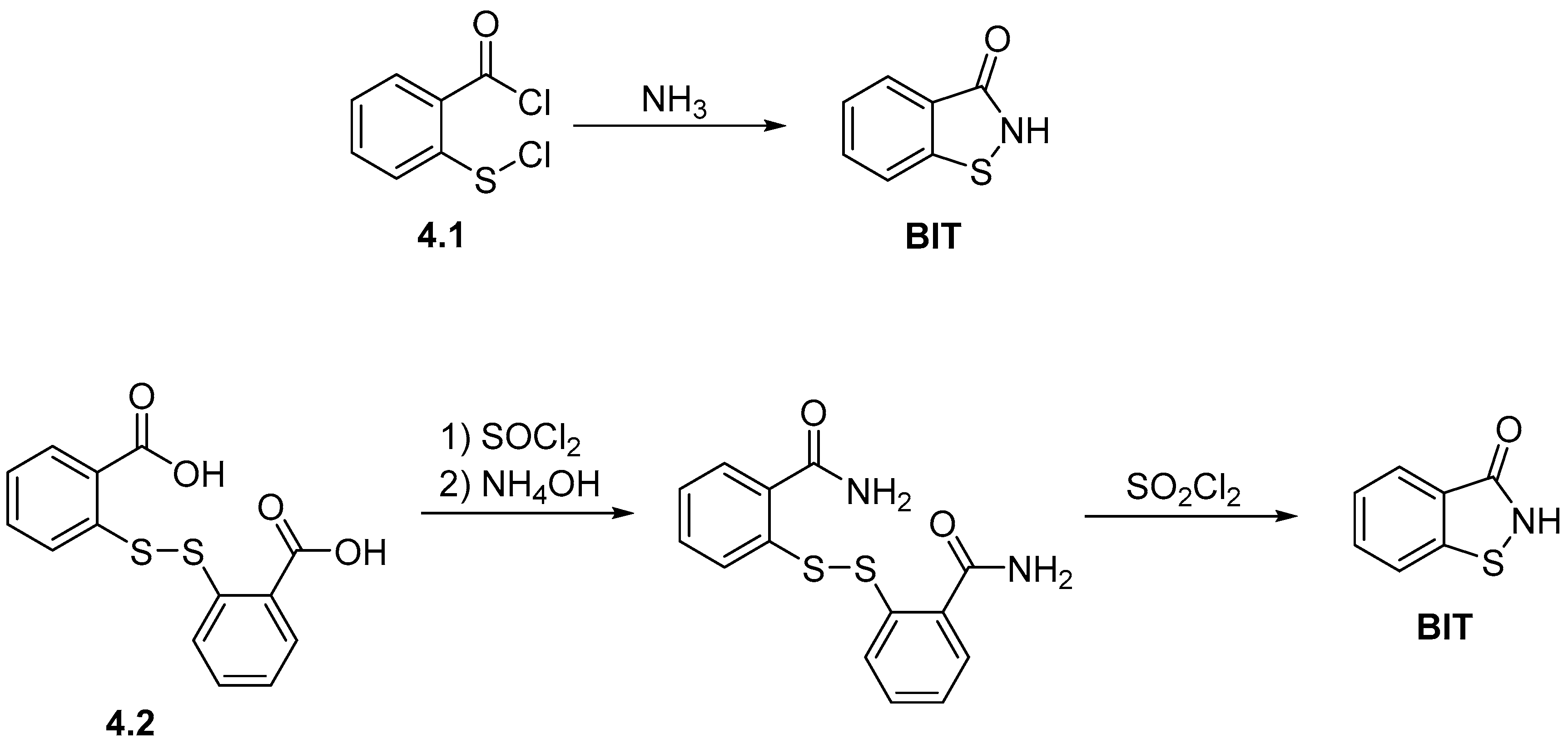
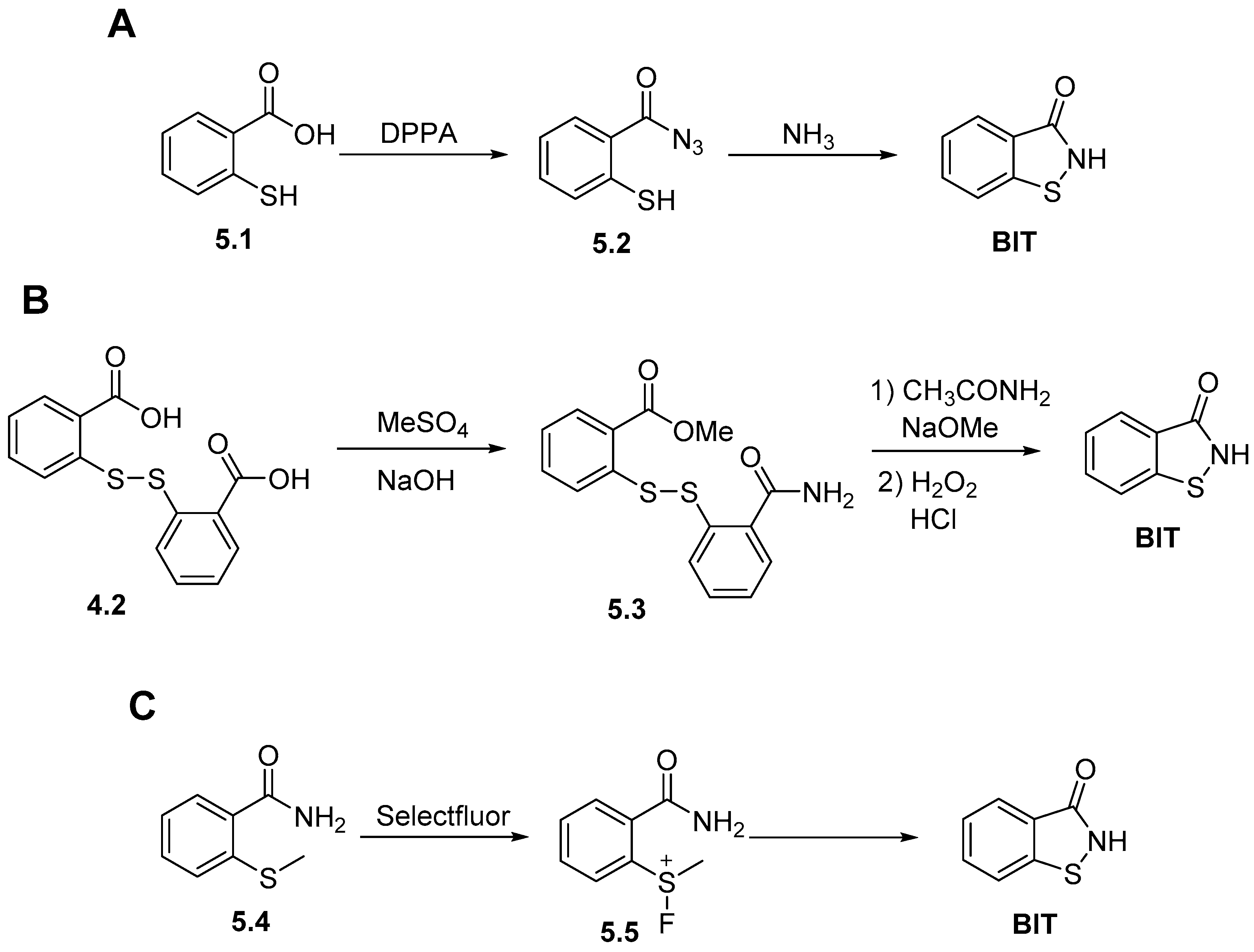
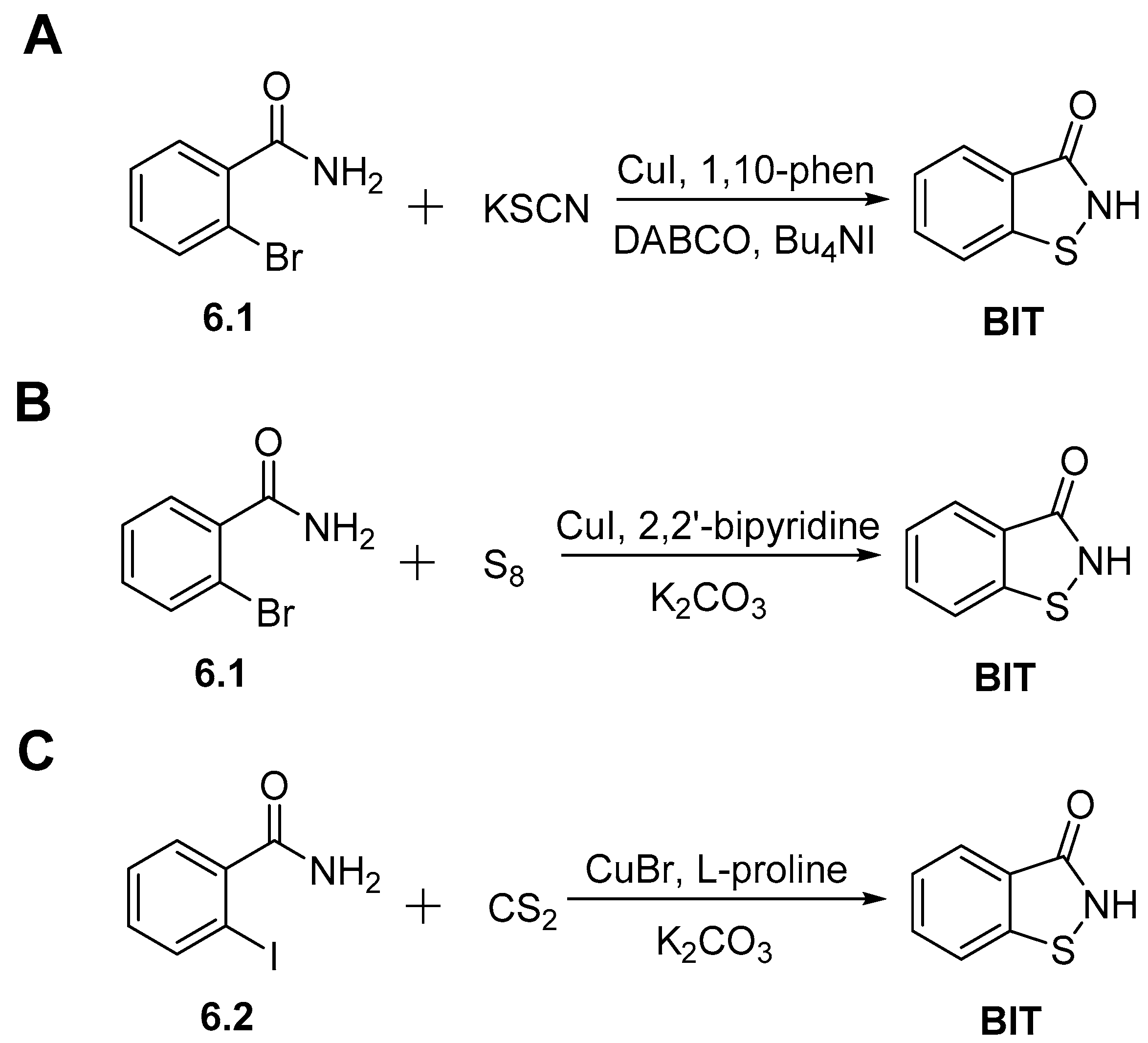
2.2. Synthesis of 1,2-Benzisothiazol-3(2H)-one (BIT)
3. Antibacterial Action and Toxicity of Isothiazolinones
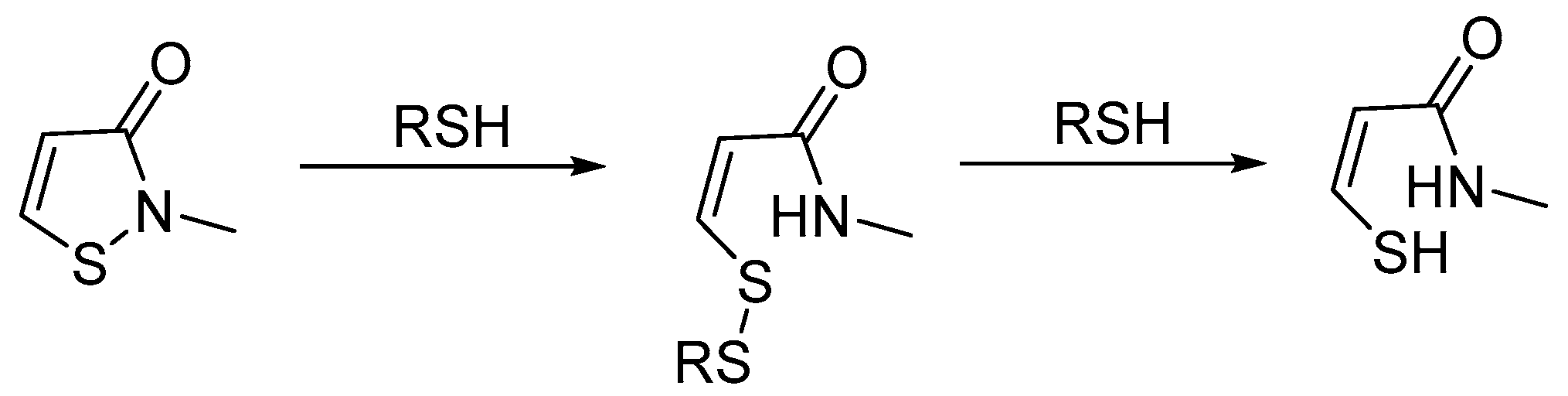
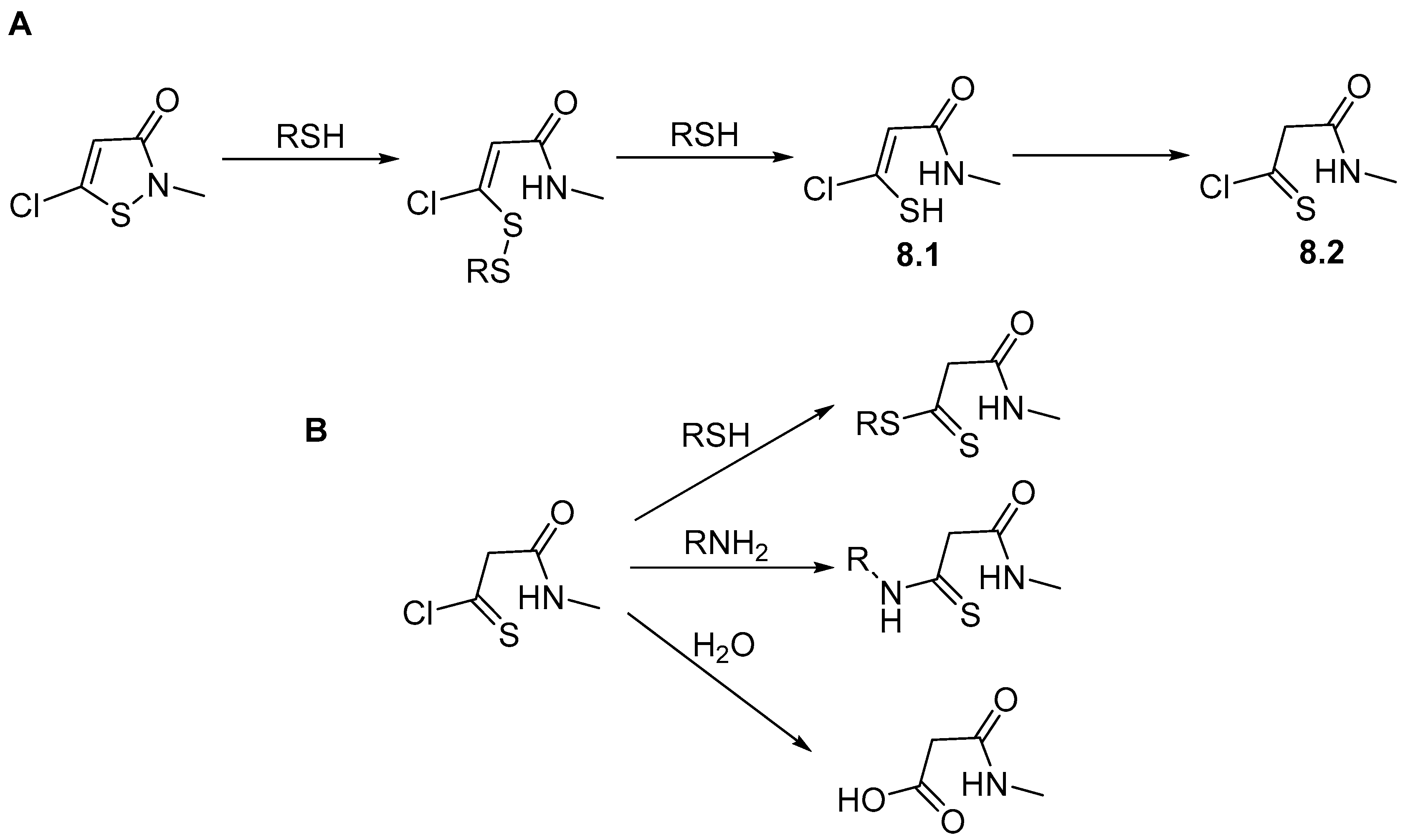
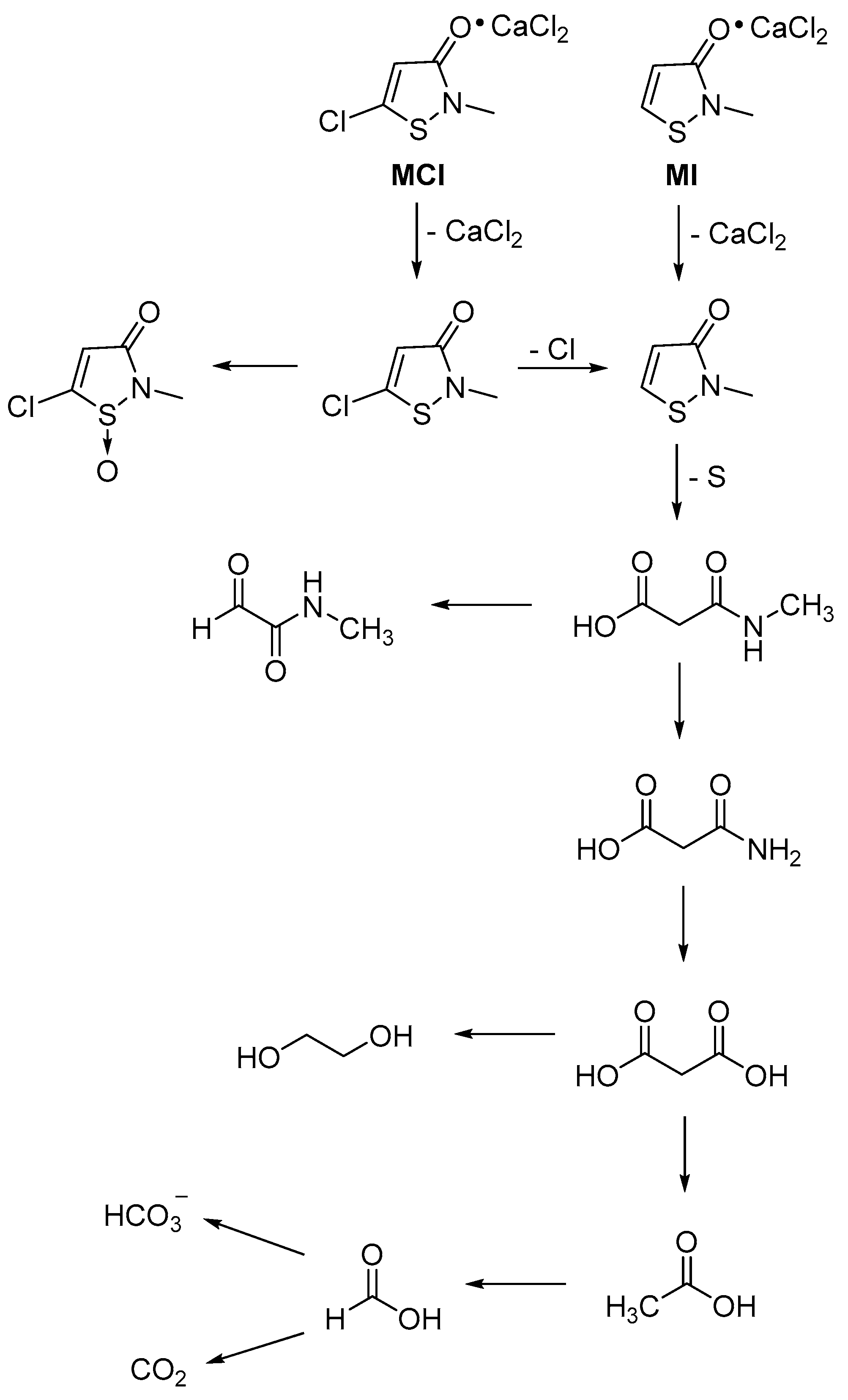
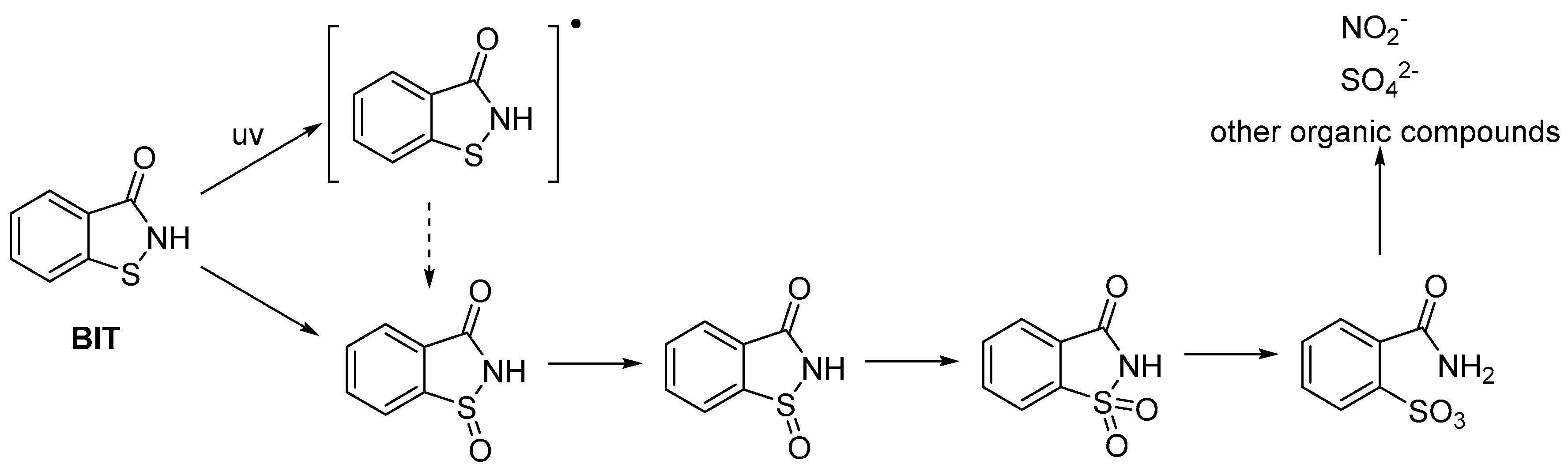
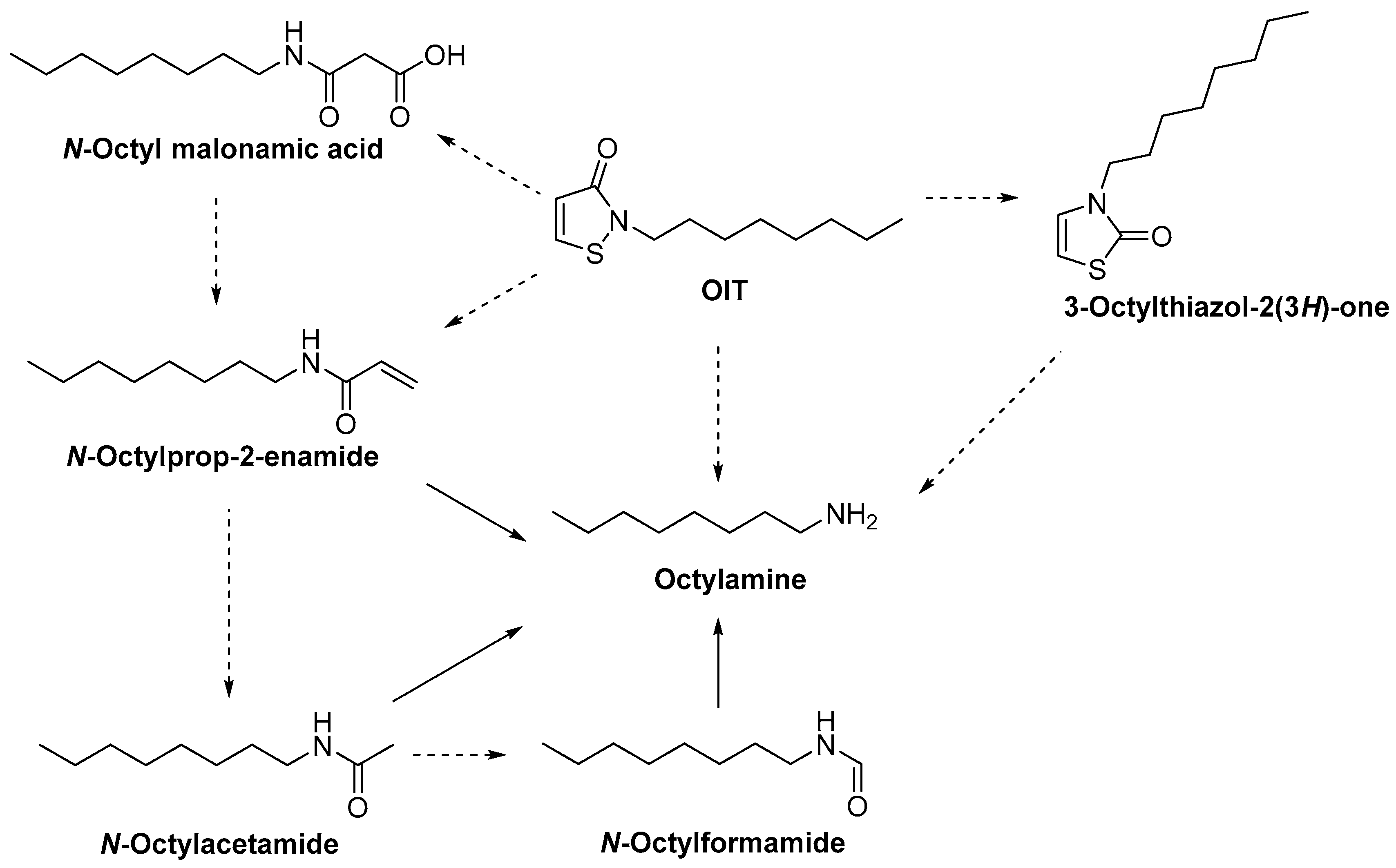
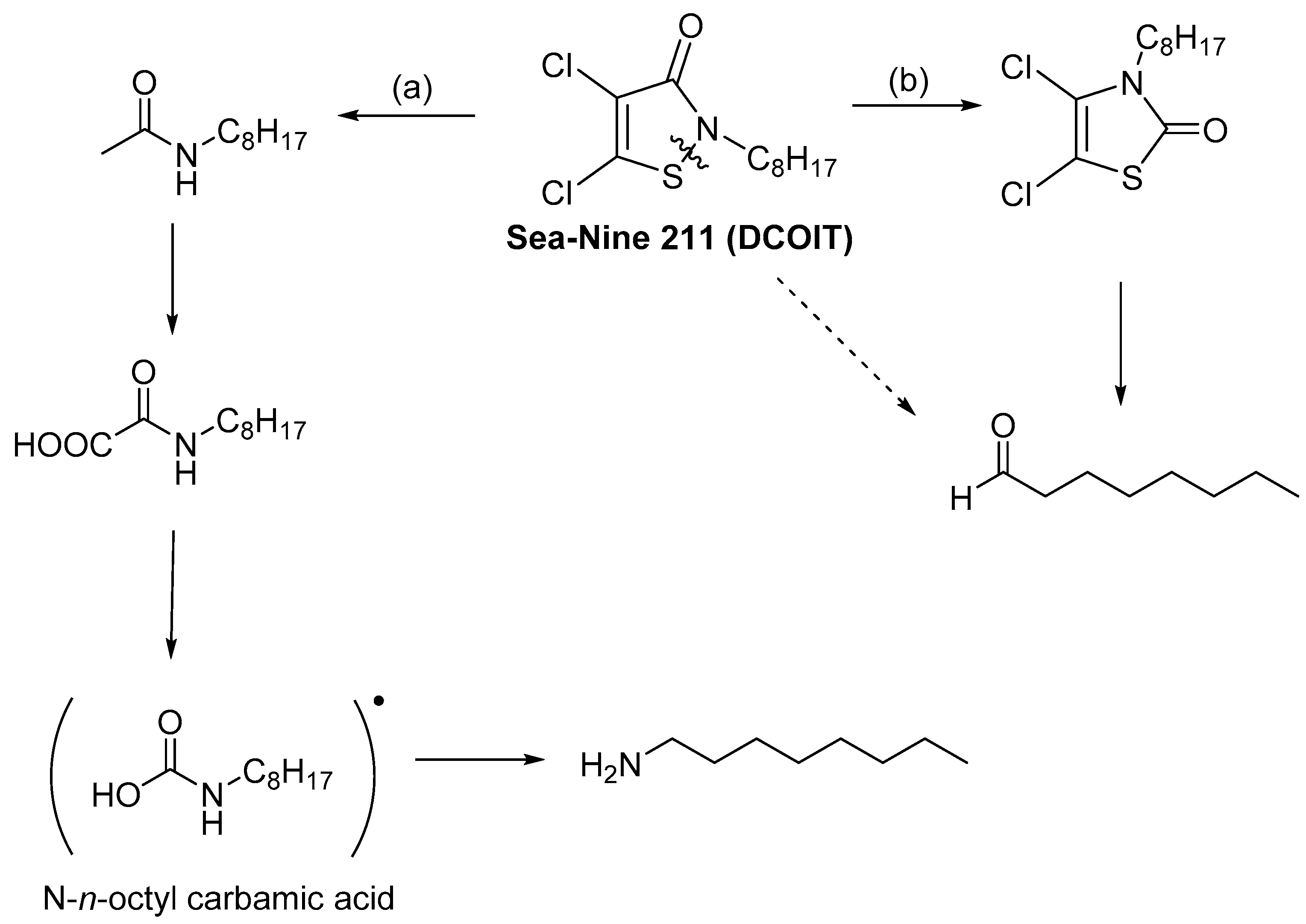
4. Stability of Isothiazolinones
5. Analysis and Determination of Isothiazolinone Biocides
6. Conclusions and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Maienfisch, P.; Edmunds, A.J.F. Thiazole and Isothiazole Ring-Containing Compounds in Crop Protection. Adv. Heterocycl. Chem. 2017, 121, 35–88. [Google Scholar]
- Ram, V.J.; Sethi, A.; Nath, M.; Pratap, R. The Chemistry of Heterocycles; Elsevier: Amsterdam, The Netherlands, 2019; pp. 149–478. [Google Scholar]
- Taubert, L.; Kraus, S.; Schulze, B. Isothiazol-3(2H)-ones, Part I: Synthesis, Reactions and Biological Activity. Sulfur Rep. 2002, 23, 79–121. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, M.; Wang, C.; Meng, X.; Zhang, W.; Chen, Z.; Crittenden, J. Electrochemical degradation of methylisothiazolinone by using Ti/SnO2-Sb2O3/a, b-PbO2 electrode: Kinetics, energy efficiency, oxidation mechanism and degradation pathway. Chem. Eng. J. 2019, 374, 626–636. [Google Scholar] [CrossRef]
- Aerts, O.; Goossens, A.; Lambert, J.; Lepoittevin, J.P. Contact allergy caused by isothiazolinone derivatives: An overview of non-cosmetic and unusual cosmetic sources. Eur. J. Dermatol. 2017, 27, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-K.; Kwon, J.-H. The fate of two isothiazolinones biocides, 5-chloro-2-methylisothiazol-3(2H)-one (CMI) and 2-methylisothiazol-3(2H)-one (MI), in liquid air fresheners and assessment of inhalation exposure. Chemosphere 2016, 144, 2270–2276. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Lam, J.C.W. SeaNine 211 as antifouling biocide: A coastal pollutant of emerging concern. J. Environ. Sci. 2017, 61, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Xu, Y.; Wang, W.; Qian, P. Degradation kinetics of a potent antifouling agent, butenolide, under various environmental conditions. Chemosphere 2015, 119, 1075–1083. [Google Scholar] [CrossRef]
- Schwensen, J.F.; Johansen, J.D. Isothiazolinones. In Kanerva’s Occupational Dermatology, 3rd ed.; Johansen, J., Rustemeyer, T., Elsner, P., Maibach, H., Eds.; Springer: Cham, Switzerland, 2019; pp. 1–14. [Google Scholar]
- Herman, A.; Aerts, O.; de Montjoye, L.; Tromme, I.; Goossens, A.; Baeck, M. Isothiazolinone derivatives and allergic contact dermatitis: A review and update. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 267–276. [Google Scholar] [CrossRef]
- Crow, W.D.; Leonard, N.J. A synthesis of 3-isothiazolones. Tetrahedron Lett. 1964, 5, 1477–1480. [Google Scholar] [CrossRef]
- Lewis, S.N.; Miller, G.A.; Hausman, M.; Szamborski, E.C. Isothiazoles I: 4-isothiazolin-3-ones. A general synthesis from 3,3′-dithiodipropionamides. J. Heterocycl. Chem. 1971, 8, 571–580. [Google Scholar] [CrossRef]
- Tsolomitis, A.; Sandris, C. Synthesis of 2-substituted 3(2H)-isothiazolones from 2-substituted 5-aroyl-3(2H)-isothiazolones. Heterocycles 1987, 25, 569–575. [Google Scholar]
- Clerici, F.; Contini, A.; Gelmi, M.L.; Pocar, D. Isothiazoles. Part 14: New 3-aminosubstituted isothiazole dioxides and their mono- and dihalogeno derivatives. Tetrahedron 2003, 59, 9399–9408. [Google Scholar] [CrossRef]
- Beeley, N.R.A.; Harwood, L.M.; Hedger, P.C. A general synthesis of N-substituted lsothiazol-3(2H)-ones. J. Chem. Soc. Perkin Trans. 1994, 1, 2245–2251. [Google Scholar] [CrossRef]
- Weiler, E.D.; Petigara, R.B.; Wolfersberger, M.H.; Miller, G.A.; Isothiazoles, I.X. Halogenation of 2-substituted-4-isothiazolin-3-ones. J. Heterocycl. Chem. 1977, 14, 627–630. [Google Scholar] [CrossRef]
- McKibben, M.; McClelland, E.W. XXI-Production and reactions of 2-dithiobenzoyl. J. Chem. Soc. Trans. 1923, 123, 170–173. [Google Scholar] [CrossRef]
- McClelland, E.W.; Gait, A.J. CXXVIII-Derivatives of 2-keto-1: 2-dihydrobenzisothiazole. J. Chem. Soc. 1926, 129, 921–925. [Google Scholar] [CrossRef]
- Katz, L.; Schroeder, W. Novel methods for the preparation of benzisothiazolones. 2-benzalaminobenzisothiazolones. J. Org. Chem. 1954, 19, 103–114. [Google Scholar] [CrossRef]
- Miura, Y. Electron Spin Resonance Spectroscopic Study of Cyclic Thiocarboxamidyl Radicals, 3-Oxo-1,2-Benzisothiazolin-2-Yls: Complete Evaluation of ESR Parameters by Measuring 17O and 33s Hyperfine Splittings and Comparison of the ESR Parameters with Acyclic Analog. J. Org. Chem. 1988, 53, 2850–2852. [Google Scholar] [CrossRef]
- Chiyoda, T.; Iida, K.; Takatori, K.; Kajiwara, M. Convenient Synthesis of 1,2-Benzisothiazol-3(2H)-ones by Cyclization Reaction of Acyl Azide. Synlett 2000, 2000, 1427–1428. [Google Scholar]
- Jin, C.K.; Moon, J.-K.; Lee, W.S.; Nam, K.S. Process Development of 1,2-Benzisothiazolin-3(2H)-one by Replacing of the Toxic Materials. Synlett 2003, 2003, 1967–1968. [Google Scholar]
- Yang, K.; Zhang, H.; Niu, B.; Tang, T.; Ge, H. Benzisothiazol-3-ones through a Metal-Free Intramolecular N–S Bond Formation. Eur. J. Org. Chem. 2018, 2018, 5520–5523. [Google Scholar] [CrossRef]
- Wang, F.; Chen, C.; Deng, G.; Xi, C. Concise approach to benzisothiazol-3(2H)-one via copper-catalyzed tandem reaction of o-bromobenzamide and potassium thiocyanate in water. J. Org. Chem. 2012, 77, 4148–4151. [Google Scholar] [CrossRef] [PubMed]
- Krasikova, V.; Katkevics, M. Preparation of benzisothiazolones from 2-bromobenzamides and sulfur under copper catalysis conditions. Chem. Heterocycl. Compd. 2013, 48, 1684–1690. [Google Scholar] [CrossRef]
- Li, T.; Yang, L.; Ni, K.; Shi, Z.; Li, F.; Chen, D. An efficient approach to construct benzisothiazol-3(2H)-ones via copper-catalyzed consecutive reaction of 2-halobenzamides and carbon disulfide. Org. Biomol. Chem. 2016, 14, 6297–6303. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.M. The mechanism of action of isothiazolone biocides. In Proceedings of the NACE-International Corrosion Conference Series; Nace International: San Diego, CA, USA, 2006; pp. 060901–0609017. [Google Scholar]
- Morley, J.O.; Kapur, A.J.O.; Charlton, M.H. Structure-activity relationships in 3-isothiazolones. Org. Biomol. Chem. 2005, 3, 3713–3719. [Google Scholar] [CrossRef] [PubMed]
- Collier, P.J.; Ramsey, A.; Waigh, R.D.; Douglas, K.T.; Austin, P.; Gilbert, P. Chemical reactivity of some isothiazolone biocides. J. Appl. Bacteriol. 1990, 69, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Collier, P.J.; Ramsey, A.J.; Austin, P.; Gilbert, P. Growth inhibitory and biocidal activity of some isothiazolonone biocides. J. Appl. Bacteriol. 1990, 69, 569–577. [Google Scholar] [CrossRef]
- Green, P.N. Efficacy of biocides on laboratory-generated Legionella biofilms. Lett. Appl. Microbiol. 1993, 17, 158–161. [Google Scholar] [CrossRef]
- Rushton, L.; Sass, A.; Baldwin, A.; Dowson, C.G.; Donoghue, D.; Mahenthiralingam, E. Key role for efflux in the preservative susceptibility and adaptive resistance of Burkholderia cepacia complex bacteria. Antimicrob. Agents Chemother. 2013, 57, 2972–2980. [Google Scholar] [CrossRef]
- Williams, T.M. Isothiazolone Biocides in Water Treatment Applications; CORROSION/2004, 2004 (New Orleans, LA, U.S.A.), Paper #04083; NACE International: Houston, TX, USA, 2004. [Google Scholar]
- Diehl, M.A.; Chapman, J.S. Association of the biocide 5-chloro-2-methylisothiazol-3-one with Pseudomonas aeruginosa and Pseudomonas fluorescens. Int. Biodeterior. Biodegrad. 1999, 44, 191–199. [Google Scholar] [CrossRef]
- Sabev, H.A.; Handley, P.S.; Robson, G.D. Fungal colonization of soil-buried plasticized polyvinyl chloride (pPVC) and the impact of incorporated biocides. Microbiology 2006, 152, 1731–1739. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Jiang, X.Q.; Ye, Y.Y.; Teng, H.K.; Wang, B.; Zhao, F.; Hu, Z.W. Preparation and antibacterial activity of one novel leather material. Adv. Mater. Res. 2014, 886, 337–340. [Google Scholar] [CrossRef]
- Willi, R.; Pfab, F.; Zilker, T.; Buters, J.; Schalock, P.; Huss-Marp, J.; Todorova, A.; Ring, J.; Darsow, U. Danger from the workplace: Allergic contact dermatitis from the first exposure to isothiazolinones. Contact Dermat. 2011, 64, 361–362. [Google Scholar] [CrossRef] [PubMed]
- Schwensen, J.F.; Menné, T.; Veien, N.K.; Funding, A.T.; Avnstorp, C.; Østerballe, M.; Andersen, K.E.; Paulsen, E.; Mørtz, C.G.; Sommerlund, M.; et al. Occupational contact dermatitis in blue-collar workers: Results from a multicentre study from the Danish Contact Dermatitis Group (2003–2012). Contact Dermat. 2014, 71, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Boonstra, M.B.; Christoffers, W.A.; Coenraads, P.J.; Schuttelaar, M.L.A. Patch test results of hand eczema patients: Relation to clinical types. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 940–947. [Google Scholar] [CrossRef] [PubMed]
- Aerts, O.; Baeck, M.; Constandt, L.; Dezfoulian, B.; Jacobs, M.C.; Kerre, S.; Lapeere, H.; Pierret, L.; Wouters, K.; Goossens, A. The dramatic increase in the rate of methylisothiazolinone contact allergy in Belgium: A multicentre study. Contact Dermat. 2014, 71, 41–48. [Google Scholar] [CrossRef]
- Urwin, R.; Warburton, K.; Carder, M.; Turner, S.; Agius, R.; Wilkinson, S.M. Methylchloroisothiazolinone and methylisothiazolinone contact allergy: An occupational perspective. Contact Dermat. 2015, 72, 381–386. [Google Scholar] [CrossRef]
- Nasir, S.; Goldsmith, P. Anogenital allergic contact dermatitis caused by methylchloroisothiazolinone, methylisothiazolinone and topical clotrimazole with subsequent generalized exanthem triggered by oral fluconazole. Contact Dermat. 2016, 74, 296–297. [Google Scholar] [CrossRef]
- Diepgen, T.L.; Ofenloch, R.F.; Bruze, M.; Bertuccio, P.; Cazzaniga, S.; Coenraads, P.J.; Elsner, P.; Goncalo, M.; Svensson; Naldi, L. Prevalence of contact allergy in the general population in different European regions. Br. J. Dermatol. 2016, 174, 319–329. [Google Scholar] [CrossRef]
- Aerts, O.; Meert, H.; Romaen, E.; Leysen, J.; Matthieu, L.; Apers, S.; Lambert, J.; Goossens, A. Octylisothiazolinone, an additional cause of allergic contact dermatitis caused by leather: Case series and potential implications for the study of cross-reactivity with methylisothiazolinone. Contact Dermat. 2016, 75, 276–284. [Google Scholar] [CrossRef]
- Todberg, T.; Opstrup, M.S.; Johansen, J.D.; Hald, M. Occupational facial contact dermatitis caused by methylchloroisothiazolinone/methylisothiazolinone in a stainless steel aerosol spray. Contact Dermat. 2017, 77, 173–174. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.J.; Park, D.U.; Yoon, J.; Lee, E.; Yang, S.I.; Kim, Y.H.; Lee, S.Y.; Hong, S.J. Effects of a mixture of chloromethylisothiazolinone and methylisothiazolinone on peripheral airway dysfunction in children. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Goodier, M.C.; Ljungberg, L.; Persson, C.; Engfeldt, M.; Bruze, M.; Warshaw, E.M. Allergic Contact Dermatitis from Methylisothiazolinone in Residential Wall Paint. Dermatitis 2017, 28, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Aalto-Korte, K.; Suuronen, K. Patterns of concomitant allergic reactions in patients suggest cross-sensitization between octylisothiazolinone and methylisothiazolinone. Contact Dermat. 2017, 77, 385–389. [Google Scholar] [CrossRef]
- Lee, E.; Son, S.K.; Yoon, J.; Cho, H.J.; Yang, S.I.; Jung, S.; Do, K.H.; Cho, Y.A.; Lee, S.Y.; Park, D.U.; et al. Two cases of chloromethylisothiazolinone and methylisothiazolinone-associated toxic lung injury. J. Korean Med. Sci. 2018, 33, e119. [Google Scholar] [CrossRef]
- Ryu, S.-H.; Park, D.-U.; Lee, E.; Park, S.; Lee, S.-Y.; Jung, S.; Hong, S.-B.; Park, J.; Hong, S.-J. Humidifier disinfectant and use characteristics associated with lung injury in Korea. Indoor Air 2019, 29, 735–747. [Google Scholar] [CrossRef]
- Neisius, U.; Brand, P.; Plochmann, S.; Saloga, J.; Knop, J.; Becker, D. Detection of increased tyrosine phosphorylation in murine Langerhans cells after stimulation with contact sensitizers. Arch. Dermatol. Res. 1999, 291, 22–27. [Google Scholar] [CrossRef]
- Schwensen, J.F.; Menné Bonefeld, C.; Zachariae, C.; Agerbeck, C.; Petersen, T.H.; Geisler, C.; Bollmann, U.E.; Bester, K.; Johansen, J.D. Cross-reactivity between methylisothiazolinone, octylisothiazolinone and benzisothiazolinone using a modified local lymph node assay. Br. J. Dermatol. 2017, 176, 176–183. [Google Scholar] [CrossRef]
- Stejskal, V.D.; Forsbeck, M.; Nilsson, R. Lymphocyte transformation test for diagnosis of isothiazolinone allergy in man. J. Investig. Dermatol. 1990, 94, 798–802. [Google Scholar] [CrossRef]
- Lee, J.; Lee, H.; Jang, S.; Hong, S.-H.; Kim, W.J.; Ryu, S.M.; Park, S.-M.; Lee, K.-H.; Cho, S.-J.; Yang, S.-R. CMIT/MIT induce apoptosis and inflammation in alveolar epithelial cells through p38/JNK/ERK1/2 signalling pathway. Mol. Cell Toxicol. 2019, 15, 41–48. [Google Scholar] [CrossRef]
- Park, E.-J.; Seong, E. Methylisothiazolinone induces apoptotic cell death via matrix metalloproteinase activation in human bronchial epithelial cells. Toxicol. Vitro 2020, 62, 104661. [Google Scholar] [CrossRef] [PubMed]
- Arning, J.; Dringen, R.; Schmidt, M.; Thiessen, A.; Stolte, S.; Matzke, M.; Bottin-Weber, U.; Caesar-Geertz, B.; Jastorff, B.; Ranke, J. Structure-activity relationships for the impact of selected isothiazol-3-one biocides on glutathione metabolism and glutathione reductase of the human liver cell line Hep G2. Toxicology 2008, 246, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Arning, J.; Matzke, M.; Stolte, S.; Nehen, F.; Bottin-Weber, U.; Böschen, A.; Abdulkarim, S.; Jastorff, B.; Ranke, J. Analyzing cytotoxic effects of selected isothiazol-3-one biocides using the toxic ratio concept and structure-activity relationship considerations. Chem. Res. Toxicol. 2009, 22, 1954–1961. [Google Scholar] [CrossRef] [PubMed]
- Tong, Z.B.; Hogberg, H.; Kuo, D.; Sakamuru, S.; Xia, M.; Smirnova, L.; Hartung, T.; Gerhold, D. Characterization of three human cell line models for high-throughput neuronal cytotoxicity screening. J. Appl. Toxicol. 2017, 37, 167–180. [Google Scholar] [CrossRef]
- Mesnage, R.; Defarge, N.; Spiroux De Vendômois, J.; Séralini, G.E. Major pesticides are more toxic to human cells than their declared active principles. BioMed Res. Int. 2014, 2014, 179691. [Google Scholar] [CrossRef]
- Friis, U.F.; Menné, T.; Flyvholm, M.A.; Bonde, J.P.E.; Lepoittevin, J.P.; Le Coz, C.J.; Johansen, J.D. Isothiazolinones in commercial products at Danish workplaces. Contact Dermat. 2014, 71, 65–74. [Google Scholar] [CrossRef]
- Regulation (EU) No 528/2012 of the European Parliament and of the Council concerning the making available on the market and use of biocidal products. Off. J. 2012, 167, 1.
- Florio, J.; Miller, D. Handbook of Coatings Additives, 2nd ed.; Marcel Dekker Inc.: New York, NY, USA, 2004. [Google Scholar]
- Lundov, M.; Kolarik, B.; Bossi, R.; Gunnarsen, L.; Johansen, J. Emission of Isothiazolinones from Water-Based Paints. Environ. Sci. Technol. 2014, 48, 6989–6994. [Google Scholar] [CrossRef]
- Hofmann, M.A.; Giménez-Arnau, A.; Aberer, W.; Bindslev Jensen, C.; Zuberbier, T. MI (2 methyl 4 isothiazolin 3 one) contained in detergents is not detectable in machine washed textiles. Clin. Transl. Allergy 2018, 8, 1. [Google Scholar] [CrossRef]
- Mardones, L.; Legnoverde, M.; Pereyra, A.; Basaldella, E. Long-lasting isothiazolinone-based biocide obtained by encapsulation in micron-sized mesoporous matrices. Prog. Org. Coat. 2018, 119, 155–163. [Google Scholar] [CrossRef]
- Tanji, Y.; Nishihara, T.; Miyanaga, K. Iron dependent degradation of an isothiazolone biocide (5-chloro-2-methyl-4-isothiazolin-3-one). Biofouling 2007, 23, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, T. Reaction of Isothiazolones with Amines. Phosphorus Sulfur Silicon Relat. Elem. 1999, 153, 367–368. [Google Scholar] [CrossRef]
- Williams, T. The Mechanism of Action of Isothiazolone Biocides. PPChem 2007, 9, 14–22. [Google Scholar]
- Krzeminski, S.F.; Brackett, C.K.; Fisher, J.D. Fate of Microbicidal 3-Isothiazolone Compounds in the Environment: Modes and Rates of Dissipation. J. Agric. Food Chem. 1975, 23, 1060–1068. [Google Scholar] [CrossRef] [PubMed]
- Krzeminski, S.F.; Brackett, C.K.; Fisher, J.D.; Spinnler, J.F. Fate of Microbicidal 3-Isothiazolone Compounds in the Environment: Products of Degradation. J. Agric. Food Chem. 1975, 23, 1068–1075. [Google Scholar] [CrossRef]
- Barman, B.N.; Preston, H.G. The effects of pH on the degradation of isothiazolone biocides. Tribol. Int. 1992, 25, 281–287. [Google Scholar] [CrossRef]
- Barman, B.N. Influence of temperature on the degradation of isothiazolone biocides in aqueous media and in a metalworking fluid concentrate. Lubr. Eng. 1993, 50, 351–355. [Google Scholar]
- Li, A.; Chen, Z.; Wu, Q.-Y.; Huang, M.-H.; Liu, Z.-Y.; Chen, P.; Mei, L.-C.; Hu, H.-Y. Study on the removal of benzisothiazolinone biocide and its toxicity: The effectiveness of ozonation. Chem. Eng. J. 2016, 300, 376–383. [Google Scholar] [CrossRef]
- Lugg, M.J. Photodegradation of the biocide 1,2-benziothiazolin-3-one used in a paper-based jointing material. Int. Biodeterior. Biodegrad. 2001, 48, 252–254. [Google Scholar] [CrossRef]
- Wang, T.; Wu, Q.; Wang, W.; Chen, Z.; Li, B.; Li, A.; Liu, Z.; Hu, H. Self-sensitized photodegradation of benzisothiazolinone by low-pressure UV-C irradiation: Kinetics, mechanisms, and the effect of media. Sep. Purif. Technol. 2017, 189, 419–424. [Google Scholar] [CrossRef]
- Bollmann, U.E.; Minelgaite, G.; Schlusener, M.; Ternes, T.; Vollertsen, J.; Bester, K. Photodegradation octylisothiazolinone and semi-field emissions from facade coatings. Sci. Rep. 2017, 7, 41501. [Google Scholar] [CrossRef] [PubMed]
- Chand, R.; Tulucan, T.; Aburlacitei, M. Investigation of biocide biodegradation in wastewater under laboratory set-up in anaerobic and aerobic with substrate conditions. J. Civ. Environ. Eng. 2018, 8, 295. [Google Scholar] [CrossRef]
- Shade, W.D.; Hurt, S.S.; Jacobson, A.H.; Reinert, K.H. Ecological Risk Assessment of a Novel Marine Antifoulant. In Environmental Toxicology and Risk Assessment; Gorsuch, J.W., Dwyer, F.W., Ingersoll, C.M., LaPoint, T.W., Eds.; American Society for Testing and Materials: West Conshohocken, PA, USA, 1993; Volume 2, pp. 381–408. [Google Scholar]
- Jacobson, A.H.; Willingham, G.L. Sea-Nine antifoulant: An environmentally acceptable alternative to organotin antifoulants. Sci. Total Environ. 2000, 258, 103–110. [Google Scholar] [CrossRef]
- Sakkas, V.A.; Konstantinou, I.K.; Triantafyllos, A.A. Aquatic phototransformation study of the antifouling agent Sea-Nine 211: Identification of byproducts and the reaction pathway by gas chromatography-mass spectroscopy. J. Chromatogr. A 2002, 959, 215–227. [Google Scholar] [CrossRef]
- Sakkas, V.A.; Konstantinou, I.K.; Albanis, T.A. Photochemical fate of organic booster biocides in the aquatic environment. In Antifouling Paint Biocides; The Handbook of Environmental Chemistry; Konstantinou, I.K., Ed.; Springer: Berlin, Germany, 2006; Volume 5, pp. 171–200. [Google Scholar]
- Bollmann, U.E.; Fernández-Calviño, D.; Brandt, K.; Storgaard, M.; Sanderson, H.; Bester, K. Biocide runoff from building facades: Degradation kinetics in soil. Environ. Sci. Technol. 2017, 51, 3694–3702. [Google Scholar] [CrossRef] [PubMed]
- Rafoth, A.; Gabriel, S.; Sacher, F.; Brauch, H. Analysis of isothiazolinones in environmental waters by gas chromatography–mass spectrometry. J. Chromatogr. A 2007, 1164, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Jeannot, M.A. Extraction-Liquid-Phase Microextraction. In Encyclopedia of Separation Science; Wilson, I.D., Ed.; Academic Press: Cambridge, MA, USA, 2007; pp. 1–5. [Google Scholar]
- Goodier, M.C.; Siegel, P.D.; Zang, L.; Warshaw, E.M. Isothiazolinone in residential interior wall paint: A high-performance liquid chromatographic-mass spectrometry analysis. Dermatitis 2018, 29, 332–338. [Google Scholar] [CrossRef]
- Nakashima, H.; Matsunaga, I.; Miyano, N.; Kitagawa, M. Determination of Antimicrobial agents in non-formalin adhesives for wallpaper. J. Health Sci. 2000, 46, 447–457. [Google Scholar] [CrossRef][Green Version]
- Heo, J.J.; Kim, U.; Oh, J. Simultaneous quantitative analysis of four isothiazolinones and 3-iodo-2-propynyl butyl carbamate in hygienic consumer products. Environ. Eng. Res. 2019, 24, 137–143. [Google Scholar] [CrossRef]
- Paul, L.D.S.; Nicolas, M.; Quivet, E. Development of quantification methods for studying the emissions of isothiazolinones from building and consumer products into indoor environments. IOP Conf. Ser. Mater. Sci. Eng. 2019, 609, 1–6. [Google Scholar]
- Baranowska, I.; Wojciechowska, I. The determination of preservatives in Cosmetics and environmental waters by HPLC. Pol. J. Environ. Stud. 2013, 22, 1609–1625. [Google Scholar]
- Chen, Z.F.; Ying, G.G.; Lai, H.J.; Chen, F.; Su, H.C.; Liu, Y.S.; Peng, F.Q.; Zhao, J.L. Determination of biocides in different environmental matrices by use of ultra-high-performance liquid chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2012, 40, 3175–3188. [Google Scholar] [CrossRef] [PubMed]
- Bester, K.; Lamani, X. Determination of biocides as well as some biocide metabolites from facada run-off waters by solid phase extraction and high performance liquid chromatographic separation and tendem mass spectrometry detection. J. Chromatogr. A 2010, 1217, 5204–5214. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Rivera, G.; Llompart, M.; Lores, M.; Garcia-Jares, C. Preservatives in Cosmetics: Regulatory Aspects and Analytical Methods. In Analysis of Cosmetic Products, 2nd ed.; Salvador, A., Chisvert, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 175–224. [Google Scholar]
- Alvarez-Rivera, G.; Dagnac, T.; Lores, M.; Garcia-Jares, C.; Sanchez-Prado, L.; Lamas, J.P.; Llompart, M. Determination of isothiazolinone preservatives in cosmetics and household products by matrix solid-phase dispersion followed by high-performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2012, 1270, 41–50. [Google Scholar] [CrossRef]
- Garcia-Hidalgo, E.; Sottas, V.; Goetz, N.V.; Hauri, U.; Bogdal, C.; Hungerbuhler, K. Occurrence and concentrations of isothiazolinones in detergents and cosmetics in switzerland. Contact Dermat. 2016, 76, 96–106. [Google Scholar] [CrossRef]
- Lin, Q.B.; Wang, T.J.; Song, H.; Li, B. Analysis of isothiazolinone biocides in paper for food packaging by ultra-high-performance liquid chromatography-tandem mass spectrometry. Food Addit. Contam. Part A 2010, 27, 1775–1781. [Google Scholar] [CrossRef]
- Wu, T.; Wang, C.; Wang, X.; Ma, Q. Simultaneous determination of 21 preservatives in cosmetics by ultra performance liquid chromatography. Int. J. Cosmet. Sci. 2008, 30, 367–372. [Google Scholar] [CrossRef]
- García, E.; Giráldez, I.; Montoya, M.R.; Morales, E. Determination of booster biocides in sediments by focused ultrasound-assisted extraction and stir bar sorptive extraction-thermal desorption-gas chromatography-mass spectrometry. Microchem. J. 2020, 152, 104445. [Google Scholar] [CrossRef]
- Zhong, H.; Li, Z.; Chen, S.; Zeng, Y.; Zheng, J.; Zeng, Y.; Li, D. Simultaneous Quantitative Analysis of Six Isothiazolinones in Water-Based Adhesive Used for Food Contact Materials by High-Performance Liquid Chromatography-Tandem Mass Spectrometry (HPLC-MS/MS). Molecules 2019, 24, 3894. [Google Scholar] [CrossRef]
- Zhou, X.; Li, X.; Chen, Z.; Ye, C.; Zhou, Y.; Meng, D. Determination of three isothiazolinone biocides in water-borne adhesives by High Performance Liquid Chromatography. SePu Chin. J. Chromatogr. 2015, 33, 75–79. [Google Scholar] [CrossRef]
- Wittenberg, J.B.; Canas, B.J.; Zhou, W.; Wang, P.G.; Rua, D.; Krynitsky, A.J. Determination of methylisothiazolinone and methylchloroisothiazolinone in cosmetic products by ultra high performance liquid chromatography with tandem mass spectrometry. J. Sep. Sci. 2015, 38, 2983–2988. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, T.; Isama, K.; Ikarashi, Y. Analysis of isothiazolinone preservatives in polyvinyl alcohol cooling towels used in Japan. Environ. Sci. Health A Toxic Hazard. Subst. Environ. Eng. 2014, 49, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Rosero-Moreano, M.; Canellas, E.; Nerín, C. Three-phase hollow-fiber liquid-phase microextraction combined with HPLC-UV for the determination of isothiazolinones biocides in adhesives used for food packaging materials. J. Sep. Sci. 2014, 37, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Baranowska, I.; Wojciechowska, I.; Solarz, N.; Krutysza, E. Determination of Preservatives in Cosmetics, Cleaning Agents and Pharmaceuticals Using Fast Liquid Chromatography. J. Chromatogr. Sci. 2014, 52, 88–94. [Google Scholar] [CrossRef]
- Kawakami, T.; Isama, K.; Nishimura, T. Analysis of Isothiazolinones and other preservatives in gel-products used for cooling in Japan. J. Environ. Chem. 2012, 22, 205–211. [Google Scholar] [CrossRef][Green Version]
- Cha, N.-R.; Lee, J.-K.; Jeong, H.-J.; Cho, J.-C.; Kim, M.-J.; Lee, S.-Y. Determination of 19 Preservatives in Various Matrices by High-Performance Liquid Chromatography. Anal. Lett. 2012, 45, 2148–2160. [Google Scholar] [CrossRef]
- Speksnijder, P.; Ravestijin, J.V.; Voogt, P. Trace analysis of isothiazolinones in water samples by large-volume direct injection liquid cromatography tandem mass spectrometry. J. Chromatogr. A 2010, 1217, 5184–5189. [Google Scholar] [CrossRef]
- Singer, H.; Jaus, S.; Hanke, I.; Lück, A.; Hollender, J.; Alder, A.C. Determination of biocides and pesticides by on-line solid phase extraction coupled with mass spectrometry and their behaviour in wastewater and surface water. Environ. Pollut. 2010, 158, 3054–3064. [Google Scholar] [CrossRef]
- Matissek, R.; Lehnguth, R. Zur analytik mikrobiocider isothiazolone. Fresenius Z. Anal. Chem. 1988, 332, 270–274. [Google Scholar] [CrossRef]

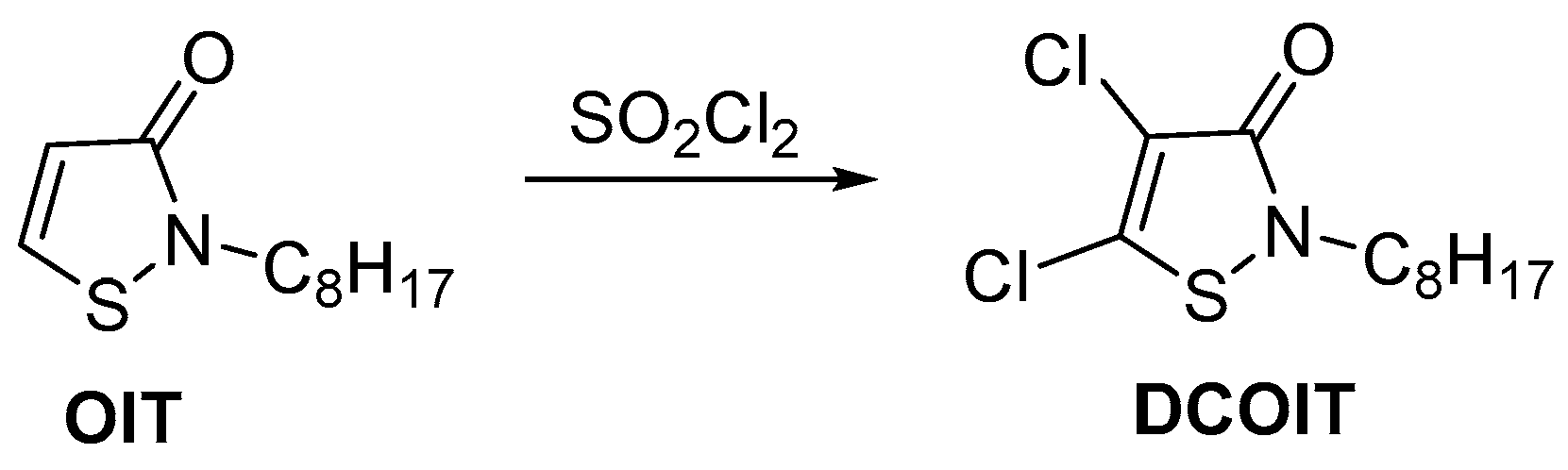
| Biocides | MIC (µg/mL) | |
|---|---|---|
| S. pombe | E. coli | |
| MI | 245 | 41 |
| MCI | 2.6 | 0.5 |
| BIT | 35.5 | 14.4 |
| Biocides | Aspergillus niger | Saccharomyces cerevisiae | ||
|---|---|---|---|---|
| MIC (mg/L) | MBC (mg/L) | MIC (mg/L) | MBC (mg/L) | |
| MI | 166 ± 52 | 300 ± 0 | 60 ± 0 | 87.0 ± 10.3 |
| MCI | 0.35 ± 0.05 | 0.42 ± 0.04 | 0.58 ± 0.10 | 0.63 ± 0.10 |
| MCI/MI | 0.40 ± 0.07 | 0.47 ± 0.05 | 0.71 ± 0.10 | 0.76 ± 0.20 |
| OIT | 0.05 ± 0.01 | 0.05 ± 0.01 | 0.56 ± 0.10 | 0.65 ± 0.20 |
| DOIT | 0.12 ± 0.01 | 0.23 ± 0.14 | 0.55 ± 0.10 | 0.79 ± 0.20 |
| Biocides | GR Inhibition in HepG2 Cells IC50 (µM) | LogP0W | EC50 HepG2 (µM) | EC50 V.fischeri (µM) | EC50 S.vacuolatus (µM) |
|---|---|---|---|---|---|
| MI | >1000 | −0.49 | 130 | 14 | 4.9 |
| MCI | 7.0 | 0.53 | 13 | 0.58 | 0.6 |
| OIT | >1000 | 3.30 | 21 | 0.94 | 0.42 |
| DCOIT | 13.2 | 4.79 | 16 | 0.43 | 0.38 |
| Commercial Products | Maximum Admitted Concentration of Biocide |
|---|---|
| Cosmetics 1 | MCI/MI < 15 ppm 3 |
| MI < 100 ppm 4 | |
| BIT forbidden 5 | |
| OIT forbidden 6 | |
| DCOIT forbidden 7 | |
| Paints 2, glues 2, detergents 1 | MCI/MI < 15 ppm |
| MI < 300 ppm | |
| BIT < 360 ppm | |
| OIT insufficient data | |
| DCOIT insufficient data | |
| Industrial biocides 2 | MI, MCI, and BIT used at high concentrations (e.g., >5000 ppm) |
| OIT insufficient data | |
| DCOIT insufficient data |
| Analyte | Matrix | Sample Preparation | Analytical Technique | Reference |
|---|---|---|---|---|
| Sea-Nine 211 (DCOIT) | Marine sediments | FUSE/SPE clean-up; SBSE/TD pre-concentration step | GC-MS/MS | [97] |
| MCI, BIT, OIT, DCOIT | Water-based adhesive/Food contact material | Vortex extraction | HPLC-MS/MS | [98] |
| MI, MCI, BIT, OIT | Human hygiene consumer products | UAE | HPLC-MS | [87] |
| MI, MCI, BIT, OIT, DCOIT | Building and consumer products | SPE; ASE; UAE | GC-MS; UPLC-MS/MS | [88] |
| MI, MCI, BIT, OIT | Residential interior wall paints | LLE | UPLC-MS/MS | [85] |
| MI, MCI, BIT, OIT | Detergents and cosmetics | UAE | LC-MS | [94] |
| MI, MCI, BIT | Water-borne adhesives | SLE | HPLC-DAD | [99] |
| MI, MCI | Cosmetic products | Mechanical shaking | UPLC-MS/MS | [100] |
| MI, MCI, BIT, OIT, DCOIT | Polyvinyl alcohol cooling towels | Mechanical shaking | LC-MS/MS | [101] |
| MI, MCI | Adhesives for food packaging | HF-LPME | HPLC-DAD | [102] |
| MI, MCI | Cosmetics, cleaning agents, pharmaceuticals | UAE | UPLC-DAD | [103] |
| MI, MCI | Cosmetics, pharmaceuticals | UAE | HPLC-DAD | [89] |
| MI, MCI, BIT, OIT | Cosmetics and household products | MSPD | HPLC-MS/MS | [93] |
| MI, CMI, OIT | Gel-products used for cooling | Mechanical shaking | LC-MS/MS | [104] |
| BIT, OIT, DCOIT | Surface water, wastewater, sediment, sludge, soil | SPE, UAE | UPLC-MS/MS | [90] |
| MI, MCI | Cosmetic products | UAE | HPLC-DAD | [105] |
| MI MCI, DCOIT | Drinking waters, surface waters, wastewater | Filtration | HPLC-MS/MS | [106] |
| MI, MCI, BIT, OIT | Paper used for food packaging | UAE | UPLC-MS/MS | [95] |
| MI, MCI, BIT, OIT, DCOIT | Facade run-off waters | SPE | HPLC-MS/MS | [91] |
| BIT, OIT | Wastewater, river water | SPE | LC-MS/MS | [107] |
| MI, MCI | Cosmetics | UAE | UPLC-DAD | [96] |
| MI, MCI, BIT, OIT, DCOI | Surface waters, groundwater, drinking water | LLE, SPE | GC-MS | [83] |
| MI, MCI, BIT, OIT | Non-formalin adhesives | LLE, SPE | GC-MS | [86] |
| MI, MCI | Cosmetics | Separation and purification by column chromatography | HPLC-DAD | [108] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, V.; Silva, C.; Soares, P.; Garrido, E.M.; Borges, F.; Garrido, J. Isothiazolinone Biocides: Chemistry, Biological, and Toxicity Profiles. Molecules 2020, 25, 991. https://doi.org/10.3390/molecules25040991
Silva V, Silva C, Soares P, Garrido EM, Borges F, Garrido J. Isothiazolinone Biocides: Chemistry, Biological, and Toxicity Profiles. Molecules. 2020; 25(4):991. https://doi.org/10.3390/molecules25040991
Chicago/Turabian StyleSilva, Vânia, Cátia Silva, Pedro Soares, E. Manuela Garrido, Fernanda Borges, and Jorge Garrido. 2020. "Isothiazolinone Biocides: Chemistry, Biological, and Toxicity Profiles" Molecules 25, no. 4: 991. https://doi.org/10.3390/molecules25040991
APA StyleSilva, V., Silva, C., Soares, P., Garrido, E. M., Borges, F., & Garrido, J. (2020). Isothiazolinone Biocides: Chemistry, Biological, and Toxicity Profiles. Molecules, 25(4), 991. https://doi.org/10.3390/molecules25040991