Copper Complexing Capacity and Trace Metal Content in Common and Balsamic Vinegars: Impact of Organic Matter
Abstract
1. Introduction
2. Results and Discussion
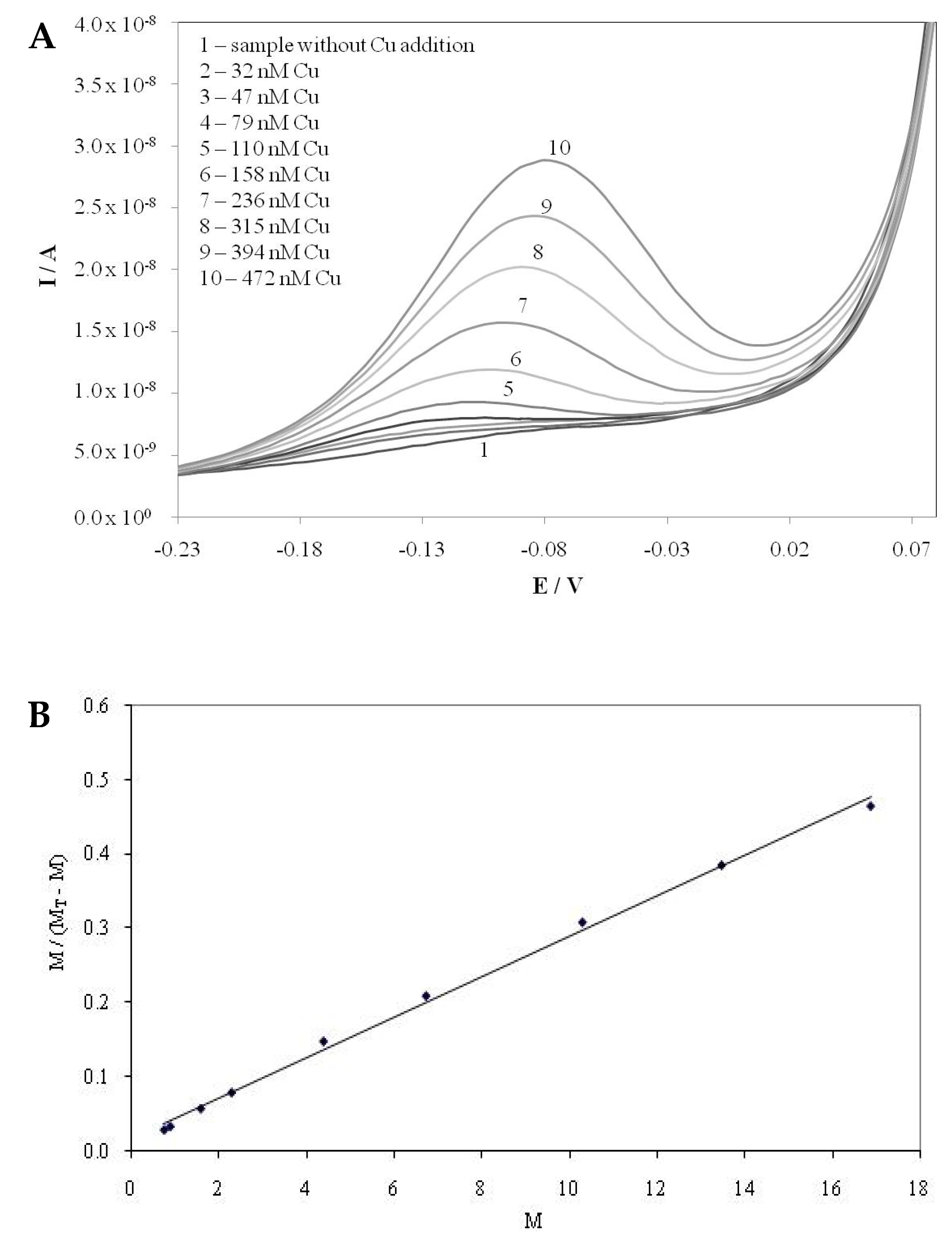
2.1. Copper Complexation
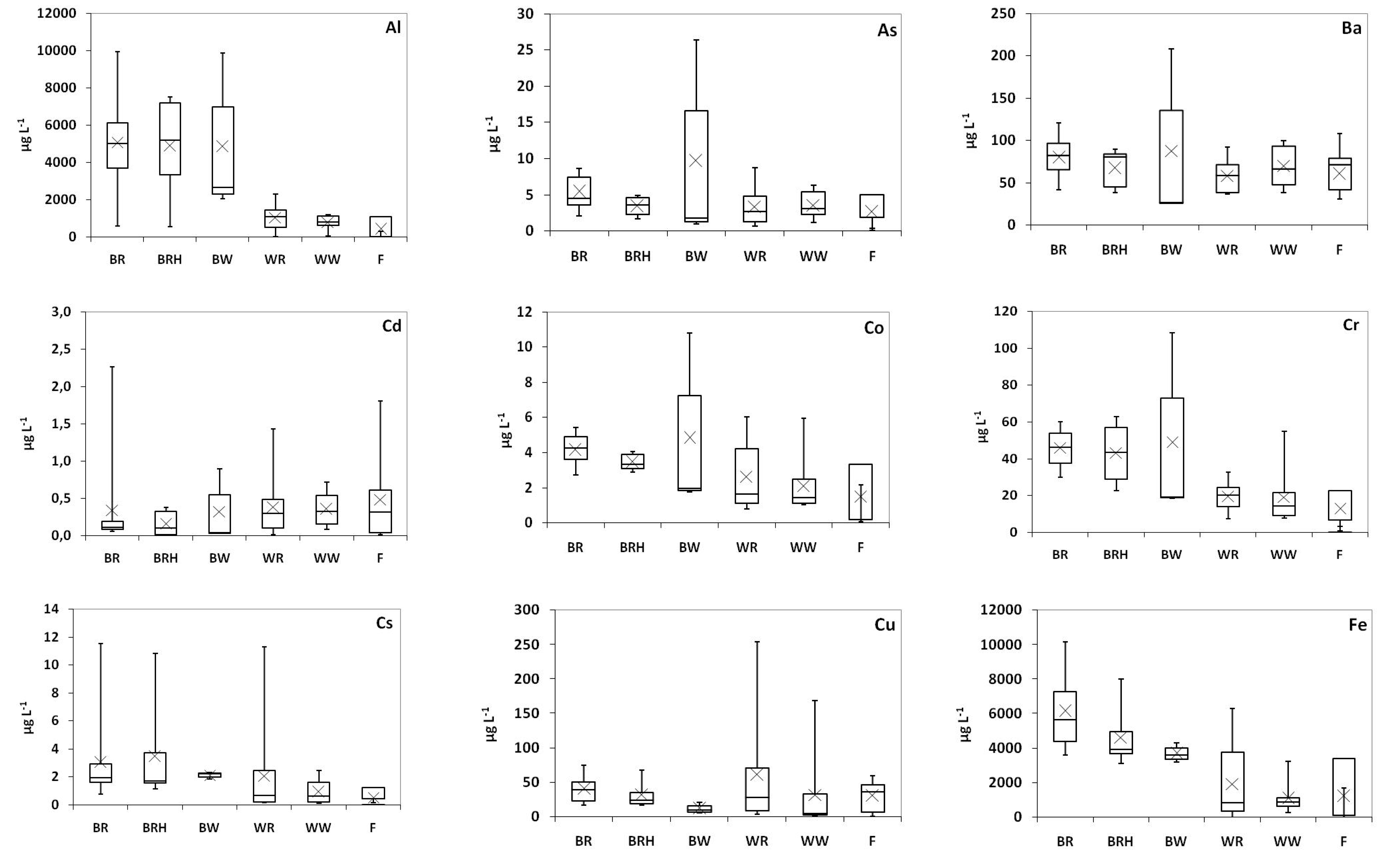
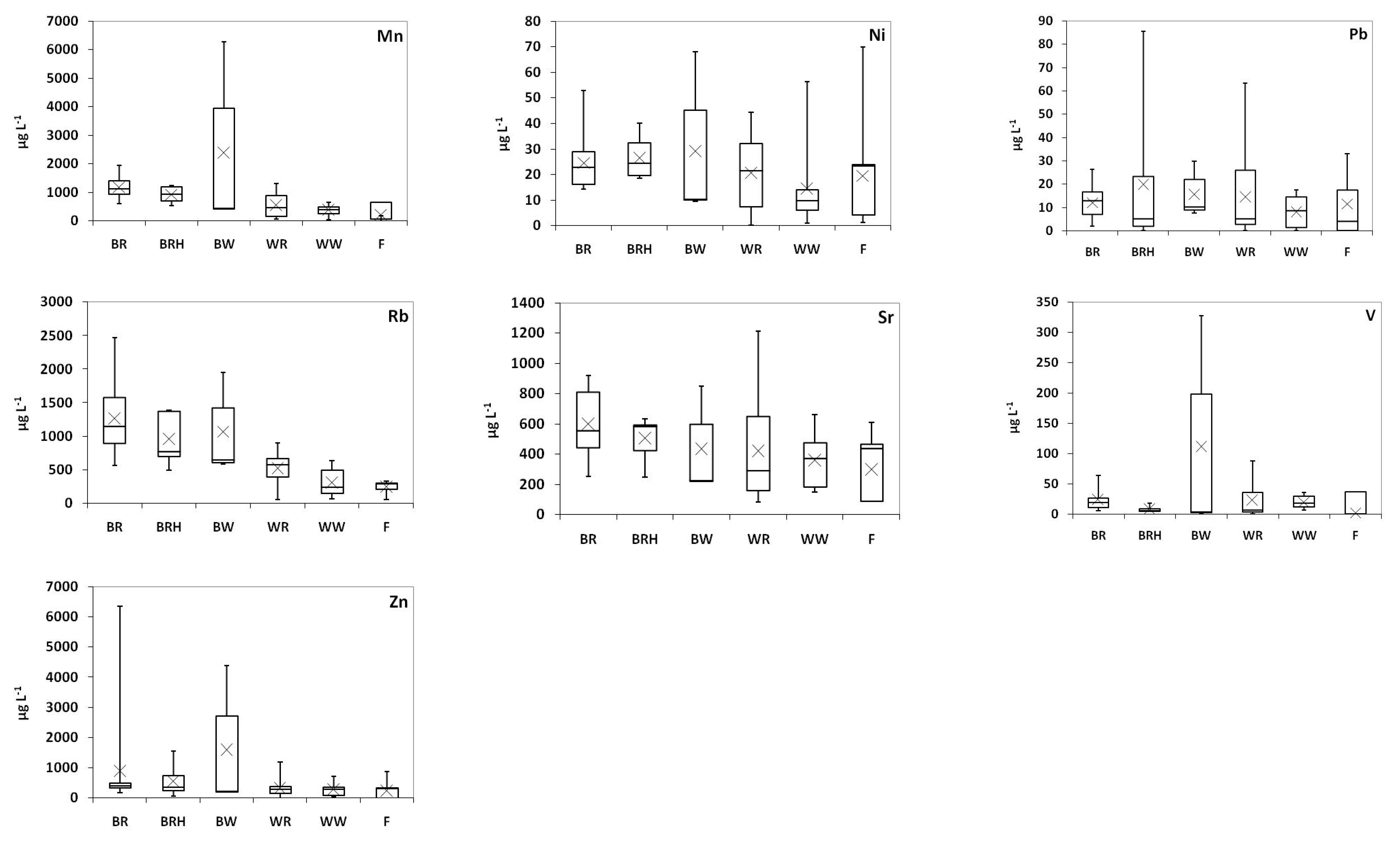
2.2. Trace Elements
3. Materials and Methods
3.1. Sampling
3.2. Electrochemical Measurements
3.2.1. Sample Preparation and Equipment
3.2.2. Copper Complexing Capacity (LT)
3.3. Trace Metals
3.4. Organic Carbon (OC) and pH
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Plessi, M. Vinegar. In Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Caballero, B., Trugo, L.C., Finglas, P.M., Eds.; Academic Press: Oxford, UK, 2003; Volume 9, pp. 5996–6004. ISBN 9780122270550. [Google Scholar]
- Budak, N.H.; Aykin, E.; Seydim, A.C.; Greene, A.K.; Guzel-Seydim, Z.B. Functional properties of vinegar. J. Food Sci. 2014, 79, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Paneque, P.; Morales, M.L.; Burgos, P.; Ponce, L.; Callejon, R.M. Elemental characterization of Andalusian wine vinegars with protected designation of origin by ICP-OES and chemometric approach. Food Control 2017, 75, 203–210. [Google Scholar] [CrossRef]
- Giudici, P.; Gullo, M.; Solieri, L. Traditional balsamic vinegar. In Vinegars of the World; Solieri, L., Giudici, P., Eds.; Springer: Milan, Italy, 2009; Volume 10, pp. 157–177. ISBN 978-0-470-0865-6. [Google Scholar]
- Mas, A.; Torija, M.J.; García-Parrilla, M.C.; Troncoso, A.M. Acetic acid bacteria and the production and quality of wine vinegar. Sci. World J. 2014, 394671. [Google Scholar] [CrossRef] [PubMed]
- Ndung’u, K.; Hibdon, S.; Veron, A.; Russell Flegal, A. Lead isotopes reveal different sources of lead in balsamic and other vinegars. Sci. Total Environ. 2011, 409, 2754–2760. [Google Scholar] [CrossRef]
- Schumann, K.; Elsenhans, B. The impact of food contaminants on the bioavailability of trace metals. J. Trace Elem. Med. Biol. 2002, 16, 139–144. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure antioxidant activity relationships of flavonoids and phenolic acids. Free Radical Bio Med. 1996, 20, 233–256. [Google Scholar] [CrossRef]
- Mira, L.; Fernandez, M.T.; Santos, M.; Rocha, R.; Florencio, M.H.; Jennings, K.R. Interactions of flavonoids with iron and copper ions: A mechanism for their antioxidant activity. Free Radic. Res. 2002, 36, 1199–1208. [Google Scholar] [CrossRef]
- Shahidi, F.; Mcdonald, J.; Chandrasekara, A.; Zhong, Y. Phytochemicals of foods, beverages and fruit vinegars: Chemistry and health effects. Asia Pac. J. Clin. Nutr. 2008, 17 (Suppl. 1), 380–382. [Google Scholar]
- Wang, Y.; Ho, C.T. Polyphenolic chemistry of tea and coffee: A century of progress. J. Agric. Food Chem. 2009, 57, 8109–8114. [Google Scholar] [CrossRef]
- Brown, P.; Markich, S. Evaluation of the free ion activity model of metal-organism interaction: Extension of the conceptual model. Aquat. Toxicol. 2000, 51, 177–194. [Google Scholar] [CrossRef]
- Chito, D.; Galceran, J.; Companys, E.; Puy, J. Determination of the complexing capacity of wine for Zn using the absence of gradients and Nernstian equilibrium stripping technique. J. Agr. Food Chem. 2013, 61, 1051–1059. [Google Scholar] [CrossRef] [PubMed]
- Vink, J.P.M. The origin of speciation: Trace metal kinetics over natural water/sediment interfaces and the consequences for bioaccumulation. Environmental Pollution 2009, 157, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Plavšić, M.; Krznarić, D.; Branica, M. Determination of the apparent copper complexing capacity of seawater by DPASV. Mar. Chem. 1982, 11, 17–31. [Google Scholar] [CrossRef]
- Steinberg, S.M.; Hodge, V.F. Copper complexation by dissolved organic matter in arid soils: A voltametric study. Environments 2018, 5, 125. [Google Scholar] [CrossRef]
- Tran, T.; Brown, B.; Nesic, S.; Tribollet, B. Investigation of the mechanisms for acetic acid corrosion of mild steel. In Proceedings of the NACE International, Corrosion Conference & Expo, Orlando, FL, USA, 17–21 March 2013. Abstract Number 2487. [Google Scholar]
- Mazinanian, N.; Odnevall Wallinder, I.; Hedberg, Y. Comparison of the influence of citric acid and acetic acid as simulant for acidic food on the release of ally constituents from stainless steel AISI 201. J. Food Eng. 2015, 145, 51–63. [Google Scholar] [CrossRef]
- Ndung’u, K.; Hibdon, S.; Flegal, A.R. Determination of lead in vinegar by ICP-MS and GFAAS: Evaluation of different sample preparation procedures. Talanta 2004, 64, 258–263. [Google Scholar] [CrossRef]
- Da Silva, J.C.J.; Cadore, S.; Nobrega, J.A.; Baccan, N. Dilute-and-shoot procedure for the determination of mineral constituents in vinegar samples by axially viewed inductively coupled plasma optical emission spectrometry (ICP OES). Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2007, 24, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Nie, X.; Xie, H.L.; Ferro, M.D. Rapid multi-element analysis of Chinese vinegar by sector field inductively coupled plasma mass spectrometry. Eur. Food Res. Technol. 2013, 237, 795–800. [Google Scholar] [CrossRef]
- Buck, Κ.Ν.; Ross, J.R.M.; Flegal, A.R.; Bruland, K.W. A review of total dissolved copper and its chemical speciation in San Francisco Bay, California. Environ. Res. 2007, 105, 5–19. [Google Scholar] [CrossRef]
- Sander, S.S.; Buck, K.N.; Wells, M. The effect of natural organic ligands on trace metal speciation in San Francisco Bay: Implications for water quality criteria. Mar. Chem. 2015, 173, 269–281. [Google Scholar] [CrossRef]
- Chen, W.B.; Smith, D.S.; Guéguen, C. Influence of water chemistry and dissolved organic matter (DOM) molecular size on copper and mercury binding determined by multiresponse fluorescence quenching. Chemosphere 2013, 92, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Karavoltsos, S.; Plavšić, M.; Kalogeropoulos, N.; Kogiannou, D.D.A.; Strrmečki, S.; Sakellari, A.; Dassenakis, M.; Scoullos, M. Copper complexing properties and physicochemical characterization of the organic matter in Greek herbal infusions. Food Chem. 2014, 160, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Grigoriou, C.; Karavoltsos, S.; Kaliora, A.; Sakellari, A.; Plavšić, M.; Dassenakis, M.; Kalogeropoulos, N. Electrochemical, photometric, and chromatographic methods for the evaluation of organic matter and bioactive compounds in coffee brews. Eur. Food Res. Technol. 2018, 244, 1953–1961. [Google Scholar] [CrossRef]
- Sakellari, A.; Karavoltsos, S.; Plavšić, M.; Bempi, E.; Papantonopoulou, G.; Dassenakis, M.; Kalogeropoulos, N. Copper complexing properties, trace metal content and organic matterphysico-chemical characterization of Greek beers. Microchem. J. 2017, 135, 66–73. [Google Scholar] [CrossRef]
- Kostova, I.; Balkansky, S. Metal complexes of biologically active ligands as potential antioxidants. Curr. Med. Chem. 2013, 20, 4508–4539. [Google Scholar] [CrossRef]
- Sinanoglou, V.; Zoumpoulakis, P.; Fotakis, C.; Kalogeropoulos, N.; Sakellari, A.; Karavoltsos, S.; Strati, I. On the characterization and correlation of compositional, antioxidant and colour profile of common and balsamic vinegars. Antioxidants 2018, 7, 139. [Google Scholar] [CrossRef]
- Alonso, A.; Guillén, D.; Barroso, C.; Puertas, B.; García, A. Determination of antioxidant activity of wine byproducts and its correlation with polyphenolic content. J. Agric. Food Chem. 2002, 50, 5832–5836. [Google Scholar] [CrossRef]
- Markoski, M.M.; Garavaglia, J.; Oliveira, A.; Olivaes, J.; Marcadenti, A. Molecular properties of red wine compounds and cardiometabolic benefits. Nutr. Metab. Insights. 2016, 9, 51–57. [Google Scholar] [CrossRef]
- Haslam, E. Plant polyphenols (syn. vegetable tannins) and chemical defense – A reappraisal. J.Chem.Ecol. 1988, 14, 1789–1805. [Google Scholar] [CrossRef]
- Kennedy, J.A.; Powell, K.J. Polyphenol interactions with aluminum (III) and iron (III): Their possible involvement in the podzolization process. Aust. J. Chem. 1985, 38, 879–888. [Google Scholar] [CrossRef]
- McDonald, M.; Mila, I.; Scalbert, A. Precipitation of metal ions by plant polyphenols: Optimal conditions and origin of precipitation. J. Agric. Food Chem. 1996, 44, 599–606. [Google Scholar] [CrossRef]
- WHO. Safety Evaluation of Certain Contaminants in Food Arsenic; WHO Food Additives Series: 63; JECFA Monographs 8; WHO: Geneva, Switzerland, 2011; pp. 153–316. [Google Scholar]
- WHO. Evaluation of Certain Food Additives and Contaminants. WHO Technical Report Series 983. In Proceedings of the 77th Report of the Joint FAO/WHO Expert Committee on Food Additives, Rome, Italy, 4–13 June 2013. [Google Scholar]
- Gupta, S.K.; Vollmer, M.K.; Krebs, R. The importance of mobile, mobilisable and pseudo total heavy metal fractions in soil for three-level risk assessment and risk management. Sci. Total Environ. 1996, 178, 11–20. [Google Scholar] [CrossRef]
- Romic, M.; Romic, D. Heavy metals distribution in agricultural topsoils in urban area. Environ. Geol. 2003, 43, 795–805. [Google Scholar] [CrossRef]
- Rudnick, R.L.; Gao, S. Composition of the Continental Crust. Treatise on Geochem. 2003, 3, 1–64. [Google Scholar]
- Walter, K.H. Manganese Fertilizers. In Manganese in Soils and Plants. Developments in Plant and Soil Sciences; Graham, R.D., Hannam, R.J., Uren, N.C., Eds.; Springer: Dordrecht, The Netherlands, 1988; Volume 33, pp. 225–241. [Google Scholar]
- Azzi, V.; El Samrani, A.; Lartiges, B.; Kobeissi, A.; Kanso, A.; Kazpard, V. Trace metals in phosphate fertilizers used in eastern Mediterranean countries. CLEAN - Soil, Air, Water 2017, 45, 1500988. [Google Scholar] [CrossRef]
- Provenzano, M.R.; El Bilali, H.; Simeone, V.; Baser, N.; Mondelli, D.; Cesari, G. Copper contents in grapes and wines from a Mediterranean organic vineyard. Food Chem. 2010, 122, 1338–1343. [Google Scholar] [CrossRef]
- Senesi, N.; Polemio, M.; Lorusso, L. Evaluation of barium, rubidium and strontium contents in commercial fertilizers. Fertil. Res. 1983, 4, 135–144. [Google Scholar] [CrossRef]
- Scoullos, M.; Vonkeman, G.; Thornton, I.; Makuch, Z. Mercury-Cadmium-Lead. Handbook for sustainable Heavy Metals Policy and Regulation, 1st ed.; Springer-Science+Business Media: Dordrecht, The Netherlands, 2001; p. 525. ISBN 978-94-010-3896-6. [Google Scholar]
- WHO. Evaluation of Certain Food Additives and Contaminants. WHO Technical Report Series 683. In Proceedings of the 26th report of the Joint FAO/WHO Expert Committee on Food Additives, Geneva, Switzerland, 19–26 October 1982. [Google Scholar]
- Lauchli, A.; Epstein, E. Transport of potassium and rubidium in plant roots. Plant Physiol. 1970, 45, 639–641. [Google Scholar] [CrossRef]
- Kot, F.S. On the rubidium and lithium content and availability in the sub-arid south-eastern Mediterranean: Potential health implications. Environ. Geochem. Health 2018, 40, 1841–1851. [Google Scholar] [CrossRef]
- Campbell, L.; Fisk, A.; Wang, X.; Kock, G.; Muir, D.C.G. Evidence for biomagnification of rubidium freshwater and marine food webs. Can. J. Fish. Aquat. Sci. 2005, 62, 1161–1167. [Google Scholar] [CrossRef]
- Guerrero, M.I.; Herce-Pagliai, C.; Cameán, A.M.; Troncoso, A.M.; Gustavo González, A. Multivariate characterization of wine vinegars from the south of Spain according to their metallic content. Talanta 1997, 45, 379–386. [Google Scholar] [CrossRef]
- Navarro-Alarcon, M.; Velasco, C.; Jordal, A.; Terrés, C.; Olalla, M.; Lopez, H.; Lopez, M.C. Copper, zinc, calcium and magnesium content of alcoholic beverages and by-products from Spain: Nutritional supply. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2007, 24, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Troncoso González, A.M.; Guzmán Chozas, M. Metallic contaminants in Andalusian vinegars. Nahrung 1988, 32, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Junior, M.M.S.; Silva, L.O.B.; Leão, D.J.; Ferreira, S.L.C. Analytical strategies for determination of cadmium in Brazilian vinegar samples using ET AAS. Food Chem. 2014, 160, 209–213. [Google Scholar] [CrossRef]
- Acosta, A.; Diaz, C.; Hardisson, A.; González, D. Levels of Cd, Pb, and Ni in different types of vinegars. Bull. Environ. Contam. Tox. 1993, 51, 852–856. [Google Scholar] [CrossRef]
- Acosta-Artiles, A.; Diaz Romero, C.; Hardisson de la Torre, A. Concentration levels of metals in different types of vinegars. Nahrung 1993, 37, 72–73. [Google Scholar] [CrossRef]
- Akpinar-Bayizit, A.; Turan, M.A.; Yilmaz-Ersan, L.; Taban, N. Inductively coupled plasma optical-emission spectroscopy determination of major and minor elements in vinegar. Not. Bot. Hort. Agrobot. Clu. J. 2010, 38, 64–68. [Google Scholar]
- Ozbek, N.; Koca, M.; Akman, S. A practical method for the determination of Al, B, Co, Cr, Cu, Fe, Mg, Mn, Pb, and Zn in different types of vinegars by microwave induced plasma optical emission spectrometry. Food Anal. Methods 2016, 9, 2246–2250. [Google Scholar] [CrossRef]
- Siavash Saei-Dehkordi, S.; Fallah, A.A.; Ghafari, E. Determination of lead, cadmium, copper, and zinc content in commercial Iranian vinegars using stripping chronopotentiometry. Food Anal. Method. 2012, 5, 767–773. [Google Scholar] [CrossRef]
- Ružić, I. Theoretical aspects of the direct titration of natural waters and its information yield for trace metal speciation. Anal. Chim. Acta 1982, 140, 99–113. [Google Scholar] [CrossRef]
- Van den Berg, C.M.G. Determination of copper complexation with natural organic ligands in seawater by equilibration with MnO2: II. Experimental procedures and application to surface seawater. Mar Chem 1982, 11, 323–342. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. Definition and Procedure for the Determination of the Method Detection Limit, Rev. 2,40 CFR, 136, App. B. 2016; pp. 1–6. Available online: https://www.epa.gov/sites/production/files/2016-12/documents/mdl-procedure_rev2_12-13-2016.pdf (accessed on 18 January 2020).
Sample Availability: Samples of the compounds are not available from the authors. |
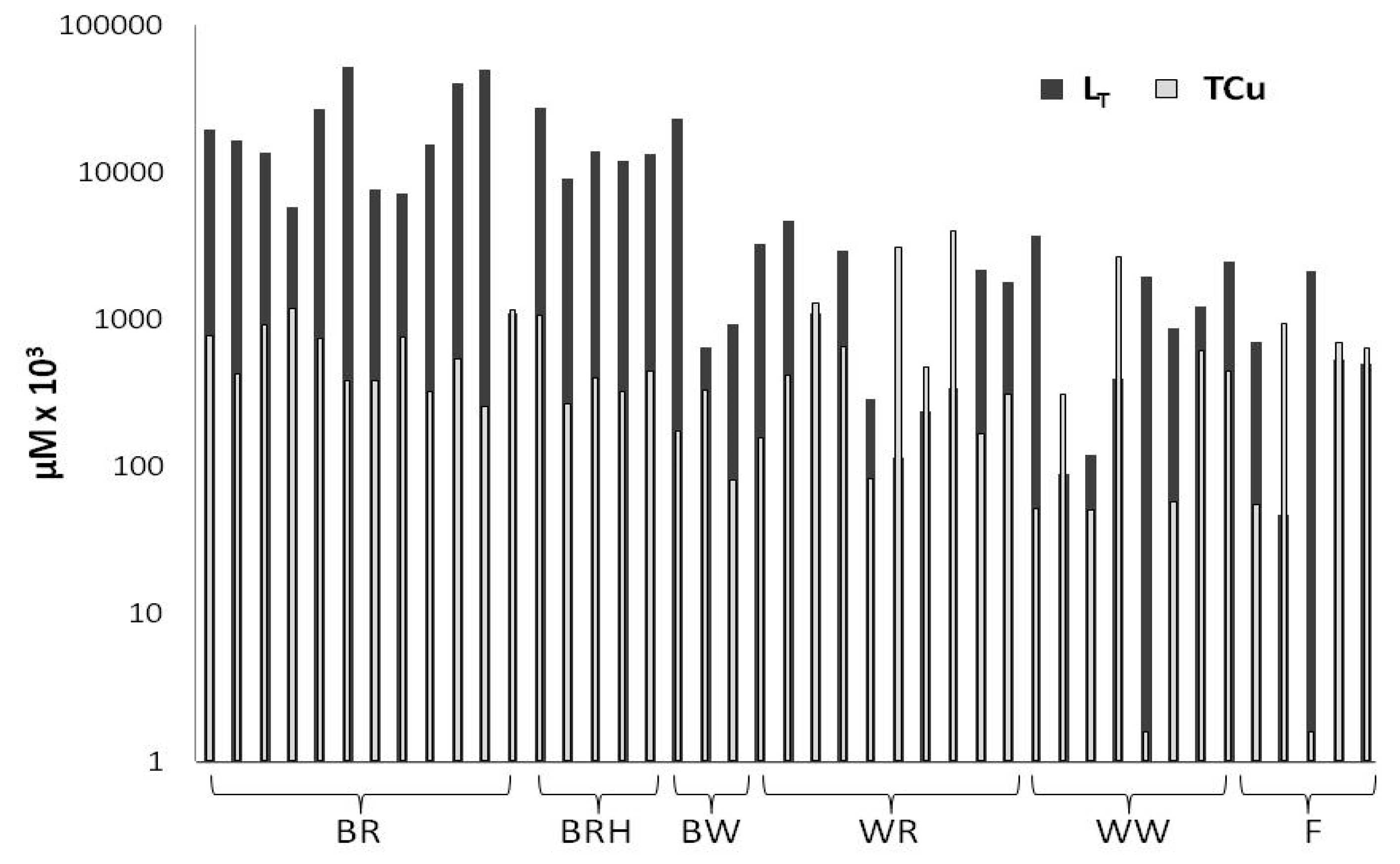
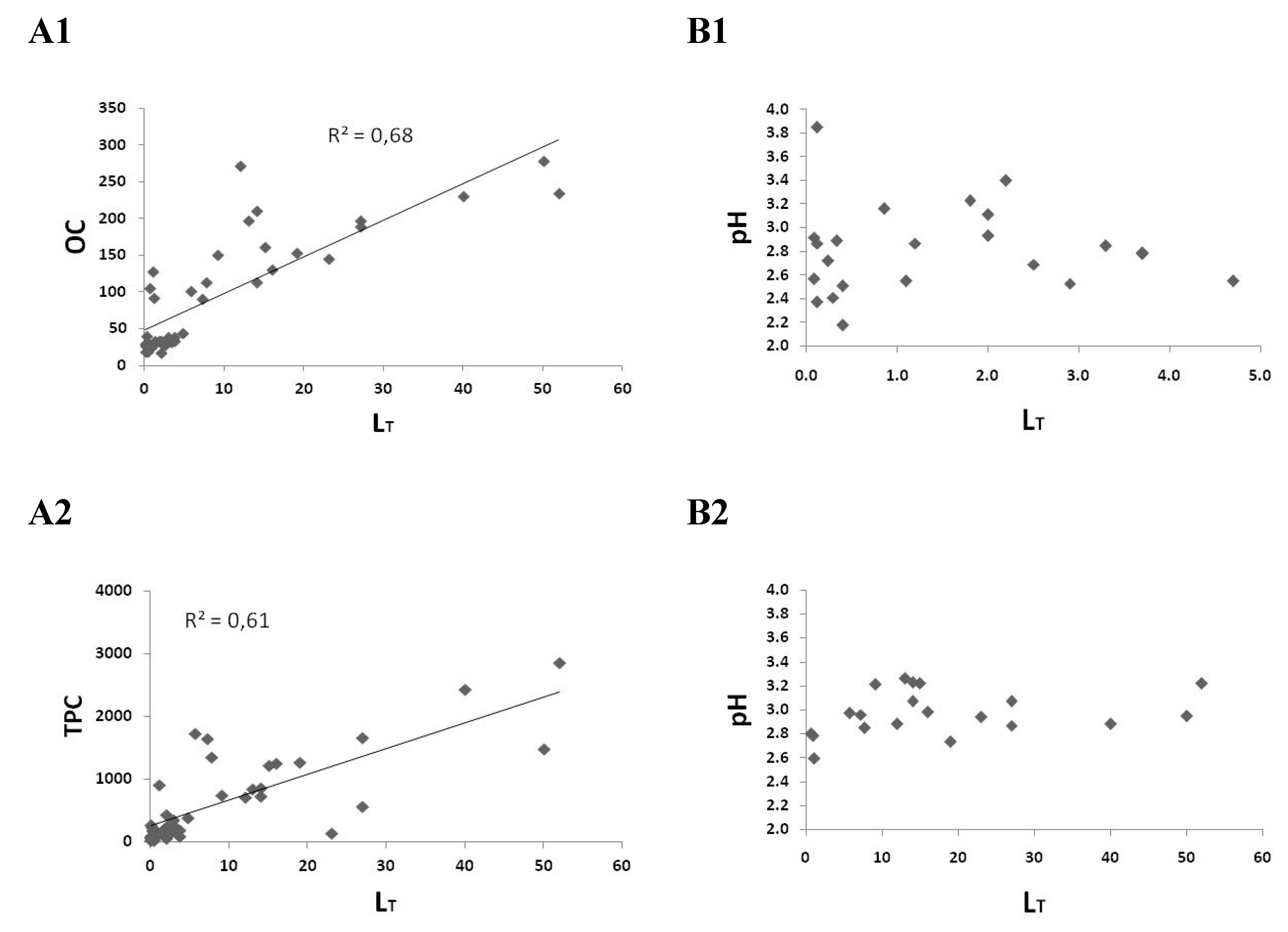
| Sample No | Vinegars | pH | LT (μM) | logKapp | Cu (μM) | OC (mg L−1) | LT/OC (nmol Cu mg−1 C) |
|---|---|---|---|---|---|---|---|
| BR (Red Grape Balsamic Vinegars) | |||||||
| BR1 | Balsamic red | 2.7 | 19 | 8.6 | 0.77 | 154 | 126 |
| BR2 | Balsamic red | 3.0 | 16 | 7.2 | 0.43 | 130 | 126 |
| BR3 | Balsamic red | 3.1 | 14 | 7.4 | 0.92 | 114 | 118 |
| BR4 | Balsamic red | 3.0 | 5.7 | 7.6 | 1.2 | 102 | 56 |
| BR5 | Balsamic red | 3.1 | 27 | 6.9 | 0.74 | 197 | 135 |
| BR6 | Balsamic red | 3.2 | 52 | 6.6 | 0.38 | 235 | 222 |
| BR7 | Balsamic red | 2.8 | 7.7 | 7.8 | 0.38 | 113 | 68 |
| BR8 | Balsamic red | 3.0 | 7.2 | 7.9 | 0.75 | 90 | 80 |
| BR9 | Balsamic red | 3.2 | 15 | 7.1 | 0.32 | 162 | 96 |
| BR10 | Balsamic red | 2.9 | 40 | 6.4 | 0.54 | 230 | 174 |
| BR11 | Balsamic red bio | 3.0 | 50 | 6.8 | 0.26 | 278 | 178 |
| BR12 | Balsamic red bio | 2.6 | 1.1 | 7.8 | 1.2 | 92 | 12 |
| mean ± SD | 3.0 ± 0.2 | 21 ± 17 | 7.3 ± 0.6 | 0.66 ± 0.33 | 158 ± 63 | 116 ± 58 | |
| median | 3.0 | 16 | 7.3 | 0.64 | 142 | 122 | |
| BRH (Red Grape Balsamic Vinegars with Honey) | |||||||
| BRH1 | Balsamic red honey | 2.9 | 27 | 7.8 | 1.1 | 189 | 144 |
| BRH2 | Balsamic red honey | 3.2 | 9.1 | 7.8 | 0.27 | 151 | 60 |
| BRH3 | Balsamic red honey | 3.2 | 14 | 7.2 | 0.40 | 211 | 66 |
| BRH4 | Balsamic red honey | 2.9 | 12 | 7.2 | 0.32 | 273 | 44 |
| BRH5 | Balsamic red honey | 3.3 | 13 | 6.7 | 0.44 | 197 | 68 |
| mean ± SD | 3.1 ± 0.2 | 15 ± 7 | 7.4 ± 0.6 | 0.51 ± 0.34 | 204 ± 44 | 76 ± 39 | |
| median | 3.2 | 13 | 7.2 | 0.40 | 197 | 66 | |
| BW (White Grape Balsamic Vinegars) | |||||||
| BW1 | Balsamic white | 2.9 | 23 | 7.0 | 0.17 | 145 | 161 |
| BW2 | Balsamic white | 2.8 | 0.64 | 8.1 | 0.33 | 106 | 6.1 |
| BW3 | Balsamic white | 2.8 | 0.94 | 8.0 | 0.08 | 128 | 7.3 |
| mean ± SD | 2.8 ± 0.1 | 8.2 ± 12.8 | 7.7 ± 0.6 | 0.19 ± 0.13 | 126 ± 20 | 58 ± 89 | |
| median | 2.8 | 0.94 | 8.0 | 0.17 | 128 | 7.3 | |
| WR (Wine Red Vinegars) | |||||||
| WR1 | Wine red | 2.8 | 3.3 | 7.8 | 0.16 | 32 | 102 |
| WR2 | Wine red | 2.6 | 4.7 | 7.2 | 0.42 | 44 | 106 |
| WR3 | Wine red | 2.6 | 1.1 | 7.3 | 1.3 | 30 | 36 |
| WR4 | Wine red | 2.5 | 2.9 | 6.4 | 0.65 | 40 | 74 |
| WR5 | Wine red | 2.4 | 0.29 | 7.3 | 0.08 | 28 | 10 |
| WR6 | Wine red | 2.9 | 0.12 | 7.1 | 3.1 | 27 | 4.2 |
| WR7 | Wine red | 2.7 | 0.24 | 7.5 | 0.47 | 41 | 5.8 |
| WR8 | Wine red | 2.9 | 0.34 | 8.4 | 4.0 | 31 | 11 |
| WR9 | Wine red | 3.4 | 2.2 | 7.6 | 0.16 | 30 | 73 |
| WR10 | Wine red | 3.2 | 1.8 | 8.0 | 0.31 | 34 | 53 |
| mean ± SD | 2.8 ± 0.3 | 1.7 ± 1.6 | 7.5 ± 0.6 | 1.1 ± 1.4 | 33.7 ± 5.8 | 48 ± 40 | |
| medium | 2.8 | 1.4 | 7.4 | 0.44 | 31.6 | 45 | |
| WW (Wine White Vinegars) | |||||||
| WW1 | Wine white | 2.8 | 3.7 | 7.7 | 0.05 | 39 | 95 |
| WW2 | Wine white | 2.6 | 0.09 | 7.2 | 0.31 | 30 | 3.0 |
| WW3 | Wine white | 2.4 | 0.12 | 7.1 | 0.05 | 28 | 4.3 |
| WW4 | Wine white | 2.5 | 0.40 | 7.2 | 2.7 | 28 | 14 |
| WW5 | Wine white | 3.1 | 2.0 | 6.7 | <0.01 | 17 | 114 |
| WW6 | Wine white | 3.2 | 0.86 | 8.5 | 0.06 | 26 | 33 |
| WW7 | Wine white with rosemary and thyme | 2.9 | 1.2 | 7.1 | 0.61 | 34 | 36 |
| WW8 | Champagne wine | 2.7 | 2.5 | 7.8 | 0.44 | 28 | 88 |
| mean ± SD | 2.8 ± 0.3 | 1.4 ± 1.3 | 7.4 ± 0.6 | 0.53 ± 0.90 | 28.8 ± 6.3 | 48 ± 44 | |
| median | 2.7 | 1.0 | 7.2 | 0.18 | 28.1 | 34 | |
| F (Fruit Vinegars) | |||||||
| F1 | Apple | 2.8 | 3.7 | 7.9 | 0.06 | 34 | 21 |
| F2 | Apple | 2.9 | 0.09 | 7.2 | 0.94 | 29 | 1.6 |
| F3 | Apple | 3.8 | 0.12 | 7.0 | <0.01 | 19 | 113 |
| F4 | Pomegranate | 2.2 | 0.40 | 7.4 | 0.70 | 19 | 28 |
| F5 | Sea Buckthorn | 2.9 | 2.0 | 7.1 | 0.63 | 33 | 15 |
| mean ± SD | 2.9 ± 0.6 | 0.78 ± 0.78 | 7.3 ± 0.4 | 0.47 ± 0.41 | 26.7 ± 7.4 | 36 ± 44 | |
| median | 2.9 | 0.54 | 7.2 | 0.63 | 29.1 | 21 | |
| Al (mg L−1) | As (μg L−1) | Ba (μg L−1) | Cd (μg L−1) | Co (μg L−1) | Cr (μg L−1) | Cu (μg L−1) | Fe (mg L−1) | Mn (mg L−1) | Ni (μg L−1) | Pb (μg L−1) | Sr (mg L−1) | V (μg L−1) | Zn (mg L−1) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Wine vinegars | ||||||||||||||
| Spain [3] | 0.85-7.11 | <20 | 40–164 | <1 | 3–77 | 17–2090 | 17–8580 | 1.18–19.0 | 0.049–1.66 | 2–903 | 35–2200 | 0.196–1.83 | 11–147 | 0.040–6.12 |
| Spain [49] | n.d.–23 | 20–6170 | 1.24–75.8 | 0.15–5.44 | 0.06–8.56 | |||||||||
| Spain [50] | 320 ± 130 | 0.72 ± 0.11 | ||||||||||||
| Spain [51] | 136 | 1350 ± 40 | 8.44 ± 0.07 | 550 ± 50 | 7.86 ± 0.51 | |||||||||
| Brazil [20] | 0.12–2.98 | 20–400 | 10–50 | 0.04–10.0 | <0.01–2.0 | |||||||||
| Brazil [52] | 0.02–0.89 | |||||||||||||
| Canary islands [53,54] | 12–245 | 1440 ± 380 | 2.08 ± 1.09 | 48–205 | 360–1280 | 1.63 ± 0.28 | ||||||||
| Turkey [55] | 130 ± 19 | 4.25 ± 1.95 | 0.78 ± 0.43 | 50 ± 27 | 20 ± 24 | 0.10 ± 0.019 | ||||||||
| Turkey [56] | 0.55–3.07 | n.d.–620 | 2950–3950 | 10–130 | 0.38–5.12 | 0.05–2.49 | 10–150 | 0.16–2.07 | ||||||
| Iran [57] | n.d. –59.3 | 12.8–584 | 3.32–167 | 0.03–1.9 | ||||||||||
| California, USA [19] | 36–50 | |||||||||||||
| California, USA [6] | 7.4–590 | |||||||||||||
| Greece * | <0.002–2.3 | 0.66–8.7 | 37–100 | <0.030–1.4 | 0.78–6.0 | 7.5–55 | <0.40–254 | <0.003–6.3 | 0.04–1.3 | <0.40–56 | <0.050–63 | 0.08–1.2 | 0.31–88 | 0.01–1.2 |
| Balsamic vinegars | ||||||||||||||
| Turkey [55] | 20 ± 5 | 420 ± 153 | 6.94 ± 1.50 | 1.31 ± 0.18 | 30 ± 19 | 20 ± 25 | 0.36 ± 0.162 | |||||||
| Turkey [56] | 0.56–2.29 | n.d.260 | 1980–2090 | 10–30 | 2.51–7.01 | 0.02–0.14 | 10 | 0.26–0.79 | ||||||
| California, USA [19] | 15–307 | |||||||||||||
| California, USA [6] | 14–720 | |||||||||||||
| Greece * | 0.56–10 | 0.93–26 | 27–208 | <0.030–2.3 | 1.8–11 | 19–109 | 5.2–75 | 3.1–10 | 0.43–6.3 | 9.6–68 | <0.050–86 | 0.23–0.92 | 1.9–328 | 0.06–6.4 |
| Fruit vinegars | ||||||||||||||
| CanaryIslands [52,53] | 12–37 | 570 ± 140 | 1.14 ± 0.45 | 48–201 | 350–550 | 0.25 ± 0.04 | ||||||||
| Turkey [55] | 10 ± 4 | 30 ± 32 | 1.31 ± 0.58 | 0.18 ± 0.13 | 10 ± 13 | 10 ± 8 | ||||||||
| Turkey [56] | 0.32–5.19 | 1970–2330 | 10–50 | 0.27–6.84 | 0.03–1.51 | 10–80 | 0.29–2.42 | |||||||
| Iran [57] | n.d. –78 | 32–1129 | 4.4–253 | 0.05–3.7 | ||||||||||
| California, USA [6] | 3.7–7.4 | |||||||||||||
| Greece * | <0.002–1.4 | <0.050–5.4 | 31–108 | <0.030–1.8 | 0.08–5.5 | 0.88–26 | <0.40–60 | <0.003–5.1 | <0.01–0.83 | 1.3–70 | <0.050–33 | 0.09–0.61 | <0.050–6.2 | <0.01–0.88 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karavoltsos, S.; Sakellari, A.; Sinanoglou, V.J.; Zoumpoulakis, P.; Plavšić, M.; Dassenakis, M.; Kalogeropoulos, N. Copper Complexing Capacity and Trace Metal Content in Common and Balsamic Vinegars: Impact of Organic Matter. Molecules 2020, 25, 861. https://doi.org/10.3390/molecules25040861
Karavoltsos S, Sakellari A, Sinanoglou VJ, Zoumpoulakis P, Plavšić M, Dassenakis M, Kalogeropoulos N. Copper Complexing Capacity and Trace Metal Content in Common and Balsamic Vinegars: Impact of Organic Matter. Molecules. 2020; 25(4):861. https://doi.org/10.3390/molecules25040861
Chicago/Turabian StyleKaravoltsos, Sotirios, Aikaterini Sakellari, Vassilia J. Sinanoglou, Panagiotis Zoumpoulakis, Marta Plavšić, Manos Dassenakis, and Nick Kalogeropoulos. 2020. "Copper Complexing Capacity and Trace Metal Content in Common and Balsamic Vinegars: Impact of Organic Matter" Molecules 25, no. 4: 861. https://doi.org/10.3390/molecules25040861
APA StyleKaravoltsos, S., Sakellari, A., Sinanoglou, V. J., Zoumpoulakis, P., Plavšić, M., Dassenakis, M., & Kalogeropoulos, N. (2020). Copper Complexing Capacity and Trace Metal Content in Common and Balsamic Vinegars: Impact of Organic Matter. Molecules, 25(4), 861. https://doi.org/10.3390/molecules25040861








