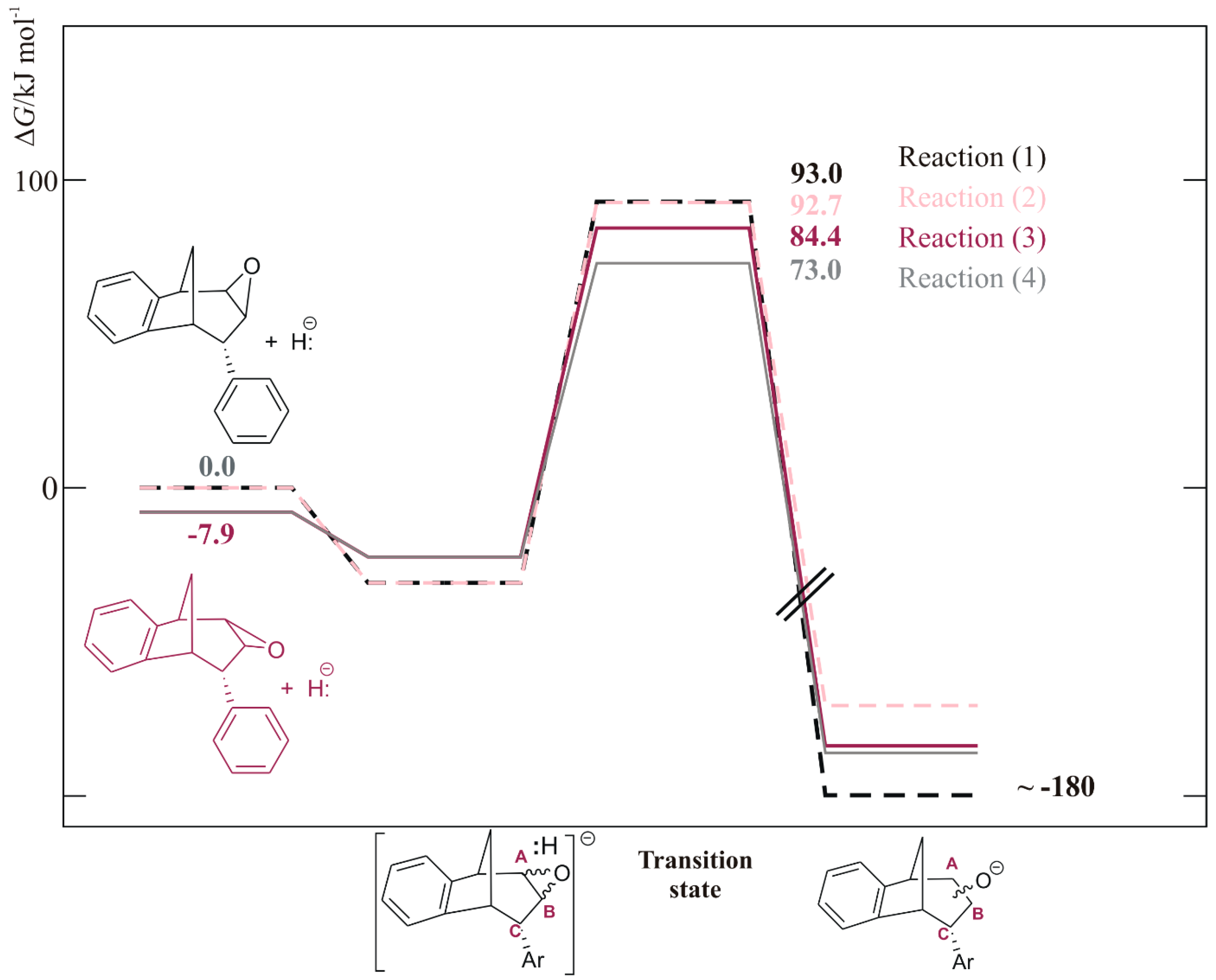
3. Materials and Methods
3.1. General
Petroleum ether (PE)(VWR Prolabo Chemicals, Fontenay-sous-Bois, France), bp 40–60 °C, was used. Solvents were purified by distillation. Column chromatography was carried out on columns with silica gel (Fluka 0.063–0.2 nm and Fluka 60 Å, technical grade, Merck, New York, NY, USA). Thin layer chromatography (TLC) was carried out using plates coated with silica gel (0.2 mm, 0.5 mm, 1.0 mm, Kiselgel 60 F
254, Merck, New York, NY, USA). Organic layers were routinely dried with anhydrous MgSO
4 and evaporated using a rotary evaporator (Heidolph, Schwabach, Germany).
1H and
13C NMR spectra were recorded on a spectrometer (Bruker AV-600 Spectrometer, Billerica, MA, USA) at 600 MHz. All NMR spectra were measured in CDCl
3 using tetramethylsilane (Merck, New York, NY, USA) as reference. The following abbreviations were used: Sh—shoulder (in UV spectra), s—singlet, d—doublet, t—triplet, q—quartet, dd—doublet of doublets, m—multiplet, br—broad, PE—petroleum ether, E—diethyl ether. UV spectra were measured on a UV/VIS spectrophotometer (Varian Cary 50 UV/VIS Spectrophotometer, Palo Alto, CA, USA). Mass spectra were obtained on a UPLC-MS system (Acquity UPLC coupled with SQD mass spectrometer, Milford, MA, USA). Melting points were obtained using a microscope equipped apparatus (Original Kofler Mikroheitztisch apparatus Reichert, Vienna, Austria) and have not been corrected. HRMS analyses were carried out on a mass spectrometer (MALDI TOF/TOF analyzer (Merck, New York, NY, USA), equipped with Nd:YAG laser (Merck, New York, NY, USA) operating at 355 nm with a firing rate of 200 Hz in the positive (H
+) or negative (-H) ion reflector mode. Compounds
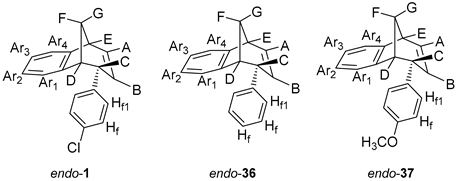
endo-
1,
endo-
36,
endo-
37 and
49 were prepared in our laboratory by photochemical strategy and published before [
31,
41,
43,
44].
3.2. Photochemistry
Photochemical Synthesis of endo-1, endo-36 and endo-37
The solutions of a mixture of isomers of corresponding Wittig reaction products were purged with argon for 20 min and irradiated at 350 nm in petroleum ether (5 × 10−3 M) in a Rayonet reactor in a Pyrex tube for 4 h. The solvent was removed in vacuum and the oily residue chromatographed on silica gel column using petroleum ether as eluent.
After column chromatography, 77% of endo-1, 90% of endo-36 and 70% of endo-37 was isolated. High-molecular-weight products remained on the column.
11-(4-Chlorophenyl)tricyclo[6.3.1.0²,7]dodeca-2,4,6,9-tetraene (
endo-
1) [
39]: colorless oil;
Rf 0.66 (petroleum ether/dichloromethane = 9:1); UV (96% EtOH)
λmax (log
ε) 276 (3.19), 268 (3.22), 224 (4.20, sh);
1H NMR (CDCl
3, 600 MHz) δ/ppm 7.14 (d,
J = 8.4 Hz, 2H, H
ar2), 7.12 (d,
J = 7.3 Hz, 1H), 7.03 (td,
J = 7.4; 1.0 Hz, 1H), 6.84 (td,
J = 7.4; 1.0 Hz, 1H), 6.65 (d,
J = 8.3 Hz, 2H, H
ar1), 6.40–6.35 (m, 1H, H
A), 6.23 (d,
J = 7.3 Hz, 1H, H
ar), 5.25 (ddd,
J = 9.6; 4.0; 1.9 Hz, 1H, H
B), 3.96–3.92 (m, 1H), 3.34 (t,
J = 9.4; 4.7 Hz, 1H), 3.29 (dd,
J = 6.1; 4.7 Hz, 1H), 2.54–2.50 (m, 1H, H
F), 2.37 (d,
J = 10.0 Hz, 1H, H
G);
13C NMR (CDCl
3, 150 MHz) δ/ppm 140.6 (s), 137.3 (s), 132.9 (s), 130.9 (s), 134.7 (d), 129.2 (2d), 127.3 (d), 125.7 (d), 125.6 (d), 125.4 (2d), 124.7 (d), 119.7 (d), 48.0 (d), 45.3 (d), 43.7 (t), 39.9 (d); HRMS (
m/z) for C
18H
15Cl: [M + H]
+calcd = 265.0789, [M + H]
+measured = 265.0786.
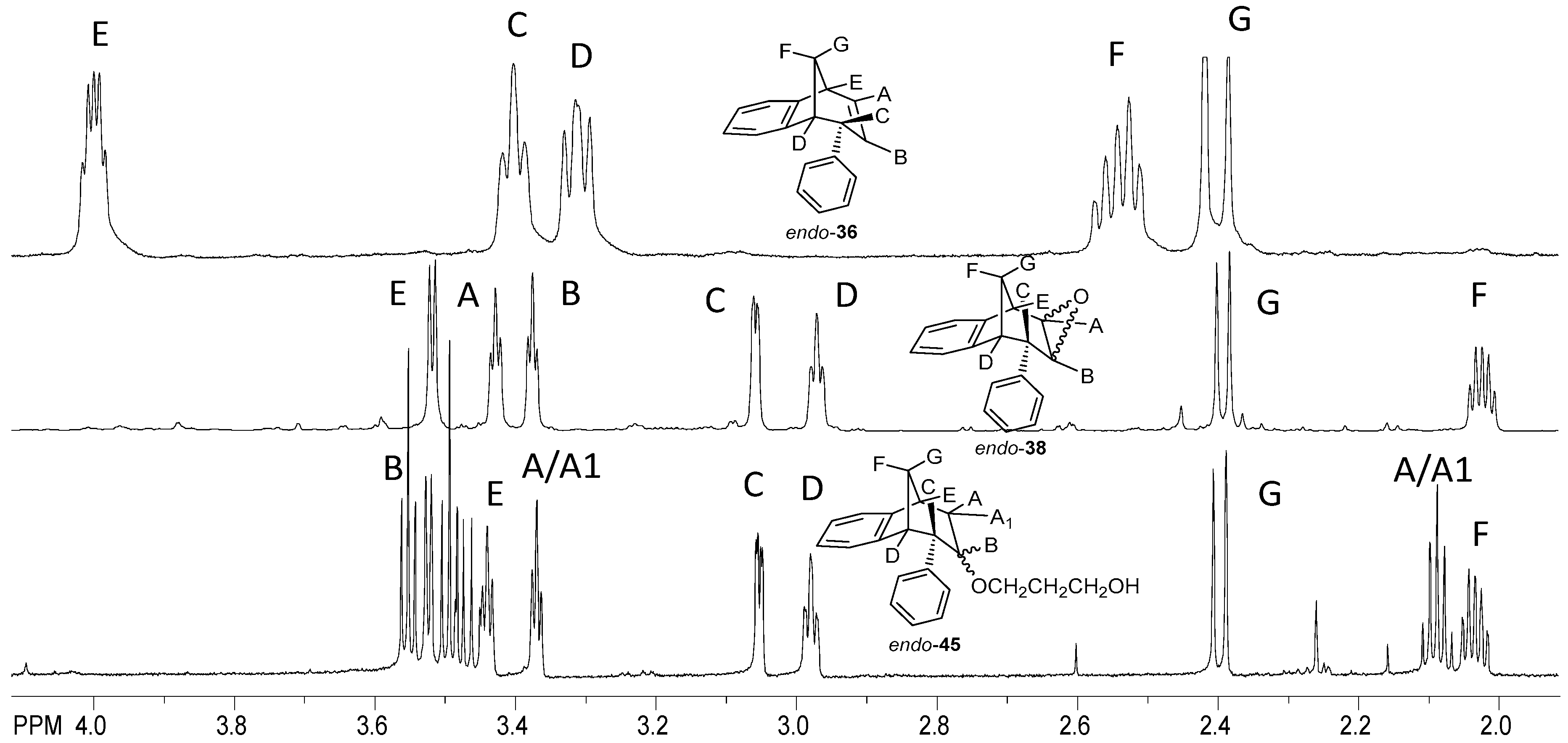
endo-6-Phenyl-6,9-dihydro-5H-5,9-methano-benzocycloheptene (
endo-36) [
38]: colorless crystals; mp 51 °C;
Rf 0.44(petroleum ether); UV (EtOH)
λmax (log
ε) 275 (2.88), 263 (2.90), 203 (4.46);
1H NMR (CDCl
3, 600 MHz)
δ 7.17–7.18 (m, 3H, H
f), 7.12 (d,
J = 7.3 Hz, 1H, H
Ar4), 7.03 (t,
J = 7.4 Hz, 1H, H
Ar3), 6.81 (t,
J = 7.4 Hz, 1H, H
Ar2), 6.73–6.76 (m, 2H, H
f1), 6.37 (ddd,
JAB = 9.5 Hz;
JAE = 6.0 Hz;
JAC = 2.5 Hz, 1H, H
A), 6.18 (d,
J = 7.3 Hz, 1H, H
Ar1), 5.33 (dt,
JAB = 9.5 Hz;
JBC = 2.5 Hz, 1H, H
B), 3.98 (m, 1H, H
C), 3.38 (t,
JCD =
JDF = 4.7 Hz, 1H, H
D), 3.29 (dd,
JAE = 6.0 Hz;
JEF = 4.7 Hz, 1H, H
E), 2.52 (dt,
JFG = 9.9 Hz;
JEF =
JDF = 4.7 Hz, 1H, H
F), 2.38 (d,
JFG = 9.9 Hz, 1H, H
G);
13C NMR (CDCl
3, 150 MHz)
δ 152.27 (s), 142.44 (s), 141.93 (s), 132.71 (d), 128.21 (d), 127.60 (d), 126.34 (d), 126.10 (d), 126.05 (d), 125.97 (d), 124.89 (d), 120.03 (d), 48.59 (d), 46.29 (d), 44.13 (t, C
FG), 40.37 (d); MS
m/z (EI) 232 (M
+, 100%), 117 (25), 115 (10); HRMS (
m/z) for C
18H
16: [M + H]
+calcd = 232.1634, [M + H]
+measured = 232.1655.
11-(4-Methoxyphenyl)tricyclo[6.3.1.0²,7]dodeca-2,4,6,9-tetraene (
endo-
37) [
40]: colorless crystals; mp 45–47 °C;
Rf (petroleum ether/dichloromethane = 8:2) = 0.27;
1H NMR (CDCl
3, 600 MHz) δ/ppm: 7.11 (d, 1H,
J = 7.3 Hz), 7.03 (dt, 1H,
J = 7.3; 1.0 Hz), 6.82 (dt, 1H,
J = 7.3; 1.0 Hz), 6.72 (d, 2H,
J = 8.6 Hz), 6.64 (d, 2H,
J = 8.6 Hz), 6.32-6.36 (m, 1H), 6.25 (d, 1H,
J = 7.3 Hz), 5.28 (dt, 1H,
J = 9.6; 2.6 Hz), 3.91-3.94 (m, 1H), 3.77 (s, 3H), 3.35 (t, 1H,
J = 4.5 Hz), 3.27 (dd, 1H,
J = 6.3; 4.7 Hz), 2.49-2.53 (m, 1H), 2.37 (d, 1H,
J = 9.9 Hz);
13C NMR (CDCl
3, 75 MHz) δ/ppm: 157.6 (s), 152.0 (s), 141.7 (s), 134.2 (s), 134.1 (d), 128.8 (2d), 126.3 (d), 125.8 (d), 125.5 (d), 124.5 (d), 119.6 (d), 112.6 (2d), 52.4 (q), 48.2 (d), 45.0 (d), 43.7 (t), 40.3 (d); MS
m/z (%, fragment): 262 (100, M
+), 154 (75), 115 (50); HRMS (
m/z) for C
19H
18O: [M + H]
+calcd = 262.1352, [M + H]
+measured = 262.1351.
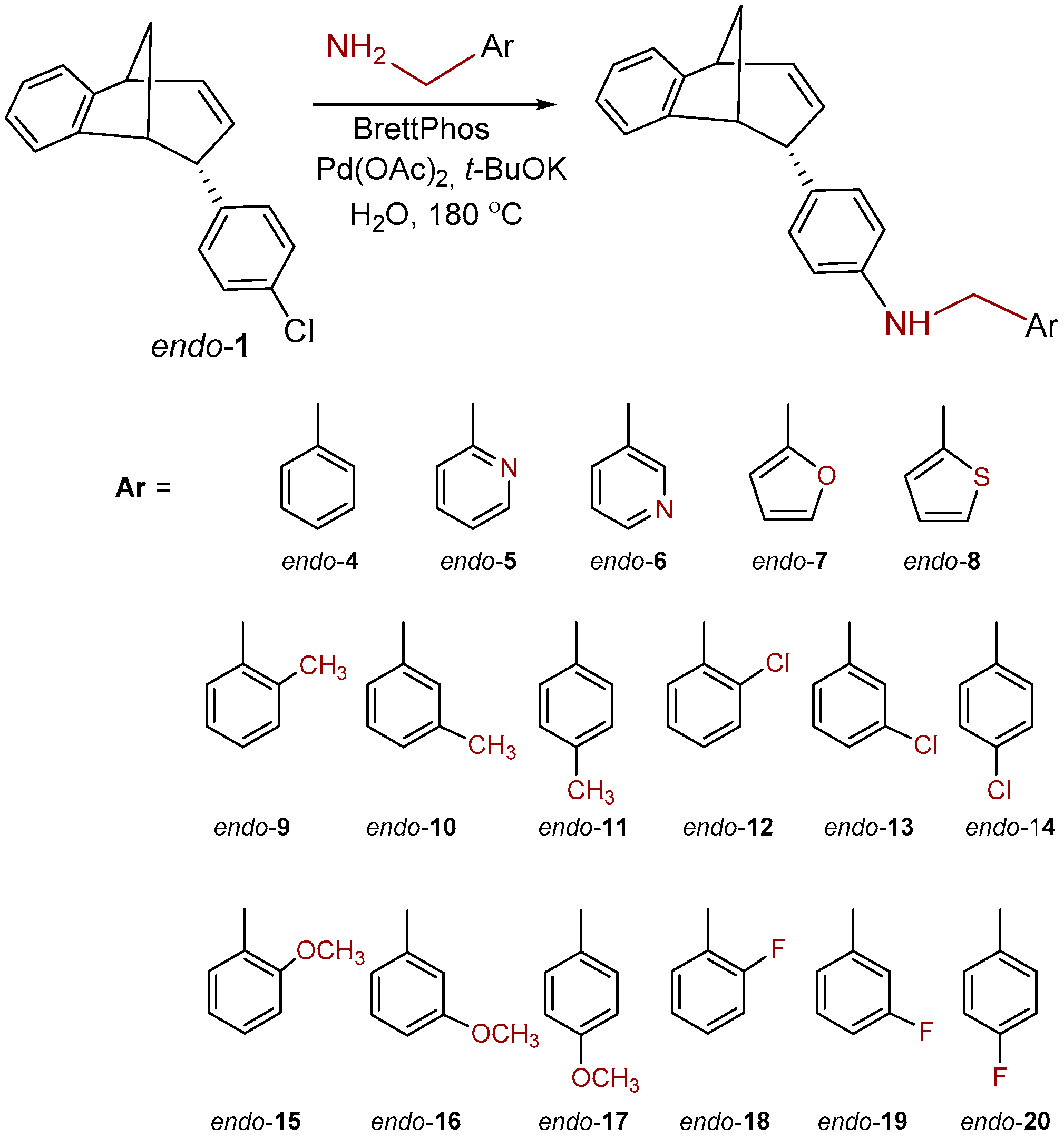
3.3. Synthesis of Benzylamine Derivatives
Amination Reactions of endo-1 with Primary Benzylic Amines
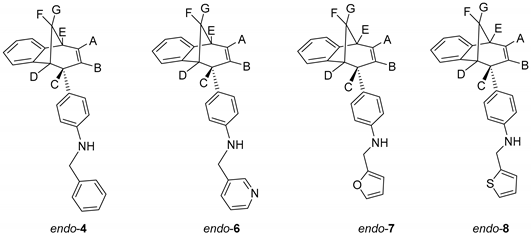
To a solution of BrettPhos (0.1 eq) and Pd(OAc)2 (0.05 eq) in dioxane, 4% of water was added. After the addition of water, the solution was heated to 120 °C for 2 min. The reaction mixture changed color from light yellow, over dark red to dark green. Compound endo-1, different amine (2 eq) and KOtBu (1.4 eq) were added into a sealed tube. The reaction mixture was stirred for 20 h on 180 °C. After the removal of the solvent, the crude product residue was purified by repeated column chromatography using petroleum ether/dichlorometane mixture as an eluent. All of the obtained compounds endo-4–20 were isolated in the first fractions as yellow oils, while BrettPhos remained on the chromatographic column. In all cases, the conversion of the amination reaction was complete. The spectroscopic data for some of the previously synthesized amines (endo-4, endo-6, endo-7 and endo-8) are given below.
![Molecules 25 04872 i002 Molecules 25 04872 i002]()
N-Benzyl-4-((5R,6S,9S)-6,9-dihydro-5H-5,9-methanobenzo[7]annulen-6-yl)aniline (endo-4): Column chromatography on silica gel using petroleum ether/dichloromethane (0%–60%) as eluent afforded 0.080 g (48.3%) of endo-4: Rf (PE / DCM = 5:1) = 0.43; UV (EtOH) λmax/nm (ε/dm3mol−1cm−1): 251 (11,312), 306 (Sh, 2527); IR νmax/cm−1 (diamond ATR): 3408.78, 1613.45, 1517.61, 1468.98; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.38–7.25 (m, 5H), 7.10 (d, 1H, J = 7.4 Hz), 7.02 (dt, 1H, J = 7.4; 1.1 Hz), 6.83 (dt, 1H, J = 7.4; 1.1 Hz), 6.54 (d, 2H, J = 8.6 Hz), 6.47 (d, 2H, J = 8.6 Hz), 6.32 (d, 2H, J = 7.4 Hz, Har, HA), 5.27 (td, 1H, J = 9.5; 1.9 Hz, HB), 4.28 (s, 2H), 3.92–3.85 (m, 2H, HC, NH), 3.35 (t, 1H, J = 4.2 Hz, HD), 3.27 (dd, 1H, J = 6.3; 4.2 Hz, HE), 2.55–2.47 (m, 1H, HF), 2.34 (d, 1H, J = 10.1 Hz, HG); 13C NMR (CDCl3, 150 MHz) δ/ppm: 152.1 (s), 146.1 (s), 141.9 (s), 139.1 (s), 133.8 (d), 130.6 (s), 128.6 (2d), 128.1 (d), 128.1 (d), 127.1 (d), 126.7 (2d), 126.0 (d), 125.9 (d), 125.4 (d), 124.4 (d), 119.5 (d), 111.9 (2d), 48.4 (d), 48.2 (t), 45.2 (d), 43.7 (t), 40.0 (d); MS m/z (%, fragment): 337 (100, M+); HRMS (m/z) for C25H23N: [M + H]+calcd = 338.1822, [M + H]+measured = 338.1902.
4-((5R,6S,9S)-6,9-Dihydro-5H-5,9-methanobenzo[7]annulen-6-yl)-N-(pyridin-3-ylmethyl)-aniline (endo-6): Column chromatography on silica gel using dichloromethane/ethanol (variable ratio) as eluent afforded 0.062 g (21.2%) of endo-6 in the first fractions: Rf (DCM) = 0.1; UV (EtOH) λmax/nm (ε/dm3mol−1cm−1): 254 (15991); IR νmax/cm−1 (diamond ATR): 3370.11, 1609.55, 1531.76, 1444.89; 1H NMR (CDCl3, 600 MHz) δ/ppm: 8.62 (s, 1H), 8.52 (dd, 1H, J = 5.4; 1.3 Hz), 7.67 (d, 1H, J = 7.4 Hz), 7.30–7.23 (m, 3H), 7.13 (d, 1H, J = 7.4 Hz), 7.09 (dt, 1H, J = 7.7; 1.2 Hz), 7.03 (dt, 1H, J = 7.7; 1.2 Hz), 6.85 (d, 1H, J = 8.5 Hz), 6.83 (d, 2H, J = 8.5 Hz), 6.60-6.58 (m, 2H, Har, HA), 5.41 (t, 1H, J = 3.7 Hz, HB), 4.36 (s, 2H), 3.79–3.76 (m, 2H, HC, NH), 3.32 (t, 1H, J = 5.1 Hz, HD), 2.69 (td, 1H, J = 8.3; 1.2 Hz, HE), (signal for HF and HG can’t be assigned because of the presence of the catalyst BrettPhos); MS m/z (%, fragment): 338 (100, M+).
Column chromatography on silica gel using dichloromethane/ethanol (variable ratio) as eluent afforded 0.051 g (43.8%) of endo-7 and 0.064 g (51.5%) of endo-8.
4-((5R,6S,9S)-6,9-Dihydro-5H-5,9-methanobenzo[7]annulen-6-yl)-N-(fur-2-ylmethyl)aniline (endo-7): Rf (PE / DCM = 5:1) = 0.62; UV (EtOH) λmax/nm (ε/dm3mol−1cm−1): 255 (28024), 358 (12474); IR νmax/cm−1 (diamond ATR): 3412.68, 1603.44, 1517.61, 1468.98; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.34 (d, 1H, J = 1.6 Hz, H5f), 7.09 (d, 1H, J = 7.2 Hz), 7.00 (t, 1H, J = 7.4 Hz), 6.80 (t, 1H, J = 7.2 Hz), 6.53 (d, 2H, J = 8.5 Hz), 6.49 (d, 2H, J = 8.5 Hz), 6.33–6.24 (m, 3H, Har, HA), 6.20 (d, 1H, J= 3.2 Hz, H3f), 5.26 (d, 1H, J = 9.4 Hz, HB), 4.26 (s, 2H), 3.88–3.83 (m, 1H, HC), 3.31 (t, 1H, J = 4.8 Hz, HD), 3.24 (t, 1H, J = 4.8 Hz, HE), 2.51–2.43 (m, 1H, HF), 2.33 (d, 1H, J = 9.7 Hz, HG); 13C NMR (CDCl3, 150 MHz) δ/ppm: 152.9 (s), 152.5 (s), 146.0 (s), 142.3 (s), 141.8 (d), 134.3 (d), 132.2 (s), 129.1 (2d), 121.1 (d), 126.4 (d), 125.9 (d), 124.9 (d), 119.9 (d), 112.8 (2d), 110.2 (d), 106.9 (d), 48.8 (d), 45.7 (d), 44.1 (t), 40.5 (d); MS m/z (%, fragment): 327 (100, M+); HRMS (m/z) for C23H21NO: [M + H]+calcd = 328.1609, [M + H]+measured = 328.1689.
4-((5R,6S,9S)-6,9-dihydro-5H-5,9-methanobenzo[7]annulen-6-yl)-N-(thien-2-ylmethyl)aniline (endo-8): Rf (PE / DCM = 5:1) = 0.70; UV (EtOH) λmax/nm (ε/dm3mol−1cm−1): 247 (18373); IR νmax/cm−1 (diamond ATR): 3413.17, 1613.45, 1516.18, 1468.98; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.48 (d, 1H, J = 4.9 Hz) 7.38 (d, 1H, J = 7.2 Hz), 7.30 (t, 1H, J = 7.1 Hz), 7.27 (broad s), 7.25–7.22(m, 2H), 7.11 (t, 1H, J = 7.2 Hz), 6.83 (d, 2H, J = 8.6 Hz), 6.78 (d, 2H, J = 8.6 Hz), 6.62-6.56 (m, 2H, Har, HA), 5.55 (d, 1H, J = 9.5 Hz, HB), 4.75 (s, 2H), 4.20–4.13 (m, 2H, HC, NH), 3.61 (t, 1H, J = 4.7 Hz, HD), 3.53 (t, 1H, J = 4.7 Hz, HE), 2.79–2.74 (m, 1H, HF), 2.62 (d, 1H, J = 9.7 Hz, HG);13C NMR (CDCl3, 150 MHz) δ/ppm: 152.5 (s), 145.9 (s), 143.1 (s), 142.3 (s), 134.4 (d), 132.3 (s), 129.1 (2d), 127.1 (d), 126.7 (d), 126.4 (d), 125.8 (d),125.0 (d), 124.9(d), 124.5 (d), 119.9 (d), 112.7 (2d), 48.8 (d), 45.7 (d), 44.1 (t), 43.7 (t), 40.4 (d); MS m/z (%, fragment): 343 (100, M+); HRMS (m/z) for C23H21NS: [M + H]+calcd = 344.1394, [M + H]+measured = 344.1474.
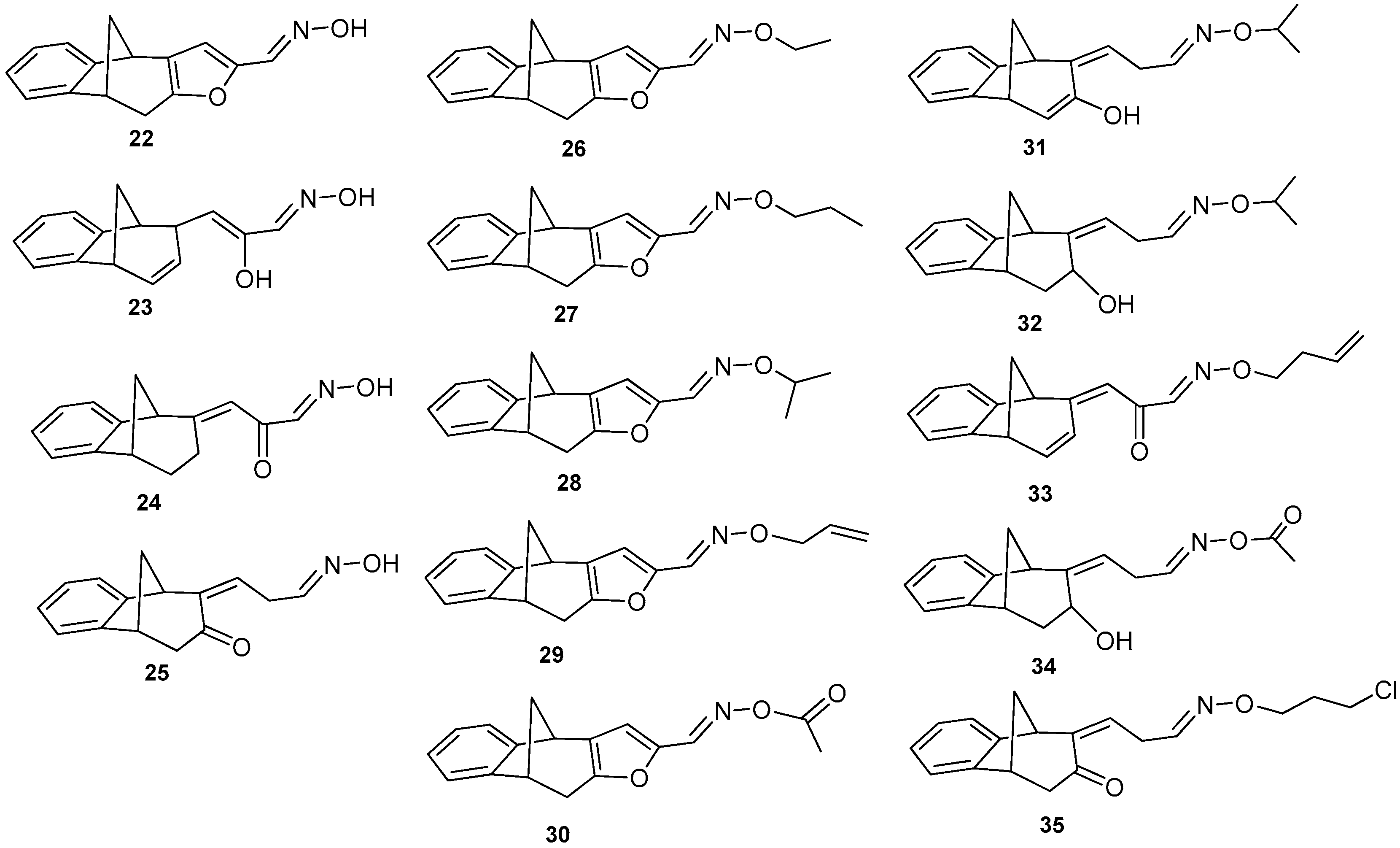
3.4. Synthesis of Oxime Derivatives
3.4.1. Synthesis of Oxime
A mixture of aldehyde 21 (0.223 mmol), hydroxylamine hydrochloride (0.112 mmol, 0.5 eq), and pyridine (0.05 mL) was refluxed in ethanol (1 mL) for 1 h on a water bath. Completion of the reaction was checked by TLC (DCM/PE, 1:3). At the end of the reaction, ethanol was evaporated to dryness. Crude material was purified on column chromatography using different polarity of solvents. The product was additionally purified using preparative TLC (DCM/PE).
3.4.2. Synthesis of Oxime Ethers
A solution of oxime 22 (0.251 mmol) and alkyl bromide (0.276 mmol, 1.1 eq) in dry acetone (2 mL) was refluxed in the presence of dry potassium carbonate (0.376 mmol, 1.5 eq) for 48 h on a water bath. Completion of the reaction was checked by TLC (DCM/PE). At the end of the reaction, acetone was evaporated to dryness. Crude material was purified on column chromatography using different polarity of solvents (DCM/PE) and the product was also additionally purified using preparative TLC.
![Molecules 25 04872 i003 Molecules 25 04872 i003]()
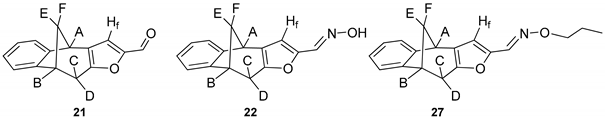
(4R,9R)-9,10-Dihydro-4H-4,9-methanobenzo[4,5]cyclohepta[1,2-b]furan-2-carbaldehyde (
21) [
33]: 180 mg (50.0%);
Rf (petroleum ether/dichloromethane = 20:1) = 0.23; UV (EtOH)
λmax (ε/dm
3 mol
−1 cm
−1): 308 (16794) nm;IR
νmax/cm
−1 (NaCl): 1666, 1504;
1H NMR (CDCl
3, 300 MHz) δ/ppm: 9.41 (s, 1H), 7.32 (dd, 1H,
J = 7.2; 1.4 Hz), 7.16–7.08 (m, 4H, 3H), 3.91 (d, 1H,
J = 4,4 Hz, H
A), 3.66 (dt, 1H,
J = 4.9; 0.5 Hz, H
B), 3.20 (dd, 1H,
J = 17.8; 4.9 Hz, H
C), 2.72 (dd, 1H,
J = 17.8; 0.5 Hz, H
D), 2.48 (ddd, 1H,
J = 10.7; 4.9; 4.4 Hz, H
E), 2.4 (d, 1H,
J = 10.7 Hz, H
F);
13C NMR (CDCl
3, 75 MHz) δ /ppm: 176.6 (d), 156.0 (s), 151.3 (s), 150.6 (s), 144.1 (s), 128.5 (s), 126.9 (d), 126.9 (d), 124.1 (d), 121.0 (2d), 42.5 (t), 39.6 (d), 39.1 (d), 31.4 (t); MS
m/
z (%, fragment): 224 (100, M
+), 167 (75), 115 (50); HRMS (
m/z) for C
19H
18O: [M + H]
+calcd = 224.3355, [M + H]
+measured = 224.3347.
(E)-9,10-Dihydro-4H-4,9-methanobenzo[4,5]cyclohepta[1,2-b]furan-2-carbaldehyde oxime (22): 16.3 mg (40%); Rf (PE / DCM = 1:1) = 0.33; IR νmax/cm−1 (NaCl): 3400, 2943, 1668, 1613, 1572, 1503, 1460; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.36 (d, 1H, J = 6.6 Hz), 7.18-7.05 (m, 4H), 6.06 (s, 1H), 4.62 (d, 1H, J = 2.5 Hz), 3.81 (d, 1H, J = 4.0 Hz), 3.59-3.54 (m, 2H), 2.40-2.37 (m, 2H); 13C NMR (CDCl3, 150 MHz) δ/ppm: 152.1 (s), 146.6 (s), 141.9 (s), 135.9 (s), 131.6 (s), 126.9 (d), 126.4 (d), 125.1 (d), 121.5 (d), 104.6 (2d), 67.4 (t), 48.9 (d), 39.7 (t), 39.6 (d); MS m/z (%, fragment) (EI): 239 (100); HRMS (m/z) for C19H19ON: [M + H]+ calcd = 240.0946; [M + H]+measured = 240.0941.
(Z)-9,10-Dihydro-4H-4,9-methanobenzo[4,5]cyclohepta[1,2-b]furan-2-carbaldehyde O-propyl oxime (27): 13.6 mg (32%); Rf (PE / DCM = 1:5) = 0.55; IR νmax/cm−1 (NaCl): 3434, 2918, 1731, 1638, 1234; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.32 (d, 1H, J = 7.4 Hz), 7.19–7.07 (m, 4H), 6.94 (s, 1H), 3.86 (d, 1H, J = 4.5 Hz), 3.68 (d, 1H, J = 5.0 Hz), 3.48 (q, 2H, J = 7.0 Hz), 3.15 (dd, 1H, J = 17.4; 5.0 Hz), 2.67 (dd, 1H, J = 17.4; 1.3 Hz), 2.51–2.47 (m, 1H), 2.03 (d, 1H, J = 11.2 Hz), 1.34–1.29 (m, 2H), 0.88 (t, 3H, J = 6.9 Hz); 13C NMR (CDCl3, 150 MHz) δ/ppm: 152.0 (s),150.5 (s), 143.8 (s), 135.6 (s), 128.1 (s), 126.9 (d), 126.9 (d), 124.1 (d), 124.1 (d), 120.2 (d), 67.0 (t), 42.5 (t), 39.6 (d), 38.9 (d), 31.2 (t), 29.6 (t), 22.7 (q); MS m/z (%, fragment) (EI): 281 (100); HRMS (m/z): [M + H]+ calcd = 282.1416; [M + H]+measured = 282.1419.
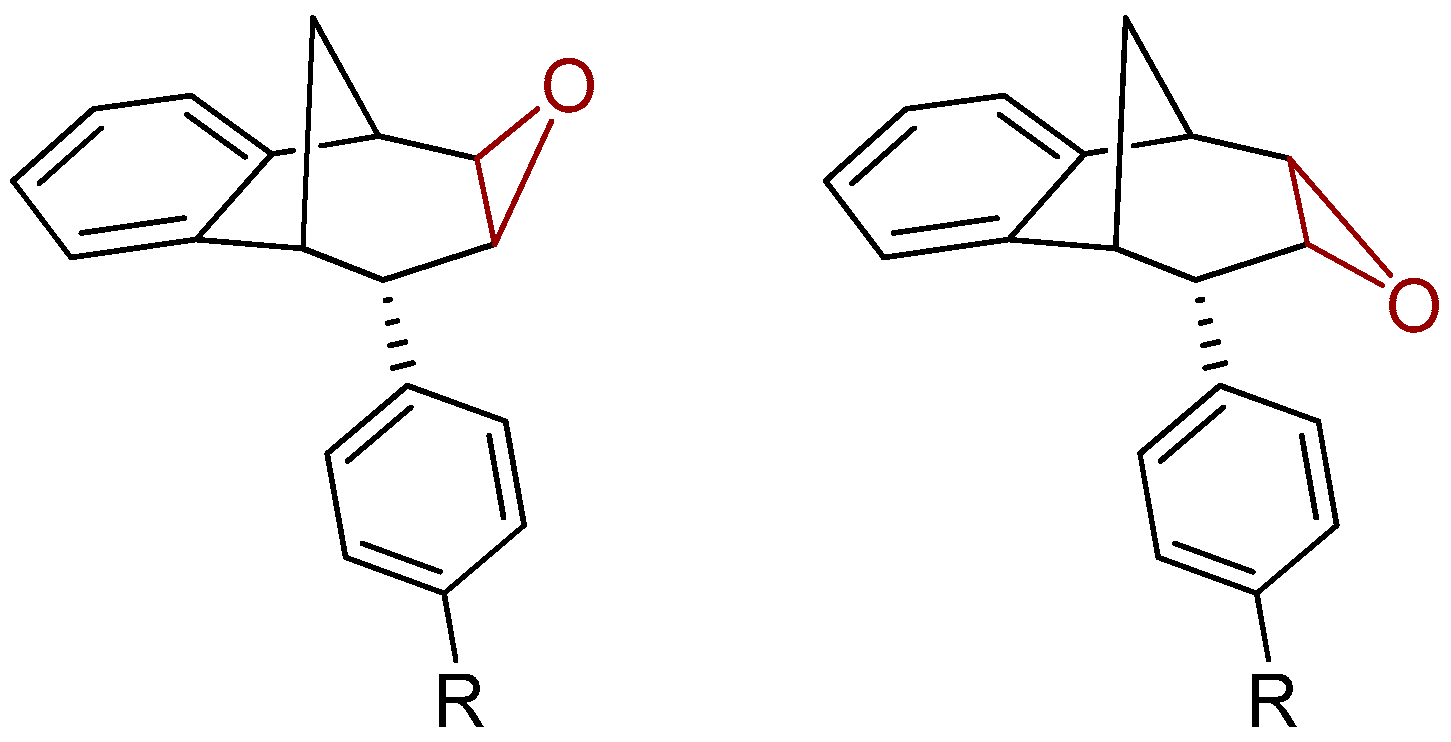
3.5. Synthesis of Epoxide Derivatives
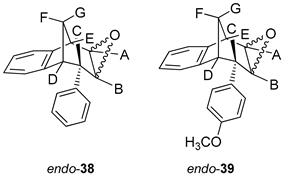
Synthesis of (2R,7R,8S,8aS)-8-Phenyl-1a,7,8,8a-tetrahydro-2H-2,7-methanobenzo[4,5]cyclohepta[1,2-b]oxirene (endo-38) and (2R,7R,8S,8aS)-8-(4-Methoxyphenyl)-1a,7,8,8a-tetrahydro-2H-2,7-methanobenzo[4,5]cyclohepta[1,2-b]oxirene (endo-39)
Epoxide derivatives endo-38 and endo-39 were prepared by epoxidation reaction using meta-chlorperbenzoic acid (m-CPBA). Photoproducts endo-36 and endo-37 were dissolved in 20 mL dry dioxane, and m-CPBA was added. The reaction mixture was stirred at room temperature overnight. After the reaction was complete, the saturated water solution of NaHCO3 was added. The organic layer was washed twice with water and dried over MgSO4, filtered and evaporated to obtain crude material. The crude material was purified using column chromatography.
![Molecules 25 04872 i004 Molecules 25 04872 i004]()
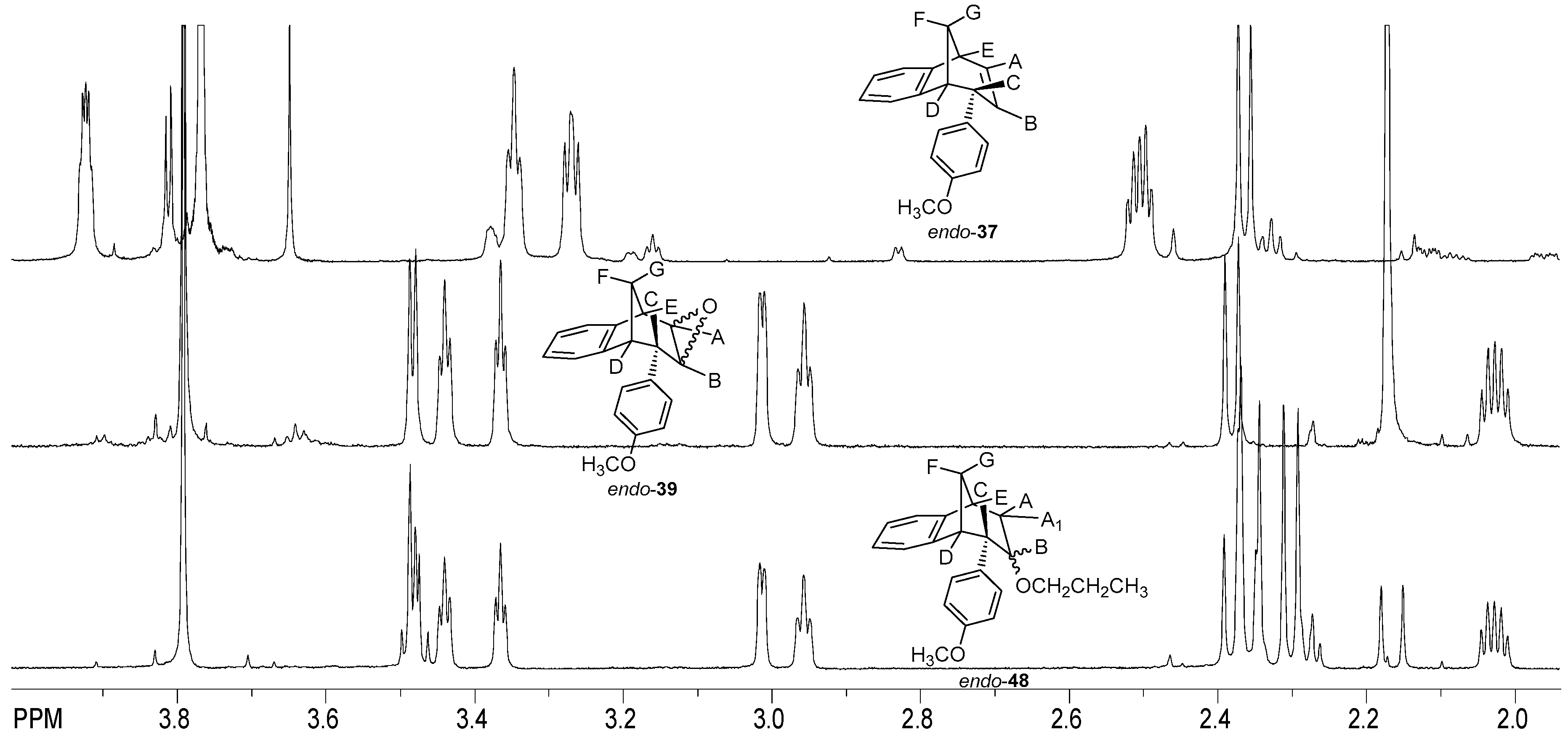
(2R,7R,8S)-8-Phenyl-1a,7,8,8a-tetrahydro-2H-2,7-methanobenzo[4,5]cyclohepta[1,2-b]-oxirene (endo-38): Yield 61%; Rf (petroleum ether/dichloromethane): 0.27; 1H NMR (cdcl3, 600 mhz) δ/ppm: 7.53 (d, 2H, J = 8.8 Hz, Ar), 7.30 (d, 1H, J = 7.4 Hz, Ar), 7.16 (t, 2H, J = 7.3 Hz, Ar), 6.93 (t, 1H, J = 7.3 Hz, Ar), 6.70 (dd, 2H, J = 8.8; 7.3 Hz, Ar), 6.26 (d, 1H, J = 7.4 Hz, Ar), 3.54 (d, 1H, J = 4.8 Hz, HE), 3.45 (t, 1H, J = 4.1 Hz, HA), 3.39 (t, 1H, J = 3.9 Hz, HB), 3.07 (d, 1H, J = 4.1 Hz, HC), 2.99 (t, 1H, J = 5.0 Hz, HD), 2.41 (d, 1H, J = 10.8 Hz, HG), 2.0–2.02 (m, 1H, HF);13C NMR (cdcl3, 75 mhz) δ/ppm: 144.6 (s), 143.2 (s), 141.2 (s), 133.2 (d), 129.3 (d), 128.2 (2d), 127.3 (2d), 126.4 (d), 125.3 (d), 122.1 (d), 54.1 (d), 52.5 (d), 45.8 (d), 43.2 (t), 40.1 (d), 34.9 (t); MS m/z (%, fragment) (EI): 248; HRMS (m/z) for C18H16O: [M + H]+calcd = 249.1201, [M + H]+measured = 249.1190.
(2R,7R,8S,8as)-8-(4-Methoxyphenyl)-1a,7,8,8a-tetrahydro-2H-2,7-methanobenzo[4,5]cyclohepta[1,2-b]oxirene (endo-39): Yield 50%; Rf (petroleum ether): 0.13; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.29 (d, 1H, J = 7.3 Hz, Ar), 7.16 (t, 1H, J = 7.2 Hz, Ar), 6.95 (t, 1H, J = 7.2 Hz, Ar), 6.75 (d, 2H, J = 8.3 Hz, Ar), 6.61 (d, 2H, J = 8.3 Hz, Ar), 6.32 (d, 1H, J = 7.3 Hz, Ar), 3.79 (s, 3H, OCH3), 3.48 (d, 1H, J = 5.0 Hz, HE), 3.44 (t, 1H, J = 4.0 Hz, HA), 3.36 (t, 1H, J = 3.8 Hz, HB), 3.00 (d, 1H, J = 4.0 Hz, HC), 2.95 (t, 1H, J = 5.0 Hz, HD), 2.38 (d, 1H, J = 10.8 Hz, HG), 2.05–2.00 (m, 1H, HF); 13C NMR (CDCl3, 150 MHz) δ/ppm: 152.5 (s), 142.9 (s), 135.9 (s), 133.8 (s), 129.6 (2d), 126.8 (d), 126.7 (d), 125.8 (d), 122.5 (d), 113.8 (2d), 55.2 (d), 54.2 (d), 53.2 (q), 44.8 (d), 42.9 (t), 40.6 (d), 35.3 (d);MS m/z (%, fragment) (EI): 278; HRMS (m/z) for C19H16O2: [M + H]+calcd = 279.1306, [M + H]+measured = 279.1325.
3.6. Epoxide Ring Opening
To a solution of starting epoxide (0.8g) in THF, lithium aluminum hydride (2.5 eq) was added. The reaction mixture was stirred at 76 °C during 24 h. After the reaction was complete, NaHCO3 was added and pH was adjusted to 7. The water layer was washed twice with diethyl ether. Organic layers were dried over MgSO4, filtered and evaporated to obtain crude material. Crude material was purified on column chromatography using petroleum ether/dichloromethane as eluent.
![Molecules 25 04872 i005 Molecules 25 04872 i005]()
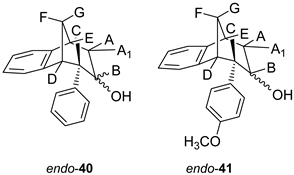
(6S)-6-Phenyl-6,7,8,9-tetrahydro-5H-5,9-methanobenzo[7]anulen-7-ol (endo-40): Yield 40%; Rf (petroleum ether/ dichloromethane 40%): 0.20; 1H NMR (CDCl3, 600 MHz) δ/ppm 8.00–7.30 (m, 4H, Ar), 6.93–6.20 (m, 5H, Ar), 4.13 (m, 1H, HB) 3.40 (dd, 1H, J = 12.7, 4.7 Hz, HA/A1/E) 3.20 (t, 1H, J = 4.5 Hz, HA/A1/E), 3.14 (d, 1H, J = 4.5 Hz, HA/A1/E), 2.56 (d, 1H, J = 10.8 Hz, HG), 2.35 (s, 1H, OH), 2.25–2.20 (m, 1H, HF) 1.63 (d, 1H, J = 4.8 Hz, HC/D), 1.60 (d, 1H, J = 4.8 Hz, HC/D); 13C NMR (CDCl3, 150 MHz) δ/ppm: 143.8 (s), 142.3 (s), 133.9 (s), 129.3 (d), 128.9 (d), 127.6 (d), 127.2 (2d), 126.5 (d), 124.3 (2d), 122.1 (d), 68.1 (d), 63.9 (t), 47.9 (d), 47.0 (d), 41.5 (d), 38.7 (t);MS m/z (%, fragment) (EI): 250; HRMS (m/z) for C18H18O: [M + H]+calcd = 251.1357, [M + H]+measured = 251.1349.
(5R,6S,9S)-6-(4-methoxyphenyl)-6,7,8,9-tetrahydro-5H-5,9-methanobenzo[7]anulen-7-ol (endo-41): Yield 35%; Rf (petroleum ether/ dichloromethane): 0.20; 1H NMR (CDCl3, 600 MHz) δ/ppm 7.85–7.32 (m, 3H, Ar), 6.98–6.24 (m, 5H, Ar), 4.31–4.28 (m, 1H, HB) 3.79 (s, 1H, OCH3) 3.65–3.63 (m, 1H, HA/A1/E) 3.44 (t, 1H, J = 4.4 Hz, HA/A1/E), 3.36 (t, 1H, J = 3.8 Hz, HA/A1/E), 3.01 (dd, 1H, J = 4.0, 1.4 Hz, HC/D), 2,96 (t, 1H, J = 5,2 Hz, HC/D) 2,38 (d, 1H, J = 11,0 Hz, HG), 2,27 (s, 1H, OH), 2,05–2,00 (m, 1H, HF); MS m/z (%, fragment) (EI): 280; HRMS (m/z) for C19H20O2: [M + H]+calcd = 281.1463, [M + H]+measured = 281.1609.
3.7. Synthesis of Ethers from Alcohols
Alcohol obtained after epoxide cleavage (0.05 g) was dissolved in acetone (2 mL), K2CO3 (1.5 eq) and corresponding alkyl bromides (1.1 eq) were added. The reaction mixture was stirred over 24 h at 56 °C. After the reaction was complete, water and diethylether were added. The organic layer was dried over MgSO4, filtered and evaporated to obtain a crude product. The crude material was purified on column chromatography using petroleum ether and dichloromethane as eluents.
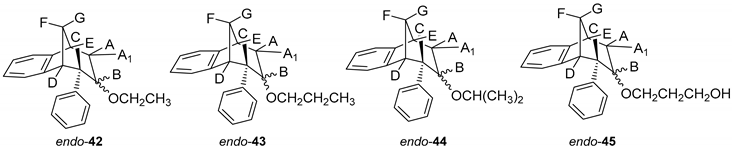
(5R,6S,9S)-7-Ethoxy-6-phenyl-6,7,8,9-tetrahydro-5H-5,9-methanobenzo[7]annulene (endo-42): Yield 20%; Rf (petroleum ether/dichloromethane): 0.52; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.72 (d, 1H, J = 7.3 Hz), 7.33–7.19 (m, 4H), 7.05–7.01(m, 2H), 7.14 (t, 1H, J = 7.4 Hz), 6.74 (t, 1H, J = 7.3 Hz), 4.16–4.11 (m, 2H), 3.35–3.30 (m, 2H), 3.01 (d, 1H, J = 11.0 Hz), 2.18–2.13 (m, 1H), 2.09–2.07 (m, 1H), 1.89 (dd, 2H, J = 15.2, 4.5 Hz), 1.76–1.73 (t, 3H, J = 6.9 Hz, CH3); MS m/z (%, fragment) (EI): 280 (100).
(5R,6S,9S)-6-Phenyl-7-propoxy-6,7,8,9-tetrahydro-5H-5,9-methanobenzo[7]annulene (endo-43): Yield 40%; Rf (petroleum ether/dichloromethane): 0.57; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.42 (t, 1H, J = 7.5 Hz), 7.33 (d,1H, J = 7.1 Hz), 7.28 (d, 2H, J = 7.9 Hz), 7.23 (t, 2H, J = 7.5 Hz), 7.14 (t, 2H, J = 7.9 Hz), 6.48 (d, 1H, J = 7.1 Hz), 4.68–4.63 (m, 2H), 4.19–4.14 (m, 2H), 3.31 (m, 2H), 3.03 (d, 1H, J = 11.0 Hz), 2.18–2.13 (m, 1H), 2.09–2.07 (m, 1H), 1.89 (dd, 2H, J = 15.2; 4.5 Hz), 1.71 (d, 1H, J = 10.5 Hz), 1.28 (t, 3H, J = 6.9 Hz, CH3); 13C NMR (CDCl3, 150 MHz) δ/ppm: 145.3 (s), 145.1 (s), 144.3 (s), 127.6 (d), 127.5 (2d), 126.9 (d), 126.6 (2d), 125.7 (d), 124.5 (d), 122.5 (d), 68.8 (d), 53.3 (d), 47.5 (t), 46.8 (d), 39.7 (t), 36.9 (t), 33.8 (t), 32.4 (d), 25.5 (q);MS m/z (%, fragment) (EI): 292 (100); HRMS (m/z) for C21H24O: [M + H]+calcd = 293.1827, [M + H]+measured = 293.1846.
(5R,6S,9S)-7-Isopropoxy-6-phenyl-6,7,8,9-tetrahydro-5H-5,9-methanobenzo[7]annulene (endo-44): Yield 50%; Rf (petroleum ether/dichloromethane) = 0.55; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.33 (t, 1H, J = 7.0 Hz), 7.30–7.28 (m, 1H), 7.23 (t, 3H, J = 7.7 Hz), 7.14 (d, 1H, J = 7.3 Hz), 6.98 (d, 1H, J = 7.3 Hz), 6.47 (d, 2H, J = 7.7 Hz), 4.19–4.14 (m, 1H), 3.50–3.46 (m, 1H, isopropyl-CH), 3.24–3.20 (m, 2H), 2.98 (d, 1H, J = 11.1 Hz), 2.17–2.12 (m, 1H), 2.06–2.01 (m, 1H), 1.81 (dd, 1H, J = 15.0; 4.3 Hz), 1.67 (d, 1H, J = 15.0 Hz), 1.29 (broad s, 6H);13C NMR (CDCl3, 150 MHz) δ /ppm: 145.4 (s), 145.2 (s), 144.3 (s), 127.5 (2d), 126.9 (d), 126.8 (d), 126.5 (2d), 125.7 (d), 124.8 (d), 122.5 (d), 68.8 (d), 53.3 (d), 46.8 (d), 43.2 (d), 39.7 (d), 36.9 (t), 34.1 (t), 27.4 (2q); MS m/z (%, fragment) (EI): 292 (100); HRMS (m/z) for C21H24O: [M + H]+calcd = 293.1827, [M + H]+measured = 293.1814.
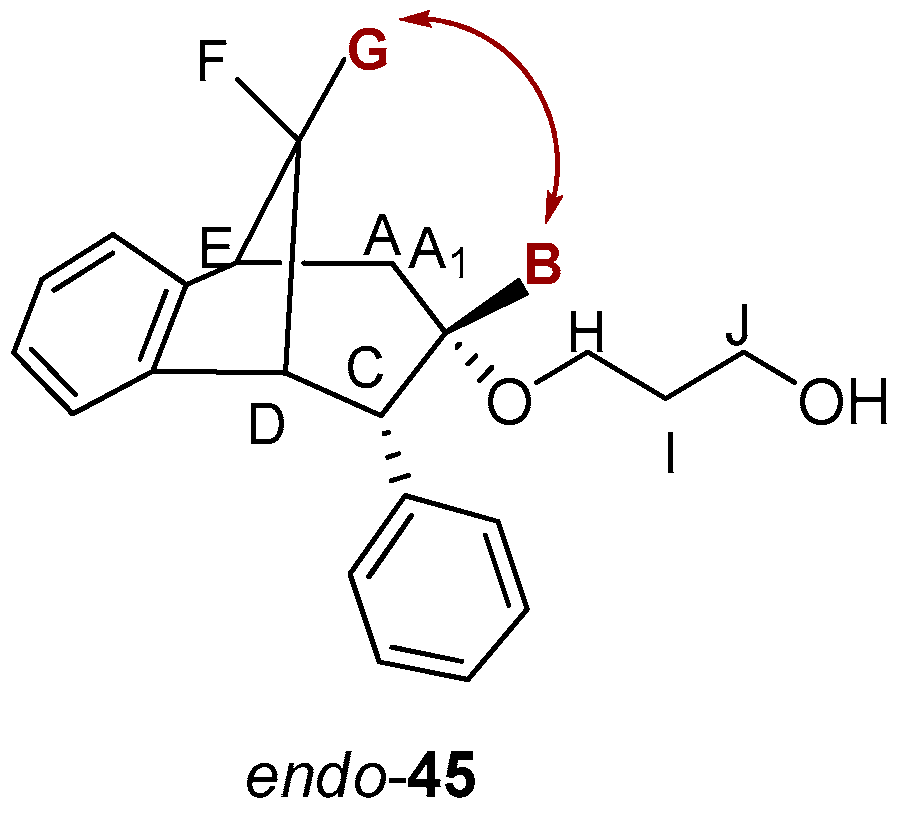
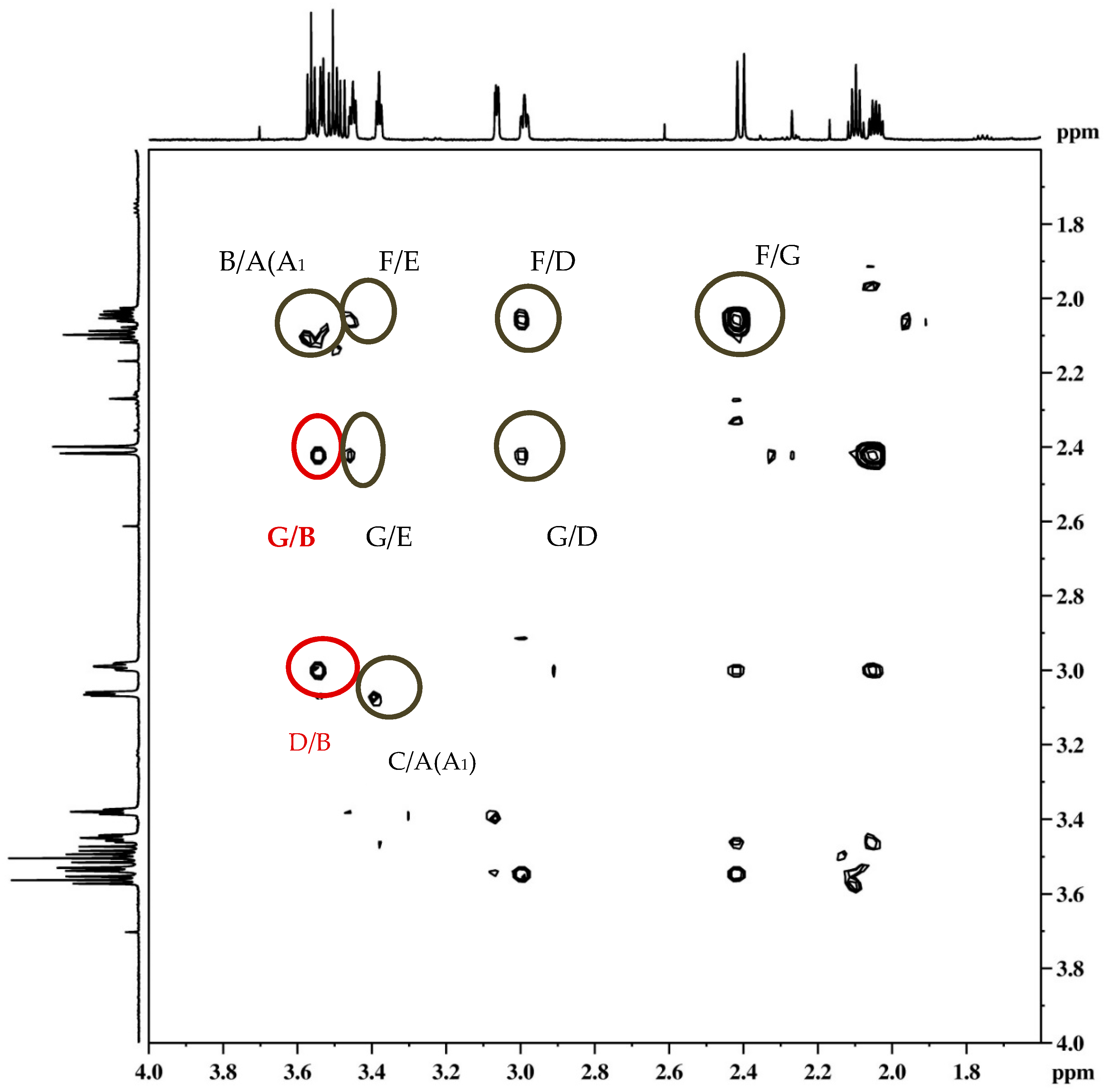
3-(((5R,6S,9S)-6-Phenyl-6,7,8,9-tetrahydro-5H-5,9-methanobenzo[7]annulen-7-il)oxy)propan-1-ol (endo-45): Yield 45%; Rf (petroleum ether/dichloromethane): 0.50; 1H NMR (CDCl3, 600 MHz) δ/ppm 7.28 (d, 1H, J = 7.3 Hz, Ar), 7.21–7.18 (m, 3H, Ar), 7.15 (t, 1H, J = 7.3 Hz, Ar), 6.92 (t, 1H, J = 7.3 Hz, Ar), 6.69 (dd, 2H, J = 6.6; 3.3 Hz, Ar), 6.24 (d, 1H, J = 7.3 Hz, Ar), 3.57–3.44 (m, 4H), 3.52 (d, 1H, J = 4.8 Hz, HB), 3.44 (t, 1H, J = 4.2 Hz, HE), 3.37 (t, 1H, J = 3.8 Hz, HA/A1), 3.05 (dd, 1H, J = 4.2 Hz, HC), 2.98 (t, 1H, J = 5.1 Hz, HD), 2.40 (d, 1H, J = 10.7 Hz, HG), 2.11–2.06 (m, 1H, HA/A1), 2.06–2.01 (m, 1H, HF), 1.42 (s, 1H, OH), 1.24 (broad s, 2H); 13C NMR (CDCl3, 150 MHz) δ /ppm: 145.2 (s), 143.5 (s), 141.7 (s), 128.6 (2d), 127.7 (2d), 126.8 (d), 126.3 (d), 126.0 (d), 125.7 (d), 122.5 (d), 68.2 (d), 54.5 (t), 52.9 (d), 50.4 (t), 45.9 (d), 43.7 (d), 40.6 (t), 39.9 (t), 35.5 (t); MS m/z (%, fragment) (EI): 308; HRMS (m/z) for C21H24O2: [M + H]+calcd = 309.1776, [M + H]+measured = 309.4140.
![Molecules 25 04872 i007 Molecules 25 04872 i007]()
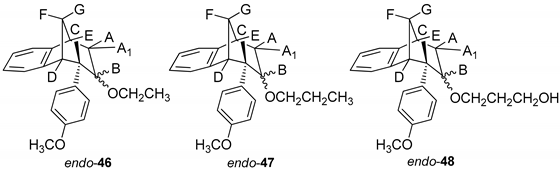
(5R,6S,9S)-7-Ethoxy-6-(4-methoxyphenyl)-6,7,8,9-tetrahydro-5H-5,9-methanobenzo-[7]annulene (endo–46): Yield 20%; Rf (petroleum ether/dichloromethane): 0.67; 1H NMR (CDCl3, 600 MHz) δ/ppm 7.30 (d, 1H, J = 7.5 Hz), 7.17 (t, 1H, J = 7.5 Hz), 6.96 (t, 1H, J = 7.5 Hz), 6.76 (d, 2H, J = 8.6 Hz), 6.62 (d, 2H, J = 8.6 Hz), 6.33 (d, 1H, J = 7.5 Hz), 3.79 (s, 3H), 3.51–3.48 (m, 2H), 3.45 (t, 1H, J = 4.8 Hz), 3.38 (t, 1H, J = 4.2 Hz), 3.03 (d, 1H, J = 4.2 Hz), 2.97 (t, 1H, J = 4.8 Hz), 2.38-2.33 (m, 2H), 2.39 (d, 1H, J = 10.8 Hz), 2.06–2.01 (m, 1H), 1.27 (s, 3H); MS m/z (%, fragment) (EI): 308 (100).
(5R,6S,9S)-6-(4-Methoxyphenyl)-7-propoxy-6,7,8,9-tetrahydro-5H-5,9-methanobenzo[7]annulene (endo-47): Yield 35%; Rf (petroleum ether/dichloromethane):0.57; 1H NMR (CDCl3, 600 MHz) δ/ppm 7.21 (d, 1H, J = 7.3 Hz), 7.08 (t, 1H, J = 7.4 Hz), 6.87 (t, 1H, J = 7.4 Hz), 6.67 (d, 2H, J = 8.6 Hz), 6.53 (d, 2H, J = 8.6 Hz), 6.24 (d, 1H, J = 7.3 Hz), 3.70 (s, 3H, OCH3), 3.51 (q, 2H, J = 6.6 Hz), 3.40 (t, 1H, J = 4.8 Hz), 3.36 (t, 1H, J = 4.3 Hz), 2.93 (d, 1H, J = 3.7 Hz), 2.87 (t, 1H, J = 4.9 Hz), 2.39–2.35 (m, 4H), 2.30 (d, 1H, J = 10.8 Hz), 1.99–1.96 (m, 1H), 1.18 (broad s, 3H); 13C NMR (CDCl3, 150 MHz) δ/ppm: 157.5 (s), 144.5 (s), 143.2 (s), 133.5 (s), 129.2 (d), 127.1 (d), 126.4 (2d), 126.2 (d), 122.1 (2d), 112.6 (d), 54.7 (d), 54.0 (d), 52.7 (d), 45.5 (d), 42.4 (d), 40.1 (d), 34.7 (t), 30.4 (q); MS m/z (%, fragment) (EI): 322; HRMS (m/z) for C22H26O2: [M + H]+calcd = 323.1932, [M + H]+measured = 323.4406.
3-(((5R,6S,9S)-6-(4-Methoxyphenyl)-6,7,8,9-tetrahydro-5H-5,9-methanobenzo-[7]annulen-7-il)oxy)propan-1-ol (endo-48): Yield 15%; Rf (petroleum ether/dichloromethane): 0.64; 1H NMR (CDCl3, 600 MHz) δ/ppm 6.95 (d, 1H, J = 7.4 Hz), 6.82 (t, 1H, J = 7.3 Hz), 6.75 (t, 1H, J = 7.3 Hz), 6,71 (d, 2H, J = 8,5 Hz, Ar), 6,63 (d, 1H, J = 7,4 Hz, Ar), 6,46 (d, 2H, J = 8,6 Hz, Ar), 4,20–4,16 (m, 1H), 3,72–3,66 (m, 4H, CH2CH2), 3,64 (s, 3H, OCH3), 3,53 (t, 1H, J = 4,8 Hz), 3,31–3,25 (m, 1H), 2,96 (s, 1H, OH), 2,57 (dt, 1H, J = 14,3; 3,4 Hz), 2,44–2,39 (m, 1H), 2,28–2,23 (m, 1H), 2,04–1,97 (m, 2H), 1,17 (broad s, 2H); MS m/z (%, fragment) (EI): 338; HRMS (m/z) for C22H26O3: [M + H]+calcd = 339.1881, [M + H]+measured = 339.4404.
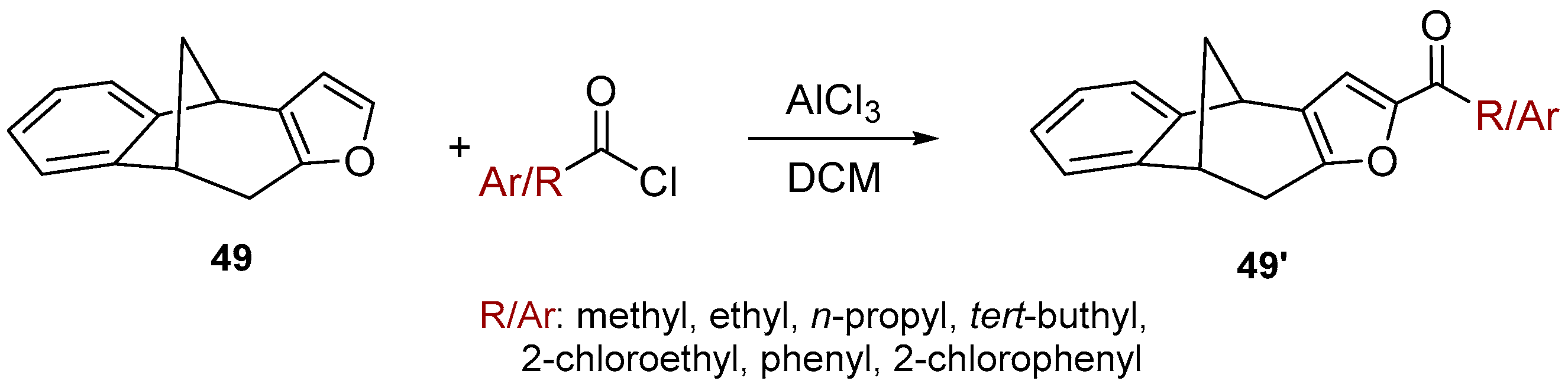
3.8. Friedel-Crafts Acylation
To the solution of compound 49 [
41] (100 mg, 0.510 mol) in dichloromethane (2 mL), AlCl
3 (68 mg, 0.510 mol) and different carbonyl chlorides (1.1 eq) were added. The reaction was performed in a sealed tube and left overnight at room temperature. After removal of the solvent, the residue crude product was purified by repeated column chromatography using petroleum ether/dichloromethane mixture as eluent. All of the obtained compounds
50–
57 were isolated in the first fractions.
![Molecules 25 04872 i008 Molecules 25 04872 i008]()
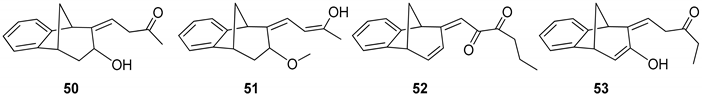
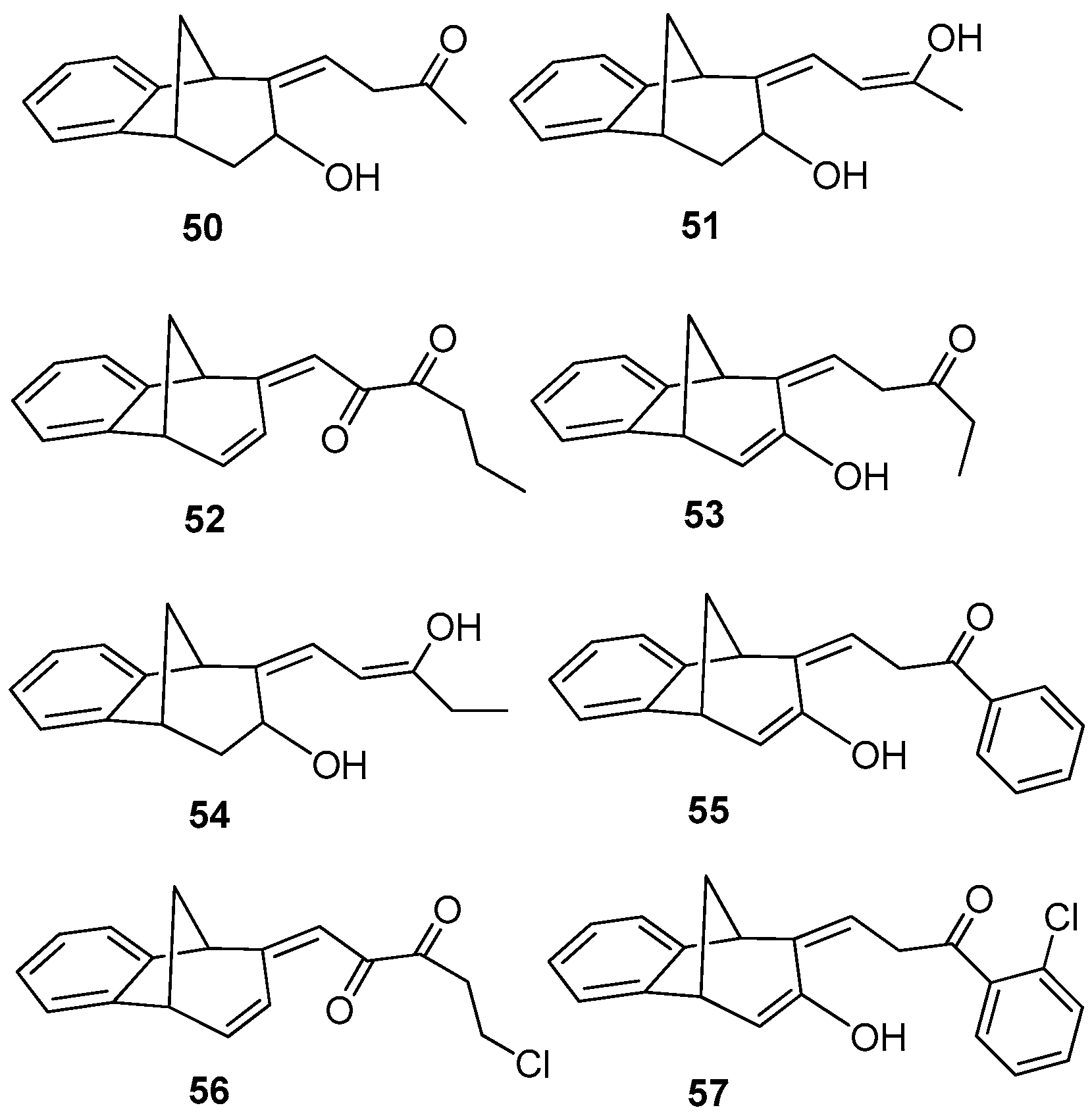
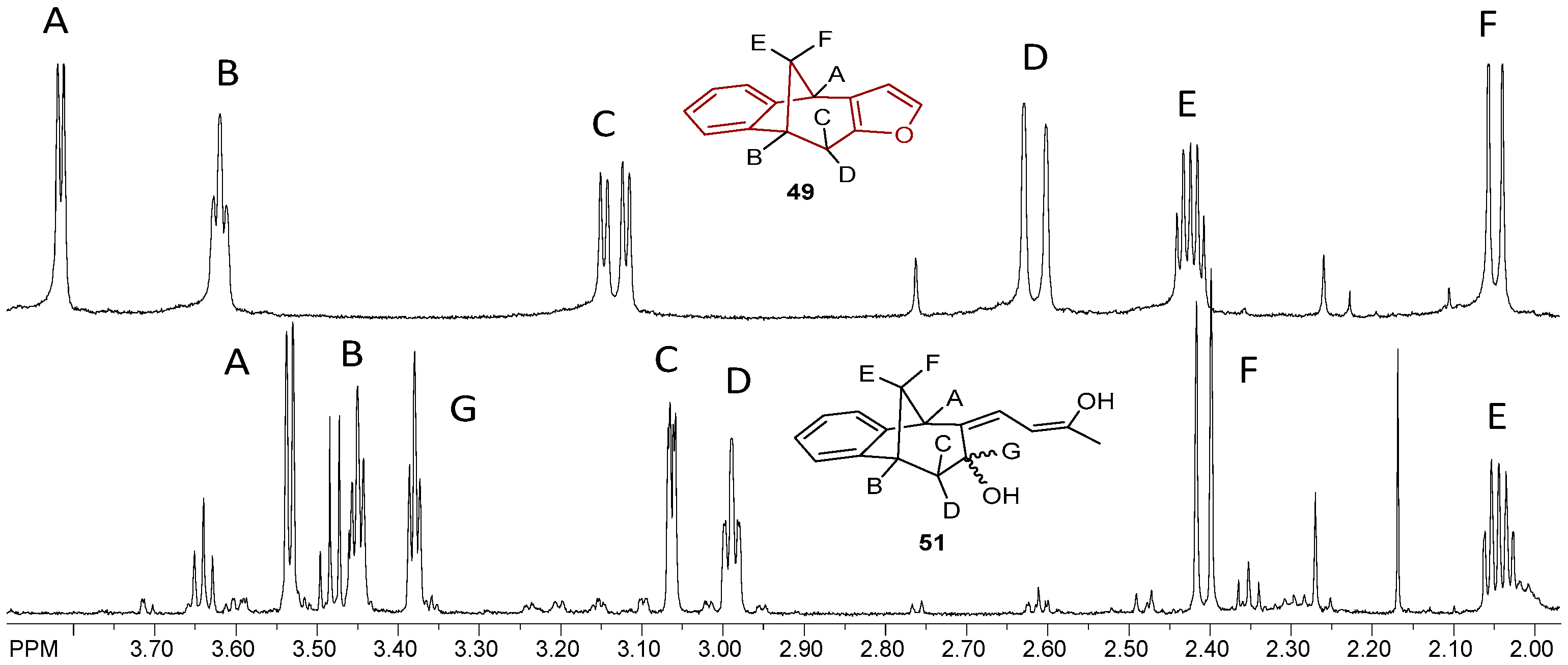
(Z)-4-((5R,9R)-7-Hydroxy-5,7,8,9-tetrahydro-6H-5,9-methanobenzo[7]annulen-6-ilydene)butan-2-one (50): 16 mg (13%); yellow oil; Rf (dichloromethane): 0.5;1H NMR (CDCl3, 600 MHz) δ/ppm: 7.54 (d, 1H, J = 7.7 Hz, HAr), 7.29 (t, 1H, J = 7.7 Hz, HAr), 7.14 (t, 1H, J = 7.7 Hz, HAr), 7.10 (d, 1H, J = 7.7 Hz, HAr), 6.79 (m, 1H), 3.83 (m, 1H), 3.62 (d, 1H, J = 3.6 Hz), 3.24 (t, 1H, J = 6.6 Hz), 3.01 (d, 1H, J = 4.6 Hz), 2.45 (m, 1H), 2.28 (s, 1H, -OH), 2.31 (d, 1H, J = 10.7 Hz,), 2.29 (m, 2H), 2.06–2.02 (m, 1H), 1.95 (s, 3H); MS m/z (%, fragment) (EI): 242 (100); HRMS (m/z) for C16H18O2: [M + H]+calcd = 243.1306, [M + H]+measured = 243.3129.
(5R,9R,Z)-6-((Z)-3-Hydroxybut-2-ene-1-ilydene)-6,7,8,9-tetrahydro-5H-5,9-methano-benzo[7]annulen-7-ol (51): 30 mg (30%); yellow oil; Rf (dichloromethane/petroleum ether 70%): 0.10; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.30 (d, 1H, J = 7.5 Hz, HAr), 7.16 (t, 1H, J = 7.5 Hz, HAr), 6.93 (t, 1H, J = 7.5 Hz, HAr), 6.70 (m, 2H), 6.26 (d, J = 7.3 Hz, 1H), 3.53 (d, 1H, J = 5.1 Hz), 3.45 (t, 1H, J = 4.4 Hz), 3.38 (t, 1H, J = 3.9 Hz), 3.06 (dd, 1H, J = 4.0; 1.5 Hz), 2.99 (t, 1H, J = 5.1 Hz), 2.41 (d, 1H, J = 10.9 Hz), 2.10 (s, 1H, OH), 2.06–2.02 (m, 1H), 1.53 (s, 3H); MS m/z (%, fragment) (EI): 242 (100); HRMS (m/z) for C16H18O2: [M + H]+calcd = 243.1306, [M + H]+measured = 243.3131.
(E)-1-((5S,9R)-5,9-Dihydro-6H-5,9-methanobenzo[7]annulen-6-ilydene)hexa-2,3-dione (52): 21 mg (15%); yellow oil; Rf (dichloromethane): 0.40; 1H NMR (CDCl3, 600 MHz) δ/ppm: 8.07 (d, 1H, J= 7.9 Hz, HAr), 7.83 (d, 1H, J = 7.9 Hz, HAr), 7.13–7.09 (m, 2H), 6.26 (dd, 1H, J = 6.8; 1.8 Hz), 6.02 (d, 1H, J = 9.6 Hz), 5.91 (d, 1H, J = 9.6 Hz), 4.25–4.19 (m, 2H), 3.55–3.51 (m, 2H), 2.71 (d, 1H, J = 11.2 Hz), 2.35–2.31 (m, 2H), 2.04–2.01 (m, 1H), 1.63 (s, 3H); MS m/z (%, fragment) (EI): 266 (100); HRMS (m/z) for C18H18O2: [M + H]+calcd = 267.1306, [M + H]+measured = 267.3343.
(Z)-1-((5R,9R)-7-Hydroxy-5,9-dihydro-6H-5,9-methanobenzo[7]annulen-6-ilydene)-pentan-3-one (53): 76 mg (59%); yellow oil; Rf (dichloromethane): 0.29; 1H NMR (CDCl3, 600 MHz) δ/ppm: 8.04 (d, 1H, J = 8.5 Hz, HAr), 7.93 (d, 1H, J = 9.1 Hz, HAr), 7.50 (t, 1H, J = 7.7 Hz, HAr), 7.16 (m, 1H, HAr), 6.30 (dd, 1H, J = 3.1; 1.9 Hz), 6.10 (d, 1H, J = 3.1 Hz), 4.59 (d, 1H, J = 6.3 Hz), 4.42 (d, 1H, J = 5.6 Hz), 4.33 (s, 1H, OH), 4.10–4.07 (m, 2H), 2.28 (d, 1H, J = 11.5 Hz), 1.93–1.83 (m, 1H) 1.80–1.69 (m, 2H), 1.00 (t, 3H, J = 5.4 Hz); MS m/z (%, fragment) (EI): 254 (100); HRMS (m/z) for C17H18O2: [M + H]+calcd = 255.1306, [M + H]+measured = 255.3236.
![Molecules 25 04872 i009 Molecules 25 04872 i009]()
(9Z)-9-[(Z)-3-Hydroxypent-2-enilydene]tricyclo[6.3.1.02,7]dodeca-2(7),3,5-trien-10-ol (54): 8.9 mg (12%); yellow oil; Rf (dichloromethane/petroleum ether 20%): 0.73; 1H NMR (CDCl3, 600 MHz) δ/ppm: 7.44–7.02 (m, 4H), 6,64 (m, 1H), 6.27 (d, 1H, J = 7.8 Hz), 3.63 (q, 2H, J = 6.8 Hz), 3.51 (d, 1H, J = 4.5 Hz), 3.44 (t, 1H, J = 4.5 Hz), 3.37 (t, 1H, J = 4.5 Hz), 2.99 (dd, 1H, J = 3.0; 1.5 Hz), 2.95 (t, 1H, J = 4.5 Hz), 2.38 (d, 1H, J = 10.6 Hz), 2.05-2.02 (m, 1H), 1.56 (t, 3H, J = 7.2 Hz); MS m/z (%, fragment) (EI): 256 (100).
(Z)-3-((5R,9R)-7-Hydroxy-5,9-dyhidro-6H-5,9-methanobenzo[7]annulen-6-ilydene)-1-phenylpropan-1-one (55): 29.4 mg (19%); colourless oil; Rf (dichloromethane): 0.38; 1H NMR (CDCl3, 600 MHz) δ/ppm: 8.01 (d, 2H, J = 8.7 Hz, HAr), 7.90 (t, 2H, J = 8.9 Hz, HAr), 7.71 (d, 1H, J = 8.3 Hz, HAr), 7.68 (d, 1H, J = 9.2 Hz, HAr), 7.50–7.43 (m, 3H, HAr), 6.30 (m, 1H), 6.10 (d, 1H, J = 3.3 Hz), 4.71 (s, 1H, OH), 4.58 (d, 1H, J = 4.3 Hz), 4.39 (d, 1H, J = 4.9 Hz), 4.12 (t, 2H, J = 4.9 Hz), 2.40 (d, 1H, J = 10.5 Hz), 2.33–2.28 (m, 1H); MS m/z (%, fragment) (EI): 302 (100).
(E)-5-Chloro-1-((5S,9R)-5,9-dyhdro-6H-5,9-methanobenzo[7]annulen-6-ilydene)penta-2,3-dione (56): 7.5 mg (7.3%); colorless oil; Rf (dichloromethane): 0.42; 1H NMR (CDCl3, 600 MHz) δ/ppm: 8.01 (d, 1H, J = 7.6 Hz, HAr), 7.13 (d, 1H, J = 7.6 Hz, HAr), 7.10–7.05 (m, 2H, HAr), 6.26 (dd, 1H, J = 6.5; 1.6 Hz), 6.14 (d, 1H, J = 9.3 Hz), 5.98 (d, 1H, J = 9.3 Hz), 4.25–4.19 (m, 2H), 3.55 (m, 2H), 2.71 (d, 1H, J = 11.1 Hz), 2.33 (m, 2H), 2.07–2.03 (m, 1H); MS m/z (%, fragment) (EI): 286 (100), 288 (32).
(Z)-1-(2-Chlorophenyl)-3-((5R,9R)-7-hydroxy-5,9-dihydro-6H-5,9-methanobenzo-[7]annulen-6-ilydene)propan-1-one (57): 9.5 mg (8%); colorless oil; Rf (dichloromethane): 0.40; 1H NMR (CDCl3, 600 MHz) δ/ppm: 8.02 (d, 1H, J = 8.9 Hz, HAr), 7.90 (t, 2H, J = 9.2 Hz, HAr), 7.81 (d, 1H, J = 8.3 Hz, HAr), 7.70 (d, 1H, J = 9.2 Hz, HAr), 7.50–7.43 (m, 3H, HAr), 6.28 (dd, 1H, J = 3.2; 1.8 Hz), 6.08 (d, 1H, J = 3.1 Hz), 4.55 (d, 1H, J = 6.1 Hz), 4.21 (d, 1H, J = 5.7 Hz), 3.98 (t, 2H, J = 6.1 Hz), 3.18 (s, 1H, -OH), 2.03–1.99 (m, 2H); MS m/z (%, fragment) (EI): 336 (100), 338 (32).
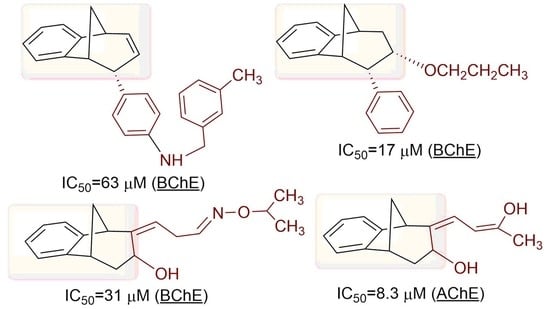
3.9. Reversible Inhibition of Cholinesterases by Novel Compounds
Inhibition of novel compounds was evaluated for recombinant human AChE and BChE derived from purified human plasma (a generous gift from Dr Florian Nachon,
Département de Toxicologie et Risques Chimiques, Institut de Recherche Biomédicale des Armées, Bretigny-sur-Orge, France). The inhibition mixture contained a 0.1 M phosphate buffer, pH 7.4, enzyme (4.5 nM BChE or 0.2 nM AChE) tested compound, and DTNB (0.3 mM; Sigma Chemical Co., St. Louis, MO, USA). Enzyme activity was measured upon addition of ATCh (0.2 or 0.1 mM; Sigma Chemical Co., St. Louis, MO, USA) by the Ellman method [
45] at 25 °C and 412 nm, on a Tecan Infinite M200PRO plate reader (Tecan Austria, GmbH, Salzburg, Austria). Due to the low solubility, a 100 mM stock solution of the tested compounds was prepared in DMSO, and the same solvent was in controls as well. The IC
50 values were determined from at least three experiments by a nonlinear fit of the compound concentration logarithm values vs.% of enzyme activity using Prism6 software (GraphPad Prism 6 Software, San Diego, USA).