New Insights into The Photoactivity of Shape-Tailored BiVO4 Semiconductors via Photocatalytic Degradation Reactions and Classical Reduction Processes
Abstract
1. Introduction
2. Results and Discussion
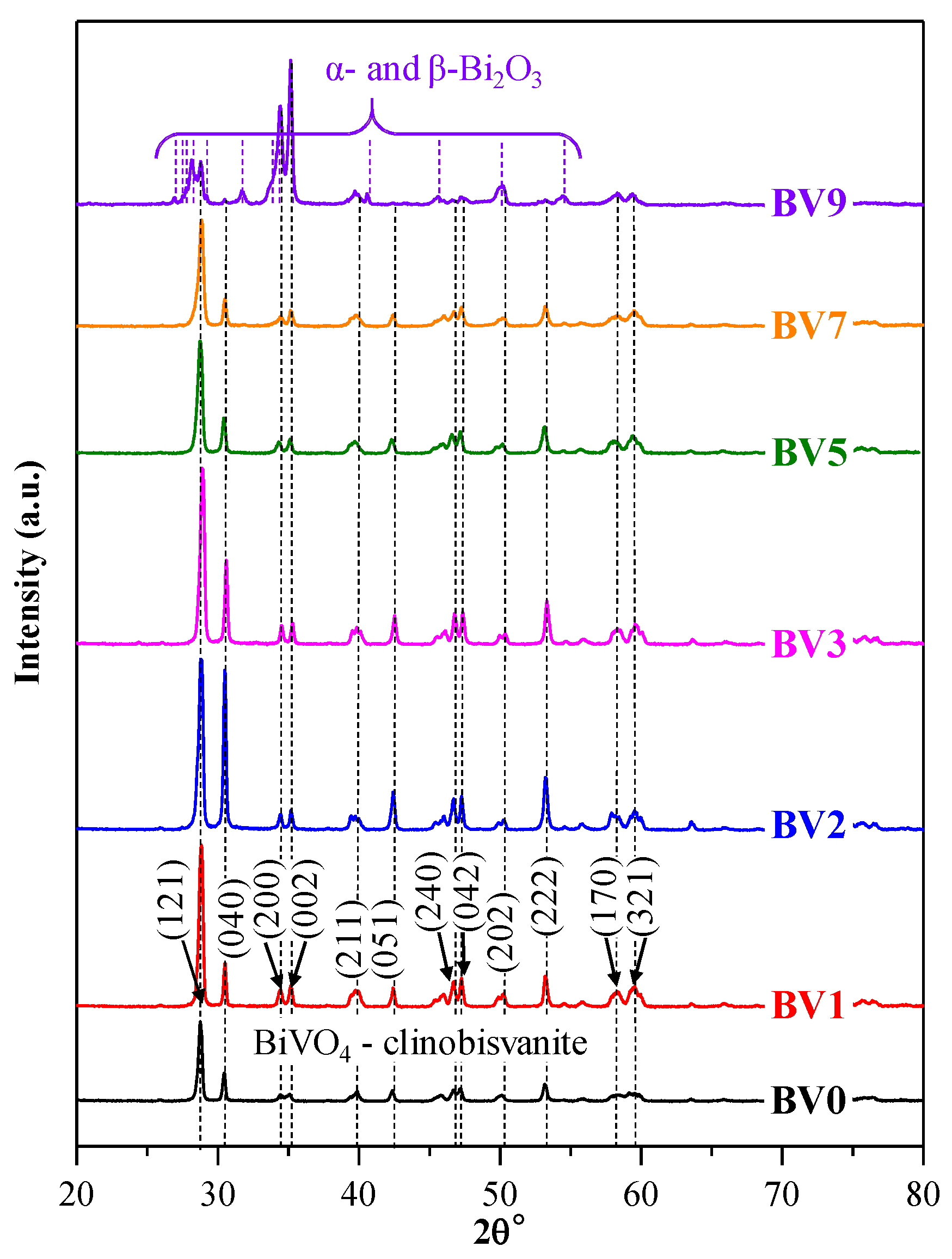
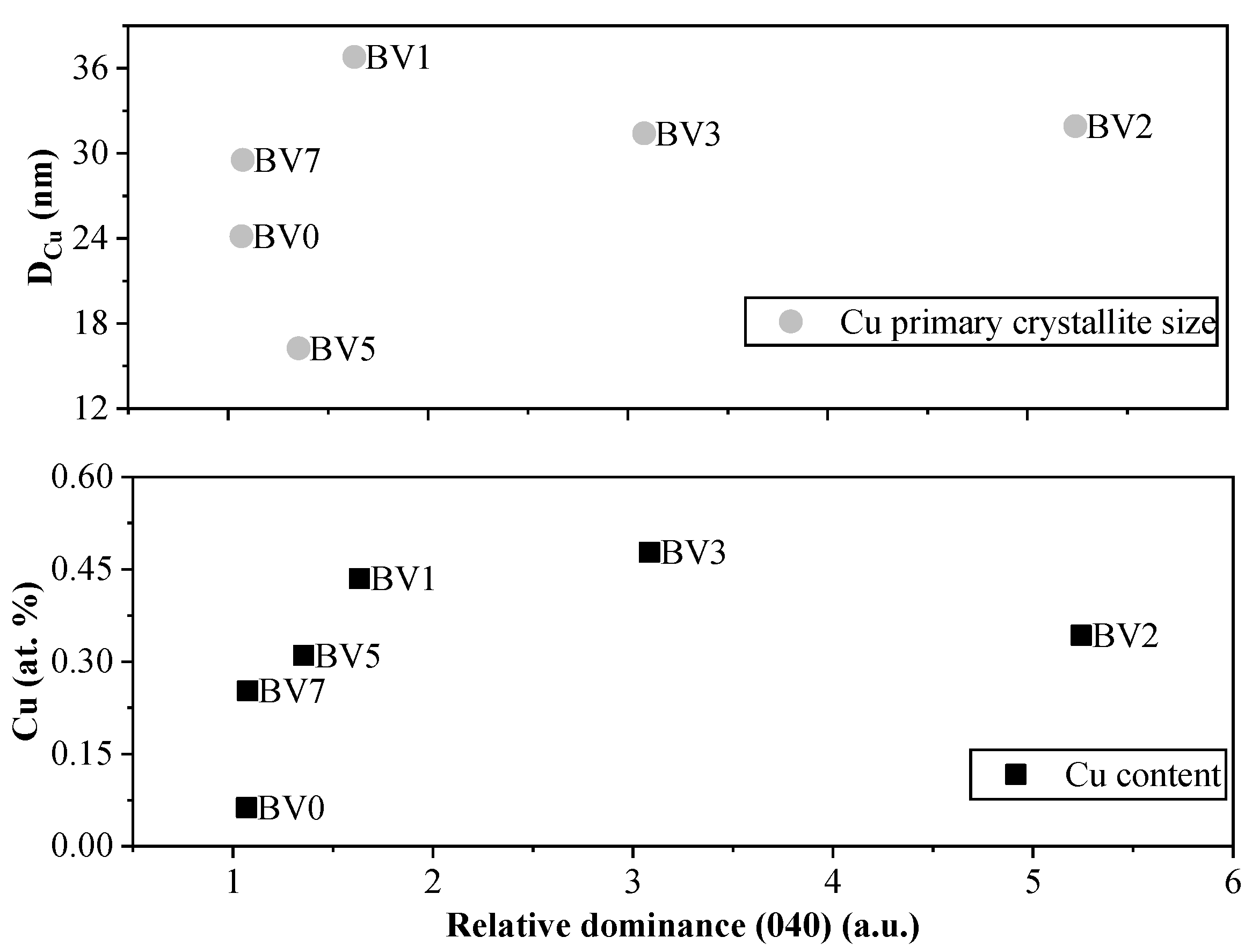
2.1. Crystal Phase Composition and Crystal Size
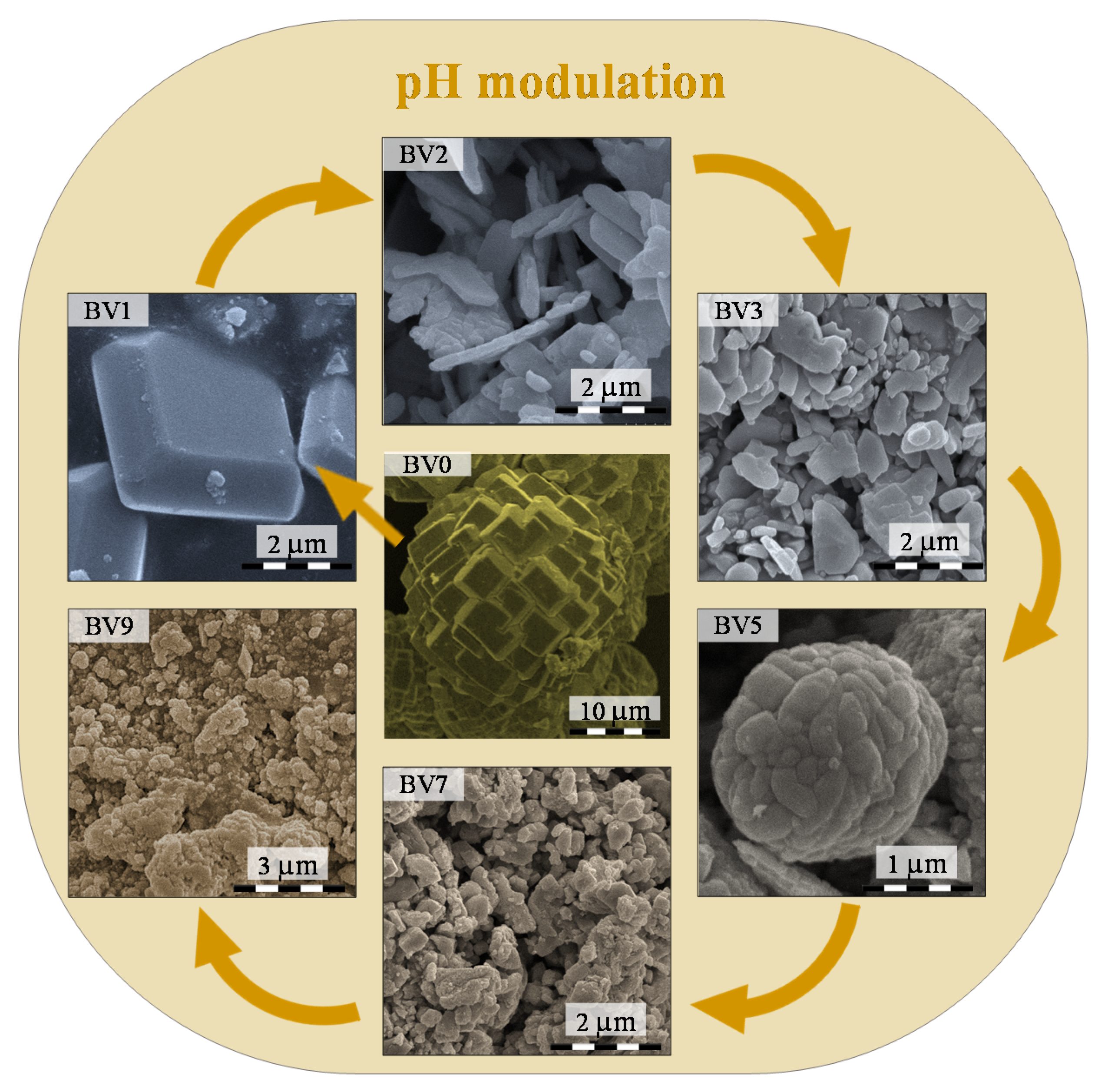
2.2. Morphological Characterization
2.3. Optical Properties
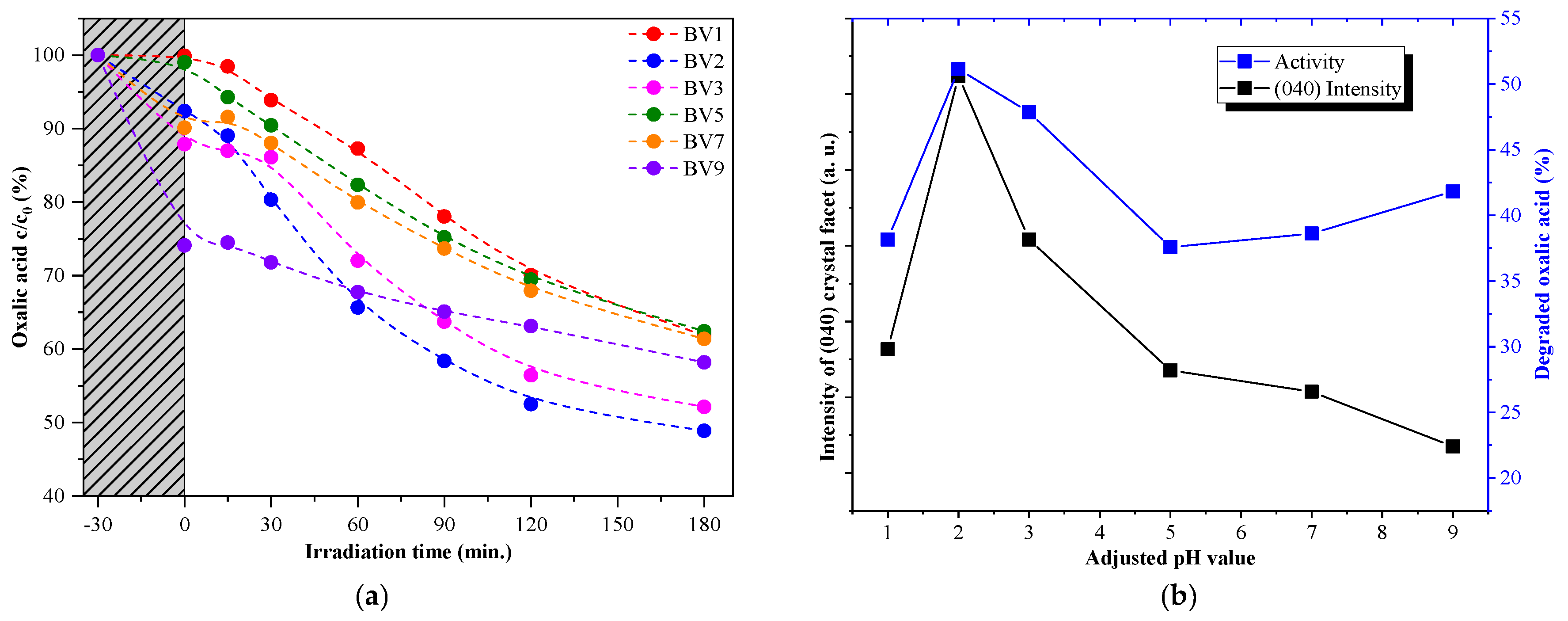
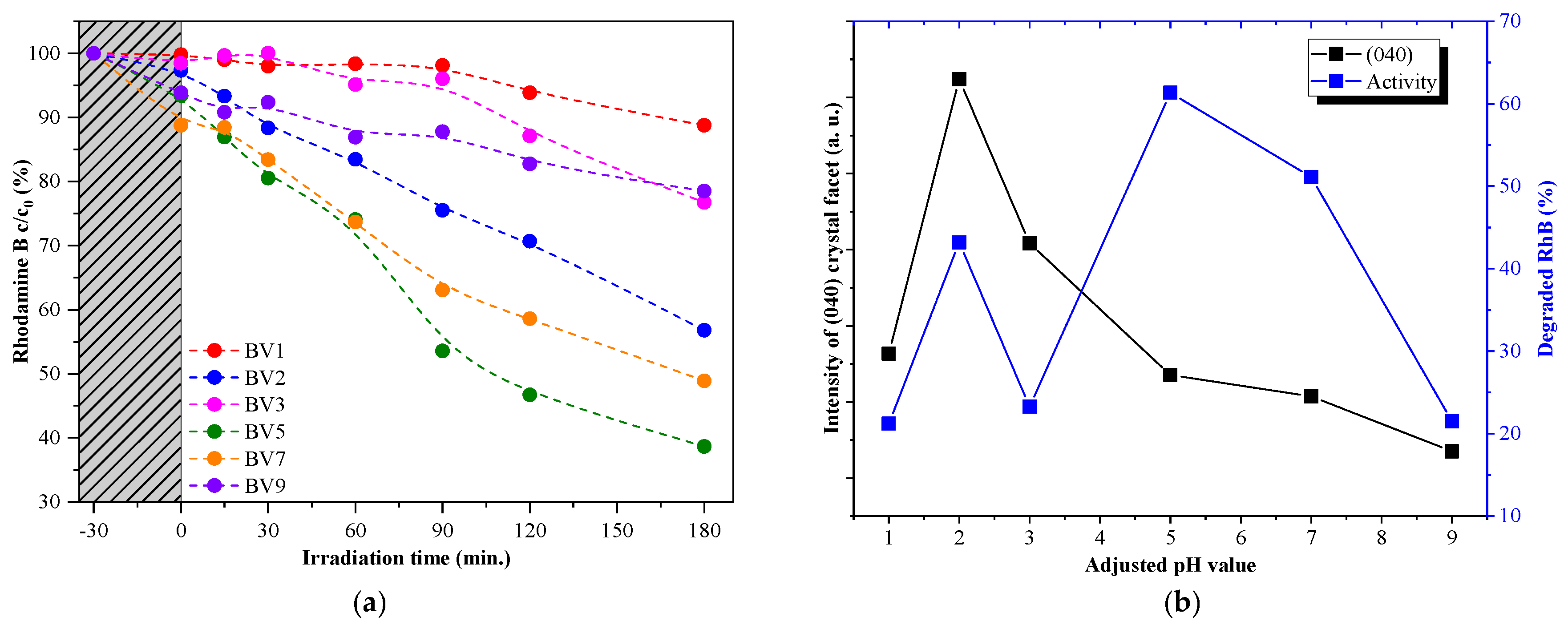
2.4. Photocatalytic Activity under Visible Light Irradiation
- Oxalic acid is usually degraded by a direct hole oxidation mechanism, as this compound can be easily adsorbed on the surface of several photocatalysts [43].
- The degradation of rhodamine B starts with an electron transfer step between the dye molecule and the photocatalyst. In addition, direct hole oxidation is an option as stated in the literature [44].
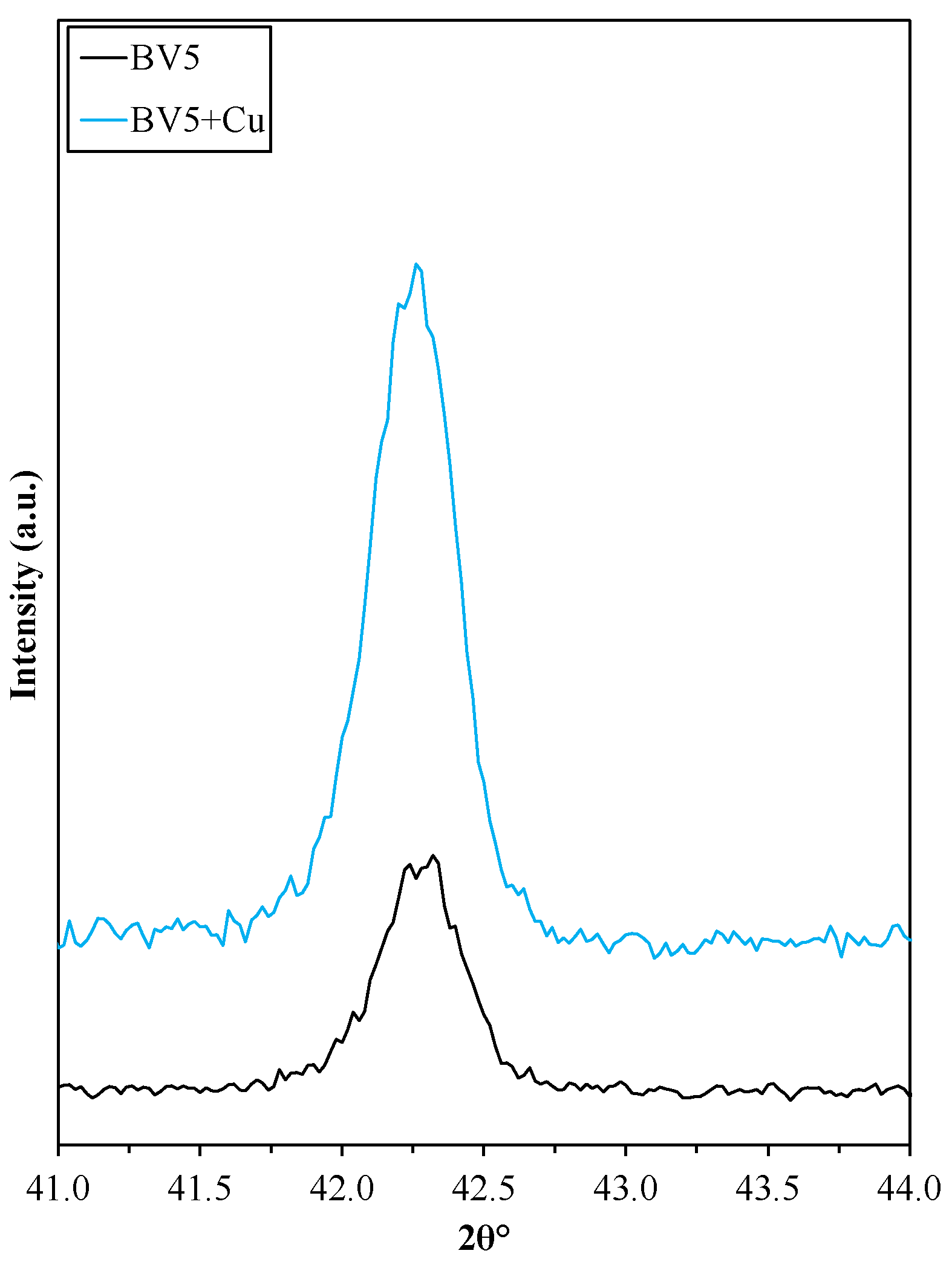
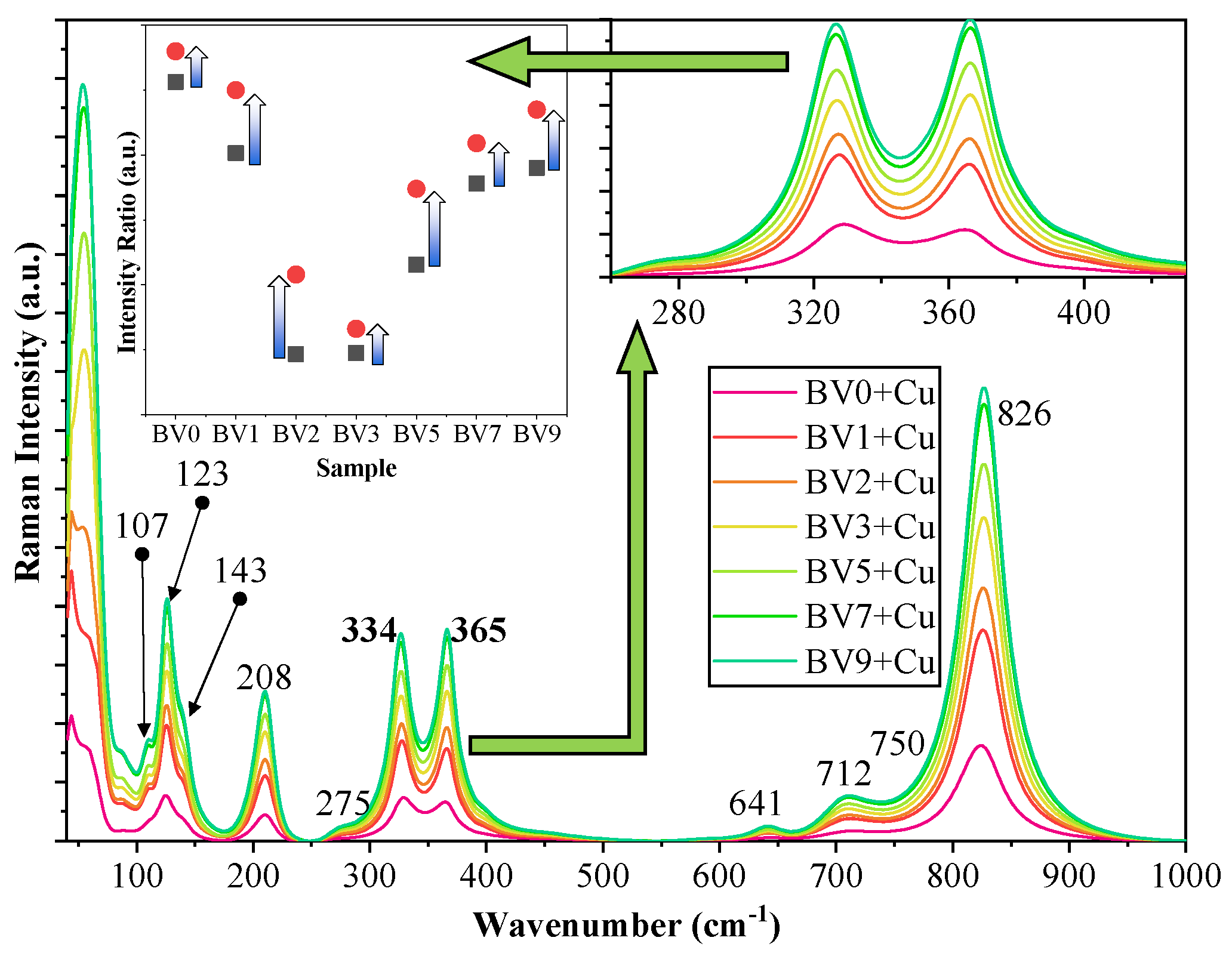
2.5. Deposition of Cu on the Surface of the Investigated Photocatalysts
- The main reaction is the reduction of Cu2+ to Cu.
- The two electrons used in the reduction process must originate from an oxidation reaction. In Bi-based photocatalysts, it is a known issue that Bi3+ can be oxidized to Bi5+ if a suitable reaction partner is present, which can be the above-mentioned reduction reaction.
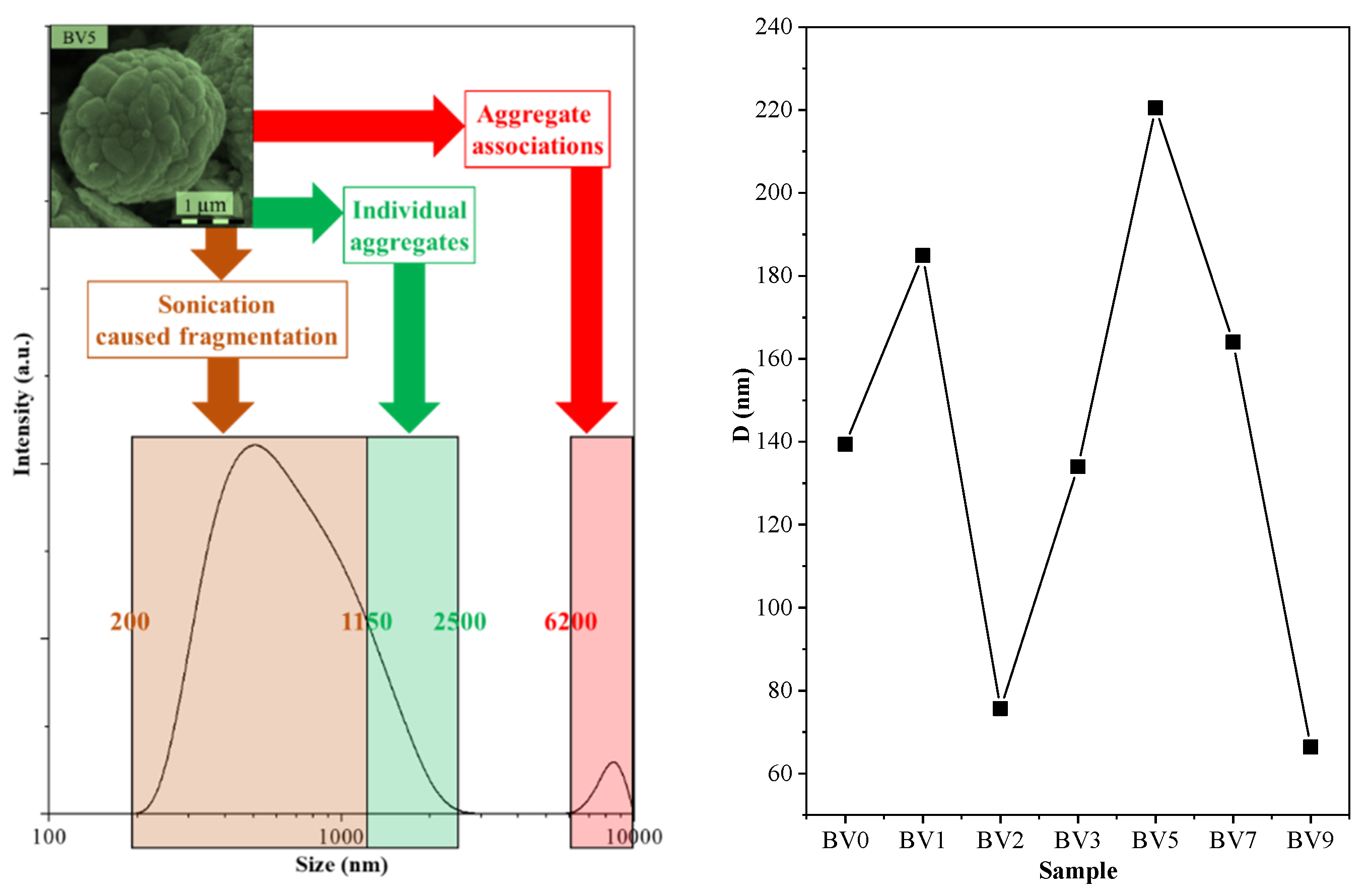
2.6. DLS Measurements
3. Materials and Methods
3.1. Materials
3.2. Preparation of Differently Shaped BiVO4 Particles
3.3. The Selective Deposition of Cu2+ on BiVO4
3.4. Methods and Instrumentation
3.5. Photocatalytic Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Andreozzi, R. Advanced oxidation processes (AOP) for water purification and recovery. Catal. Today 1999, 53, 51–59. [Google Scholar] [CrossRef]
- Chong, M.N.; Jin, B.; Chow, C.W.; Saint, C. Recent developments in photocatalytic water treatment technology: A review. Water Res. 2010, 44, 2997–3027. [Google Scholar] [CrossRef] [PubMed]
- Robinson, T.; McMullan, G.; Marchant, R.; Nigam, P. Remediation of dyes in textile effluent: A critical review on current treatment technologies with a proposed alternative. Bioresour. Technol. 2001, 77, 247–255. [Google Scholar] [CrossRef]
- Hachem, C.; Bocquillon, F.; Zahraa, O.; Bouchy, M. Decolourization of textile industry wastewater by the photocatalytic degradation process. Dye Pigment. 2001, 49, 117–125. [Google Scholar] [CrossRef]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 Photocatalysis: Mechanisms and Materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef]
- Wang, C.-C.; Li, J.-R.; Lv, X.-L.; Zhang, Y.-Q.; Guo, G. Photocatalytic organic pollutants degradation in metal-organic frameworks. Energy Environ. Sci. 2014, 7, 2831–2867. [Google Scholar] [CrossRef]
- Da Silva, J.C.C.; Teodoro, J.A.R.; Afonso, R.J.D.C.F.; Aquino, S.F.; Augusti, R. Photolysis and photocatalysis of ibuprofen in aqueous medium: Characterization of by-products via liquid chromatography coupled to high-resolution mass spectrometry and assessment of their toxicities againstArtemia Salina. J. Mass Spectrom. 2014, 49, 145–153. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, J.; Li, D.; Guo, W.; Qin, Q.; Zhu, L.; Zheng, W. Template-free facile preparation of monoclinic WO3 nanoplates and their high photocatalytic activities. Appl. Surf. Sci. 2014, 305, 274–280. [Google Scholar] [CrossRef]
- Liu, Y.; Yuan, X.; Wang, H. Novel visible light-induced g-C3N4-Sb2S3/Sb4O5Cl2 composite photocatalysts for efficient degradation of methyl orange. Catal. Commun. 2015, 70, 17–20. [Google Scholar] [CrossRef]
- Shao, J.; Sheng, W.; Wang, M.; Li, S.; Chen, J.; Zhang, Y.; Cao, S. In situ synthesis of carbon-doped TiO2 single-crystal nanorods with a remarkably photocatalytic efficiency. Appl. Catal. B Environ. 2017, 209, 311–319. [Google Scholar] [CrossRef]
- Bojinova, A.; Kaneva, N.; Papazova, K. Green synthesis of UV and visible light active TiO2/WO3 powders and films for malachite green and ethylene photodegradation. React. Kinet. Mech. Catal. 2017, 120, 821–832. [Google Scholar] [CrossRef]
- Mao, Z.; Xie, R.; Fu, D.; Zhang, L.; Xu, H.; Zhong, Y.; Sui, X. PAN supported Ag-AgBr@Bi20TiO32 electrospun fiber mats with efficient visible light photocatalytic activity and antibacterial capability. Sep. Purif. Technol. 2017, 176, 277–286. [Google Scholar] [CrossRef]
- Ismail, A.A.; Bahnemann, D.W. Photochemical splitting of water for hydrogen production by photocatalysis: A review. Sol. Energy Mater. Sol. Cells 2014, 128, 85–101. [Google Scholar] [CrossRef]
- Bai, J.; Zhou, B. Titanium Dioxide Nanomaterials for Sensor Applications. Chem. Rev. 2014, 114, 10131–10176. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Zheng, G.; Li, W.; Seh, Z.W.; Yao, H.; Yan, K.; Kong, D.; Cui, Y. Sulfur Cathodes with Hydrogen Reduced Titanium Dioxide Inverse Opal Structure. ACS Nano 2014, 8, 5249–5256. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Mora-Seró, I.; De Angelis, F.; Bisquert, J.; Wang, P. Titanium Dioxide Nanomaterials for Photovoltaic Applications. Chem. Rev. 2014, 114, 10095–10130. [Google Scholar] [CrossRef]
- Reddy, K.R.; Hassan, M.; Gomes, V.G. Hybrid nanostructures based on titanium dioxide for enhanced photocatalysis. Appl. Catal. A Gen. 2015, 489, 1–16. [Google Scholar] [CrossRef]
- Khan, M.M.; Ansari, S.A.; Pradhan, D.K.; Ansari, M.O.; Han, D.H.; Lee, J.; Cho, M.H. Band gap engineered TiO2 nanoparticles for visible light induced photoelectrochemical and photocatalytic studies. J. Mater. Chem. A 2014, 2, 637–644. [Google Scholar] [CrossRef]
- Li, D.; Haneda, H.; Labhsetwar, N.K.; Hishita, S.; Ohashi, N. Visible-light-driven photocatalysis on fluorine-doped TiO2 powders by the creation of surface oxygen vacancies. Chem. Phys. Lett. 2005, 401, 579–584. [Google Scholar] [CrossRef]
- Cho, Y.; Choi, W.; Lee, C.-H.; Hyeon, T.; Lee, H.-I. Visible Light-Induced Degradation of Carbon Tetrachloride on Dye-Sensitized TiO2. Environ. Sci. Technol. 2001, 35, 966–970. [Google Scholar] [CrossRef]
- Dawson, A.; Kamat, P.V. Semiconductor—metal nanocomposites. Photoinduced fusion and photocatalysis of gold-capped TiO2 (TiO2/gold) nanoparticles. J. Phys. Chem. B 2001, 105, 960–966. [Google Scholar]
- Chen, C.; Cai, W.; Long, M.; Zhou, B.; Wu, Y.; Wu, D.; Feng, Y. Synthesis of Visible-Light Responsive Graphene Oxide/TiO2 Composites with p/n Heterojunction. ACS Nano 2010, 4, 6425–6432. [Google Scholar] [CrossRef]
- Shang, M.; Wang, W.; Sun, S.; Zhou, L.; Zhang, L. Bi2WO6 Nanocrystals with High Photocatalytic Activities under Visible Light. J. Phys. Chem. C 2008, 112, 10407–10411. [Google Scholar] [CrossRef]
- Ma̧czka, M.; Macalik, L.; Hermanowicz, K.; Kȩpiński, L.; Tomaszewski, P.E. Phonon properties of nanosized bismuth layered ferroelectric material-Bi2WO6. J. Raman Spectrosc. 2009, 41, 1059–1066. [Google Scholar] [CrossRef]
- Tokunaga, S.; Kato, H.; Kudo, A. Selective Preparation of Monoclinic and Tetragonal BiVO4 with Scheelite Structure and Their Photocatalytic Properties. Chem. Mater. 2001, 13, 4624–4628. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Mallick, K.; Hartridge, A. Phase transition in BiVO4. Mater. Lett. 1997, 30, 7–13. [Google Scholar] [CrossRef]
- Zhou, L.; Wang, W.; Liu, S.; Zhang, L.; Xu, H.; Zhu, W. A sonochemical route to visible-light-driven high-activity BiVO4 photocatalyst. J. Mol. Catal. A Chem. 2006, 252, 120–124. [Google Scholar] [CrossRef]
- Jia, T.; Fu, F.; Long, F. Synthesis, characterization and enhanced visible-light photocatalytic activity of Zn2SnO4/C nanocomposites with truncated octahedron morphology. Ceram. Int. 2016, 42, 13893–13899. [Google Scholar] [CrossRef]
- Yu, J.; Kudo, A. Effects of Structural Variation on the Photocatalytic Performance of Hydrothermally Synthesized BiVO4. Adv. Funct. Mater. 2006, 16, 2163–2169. [Google Scholar] [CrossRef]
- Wang, M.; Che, Y.; Niu, C.; Dang, M.; Dong, D. Effective visible light-active boron and europium co-doped BiVO4 synthesized by sol-gel method for photodegradion of methyl orange. J. Hazard. Mater. 2013, 262, 447–455. [Google Scholar] [CrossRef]
- Dunkle, S.S.; Helmich, R.J.; Suslick, K.S. BiVO4 as a Visible-Light Photocatalyst Prepared by Ultrasonic Spray Pyrolysis. J. Phys. Chem. C 2009, 113, 11980–11983. [Google Scholar] [CrossRef]
- Tan, H.L.; Amal, R.; Ng, Y.H. Exploring the Different Roles of Particle Size in Photoelectrochemical and Photocatalytic Water Oxidation on BiVO4. ACS Appl. Mater. Interfaces 2016, 8, 28607–28614. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Liu, W.; Dong, M.; Li, W.; Chang, L. Synthesis of monoclinic sheet-like BiVO4 with preferentially exposed (040) facets as a new yellow-green pigment. Dye. Pigment. 2016, 134, 91–98. [Google Scholar] [CrossRef]
- Shang, M.; Wang, W.; Ren, J.; Sun, S.; Zhang, L. A novel BiVO4 hierarchical nanostructure: Controllable synthesis, growth mechanism, and application in photocatalysis. CrystEngComm 2010, 12, 1754–1758. [Google Scholar] [CrossRef]
- Han, M.; Chen, X.; Sun, T.; Tan, O.K.; Tse, M.S. Synthesis of mono-dispersed m-BiVO4 octahedral nano-crystals with enhanced visible light photocatalytic properties. CrystEngComm 2011, 13, 6674. [Google Scholar] [CrossRef]
- Kim, T.-H.; Jo, Y.-H.; Matsuoka, M.; Cho, S.-H.; Kim, H.-S.; Lee, S.W. Effect of pH on the Microwave Hydrothermal Synthesis of BiVO4 Photocatalysts and Their Photocatalytic Activity Under Simulated Solar Light. J. Nanosci. Nanotechnol. 2016, 16, 11148–11152. [Google Scholar] [CrossRef]
- Li, W.; Wang, X.; Wang, Z.; Meng, Y.; Sun, X.; Yan, T.; You, J.; Kong, D. Relationship between crystalline phases and photocatalytic activities of BiVO4. Mater. Res. Bull. 2016, 83, 259–267. [Google Scholar] [CrossRef]
- Suarez, C.M.; Hernández, S.; Russo, N. BiVO4 as photocatalyst for solar fuels production through water splitting: A short review. Appl. Catal. A Gen. 2015, 504, 158–170. [Google Scholar] [CrossRef]
- Zhu, J.; Fan, F.; Chen, R.; An, H.; Feng, Z.; Li, C. Direct Imaging of Highly Anisotropic Photogenerated Charge Separations on Different Facets of a Single BiVO4 Photocatalyst. Angew. Chem. Int. Ed. 2015, 54, 9111–9114. [Google Scholar] [CrossRef]
- Xia, J.; Liu, L.; Jin, S.; Xia, Z.; Tang, M. Preparation and growth mechanism of α-Bi2O3 crystal whiskers. Procedia Eng. 2012, 27, 1441–1447. [Google Scholar] [CrossRef]
- Pap, Z.; Mogyorosi, K.; Veréb, G.; Dombi, A.; Hernadi, K.; Danciu, V.; Baia, L.; Pap, Z. Commercial and home-made nitrogen modified titanias. A short reflection about the advantageous/disadvantageous properties of nitrogen doping in the frame of their applicability. J. Mol. Struct. 2014, 1073, 157–163. [Google Scholar] [CrossRef]
- Pan, J.; Liu, G.; Lu, G.Q. On the true photoreactivity order of {001}, {010}, and {101} facets of anatase TiO2 crystals. Angew. Chem. Int. Ed. 2011, 50, 2133–2137. [Google Scholar] [CrossRef] [PubMed]
- Veréb, G.; Ambrus, Z.; Pap, Z.; Kmetykó, Á.; Dombi, A.; Danciu, V.; Cheesman, A.; Mogyorosi, K.; Pap, Z. Comparative study on UV and visible light sensitive bare and doped titanium dioxide photocatalysts for the decomposition of environmental pollutants in water. Appl. Catal. A Gen. 2012, 417, 26–36. [Google Scholar] [CrossRef]
- Kása, Z.; Saszet, K.; Dombi, A.; Hernadi, K.; Baia, L.; Magyari, K.; Pap, Z. Thiourea and Triton X-100 as shape manipulating tools or more for Bi2WO6 photocatalysts? Mater. Sci. Semicond. Process. 2018, 74, 21–30. [Google Scholar] [CrossRef]
- Vajda, K.; Saszet, K.; Kedves, E.Z.; Kása, Z.; Danciu, V.; Baia, L.; Magyari, K.; Hernádi, K.; Kovács, G.; Pap, Z. Shape-controlled agglomeration of TiO2 nanoparticles. New insights on polycrystallinity vs. single crystals in photocatalysis. Ceram. Int. 2016, 42, 3077–3087. [Google Scholar]
- Nishikawa, M.; Hiura, S.; Mitani, Y.; Nosaka, Y. Enhanced photocatalytic activity of BiVO4 by co-grafting of metal ions and combining with CuBi2O4. J. Photochem. Photobiol. A Chem. 2013, 262, 52–56. [Google Scholar] [CrossRef]
- Holzwarth, U.; Gibson, N. The Scherrer equation versus the ‘Debye-Scherrer equation’. Nat. Nanotechnol. 2011, 6, 534. [Google Scholar] [CrossRef]
- Gillespie, J.B.; Lindberg, J.D.; Laude, L.S. Kubelka-Munk Optical Coefficients for a Barium Sulfate White Reflectance Standard. Appl. Opt. 1975, 14, 807–809. [Google Scholar] [CrossRef]
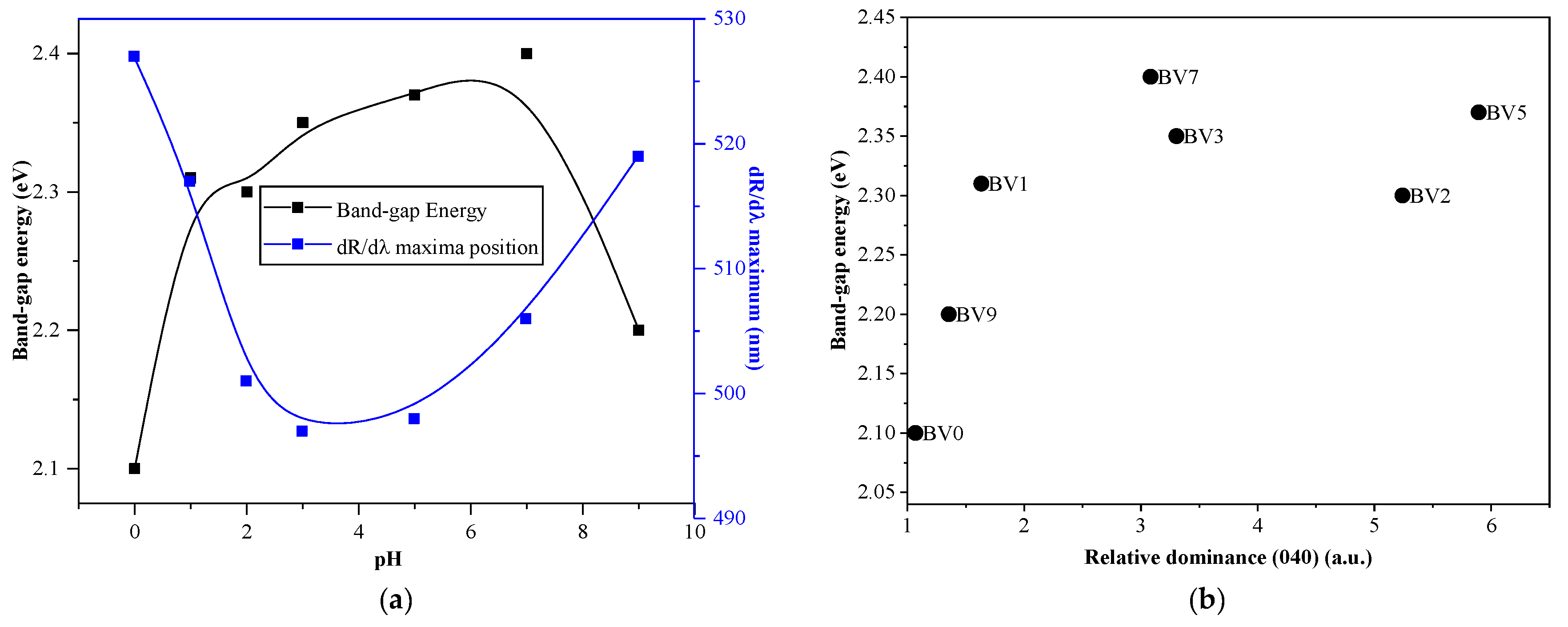

| Sample Name | Crystal Size (nm) | Band Gap (ev) | dR/dλmax (nm) | Degraded RhB (%) | Degraded Oxalic Acid (%) |
|---|---|---|---|---|---|
| BV0 | 42.8 | 2.10 | 527 | - | - |
| BV1 | 37.4 | 2.31 | 517 | 21.21 | 38.16 |
| BV2 | 36.3 | 2.30 | 501 | 43.20 | 51.13 |
| BV3 | 36.9 | 2.35 | 497 | 23.27 | 47.86 |
| BV5 | 29.8 | 2.37 | 498 | 61.34 | 37.58 |
| BV7 | 33.0 | 2.40 | 506 | 51.11 | 38.62 |
| BV9 | 43.4 | 2.20 | 519 | 21.49 | 41.82 |
Sample Availability: Samples of the compounds are available from the authors. | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kása, Z.; Almási, E.E.; Hernádi, K.; Gyulavári, T.; Baia, L.; Veréb, G.; László, Z.; Pap, Z. New Insights into The Photoactivity of Shape-Tailored BiVO4 Semiconductors via Photocatalytic Degradation Reactions and Classical Reduction Processes. Molecules 2020, 25, 4842. https://doi.org/10.3390/molecules25204842
Kása Z, Almási EE, Hernádi K, Gyulavári T, Baia L, Veréb G, László Z, Pap Z. New Insights into The Photoactivity of Shape-Tailored BiVO4 Semiconductors via Photocatalytic Degradation Reactions and Classical Reduction Processes. Molecules. 2020; 25(20):4842. https://doi.org/10.3390/molecules25204842
Chicago/Turabian StyleKása, Zsolt, Enikő Eszter Almási, Klára Hernádi, Tamás Gyulavári, Lucian Baia, Gábor Veréb, Zsuzsanna László, and Zsolt Pap. 2020. "New Insights into The Photoactivity of Shape-Tailored BiVO4 Semiconductors via Photocatalytic Degradation Reactions and Classical Reduction Processes" Molecules 25, no. 20: 4842. https://doi.org/10.3390/molecules25204842
APA StyleKása, Z., Almási, E. E., Hernádi, K., Gyulavári, T., Baia, L., Veréb, G., László, Z., & Pap, Z. (2020). New Insights into The Photoactivity of Shape-Tailored BiVO4 Semiconductors via Photocatalytic Degradation Reactions and Classical Reduction Processes. Molecules, 25(20), 4842. https://doi.org/10.3390/molecules25204842










