Effect of Enzymatic, Ultrasound, and Reflux Extraction Pretreatments on the Yield and Chemical Composition of Essential Oils
Abstract
1. Introduction
2. Results
2.1. Cellulase, Xylanase, and Pectinase Can Degrade Their Respective Substrates under the Reaction Conditions
2.2. Optimization of Ultrasound Extraction Conditions
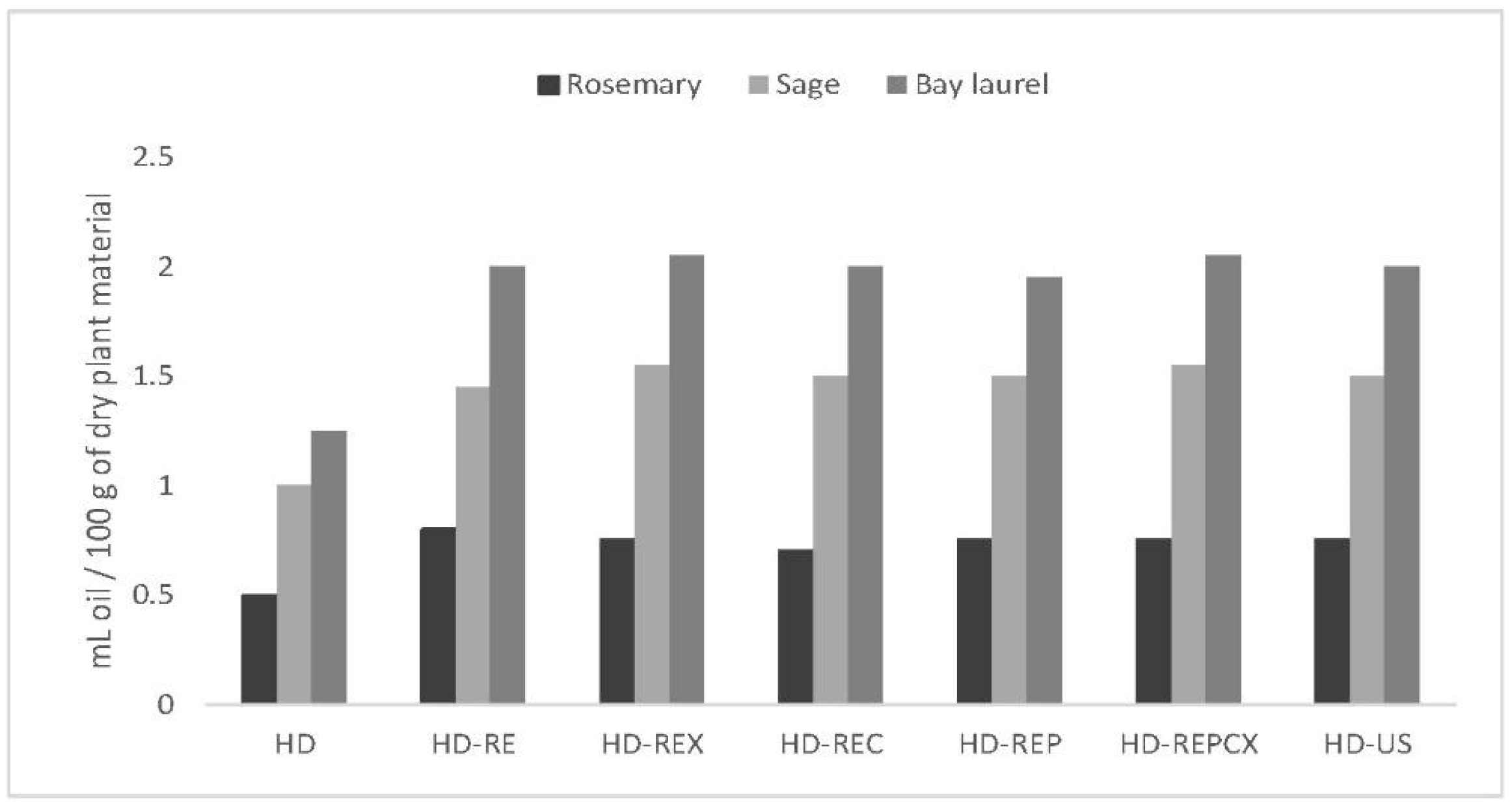
2.3. Different Pretreatments Increased the Essential Oil Yield
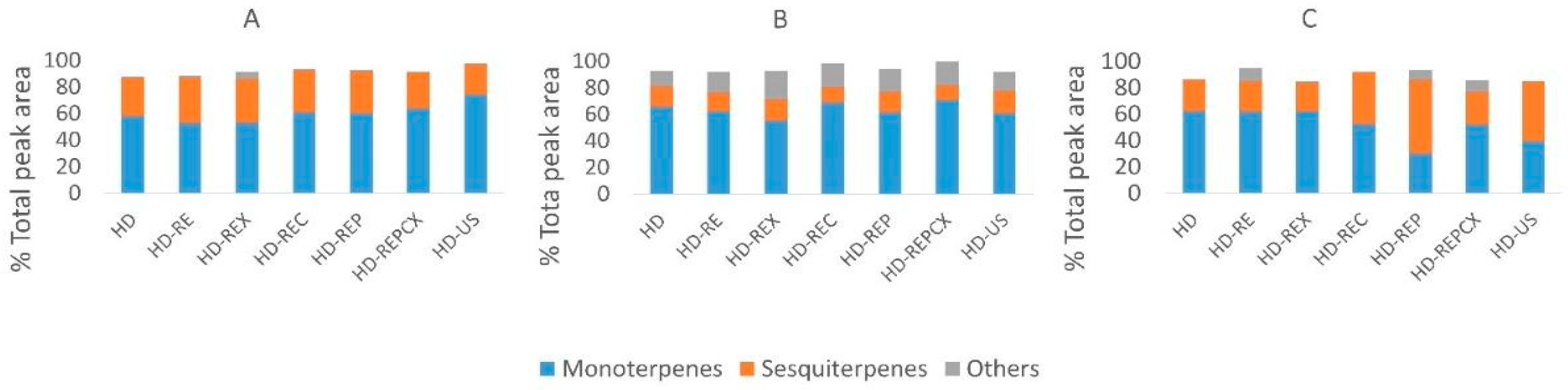
2.4. Overall Chemical Composition of Essential Oils Was Not Significantly Affected by Different Hydrodistillation Pretreatments
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Chemicals
4.3. 3,5-Dinitrosalicylic Acid (DNSA) Assay for the Determination of Enzyme Activity
4.4. Determination of Total Phenols for Optimization of Ultrasonic Parameters
4.5. Extraction Procedures
4.6. Gas Chromatograph/Mass Spectrometer (GC-MS) Analysis
4.7. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dimaki, V.D.; Iatrou, G.; Lamari, F.N. Effect of acidic and enzymatic pretreatment on the analysis of mountain tea (Sideritis spp.) volatiles via distillation and ultrasound-assisted extraction. J. Chromatogr. A 2017, 1524, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Hosni, K.; Hassen, I.; Chaâbane, H.; Jemli, M.; Dallali, S.; Sebei, H.; Casabianca, H. Enzyme-assisted extraction of essential oils from thyme (Thymus capitatus L.) and rosemary (Rosmarinus officinalis L.): Impact on yield, chemical composition and antimicrobial activity. Ind. Crop. Prod. 2013, 47, 291–299. [Google Scholar] [CrossRef]
- Olmedo, R.H.; Asensio, C.M.; Grosso, N.R. Thermal stability and antioxidant activity of essential oils from aromatic plants farmed in Argentina. Ind. Crop. Prod. 2015, 69, 21–28. [Google Scholar] [CrossRef]
- Amorati, R.; Foti, M.C.; Valgimigli, L. Antioxidant activity of essential oils. J. Agric. Food Chem. 2013, 61, 10835–10847. [Google Scholar] [CrossRef]
- Lesgards, J.F.; Baldovini, N.; Vidal, N.; Pietri, S. Anticancer activities of essential oils constituents and synergy with conventional therapies: A review. Phyther. Res. 2014, 28, 1423–1446. [Google Scholar] [CrossRef]
- Martins, I.M.; Barreiro, M.F.; Coelho, M.; Rodrigues, A.E. Microencapsulation of essential oils with biodegradable polymeric carriers for cosmetic applications. Chem. Eng. J. 2014. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; Carvalho-Costa, D.; Cavaleiro, C.; Costa, H.S.; Albuquerque, T.G.; Castilho, M.C.; Ramos, F.; Melo, N.R.; Sanches-Silva, A. A novel insight on an ancient aromatic plant: The rosemary (Rosmarinus officinalis L.). Trends Food Sci. Technol. 2015. [Google Scholar] [CrossRef]
- Trongtokit, Y.; Rongsriyam, Y.; Komalamisra, N.; Apiwathnasorn, C. Comparative repellency of 38 essential oils against mosquito bites. Phyther. Res. 2005, 19, 303–309. [Google Scholar] [CrossRef]
- Périno-Issartier, S.; Ginies, C.; Cravotto, G.; Chemat, F. A comparison of essential oils obtained from lavandin via different extraction processes: Ultrasound, microwave, turbohydrodistillation, steam and hydrodistillation. J. Chromatogr. A 2013, 1305, 41–47. [Google Scholar] [CrossRef]
- Toma, M.; Vinatoru, M.; Paniwnyk, L.; Mason, T.J. Investigation of the effects of ultrasound on vegetal tissues during solvent extraction. Ultrason. Sonochem. 2001, 8, 137–142. [Google Scholar] [CrossRef]
- Assami, K.; Pingret, D.; Chemat, S.; Meklati, B.Y.; Chemat, F. Ultrasound induced intensification and selective extraction of essential oil from Carum carvi L. seeds. Chem. Eng. Process. Process Intensif. 2012, 62, 99–105. [Google Scholar] [CrossRef]
- Lilia, C.; Abdelkader, A.; Karima, A.K.A.; Tarek, B. The Effect of Ultrasound Pre-treatment on the Yield, Chemical Composition and Antioxidant Activity of Essential Oil from Wild Lavandula stoechas L. J. Essent. Oil-Bear. Plants 2018, 21, 253–263. [Google Scholar] [CrossRef]
- Morsy, N.F.S. A short extraction time of high quality hydrodistilled cardamom (Elettaria cardamomum L. Maton) essential oil using ultrasound as a pretreatment. Ind. Crop. Prod. 2015, 62, 287–292. [Google Scholar] [CrossRef]
- Seidi Damyeh, M.; Niakousari, M.; Saharkhiz, M.J. Ultrasound pretreatment impact on Prangos ferulacea Lindl. and Satureja macrosiphonia Bornm. essential oil extraction and comparing their physicochemical and biological properties. Ind. Crop. Prod. 2016, 10, 22–28. [Google Scholar] [CrossRef]
- Kowalski, R.; Wawrzykowski, J. Effect of ultrasound-assisted maceration on the quality of oil from the leaves of thyme Thymus vulgaris L. Flavour Fragr. J. 2009, 24, 214–220. [Google Scholar] [CrossRef]
- Kowalski, R.; Kowalska, G.; Jamroz, J.; Nawrocka, A.; Metyk, D. Effect of the ultrasound-assisted preliminary maceration on the efficiency of the essential oil distillation from selected herbal raw materials. Ultrason. Sonochem. 2015, 24, 214–220. [Google Scholar] [CrossRef]
- Smigielski, K.B.; Majewska, M.; Kunicka-Styczyñska, A.; Gruska, R. The Effect of Ultrasound-Assisted Maceration on the Bioactivity, Chemical Composition and Yield of Essential Oil from Waste Carrot Seeds (Daucus carota). J. Essent. Oil-Bear. Plants 2014, 17, 1075–1086. [Google Scholar] [CrossRef]
- Balasubramaniam, V.G.; Ayyappan, P.; Sathvika, S.; Antony, U. Effect of enzyme pretreatment in the ultrasound assisted extraction of finger millet polyphenols. J. Food Sci. Technol. 2019, 74, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Boulila, A.; Hassen, I.; Haouari, L.; Mejri, F.; Amor, I.B.; Casabianca, H.; Hosni, K. Enzyme-assisted extraction of bioactive compounds from bay leaves (Laurus nobilis L.). Ind. Crop. Prod. 2015, 74, 485–493. [Google Scholar] [CrossRef]
- Puri, M.; Sharma, D.; Barrow, C.J. Enzyme-assisted extraction of bioactives from plants. Trends Biotechnol. 2012, 30, 37–44. [Google Scholar] [CrossRef]
- Sowbhagya, H.B.; Srinivas, P.; Purnima, K.T.; Krishnamurthy, N. Enzyme-assisted extraction of volatiles from cumin (Cuminum cyminum L.) seeds. Food Chem. 2011, 127, 1856–1861. [Google Scholar] [CrossRef]
- Sowbhagya, H.B.; Srinivas, P.; Krishnamurthy, N. Effect of enzymes on extraction of volatiles from celery seeds. Food Chem. 2010, 120, 230–234. [Google Scholar] [CrossRef]
- Baby, K.C.; Ranganathan, T.V. Effect of enzyme pre-treatment on extraction yield and quality of cardamom (Elettaria cardamomum maton.) volatile oil. Ind. Crop. Prod. 2016, 89, 200–206. [Google Scholar] [CrossRef]
- Chandran, J.; Amma, K.P.P.; Menon, N.; Purushothaman, J.; Nisha, P. Effect of enzyme assisted extraction on quality and yield of volatile oil from black pepper and cardamom. Food Sci. Biotechnol. 2012, 21, 1611–1617. [Google Scholar] [CrossRef]
- Chávez-González, M.L.; López-López, L.I.; Rodríguez-Herrera, R.; Contreras-Esquivel, J.C.; Aguilar, C.N. Enzyme-assisted extraction of citrus essential oil. Chem. Pap. 2016, 70, 412–417. [Google Scholar] [CrossRef]
- Śmigielski, K.B.; Majewska, M.; Kunicka-Styczyńska, A.; Szczesna-Antczak, M.; Gruska, R.; Stańczyk, Ł. The effect of enzyme-assisted maceration on bioactivity, quality and yield of essential oil from waste carrot (Daucus carota) seeds. J. Food Qual. 2014, 37, 219–228. [Google Scholar] [CrossRef]
- Hamrouni-Sellami, I.; Rahali, F.Z.; Rebey, I.B.; Bourgou, S.; Limam, F.; Marzouk, B. Total Phenolics, Flavonoids, and Antioxidant Activity of Sage (Salvia officinalis L.) Plants as Affected by Different Drying Methods. Food Bioprocess Technol. 2013, 59, 257–268. [Google Scholar] [CrossRef]
- Ali, A.; Chua, B.L.; Chow, Y.H. An insight into the extraction and fractionation technologies of the essential oils and bioactive compounds in Rosmarinus officinalis L.: Past, present and future. TrAC Trends Anal. Chem. 2019, 118, 338–351. [Google Scholar] [CrossRef]
- Ali, M.B.; Mnafgui, K.; Feki, A.; Damak, M.; Allouche, N. In vitro Antidiabetic, anti-obesity and antioxidant proprities of Rosemary extracts. J. Adv. Chem. 2014, 10, 2305–2316. [Google Scholar] [CrossRef]
- Fidan, H.; Stefanova, G.; Kostova, I.; Stankov, S.; Damyanova, S.; Stoyanova, A.; Zheljazkov, V.D. Chemical Composition and Antimicrobial Activity of Laurus nobilis L. Essential oils from Bulgaria. Molecules 2019, 24, 804. [Google Scholar] [CrossRef]
- Soares, I.H.; Loreto, S.; Rossato, L.; Mario, D.N.; Venturini, T.P.; Baldissera, F.; Santurio, J.M.; Alves, S.H. In vitro activity of essential oils extracted from condiments against fluconazole-resistant and -sensitive Candida glabrata. J. Mycol. Med. 2015, 54, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Fernández, N.J.; Damiani, N.; Podaza, E.A.; Martucci, J.F.; Fasce, D.; Quiroz, F.; Meretta, P.E.; Quintana, S.; Eguaras, M.J.; Gende, L.B. Laurus nobilis L. Extracts against Paenibacillus larvae: Antimicrobial activity, antioxidant capacity, hygienic behavior and colony strength. Saudi J. Biol. Sci. 2019. [Google Scholar] [CrossRef]
- Dammak, I.; Hamdi, Z.; Kammoun El Euch, S.; Zemni, H.; Mliki, A.; Hassouna, M.; Lasram, S. Evaluation of antifungal and anti-ochratoxigenic activities of Salvia officinalis, Lavandula dentata and Laurus nobilis essential oils and a major monoterpene constituent 1,8-cineole against Aspergillus carbonarius. Ind. Crop. Prod. 2019, 128, 85–93. [Google Scholar] [CrossRef]
- Ferreira, D.F.; Lucas, B.N.; Voss, M.; Santos, D.; Mello, P.A.; Wagner, R.; Cravotto, G.; Barin, J.S. Solvent-free simultaneous extraction of volatile and non-volatile antioxidants from rosemary (Rosmarinus officinalis L.) by microwave hydrodiffusion and gravity. Ind. Crop. Prod. 2020, 145, 112094. [Google Scholar] [CrossRef]
- Al Zuhairi, J.J.M.J.; Jookar Kashi, F.; Rahimi-Moghaddam, A.; Yazdani, M. Antioxidant, cytotoxic and antibacterial activity of Rosmarinus officinalis L. essential oil against bacteria isolated from urinary tract infection. Eur. J. Integr. Med. 2020, 38, 101192. [Google Scholar] [CrossRef]
- Vosoughi, N.; Gomarian, M.; Ghasemi Pirbalouti, A.; Khaghani, S.; Malekpoor, F. Essential oil composition and total phenolic, flavonoid contents, and antioxidant activity of sage (Salvia officinalis L.) extract under chitosan application and irrigation frequencies. Ind. Crop. Prod. 2018, 117, 366–374. [Google Scholar] [CrossRef]
- Khiva, Z.; Havani, M.; Garnar, A.; Kharchouf, S.; Amine, S.; Berrekhis, F.; Bouzoubae, A.; Zair, T.; Elhilali, F. Valorization of the Salvia officinalis L of the Morocco bioactive extracts: Phytochemistry, antioxidant and anticorrosive activities. J. King Saud Univ. Sci. 2019. [Google Scholar] [CrossRef]
- Hatipoglu, S.D.; Zorlu, N.; Dirmenci, T.; Goren, A.C.; Ozturk, T.; Topcu, G. Determination of volatile organic compounds in fourty five salvia species by thermal desorption-GC-MS technique. Rec. Nat. Prod. 2016, 10, 659–700. [Google Scholar]
- Russo, A.; Formisano, C.; Rigano, D.; Senatore, F.; Delfine, S.; Cardile, V.; Rosselli, S.; Bruno, M. Chemical composition and anticancer activity of essential oils of Mediterranean sage (Salvia officinalis L.) grown in different environmental conditions. Food Chem. Toxicol. 2013, 55, 42–47. [Google Scholar] [CrossRef]
- Awada, F.; Kobaissi, A.; Chokr, A.; Hamze, K.; Hayar, S.; Mortada, A. Factors affecting quantitative and qualitative variation of thyme (Origanum syriacum L.) essential oil in Lebanon. Adv. Environ. Biol. 2012, 6, 1509–1514. [Google Scholar]
- Nor Fazila, K.; Ku Halim, K.H. Effects of soaking on yield and quality of agarwood oil. J. Trop. For. Sci. 2012, 24, 557–564. [Google Scholar]
- Ghose, T.K. Measurement of cellulase activities. Pure Appl. Chem. 1987, 59, 257–268. [Google Scholar] [CrossRef]
- Miller, G.L. Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total Phenolics with Phosphomolybdic-Phosphotungstic Acid reagents. Am. J. Enol. Vitic. 1965, 16, 144. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Development Core Team: Vienna, Austria, 2017. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
| Time | Total Phenol Yield (mg/g) | |||
|---|---|---|---|---|
| 30% of Max. Ultrasonic Power | 60% of Max. Ultrasonic Power | 90% of Max. Ultrasonic Power | ||
| Bay laurel | 5 min | 16.87 ± 0.18 | 8.86 ± 0.05 | 7.32 ± 0.14 |
| 10 min | 17.37 ± 0.53 | 8.27 ± 0.21 | 5.99 ± 0.00 | |
| 15 min | 16.86 ± 0.18 | 8.30 ± 0.21 | 6.38 ± 0.15 | |
| Sage | 5 min | 29.46 ± 3.14 | 32.95 ± 2.6 | 32.20 ± 2.9 |
| 10 min | 35.71 ± 5.6 | 34.24 ± 7.2 | 36.83 ± 3.9 | |
| 15 min | 31.25 ± 5.4 | 31.76 ± 3.4 | 31.82 ± 0.32 | |
| Rosemary | 5 min | 93.98 ± 1.41 | 46.35 ± 1.06 | 50.16 ± 2.75 |
| 10 min | 97.44 ± 2.12 | 45.27 ± 0.07 | 51.22 ± 0.78 | |
| 15 min | 103.44 ± 2.12 | 43.08 ± 0.14 | 43.12 ± 0.14 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miljanović, A.; Bielen, A.; Grbin, D.; Marijanović, Z.; Andlar, M.; Rezić, T.; Roca, S.; Jerković, I.; Vikić-Topić, D.; Dent, M. Effect of Enzymatic, Ultrasound, and Reflux Extraction Pretreatments on the Yield and Chemical Composition of Essential Oils. Molecules 2020, 25, 4818. https://doi.org/10.3390/molecules25204818
Miljanović A, Bielen A, Grbin D, Marijanović Z, Andlar M, Rezić T, Roca S, Jerković I, Vikić-Topić D, Dent M. Effect of Enzymatic, Ultrasound, and Reflux Extraction Pretreatments on the Yield and Chemical Composition of Essential Oils. Molecules. 2020; 25(20):4818. https://doi.org/10.3390/molecules25204818
Chicago/Turabian StyleMiljanović, Anđela, Ana Bielen, Dorotea Grbin, Zvonimir Marijanović, Martina Andlar, Tonči Rezić, Sunčica Roca, Igor Jerković, Dražen Vikić-Topić, and Maja Dent. 2020. "Effect of Enzymatic, Ultrasound, and Reflux Extraction Pretreatments on the Yield and Chemical Composition of Essential Oils" Molecules 25, no. 20: 4818. https://doi.org/10.3390/molecules25204818
APA StyleMiljanović, A., Bielen, A., Grbin, D., Marijanović, Z., Andlar, M., Rezić, T., Roca, S., Jerković, I., Vikić-Topić, D., & Dent, M. (2020). Effect of Enzymatic, Ultrasound, and Reflux Extraction Pretreatments on the Yield and Chemical Composition of Essential Oils. Molecules, 25(20), 4818. https://doi.org/10.3390/molecules25204818










