Multifunctional Hydroxyapatite Coated with Arthemisia absinthium Composites
Abstract
1. Introduction
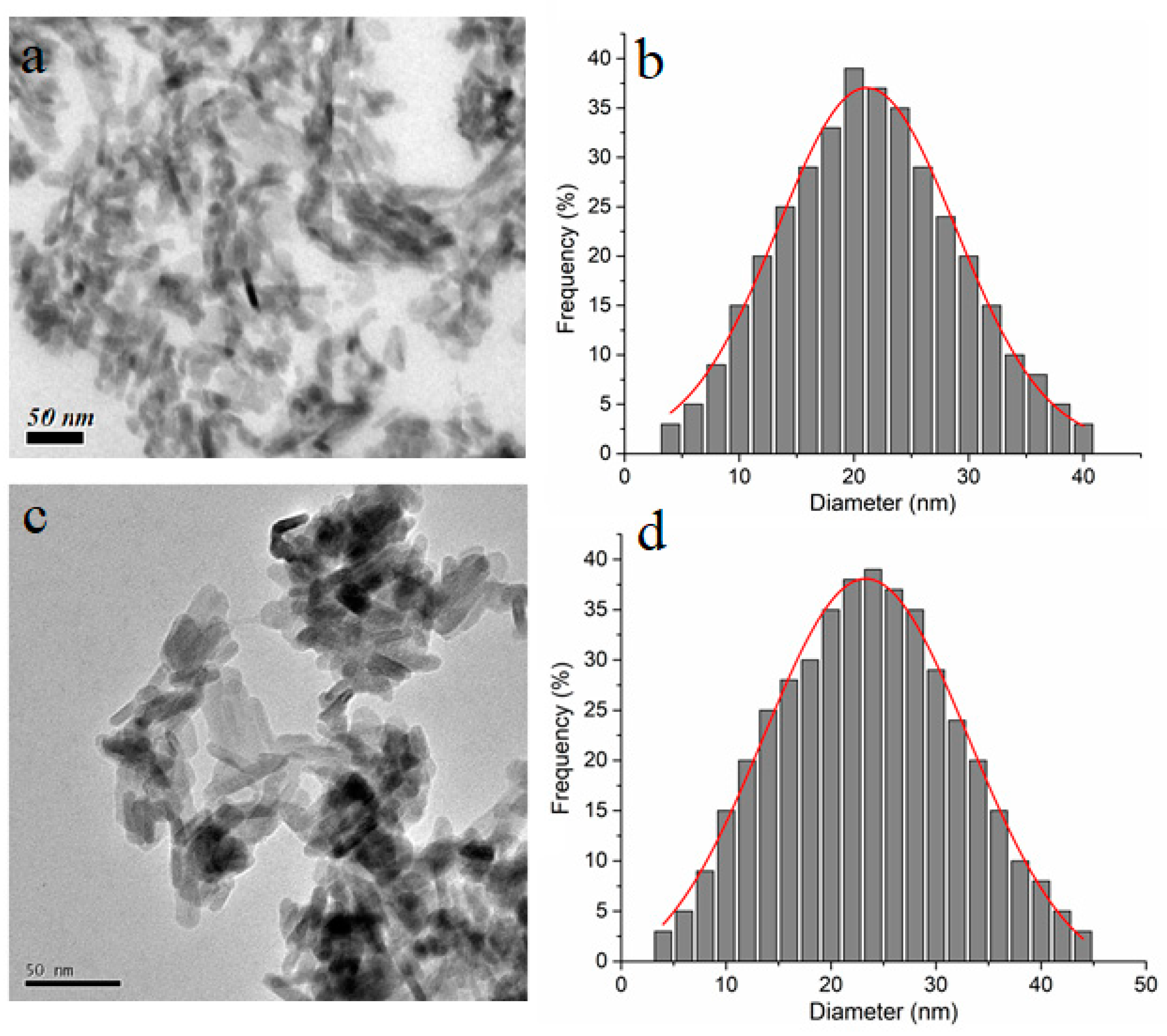
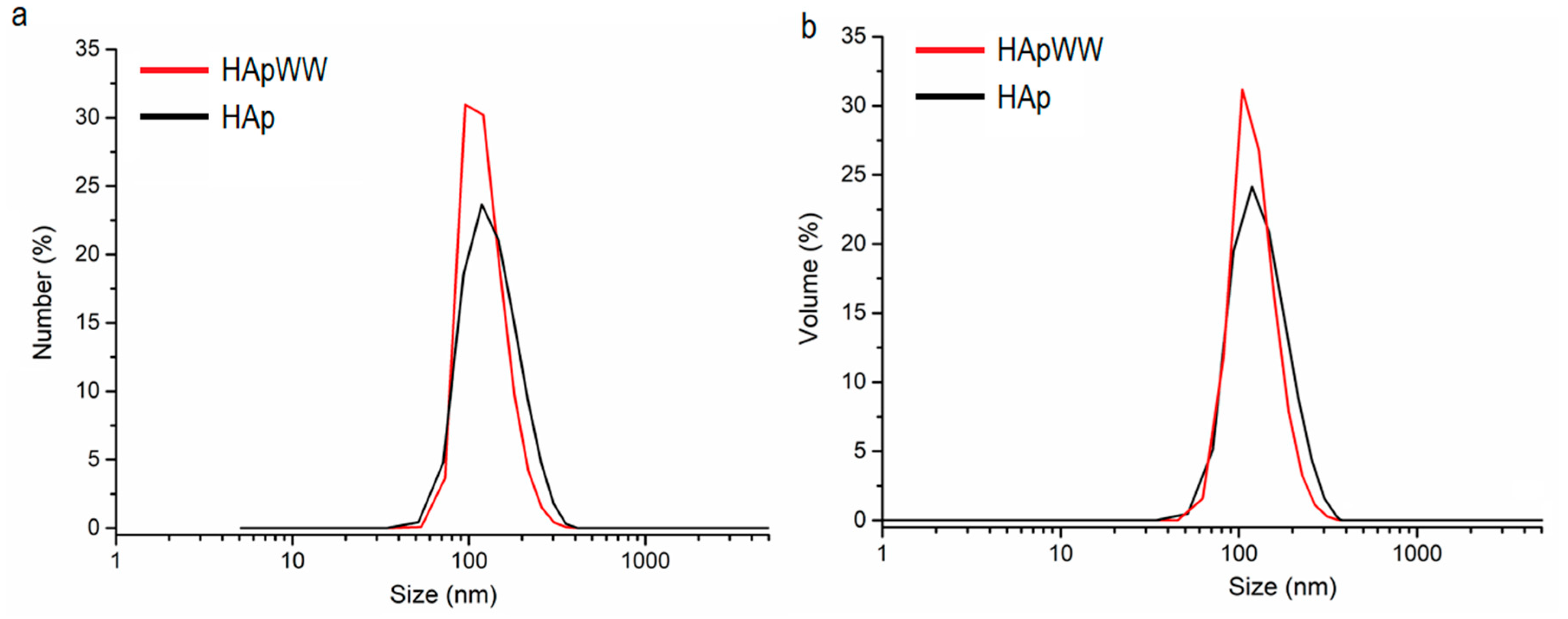
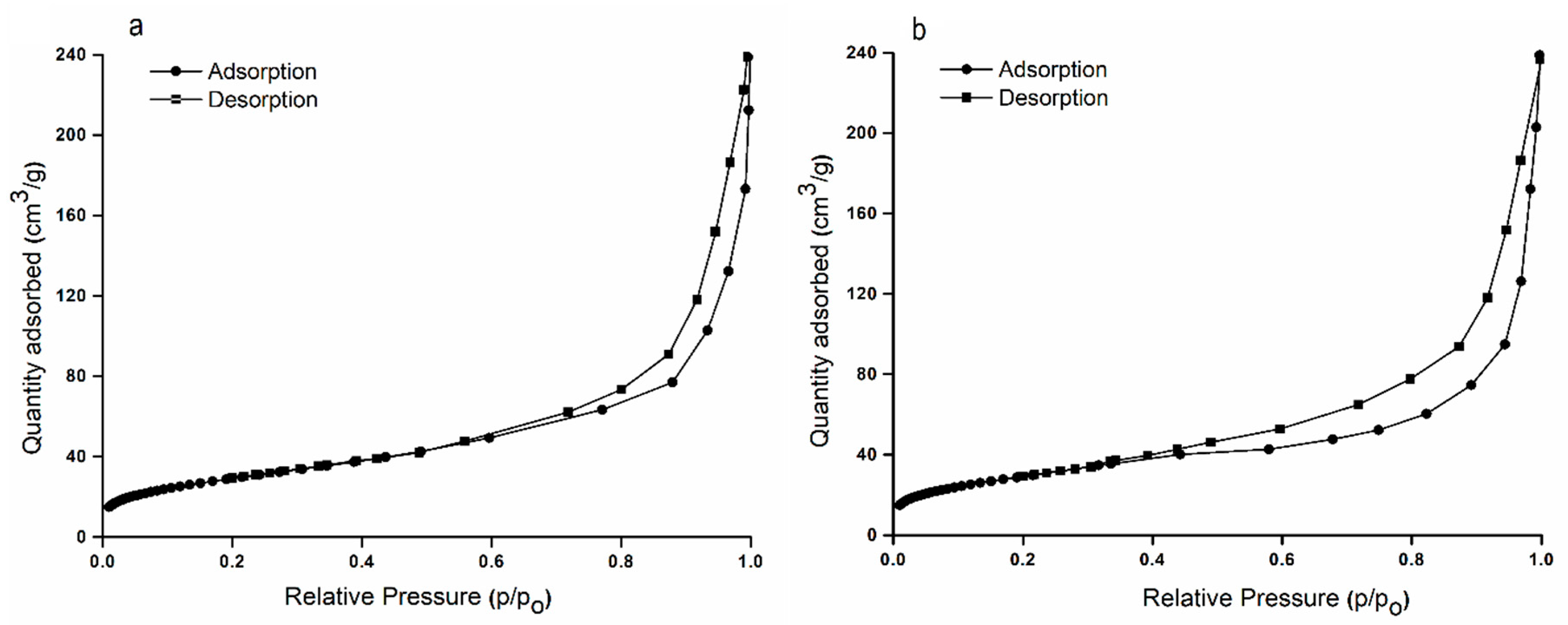
2. Results and Discussions
3. Materials and Methods
3.1. Sample Preparation
3.2. Physico-Chemical Characterization
3.3. Antimicrobial Assays
3.3.1. Microbial Strains and Culture
3.3.2. Agar Diffusion Method
3.3.3. Broth Micro-Dilution Assay
3.3.4. Flow Cytometric Testing
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Darouiche, R.O. Treatment of infections associated with surgical implants. N. Engl. J. Med. 2004, 350, 1422–1429. [Google Scholar] [CrossRef]
- Raho, G.B.; Abouni, B. Escherichia coli and Staphylococcus aureus most common source of infection. In The Battle against Microbial Pathogens: Basic Science, Technological Advances and Educational Programs; Méndez-Vilas, A., Ed.; Formatex Research Center: Badajoz, Spain, 2015. [Google Scholar]
- Tiemersma, E.W.; Bronzwaers, S.L.; Lyytikäinen, O.; Degener, J.E.; Schrijnemakers, P.; Bruinsma, N.; Monen, J.; Witte, W.; Grundman, H.; European Antimicrobial Resistance Surveillance System Participants. European antimicrobial resistance surveillance system participants: Methicillin-resistant Staphylococcus aureus in Europe. Emerg. Infect. Dis. 2004, 10, 1627–1633. [Google Scholar] [CrossRef]
- Walsh, S.E.; Maillard, J.Y.; Russel, A.D.; Catrenich, C.E.; Charbonneau, D.L.; Bartolo, R.J. Activity and mechanism of action of selected biocidal agents on Gram-positive and negative bacteria. J. Appl. Microbiol. 2003, 94, 240–247. [Google Scholar] [CrossRef]
- Altiok, D.; Altiok, E.; Tihminlioglu, F. Physical, antibacterial and antioxidant properties of chitosan films incorporated with thyme oil for potential wound healing applications. J. Mater. Sci. Mater. Med. 2010, 21, 2227–2236. [Google Scholar] [CrossRef]
- Bora, K.S.; Sharma, A. Phytochemical and pharmacological potential of Artemisia absinthium Linn. and Artemisia asiatica Nakai: A Review. J. Pharm. Res. 2010, 3, 325–328. [Google Scholar]
- Chouhan, S.; Sharma, K.; Guleria, S. Antimicrobial Activity of Some Essential Oils—Present Status and Future Perspectives. Medicines 2017, 4, 58. [Google Scholar] [CrossRef]
- Bora, K.S.; Sharma, A. The genus Artemisia: A comprehensive review. Pharm. Biol. 2011, 49, 101–109. [Google Scholar] [CrossRef]
- Msaada, K.; Salem, N.; Bachrouch, O.; Bousselmi, S.; Tammar, S.; Alfaify, A.; Al Sane, K.; Ben Ammar, W.; Azeiz, S.; Brahim, A.H.; et al. Chemical Composition and Antioxidant and Antimicrobial Activities of Wormwood (Artemisia absinthium L.) Essential Oils and Phenolics. J. Chem. 2015, 2015, 804658. [Google Scholar] [CrossRef]
- Juteau, F.; Jerkovic, I.; Masotti, V.; Milos, M.; Mastelic, J.; Bessière, J.M.; Viano, J. Composition and antimicrobial activity of the essential oil of Artemisia absinthiumfrom Croatia and France. Planta Med. 2003, 69, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Irshad, S.; Butt, M.; Hira, Y. In-vitro antibacterial activity of two medicinal plants neem (Azadirachta indica) and peppermint. Int. Res. J. Pharm. 2011, 1, 9–14. [Google Scholar]
- Gilani, A.-U.H.; Janbaz, K.H. Preventive and curative effects of Artemisia absinthium on acetaminophen and CCl4- induced hepatotoxicity. Gen. Pharm. 1995, 26, 309–315. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Ebrahimzadeh, M.A.; Ansaroudi, F.; Nabavi, S.F.; Nabavi, S.M. Antidepressant and antioxidant activities of Artemisia absinthium L. at flowering stage. Afr. J. Biotechnol. 2009, 8, 7170–7175. [Google Scholar]
- Erturk, O.; Uslu, U. Antifeedant, growth and toxic effects of some plant extracts on Leptinotarsa decemlineata (Say.) (Coleoptera, Chrysomelidae). Fresenius Environ. Bull. 2007, 16, 601–607. [Google Scholar]
- Lachenmeier, D.W. Wormwood (Artemisia absinthium L.)—A curious plant with both neurotoxic and neuroprotective properties. J. Ethnopharmacol. 2010, 131, 224–227. [Google Scholar] [CrossRef] [PubMed]
- Bora, K.S.; Sharma, A. Neuroprotective effect of Artemisia absinthium L. on focal ischemia and reperfusion-induced cerebral injury. J. Ethnopharmacol. 2010, 129, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Amat, N.; Upur, H.; Blazekovi, B. In vivo hepatoprotective activity of the aqueous extract of Artemisia absinthium L. against chemically and immunologically induced liver injuries in mice. J. Ethnopharmacol. 2010, 131, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Bhat, R.R.; Rehman, M.U.; Shabir, A.; Rahman Mir, M.U.; Ahmad, A.; Khan, R.; Masoodi, M.H.; Madkhali, H.; Ganaie, M.A. Chemical Composition and Biological Uses of Artemisia absinthium (Wormwood). Plant Hum. Health 2019, 3, 37–63. [Google Scholar] [CrossRef]
- Hadi, A.; Hossein, N.; Shirin, P.; Najmeh, N.; Abolfazl, M. Anti-inflammatory and Analgesic Activities of Artemisia absinthium. Int. J. Pharm. Sci. Rev. Res. 2014, 24, 237–244. [Google Scholar]
- Predoi, D.; Iconaru, S.L.; Buton, N.; Badea, M.L.; Marutescu, L. Antimicrobial Activity of New Materials Based on Lavender and Basil Essential Oils and Hydroxyapatite. Nanomaterials 2018, 8, 291. [Google Scholar] [CrossRef]
- Kennedy, A.I.; Deans, S.G.; Svoboda, K.P.; Gray, A.I.; Waterman, P.G. Volatile oils from normal and transformed root of Artemisia absinthium. Phytochemistry 1993, 32, 1449–1451. [Google Scholar] [CrossRef]
- Nin, S.; Bennici, A.; Roselli, G.; Mariotti, D.; Schiff, S.; Magherini, R. Agrobacterium-mediated transformation of Artemisia absinthium L. (wormwood) and production of secondary metabolites. Plant Cell Rep. 1997, 16, 725–730. [Google Scholar] [CrossRef]
- Blagojevicä, P.; Radulovicä, N.; Palicä, R.; Stojanovicä, G. Chemical Composition of the Essential Oils of Serbian Wild-Growing Artemisia absinthium and Artemisia vulgaris. J. Agric. Food Chem. 2006, 54, 4780–4789. [Google Scholar] [CrossRef]
- Available online: http://imagej.nih.gov/ij (accessed on 10 January 2018).
- Predoi, D.; Groza, A.; Iconaru, S.L.; Predoi, G.; Barbuceanu, F.; Guegan, R.; Motelica-Heino, M.S.; Cimpeanu, C. Properties of Basil and Lavender Essential Oils Adsorbed on the Surface of Hydroxyapatite. Materials 2018, 11, 652. [Google Scholar] [CrossRef] [PubMed]
- Sahu, S.C.; Casciano, D.A. (Eds.) Nanotoxicity: From In Vivo and In Vitro Models to Health Risks; Wiley: Hoboken, NJ, USA, 2009; ISBN 978-0-470-74779-7. [Google Scholar]
- Kruk, M.; Jaroniec, M. Gas adsorption characterization of ordered organic inorganic nanocomposite materials. Chem. Mater. 2001, 13, 3169–3183. [Google Scholar] [CrossRef]
- Wypych, A.; Bobowska, I.; Tracz, M.; Opasinska, A.; Kadlubowski, S.; Krzywania-Kaliszewska, A.; Grobelny, J.; Wojciechowski, P. Dielectric Properties and Characterisation of Titanium Dioxide Obtained by Different Chemistry Methods. J. Nanomater. 2014, 2014, 124814. [Google Scholar] [CrossRef]
- Iconaru, S.L.; Chifiriuc, M.C.; Groza, A. Structural and Antimicrobial Evaluation of Silver Doped Hydroxyapatite-Polydimethylsiloxane Thin Layers. J. Nanomater. 2017, 2017, 7492515. [Google Scholar] [CrossRef]
- Popa, C.L.; Groza, A.; Chapon, P.; Ciobanu, C.S.; Ghita, R.V.; Trusca, R.; Ganciu, M.; Predoi, D. Physicochemical Analysis of the Polydimethylsiloxane Interlayer Influence on a Hydroxyapatite Doped with Silver Coating. J. Nanomater. 2015, 2015, 250617. [Google Scholar] [CrossRef]
- Morar, M.I.; Fetea, F.; Rotar, A.M.; Nagy, M.; Semeniuc, C.A. Characterization of essential oils extracted from different aromatic plants by FTIR spectroscopy. Bull. UASVM Food Sci. Technol. 2017, 74, 1–2. [Google Scholar] [CrossRef]
- Nowak, A.; Kalemba, D.; Piotrowska, M.; Czyżowska, A. Effects of thyme (Thymus vulgaris L.) and rosemary (Rosmarinus officinalis L.) essential oils on growth of Brochothrix. Afr. J. Microbiol. Res. 2013, 7, 3396–3404. [Google Scholar]
- Coates, J. Interpretation of Infrared Spectra, A Practical Approach. In Encyclopedia of Analytical Chemistry; Meyers, R.A., Ed.; JohnWiley & Sons Ltd.: Chichester, UK, 2000; pp. 10815–10837. [Google Scholar]
- Samfira, I.; Rodino, S.; Petrache, P.; Cristina, R.T.; Butu, M.; Butnariu, M. Characterization and identity confirmation of essential oils by mid infrared absorbtion spectrophometry. Dig. J. Nanomater. Biostruct. 2015, 10, 557–566. [Google Scholar]
- Lafhal, S.; Vanloot, P.; Bombarda, I.; Kister, J.; Dupuy, N. Identification of metabolomic markers of lavender and lavandin essential oils using mid-infrared spectroscopy. Vib. Spectrosc. 2016, 85, 79–90. [Google Scholar] [CrossRef]
- Wright, C.W. Artemisia; Taylor & Francis: New York, NY, USA, 2002. [Google Scholar]
- Susan, K.; Mikota, D.V.M.; Donald, C.; Plumb, P.D. Dimethyl Sulfoxide; Elephant Care International: Hohenwald, TN, USA, 2003. [Google Scholar]
- Hassan, A.S. The Antibacterial Activity of Dimethyl Sulfoxide (DMSO) withand without of Some Ligand Complexes of the Transitional MetalIons of Ethyl Coumarin against Bacteria Isolate from Burn and Wound Infection. J. Nat. Sci. Res. 2014, 4, 2224–3186. [Google Scholar]
- Lopes-Lutz, D.; Alviano, D.S.; Alviano, C.S.; Kolodziejczyk, P.P. Screening of chemical composition, antimicrobial and antioxidant activities of Artemisia essential oils. Phytochemistry 2008, 69, 1732–1738. [Google Scholar] [CrossRef] [PubMed]
- Janaki, A.C.; Sailatha, E.; Gunasekaran, S. Synthesis, characteristics and antimicrobial activity of ZnO nanoparticles. Spectrochim. Acta Part A. 2015, 144, 17–22. [Google Scholar] [CrossRef]
- Weete, J.D.; Abril, M.; Blackwell, M. Phylogenetic distribution of fungal sterols. PLoS ONE 2010, 5, e10899. [Google Scholar] [CrossRef]
- Wang, L.; Hu, C.; Shao, L. The antimicrobial activity of nanoparticles: Present situation and prospects for the future. Int. J. Nanomed. 2017, 14, 1227–1249. [Google Scholar] [CrossRef]
- Leung, Y.H.; Ng, A.M.; Xu, X.; Shen, Z.; Gethings, L.A.; Wong, M.T.; Chan, C.M.; Guo, M.Y.; Ng, Y.H.; Djurišić, A.B.; et al. Mechanisms of antibacterial activity of MgO: Non-ROS mediated toxicity of MgO nanoparticles towards Escherichia coli. Small 2014, 10, 1171–1183. [Google Scholar] [CrossRef]
- Beyth, N.; Houri-Haddad, Y.; Domb, A.; Khan, W.; Hazan, R. Alternative antimicrobial approach: Nano-antimicrobial materials. Evid. Based. Complement. Altern. Med. 2015, 2015, 246012. [Google Scholar] [CrossRef]
- Poole, K. Mechanisms of bacterial biocide and antibiotic resistance. J. Appl. Microbiol. 2002, 92, 55S–64S. [Google Scholar] [CrossRef]
- Peacock, S.J.; Paterson, G.K. Mechanisms of Methicillin Resistance in Staphylococcus aureus. Annu. Rev. Biochem. 2015, 84, 577–601. [Google Scholar] [CrossRef]
- Niland, C.N.; Anderson, D.R.; Jankowsky, E.; Harris, M.E. The contribution of the C5 protein subunit of Escherichia coli ribonuclease P to specificity for precursor tRNA is modulated by proximal 5′ leader sequences. RNA 2017, 23, 1502–1511. [Google Scholar] [CrossRef] [PubMed]
- Al-Tahhan, R.A.; Sandrin, T.R.; Bodour, A.A.; Maier, R.M. Rhamnolipid-induced removal of lipopolysaccharide from Pseudomonas aeruginosa: Effect on cell surface properties and interaction with hydrophobic substrates. Appl. Environ. Microbiol. 2000, 66, 3262–3268. [Google Scholar] [CrossRef] [PubMed]
- Bassolé, I.H.N.; Lamien-Meda, A.; Bayala, B.; Tirogo, S.; Franz, C.; Novak, J.; Nebié, R.C.; Dicko, M.H. Composition and antimicrobial activities of Lippia multi-flora Moldenke, Mentha piperita L. and Ocimum basilicum L. essential oils and their major monoterpene alcohols alone and in combination. Molecules 2010, 15, 7825–7839. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, W.; Li, H.; Liu, S.; Li, S.; Zhang, Y. Formation of dicalcium phosphate dehydrate coating on carbon fibers with in-situ grown graphene nanosheet interlayer. Ceram. Int. 2017, 43, 5968–5973. [Google Scholar]
- Donlan, R.M. Biofilms: Microbial life on surfaces. Emerg. Infect. Dis. 2002, 8, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Katsikogianni, M.; Missirlis, Y.F. Concise review of mechanisms of bacterial adhesion to biomaterials and of techniques used in estimating bacteria-material interactions. Eur. Cells Mater. 2004, 8, 37–57. [Google Scholar] [CrossRef]
- Katainen, J.; Paajanen, M.; Ahtola, E.; Pore, V.; Lahtinen, J. Adhesion as an interplay between particle size and surface roughness. J. Colloid Interface Sci. 2006, 304, 524–529. [Google Scholar] [CrossRef]
- Kawai, K.; Urano, M.; Ebisu, S. Effect of surface roughness of porcelain on adhesion of bacteria and their synthesizing glucans. J. Prosthet. Dent. 2000, 83, 664–667. [Google Scholar] [CrossRef]
- Carlén, A.; Nikdel, K.; Wennerberg, A.; Holmberg, K.; Olsson, J. Surface characteristics and in vitro biofilm formation on glass ionomer and composite resin. Biomaterials 2001, 22, 481–487. [Google Scholar] [CrossRef]
- Buergers, R.; Rosentritt, M.; Handel, G. Bacterial adhesion of Streptococcus mutans to provisional fixed prosthodontic material. J. Prosthet. Dent. 2007, 98, 461–469. [Google Scholar] [CrossRef]
- Zhou, S.; Wei, C.; Zhang, C.; Han, C.; Kuchkarova, N.; Shao, H. Chemical Composition, Phytotoxic, Antimicrobial and Insecticidal Activity of the Essential Oils of Dracocephalum integrifolium. Toxins 2019, 11, 598. [Google Scholar] [CrossRef] [PubMed]
- Ghaffari, T.; Kafil, H.S.; Asnaashari, S.; Farajnia, S.; Delazar, A.; Baek, S.C.; Hamishehkar, H.; Kim, K.H. Chemical Composition and Antimicrobial Activity of Essential Oils from the Aerial Parts of Pinus eldarica Grown in Northwestern Iran. Molecules 2019, 24, 3203. [Google Scholar] [CrossRef] [PubMed]
- Şimşek, M.; Duman, R. Investigation of Effect of 1,8-cineole on Antimicrobial Activity of Chlorhexidine Gluconate. Pharmacogn. Res. 2017, 9, 234–237. [Google Scholar] [CrossRef] [PubMed]
- Indrayan, A.K.; Tyagi, P.K.; Agrawal, N.K. Chemical Composition and Antimicrobial Activity of the Essential Oil of Alpinia speciosa K. Schum. Rhizome from India. J. Essent. Oil Res. 2010, 22, 179–182. [Google Scholar] [CrossRef]
- Kordali, S.; Kotan, R.; Mavi, A.; Cakir, A.; Ala, A.; Yildirim, A. Determination of the chemical composition and antioxidant activity of the essential oil of Artemisia dracunculus and of the antifungal and antibacterial activities of Turkish Artemisia absinthium, A. dracunculus, Artemisia santonicum, and Artemisia spicigera essential oils. J. Agric. Food Chem. 2005, 53, 9452–9458. [Google Scholar]
- Umpierrez, M.L.; Lagreca, M.E.; Cabrera, R.; Grille, G.; Rossini, C. Essential oils from Asteraceae as potential biocontrol tools for tomato pests and diseases. Phytochem. Rev. 2012, 11, 339–350. [Google Scholar] [CrossRef]
- Ciobanu, C.S.; Constantin, L.V.; Predoi, D. Structural and physical properties of antibacterial Ag-doped nano-hydroxyapatite synthesized at 100 °C. Nanoscale Res. Lett. 2011, 6, 613. [Google Scholar] [CrossRef]
- Jennings, W.; Shibamoto, T. Qualitative Analysis of Flavor and Fragrance Volatile by Glass Capillary Gas Chromatography; Academic Press: New York, NY, USA, 1980. [Google Scholar]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Quadruple Mass Spectroscopy; Allured Publishing Corporation: Carol Stream, IL, USA, 2001. [Google Scholar]
- Groza, A.; Surmeian, A. Characterization of the oxides present in a polydimethylsiloxane layer obtained by polymerisation of its liquid precursor in corona discharge. J. Nanomater. 2015, 2015, 204296. [Google Scholar] [CrossRef]
- Prodan, A.M.; Beuran, M.; Turculet, C.S.; Popa, M.; Andronescu, E.; Bleotu, C.; Raita, S.M.; Soare, M.; Lupescu, O. In vitro evaluation of glycerol coated iron oxide nanoparticles in solution. Rom. Biotechnol. Lett. 2018, 23, 13901–13908. [Google Scholar]
- Nikolić, M.; Jovanović, K.K.; Marković, T.; Marković, D.; Gligorijević, N.; Radulović, S.; Soković, M. Chemical composition, antimicrobial, and cytotoxic properties of five Lamiaceae essential oils. Ind. Crop. Prod. 2014, 61, 225–232. [Google Scholar] [CrossRef]
- Prodan, A.M.; Iconaru, S.L.; Chifiriuc, M.C.; Bleotu, C.; Ciobanu, C.S.; Motelica-Heino, M.; Sizaret, S.; Predoi, D. Magnetic Properties and Biological Activity Evaluation of Iron Oxide Nanoparticles. J. Nanomater. 2013, 2013, 893970. [Google Scholar] [CrossRef]
- Turculet, C.S.; Prodan, A.M.; Negoi, I.; Teleanu, G.; Popa, M.; Andronescu, E.; Beuran, M.; Stanciu, G.A.; Hristu, R.; Badea, M.L.; et al. Preliminary evaluation of the antifungal activity of samarium doped hydroxyapatite thin films. Rom. Biotechnol. Lett. 2018, 23, 13928–13932. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |


| Compounds | Percentage of Chemical Composition (%) |
|---|---|
| α-Fenchene | 52.29 |
| Sabinene | 7.2 |
| β-Thujene | 6.28 |
| β-myrcene | 5.47 |
| methyl salicylate | 4.2 |
| endo-bornylacetate | 2.21 |
| ϒ-isogeraniol | 1.95 |
| neryl isobutyrate | 1.9 |
| dyhidrocarveol acetate | 1.29 |
| nerol | 1.28 |
| 1,8-Cineole | 1.05 |
| ϒ-Terpinene | 1.05 |
| β-Pinene | 1.02 |
| camphene | tr |
| α-pinene | tr |
| hexanol | tr |
| limonene | tr |
| neryl acetate | tr |
| bornyl isovalerate | tr |
| neryl propionate | tr |
| Sample | Single Point Surface Area (nm) | BET Surface Area (nm) | Langmuir Surface Area (nm) | t-Plot External Surface Area (nm) |
|---|---|---|---|---|
| HAp | 101.12 | 102.54 | 153.50 | 112.10 |
| HApWW | 115.24 | 116.72 | 173.40 | 123.60 |
| HAp Solution IR Band Wavenumber | IR Band Assignment | WW EO IR Band Wavenumber | IR Band Assignment | HApWW IR Band Wavenumber | IR Band Assignment |
|---|---|---|---|---|---|
| 588, 602, 633 | vibrations in [PO4]3− | 816, 890 | C-H vibrations | 563, 602, 630 | vibrations in [PO4]3− |
| 1418 | vibrations in [CO3]2− | 1014, 1070, 1108, 1154, 1185,1236 | C-O vibrations | 890 | C-H vibrations |
| 960, 1027, 1090 | vibrations in [PO4]3− | 1375,1449 | C-H vibrations | 960, 1027, 1092 | vibrations in [PO4]3− |
| 1631, 3309 | O-H vibrations | 1516,1589 | C=C-C stretching vibrations | 1375, 1449 | C-H vibrations in CH3 |
| 2140 | 1648 | C=C vibrations | 1648 | C=C vibrations | |
| 1737 | C=O stretching vibrations | 1737 | C=O stretching vibrations | ||
| 2872, 2961 | C-H vibrations in CH3 | 2872, 2961 | C-H vibrations in CH3 | ||
| 2930 | C-H vibrations in CH2 | 2930 | C-H vibrations in CH2 | ||
| 3078 | C-H vibrations | 3078 | C-H vibrations | ||
| 3408 | O-H vibrations | 3377 | O-H vibrations |
| Tested EOs | WW EO | HApWW | HAp | DMSO |
|---|---|---|---|---|
| Bacterial Strains | ||||
| E. coli ATCC 25922 | MBC 125 MIC 31.25 | 125 125 | > 50 | >250 |
| E. coli C5 | MBC 125 MIC 62.5 | 125 62.5 | >250 | >250 |
| P. aeruginosa ATCC 27853 | MBC 62.5 MIC 62.5 | 62.5 62.5 | >250 | >250 |
| P. aeruginosa ATCC 9027 | MBC 62.5 MIC 62.5 | 62.5 62.5 | >250 | >250 |
| S. aureus ATCC 25923 | MBC 62.5 MIC 62.5 | 250 62.5 | >250 | >250 |
| S. aureus ATCC 6538 | MBC 62.5 MIC 62.5 | 250 62.5 | >250 | >250 |
| S. aureus 388 (MRSA) | MBC 62.5 MIC 62.5 | 250 62.5 | >250 | >250 |
| E. faecium DSM 13590 | MBC 250 MIC 31.2 | 250 31.2 | >250 | >250 |
| C. parapsilosis ATCC 22019 | MBC 7.81 MIC 7.81 | 15.62 15.62 | 250 125 | >250 |
| Samples | WW EO | HapWW | Control |
|---|---|---|---|
| Bacterial Strains | |||
| E. coli ATCC 25922 | 9.7% | 0.2% | 0% |
| P. aeruginosa ATCC 27853 | 34.2% | 5.4% | 0.1% |
| S. aureus ATCC 25923 | 3.5% | 0.9% | 0.6% |
| E. faecium DSM 13590 | 2.1% | 8.2% | 0.3% |
| C. parapsilosis ATCC 22019 | 6.4% | 14.1% | 1.3% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raita, M.S.; Iconaru, S.L.; Groza, A.; Cimpeanu, C.; Predoi, G.; Ghegoiu, L.; Badea, M.L.; Chifiriuc, M.C.; Marutescu, L.; Trusca, R.; et al. Multifunctional Hydroxyapatite Coated with Arthemisia absinthium Composites. Molecules 2020, 25, 413. https://doi.org/10.3390/molecules25020413
Raita MS, Iconaru SL, Groza A, Cimpeanu C, Predoi G, Ghegoiu L, Badea ML, Chifiriuc MC, Marutescu L, Trusca R, et al. Multifunctional Hydroxyapatite Coated with Arthemisia absinthium Composites. Molecules. 2020; 25(2):413. https://doi.org/10.3390/molecules25020413
Chicago/Turabian StyleRaita, Mariana Stefania, Simona Liliana Iconaru, Andreea Groza, Carmen Cimpeanu, Gabriel Predoi, Liliana Ghegoiu, Monica Luminita Badea, Mariana Carmen Chifiriuc, Luminita Marutescu, Roxana Trusca, and et al. 2020. "Multifunctional Hydroxyapatite Coated with Arthemisia absinthium Composites" Molecules 25, no. 2: 413. https://doi.org/10.3390/molecules25020413
APA StyleRaita, M. S., Iconaru, S. L., Groza, A., Cimpeanu, C., Predoi, G., Ghegoiu, L., Badea, M. L., Chifiriuc, M. C., Marutescu, L., Trusca, R., Furnaris, C. F., Turculet, C. S., Enache, D. V., & Predoi, D. (2020). Multifunctional Hydroxyapatite Coated with Arthemisia absinthium Composites. Molecules, 25(2), 413. https://doi.org/10.3390/molecules25020413











