Significance of Artemisia Vulgaris L. (Common Mugwort) in the History of Medicine and Its Possible Contemporary Applications Substantiated by Phytochemical and Pharmacological Studies
Abstract
1. Introduction
2. Distribution and Taxonomic Consideration of Artemisia
3. Genetic Issues
4. Botanical Characteristics
5. Natural Habitats and Cultivation
6. Status of the Plant in Official Phytotherapy in Europe
7. Phytochemical Characteristics
8. History of Medicinal Use
9. Applications in Traditional Medicine Worldwide
10. Applications in Modern Phytotherapy and Position in Official European Medicine
11. Biological Activities of Extracts Confirmed by Scientific Research
11.1. Antioxidant Effect
11.2. Hypolipemic Effect
11.3. Hepatoprotective Effect
11.4. Antispasmodic Effect
11.5. Bronchodilatory Effect
11.6. Analgesic Effect
11.7. MAO Inhibitory Effect
11.8. Antihypertensive Effect
11.9. Estrogenic Effect
11.10. Cytotoxic Effect
11.11. Antifungal and Antibacterial Activities
11.12. Anti-Inflammatory Activity
11.13. Antiallergenic Effect
11.14. Antimalarial Activity
11.15. Anthelmintic Activity
11.16. Insecticidal Activity
12. Applications in Cosmetology
13. Applications in the Food Industry
14. Safety of Use
15. Biotechnological Research on Micropropagation
16. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Haq, F.U.; Roman, M.; Ahmad, K.; Rahman, S.U.; Shah, S.M.A.; Suleman, N.; Ullah, S.; Ahmad, I.; Ullah, W. Artemisia annua: Trials are needed for COVID-19. Phyther. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T.; Zacchino, S.; Georgiev, M.I.; Liu, L.; Wagner, H.; Panossian, A. Nobel Prize for artemisinin brings phytotherapy into the spotlight. Phytomedicine Int. J. Phyther. Phytopharm. 2015, 22, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Wichtl, M. Herbal Drugs and Phytopharmaceuticals: A Handbook for Practice on a Scientific Basis, 3rd ed.; Medpharm: Marburg, Germany, 2004. [Google Scholar]
- The Plant List. Available online: http://www.theplantlist.org (accessed on 10 March 2020).
- Holm, L.; Doll, J.; Holm, E.; Pnacho, J.; Herberger, J. World Weeds: Natural Histories and Distribution; John Wiley and Sons: New York, NY, USA, 1997. [Google Scholar]
- Chevallier, A. The Encyclopedia of Medicinal Plants: A Practical Reference Guide to more than 500 Key Medicinal Plants and Their Uses; DK Publishing: New York, NY, USA, 1996. [Google Scholar]
- Barney, J.N.; DiTommaso, A. The biology of Canadian weeds. 118. Artemisia vulgaris L. Can. J. Plant. Sci. 2003, 83, 205–215. [Google Scholar] [CrossRef]
- Temraz, A.; El-Tantawy, W.H. Characterization of antioxidant activity of extract from Artemisia vulgaris. Pak. J. Pharm. Sci. 2008, 21, 321–326. [Google Scholar] [PubMed]
- Khan, K.A. A preclinical antihyperlipidemic evaluation of Artemisia vulgaris root in diet induced hyperlipidemic animal model. Int. J. Pharmacol. Res. 2015, 5, 110–114. [Google Scholar]
- Natividad, G.M.; Broadley, K.J.; Kariuki, B.; Kidd, E.J.; Ford, W.R.; Simons, C. Actions of Artemisia vulgaris extracts and isolated sesquiterpene lactones against receptors mediating contraction of guinea pig ileum and trachea. J. Ethnopharmacol. 2011, 137, 808–816. [Google Scholar] [CrossRef]
- Pires, J.M.; Mendes, F.R.; Negri, G.; Duarte-almeida, J.M.; Carlini, E.A. Antinociceptive Peripheral Effect of Achillea millefolium L. and Artemisia vulgaris L.: Both Plants known popularly by Brand Names of Analgesic Drugs. Phyther. Res. 2009, 219, 212–219. [Google Scholar] [CrossRef]
- Tigno, X.T.; de Guzman, F.; Flora, A.M.; Theresa, V. Phytochemical analysis and hemodynamic actions of Artemisia vulgaris L. Clin. Hemorheol. Microcirc. 2000, 23, 167–175. [Google Scholar]
- Lee, S.J.; Chung, H.Y.; Maier, C.G.A.; Wood, A.R.; Dixon, R.A.; Mabry, T.J. Estrogenic Flavonoids from Artemisia vulgaris L. J. Agric. Food Chem. 1998, 46, 3325–3329. [Google Scholar] [CrossRef]
- Erel, B.; Aydin, F.; Ballar, P. In vitro cytotoxic properties of six Artemisia L. species. Turkish J. Pharm. Sci. 2011, 8, 247–251. [Google Scholar]
- Obistioiu, D.; Cristina, R.T.; Schmerold, I.; Chizzola, R.; Stolze, K.; Nichita, I.; Chiurciu, V. Chemical characterization by GC-MS and in vitro activity against Candida albicans of volatile fractions prepared from Artemisia dracunculus, Artemisia abrotanum, Artemisia absinthium and Artemisia vulgaris. Chem. Cent. J. 2014, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Blagojević, P.; Radulović, N.; Palić, R.; Stojanović, G. Chemical composition of the essential oils of Serbian wild-growing Artemisia absinthium and Artemisia vulgaris. J. Agric. Food Chem. 2006, 54, 4780–4789. [Google Scholar] [CrossRef] [PubMed]
- Govindaraj, S.; Ranjitha Kumari, B.D. Composition and Larvicidal Activity of Artemisia vulgaris L. Stem Essential Oil against Aedes Aegypti. Jordan J. Biol. Sci. 2013, 6, 11–16. [Google Scholar] [CrossRef][Green Version]
- European Food Safety Authority. Artemisia Vulgaris Basic Substance Application; EFSA Supporting Publications: Parma, Italy, 2013. [Google Scholar]
- European Commission. CosIng-Cosmetic Database. Available online: https://ec.europa.eu/growth/sectors/cosmetics/cosing_en (accessed on 8 March 2020).
- Anwar, S.; Asif, N.; Naqvi, S.A.H.; Malik, S. Evaluation of multiple risk factors involved in the development of diabetic retinopathy. Pakistan J. Med. Sci. 2019, 35, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Abiri, R.; Silva, A.L.M.; de Mesquita, L.S.S.; de Mesquita, J.W.C.; Atabaki, N.; de Almeida, E.B.; Shaharuddin, N.A.; Malik, S. Towards a better understanding of Artemisia vulgaris: Botany, phytochemistry, pharmacological and biotechnological potential. Food Res. Int. 2018, 109, 403–415. [Google Scholar] [CrossRef]
- Abad, M.J.; Bedoya, L.M.; Apaza, L.; Bermejo, P. The Artemisia L. genus: A review of bioactive essential oils. Molecules 2012, 17, 2542–2566. [Google Scholar] [CrossRef]
- Tan, R.X.; Zheng, W.F.; Tang, H.Q. Biologically Active Substances from the Genus Artemisia. Planta Med. 1998, 64, 295–302. [Google Scholar] [CrossRef]
- Bora, K.S.; Sharma, A. The genus Artemisia: A comprehensive review. Pharm. Biol. 2011, 49, 101–109. [Google Scholar] [CrossRef]
- Hegnauer, R. Chemotaxonomie der Pflanzen; Springer: Basel, Switzerland, 1962. [Google Scholar]
- Tutin, T.G.; Heywood, V.H.; Burges, N.A.; Valentine, D.H. Flora Europaea: Volume 4. Plantaginaceae to Composite (and Rubiaceae); Cambridge University Press: Cambridge, UK, 1976; Volume 4. [Google Scholar]
- Szafer, W.; Kulczyński, S.; Pawłowski, B. Rośliny Polskie. Vol. I-III.; Państwowe Wydawnictwo Naukowe: Warszawa, Poland, 1986. [Google Scholar]
- Zarzycki, K.; Kaźmierczakowa, R.; Mirek, Z. Polska Czerwona Księga Roślin. Paprotniki i rośliny kwiatowe; Wyd. III.; Instytut Ochrony Przyrody PAN: Kraków, Poland, 2014. [Google Scholar]
- Missouri Botanical Garden. Available online: www.tropicos.org (accessed on 7 March 2020).
- Weston, L.A.; Barney, J.N.; DiTommaso, A. A review of the biology and ecology of three invasive perennials in New York State: Japanese knotweed (Polygonum cuspidatum), mugwort (Artemisia vulgaris) and pale swallow-wort (Vincetoxicum rossicum). Plant. Soil 2005, 277, 53–69. [Google Scholar] [CrossRef]
- van Wyk, B.-E.; Wink, M. Medicinal Plants of the World; Timber Press: Portland, OR, USA, 2004. [Google Scholar]
- Vallès, J.; McArthur, D.E. Artemisia Systematics and Phylogeny: Cytogenetic and Molecular Insights. In Shrubland Ecosystem Genetics and Biodiversity: Proceedings; McArthur, E.D., Fairbanks, D.J., Eds.; Department of Agriculture, Forest Service, Rocky Mountain Research Station: Ogden, UT, USA, 2001; pp. 13–15. [Google Scholar]
- Oliva, M.; Vallès, J. Karyological studies in some taxa of the genus Artemisia (Asteraceae). Can. J. Bot. 1994, 72, 1126–1135. [Google Scholar]
- Tabur, S.; Civelek, Ş.; Öney, S.; Ergün, Ş.B.Y. Chromosome counts and karyomorphology of some species of Artemisia (Asteraceae) from Turkey. Turk. J. Bot. 2012, 36, 235–246. [Google Scholar]
- Vallès, J.; Garcia, S.; Hidalgo, O.; Martín, J.; Pellicer, J.; Sanz, M.; Garnatje, T. Biology, genome evolution, biotechnological issues and research including applied perspectives in Artemisia (Asteraceae). In Advances in Botanical Research; Academic Press Inc.: Cambridge, MA, USA, 2011; Volume 60, pp. 349–419. [Google Scholar]
- Gupta, R.C.; Goyal, H.; Singh, V. Cytology of the genus Artemisia (Anthemidae, Asteraceae) in the Western Himalayas. Biologia 2014, 69, 1134–1141. [Google Scholar] [CrossRef]
- Pellicer, J.; Garcia, S.; Garnatje, T.; Hidalgo, O.; Korobkov, A.A.; Dariimaa, S.; Vallès, J. Chromosome counts in Asian Artemisia L. (Asteraceae) species: From diploids to the first report of the highest polyploid in the genus. Bot. J. Linn. Soc. 2007, 153, 301–310. [Google Scholar] [CrossRef]
- Korobkov, A.A.; Kotseruba, V.V.; Probatova, N.S. Chromosome numbers of some species of Artemisia L. from Altai region, South Siberia. Bot. Pacifica 2014, 3, 61–66. [Google Scholar] [CrossRef]
- Koul, M.L.H. Cytogenetics of Polyploids I. Cytology of polyploid Artemisia vulgaris. Cytologia 1964, 29, 407–414. [Google Scholar] [CrossRef][Green Version]
- Xirau, J.V.; Siljak-Yakovlev, S. Cytogenetic studies in the genus Artemisia L. (Asteraceae): Fluorochrome-banded karyotypes of five taxa, including the Iberian endemic species Artemisia barrelieri Besser. Can. J. Bot. 1997, 75, 595–606. [Google Scholar] [CrossRef]
- Kim, G.B.; Lim, C.E.; Kim, J.S.; Kim, K.; Lee, J.H.; Yu, H.J.; Mun, J.H. Comparative chloroplast genome analysis of Artemisia (Asteraceae) in East Asia: Insights into evolutionary divergence and phylogenomic implications. BMC Genomics 2020, 21, 415. [Google Scholar] [CrossRef]
- Barney, J.N.; Hay, A.G.; Weston, L.A. Isolation and characterization of allelopathic volatiles from mugwort (Artemisia vulgaris). J. Chem. Ecol. 2005, 31, 247–265. [Google Scholar] [CrossRef]
- The Herb Society of America. Artemisia—An Essential Facts. 2014. Available online: https://www.herbsociety.org/file_download/inline/d52eae8c-be89-497d-94b3-7fc8da4105f1 (accessed on 7 March 2020).
- Borzabad, R.K.; Sudarshana, M.S.; Niranjan, M.H. In vitro Plant Regeneration from Leaf Explants of Artemisia vulgaris L.–A Medicinal Herb. Mod. Appl. Sci. 2010, 4, 130–134. [Google Scholar] [CrossRef]
- Uva, R.H.; Neal, J.C.; DiTomaso, J.M. Weeds of the Northeast; Cornell University Press: Ithaca, NY, USA, 1997. [Google Scholar]
- Anwar, F.; Ahmad, N.; Alkharfy, K.M.; Gilani, A.H. Mugwort (Artemisia vulgaris) oils. In Essential Oils in Food Preservation, Flavor and Safety; Academic Press: London, UK, 2016; pp. 573–579. ISBN 9780124166448. [Google Scholar]
- Garnock-Jones, P.J. Floret specialization, seed production and gender in Artemisia vulgaris L. (Asteraceae, Anthemideae). Bot. J. Linn. Soc. 1986, 92, 285–302. [Google Scholar] [CrossRef]
- Gleason, H.A.; Cronquist, A. Manual of Vascular Plants of Northeastern United States and Adjacent Canada, 2nd ed.; The New York Botanical Garden: New York, NY, USA, 1991. [Google Scholar]
- Pawłowski, F.; Kapeluszny, T.; Kolasa, A.; Lecyk, Z. Fertility of some species of ruderal weeds. Ann. Univ. Mariae Curie-Slodowska 1968, 22, 221–223. [Google Scholar]
- Hale, M. Allelopathic potential of Artemisia vulgaris rhizomes. Plant. Physiol. 1982, 69, S126. [Google Scholar]
- Bundesinstitut für Arzneimittel und Medizinprodukt. German Commission E Monograph; Blaumenthal, M.T., Hall, R., Rister, B., Eds.; American Botanical Council: Austin, TX, USA, 1988. [Google Scholar]
- European Directorate for the Quality of Medicine. European Pharmacopoeia 10.0; Council of Europe: Strasbourg, France, 2020. [Google Scholar]
- Française Pharmacopée. Pharmacopée Française, 11th ed.; Noculak, A., Ed.; Georg Olms Verlag: Hildesheim, France, 2020; Volume 37. [Google Scholar]
- Marco, J.A.; Sanz, T.J.; Del Hierro, P. Two eudesmane acids from Artemisia vulgaris. Pytochemistry 1991, 30, 2403–2404. [Google Scholar] [CrossRef]
- Geissmax, T.A.; Ellestad, A. Vulgarin, a Sesquiterpene Lactone from Artemisia vulgaris L. J. Org. Chem. 1961, 27, 1855–1859. [Google Scholar] [CrossRef]
- Numonov, S.; Sharopov, F.; Salimov, A.; Sukhrobov, P.; Atolikshoeva, S.; Safarzoda, R.; Habasi, M.; Aisa, H. Assessment of Artemisinin Contents in Selected Artemisia Species from Tajikistan (Central Asia). Medicines 2019, 6, 23. [Google Scholar] [CrossRef]
- Nganthoi, M.; Sanatombi, K. Artemisinin content and DNA profiling of Artemisia species of Manipur. South Afr. J. Bot. 2019, 125, 9–15. [Google Scholar] [CrossRef]
- Lee, K.H.; Jung, M.Y.; Kim, S.Y. Effects of Ascorbic Acid on the Light-Induced Riboflavin Degradation and Color Changes in Milks. J. Agric. Food Chem. 1998, 46, 407–410. [Google Scholar] [CrossRef]
- Melguizo-Melguizo, D.; Diaz-de-Cerio, E.; Quirantes-Piné, R.; Švarc-Gajić, J.; Segura-Carretero, A. The potential of Artemisia vulgaris leaves as a source of antioxidant phenolic compounds. J. Funct. Foods 2020, 5, 192–200. [Google Scholar] [CrossRef]
- Wallnofer, B.; Hofner, O.; Greger, H. Polyacetylenes from the Artemisia ‘Vulgares’ group. Phytochemistry 1989, 28, 2687–2691. [Google Scholar] [CrossRef]
- Carnat, A.; Heitz, A.; Fraisse, D.; Carnat, A.P.; Lamaison, J.L. Major dicaffeoylquinic acids from Artemisia vulgaris. Fitoterapia 2000, 71, 587–589. [Google Scholar] [CrossRef]
- Judžentien, A.; Buzelyte, J. Chemical composition of essential oils of Artemisia vulgaris L. (mugwort ) from North Lithuania. Chemija 2006, 17, 12–15. [Google Scholar]
- Mizushina, Y.; Takahashi, N.; Ogawa, A.; Tsurugaya, K.; Koshino, H.; Takemura, M.; Yoshida, S.; Matsukage, A.; Sugawara, F.; Sakaguchi, K. The cyanogenic glucoside, prunasin (D-mandelonitrile-β-D-glucoside), is a novel inhibitor of DNA polymerase β. J. Biochem. 1999, 126, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Govindaraj, S.; Kumari, B.D.R.; Cioni, P.L.; Flamini, G. Mass propagation and essential oil analysis of Artemisia vulgaris. J. Biosci. Bioeng. 2008, 105, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Madhav, K.; Kunal, M.; Zafar, H.; Ujjwal, B.; Gaurav, N. Antioxidant analysis of essential oils and methanolic extracts of Artemisia vulgaris. Int. J. Agric. Sci. 2018, 10, 5710–5713. [Google Scholar]
- El-Sahhar, K.F.; Nassar, R.M.; Farag, H.M. Morphological and anatomical studies of Artemisia vulgaris L. (Asteraceae) II. Anatomical characteristics and volatile oil. Aust. J. Basic Appl. Sci. 2015, 5, 56–68. [Google Scholar]
- Malik, S.; de Mesquita, L.; Silva, C.; de Mesquita, J.; de Sá Rocha, E.; Bose, J.; Abiri, R.; de Maria Silva Figueiredo, P.; Costa-Júnior, L. Chemical Profile and Biological Activities of Essential Oil from Artemisia vulgaris L. cultivated in Brazil. Pharmaceuticals 2019, 12, 49. [Google Scholar] [CrossRef]
- European Food Safety Authority. Botanical Summary Report. Available online: https://www.efsa.europa.eu/ (accessed on 1 March 2020).
- Koul, B.; Taak, P. The Artemisia Genus: A Review on Traditional Uses, Phytochemical Constituents, Pharmacological Properties and Germplasm Conservation. J. Glycomics Lipidomics 2017, 7, 142. [Google Scholar] [CrossRef]
- Duke, J.A.; Bogenshutz-Godwin, M.J.; DuCellier, M.J.; Duke, P.A. Handbook of Medicinal Herbs, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Pelkonen, O.; Abass, K.; Wiesner, J. Thujone and thujone-containing herbal medicinal and botanical products: Toxicological assessment. Regul. Toxicol. Pharmacol. 2013, 65, 100–107. [Google Scholar] [CrossRef]
- European Union Reference Laboratory for Feed Additives. Evaluation Report on the Analytical Methods Submitted in Connection with the Application for Authorisation of a Feed Additive According to Regulation (EC) No 1831/2003. Available online: https://ec.europa.eu/jrc/sites/jrcsh/files/finrep-fad-2017-0021-ronozyme_hiphos.pdf (accessed on 1 March 2020).
- Jerkovic, I.; Mastelic, J.; Milos, M.; Juteau, F.; Masotti, V.; Viano, J. Chemical variability of Artemisia vulgaris L. essential oils originated from the Mediterranean area of France and Croatia. Flavour Fragr. J. 2003, 18, 436–440. [Google Scholar] [CrossRef]
- Michaelis, K.; Vostrowsky, O.; Paulini, H.; Zintl, R.; Knobloch, K. On the Essential Oil Components from Blossoms of Artemisia vulgaris L. Z. Naturforsch. C Biosci. 1982, 37, 152–158. [Google Scholar] [CrossRef]
- Misra, L.N. σ-Thujone, the major component of the essential oil from Artemisia vulgaris growing wild in Nilgiri hills. J. Nat. Prod. 1986, 3, 941. [Google Scholar] [CrossRef]
- Mucciarelli, M.; Caramiello, R.; Maffei, M.; Chialva, F. Essential oils from some Artemisia species growing spontaneously in North-West Italy. Flavour Fragr. J. 1995, 10, 25–32. [Google Scholar] [CrossRef]
- Nano, M.G.; Bicchini, C.; Frattini, C.; Gallino, M. On the composition of some oils from Artemisia vulgaris. Plante Med. 1976, 30, 209–215. [Google Scholar] [CrossRef]
- Näf-Müller, R.; Pickenhagen, W.; Willhalm, B. New Irregular Monoterpenes in Artemisia vulgaris. Helv. Chim. Acta 1981, 64, 1424–1430. [Google Scholar] [CrossRef]
- Dũng, N.X.; Nam, V.V.; Huóng, H.T.; Leclercq, P.A. Chemical composition of the essential oil of Artemisia vulgaris L. var. indica Maxim. from Vietnam. J. Essent. Oil Res. 1992, 4, 433–434. [Google Scholar] [CrossRef]
- Thi Phuong Thao, N.; Thi Thuy, N.; Minh Hoi, T.; Huy Thai, T.; Muselli, A.; Bighelli, A.; Castola, V.; Casanova, J. Artemisia vulgaris L. from Vietnam: Chemical variability and composition of the oil along the vegetative life of the plant. J. Essent. Oil Res. 2004, 16, 358–361. [Google Scholar] [CrossRef]
- Des Berendes, J. Pedanios Dioskurides aus Anazarbos Arzneimittellehre in fünf Büchern. Übersetzt und mit Erklärungen Versehen; Band III.; Ferdinand Enke: Stuttgart, Germany, 1902. [Google Scholar]
- König, R.; Winkler, G.; Plinius Secundus, D.Ä.C. Naturkunde Lateinisch-Deutsch. 37 Bücher (und Register) in 32 Bänden; Artemis & Winkler: München, Germany, 1977. [Google Scholar]
- Karl Gottlob Kühn. Claudi Galeni Opera Omnia, Bd. 11, Leipzig 1826; Olms-Verlag: Hildesheim, Germany, 1965. [Google Scholar]
- Stoll, U. Das “Lorscher Arzneibuch“ (Codex Bambergis Medicinalis 1); Franz Steiner Verlag: Stuttgart, Germany, 1902. [Google Scholar]
- Wölfel, H. Das Arzneidrogenbuch Circa Instans in einer Fassung des XIII. Jahrhunderts aus der Universitätsbibliothek Erlangen: Text. und Kommentar als Beitrag zur Pflanzen- und Drogenkunde des Mittelalters; Mathematisch-Naturwissenschaftliche Dissertation: Berlin, Germany, 1939. [Google Scholar]
- Choulant (Hrsg.), J.L. Macer Floridus, De viribus Herbarum ‘una cum Walafridi Strabonis, Othonis Cremonensis et Ioannis Folcz’ ‘Carminibus Similis Argumenti Secundum Codices Manuscriptos et Veteres Editione Srecensuit, Supplevit et Adnotatione Criticains Truxit Ludovicus Choulan’; Leopold Voss: Leipzig, Germany, 1832. [Google Scholar]
- Wonnecke von Kaub, J.; Schöffer, P. Gart der Gesundheit, Mainz 1485, Kap. 1; Konrad Kölbl: München, Germany, 1996. [Google Scholar]
- Lonitzer, A.; Uffenbach, P. Kräuter-Buch und Künstliche Conterfeyungen der Bäumen, Stauden, Hecken, Kräuter; Verlag Bartholomae: Ulm, Germany, 1703. [Google Scholar]
- Brunfels, O. Contrafayt Kreüterbuch (mit naturgetreuen Abb. Hans Weidnitz), 2 Teile.; Basel, Switzerland, 1532.
- Madaus, G. Lehrbuch der Biologischen Heilmittel, Band I.; Georg Olms Verlag: Hildesheim, Germany, 1976. [Google Scholar]
- Tusaie, K.R.; Fitzpatric, J.J. Advanced Practice Psychiatric Nursing, 2nd ed.; Springer Publishing Company: Berlin, Germany, 2017. [Google Scholar]
- Ulbricht, C.E. Natural Standard, Herb and Supplement Guide; An. Evidence-Based Reference; Elsevier: Amsterdam, The Netherlands, 2010. [Google Scholar]
- Wheeler, J.; Coppock, B.; Chen, C. Does the burning of moxa (Artemisia vulgaris) in traditional Chinese medicine constitute a health hazard? Acupunct. Med. 2009, 27, 16–20. [Google Scholar] [CrossRef]
- Khare, C.P. Indian Herbal Remedies; Springer: Berlin, Germany, 2004. [Google Scholar]
- Rodrigues, E. Plants and animals utilized as medicines in the Jaú National Park (JNP), Brazilian Amazon. Phyther. Res. 2006, 20, 378–391. [Google Scholar] [CrossRef]
- de Albuquerque, U.P.; Monteiro, J.M.; Ramos, M.A.; de Amorim, E.L.C. Medicinal and magic plants from a public market in northeastern Brazil. J. Ethnopharmacol. 2007, 110, 76–91. [Google Scholar] [CrossRef]
- Milliken, W. Malaria and antimalarial plants in Roraima, Brazil. Trop. Doct. 1997, 27 (Suppl. S1), 20–25. [Google Scholar] [CrossRef]
- Quisumbing, E. Medicinal Plants of the Philippines; Bureau of Printing: Manila, Philippines, 1978. [Google Scholar]
- Lockie, A. Encyclopedia of Homeopathy; DK Publishing: New York, NY, USA, 2006; ISBN 9780756618711. [Google Scholar]
- Agence Nationale de sécurité du Médicamen. Mugwort for Homoeopathic Preparations Artemisia vulgaris 2004, 3–5. Available online: https://ansm.sante.fr/var/ansm_site/storage/original/application/a21d700c1a71f21672602ada7e998774.pdf (accessed on 15 March 2020).
- Oyedemi, S.; Coopoosamy, R. Preliminary Studies on the Antibacterial and Antioxidative Potentials of Hydroalcoholic Extract from the Whole Parts of Artemisia vulgaris L. Int. J. Pharmacol. 2015, 2, 1–9. [Google Scholar] [CrossRef]
- Baykan Erel, Ş.; Reznicek, G.; Şenol, S.G.; Karabay Yavaşoğulu, N.Ü.; Konyalioğlu, S. Antimicrobial and antioxidant properties of Artemisia L. species from western Anatolia. Turk. J. Biol 2012, 75, 75–84. [Google Scholar] [CrossRef]
- Ben Nasr, S.; Aazza, S.; Mnif, W.; Miguel, M. In-vitro antioxidant and anti-inflamatory activities of Pituranthos chloranthus and Artemisia vulgaris from Tunisia. Int. J. Appl. Pharm. Sci. Res. 2020, 11, 605–614. [Google Scholar] [CrossRef]
- El-Tantawy, W.H. Biochemical effects, hypolipidemic and anti-inflammatory activities of Artemisia vulgaris extract in hypercholesterolemic rats. J. Clin. Biochem. Nutr. 2015, 57, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Gilani, A.H.; Yaeesh, S.; Jamal, Q.; Ghayur, M.N. Hepatoprotective activity of aqueous-methanol extract of Artemisia vulgaris. Phyther. Res. 2005, 19, 170–172. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.U.; Gilani, A.H. Antispasmodic and bronchodilator activities of Artemisia vulgaris are mediated through dual blockade of muscarinic receptors and calcium influx. J. Ethnopharmacol. 2009, 126, 480–486. [Google Scholar] [CrossRef]
- Lee, S.-J.; Chung, H.-Y.; Lee, I.-K.; Oh, S.-U.; Yoo, I.-D. Phenolics with Inhibitory Activity on Mouse Brain Monoamine Oxidase (MAO) from Whole Parts of Artemisia vulgaris L. (Mugwort). Food Sci. Biotechnol. 2000, 9, 179–182. [Google Scholar]
- Yang, X.; Xiong, X.; Yang, G.; Wang, J. Effectiveness of Stimulation of Acupoint KI 1 by Artemisia vulgaris (Moxa) for the Treatment of Essential Hypertension: A Systematic Review of Randomized Controlled Trials. Evid. Based Complement Altern. Med. 2014, 2014, 187484. [Google Scholar] [CrossRef]
- Shaik, A.; Kanhere, R.S.; Cuddapah, R.; Nelson, K.S.; Vara, P.R.; Sibyala, S. Antifertility activity of Artemisia vulgaris leaves on female Wistar rats. Chin. J. Nat. Med. 2014, 12, 180–185. [Google Scholar] [CrossRef]
- Jakovljević, M.R.; Grujičić, D.; Vukajlović, J.T.; Marković, A.; Milutinović, M.; Stanković, M.; Vuković, N.; Vukić, M.; Milošević-Djordjević, O. In vitro study of genotoxic and cytotoxic activities of methanol extracts of Artemisia vulgaris L. and Artemisia alba Turra. South Afr. J. Bot. 2020, 132, 117–126. [Google Scholar] [CrossRef]
- Saleh, A.M.; Aljada, A.; Rizvi, S.A.A.; Nasr, A.; Alaskar, A.S.; Williams, J.D. In vitro cytotoxicity of Artemisia vulgaris L. essential oil is mediated by a mitochondria-dependent apoptosis in HL-60 leukemic cell line. BMC Complement. Altern. Med. 2014, 14, 226. [Google Scholar] [CrossRef]
- Raj Singh, B.; Singh, V.; Karan Singh, R.; Toppo, S.; Haque, N.; Ebibeni, N. Antimicrobial effect of Artemisia vulgaris essential oil. Nat. Prod. Indian J. 2011, 5, 1–7. [Google Scholar]
- Hiremath, S.K.; Kolume, D.G.; Muddapur, U.M. Antimicrobial activity of Artemisia vulgaris Linn. (Damanaka). Int. J. Res. Ayurveda Pharm. 2011, 2, 1674–1675. [Google Scholar]
- Singh, R.; Verma, P.; Singh, G. Total phenolic, flavonoids and tannin contents in different extracts of Artemisia absinthium. J. Intercult. Ethnopharmacol. 2012, 1, 101. [Google Scholar] [CrossRef]
- Afsar, S.K.; Rajesh Kumar, K.; Venu Gopal, J.; Raveesha, P. Assessment of anti-inflammatory activity of Artemisia vulgaris leaves by cotton pellet granuloma method in Wistar albino rats. J. Pharm. Res. 2013, 7, 463–467. [Google Scholar] [CrossRef]
- Olsen, O.T.; Frolund, L.; Heinig, J.; Jacobsen, L.; Svendsen, U.G. A double-blind, randomized study investigating the efficacy and specificity of immunotherapy with Artemisia vulgaris or Phleum pratense/Betula verrucosa. Allergol. Immunopathol. (Madr). 1995, 23, 73–78. [Google Scholar]
- Kodippili, K.; Ratnasooriya, W.D.; Premakumara, S.; Udagama, P.V. An investigation of the antimalarial activity of Artemisia vulgaris leaf extract in a rodent malaria model. Int. J. Green Pharm. 2011, 5, 1–7. [Google Scholar] [CrossRef]
- Bamunuarachchi, G.S.; Ratnasooriya, W.D.; Premakumara, S.; Udagama, P.V. Antimalarial properties of Artemisia vulgaris L. ethanolic leaf extract in a Plasmodium berghei murine malaria model. J. Vector Borne Dis. 2013, 50, 278–284. [Google Scholar]
- Caner, A.; Döşkaya, M.; Deǧirmenci, A.; Can, H.; Baykan, Ş.; Üner, A.; Başdemir, G.; Zeybek, U.; Gürüz, Y. Comparison of the effects of Artemisia vulgaris and Artemisia absinthium growing in western Anatolia against trichinellosis (Trichinella spiralis) in rats. Exp. Parasitol. 2008, 119, 173–179. [Google Scholar] [CrossRef]
- Teixeira, B.; Marques, A.; Ramos, C.; Neng, N.R.; Nogueira, J.M.F.; Saraiva, J.A.; Nunes, M.L. Chemical composition and antibacterial and antioxidant properties of commercial essential oils. Ind. Crops Prod. 2013, 43, 587–595. [Google Scholar] [CrossRef]
- Singh, B.R.; Singh, V.; Singh, R.K.; Toppo, S.; Haque, N.; Ebibeni, N. Comparative evaluation of antimicrobial effect of Artemisia vulgaris essential oils extracted from fresh and dried herb. Med. Plants 2012, 4, 76–82. [Google Scholar] [CrossRef]
- Lavor, P.L.; Santiago, G.M.P.; Da Silva Gois, R.W.; De Sousa, L.M.; Bezerra, G.D.P.; Romero, N.R.; Arriaga, Â.M.C.; Lemos, T.L.G.; Alves, P.B.; Gomes, P.C.S. Larvicidal activity against Aedes aegypti of essential oils from northeast Brazil. Nat. Prod. Commun. 2012, 7, 1391–1392. [Google Scholar] [CrossRef]
- Balasubramani, S.; Sabapathi, G.; Moola, A.K.; Solomon, R.V.; Venuvanalingam, P.; Bollipo Diana, R.K. Evaluation of the Leaf Essential Oil from Artemisia vulgaris and Its Larvicidal and Repellent Activity against Dengue Fever Vector Aedes aegypti-An Experimental and Molecular Docking Investigation. ACS Omega 2018, 3, 15657–15665. [Google Scholar] [CrossRef]
- Ilahi, I.; Ullah, F. Larvicidal Activities of Different Parts of Artemisia vulgaris Linn. against Culex quinquefasciatus Say. (Diptera: Culicidae). Int. J. Innov. Appl. Stud. 2013, 2, 189–195. [Google Scholar]
- Sharifian, I.; Hashemi, S.M.; Darvishzadeh, A. Fumigant toxicity of essential oil of Mugwort (Artemisia vulgaris L.) against three major stored product beetles. Arch. Phytopathol. Plant. Prot. 2013, 46, 445–450. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, F.; Zhou, X.M.; Niu, C.Y.; Lei, C.L. Repellent and fumigant activity of essential oil from Artemisia vulgaris to Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). J. Stored Prod. Res. 2006, 42, 339–347. [Google Scholar] [CrossRef]
- Seul Park, Y.; He Nam, G.; Jo Jo, K.; Won Kawk, H.; Yoo, J.-G.; Dong Jang, J.; Moon Kang, S.; Yong Kim, S.; Min Kim, Y. Adequacy of the Anti-aging and Anti-wrinkle Effects of the Artemisia vulgaris Fermented Solvent Fraction. Korean Soc. Biotechnol. Bioeng. J. 2019, 34, 199–206. [Google Scholar] [CrossRef]
- Bampidis, V.; Azimonti, G.; de Lourdes Bastos, M.; Christensen, H.; Kouba, M.; Kos Durjava, M.; López-Alonso, M.; López Puente, S.; Marcon, F.; Mayo, B.; et al. Safety and efficacy of a tincture derived from Artemisia vulgaris L. (Mugwort tincture) when used as a sensory additive in feed for all animal species. EFSA J. 2019, 17, 5879. [Google Scholar] [CrossRef]
- European Food Safety Authority. EFSA Supporting Publications. 2014, Volume 11. Available online: https://efsa.onlinelibrary.wiley.com/toc/23978325/2014/11/1 (accessed on 1 April 2020).
- Food and Drug Administration Pollens-Weeds and Garden Plants 2011. Available online: https://www.fda.gov/media/81288/download (accessed on 17 March 2020).
- Ipsen, H.; Formgren, H.; Løswenstein, H.; Ingemann, L. Immunochemical and Biological Characterization of a Mugwort (Artemisia vulgaris) Pollen Extract. Allergy 1985, 40, 289–294. [Google Scholar] [CrossRef]
- Yong, W.L.; Soo, Y.C.; Eun, K.L.; Jung, H.S.; Park, J.W.; Hong, C.S. Cross-allergenicity of pollens from the Compositae family: Artemisia vulgaris, Dendranthema grandiflorum, and Taraxacum officinale. Ann. Allergy Asthma Immunol. 2007, 99, 526–533. [Google Scholar] [CrossRef]
- Wrangsjö, K.; Ros, A.M.; Wahlberg, J.E. Contact allergy to Compositae plants in patients with summer-exacerbated dermatitis. Contact Dermatitis 1990, 22, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Haw, S.; Cho, H.-R.; Lee, M.-H. Allergic contact dermatitis associated with mugwort (Artemisia vulgaris). Contact Dermatitis 2010, 62, 61–63. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority Compendium of botanicals reported to contain naturally occuring substances of possible concern for human health when used in food and food supplements. EFSA J. 2012, 10. [CrossRef]
- Journal, T.E. Opinion of the Scientific Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food (AFC) on hydrocyanic acid in flavourings and other food ingredients with flavouring properties. EFSA J. 2004, 2, 1–28. [Google Scholar] [CrossRef]
- Sudarshana, M.S. In vitro regeneration of multiple shoots from encapsulated somatic embryos of Artemisia vulgaris L. IOSR J. Pharm. Biol. Sci. 2013, 6, 11–15. [Google Scholar] [CrossRef]
| Artemisia vulgaris Burm.f. [Illegitimate] |
| Artemisia vulgaris Mattf. [Illegitimate] |
| Artemisia vulgaris C.B.Clarke |
| Artemisia vulgaris var. americana Besser |
| Artemisia vulgaris f. angustisecta Fiori |
| Artemisia vulgaris var. aromatica Sacc. |
| Artemisia vulgaris subsp. candicans (Rydb.) H.M.Hall & Clem. |
| Artemisia vulgaris var. candicans (Rydb.) M.Peck |
| Artemisia vulgaris subsp. coarctata (Fors ex Besser) Ameljcz. |
| Artemisia vulgaris var. coarctica Besser |
| Artemisia vulgaris var. douglasiana H.St.John |
| Artemisia vulgaris subsp. flodmanii (Rydb.) H.M.Hall & Clem. |
| Artemisia vulgaris var. flodmanii (Rydb.) M.Peck |
| Artemisia vulgaris var. gilvescens (Miq.) Nakai |
| Artemisia vulgaris var. glabra Ledeb. |
| Artemisia vulgaris var. glandulifera (L.F.Hend.) M.Peck |
| Artemisia vulgaris subsp. gnaphalodes (Nutt.) H.M.Hall & Clem. |
| Artemisia vulgaris var. gnaphalodes (Nutt.) Kuntze |
| Artemisia vulgaris subsp. heterophylla (Nutt.) H.M.Hall & Clem. |
| Artemisia vulgaris var. incana Maxim. |
| Artemisia vulgaris var. incanescens Franch. |
| Artemisia vulgaris var. incompta (Nutt.) H.St.John |
| Artemisia vulgaris var. indica (Willd.) Hassk. |
| Artemisia vulgaris var. kamtschatica Besser |
| Artemisia vulgaris var. kiusiana Makino |
| Artemisia vulgaris var. latiloba Ledeb. |
| Artemisia vulgaris var. leucophylla Turcz. ex Besser |
| Artemisia vulgaris var. littoralis Suksd. |
| Artemisia vulgaris var. longifolia (Nutt.) M.Peck |
| Artemisia vulgaris subsp. longifolia (Nutt.) H.M.Hall & Clem. |
| Artemisia vulgaris subsp. ludoviciana (Nutt.) H.M.Hall & Clem. |
| Artemisia vulgaris var. ludoviciana (Nutt.) Kuntze |
| Artemisia vulgaris var. maximowiczii Nakai |
| Artemisia vulgaris var. mexicana (Willd. ex Spreng.) Torr. & A.Gray |
| Artemisia vulgaris subsp. mexicana (Willd. ex Spreng.) H.M.Hall & Clem. |
| Artemisia vulgaris subsp. michauxiana (Besser) H.St.John |
| Artemisia vulgaris var. minor Ledeb. |
| Artemisia vulgaris var. mongolica Fisch. ex Besser |
| Artemisia vulgaris f. montana Nakai |
| Artemisia vulgaris var. nilagirica C.B.Clarke |
| Artemisia vulgaris var. nipponica Nakai |
| Artemisia vulgaris f. nipponica Nakai |
| Artemisia vulgaris subsp. redolens (A.Gray) H.M.Hall & Clem. |
| Artemisia vulgaris var. rubriflora Turcz. ex Besser |
| Artemisia vulgaris subsp. selengensis Thell. |
| Artemisia vulgaris var. selengensis (Turcz. ex Besser) Maxim. |
| Artemisia vulgaris subsp. serrata (Nutt.) H.M.Hall & Clem. |
| Artemisia vulgaris var. stolonifera Maxim. |
| Artemisia vulgaris var. suaveolens Bég. |
| Artemisia vulgaris var. tenuifolia Turcz. ex DC. |
| Artemisia vulgaris subsp. tilesii (Ledeb.) H.M.Hall & Clem. |
| Artemisia vulgaris var. umbrosa Turcz. ex Besser |
| Artemisia vulgaris subsp. urjanchaica Ameljcz. |
| Artemisia vulgaris var. verbenacea Kom. |
| Artemisia vulgaris var. viridissima Kom. |
| Artemisia vulgaris var. vulgaris |
| Artemisia vulgaris var. wrightii (A.Gray) H.M.Hall & Clem. |
| Artemisia vulgaris subsp. wrightii (A.Gray) H.M.Hall & Clem. |
| Chemical Group | Compound | Content | References |
|---|---|---|---|
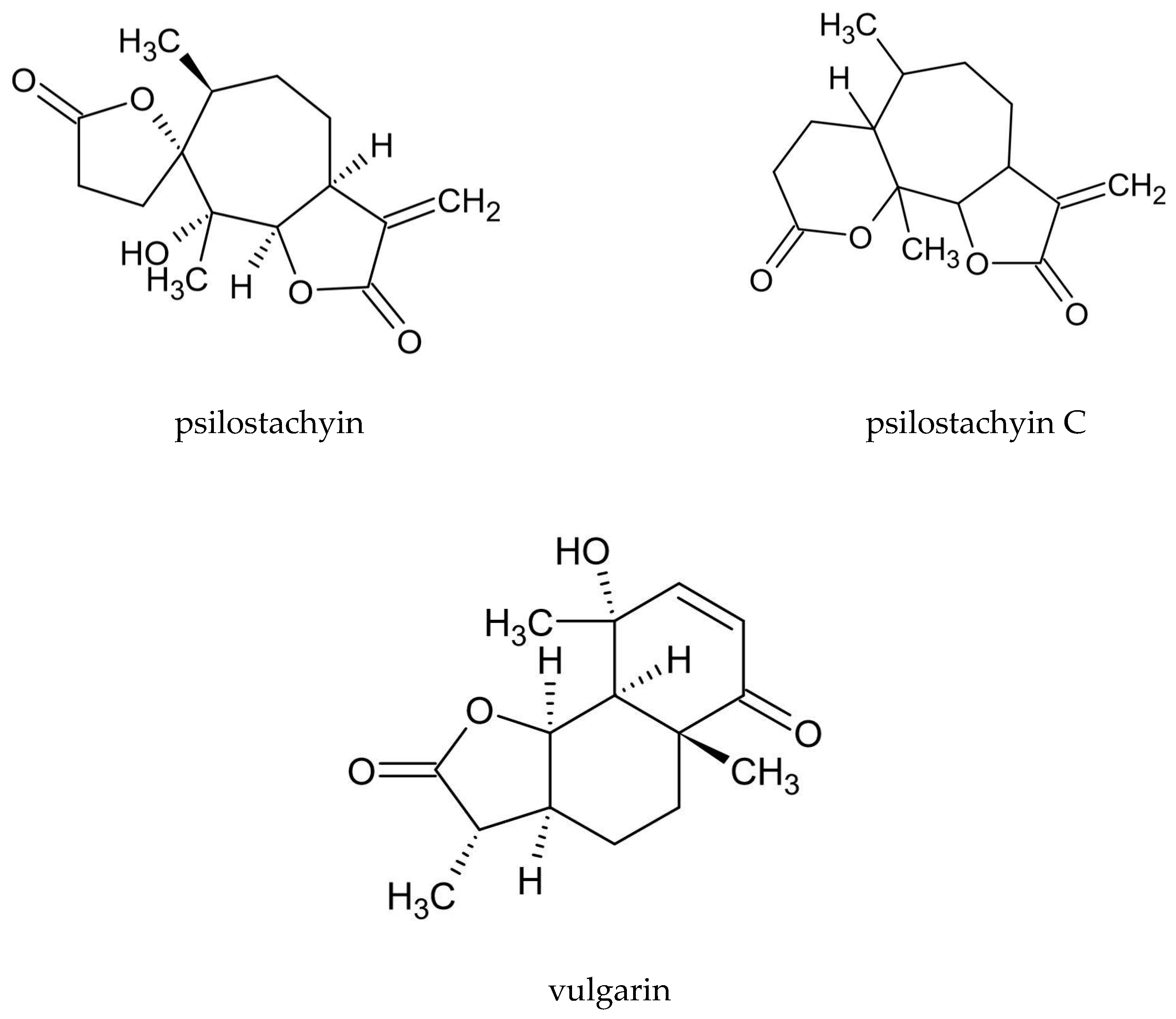
| Sesquiterpenoid lactones | 1,2,3,4-diepoxy-11(13)-eudesmen- 12,8-olide; yomogin | na * | [10] |
| psilostachyin, psilostachyin C | na | [3,54] | |
| vulgarin | 0.15% d.w. | [3,30,55] | |
| artemisinin | 0–2.3% dw. | [56,57] | |
| Flavonoids | quercetin 3-galactoside, quercetin 3-glucoside, kaempferol 3-glucoside, kaempferol 3-rhamnoside, kaempferol 3-rutinoside, kaempferol 7-glucoside, luteolin 7-glucoside | ~6 mg/kg d.w. ~11 mg/kg d.w. ~10 mg/kg d.w. ~5 mg/kg d.w. ~5 mg/kg d.w. ~5 mg/kg d.w. ~5 mg/kg d.w. | [13] |
| apigenin | ~6 mg/kg d.w. | [3,11,13,58] | |
| chrysoeriol | ~2.5 mg/kg d.w. | [13] | |
| eriodictyol, diosmetin, isorhamnetin | ~40 mg/kg d.w. ~5 mg/kg d.w. ~2.5 mg/kg d.w. | [3,13,58] | |
| eupafolin, homoeriodictyol | ~5 mg/kg d.w. ~10 mg/kg d.w. | [13] | |
| hyperoside | 0.5 mg/g d.w. | [3,58,59] | |
| jaceosidin | ~3 mg/kg d.w. | [13] | |
| quercetin | ~3 mg/kg d.w. | [3,11,30,58] | |
| luteolin | ~40 mg/kg d.w. | [3,13,58] | |
| rutoside | ~7–20 mg/kg d.w. | [11,13,59] | |
| tricine, | ~3 mg/kg d.w. | [13] | |
| vitexin | ~4 mg/kg d.w. | [3,58] | |
| Coumarins | esculin, esculetin, umbelliferone | na | [3,60] |
| Phenolic acids | 1,5-di-O-caffeoylquinic acid, 3,5-di-O-caffeoylquinic acid | 0.3% d.w. 0.2% d.w. | [61] |
| 5-feruloylquinic acid quinic acid protocatechuic acid glucoside 3-O-caffeoylquinic acid 5-O-caffeoylquinic acid 4,5-O-di-caffeoylquinic acid | 0.37 mg/g d.w. 1.5 mg/g d.w. 3.2 mg/g d.w. 0.44 mg/g d.w. 2.8 mg/g d.w. 11 mg/g d.w. | [59] | |
| caffeic acid | na | [3,11,61] | |
| Sterols | sitosterol, stigmasterol | [3] | |
| Fatty acids | na | 13.3 mg/g f.w. | [46] |
| Carotenoids | (E)-β-ionone | na | [3,62] |
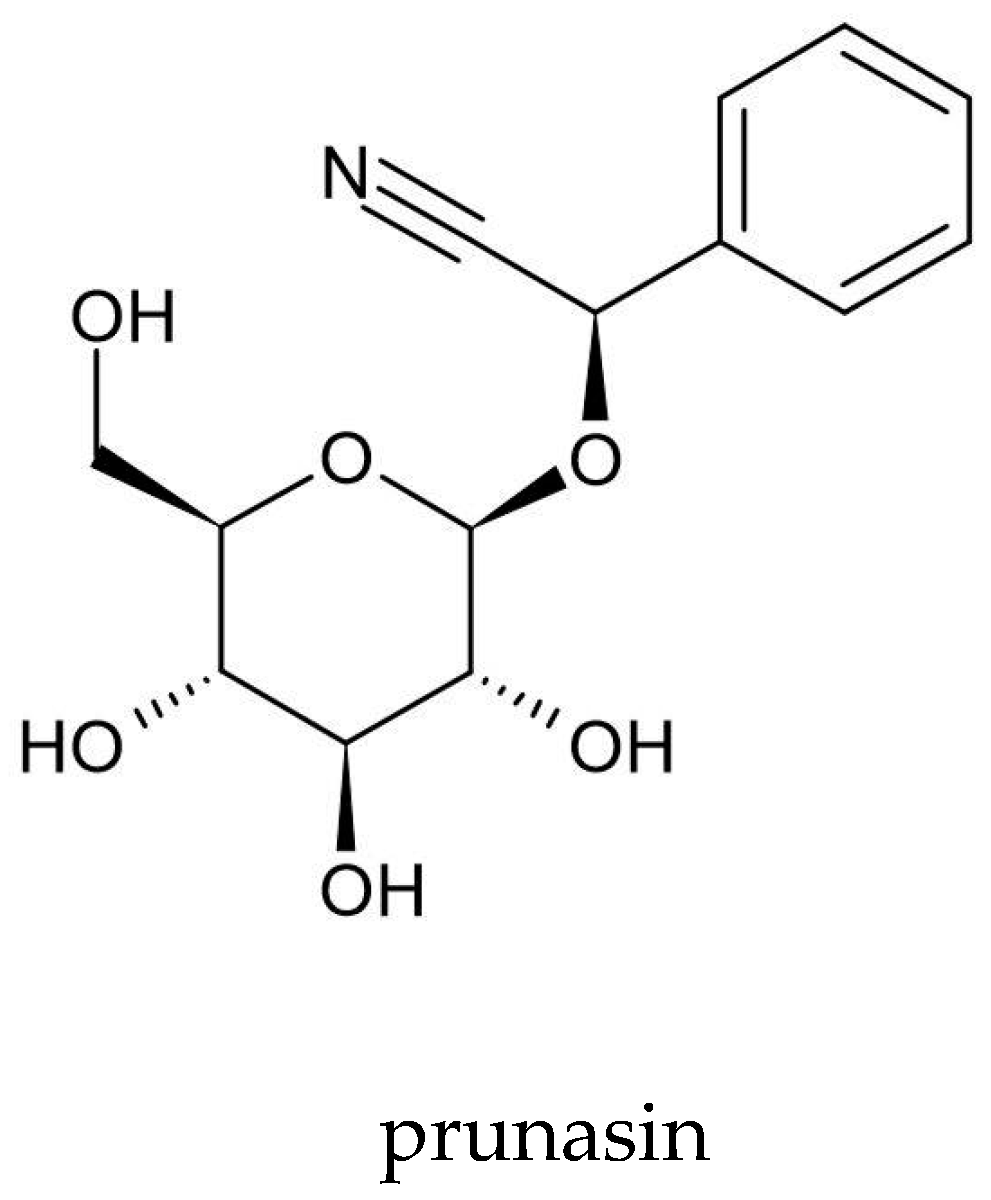
| Cyanogenic glycosides | prunasin | na | [3,63] |
| Vitamins | ascorbic acid | na | [30] |
| Polyacetylenes | na | na | [3,60] |
| Tannins | na | na | [11] |
| Chemical Groups/Compounds | Estimated Content (%) | References |
|---|---|---|
| Monoterpenoids | ||
| artemisia alcohol, artemisyl acetate, isobornyl acetate, (Z)-β-ocymen, terpinolene | 0–2.6 | [62] |
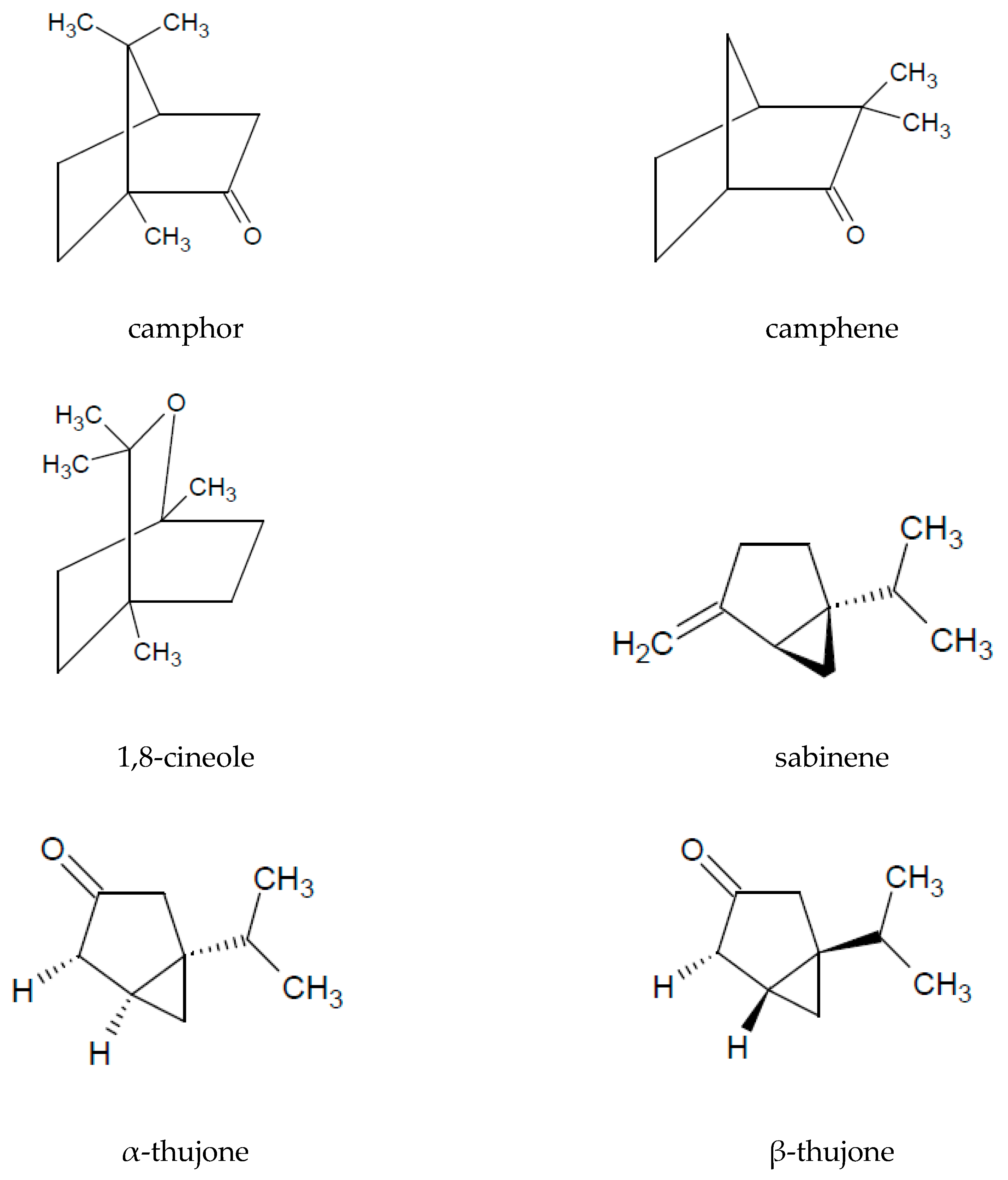
| artemisia ketone | 0–2.89 | [62,64,65] |
| borneol | 0.4–9.8 | [3,15,17,24,62,64,65,66,67] |
| bornyl acetate | 0–6.29 | [15,16,17,65,67] |
| camphene | 1.8–9.1 | [15,16,17,46,62,64,65] |
| camphor | 0–47.7 | [16,17,18,24,46,62,64,65,66,68,69] |
| carvone | 0–0.38 | [65] |
| trans-carveol, trans-pinocarveol | 0–0.77 | [15,64,65] |
| 1,8-cineol | 2.6–17.6 | [3,16,17,18,24,30,46,62,64,66,70] |
| cis-chrysanthenol, dehydrosabinaketone, methyleugenol, verbenyl acetate, p-cymene-8-ol, piperitone, p-mentha-1,4-dien-7-ol, sabinaketone, trans-verbenol, cuminol | 0–7.0 | [15] |
| cymene | 0–1.14 | [17,62,65] |
| isoborneol | 0.3–8.2 | [17,62,64] |
| isobornyl 2-methylbutyrate, menthol | 0–5 | [46] |
| iso-3-thujanol | 0–1.4 | [62,64,65] |
| limonene | 0–0.46 | [65] |
| (E)-β-ocymen | 0.5–2.7 | [62] |
| 3-thujanol | [17,64] | |
| 4-terpineol | 0–1.4 | [3,15,16,24,62,64,66] |
| cis-thujone | 0–12.9 | [46,62] |
| linalool | 0–0.4 | [3,62,64,66] |
| chrysanthenyl acetate | 0–23.6 | [15,46,62] |
| β-myrcene | 0.1–8.8 | [16,62] |
| sabinene | 0–0.67 | [15,16,17,62,64,65] |
| cis-sabinene hydrate, | 0–1.08 | [62,64,65] |
| trans-sabinene hydrate | 0–0.55 | [15,62,64,65] |
| santolina triene | 0–0.6 | [15,16,17,62,64] |
| α-thujone | 0–3.18 | [15,16,17,24,46,64,71] |
| β-thujone | 0–1.19 | [15,16,17,24,46,64,68,71,72] |
| α-fenchen | [16,17,64] | |
| α-pinene | 0–0.9 | [15,16,46,62,64,65] |
| α-terpinene | 0–0.4 | [62,64] |
| α-terpineol | 0–1.6 | [15,24,62] |
| α-thujene | 0.2–4.1 | [15,16,62] |
| β-pinene | 0.1–12.9 | [15,17,24,46,62,64] |
| γ-terpinene | 0–0.54 | [16,62,64,65] |
| thymol | 0–0.39 | [65] |
| Sesquiterpenoids | ||
| aromadendrene | 0–0.2 | [16,64] |
| bicyclogermacrene | 0.9–2.2 | [16,17,62,64] |
| α-cadinol | 0–1.99 | [3,17,66,67] |
| caryophyllene | 0–37.45 | [24,46,62,67] |
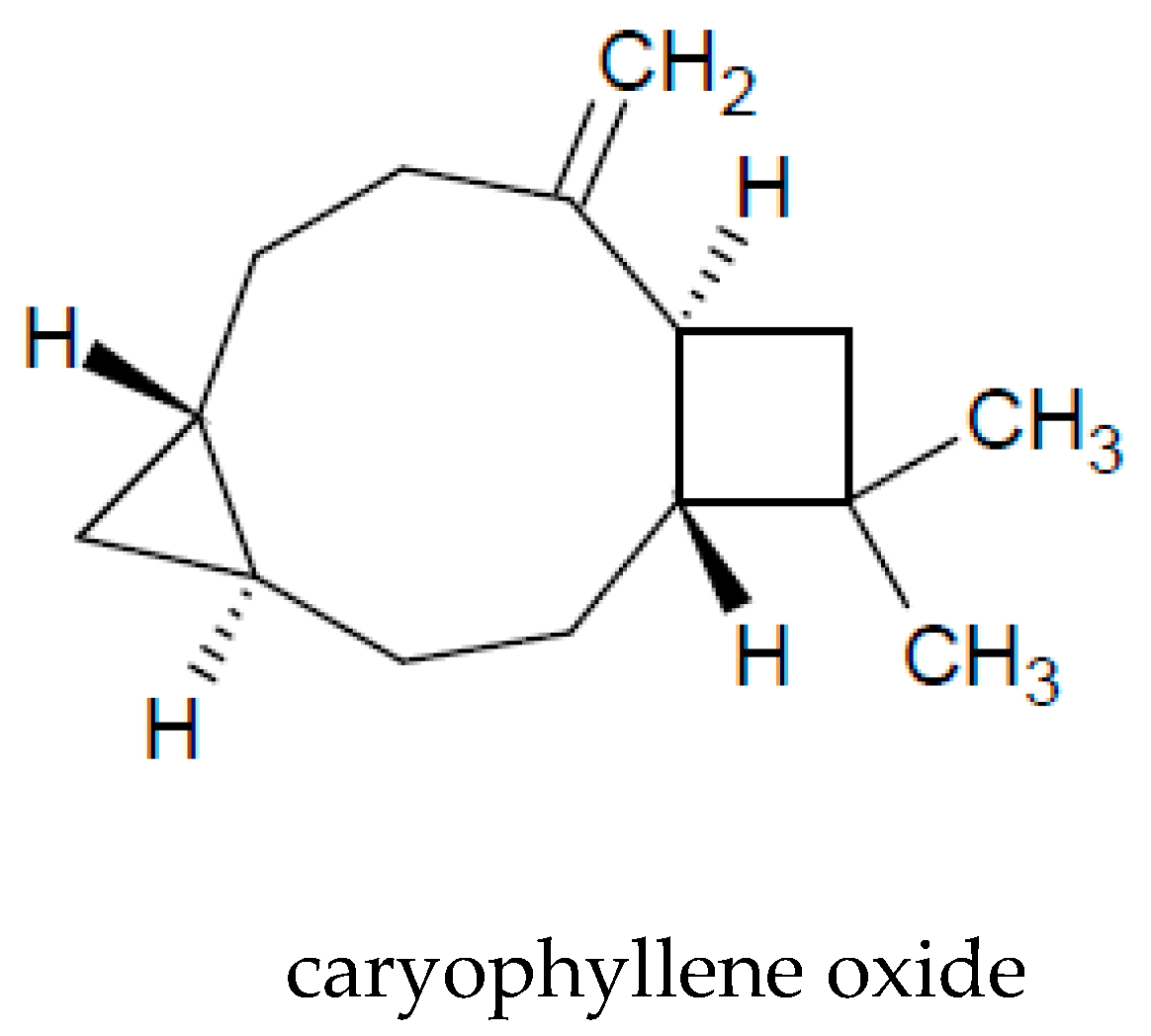
| caryophyllene oxide | 1.52–5.5 | [3,15,16,17,46,62,64,65,66] |
| trans-caryophyllene, trans-salvene | 2.5–12.2 | [46,65] |
| caryophylla-4(14),8(15)-diene-5-α-ol, (E)-nerolidol, humulene epoxide II, germacrene d-4-ol, ledol, farnesyl acetate, lanceol acetate, salvial-4(14)-en-1-one, silphiperfol-5-en-3-ol (Z)- β-farnesene, α-calacorene, β-chamigrene, β-longipinene | 0–0.5 | [62,67] |
| α-copaen | 0–1.0 | [15,16,17,62,64] |
| cubebene | 0–12 | [3] |
| davanone, silphiperfol-4,7(14)-diene, β-burbonen | 0–0.15 | [62,64] |
| β-elemene | 0–8 | [17], [64] |
| β-eudesmol | 0–8.95 | [16,46,64,65] |
| α-elemene, β-bisabolene | 0–8.8 | [16,62] |
| farnesene | 0–0.88 | [15,65] |
| germacrene D | 5.3–15.1 | [15,46,62,64] |
| α-humulene | 0.2–8.8 | [16,62,64,65] |
| epi-α-muurolol | 0.4–1.4 | [15,62] |
| spathulenol | 1–2.5 | [15,17,46,62] |
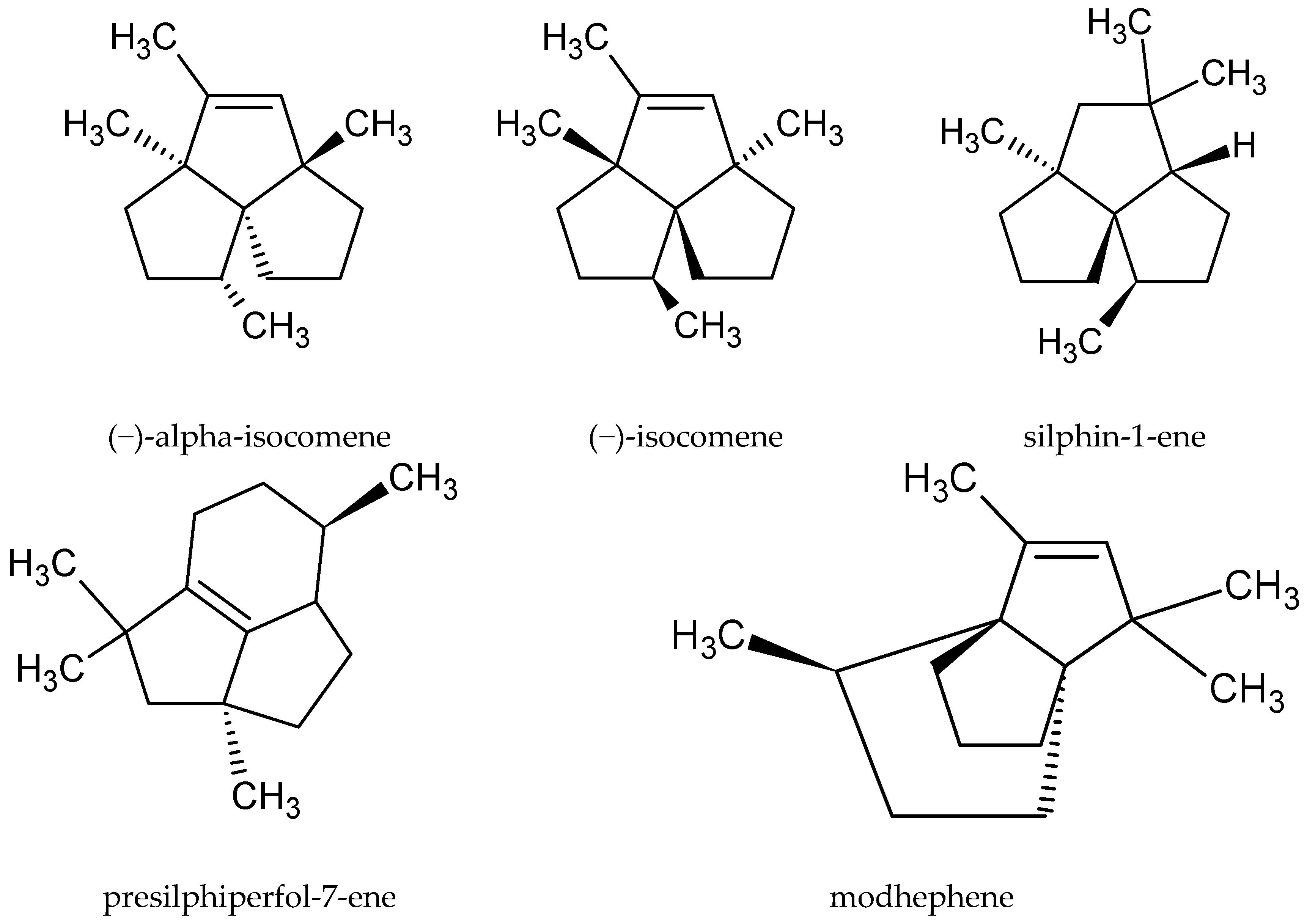
| 7-α-silphiperfol-5-ene, epi-β-santalene, modhephene, petasitene, presilphiperfol-7-ene, silphin-1-ene, valeranone, humulene oxide, α-bisabololene, α-cedrene, α-isocomene, α-trans-bergamotene | 0–0.5 | [16] |
| Diterpenoids | ||
| phytol | 0–2.94 | [62,67] |
| γ-terpineol | 0–1.44 | [65] |
| Origin of Plants | Main Components | References |
|---|---|---|
| Brazil | caryophyllene (37.45%) germacrene D (16.17%) humulene (13.66%) borneol (6.80%) caryophyllene oxide (5.67%) | [67] |
| France | camphor (1–13%) 1,8-cineole (1–23%) terpinen-4-ol (1–19%) | [73] |
| Germany | sabinene (16%) myrcene (14%) 1,8-cineole (10%) | [74] |
| India | α-thujone and thujone isomer (β-thujone) camphor | [75] |
| Italy | camphor (47%) | [76] |
| camphor (2–20%) myrcene (9–70%) 1,8-cineole (1–27%) borneol (3–18%) | [77] | |
| Lithuania (North part) | sabinene (0–8.4%) β-pinene (0.1–12.9%) 1,8-cineole (2.6–17.6%) cis-thujone (0–12.9%) trans-thujone (0–20.2%) chrysanthenyl acetate (0–23.6%) caryophyllene (2.5–12.2%) germacrene D (5.3–15.1%) | [62] |
| Morocco | thujone/isothujone camphor | [78] |
| Vietnam | β-caryophyllene (24%) β-cubebene (12%) β-elemene (6%) | [79] |
| 1,8-cineole camphor α-terpineol | [80] |
| Activity * | Information | Compounds Supposed to be Responsible | References |
|---|---|---|---|
| Antioxidant | Proved by different methods: DPPH, lipid peroxidation, protein glycation, xanthine oxidases, ABTS, hydroxyl superoxide, nitric oxide, ferric reducing power activity and inhibition of lipid peroxidation by thiobarbituric acid reactive species assays. Increasing the level of ascorbic acid and glutathione. | flavonoids, flavonols, phenolic acids | [8,59,101,102,103] |
| Hypolipemic | Normalized serum lipid profile, a significant increase in paraoxonase-1 activity and decrease in serum malondialdehyde, nitric oxide, tumor necrosis factor-α level and decrease in hydroxymethylglutaryl-CoA reductase activity. Lowering total cholesterol, triglycerides, LDL, VLDL, and increasing HDL and atherogenicity indicator (aqueous extract of A. vulgaris roots) *. | [9,104] | |
| Hepatoprotective | Prophylactic protective effect limiting inflammation, cellular oedema, apoptotic cell count, and hyperaemia of the hepatic parenchyma. | [105] | |
| Antispasmolytic | Antagonism towards H1 histamine receptors. | [10,11] | |
| Bronchodilatory | Anticholinergic and Ca2+ antagonist mechanisms. Histamine H1 antagonism in the ileum and trachea. | yomogin (sesquiterpene lactone), alkaloids, coumarins, flavonoids, saponins, sterols, tannins, terpenoids | [10,106] |
| Analgesic | Mild peripheral anti-nociceptive effect. | probably induced by rutoside, hydroxybenzoic acid derivatives, and caffeic acid and its derivatives | [11] |
| MAO inhibition | Inhibition of mouse brain monoamine oxidase (MAO) enzyme. | flavonoids: jaceosidine, eupafolin, luteolin, quercetin, apigenine; coumarins: esculetin, esculetin-6-methylether, scopoletin | [107] |
| Antihypertensive | Inhibiting the hypertensive effect of noradrenaline. Moxibustion showed lowering the blood pressure compared to antihypertensive drugs by stimulation of acupoint KI 1. | na ** Moxibustion—a traditional Chinese method that uses the heat generated by burning herbal preparations containing A. vulgaris to stimulate acupuncture points | [12,108] |
| Estrogenic | Antagonism towards the estrogen receptor and activation of gene transcription. Induction of gene transcription by eriodictyol and apigenin. Anti-implantation activity and estrogenic activity on female Wistar rats. | flavonoids | [13,109] |
| Cytotoxic | Inhibition of tumour cell growth in cancer cell lines: MCF7, HeLa, A7R5, 293T, HL-60 and SW-480. | phenolic compounds, flavonoids, essential oil | [14,110,111] |
| Antifungal and antibacterial | Inhibitory effect of the oil fraction on the development of Candida albicans. Inhibitory effect of the oil fraction on the development of Escherichia coli, Salmonella enteritidis, Pseudomonas aeruginosa, Klebsiella pneumoniae, Staphylococcus aureus, Streptococcus mutans, Candida albicans, and Aspergillus niger. | probably associated with the presence of essential oils, 1,8-cineole, α-thujone and camphene | [15,16,67,102,112,113,114] |
| Anti-inflammatory | Normalization of serum lipid profile, increase in paraoxonase-1 activity and decrease in serum malondialdehyde, nitric oxide and tumor necrosis factor-α level. Proved by lipoxygenase (LOX) inhibitory activity assay and “Cotton Pellet Granuloma Method”. | na | [103,104,115] |
| Antialergenic | Decrease in skin sensitivity and eye sensitivity. | na | [116] |
| Antimalarial | Activity angainst Plasmodium yoelii and P. berghei. | na | [117,118] |
| Anthelmintic | Activity against Trichinella spiralis. | na | [119] |
| Insect Species | Pathogenity | Raw Material of A. Vulgaris | References |
|---|---|---|---|
| Aedes aegypti (Egyptian mosquito) | denge fever virus vector | essential oil from the leaves | [122,123] |
| essential oil from the herb | [17] | ||
| nanoparticles with leaf extract | [123] | ||
| Culex quinquefasciatus Say. (Culex fatigans) (arbo virus) | vector of avian malaria, vector of wuchereria bancrofti | leaves extracts | [124] |
| stem extracts | |||
| root extracts | |||
| Tribolium castaneum (Herbst) | stored–product insect pests | essential oil from leaves | [126] |
| essential oil from aerial parts | [125] | ||
| Callosobruchus maculatus (F.) | stored–product beetles | essential oil from aerial parts | [125] |
| Rhyzopertha dominica | stored–product beetles | essential oil from aerial parts | [125] |
| Name in CosIng Database | Functions |
|---|---|
| Artemisia vulgaris extract | skin conditioning |
| Artemisia vulgaris herb extract | perfuming |
| Artemisia vulgaris oil | perfuming, skin conditioning |
| Bacillus/ Artemisia vulgaris extract/ferment filtrate | skin conditioning |
| hydrolyzed Artemisia vulgaris leaf | humectant, skin conditioning |
| Lactobacillus/Chrysanthemum zawadskii flower/Gleditsia japonica fruit/Thuja orientalis leaf/Morus alba bark/Panax ginseng root/Artemisia vulgaris extract ferment filtrate | skin conditioning |
| Bacillus/Cinnamomum cassia bark/Momordica charantia/Opuntia humifusa fruit/Aloevera/Artemisia vulgaris/Camellia sinensis/Nelum bonucifera leaf/Pueraria thunbergiana root ferment filtrate | antioxidant, skin protecting |
| Saccharomyces/Aloe barbadensis leaf/Artemisia vulgaris leaf/Prunus mume fruit ferment filtrate | skin conditioning |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ekiert, H.; Pajor, J.; Klin, P.; Rzepiela, A.; Ślesak, H.; Szopa, A. Significance of Artemisia Vulgaris L. (Common Mugwort) in the History of Medicine and Its Possible Contemporary Applications Substantiated by Phytochemical and Pharmacological Studies. Molecules 2020, 25, 4415. https://doi.org/10.3390/molecules25194415
Ekiert H, Pajor J, Klin P, Rzepiela A, Ślesak H, Szopa A. Significance of Artemisia Vulgaris L. (Common Mugwort) in the History of Medicine and Its Possible Contemporary Applications Substantiated by Phytochemical and Pharmacological Studies. Molecules. 2020; 25(19):4415. https://doi.org/10.3390/molecules25194415
Chicago/Turabian StyleEkiert, Halina, Joanna Pajor, Paweł Klin, Agnieszka Rzepiela, Halina Ślesak, and Agnieszka Szopa. 2020. "Significance of Artemisia Vulgaris L. (Common Mugwort) in the History of Medicine and Its Possible Contemporary Applications Substantiated by Phytochemical and Pharmacological Studies" Molecules 25, no. 19: 4415. https://doi.org/10.3390/molecules25194415
APA StyleEkiert, H., Pajor, J., Klin, P., Rzepiela, A., Ślesak, H., & Szopa, A. (2020). Significance of Artemisia Vulgaris L. (Common Mugwort) in the History of Medicine and Its Possible Contemporary Applications Substantiated by Phytochemical and Pharmacological Studies. Molecules, 25(19), 4415. https://doi.org/10.3390/molecules25194415








