Impact of Different Stoppers on the Composition of Red and Rosé Lagrein, Schiava (Vernatsch) and Merlot Wines Stored in Bottle
Abstract
1. Introduction
2. Results
2.1. Evolution of Phenolic Compounds
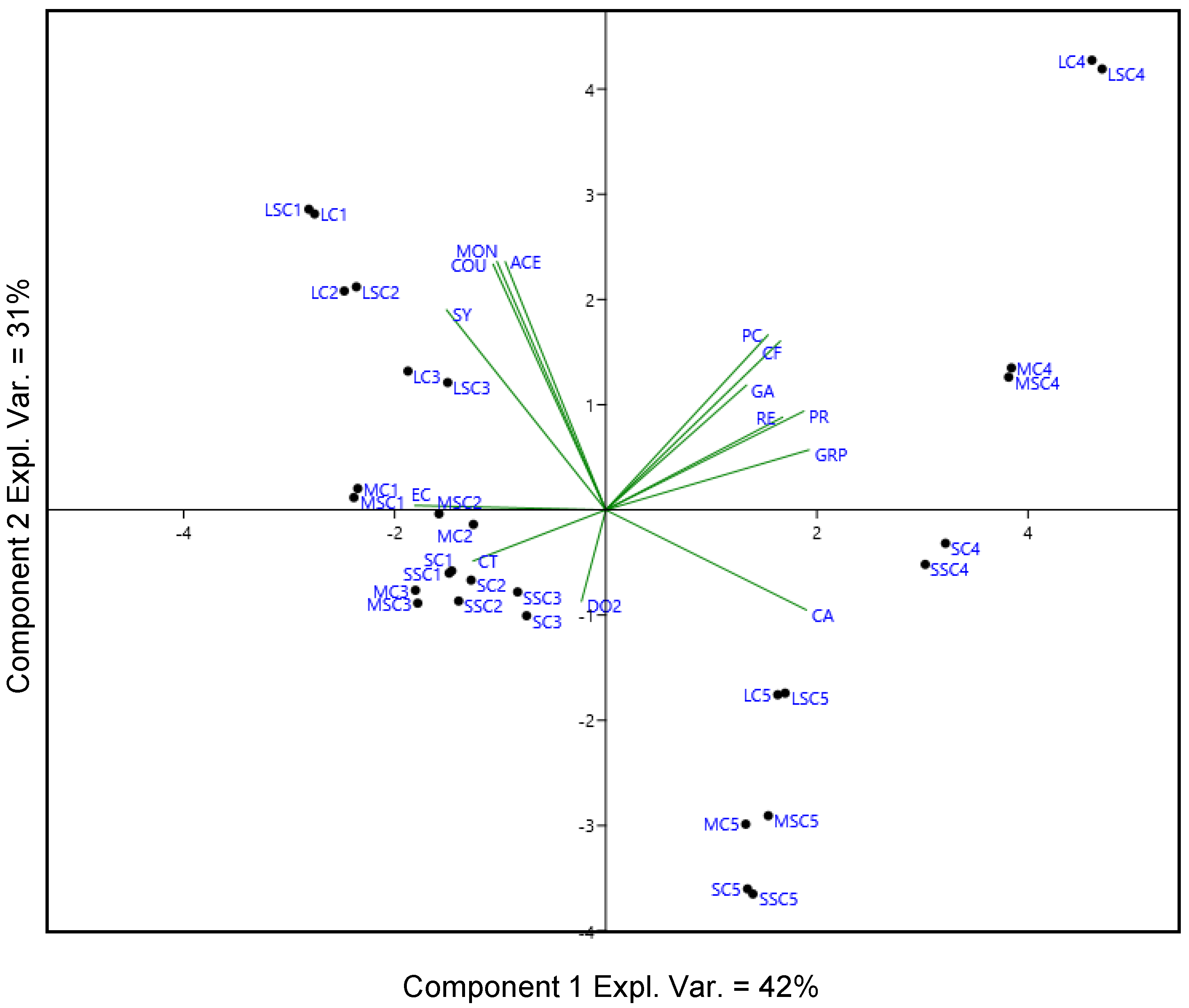
2.2. Effect of the Type of Stopper and Storage Time on the Phenolic Profile
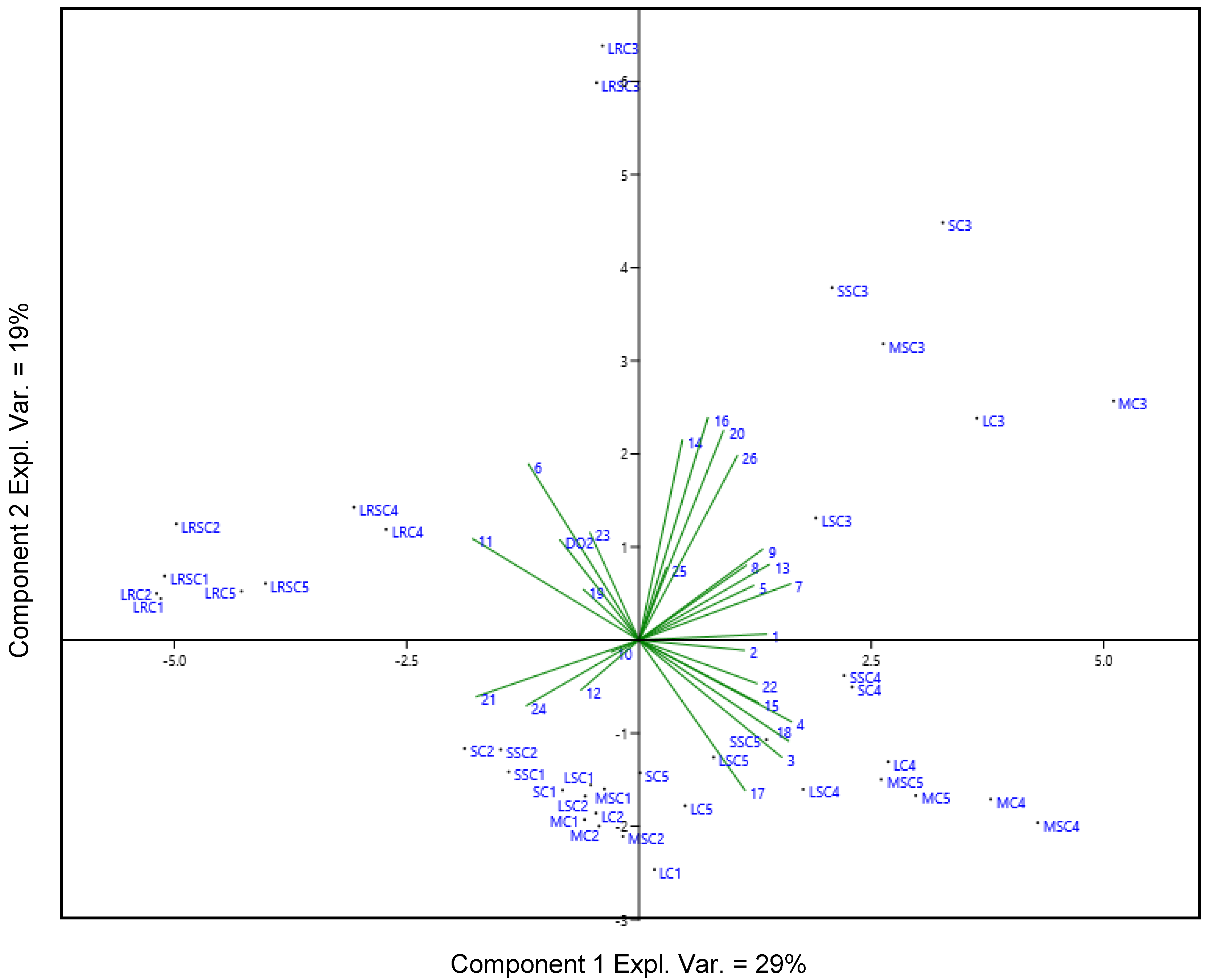
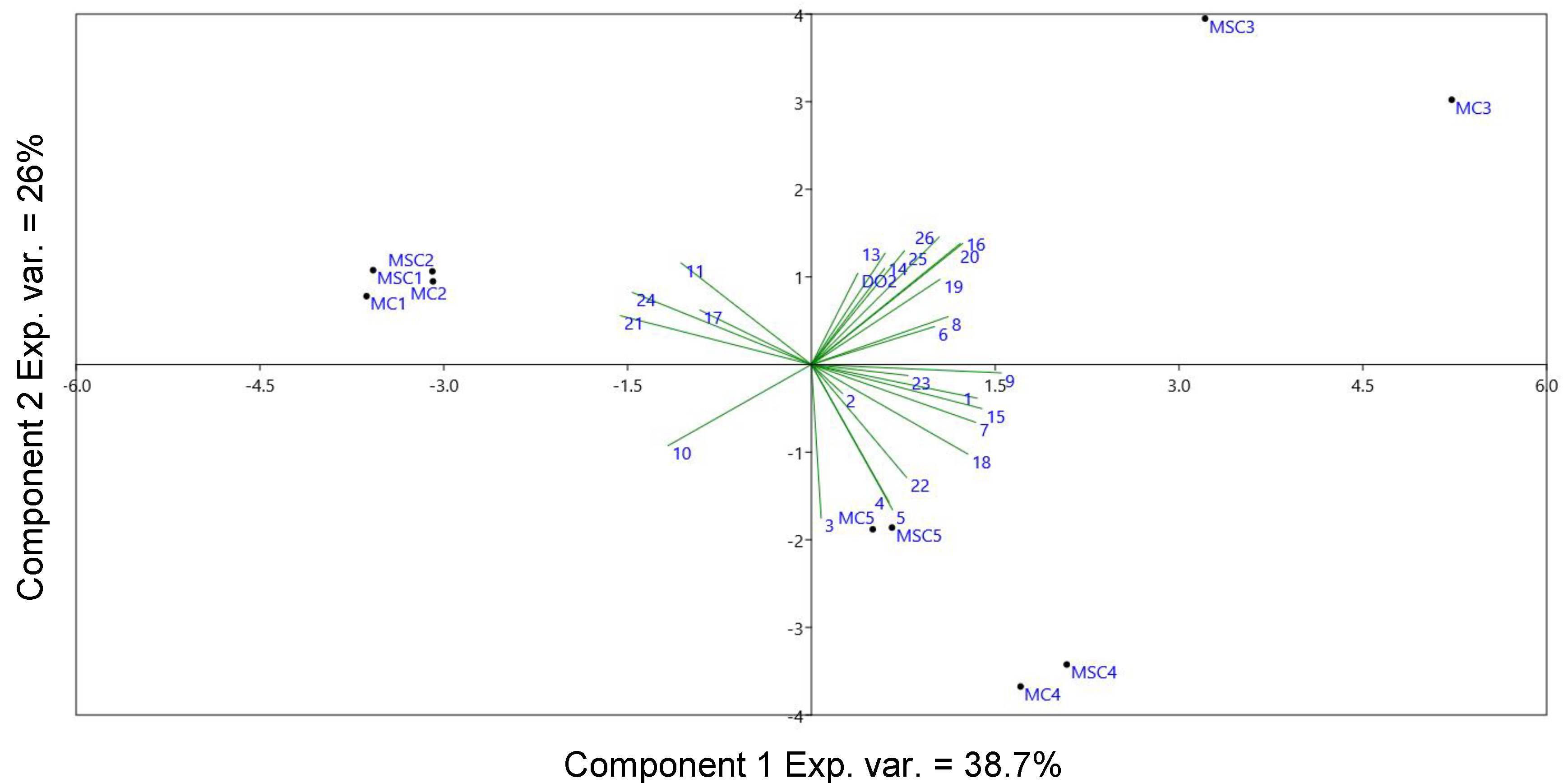
2.3. Evolution of Volatile Compounds during the Storage in Bottle
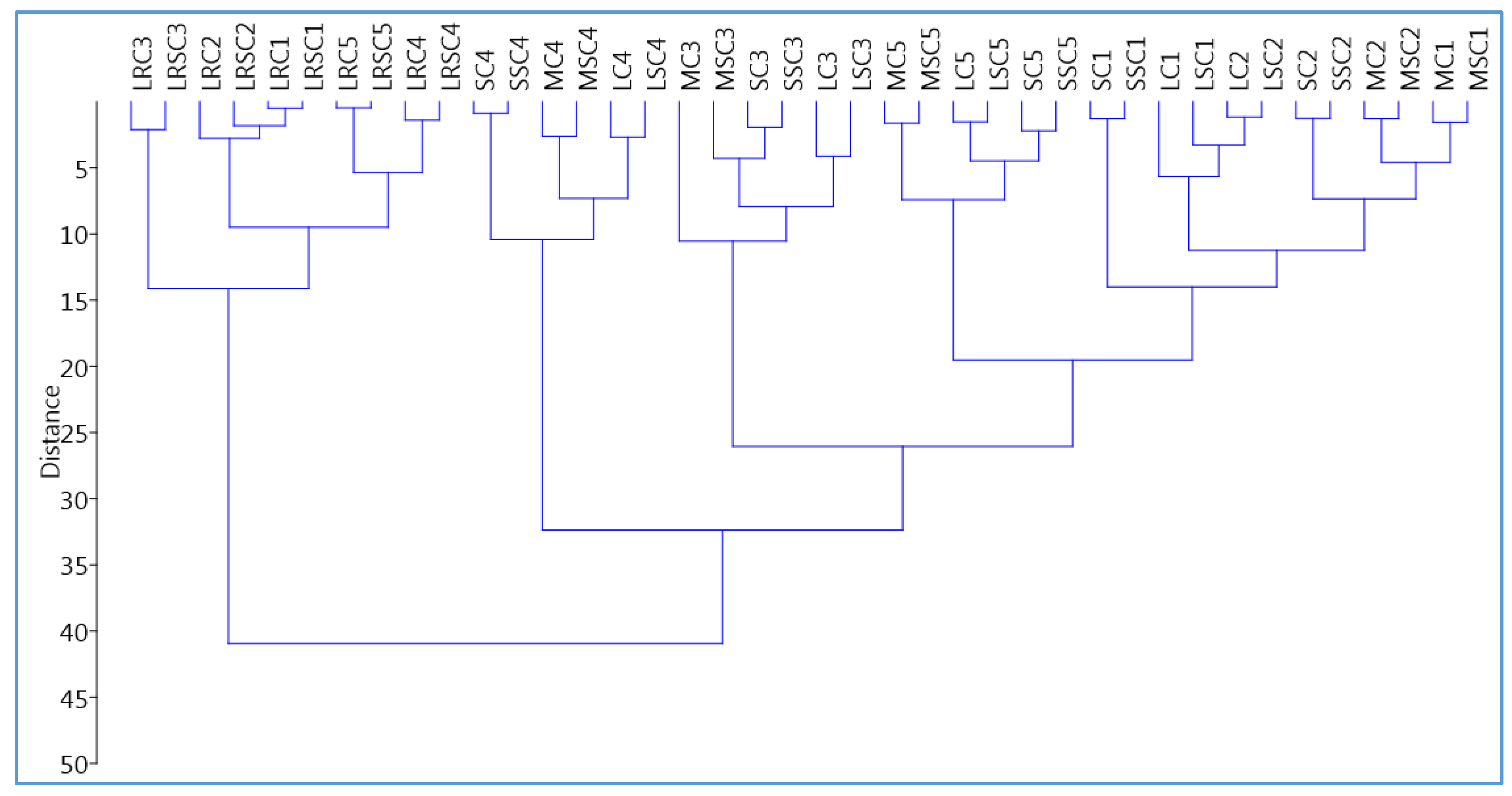
2.4. Hierarchical Clustering Analysis (HCA)
2.5. Sensory Analysis
3. Discussion
4. Materials and Methods
4.1. Wine Samples and Stoppers
4.2. Chemicals and Reagents
4.3. Non-Anthocyanin Phenolic Compounds
4.4. Anthocyanins
4.5. Volatile Compounds
4.6. Dissolved Oxygen Content
4.7. Sensory Analysis
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ribéreau-Gayon, P.; Glories, Y.; Maujean, A.; Dubourdieu, D. Handbook of Enology—The Chemistry of Wine Stabilization and Treatments; John Wiley & Sons, Ltd.: Chichester, UK, 2006. [Google Scholar]
- Skouroumounis, G.; Kwiatkowski, M.; Francis, I.; Oakey, H.; Capone, D.; Duncan, B.; Sefton, M.; Waters, E. The impact of closure type and storage conditions on the composition, colour and flavour properties of a Riesling and a wooded Chardonnay wine during five years’ storage. Aust. J. Grape Wine Res. 2005, 11, 369–377. [Google Scholar] [CrossRef]
- Puškaš, V.; Miljić, U.D. The application of D-optimal design for modelling the red wine ageing process. Food Control. 2012, 28, 362–367. [Google Scholar] [CrossRef]
- Wirth, J.; Morel-Salmi, C.; Souquet, J.M.; Dieval, J.; Aagaard, O.; Vidal, S.; Fulcrand, H.; Cheynier, V. The impact of oxygen exposure before and after bottling on the polyphenolic composition of red wines. Food Chem. 2010, 123, 107–116. [Google Scholar] [CrossRef]
- Somers, T. The polymeric nature of wine pigments. Phytochemistry 1971, 10, 2175–2186. [Google Scholar] [CrossRef]
- Escudero, A.; Asensio, E.; Cacho, J.; Ferreira, V. Sensory and chemical changes of young white wines stored under oxygen. An assessment of the role played by aldehydes and some other important odorants. Food Chem. 2002, 77, 325–331. [Google Scholar] [CrossRef]
- Mozzon, M.; Savini, S.; Boselli, E.; Thorngate, J. The herbaceous character of wines. Italy J. Food Sci. 2016, 28, 190–207. [Google Scholar]
- De Souza, J.F.; Nascimento, A.M.D.S.; Linhares, M.D.S.S.; Dutra, M.D.C.P.; Lima, M.D.S.; Pereira, G.E. Evolution of Phenolic Compound Profiles and Antioxidant Activity of Syrah Red and Sparkling Moscatel Wines Stored in Bottles of Different Colors. Beverages 2018, 4, 89. [Google Scholar] [CrossRef]
- Lopes, P.; Saucier, C.; Teissedre, P.-L.; Glories, Y. Impact of storage position on oxygen ingress through different closures into wine bottles. J. Agric. Food Chem. 2006, 54, 6741–6746. [Google Scholar] [CrossRef]
- Lopes, P.; Saucier, C.; Teissedre, P.-L.; Glories, Y. Main Routes of Oxygen Ingress through Different Closures into Wine Bottles. J. Agric. Food Chem. 2007, 55, 5167–5170. [Google Scholar] [CrossRef]
- Diéval, J.-B.; Vidal, S.; Aagaard, O. Measurement of the Oxygen Transmission Rate of Co-extruded Wine Bottle Closures Using a Luminescence-Based Technique. Packag. Technol. Sci. 2011, 24, 375–385. [Google Scholar] [CrossRef]
- Godden, P.; Francis, L.; Field, J.; Gishen, M.; Coulter, A.; Valente, P.; Høj, P.; Robinson, E. Wine bottle closures: Physical characteristics and effect on composition and sensory properties of a Semillon wine 1. Performance up to 20 months post-bottling. Aust. J. Grape Wine Res. 2001, 7, 64–105. [Google Scholar] [CrossRef]
- Boselli, E.; Di Lecce, G.; Alberti, F.; Frega, N.G. Nitrogen gas affects the quality and the phenolic profile of must obtained from vacuum-pressed white grapes. LWT 2010, 43, 1494–1500. [Google Scholar] [CrossRef]
- Day, M.; Schmidt, S.A.; Smith, P.; Wilkes, E. Use and impact of oxygen during winemaking. Aust. J. Grape Wine Res. 2015, 21, 693–704. [Google Scholar] [CrossRef]
- Trivellin, N.; Barbisan, D.; Badocco, D.; Pastore, P.; Meneghesso, G.; Meneghini, M.; Zanoni, E.; Belgioioso, G.; Cenedese, A. Study and Development of a Fluorescence Based Sensor System for Monitoring Oxygen in Wine Production: The WOW Project. Sensors 2018, 18, 1130. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.; Lambri, M. Oxygen measures and consumption in must and wine. Anal. Chim. Acta 2006, 563, 391–395. [Google Scholar] [CrossRef]
- Crouvisier-Urion, K.; Bellat, J.-P.; Gougeon, R.D.; Karbowiak, T. Gas transfer through wine closures: A critical review. Trends Food Sci. Technol. 2018, 78, 255–269. [Google Scholar] [CrossRef]
- Silva, M.A.; Julien, M.; Jourdes, M.; Teissedre, P.-L. Impact of closures on wine post-bottling development: A review. Eur. Food Res. Technol. 2011, 233, 905–914. [Google Scholar] [CrossRef]
- Gao, Y.; Tian, Y.; Liu, D.; Li, Z.; Zhang, X.-X.; Li, J.; Huang, J.-H.; Wang, J.; Pan, Q.-H. Evolution of phenolic compounds and sensory in bottled red wines and their co-development. Food Chem. 2015, 172, 565–574. [Google Scholar] [CrossRef]
- Xing, R.-R.; Liu, D.; Li, Z.; Tian, Y.; Zhang, X.-X.; Li, J.-M.; Pan, Q.-H. Impact of different types of stoppers on sensorial and phenolic characteristics evolution during a bottle storage time of a white wine from Chardonnay grape variety. J. Food Sci. Technol. 2016, 53, 4043–4055. [Google Scholar] [CrossRef]
- Sáenz-Navajas, M.-P.; Avizcuri, J.-M.; Ferreira, V.; Fernandez-Zurbano, P. Sensory changes during bottle storage of Spanish red wines under different initial oxygen doses. Food Res. Int. 2014, 66, 235–246. [Google Scholar] [CrossRef]
- Ugliano, M. Oxygen Contribution to Wine Aroma Evolution during Bottle Aging. J. Agric. Food Chem. 2013, 61, 6125–6136. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Xing, R.-R.; Li, Z.; Yang, D.-M.; Pan, Q.-H. Evolution of volatile compounds, aroma attributes, and sensory perception in bottle-aged red wines and their correlation. Eur. Food Res. Technol. 2016, 242, 1937–1948. [Google Scholar] [CrossRef]
- Castellari, M.; Matricardi, L.; Arfelli, G.; Galassi, S.; Amati, A. Level of single bioactive phenolics in red wine as a function of the oxygen supplied during storage. Food Chem. 2000, 69, 61–67. [Google Scholar] [CrossRef]
- Cheynier, V.; Rigaud, J.; Souquet, J.; Duprat, F.; Moutounet, M. Must browning in relation to the behaviour of phenolic compounds during oxidation. Am. J. Enol. Vitic. 1990, 41, 346–349. [Google Scholar]
- Gómez-Plaza, E.; Gil-Muñoz, R.; López-Roca, J.; Martínez-Cutillas, A.; Fernández-Fernández, J. Maintenance of Colour Composition of a Red Wine during Storage. Influence of Prefermentative Practices, Maceration Time and Storage. LWT 2002, 35, 46–53. [Google Scholar] [CrossRef]
- Zafrilla, P.; Morillas, J.; Mulero, J.; Cayuela, J.M.; Martínez-Cachá, A.; Pardo, F.; López-Nicolás, J. Changes during Storage in Conventional and Ecological Wine: Phenolic Content and Antioxidant Activity. J. Agric. Food Chem. 2003, 51, 4694–4700. [Google Scholar] [CrossRef] [PubMed]
- Klesk, K.; Qian, M. Aroma Extract Dilution Analysis of Cv. Marion (Rubusspp.hyb) and Cv. Evergreen (R. laciniatus L.) Blackberries. J. Agric. Food Chem. 2003, 51, 3436–3441. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Shin, J.H.; Yu, E.J.; Kim, Y.-S.; Lee, H.J.; Kim, T.H. Characteristics of aroma-active compounds in the pectin-elicited suspension culture of Zanthoxylum piperitum (prickly ash). Biotechnol. Lett. 2002, 24, 551–556. [Google Scholar] [CrossRef]
- Beaulieu, J.C.; Grimm, C.C. Identification of volatile compounds in cantaloupe at various developmental stages using solid phase microextraction. J. Agric. Food Chem. 2001, 49, 1345–1352. [Google Scholar] [CrossRef]
- Xu, X.; Stee, L.L.P.; Williams, J.; Beens, J.; Adahchour, M.; Vreuls, R.J.J.; Brinkman, U.A.; Lelieveld, J. Comprehensive two-dimensional gas chromatography (GC × GC) measurements of volatile organic compounds in the atmosphere. Atmos. Chem. Phys. Discuss. 2003, 3, 665–682. [Google Scholar] [CrossRef]
- Schirack, A.; Drake, M.; Sanders, T.; Sandeep, K. Characterization of Aroma-Active Compounds in Microwave Blanched Peanuts. J. Food Sci. 2006, 71, C513–C520. [Google Scholar] [CrossRef]
- Su, Y.-C.; Ho, C.L.; Wang, E.I.-C.; Chang, S.-T. Antifungal activities and chemical compositions of essential oils from leaves of four eucalypts. Taiwan J. Sci. 2006, 21, 49–61. [Google Scholar]
- Flamini, G.; Cioni, P.L.; Morelli, I. Volatiles from Leaves, Fruits, and Virgin Oil fromOlea europaea Cv. Olivastra Seggianese from Italy. J. Agric. Food Chem. 2003, 51, 1382–1386. [Google Scholar] [CrossRef] [PubMed]
- Boulanger, R.; Crouzet, J. Free and bound flavour components of Amazonian fruits 3-glycosidically bound components of cupuacu. Food Chem. 2000, 70, 463–470. [Google Scholar] [CrossRef]
- Sampaio, T.S.; Nogueira, P.C.L. Volatile components of mangaba fruit (Hancornia speciosa Gomes) at three stages of maturity. Food Chem. 2006, 95, 606–610. [Google Scholar] [CrossRef]
- Passos, X.S.; Castro, A.C.M.; Pires, J.S.; Garcia, A.C.F.; Campos, F.C.; Fernandes, O.F.L.; Paula, J.R.; Ferreira, H.D.; Santos, S.; Ferri, P.H.; et al. Composition and Antifungal Activity of the Essential Oils of Caryocar brasiliensis. Pharm. Boil. 2003, 41, 319–324. [Google Scholar] [CrossRef]
- Moio, L.; Ugliano, M.; Genovese, A.; Gambuti, A.; Pessina, R.; Piombino, P. Effect of Antioxidant Protection of Must on Volatile Compounds and Aroma Shelf Life of Falanghina (Vitis vinifera L.) Wine. J. Agric. Food Chem. 2004, 52, 891–897. [Google Scholar] [CrossRef]
- Ugliano, M.; Siebert, T.; Mercurio, M.; Capone, D.; Henschke, P.A. Volatile and Color Composition of Young and Model-Aged Shiraz Wines as Affected by Diammonium Phosphate Supplementation Before Alcoholic Fermentation. J. Agric. Food Chem. 2008, 56, 9175–9182. [Google Scholar] [CrossRef]
- Culleré, L.; Cacho, A.J.; Ferreira, V. An Assessment of the Role Played by Some Oxidation-Related Aldehydes in Wine Aroma. J. Agric. Food Chem. 2007, 55, 876–881. [Google Scholar] [CrossRef]
- Escudero, A.; Hernández-Orte, P.; Cacho, J.; Ferreira, V.; Hernández-Orte, P. Clues about the Role of Methional As Character Impact Odorant of Some Oxidized Wines. J. Agric. Food Chem. 2000, 48, 4268–4272. [Google Scholar] [CrossRef]
- Fedrizzi, B.; Zapparoli, G.; Finato, F.; Tosi, E.; Turri, A.; Azzolini, M.; Versini, G. Model Aging and Oxidation Effects on Varietal, Fermentative, and Sulfur Compounds in a Dry Botrytized Red Wine. J. Agric. Food Chem. 2011, 59, 1804–1813. [Google Scholar] [CrossRef]
- Di Lecce, G.; Boselli, E.; D’Ignazi, G.; Frega, N.G. Evolution of phenolics and glutathione in Verdicchio wine obtained with maceration under reductive conditions. LWT 2013, 53, 54–60. [Google Scholar] [CrossRef]
- Ferreira, V.; Escudero, A.; Fernández, P.; Cacho, J. Changes in the profile of volatile compounds in wines stored under oxygen and their relationship with the browning process. Z. Lebensm. Forsch. A 1997, 205, 392–396. [Google Scholar] [CrossRef]
- Hernanz, D.; Gallo, V.; Fernández-Recamales, M.Á.; Meléndez-Martínez, A.J.; Gonzalez-Miret, M.; Heredia, F.J. Effect of storage on the phenolic content, volatile composition and colour of white wines from the varieties Zalema and Colombard. Food Chem. 2009, 113, 530–537. [Google Scholar] [CrossRef]
- Makhotkina, O.; Pineau, B.; Kilmartin, P.A. Effect of storage temperature on the chemical composition and sensory profile of Sauvignon Blanc wines. Aust. J. Grape Wine Res. 2012, 18, 91–99. [Google Scholar] [CrossRef]
- Wang, J.; Capone, D.L.; Wilkinson, K.; Jeffery, D.W. Rosé wine volatile composition and the preferences of Chinese wine professionals. Food Chem. 2016, 202, 507–517. [Google Scholar] [CrossRef]
- Chira, K. In Structures Moléculaires et Perception Tannique Des Raisins et Des Vins (Cabernet-Sauvignon, Merlot) du bordelais. Ph.D. Thesis, University of Bordeaux II, Bordeaux, France, 11 December 2009. Available online: http://www.sudoc.fr/14547027X (accessed on 16 September 2020).
- Paissoni, M.A.; Waffo-Teguo, P.; Ma, W.; Jourdes, M.; Rolle, L.; Teissedre, P.-L. Chemical and sensorial investigation of in-mouth sensory properties of grape anthocyanins. Sci. Rep. 2018, 8, 17098. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, F.; Caldeira, M.; Câmara, J. Development of a dynamic headspace solid-phase microextraction procedure coupled to GC-qMSD for evaluation the chemical profile in alcoholic beverages. Anal. Chim. Acta 2008, 609, 82–104. [Google Scholar] [CrossRef]
- Mozzon, M.; Foligni, R.; Mannozzi, C. Brewing Quality of Hop Varieties Cultivated in Central Italy Based on Multivolatile Fingerprinting and Bitter Acid Content. Foods 2020, 9, 541. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]

| Gallic Acid | Protocatechuic Acid | Caftaric Acid | GRP | (+)-Catechin | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time | c | Blend | F1 | F2 | F3 | c | Blend | F1 | F2 | F3 | c | Blend | F1 | F2 | F3 | c | Blend | F1 | F2 | F3 | c | Blend | F1 | F2 | F3 | |
| Lagrein Red | T1 | 16.2 ± 0.5 | 16.3 ± 0.1 | ns | 1932 * | ns | 1.4 ± 0 | 1.3 ± 0.1 | ns | 1143 * | ns | 19.9 ± 1.2 | 18.8 ± 0.7 | ns | 356.5 * | ns | 1 ± 0.1 | 1 ± 0.1 | ns | 626.7 * | ns | 23.7 ± 0.1 | 24.2 ± 2.5 | 14.1 * | 81.7 * | ns |
| T2 | 17 ± 0 | 18 ± 0.5 | ns | 1.2 ± 0.1 | 0.9 ± 0 | ns | 20.3 ± 0.2 | 23.7 ± 1.2 | ns | 1 ± 0.1 | 0.8 ± 0 | ns | 24.9 ± 1.2 | 24.6 ± 2.2 | ns | |||||||||||
| T3 | 18.7 ± 1.4 | 18.4 ± 0.1 | ns | 1.4 ± 0 | 2.2 ± 0.7 | ns | 22.1 ± 0.1 | 20.6 ± 0.8 | ns | 1 ± 0 | 1 ± 0.1 | ns | 23.9 ± 0.3 | 18 ± 0.4 | * | |||||||||||
| T4 | 47.6 ± 0.7 | 46.5 ± 0.1 | ns | 9.9 ± 0.1 | 10.3 ± 0.3 | ns | 41.5 ± 3.5 | 38.8 ± 0 | ns | 17.3 ± 1.8 | 16.3 ± 0.4 | ns | 20 ± 0.2 | 12.5 ± 0.5 | * | |||||||||||
| T5 | 21.7 ± 0.2 | 21.4 ± 0.4 | ns | 3.3 ± 0.1 | 3.5 ± 0.1 | ns | 47.5 ± 0.1 | 47.2 ± 0 | ns | 1.5 ± 0.1 | 1.5 ± 0.1 | ns | 15.7 ± 0.2 | 14.7 ± 0.5 | ns | |||||||||||
| Lagrein Rosè | T1 | 3.8 ± 0.1 | 4.1 ± 0.1 | ns | 751 * | ns | 0.5 ± 0 | 0.5 ± 0 | ns | 27 * | ns | 3.8 ± 0 | 3.8 ± 0.2 | ns | 786 * | ns | 1.4 ± 0 | 1.3 ± 0.1 | ns | 8570 * | ns | 7.1 ± 0.2 | 7.4 ± 0.6 | ns | 29 * | ns |
| T2 | 3.7 ± 0.2 | 4 ± 0.2 | ns | 0.4 ± 0 | 0.4 ± 0 | ns | 4.2 ± 0.2 | 4.5 ± 0.1 | ns | 1.3 ± 0.1 | 1.3 ± 0.1 | ns | 5.4 ± 0.4 | 5.6 ± 0.2 | ns | |||||||||||
| T3 | 4.6 ± 0.1 | 4.9 ± 0 | ns | 0.6 ± 0.1 | 0.5 ± 0 | ns | 3.7 ± 0.1 | 3.8 ± 0.1 | ns | 1.2 ± 0 | 1.2 ± 0 | ns | 5 ± 1.1 | 5 ± 0 | ns | |||||||||||
| T4 | 6 ± 0.1 | 5.9 ± 0.1 | ns | 0.6 ± 0.1 | 0.5 ± 0 | ns | 6.8 ± 0 | 6.8 ± 0 | ns | 8.2 ± 0 | 8.2 ± 0.1 | ns | 6.8 ± 0 | 6.8 ± 0 | ns | |||||||||||
| T5 | 1.6 ± 0.1 | 1.6 ± 0.1 | ns | 0.8 ± 0.1 | 0.8 ± 0.1 | ns | 7.1 ± 0.1 | 7.1 ± 0.1 | ns | 4.7 ± 0 | 4.7 ± 0 | ns | 4.5 ± 0.1 | 4.2 ± 0.1 | ns | |||||||||||
| Merlot | T1 | 19.9 ± 0.2 | 21.1 ± 0.7 | ns | 1526 * | ns | 1.7 ± 0.1 | 1.7 ± 0.2 | ns | 534 * | ns | 18.1 ± 0.2 | 17.5 ± 1.1 | ns | 292.4 * | ns | 1.5 ± 0.3 | 1.4 ± 0.2 | ns | 3263 * | ns | 35 ± 2 | 37.8 ± 2.4 | ns | 21.56 * | ns |
| T2 | 24.1 ± 0.6 | 26 ± 1 | ns | n.d. | n.d. | ns | 21.4 ± 0.5 | 23.4 ± 0.2 | ns | 1 ± 0.1 | 1.2 ± 0 | ns | 39.3 ± 0.3 | 38 ± 6.7 | ns | |||||||||||
| T3 | 26.8 ± 0.4 | 26.8 ± 0.2 | ns | 1.8 ± 0.3 | 1.9 ± 0.1 | ns | 20.3 ± 1.5 | 20.2 ± 0.4 | ns | 1.1 ± 0 | 1.1 ± 0 | ns | 37.7 ± 0.7 | 37.1 ± 5 | ns | |||||||||||
| T4 | 59.9 ± 0.5 | 59.3 ± 0.2 | ns | 4.8 ± 0.1 | 4.8 ± 0.1 | ns | 35.7 ± 0.1 | 35.4 ± 0.2 | ns | 12.7 ± 0.3 | 12.8 ± 0 | ns | 24.5 ± 0.1 | 23.6 ± 0.3 | ns | |||||||||||
| T5 | 28.7 ± 1.6 | 29.8 ± 0.5 | ns | 2.4 ± 0.3 | 2.7 ± 0.1 | ns | 42.5 ± 2.1 | 44.2 ± 2.3 | ns | 4.7 ± 0.2 | 4.9 ± 0.2 | ns | 24.2 ± 0.8 | 24.5 ± 0.1 | ns | |||||||||||
| St Magdalener | T1 | 4.8 ± 0.3 | 4.7 ± 0.1 | ns | 404 * | ns | 3.3 ± 0.2 | 3.3 ± 0.5 | ns | 21.9 * | ns | 22.4 ± 1.9 | 20.3 ± 1.2 | ns | 474 * | ns | 1.6 ± 0.2 | 1.5 ± 0 | ns | 2861 * | ns | 30.2 ± 0.9 | 30.7 ± 1.1 | ns | 56.4 * | ns |
| T2 | 5.4 ± 1 | 4.5 ± 0.2 | ns | 2.7 ± 0.2 | 1.8 ± 0.4 | ns | 23 ± 1.9 | 20.3 ± 1.1 | ns | 3.6 ± 0.2 | 3.3 ± 0 | ns | 40.4 ± 1.2 | 38.6 ± 1.2 | ns | |||||||||||
| T3 | 4.6 ± 0.2 | 4.3 ± 0.4 | ns | 2.4 ± 0.3 | 2.1 ± 0.2 | ns | 21.4 ± 0.3 | 20.2 ± 0.2 | ns | 1.5 ± 0.1 | 1.4 ± 0.3 | ns | 31.3 ± 0.1 | 28.6 ± 1.4 | ns | |||||||||||
| T4 | 15.2 ± 0.4 | 14.2 ± 0 | ns | 4.6 ± 0.9 | 4.2 ± 0.1 | ns | 39.4 ± 0.9 | 39.8 ± 0.4 | ns | 22.2 ± 0.8 | 21.1 ± 0 | * | 24.7 ± 5 | 27.3 ± 0.3 | ns | |||||||||||
| T5 | 6.2 ± 0.1 | 5.9 ± 0.6 | ns | 3.1 ± 0.1 | 3.2 ± 0.1 | ns | 46.9 ± 1 | 47.1 ± 0.6 | ns | 6.1 ± 0.1 | 6 ± 0.2 | ns | 22.1 ± 0.8 | 21.6 ± 0.2 | ns | |||||||||||
| Caffeic Acid | Syringic Acid | (−)-Epicatechin | P-coumaric Acid | Trans-Resveratrol | ||||||||||||||||||||||
| Time | c | blend | F1 | F2 | F3 | c | blend | F1 | F2 | F3 | c | blend | F1 | F2 | F3 | c | blend | F1 | F2 | F3 | c | blend | F1 | F2 | F3 | |
| Lagrein Red | T1 | 3.7 ± 0.1 | 3.6 ± 0 | ns | 1132 * | ns | 7.7 ± 1 | 8.2 ± 0.4 | ns | 144 * | ns | 26.2 ± 1.2 | 24.7 ± 0.5 | ns | 80 * | ns | 6.1 ± 0.1 | 5.7 ± 0.1 | 8.2 * | 4297 * | ns | 3.4 ± 1.4 | 4 ± 0 | ns | 160.5 * | ns |
| T2 | 3.8 ± 0 | 4.6 ± 0 | ns | 6.9 ± 0.1 | 6.5 ± 0.1 | ns | 28.2 ± 0.2 | 25 ± 0.3 | ns | 5.3 ± 0 | 4 ± 0.1 | * | 4.1 ± 0 | 4.2 ± 0.1 | ns | |||||||||||
| T3 | 4.1 ± 0.6 | 1.7 ± 0.1 | * | 6.1 ± 0.4 | 6.3 ± 0.5 | ns | 25.2 ± 1.8 | 20.5 ± 6.3 | ns | 6.1 ± 0.2 | 6.2 ± 0.5 | ns | 3.4 ± 0.5 | 3.6 ± 0 | ns | |||||||||||
| T4 | 27.4 ± 1.7 | 25.3 ± 0.2 | * | 3.9 ± 0 | 4.2 ± 0.1 | ns | 5.3 ± 0 | 4.7 ± 0.1 | ns | 24.7 ± 0.7 | 25.6 ± 0 | ns | 12.4 ± 0.8 | 11.8 ± 0.4 | ns | |||||||||||
| T5 | 2.1 ± 0.1 | 2.1 ± 0.4 | ns | 1.9 ± 0.3 | 1.8 ± 0 | ns | 10.7 ± 0.1 | 10.2 ± 0.4 | 3.7 ± 0 | 3.6 ± 0.1 | ns | 8.6 ± 0.2 | 8.7 ± 0.2 | ns | ||||||||||||
| Lagrein Rosè | T1 | 0.8 ± 0 | 0.8 ± 0 | ns | 41 * | ns | 1.5 ± 0 | 1.5 ± 0.4 | ns | 24 * | ns | 2.2 ± 0.3 | 1.1 ± 0.1 | 5.6 * | 15 * | ns | 2.8 ± 0.1 | 2.6 ± 0.2 | 6.8 * | 1328 * | ns | 2.4 ± 0.1 | 2.5 ± 0 | ns | 1899 * | ns |
| T2 | 0.8 ± 0 | 0.7 ± 0 | ns | 1.3 ± 0.2 | 1.6 ± 0.4 | ns | 2.1 ± 0.6 | 2.8 ± 0.3 | ns | 3.5 ± 0.1 | 3.9 ± 0.2 | * | 2.5 ± 0 | 2.5 ± 0 | ns | |||||||||||
| T3 | 5.5 ± 1.7 | 4.1 ± 0.9 | ns | 1.3 ± 0.1 | 1.2 ± 0.2 | ns | 2.8 ± 0.9 | 1.5 ± 0 | ns | 2.9 ± 0 | 2.9 ± 0 | ns | 2.4 ± 0 | 2.5 ± 0 | ns | |||||||||||
| T4 | 2.4 ± 0.1 | 2.4 ± 0 | ns | 0.2 ± 0 | 0.2 ± 0 | ns | 1.9 ± 0 | 1.9 ± 0 | ns | 1.9 ± 0 | 1.8 ± 0 | ns | 0.4 ± 0 | 0.5 ± 0 | ns | |||||||||||
| T5 | 0 ± 0 | 0 ± 0 | ns | 0.9 ± 0 | 0.9 ± 0 | ns | 3.3 ± 0.1 | 3.7 ± 0.6 | ns | 0.1 ± 0 | 0.1 ± 0 | ns | 0.3 ± 0.1 | 0.2 ± 0 | ns | |||||||||||
| Merlot | T1 | 1.9 ± 0.1 | 2.3 ± 0.1 | ns | 3435 * | ns | 4.6 ± 1.1 | 3.9 ± 1.4 | ns | 30.3 * | ns | 41.7 ± 0.2 | 42.7 ± 6.5 | ns | 274.1 * | ns | 7.9 ± 0 | 7.6 ± 1 | ns | 282.7 * | ns | 2.9 ± 0.7 | 3.5 ± 0.1 | ns | 143.3 * | ns |
| T2 | 5.8 ± 0.2 | 6.2 ± 0.2 | ns | 3.8 ± 0.5 | 4.7 ± 0.8 | ns | 19.8 ± 1.6 | 21 ± 0.2 | ns | 8.6 ± 0.9 | 8.8 ± 0.8 | ns | 3.7 ± 0.1 | 3.4 ± 0.7 | ns | |||||||||||
| T3 | 1.7 ± 0 | 1.5 ± 0.1 | ns | 3.3 ± 0.4 | 3.2 ± 0.3 | ns | 41.2 ± 1.2 | 39.7 ± 5.1 | ns | 7.6 ± 0.4 | 7.5 ± 1.5 | ns | 3.6 ± 0 | 3.5 ± 0.1 | ns | |||||||||||
| T4 | 16 ± 0.1 | 16.1 ± 0.5 | ns | 2.3 ± 0.1 | 2.3 ± 0.1 | ns | 10.5 ± 0 | 10.1 ± 0.3 | ns | 19.4 ± 0.1 | 19.4 ± 0.2 | ns | 16 ± 0.1 | 15.4 ± 0.8 | ns | |||||||||||
| T5 | 0.7 ± 0.2 | 0.6 ± 0.2 | ns | 0.4 ± 0.1 | 0.5 ± 0.1 | ns | 16.7 ± 1 | 17 ± 0.4 | ns | 1.5 ± 0.1 | 1.6 ± 0 | ns | 8.1 ± 2.3 | 9.5 ± 0.4 | ns | |||||||||||
| St Magdalener | T1 | 1.2 ± 0 | 1.2 ± 0.1 | ns | 517 * | ns | 5.5 ± 0.4 | 5.4 ± 0.1 | ns | 319 * | ns | 22.7 ± 1.1 | 21.1 ± 0.6 | ns | 101 * | ns | 3.5 ± 0 | 3.7 ± 0.2 | ns | 705 * | ns | 2.7 ± 0 | 2.7 ± 0 | ns | 22.8 * | ns |
| T2 | 3.6 ± 0.5 | 2.5 ± 0.3 | ns | 4.3 ± 0.2 | 3.8 ± 0.5 | ns | 22.8 ± 0.4 | 20.1 ± 1 | ns | 6.4 ± 0.3 | 6 ± 0.4 | ns | 3.3 ± 0.1 | 3.4 ± 0.3 | ns | |||||||||||
| T3 | 1 ± 0 | 1 ± 0.1 | ns | 3.3 ± 0.3 | 3.2 ± 0.1 | ns | 18.3 ± 0.6 | 16.5 ± 0.2 | ns | 4.3 ± 0 | 3.8 ± 0 | ns | 2.6 ± 0 | 2.7 ± 0 | ns | |||||||||||
| T4 | 16.6 ± 1.8 | 17.6 ± 0.1 | ns | 0.8 ± 0.1 | 0.9 ± 0 | ns | 7.8 ± 2.8 | 9.3 ± 0 | ns | 15.4 ± 1 | 16.2 ± 0.2 | ns | 4.5 ± 1.2 | 3.5 ± 0.1 | ns | |||||||||||
| T5 | 0 ± 0 | 0 ± 0 | ns | 0.3 ± 0 | 0.3 ± 0 | ns | 12.1 ± 0.2 | 11.4 ± 0.6 | ns | 3.2 ± 0 | 3.2 ± 0.1 | ns | 1.4 ± 0 | 1.3 ± 0.4 | ns | |||||||||||
| Glucoside | Acetyl-Glucoside | Cumaroyl-Glucoside | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time | c | Blend | F1 | F2 | F3 | c | Blend | F1 | F2 | F3 | c | Blend | F1 | F2 | F3 | |
| Lagrein red | T1 | 270.3 ± 6.2 | 266.2 ± 0.4 | ns | 1481 * | ns | 87 ± 0.9 | 85.7 ± 0.1 | ns | 1694 * | ns | 27.2 ± 0.6 | 27.6 ± 0.2 | ns | 1130 * | ns |
| T2 | 253.3 ± 4 | 249.6 ± 3.3 | ns | 80.5 ± 0.6 | 79 ± 1.9 | ns | 26.1 ± 0.3 | 25.3 ± 0.5 | ns | |||||||
| T3 | 206.4 ± 0 | 193.1 ± 3.4 | * | 61.4 ± 0 | 58.9 ± 0.7 | ns | 20.7 ± 0.1 | 19.6 ± 0.4 | ns | |||||||
| T4 | 206 ± 0.4 | 198 ± 5.6 | ns | 69.8 ± 1 | 64.3 ± 2.7 | * | 21.1 ± 0 | 20.3 ± 0.8 | ns | |||||||
| T5 | 83.3 ± 5 | 80 ± 1.2 | ns | 17.1 ± 1.3 | 17.2 ± 0.4 | ns | 9.8 ± 0.2 | 10 ± 0.3 | ns | |||||||
| Lagrein rosè | T1 | 28.2 ± 0.2 | 28.1 ± 0.2 | ns | 34170 * | ns | 14.2 ± 0.1 | 14.3 ± 0 | ns | 20423 * | ns | 7.4 ± 0 | 7.3 ± 0.1 | ns | 10927 * | ns |
| T2 | 27.8 ± 0 | 27.8 ± 0.2 | ns | 14 ± 0.2 | 13.9 ± 0.1 | ns | 3.9 ± 0.1 | 4 ± 0.1 | ns | |||||||
| T3 | 25.7 ± 0 | 25.3 ± 0 | ns | 13 ± 0.1 | 13 ± 0 | ns | 3.8 ± 0 | 3.8 ± 0 | ns | |||||||
| T4 | 12.8 ± 0 | 12.6 ± 0.3 | ns | 4.1 ± 0.1 | 4 ± 0.1 | ns | 0.8 ± 0 | 0.7 ± 0.1 | ns | |||||||
| T5 | 5.8 ± 0 | 5.7 ± 0 | ns | 1 ± 0 | 1 ± 0 | ns | 0.4 ± 0 | 0.4 ± 0 | ns | |||||||
| Merlot | T1 | 163.8 ± 1.4 | 159.7 ± 2.6 | ns | 3439 * | ns | 46 ± 0.5 | 51.6 ± 0.9 | 15.5 * | 1941 * | * | 19.2 ± 0.3 | 18.9 ± 0.1 | ns | 979 * | ns |
| T2 | 152.7 ± 0.7 | 149.9 ± 0 | ns | 42.8 ± 0.3 | 47.6 ± 0.5 | * | 18.6 ± 0.3 | 18.1 ± 0.1 | ns | |||||||
| T3 | 123.1 ± 0.2 | 121.5 ± 0.4 | ns | 35.7 ± 0.6 | 38.1 ± 0.9 | * | 15.6 ± 0.3 | 15.3 ± 0.1 | ns | |||||||
| T4 | 117.1 ± 0.7 | 114.4 ± 3.9 | ns | 30.4 ± 0.2 | 30.3 ± 1 | ns | 13.5 ± 0.1 | 13.1 ± 0.8 | ns | |||||||
| T5 | 48.4 ± 1.1 | 47.6 ± 0.4 | ns | 9.9 ± 0.1 | 9.6 ± 0.4 | ns | 7.2 ± 0.1 | 6.8 ± 0 | ns | |||||||
| St Magdalener | T1 | 142.5 ± 1.7 | 142.7 ± 0.4 | 4.5 * | 4767 * | ns | 29.6 ± 0.1 | 28.9 ± 0.1 | 5.6 * | 2971 * | ns | 13.5 ± 0.2 | 13.4 ± 0 | ns | 1867 * | ns |
| T2 | 138.6 ± 0 | 137.2 ± 0.1 | ns | 28.9 ± 0.1 | 28.6 ± 0.1 | ns | 13.4 ± 0.3 | 13.6 ± 0.2 | ns | |||||||
| T3 | 118.2 ± 1.1 | 116.2 ± 1.2 | ns | 24.6 ± 0.1 | 24.5 ± 0.4 | ns | 12.1 ± 0.1 | 12 ± 0.1 | ns | |||||||
| T4 | 113 ± 2.5 | 103.1 ± 2.9 | * | 16.2 ± 0.5 | 13.9 ± 0.7 | * | 10.6 ± 0.1 | 9.8 ± 0.7 | ns | |||||||
| T5 | 47.6 ± 0.3 | 43.1 ± 3.9 | ns | 5.2 ± 0 | 4.6 ± 0.4 | ns | 0.9 ± 0 | 1 ± 0.1 | ns | |||||||
| Time | Blend | c | Significant Difference (95% Confidence) | |
|---|---|---|---|---|
| Lagrein Red | T1 | 0.40 ± 0.00 | 0.25 ± 0.07 | |
| T2 | 0.30 ± 0.00 | 0.20 ± 0.00 | * | |
| T3 | 0.20 ± 0.00 | 0.25 ± 0.07 | ||
| T4 | 0.20 ± 0.00 | 0.20 ± 0.14 | ||
| T5 | 0.45 ± 0.07 | 0.50 ± 0.00 | ||
| Lagrein Rosé | T1 | 3.10 ± 0.00 | 1.40 ± 0.00 | * |
| T2 | 3.05 ± 0.07 | 2.20 ± 0.14 | * | |
| T3 | 3.65 ± 0.07 | 2.45 ± 0.07 | * | |
| T4 | 2.65 ± 0.07 | 1.90 ± 0.00 | * | |
| T5 | 2.75 ± 0.07 | 2.90 ± 0.00 | ||
| Merlot | T1 | 1.70 ± 0.14 | 1.3 ± 0.28 | |
| T2 | 2.35 ± 0.21 | 1.80 ± 0.00 | ||
| T3 | 2.75 ± 0.07 | 2.15 ± 0.07 | * | |
| T4 | 1.25 ± 0.07 | 1.10 ± 0.00 | ||
| T5 | 2.35 ± 0.07 | 2.55 ± 0.07 | ||
| St. Magdalener | T1 | 0.35 ± 0.07 | 0.15 ± 0.07 | |
| T2 | 0.30 ± 0.00 | 0.10 ± 0.00 | * | |
| T3 | 0.40 ± 0.00 | 0.20 ± 0.00 | * | |
| T4 | 0.10 ± 0.00 | 0.10 ± 0.00 | ||
| T5 | 0.50 ± 0.14 | 0.45 ± 0.07 |
| No. | Esters | LRI [Ref.] |
|---|---|---|
| 2 | Ethyl butanoate | 803 [28] |
| 3 | 2-methylbutanoic acid, ethyl ester | 846 [28] |
| 4 | 3-methylbutanoic acid, ethyl ester | 859 [29] |
| 6 | Isopentyl acetate | 876 [30] |
| 7 | 4-Ethylbenzoic acid, 2-butylester | - |
| 10 | Ethyl hexanoate | 999 [30] |
| 11 | Hexyl acetate | 1011 [30] |
| 14 | 4-Methylbenzaldehyde | 1076 [31] |
| 16 | 4-Ethylbenzaldehyde | 1163 [32] |
| 18 | Diethyl succinate | 1179 [33] |
| 20 | Methyl salicylate | 1192 [30] |
| 21 | Ethyl octanoate | 1194 [34] |
| 22 | Benzenacetic acid ethyl ester | 1243 [30] |
| 23 | 2-Phenylethylacetate | 1255 [30] |
| 24 | Ethyl decanoate | 1392 [30] |
| 25 | Ethyl dodecanoate | 1554 [30] |
| 26 | Ethyl hexadecanoate | 1992 [35] |
| No. | Alcohols | LRI [Ref.] |
| 5 | 1-Hexanol | 865 [30] |
| 8 | 1-Heptanol | 969 [30] |
| 9 | 1-Octen-3ol | 980 [28] |
| 13 | 2-Ethyl hexanol | 1028 [36] |
| 15 | Octanol | 1070 [30] |
| 17 | 2-Phenylethyl alcohol | 1112 [28] |
| No. | Acids | |
| 1 | Acetic acid | 599 [28] |
| 19 | Octanoic acid | 1180 [37] |
| No. | Terpens | |
| 12 | Limonene | 1020 [30] |
| Lagrein Red | Lagrein Rosé | Merlot | St. Magdalener | |
|---|---|---|---|---|
| T1 (Bottling time) | ns | ns | * | * |
| T2 (1 month) | ns | ns | ns | ns |
| T3 (3 months) | ns | ns | ns | ns |
| T4 (6 months) | ns | ns | * | ns |
| T5 (12 months) | ns | ns | ns | ns |
| Wines | Control | Conventional Stoppers |
|---|---|---|
| Lagrein red | Blend a | One-piece natural cork |
| Lagrein Rosé | Blend | Technical cork 1+1 |
| Merlot | Blend | Technical cork 1+1 |
| St Magdalener | Blend | Agglomerated natural cork |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossetti, F.; Jouin, A.; Jourdes, M.; Teissedre, P.-L.; Foligni, R.; Longo, E.; Boselli, E. Impact of Different Stoppers on the Composition of Red and Rosé Lagrein, Schiava (Vernatsch) and Merlot Wines Stored in Bottle. Molecules 2020, 25, 4276. https://doi.org/10.3390/molecules25184276
Rossetti F, Jouin A, Jourdes M, Teissedre P-L, Foligni R, Longo E, Boselli E. Impact of Different Stoppers on the Composition of Red and Rosé Lagrein, Schiava (Vernatsch) and Merlot Wines Stored in Bottle. Molecules. 2020; 25(18):4276. https://doi.org/10.3390/molecules25184276
Chicago/Turabian StyleRossetti, Fabrizio, Alicia Jouin, Michael Jourdes, Pierre-Louis Teissedre, Roberta Foligni, Edoardo Longo, and Emanuele Boselli. 2020. "Impact of Different Stoppers on the Composition of Red and Rosé Lagrein, Schiava (Vernatsch) and Merlot Wines Stored in Bottle" Molecules 25, no. 18: 4276. https://doi.org/10.3390/molecules25184276
APA StyleRossetti, F., Jouin, A., Jourdes, M., Teissedre, P.-L., Foligni, R., Longo, E., & Boselli, E. (2020). Impact of Different Stoppers on the Composition of Red and Rosé Lagrein, Schiava (Vernatsch) and Merlot Wines Stored in Bottle. Molecules, 25(18), 4276. https://doi.org/10.3390/molecules25184276









