Polymer-Supported Phosphoric, Phosphonic and Phosphinic Acids—From Synthesis to Properties and Applications in Separation Processes
Abstract
1. Introduction
2. Uniqueness of Phosphorus-Containing Compounds
3. Resins Preparation
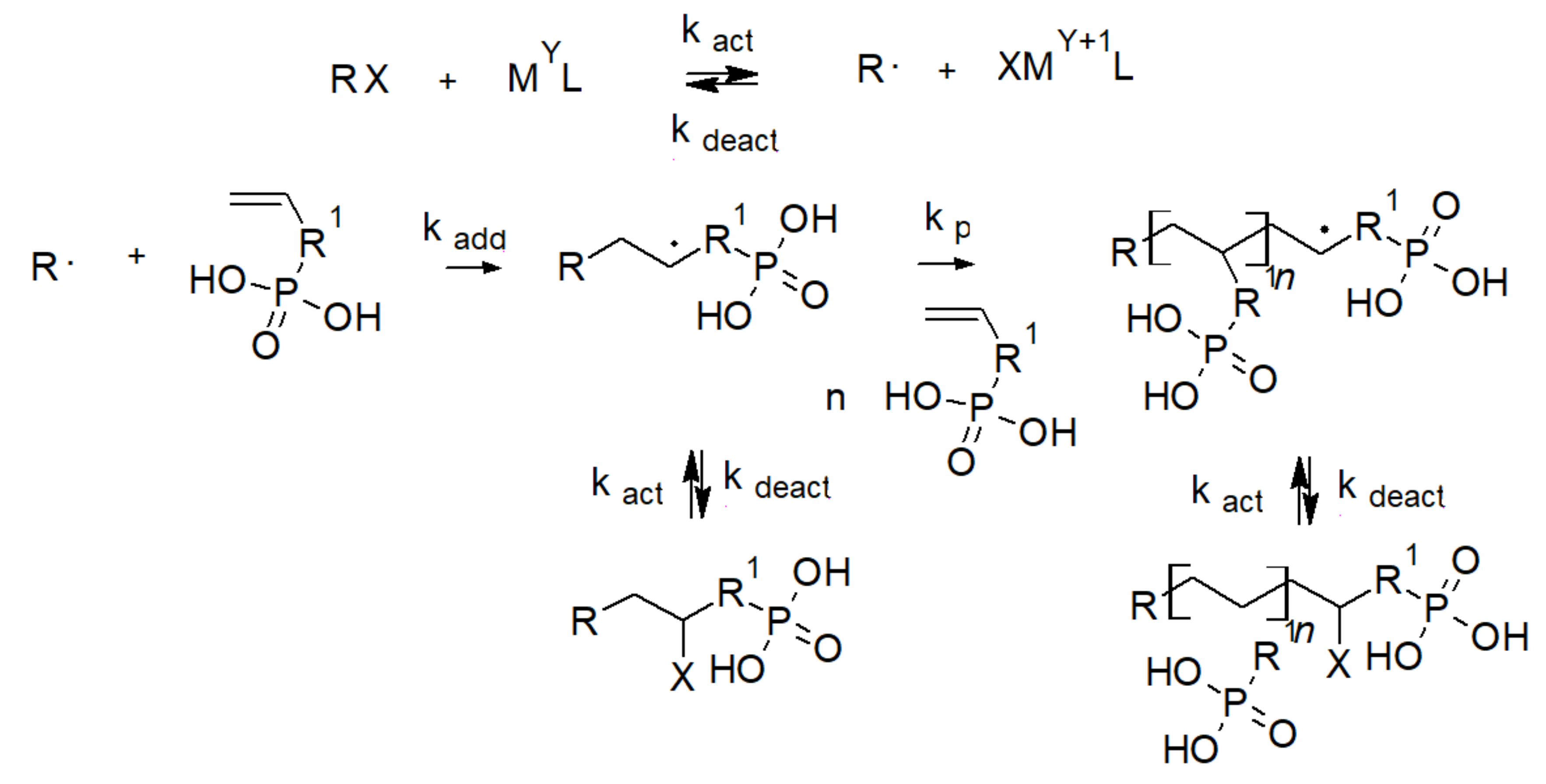
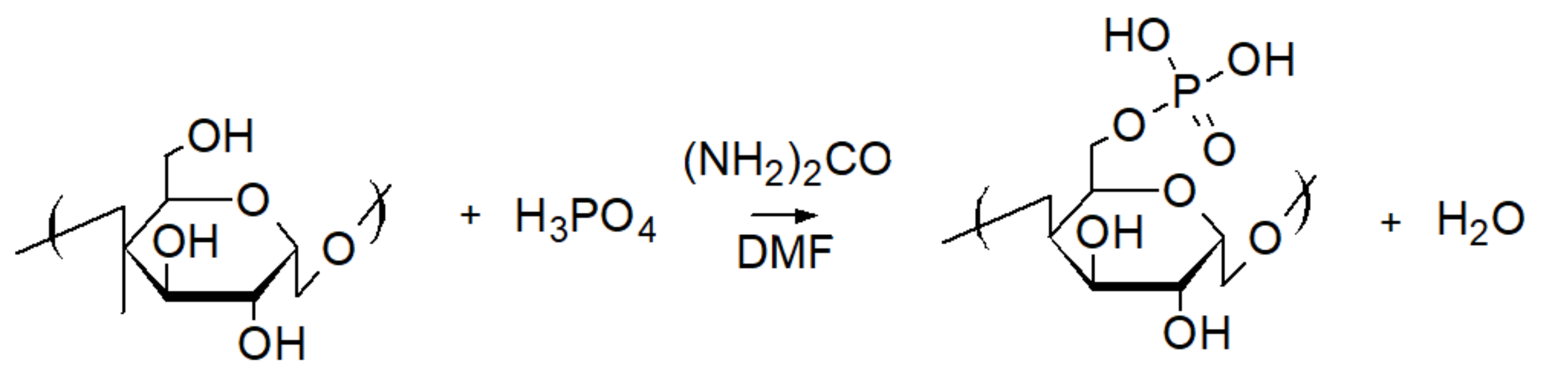
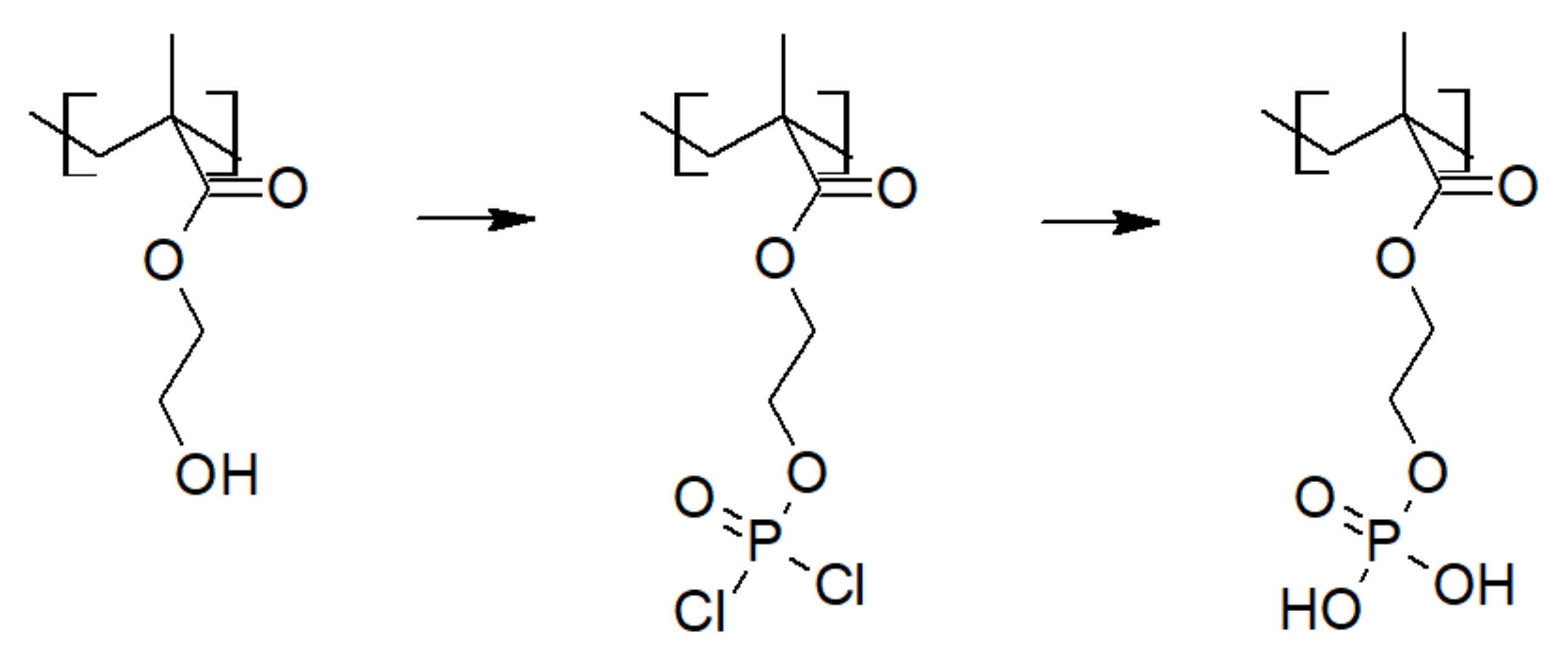
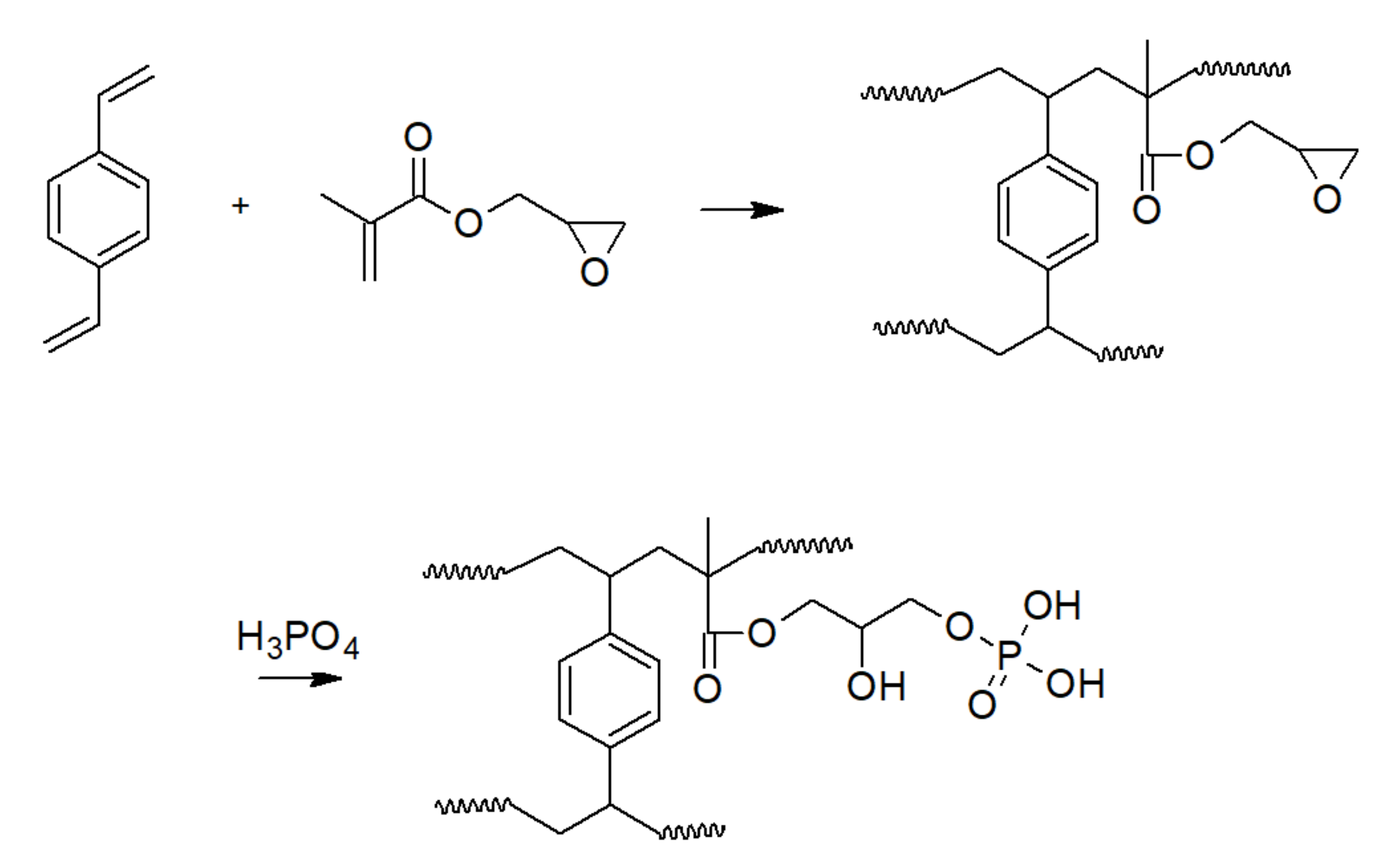
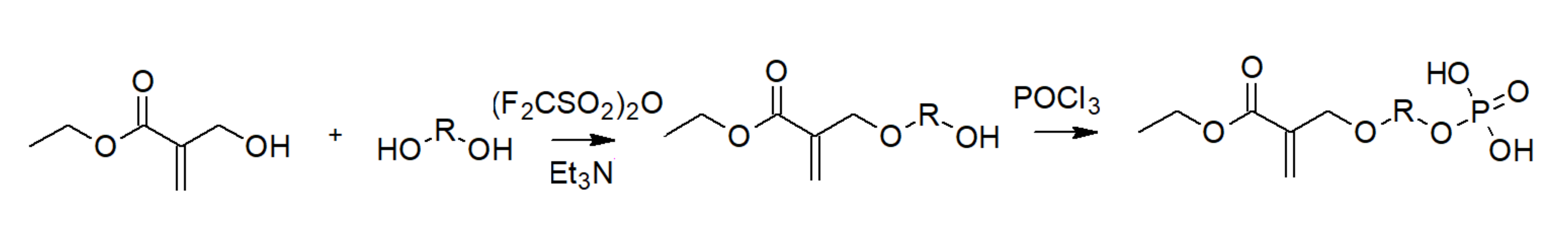
3.1. Phosphoric Acid Polymers
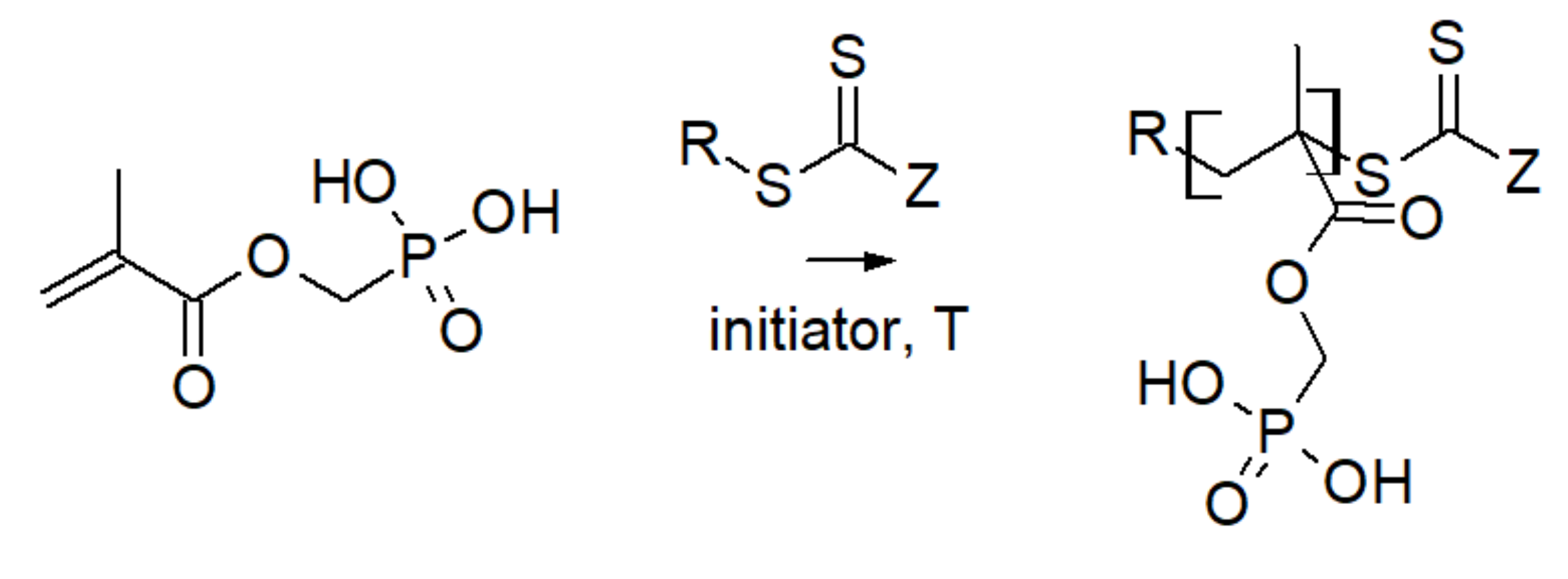
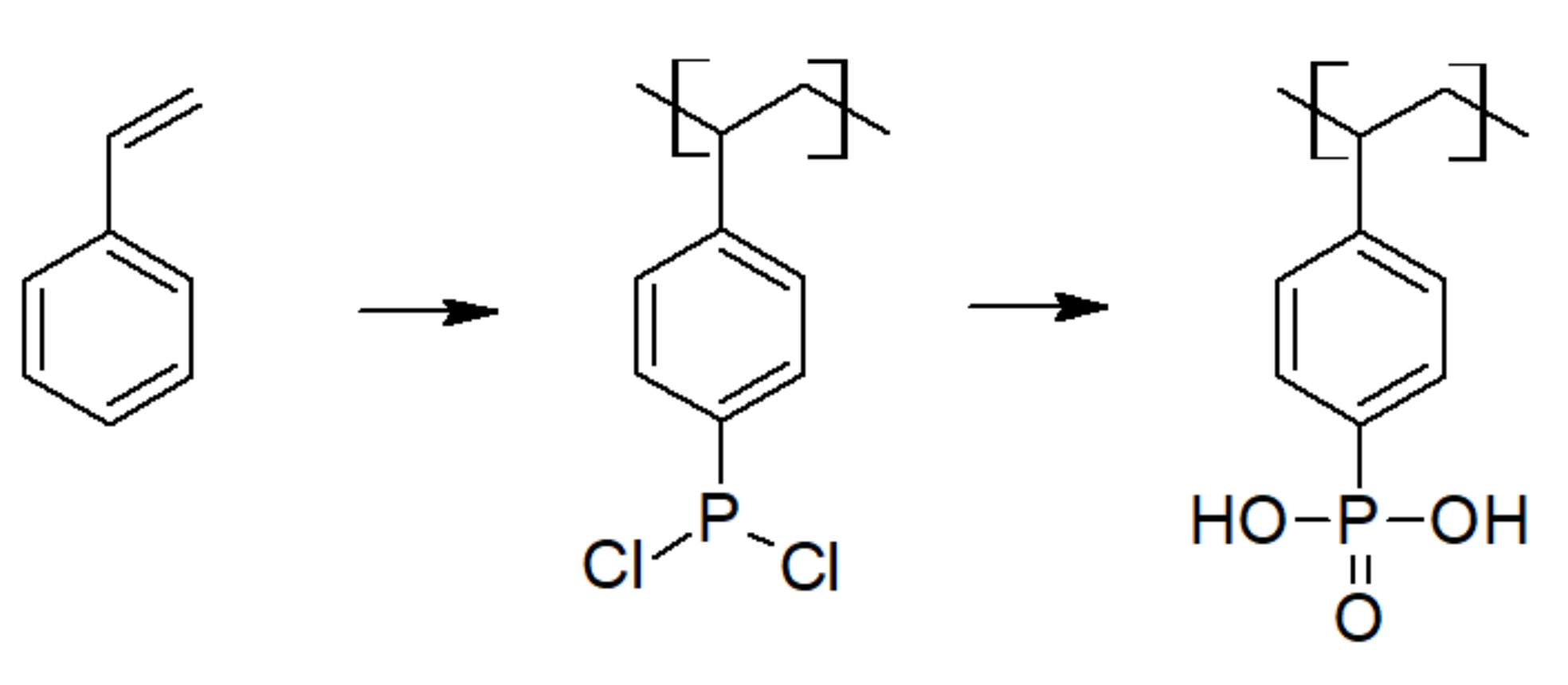
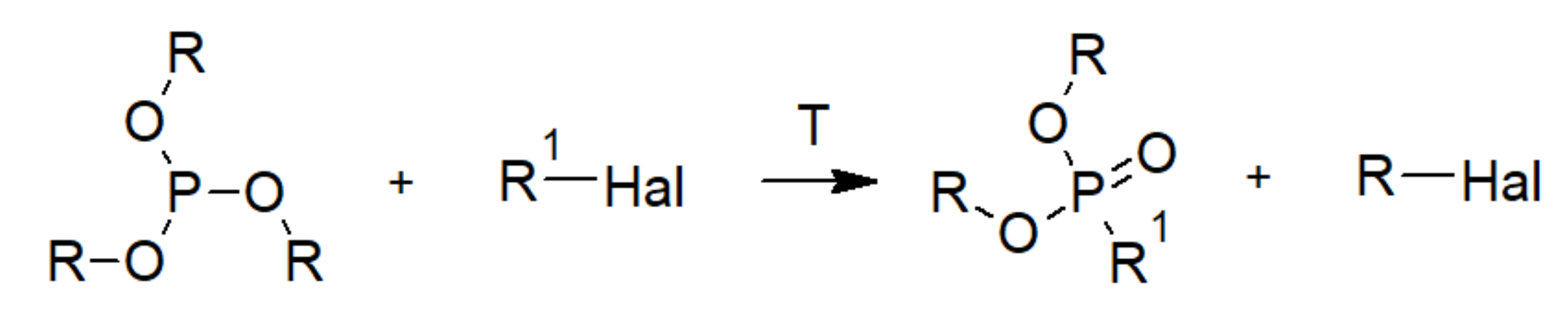
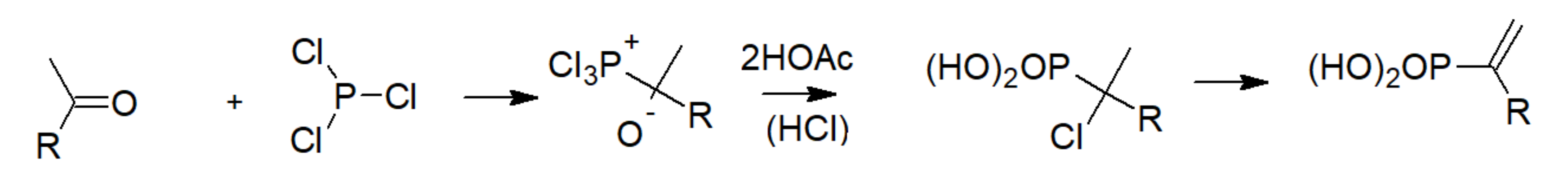
3.2. Phosphonic Acid Polymers
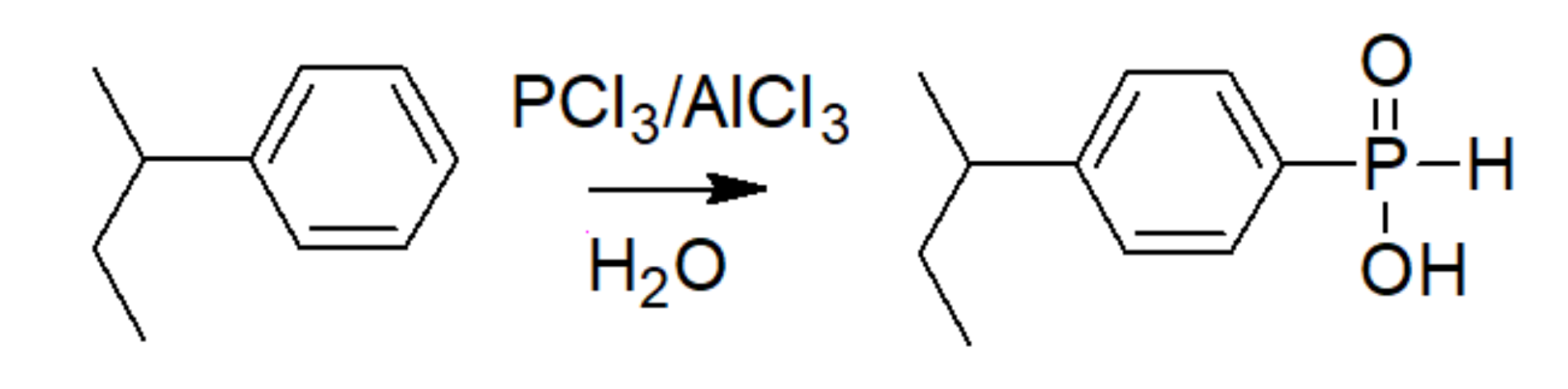
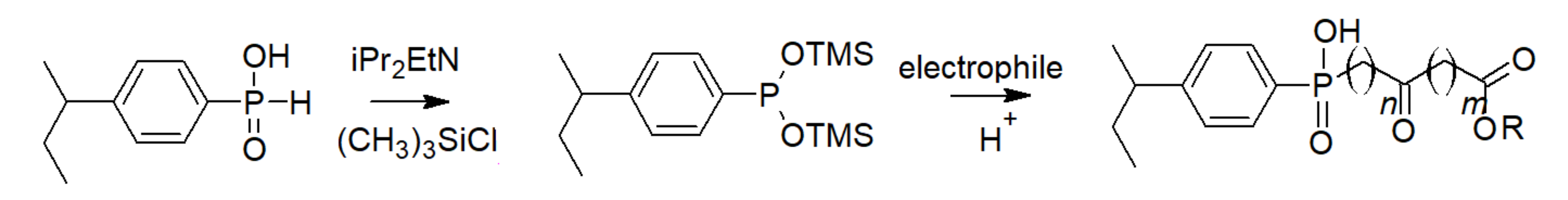
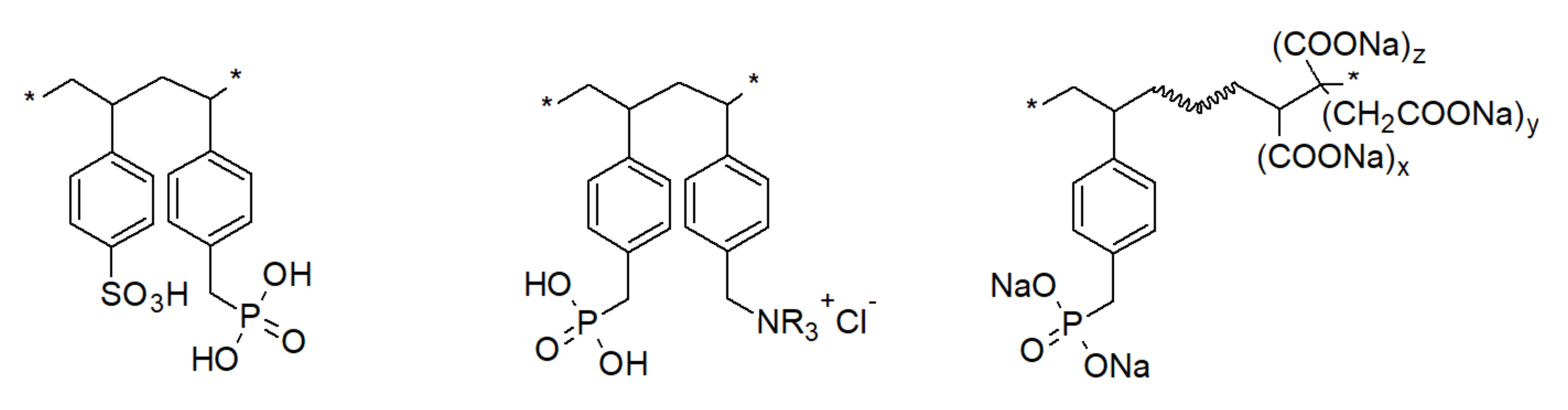
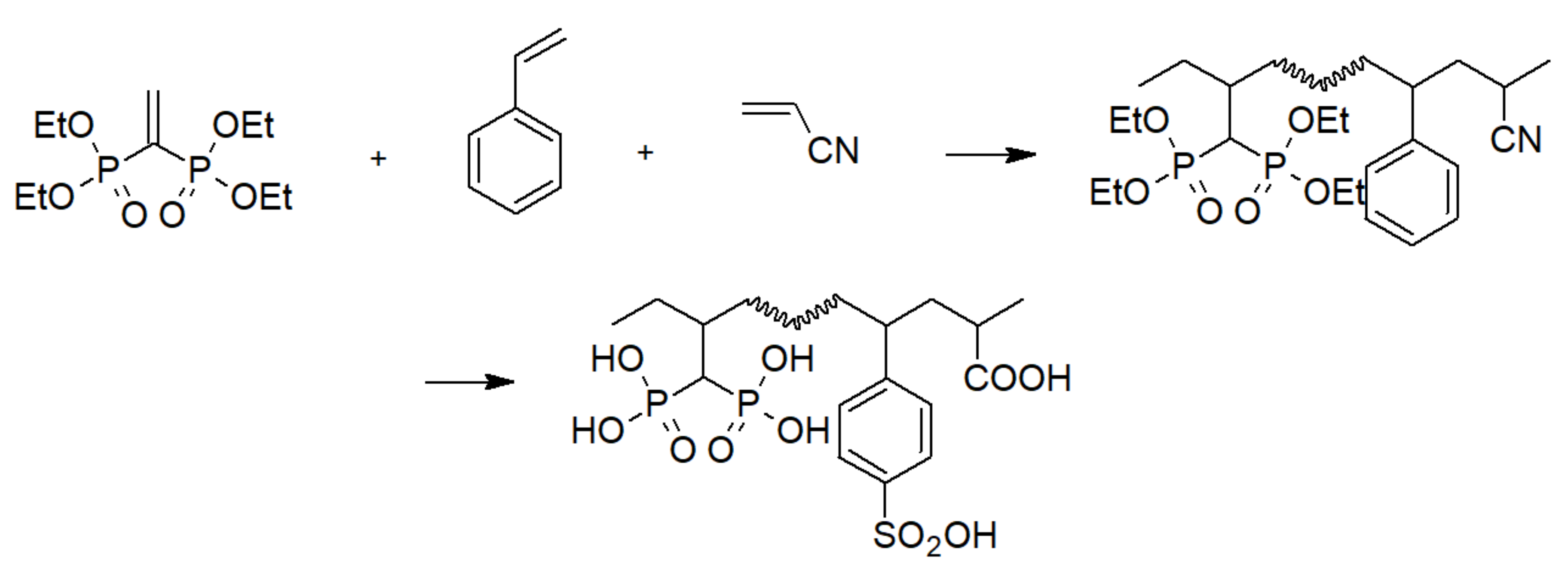
3.3. Phosphinic Acid Polymers
4. Organophosphorus Polymers in Separation Science
4.1. Solvent Extraction and Organophosphorus Polymers in Separation Processes
4.2. Developments in Organophosphorus Resins
- class I—ion exchange/redox resins, e.g., phosphinic acid resin is capable of ion exchange with any metal ion, but also of reducing the limited number of ions such as Hg (II), Ag (I), Cu (II), Pd(II) to free metal, thus oxidizing the ligand to phosphonic acid;
- class II—ion exchange/coordination resins; DMBPs composed of phosphonic acid (access mechanism) and coordinating ligand (amine, ester—recognition mechanism), e.g., phosphonate diester/monoester resin, where a synergistic enhancement in the amount of complexed Ag(I) in comparison to corresponding monofunctional resin was observed;
- class III—ion exchange/precipitation resins; e.g., resin composed of phosphonic acid ligands and quaternary ammonium ligand. The former ligand allows ions into the polymer network, the latter carrying an associated anion leads to the formation of insoluble salt when a given cation comes into close proximity. Process occurs rapidly and the precipitate is retained within polymer bead.
5. Summary
Funding
Conflicts of Interest
References
- Hanson, P.R. Organophosphorus chemistry. Beilstein J. Org. Chem. 2014, 10, 2087–2088. [Google Scholar] [CrossRef] [PubMed]
- Quin, L.D. A Guide to Organophosphorus Chemistry; A John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2000; ISBN 0-471-31824-8. [Google Scholar]
- Maiti, S.; Banerjee, S.; Palit, S.R. Phosphorus-containing polymers. Prog. Polym. Sci. 1993, 18, 227–261. [Google Scholar] [CrossRef]
- Ripperger, K.P.; Alexandratos, S.D. Polymer-Supported Phosphorus-Containing Ligands for Selective Metal Ion Complexation. In Adsorption and Its Application in Industry and Environmental Protection Vol II: Appliactions in Environmental Protecion; Elsevier Science: Amsterdam, The Netherlands, 1999; Volume 120, pp. 473–495. [Google Scholar]
- Monge, S.; David, G. Phosphorus-Based Polymers–From Synthesis to Applications; Royal Society of Chemistry: Cambridge, UK, 2014; ISBN 978-1-84973-646-6. [Google Scholar]
- Yang, Y.; Alexandratos, S.D. Affinity of Polymer-Supported Reagents for Lanthanides as a Function of Donor Atom Polarizability. Ind. Eng. Chem. Res. 2009, 48, 6173–6187. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Crick, D.W. Polymer-Supported Reagents: Application to Separation Science. Ind. Eng. Chem. Res. 1996, 35, 635–644. [Google Scholar] [CrossRef]
- Janauer, G.E.; Ramseyer, G.O.; Lin, J.W. Selective Separations by Reactive Ion Exchange with Common Polystyrene-Type Resins. Part, I. General Considerations. Anal. Chim. Acta 1974, 73, 311. [Google Scholar] [CrossRef]
- El Asri, Z.; Chougrani, K.; Negrell-Guirao, C.; David, G.; Boutevin, B.; Loubat, C. An Efficient Process for Synthesizing and Hydrolyzing a Phosphonated Methacrylate: Investigation of the Adhesive and Anticorrosive Properties. J. Polym. Sci. A1 Polym. Chem. 2008, 46, 4794–4803. [Google Scholar] [CrossRef]
- Markova, D.; Opper, K.L.; Wagner, M.; Klapper, M.; Wagener, K.B.; Mullen, K. Synthesis of proton conducting phosphonic acid-functionalized polyolefins by the combination of ATRP and ADMET. Polym. Chem. 2013, 4, 1351–1363. [Google Scholar] [CrossRef]
- David, G.; Negrell, C.; Manseri, A.; Boutevin, B. Poly(MMA)-b-poly(monophosphonic acrylate) Diblock Copolymers Obtained by ATRP and Used as Additives for Anticorrosive Coatings. J. Appl. Polym. Sci. 2009, 114, 2213–2220. [Google Scholar] [CrossRef]
- Barthélémy, B.; Devillers, S.; Minet, I.; Delhalle, J.; Mekhalif, Z. Surface-initiated ATRP of 2-(methacryloyloxy)ethyl 2-(trimethylammonio)ethyl phosphate on Phynox. Appl. Surf. Sci. 2011, 258, 466–473. [Google Scholar] [CrossRef]
- Barnard, Z.Z.; Keen, I.; David, J.T.H.; Traian, V.; Chirila, D.; Harkin, G. PHEMA Hydrogels Modified through the Grafting of Phosphate Groups by ATRP Support the Attachment and Growth of Human Corneal Epithelial Cells. J. Biomater. Appl. 2008, 23, 147–168. [Google Scholar]
- Yuan, B.; Chen, Q.; Ding, W.-Q.; Liu, P.-S.; Wu, S.-S.; Lin, S.-C.; Shen, J.; Gai, Y. Copolymer Coatings Consisting of 2-Methacryloyloxyethyl Phosphorylcholine and 3-Methacryloxypropyl Trimethoxysilane via ATRP To Improve Cellulose Biocompatibility. Appl. Mater. Interfaces 2012, 4, 4031–4039. [Google Scholar]
- Zhou, F.; Huck, W.T.S. Three-stage switching of surface wetting using phosphate-bearing polymer brushes. Chem. Commun. 2005, 48, 5999–6001. [Google Scholar]
- Yu, X.; Yang, X.; Horte, S.; Kizhakkedathu, J.N.; Brooks, D.E. ATRP synthesis of poly(2-(methacryloyloxy)ethyl choline phosphate): A multivalent universal biomembrane adhesive. Chem. Commun. 2013, 49, 6831–6833. [Google Scholar]
- Huang, J.; Matyjaszewski, K. Atom Transfer Radical Polymerization of Dimethyl(1-ethoxycarbonyl)vinyl Phosphate and Corresponding Block Copolymers. Macromolecules 2005, 38, 3577–3583. [Google Scholar]
- Ma, Y.; Tang, Y.N.C.; Armes, S.P.; Lewis, A.L.; Lloyd, A.W.; Salvage, J.P. Well-Defined Biocompatible Block Copolymers via Atom Transfer Radical Polymerization of 2-Methacryloyloxyethyl Phosphorylcholine in Protic Media. Macromolecules 2003, 36, 3475–3484. [Google Scholar]
- Canniccioni, B.; Monge, S.; David, G.; Robin, J.J. RAFT polymerization of dimethyl(methacryloyloxy)-methyl phosphonate and its phosphonic acid derivative: A new opportunity for phosphorus-based materials. Polym. Chem. 2013, 4, 3676. [Google Scholar] [CrossRef]
- David, G.; El Asri, Z.; Rich, S.; Castignolles, P.; Guillaneuf, Y.; Lacroix-Desmazes, P.; Boutevin, B. Peculiar Behavior of Degenerative Chain Transfer Polymerization of a Phosphonated Methacrylate. Macromol. Chem. Phys. 2009, 210, 631–639. [Google Scholar]
- Baratha, K.V.; Nourry, A.; Pilard, J.-F. Synthesis of NR based polyurethanes containing phosphorylated polymers as chain extenders. Eur. Polym. J. 2015, 70, 317–330. [Google Scholar]
- Yusa, S.; Fukuda, K.; Yamamoto, T.; Ishihara, K.; Morishima, Y. Synthesis of Well-Defined Amphiphilic Block Copolymers Having Phospholipid Polymer Sequences as a Novel Biocompatible Polymer Micelle Reagent. Biomacromolecules 2005, 6, 663–670. [Google Scholar] [CrossRef]
- Stenzel, M.H.; Barner-Kowollik, C.; Davis, T.P.; Dalton, H.M. Amphiphilic Block Copolymers Based on Poly(2-acryloyloxyethyl phosphorylcholine) Prepared via RAFT Polymerisation as Biocompatible Nanocontainers. Macromol. Biosci. 2004, 4, 445–453. [Google Scholar] [PubMed]
- Bhuchar, N.; Deng, Z.; Ishihara, K.; Narain, R. Detailed study of the reversible addition–fragmentation chain transfer polymerization and co-polymerization of 2-methacryloyloxyethyl phosphorylcholine. Polym. Chem. 2011, 2, 632–639. [Google Scholar] [CrossRef]
- Uchiyama, M.; Sato, K.; Kamigaito, M. A phosphonium intermediate for cationic RAFT polymerization. Polym. Chem. 2016, 7, 1387–1396. [Google Scholar] [CrossRef]
- Alexandratos, S.D. Polymer-Supported Reagents with Enhanced Metal Ion Recognition: Application to Separations Science. Separ. Purif. Method. 1992, 21, 1–22. [Google Scholar]
- Alexandratos, S.D.; Hussain, L.A. Bifunctionality as a Means of Enhancing Complexation Kinetics in Selective Ion Exchange Resins. Ind. Eng. Chem. Res. 1996, 34, 251–254. [Google Scholar] [CrossRef]
- Jyo, A.; Jamabe, K.; Kimura, G. Properties of phosphoric acid resins derived from poly(glycidyl methacrylates) crosslinked with dimethacrylates of oligo(ethylene glycols), Special Publication. R. Soc. Chem. 1999, 239, 246–252. [Google Scholar]
- Head, A.J.; Kember, N.F.; Miller, R.P.; Wells, R.A. Ion-exchange properties of cellulose phosphate. J. Chem. Soc. 1958, 3418–3425. [Google Scholar] [CrossRef]
- Jurgens, J.F.; Reid, D.; Guthrie, J.D. Phosphorylated Cotton Cellulose as a Cation-Exchange Material. Text. Res. J. 1948, 18, 42–44. [Google Scholar] [CrossRef]
- Hoffpauir, C.L.; Guthrie, J.D. Ion-Exchange Characteristics of Chemically Modified Cotton Fabrics. Text. Res. J. 1950, 20, 617–620. [Google Scholar] [CrossRef]
- Guthrie, J.D. Ion Exchange Cottons. Ind. Eng. Chem. 1952, 44, 2187–2189. [Google Scholar] [CrossRef]
- Peterson, E.A.; Sober, H.A. Chromatography of Proteins. I. Cellulose Ion-exchange Adsorbents. J. Am. Chem. 1956, 78, 751–755. [Google Scholar] [CrossRef]
- Reid, J.D.; Mazzeno, L.W. Preparation and Properties of Cellulose Phosphates. Ind. Eng. Chem. 1949, 41, 2828–2831. [Google Scholar] [CrossRef]
- Ford, F.M.; Hall, W.P. Flameproofing of Fibrous Materials. U.S. Patent 2,482,755, 29 May 1949. [Google Scholar]
- Jayakumar, R.; Reis, R.L.; Mano, J.F. Chemistry and Applications of Phosphorylated Chitin and Chitosan. e-Polymers 2006, 35. [Google Scholar] [CrossRef]
- Furukawa, J.; Kobayashi, E.; Wakui, T. Preparations and Chelating Properties of Ethyl-(p-vinylphenyl)-phosphoric Acid or Ethyl-(p-vinylphenyl)thiophosphoric-Acid Polymer and Copolymer. Polymer. J. 1980, 12, 293–303. [Google Scholar]
- Jyo, A.; Matsufune, S.; Ono, H.; Egawa, H. Preparation of phosphoric acid resins with large cation exchange capacities from Macroreticular poly(glycidyl methacrylate-co-divinylbenzene) beads and their behavior in uptake of metal ions. J. Appl. Polym. Sci. 1997, 63, 1327–1334. [Google Scholar] [CrossRef]
- Jyo, A.; Zhu, X. Metal-Ion Selectivity of Phosphoric Acid Resin in Aqueous Nitric Acid Media. In Chemistry for the Protection of the Environment 3; Pawłowski, L., Gonzales, M.A., Dudzińska, M.R., Lacy, W.J., Eds.; Environmental Science Research; Springer: Berlin/Heidelberg, Germany, 1998. [Google Scholar]
- Egawa, H.; Nonaka, T.; Maeda, H. Studies on selective adsorption resins. XXI. Preparation and properties of macroreticular chelating ion exchange resins containing phosphoric acid groups. J. Appl. Polym. Sci. 1985, 30, 3239–3247. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Hussain, L.A. Synthesis of α-, β-, and γ-Ketophosphonate Polymer-Supported Reagents: The Role of Intra-ligand Cooperation in the Complexation of Metal Ions. Macromolecules 1998, 31, 3235–3238. [Google Scholar] [CrossRef]
- Klee, J.E.; Lehmann, U. Novel 2-(ω-phosphonooxy-2-oxaalkyl)acrylate monomers for self-etching self-priming one part adhesive. Beilstein J. Org. Chem. 2010, 6, 766–772. [Google Scholar] [CrossRef]
- Moszner, N.; Salz, U.; Zimmermann, J. Chemical aspects of self-etching enamel–dentin adhesives: A systematic review. Dent. Mater. 2005, 21, 895–910. [Google Scholar] [CrossRef]
- Salz, U.; Zimmermann, J.; Zeuner, F.; Moszner, N. Hydrolytic stability of self-etching adhesive systems. J. Adhes. Dent. 2005, 7, 107–116. [Google Scholar] [PubMed]
- Moszner, N.; Zeuner, F.; Pfeiffer, S.; Schurte, I.; Rheinberger, V.; Drache, M. Monomers for Adhesive Polymers, 3 - Synthesis, Radical Polymerization and Adhesive Properties of Hydrolytically Stable Phosphonic Acid Monomers. Macromol. Mater. Eng. 2001, 286, 225–231. [Google Scholar] [CrossRef]
- Sahin, G.; Albayrak, A.Z.; Bilgici, Z.S.; Avci, D. Synthesis and Evaluation of New Dental Monomers with Both Phosphonic and Carboxylic Acid Functional Groups. J. Polym. Sci. Part A 2009, 47, 1953–1965. [Google Scholar] [CrossRef]
- David, G.; Boutevin, B.; Seabrook, S.; Destarac, M.; Woodward, G.; Otter, G. Radical Telomerisation of Vinyl Phosphonic Acid with a Series of Chain Transfer Agents. Macromol. Chem. Phys. 2007, 208, 635–642. [Google Scholar] [CrossRef]
- David, G.; Boyer, C.; Tayouo, R.; Seabrook, S.; Ameduri, B.; Boutevin, B.; Woodward, G.; Destarac, M. A Process for Polymerizing Vinyl Phosphonic Acid with C6F13I Perfluoroalkyl Iodide Chain-Transfer Agent. Macromol. Chem. Phys. 2008, 209, 75–83. [Google Scholar] [CrossRef]
- Michaelis, A.; Arbuzov, B.A. Reactions of alkyl halides with phosphites. J. Russ. Phys. Chem. Soc. 1906, 38, 687. [Google Scholar]
- Bhattacharya, A.K.; Thyagarajan, O. The Michaelis-Arbuzov Rearrangement. Chem. Rev. 1981, 81, 415–430. [Google Scholar] [CrossRef]
- Trochimczuk, A.W.; Alexandratos, S.D. Synthesis of bifunctional ion-exchange resins through the Arbusov reaction: Effect on selectivity and kinetics. J. Appl. Polym. Sci. 1994, 52, 1273–1277. [Google Scholar] [CrossRef]
- Yu, Z.; Zhu, W.X.; Cabasso, I. Synthesis and polymerization of vinylbenzylphosphonate diethyl ester. J. Polym. Sci. Part A 1990, 28, 227–230. [Google Scholar] [CrossRef]
- Cabasso, I.; Jagur-Grodzinski, J.; Vofsi, D. Synthesis and Characterization of Polymers with Pendent Phosphonate Groups. J. Appl. Polym. Sci. 1974, 18, 1969–1986. [Google Scholar] [CrossRef]
- Kennedy, J.; Burford, F.A.; Sammes, P.G. The selective adosption of hexavalent uranium by a non-ionic phosphorylated resin from solutions of di-normal-butylphosphoric acid in benzene. J. Inorg. Nucl. Chem. 1960, 14, 114. [Google Scholar] [CrossRef]
- Barney, R.J.; Richardson, R.M.; Wiemer, D.F. Direct Conversion of Benzylic and Allylic Alcohols to Phosphonates. J. Org. Chem. 2011, 76, 2875–2879. [Google Scholar] [CrossRef]
- Wyman, P.; Crook, V.L.; Hunt, B.J.; Ebdon, J.R. Improved synthesis of phosphorus-containing styrenic monomers. Des. Monomers Polym. 2004, 7, 301–309. [Google Scholar] [CrossRef]
- Rajalakshmi, K.; Santhana Gopala Krishnan, P.; Nayak, S.K. Synthesis of Dialkyl 2-(Methacryloyloxyethyl) Phosphonates, Their Characterization and Polymerization. Polym. Sci. Ser. B 2015, 57, 408–416. [Google Scholar] [CrossRef]
- Meziane, D.; Hardouin, J.; Elias, A.; Guénin, E.; Lecouvey, M. Microwave Michaelis–Becker Synthesis of Diethyl Phosphonates, Tetraethyl Diphosphonates, and Their Total or Partial Dealkylation. Heteroat. Chem. 2009, 20, 369–377. [Google Scholar] [CrossRef]
- Souzy, R.; Ameduri, B.; Boutevin, B.; Virieux, D. Synthesis of new aromatic perfluorovinyl ether monomers containing phosphonic acid functionality. J. Fluor. Chem. 2004, 125, 1317–1324. [Google Scholar] [CrossRef]
- Queffeélec, C.; Petit, M.; Janvier, P.; Knight, D.A.; Bujoli, B. Surface Modification Using Phosphonic Acids and Esters. Chem. Rev. 2012, 112, 3777–3807. [Google Scholar] [CrossRef]
- Hirao, T.; Masunaga, T.; Ohshiro, Y.; Agawa, T. Stereoselective synthesis of vinylphosphonate. Tetrahedron. Lett. 1980, 21, 3595–3598. [Google Scholar] [CrossRef]
- Hirao, T.; Masunaga, T.; Ohshiro, Y.; Agawa, T. Palladium-catalyzed New Carbon-Phosphorus Bond Formation. Bull. Chem. Soc. Jpn. 1982, 55, 909–913. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Hong, M.-J. Enhanced metal ion affinities by supported ligand synergistic interaction in bifunctional polymer-supported aminomethylphosphonates. Sep. Sci. Technol. 2002, 37, 2587–2605. [Google Scholar] [CrossRef]
- Trochimczuk, A.W.; Jezierska, J. Modification of Malonamide Ion-Exchange Chelating Resins Using the Fields Kabatschnik Reaction and Their Application to Metal Ion Removal from Aqueous Solutions. J. Inorg. Organomet. P 2000, 10, 81–91. [Google Scholar] [CrossRef]
- Papathanasiou, K.E.; Demadis, K.D. Phosphonates in Matrices. In Layered Materials Chemistry: Techniques to Tailor New Enabling Organic-Inorganic Materials; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2015. [Google Scholar]
- Trochimczuk, A.W.; Streat, M. Novel chelating resins with aminothiophosphonate ligands. React. Funct. Polym. 1999, 40, 205–213. [Google Scholar] [CrossRef]
- Jeanmaire, T.; Hervaud, Y.; Boutevin, B. Synthesis of Dialkyl-Hydroxymethylphosphonates in Heterogeneous Conditions. Phosphorus Sulfur Silicon 2002, 177, 1137–1145. [Google Scholar] [CrossRef]
- Kabachnik, M.I.; Medved, T. Vinylphosphonic acid and some of its derivatives. Russ. Chem. Bull. 1959, 8, 2043–2045. [Google Scholar] [CrossRef]
- Macarie, L.; Ilia, G. Poly(vinylphosphonic acid) and its derivatives. Prog. Polym. Sci. 2010, 35, 1078–1092. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Strand, M.A.; Quillen, D.R.; Walder, A.J. Synthesis and Characterization of Bifunctional Phosphinic Acid Resins. Macromolecules 1985, 18, 829. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Wilson, D.L.; Kaiser, P.T. Dual mechanism bifunctional polymers: Phosphinic acid ion-exchange/redox resins and the kinetics of metal-ion complexation. React. Polym. Ion Exch. Sorbents 1987, 5, 23–35. [Google Scholar] [CrossRef]
- Trochimczuk, A.W. Synthesis of functionalized phenylphosphinic acid resins through Michael reaction. Part II. Resins with multidentate ligands. React. Funct. Polym. 2001, 48, 141–148. [Google Scholar] [CrossRef]
- Tavlaridesm, L.L.; Bae, J.H.; Lee, C.K. Solvent Extraction, Membranes, and Ion Exchange in Hydrometallurgical Dilute Metals Separation. Sep. Sci. Technol. 1987, 22, 581–617. [Google Scholar] [CrossRef]
- Beauvais, R.A.; Alexandratos, S.D. Polymer-supported reagents for the selective complexation metal ions: An overview. React. Funct. Polym. 1998, 36, 113–123. [Google Scholar] [CrossRef]
- Ferrel, R.E.; Olcott, H.S.; Fraenkel-Conrat, H. Phosphorylation of Proteins with Phosphoric Acid Containing Excess Phosphorus Pentoxide. J. Am. Chem. Soc. 1948, 70, 2101. [Google Scholar] [CrossRef]
- Trochimczuk, A.W. Synthesis of functionalized phenylphosphinic acid resins through Michael reaction and their ion-exchange properties. React. Funct. Polym. 2000, 44, 9–19. [Google Scholar] [CrossRef]
- Bregman, J.I.Y. Phosphonous and Phosphonic Cation Exchange Resins. J. Am. Chem. Soc. 1952, 74, 1867. [Google Scholar] [CrossRef]
- Boyd, G.E.; Vaslow, F.; Lindenbaum, S.J. Thermodynamic Properties at 25° of Aqueous Solutions of p-EthylbenzenesuIfonic Acid and Its Alkali Metal Salts. Comparisons with Cross-Linked Polystyrenesulfonate Type Cation Exchangers. Phys. Chem. 1967, 71, 3879–3887. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Quillen, D.R.; Bates, M.E. Synthesis and Characterization of Bifunctional Ion-Exchange /Coordination Resins. Macromolecules 1987, 20, 1191–1196. [Google Scholar] [CrossRef]
- Sessler, J.L.; Melfi, P.J.; Pantos, G.D. Uranium complexes of multidentate N-donor ligands. Coordin. Chem. Rev. 2006, 250, 816–843. [Google Scholar] [CrossRef]
- Allard, B.; Olofsson, U.; Torstenfelt, B. Environmental Actinide Chemistry. Inorgan. Chim. Acta 1984, 94, 205–221. [Google Scholar] [CrossRef]
- Tomiyasu, H.; Asano, Y. Environmentally acceptable nuclear fuel cycle—development of a new reprocessing system. Prog. Nucl. Energ. 1998, 32, 421–427. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Zhu, X. High-affinity ion-complexing polymer-supported reagents: Immobilized phosphate ligands and their affinity for the uranyl ion. React. Funct. Polym. 2007, 67, 375–382. [Google Scholar] [CrossRef]
- Aly, M.M.; Hamza, M.F. A Review: Studies on Uranium Removal Using Different Techniques. Overview. J. Disper. Sci. Technol. 2013, 34, 182–213. [Google Scholar] [CrossRef]
- Mincher, B.J.; Modolo, G.; Mezyk, S.P. Review Article: The Effects of Radiation Chemistry on Solvent Extraction: 1. Conditions in Acidic Solution and a Review of TBP Radiolysis, Radiolysis. Solv. Extr. Ion Exc. 2009, 27, 1–25. [Google Scholar] [CrossRef]
- Egawa, H.T.; Ikari, M. Preparation of macroreticular chelating resins containing dihydroxyphosphino and/or phosphono groups and their adsorption ability for uranium. J. Appl. Polym. Sci. 1984, 29, 2045–2055. [Google Scholar] [CrossRef]
- Egawa, H.; Nonaka, T. Recovery of Uranium from Seawater. 7. Concentration and Separation of Uranium in Acidic Eluate. Ind. Eng. Chem. Res. 1990, 29, 2273–2277. [Google Scholar] [CrossRef]
- Kabay, N.; Demircioglu, M.; Yaylı, S.; Gunay, E.; Yuksel, M.; Saglam, M.; Streat, M. Recovery of Uranium from Phosphoric Acid Solutions Using Chelating Ion-Exchange Resins. Ind. Eng. Chem. Res. 1998, 37, 1983–1990. [Google Scholar] [CrossRef]
- Gonzalez-Luque, S.; Streat, M. Uranium sorption from phosphoric acid solutions using selective ion exchange resins: Part, I. Isotherms of extraction and desorption. Hydrometallurgy 1983, 11, 207–225. [Google Scholar] [CrossRef]
- Gonzalez-Luque, S.; Streat, M. Uranium sorption from phosphoric acid solutions using selective ion exchange resins: Part II. Kinetic studies. Hydrometallurgy 1983, 11, 227–245. [Google Scholar] [CrossRef]
- Kim, J.; Tsouris, C.; Mayes, R.T.; Oyola, Y.; Saito, T.; Janke, C.J.; Dai, S.; Schneider, E.; Sachde, D. Recovery of Uranium from Seawater: A Review of Current Status and Future Research Needs. Sep. Sci. Technol. 2013, 48, 367–387. [Google Scholar] [CrossRef]
- Jordens, A.; Cheng, Y.P.; Waters, K.E. A review of the beneficiation of rare earth element bearing minerals. Miner. Eng. 2013, 41, 97–114. [Google Scholar] [CrossRef]
- Jha, M.K.; Kumari, A.; Panda, R.; Kumar, J.R.; Yo, K.K.; Lee, J.Y. Review on hydrometallurgical recovery of rare earth metals. Hydrometallurgy 2016, 165, 2–26. [Google Scholar] [CrossRef]
- Xie, F.; Zhang, T.A.; Dreisinger, D.; Doyle, F. A critical review on solvent extraction of rare earths from aqueous solutions. Miner. Eng. 2014, 56, 10–28. [Google Scholar] [CrossRef]
- Powell, J.E. The Separation of Rare Earths by Ion Exchange. In Progress in the Science and Technology of the Rare Earths; Eyring, L., Ed.; Pergamon Press: Oxford, UK, 1964. [Google Scholar]
- Kabay, N.; Cortina, J.L.; Trochimczuk, A.; Streat, M. Solvent-impregnated resins (SIRs)–Methods of preparation and their applications. React. Funct. Polym. 2010, 70, 484–496. [Google Scholar] [CrossRef]
- Lederer, M. Chromatography on paper impregnated with ion-exchange resins: III. The separation of some cation mixtures. J. Chromatogr. A 1958, 1, 314–316. [Google Scholar] [CrossRef]
- Juang, R.S. Preparation, Properties and Sorption Behavior of Impregnated Resins Containing Acidic Organophosphorus Extractants. Proc. Nat. Sci. Counc. Rep. Chin. Part A 1999, 23, 353–364. [Google Scholar]
- Alexandratos, S.D.; Bates, M.E. Enhanced Ionic Recognition by Polymer-Supported Reagents: Synthesis and Characterization of Ion-Exchange/ Precipitation Resins. Macromolecules 1988, 21, 2905–2910. [Google Scholar] [CrossRef]
- Park, I.-H.; Rhee, J.M.; Jung, Y.S. Synthesis and heavy metal ion adsorptivity of macroreticular chelating resins containing phosphono and carboxylic acid groups. Die Angew. Makromol. Chem. 1999, 267, 27–34. [Google Scholar] [CrossRef]
- Horwitz, E.P.; Chiarizia, R.; Diamond, H.; Gatrone, R.C.; Alexandratos, S.D.; Trochimczuk, A.W.; Crick, D.W. Uptake of Metal Ions by A New Chelating Ion Exchange Resin Part 1: Acid Dependencies of Actinide Ions. Solvent Extr. Ion Exc. 1993, 11, 943–966. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Crick, D.W.; Quillen, D.R. Development of Bifunctional Polymers for Metal Ion Separations: Ionic Recognition with Polymer-Supported Reagents. Ind. Eng. Chem. Res. 1991, 30, 772–778. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Bates, M.; Walder, J.; McDowell, W.J. Novel bifunctional resins in metal ion separations: Ion exchange/coordination resins and ion exchange/precipitation resins. Sep. Sci. Technol. 1988, 23, 1915–1927. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Trochimczuk, A.W.; Horwitz, E.P.; Gatrone, R.C. Synthesis and Characterization of a Bifunctional Ion Exchange Resin with Polystyrene-Immobilized Diphosphonic Acid Ligands. J. Appl. Polym. Sci. 1996, 61, 273–278. [Google Scholar] [CrossRef]
- Trochimczuk, A.W.; Horwitz, E.P.; Alexandratos, S.D. Complexing Properties of Diphonix, a New Chelating Resin with Diphosphonate Ligands, Toward Ga(III) and in(III). Sep. Sci. Technol. 1994, 29, 543–549. [Google Scholar] [CrossRef]
- Chiarizia, R.; Horwitz, E.P.; D’Arcy, K.A.; Alexandratos, S.D.; Trochimczuk, A.W. Uptake of Metal Ions by A New Chelating Ion Exchange Resin. Part 9: Silica Grafted Diphosphonic Acid. Solvent Extr. Ion Exc. 1996, 14, 1077–1100. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Trochimczuk, A.W.; Crick, D.W.; Horwitz, E.P.; Gatrone, R.C.; Chiarizia, R. Synthesis and Ion-Complexing Properties of a Novel Polymer-Supported Reagent with Diphosphonate Ligands. Macromolecules 1996, 29, 1021–1026. [Google Scholar] [CrossRef]
- Chiarizia, R.; Horwitz, E.P.; D’Arcy, K.A.; Alexandratos, S.D.; Trochimczuk, A.W. Uptake of metal ions by a new chelating ion exchange resin. Part 8. Simultaneous uptake of cationic and anionic species. Solvent Extr. Ion Exc. 1996, 14, 519–542. [Google Scholar] [CrossRef]
- Alexandratos, S.D.; Zhu, X. The role of polarizability in determining metal ion affinities in polymer-supported reagents: Monoprotic phosphates and the effect of hydrogen bonding. New J. Chem. 2015, 39, 5366. [Google Scholar] [CrossRef]
- Nash, K.L.; Rickert, P.G.; Muntean, J.V.; Alexandratos, S.D. Uptake of Metal Ions by A New Chelating Ion Exchange Resin Part 3: Protonation Constant Via Potentiometric Titration and Solid State 31P NMR Spectroscopy. Solvent Extr. Ion Exc. 1994, 12, 193–209. [Google Scholar] [CrossRef]
- Chiarizia, R.; Horwitz, E.P.; Alexandratos, S.D. Uptake of Metal Ions by A New Chelating Ion Exchange Resin Part 4: Kinetics. Solvent Extr. Ion Exc. 1994, 12, 211–237. [Google Scholar] [CrossRef]
- Chiarizia, R.; Horwitz, E.P.; Alexandratos, S.D. Uptake of Metal Ions by A New Chelating Ion Exchange Resin Part 5: The Effect of Solution Matrix on Actinides. Solvent Extr. Ion Exc. 1994, 12, 831–845. [Google Scholar]
- Chiarizia, R.; Horwitz, E.P.; Gatrone, R.C.; Alexandratos, S.D.; Trochimczuk, A.W.; Crick, D.W. Uptake of Metal Ions by A New Chelating Ion Exchange Resin, Part 2: Acid Dependencies of Transition And Posttransition Metal-Ions. Solvent Extr. Ion Exc. 1993, 11, 967–985. [Google Scholar] [CrossRef]
- Kilislioglu, A. Ion Exchange Technologies; IntechOpen: London, UK, 2012; ISBN 978-953-51-0836-8. [Google Scholar]
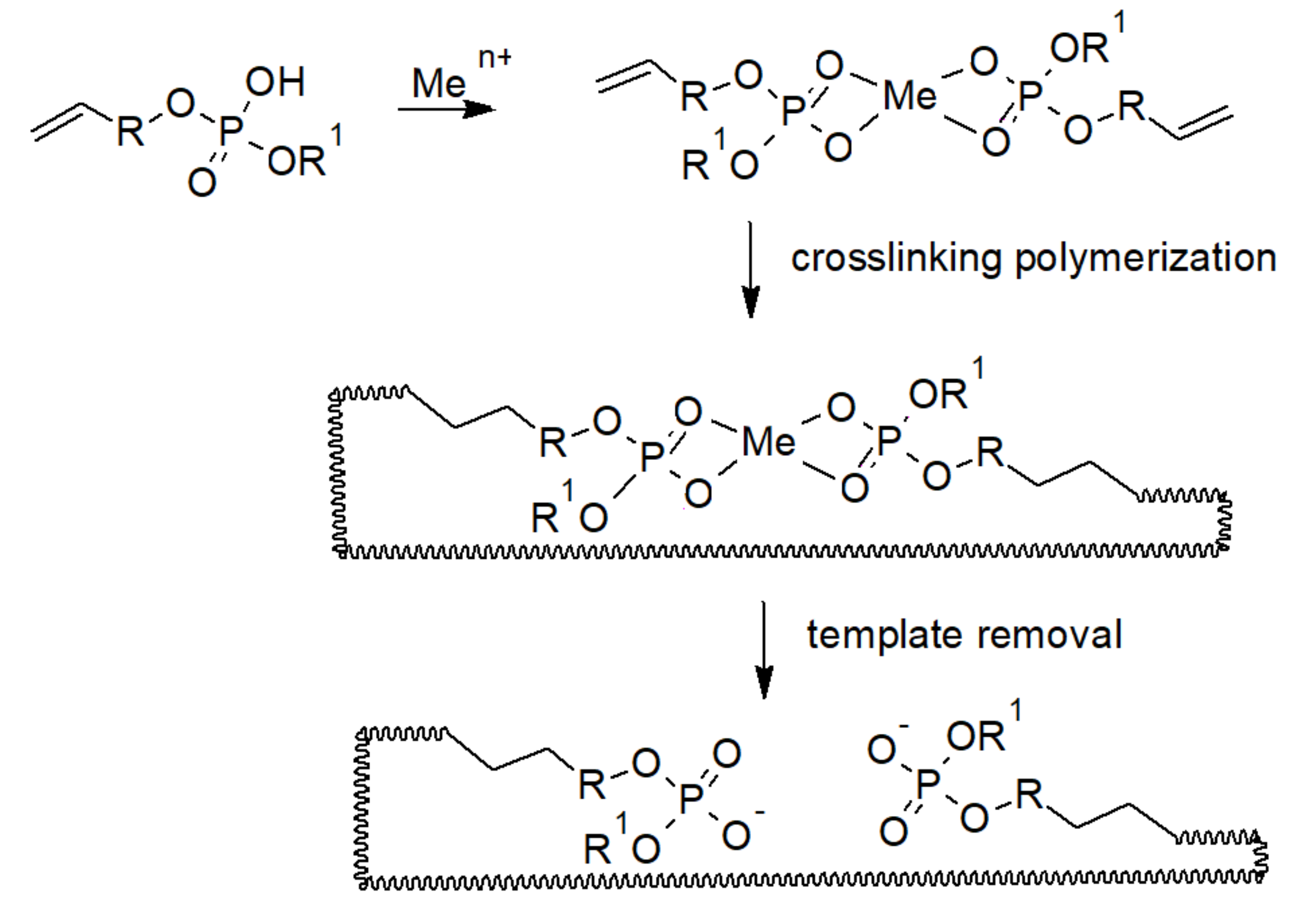
- Yoshida, M.; Uezu, K.; Goto, M.; Furusaki, S. Metal Ion Imprinted Microsphere Prepared by Surface Molecular Imprinting Technique Using Water-in-Oil-in-Water Emulsions. J. Appl. Polym. Sci. 1999, 73, 1223–1230. [Google Scholar] [CrossRef]
- Yoshida, M.; Hatate, Y.; Uezu, K.; Goto, M.; Furusaki, S. Metal-Imprinted Microsphere Prepared by Surface Template Polymerization and Its Application to Chromatography. J. Polym. Sci. Part A 2000, 38, 689–696. [Google Scholar] [CrossRef][Green Version]


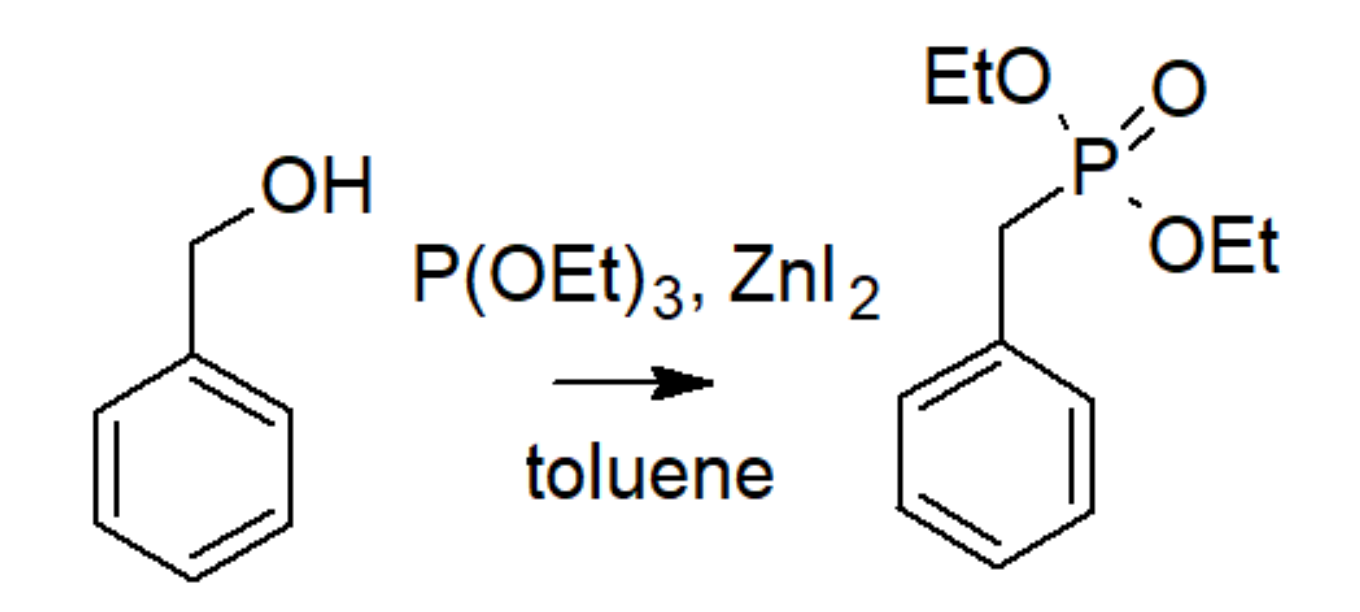
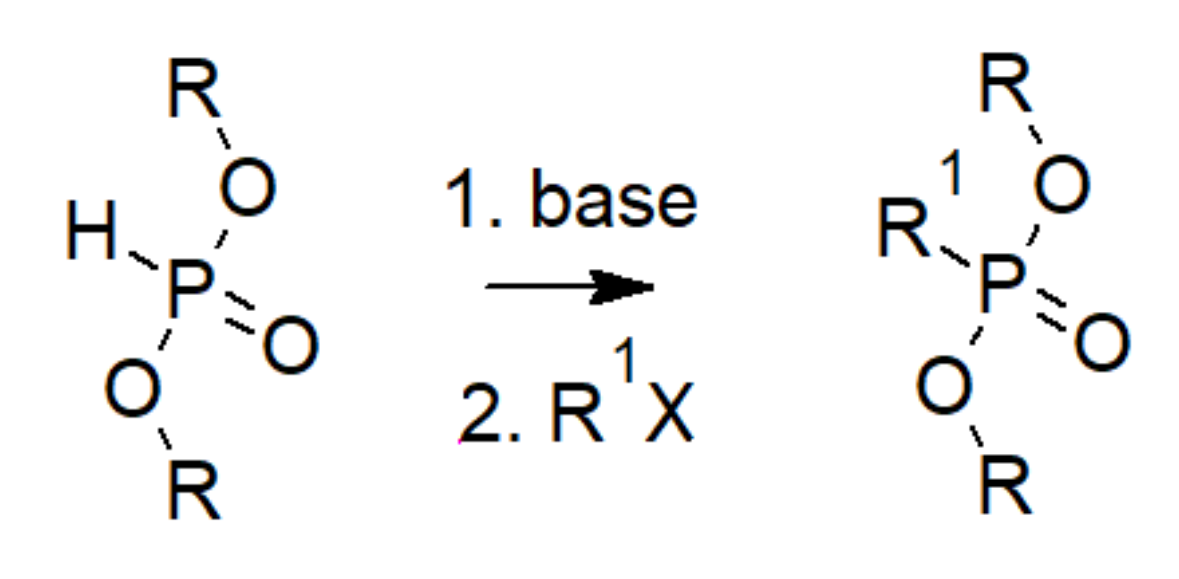
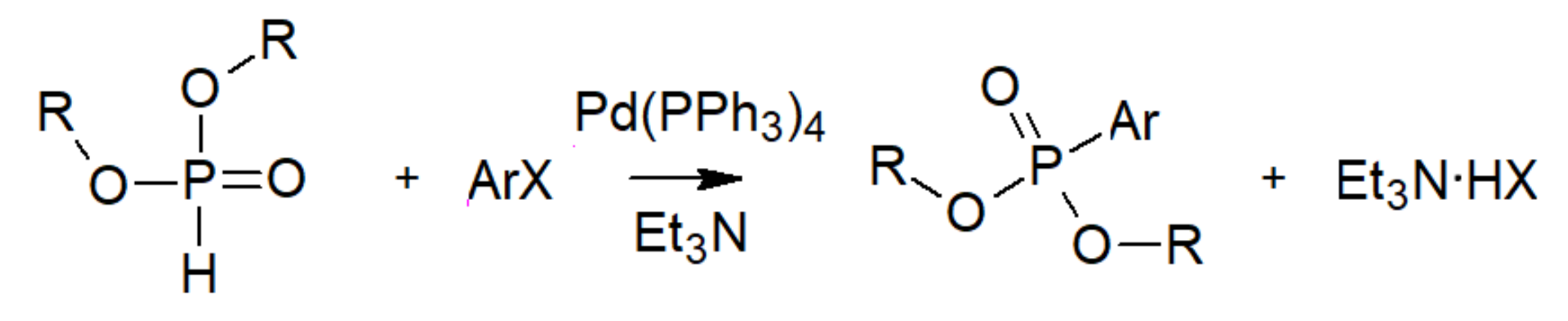

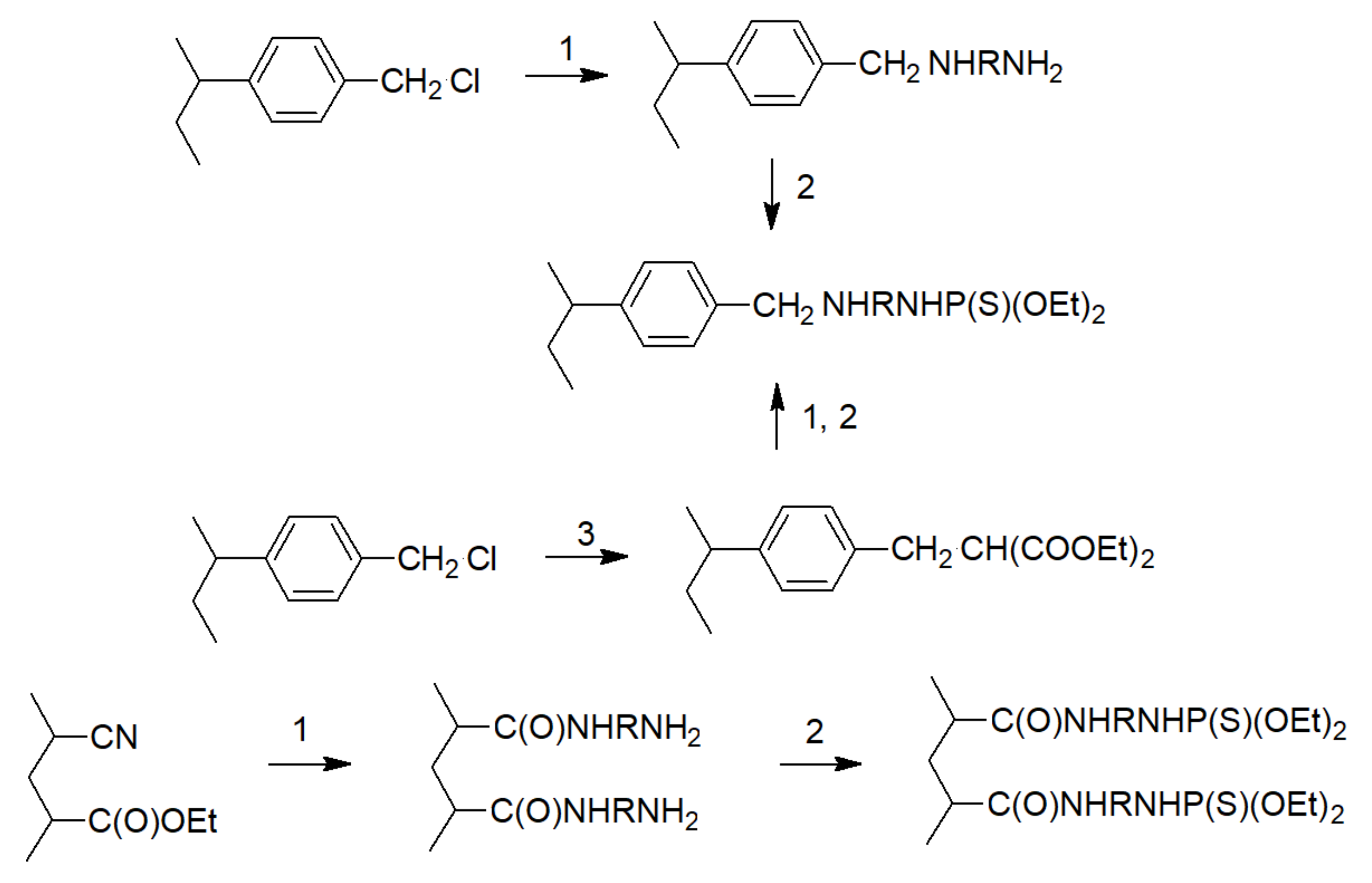
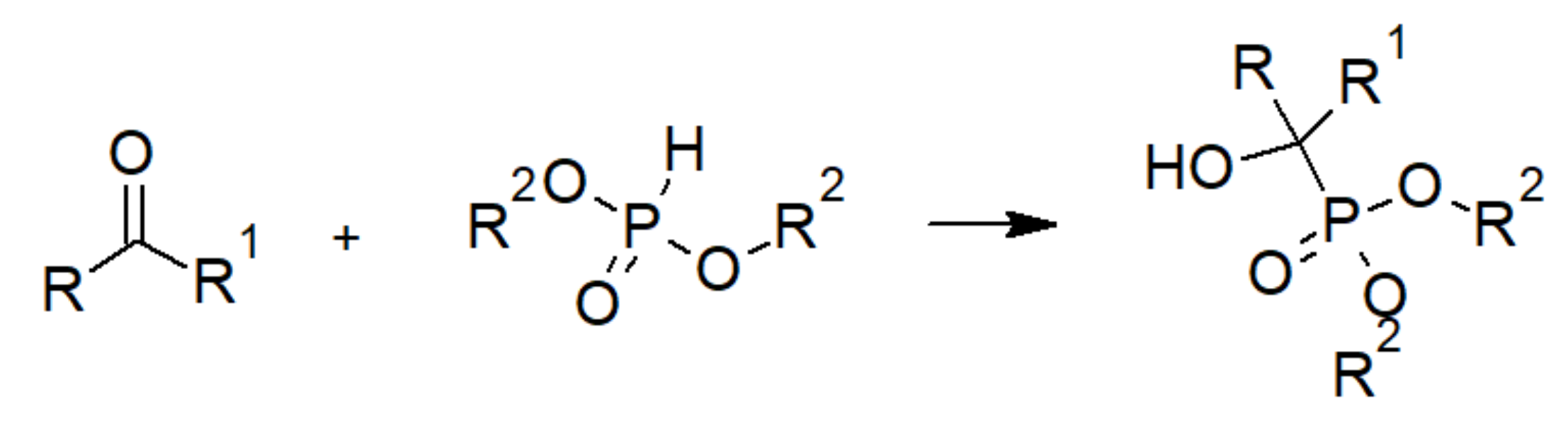

| Bond | Bond Energy [kJ/mol] |
|---|---|
| P-H | 322 |
| P-P | 209 |
| P-C | 272 |
| P-O | 360 |
| P=O | 586 |
| P-N | 230 |
| P=C | 578 |
| P≡P | 490 |
| P=N | 687 |
| P-Cl | 321 |
| P-F | 527 |
| P-I | 184 |
| P-Br | 264 |
| P-Containing Functional Group | Polymer | Application/Features | References |
|---|---|---|---|
| ‘Phosphoric acid | Cellulose phosphate | Sorption from 4N-acid: high affinity for Th4+, Ti4+, U4+, Ce4+, Fe3+, ZrO2+, UO22+ | [29] |
| Cation exchanger in calcium-hydrogen cycle | [30] | ||
| Adsorption of proteins | [33] | ||
| Poly(glycidyl methacrylate-co-divinylbenzene) functionalized with phosphoric acid | Selective resins (lithium adsorbed in preference to sodium and potassium, better adsorption of hard-Lewis cations from acidic media, highest selectivity towards lead) | [38] | |
| Poly(glycidyl methacrylate-co-divinylbenzene) and poly(glycidyl methacrylate) functionalized with phosphoric acid or phosphorus oxychloride | Resins with high cation exchange capacity, salt splitting capacity, and adsorption capacity for Cu2+, Zn2+, Cd2+, Ca2+, and Ag+; RGP obtained from poly(glycidyl methacrylate) – high adsorption capacity for Al3+, Fe3+, and UO22+; phosphoric acid functionalization – higher selective adsorption for Li+ than for Na+ | [40] | |
| Phosphonic acid | D2EHPA/TOPO impregnated resins, ion exchange resin containing aminophosphonic acid groups | Uranium removal – D2EHPA/TOPO resins are not affected by the presence of Fe2+, which reduces uranium to the tetravalent oxidation state, prevents uranium sorption. | [89,90] |
| α-, β -, and γ-ketophosphonate polymer-supported resins | Sorption of metal ions (Eu(III), Cu(II), Pb(II), Cd(II), Co(II), Ag(II)) from nitric acid | [41] | |
| Bifunctional ion-exchange resins synthesized from vinylbenzyl chloride– styrene copolymers: Arbuzov reaction followed by sulfonation | Eu(III) sorption from acidic solutions | [51] | |
| Non-ionic phosphorylated resin diethyl polystyrene-methylenephosphonate (EPMP) | Selective adsorption of hexavalent uranium | [54] | |
| Bifunctional aminomethylphosphonic acid resins (Mannich reaction) | Sorption of Cu(II), Cd(II), Pb(II), and Eu(III)—cadmium ion complexation shows the greatest bifunctional effect, | [63] | |
| Ion-exchange/chelating resins with aminomethylphosphonate groups (Fields-Kabatschnik reaction) | The removal of Cu(II), Cd(II), Ni(II), and Zn(II) | [64] | |
| Diphonix® | Uranium removal (after 2h—20.7%, 24h—18.0% from 40% H3PO4) | [68] | |
| Actinides ion removal from very acidic solutions | [101] | ||
| Uptake of various transition and post-transition metal ions; a very high affinity for Fe(III) and Cr(III) in very acidic solutions | [113] | ||
| Gallium(III) and indium(III) recovery | [105] | ||
| Chelating diphosphonic acid groups are grafted to a silica support to overcome some of the problems associated with the stripping of actinide elements from the resin (Diphosil) | [106] | ||
| Diphonix A – additional strong base anion exchange groups; the uptake of Se(IV), tri-, tetra- and hexa-valent actinides and pertechnetate anions | [108] | ||
| The of uptake of several actinide ions [Am(III), U(VI), Th(IV), Np(IV) and Pu(IV)] and of some transition-metal ions [Co(II), Zn(II), Fe(III) and Cr(III)] at tracer concentration level; | [111] | ||
| Bifunctional resin consisting of phosphonic acid and quaternary amine groups | Sorption of barium, calcium, silver cations; changing the associated anion allows for control of the ionic recognition process | [99] | |
| Poly(vinylbenzyl chloride-co-styrene-co-divinylbenzene) functionalized with the sodium salt of tetra(isopropyl) methylene diphosphonate | Eu(III) sorption; highly selective resins with rapid complexation kinetics: 99.7% Eu(III) complexed from a 1M nitric acid solution with a 30-min contact time | [104] | |
| Diphosphonate | Chelating resin with diphosphonate groups synthesized from the tetraethylester of 1,1-vinylidenediphosphonic acid | Eu(III) sorption: sulfonic acid ion exchange resin complexes 14.3% Eu(III), diphosphonate-based polymer complexes 96.5% from a 1N HNO3/0.4N NaNO3 solution | [107] |
| Phosphate and phosphonate diesters | Polymer-supported phosphonate and phosphate diester ligands | The complexation of uranium(VI) e.g., the expanded gel resin - phosphorylated pentaerythritol: 99% complexation within 20 min | [83] |
| Polystyrene-divinylbenzene matrix with methylenediphosphonate, carboxylate, and sulfonate functional groups | Exceptionally high affinity for polyvalent cations even from moderately acidic aqueous media | [110] | |
| Phosphonic acid, phosphonate ester | Resins with phosphonic acid/phosphonate ester ligands and phosphonic acid/tertiary amine ligand on PS support | Americum extraction | [79] |
| Phosphonates, phosphinic acid | Bifunctional polymer resins (I DMBP, II DMBP, compared with monofunctional resins) | Metal-specific reactions, polymers with high level of ionic recognition; phosphinic ligands—ability to control the recognition mechanism through the reduction reaction with Hg(II); phosphonic resin—sorption of Ag, Hg and Fe in different conditions—high complexation of Fe; complexation of Fe is adversely affected by the presence of diester ligands | [102] |
| Phosphinic acid | Bifunctional phosphinic acid ion-exchange/redox resins | Dual mechanism sorption (ion-exchange and metal-ion reduction) of Ag(I) and Hg(II): after a 9-h contact time with Ag(I) ions and a 2-h contact time with Hg(II) ions all of the primary acid sites are oxidized | [71] |
| Phenylphosphinic acid resins (Michael reaction) | the removal of Cu(II), Cd(II), Ni(II), Zn(II), Mg(II) and Ca(II) from nitric acid solutions | [72,76] | |
| Dihydroxyphosphino- and/or phosphono groups | The macroreticular chelating resins containing dihydroxy-phosphino and/or phosphono groups | Recovery of uranium from sea water—e.g., the average recovery ratios of uranium from sea water with Na-form and H-form RCSP on 10 recycles: 84.9% and 90.5%, respectively | [86,87] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Głowińska, A.; Trochimczuk, A.W. Polymer-Supported Phosphoric, Phosphonic and Phosphinic Acids—From Synthesis to Properties and Applications in Separation Processes. Molecules 2020, 25, 4236. https://doi.org/10.3390/molecules25184236
Głowińska A, Trochimczuk AW. Polymer-Supported Phosphoric, Phosphonic and Phosphinic Acids—From Synthesis to Properties and Applications in Separation Processes. Molecules. 2020; 25(18):4236. https://doi.org/10.3390/molecules25184236
Chicago/Turabian StyleGłowińska, Agnieszka, and Andrzej W. Trochimczuk. 2020. "Polymer-Supported Phosphoric, Phosphonic and Phosphinic Acids—From Synthesis to Properties and Applications in Separation Processes" Molecules 25, no. 18: 4236. https://doi.org/10.3390/molecules25184236
APA StyleGłowińska, A., & Trochimczuk, A. W. (2020). Polymer-Supported Phosphoric, Phosphonic and Phosphinic Acids—From Synthesis to Properties and Applications in Separation Processes. Molecules, 25(18), 4236. https://doi.org/10.3390/molecules25184236







