The Impact of Electronic Nicotine Delivery System (ENDS) Flavors on Nicotinic Acetylcholine Receptors and Nicotine Addiction-Related Behaviors
Abstract
1. Introduction
2. Background of Nicotine Addiction
2.1. Neuronal nAChRs: Structure and Function
2.2. Nicotine’s Actions in the Brain
2.3. nAChR Subtypes
2.3.1. α4β2* nAChRs
2.3.2. α6β2β3* nAChRs
2.3.3. α4α6β2* nAChRs
2.3.4. α7 nAChRs
2.3.5. α3* and α5* nAChRs
3. Neurocircuitry Involved in Nicotine Addiction
3.1. Ventral Tegmental Area (VTA)
3.2. Substantia Nigra (SN)
3.3. Nucleus Accumbens (NAc)
3.4. Prefrontal Cortex (PFC)
3.5. Hippocampus (HIPP)
3.6. Habenula (Hb)
4. Flavoring Chemicals in Nicotine Addiction
5. Future Directions
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- SGR. Reports of the Surgeon General. In The Health Consequences of Smoking-50 Years of Progress: A Report of the Surgeon General; Centers for Disease Control and Prevention (US): Atlanta, GA, USA, 2014. [Google Scholar]
- Babb, S.; Malarcher, A.; Schauer, G.; Asman, K.; Jamal, A. Quitting Smoking among Adults—United States, 2000–2015. Mmwr. Morb. Mortal. Wkly. Rep. 2017, 65, 1457–1464. [Google Scholar] [CrossRef] [PubMed]
- Office on Smoking and Health. Publications and Reports of the Surgeon General. In Women and Smoking: A Report of the Surgeon General; Centers for Disease Control and Prevention (US): Atlanta, GA, USA, 2001. Available online: https://www.hhs.gov/surgeongeneral/reports-and-publications/tobacco/index.html (accessed on 12 May 2020).
- FTC Cigarette Report for 2018. Available online: https://www.ftc.gov/system/files/documents/reports/federal-trade-commission-cigarette-report-2018-smokeless-tobacco-report-2018/p114508cigarettereport2018.pdf (accessed on 27 March 2020).
- Ai, J.; Taylor, K.M.; Lisko, J.G.; Tran, H.; Watson, C.H.; Holman, M.R. Menthol Content in US Marketed Cigarettes. Nicotine Tob. Res. 2015, 18, 1575–1580. [Google Scholar] [CrossRef] [PubMed]
- Glasser, A.M.; Collins, L.; Pearson, J.L.; Abudayyeh, H.; Niaura, R.S.; Abrams, D.B.; Villanti, A.C.; Katz, L. Overview of Electronic Nicotine Delivery Systems: A Systematic Review. Am. J. Prev. Med. 2017, 52, e33–e66. [Google Scholar] [CrossRef]
- SGR. Reports of the Surgeon General. In Facing Addiction in America: The Surgeon General’s Report on Alcohol, Drugs, and Health; US Department of Health and Human Services: Washington, DC, USA, 2016. [Google Scholar]
- SGR. Publications and Reports of the Surgeon General. In E-Cigarette Use Among Youth and Young Adults: A Report of the Surgeon General; Centers for Disease Control and Prevention (US): Atlanta, GA, USA, 2016. [Google Scholar]
- Omaiye, E.E.; McWhirter, K.J.; Luo, W.; Tierney, P.A.; Pankow, J.F.; Talbot, P. High concentrations of flavor chemicals are present in electronic cigarette refill fluids. Sci. Rep. 2019, 9, 2468. [Google Scholar] [CrossRef] [PubMed]
- Office on Smoking and Health. 2019 National Youth Tobacco Survey: Methodology Report; Department of Health and Human Services, Centers for Disease Control and Prevention National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health: Atlanta, GA, USA, 2019. Available online: https://www.cdc.gov/tobacco/data_statistics/surveys/nyts/index.htm (accessed on 8 September 2020).
- Cullen, K.A.; Gentzke, A.S.; Sawdey, M.D.; Chang, J.T.; Anic, G.M.; Wang, T.W.; Creamer, M.R.; Jamal, A.; Ambrose, B.K.; King, B.A. E-cigarette Use Among Youth in the United States, 2019. JAMA 2019, 322, 2095. [Google Scholar] [CrossRef]
- Mead, E.; Duffy, V.B.; Oncken, C.; Litt, M.D. E-cigarette palatability in smokers as a function of flavorings, nicotine content and propylthiouracil (PROP) taster phenotype. Addict. Behav. 2019, 91, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Willis, D.N.; Liu, B.; Ha, M.A.; Jordt, S.-E.; Morris, J.B. Menthol attenuates respiratory irritation responses to multiple cigarette smoke irritants. FASEB J. 2011, 25, 4434–4444. [Google Scholar] [CrossRef]
- Zhu, S.-H.; Sun, J.Y.; Bonnevie, E.E.; Cummins, S.; Gamst, A.; Yin, L.; Lee, M. Four hundred and sixty brands of e-cigarettes and counting: Implications for product regulation. Tob. Control. 2014, 23, iii3–iii9. [Google Scholar] [CrossRef]
- Hsu, G.; Sun, J.Y.; Zhu, S.-H.; Allem, J.-P.; Chen, J.C.; Gundel, L. Evolution of Electronic Cigarette Brands From 2013–2014 to 2016–2017: Analysis of Brand Websites. J. Med Internet Res. 2018, 20, e80. [Google Scholar] [CrossRef]
- Wang, T.; Wang, B.; Chen, H. Menthol facilitates the intravenous self-administration of nicotine in rats. Front. Behav. Neurosci. 2014, 8, 437. [Google Scholar] [CrossRef]
- Biswas, L.; Harrison, E.; Gong, Y.; Avusula, R.; Lee, J.; Zhang, M.; Rousselle, T.; Lage, J.; Liu, X. Enhancing effect of menthol on nicotine self-administration in rats. Psychopharmacology 2016, 233, 3417–3427. [Google Scholar] [CrossRef] [PubMed]
- Henderson, B.J.; Wall, T.R.; Henley, B.M.; Kim, C.H.; McKinney, S.; Lester, H.A. Menthol Enhances Nicotine Reward-Related Behavior by Potentiating Nicotine-Induced Changes in nAChR Function, nAChR Upregulation, and DA Neuron Excitability. Neuropsychopharmacology 2017, 42, 2285–2291. [Google Scholar] [CrossRef] [PubMed]
- Cooper, S.Y.; Akers, A.T.; Henderson, B.J. Flavors enhance nicotine vapor self-administration in male mice. Nicotine Tob. Res. 2020. [Google Scholar] [CrossRef]
- Henderson, B.J.; Wall, T.R.; Henley, B.M.; Kim, C.H.; Nichols, W.A.; Moaddel, R.; Xiao, C.; Lester, H.A. Menthol Alone Upregulates Midbrain nAChRs, Alters nAChR Subtype Stoichiometry, Alters Dopamine Neuron Firing Frequency, and Prevents Nicotine Reward. J. Neurosci. 2016, 36, 2957–2974. [Google Scholar] [CrossRef]
- Brody, A.L.; Mukhin, A.G.; La Charite, J.; Ta, K.; Farahi, J.; Sugar, C.A.; Mamoun, M.S.; Vellios, E.; Archie, M.; Kozman, M.; et al. Up-regulation of nicotinic acetylcholine receptors in menthol cigarette smokers. Int. J. Neuropsychopharmacol. 2013, 16, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Henderson, B.J.; Grant, S.; Chu, B.W.; Shahoei, R.; Huard, S.M.; Saladi, S.S.M.; Tajkhorshid, E.; Dougherty, D.A.; Lester, H.A. Menthol stereoisomers exhibit different effects on a4b2 nAChR upregulation and dopamine neuron spontaneous firing. eNeuro 2018. [Google Scholar] [CrossRef]
- Zhang, M.; Harrison, E.; Biswas, L.; Tran, T.; Liu, X. Menthol facilitates dopamine-releasing effect of nicotine in rat nucleus accumbens. Pharmacol. Biochem. Behav. 2018, 175, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Balakrishna, S.; Jabba, S.V.; Bonner, P.E.; Taylor, S.R.; Picciotto, M.R.; Jordt, S.-E. Menthol decreases oral nicotine aversion in C57BL/6 mice through a TRPM8-dependent mechanism. Tob. Control. 2016, 25, ii50–ii54. [Google Scholar] [CrossRef]
- Espino-Díaz, M.; Sepúlveda, D.R.; González-Aguilar, G.; Olivas, G.I. Biochemistry of Apple Aroma: A Review. Food Technol. Biotechnol. 2016, 54, 375–397. [Google Scholar] [CrossRef]
- Avelar, A.J.; Akers, A.T.; Baumgard, Z.J.; Cooper, S.Y.; Casinelli, G.P.; Henderson, B.J. Why flavored vape products may be attractive: Green apple tobacco flavor elicits reward-related behavior, upregulates nAChRs on VTA dopamine neurons, and alters midbrain dopamine and GABA neuron function. Neuropharmacology 2019, 158, 107729. [Google Scholar] [CrossRef]
- Cooper, S.Y.; Akers, A.T.; Henderson, B.J. Green apple e-cigarette flavorant farnesene triggers reward-related behavior by promoting high-sensitivity nAChRs in the ventral tegmental area. Eneuro 2020. [Google Scholar] [CrossRef] [PubMed]
- Sine, S.M.; Engel, A.G. Recent advances in Cys-loop receptor structure and function. Nature 2006, 440, 448–455. [Google Scholar] [CrossRef]
- Le Novère, N.; Changeux, J.-P. Molecular evolution of the nicotinic acetylcholine receptor: An example of multigene family in excitable cells. J. Mol. Evol. 1995, 40, 155–172. [Google Scholar] [CrossRef]
- Albuquerque, E.X.; Pereira, E.F.R.; Alkondon, M.; Rogers, S.W. Mammalian Nicotinic Acetylcholine Receptors: From Structure to Function. Physiol. Rev. 2009, 89, 73–120. [Google Scholar] [CrossRef]
- Dang, N.; Meng, X.; Song, H. Nicotinic acetylcholine receptors and cancer. Biomed. Rep. 2016, 4, 515–518. [Google Scholar] [CrossRef] [PubMed]
- Zoli, M.; Pistillo, F.; Gotti, C. Diversity of native nicotinic receptor subtypes in mammalian brain. Neuropharmacology 2015, 96, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Penton, R.E.; Lester, R.A. Cellular events in nicotine addiction. Semin. Cell Dev. Boil. 2009, 20, 418–431. [Google Scholar] [CrossRef]
- Gregg, B.W.; Wells, G.B. Structural answers and persistent questions about how nicotinic receptors work. Front. Biosci. 2008, 13, 5479–5510. [Google Scholar] [CrossRef]
- Henderson, B.J.; Srinivasan, R.; Nichols, W.A.; Dilworth, C.N.; Gutierrez, D.F.; Mackey, E.D.; McKinney, S.; Drenan, R.M.; Richards, C.I.; Lester, H.A. Nicotine exploits a COPI-mediated process for chaperone-mediated up-regulation of its receptors. J. Gen. Physiol. 2013, 143, 51–66. [Google Scholar] [CrossRef]
- Wiesner, A.; Führer, C. Regulation of nicotinic acetylcholine receptors by tyrosine kinases in the peripheral and central nervous system: Same players, different roles. Cell. Mol. Life Sci. 2006, 63, 2818–2828. [Google Scholar] [CrossRef]
- Srinivasan, R.; Pantoja, R.; Moss, F.J.; Mackey, E.D.; Son, C.D.; Miwa, J.M.; Lester, H.A. Nicotine up-regulates α4β2 nicotinic receptors and ER exit sites via stoichiometry-dependent chaperoning. J. Gen. Physiol. 2010, 137, 59–79. [Google Scholar] [CrossRef] [PubMed]
- Gotti, C.; Clementi, F.; Fornari, A.; Gaimarri, A.; Guiducci, S.; Manfredi, I.; Moretti, M.; Pedrazzi, P.; Pucci, L.; Zoli, M. Structural and functional diversity of native brain neuronal nicotinic receptors. Biochem. Pharmacol. 2009, 78, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Tapia, L.; Kuryatov, A.; Lindström, J. Ca2+ Permeability of the (α4)3(β2)2 Stoichiometry Greatly Exceeds That of (α4)2(β2)3 Human Acetylcholine Receptors. Mol. Pharmacol. 2006, 71, 769–776. [Google Scholar] [CrossRef]
- Zhao, L.; Kuo, Y.-P.; George, A.A.; Peng, J.-H.; Purandare, M.S.; Schroeder, K.M.; Lukas, R.J.; Wu, J. Functional Properties of Homomeric, Human α7-Nicotinic Acetylcholine Receptors Heterologously Expressed in the SH-EP1 Human Epithelial Cell Line. J. Pharmacol. Exp. Ther. 2003, 305, 1132–1141. [Google Scholar] [CrossRef] [PubMed]
- Conroy, W.G.; Ogden, L.F.; Berg, D.K. Cluster formation of α7-containing nicotinic receptors at interneuronal interfaces in cell culture. Neuropharmacology 2000, 39, 2699–2705. [Google Scholar] [CrossRef]
- Zoli, M.; Pucci, S.; Vilella, A.; Gotti, C. Neuronal and Extraneuronal Nicotinic Acetylcholine Receptors. Curr. Neuropharmacol. 2018, 16, 338–349. [Google Scholar] [CrossRef]
- McGehee, D.; Heath, M.; Gelber, S.; DeVay, P.; Role, L. Nicotine enhancement of fast excitatory synaptic transmission in CNS by presynaptic receptors. Science 1995, 269, 1692–1696. [Google Scholar] [CrossRef]
- Jensen, A.A.; Froelund, B.; Liljefors, T.; Krogsgaard-Larsen, P. Neuronal Nicotinic Acetylcholine Receptors: Structural Revelations, Target Identifications, and Therapeutic Inspirations. J. Med. Chem. 2005, 48, 4705–4745. [Google Scholar] [CrossRef]
- Picciotto, M.R.; Addy, N.A.; Mineur, Y.S.; Brunzell, D.H. It is not “either/or”: Activation and desensitization of nicotinic acetylcholine receptors both contribute to behaviors related to nicotine addiction and mood. Prog. Neurobiol. 2008, 84, 329–342. [Google Scholar] [CrossRef]
- Alkondon, M.; Albuquerque, E.X. Nicotinic acetylcholine receptor alpha7 and alpha4beta2 subtypes differentially control GABAergic input to CA1 neurons in rat hippocampus. J. Neurophysiol. 2001, 86, 3043–3055. [Google Scholar] [CrossRef]
- Dani, J.A. Neuronal Nicotinic Acetylcholine Receptor Structure and Function and Response to Nicotine. Int. Rev. Neurobiol. 2015, 124, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Picciotto, M.R.; Kenny, P.J. Molecular Mechanisms Underlying Behaviors Related to Nicotine Addiction. Cold Spring Harb. Perspect. Med. 2012, 3, a012112. [Google Scholar] [CrossRef] [PubMed]
- Molas, S.; Dierssen, M. The role of nicotinic receptors in shaping and functioning of the glutamatergic system: A window into cognitive pathology. Neurosci. Biobehav. Rev. 2014, 46, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Shivange, A.V.; Borden, P.M.; Muthusamy, A.K.; Nichols, A.L.; Bera, K.; Bao, H.; Bishara, I.; Jeon, J.; Mulcahy, M.J.; Cohen, B.; et al. Determining the pharmacokinetics of nicotinic drugs in the endoplasmic reticulum using biosensors. J. Gen. Physiol. 2019, 151, 738–757. [Google Scholar] [CrossRef] [PubMed]
- Lester, H.A.; Xiao, C.; Srinivasan, R.; Son, C.D.; Miwa, J.M.; Pantoja, R.; Banghart, M.R.; Dougherty, D.A.; Goate, A.; Wang, J.C. Nicotine is a Selective Pharmacological Chaperone of Acetylcholine Receptor Number and Stoichiometry. Implications for Drug Discovery. AAPS J. 2009, 11, 167–177. [Google Scholar] [CrossRef]
- Kuryatov, A.; Luo, J.; Cooper, J.; Lindstrom, J. Nicotine acts as a pharmacological chaperone to up-regulate human α4β2 acetylcholine receptors. Mol. Pharmacol. 2005, 68, 1839–1851. [Google Scholar] [CrossRef]
- Nashmi, R.; Xiao, C.; Deshpande, P.; McKinney, S.; Grady, S.R.; Whiteaker, P.; Huang, Q.; McClure-Begley, T.; Lindstrom, J.M.; Labarca, C.; et al. Chronic Nicotine Cell Specifically Upregulates Functional α4* Nicotinic Receptors: Basis for Both Tolerance in Midbrain and Enhanced Long-Term Potentiation in Perforant Path. J. Neurosci. 2007, 27, 8202–8218. [Google Scholar] [CrossRef]
- Sallette, J.; Pons, S.; Devillers-Thiéry, A.; Soudant, M.; De Carvalho, L.P.; Changeux, J.-P.; Corringer, P.J. Nicotine Upregulates Its Own Receptors through Enhanced Intracellular Maturation. Neuron 2005, 46, 595–607. [Google Scholar] [CrossRef]
- Tapper, A.R.; McKinney, S.L.; Nashmi, R.; Schwarz, J.; Deshpande, P.; Labarca, C.; Whiteaker, P.; Marks, M.J.; Collins, A.C.; Lester, H.A. Nicotine Activation of α4* Receptors: Sufficient for Reward, Tolerance, and Sensitization. Science 2004, 306, 1029–1032. [Google Scholar] [CrossRef]
- Pons, S.; Fattore, L.; Cossu, G.; Tolu, S.; Porcu, E.; McIntosh, J.M.; Changeux, J.P.; Maskos, U.; Fratta, W. Crucial role of α4 and α6 nicotinic acetylcholine receptor subunits from ventral tegmental area in systemic nicotine self-administration. J. Neurosci. 2008, 28, 12318–12327. [Google Scholar] [CrossRef]
- Dani, J.A.; Ji, D.; Zhou, F.-M. Synaptic plasticity and nicotine addiction. Neuron 2001, 31, 349–352. [Google Scholar] [CrossRef]
- McKay, B.E.; Placzek, A.N.; Dani, J.A. Regulation of synaptic transmission and plasticity by neuronal nicotinic acetylcholine receptors. Biochem. Pharmacol. 2007, 74, 1120–1133. [Google Scholar] [CrossRef]
- Mansvelder, H.D.; Mertz, M.; Role, L. Nicotinic modulation of synaptic transmission and plasticity in cortico-limbic circuits. Semin. Cell Dev. Boil. 2009, 20, 432–440. [Google Scholar] [CrossRef]
- Mansvelder, H.D.; McGehee, D.S. Long-term potentiation of excitatory inputs to brain reward areas by nicotine. Neuron 2000, 27, 349–357. [Google Scholar] [CrossRef]
- Xiao, C.; Nashmi, R.; McKinney, S.; Cai, H.; McIntosh, J.M.; Lester, H.A. Chronic nicotine selectively enhances α4β2* nicotinic acetylcholine receptors in the nigrostriatal dopamine pathway. J. Neurosci. 2009, 29, 12428–12439. [Google Scholar] [CrossRef]
- Mansvelder, H.D.; Keath, J.R.; McGehee, D.S. Synaptic mechanisms underlie nicotine-induced excitability of brain reward areas. Neuron 2002, 33, 905–919. [Google Scholar] [CrossRef]
- Yan, Y.; Peng, C.; Arvin, M.C.; Jin, X.-T.; Kim, V.; Ramsey, M.D.; Wang, Y.; Banala, S.; Wokosin, D.L.; McIntosh, J.M.; et al. Nicotinic Cholinergic Receptors in VTA Glutamate Neurons Modulate Excitatory Transmission. Cell Rep. 2018, 23, 2236–2244. [Google Scholar] [CrossRef] [PubMed]
- Corrigall, W.A.; Coen, K.M. Nicotine maintains robust self-administration in rats on a limited-access schedule. Psychopharmacology 1989, 99, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, A.; Fujiyoshia, Y.; Unwin, N. Structure and gating mechanism of the acetylcholine receptor pore. Nature 2003, 423, 949–955. [Google Scholar] [CrossRef]
- Unwin, N.; Miyazawa, A.; Li, J.; Fujiyoshia, Y. Activation of the Nicotinic Acetylcholine Receptor Involves a Switch in Conformation of the α Subunits. J. Mol. Boil. 2002, 319, 1165–1176. [Google Scholar] [CrossRef]
- Picciotto, M.R.; Zoli, M.; Rimondini, R.; Lena, C.; Marubio, L.M.; Pich, E.M.; Fuxe, K.; Changeux, J.P. Acetylcholine receptors containing the beta2 subunit are involved in the reinforcing properties of nicotine. Nature 1998, 391, 173–177. [Google Scholar] [CrossRef]
- Pidoplichko, V.I.; DeBiasi, M.; Williams, J.T.; Dani, J.A. Nicotine activates and desensitizes midbrain dopamine neurons. Nature 1997, 390, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Engle, S.E.; Shih, P.Y.; McIntosh, J.M.; Drenan, R.M. α4α6β2* nicotinic acetylcholine receptor activation on ventral tegmental area dopamine neurons is sufficient to stimulate a depolarizing conductance and enhance surface AMPA receptor function. Mol. Pharmacol. 2013, 84, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhao-Shea, R.; McIntosh, J.M.; Gardner, P.D.; Tapper, A.R. Nicotine persistently activates ventral tegmental area dopaminergic neurons via nicotinic acetylcholine receptors containing α4 and α6 subunits. Mol. Pharmacol. 2012, 81, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Mansvelder, H.D.; McGehee, D.S. Cellular and synaptic mechanisms of nicotine addiction. J. Neurobiol. 2002, 53, 606–617. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.W.A.; North, R. Two types of neurone in the rat ventral tegmental area and their synaptic inputs. J. Physiol. 1992, 450, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Beckley, N.A.; Kim, V.; Drenan, R.M. Differential Nicotinic Modulation of Glutamatergic and GABAergic VTA Microcircuits. Eneuro 2019, 6. [Google Scholar] [CrossRef]
- Brody, A.L.; Mandelkern, M.A.; London, E.D.; Olmstead, R.E.; Farahi, J.; Scheibal, D.; Jou, J.; Allen, V.; Tiongson, E.; Chefer, S.I.; et al. Cigarette smoking saturates brain α4β2 nicotinic acetylcholine receptors. Arch Gen Psychiatry 2006, 63, 907–915. [Google Scholar] [CrossRef] [PubMed]
- Marks, M.; Pauly, J.; Gross, S.D.; Deneris, E.; Hermans-Borgmeyer, I.; Heinemann, S.; Collins, A. Nicotine binding and nicotinic receptor subunit RNA after chronic nicotine treatment. J. Neurosci. 1992, 12, 2765–2784. [Google Scholar] [CrossRef]
- Henderson, B.J.; Lester, H.A. Inside-out neuropharmacology of nicotinic drugs. Neuropharmacology 2015, 96, 178–193. [Google Scholar] [CrossRef]
- Hurst, R.; Rollema, H.; Bertrand, D. Nicotinic acetylcholine receptors: From basic science to therapeutics. Pharmacol. Ther. 2013, 137, 22–54. [Google Scholar] [CrossRef]
- Dani, J.A.; Heinemann, S. Molecular and Cellular Aspects of Nicotine Abuse. Neuron 1996, 16, 905–908. [Google Scholar] [CrossRef]
- Mukhin, A.G.; Kimes, A.S.; Chefer, S.I.; Matochik, J.A.; Contoreggi, C.S.; Horti, A.G.; Vaupel, D.B.; Pavlova, O.; Stein, E.A. Greater nicotinic acetylcholine receptor density in smokers than in nonsmokers: A PET study with 2-18F-FA-85380. J. Nucl. Med. 2008, 49, 1628–1635. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, R.; Richards, C.I.; Xiao, C.; Rhee, D.; Pantoja, R.; Dougherty, D.A.; Miwa, J.M.; Lester, H.A. Pharmacological chaperoning of nicotinic acetylcholine receptors reduces the endoplasmic reticulum stress response. Mol. Pharmacol. 2012, 81, 759–769. [Google Scholar] [CrossRef] [PubMed]
- Omaiye, E.E.; McWhirter, K.J.; Luo, W.; Pankow, J.F.; Talbot, P. High-Nicotine Electronic Cigarette Products: Toxicity of JUUL Fluids and Aerosols Correlates Strongly with Nicotine and Some Flavor Chemical Concentrations. Chem. Res. Toxicol. 2019, 32, 1058–1069. [Google Scholar] [CrossRef] [PubMed]
- Grieder, T.E.; Besson, M.; Maal-Bared, G.; Pons, S.; Maskos, U.; van der Kooy, D. beta2* nAChRs on VTA dopamine and GABA neurons separately mediate nicotine aversion and reward. Proc. Natl. Acad. Sci. USA 2019, 116, 25968–25973. [Google Scholar] [CrossRef]
- Picciotto, M.R.; Caldarone, B.J.; Brunzell, D.H.; Zachariou, V.; Stevens, T.R.; King, S.L. Neuronal nicotinic acetylcholine receptor subunit knockout mice: Physiological and behavioral phenotypes and possible clinical implications. Pharmacol. Ther. 2001, 92, 89–108. [Google Scholar] [CrossRef]
- Watkins, S.S.; Epping-Jordan, M.P.; Koob, G.F.; Markou, A. Blockade of Nicotine Self-Administration with Nicotinic Antagonists in Rats. Pharmacol. Biochem. Behav. 1999, 62, 743–751. [Google Scholar] [CrossRef]
- Corrigall, W.A.; Coen, K.M.; Adamson, K.L. Self-administered nicotine activates the mesolimbic dopamine system through the ventral tegmental area. Brain Res. 1994, 653, 278–284. [Google Scholar] [CrossRef]
- Nelson, M.E.; Kuryatov, A.; Choi, C.H.; Zhou, Y.; Lindstrom, J. Alternate stoichiometries of α4β2 nicotinic acetylcholine receptors. Mol. Pharmacol. 2003, 63, 332–341. [Google Scholar] [CrossRef]
- Kuryatov, A.; Onksen, J.; Lindstrom, J. Roles of Accessory Subunits in α4β2* Nicotinic Receptors. Mol. Pharmacol. 2008, 74, 132–143. [Google Scholar] [CrossRef]
- Schwartz, R.; Kellar, K. Nicotinic cholinergic receptor binding sites in the brain: Regulation In Vivo. Science 1983, 220, 214–216. [Google Scholar] [CrossRef]
- Liu, M.; Jiang, Y.; Wedow, R.; Li, Y.; Brazel, D.M.; Chen, F.; Datta, G.; Davila-Velderrain, J.; McGuire, D.; Tian, C.; et al. Association studies of up to 1.2 million individuals yield new insights into the genetic etiology of tobacco and alcohol use. Nat. Genet. 2019, 51, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Champtiaux, N.; Han, Z.-Y.; Bessis, A.; Rossi, F.M.; Zoli, M.; Marubio, L.; McIntosh, J.M.; Changeux, J.-P. Distribution and Pharmacology of α6-Containing Nicotinic Acetylcholine Receptors Analyzed with Mutant Mice. J. Neurosci. 2002, 22, 1208–1217. [Google Scholar] [CrossRef] [PubMed]
- Champtiaux, N.; Gotti, C.; Cordero-Erausquin, M.; David, D.J.; Przybylski, C.; Léna, C.; Clementi, F.; Moretti, M.; Rossi, F.M.; Le Novère, N.; et al. Subunit Composition of Functional Nicotinic Receptors in Dopaminergic Neurons Investigated with Knock-Out Mice. J. Neurosci. 2003, 23, 7820–7829. [Google Scholar] [CrossRef] [PubMed]
- Salminen, O.; Murphy, K.L.; McIntosh, J.M.; Drago, J.; Marks, M.J.; Collins, A.C.; Grady, S.R. Subunit Composition and Pharmacology of Two Classes of Striatal Presynaptic Nicotinic Acetylcholine Receptors Mediating Dopamine Release in Mice. Mol. Pharmacol. 2004, 65, 1526–1535. [Google Scholar] [CrossRef]
- Wonnacott, S.; Sidhpura, N.; Balfour, D.J. Nicotine: From molecular mechanisms to behaviour. Curr. Opin. Pharmacol. 2005, 5, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Mackey, E.D.; Engle, S.E.; Kim, M.R.; O’Neill, H.C.; Wageman, C.R.; Patzlaff, N.E.; Wang, Y.; Grady, S.R.; McIntosh, J.M.; Marks, M.J.; et al. α6* nicotinic acetylcholine receptor expression and function in a visual salience circuit. J. Neurosci. 2012, 32, 10226–10237. [Google Scholar] [CrossRef] [PubMed]
- Walters, C.L.; Brown, S.; Changeux, J.-P.; Martin, B.; Damaj, M.I. The β2 but not α7 subunit of the nicotinic acetylcholine receptor is required for nicotine-conditioned place preference in mice. Psychopharmacology 2006, 184, 339–344. [Google Scholar] [CrossRef]
- Xiao, C.; Srinivasan, R.; Drenan, R.M.; Mackey, E.D.W.; McIntosh, J.M.; Lester, H.A. Characterizing functional α6β2 nicotinic acetylcholine receptors in vitro: Mutant β2 subunits improve membrane expression, and fluorescent proteins reveal responsive cells. Biochem. Pharmacol. 2011, 82, 852–861. [Google Scholar] [CrossRef]
- Wang, J.; Kuryatov, A.; Sriram, A.; Jin, Z.; Kamenecka, T.M.; Kenny, P.J.; Lindstrom, J. An accessory agonist binding site promotes activation of α4β2* nicotinic acetylcholine receptors. J. Biol. Chem. 2015, 290, 13907–13918. [Google Scholar] [CrossRef] [PubMed]
- Alkondon, M.; Albuquerque, E.X. Initial Characterization of the Nicotinic Acetylcholine Receptors in Rat Hippocampal Neurons. J. Recept. Res. 1991, 11, 1001–1021. [Google Scholar] [CrossRef] [PubMed]
- Swanson, L.W. The projections of the ventral tegmental area and adjacent regions: A combined fluorescent retrograde tracer and immunofluorescence study in the rat. Brain Res. Bull. 1982, 9, 321–353. [Google Scholar] [CrossRef]
- Lüscher, C.; Malenka, R.C. Drug-Evoked Synaptic Plasticity in Addiction: From Molecular Changes to Circuit Remodeling. Neuron 2011, 69, 650–663. [Google Scholar] [CrossRef]
- Van Huijstee, A.N.; Mansvelder, H.D. Glutamatergic synaptic plasticity in the mesocorticolimbic system in addiction. Front. Cell. Neurosci. 2015, 8, 466. [Google Scholar] [CrossRef] [PubMed]
- Berrettini, W.; Yuan, X.; Tozzi, F.; Song, K.; Francks, C.; Chilcoat, H.; Waterworth, D.; Muglia, P.; Mooser, V. α5/α3 nicotinic receptor subunit alleles increase risk for heavy smoking. Mol. Psychiatry 2008, 13, 368–373. [Google Scholar] [CrossRef]
- Saccone, S.F.; Hinrichs, A.L.; Saccone, N.L.; Chase, G.A.; Konvicka, K.; Madden, P.A.; Breslau, N.; Johnson, E.O.; Hatsukami, D.; Pomerleau, O.; et al. Cholinergic nicotinic receptor genes implicated in a nicotine dependence association study targeting 348 candidate genes with 3713 SNPs. Hum. Mol. Genet. 2006, 16, 36–49. [Google Scholar] [CrossRef]
- Bierut, L.J.; Stitzel, J.A.; Wang, J.C.; Hinrichs, A.L.; Grucza, R.A.; Xuei, X.; Saccone, N.L.; Saccone, S.F.; Bertelsen, S.; Fox, L.; et al. Variants in nicotinic receptors and risk for nicotine dependence. Am. J. Psychiatry 2008, 165, 1163–1171. [Google Scholar] [CrossRef]
- Wang, J.C.; Grucza, R.A.; Cruchaga, C.; Hinrichs, A.L.; Bertelsen, S.; Budde, J.; Fox, L.; Goldstein, E.; Reyes, O.; Saccone, N.; et al. Genetic variation in the CHRNA5 gene affects mRNA levels and is associated with risk for alcohol dependence. Mol. Psychiatry 2008, 14, 501–510. [Google Scholar] [CrossRef]
- Fowler, C.D.; Lu, Q.; Johnson, P.M.; Marks, M.J.; Kenny, P.J. Habenular α5 nicotinic receptor subunit signalling controls nicotine intake. Nature 2011, 471, 597–601. [Google Scholar] [CrossRef]
- Fowler, C.D.; Kenny, P.J. Nicotine aversion: Neurobiological mechanisms and relevance to tobacco dependence vulnerability. Neuropharmacology 2013, 76, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Nair-Roberts, R.; Chatelain-Badie, S.; Benson, E.; White-Cooper, H.; Bolam, J.P.; Ungless, M.A. Stereological estimates of dopaminergic, GABAergic and glutamatergic neurons in the ventral tegmental area, substantia nigra and retrorubral field in the rat. Neuroscience 2008, 152, 1024–1031. [Google Scholar] [CrossRef] [PubMed]
- Di Chiara, G.; Imperato, A. Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc. Natl. Acad. Sci. USA 1988, 85, 5274–5278. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F.; Le Moal MD, M. Drug Addiction, Dysregulation of Reward, and Allostasis. Neuropsychopharmacology 2001, 24, 97–129. [Google Scholar] [CrossRef]
- Wise, R.A.; Rompre, P.P. Brain dopamine and reward. Annu. Rev. Psychol. 1989, 40, 191–225. [Google Scholar] [CrossRef]
- Wise, R.A. Dopamine and reward: The anhedonia hypothesis 30 years on. Neurotox. Res. 2008, 14, 169–183. [Google Scholar] [CrossRef]
- Corrigall, W.A.; Franklin, K.B.J.; Coen, K.M.; Clarke, P.B.S. The mesolimbic dopaminergic system is implicated in the reinforcing effects of nicotine. Psychopharmacology 1992, 107, 285–289. [Google Scholar] [CrossRef]
- Louis, M.; Clarke, P.B. Effect of ventral tegmental 6-hydroxydopamine lesions on the locomotor stimulant action of nicotine in rats. Neuropharmacology 1998, 37, 1503–1513. [Google Scholar] [CrossRef]
- Capriles, N.; Rodaros, D.; Sorge, R.E.; Stewart, J. A role for the prefrontal cortex in stress- and cocaine-induced reinstatement of cocaine seeking in rats. Psychopharmacology 2002, 168, 66–74. [Google Scholar] [CrossRef]
- Counotte, D.S.; Goriounova, N.A.; Moretti, M.; Smoluch, M.T.; Irth, H.; Clementi, F.; Schoffelmeer, A.N.M.; Mansvelder, H.D.; Smit, A.B.; Gotti, C.; et al. Adolescent nicotine exposure transiently increases high-affinity nicotinic receptors and modulates inhibitory synaptic transmission in rat medial prefrontal cortex. FASEB J. 2012, 26, 1810–1820. [Google Scholar] [CrossRef]
- Lodge, D.J.; Grace, A.A. The laterodorsal tegmentum is essential for burst firing of ventral tegmental area dopamine neurons. Proc. Natl. Acad. Sci. USA 2006, 103, 5167–5172. [Google Scholar] [CrossRef] [PubMed]
- Omelchenko, N.; Sesack, S.R. Laterodorsal tegmental projections to identified cell populations in the rat ventral tegmental area. J. Comp. Neurol. 2005, 483, 217–235. [Google Scholar] [CrossRef] [PubMed]
- Paladini, C.; Roeper, J. Generating bursts (and pauses) in the dopamine midbrain neurons. Neuroscience 2014, 282, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Wolf, M.E. Addiction: Making the Connection Between Behavioral Changes and Neuronal Plasticity in Specific Pathways. Mol. Interv. 2002, 2, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Lammel, S.; Lim, B.; Ran, C.; Huang, K.W.; Betley, M.J.; Tye, K.M.; Deisseroth, K.; Malenka, R.C. Input-specific control of reward and aversion in the ventral tegmental area. Nature 2012, 491, 212–217. [Google Scholar] [CrossRef]
- Matsui, A.; Williams, J.T. Opioid-sensitive GABA inputs from rostromedial tegmental nucleus synapse onto midbrain dopamine neurons. J. Neurosci. 2011, 31, 17729–17735. [Google Scholar] [CrossRef]
- Fowler, C.D.; Tuesta, L.; Kenny, P.J. Role of α5* nicotinic acetylcholine receptors in the effects of acute and chronic nicotine treatment on brain reward function in mice. Psychopharmacology 2013, 229, 503–513. [Google Scholar] [CrossRef]
- Hikosaka, O. The habenula: From stress evasion to value-based decision-making. Nat. Rev. Neurosci. 2010, 11, 503–513. [Google Scholar] [CrossRef]
- Sulzer, D. How Addictive Drugs Disrupt Presynaptic Dopamine Neurotransmission. Neuron 2011, 69, 628–649. [Google Scholar] [CrossRef]
- Maskos, U.; Molles, B.E.; Pons, S.; Besson, M.; Guiard, B.P.; Guilloux, J.-P.; Evrard, A.; Cazala, P.; Cormier, A.; Mameli-Engvall, M.; et al. Nicotine reinforcement and cognition restored by targeted expression of nicotinic receptors. Nature 2005, 436, 103–107. [Google Scholar] [CrossRef]
- Guan, Y.-Z.; Ye, J.-H. Glycine blocks long-term potentiation of GABAergic synapses in the ventral tegmental area. Neuroscience 2016, 318, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.H.; Wang, F.; Krnjevic, K.; Wang, W.; Xiong, Z.-G.; Zhang, J. Presynaptic Glycine Receptors on GABAergic Terminals Facilitate Discharge of Dopaminergic Neurons in Ventral Tegmental Area. J. Neurosci. 2004, 24, 8961–8974. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Nie, H.; Bian, W.; Dave, V.; Janak, P.H.; Ye, J.H. Microinjection of glycine into the ventral tegmental area selectively decreases ethanol consumption. J. Pharmacol. Exp. Ther. 2012, 341, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Tritsch, N.X.; Sabatini, B.L. Dopaminergic Modulation of Synaptic Transmission in Cortex and Striatum. Neuron 2012, 76, 33–50. [Google Scholar] [CrossRef] [PubMed]
- Drenan, R.M.; Grady, S.R.; Steele, A.D.; McKinney, S.; Patzlaff, N.E.; McIntosh, J.M.; Marks, M.J.; Miwa, J.M.; Lester, H.A. Cholinergic Modulation of Locomotion and Striatal Dopamine Release Is Mediated by α6α4* Nicotinic Acetylcholine Receptors. J. Neurosci. 2010, 30, 9877–9889. [Google Scholar] [CrossRef]
- Cooper, S.; Robison, A.J.; Mazei-Robison, M.S. Reward Circuitry in Addiction. Neurotherapeutics 2017, 14, 687–697. [Google Scholar] [CrossRef]
- McFarland, K.; Kalivas, P.W. The Circuitry Mediating Cocaine-Induced Reinstatement of Drug-Seeking Behavior. J. Neurosci. 2001, 21, 8655–8663. [Google Scholar] [CrossRef]
- Knackstedt, L.A.; Kalivas, P.W. Glutamate and reinstatement. Curr. Opin. Pharmacol. 2009, 9, 59–64. [Google Scholar] [CrossRef]
- Yang, H.; De Jong, J.W.; Tak, Y.; Peck, J.; Bateup, H.S.; Lammel, S. Nucleus Accumbens Subnuclei Regulate Motivated Behavior via Direct Inhibition and Disinhibition of VTA Dopamine Subpopulations. Neuron 2018, 97. [Google Scholar] [CrossRef]
- Bocklisch, C.; Pascoli, V.; Wong, J.; House, D.R.C.; Yvon, C.; De Roo, M.; Tan, K.R.; Lüscher, C. Cocaine Disinhibits Dopamine Neurons by Potentiation of GABA Transmission in the Ventral Tegmental Area. Science 2013, 341, 1521–1525. [Google Scholar] [CrossRef]
- Xia, Y.; Driscoll, J.R.; Wilbrecht, L.; Margolis, E.B.; Fields, H.L.; Hjelmstad, G.O. Nucleus Accumbens Medium Spiny Neurons Target Non-Dopaminergic Neurons in the Ventral Tegmental Area. J. Neurosci. 2011, 31, 7811–7816. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Chen, H.; Steketee, J.D.; Sharp, B.M. Upregulation of ionotropic glutamate receptor subunits within specific mesocorticolimbic regions during chronic nicotine self-administration. Neuropsychopharmacology 2006, 32, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Rezvani, A.H.; Eddins, N.; Slade, S.; Hampton, D.S.; Christopher, N.C.; Petro, A.; Horton, K.; Johnson, M.; Levin, E.D. Neonatal 6-hydroxydopamine lesions of the frontal cortex in rats: Persisting effects on locomotor activity, learning and nicotine self-administration. Neuroscience 2008, 154, 885–897. [Google Scholar] [CrossRef] [PubMed]
- Kalivas, P.W.; Volkow, N.D. The Neural Basis of Addiction: A Pathology of Motivation and Choice. Am. J. Psychiatry 2005, 162, 1403–1413. [Google Scholar] [CrossRef]
- Cornish, J.L.; Kalivas, P.W. Glutamate Transmission in the Nucleus Accumbens Mediates Relapse in Cocaine Addiction. J. Neurosci. 2000, 20, RC89. [Google Scholar] [CrossRef]
- Due, D.L.; Huettel, S.A.; Hall, W.G.; Rubin, D.C. Activation in Mesolimbic and Visuospatial Neural Circuits Elicited by Smoking Cues: Evidence From Functional Magnetic Resonance Imaging. Am. J. Psychiatry 2002, 159, 954–960. [Google Scholar] [CrossRef]
- Taber, M.T.; Das, S.; Fibiger, H.C. Cortical Regulation of Subcortical Dopamine Release: Mediation via the Ventral Tegmental Area. J. Neurochem. 2002, 65, 1407–1410. [Google Scholar] [CrossRef]
- Deas, D.; Brown, E.S. Adolescent substance abuse and psychiatric comorbidities. J. Clin. Psychiatry 2006, 67, 2. [Google Scholar] [CrossRef]
- Brown, R.A.; Lewinsohn, P.M.; Seeley, J.R.; Wagner, E.F. Cigarette Smoking, Major Depression, and Other Psychiatric Disorders among Adolescents. J. Am. Acad. Child. Adolesc. Psychiatry 1996, 35, 1602–1610. [Google Scholar] [CrossRef]
- Yuan, M.; Cross, S.J.; Loughlin, S.E.; Leslie, F.M. Nicotine and the adolescent brain. J. Physiol. 2015, 593, 3397–3412. [Google Scholar] [CrossRef]
- Pascoli, V.; Terrier, J.; Espallergues, J.; Valjent, E.; O’Connor, E.C.; Lüscher, C. Contrasting forms of cocaine-evoked plasticity control components of relapse. Nature 2014, 509, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.H.; Schlüter, O.M.; Dong, Y. Silent Synapses Speak Up. Neuroscience 2015, 21, 451–459. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, I.; Dani, J.A.; De Biasi, M. The medial habenula and interpeduncular nucleus circuitry is critical in addiction, anxiety, and mood regulation. J. Neurochem. 2017, 142, 130–143. [Google Scholar] [CrossRef] [PubMed]
- Salas, R.; Sturm, R.M.; Boulter, J.; De Biasi, M. Nicotinic receptors in the habenulo-interpeduncular system are necessary for nicotine withdrawal in mice. J. Neurosci. 2009, 29, 3014–3018. [Google Scholar] [CrossRef]
- Salas, R.; Pieri, F.; De Biasi, M. Decreased signs of nicotine withdrawal in mice null for the β4 nicotinic acetylcholine receptor subunit. J. Neurosci. 2004, 24, 10035–10039. [Google Scholar] [CrossRef]
- Ji, H.; Shepard, P.D. Lateral habenula stimulation inhibits rat midbrain dopamine neurons through a GABA(A) receptor-mediated mechanism. J. Neurosci. 2007, 27, 6923–6930. [Google Scholar] [CrossRef]
- Matsumoto, M.; Hikosaka, O. Lateral habenula as a source of negative reward signals in dopamine neurons. Nature 2007, 447, 1111–1115. [Google Scholar] [CrossRef]
- Wellmann, K.F. Smoking and health. On the report of the advisory committee to the surgeon general of the public health service. DMW—Dtsch. Med. Wochenschr. 1964, 89, 45–46. [Google Scholar]
- Lundeen, R.E. Tobacco under the FDA: A summary of the Family Smoking Prevention and Tobacco Control Act. Health Care Law Mon. 2009, 9, 2–9. [Google Scholar]
- Odani, S.; Armour, B.S.; Graffunder, C.M.; Willis, G.; Hartman, A.M.; Agaku, I.T. The 2014–2015 Tobacco Use Supplement to the Current Population Survey. MMWR Morb Mortal Wkly Rep. 2018, 67, 97–102. [Google Scholar] [CrossRef]
- Kozlitina, J.; Risso, D.; Lansu, K.; Olsen, R.H.J.; Sainz, E.; Luiselli, D.; Barik, A.; Domingues, C.E.F.; Pagani, L.; Wooding, S.; et al. An African-specific haplotype in MRGPRX4 is associated with menthol cigarette smoking. PLoS Genet. 2019, 15, e1007916. [Google Scholar] [CrossRef] [PubMed]
- Wickham, R. How Menthol Alters Tobacco-Smoking Behavior: A Biological Perspective. Yale J. Boil. Med. 2015, 88, 279–287. [Google Scholar]
- DeVito, E.E.; Krishnan-Sarin, S. E-cigarettes: Impact of E-Liquid Components and Device Characteristics on Nicotine Exposure. Curr. Neuropharmacol. 2018, 16, 438–459. [Google Scholar] [CrossRef]
- Baker, T.B.; Piper, M.E.; McCarthy, D.E.; Bolt, D.M.; Smith, S.S.; Kim, S.-Y.; Colby, S.; Conti, D.; Giovino, G.A.; Hatsukami, D.; et al. Time to first cigarette in the morning as an index of ability to quit smoking: Implications for nicotine dependence. Nicotine Tob. Res. 2007, 9, 555–570. [Google Scholar] [CrossRef]
- Ahijevych, K.; Parsley, L.A. Smoke constituent exposure and stage of change in black and white women cigarette smokers. Addict. Behav. 1999, 24, 115–120. [Google Scholar] [CrossRef]
- Ahijevych, K.; Garrett, B.E. Menthol pharmacology and its potential impact on cigarette smoking behavior. Nicotine Tob. Res. 2004, 6, 17–28. [Google Scholar] [CrossRef]
- Caggiula, A.R.; Donny, E.C.; White, A.R.; Chaudhri, N.; Booth, S.A.; Gharib, M.; Hoffman, A.; Perkins, K.A.; Sved, A.F. Cue dependency of nicotine self-administration and smoking. Pharmacol. Biochem. Behav. 2001, 70, 515–530. [Google Scholar] [CrossRef]
- Harrison, E.; Biswas, L.; Avusula, R.; Zhang, M.; Gong, Y.; Liu, X. Effects of menthol and its interaction with nicotine-conditioned cue on nicotine-seeking behavior in rats. Psychopharmacology 2017, 234, 3443–3453. [Google Scholar] [CrossRef]
- Wickham, R.J.; Nunes, E.J.; Hughley, S.; Silva, P.; Walton, S.N.; Park, J.; Addy, N.A. Evaluating oral flavorant effects on nicotine self-administration behavior and phasic dopamine signaling. Neuropharmacology 2017, 128, 33–42. [Google Scholar] [CrossRef]
- Hans, M.; Wilhelm, M.; Swandulla, D. Menthol Suppresses Nicotinic Acetylcholine Receptor Functioning in Sensory Neurons via Allosteric Modulation. Chem. Senses 2012, 37, 463–469. [Google Scholar] [CrossRef]
- Ashoor, A.; Nordman, J.C.; Veltri, D.; Yang, K.-H.S.; Al Kury, L.; Shuba, Y.; Mahgoub, M.; Howarth, F.C.; Sadek, B.; Shehu, A.; et al. Menthol Binding and Inhibition of α7-Nicotinic Acetylcholine Receptors. PLoS ONE 2013, 8, e67674. [Google Scholar] [CrossRef]
- Ton, H.T.; Smart, A.E.; Aguilar, B.L.; Olson, T.T.; Kellar, K.J.; Ahern, G.P. Menthol Enhances the Desensitization of Human alpha3beta4 Nicotinic Acetylcholine Receptors. Mol. Pharmacol. 2015, 88, 256–264. [Google Scholar] [CrossRef]
- Shahoei, R.; Tajkhorshid, E. Menthol Binding to the Human α4β2 Nicotinic Acetylcholine Receptor Facilitated by Its Strong Partitioning in the Membrane. J. Phys. Chem. B 2020, 124, 1866–1880. [Google Scholar] [CrossRef] [PubMed]
- AlSharari, S.D.; King, J.R.; Nordman, J.C.; Muldoon, P.P.; Jackson, A.; Zhu, A.Z.X.; Tyndale, R.F.; Kabbani, N.; Damaj, M.I. Effects of Menthol on Nicotine Pharmacokinetic, Pharmacology and Dependence in Mice. PLoS ONE 2015, 10, e0137070. [Google Scholar] [CrossRef] [PubMed]
- Wickham, R.J. The Biological Impact of Menthol on Tobacco Dependence. Nicotine Tob. Res. 2019. [Google Scholar] [CrossRef]
- Valentine, G.E.; DeVito, E.; Jatlow, P.; Gueorguieva, R.V.; Sofuoglu, M. Acute effects of inhaled menthol on the rewarding effects of intravenous nicotine in smokers. J. Psychopharmacol. 2018, 32, 986–994. [Google Scholar] [CrossRef]
- Villanti, A.C.; Collins, L.K.; Niaura, R.; Gagosian, S.Y.; Abrams, D.B. Menthol cigarettes and the public health standard: A systematic review. BMC Public Health 2017, 17, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Tierney, P.A.; Karpinski, C.D.; Brown, J.E.; Luo, W.; Pankow, J.F. Flavour chemicals in electronic cigarette fluids. Tob. Control. 2015, 25, e10–e15. [Google Scholar] [CrossRef] [PubMed]
- Keath, J.R.; Iacoviello, M.P.; Barrett, L.E.; Mansvelder, H.D.; McGehee, D.S. Differential Modulation by Nicotine of Substantia Nigra Versus Ventral Tegmental Area Dopamine Neurons. J. Neurophysiol. 2007, 98, 3388–3396. [Google Scholar] [CrossRef]
- Chen, H.; Matta, S.G.; Sharp, B.M. Acquisition of Nicotine Self-Administration in Adolescent Rats Given Prolonged Access to the Drug. Neuropsychopharmacology 2006, 32, 700–709. [Google Scholar] [CrossRef]
- Kota, D.; Martin, B.R.; Robinson, S.E.; Damaj, M.I. Nicotine Dependence and Reward Differ between Adolescent and Adult Male Mice. J. Pharmacol. Exp. Ther. 2007, 322, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Goriounova, N.A.; Mansvelder, H.D. Short- and Long-Term Consequences of Nicotine Exposure during Adolescence for Prefrontal Cortex Neuronal Network Function. Cold Spring Harb. Perspect. Med. 2012, 2, a012120. [Google Scholar] [CrossRef] [PubMed]
- Goriounova, N.A.; Mansvelder, H.D. Nicotine exposure during adolescence alters the rules for prefrontal cortical synaptic plasticity during adulthood. Front. Synaptic Neurosci. 2012, 4, 3. [Google Scholar] [CrossRef] [PubMed]
- Romoli, B.; Lozada, A.F.; Sandoval, I.M.; Manfredsson, F.P.; Hnasko, T.S.; Berg, D.K.; Dulcis, D. Neonatal Nicotine Exposure Primes Midbrain Neurons to a Dopaminergic Phenotype and Increases Adult Drug Consumption. Boil. Psychiatry 2019, 86, 344–355. [Google Scholar] [CrossRef]
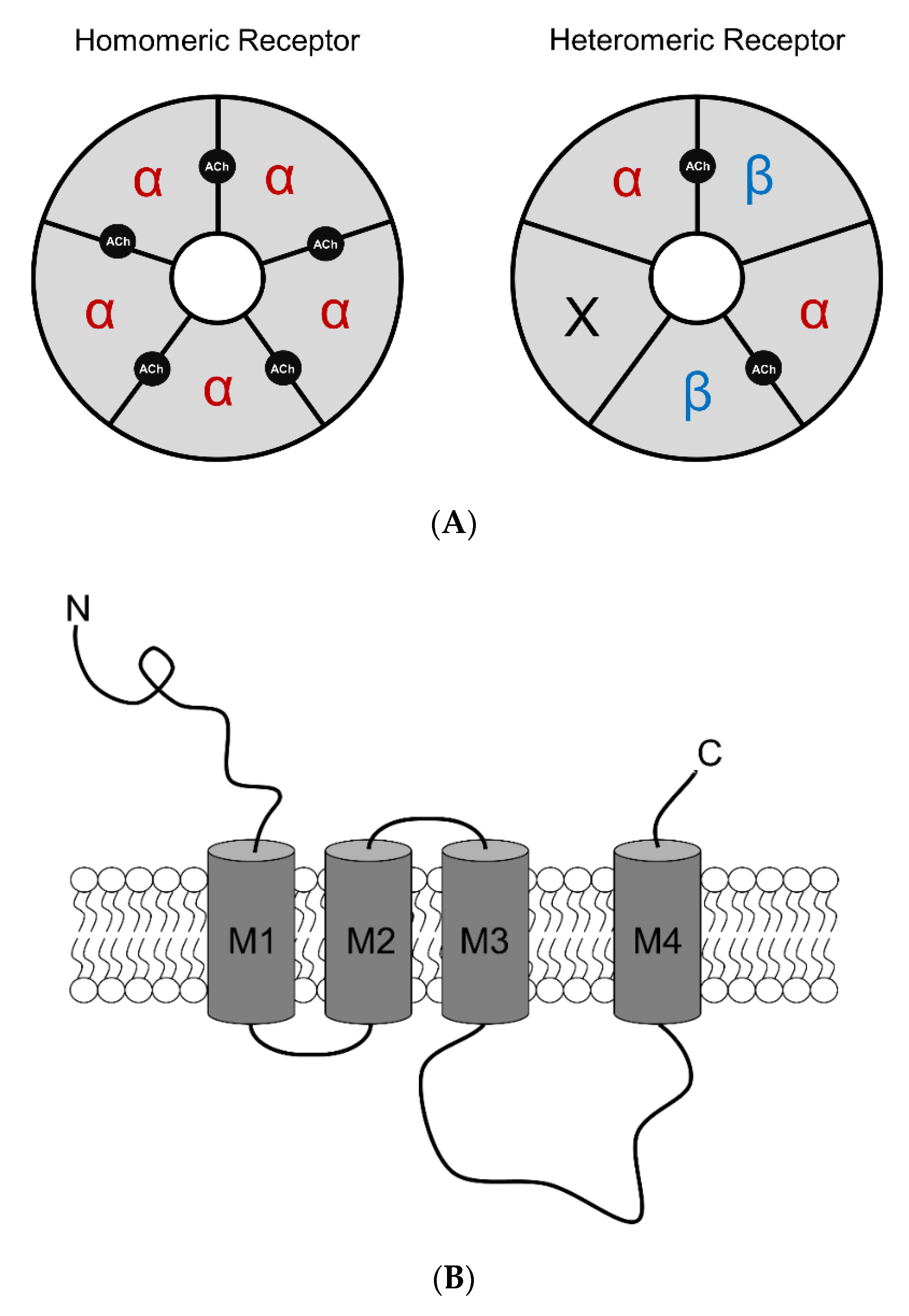
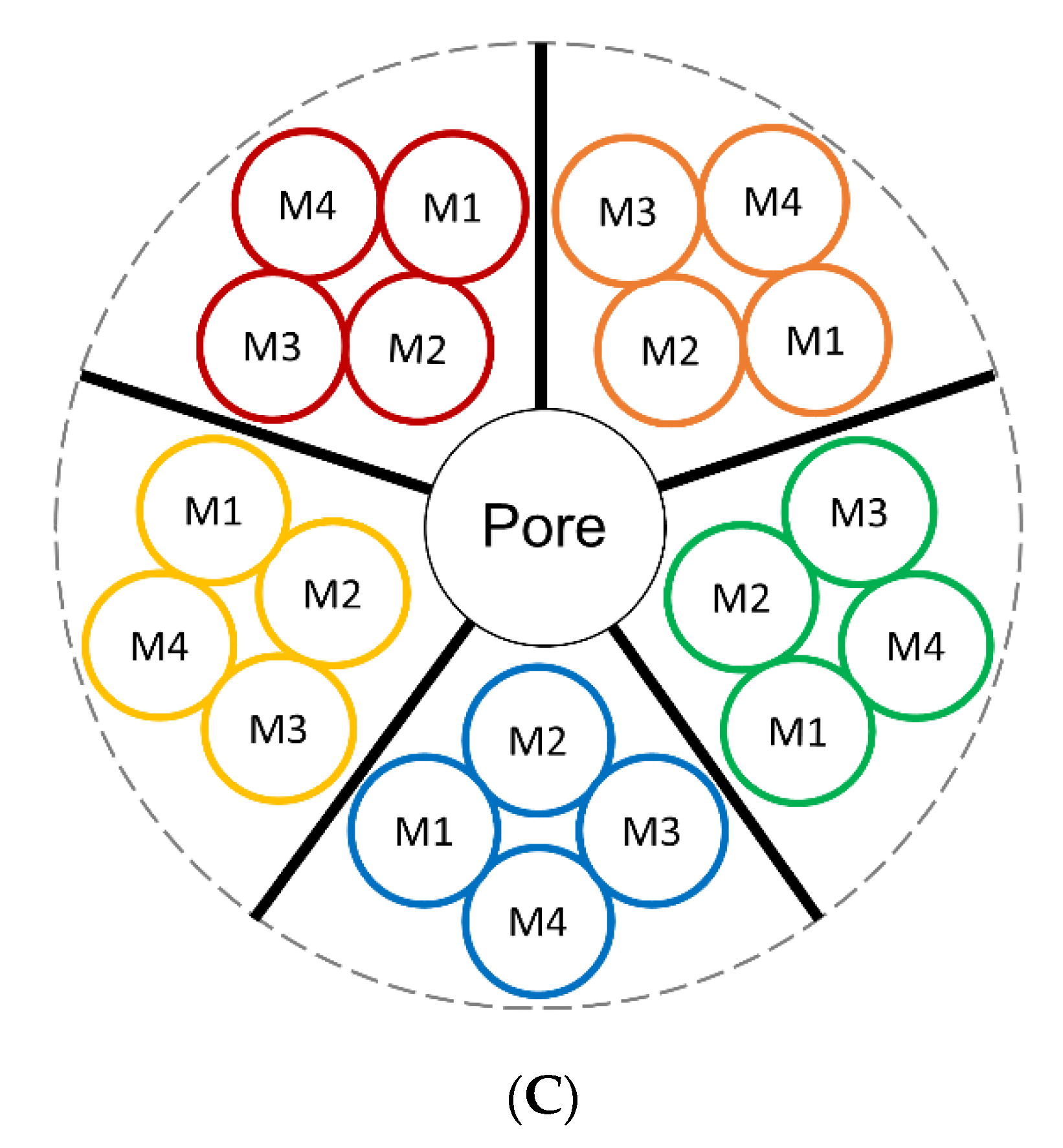
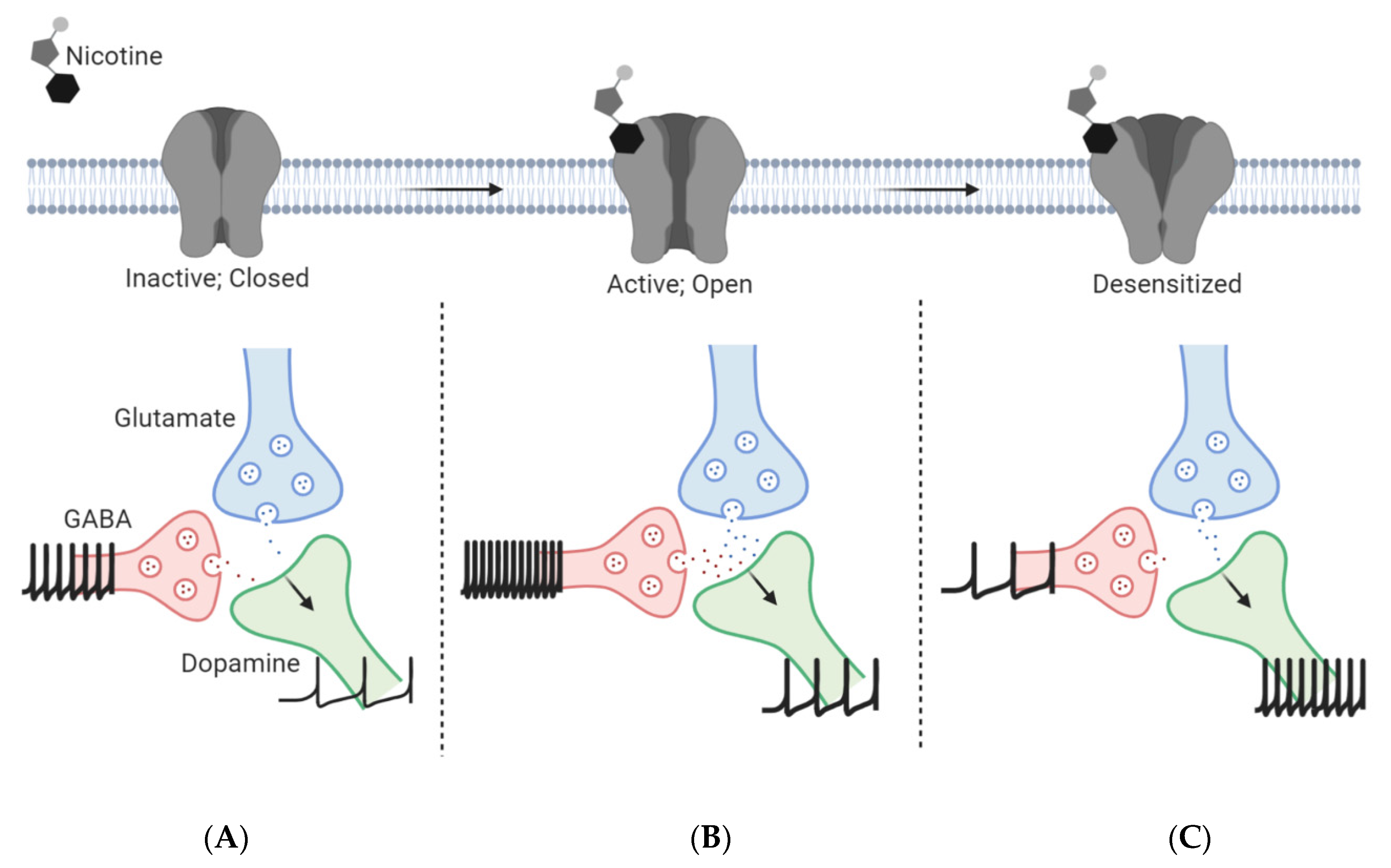

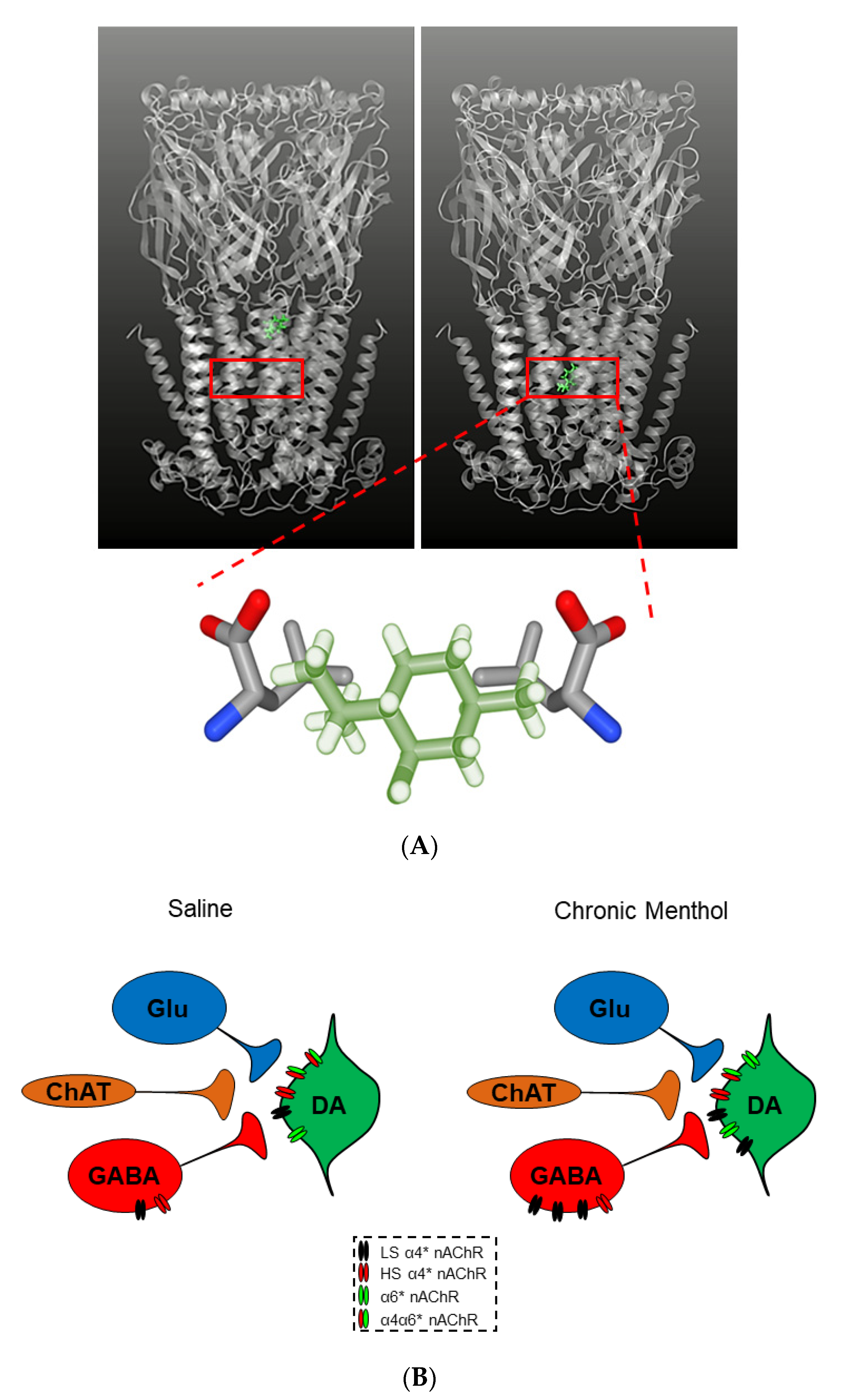
| Steps of Nicotine Addiction | nAChR Subunits | Brain Regions | Nicotine Concentration | Duration of Administration |
|---|---|---|---|---|
| Nicotine binding promotes a conformational transition of nAChRs from a resting, closed state to an open state, allowing signal transduction to occur [65,66]. | α4β2*, α4α6β2*, α6β2β3, α7 | Midbrain | Small (<1 µM) | Acute |
| When an individual becomes a long-term nicotine user, nAChR upregulation occurs. | α4, α6, β2, and β3 | VTA | 20–500 nM | Chronic |
| Upregulated α4β2* nAChRs found on GABA neurons of the midbrain elicit an inhibitory tone to VTA DA neurons, reducing the transmission of DA and contribute to nicotine-seeking behaviors [72]. | α4β2* | Midbrain GABA | ||
| After long-term exposure to nicotine, α4β2* nAChRs desensitize quickly, which decreases the GABAergic transmission onto DA neurons resulting in the disinhibition of DA neurons. | α4β2* | Midbrain GABA | 20–500 nM | Chronic |
| The activation of α4*, α6*, and α7 nAChRs on midbrain DA and glutamate neurons, respectively, promotes a net excitatory effect on DA neurotransmission from the VTA to the NAc and PFC, leading to the rewarding and reinforcing aspects of nicotine use [71]. | α4β2*, α6β2β3, α4α6β2*, α7 | VTA | ||
| These effects are reinforced by the enhanced glutamate neurotransmission from distal regions and local mVTA glutamate neurons to lateral VTA (latVTA) DA neurons [63,73], leading to long-term potentiation (LTP). | α7 | mVTA, PFC |
| Flavor Chemical | Chemical Class | Flavor Profile |
|---|---|---|
| Vanillin | Aldehyde | Vanilla, Chocolate, Cotton Candy, Mint, Coffee, Tobacco |
| Ethyl Vanillin | Aldehyde | Vanilla, Chocolate, Cotton Candy, Coffee, Tobacco |
| Ethyl Maltol | Alcohol | Vanilla, Chocolate, Cotton Candy, Mint, Coffee, Tobacco, Grape, Cherry |
| Maltol | Alcohol | Vanilla, Chocolate, Mint, Coffee, Tobacco, Grape |
| Benzaldehyde | Aldehyde | Cherry, Bubble Gum |
| Benzyl Alcohol | Alcohol | Cherry, Vanilla, Coffee, Tobacco |
| Ethyl Butyrate | Ester | Vanilla, Cherry, Bubble Gum, Apple, Tobacco, Grape |
| Menthol | Alcohol | Mint |
| Hexyl Acetate | Ester | Apple |
| Ethyl Acetate | Ester | Bubble Gum, Apple, Grape, Tobacco |
| Methylbutyl Acetate | Ester | Bubble Gum, Apple |
| Farnesol | Sesquiterpene | Apple |
| Farnesene | Sesquiterpene | Apple |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cooper, S.Y.; Henderson, B.J. The Impact of Electronic Nicotine Delivery System (ENDS) Flavors on Nicotinic Acetylcholine Receptors and Nicotine Addiction-Related Behaviors. Molecules 2020, 25, 4223. https://doi.org/10.3390/molecules25184223
Cooper SY, Henderson BJ. The Impact of Electronic Nicotine Delivery System (ENDS) Flavors on Nicotinic Acetylcholine Receptors and Nicotine Addiction-Related Behaviors. Molecules. 2020; 25(18):4223. https://doi.org/10.3390/molecules25184223
Chicago/Turabian StyleCooper, Skylar Y., and Brandon J. Henderson. 2020. "The Impact of Electronic Nicotine Delivery System (ENDS) Flavors on Nicotinic Acetylcholine Receptors and Nicotine Addiction-Related Behaviors" Molecules 25, no. 18: 4223. https://doi.org/10.3390/molecules25184223
APA StyleCooper, S. Y., & Henderson, B. J. (2020). The Impact of Electronic Nicotine Delivery System (ENDS) Flavors on Nicotinic Acetylcholine Receptors and Nicotine Addiction-Related Behaviors. Molecules, 25(18), 4223. https://doi.org/10.3390/molecules25184223







