Flavonoids as Phytoestrogenic Components of Hops and Beer
Abstract
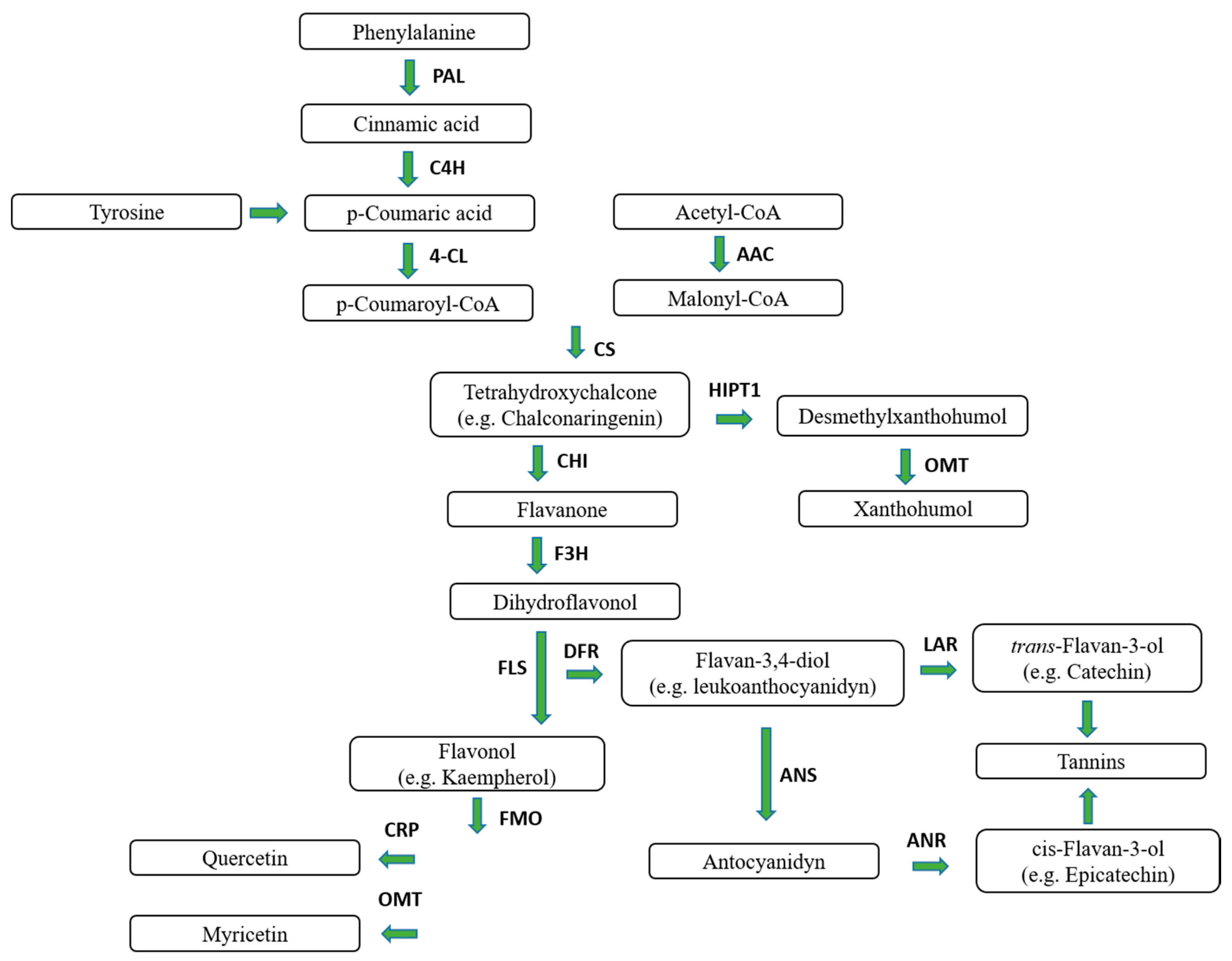
1. Hop
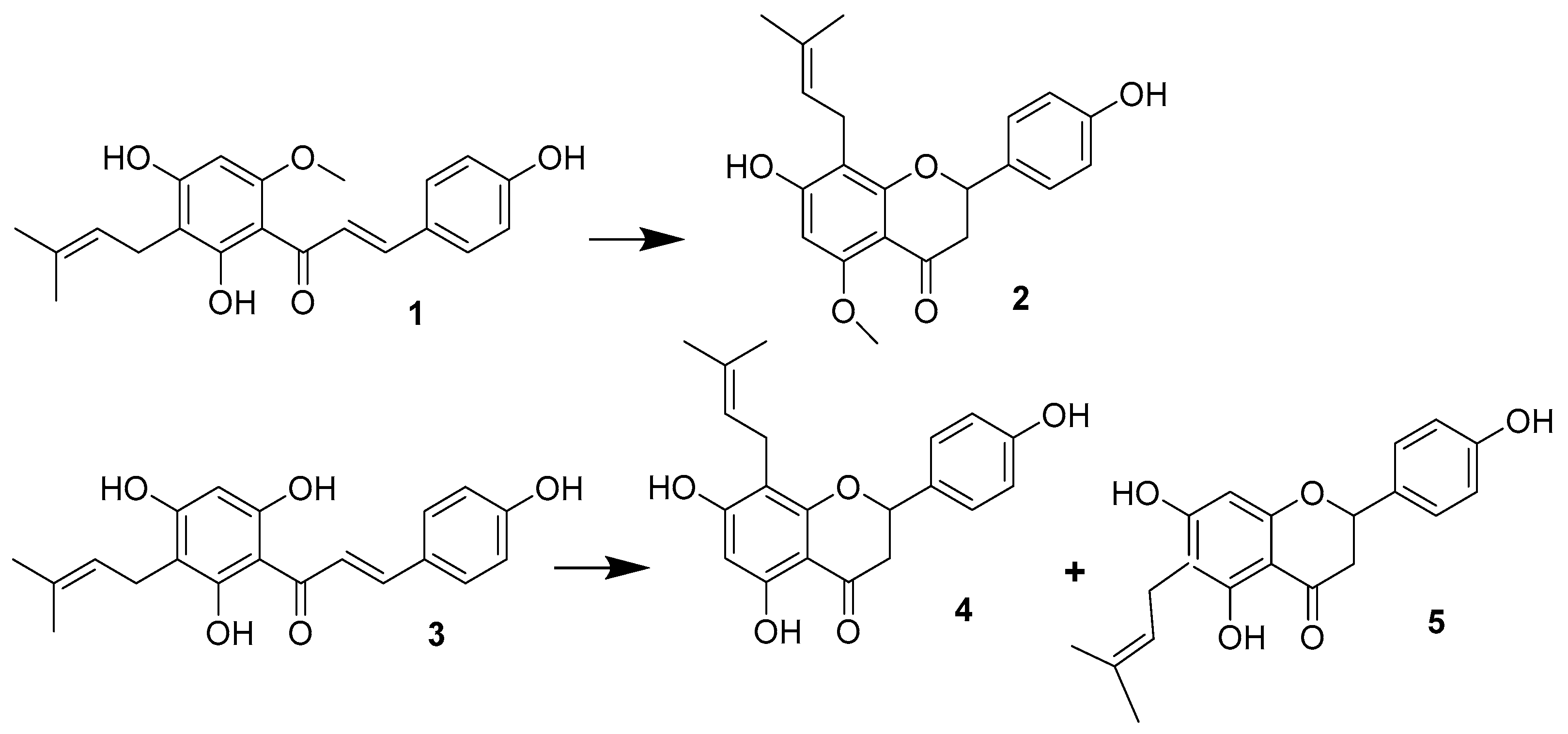
1.1. Hop Prenylflavonoids
1.2. Prenylflavonoids in Beer
2. Metabolism
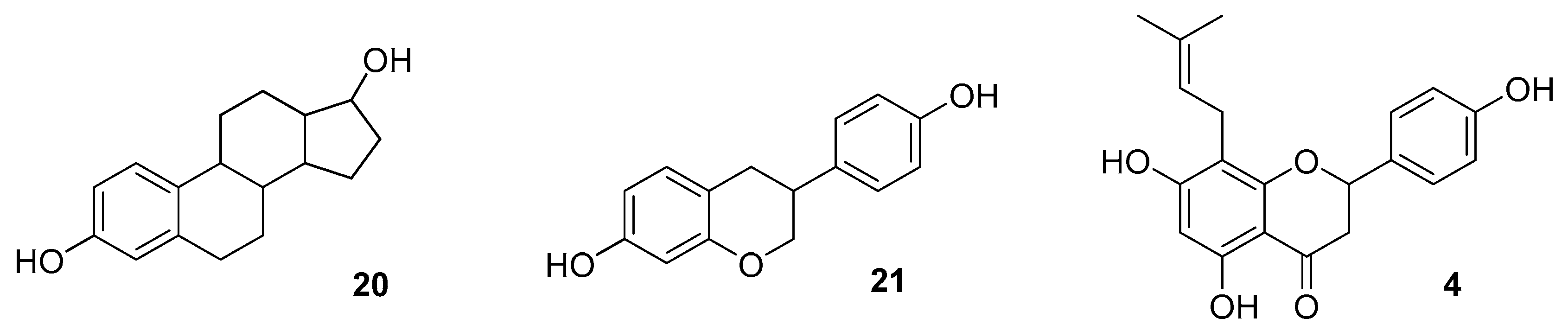
2.1. Metabolism of IX (2)
2.2. Metabolism and Bioavailability of Hop Estrogen
3. Phytoestrogens
3.1. Hop Phytoestrogens and Their Activities
3.1.1. Protective Function on the Cardiovascular System
3.1.2. Neuroprotection
3.1.3. Anticancerogenic Activities
3.1.4. Anti-Inflammatory Activity
3.1.5. Anti-Diabetic Activity
3.1.6. Antimicrobial Activity
3.2. Application
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Humulus lupulus | International Plant Names Index. Available online: https://www.ipni.org/n/303502-2 (accessed on 1 July 2020).
- Koetter, U.; Biendl, M. Hops (Humulus lupulus): A Review of its Historic and Medicinal Uses. J. Am. Bot. Counc. 2010, 87, 44–57. [Google Scholar]
- Zanoli, P.; Zavatti, M. Pharmacognostic and pharmacological profile of Humulus lupulus L. J. Ethnopharmacol. 2008, 116, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Xiang, D.; Chen, X.; Huo, H. Role of Characteristic Components of Humulus lupulus in Promoting Human Health. J. Agric. Food Chem. 2019, 67, 8291–8302. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.W.; Barofsky, E.; Kennedy, J.A.; Deinzer, M.L. Hop (Humulus lupulus L.) proanthocyanidins characterized by mass spectrometry, acid catalysis, and gel permeation chromatography. J. Agric. Food Chem. 2003, 51, 4101–4110. [Google Scholar] [CrossRef]
- Quifer-Rada, P.; Vallverdú-Queralt, A.; Martínez-Huélamo, M.; Chiva-Blanch, G.; Jáuregui, O.; Estruch, R.; Lamuela-Raventós, R. A comprehensive characterisation of beer polyphenols by high resolution mass spectrometry (LC-ESI-LTQ-Orbitrap-MS). Food Chem. 2015, 169, 336–343. [Google Scholar] [CrossRef]
- Perez-vizcaino, F.; Fraga, C.G. Research trends in flavonoids and health. Arch. Biochem. Biophys. 2018, 646, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Vogel, S.; Barbic, M.; Ju, G. European Journal of Medicinal Chemistry Synthesis, cytotoxicity, anti-oxidative and anti-inflammatory activity of chalcones and influence of A-ring modifications on the pharmacological effect. Eur. J. Med. Chem. 2010, 45, 2206–2213. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Jiang, Y.; Yang, J.; He, J.; Sun, J.; Chen, F.; Zhang, M.; Yang, B. Prenylated flavonoids, promising nutraceuticals with impressive biological activities. Trends Food Sci. Technol. 2015, 44, 93–104. [Google Scholar] [CrossRef]
- Yazaki, K.; Sasaki, K.; Tsurumaru, Y. Prenylation of aromatic compounds, a key diversification of plant secondary metabolites. Phytochemistry 2009, 70, 1739–1745. [Google Scholar] [CrossRef]
- Stevens, J.F.; Page, J.E. Xanthohumol and related prenylflavonoids from hops and beer: To your good health! Phytochemistry 2004, 65, 1317–1330. [Google Scholar] [CrossRef]
- Araújo, J.R.; Gonçalves, P.; Martel, F. Chemopreventive effect of dietary polyphenols in colorectal cancer cell lines. Nutr. Res. 2011, 31, 77–87. [Google Scholar] [CrossRef]
- Miranda, C.L.; Stevens, J.F.; Helmrich, A.; Henderson, M.C.; Rodriguez, R.J.; Yang, Y.H.; Deinzer, M.L.; Barnes, D.W.; Buhler, D.R. Antiproliferative and cytotoxic effects of prenylated flavonoids from hops (Humulus lupulus) in human cancer cell lines. Food Chem. Toxicol. 1999, 37, 271–285. [Google Scholar] [CrossRef]
- Delmulle, L.; Bellahcène, A.; Dhooge, W.; Comhaire, F.; Roelens, F.; Huvaere, K.; Heyerick, A.; Castronovo, V.; De Keukeleire, D. Anti-proliferative properties of prenylated flavonoids from hops (Humulus lupulus L.) in human prostate cancer cell lines. Phytomedicine 2006, 13, 732–734. [Google Scholar] [CrossRef] [PubMed]
- Colgate, E.C.; Miranda, C.L.; Stevens, J.F.; Bray, T.M.; Ho, E. Xanthohumol, a prenylflavonoid derived from hops induces apoptosis and inhibits NF-kappaB activation in prostate epithelial cells. Cancer Lett. 2007, 246, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Harikumar, K.B.; Kunnumakkara, A.B.; Ahn, K.S.; Anand, P.; Krishnan, S.; Guha, S.; Aggarwal, B.B. Modification of the cysteine residues in IκBα kinase and NF-κB (p65) by xanthohumol leads to suppression of NF-κB-regulated gene products and potentiation of apoptosis in leukemia cells. Blood 2009, 113, 2003–2013. [Google Scholar] [CrossRef] [PubMed]
- Deeb, D.; Gao, X.; Jiang, H.; Arbab, A.S.; Dulchavsky, S.A.; Gautam, S.C. Growth inhibitory and apoptosis-inducing effects of xanthohumol, a prenylated chalone present in hops, in human prostate cancer cells. Anticancer Res. 2010, 30, 3333–3339. [Google Scholar]
- Miranda, C.L.; Elias, V.D.; Hay, J.J.; Choi, J.; Reed, R.L.; Stevens, J.F. Xanthohumol improves dysfunctional glucose and lipid metabolism in diet-induced obese C57BL/6J mice. Arch. Biochem. Biophys. 2016, 599, 22–30. [Google Scholar] [CrossRef]
- Legette, L.L.; Moreno Luna, A.Y.; Reed, R.L.; Miranda, C.L.; Bobe, G.; Proteau, R.R.; Stevens, J.F. Xanthohumol lowers body weight and fasting plasma glucose in obese male Zucker fa/fa rats. Phytochemistry 2013, 91, 236–241. [Google Scholar] [CrossRef]
- Mahli, A.; Seitz, T.; Freese, K.; Frank, J.; Weiskirchen, R.; Abdel-Tawab, M.; Behnam, D.; Hellerbrand, C. Therapeutic Application of Micellar Solubilized Xanthohumol in a Western-Type Diet-Induced Mouse Model of Obesity, Diabetes and Non-Alcoholic Fatty Liver Disease. Cells 2019, 8, 359. [Google Scholar] [CrossRef]
- Gerhäuser, C. Beer constituents as potential cancer chemopreventive agents. Eur. J. Cancer 2005, 41, 1941–1954. [Google Scholar] [CrossRef]
- Liu, M.; Hansen, P.E.; Wang, G.; Qiu, L.; Dong, J.; Yin, H.; Qian, Z.; Yang, M.; Miao, J. Pharmacological profile of xanthohumol, a prenylated flavonoid from hops (Humulus lupulus). Molecules 2015, 20, 754–779. [Google Scholar] [CrossRef]
- Racolta, E.; Tofana, M.; Muresan, C.C.; Socaci, S.; Florin, G.G.; Vlad, M. A Hop-Derived Prenylflavonoids and Their Importance in Brewing Technology: A Review. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca Food Sci. Technol. 2015, 72, 107–113. [Google Scholar] [CrossRef]
- Prencipe, F.; Brighenti, V.; Rodolfi, M.; Mongelli, A.; Ganino, T.; Bruni, R.; Pellati, F. Development of a new high-performance liquid chromatography method with diode array and electrospray ionization-mass spectrometry detection for the metabolite fingerprinting of bioactive compounds in Humulus lupulus L. J. Chromatogr. A 2014, 1349, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Intelmann, D.; Haseleu, G.; Hofmann, T. LC-MS/MS quantitation of hop-derived bitter compounds in beer using the ECHO technique. J. Agric. Food Chem. 2009, 57, 1172–1182. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.F.; Taylor, A.W.; Deinzer, M.L. Quantitative analysis of xanthohumol and related prenylflavonoids in hops and beer by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 1999, 832, 97–107. [Google Scholar] [CrossRef]
- Sandoval-Ramírez, B.A.; Lamuela-Raventós, R.M.; Estruch, R.; Sasot, G.; Doménech, M.; Tresserra-Rimbau, A. Beer Polyphenols and Menopause: Effects and Mechanisms—A Review of Current Knowledge. Oxid. Med. Cell. Longev. 2017, 2017. [Google Scholar] [CrossRef]
- Rothwell, J.A.; Perez-Jimenez, J.; Neveu, V.; Medina-Remón, A.; M’Hiri, N.; García-Lobato, P.; Manach, C.; Knox, C.; Eisner, R.; Wishart, D.S.; et al. Phenol-Explorer 3.0: A major update of the Phenol-Explorer database to incorporate data on the effects of food processing on polyphenol content. Database 2013, 2013, 1–8. [Google Scholar] [CrossRef]
- Karabin, M.; Hudcova, T.; Jelinek, L.; Dostalek, P. Biotransformations and biological activities of hop flavonoids. Biotechnol. Adv. 2015, 33, 1063–1090. [Google Scholar] [CrossRef]
- Wesołowska, O.; Gąsiorowska, J.; Petrus, J.; Czarnik-Matusewicz, B.; Michalak, K. Interaction of prenylated chalcones and flavanones from common hop with phosphatidylcholine model membranes. Biochim. Biophys. Acta-Biomembr. 2014, 1838, 173–184. [Google Scholar] [CrossRef]
- Schmandke, H. Prenyl flavonoids in hops and beer—biochemical and biological activities. Ernährungs-Umschau 2006, 53, 225–229. [Google Scholar]
- Fang, J.; Nikolić, D.; Chen, S.; Alvarenga, R.R.; Pauli, G.; van Breemen, R. Structure Determination of Isoxanthohumol and 8-Prenylnaringenin Glucuronides Formed by Human Liver Microsomes. Planta Med. 2013, 79, PR4. [Google Scholar] [CrossRef]
- Nikolic, D.; Li, Y.; Chadwick, L.R.; Pauli, G.F.; Van Breemen, R.B. Metabolism of xanthohumol and isoxanthohumol, prenylated flavonoids from hops (Humulus lupulus L.), by human liver microsomes. J. Mass Spectrom. 2005, 40, 289–299. [Google Scholar] [CrossRef]
- Possemiers, S.; Bolca, S.; Grootaert, C.; Heyerick, A.; Decroos, K.; Dhooge, W.; De Keukeleire, D.; Rabot, S.; Verstraete, W.; Van de Wiele, T. The Prenylflavonoid Isoxanthohumol from Hops (Humulus lupulus L.) Is Activated into the Potent Phytoestrogen 8-Prenylnaringenin In Vitro and in the Human Intestine. J. Nutr. 2006, 136, 1862–1867. [Google Scholar] [CrossRef] [PubMed]
- Jian, G.; Dejan, N.; Lucas, R.C.; Guido, F.P.; van Breemen, R.B. Identification of human hepatic cytochrome P450 enzymes involved in the metabolism of 8-prenylnaringenin and isoxanthohumol from hops (Humulus lupulus L.). Am. Soc. Pharmacol. Exp. Ther. 2006, 34, 1152–1159. [Google Scholar] [CrossRef]
- Bolca, S.; Possemiers, S.; Maervoet, V.; Huybrechts, I.; Heyerick, A.; Vervarcke, S.; Depypere, H.; De Keukeleire, D.; Bracke, M.; De Henauw, S.; et al. Microbial and dietary factors associated with the 8-prenylnaringenin producer phenotype: A dietary intervention trial with fifty healthy post-menopausal Caucasian women. Br. J. Nutr. 2007, 98, 950–959. [Google Scholar] [CrossRef]
- Bolca, S.; Wyns, C.; Possemiers, S.; Depypere, H.; De Keukeleire, D.; Bracke, M.; Verstraete, W.; Heyerick, A. Cosupplementation of Isoflavones, Prenylflavonoids, and Lignans Alters Human Exposure to Phytoestrogen-Derived. Nutr. Physiol. Metab. Nutr. Interact. 2009, 139, 2293–2300. [Google Scholar] [CrossRef]
- Bolca, S.; Li, J.; Nikolic, D.; Roche, N.; Blondeel, P.; Possemiers, S.; De Keukeleire, D.; Bracke, M.; Heyerick, A.; Van Breemen, R.B.; et al. Disposition of hop prenylflavonoids in human breast tissue. Mol. Nutr. Food Res. 2010, 54, 284–294. [Google Scholar] [CrossRef]
- Schaefer, O.; Bohlmann, R.; Schleuning, W.D.; Schulze-Forster, K.; Hümpel, M. Development of a radioimmunoassay for the quantitative determination of 8-prenylnaringenin in biological matrices. J. Agric. Food Chem. 2005, 53, 2881–2889. [Google Scholar] [CrossRef]
- van Breemen, R.B.; Yuan, Y.; Banuvar, S.; Shulman, L.P.; Qiu, X.; Ramos Alvarenga, R.F.; Chen, S.N.; Dietz, B.M.; Bolton, J.L.; Pauli, G.F.; et al. Pharmacokinetics of prenylated hop phenols in women following oral administration of a standardized extract of hops. Mol. Nutr. Food Res. 2014, 58, 1962–1969. [Google Scholar] [CrossRef]
- Fang, J.B.; Nikolić, D.; Lankin, D.C.; Simmler, C.; Chen, S.N.; Ramos Alvarenga, R.F.; Liu, Y.; Pauli, G.F.; Van Breemen, R.B. Formation of (2R)- And (2S)-8-Prenylnaringenin Glucuronides by Human UDP-Glucuronosyltransferases. J. Agric. Food Chem. 2019, 67, 11650–11656. [Google Scholar] [CrossRef]
- Nikolic, D.; Li, Y.; Chadwick, L.R.; Grubjesic, S.; Schwab, P.; Metz, P.; Van Breemen, R.B. Metabolism of 8-prenylnaringenin, a potent phytoestrogen from hops (Humulus lupulus), by human liver microsomes. Drug Metab. Dispos. 2004, 32, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Rad, M.; Hümpel, M.; Schaefer, O.; Schoemaker, R.C.; Schleuning, W.D.; Cohen, A.F.; Burggraaf, J. Pharmacokinetics and systemic endocrine effects of the phyto-oestrogen 8-prenylnaringenin after single oral doses to postmenopausal women. Br. J. Clin. Pharmacol. 2006, 62, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Connell, O. Pharmacokinetic and Pharmacologic Variation Between Different Estrogen Products. J. Clin. Pharmacol. 1995, 35, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Castro, L.A.; Burkard, M.; Sus, N.; Scheubeck, G.; Leischner, C.; Lauer, U.M.; Bosy-Westphal, A.; Hund, V.; Busch, C.; Venturelli, S.; et al. The Oral Bioavailability of 8-Prenylnaringenin from Hops (Humulus lupulus L.) in Healthy Women and Men is Significantly Higher than that of its Positional Isomer 6-Prenylnaringenin in a Randomized Crossover Trial. Mol. Nutr. Food Res. 2018, 62, 1–27. [Google Scholar] [CrossRef]
- Mueller, S.O. Overview of in vitro tools to assess the estrogenic and antiestrogenic activity of phytoestrogens. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2002, 777, 155–165. [Google Scholar] [CrossRef]
- Gruber, C.J.; Tschugguel, W.; Schneeberger, C.; Huber, J.C. Produkction and Actions of Esterogen. J. Med. 2002, 346, 340–352. [Google Scholar]
- Farzaneh, S.; Zarghi, A. Estrogen receptor ligands: A review (2013–2015). Sci. Pharm. 2016, 84, 409–427. [Google Scholar] [CrossRef]
- Štulíková, K.; Karabín, M.; Nešpor, J.; Dostálek, P. Therapeutic perspectives of 8-prenylnaringenin, a potent phytoestrogen from hops. Molecules 2018, 23, 660. [Google Scholar] [CrossRef]
- Messina, M.; Kucuk, O.; Lampe, J.W. An overview of the health effects of isoflavones with an emphasis on prostate cancer risk and prostate-specific antigen levels. J. AOAC Int. 2006, 89, 1121–1134. [Google Scholar] [CrossRef]
- Milligan, S.R. Reproductive and estrogenic effects of 8-prenylnaringenin in hops. In Beer in Health and Disease Prevention; Preedy, V.R., Ed.; Academic Press: Cambridge, MA, USA, 2009; pp. 711–723. [Google Scholar]
- Milligan, S.R.; Kalita, J.C.; Heyerick, A.; Rong, H.; De Cooman, L.; De Keukeleire, D. Identification of a potent phytoestrogen in hops (Humulus lupulus L.) and beer. J. Clin. Endocrinol. Metab. 1999, 83, 2249–2252. [Google Scholar] [CrossRef]
- Koch, W.; Heim, G. Östrogene Hormone in Hopfen und Bier. München. Med. Wchnschr. 1953, 95, 845. [Google Scholar]
- Bednar, I.; Zenisek, A. Identification of the estrogenic activity of hops. Brauwissenschaft 1961, 14, 4–7. [Google Scholar]
- Nastainczyk, W. Untersuchung über die östrogene Wirkung des Hopfens und des Bieres. Ph.D. Thesis, Universitat des Saarlandes, Saarbrucken, Germany, 1972. [Google Scholar]
- Hänsel, R.; Schulz, J. Desmethylxanthohumol: Isolierung aus Hopfen und Cyclisierung zu Flavanonen. Arch. Pharm. 1988, 321, 37–40. [Google Scholar] [CrossRef]
- Milligan, S.R.; Kalita, J.C.; Heyerick, A.; Rong, H.; De Cooman, L.; Keukeleire, D. De Identification of the estrogenic activity of hops. J. Clin. Endocrinol. Metab. 1999, 84, 2249–2252. [Google Scholar] [CrossRef] [PubMed]
- Milligan, S.; Kalita, J.; Pocock, V.; Heyerick, A.; De Cooman, L.; Rong, H.; De Keukeleire, D. Oestrogenic activity of the hop phyto-oestrogen, 8-prenylnaringenin. Reproduction 2002, 123, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Milligan, S.R.; Kalita, J.C.; Pocock, V.; Van De Kauter, V.; Stevens, J.F.; Deinzer, M.L.; Rong, H.; De Keukeleire, D. The endocrine activities of 8-prenylnaringenin and related hop (Humulus lupulus L.) flavonoids. J. Clin. Endocrinol. Metab. 2000, 85, 4912–4915. [Google Scholar] [CrossRef] [PubMed]
- Rong, H.; Boterberg, T.; Maubach, J.; Stove, C.; Depypere, H.; Van Slambrouck, S.; Serreyn, R.; De Keukeleire, D.; Mareel, M.; Bracke, M. 8-prenylnaringenin, the phytoestrogen in hops and beer, upregulates the function of the E-cadherin/catenin complex in human mammary carcinoma cells. Eur. J. Cell Biol. 2001, 80, 580–585. [Google Scholar] [CrossRef]
- Coldham, N.G.; Sauer, M.J. Identification, quantitation and biological activity of phytoestrogens in a dietary supplement for breast enhancement. Food Chem. Toxicol. 2001, 39, 1211–1224. [Google Scholar] [CrossRef]
- Zierau, O.; Gester, S.; Schwab, P.; Metz, P.; Kolba, S.; Wulf, M.; Vollmer, G. Estrogenic activity of the phytoestrogens naringenin, 6-(1,1-dimethylallyl)naringenin and 8-prenylnaringenin. Planta Med. 2002, 68, 449–451. [Google Scholar] [CrossRef]
- Setchell, K.D.R.; Cassidy, A. Symposium on Phytochemicals: Biochemistry and Physiology Dietary Isoflavones: Biological Effects and Relevance to Human Health. J. Nutr. 1999, 129, 758–767. [Google Scholar] [CrossRef]
- Hümpel, M.; Schleuning, W.-D.; Schaefer, O.; Isaksson, P.; Bohlmann, R. Use of 8-Penylnaringenin for Hormone Replacement Therapy. EP1524269, 7 October 2003.
- Kitaoka, M.; Kadokawa, H.; Sugano, M.; Ichikawa, K.; Taki, M.; Takaishi, S.; Tsutsumi, S.; Boriboon, M.; Akiyama, T. Prenylflavonoids: A new class of non-steroidal phytoestrogen (Part 1). Isolation of 8-isopentenylnaringenin and an initial study on its structure-activity relationship. Planta Med. 1998, 64, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Kretzschmar, G.; Zierau, O.; Wober, J.; Tischer, S.; Metz, P.; Vollmer, G. Prenylation has a compound specific effect on the estrogenicity of naringenin and genistein. J. Steroid Biochem. Mol. Biol. 2010, 118, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Mao, Z.; Brinton, R.D. A Select Combination of Clinically Relevant. Neuroendocrinology 2015, 150, 770–783. [Google Scholar] [CrossRef]
- Simons, R.; Gruppen, H.; Bovee, T.F.H.; Verbruggen, M.A.; Vincken, J.P. Prenylated isoflavonoids from plants as selective estrogen receptor modulators (phytoSERMs). Food Funct. 2012, 3, 810–827. [Google Scholar] [CrossRef] [PubMed]
- Bolego, C.; Poli, A.; Cignarella, A.; Paoletti, R. Phytoestrogens: Pharmacological and Therapeutic Perspectives. Curr. Drug Targets 2005, 4, 77–87. [Google Scholar] [CrossRef]
- Dietz, B.M.; Hagos, G.K.; Eskra, J.N.; Wijewickrama, G.T.; Anderson, J.R.; Nikolic, D.; Guo, J.; Wright, B.; Chen, S.N.; Pauli, G.F.; et al. Differential regulation of detoxification enzymes in hepatic and mammary tissue by hops (Humulus lupulus) in vitro and in vivo. Mol. Nutr. Food Res. 2013, 57, 1055–1066. [Google Scholar] [CrossRef]
- Hoffman, B.; Schorge, J.; Schaffer, J.; Halvorson, L.; Bradshaw, K. Evaluation of the Infertile Couple in: Williams Gynecology, 2nd ed.; McGraw-Hill: New York, NY, USA, 2012; ISBN 978-0-07-171672-7. [Google Scholar]
- Baig, L.; Mansuri, F.A.; Karim, S.A. Association of menopause with osteopenia and osteoporosis: Results from population based study done in Karachi. J. Coll. Physicians Surg. Pak. 2009, 19, 240–244. [Google Scholar]
- Ebong, I.A.; Watson, K.E.; Goff, D.C.; Bluemke, D.A.; Srikanthan, P.; Horwich, T.; Bertoni, A.G. Age at menopause and incident heart failure: The Multi-Ethnic Study of Atherosclerosis. Menopause 2014, 21, 585–591. [Google Scholar] [CrossRef]
- Wellons, M.; Ouyang, P.; Schreiner, P.J.; Herrington, D.M.; Vaidya, D. Early menopause predicts future coronary heart disease and stroke: The Multi-Ethnic Study of Atherosclerosis. Menopause 2012, 19, 1081–1087. [Google Scholar] [CrossRef]
- Velders, M.; Diel, P. How Sex Hormones Promote Skeletal Muscle Regeneration. Sports Med. 2013, 43, 1089–1100. [Google Scholar] [CrossRef]
- Jacobsen, D.E.; Samson, M.M.; Kezic, S.; Verhaar, H.J.J. Postmenopausal HRT and tibolone in relation to muscle strength and body composition. Maturitas 2007, 58, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Mukai, R.; Horikawa, H.; Lin, P.; Tsukumo, N.; Nikawa, T.; Kawamura, T.; Nemoto, H.; Terao, J.; Nemoto, H.; Terao, J. 8-Prenylnaringenin promotes recovery from immobilization-induced disuse muscle atrophy through activation of the Akt phosphorylation pathway in mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2016, 311, 1022–1031. [Google Scholar] [CrossRef] [PubMed]
- Howard, B.V.; Van Horn, L.; Hsia, J.; Manson, J.E.; Stefanick, M.L.; Wassertheil-Smoller, S.; Kuller, L.H.; LaCroix, A.Z.; Langer, R.D.; Lasser, N.L.; et al. Risks and Benefits of Estrogen Plus Progestin in Healthy Postmenopausal Women: Principal results from the women’s health initiative randomized controlled trial. J. Am. Med. Assoc. 2002, 288, 321–333. [Google Scholar] [CrossRef]
- Dietz, B.M.; Hajirahimkhan, A.; Dunlap, T.L.; Bolton, J.L. Botanicals and their bioactive phytochemicals for women’s health. Pharmacol. Rev. 2016, 68, 1026–1073. [Google Scholar] [CrossRef] [PubMed]
- Pacifi, R. Editorial: Cytokines, estrogen, and postmenopausal osteoporosis—The second decade. Endocrinology 1998, 139, 2659–2661. [Google Scholar] [CrossRef]
- Osterkamp, R. Bevölkerungsentwicklung in Deutschland bis 2050: Demografische und ökonomische konsequenzen für die alterschirurgie. Chirurg 2005, 76, 10–18. [Google Scholar] [CrossRef]
- Miyamoto, M.; Matsushita, Y.; Kiyokawa, A.; Fukuda, C.; Iijima, Y.; Sugano, M.; Akiyama, T. Prenylflavonoids: A new class of non-steroidal phytoestrogen (Part 2). Estrogenic effects of 8-isopentenylnaringenin on bone metabolism. Planta Med. 1998, 64, 516–519. [Google Scholar] [CrossRef]
- Sehmisch, S.; Hammer, F.; Christoffel, J.; Seidlova-Wuttke, D.; Tezval, M.; Wuttke, W.; Stuermer, K.M.; Stuermer, E.K. Comparison of the phytohormones genistein, resveratrol and 8-prenylnaringenin as agents for preventing osteoporosis. Planta Med. 2008, 74, 794–801. [Google Scholar] [CrossRef]
- Komrakova, M.; Rechholtz, C.; Pohlmann, N.; Lehmann, W.; Schilling, A.F.; Wigger, R.; Sehmisch, S.; Hoffmann, D.B. Effect of alendronate or 8-prenylnaringenin applied as a single therapy or in combination with vibration on muscle structure and bone healing in ovariectomized rats. Bone Rep. 2019, 11, 100224. [Google Scholar] [CrossRef]
- Hoffmann, D.B.; Griesel, M.H.; Brockhusen, B.; Tezval, M.; Komrakova, M.; Menger, B.; Wassmann, M.; Stuermer, K.M.; Sehmisch, S. Effects of 8-Prenylnaringenin and Whole-Body Vibration Therapy on a Rat Model of Osteopenia. J. Nutr. Metab. 2016, 2016. [Google Scholar] [CrossRef]
- Keiler, A.M.; Helle, J.; Bader, M.I.; Ehrhardt, T.; Nestler, K.; Kretzschmar, G.; Bernhardt, R.; Vollmer, G.; Nikolić, D.; Bolton, J.L.; et al. A standardized Humulus lupulus (L.) ethanol extract partially prevents ovariectomy-induced bone loss in the rat without induction of adverse effects in the uterus. Phytomedicine 2017, 34, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Freedman, R.R. Hot flashes: Behavioral treatments, mechanisms, and relation to sleep. Am. J. Med. 2005, 118, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Bowe, J.; Li, X.F.; Kinsey-jones, J.; Heyerick, A.; Brain, S.; Milligan, S.; Byrne, K.O. The hop phytoestrogen, 8-prenylnaringenin, reverses the ovariectomy-induced rise in skin temperature in an animal model of menopausal hot flushes. J. Endocrinol. 2006, 191, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Heyerick, A.; Vervarcke, S.; Depypere, H.; Bracke, M.; De Keukeleire, D. A first prospective, randomized, double-blind, placebo-controlled study on the use of a standardized hop extract to alleviate menopausal discomforts. Maturitas 2006, 54, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Erkkola, R.; Vervarcke, S.; Vansteelandt, S.; Rompotti, P.; De Keukeleire, D.; Heyerick, A. A randomized, double-blind, placebo-controlled, cross-over pilot study on the use of a standardized hop extract to alleviate menopausal discomforts. Phytomedicine 2010, 17, 389–396. [Google Scholar] [CrossRef]
- Aghamiri, V.; Mirghafourvand, M.; Mohammad-Alizadeh-Charandabi, S.; Nazemiyeh, H. The effect of Hop (Humulus lupulus L.) on early menopausal symptoms and hot flashes: A randomized placebo-controlled trial. Complement. Ther. Clin. Pract. 2016, 23, 130–135. [Google Scholar] [CrossRef]
- Fennell, T.R.; Mathews, J.M.; Snyder, R.W.; Hong, Y.; Watson, S.L.; Black, S.R.; McIntyre, B.S.; Waidyanatha, S. Metabolism and disposition of 2-ethylhexyl-p-methoxycinnamate following oral gavage and dermal exposure in Harlan Sprague Dawley rats and B6C3F1/N mice and in hepatocytes in vitro. Xenobiotica 2018, 48, 1142–1156. [Google Scholar] [CrossRef]
- Taylor, M. Complementary and Alternative Approaches to Menopause. Endocrinol. Metab. Clin. N. Am. 2015, 44, 619–648. [Google Scholar] [CrossRef]
- Keiler, A.M.; Zierau, O.; Kretzschmar, G. Hop extracts and hop substances in treatment of menopausal complaints. Planta Med. 2013, 79, 576–579. [Google Scholar] [CrossRef]
- Solak, K.A.; Santos, R.R.; van den Berg, M.; Blaauboer, B.J.; Roelen, B.A.J.; van Duursen, M.B.M. Naringenin (NAR) and 8-prenylnaringenin (8-PN) reduce the developmental competence of porcine oocytes in vitro. Reprod. Toxicol. 2014, 49, 1–11. [Google Scholar] [CrossRef]
- Fugh-Berman, A. “Bust enhancing” herbal products. Obstet. Gynecol. 2003, 101, 1345–1349. [Google Scholar] [CrossRef] [PubMed]
- Di Vito, C.; Bertoni, A.; Nalin, M.; Sampietro, S.; Zanfa, M.; Sinigaglia, F. The phytoestrogen 8-prenylnaringenin inhibits agonist-dependent activation of human platelets. Biochim. Biophys. Acta Gen. Subj. 2012, 1820, 1724–1733. [Google Scholar] [CrossRef] [PubMed]
- Myasoedova, V.A.; Kirichenko, T.V.; Melnichenko, A.A.; Orekhova, V.A.; Ravani, A.; Poggio, P.; Sobenin, I.A.; Bobryshev, Y.V.; Orekhov, A.N. Anti-atherosclerotic effects of a phytoestrogen-rich herbal preparation in postmenopausal women. Int. J. Mol. Sci. 2016, 17, 1318. [Google Scholar] [CrossRef] [PubMed]
- Benkherouf, A.Y.; Soini, S.L.; Stompor, M.; Uusi-Oukari, M. Positive allosteric modulation of native and recombinant GABAA receptors by hops prenylflavonoids. Eur. J. Pharmacol. 2019, 852, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Mukai, R.; Horikawa, H.; Fujikura, Y.; Kawamura, T.; Nemoto, H.; Nikawa, T.; Terao, J. Prevention of Disuse Muscle Atrophy by Dietary Ingestion of 8-Prenylnaringenin in Denervated Mice. PLoS ONE 2012, 7, e45048. [Google Scholar] [CrossRef]
- Urmann, C.; Oberbauer, E.; Couillard-Després, S.; Aigner, L.; Riepl, H. Neurodifferentiating potential of 8-prenylnaringenin and related compounds in neural precursor cells and correlation with estrogen-like activity. Planta Med. 2015, 81, 305–311. [Google Scholar] [CrossRef]
- Miranda, C.L.; Stevens, J.F.; Ivanov, V.; McCall, M.; Frei, B.; Deinzer, M.L.; Buhler, D.R. Antioxidant and prooxidant actions of prenylated and nonprenylated chalcones and flavanones in vitro. J. Agric. Food Chem. 2000, 48, 3876–3884. [Google Scholar] [CrossRef]
- Gerhauser, C.; Alt, A.; Heiss, E.; Gamal-Eldeen, A.; Klimo, K.; Knauft, J.; Neumann, I.; Scherf, H.R.; Frank, N.; Bartsch, H.; et al. Cancer chemopreventive activity of xanthohumol, a natural product derived from hop. Mol. Cancer Ther. 2002, 1, 959–969. [Google Scholar]
- Santos, C.M.M.; Silva, A.M.S. The antioxidant activity of prenylflavonoids. Molecules 2020, 25, 696. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Stubert, J.; Gerber, B. Isoflavones—Mechanism of action and impact on breast cancer risk. Breast Care 2009, 4, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, A.; Ghosh, A.; Pope, G.S.; Darbre, P.D. Comparative study of oestrogenic properties of eight phytoestrogens in MCF7 human breast cancer cells. J. Steroid Biochem. Mol. Biol. 2005, 94, 431–443. [Google Scholar] [CrossRef] [PubMed]
- Piersen, C.E. Phytoestrogens in Botanical Dietary Supplements: Implications for Cancer legumes. Integr. Cancer Ther. 2003, 2, 120–138. [Google Scholar] [CrossRef] [PubMed]
- Wuttke, W.; Jarry, H.; Seidlová-Wuttke, D. Isoflavones-Safe food additives or dangerous drugs? Ageing Res. Rev. 2007, 6, 150–188. [Google Scholar] [CrossRef]
- Powers, C.N.; Setzer, W.N. A molecular docking study of phytochemical estrogen mimics from dietary herbal supplements. In Silico Pharmacol. 2015, 3, 4. [Google Scholar] [CrossRef]
- Bartmańska, A.; Tronina, T.; Popłoński, J.; Milczarek, M.; Filip-Psurska, B.; Wietrzyk, J. Highly cancer selective antiproliferative activity of natural prenylated flavonoids. Molecules 2018, 23, 2922. [Google Scholar] [CrossRef]
- Brunelli, E.; Minassi, A.; Appendino, G.; Moro, L. 8-Prenylnaringenin, inhibits estrogen receptor-α mediated cell growth and induces apoptosis in MCF-7 breast cancer cells. J. Steroid Biochem. Mol. Biol. 2007, 107, 140–148. [Google Scholar] [CrossRef]
- Helle, J.; Kräker, K.; Bader, M.I.; Keiler, A.M.; Zierau, O.; Vollmer, G.; Welsh, J.E.; Kretzschmar, G. Assessment of the proliferative capacity of the flavanones 8-prenylnaringenin, 6-(1.1-dimethylallyl)naringenin and naringenin in MCF-7 cells and the rat mammary gland. Mol. Cell. Endocrinol. 2014, 392, 125–135. [Google Scholar] [CrossRef]
- Wang, S.; Dunlap, T.L.; Howell, C.E.; Mbachu, O.C.; Rue, E.A.; Phansalkar, R.; Chen, S.N.; Pauli, G.F.; Dietz, B.M.; Bolton, J.L. Hop (Humulus lupulus L.) Extract and 6-Prenylnaringenin Induce P450 1A1 Catalyzed Estrogen 2-Hydroxylation. Chem. Res. Toxicol. 2016, 29, 1142–1150. [Google Scholar] [CrossRef]
- Dietz, B.M.; Chen, S.N.; Alvarenga, R.F.R.; Dong, H.; Nikolić, D.; Biendl, M.; Van Breemen, R.B.; Bolton, J.L.; Pauli, G.F. DESIGNER Extracts as Tools to Balance Estrogenic and Chemopreventive Activities of Botanicals for Women’s Health. J. Nat. Prod. 2017, 80, 2284–2294. [Google Scholar] [CrossRef]
- Venturelli, S.; Niessner, H.; Sinnberg, T.; Berger, A.; Burkard, M.; Urmann, C.; Donaubauer, K.; Böcker, A.; Leischner, C.; Riepl, H.; et al. 6- and 8-Prenylnaringenin, novel natural histone deacetylase inhibitors found in hops, exert antitumor activity on melanoma cells. Cell. Physiol. Biochem. 2018, 51, 543–556. [Google Scholar] [CrossRef]
- Busch, C.; Noor, S.; Leischner, C.; Burkard, M.; Lauer, U.M.; Venturelli, S. Antiproliferative Aktivität gegenüber humanen Krebszellen von Prenylflavonoiden aus dem Hopfen. Wien. Med. Wochenschr. 2015, 165, 258–261. [Google Scholar] [CrossRef] [PubMed]
- Delmulle, L.; Vanden Berghe, T.; De Keukeleire, D.; Vandenabeele, P. Treatment of PC-3 and DU145 Prostate Cancer Cells by Prenylflavonoids from Hop (Humulus lupulus L.) induces a Caspase-independent Form of Cell Death. Phytother. Res. 2008, 203, 197–203. [Google Scholar] [CrossRef]
- Miranda, C.L.; Aponso, G.L.M.; Stevens, J.F.; Deinzer, M.L.; Buhler, D.R. Prenylated chalcones and flavanones as inducers of quinone reductase in mouse Hepa 1c1c7 cells. Cancer Lett. 2000, 149, 21–29. [Google Scholar] [CrossRef]
- Gerhäuser, C.; Alt, A.P.; Klimo, K.; Knauft, J.; Frank, N.; Becker, H. Isolation and potential cancer chemopreventive activities of phenolic compounds of beer. Phytochem. Rev. 2002, 1, 369–377. [Google Scholar] [CrossRef]
- Rodriguez, R.J.; Miranda, C.L.; Stevens, J.F.; Deinzer, M.L.; Buhler, D.R. Influence of prenylated and non-prenylated flavonoids on liver microsomal lipid peroxidation and oxidative injury in rat hepatocytes. Food Chem. Toxicol. 2001, 39, 437–445. [Google Scholar] [CrossRef]
- Henderson, M.C.; Miranda, C.L.; Stevens, J.F.; Deinzer, M.L.; Buhler, D.R. In vitro inhibition of human P450 enzymes by prenylated flavonoids from hops, Humulus lupulus. Xenobiotica 2000, 30, 235–251. [Google Scholar] [CrossRef]
- Bertl, E.; Klimo, K.; Heiss, E.; Klenke, F.; Peschke, P.; Becker, H.; Eicher, T.; Herhaus, C.; Kapadia, G.; Bartsch, H.; et al. Identification of Novel Inhibitors of Angiogenesis using a Human In Vitro Anti-angiogenic Assay. Int. J. Cancer Res. Prev. 2004, 1, 47–61. [Google Scholar]
- Koosha, S.; Mohamed, Z.; Sinniah, A.; Ibrahim, Z.A.; Seyedan, A.; Alshawsh, M.A. Antiproliferative and apoptotic activities of 8-prenylnaringenin against human colon cancer cells. Life Sci. 2019, 232, 116633. [Google Scholar] [CrossRef]
- Tan, K.W.; Cooney, J.; Jensen, D.; Li, Y.; Paxton, J.W.; Birch, N.P.; Scheepens, A. Hop-derived prenylflavonoids are substrates and inhibitors of the efflux transporter breast cancer resistance protein (BCRP/ABCG2). Mol. Nutr. Food Res. 2014, 58, 2099–2110. [Google Scholar] [CrossRef]
- Natarajan, K.; Xie, Y.; Baer, M.R.; Ross, D.D. Role of breast cancer resistance protein (BCRP/ABCG2) in cancer drug resistance. Biochem. Pharmacol. 2012, 83, 1084–1103. [Google Scholar] [CrossRef]
- Antman, E.M.; Bennett, J.S.; Daugherty, A.; Furberg, C.; Roberts, H.; Taubert, K.A. Use of nonsteroidal antiinflammatory drugs: An update for clinicians: A scientific statement from the American Heart Association. Circulation 2007, 115, 1634–1642. [Google Scholar] [CrossRef] [PubMed]
- Tolbert, T.; Thompson, J.A.; Bouchard, P.; Oparil, S. Estrogen-induced vasoprotection is independent of inducible nitric oxide synthase expression: Evidence from the mouse carotid artery ligation model. Circulation 2001, 104, 2740–2745. [Google Scholar] [CrossRef]
- Biesalski, H.K. Polyphenols and inflammation: Basic interactions. Curr. Opin. Clin. Nutr. Metab. Care 2007, 10, 724–728. [Google Scholar] [CrossRef]
- Oseni, T.; Patel, R.; Pyle, J.; Jordan, V.C. Selective estrogen receptor modulators and phytoestrogens. Planta Med. 2008, 74, 1656–1665. [Google Scholar] [CrossRef]
- Hermenegildo, C.; Oviedo, P.J.; Garcı, M.A.; Tarı, J.J.; Cano, A. Effects of Phytoestrogens Genistein and Daidzein on Prostacyclin Production by Human Endothelial Cells. J. Pharmacol. Exp. Ther. 2005, 315, 722–728. [Google Scholar] [CrossRef]
- Negrão, R.; Costa, R.; Duarte, D.; Taveira Gomes, T.; Mendanha, M.; Moura, L.; Vasques, L.; Azevedo, I.; Soares, R. Angiogenesis and inflammation signaling are targets of beer polyphenols on vascular cells. J. Cell. Biochem. 2010, 111, 1270–1279. [Google Scholar] [CrossRef]
- Paoletti, T.; Fallarini, S.; Gugliesi, F.; Minassi, A.; Appendino, G.; Lombardi, G. Anti-inflammatory and vascularprotective properties of 8-prenylapigenin. Eur. J. Pharmacol. 2009, 620, 120–130. [Google Scholar] [CrossRef]
- Costa, R.; Rodrigues, I.; Guardão, L.; Rocha-Rodrigues, S.; Silva, C.; Magalhães, J.; Ferreira-de-Almeida, M.; Negrão, R.; Soares, R. Xanthohumol and 8-prenylnaringenin ameliorate diabetic-related metabolic dysfunctions in mice. J. Nutr. Biochem. 2017, 45, 39–47. [Google Scholar] [CrossRef]
- Costa, R.; Rodrigues, I.; Guardão, L.; Lima, J.Q.; Sousa, E.; Soares, R.; Negrão, R. Modulation of VEGF signaling in a mouse model of diabetes by xanthohumol and 8-prenylnaringenin: Unveiling the angiogenic paradox and metabolism interplay. Mol. Nutr. Food Res. 2017, 61, 1–27. [Google Scholar] [CrossRef]
- Sakamoto, K.; Konings, W.N. Beer spoilage bacteria and hop resistance. Int. J. Food Microbiol. 2003, 89, 105–124. [Google Scholar] [CrossRef]
- Schmalreck, A.; Teuber, M.; Reininger, W.; Hartl, A. Structural features determining the antibiotic potencies of natural and synthetic hop bitter resins, their precursors and derivatives. Can. J. Microbiol. 2011, 21, 205–2012. [Google Scholar] [CrossRef] [PubMed]
- Rozalski, M.; Micota, B.; Sadowska, B.; Stochmal, A.; Jedrejek, D.; Wieckowska-Szakiel, M.; Rozalska, B. Antiadherent and antibiofilm activity of Humulus lupulus L. derived products: New pharmacological properties. BioMed Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Mizobuchi, S.; Sato, Y. A New Flavanone with Antifungal Activity Isolated from Hops. Agric. Biol. Chem. 1984, 48, 2771–2775. [Google Scholar] [CrossRef]
- Bartmańska, A.; Wałecka-Zacharska, E.; Tronina, T.; Popłoński, J.; Sordon, S.; Brzezowska, E.; Bania, J.; Huszcza, E. Antimicrobial properties of spent hops extracts, flavonoids isolated therefrom, and their derivatives. Molecules 2018, 23, 2059. [Google Scholar] [CrossRef]
- Frolich, S.; Schubert, C.; Bienzle, U.; Jenett-Siems, K. In vitro antiplasmodial activity of prenylated chalcone derivatives of hops (Humulus lupulus) and their interaction with haemin. J. Antimicrob. Chemother. 2005, 55, 883–887. [Google Scholar] [CrossRef]
- Aichinger, G.; Beisl, J.; Marko, D. The Hop Polyphenols Xanthohumol and 8-Prenyl-Naringenin Antagonize the Estrogenic Effects of Fusarium Mycotoxins in Human Endometrial Cancer Cells. Front. Nutr. 2018, 5, 1–11. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1, 2, 4 are available from the authors. |


| Contents (μg/L) and Type of Beer | XN (1) | IX (2) | 8-PN (4) | Minor Prenylflavonoids (Including 6-PN (5) and 6-/8-GN (7)) | 6-PN (5) | References | |
|---|---|---|---|---|---|---|---|
| European Beers | Stout | 340 | 2100 | 69 | 2680 | [11,28] | |
| Lager | 2 | 40 | 1 | 43 | |||
| Pilsner | 28 | 570 | 21 | 680 | |||
| 12 | 1060 | 8 | 1100 | ||||
| US Beers | Lager/pilsner | 34 | 500 | 13 | 590 | ||
| 9 | 680 | 14 | 750 | ||||
| 14 | 400 | 17 | 460 | ||||
| Porter | 690 | 1330 | 240 | 2900 | |||
| Wheat | 5 | 300 | 8 | 330 | |||
| Strong ale | 240 | 3440 | 110 | 4000 | |||
| India pale ale | 160 | 800 | 39 | 1160 | |||
| Other | Alcohol-free | 3 | 110 | 3 | 120 | ||
| 3 | 100 | 3 | 7 | ||||
| Ale | 100 | 2100 | 44 | 11 (6-GN) | 200 | ||
| Dark | 300 | 1200 | 92 | 27 (6-GN) | 200 | ||
| Regular | 14 | 400 | 10 | 46 (GN) | 26 |
| Biological Activity | Cell Line/Substrate | Main Active Components | Reference |
|---|---|---|---|
| Antiproliferative Activity | MCF-7, HT-29, A2780 | IX | [13] |
| PC-3, DU 145 | DMX, IX, 6-PN, 8-PN | [14] | |
| T-47D A2780cis | 6-PN, IX | [111] | |
| SK-MEL-28, BLM | 6-PN, 8-PN | [116] | |
| PC-3, UO.31 | 6-PN, 8-PN | [117] | |
| Induced Caspase-Independent Form of Cell Death | PC-3 | IX, 6-PN | [118] |
| DU 145 | IX, 6-PN, 8-PN | ||
| Induction of Quinone Reductase (QR) Activity | Mouse hepatoma Hepa 1c1c7 cells | 6-PN, 8-PN | [119] |
| IX | [120] | ||
| Antioxidant and Antiperoxidant | Isolated human LDL | DMX | [102] |
| Inhibition of Metabolic Activation of Procarcinogens | Rat liver microsomes CYP1A1, CYP1B1, CYP1A2, CYP3A4,CYP2E1 | DMX | [121] |
| P450 1A1 and 1B1 | 6-PN | [114] | |
| CYP1A2 | IX, 8-PN | [122] | |
| CYP1A2 | IX, 8-PN | [119] | |
| CYP1A | IX, 6PN, 8-PN | [120] | |
| MCF-7, CYP1A1 | 6-PN | [114] | |
| Inhibition of Nitric Oxide Synthase (iNos) | Mouse macrophage cells | IX | [120] |
| Inhibition of Angiogenesis | Human placental vessels | IX | [123] |
| Induction of Apoptosis in Tumor Cells | MCF-7 | 8-PN | [112] |
| HCT-116 | [124] | ||
| Inhibition of Cyclooxygenase Enzymes: COX1 | Sheep seminal vesicle microsomes | 8-PN | [120] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tronina, T.; Popłoński, J.; Bartmańska, A. Flavonoids as Phytoestrogenic Components of Hops and Beer. Molecules 2020, 25, 4201. https://doi.org/10.3390/molecules25184201
Tronina T, Popłoński J, Bartmańska A. Flavonoids as Phytoestrogenic Components of Hops and Beer. Molecules. 2020; 25(18):4201. https://doi.org/10.3390/molecules25184201
Chicago/Turabian StyleTronina, Tomasz, Jarosław Popłoński, and Agnieszka Bartmańska. 2020. "Flavonoids as Phytoestrogenic Components of Hops and Beer" Molecules 25, no. 18: 4201. https://doi.org/10.3390/molecules25184201
APA StyleTronina, T., Popłoński, J., & Bartmańska, A. (2020). Flavonoids as Phytoestrogenic Components of Hops and Beer. Molecules, 25(18), 4201. https://doi.org/10.3390/molecules25184201






