Calix[3]arene-Analogous Metacyclophanes: Synthesis, Structures and Properties with Infinite Potential
Abstract
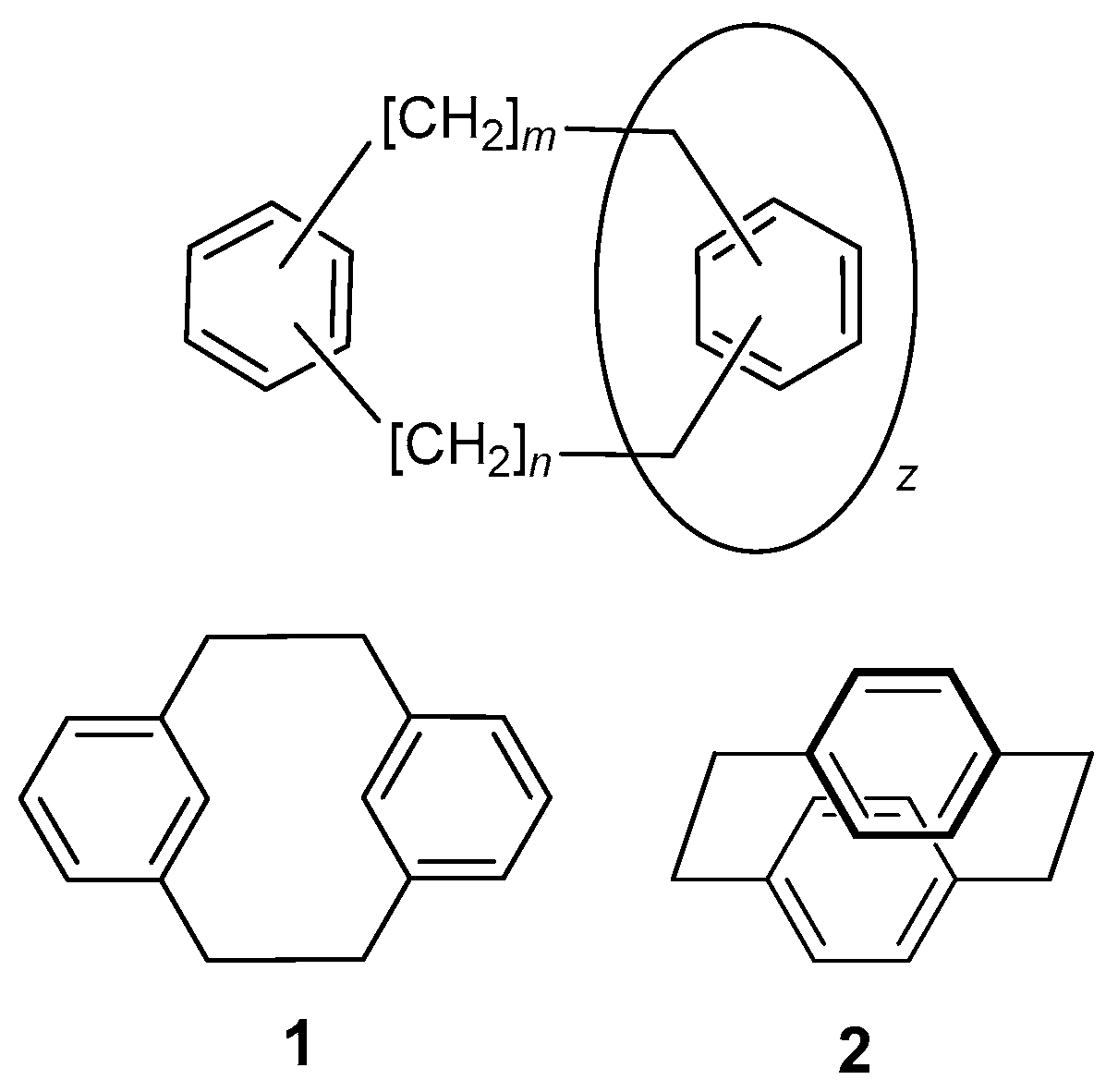
1. Introduction
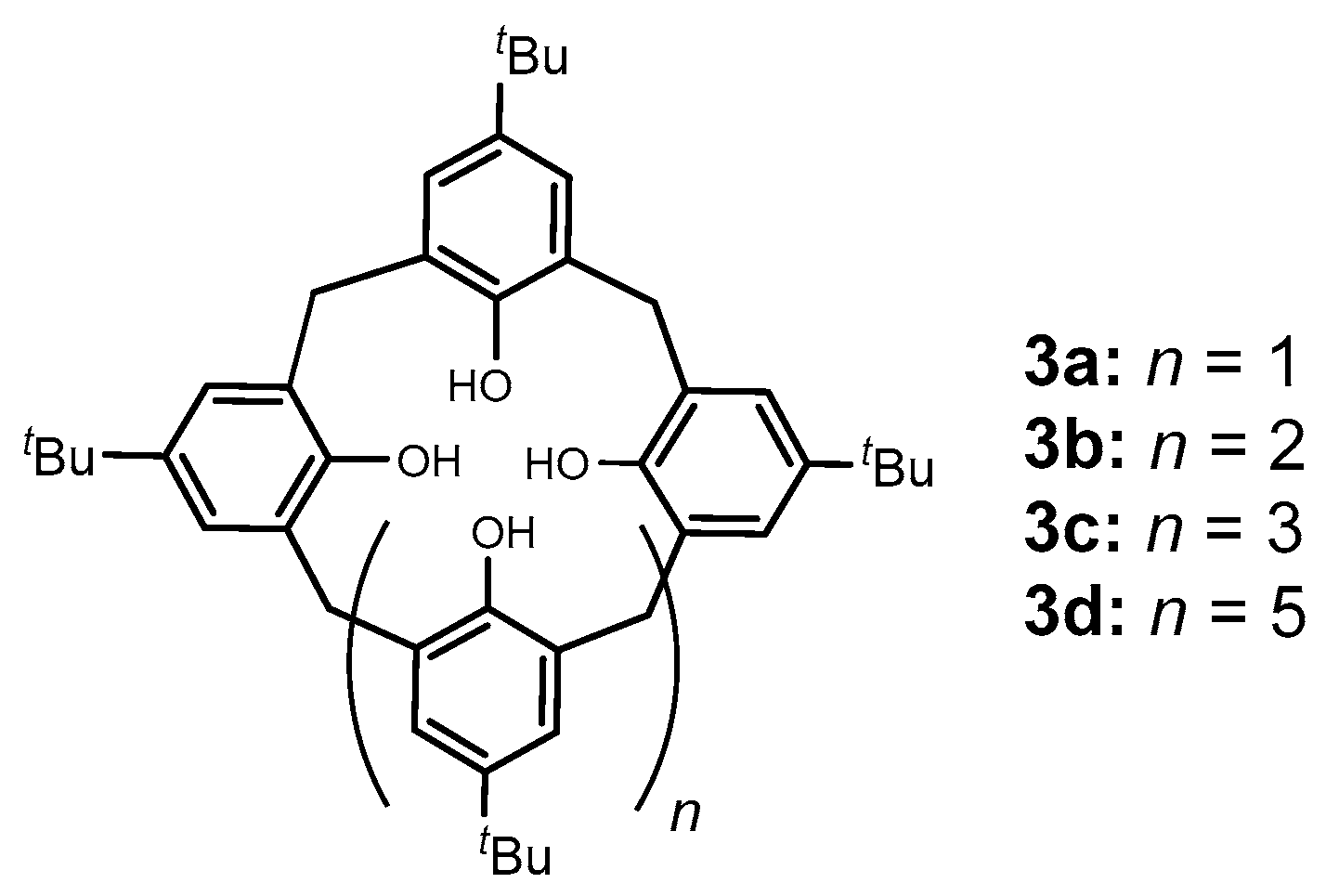
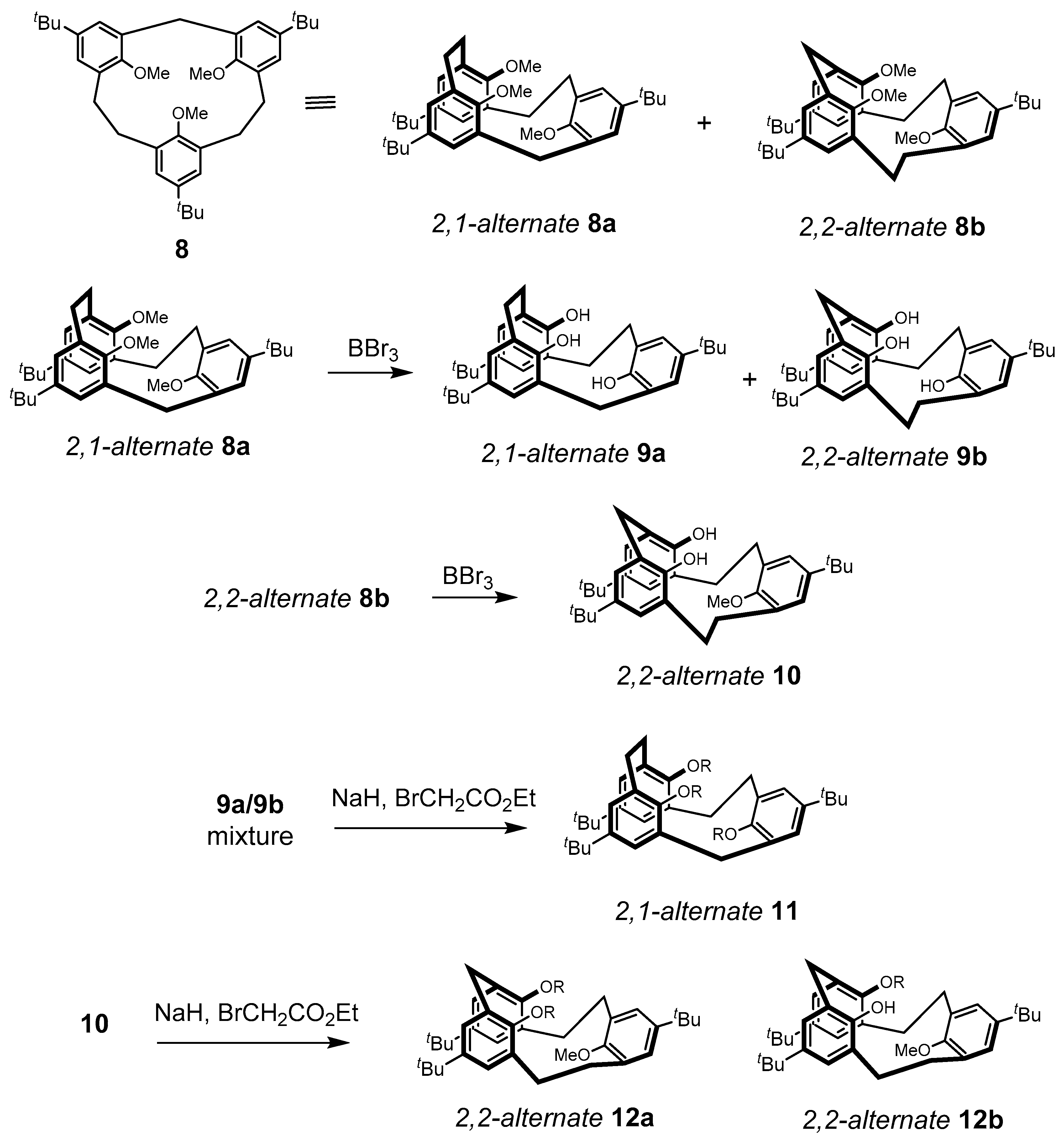
2. Calixarene-Analogous MCPs Containing Three Aromatic Rings: Calix[3]arene Analogues
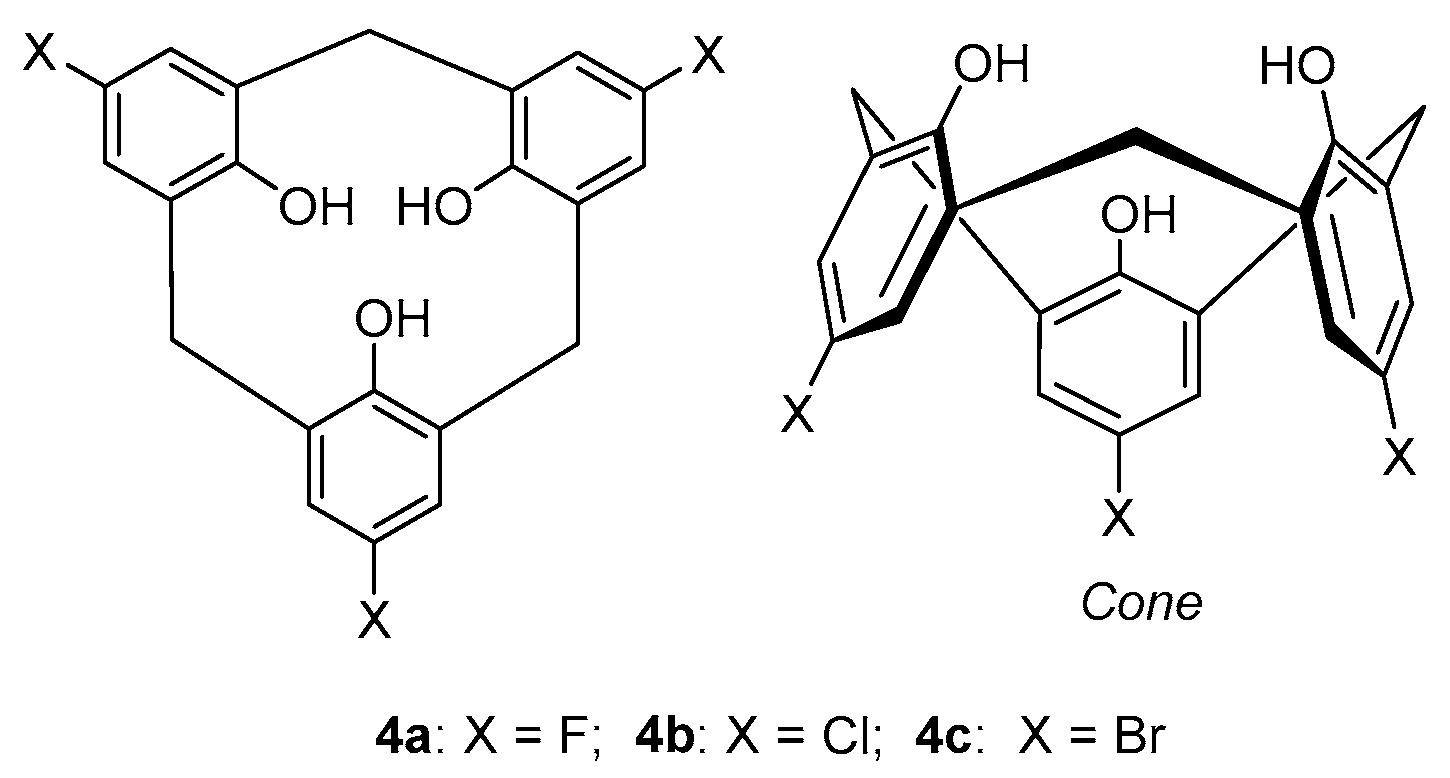
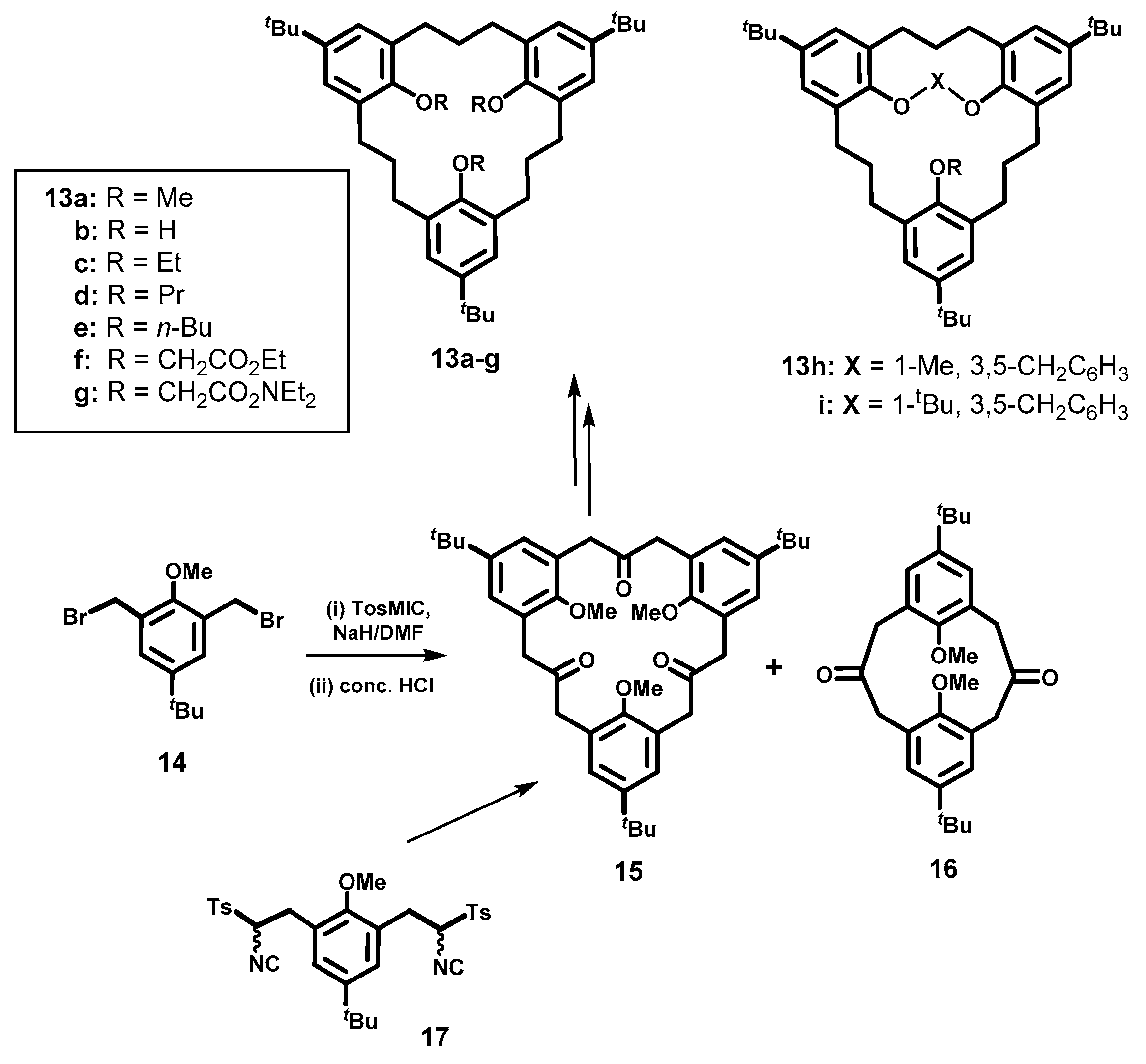
3. Homocalixarene-Analogous MCPs Containing Three Aromatic Rings: Homocalix[3]arene Analogues
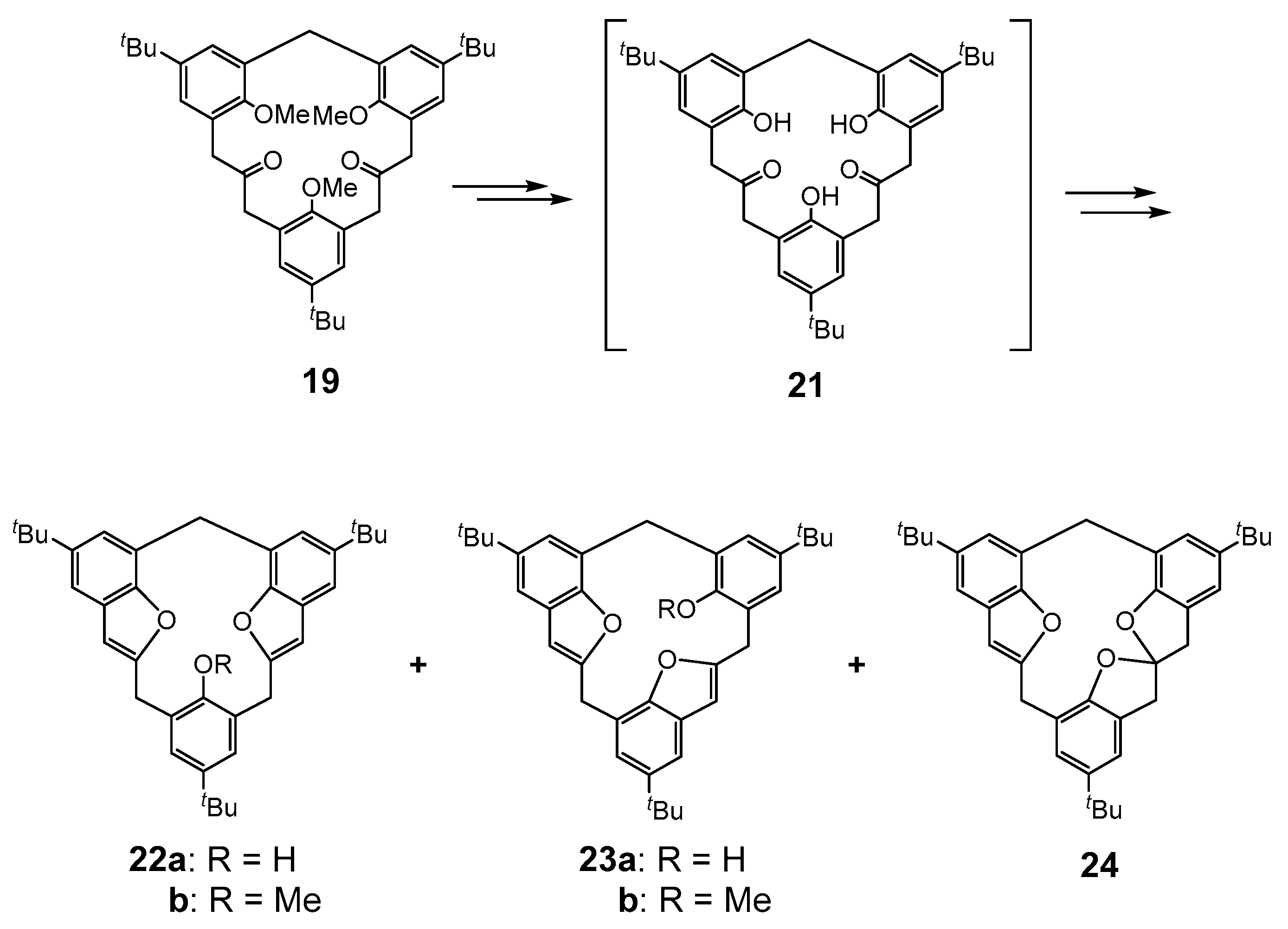
4. Calixbenzofuran-Analogous MCPs: Calix[3]benzofuran Analogues
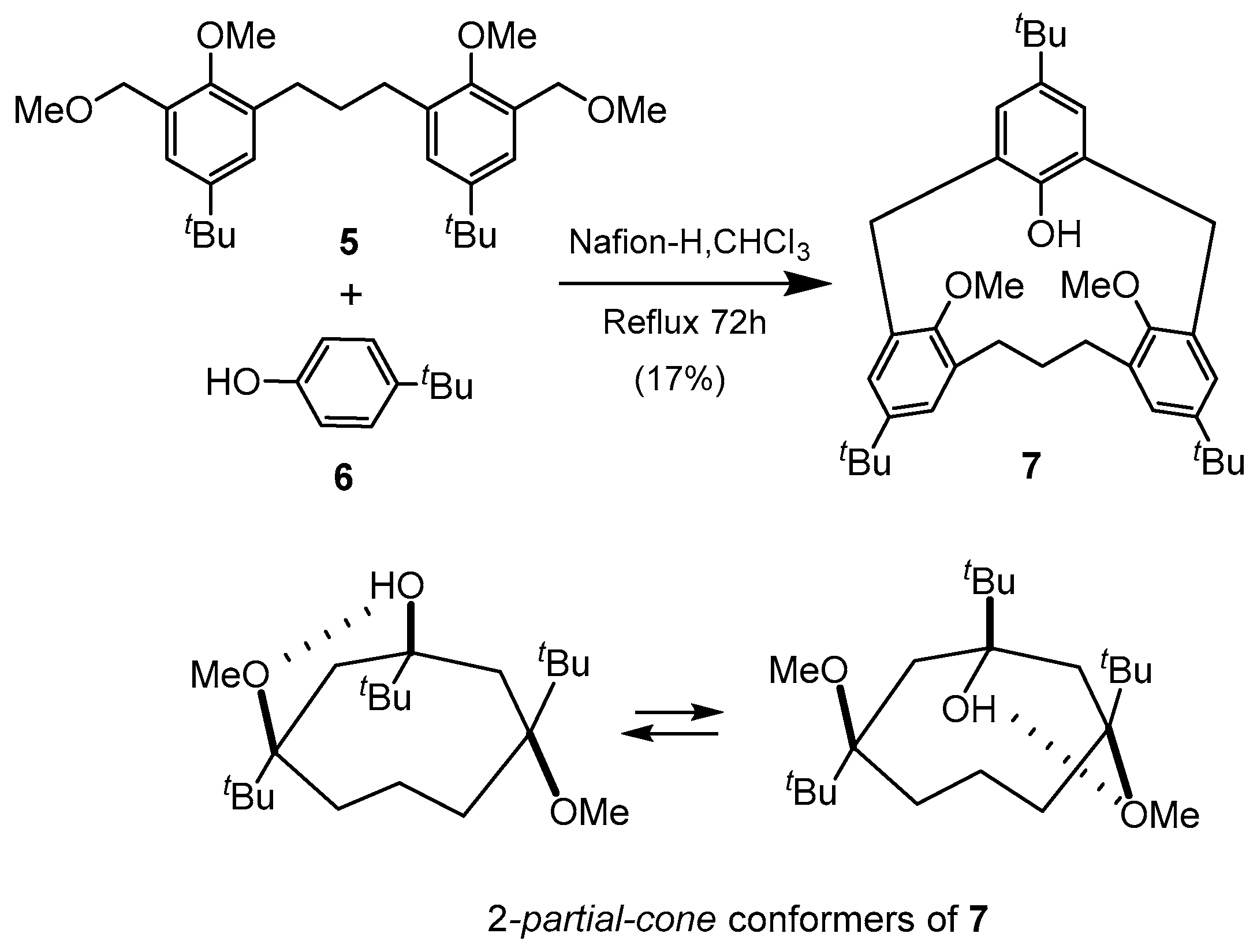
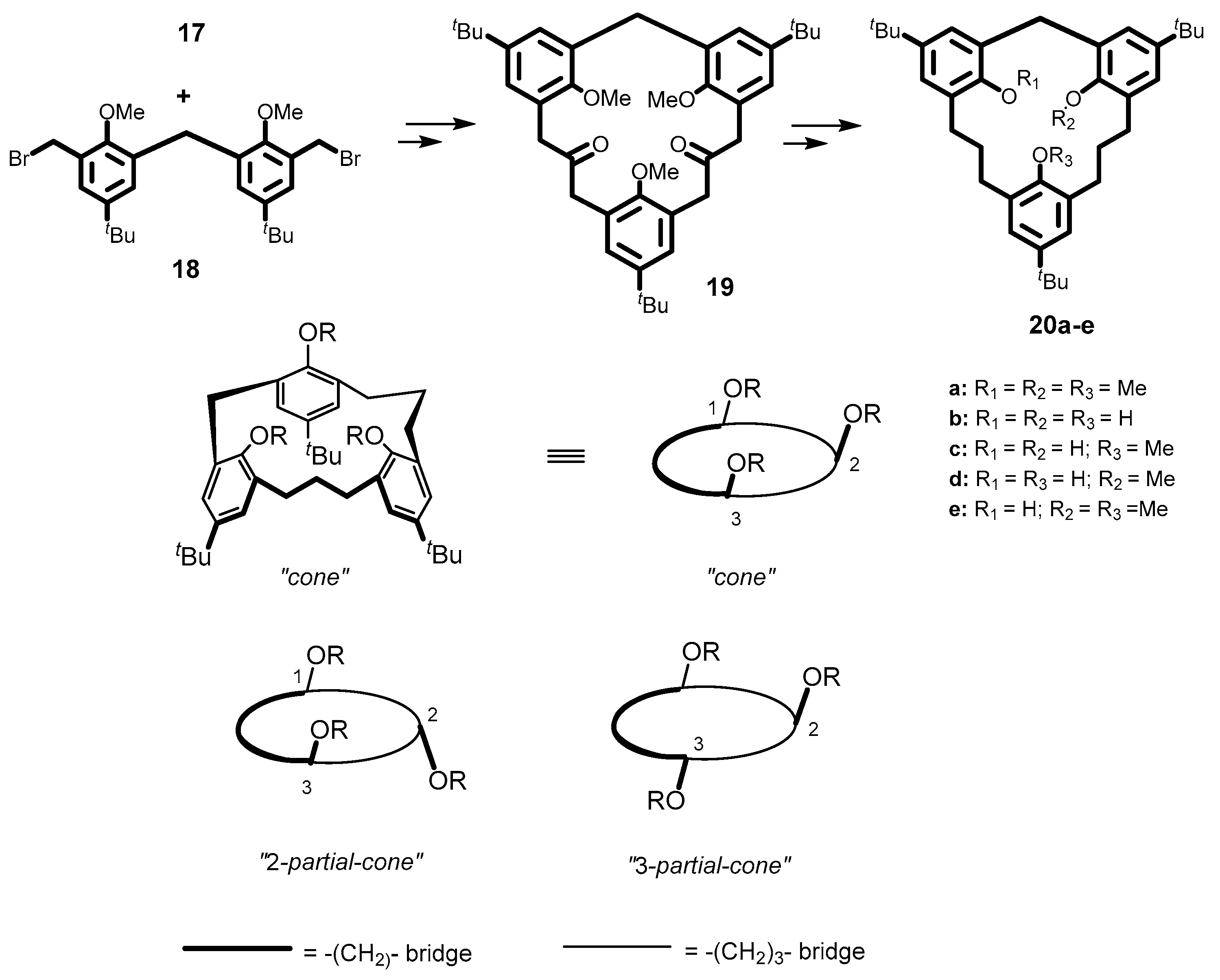
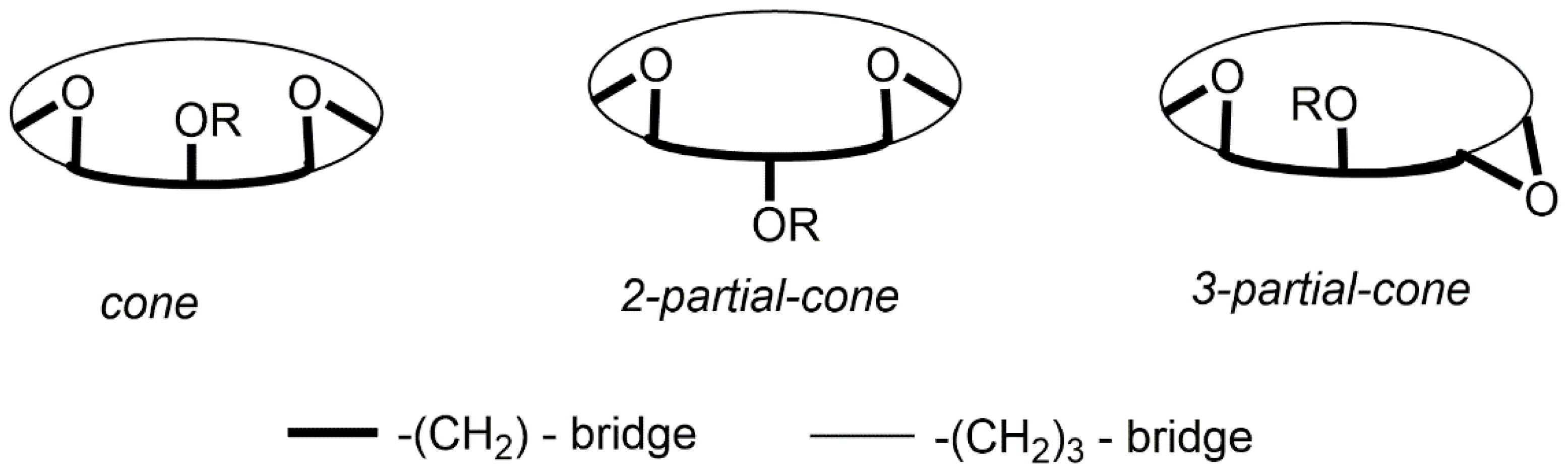
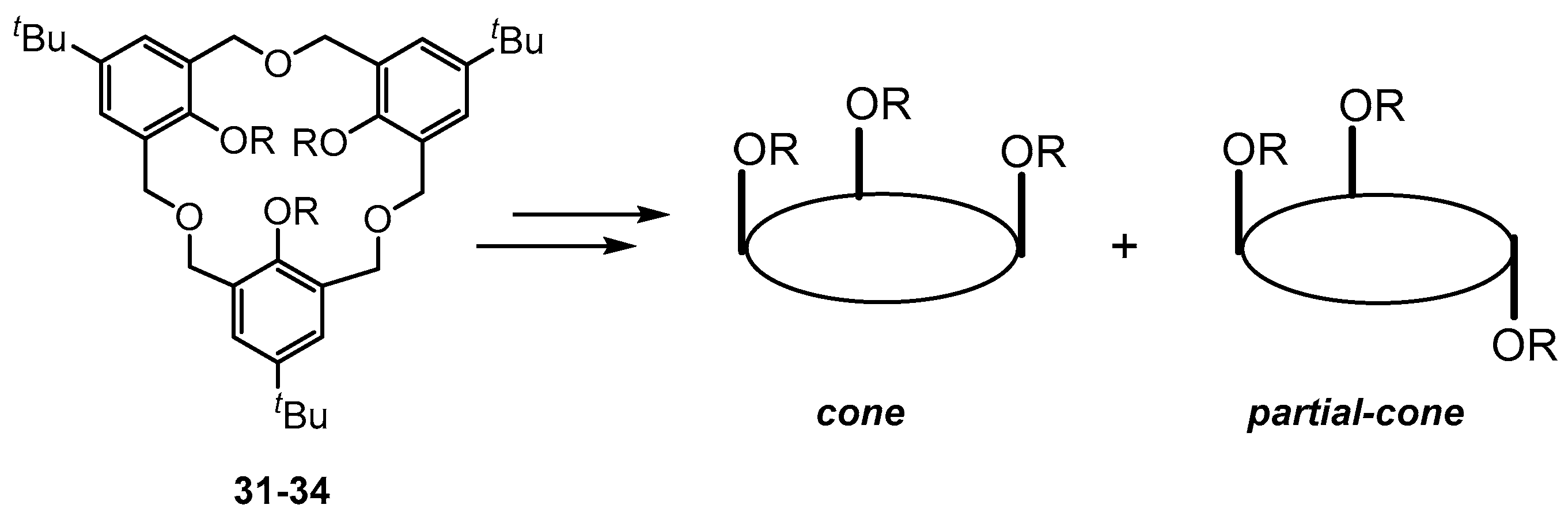
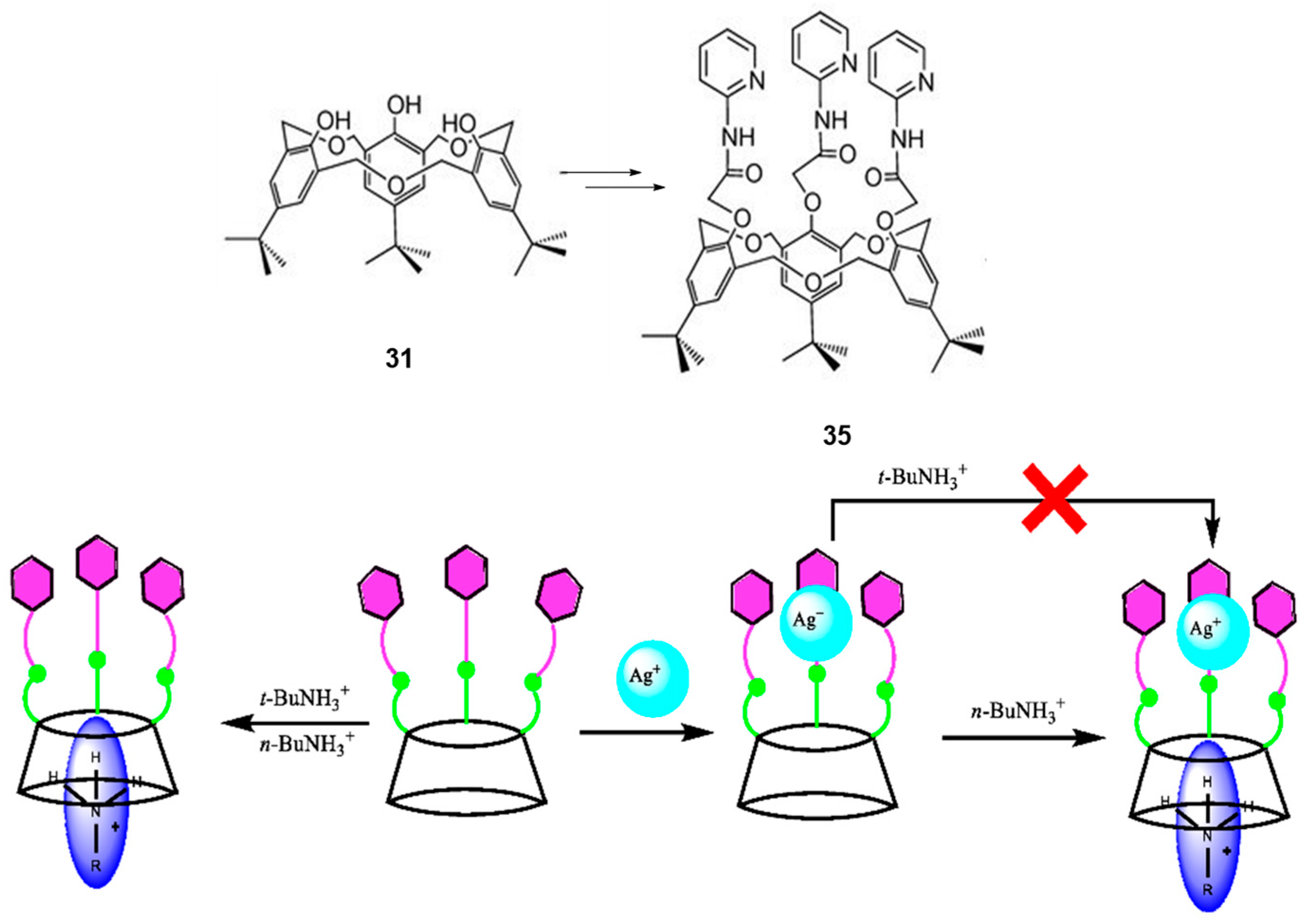
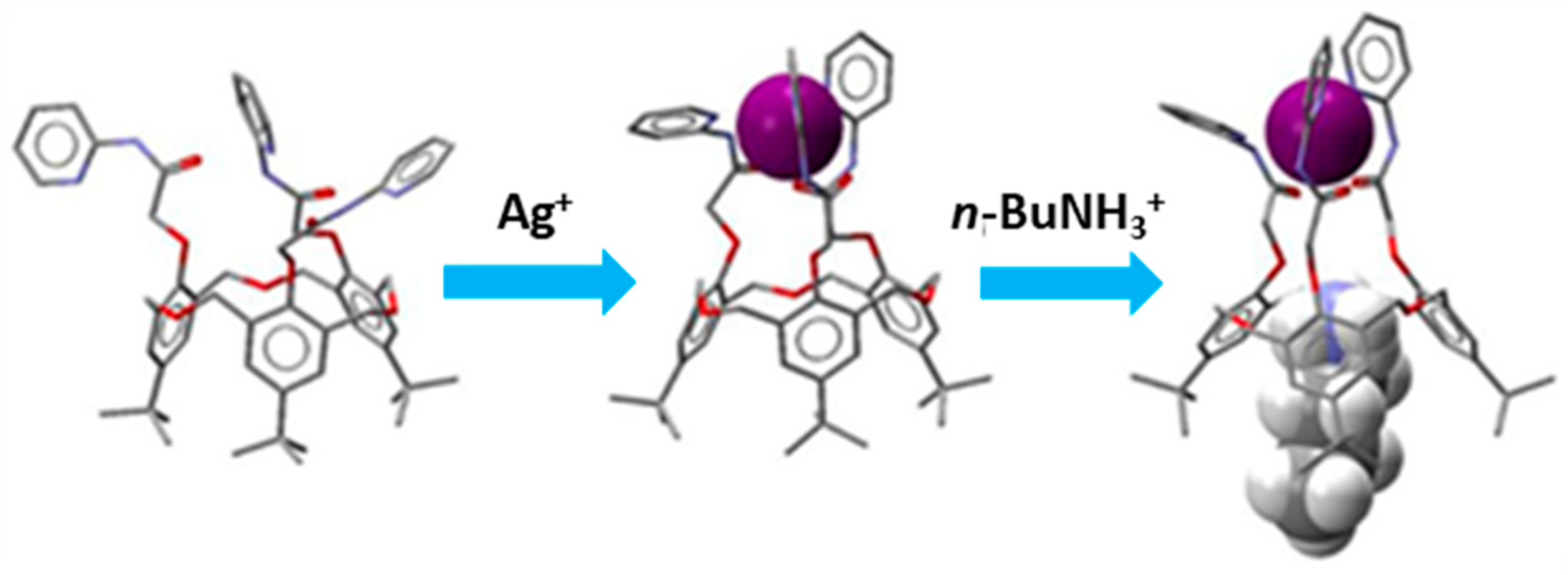
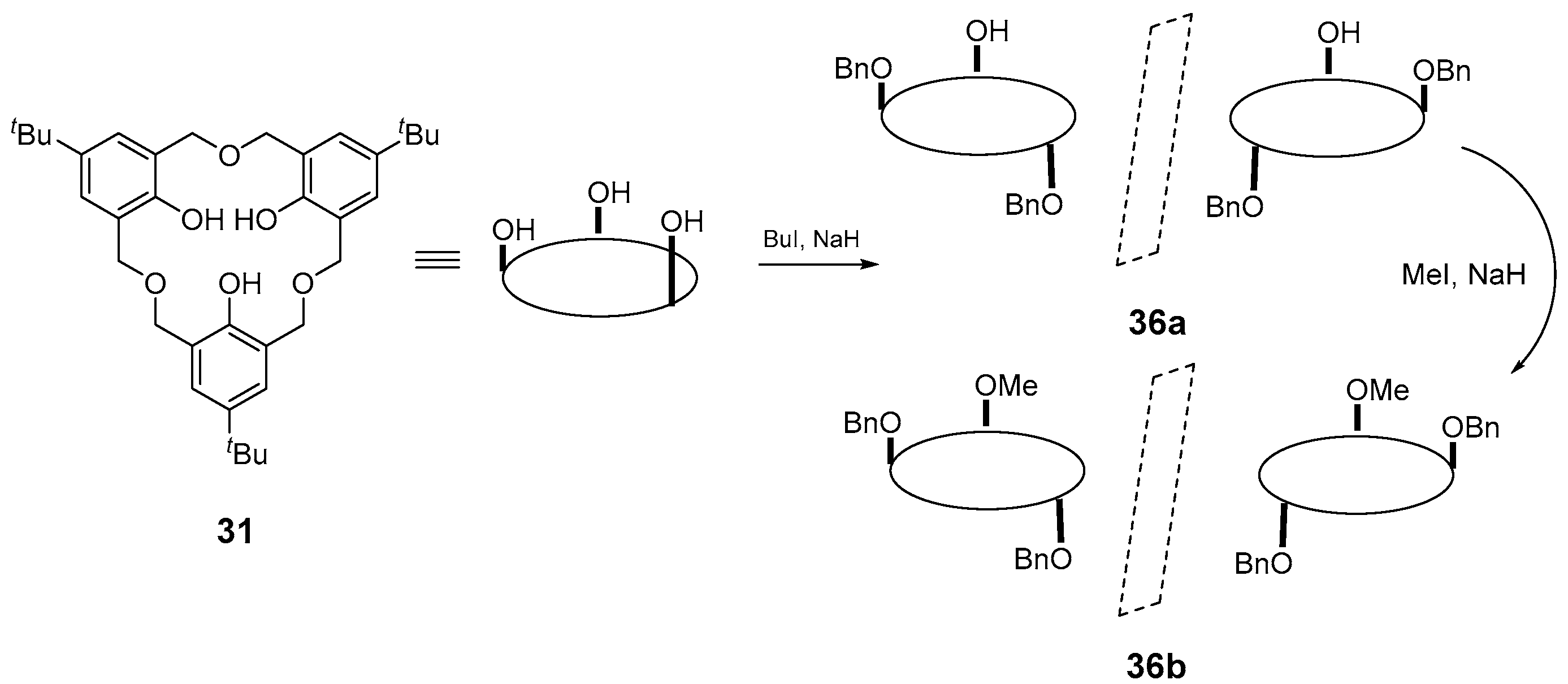
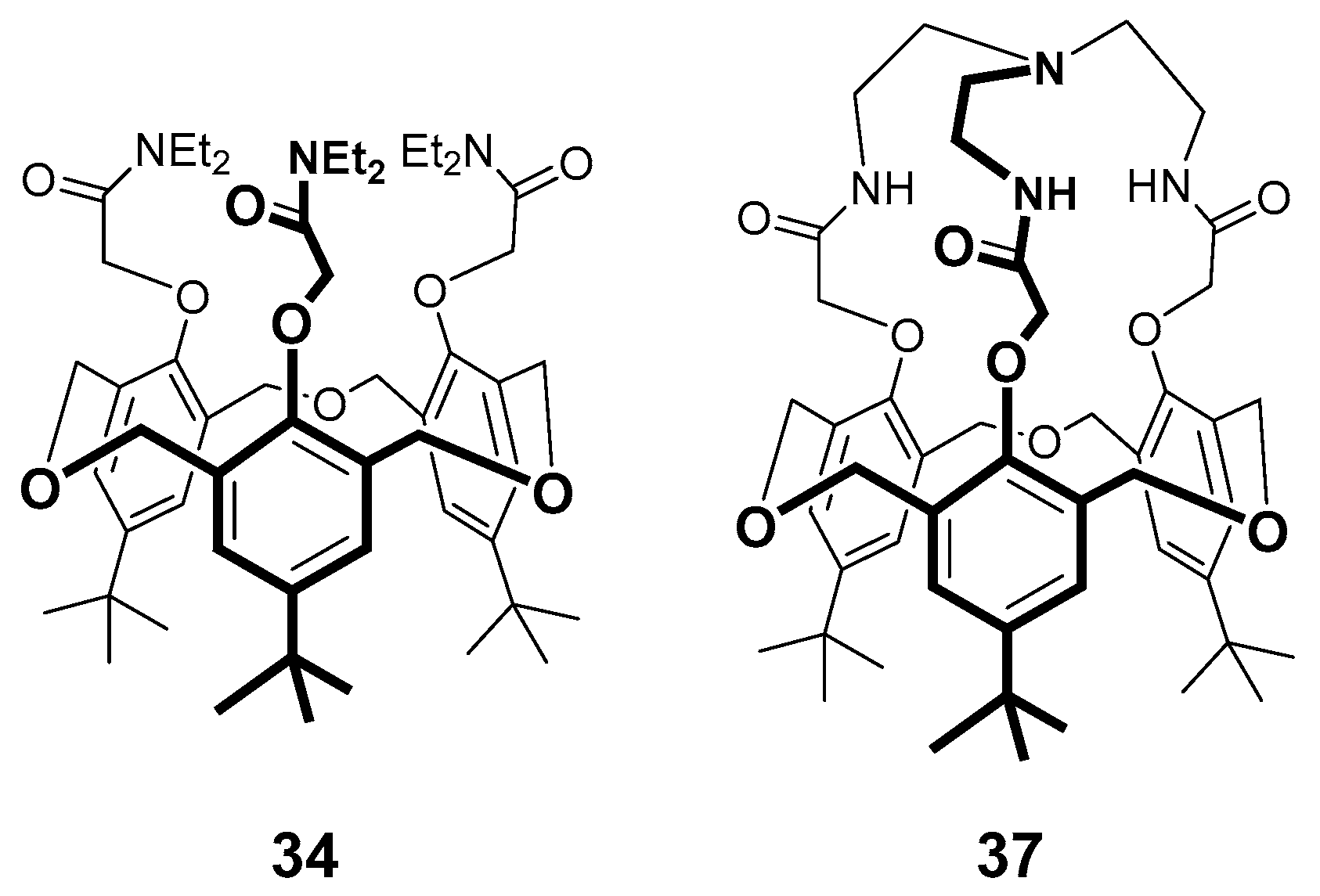
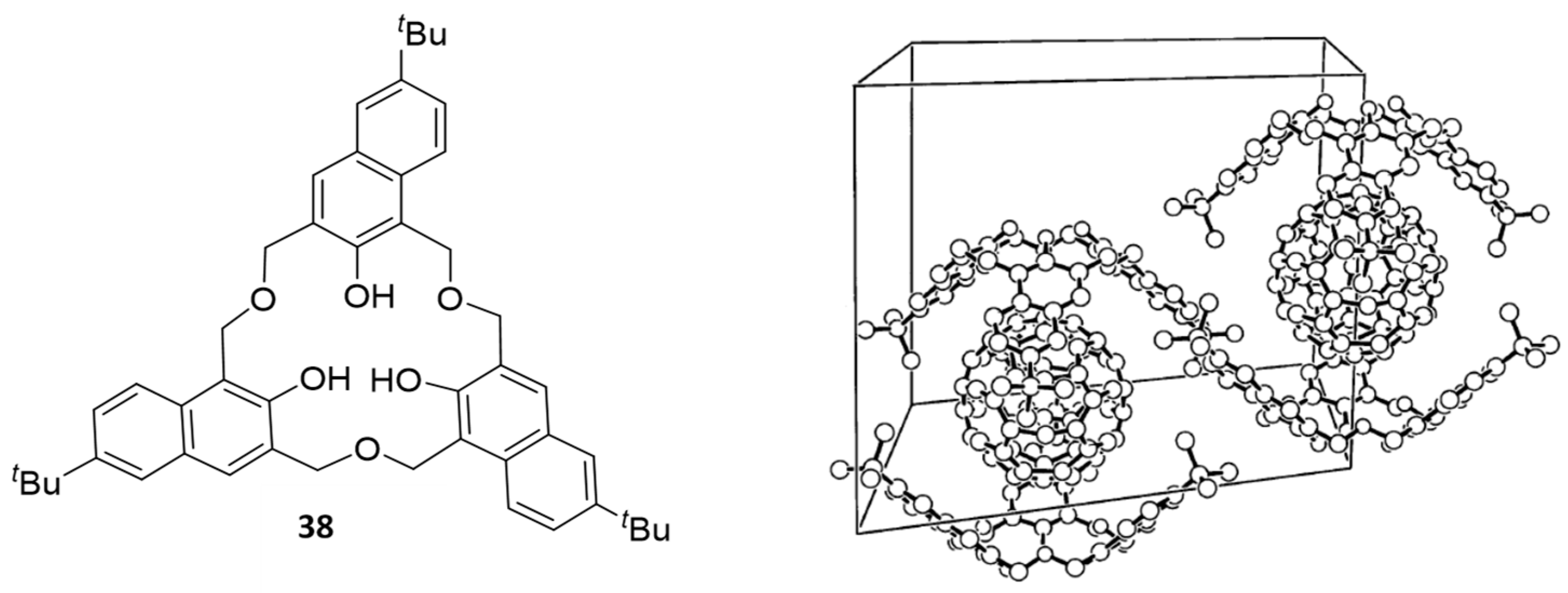
5. Homooxacalixarene-Analogue MCPs Containing Three Aromatic Rings: “Oxacalix[3]arene” Analogues
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Gleiter, R.; Hopf, H. Modern Cyclophane Chemistry; Wiley-VCH: Weinheim, Germany, 2004; ISBN 978-3-527-30713-5. [Google Scholar]
- Ghasemabadi, P.G.; Yao, T.; Bodwell, G.J. Cyclophanes containing large polycyclic aromatic hydrocarbons. Chem. Soc. Rev. 2015, 44, 6494–6518. [Google Scholar] [CrossRef]
- Pellegrin, M.M. Contribution à l’étude de la réaction de Fittig. Recl. Trav. Chim. Pays-Bas 1899, 18, 457–465. [Google Scholar] [CrossRef]
- Cram, D.J.; Steinberg, H. Macro rings. I. Preparation and spectra of the paracyclophanes. J. Am. Chem. Soc. 1951, 73, 5691–5704. [Google Scholar] [CrossRef]
- Zincke, A.; Ziegler, E. Zur kenntnis des härtungsprozesses von phenol-formaldehyd-harzen, X. mitteilung. Ber. Dtsch. Chem. Ges. 1944, 77, 264–272. [Google Scholar] [CrossRef]
- Gutsche, C.D.; Iqbal, M. p-tert-Butylcalix[4]arene. Org. Synth. 1990, 68, 234. [Google Scholar] [CrossRef]
- Stewart, D.R.; Gutsche, C.D. The one-step synthesis of p-tert-butylcalix[5]arene. Org. Prep. Proc. Intl. 1993, 25, 137–139. [Google Scholar] [CrossRef]
- Gutsche, C.D.; Dhawan, B.; Leonis, M.; Stewart, D.R. p-tert-Butylcalix[6]arene. Org. Synth. 1990, 68, 238. [Google Scholar] [CrossRef]
- Munch, J.H.; Gutsche, C.D. p-tert-Butylcalix[8]arene. Org. Synth. 1990, 68, 243. [Google Scholar] [CrossRef]
- Haase, C.H.W. Path to industrial production of calix[8 and 4]arenes. J. Org. Chem. 2020, 85, 603–611. [Google Scholar] [CrossRef]
- Neri, P.; Sessler, J.; Wang, M.-X. Calixarenes and Beyond; Springer International Publishing: Cham, Switzerland, 2016; ISBN 978-3-319-31867-7. [Google Scholar]
- Agrawal, Y.K.; Kunji, S.; Menon, S.K. Analytical applications of calixarenes. Rev. Anal. Chem. 1998, 17, 69–139. [Google Scholar] [CrossRef]
- Van Loon, J.D.; Verboom, W.; Reinhoudt, D.N. Selective functionalization and conformational properties of calix[4]arenes, a review. Org. Prep. Proced. Int. 1992, 24, 437–462. [Google Scholar] [CrossRef]
- Gaeta, C.; Talotta, C.; Neri, P. The calixarene p-bromodienone route: From a chemical curiosity to an useful synthetic tool. J. Incl. Phenom. Macrocycl. Chem. 2014, 79, 23–46. [Google Scholar] [CrossRef]
- Tlusty, M.; Eigner, V.; Babor, M.; Kohouta, M.; Lhotak, P. Synthesis of upper rim-double-bridged calix[4]arenes bearing seven membered rings and related compounds. RSC Adv. 2019, 9, 22017–22030. [Google Scholar] [CrossRef]
- Nakamura, Y.; Fujii, T.; Inokuma, S.; Nishimura, J. Homocalixarenes. In Calixarenes 2001; Asfari, Z., Böhmer, V., Harrowfield, J., Vicens, J., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; pp. 219–234. [Google Scholar] [CrossRef]
- Cottet, K.; Marcos, P.M.; Cragg, P.J. Fifty years of oxacalix[3]arenes: A review. Beilstein J. Org. Chem. 2012, 8, 201–226. [Google Scholar] [CrossRef] [PubMed]
- Masci, B. Homooxa- and homoaza-calixarenes. In Calixarenes 2001; Asfari, Z., Böhmer, V., Harrowfield, J., Vicens, J., Saadioui, M., Eds.; Springer: Dordrecht, The Netherlands, 2001; pp. 235–249. [Google Scholar] [CrossRef]
- Wang, D.-X.; Wang, M.-X. Azacalixaromatics. In Calixarenes and Beyond; Neri, P., Sessler, J., Wang, M.-X., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 363–397. [Google Scholar] [CrossRef]
- Kaewtong, C.; Pulpoka, P. Azacalix[3]arenes: Chemistry and recent developments in functionalization for specific anion and cation recognition. J. Incl. Phenom. Macrocycl. Chem. 2009, 65, 129–136. [Google Scholar] [CrossRef]
- Chen, C.-F.; Han, Y. Triptycene-derived macrocyclic arenes: From calixarenes to helicarenes. Acc. Chem. Res. 2018, 51, 2093–2106. [Google Scholar] [CrossRef]
- Kohnke, F.H. Calixpyrroles: From anion ligands to potential anticancer drugs. Eur. J. Org. Chem. 2020, 2020, 4261–4272. [Google Scholar] [CrossRef]
- Moshfegh, A.A.; Beladi, E.; Radnia, L.; Hosseini, A.S.; Tofigh, S.; Hakimelahi, G.H. The synthesis of 5,11,17-trihalotetracyclo [13.3.1.13,7.19,13]henicosa-1 (19),3,5,7,9,11,13, 15,17-nonaene-19,20,21-triols and 5,11,17-trihalo-19,20,21-trihydroxytetracyclo [13.3.1.13,7. 19,13]henicosa-1(19),3,5,7(20),9,11,13(21), 15,17-nonaene-8,14-dione. Cyclo-derivatives of phloroglucide analogues. Helv. Chim. Acta 1982, 65, 1264–1270. [Google Scholar] [CrossRef]
- Yamato, T.L.; Doamekpor, K.; Tsuzuki, H.; Tashiro, M. A novel calixarene-analogous macrocyclic metacyclophane ‘molecular pendulum’. Chem. Lett. 1995, 24, 89–90. [Google Scholar] [CrossRef]
- Yamato, T.L.; Doamekpor, K.; Tsuzuki, H. Synthesis and conformational studies of novel calixarene-analogous macrocyclic [3.1.1]metacyclophanes. Liebigs Ann. Recl. 1997, 1997, 1537–1544. [Google Scholar] [CrossRef]
- Tsuge, A.; Sawada, T.; Mataka, S.; Nishiyama, N.; Sakashita, H.; Tashiro, M. Preparation and structural properties of hydroxy- and alkoxy-[2.2.1]metacyclophanes. J. Chem. Soc. Perkin Trans. 1 1992, 1489–1494. [Google Scholar] [CrossRef]
- Laufenberg, S.; Feuerbacher, N.; Pischel, I.; Börsch, O.; Nieger, M.; Vögtle, F. “New biphenylenophanes and biphenylophanes–1,8-dimethylbiphenylene by continuous vacuum pyrolysis. Liebigs Ann. Chem. 1997, 1901–1906. [Google Scholar] [CrossRef]
- Vögtle, F. Cyclophane Chemistry: Synthetics, Structures and Reactions; John Wiley & Sons: Chichester, UK, 1993; ISBN 10: 0471931993. [Google Scholar]
- Tashiro, M.; Watanabe, T.; Tsuge, A.; Sawada, T.; Mataka, S. Metacyclophanes and related compounds. 24. Preparation and reaction of trimethyl[2.2.2]- and tetramethyl[2.2.2.2]metacyclophane. J. Org. Chem. 1989, 54, 2632–2638. [Google Scholar] [CrossRef]
- Possel, O.; van Leusen, A.M. Tosylmethyl isocyanide employed in a novel synthesis of ketones. A new masked formaldehyde reagent. Tetrahedron Lett. 1977, 18, 4229–4231. [Google Scholar] [CrossRef]
- van Leusen, A.M.; Boerna, G.J.; Helholdt, R.B.; Siderius, H.; Strating, J. Chemistry of sulfonylmethylisocyanides. Simple synthetic approaches to a new versatile chemical building block. Tetrahedron Lett. 1972, 13, 2367–2368. [Google Scholar] [CrossRef]
- Breitenbach, J.; Vögtle, F. Macrocyclizations with TosMIC-Yielding [3n]metacyclophanes. Synthesis 1992, 41–43. [Google Scholar] [CrossRef]
- Yamato, T.; Doamekpor, L.K.; Koizumi, K.; Kishi, K.; Haraguchi, M.; Tashiro, M. Synthesis and conformational studies of calixarene-analogous trihydroxy[3.3.3]metacyclophanes and their O-alkylated derivatives. Liebigs Ann. 1995, 7, 1259–1267. [Google Scholar] [CrossRef]
- Yamato, T.; Zhang, F. The synthesis and probable conformation of m-xylenyl capped homocalix[3]arenes derived from a trihydroxy[3.3.3]metacyclophane. J. Chem. Res. 1999, 23, 34–35. [Google Scholar] [CrossRef]
- Yamato, T.; Kohno, K.; Tsuchihashi, K. Synthesis, structures and ion selectivity of homocalix[3]arene thioketals derived from homocalix[3]arene ketones. J. Incl. Phenom. Macrocycl. Chem. 2002, 43, 137–144. [Google Scholar] [CrossRef]
- Yamato, T.; Haraguchi, M.; Nishikawa, J.; Ide, S. Synthesis, conformational studies and inclusion properties of O-benzylated calixarene analogues of trihydroxy[3.3.3]metacyclophanes. J. Chem. Soc. Perkin Trans. 1 1998, 609–614. [Google Scholar] [CrossRef]
- Yamato, T. Synthesis, conformations and inclusion properties of homocalix[3]arenes. J. Incl. Phenom. Macrocycl. Chem. 1998, 32, 195–207. [Google Scholar] [CrossRef]
- Islam, M.M.; Tomiyasu, H.; Thuery, P.; Matsumoto, T.; Tanaka, J.; Elsegood, M.R.J.; Redshaw, C.; Yamato, T. Synthesis and conformational studies of calixarene analogue chiral [3.3.1]metacyclophane. J. Mol. Struct. 2015, 1098, 47–54. [Google Scholar] [CrossRef]
- Islam, M.M.; Tomiyasu, H.; Matsumoto, T.; Tanaka, J.; Rahman, S.; Georghiou, P.E.; Redshaw, C.; Yamato, T. Synthesis and conformational studies of chiral macrocyclic [1.1.1]metacyclophanes containing benzofuran rings. Org. Biomol. Chem. 2015, 13, 9055–9064. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.M.; Akther, T.; Ikejiri, Y.; Matsumoto, T.; Tanaka, J.; Rahman, S.; Georghiou, P.E.; Hughes, D.L.; Redshaw, C.; Yamato, T. Synthesis, structural properties, electrophilic substitution reactions and DFT computational studies of calix[3]benzofurans. RSC Adv. 2016, 6, 50808–50817. [Google Scholar] [CrossRef]
- Islam, M.M.; Wang, C.-Z.; Feng, X.; Rahman, S.; Georghiou, P.E.; Alodhayb, A.; Yamato, T. Synthesis, structures and DFT computational studies of [3.1.1]metacyclophanes containing benzofuran rings. ChemistrySelect 2018, 3, 13542–13547. [Google Scholar] [CrossRef]
- Sawada, T.; Hongo, T.; Matsuo, N.; Konishi, M.; Kawaguchi, T.; Ihara, H. Hemisphere-shaped calixarenes and their analogs: Synthesis, structure, and chiral recognition ability. Tetrahedron 2011, 67, 4716–4722. [Google Scholar] [CrossRef]
- Sawada, T.; Nishiyama, Y.; Tabuchi, W.; Ishikawa, M.; Tsutsumi, E.; Kuwahara, Y.; Shosenji, H. Novel calixarene hemisphere synthesized via pinacol rearrangement of [2.1.2.1]metacyclophane. Org. Lett. 2006, 8, 1995–1997. [Google Scholar] [CrossRef]
- Szumna, A. Inherently chiral concave molecules-from synthesis to applications. Chem. Soc. Rev. 2010, 39, 4274–4285. [Google Scholar] [CrossRef]
- Wierzbicki, M.; Jędrzejewska, H.; Szumna, A. Chiral calixarenes and resorcinarenes. In Calixarenes and Beyond; Neri, P., Sessler, J., Wang, M.-X., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 13–42. ISBN 978-3-319-31867-7. [Google Scholar] [CrossRef]
- Wolff, A.; Böhmer, V.; Fogt, W.; Ugozzoli, F.; Andreetti, G.D. Dissymmetric calix[4]arenes with C2 and C4 symmetry. J. Org. Chem. 1990, 55, 5665–5667. [Google Scholar] [CrossRef]
- Pickard, S.T.; Pirkle, W.H.; Tabatabai, M.; Vogt, W.; Böhmer, V. Dissymmetric calix[4]arenes: Optical resolution of some conformationally fixed derivatives. Chirality 1993, 5, 310–314. [Google Scholar] [CrossRef]
- Mandolini, L.; Ungaro, R. Calixarenes in Action; Imperial College Press: London, UK, 2000. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 09, Revision D.01; Gaussian, Inc.: Wallingford, CT, USA, 2013. [Google Scholar]
- Hultzsch, K. Ring condensates in alkyl phenol resins. Kunststoffe 1962, 52, 19–24. [Google Scholar]
- Dhawan, B.; Gutsche, C.D. Calixarenes. 10. Oxacalixarenes. J. Org. Chem. 1983, 48, 1536–1539. [Google Scholar] [CrossRef]
- Hampton, P.D.; Bencze, Z.; Tong, W.; Daitch, C.E. A new synthesis of oxacalix[3]arene macrocycles and alkali-metal-binding studies. J. Org. Chem. 1994, 59, 4838–4843. [Google Scholar] [CrossRef]
- Araki, K.; Inada, K.; Otsuka, H.; Shinkai, S. Conformational isomerism in and binding properties to alkali-metals and an ammonium salt of O-alkylated homooxacalix[3]arenes. Tetrahedron 1993, 49, 9465–9478. [Google Scholar] [CrossRef]
- Araki, K.; Hashimoto, N.; Otsuka, H.; Shinkai, S. Synthesis and ion selectivity of conformers derived from hexahomotrioxacalix[3]arene. J. Org. Chem. 1993, 58, 5958–5963. [Google Scholar] [CrossRef]
- Matsumoto, H.; Nishio, S.; Takeshita, M.; Shinkai, S. Syntheses and ion selectivities of tri-amide derivatives of hexahomotrioxacalix[3]arene. Remarkably large metal template effect on the ratio of cone vs. partial-cone conformers. Tetrahedron 1995, 51, 4647–4654. [Google Scholar] [CrossRef]
- Takeshita, M.; Inokuchi, F.; Shinkai, S. C3-Symmetrically-capped homotrioxacalix[3]arene. A preorganized host molecule for inclusion of primary ammonium ions. Tetrahedron Lett. 1995, 36, 3341–3344. [Google Scholar] [CrossRef]
- Jiang, X.-K.; Ikejiri, Y.; Wu, C.; Rahman, S.; Georghiou, P.E.; Zeng, X.; Elsegood, M.R.J.; Redshaw, C.; Teat, S.J.; Yamato, T. A hexahomotrioxacalix[3]arene-based ditopic receptor for alkylammonium ions controlled by Ag+ ions. Molecules 2018, 23, 467. [Google Scholar] [CrossRef]
- Araki, K.; Inada, K.; Shinkai, S. Chiral recognition of α-amino acid derivatives with a homooxacalix[3]arene: Construction of a pseudo-C2-symmetrical compound from a C3-symmetrical macrocycle. Angew. Chem. Int. Ed. Engl. 1996, 35, 72–74. [Google Scholar] [CrossRef]
- Teixeira, F.A.; Ascenso, J.R.; Cragg, P.J.; Hickey, N.; Geremia, S.; Marcos, P.M. Recognition of anions, monoamine neurotransmitter and trace amine hydrochlorides by ureido-hexahomotrioxacalix[3]arene ditopic receptors. Eur. J. Org. Chem. 2020, 1930–1940. [Google Scholar] [CrossRef]
- Lambert, S.; Bartik, K.; Jabin, I. Specific binding of primary ammonium ions and lysine-containing peptides in protic solvents by hexahomotrioxacalix[3]arenes. J. Org. Chem. 2020, 85, 10062–10071. [Google Scholar] [CrossRef]
- Jiang, X.-K.; Deng, M.; Mu, L.; Zeng, X.; Zhang, J.X.; Yamato, T. Synthesis and crystal structure of a novel hexahomotrioxacalix[3]cryptand. Asian J. Chem. 2013, 25, 515–517. [Google Scholar] [CrossRef]
- Zahim, S.; Ajami, D.; Laurent, P.; Valkenier, H.; Reinaud, O.; Luhmer, M.; Jabin, I. Synthesis and binding properties of a tren-capped hexahomotrioxacalix[3]arene. ChemPhysChem 2019, 21, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Ashram, M.; Mizyed, S.; Georghiou, P.E. Synthesis of hexahomotrioxacalix[3]naphthalenes and a study of their alkali-metal cation binding properties. J. Org. Chem. 2001, 66, 1473–1479. [Google Scholar] [CrossRef] [PubMed]
- Mizyed, S.; Ashram, M.; Miller, D.O.; Georghiou, P.E. Supramolecular complexation of [60]fullerene with hexahomotrioxacalix[3]naphthalenes: A new class of naphthalene-based calixarenes. J. Chem. Soc. Perkin Trans. 2 2001, 1916–1919. [Google Scholar] [CrossRef]
- Tsubaki, K.; Tanaka, K.; Kinoshita, T.; Fuji, K. Complexation of C60 with hexahomooxacalix[3]arenes and supramolecular structures of complexes in the solid state. Chem. Commun. 1998, 895–896. [Google Scholar] [CrossRef]


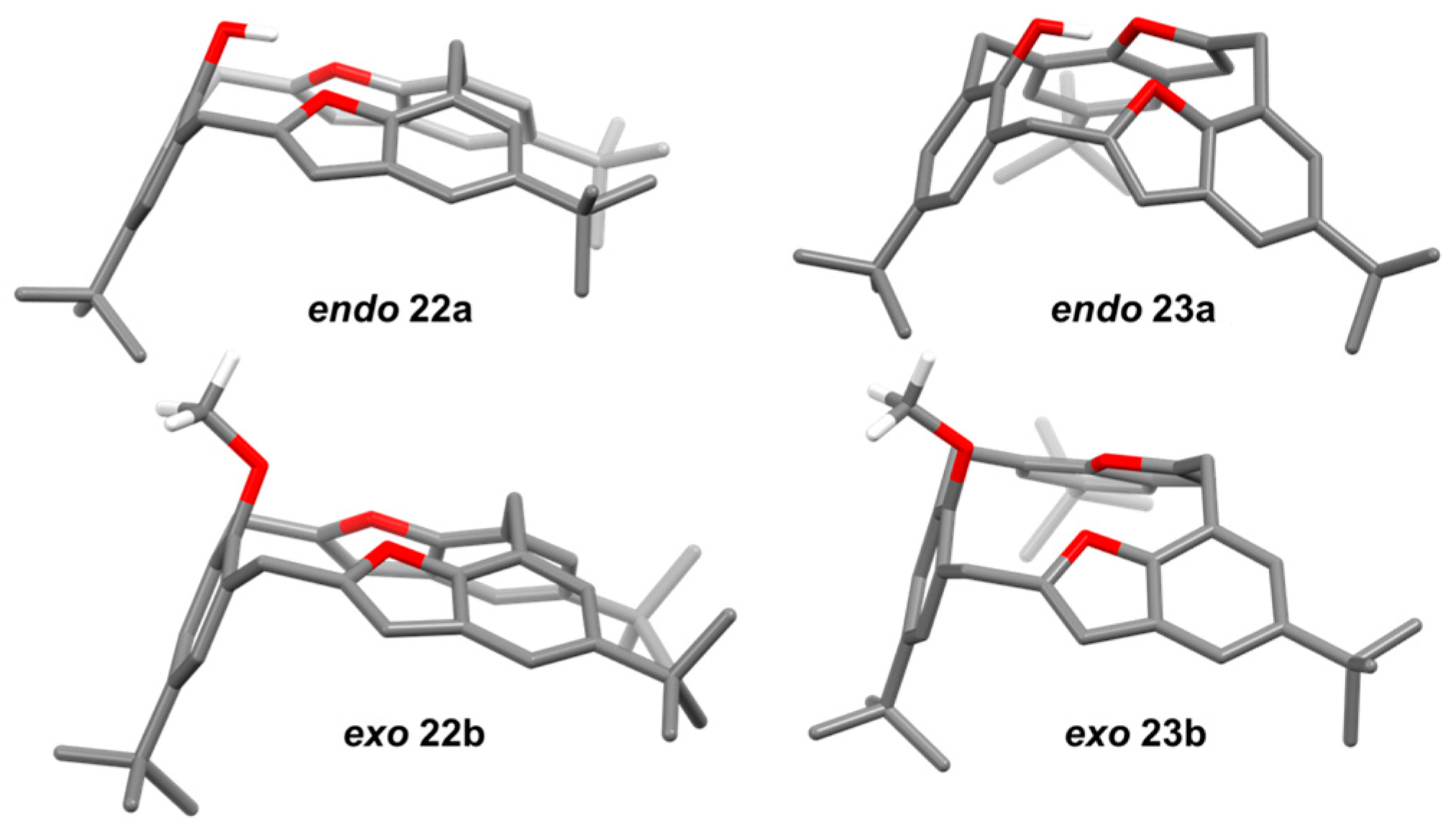
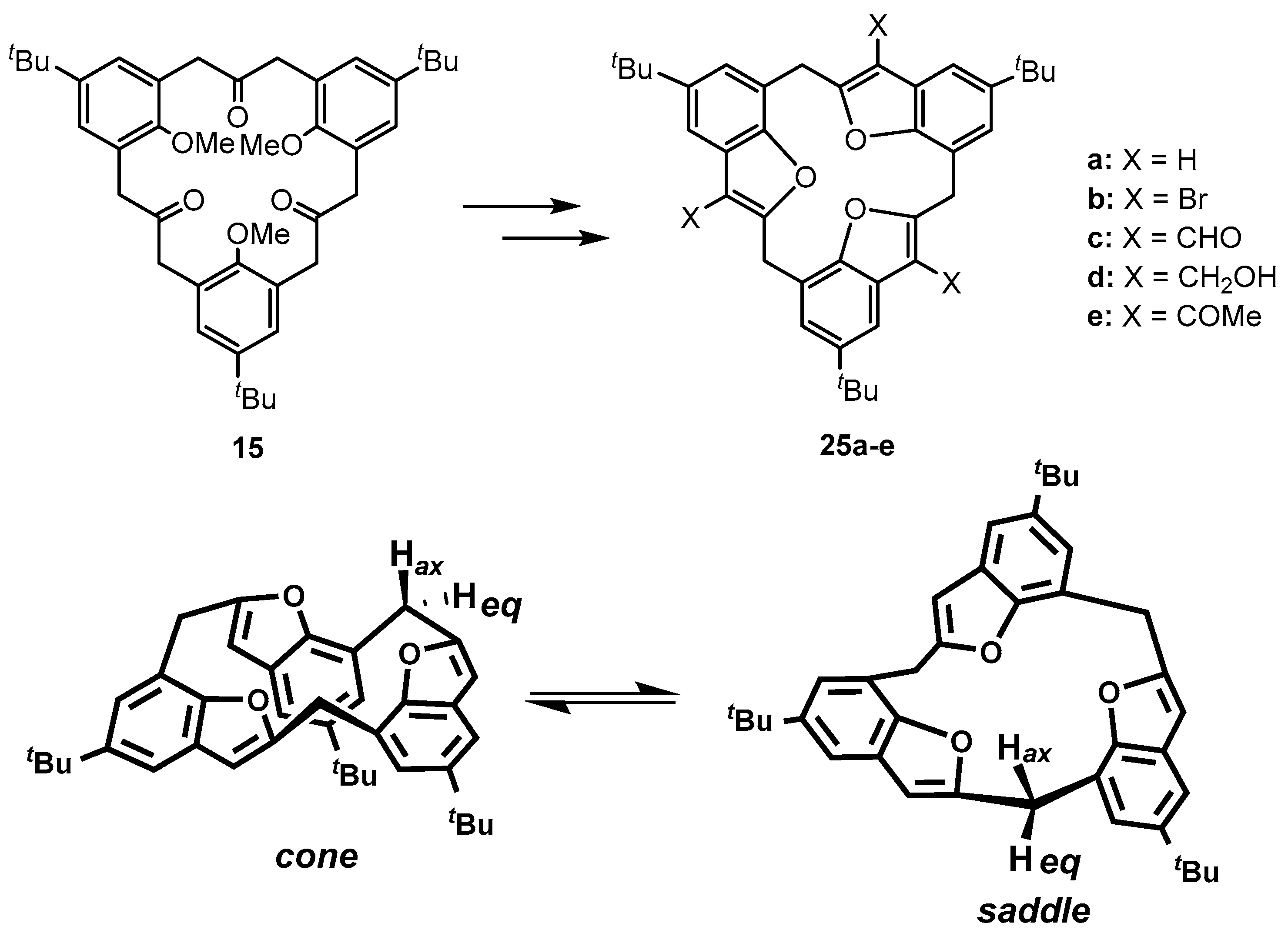
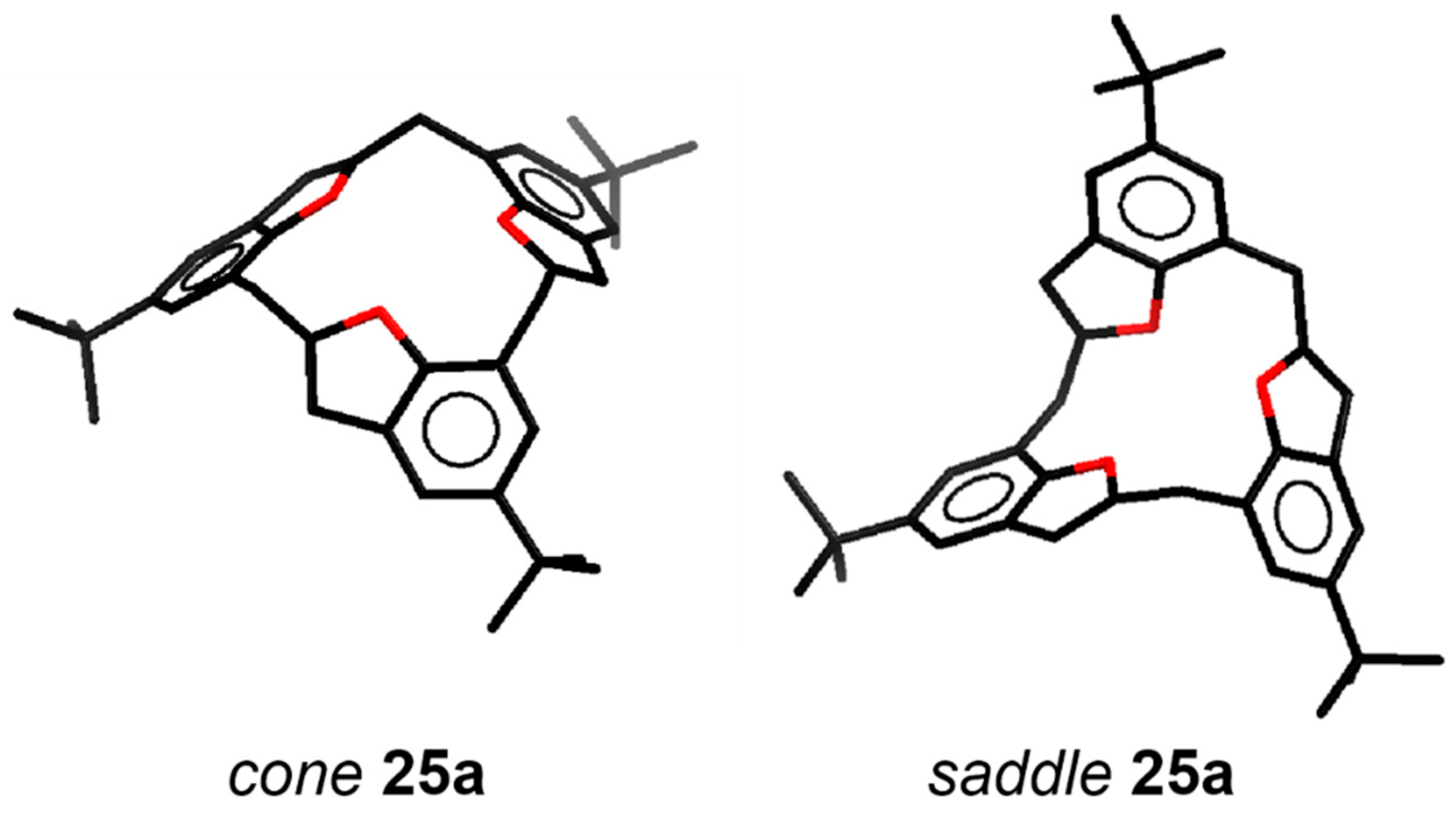
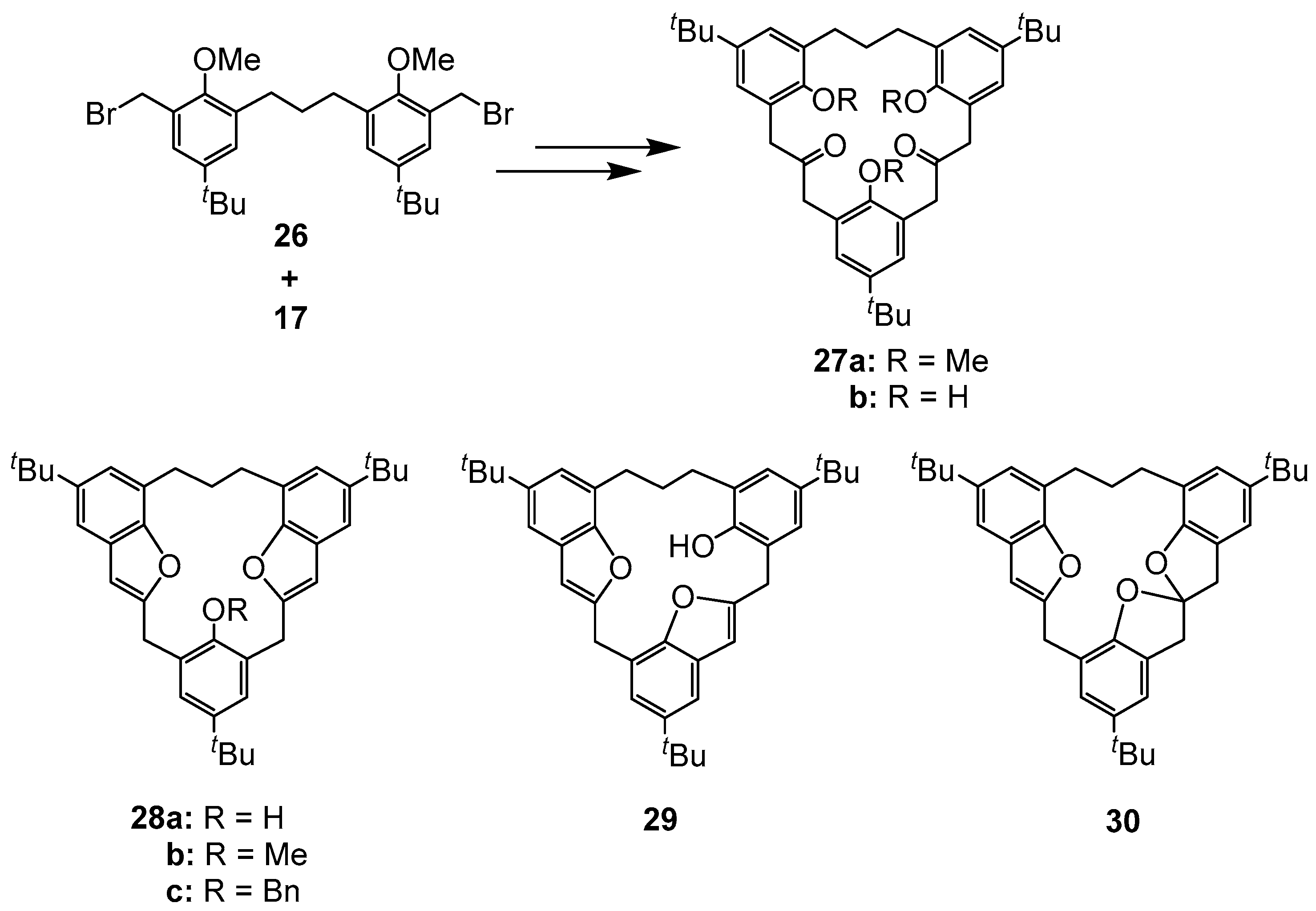
| Compound | Tc | ΔG≠ kJ mol−1 | Cone:saddle(−30 °C) |
|---|---|---|---|
| 25a | 40 a | 61.9 | 80:20 |
| 25b | 45 a | 62.8 | 100:0 |
| 25c | 28 a | 58.6 | 40:60 |
| 25d | 50 a | 68.2 | 100:0 |
| 25e | 75 b | 69.0 | 20:80 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Islam, M.M.; Georghiou, P.E.; Rahman, S.; Yamato, T. Calix[3]arene-Analogous Metacyclophanes: Synthesis, Structures and Properties with Infinite Potential. Molecules 2020, 25, 4202. https://doi.org/10.3390/molecules25184202
Islam MM, Georghiou PE, Rahman S, Yamato T. Calix[3]arene-Analogous Metacyclophanes: Synthesis, Structures and Properties with Infinite Potential. Molecules. 2020; 25(18):4202. https://doi.org/10.3390/molecules25184202
Chicago/Turabian StyleIslam, Md. Monarul, Paris E. Georghiou, Shofiur Rahman, and Takehiko Yamato. 2020. "Calix[3]arene-Analogous Metacyclophanes: Synthesis, Structures and Properties with Infinite Potential" Molecules 25, no. 18: 4202. https://doi.org/10.3390/molecules25184202
APA StyleIslam, M. M., Georghiou, P. E., Rahman, S., & Yamato, T. (2020). Calix[3]arene-Analogous Metacyclophanes: Synthesis, Structures and Properties with Infinite Potential. Molecules, 25(18), 4202. https://doi.org/10.3390/molecules25184202








