Imaging of Fibroblast Activation Protein Alpha Expression in a Preclinical Mouse Model of Glioma Using Positron Emission Tomography
Abstract
1. Introduction
2. Results
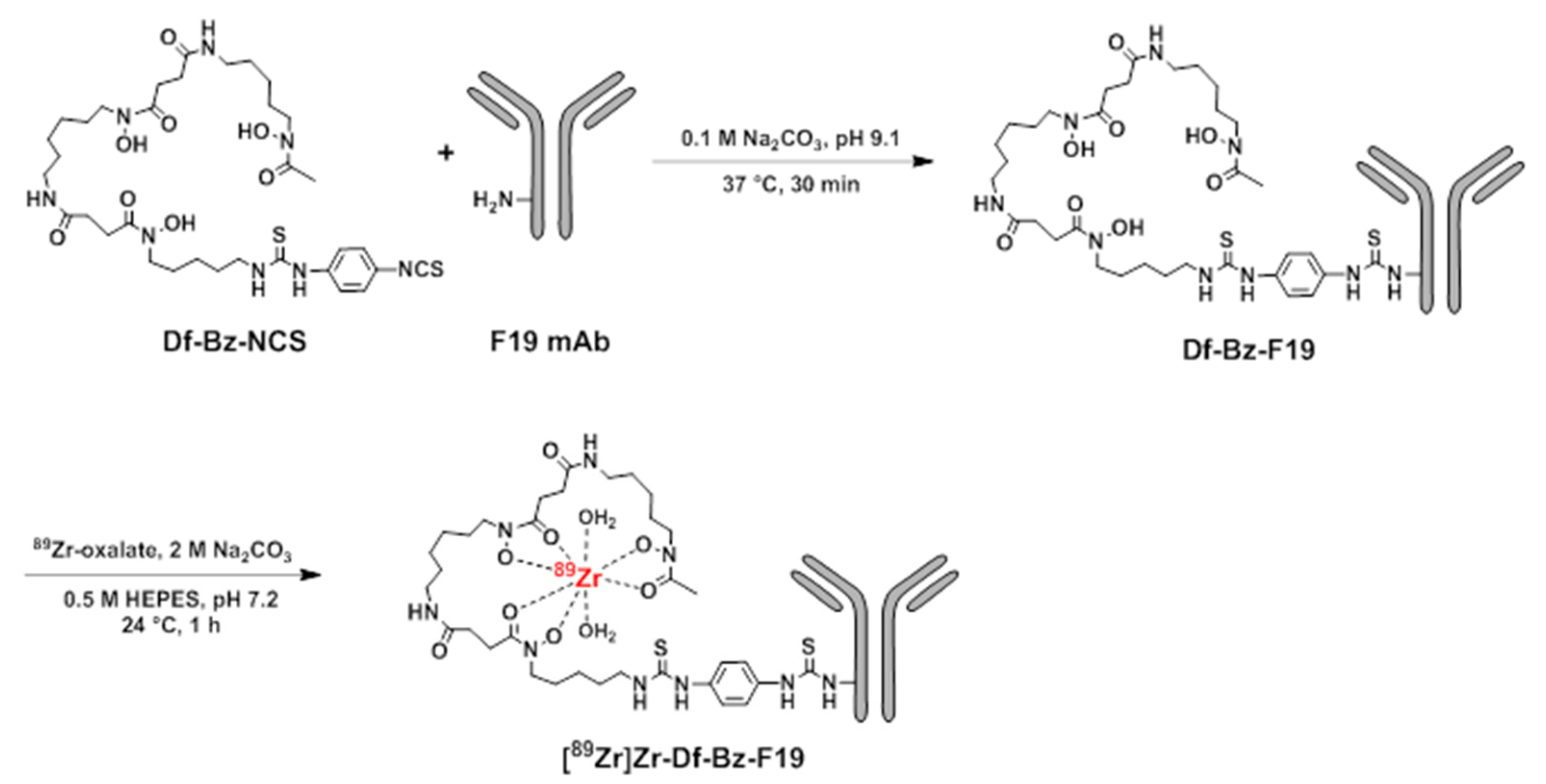
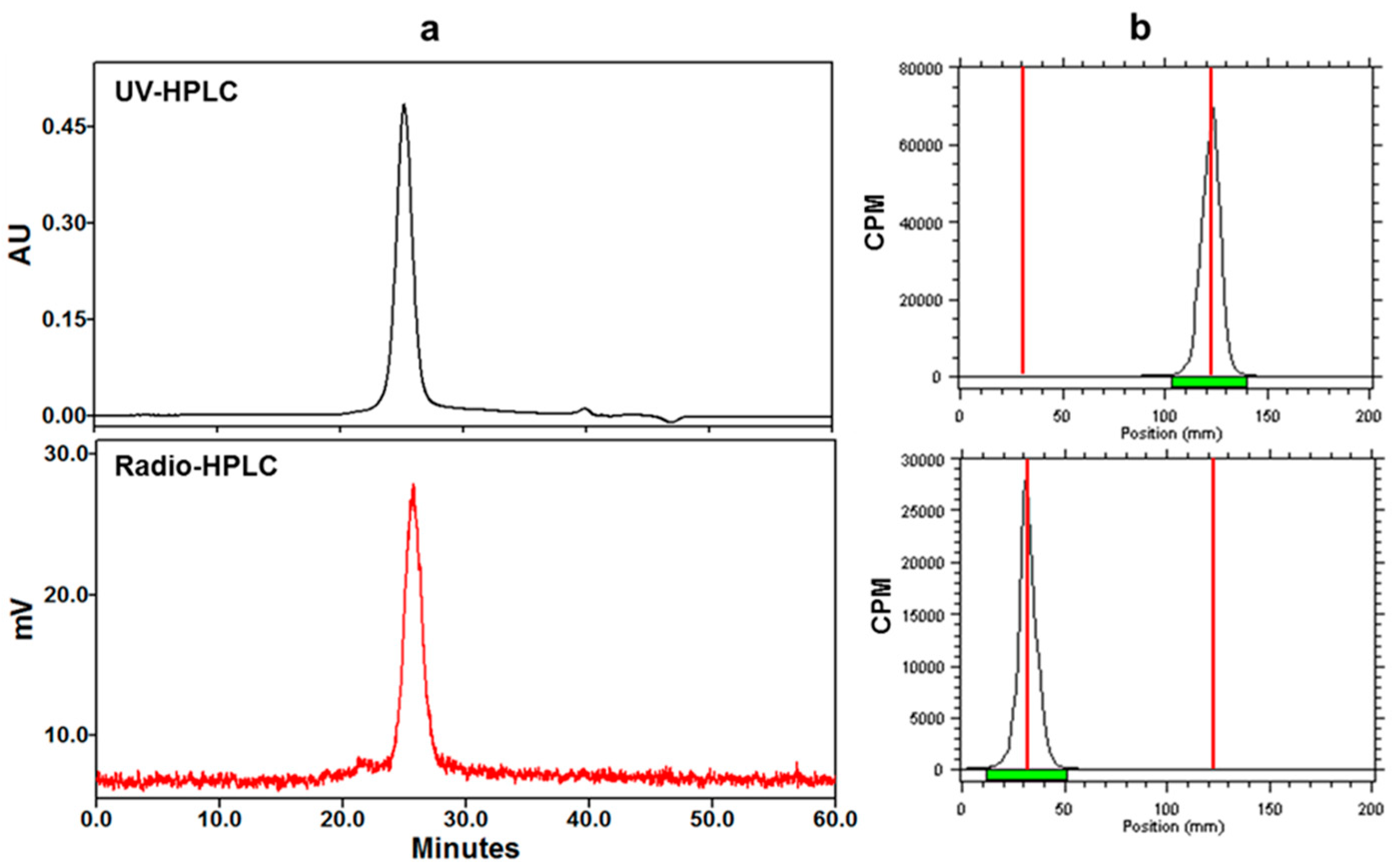
2.1. [89Zr]Zr-Df-Bz-F19 Preparation and In Vitro Characterization
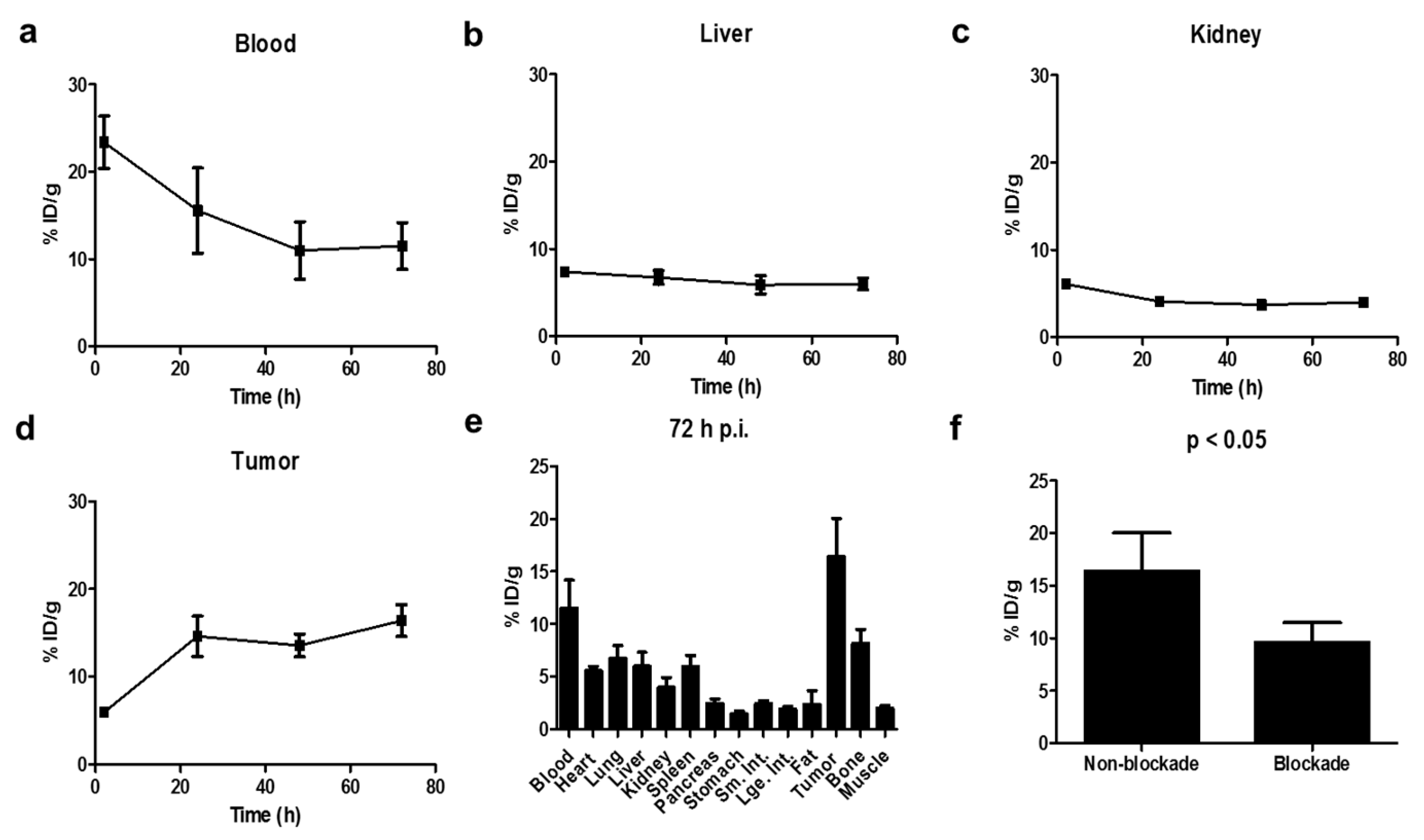
2.2. Biodistribution Studies
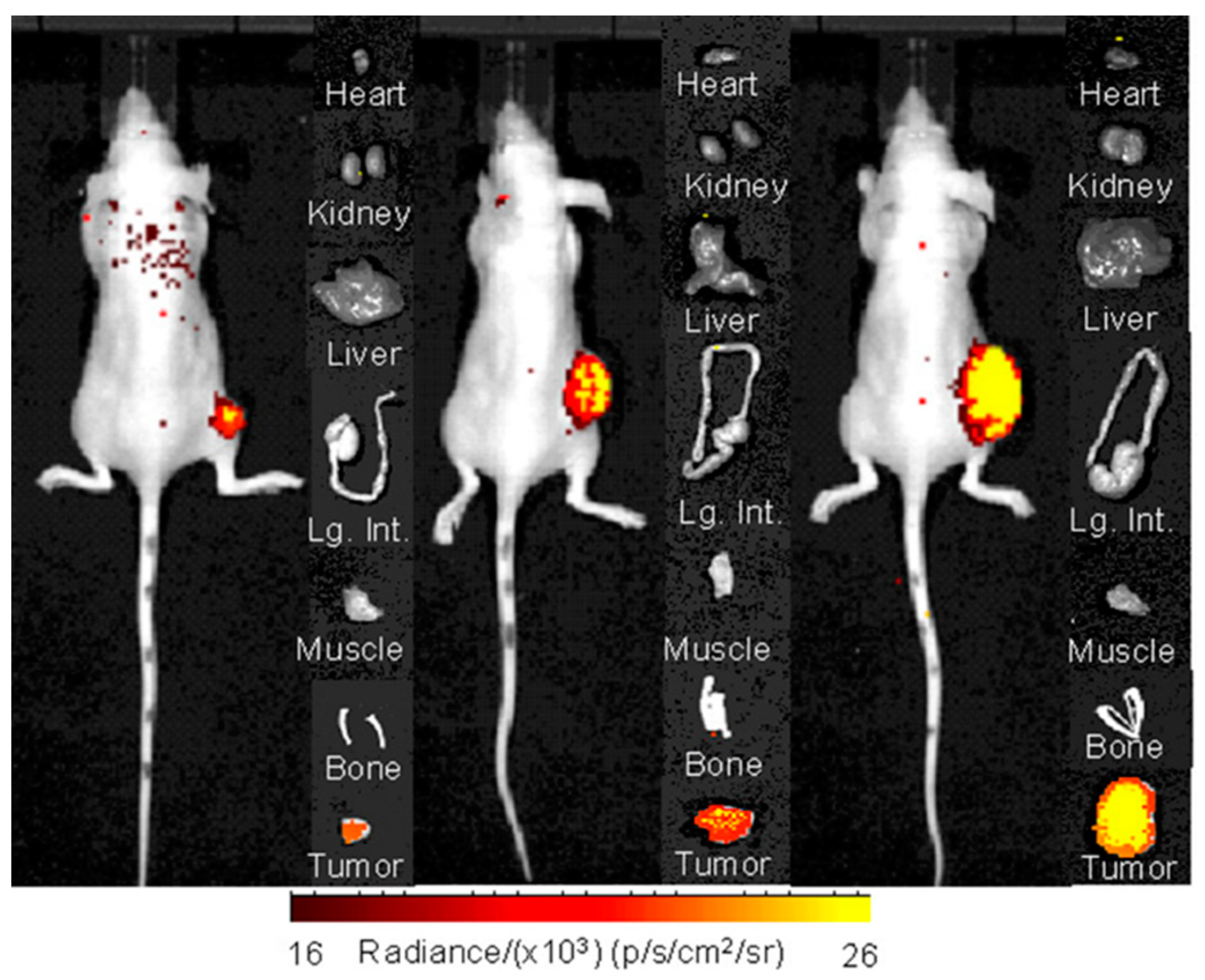
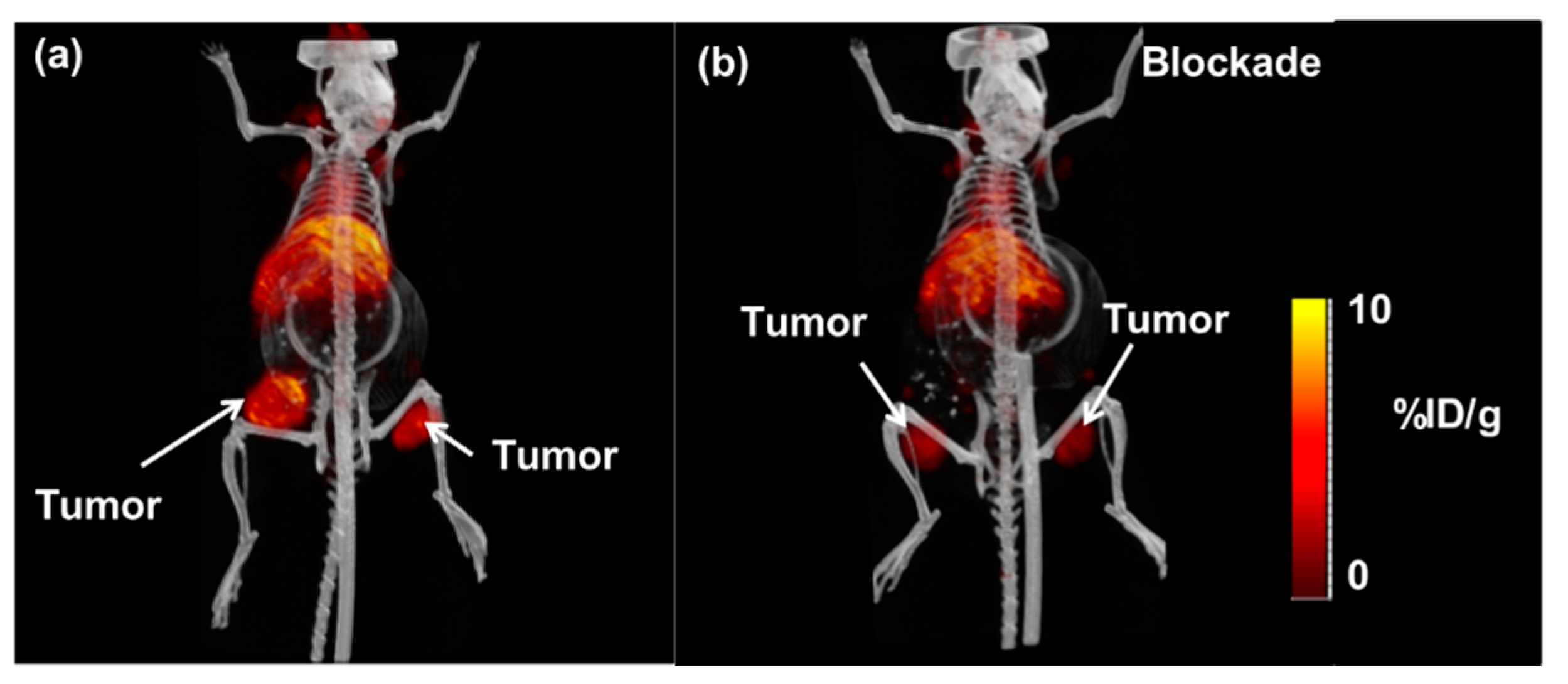
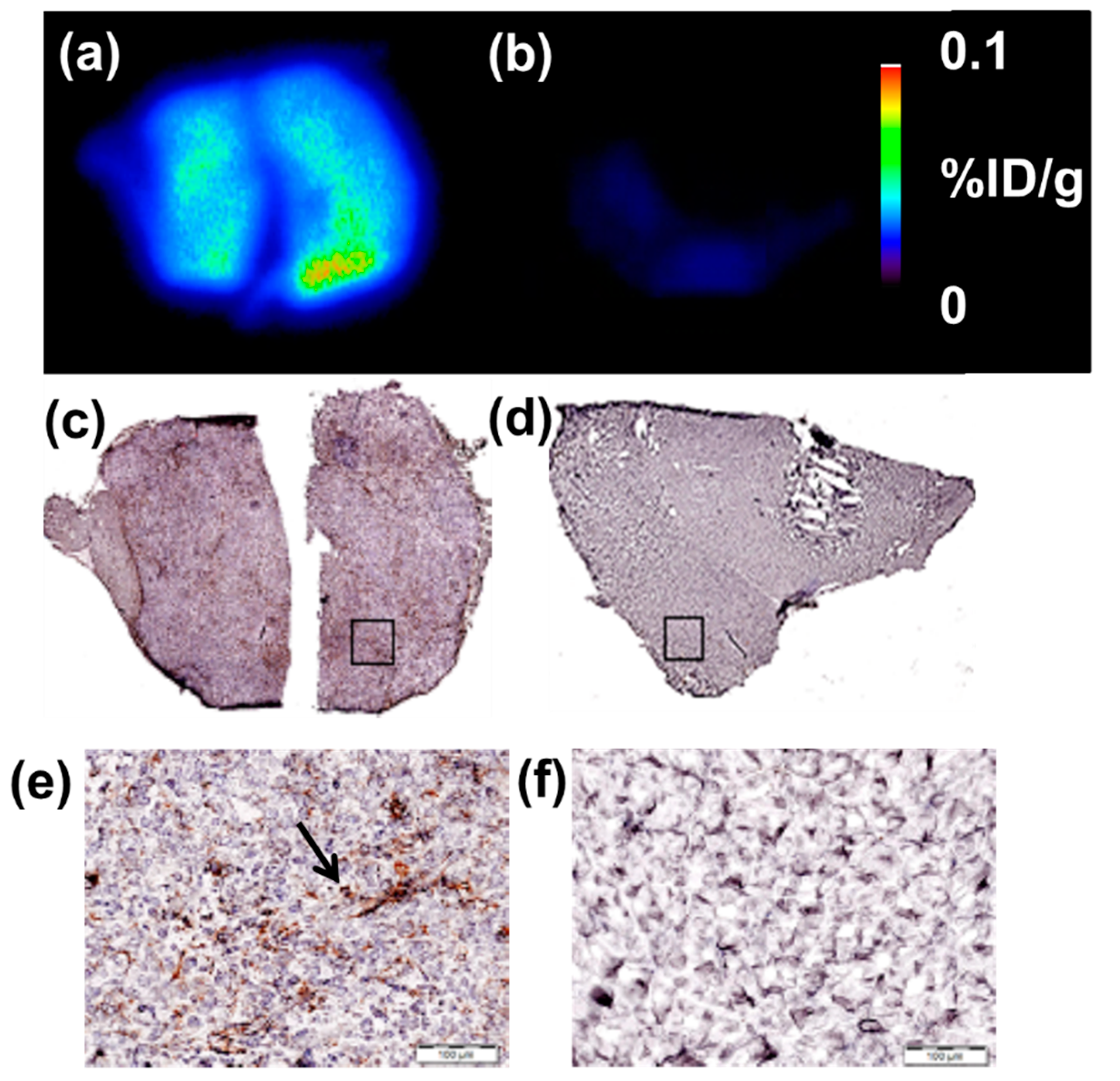
2.3. Small Animal Imaging Studies
3. Discussion
4. Materials and Methods
4.1. Reagents and Equipment
4.2. Conjugation, 89Zr-Radiolabeling, and Quality Control
4.3. In Vitro Serum Stability of [89Zr]Zr-Df-Bz-F19
4.4. Immunoreactivity (IR) Studies
4.5. Xenograft Models
4.6. Biodistribution
4.7. Small Animal Optical Imaging Using Cerenkov Luminescence
4.8. Small Animal PET/CT Imaging
4.9. Autoradiography (AR) and Immunohistochemistry (IHC)
4.10. Statistical Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jackson, C.M.; Lim, M. Immunotherapy for Glioblastoma: Playing Chess, Not Checkers. Clin. Cancer Res. 2018, 24, 4059–4061. [Google Scholar] [CrossRef] [PubMed]
- Lim, M.; Xia, Y.; Bettegowda, C.; Weller, M. Current state of immunotherapy for glioblastoma. Nat. Rev. Clin. Oncol. 2018, 15, 422–442. [Google Scholar] [CrossRef] [PubMed]
- Shergalis, A.; Bankhead, A.; Luesakul, U.; Muangsin, N.; Neamati, N. Current Challenges and Opportunities in Treating Glioblastoma. Pharmacol. Rev. 2018, 70, 412–445. [Google Scholar] [CrossRef] [PubMed]
- Jacob, M.; Chang, L.; Pure, E. Fibroblast activation protein in remodeling tissues. Curr. Mol. Med. 2012, 12, 1220–1243. [Google Scholar] [CrossRef] [PubMed]
- Pure, E.; Blomberg, R. Pro-tumorigenic roles of fibroblast activation protein in cancer: Back to the basics. Oncogene 2018, 37, 4343–4357. [Google Scholar] [CrossRef]
- Laverman, P.; van der Geest, T.; Terry, S.Y.; Gerrits, D.; Walgreen, B.; Helsen, M.M.; Nayak, T.K.; Freimoser-Grundschober, A.; Waldhauer, I.; Hosse, R.J.; et al. Immuno-PET and Immuno-SPECT of Rheumatoid Arthritis with Radiolabeled Anti-Fibroblast Activation Protein Antibody Correlates with Severity of Arthritis. J. Nucl. Med. 2015, 56, 778–783. [Google Scholar] [CrossRef]
- van der Geest, T.; Roeleveld, D.M.; Walgreen, B.; Helsen, M.M.; Nayak, T.K.; Klein, C.; Hegen, M.; Storm, G.; Metselaar, J.M.; van den Berg, W.B.; et al. Imaging fibroblast activation protein to monitor therapeutic effects of neutralizing interleukin-22 in collagen-induced arthritis. Rheumatology (Oxford) 2018, 57, 737–747. [Google Scholar] [CrossRef]
- Acharya, P.S.; Zukas, A.; Chandan, V.; Katzenstein, A.L.; Pure, E. Fibroblast activation protein: A serine protease expressed at the remodeling interface in idiopathic pulmonary fibrosis. Hum. Pathol. 2006, 37, 352–360. [Google Scholar] [CrossRef]
- Wang, X.M.; Yao, T.W.; Nadvi, N.A.; Osborne, B.; McCaughan, G.W.; Gorrell, M.D. Fibroblast activation protein and chronic liver disease. Front. Biosci. 2008, 13, 3168–3180. [Google Scholar] [CrossRef]
- Chopra, A. Fibroblast activation protein alpha-specific, near-infrared peptide probe (KGPGPNQC) linked to Cy5.5 and a quencher dye, QSY21. In Molecular Imaging and Contrast Agent Database (MICAD); National Center for Biotechnology Information: Bethesda, MD, USA, 2004. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23193619 (accessed on 4 July 2020).
- Li, J.; Chen, K.; Liu, H.; Cheng, K.; Yang, M.; Zhang, J.; Cheng, J.D.; Zhang, Y.; Cheng, Z. Activatable near-infrared fluorescent probe for in vivo imaging of fibroblast activation protein-alpha. Bioconjug. Chem. 2012, 23, 1704–1711. [Google Scholar] [CrossRef]
- Lo, P.C.; Chen, J.; Stefflova, K.; Warren, M.S.; Navab, R.; Bandarchi, B.; Mullins, S.; Tsao, M.; Cheng, J.D.; Zheng, G. Photodynamic molecular beacon triggered by fibroblast activation protein on cancer-associated fibroblasts for diagnosis and treatment of epithelial cancers. J. Med. Chem. 2009, 52, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Baird, S.K.; Allan, L.; Renner, C.; Scott, F.E.; Scott, A.M. Fibroblast activation protein increases metastatic potential of fibrosarcoma line HT1080 through upregulation of integrin-mediated signaling pathways. Clin. Exp. Metastasis 2015, 32, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Baird, S.K.; Rigopoulos, A.; Cao, D.; Allan, L.; Renner, C.; Scott, F.E.; Scot, A.M. Integral membrane protease fibroblast activation protein sensitizes fibrosarcoma to chemotherapy and alters cell death mechanisms. Apoptosis 2015, 20, 1483–1498. [Google Scholar] [CrossRef] [PubMed]
- Fischer, E.; Chaitanya, K.; Wuest, T.; Wadle, A.; Scott, A.M.; van den Broek, M.; Schibli, R.; Bauer, S.; Renner, C. Radioimmunotherapy of fibroblast activation protein positive tumors by rapidly internalizing antibodies. Clin. Cancer Res. 2012, 18, 6208–6218. [Google Scholar] [CrossRef] [PubMed]
- Kloft, C.; Graefe, E.U.; Tanswell, P.; Scott, A.M.; Hofheinz, R.; Amelsberg, A.; Karlsson, M.O. Population pharmacokinetics of sibrotuzumab, a novel therapeutic monoclonal antibody, in cancer patients. Invest. New Drugs 2004, 22, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Scott, A.M.; Wiseman, G.; Welt, S.; Adjei, A.; Lee, F.T.; Hopkins, W.; Divgi, C.R.; Hanson, L.H.; Mitchell, P.; Gansen, D.N.; et al. A Phase I dose-escalation study of sibrotuzumab in patients with advanced or metastatic fibroblast activation protein-positive cancer. Clin. Cancer Res. 2003, 9, 1639–1647. [Google Scholar] [PubMed]
- Tahtis, K.; Lee, F.T.; Wheatley, J.M.; Garin-Chesa, P.; Park, J.E.; Smyth, F.E.; Obata, Y.; Stockert, E.; Hall, C.M.; Old, L.J. Expression and targeting of human fibroblast activation protein in a human skin/severe combined immunodeficient mouse breast cancer xenograft model. Mol. Cancer Ther. 2003, 2, 729–737. [Google Scholar]
- Tanswell, P.; Garin-Chesa, P.; Rettig, W.J.; Welt, S.; Divgi, C.R.; Casper, E.S.; Finn, R.D.; Larson, S.M.; Old, L.J.; Scott, A.M. Population pharmacokinetics of antifibroblast activation protein monoclonal antibody F19 in cancer patients. Br. J. Clin. Pharmacol. 2001, 51, 177–180. [Google Scholar] [CrossRef]
- Welt, S.; Divgi, C.R.; Scott, A.M.; Garin-Chesa, P.; Finn, R.D.; Graham, M.; Carswell, E.A.; Cohen, A.; Larson, S.M.; Old, L.J. Antibody targeting in metastatic colon cancer: A phase I study of monoclonal antibody F19 against a cell-surface protein of reactive tumor stromal fibroblasts. J. Clin. Oncol. 1994, 12, 1193–1203. [Google Scholar] [CrossRef]
- Rabenhold, M.; Steiniger, F.; Fahr, A.; Kontermann, R.E.; Ruger, R. Bispecific single-chain diabody-immunoliposomes targeting endoglin (CD105) and fibroblast activation protein (FAP) simultaneously. J. Control. Release 2015, 201, 56–67. [Google Scholar] [CrossRef]
- Tansi, F.L.; Ruger, R.; Bohm, C.; Steiniger, F.; Kontermann, R.E.; Teichgraeber, U.K.; Fahr, A.; Hilger, I. Activatable bispecific liposomes bearing fibroblast activation protein directed single chain fragment/Trastuzumab deliver encapsulated cargo into the nuclei of tumor cells and the tumor microenvironment simultaneously. Acta Biomater. 2017, 54, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Tansi, F.L.; Ruger, R.; Kollmeier, A.M.; Rabenhold, M.; Steiniger, F.; Kontermann, R.E.; Teichgraeber, U.K.; Fahr, A.; Hilger, I. Targeting the Tumor Microenvironment with Fluorescence-Activatable Bispecific Endoglin/Fibroblast Activation Protein Targeting Liposomes. Pharmaceutics 2020, 12, 370. [Google Scholar] [CrossRef] [PubMed]
- Baum, P.; Muller, D.; Ruger, R.; Kontermann, R.E. Single-chain Fv immunoliposomes for the targeting of fibroblast activation protein-expressing tumor stromal cells. J. Drug Target 2007, 15, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhao, L.; Ruan, D.; Sun, L.; Lin, Q. 68Ga-FAPI PET/CT Improves Therapeutic Strategy by Detecting a Second Primary Malignancy in a Patient with Rectal Cancer. Clin. Nucl. Med. 2020, 45, 468–470. [Google Scholar] [CrossRef] [PubMed]
- Giesel, F.L.; Kratochwil, C.; Lindner, T.; Marschalek, M.M.; Loktev, A.; Lehnert, W.; Debus, J.; Jager, D.; Flechsig, P.; Mier, W.; et al. (68)Ga-FAPI PET/CT: Biodistribution and Preliminary Dosimetry Estimate of 2 DOTA-Containing FAP-Targeting Agents in Patients with Various Cancers. J. Nucl. Med. 2019, 60, 386–392. [Google Scholar] [CrossRef] [PubMed]
- Koerber, S.A.; Staudinger, F.; Kratochwil, C.; Adeberg, S.; Haefner, M.F.; Ungerechts, G.; Rathke, H.; Winter, E.; Lindner, T.; Syed, M.; et al. The role of FAPI-PET/CT for patients with malignancies of the lower gastrointestinal tract - first clinical experience. J. Nucl. Med. 2020. [Google Scholar] [CrossRef]
- Kratochwil, C.; Flechsig, P.; Lindner, T.; Abderrahim, L.; Altmann, A.; Mier, W.; Adberg, S.; Rathke, H.; Rohrich, M.; Winter, H.; et al. (68)Ga-FAPI PET/CT: Tracer Uptake in 28 Different Kinds of Cancer. J. Nucl. Med. 2019, 60, 801–805. [Google Scholar] [CrossRef]
- Lindner, T.; Altmann, A.; Kraemer, S.; Kleist, C.; Loktev, A.; Kratochwil, C.; Giesel, F.; Mier, W.; Marme, F.; Debus, J.; et al. Design and development of (99m)Tc labeled FAPI-tracers for SPECT-imaging and 188Re therapy. J. Nucl. Med. 2020. [Google Scholar] [CrossRef]
- Luo, Y.; Pan, Q.; Yang, H.; Peng, L.; Zhang, W.; Li, F. Fibroblast activation protein targeted PET/CT with (68)Ga-FAPI for imaging IgG4-related disease: Comparison to (18)F-FDG PET/CT. J. Nucl. Med. 2020. [Google Scholar] [CrossRef]
- Meyer, C.; Dahlbom, M.; Lindner, T.; Vauclin, S.; Mona, C.; Slavik, R.; Czernin, J.; Haberkorn, U.; Calais, J. Radiation dosimetry and biodistribution of (68)Ga-FAPI-46 PET imaging in cancer patients. J. Nucl. Med. 2019. [Google Scholar] [CrossRef]
- Syed, M.; Flechsig, P.; Liermann, J.; Windisch, P.; Staudinger, F.; Akbaba, S.; Koerber, S.A.; Freudlsperger, C.; Plinkert, P.K.; Debus, J.; et al. Fibroblast activation protein inhibitor (FAPI) PET for diagnostics and advanced targeted radiotherapy in head and neck cancers. Eur. J. Nucl. Med. Mol. Imaging 2020. [Google Scholar] [CrossRef] [PubMed]
- Varasteh, Z.; Mohanta, S.; Robu, S.; Braeuer, M.; Li, Y.; Omidvari, N.; Topping, G.; Sun, T.; Nekolla, S.G.; Richter, A.; et al. Molecular Imaging of Fibroblast Activity After Myocardial Infarction Using a (68)Ga-Labeled Fibroblast Activation Protein Inhibitor, FAPI-04. J. Nucl. Med. 2019, 60, 1743–1749. [Google Scholar] [CrossRef]
- Watabe, T.; Liu, Y.; Kaneda-Nakashima, K.; Shirakami, Y.; Lindner, T.; Ooe, K.; Toyoshima, A.; Nagata, K.; Shimosegawa, E.; Haberkorn, U.; et al. Theranostics Targeting Fibroblast Activation Protein in the Tumor Stroma: (64)Cu- and (225)Ac-Labeled FAPI-04 in Pancreatic Cancer Xenograft Mouse Models. J. Nucl. Med. 2020, 61, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Mentlein, R.; Hattermann, K.; Hemion, C.; Jungbluth, A.A.; Held-Feindt, J. Expression and role of the cell surface protease seprase/fibroblast activation protein-alpha (FAP-alpha) in astroglial tumors. Biol. Chem. 2011, 392, 199–207. [Google Scholar] [CrossRef]
- Balaziova, E.; Busek, P.; Stremenova, J.; Sromova, L.; Krepela, E.; Lizcova, L.; Sedo, A. Coupled expression of dipeptidyl peptidase-IV and fibroblast activation protein-alpha in transformed astrocytic cells. Mol. Cell Biochem. 2011, 354, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Busek, P.; Balaziova, E.; Matrasova, I.; Hilser, M.; Tomas, R.; Syrucek, M.; Zemanova, Z.; Krepela, E.; Belacek, J.; Sedo, A. Fibroblast activation protein alpha is expressed by transformed and stromal cells and is associated with mesenchymal features in glioblastoma. Tumour Biol. 2016, 37, 13961–13971. [Google Scholar] [CrossRef] [PubMed]
- Busek, P.; Mateu, R.; Zubal, M.; Kotackova, L.; Sedo, A. Targeting fibroblast activation protein in cancer—Prospects and caveats. Front. Biosci. (Landmark Ed.) 2018, 23, 1933–1968. [Google Scholar]
- Matrasova, I.; Busek, P.; Balaziova, E.; Sedo, A. Heterogeneity of molecular forms of dipeptidyl peptidase-IV and fibroblast activation protein in human glioblastomas. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2017, 161, 252–260. [Google Scholar] [CrossRef]
- Bhatt, N.B.; Pandya, D.N.; Wadas, T.J. Recent Advances in Zirconium-89 Chelator Development. Molecules 2018, 23, 638. [Google Scholar] [CrossRef]
- Wadas, T.J.; Wong, E.H.; Weisman, G.R.; Anderson, C.J. Coordinating radiometals of copper, gallium, indium, yttrium, and zirconium for PET and SPECT imaging of disease. Chem. Rev. 2010, 110, 2858–2902. [Google Scholar] [CrossRef]
- Das, S.; Thorek, D.L.; Grimm, J. Cerenkov imaging. Adv. Cancer Res. 2014, 124, 213–234. [Google Scholar] [CrossRef] [PubMed]
- Tamura, R.; Pratt, E.C.; Grimm, J. Innovations in Nuclear Imaging Instrumentation: Cerenkov Imaging. Semin. Nucl. Med. 2018, 48, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, A.; Holland, J.P.; Lewis, J.S.; Grimm, J. Cerenkov luminescence imaging of medical isotopes. J. Nucl. Med. 2010, 51, 1123–1130. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, N.B.; Pandya, D.N.; Rideout-Danner, S.; Gage, H.D.; Marini, F.C.; Wadas, T.J. A comprehensively revised strategy that improves the specific activity and long-term stability of clinically relevant 89Zr-immuno-PET agents. Dalton Trans. 2018, 47, 13214–13221. [Google Scholar] [CrossRef]
- Tinianow, J.N.; Pandya, D.N.; Pailloux, S.L.; Ogasawara, A.; Vanderbilt, A.N.; Gill, H.S.; Williams, S.P.; Wadas, T.J.; Magda, D.; Marik, J. Evaluation of a 3-hydroxypyridin-2-one (2,3-HOPO) Based Macrocyclic Chelator for 89Zr4+ and Its Use for ImmunoPET Imaging of HER2 Positive Model of Ovarian Carcinoma in Mice. Theranostics 2016, 6, 511–521. [Google Scholar] [CrossRef]
- Pandya, D.N.; Bhatt, N.B.; Almaguel, F.; Rideout-Danner, S.; Gage, H.D.; Solingapuram Sai, K.K.; Wadas, T.J. 89Zr-Chloride Can Be Used for Immuno-PET Radiochemistry Without Loss of Antigen Reactivity In Vivo. J. Nucl. Med. 2019, 60, 696–701. [Google Scholar] [CrossRef]
- Rana, T.M.; Meares, C.F. N-terminal modification of immunoglobulin polypeptide chains tagged with isothiocyanato chelates. Bioconjug. Chem. 1990, 1, 357–362. [Google Scholar] [CrossRef]
- Messerschmidt, S.K.; Kolbe, A.; Muller, D.; Knoll, M.; Pleiss, J.; Kontermann, R.E. Novel single-chain Fv’ formats for the generation of immunoliposomes by site-directed coupling. Bioconjug. Chem. 2008, 19, 362–369. [Google Scholar] [CrossRef]
- Sarav, M.; Wang, Y.; Hack, B.K.; Chang, A.; Jensen, M.; Bao, L.; Quigg, R.J. Renal FcRn reclaims albumin but facilitates elimination of IgG. J. Am. Soc. Nephrol. 2009, 20, 1941–1952. [Google Scholar] [CrossRef]
- Morita, H.; Yoshimura, A.; Kimata, K. The role of heparan sulfate in the glomerular basement membrane. Kidney Int. 2008, 73, 247–248. [Google Scholar] [CrossRef]
- van den Hoven, M.J.; Wijnhoven, T.J.; Li, J.P.; Zcharia, E.; Dijkman, H.B.; Wismans, R.G.; Rops, A.L.; Lensen, J.F.; van den Heuvel, L.P.; van Kuppevelt, T.H.; et al. Reduction of anionic sites in the glomerular basement membrane by heparanase does not lead to proteinuria. Kidney Int. 2008, 73, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Wadas, T.J.; Wong, E.H.; Weisman, G.R.; Anderson, C.J. Copper chelation chemistry and its role in copper radiopharmaceuticals. Curr. Pharm. Des. 2007, 13, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, N.B.; Pandya, D.N.; Dezarn, W.A.; Marini, F.C.; Zhao, D.; Gmeiner, W.H.; Triozzi, P.L.; Wadas, T.J. Practical Guidelines for Cerenkov Luminescence Imaging with Clinically Relevant Isotopes. Methods Mol. Biol. 2018, 1790, 197–208. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, T.M.; Drain, C.M.; Grimm, J. Optical Imaging of Ionizing Radiation from Clinical Sources. J. Nucl. Med. 2016, 57, 1661–1666. [Google Scholar] [CrossRef]
- Hamson, E.J.; Keane, F.M.; Tholen, S.; Schilling, O.; Gorrell, M.D. Understanding fibroblast activation protein (FAP): Substrates, activities, expression and targeting for cancer therapy. Proteomics Clin. Appl. 2014, 8, 454–463. [Google Scholar] [CrossRef]
- Arvantis, C.D.; Askoxylakis, V.; Guo, Y.; Datta, M.; Kloepper, J.; Ferraro, G.B.; Bernabeu, M.O.; Fukumura, D.; McDannold, N.; Jain, R.K. Mechanisms of enhanced drug delivery in brain metastases with focused ultrasound-induced blood–tumor barrier disruption. Proc. Natl. Acad. Sci. USA 2018, 115, E8717–E8726. [Google Scholar] [CrossRef]
- Arvanitis, C.D.; Ferraro, G.B.; Jain, R.K. The blood-brain barrier and blood-tumour barrier in brain tumours and metastases. Nat. Rev. Cancer 2020, 20, 26–41. [Google Scholar] [CrossRef]
- Sattiraju, A.; Solingapuram Sai, K.K.; Xuan, A.; Pandya, D.N.; Almaguel, F.G.; Wadas, T.J.; Herpai, D.M.; Debinski, W.; Mintz, A. IL13RA2 targeted alpha particle therapy against glioblastomas. Oncotarget 2017, 8, 42997–43007. [Google Scholar] [CrossRef]
- Sattiraju, A.; Sun, Y.; Solingapuram Sai, K.K.; Li, K.C.P.; Mintz, A. Part IV: Image-Guided, Remote-Controlled Opening of the Blood-Brain Barrier for Systemic Brain Tumor Therapy. In Maximizing Local Access to Therapeutic Deliveries in Glioblastoma; De Vleeschouwer, S., Ed.; Glioblastoma: Brisbane, Australia, 2017. [Google Scholar]
- Sattiraju, A.; Xiong, X.; Pandya, D.N.; Wadas, T.J.; Xuan, A.; Sun, Y.; Jung, Y.; Sai, K.K.S.; Dorsey, J.F.; Li, K.; et al. Alpha Particle Enhanced Blood Brain/Tumor Barrier Permeabilization in Glioblastomas Using Integrin Alpha-v Beta-3-Targeted Liposomes. Mol. Cancer Ther. 2017, 16, 2191–2200. [Google Scholar] [CrossRef]
- Sun, Y.; Xiong, X.; Pandya, D.; Jung, Y.; Mintz, A.; Hayasaka, S.; Wadas, T.J.; Li, K.C.P. Enhancing tissue permeability with MRI guided preclinical focused ultrasound system in rabbit muscle: From normal tissue to VX2 tumor. J. Control. Release 2017, 256, 1–8. [Google Scholar] [CrossRef]
- Wang, L.; Habib, A.A.; Mintz, A.; Li, K.C.; Zhao, D. Phosphatidylserine-Targeted Nanotheranostics for Brain Tumor Imaging and Therapeutic Potential. Mol. Imaging 2017, 16. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.; Sun, Y.; Sattiraju, A.; Jung, Y.; Mintz, A.; Hayasaka, S.; Li, K.C.P. Remote spatiotemporally controlled and biologically selective permeabilization of blood-brain barrier. J. Control. Release 2015, 217, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Ciarrocchi, E.; Belcari, N. Cerenkov luminescence imaging: Physics principles and potential applications in biomedical sciences. EJNMMI Phys. 2017, 4. [Google Scholar] [CrossRef] [PubMed]
- Grootendorst, M.R.; Cariati, M.; Kothari, A.; Tuch, D.S.; Purushotham, A. Cerenkov luminescence imaging (CLI) for image-guided cancer surgery. Clin. Transl. Imaging 2016, 4, 353–366. [Google Scholar] [CrossRef] [PubMed]
- Pandya, D.N.; Hantgan, R.; Budzevich, M.M.; Kock, N.D.; Morse, D.L.; Batista, I.; Mintz, A.; Li, K.C.P.; Wadas, T.J. Preliminary Therapy Evaluation of (225)Ac-DOTA-c(RGDyK) Demonstrates that Cerenkov Radiation Derived from (225)Ac Daughter Decay Can Be Detected by Optical Imaging for In Vivo Tumor Visualization. Theranostics 2016, 6, 698–709. [Google Scholar] [CrossRef] [PubMed]
- Schwenck, J.; Fuchs, K.; Eilenberger, S.H.; Rolle, A.M.; Castaneda Vega, S.; Thaiss, W.M.; Maier, F.C. Fluorescence and Cerenkov luminescence imaging. Applications in small animal research. Nuklearmedizin 2016, 55, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Liu, H.; Cheng, Z. Harnessing the power of radionuclides for optical imaging: Cerenkov luminescence imaging. J. Nucl. Med. 2011, 52, 2009–2018. [Google Scholar] [CrossRef]
- Zhang, J.G.; Liu, H.F. Functional imaging and endoscopy. World J. Gastroenterol 2011, 17, 4277–4282. [Google Scholar] [CrossRef]
- Aras, G.; Arican, P.; Cam, R.; Kucuk, N.O.; Ibis, E.; Tuzuner, A.; Soylu, A. Identification of sentinel lymph node in breast cancer by lymphoscintigraphy and surgical gamma probe with peritumoral injection of scintimammographic agent “99mTc MIbI”. Ann. Nucl. Med. 2002, 16, 121–126. [Google Scholar] [CrossRef]
- Arcan, P.; Ibis, E.; Aras, G.; Cam, R.; Kucuk, N.O. Identification of sentinel lymph node in stage I-II breast cancer with lymphoscintigraphy and surgical gamma probe: Comparison of Tc-99m MIBI and Tc-99m sulfur colloid. Clin. Nucl. Med. 2005, 30, 317–321. [Google Scholar] [CrossRef]
- Atinkaya, C.; Ozlem Kucuk, N.; Koparal, H.; Aras, G.; Sak, S.D.; Ozdemir, N. Mediastinal intraoperative radioisotope sentinel lymph node mapping in non-small-cell lung cancer. Nucl. Med. Commun. 2005, 26, 717–720. [Google Scholar] [CrossRef] [PubMed]
- Zulfikaroglu, B.; Koc, M.; Ozmen, M.M.; Kucuk, N.O.; Ozalp, N.; Aras, G. Intraoperative lymphatic mapping and sentinel lymph node biopsy using radioactive tracer in gastric cancer. Surgery 2005, 138, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Holland, J.P.; Sheh, Y.; Lewis, J.S. Standardized methods for the production of high specific-activity zirconium-89. Nucl. Med. Biol. 2009, 36, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Rettig, W.J.; Garin-Chesa, P.; Beresford, H.R.; Oettgen, H.F.; Melamed, M.R.; Old, L.J. Cell-surface glycoproteins of human sarcomas: Differential expression in normal and malignant tissues and cultured cells. Proc. Natl. Acad. Sci. USA 1988, 85, 3110–3114. [Google Scholar] [CrossRef]
- Rettig, W.J.; Su, S.L.; Fortunato, S.R.; Scanlan, M.J.; Raj, B.K.; Garin-Chesa, P.; Healey, J.H.; Old, L.J. Fibroblast activation protein: Purification, epitope mapping and induction by growth factors. Int. J. Cancer 1994, 58, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Boven, E.; Lindmo, T.; Mitchell, J.B.; Bunn, P.A., Jr. Selective cytotoxicity of 125I-labeled monoclonal antibody T101 in human malignant T cell lines. Blood 1986, 67, 429–435. [Google Scholar] [CrossRef]
- Pandya, D.N.; Bhatt, N.; Yuan, H.; Day, C.S.; Ehrmann, B.M.; Wright, M.; Bierbach, U.; Wadas, T.J. Zirconium tetraazamacrocycle complexes display extraordinary stability and provide a new strategy for zirconium-89-based radiopharmaceutical development. Chem. Sci. 2017, 8, 2309–2314. [Google Scholar] [CrossRef]
- Gambhir, S.S.; Barrio, J.R.; Wu, L.; Iyer, M.; Namavari, M.; Satyamurthy, N.; Bauer, E.; Parrish, C.; MacLaren, D.G.; Borghei, A.R.; et al. Imaging of adenoviral-directed herpes simplex virus type 1 thymidine kinase reporter gene expression in mice with radiolabeled ganciclovir. J. Nucl. Med. 1998, 39, 2003–2011. [Google Scholar]
- Hoang, L.L.; Tacha, D.E.; Qi, W.; Yu, C.; Bremer, R.E.; Chu, J.; Haas, T.S.; Cheng, L. A newly developed uroplakin II antibody with increased sensitivity in urothelial carcinoma of the bladder. Arch. Pathol. Lab. Med. 2014, 138, 943–949. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pandya, D.N.; Sinha, A.; Yuan, H.; Mutkus, L.; Stumpf, K.; Marini, F.C.; Wadas, T.J. Imaging of Fibroblast Activation Protein Alpha Expression in a Preclinical Mouse Model of Glioma Using Positron Emission Tomography. Molecules 2020, 25, 3672. https://doi.org/10.3390/molecules25163672
Pandya DN, Sinha A, Yuan H, Mutkus L, Stumpf K, Marini FC, Wadas TJ. Imaging of Fibroblast Activation Protein Alpha Expression in a Preclinical Mouse Model of Glioma Using Positron Emission Tomography. Molecules. 2020; 25(16):3672. https://doi.org/10.3390/molecules25163672
Chicago/Turabian StylePandya, Darpan N., Akesh Sinha, Hong Yuan, Lysette Mutkus, Kristina Stumpf, Frank C. Marini, and Thaddeus J. Wadas. 2020. "Imaging of Fibroblast Activation Protein Alpha Expression in a Preclinical Mouse Model of Glioma Using Positron Emission Tomography" Molecules 25, no. 16: 3672. https://doi.org/10.3390/molecules25163672
APA StylePandya, D. N., Sinha, A., Yuan, H., Mutkus, L., Stumpf, K., Marini, F. C., & Wadas, T. J. (2020). Imaging of Fibroblast Activation Protein Alpha Expression in a Preclinical Mouse Model of Glioma Using Positron Emission Tomography. Molecules, 25(16), 3672. https://doi.org/10.3390/molecules25163672






