Hydrogels for the Delivery of Plant-Derived (Poly)Phenols
Abstract
1. Introduction
2. Potential Biomedical Applications of (Poly)Phenols Loaded-Hydrogels
2.1. Skin and Epithelial Diseases
2.1.1. Skin Wound Healing
2.1.2. Epithelial Pathologies
2.1.3. Epithelial Cancer
2.2. Injectable and Targeted Hydrogels
2.3. Hydrogels for Oral and Systemic Administration
3. Hydrogels Projected for Topical Applications
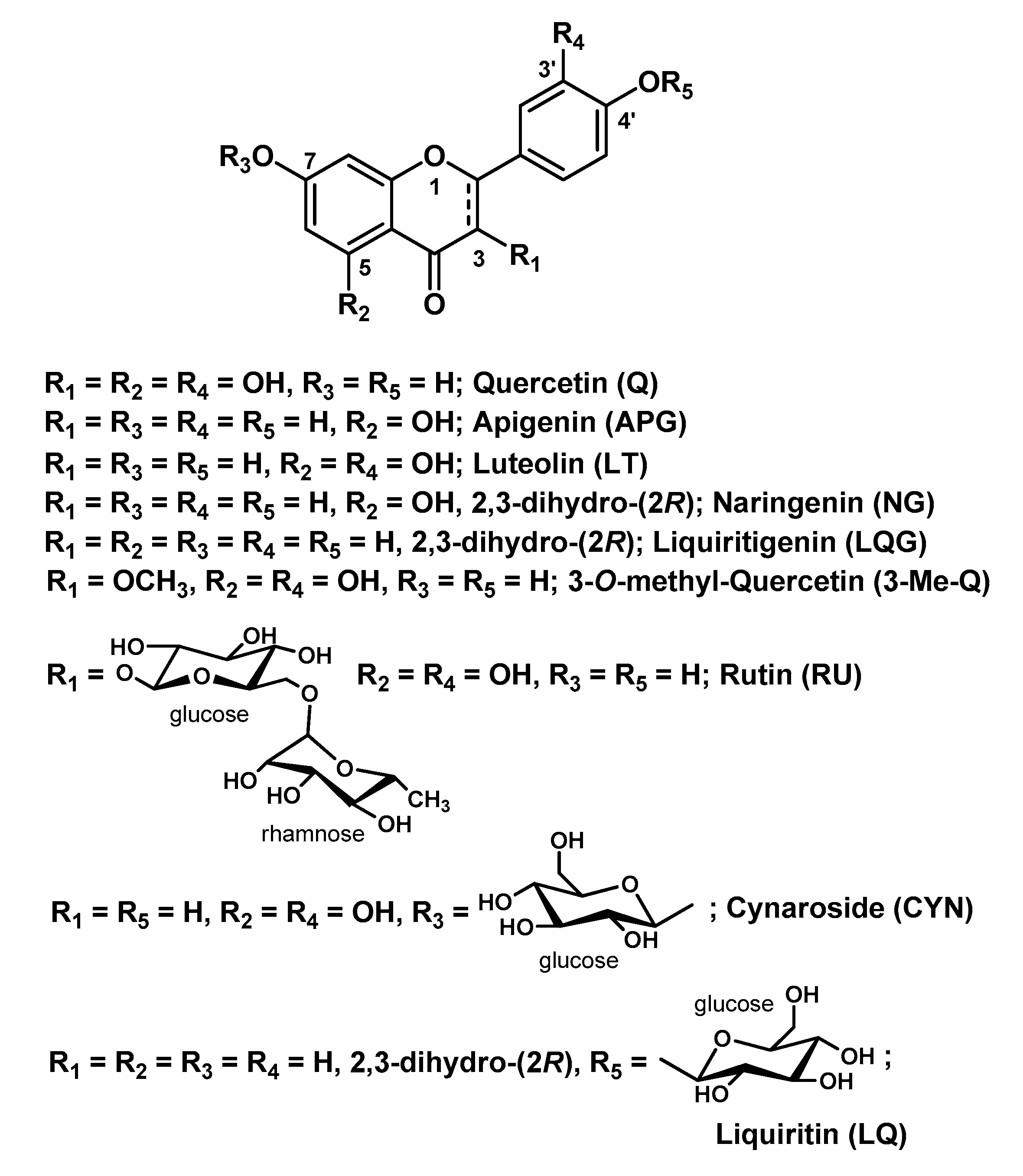
3.1. Quercetin-Containing Hydrogels
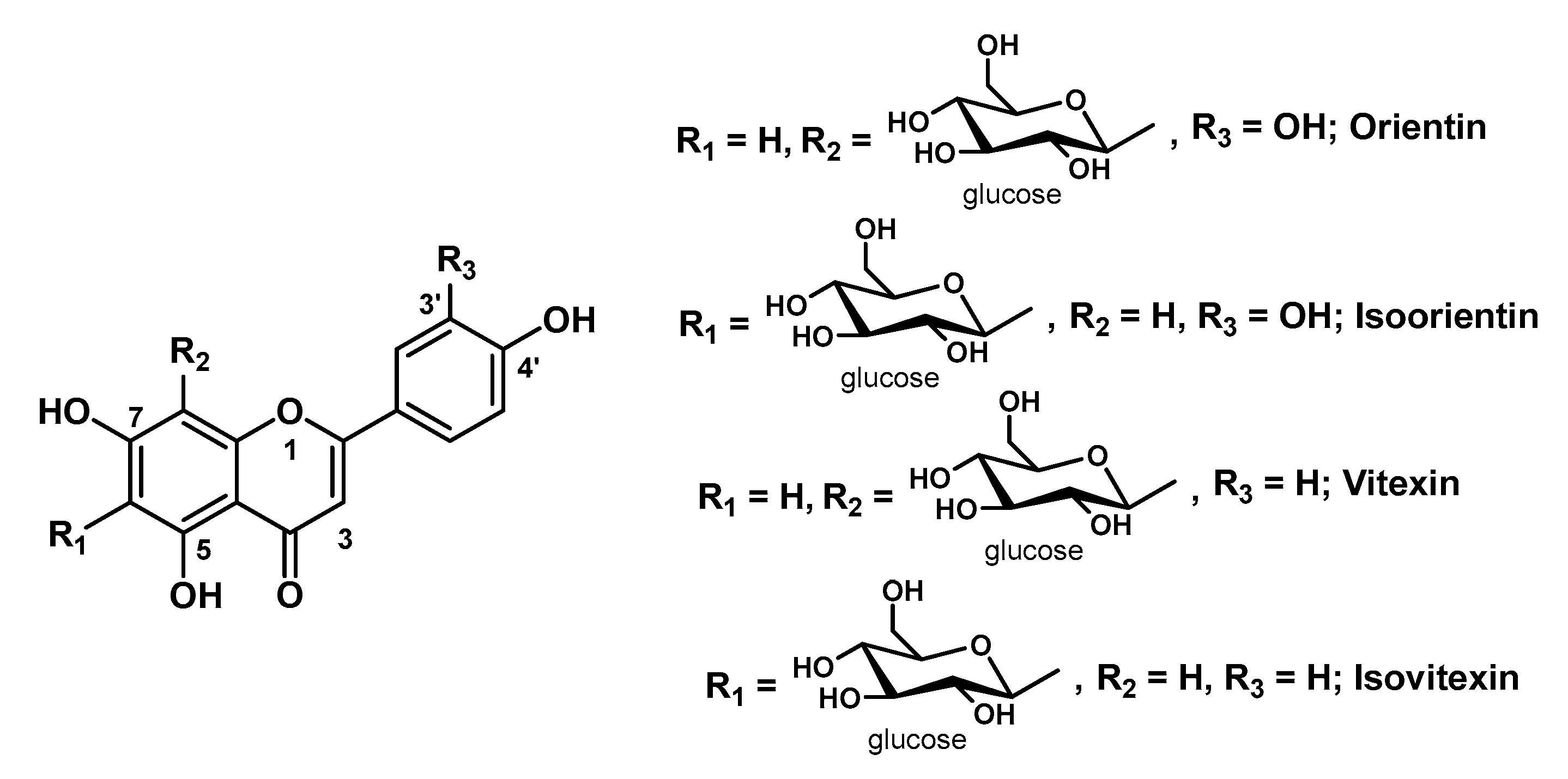
3.2. Hydrogels Containing Other Flavonoids
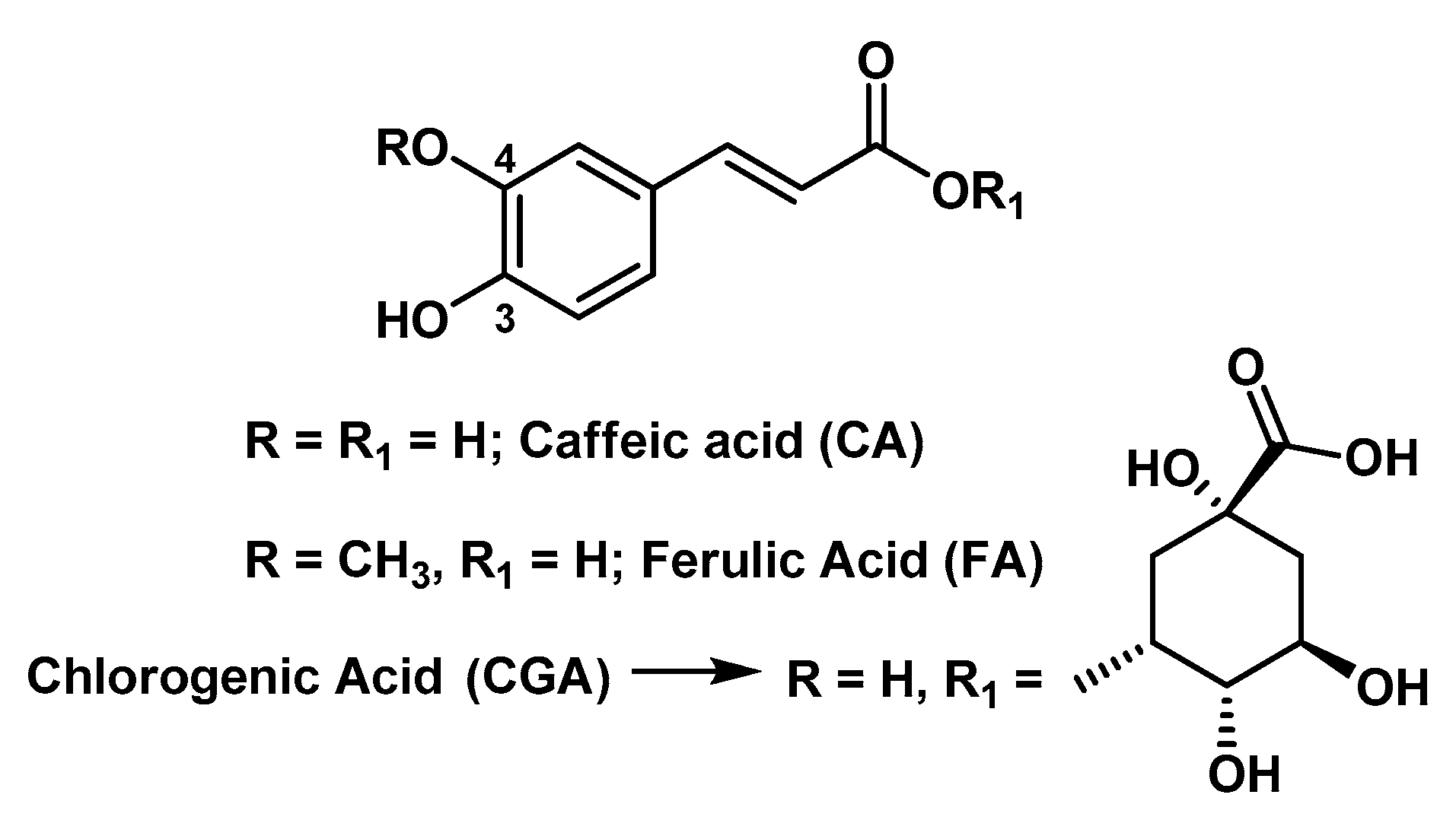
3.3. Hydrogels Containing Phenols with Trans-Cinnamic Acid Skeleton
3.4. Thymol-Containing Hydrogels
3.5. Plant Extract-Containing Hydrogels
4. Injectable Hydrogels
5. Hydrogels Projected for Oral and Systemic Administration.
5.1. Quercetin-Containing Hydrogels
5.2. Hydrogels Containing other (Poly)Phenols
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, Y.S.; Khademhosseini, A. Advances in engineering hydrogels. Science 2017, 356, 6337. [Google Scholar] [CrossRef] [PubMed]
- Chai, Q.; Jiao, Y.; Yu, X. Hydrogels for biomedical applications: Their characteristics and the mechanisms behind them. Gels 2017, 3, 6. [Google Scholar] [CrossRef]
- Ahmed, E.M. Hydrogel: Preparation, characterization and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Raghuwanshi, V.S.; Garnier, G. Characterisation of hydrogels: Linking the nano to the microscale. Adv. Colloid Interface Sci. 2019, 274, 102044. [Google Scholar] [CrossRef] [PubMed]
- Chyzy, A.; Tomczykowa, M.; Plonska-Brzezinska, M.E. Hydrogels as potential nano-, micro- and macro-scale systems for controlled drug delivery. Materials 2020, 13, 188. [Google Scholar] [CrossRef] [PubMed]
- Thoniyot, P.; Tan, M.J.; Karim, A.A.; Young, D.J.; Loh, X.J. Nanoparticle-hydrogel composites: Concept, design, and applications of these promising, multi-functional materials. Adv. Sci. 2015, 2, 1–13. [Google Scholar] [CrossRef]
- Narayanaswamy, R.; Torchilin, V.P. Hydrogels and their applications in targeted drug delivery. Molecules 2019, 24, 603. [Google Scholar] [CrossRef]
- Echeverria, C.; Fernandes, S.N.; Godinho, M.H.; Borges, J.P.; Soares, P.I.P. Functional stimuli-responsive gels: Hydrogels and microgels. Gels 2018, 4, 54. [Google Scholar] [CrossRef]
- Leri, M.; Scuto, M.; Ontario, M.L.; Calabrese, V.; Calabrese, E.J.; Bucciantini, M.; Stefani, M. Healthy effects of plant polyphenols: Molecular mechanisms. Int. J. Mol. Sci. 2020, 21, 1250. [Google Scholar] [CrossRef]
- Vitorino, C.; Sousa, J.; Pais, A. Overcoming the skin permeation barrier: Challenges and opportunities. Curr. Pharm. Des. 2015, 21, 2698–2712. [Google Scholar] [CrossRef]
- Flaten, G.E.; Palac, Z.; Engesland, A.; Filipović-Grčić, J.; Vanić, Ž.; Škalko-Basnet, N. In vitro skin models as a tool in optimization of drug formulation. Eur. J. Pharm. Sci. 2015, 75, 10–24. [Google Scholar] [CrossRef] [PubMed]
- Schwingel, L.C.; Bianchi, S.E.; Zorzi, G.K.; Gonçalves, P.; Teixeira, H.F.; Bassani, V.L. Quercetin and 3-O-methylquercetin in vitro skin layers permeation/retention from hydrogels: Why only a methoxy group difference determines different behaviors? J. Pharm. Pharmacol. 2019, 71, 733–745. [Google Scholar] [CrossRef] [PubMed]
- Park, S.N.; Lee, M.H.; Kim, S.J.; Yu, E.R. Preparation of quercetin and rutin-loaded ceramide liposomes and drug-releasing effect in liposome-in-hydrogel complex system. Biochem. Biophys. Res. Commun. 2013, 435, 361–366. [Google Scholar] [CrossRef]
- Park, S.H.; Shin, H.S.; Park, S.N. A novel pH-responsive hydrogel based on carboxymethyl cellulose/2-hydroxyethyl acrylate for transdermal delivery of naringenin. Carbohydr. Polym. 2018, 200, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kwon, S.S.; Jeon, S.H.; Yu, E.R.; Park, S.N. Enhanced skin delivery of liquiritigenin and liquiritin-loaded liposome-in-hydrogel complex system. Int. J. Cosmet. Sci. 2014, 36, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Lu, Y.; Li, P.Y.; Liu, C.M.; Wu, H.C.; Wen, R.; Du, S.Y. Development and in vitro evaluation of a transdermal hydrogel patch for ferulic acid. Pak. J. Pharm. Sci. 2014, 27, 369–375. [Google Scholar]
- Sorg, H.; Tilkorn, D.J.; Hager, S.; Hauser, J.; Mirastschijski, U. Skin wound healing: An update on the current knowledge and concepts. Eur. Surg. Res. 2017, 58, 81–94. [Google Scholar] [CrossRef]
- Patel, S.; Srivastava, S.; Singh, M.R.; Singh, D. Mechanistic insight into diabetic wounds: Pathogenesis, molecular targets and treatment strategies to pace wound healing. Biomed. Pharmacother. 2019, 112, 108615. [Google Scholar] [CrossRef]
- Grada, A.; Mervis, J.; Falanga, V. Research techniques made simple: Animal models of wound healing. J. Investig. Dermatol. 2018, 138, 2095–2105. [Google Scholar] [CrossRef]
- Gaspar-Pintiliescu, A.; Stanciuc, A.M.; Craciunescu, O. Natural composite dressings based on collagen, gelatin and plant bioactive compounds for wound healing: A review. Int. J. Biol. Macromol. 2019, 138, 854–865. [Google Scholar] [CrossRef]
- Vigani, B.; Rossi, S.; Sandri, G.; Bonferoni, M.C.; Caramella, C.M.; Ferrari, F. Hyaluronic acid and chitosan-based nanosystems: A new dressing generation for wound care. Expert. Opin. Drug Deliv. 2019, 16, 715–740. [Google Scholar] [CrossRef] [PubMed]
- Działo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The potential of plant phenolics in prevention and therapy of skin disorders. Int. J. Mol. Sci. 2016, 17, 160. [Google Scholar] [CrossRef] [PubMed]
- Hajialyani, M.; Tewari, D.; Sobarzo-Sánchez, E.; Nabavi, S.M.; Farzaei, M.H.; Abdollahi, M. Natural product-based nanomedicines for wound healing purposes: Therapeutic targets and drug delivery systems. Int. J. Nanomedicine 2018, 13, 5023–5043. [Google Scholar] [CrossRef] [PubMed]
- Almeida, J.S.; Benvegnú, D.M.; Boufleur, N.; Reckziegel, P.; Barcelos, R.C.; Coradini, K.; de Carvalho, L.M.; Bürger, M.E.; Beck, R.C. Hydrogels containing rutin intended for cutaneous administration: Efficacy in wound healing in rats. Drug Dev. Ind. Pharm. 2012, 38, 792–799. [Google Scholar] [CrossRef]
- Tran, N.Q.; Joung, Y.K.; Lih, E.; Park, K.D. In situ forming and rutin-releasing chitosan hydrogels as injectable dressings for dermal wound healing. Biomacromolecules 2011, 12, 2872–2880. [Google Scholar] [CrossRef]
- Lopes Affonso, R.C.; Lorenzen Voytena, A.P.; Fanan, S.; Pitz, H.; Sousa Coelho, D.; Horstmann, A.L.; Pereira, A.; Gavicho Uarrota, V.; Hillmann, M.C.; Calbusch Varela, L.A.; et al. Phytochemical composition, antioxidant activity, and the effect of the aqueous extract of coffee (Coffea arabica L.) bean residual press cake on the skin wound healing. Oxid. Med. Cell. Longev. 2016, 2016, 1923754. [Google Scholar]
- Jangde, R.; Srivastava, S.; Singh, M.R.; Singh, D. In vitro and In vivo characterization of quercetin loaded multiphase hydrogel for wound healing application. Int. J. Biol. Macromol. 2018, 115, 1211–1217. [Google Scholar] [CrossRef]
- Bairagi, U.; Mittal, P.; Singh, J.; Mishra, B. Preparation, characterization, and in vivo evaluation of nano formulations of ferulic acid in diabetic wound healing. Drug Dev. Ind. Pharm. 2018, 44, 1783–1796. [Google Scholar] [CrossRef]
- Ma, H.; Zhou, Q.; Chang, J.; Wu, C. Grape seed-inspired smart hydrogel scaffolds for melanoma therapy and wound healing. ACS Nano 2019, 13, 4302–4311. [Google Scholar] [CrossRef]
- Carmignan, F.; Matias, R.; Carollo, C.A.; Dourado, D.M.; Fermiano, M.H.; Silva, B.A.K.; Bastos, P.R.H.O. Efficacy of application of Equisetum pyramidale Goldm. hydrogel for tissue restoration of induced skin lesions in Wistar rats. Braz. J. Biol. 2020, 80, 12–22. [Google Scholar] [CrossRef]
- Soares, R.D.F.; Campos, M.G.N.; Ribeiro, G.P.; Salles, B.C.C.; Cardoso, N.S.; Ribeiro, J.R.; Souza, R.M.; Leme, K.C.; Soares, C.B.; de Oliveira, C.M.; et al. Development of a chitosan hydrogel containing flavonoids extracted from Passiflora edulis leaves and the evaluation of its antioxidant and wound healing properties for the treatment of skin lesions in diabetic mice. J. Biomed. Mater. Res. 2020, 108, 654–662. [Google Scholar] [CrossRef] [PubMed]
- Jee, J.-P.; Pangeni, R.; Jha, S.K.; Byun, Y.; Park, J.-W. Preparation and in vivo evaluation of a topical hydrogel system incorporating highly skin-permeable growth factors, quercetin, and oxygen carriers for enhanced diabetic wound-healing therapy. Int. J. Nanomed. 2019, 14, 5449–5475. [Google Scholar] [CrossRef] [PubMed]
- Gallelli, G.; Cione, E.; Serra, R.; Leo, A.; Citraro, R.; Matricardi, P.; Di Meo, C.; Bisceglia, F.; Caroleo, M.C.; Basile, S.; et al. Nano-hydrogel embedded with quercetin and oleic acid as a new formulation in the treatment of diabetic foot ulcer: A pilot study. Int. Wound J. 2020, 17, 485–490. [Google Scholar] [CrossRef]
- Jiji, S.; Udhayakumar, S.; Rose, C.; Muralidharan, C.; Kadirvelu, K. Thymol enriched bacterial cellulose hydrogel as effective material for third degree burn wound repair. Int. J. Biol. Macromol. 2019, 122, 452–460. [Google Scholar] [CrossRef]
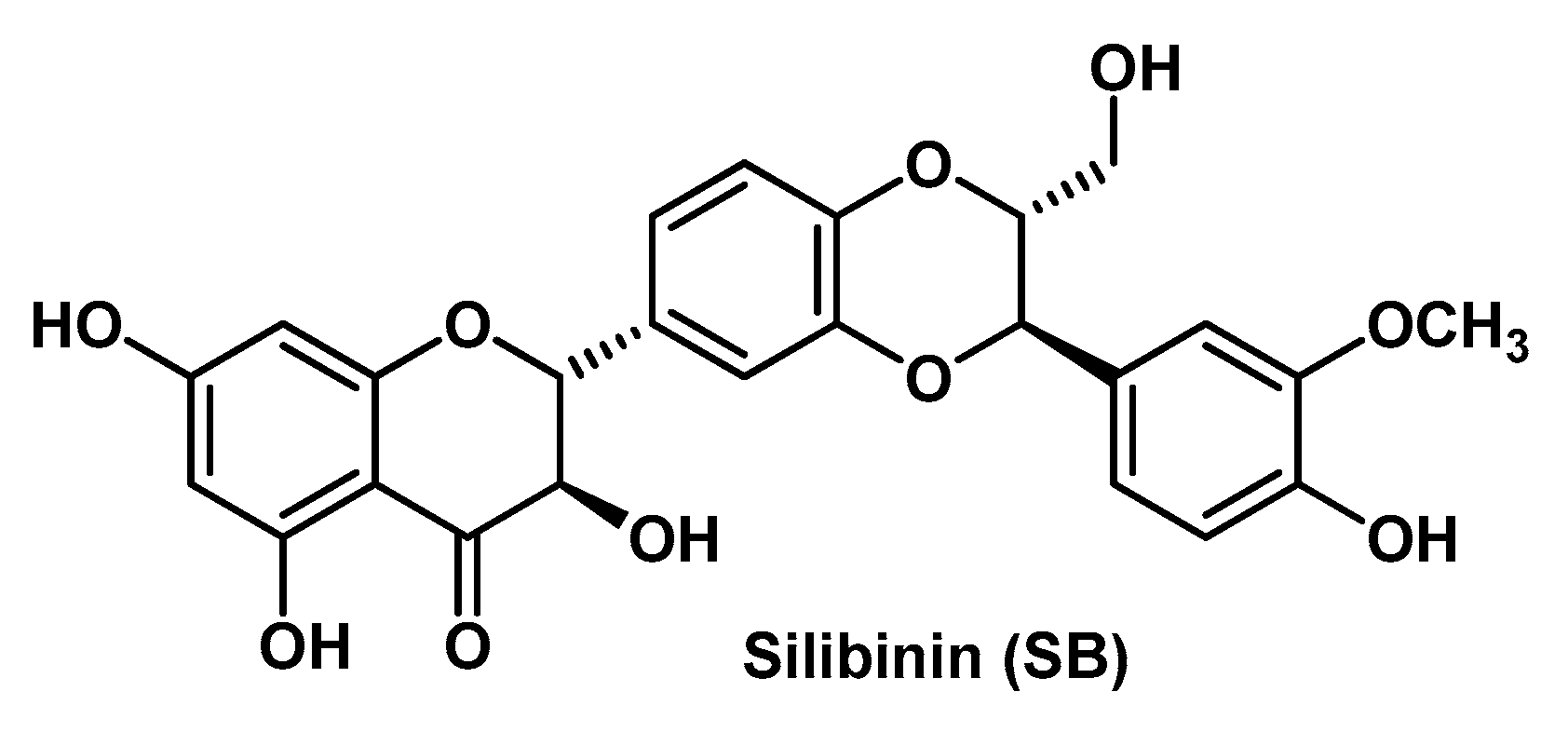
- Rigon, C.; Marchiori, M.C.L.; da Silva Jardim, F.; Pegoraro, N.S.; Chaves, P.D.S.; Velho, M.C.; Beck, R.C.R.; Ourique, A.F.; Sari, M.H.M.; Oliveira, S.M.; et al. Hydrogel containing silibinin nanocapsules presents effective anti-inflammatory action in a model of irritant contact dermatitis in mice. Eur. J. Pharm. Sci. 2019, 137, 104969. [Google Scholar] [CrossRef] [PubMed]
- Shrotriya, S.N.; Vidhate, B.V.; Shukla, M.S. Formulation and development of Sylibin loaded solid lipid nanoparticle enriched gel for irritant contact dermatitis. J. Drug Deliv. Sci. Technol. 2017, 41, 164–173. [Google Scholar] [CrossRef]
- Kim, A.R.; Lee, S.L.; Park, S.N. Properties and in vitro drug release of pH- and temperature-sensitive double cross-linked interpenetrating polymer network hydrogels based on hyaluronic acid/poly (N-isopropylacrylamide) for transdermal delivery of luteolin. Int. J. Biol. Macromol. 2018, 118, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Makhmalzadeh, B.S.; Molavi, O.; Vakili, M.R.; Zhang, H.F.; Solimani, A.; Abyaneh, H.S.; Loebenberg, R.; Lai, R.; Lavasanifar, A. Functionalized caprolactone-polyethylene glycol based thermo-responsive hydrogels of silibinin for the treatment of malignant melanoma. J. Pharm. Pharm. Sci. 2018, 21, 143–159. [Google Scholar] [CrossRef]
- Balestrin, L.A.; Bidone, J.; Bortolin, R.C.; Moresco, K.; Moreira, J.C.; Teixeira, H.F. Protective effect of a hydrogel containing Achyrocline satureioides extract-loaded nanoemulsion against UV-induced skin damage. J. Photochem. Photobiol. 2016, 163, 269–276. [Google Scholar] [CrossRef]
- Tsai, C.-Y.; Woung, L.-C.; Yen, J.-C.; Tseng, P.-C.; Chiou, S.-H.; Sung, Y.-J.; Liu, K.-T.; Cheng, Y.-H. Thermosensitive chitosan-based hydrogels for sustained release of ferulic acid on corneal wound healing. Carbohyd. Polym. 2016, 135, 308–315. [Google Scholar] [CrossRef]
- Pinho, E.; Henriques, M.; Soares, G. Caffeic acid loading wound dressing: Physicochemical and biological characterization. Ther. Deliv. 2014, 5, 1063–1075. [Google Scholar] [CrossRef] [PubMed]
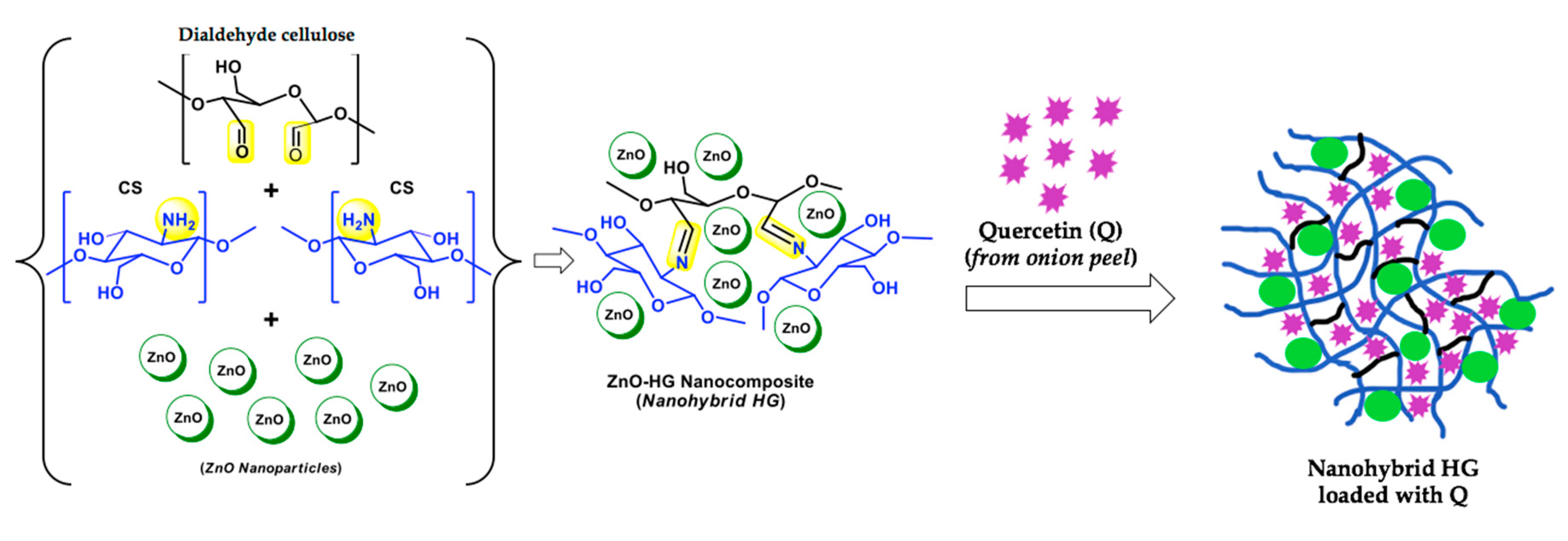
- George, D.; Maheswari, P.U.; Begum, K.M.M.S. Synergic formulation of onion peel quercetin loaded chitosan-cellulose hydrogel with green zinc oxide nanoparticles towards controlled release, biocompatibility, antimicrobial and anticancer activity. Int. J. Biol. Macromol. 2019, 132, 784–794. [Google Scholar] [CrossRef] [PubMed]
- Alvarez Echazú, M.I.; Olivetti, C.E.; Anesini, C.; Perez, C.J.; Alvarez, G.S.; Desimone, M.F. Development and evaluation of thymol-chitosan hydrogels with antimicrobial-antioxidant activity for oral local delivery. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 81, 588–596. [Google Scholar] [CrossRef]
- Moghadamnia, A.A.; Motallebnejad, M.; Khanian, M. The efficacy of the bioadhesive patches containing licorice extract in the management of recurrent aphthous stomatitis. Phytother. Res. 2009, 23, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Delparastan, P.; Burch, P.; Cheng, J.; Cao, Y.; Messersmith, P.B. Injectable dynamic covalent hydrogels of boronic acid polymers cross-linked by bioactive plant-derived polyphenols. Biomater. Sci. 2018, 6, 2487–2495. [Google Scholar] [CrossRef]
- Dong, G.-C.; Kuan, C.-Y.; Subramaniam, S.; Zhao, J.-Y.; Sivasubramaniam, S.; Chang, H.-Y.; Lin, F.-H. A potent inhibition of oxidative stress induced gene expression in neural cells by sustained ferulic acid release from chitosan based hydrogel. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 49, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.-H.; Yang, S.-H.; Liu, C.-C.; Gefen, A.; Lin, F.-H. Thermosensitive hydrogel made of ferulic acid-gelatin and chitosan glycerophosphate. Carbohyd. Polym. 2013, 92, 1512–1519. [Google Scholar] [CrossRef]
- Mok, S.-W.; Fu, S.-C.; Cheuk, Y.-C.; Chu, I.-M.; Chan, K.-M.; Qin, L.; Yung, S.-H.; Kevin Ho, K.-W. Intra-articular delivery of quercetin using thermosensitive hydrogel attenuate cartilage degradation in an osteoarthritis rat model. Cartilage 2018. [Google Scholar] [CrossRef]
- Cheng, Y.-H.; Lin, F.-H.; Wang, C.-Y.; Hsiao, C.-Y.; Chen, H.-C.; Kuo, H.-Y.; Tsai, T.-F.; Chiou, S.-H. Recovery of oxidative stress-induced damage in Cisd2-deficient cardiomyocytes by sustained release of ferulic acid from injectable hydrogel. Biomaterials 2016, 103, 207–218. [Google Scholar] [CrossRef]
- Barbarisi, M.; Iaffaioli, R.V.; Armenia, E.; Schiavo, L.; De Sena, G.; Tafuto, S.; Barbarisi, A.; Quagliariello, V. Novel nanohydrogel of hyaluronic acid loaded with quercetin alone and in combination with temozolomide as new therapeutic tool, CD44 targeted based, of glioblastoma multiforme. J. Cell. Physiol. 2018, 233, 6550–6564. [Google Scholar] [CrossRef]
- Quagliariello, V.; Iaffaioli, R.V.; Armenia, E.; Clemente, O.; Barbarisi, M.; Nasti, G.; Berretta, M.; Ottaiano, A.; Barbarisi, A. Hyaluronic acid nanohydrogel loaded with quercetin alone or in combination to a macrolide derivative of Rapamycin RAD001 (Everolimus) as a new treatment for hormone-responsive human breast cancer. J. Cell. Physiol. 2017, 232, 2063–2074. [Google Scholar] [CrossRef]
- Quagliariello, V.; Armenia, E.; Aurilio, C.; Rosso, F.; Clemente, O.; de Sena, G.; Barbarisi, M.; Barbarisi, A. New treatment of medullary and papillary human thyroid cancer: Biological effects of hyaluronic acid hydrogel loaded with quercetin alone or in combination to an inhibitor of Aurora Kinase. J. Cell. Physiol. 2016, 231, 1784–1795. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wang, Z. A pH-sensitive microemulsion-filled gellan gum hydrogel encapsulated apigenin: Characterization and in vitro release kinetics. Colloids Surf. B Biointerfaces 2019, 178, 245–252. [Google Scholar] [CrossRef]
- Hu, X.; Wang, Y.; Zhang, L.; Xu, M. Construction of self-assembled polyelectrolyte complex hydrogel based on oppositely charged polysaccharides for sustained delivery of green tea polyphenols. Food Chem. 2020, 306, 125632. [Google Scholar] [CrossRef] [PubMed]
- Abdel Ghaffar, A.M.; Radwan, R.R.; Ali, H.E. Radiation synthesis of poly(starch/acrylic acid) pH sensitive hydrogel for rutin controlled release. Int. J. Biol. Macromol. 2016, 92, 957–964. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Shen, Y.; Adamcik, J.; Fischer, P.; Schneider, M.; Loessner, M.J.; Mezzenga, R. Polyphenol-binding amyloid fibrils self-assemble into reversible hydrogels with antibacterial activity. ACS Nano 2018, 12, 3385–3396. [Google Scholar] [CrossRef]
- Kunjiappan, S.; Theivendran, P.; Baskararaj, S.; Sankaranarayanan, B.; Palanisamy, P.; Saravanan, G.; Arunachalam, S.; Sankaranarayanan, M.; Natarajan, J.; Somasundaram, B.; et al. Modeling a pH-sensitive Zein-co-acrylic acid hybrid hydrogels loaded 5-fluorouracil and rutin for enhanced anticancer efficacy by oral delivery. 3 Biotech 2019, 9, 185. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Dong, X.; Yan, X.; Liu, Y.; Zhang, L.; Sun, Y. Nanogels of dual inhibitor-modified hyaluronic acid function as a potent inhibitor of amyloid β-protein aggregation and cytotoxicity. Sci. Rep. 2018, 8, 3505. [Google Scholar] [CrossRef]
- Shi, C.; Wang, C.; Liu, H.; Li, Q.; Li, R.; Zhang, Y.; Liu, Y.; Shao, Y.; Wang, J. Selection of appropriate wound dressing for various wounds. Front. Bioeng. Biotechnol. 2020, 8, 182. [Google Scholar] [CrossRef]
- Liu, W.-C.; Wang, H.-Y.; Lee, T.-H.; Chung, R.-J. Gamma-poly glutamate/gelatin composite hydrogels crosslinked by proanthocyanidins for wound healing. Mater. Sci. Eng. C 2019, 101, 630–639. [Google Scholar] [CrossRef]
- Zack, B.; Arrandale, V.; Holness, D.L. Skin-specific training experience of workers assessed for contact dermatitis. Occup. Med. 2018, 68, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Szekalska, M.; Sosnowska, K.; Tomczykowa, M.; Winnicka, K.; Kasacka, I.; Tomczyk, M. In vivo anti-inflammatory and anti-allergic activities of cynaroside evaluated by using hydrogel formulations. Biomed. Pharmacother. 2020, 121, 109681. [Google Scholar] [CrossRef] [PubMed]
- Marchiori, M.C.L.; Rigon, C.; Camponogara, C.; Oliveira, S.M.; Cruz, L. Hydrogel containing silibinin-loaded pomegranate oil based nanocapsules exhibits anti-inflammatory effects on skin damage UVB radiation-induced in mice. J. Photochem. Photobiol. 2017, 170, 25–32. [Google Scholar] [CrossRef]
- Spizzirri, U.G.; Cirillo, G.; Curcio, M.; Picci, N.; Iemma, F. Flavonoid-based pH-responsive hydrogels as carrier of unstable drugs in oxidative conditions. Pharm. Dev. Technol. 2015, 20, 288–296. [Google Scholar] [CrossRef]
- Barrientos, S.; Stojadinovic, O.; Golinko, M.S.; Brem, H.; Tomic-Canic, M. Growth factors and cytokines in wound healing. Wound Repair Regen. 2008, 16, 585–601. [Google Scholar] [CrossRef]
- Sen, C.K. Wound healing essentials: Let there be oxygen. Wound Repair Regen. 2019, 17, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, C.; Li, C.; Qin, Y.; wang, Z.; Yang, F.; Li, Z.; Wang, J. A functional chitosan-based hydrogel as a wound dressing and drug delivery system in the treatment of wound healing. RSC Adv. 2018, 8, 7533–7549. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Khan, I.A.; ur-Rheman, M.; Gilani, S.A.; Mehmood, Z.; Mubarak, M.S. Anticancer potential of quercetin: A comprehensive review. Phyther. Res. 2018, 32, 2109–2130. [Google Scholar] [CrossRef]
- Haimhoffer, Á.; Rusznyák, Á.; Réti-Nagy, K.; Vasvári, G.; Váradi, J.; Vecsernyés, M.; Báckskay, I.; Fehér, P.; Ujhelyi, Z.; Fenyvesi, F. Cyclodextrins in drug delivery systems and their effects on biological barriers. Sci. Pharm. 2019, 87, 33. [Google Scholar] [CrossRef]
- Varghese, J.S.; Chellappa, N.; Fathima, N.N. Gelatin-carrageenan hydrogels: Role of pore size distribution on drug delivery process. Colloids Surf. B Biointerfaces 2014, 113, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Rattanaruengsrikul, V.; Pimpha, N.; Supaphol, P. In vitro efficacy and toxicology evaluation of silver nanoparticle-loaded gelatin hydrogel pads as antibacterial wound dressings. J. Appl. Polym. Sci. 2012, 124, 1668–1682. [Google Scholar] [CrossRef]
- Yu, Y.; Feng, R.; Yu, S.; Li, J.; Wang, Y.; Song, Y.; Yang, X.; Pan, W.; Li, S. Nanostructured lipid carrier-based pH and temperature dual-responsive hydrogel composed of carboxymethyl chitosan and poloxamer for drug delivery. Int. J. Biol. Macromol. 2018, 114, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Wattamwar, P.P.; Biswal, D.; Cochran, D.B.; Lyvers, A.C.; Eitel, R.E.; Anderson, K.W.; Hilt, J.Z.; Dziubla, T.D. Synthesis and characterization of poly(antioxidant β-amino esters) for controlled release of polyphenolic antioxidants. Acta Biomater. 2012, 8, 2529–2537. [Google Scholar] [CrossRef]
- Tang, S.; Bhandari, R.; Delaney, S.P.; Munson, E.J.; Dziubla, T.D.; Hilt, J.Z. Synthesis and characterization of thermally responsive N-isopropylacrylamide hydrogels copolymerized with novel hydrophobic polyphenolic crosslinkers. Mater. Today Commun. 2017, 10, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kwon, S.S.; Yu, E.R.; Park, S.N. Development od porous cellulose hydrogel for enhanced transdermal delivery of liquiritin and liquitigenin as locorice flavoniods. Polym. Korea 2014, 38, 676–681. [Google Scholar] [CrossRef][Green Version]
- Weng, Z.; Pate, A.B.; Vasiadi, M.; Therianou, A.; Theoharides, T.C. Luteon inhibits human keratinocyte activation and decreases NF-κB induction that is increased in psoriatic skin. PLoS ONE 2014, 9, 90739. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.R.; Seo, H.S.; Ku, J.M.; Kim, G.J.; Jeon, C.Y.; Park, J.H.; Jang, B.H.; Park, S.J.; Shin, Y.C.; Ko, S.G. Silibinin inhibits the production of pro-inflammatory cytokines through inhibition of NF-κB signaling pathway in HMC-1 mast cells. Inflam. Res. 2013, 62, 941–950. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.; Bhatia, D.; Bishayee, A. Anti-inflammatory mechanism involved in pomegranate-mediated prevention of breast cancer: The role of NF-κB and Nrf2 signaling pathways. Nutrients 2017, 9, 436. [Google Scholar] [CrossRef] [PubMed]
- Marchiori, M.C.L.; Rigon, C.; Copetti, P.M.; Sagrillo, M.R.; Cruz, L. Nanoencapsulation improves scavenging capacity and decreases cytotoxicity of silibinin and pomegranate oil association. AAPS PharmSciTech 2017, 18, 3236–3246. [Google Scholar] [CrossRef]
- Bosch-Barrera, J.; Menendez, J.A. Silibinin and STAT3: A natural way of targeting transcription factors for cancer therapy. Cancer Treat. Rev. 2015, 41, 540–546. [Google Scholar] [CrossRef]
- Ghosh, S.; Basak, P.; Dutta, S.; Chowdhury, S.; Sil, P.C. New insights into the ameliorative effects of ferulic acid in pathophysiological conditions. Food Chem. Toxicol. 2017, 103, 41–55. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Jain, G.K.; Javed, S.; Iqbal, Z.; Talegaonkar, S.; Ahmad, F.J.; Khar, R.K. Recent approaches for the treatment of periodontitis. Drug Discov. Today 2008, 13, 932–943. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.; Selmin, F.; Baldassari, S.; Gennari, C.G.M.; Caviglioli, G.; Cilurzo, F.; Minghetti, P.; Parodi, B. A focus on mucoadhesive polymers and their application in buccal dosage forms. J. Drug Deliv. Sci. Technol. 2016, 32, 113–125. [Google Scholar] [CrossRef]
- Hosseinnejad, M.; Jafari, S.M. Evaluation of different factors affecting antimicrobial properties of chitosan. Int. J. Biol. Macromol. 2016, 85, 467–475. [Google Scholar] [CrossRef]
- Alvarez Echazú, M.I.; Tuttolomondo, M.V.; Foglia, M.L.; Mebert, A.M.; Alvarez, G.S.; Desimone, M.F. Advances in collagen, chitosan and silica biomaterials for oral tissue regeneration: From basics to clinical trials. J. Mater. Chem. B 2016, 4, 6913–6929. [Google Scholar] [CrossRef] [PubMed]
- Retta, D.; Dellacassa, E.; Villamil, J.; Suárez, S.A.; Bandoni, A.L. A. satureioides, a promising medicinal and aromatic plant from Latin America: A review. Ind. Crop. Prod. 2012, 38, 27–38. [Google Scholar] [CrossRef]
- Choi, Y.; Kim, H.; Min, K. Effects of proanthocyanidin, a cross.linking agent, on physical and biological properties of collagen hydrogel scaffold. Restor. Dent. Endod. 2016, 41, 296–303. [Google Scholar] [CrossRef]
- Wu, C.-Y.; Chen, Y.-F.; Wang, C.-H.; Kao, C.-H.; Zhuang, H.-W.; Chen, C.-C.; Chen, L.-K.; Kirby, R.; Wei, Y.-H.; Tsai, S.-F.; et al. A persistent level of Cisd2 extends healthy lifespan and delays aging in mice. Hum. Mol. Genet. 2012, 21, 3956–3968. [Google Scholar] [CrossRef]
- Cheng, Y.-H.; Yang, S.-H.; Lin, F.-H. Thermosensitive chitosan-gelatin-glycerol phosphate hydrogel as a controlled release system of ferulic acid for nucleus pulposus regeneration. Biomaterials 2011, 32, 6953–6961. [Google Scholar] [CrossRef]
- Walker, M.H.; Anderson, D.G. Molecular basis of intervertebral disc degeneration. Spine J. 2004, 4, 158–166. [Google Scholar] [CrossRef]
- Na, J.Y.; Song, K.; Kim, S.; Kwon, J. Rutin protects rat articular chondrocytes against oxidative stress induced by hydrogen peroxide through SIRT1 activation. Biochem. Biophys. Res. Commun. 2016, 473, 1301–1308. [Google Scholar] [CrossRef]
- Pan, F.; Zhu, L.; Lv, H.; Pei, C. Quercetin promotes the apoptosis of fibroblast-like synoviocytes in rheumatoid arthritis by upregulating lncRNA MALAT1. Int. J. Mol. Med. 2016, 38, 1507–1514. [Google Scholar] [CrossRef]
- Sharpe, L.A.; Daily, A.M.; Horava, S.D.; Peppas, N.A. Therapeutic applications of hydrogels in oral drug delivery. Expert Opin. Drug Deliv. 2014, 11, 901–915. [Google Scholar] [CrossRef]
- Chen, C.; Zhao, S.; Karned, A.; Freeman, J.W. The biology and role of CD44 in cancer progression: Therapeutic implications. J. Hematol. Oncol. 2018, 11, 64. [Google Scholar] [CrossRef]
- Gavriilidis, P.; Giakoustidis, A.; Giakoustidis, D. Aurora kinases and potential medical applications of aurora: A review. J. Clin. Med. Res. 2015, 7, 742–751. [Google Scholar] [CrossRef]
- Wang, H.-H.; Liao, C.-C.; Chow, N.-H.; Huang, L.-L.; Chuang, J.-I.; Wei, K.-C.; Shin, J.-W. Whether CD44 is an applicable marker for glioma stem cells. Am. J. Transl. Res. 2017, 9, 4785–4806. [Google Scholar] [PubMed]
- Doosti, M.; Dorraji, M.S.S.; Mousavi, S.N.; Rasoulifard, M.H.; Hosseini, S.H. Enhancing quercetin bioavailability by super paramagnetic starch-based hydrogel grafted with fumaric acid: An in vitro and in vivo study. Colloids Surf. B Biointerfaces 2019, 183, 110487. [Google Scholar] [CrossRef] [PubMed]
- Andrich, K.; Bieschke, J. The Effect of (−)-Epigallo-catechin-(3)-gallate on Amyloidogenic Proteins Suggests a Common Mechanism. Adv. Exp. Med. Biol. 2015, 863, 139–161. [Google Scholar]
- Reddy, P.H.; Manczak, M.; Yin, X.; Grady, M.C.; Mitchell, A.; Tonk, S.; Kuruva, C.S.; Bhatti, J.S.; Kandimalla, R.; Vijayan, M.; et al. Protective effects of indian spice curcumin against amyloid-β in Alzheimer’s disease. J. Alzheimers Dis. 2018, 61, 843–866. [Google Scholar] [CrossRef] [PubMed]
- Palhano, F.L.; Lee, J.; Grimster, N.P.; Kelly, J.W. Toward the molecular mechanism(s) by which EGCG treatment remodels mature amyloid fibrils. J. Am. Chem. Soc. 2013, 135, 7503–7510. [Google Scholar] [CrossRef]
- Thapa, A.; Jett, S.D.; Chi, E.Y. Curcumin attenuates amyloid-β aggregate toxicity and modulates amyloid-β aggregation pathway. ACS Chem. Neurosci. 2015, 7, 56–68. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Wei, W.; Shen, J.; Dong, W. Salecan polysaccharide-based hydrogels and their applications: A review. J. Mater. Chem. B 2019, 7, 2577–2587. [Google Scholar] [CrossRef] [PubMed]

| Compound | System | Disease | Experimental Model | Reference | |
|---|---|---|---|---|---|
| In Vitro and Ex Vivo Models | In Vivo Models | ||||
| Skin Wound Healing | |||||
| RU | Carbopol Ultrez® 10 NF HG | Skin wound healing | Skin wounds in Wistar rats | [24] | |
| RU | CS-PEG-TY in presence of HRP and H2O2 | Injectable wound dressing for skin wounds | Biocompatibility on L929 mouse fibroblasts | Skin wounds in Sprague Dawley rats | [25] |
| CGA | Carbopol 940 HG | Skin wound healing | Biocompatibility on L929 mouse fibroblasts | Skin wounds in Swiss mice | [26] |
| Q | Carbopol HG | Skin wound healing | Skin wounds in Sprague Dawley rats | [27] | |
| FA | Carbopol 980 HG | Diabetic wound healing | Skin wounds in streptozotocin-diabetic Wistar rats | [28] | |
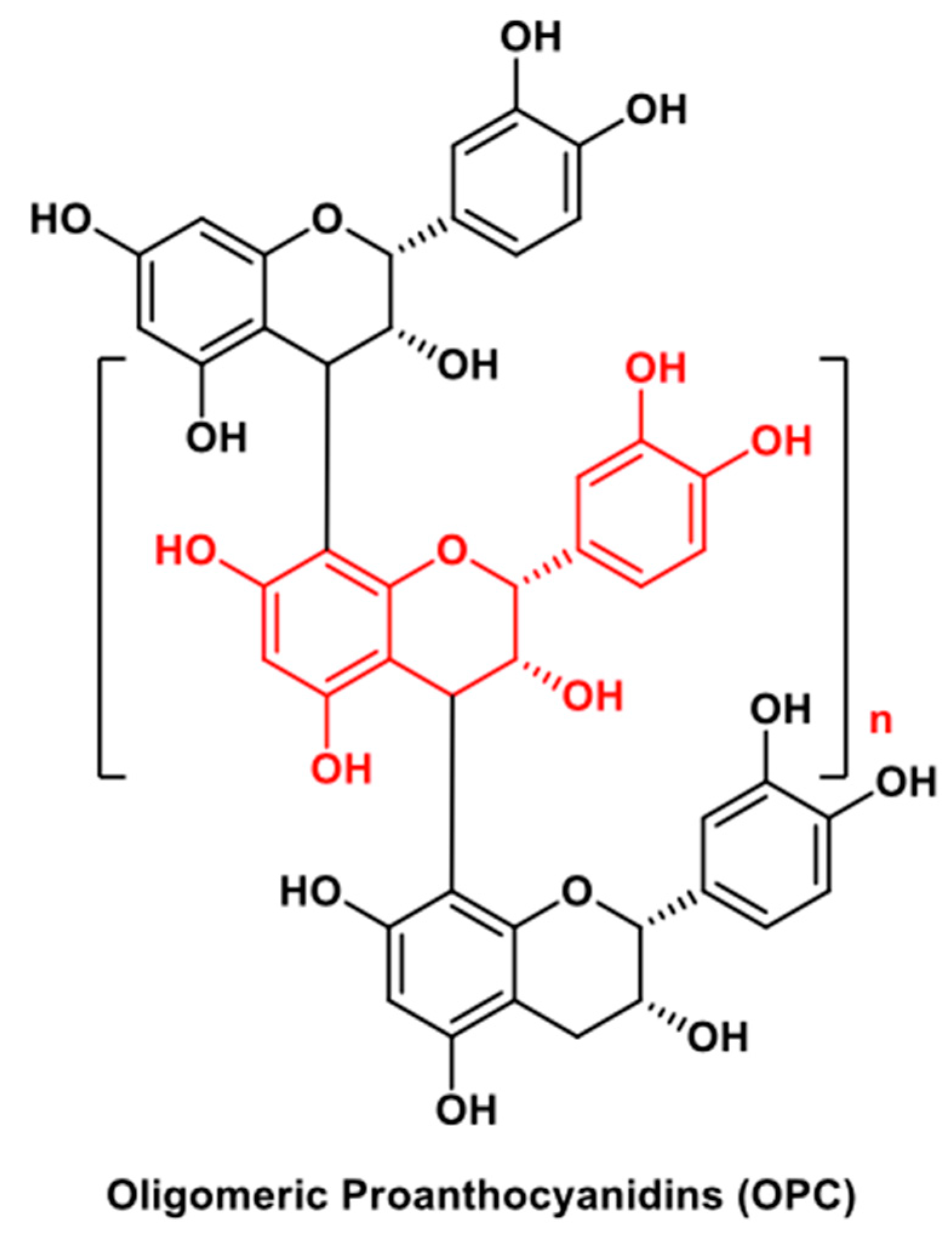
| OPC | Light-responsive collagen-based HG | Wound healing | Regenerative capability on HUVECs and human dermal fibroblasts (HDFs) | Skin wounds in streptozocin-diabetic C57BL/6 mice | [29] |
| Equisetum pyramidales hoots extracts | Carbopol HG | Wound healing | Skin wounds in Wistar rats | [30] | |
| Passiflora edulis Sims leaves extracts | CS-HG | Wound healing | Biocompatibility on L929 mouse fibroblasts | Skin wounds in alloxan- diabetic Wistar rats | [31] |
| Q, GFs and 1-bromoperfluorooctane | Carbopol 981 HG | Diabetic wound healing | Biocompatibility on HaCaT human keratinocytes and CCD-986sk human fibroblasts | Skin wounds in streptozotocin-diabetic C57BL/6 mice | [32] |
| Q and OA | HA-based nano-HG | Diabetic wound healing | Patients with diabetic foot ulcers | [33] | |
| Thymol | BCT-HG | Wound healing | Antimicrobial activity against Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, Klebsiella pneumoniae; Biocompatibility on NIH 3T3 mouse fibroblasts | Wistar rats bearing third degree burn wounds | [34] |
| Skin Permeation | |||||
| Q and RU | Ceramide liposomes incorporated into cellulose HG using ECH as cross-linking agent | Improvement of drug skin permeation | Skin permeation study on ICR hairy albino mouse skin | [13] | |
| NR | pH-responsive cl-CMC-g-pHEA | Improvement of drug skin permeation | Biocompatibility on HaCaT cells; Skin permeation study on hairless mouse skin | [14] | |
| LQG and LQ | Ceramide Liposomes incorporated into cellulose HG | Improvement of drug skin permeation | Skin permeation study on dorsal skin of ICR hairy albino mice | [15] | |
| FA | NP700 HG | Improvement of drug skin permeation | Skin permeation test on hairless mouse skin | [16] | |
| SB and pomegranate oil | Pemulen® TR2 HG system containing pomegranate oil-based nanocapsules | Improvement of drug skin permeation | Skin permeation study on human skin | [35] | |
| SB | Carbopol 940 HG | Improvement of drug skin permeation | Skin diffusion study on human cadaver skin | [36] | |
| LT | pH and temperature sensitive poly (N-isopropylacrylamide)/HA-based HG | Improvement of drug skin permeation | Skin permeation study on micropig dorsal skin | [37] | |
| SB | Thermo-responsive hydrogels based on triblock co-polymers (PolyGelTM) | Improvement of drug skin permeation | Skin permeation study on mouse skin | [38] | |
| Epithelial Pathologies | |||||
| SB and pomegranate oil | Pemulen® TR2 HG system containing pomegranate oil-based nanocapsules | Irritant contact dermatitis | Croton oil-induced ear edema in Swiss mice; Cutaneous safety in humans | [35] | |
| SB | Carbopol 940 HG | Irritant contact dermatitis | Skin irritancy in White New Zealand rabbits; Dinitrochlorobenzene - induced ICD in BALB/c mice | [36] | |
| Achyrocline satureioides inflorescences extracts | Carbopol® Ultrez 20 HG | UVA/UVB radiation-induced skin damage | UVA/UVB light-induced oxidative stress on porcine ears skin | [39] | |
| FA | Thermosensitive CS/G/GP-HG | Corneal wound healing | H2O2-induced oxidative stress in rabbit corneal epithelial CCL-60 cells | Corneal alkali burn in New Zealand albino rabbits | [40] |
| LT | pH and temperature sensitive poly (N-isopropylacrylamide)/HA-based HG | Psoriasis | Cytotoxicity assays on HaCaT human keratinocytes | [37] | |
| CA | Cyclodextrin-based HG | Epithelial infections | Antibacterial activity against Staphylococcus epidermidis, Staphylococcus aureus and Klebsiella pneumonia; Proliferation study on NIH 3T3 mouse fibroblasts | [41] | |
| Q | CHGZ-HGs | Skin and epithelial infections | Activity against Staphylococcus aureus and Trichophyton rubrum; Biocompatibility on L929 murine fibroblasts | [42] | |
| Thymol | CS-HG | Periodontal diseases | Biocompatibility on NIH 3T3 mouse fibroblasts Antibacterial activity against Streptococcus mutans and Staphylococcus aureus | [43] | |
| Licorice (Glycyrrhiza glabra) extracts | Tragacanth gum based HG | Recurrent aphthous stomatitis | Patients with aphthous ulcers | [44] | |
| Epithelial Cancer | |||||
| OPC | Light-responsive collagen-based HG | Melanoma | B16F10 murine melanoma cells | Photothermal therapy in Balb/c mice bearing B16F10 cell tumor | [29] |
| Q | CHGZ-HGs | Skin cancer | Human skin carcinoma A431 cells | [42] | |
| SB (also in combination with doxorubicin) | Thermo-responsive HG based on triblock co-polymers (PolyGelTM) | Melanoma | B16-F10 murine melanoma cells | [38] | |
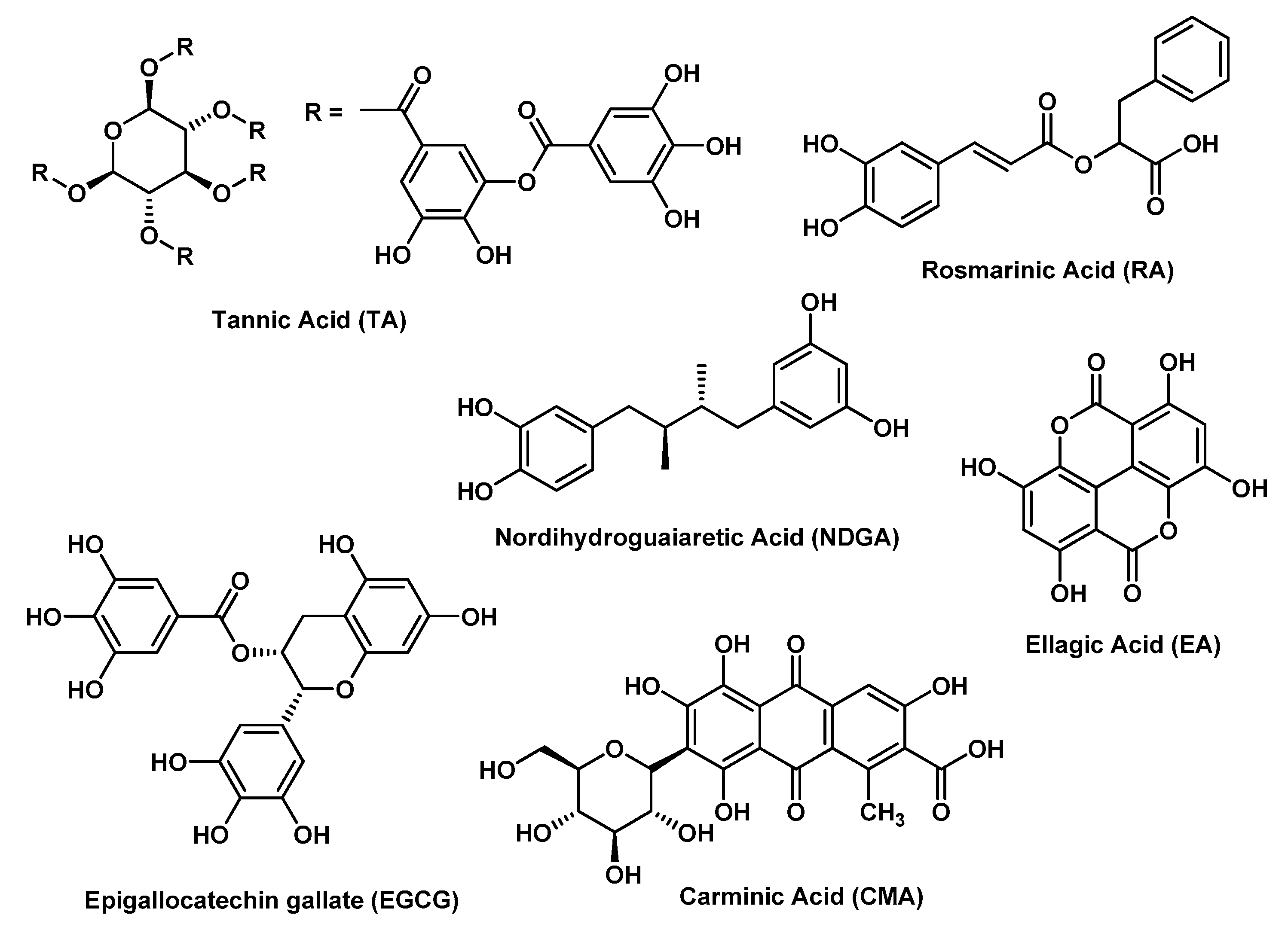
| EA, EGCG, TA, NDGA, RT, RA, and CMA | PEG-HG | Oral cancer | Human oral cancer CAL-27 cells | [45] | |
| Injectable and Targeted Hydrogels | |||||
| FA | Thermosensitive CS/G/GP-HG | Secondary brain injury | H2O2-induced oxidative stress in Neuro-2a cells | [46] | |
| FA | Thermosensitive CS/G/GP-HG | Intervertebral disc degeneration | H2O2-induced oxidative stress in nucleus pulposus cells from New Zealand rabbits | [47] | |
| Q | Thermosensitive hydrogel based on mPEG-PA-HG | Osteoarthritis | Human chondrocytes from patients undergone knee arthroplasty | Rats undergone anterior cruciate ligament transection | [48] |
| FA | Thermosensitive CS/G/HG | Cardiovascular diseases | Cisd2-deficient (Cisd2−/−) cardiomyocytes and cardiac tissue of Cisd2 knockout mice | Biocompatibility in subcutaneously injected New Zealand albino rabbits and intramyocardially injected Cisd2 deficient (Cisd2−/−) and wild-type (Cisd2+/+)] rats | [49] |
| Q (also in combination with temozolide) | HA-HG | Glioblastoma multiforme | Human glioblastoma A172 and T98MG cells | [50] | |
| Q (also in combination with everolimus) | HA-HG | Hormone-responsive human breast cancer | Human breast cancer MCF7 cells | [51] | |
| Q (also in combination with SNS-314) | HA-HG | Medullary and papillary human thyroid cancer | Human medullary and papillary cancer thyroid B-CPAP and TT cells; Biocompatibility on NIH 3T3 mouse fibroblasts | [52] | |
| Hydrogels for Oral and Systemic Administration | |||||
| APG | pH-sensitive gellan gum HGs | Oral delivery system for the controlled release of hydrophobic molecules | Drug release in different pH environments | [53] | |
| GTP | Poly electrolyte complex HG | Oral delivery | Media simulating the gastric and the intestinal tracts | [54] | |
| RU | pH-sensitive poly(starch/acrylic acid) HG | Ulcerative colitis | Dextran sulphate sodium-induced colitis in Wistar rats | [55] | |
| EGCG | Polyphenol-binding amyloid fibrils self-assemble into reversible HGs | Infection of small intestine | Antibacterial activity against Listeria monocytogenes, methicillin resistant Staphylococcus aureus, Streptococcus oralis, Escherichia coli, Klebsiella pneumoniae and Pseudomonas aeruginosa; Biocompatibility on human colonic epithelial cells | [56] | |
| RU and 5-FU | pH-responsive Zein-co-acrylic acid HGs | Anticancer activity | Breast cancer MDA-MB-231and MCF-7 cells | [57] | |
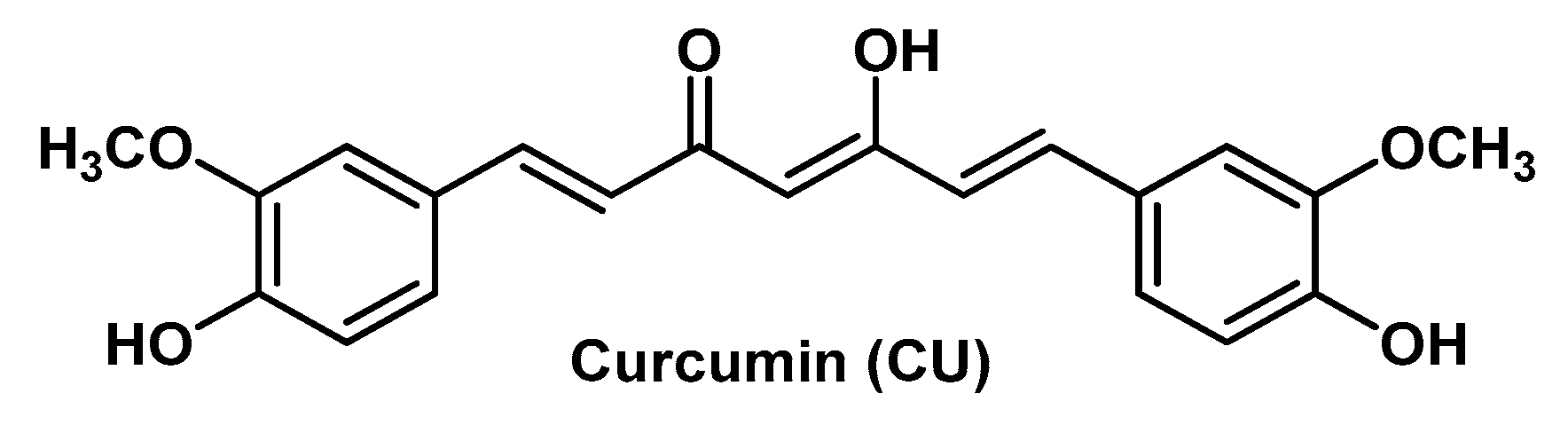
| EGCG and CU | HA-HG | Alzheimer’s disease | Human neuroblastoma SH-SY5Y cells | [58] | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Micale, N.; Citarella, A.; Molonia, M.S.; Speciale, A.; Cimino, F.; Saija, A.; Cristani, M. Hydrogels for the Delivery of Plant-Derived (Poly)Phenols. Molecules 2020, 25, 3254. https://doi.org/10.3390/molecules25143254
Micale N, Citarella A, Molonia MS, Speciale A, Cimino F, Saija A, Cristani M. Hydrogels for the Delivery of Plant-Derived (Poly)Phenols. Molecules. 2020; 25(14):3254. https://doi.org/10.3390/molecules25143254
Chicago/Turabian StyleMicale, Nicola, Andrea Citarella, Maria Sofia Molonia, Antonio Speciale, Francesco Cimino, Antonella Saija, and Mariateresa Cristani. 2020. "Hydrogels for the Delivery of Plant-Derived (Poly)Phenols" Molecules 25, no. 14: 3254. https://doi.org/10.3390/molecules25143254
APA StyleMicale, N., Citarella, A., Molonia, M. S., Speciale, A., Cimino, F., Saija, A., & Cristani, M. (2020). Hydrogels for the Delivery of Plant-Derived (Poly)Phenols. Molecules, 25(14), 3254. https://doi.org/10.3390/molecules25143254











