Hydration and Barrier Potential of Cosmetic Matrices with Bee Products
Abstract
1. Introduction
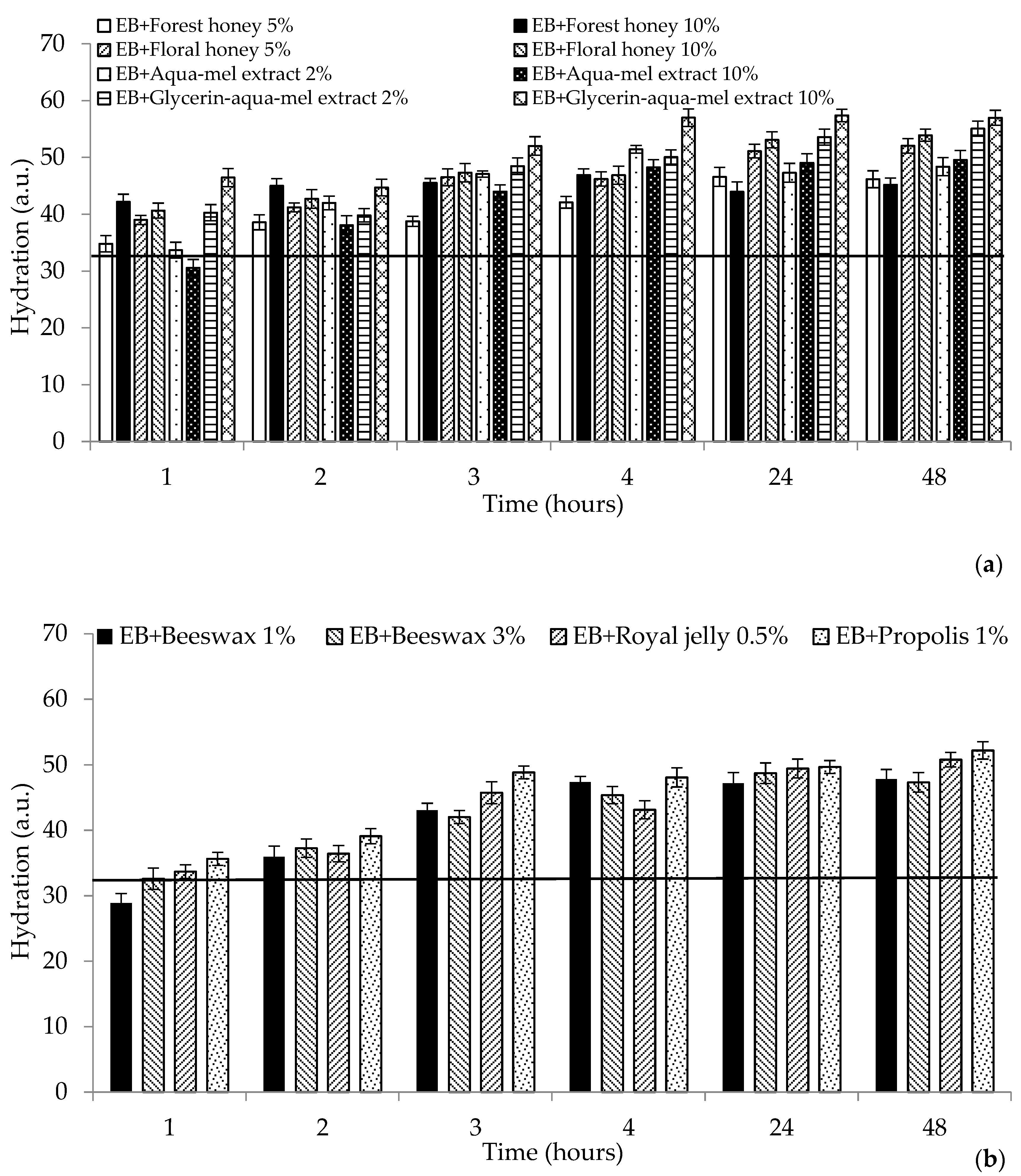
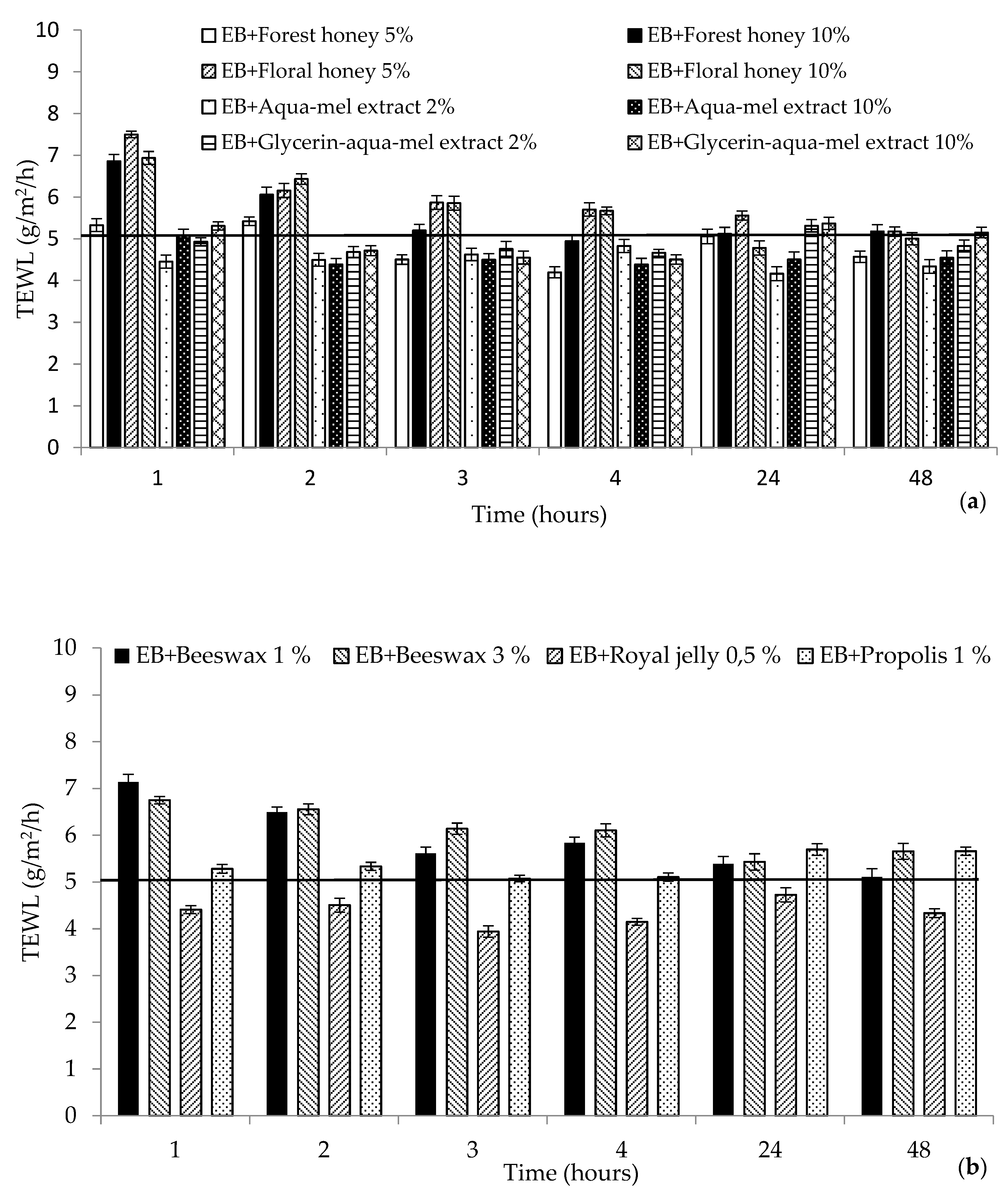
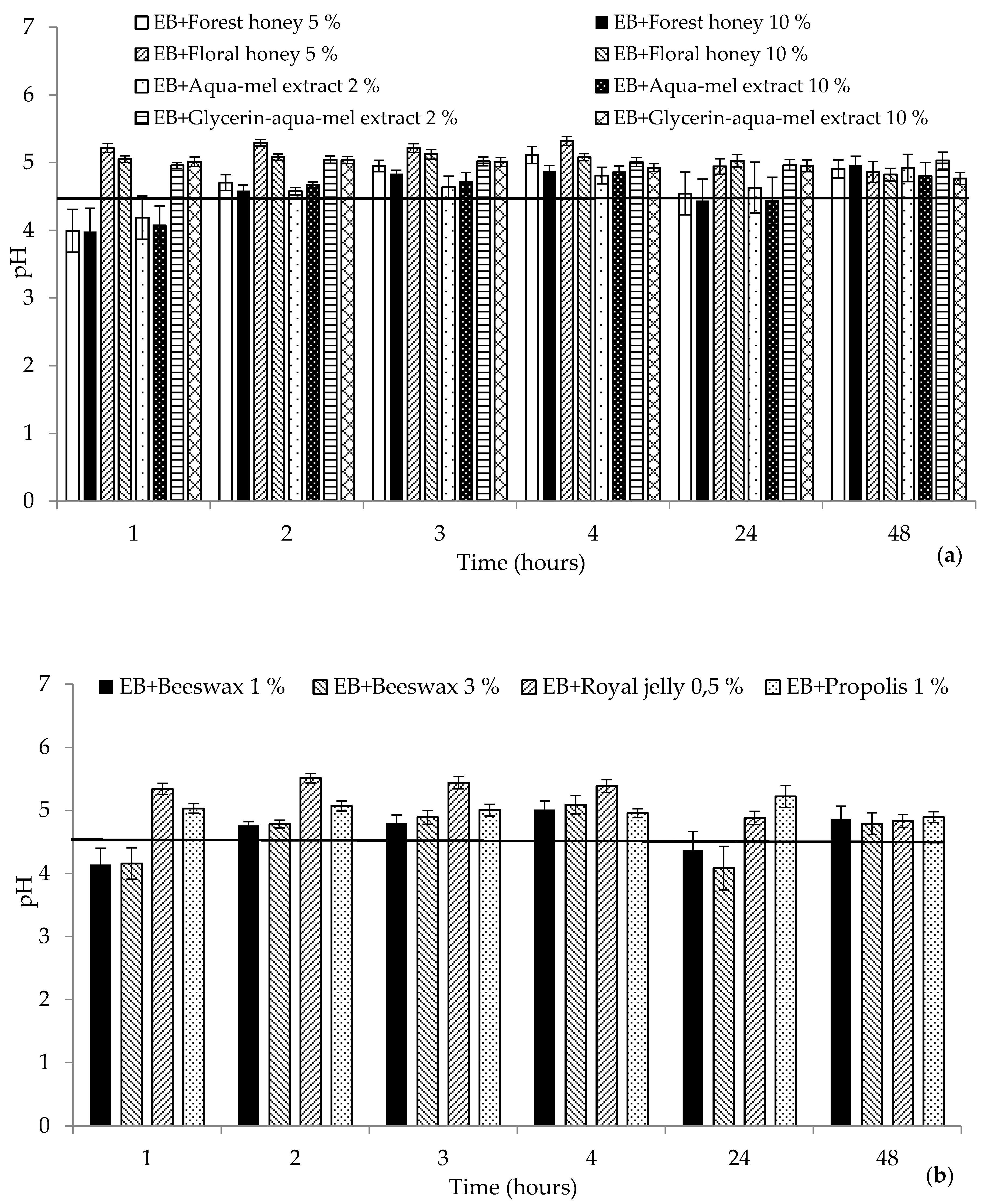
2. Results and Discussion
2.1. Biophysical Characteristics
2.2. Sensory Analysis
2.3. Questionnairre Survey
3. Materials and Methods
3.1. Analysis of Forest and Flower Honey
3.2. Procedure of Preparation of Cosmetics Matrices
3.3. Instrumental Techniques and Study Design
3.4. Sensory Analysis and Questionnairre Survey
3.5. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hajdušová, J. Bee Products from the Physician Sight [in Czech], 1st ed.; Český svaz včelařů: Praha, Czech Republic, 2006; pp. 10–45. [Google Scholar]
- Přidal, A. Bee Products [in Czech], 1st ed.; MZLU: Brno, Czech Republic, 2005; pp. 48–80. [Google Scholar]
- Gottschlack, T.E.; McEwen, G.N. International Cosmetic Ingredient Dictionary and Handbook, 13th ed.; The Personal Care Products Council: Washington, DC, USA, 2010. [Google Scholar]
- Crane, E. A Book of Honey; Oxford University Press: Oxford, UK, 1980; pp. 39–63. [Google Scholar]
- Nilforoushzadech, M.A.; Amirkhani, M.A.; Zarrintaj, P.; Salehi Moghaddam, A.; Mehrabi, T.; Alavi, S.; Mollapour, S.M. Skin care and rejuvenation by cosmeceutical facial mask. J. Cosmet. Dermatol. 2018, 17, 693–702. [Google Scholar] [CrossRef] [PubMed]
- McLoone, P.; Warnock, M.; Fyfe, L. Honey: A realistic antimicrobial for disorders of the skin. J. Microbial. Immunol. Infect. 2016, 49, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Burlando, B.; Cornara, L. Honey in dermatology and skin care: A review. J. Cosmet. Dermatol. 2013, 12, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Gordon, L.A. Compositae dermatitis. Australas J. Derm. 1999, 40, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, S. Royal Jelly, Bee Brood: Composition, Health, Medicine: A Review. Available online: https://www.bee-hexagon.net/files/file/fileE/Health/RJBookReview.pdf (accessed on 15 September 2019).
- Saewan, N.; Jimtaisong, A. Natural products as photoprotection. J. Cosmet. Dermatol. 2015, 14, 47–63. [Google Scholar] [CrossRef]
- Banskota, A.H.; Tezuka, Y.; Kadota, S. Recent progress in pharmacological research of propolis. Phytother. Res. 2001, 15, 561–571. [Google Scholar] [CrossRef]
- Krell, L. Value-Added Products from Beekeeping. Available online: http://www.fao.org/3/w0076e/w0076e00.htm (accessed on 16 September 2019).
- Meo, S.A.; Al-Asiri, S.A.; Mahesar, A.L.; Ansari, M.J. Role of honey in modern medicine. Saudi J. Biol. Sci. 2017, 24, 975–978. [Google Scholar] [CrossRef]
- De Paepe, K.; Derde, M.P.; Roseeuw, D.; Rogiers, V. Claim substantion and efficiency of hydrating body lotions and protective creams. Contact Dermat. 2000, 42, 227–234. [Google Scholar] [CrossRef]
- Lodén, M.; Wessman, W. The influence of a cream containing 20% glycerin and its vehicle on skin barrier properties. Int. J. Cosmet. Sci. 2001, 23, 115–119. [Google Scholar] [CrossRef]
- Polaskova, J.; Pavlackova, J.; Egner, P. Effect of vehicle on the performance of active moisturizing substances. Skin Res. Technol. 2015, 21, 403–412. [Google Scholar] [CrossRef]
- Jiménez, M.M.; Fresno, M.J.; Sellés, E. The galenic behaviour of a dermopharmaceutical excipient containing honey. Int. J. Cosmet. Sci. 1994, 16, 211–226. [Google Scholar] [CrossRef] [PubMed]
- Ahshawat, M.S.; Saraf, S.; Saraf, S. Preparation and characterization of herbal creams for improvement of skin viscoelastic properties. Int. J. Cosmet. Sci. 2008, 30, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Ediriweera, E.R.H.S.S.; Premarathna, N.Y.S. Medicinal and cosmetic uses of bee’s honey−A review. Ayu 2012, 33, 178–182. [Google Scholar] [CrossRef]
- Isla, M.; Cordero, A.; Díaz, L.; Pérez-Pérez, E.M.; Vit, P. Cosmetic properties of honey. 1. Antioxidant activity. In Stingless Bees Process Honey and Pollen in Cerumen Pots; Vit, P., Roubik, D.W., Eds.; Universidad de los Andes: Mérida, Venezuela, 2013; pp. 1–8. [Google Scholar]
- Márquez, R.; Bálsamo, S.; Morales, F.; Ruiz, N.; García, A.; León, R.; Montes, A.; Nava, N.; Noguera, Y.; Quintero, A.; et al. Technological use of beeswax for obtaining organic products, non-toxic for the human being. Revista Ciencia e Ingeniería 2019, 40, 17–26. [Google Scholar]
- Tupker, R.A.; Willis, C.; Berardesca, E.; Lee, C.H.; Fartasch, M.; Agner, T.; Serup, J. Guidelines on sodium lauryl sulfate (SLS) exposure tests. A report from the Standardization Group of the European Society of Contact Dermatitis. Contact Dermat. 1997, 37, 53–69. [Google Scholar] [CrossRef] [PubMed]
- Seweryn, A.; Bujak, T. Application of Anionic Phosphorus Derivatives of Alkyl Polyglucosides for the Production of Sustainable and Mild Body Wash Cosmetics. ACS Sustain. Chem. Eng. 2018, 6, 17294–17301. [Google Scholar] [CrossRef]
- Park, K.B.; Eun, H.C. A study of skin responses to follow-up, rechallenge and combined effects of irritants using non-invasive measurements. J. Derm. Sci. 1995, 10, 159–165. [Google Scholar] [CrossRef]
- Atrux-Tallau, N.; Romagny, C.; Padois, K.; Denis, A.; Haftek, M.; Falson, F.; Pirot, F.; Maibach, H.I. Effects of glycerol on human skin damaged by acute sodium lauryl sulphate treatment. Arch. Derm. Res. 2010, 302, 435–441. [Google Scholar] [CrossRef]
- Zahmatkesh, M.; Manesh, M.J.; Babashahabi, R. Effect of olea ointment and acetate mafenide on burn wounds–a randomized clinical trial. Iran. J. Nurs. Midwifery Res. 2015, 20, 599–603. [Google Scholar] [PubMed]
- Polášková, S. Year-round care of a child’s skin [in Czech]. Pediatr. Praxi 2012, 13, 96–100. [Google Scholar]
- Seweryn, A.; Klimaszewska, E.; Ogorzałek, M. Improvement in the Safety of Use of Hand Dishwashing Liquids through the Addition of Sulfonic Derivatives of Alkyl Polyglucosides. J. Surfact. Deterg. 2019, 22, 743–750. [Google Scholar] [CrossRef]
- Kiistala, R.; Hannuksela, M.; Mäkinen-Kiljunen, S.; Niinimäki, A.; Haahtela, T. Honey allergy is rare in patients sensitive to pollens. Allergy 1995, 50, 844–847. [Google Scholar] [CrossRef] [PubMed]
- Rosmilah, M.; Shahnaz, M.; Patel, G.; Lock, J.; Rahman, D.; Masita, A.; Noormalin, A. Characterization of major allergens of royal jelly Apis mellifera. Trop Biomed. 2008, 25, 243–251. [Google Scholar] [PubMed]
- Deutsche Industrie Norm. Bestimmung des Wassergehalts in Honig. DIN 10752. 1992.
- Lord, D.W.; Scotter, M.J.; Whittaker, A.D.; Wood, R. The determination of acidity, apparent reducing sugar and sucrose, hydroxymethylfurfural, mineral, moisture, water-insoluble solids contents in honey; collaborative study. J. Assoc. Publ. Anal. 1988, 26, 51–76. [Google Scholar]
- Akharaiyi, F.C.; Lawal, H.A. Physicochemical analysis and mineral contents of honey from farmers in western states of Nigeria. J. Nat. Sci. Res. 2016, 6, 78–84. [Google Scholar]
- Bosi, G.; Battaglini, M. Gas chromatographic analysis of free and protein amino acids in some unifloral honeys. J. Apicult. Res. 1978, 17, 152–166. [Google Scholar] [CrossRef]
- Nozal, M.J.; Bernal, J.L.; Toribio, M.L.; Diego, J.C.; Ruiz, A. Rapid and sensitive method for determining free amino acids in honey by gas chromatography with flame ionization or mass spectrometric detection. J. Chromatogr. A 2004, 1047, 137–146. [Google Scholar] [CrossRef]
- Bogdanov, S.; Baumann, S.E. Bestimmung von Honigzucker mit HPLC. Mitt. Gebiete Lebensm. Hyg. 1988, 79, 198–206. [Google Scholar]
- Da Costa Leite, J.M.; Trugo, L.C.; Costa, L.S.M.; Quinteiro, L.M.C.; Barth, O.M.; Dutra, V.L.M.; De Maria, C.A.B. Determination of oligosaccharides in Brazilian honeys of different botanical origin. Food Chem. 2000, 70, 93–98. [Google Scholar] [CrossRef]
- Rybak-Chmielewska, H.; Szczesna, T. Determination of saccharides in multifloral honey by means of HPLC. J. Apicult. Sci. 2003, 47, 93–101. [Google Scholar]
- Ciulu, M.; Solinas, S.; Floris, I.; Panzanelli, A.; Pilo, M.I.; Piu, P.C.; Spano, N.; Sanna, G. RP-HPLC determination of water-soluble vitamins in honey. Talanta 2011, 83, 924–929. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R. A Department of Food Technology, University of New South Wales, Kensington, NSW, Australia.; Faraji-Haremi, R Department of Food Technology, University of New South Wales, Kensington, NSW, Australia.; Wootton, M Department of Food Technology, University of New South Wales, Kensington, NSW, Australia. A rapid method for determining the diastase activity in honey. J. Apicult. Res. 1975, 14, 47–50. [Google Scholar]
- Hadorn, H. On the problem of the quantitative determination of diastase in honey. Mitt. Gebiete Lebensm. Hyg. 1961, 52, 67–103. [Google Scholar]
- Bogdanov, S. Beeswax: Production, Properties, Composition and Control. Available online: http://www.bee-hexagon.net/files/file/fileE/Wax/WaxBook1.pdf (accessed on 25 September 2019).
- Přidal, A. Beeswa–composition and utilization [in Czech]. Moderní Včelař 2007, 5, 20–21. [Google Scholar]
- Mathivanan, V.; Shah, G.N.; Manzoor, M.; Mir, G.M. A review on propolis-as a novel folk medicine. Indian J. Sci. 2013, 2, 23–30. [Google Scholar]
- Alban Muller International. Phytami® Honey; Raw material information data sheet; Alban Muller International: Chartres, France, 2009. [Google Scholar]
- Crodarom. Phytami® Honey; Safety data sheet; Crodarom: East Yorkshire, UK, 2013. [Google Scholar]
- CIOMS. International Ethical Guidelines for Health-related Research Involving Humans; Council for International Organizations of Medical Science (CIOMS): Geneva, Switzerland, 2016; Available online: https://cioms.ch/publications/product/international-ethical-guidelines-for-health-related-research-involving-humans/ (accessed on 20 September 2019).
- International Organization for Standardization. Sensory Analysis–Methodology–General Guidance; ISO 6658; International Organization for Standardization: Geneva, Switzerland, 2017. [Google Scholar]
- International Organization for Standardization. Sensory Analysis General Guidance for the Design of Test Rooms; ISO 8589; International Organization for Standardization: Geneva, Switzerland, 2017. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
| Component | Forest Honey (% ± SD) | Flower Honey (% ± SD) |
|---|---|---|
| Fructose | 31.30 ± 1.95 | 38.40 ± 2.01 |
| Glucose | 36.80 ± 4.60 | 33.20 ± 4.08 |
| Sucrose | 2.30 ± 0.59 | 2.90 ± 0.55 |
| Maltose | 5.50 ± 0.48 | 3.10 ± 0.34 |
| Oligosaccharides | 3.70 ± 0.61 | 2.20 ± 0.41 |
| Moisture | 18.60 ± 2.17 | 18.20 ± 2.04 |
| Enzymes | 0.16 ± 0.02 | 0.56 ± 0.03 |
| Vitamins | 0.11 ± 0.02 | 0.21 ± 0.02 |
| Ash | 1.04 ± 0.04 | 0.45 ± 0.03 |
| Free acids | 0.15 ± 0.02 | 0.35 ± 0.03 |
| Amino acids | 0.34 ± 0.02 | 0.43 ± 0.02 |
| Volar Side of the Left Forearm | Volar Side of the Right Forearm | ||
|---|---|---|---|
| 1st group of volunteers | 2nd group of volunteers | 1st group of volunteers | 2nd group of volunteers |
| Control | Aqua-mel extract 2% | Floral honey 10% | |
| SLS | Aqua-mel extract 10% | Glycerin–aqua–mel extract 2% | |
| Forest honey 5% | Royal jelly 0.5% | Beeswax 1% | Glycerin–aqua–mel extract 10% |
| Forest honey 10% | Floral honey 5% | Beeswax 3% | Propolis 1% |
| No. | Question |
|---|---|
| 1 | Which products do you use? Answer possibility: honey, propolis, royal jelly, wax, bee venom, pollen |
| 2 | What reasons led you to use honey and bee products? Answer possibility: physician recommendation, friend’s recommendation, advertising, skin problems, others |
| 3 | Do you think bee products has healing effects? Answer possibility: yes/no/don’t know |
| 4 | Are you allergic to any of the listed products: honey, propolis, royal jelly, wax, bee venom, pollen? Answer possibility: yes/no |
| 5 | Do you process honey or bee products for the production of ointments, tinctures, emulsions, etc. for home use? Answer possibility: yes/no |
| 6 | Do you use honey cosmetics? Answer possibility:/no |
| (a) | What is the reason for using honey cosmetics? Answer possibility: pain relief, wound healing, acne, inflammation, eczema, psoriasis, dry skin, strengthening immunity |
| (b) | What types of honey cosmetics do you use? Answer possibility: healing cosmetics (drops/tincture/spray/gel/ointment/balm/cream), cleaning cosmetics (wipes/water/milk/peeling/mask/toothpaste/mouthwash), cosmetics for men (shampoo/after shave/men’s cream), body cosmetics (milk/balm/bath foam/soap/shower shampoo), hair cosmetic (shampoo/hair lotion/balm) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pavlačková, J.; Egner, P.; Slavík, R.; Mokrejš, P.; Gál, R. Hydration and Barrier Potential of Cosmetic Matrices with Bee Products. Molecules 2020, 25, 2510. https://doi.org/10.3390/molecules25112510
Pavlačková J, Egner P, Slavík R, Mokrejš P, Gál R. Hydration and Barrier Potential of Cosmetic Matrices with Bee Products. Molecules. 2020; 25(11):2510. https://doi.org/10.3390/molecules25112510
Chicago/Turabian StylePavlačková, Jana, Pavlína Egner, Roman Slavík, Pavel Mokrejš, and Robert Gál. 2020. "Hydration and Barrier Potential of Cosmetic Matrices with Bee Products" Molecules 25, no. 11: 2510. https://doi.org/10.3390/molecules25112510
APA StylePavlačková, J., Egner, P., Slavík, R., Mokrejš, P., & Gál, R. (2020). Hydration and Barrier Potential of Cosmetic Matrices with Bee Products. Molecules, 25(11), 2510. https://doi.org/10.3390/molecules25112510








