Phytochemical Analysis, Network Pharmacology and in Silico Investigations on Anacamptis pyramidalis Tuber Extracts
Abstract
1. Introduction
2. Results and Discussion
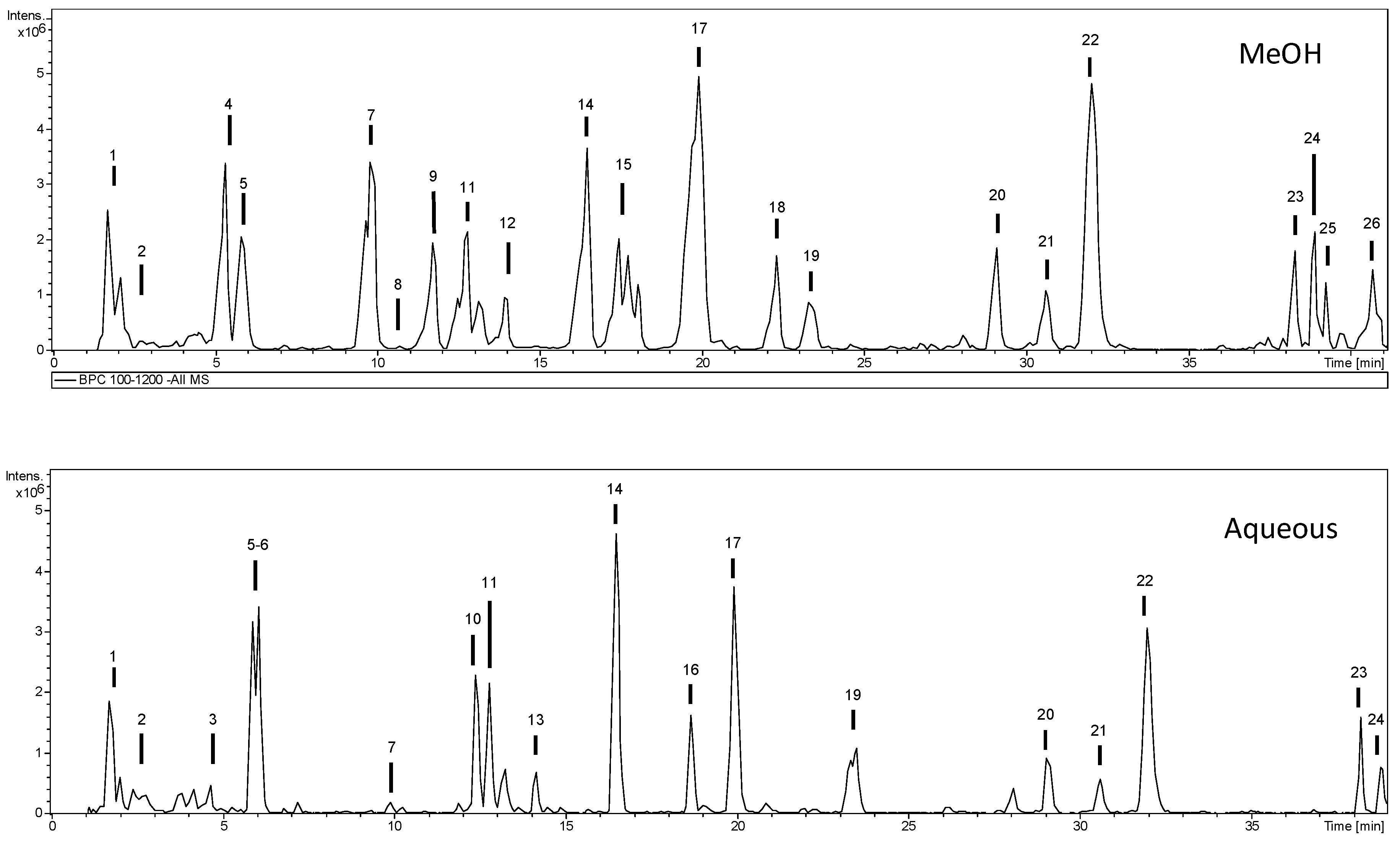
2.1. Phytochemical Profile
2.2. Enzyme Inhibition
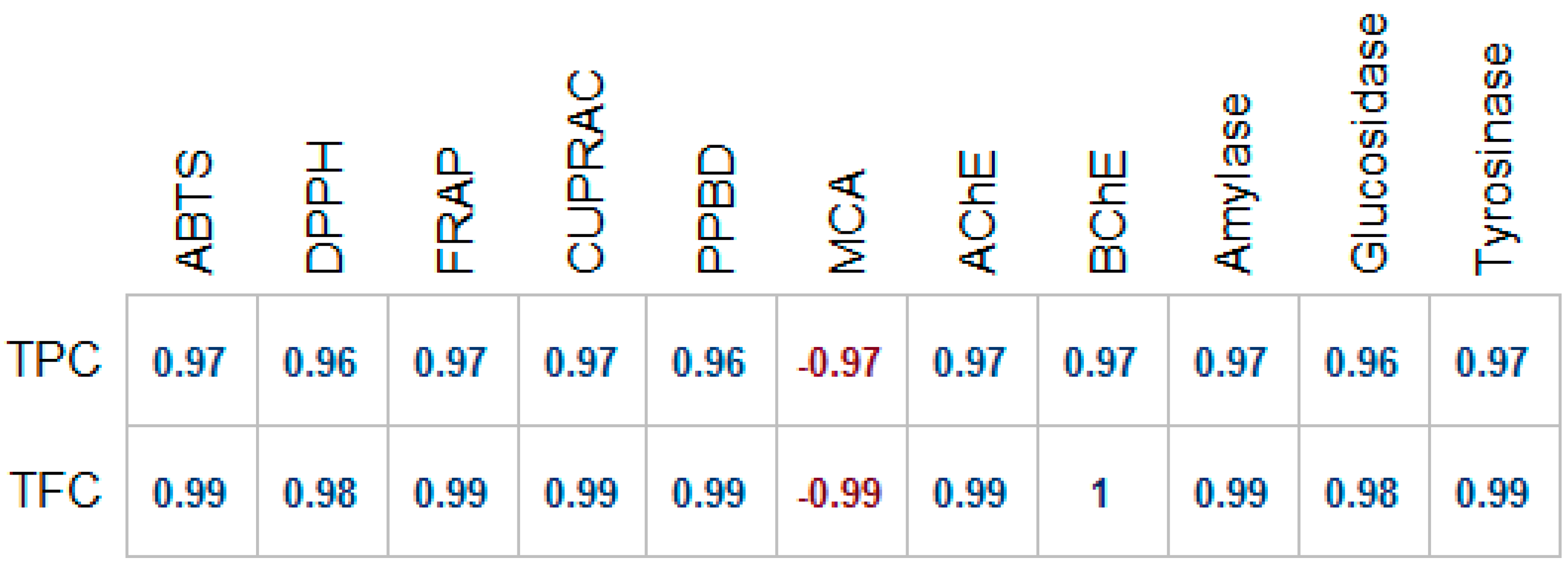
2.3. Antioxidant Activity
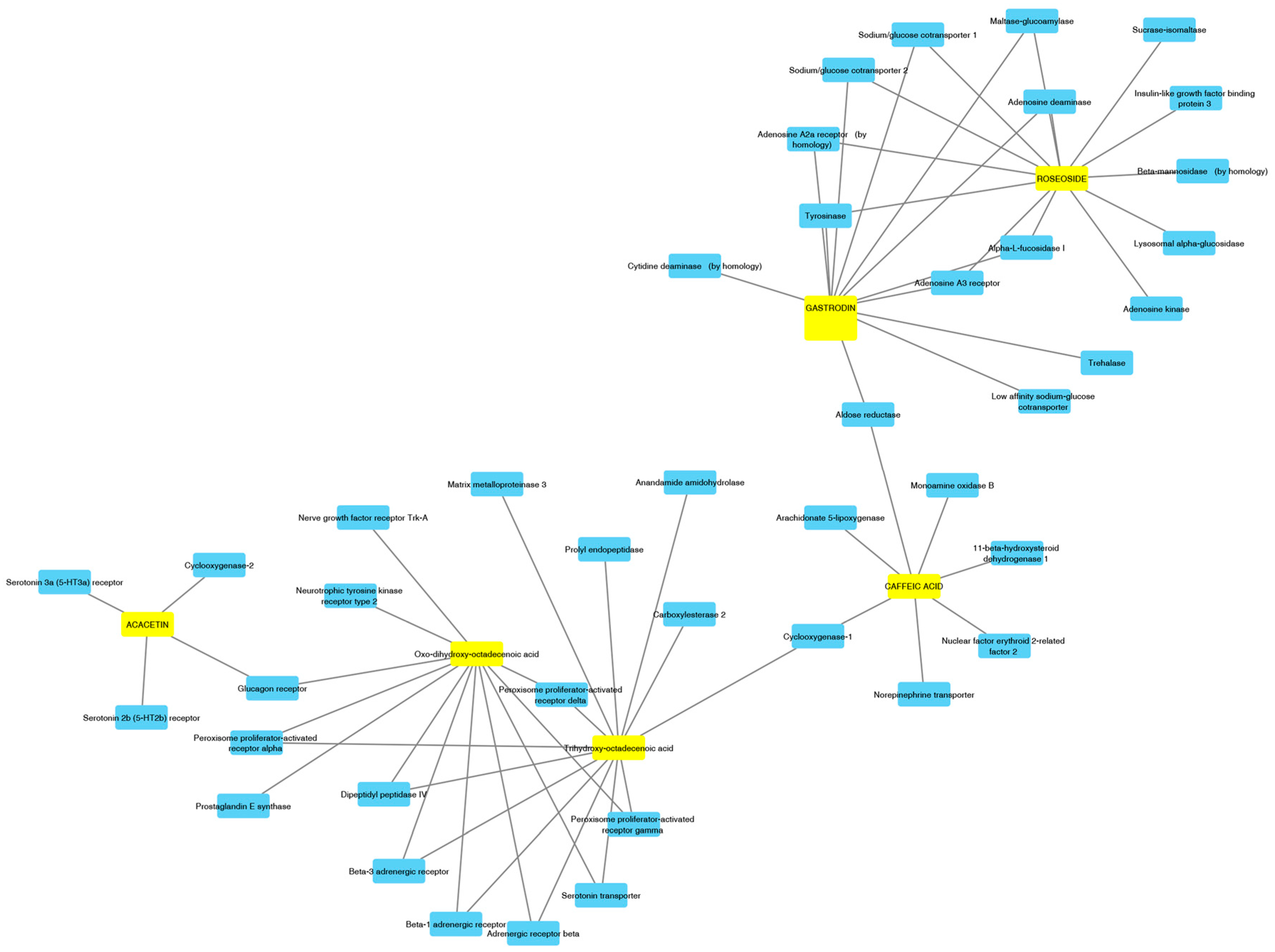
2.4. Prediction of Pharmacologic Targets and Pharmacokinetic Profile
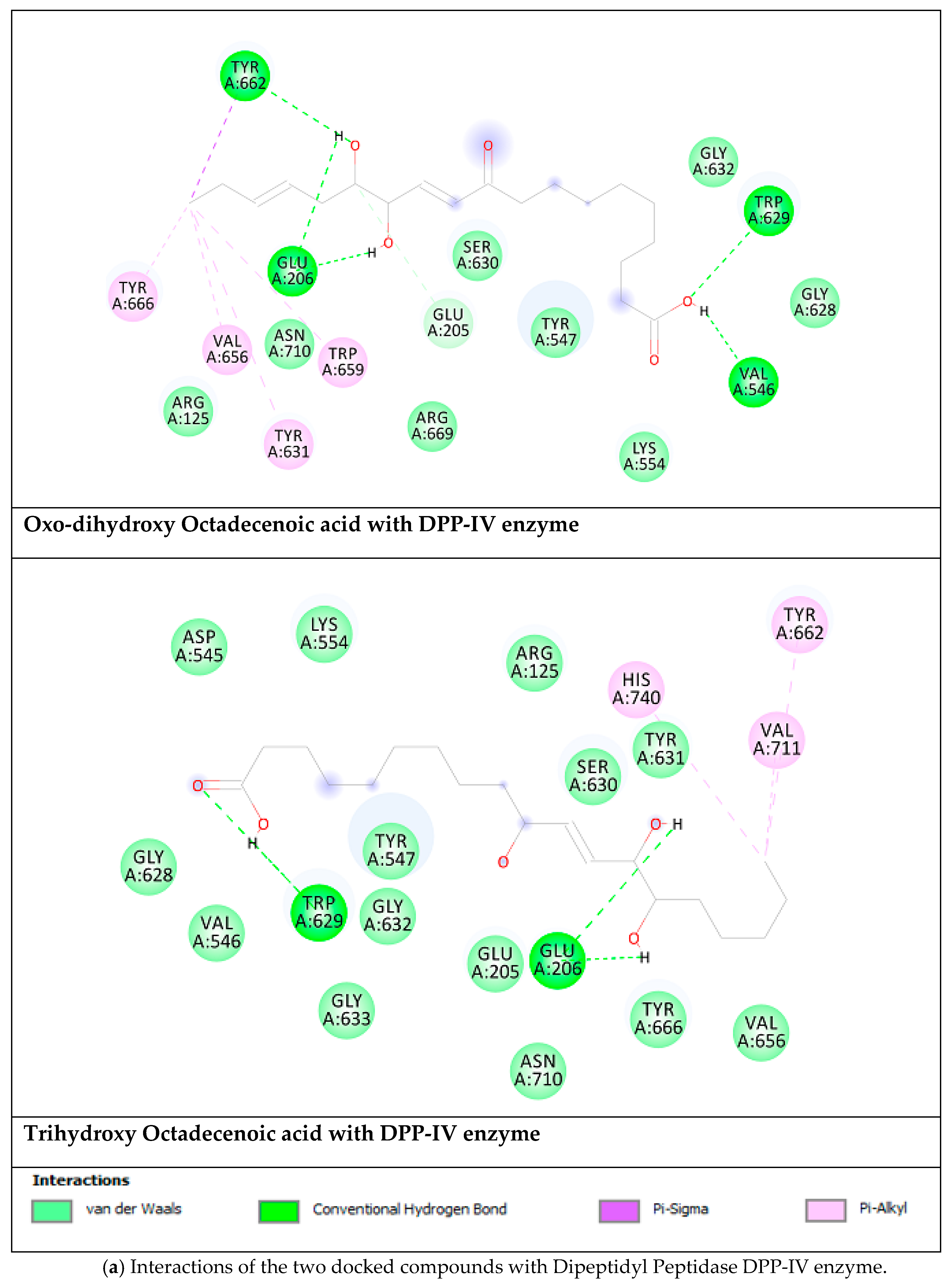
2.5. Docking Results
3. Materials and Methods
3.1. Plant Material and Preparation of Extracts
3.2. Profile of Bioactive Compounds
3.3. Chromatographic Analysis
3.4. Determination of Antioxidant and Enzyme Inhibitory Effects
3.5. Prediction of Putative Targets and Pharmacokinetics
3.6. Docking Calculations
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Hao, D.C.; Xiao, P.G. Pharmaceutical resource discovery from traditional medicinal plants: Pharmacophylogeny and pharmacophylogenomics. Chin. Herb. Med. 2020, 12, 104–117. [Google Scholar] [CrossRef]
- Chinsamy, M.; Finnie, J.F.; Van Staden, J. The ethnobotany of South African medicinal orchids. South Afr. J. Bot. 2011, 77, 2–9. [Google Scholar] [CrossRef]
- Bhattacharyya, P.; Van Staden, J. Ansellia africana (Leopard orchid): A medicinal orchid species with untapped reserves of important biomolecules—A mini review. South Afr. J. Bot. 2016, 106, 181–185. [Google Scholar] [CrossRef]
- Bhattacharyya, P.; Kumaria, S.; Tandon, P. Applicability of ISSR and DAMD markers for phyto-molecular characterization and association with some important biochemical traits of Dendrobium nobile, an endangered medicinal orchid. Phytochemistry 2015, 117, 306–316. [Google Scholar] [CrossRef]
- Sargin, S.A.; Büyükcengiz, M. Plants used in ethnomedicinal practices in Gulnar district of Mersin, Turkey. J. Herb. Med. 2019, 15, 100224. [Google Scholar] [CrossRef]
- Sargin, S.A.; Selvi, S.; Büyükcengiz, M. Ethnomedicinal plants of Aydıncık District of Mersin, Turkey. J. Ethnopharmacol. 2015, 174, 200–216. [Google Scholar] [CrossRef] [PubMed]
- Tekinşen, K.K.; Güner, A. Chemical composition and physicochemical properties of tubera salep produced from some Orchidaceae species. Food Chem. 2010, 121, 468–471. [Google Scholar] [CrossRef]
- Ghorbani, A.; Gravendeel, B.; Naghibi, F.; de Boer, H. Wild orchid tuber collection in Iran: A wake-up call for conservation. Biodivers. Conserv. 2014, 23, 2749–2760. [Google Scholar] [CrossRef]
- Masters, S.; van Andel, T.; de Boer, H.J.; Heijungs, R.; Gravendeel, B. Patent analysis as a novel method for exploring commercial interest in wild harvested species. Biol. Conserv. 2020, 243, 108454. [Google Scholar] [CrossRef]
- Swarts, N.D.; Dixon, K.W. Terrestrial orchid conservation in the age of extinction. Ann. Bot. 2009, 104, 543–556. [Google Scholar] [CrossRef] [PubMed]
- Kreziou, A.; de Boer, H.; Gravendeel, B. Harvesting of salep orchids in north-western Greece continues to threaten natural populations. Oryx 2016, 50, 393–396. [Google Scholar] [CrossRef]
- Wang, L.; Xiao, H.B.; Yang, L.; Wang, Z.T. Two new phenolic glycosides from the rhizome of Gastrodia elata. J. Asian Nat. Prod. Res. 2012, 14, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xiao, H.; Liang, X.; Wei, L. Identification of phenolics and nucleoside derivatives in Gastrodia elata by HPLC-UV-MS. J. Sep. Sci. 2007, 30, 1488–1495. [Google Scholar] [CrossRef]
- Spínola, V.; Llorent-Martínez, E.J.; Gouveia, S.; Castilho, P.C. Myrica faya: A new source of antioxidant phytochemicals. J. Agric. Food Chem. 2014, 62, 9722–9735. [Google Scholar] [CrossRef]
- Van Hoyweghen, L.; De Bosscher, K.; Haegeman, G.; Deforce, D.; Heyerick, A. In vitro inhibition of the transcription factor NF-κB and cyclooxygenase by Bamboo extracts. Phytother. Res. 2014, 28, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Pattan, S.; Shirote, P.; Pattan, J.; Manvi, F.V. Treatment of chronic diseases through enzyme inhibition. Indian Drugs 2008, 45, 169–174. [Google Scholar]
- Barbagallo, M.; Dominguez, L.J. Type 2 diabetes mellitus and Alzheimer’s disease. World J. Diabetes 2014, 5, 889–893. [Google Scholar] [CrossRef]
- Kandimalla, R.; Thirumala, V.; Reddy, P.H. Is Alzheimer’s disease a Type 3 Diabetes? A critical appraisal. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2017, 1863, 1078–1089. [Google Scholar] [CrossRef]
- Darvesh, S. Butyrylcholinesterase as a diagnostic and therapeutic target for Alzheimer’s disease. Curr. Alzheimer Res. 2016, 13, 1173–1177. [Google Scholar] [CrossRef]
- Agunloye, O.M.; Oboh, G. Modulatory effect of caffeic acid on cholinesterases inhibitory properties of donepezil. J. Complement. Integr. Med. 2017, 15, 20170016. [Google Scholar] [CrossRef]
- Nugroho, A.; Park, J.H.; Choi, J.S.; Park, K.S.; Hong, J.P.; Park, H.J. Structure determination and quantification of a new flavone glycoside with anti-acetylcholinesterase activity from the herbs of Elsholtzia ciliata. Nat. Prod. Res. 2019, 33, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Pei, C.J.; Lee, J.; Si, Y.X.; Oh, S.; Xu, W.A.; Yin, S.J.; Qian, G.Y.; Han, H.Y. Inhibition of tyrosinase by gastrodin: An integrated kinetic-computational simulation analysis. Process. Biochem. 2013, 48, 162–168. [Google Scholar] [CrossRef]
- Kim, N.Y.; Kwon, H.S.; Lee, H.Y. Effect of inhibition on tyrosinase and melanogenesis of Agastache rugosa Kuntze by lactic acid bacteria fermentation. J. Cosmet. Dermatol. 2017, 16, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Wang, X. Antioxidant therapies for Alzheimer’s disease. Oxid Med. Cell Longev. 2012, 2012, 472932. [Google Scholar] [CrossRef]
- Hajhashemi, V.; Vaseghi, G.; Pourfarzam, M.; Abdollahi, A. Are antioxidants helpful for disease prevention? Res. Pharm. Sci. 2010, 5, 1–8. [Google Scholar]
- Young, I.S.; Woodside, J.V. Antioxidants in health and disease. J. Clin. Pathol. 2001, 54, 176. [Google Scholar] [CrossRef]
- Aroor, A.R.; Manrique-Acevedo, C.; DeMarco, V.G. The role of dipeptidylpeptidase-4 inhibitors in management of cardiovascular disease in diabetes; focus on linagliptin. Cardiovasc Diabetol. 2018, 17, 59. [Google Scholar] [CrossRef]
- Valassi, E.; Scacchi, M.; Cavagnini, F. Neuroendocrine control of food intake. Nutr. Metab. Cardiovasc Dis. 2008, 18, 158–168. [Google Scholar] [CrossRef]
- Chiavaroli, A.; Recinella, L.; Ferrante, C.; Locatelli, M.; Macchione, N.; Zengin, G.; Leporini, L.; Leone, S.; Martinotti, S.; Brunetti, L.; et al. Crocus sativus, Serenoa repens and Pinus massoniana extracts modulate inflammatory response in isolated rat prostate challenged with LPS. J. Boil. Regul. Homeost. Agents 2017, 31, 531–541. [Google Scholar]
- Llorent-Martínez, E.J.; Zengin, G.; Lobine, D.; Molina-García, L.; Mollica, A.; Mahomoodally, M.F. Phytochemical characterization, in vitro and in silico approaches for three Hypericum species. New J. Chem. 2018, 42, 5204–5214. [Google Scholar] [CrossRef]
- Zengin, G.; Locatelli, M.; Stefanucci, A.; Macedonio, G.; Novellino, E.; Mirzaie, S.; Dvorácskó, S.; Carradori, S.; Brunetti, L.; Orlando, G.; et al. Chemical characterization, antioxidant properties, anti-inflammatory activity, and enzyme inhibition of Ipomoea batatas L. leaf extracts. Int. J. Food Prop. 2017, 20, 1907–1919. [Google Scholar] [CrossRef]
- Gu, L.; Lu, J.; Li, Q.; Wu, N.; Zhang, L.; Li, H.; Xing, W.; Zhang, X. A network-based analysis of key pharmacological pathways of Andrographis paniculata acting on Alzheimer’s disease and experimental validation. J. Ethnopharmacol. 2020, 251, 112488. [Google Scholar] [CrossRef] [PubMed]
- The Molinspiration Database. Available online: http://www.molinspiration.com (accessed on 14 May 2020).
- Frisch, M.; Trucks, G.; Schlegel, H.; Scuseria, G.; Robb, M.; Cheeseman, J.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. Gaussian 09; Gaussian, Inc.: Wallingford, CT, USA, 2009; Volume 32, pp. 5648–5652. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |

| Extracts | Extraction Yield (%) | Total Phenolic Content (mg·gallic acid Equivalent (GAE)/g Extract) | Total Flavonoid Content (mg·rutin Equivalent (RE)/g Extract) |
|---|---|---|---|
| Methanol | 13.24 | 17.03 ± 0.06 a | 0.48 ± 0.05 a |
| Water | 13.08 | 16.64 ± 0.06 a | 0.02 ± 0.01 b |
| No. | tR (min) | [M − H]− m/z | m/z (% Base Peak) | Assigned Identification | Methanol | Water |
|---|---|---|---|---|---|---|
| 1 | 1.9 | 341 | MS2 [341]: 179 (100), 161 (26), 149 (8), 143 (13), 131 (7), 119 (8), 113 (15) | Disaccharide | ✓ | ✓ |
| 2 | 2.7 | 191 | MS2 [191]: 173 (62), 111 (100) | Citric acid | ✓ | ✓ |
| 3 | 4.7 | 459 | MS2 [459]: 173 (100) MS3 [459→173]: 111 (100) | Parishin G isomer-1 | ✓ | |
| 4 | 5.3 | 367 | MS2 [367]: 293 (21), 187 (20), 143 (100) MS3 [367→143]: 125 (100) | Unknown | ✓ | |
| 5 | 5.9 | 433 | MS2 [433]: 227(7), 205 (100) MS3 [433→205]: 143 (62), 115 (100) | Unknown | ✓ | ✓ |
| 6 | 6.1 | 459 | MS2 [459]: 173 (100) MS3 [459→173]: 111 (100) | Parishin G isomer-2 | ✓ | |
| 7 | 9.8 | 351 | MS2 [351]: 171 (100), 127 (22) MS3 [351→171]: 127 (100) | Unknown | ✓ | ✓ |
| 8 | 10.7 | 431 | MS2 [431]: 385 (100), 223 (15) MS3 [431→385]: 223 (42), 153 (100), 138 (43) | Roseoside (formate adduct) | ✓ | |
| 9 | 11.7 | 635 | MS2 [635]: 349 (100), 277 (56) MS3 [635→349]: 305 (19), 277 (100), 169 (89), 143 (43) | Gastrodin derivative | ✓ | |
| 10 | 12.4 | 727 | MS2 [727]: 459 (4), 441 (38), 423 (100), 397 (21), 369 (18), 263 (3) | Parishin B | ✓ | |
| 11 | 12.8 | 473 | MS2 [473]: 285 (100), 187 (27), 159 (53), 143 (63) MS3 [473→285]: 161 (39), 123 (100) | Gastrodin derivative | ✓ | ✓ |
| 12 | 13.9 | 635 | MS2 [635]: 349 (100), 277 (38) MS3 [635→349]: 277 (100), 169 (66), 143 (50) | Gastrodin derivative | ✓ | |
| 13 | 14.1 | 727 | MS2 [727]: 459 (3), 441 (46), 423 (100), 397 (26), 369 (25), 263 (5) | Parishin C | ✓ | |
| 14 | 16.5 | 473 | MS2 [473]: 285 (100), 187 (11), 169 (20), 159 (36), 143 (48) MS3 [473→285]: 161 (23), 123 (100) | Gastrodin derivative | ✓ | ✓ |
| 15 | 17.7 | 619 | MS2 [619]: 439 (100) MS3 [619→439]: 171 (32), 153 (100) MS4 [619→439→153]: 138 (100), 109 (45) | Dihydroxybenzoic acid derivative | ✓ | |
| 16 | 18.6 | 995 | MS2 [995]: 727 (100) MS3 [995→727]: 459 (7), 441 (28), 423 (100), 397 (27), 369 (18), 263 (1) | Parishin | ✓ | |
| 17 | 19.9 | 741 | MS2 [741]: 473 (100) MS3 [741→473]: 285 (100), 187 (13), 159 (24), 143 (62) MS4 [741→473→285]: 161 (65), 123 (100) | Gastrodin derivative | ✓ | ✓ |
| 18 | 22.3 | 887 | MS2 [887]: 619 (100), 439 (32) MS3 [887→619]: 439 (100) MS4 [887→619→439]: 171 (33), 153 (100) | Unknown | ✓ | |
| 19 | 23.3 | 457 | MS2 [457]: 153 (100) MS3 [457→153]: 109 (100) | Dihydroxybenzoic acid derivative | ✓ | ✓ |
| 20 | 29.0 | 725 | MS2 [725]: 457 (100) MS3 [725→457]: 285 (39), 153 (100) MS4 [725→457→153]: 109 (100) | Dihydroxybenzoic acid derivative | ✓ | ✓ |
| 21 | 30.6 | 282 | MS2 [282]: 145 (100), 119 (73) | Unknown | ✓ | ✓ |
| 22 | 32.0 | 312 | MS2 [312]: 179 (61), 135 (100) | Caffeic acid derivative | ✓ | ✓ |
| 23 | 38.2 | 623 | MS2 [623]: 461 (100), 283 (38) MS3 [623→461]: 283 (100) MS4 [623→461→283]: 268 (100) | Acacetin derivative | ✓ | ✓ |
| 24 | 38.8 | 623 | MS2 [623]: 461 (100) MS3 [623→461]: 283 (100) MS4 [623→461→283]: 268 (100) | Acacetin derivative | ✓ | ✓ |
| 25 | 39.2 | 327 | MS2 [327]: 309 (27), 291 (55), 229 (48), 211 (48), 171 (100) | Oxo-dihydroxy-octadecenoic acid | ✓ | |
| 26 | 40.6 | 329 | MS2 [329]: 311 (47), 229 (72), 211 (56), 171 (100) | Trihydroxy-octadecenoic acid | ✓ |
| Peak | Compound | Methanol | Water |
|---|---|---|---|
| 1 | Disaccharide | 2.02 | 1.83 |
| 2 | Citric acid | 0.14 | 1.65 |
| 3 | Parishin G isomer-1 | 0.00 | 1.11 |
| 4 | Unknown | 6.50 | 0.00 |
| 5 | Unknown | 0.80 | 1.08 |
| 6 | Parishin G isomer-2 | 0.00 | 10.96 |
| 7 | Unknown | 10.03 | 0.48 |
| 8 | Roseoside | 0.16 | 0.00 |
| 9 | Gastrodin derivative | 4.35 | 0.00 |
| 10 | Parishin B | 0.00 | 7.25 |
| 11 | Gastrodin derivative | 5.14 | 6.28 |
| 12 | Gastrodin derivative | 1.41 | 0.00 |
| 13 | Parishin C | 0.00 | 1.53 |
| 14 | Gastrodin derivative | 9.58 | 16.03 |
| 15 | Dihydroxybenzoic acid derivative | 5.04 | 0.00 |
| 16 | Parishin | 0.00 | 5.90 |
| 17 | Gastrodin derivative | 18.01 | 13.34 |
| 18 | Unknown | 3.19 | 0.00 |
| 19 | Dihydroxybenzoic acid derivative | 2.75 | 6.11 |
| 20 | Dihydroxybenzoic acid derivative | 3.57 | 3.46 |
| 21 | Unknown | 2.28 | 1.73 |
| 22 | Caffeic acid derivative | 15.94 | 16.20 |
| 23 | Acacetin derivative | 2.76 | 2.85 |
| 24 | Acacetin derivative | 3.23 | 2.20 |
| 25 | Oxo-dihydroxy-octadecenoic acid | 0.19 | 0.00 |
| 26 | Trihydroxy-octadecenoic acid | 2.91 | 0.00 |
| Extracts | AChE (mg·GALAE/g Extract) | BChE (mg·GALAE/g Extract) | Tyrosinase (mg·KAE/g Extract) | α-Amylase (mg·ACAE/g Extract) | α-Glucosidase (mg·ACAE/g Extract) |
|---|---|---|---|---|---|
| Methanol | 0.97 ± 0.01 a | 0.78 ± 0.03 a | 69.69 ± 0.29 a | 15.76 ± 0.25 a | 20.07 ± 4.29 a |
| Water | 0.04 ± 0.01 b | 0.18 ± 0.02 b | 11.09 ± 1.40 b | 5.23 ± 0.10 b | 5.23 ± 0.40 b |
| Extracts | Phosphomolybdenum (mmol·TE/g) | DPPH (mg·TE/g Extract) | ABTS (mg·TE/g Extract) | CUPRAC (mg·TE/g Extract) | FRAP (mg·TE/g Extract) | Metal Chelating Ability (mg·EDTAE/g) |
|---|---|---|---|---|---|---|
| Methanol | 0.73 ± 0.03 a | 22.12 ± 0.69 a | 44.23 ± 0.29 a | 45.56 ± 0.81 a | 29.38 ± 0.57 a | 11.10 ± 0.44 b |
| Water | 0.42 ± 0.02 b | 9.73 ± 0.11 b | 29.83 ± 0.63 b | 26.99 ± 0.19 b | 21.70 ± 0.25 b | 21.14 ± 0.44 a |
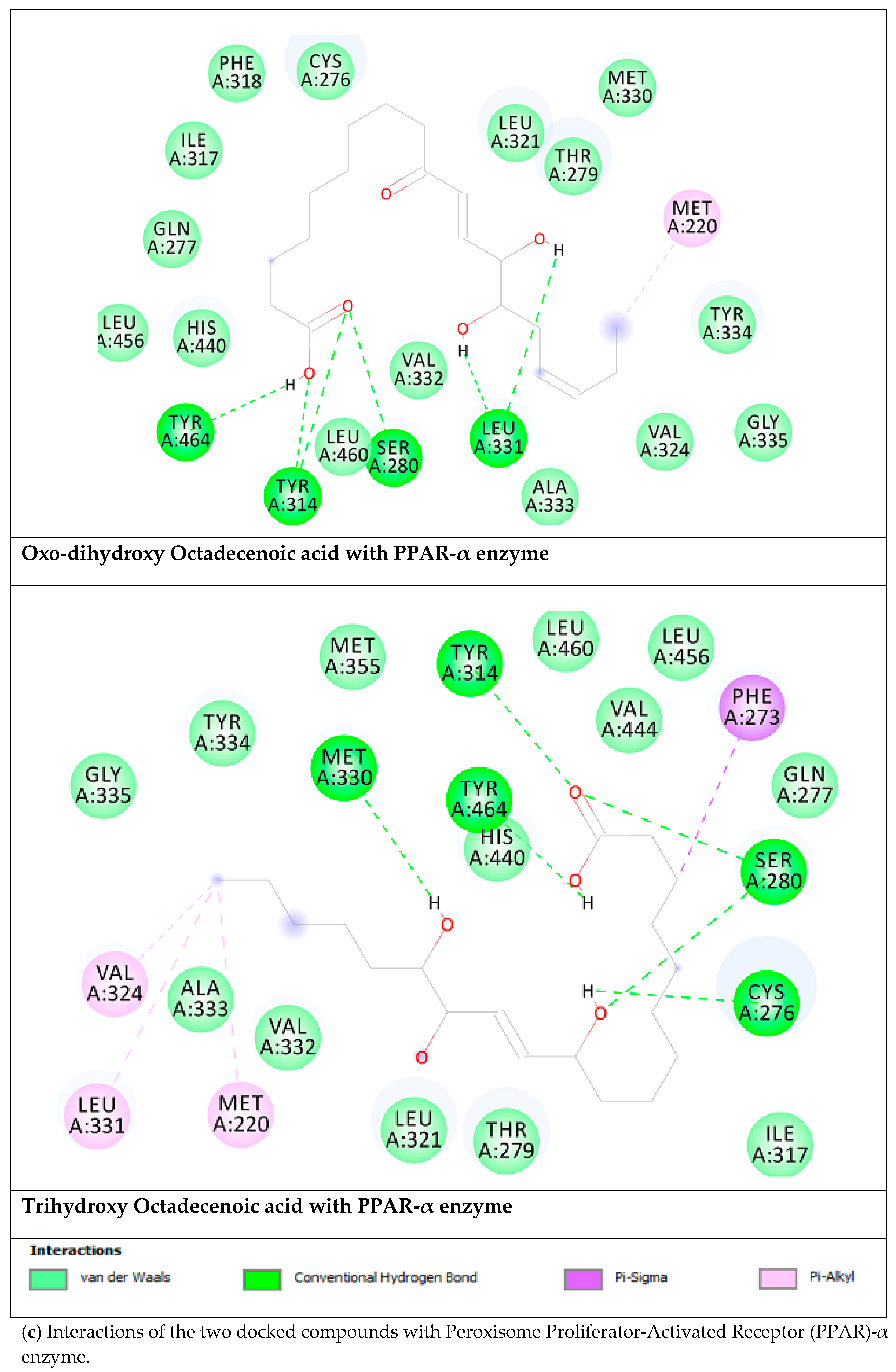
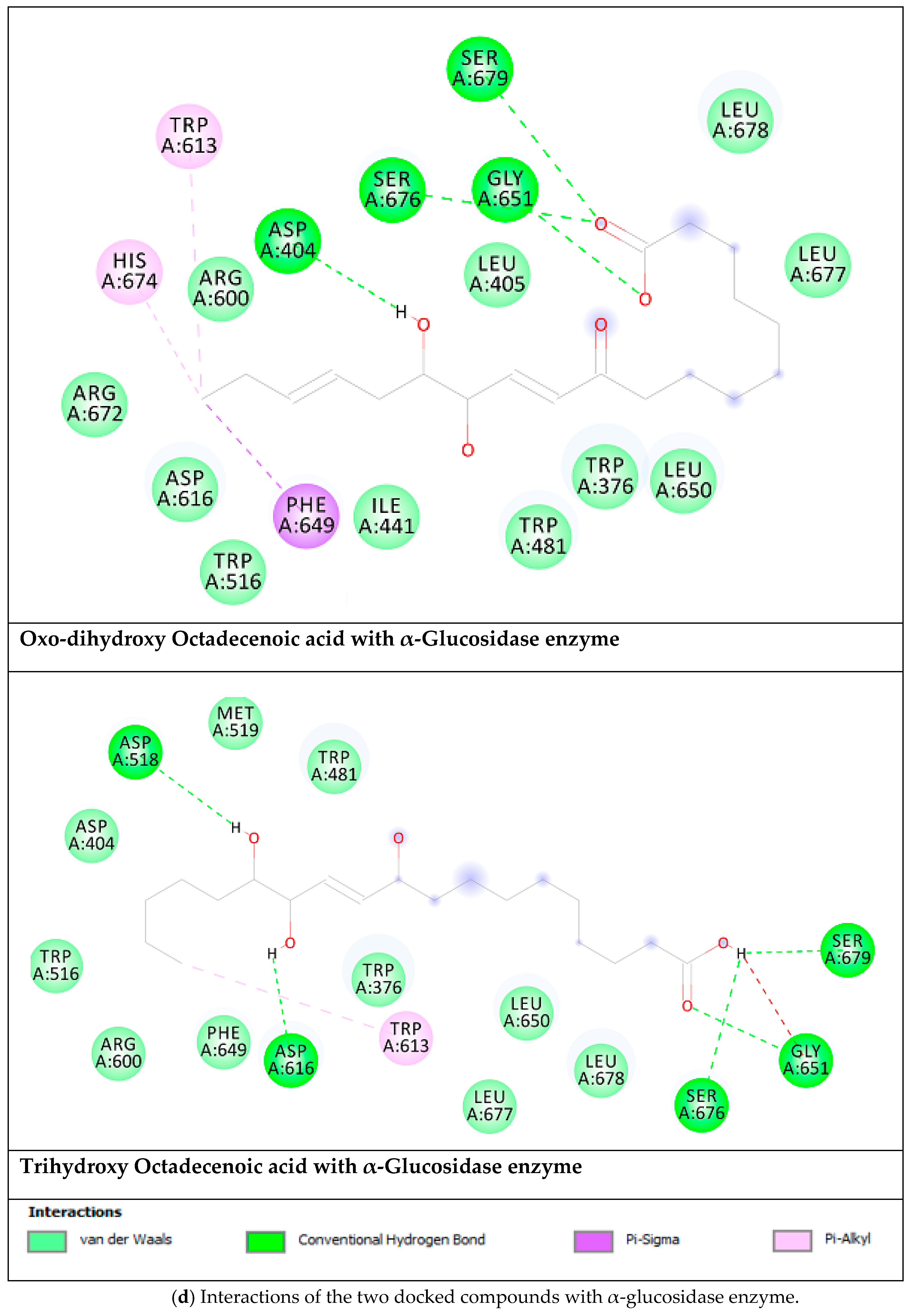
| Targets | ∆G (Ki) | Key Residues | no. of HB |
|---|---|---|---|
| Oxo-Dihydroxy Octadecenoic acid | |||
| Aldose reductase | −9.92 (53.7 nM) | Ala299(HB), Trp20 (HB), Thr19 (HB), Asp43(HB), Trp111, Phe311, Tyr309 | 4 |
| DPP-IV | −6.06 (35.9 µM) | Tyr662 (HB), Trp629 (HB), Val546 (HB), Glu206 (HB), Trp659, Tyr631, Val656, Tyr666. | 5 |
| PPAR-α | −7.04 (6.9 µM) | Tyr464 (HB), Tyr314 (HB), Ser280 (HB), Leu331 (HB), Met220 | 6 |
| α-Glucosidase | −6.06 (36.1 µM) | Ser679 (HB), Gly651 (HB), Ser676 (HB), Asp404 (HB), Trp613, His674, Phe649 | 4 |
| Trihydroxy Octadecenoic acid | |||
| Aldose reductase | −9.08 (222.3 nM) | Ala299 (HB), Trp111 (HB), Asp43 (HB),Thr9 (HB),Trp20 (HB), Cys303, Cys80, Phe115 | 6 |
| DPP-IV | −5.82 (54.2 µM) | Trp629 (HB), Glu206 (HB), His740, Val711, Tyr662. | 3 |
| PPAR-α | −6.74 (11.4 µM) | Met330 (HB), Tyr314 (HB), Tyr464 (HB), Ser280 (HB), Cys276 (HB), Phe273, Met220, Leu331, Val324 | 6 |
| α-Glucosidase | −4.63 (400.5 µM) | Asp518 (HB), Asp616 (HB), Ser679 (HB), Gly651 (HB), Ser676 (HB), Trp613 | 5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fawzi Mahomoodally, M.; Picot-Allain, M.C.N.; Zengin, G.; Llorent-Martínez, E.J.; Abdullah, H.H.; Ak, G.; Senkardes, I.; Chiavaroli, A.; Menghini, L.; Recinella, L.; et al. Phytochemical Analysis, Network Pharmacology and in Silico Investigations on Anacamptis pyramidalis Tuber Extracts. Molecules 2020, 25, 2422. https://doi.org/10.3390/molecules25102422
Fawzi Mahomoodally M, Picot-Allain MCN, Zengin G, Llorent-Martínez EJ, Abdullah HH, Ak G, Senkardes I, Chiavaroli A, Menghini L, Recinella L, et al. Phytochemical Analysis, Network Pharmacology and in Silico Investigations on Anacamptis pyramidalis Tuber Extracts. Molecules. 2020; 25(10):2422. https://doi.org/10.3390/molecules25102422
Chicago/Turabian StyleFawzi Mahomoodally, Mohamad, Marie Carene Nancy Picot-Allain, Gokhan Zengin, Eulogio J. Llorent-Martínez, Hassan H. Abdullah, Gunes Ak, Ismail Senkardes, Annalisa Chiavaroli, Luigi Menghini, Lucia Recinella, and et al. 2020. "Phytochemical Analysis, Network Pharmacology and in Silico Investigations on Anacamptis pyramidalis Tuber Extracts" Molecules 25, no. 10: 2422. https://doi.org/10.3390/molecules25102422
APA StyleFawzi Mahomoodally, M., Picot-Allain, M. C. N., Zengin, G., Llorent-Martínez, E. J., Abdullah, H. H., Ak, G., Senkardes, I., Chiavaroli, A., Menghini, L., Recinella, L., Brunetti, L., Leone, S., Orlando, G., & Ferrante, C. (2020). Phytochemical Analysis, Network Pharmacology and in Silico Investigations on Anacamptis pyramidalis Tuber Extracts. Molecules, 25(10), 2422. https://doi.org/10.3390/molecules25102422















