Improving Regulation of Enzymatic and Non-Enzymatic Antioxidants and Stress-Related Gene Stimulation in Cucumber mosaic cucumovirus-Infected Cucumber Plants Treated with Glycine Betaine, Chitosan and Combination
Abstract
1. Introduction
2. Results
2.1. Disease Severity and Reduction of Virus Infectivity
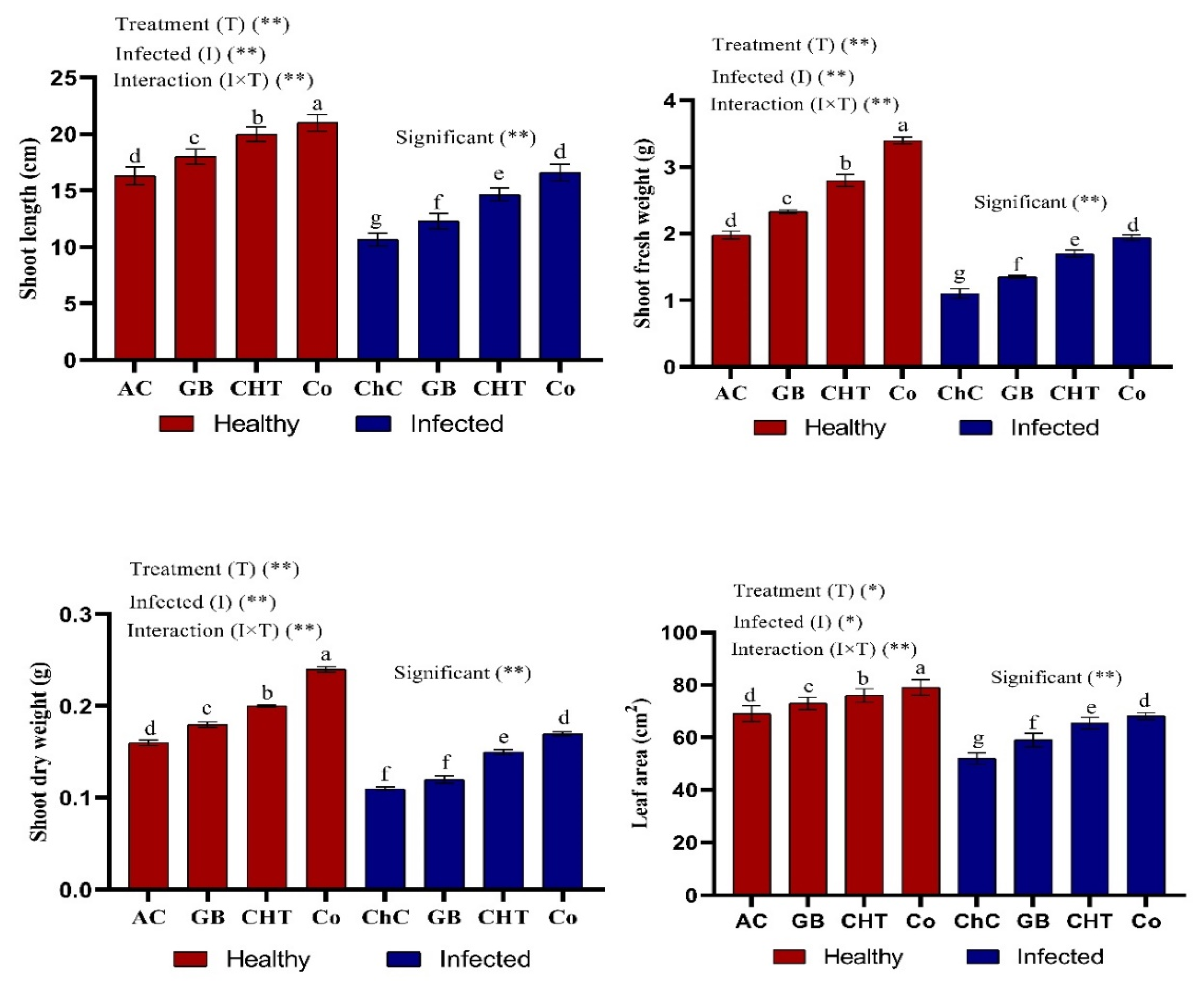
2.2. Plant Growth
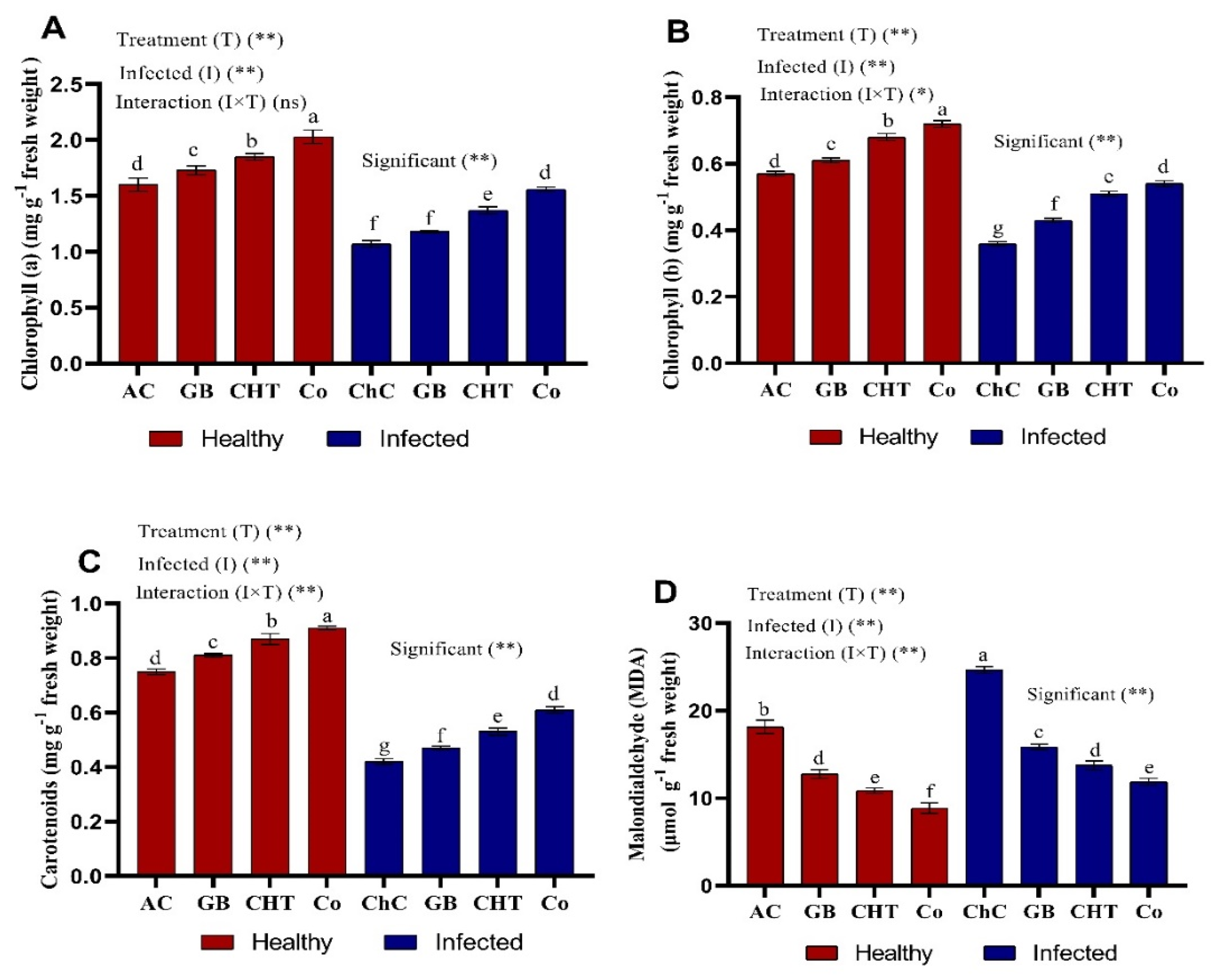
2.3. Photosynthetic Pigments and MDA Content
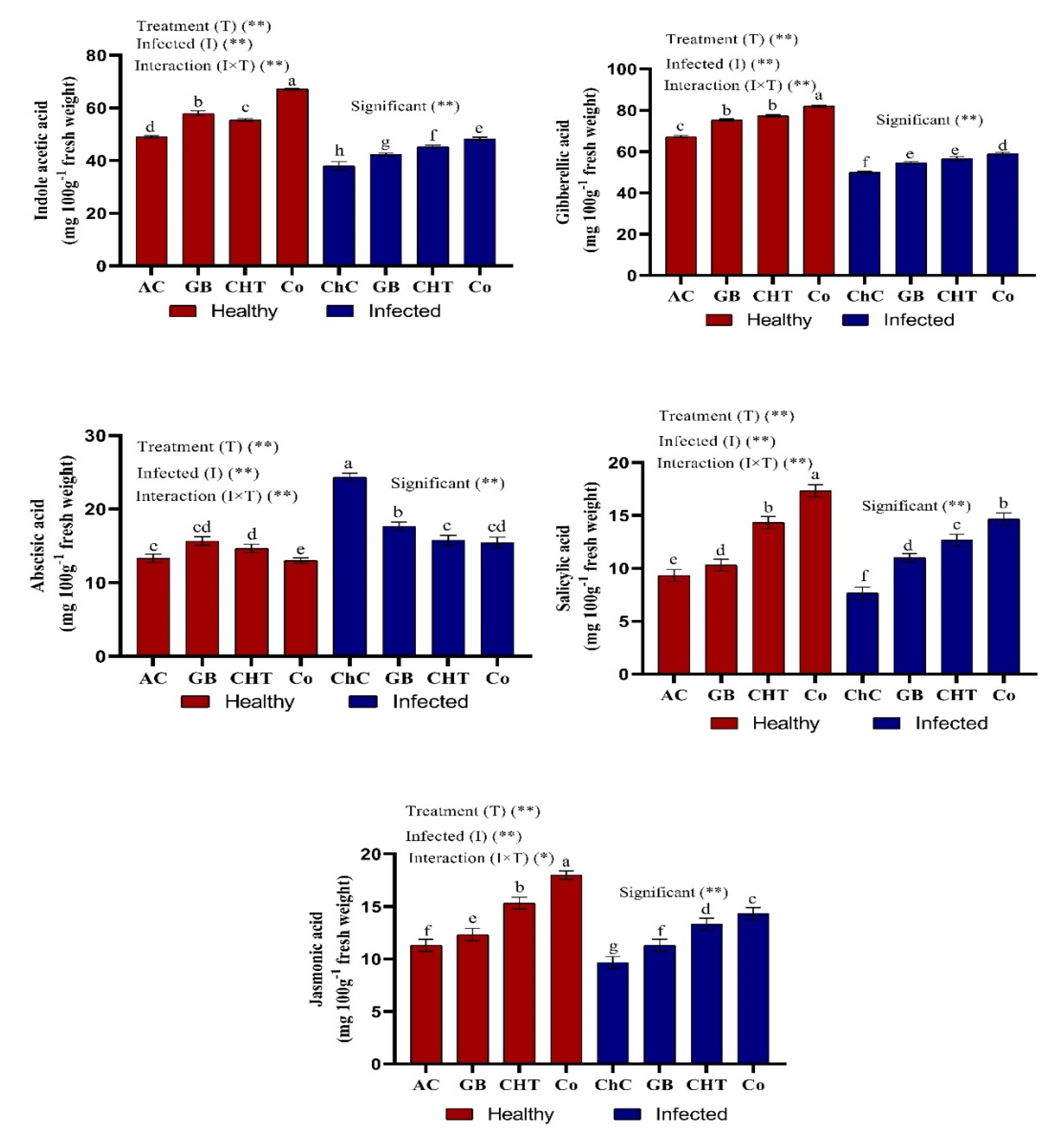
2.4. Endogenous Phytohormones
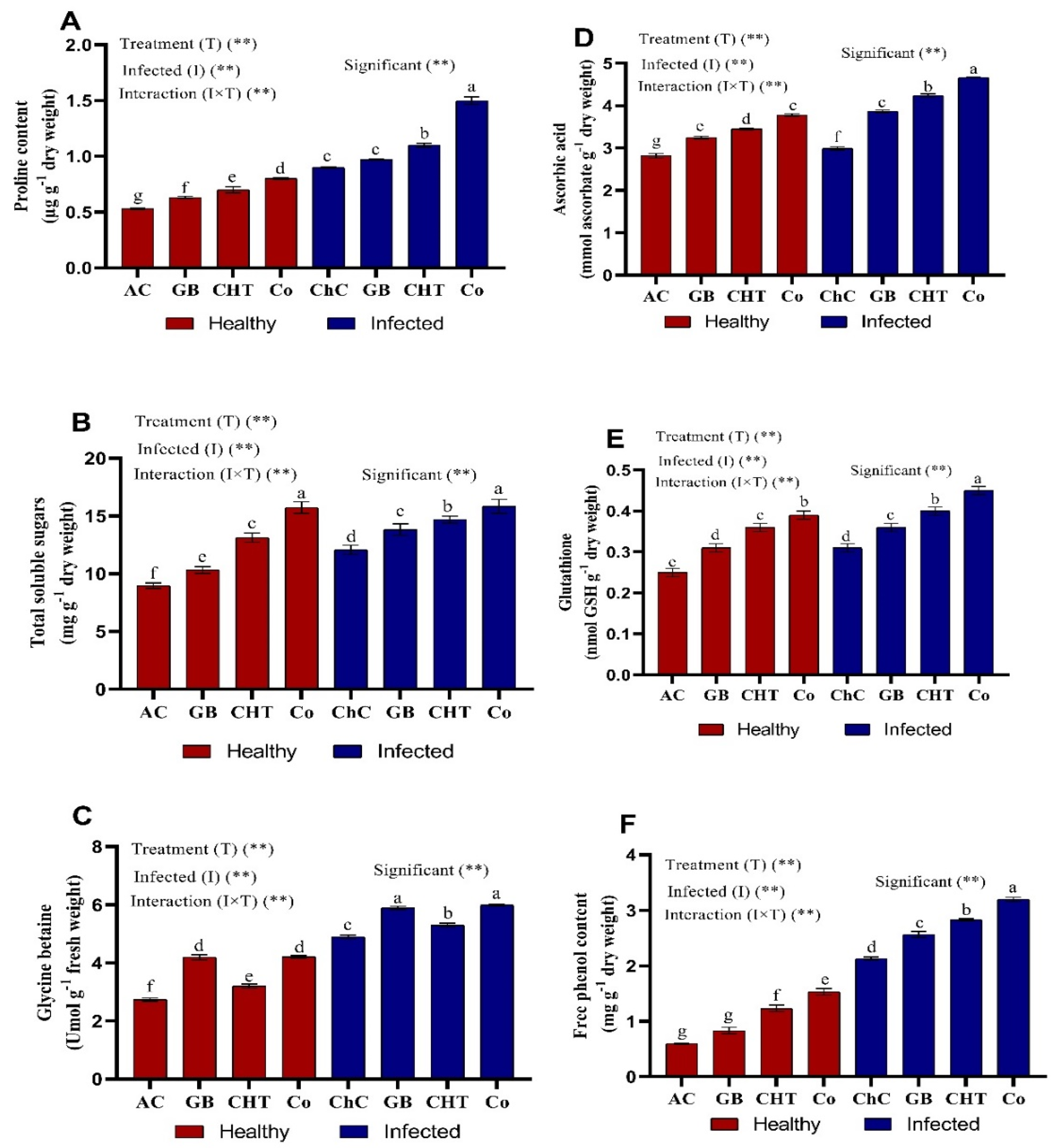
2.5. Osmolytes and Non-Enzymatic Antioxidants
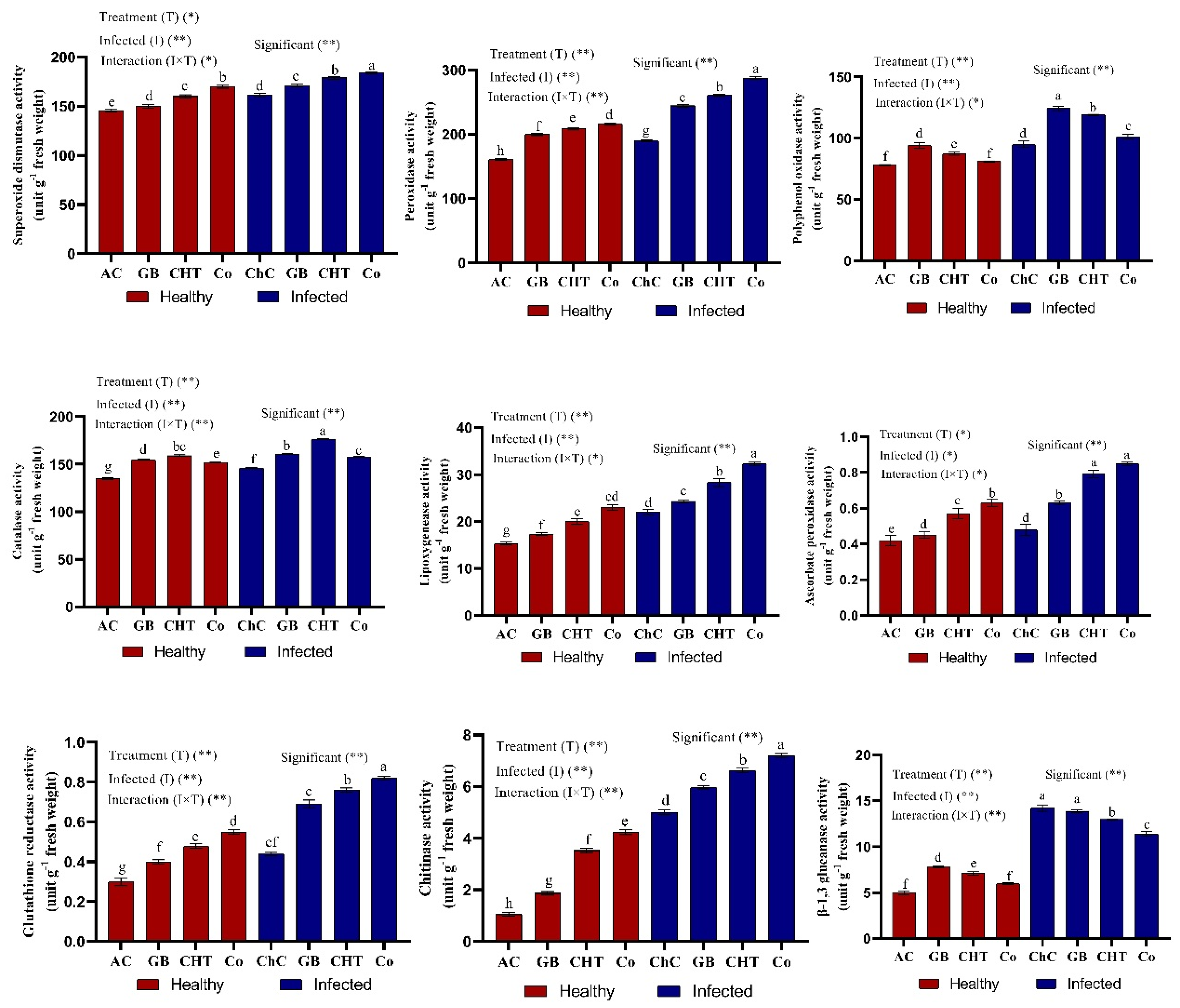
2.6. Antioxidant Enzymes
2.7. Gene Expression
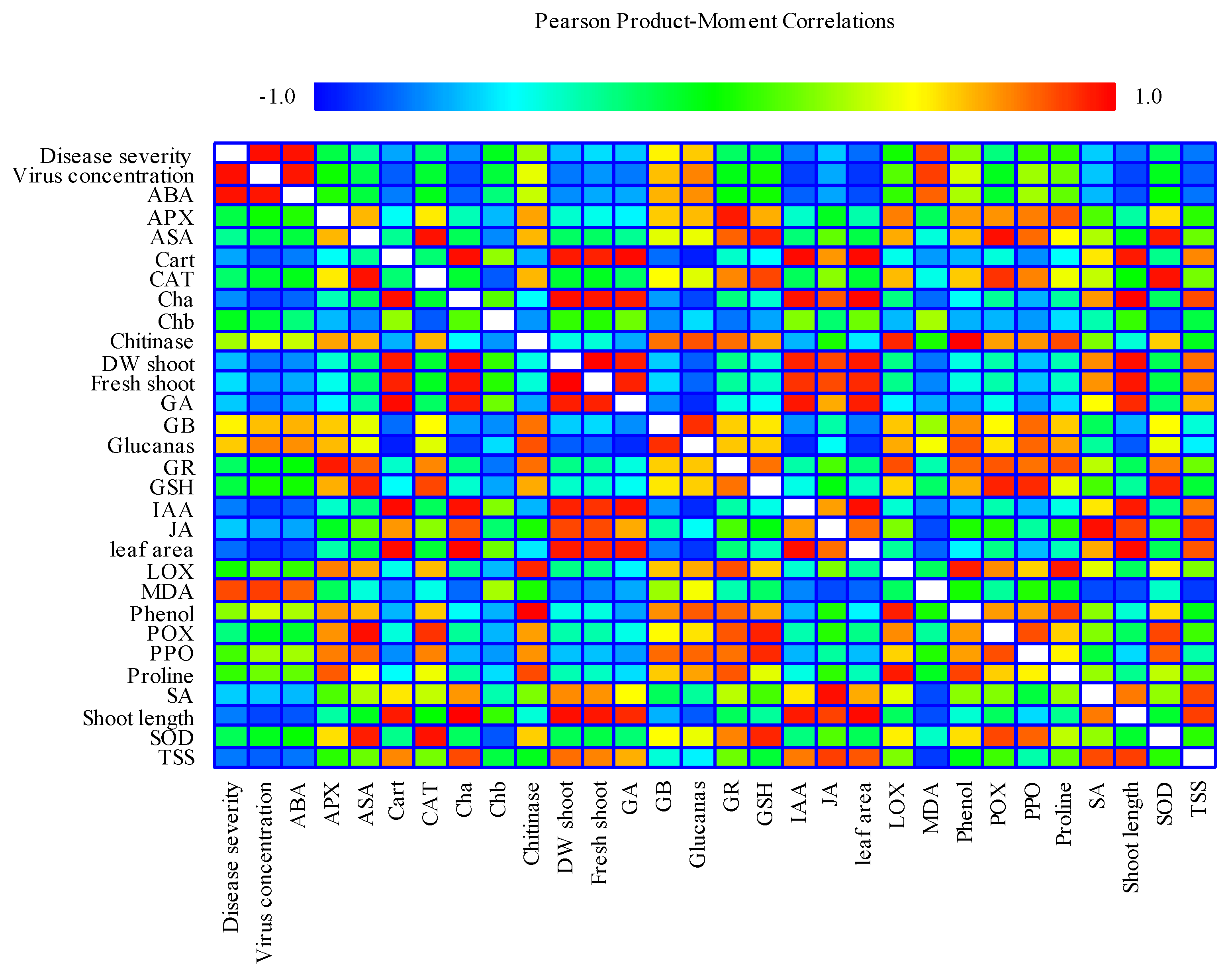
2.8. Correlation
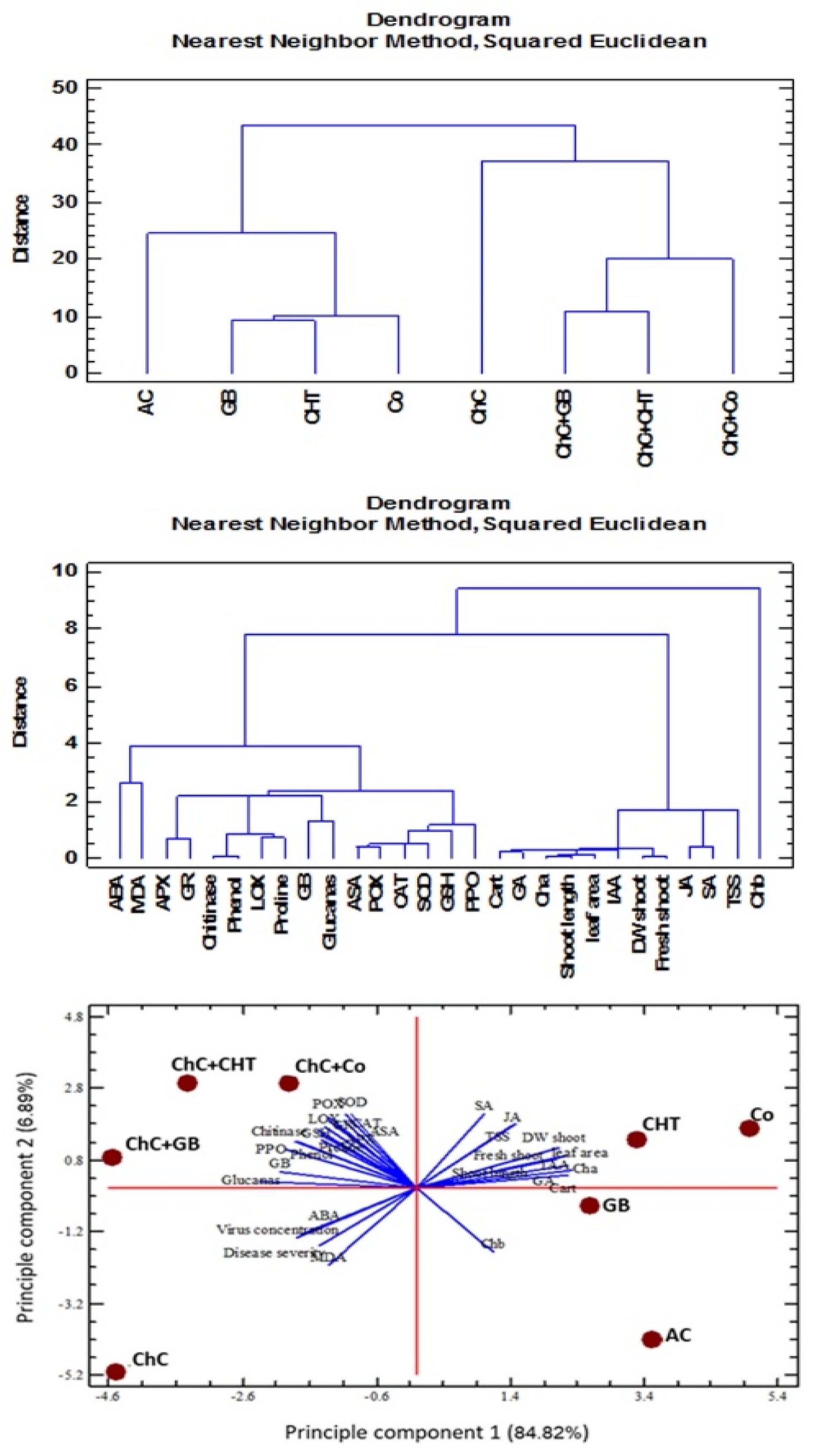
2.9. Multivariate Statistical Approach
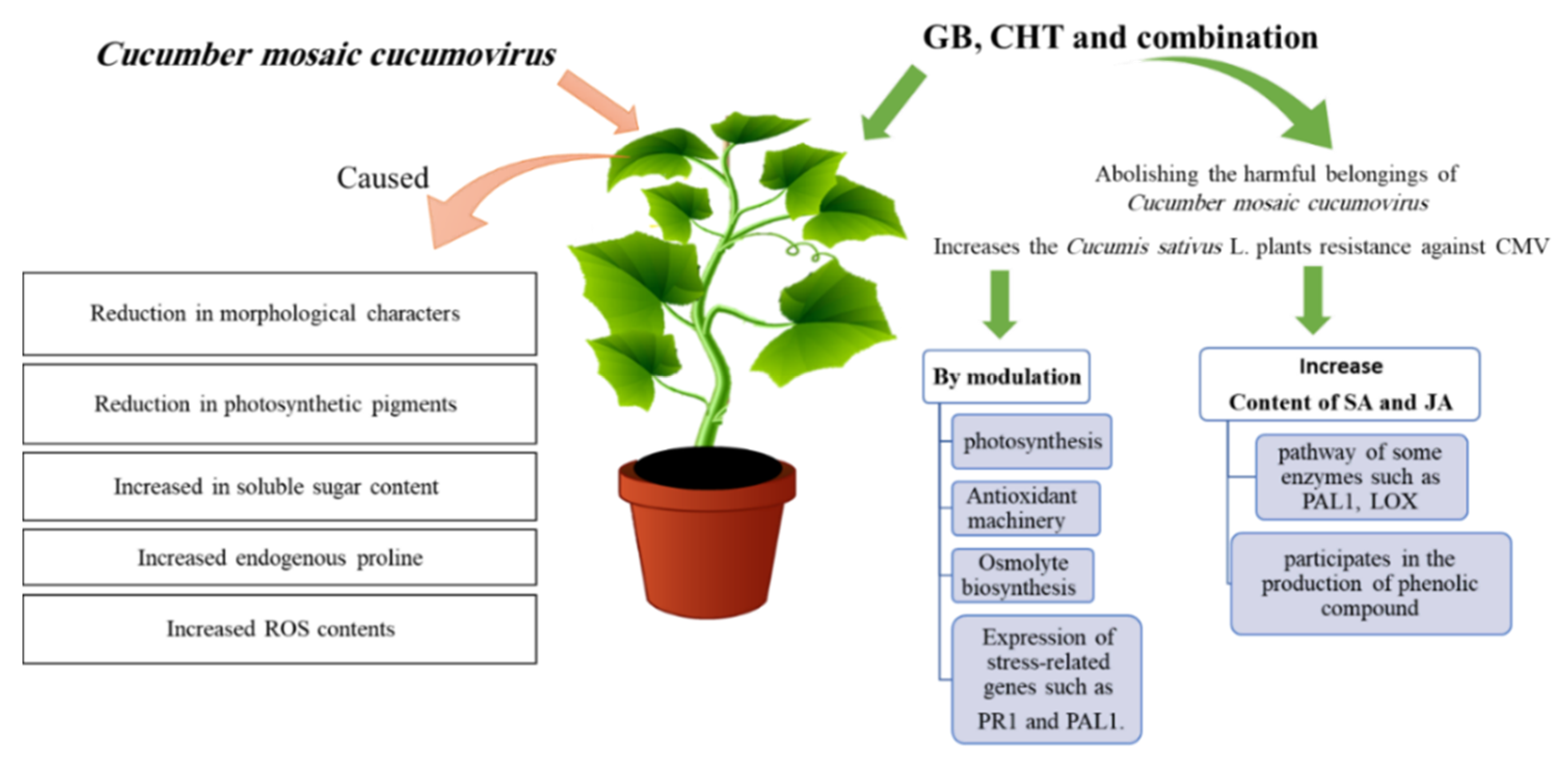
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Chemicals
4.2. CMV Inoculation
4.3. Experimental Design
4.4. Enzyme-Linked Immunosorbent Assay (ELISA)
4.5. qRT-PCR Analysis
4.6. Measurements
4.6.1. Growth Indices
4.6.2. Biochemical Measurements
Estimation of Photosynthetic Pigments
4.7. Determination of Lipid Peroxidation
4.8. Extraction, Separation and Estimation of Hormonal Content
4.8.1. Extraction
4.8.2. Separation
4.8.3. Estimation
4.9. Proline Content
4.10. Total Soluble Sugars Analysis
4.11. Glycine Betaine Analysis
4.12. Ascorbic Acid Analysis
4.13. Glutathione (GSH) Content
4.14. Free Phenol Analysis
4.15. Enzymes Activity
Extraction and Analysis
4.16. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roossinck, M.J. Cucumber mosaic virus, a model for RNA virus evolution. Mol. Plant. Pathol. 2001, 2, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Agrios, G.N. Plant disease caused by viruses. In Plant Pathology; Agrios, G.N., Ed.; Elsevier-Academic Press: San Diego, CA, USA, 2005; pp. 724–822. [Google Scholar]
- Montasser, M.S.; Dashti, N.H.; Ali, N.Y.A. Attenuation of viral symptoms and yield reduction responses to CMV infection in tomato crops subjected to plant growth promoting rhizobacteria. Fed. Am. Soc. Exp. Biol. J. 2011, 25, 714–765. [Google Scholar]
- Sofy, A.R.; Sofy, M.R.; Hmed, A.A.; El-Dougdoug, N.K. Potential effect of plant growth-promoting rhizobacteria (PGPR) on enhancing protection against viral diseases. In Field Crops: Sustainable Management by PGPR; Springer International Publishing: Cham, Switzerland, 2019; pp. 411–445. [Google Scholar] [CrossRef]
- Radwan, D.E.M.; Fayez, K.A.; Younis Mahmoud, S.; Hamad, A.; Lu, G. Physiological and metabolic changes of Cucurbita pepo leaves in response to Zucchini yellow mosaic virus (ZYMV) infection and salicylic acid treatments. Plant Physiol. Biochem. 2007, 45, 480–489. [Google Scholar] [CrossRef]
- Radwan, D.E.M.; Fayez, K.A.; Younis Mahmoud, S.; Lu, G. Modifications of antioxidant activity and protein composition of bean leaf due to Bean yellow mosaic virus infection and salicylic acid treatments. Acta Physiol. Plant. 2010, 32, 891–904. [Google Scholar] [CrossRef]
- Farahat, A.S.; El-Morsi, A.A.; Soweha, H.E.; Sofy, A.R.; Refaey, E.E. Metabolic changes of cucumber plants due to two CMV Egyptian isolates. Arab Univ. J. Agric. Sci. 2018, 26, 2019–2028. [Google Scholar] [CrossRef]
- Sofy, M.R.; Sharaf, A.M.A.; Noufl, M.; Sofy, A.R. Physiological and biochemical responses in Cucurbita pepo leaves associated with some elicitors-induced systemic resistance against Zucchhini yellow mosaic virus. Int. J. Mod. Bot. 2014, 4, 61–74. [Google Scholar]
- Sofy, A.R.; Mahfouze, S.A.; El-Enany, M.A. Isozyme markers for response of wild potato species to potato spindle tuber viroid Egyptian isolate. World Appl. Sci. J. 2013, 27, 1010–1022. [Google Scholar] [CrossRef]
- Riedle-Bauer, M. Role of reactive oxygen species and antioxidant enzymes in systemic Virus infections of plants. J. Phytopathol. 2000, 148, 297–302. [Google Scholar] [CrossRef]
- Wu, J.; Yang, R.; Yang, Z.; Yao, S.; Zhao, S.; Wang, Y.; Li, P.; Song, X.; Jin, L.; Zhou, T.; et al. ROS accumulation and antiviral defence control by microRNA528 in rice. Nat. Plants 2017, 3, 16203. [Google Scholar] [CrossRef]
- Treutter, D. Significance of flavonoids in plant resistance and enhancement of their biosynthesis. Plant Biol. 2005, 7, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Aly, A.A.; Mansour, M.T.M.; Mohamed, H.I. Association of increase in some biochemical components with flax resistance to powdery mildew. Gesunde Pflanzen 2017, 69, 47–52. [Google Scholar] [CrossRef]
- Asran, A.A.; Mohamed, H.I. Use of phenols, peroxidase, and polyphenoloxidase of seed to quantify resistance of cotton genotypes to fusarium wilt disease. Bangladesh J. Bot. 2014, 43, 353–357. [Google Scholar] [CrossRef]
- Aly, A.A.; Mansour, M.T.M.; Mohamed, H.I.; Abd-Elsalam, K.A. Examination of correlations between several biochemical components and powdery mildew resistance of flax cultivars. Plant Pathol. J. 2012, 28, 149–155. [Google Scholar] [CrossRef]
- Mohamed, H.I.; Aly, A.A.; Mansour, M.T.M.; El-Samawaty, A.M.A. Association of oxidative stress components with resistance to flax powdery mildew. Trop. Plant Pathol. 2012, 37, 386–392. [Google Scholar] [CrossRef]
- Boobis, A.R.; Ossendorp, B.C.; Banasiak, U.; Hamey, P.Y.; Sebestyen, I.; Moretto, A. Cumulative risk assessment of pesticide residues in food. Toxicol. Lett. 2008, 137–150. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, H.I.; Akladious, S.A. Changes in antioxidants potential, secondary metabolites and plant hormones induced by different fungicides treatment in cotton plants. Pest Biochem Physiol. 2017, 142, 117–122. [Google Scholar] [CrossRef]
- Mohamed, H.I.; El-Beltagi, H.S.; Aly, A.A.; Latif, H.H. The role of systemic and non-systemic fungicides on the physiological and biochemical parameters in Gossypium hirsutum plant, implications for defense responses. Fresenius Environ. Bull. 2018, 27, 8585–8593. [Google Scholar]
- Aly, A.A.; Mohamed, H.I.; Mansour, M.T.M.; Omar, M.R. Suppression of powdery mildew on flax by foliar application of essential oils. J. Phytopathol. 2013, 376–381. [Google Scholar] [CrossRef]
- Aktar, M.W.; Sengupta, D.; Chowdhury, A. Impact of pesticides use in agriculture: Their benefits and hazards. Interdiscip. Toxicol. 2009, 2, 1–12. [Google Scholar] [CrossRef]
- De Zwart, F.J.; Slow, S.; Payne, R.J.; Lever, M.; George, P.M.; Gerrard, J.A.; Chambers, S.T. Glycine betaine and glycine betaine analogues in common foods. Food Chem. 2003, 83, 197–204. [Google Scholar] [CrossRef]
- Ashraf, M.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Wang, S.; Yao, Q.; Tao, J.; Qiao, Y.; Zhang, Z. Co-ordinate expression of glycine betaine synthesis genes linked by the FMDV 2A region in a single open reading frame in Pichia pastoris. Appl. Microbiol. Biotechnol. 2007, 77, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Sui, Y.; Liu, J.; Wisniewski, M.; Droby, S.; Norelli, J.; Hershkovitz, V. Pretreatment of the yeast antagonist, Candida oleophila, with glycine betaine increases oxidative stress tolerance in the microenvironment of apple wounds. Int. J. Food Microbiol. 2012, 157, 45–51. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, G.; Li, P.; Yang, Q.; Chen, K.; Zhao, L.; Apaliya, M.T.; Gu, X.; Zhang, H. Mechanisms of glycine betaine enhancing oxidative stress tolerance and biocontrol efficacy of Pichia caribbica against blue mold on apples. Biol. Control. 2017, 108, 55–63. [Google Scholar] [CrossRef]
- Kochkina, Z.M.; Chirkov, S.N. Influence of the chitosan oligomer on the phage particles and reproduction of phage 1-97A in the culture of Bacillus thuringiensis. Microbiology 2001, 70, 706–710. [Google Scholar] [CrossRef]
- Chirkov, S.N. The antiviral activity of chitosan (review). Appl. Biochem. Microbiol. 2002, 38, 1–8. [Google Scholar] [CrossRef]
- Barber, M.S.; Bertram, R.E.; Ride, J.P. Chitin oligosaccharides elicit lignification in wounded wheat leaves. Physiol. Mol. Plant Pathol. 1989, 34, 3–12. [Google Scholar] [CrossRef]
- Felix, G.; Regenass, M.; Boller, T. Specific perception of subnanomolar concentrations of chitin fragments by tomato cells: Induction of extracellular alkalinization, changes in protein phosphorylation, and establishment of a refractory state. Plant J. 1993, 4, 307–316. [Google Scholar] [CrossRef]
- Kaku, H.; Shibuya, N.; Xu, P.; Aryan, A.P.; Fincher, G.B. N-acetylchitooligosaccharides elicit expression of a single (13)-beta-glucanase gene in suspension-cultured cells from barley (Hordeum vulgare). Physiol. Plant. 1997, 100, 111–118. [Google Scholar] [CrossRef]
- Yamada, A.; Shibuya, N.; Kodama, O.; Akatsuka, T. Induction of phytoalexin formation in suspension-cultured rice cells by N-Acetyl-chitooligosaccharides. Biosci. Biotechnol. Biochem. 1993, 57, 405–409. [Google Scholar] [CrossRef]
- Kuchitsu, K.; Kosaka, H.; Shiga, T.; Shibuya, N. EPR evidence for generation of hydroxyl radical triggered by N-acetylchitooligosaccharide elicitor and a protein phosphatase inhibitor in suspension-cultured rice cells. Protoplasma 1995, 188, 138–142. [Google Scholar] [CrossRef]
- Nojiri, H.; Sugimori, M.; Yamane, H.; Nishimura, Y.; Yamada, A.; Shibuya, N.; Kodama, O.; Murofushi, N.; Omori, T. Involvement of jasmonic acid in elicitor-induced phytoalexin production in suspension-cultured rice cells. Plant Physiol. 1996, 110, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Megahed, A.A.; El-Dougdoug, K.A.; Othman, B.A.; Lashin, S.M.; Ibrahim, M.A.; Sofy, A.R. A new Egyptian satellite strain of Cucumber mosaic cucumovirus. Int. J. Virol. 2012, 8, 240–257. [Google Scholar] [CrossRef][Green Version]
- Sofy, A.R.; Soliman, A.M. Molecular identification of a Cucumber mosaic virus subgroup I Egyptian isolate from geranium based on bioinformatics analysis of CP gene sequence Egypt. Egypt. J. Virol. 2011, 8, 178–194. [Google Scholar]
- El-Borollosy, A.M.; Oraby, M.M. Induced systemic resistance against Cucumber mosaic cucumovirus and promotion of cucumber growth by some plant growth-promoting rhizobacteria. Ann. Agric. Sci. 2012, 57, 91–97. [Google Scholar] [CrossRef]
- Megahed, A.A.; El-Dougdoug, K.A.; Othman, B.A.; Lashin, S.M.; Hassanin, M.D.; Ibrahim, M.A.; Sofy, A.R. Molecular identification and analysis of coat protein gene of Cucucmber mosaic cucumovirus sugar beet Egyptian isolate. Int. J. Plant Pathol. 2014, 5, 70–83. [Google Scholar] [CrossRef][Green Version]
- Abd El-Aziz, M.H.; Younes, H.A. Detection of Cucumber mosaic cucumovirus in infected cowpea plants (Vigna unguiculata L.) from northern Egypt. Nov. Res. Microbiol. J. 2019, 3, 326–340. [Google Scholar] [CrossRef]
- Bari, R.; Jones, J.D.G. Role of plant hormones in plant defence responses. Plant Mol. Biol. 2009, 69, 473–488. [Google Scholar] [CrossRef]
- Vitti, A.; Nuzzaci, M.; Scopa, A.; Tataranni, G.; Remans, T.; Vangronsveld, J.; Sofo, A. Auxin and cytokinin metabolism and root morphological modifications in Arabidopsis thaliana seedlings infected with Cucumber mosaic virus (CMV) or exposed to cadmium. Int. J. Mol. Sci. 2013, 14, 6889–6902. [Google Scholar] [CrossRef]
- Lavanya, N.S.; Amruthesh, N.K. Glycine betaine mediated disease resistance against Sclerospora graminicola in Pearl Millet. J. Appl. Biol. Biotechnol. 2017, 5. [Google Scholar] [CrossRef]
- Beffa, R.S.; Hofer, R.M.; Thomas, M.; Meins, F. Decreased susceptibility to viral disease of [beta]-1,3-glucanase-deficient plants generated by antisense transformation. Plant Cell 1996, 8, 1001–1011. [Google Scholar] [CrossRef] [PubMed]
- Vitti, A.; Pellegrini, E.; Nali, C.; Lovelli, S.; Sofo, A.; Valerio, M.; Scopa, A.; Nuzzaci, M. Trichoderma harzianum T-22 induces systemic resistance in tomato infected by Cucumber Mosaic Virus. Front. Plant Sci. 2016, 7, 1520. [Google Scholar] [CrossRef] [PubMed]
- Sofy, M.R.; Sharaf, A.M.A.; El-Nosary, M.E.; Sofy, A.R. Salix alba extract induces systemic resistance in Cucumis sativus infected by Cucumber mosaic virus. Nature and Science 2018, 16, 107–113. [Google Scholar]
- Rahoutei, J.; Garcia-Luque, I.; Baron, M. Inhibition of photosynthesis by viral infection: Effect on PSII structure and function. Physiol. Plant. 2000, 110, 286–292. [Google Scholar] [CrossRef]
- Balachandran, S.; Hurry, V.M.; Kelley, S.E.; Osmond, C.B.; Robinson, S.A.; Rohozinski, J.; Seaton, G.G.R.; Sims, D.A. Concepts of plant biotic stress. Some insights into the stress physiology of virus-infected plants, from the perspective of photosynthesis. Physiol. Plant. 1997, 100, 203–213. [Google Scholar] [CrossRef]
- Fan, H.; Song, B.; Bhadury, P.S.; Jin, L.; Hu, D.; Yang, S. Antiviral activity and mechanism of action of novel thiourea containing chiral phosphonate on Tob. Mosaic Virus. Int. J. Mol. Sci. 2011, 12, 4522–4535. [Google Scholar] [CrossRef]
- Zhao, N.; Zhang, Y.; Li, Q.; Li, R.; Xia, X.; Qin, X.; Guo, H. Identification and expression of a stearoyl-ACP desaturase gene responsible for oleic acid accumulation in Xanthoceras sorbifolia seeds. Plant Physiol. Biochem. 2015, 87, 9–16. [Google Scholar] [CrossRef]
- Hadwiger, L.A. Anatomy of a nonhost disease resistance response of pea to Fusarium solani: PR gene elicitation via DNase, chitosan and chromatin alterations. Front. Plant Sci. 2015, 6, 373. [Google Scholar] [CrossRef]
- Malekzadeh, P. Influence of exogenous application of glycinebetaine on antioxidative system and growth of salt-stressed soybean seedlings (Glycine max L.). Physiol. Mol. Biol. Plants 2015, 21, 225–232. [Google Scholar] [CrossRef]
- Bargmann, B.O.; Laxalt, A.M.; ter Riet, B.; Testerink, C.; Merquiol, E.; Mosblech, A.; Leon-Reyes, A.; Pieterse, C.M.; Haring, M.A.; Heilmann, I.; et al. Reassessing the role of phospholipase D in the Arabidopsis wounding response. Plant Cell Env. 2009, 32, 837–850. [Google Scholar] [CrossRef]
- Tian, M.; Sasvari, Z.; Gonzalez, P.A.; Friso, G.; Rowland, E.; Liu, X.M.; van Wijk, K.J.; Nagy, P.D.; Klessig, D.F. Salicylic acid inhibits the replication of Tomato bushy stunt virus by directly targeting a host component in the replication complex. Mol. Plant Microbe Interact. 2015, 28, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Faoro, F.; Gozzo, F. Is modulating virus virulence by induced systemic resistance realistic? Plant Sci. 2015, 234, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Qin, Q.; Wang, Y.; Pu, Y.; Liu, L.; Wen, X.; Ji, S.; Wu, J.; Wei, C.; Ding, B.; et al. Rice dwarf virus P2 protein hijacks auxin signaling by directly targeting the rice OsIAA10 protein, enhancing viral infection and disease development. PLoS Pathog. 2016, 12, e1005847. [Google Scholar] [CrossRef] [PubMed]
- De Saint Germain, A.; Ligerot, Y.; Dun, E.A.; Pillot, J.P.; Ross, J.J.; Beveridge, C.A.; Rameau, C. Strigolactones stimulate internode elongation independently of gibberellins. Plant Physiol. 2013, 163, 1012–1025. [Google Scholar] [CrossRef]
- Oka, K.; Kobayashi, M.; Mitsuhara, I.; Seo, S. Jasmonic acid negatively regulates resistance to Tobacco mosaic virus in tobacco. Plant Cell Physiol. 2013, 54, 1999–2010. [Google Scholar] [CrossRef]
- Creelman, R.A.; Mullet, J.E. Biosynthesis and action of jasmonates in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1997, 48, 355–381. [Google Scholar] [CrossRef]
- Alazem, M.; Lin, K.-Y.; Lin, N.-S. The abscisic acid pathway has multifaceted effects on the accumulation of Bamboo Mosaic Virus. Mol. Plant Microbe Interact. 2014, 27, 177–189. [Google Scholar] [CrossRef]
- Alazem, M.; Lin, N.-S. Antiviral roles of abscisic acid in plants. Front. Plant Sci. 2017, 8, 1760. [Google Scholar] [CrossRef]
- De Storme, N.; Geelen, D. Callose homeostasis at plasmodesmata: Molecular regulators and developmental relevance. Front. Plant Sci. 2014, 5, 138. [Google Scholar] [CrossRef]
- Oide, S.; Bejai, S.; Staal, J.; Guan, N.; Kaliff, M.; Dixelius, C. A novel role of PR2 in abscisic acid (ABA) mediated, pathogen-induced callose deposition in Arab. Thaliana. New Phytol. 2013, 200, 1187–1199. [Google Scholar] [CrossRef]
- Durner, J.; Wendehenne, D.; Klessig, D.F. Defense gene induction in tobacco by nitric oxide, cyclic GMP, and cyclic ADP-ribose. Proc. Natl. Acad. Sci. USA 1998, 95, 10328–10333. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zheng, Z.; Huang, J.; Lai, Z.; Fan, B. Biosynthesis of salicylic acid in plants. Plant Signal. Behav. 2009, 4, 493–496. [Google Scholar] [CrossRef] [PubMed]
- Malamy, J.; Klessig, D.F. Salicylic acid and plant disease resistance. Plant J. 1992, 2, 643–654. [Google Scholar] [CrossRef]
- Metraux, J.P.; Signer, H.; Ryals, J.; Ward, E.; Wyss-Benz, M.; Gaudin, J.; Raschdorf, K.; Schmid, E.; Blum, W.; Inverardi, B. Increase in salicylic acid at the onset of systemic acquired resistance in cucumber. Science 1990, 250, 1004–1006. [Google Scholar] [CrossRef]
- Hakmaoui, A.; Perez-Bueno, M.L.; Garcia-Fontana, B.; Camejo, D.; Jimenez, A.; Sevilla, F.; Baron, M. Analysis of the antioxidant response of Nicotiana benthamiana to infection with two strains of Pepper Mild Mottle Virus. J. Exp. Bot. 2012, 63, 5487–5496. [Google Scholar] [CrossRef]
- Gullner, C.; Tóbiás, I.; Fodor, J.; Kömives, U. Elevation of glutathione level and activation of glutathione-related enzymes affect virus infection in tobacco. Free Radic. Res. 2016, 31, 155–161. [Google Scholar] [CrossRef]
- Pavet, V.; Olmos, E.; Kiddle, G.; Mowla, S.; Kumar, S.; Antoniw, J.; Alvarez, M.E.; Foyer, C.H. Ascorbic acid deficiency activates cell death and disease resistance responses in Arab. Plant Physiol. 2005, 139, 1291–1303. [Google Scholar] [CrossRef]
- Anuradha, C.; Selvarajan, R.; Vasantha, S.; Suresha, G.S. Biochemical Characterization of compatible plant virus interaction: A case study with Bunchy top virus-banana host-pathosystem. Plant Pathol. J. 2015, 14, 212–222. [Google Scholar] [CrossRef]
- Hammerschmidt, R.; Nuckles, E.M.; Kuć, J. Association of enhanced peroxidase activity with induced systemic resistance of cucumber to Colletotrichum Lagenarium. Physiol. Plant Pathol. 1982, 20, 73–82. [Google Scholar] [CrossRef]
- Campos-Vargas, R.; Saltveit, M.E. Involvement of putative chemical wound signals in the induction of phenolic metabolism in wounded lettuce. Physiol. Plant. 2002, 114, 73–84. [Google Scholar] [CrossRef]
- Wang, F.; Feng, G.; Chen, K. Defense responses of harvested tomato fruit to burdock fructooligosaccharide, a novel potential elicitor. Postharvest Biol. Technol. 2009, 52, 110–116. [Google Scholar] [CrossRef]
- Klessig, D.F.; Malamy, J. The salicylic acid signal in plants. Plant Mol. Biol. 1994, 26, 1439–1458. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef] [PubMed]
- Mathioudakis, M.M.; Veiga, R.S.L.; Canto, T.; Medina, V.; Mossialos, D.; Makris, A.M.; Livieratos, I. Pepino mosaic virus triple gene block protein 1 (TGBp1) interacts with and increases tomato catalase 1 activity to enhance virus accumulation. Mol. Plant Pathol. 2013, 14, 589–601. [Google Scholar] [CrossRef]
- Leon, J.; Lawton, M.A.; Raskin, I. Hydrogen peroxide stimulates salicylic acid biosynthesis in tobacco. Plant Physiol. 1995, 108, 1673–1678. [Google Scholar] [CrossRef]
- Ryu, C.-M.; Murphy, J.F.; Mysore, K.S.; Kloepper, J.W. Plant growth-promoting rhizobacteria systemically protect Arabidopsis thaliana against Cucumber mosaic virus by a salicylic acid and NPR1-independent and jasmonic acid-dependent signaling pathway. Plant J. 2004, 39, 381–392. [Google Scholar] [CrossRef]
- Yang, X.; Kang, L.; Tien, P.O. Resistance of tomato infected with cucumber mosaic virus satellite RNA to potato spindle tuber viroid. Ann. Appl. Biol. 1996, 129, 543–551. [Google Scholar] [CrossRef]
- Zehnder, G.W.; Yao, C.; Murphy, J.F.; Sikora, E.R.; Kloepper, J.W. Induction of resistance in tomato against Cucumber mosaic cucumovirus by plant growth-promoting rhizobacteria. BioControl 2000, 45, 127–137. [Google Scholar] [CrossRef]
- Derbalah, A.S.H.; Elsharkawy, M.M. A new strategy to control Cucumber mosaic virus using fabricated NiO-nanostructures. J. Biotechnol. 2019, 306, 134–141. [Google Scholar] [CrossRef]
- Wang, X.; Tang, C.; Zhang, G.; Li, Y.; Wang, C.; Liu, B.; Qu, Z.; Zhao, J.; Han, Q.; Huang, L.; et al. cDNA-AFLP analysis reveals differential gene expression in compatible interaction of wheat challenged with Puccinia striiformis f. sp. tritici. Bmc Genom. 2009, 10, 289. [Google Scholar] [CrossRef]
- Pu, X.; Xie, B.; Li, P.; Mao, Z.; Ling, J.; Shen, H.; Zhang, J.; Huang, N.; Lin, B. Analysis of the defence-related mechanism in cucumber seedlings in relation to root colonization by nonpathogenic Fusarium oxysporum CS-20. Fems Microbiol. Lett. 2014, 355, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Vernon, L.; Seely, G. The Chlorophylls; Academic Press: New York, NY, USA, 1966. [Google Scholar]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1987; Volume 148, pp. 350–382. [Google Scholar]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Knegt, E.; Bruinsma, J. A rapid, sensitive and accurate determination of indolyl-3-acetic acid. Phytochemistry 1973, 12, 753–756. [Google Scholar] [CrossRef]
- Lee, B.; Martin, P.; Bangerth, F. The effect of sucrose on the levels of abscisic acid, indoleacetic acid and zeatin/zeatin riboside in wheat ears growing in liquid culture. Physiol. Plant. 1989, 77, 73–80. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Irigoyen, J.J.; Einerich, D.W.; Sanchez-Diaz, M. Water stress induced changes in concentrations of proline and total soluble sugars in nodulated alfalfa (Medicago sativd) plants. Physiol. Plant. 1992, 84, 55–60. [Google Scholar] [CrossRef]
- Grieve, C.M.; Grattan, S.R. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil 1983, 70, 303–307. [Google Scholar] [CrossRef]
- Mukherjee, S.P.; Choudhuri, M.A. Implications of water stress-induced changes in the levels of endogenous ascorbic acid and hydrogen peroxide in Vigna seedlings. Physiol. Plant. 1983, 58, 166–170. [Google Scholar] [CrossRef]
- Griffith, O.W. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal. Biochem. 1980, 106, 207–212. [Google Scholar] [CrossRef]
- Galicia, L.N.; Rosales, E.; A Palacios Rojas, N. Laboratory Protocols: Maize Nutrition Quality and Plant Tissue Analysis Laboratory; CIMMYT: Mexico City, Mexico, 2009. [Google Scholar]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Bergmeyer, H.U.; Gawehn, K. Methods of Enzymatic Analysis; Academic Press: Cambridge, MA, USA, 1974; Volume 2. [Google Scholar]
- Todd, J.F.; Paliyath, G.; Thompson, J.E. Characteristics of a membrane-associated lipoxygenase in tomato fruit. Plant Physiol. 1990, 94, 1225–1232. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Cao, X.; Lu, Y.; Wang, X. Effects of rare earth metal ions and their EDTA complexes on antioxidant enzymes of fish liver. Bull. Environ. Contam. 2000, 65, 357–365. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in Spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar] [CrossRef]
- Jiang, M.; Zhang, J. Water stress-induced abscisic acid accumulation triggers the increased generation of reactive oxygen species and up-regulates the activities of antioxidant enzymes in maize leaves. J. Exp. Bot. 2002, 53, 2401–2410. [Google Scholar] [CrossRef] [PubMed]
- Monreal, J.; Reese, E.T. The chitinase of Serratia marcescens. Can. J. Microbiol. 1969, 15, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Abeles, F.B.; Forrence, L.E. Temporal and hormonal control of β-1,3-glucanase in Phaseolus vulgaris L. Plant Physiol. 1970, 45, 395–400. [Google Scholar] [CrossRef]
- Gomez, K.A.; Gomez, A.A. Statistical Procedures for Agricultural Research; John Wiley & Sons Inc.: Singapore, 1984. [Google Scholar]
Sample Availability: Not available. |


| Treatments | Virus Concentration | Percentage of Infection (%) | Disease Severity (%) |
|---|---|---|---|
| Challenge control (ChC) | 1.30 ± 0.04 a | 95 ± 0.33 a | 70 ± 0.24 a |
| GB + Virus | 0.48 ± 0.01 b | 24 ± 0.41 b | 14.33 ± 0.38 b |
| CHT + Virus | 0.35± 0.03 c | 15 ± 0.25 c | 6.22 ± 0.52 c |
| Combination (GB+CHT) + Virus | 0.28 ± 0.02 d | 10± 0.20 d | 2.88± 0.33 d |
| Gene | Forward Primer (´5–´3) | Reverse Primer (´5–´3) |
|---|---|---|
| PR1 | TGCTCAACAA ATGCGAACC | TCATCCACCCACAACTGAAC |
| PAL1 | ATGGAGGCAACTTCCAAGGA | CCATGGCAATCTCAGCACCT |
| ef1-∝ | ACTGTGCTGTCCTCATTATTG | AGGGTGAAAGCAAGAAGAGC |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sofy, A.R.; Dawoud, R.A.; Sofy, M.R.; Mohamed, H.I.; Hmed, A.A.; El-Dougdoug, N.K. Improving Regulation of Enzymatic and Non-Enzymatic Antioxidants and Stress-Related Gene Stimulation in Cucumber mosaic cucumovirus-Infected Cucumber Plants Treated with Glycine Betaine, Chitosan and Combination. Molecules 2020, 25, 2341. https://doi.org/10.3390/molecules25102341
Sofy AR, Dawoud RA, Sofy MR, Mohamed HI, Hmed AA, El-Dougdoug NK. Improving Regulation of Enzymatic and Non-Enzymatic Antioxidants and Stress-Related Gene Stimulation in Cucumber mosaic cucumovirus-Infected Cucumber Plants Treated with Glycine Betaine, Chitosan and Combination. Molecules. 2020; 25(10):2341. https://doi.org/10.3390/molecules25102341
Chicago/Turabian StyleSofy, Ahmed R., Rehab A. Dawoud, Mahmoud R. Sofy, Heba I. Mohamed, Ahmed A. Hmed, and Noha K. El-Dougdoug. 2020. "Improving Regulation of Enzymatic and Non-Enzymatic Antioxidants and Stress-Related Gene Stimulation in Cucumber mosaic cucumovirus-Infected Cucumber Plants Treated with Glycine Betaine, Chitosan and Combination" Molecules 25, no. 10: 2341. https://doi.org/10.3390/molecules25102341
APA StyleSofy, A. R., Dawoud, R. A., Sofy, M. R., Mohamed, H. I., Hmed, A. A., & El-Dougdoug, N. K. (2020). Improving Regulation of Enzymatic and Non-Enzymatic Antioxidants and Stress-Related Gene Stimulation in Cucumber mosaic cucumovirus-Infected Cucumber Plants Treated with Glycine Betaine, Chitosan and Combination. Molecules, 25(10), 2341. https://doi.org/10.3390/molecules25102341








