1. Introduction
Curcumin is a natural phenol and has been extensively investigated as potential drug candidate for various illnesses and medical conditions [
1]. However, the compound has been classified as a pan-assay interference compound (PAINS) and an invalid metabolic panacea (IMPS) [
1]. PAINS are compounds that show activity in different types of assay mainly through interfering with the assay itself while IMPS are reagents that display activity against virtually any biological target. Despite this and other drawbacks (chemical instability, low bioavailability, non-selectivity and toxicity), curcumin is still subject of intense research and about 50 papers are published each week on biological interactions of the compound [
1].
Previously, it was shown that curcumin display anti-proliferative activity against the protozoan parasite
Trypanosoma brucei, the causative agent of sleeping sickness in humans and nagana disease in livestock [
2]. In search for the biological target involved in the trypanocidal activity of curcumin, the effect of the compound on rhodesain, the major lysosomal cathepsin L cysteine protease in
T. brucei, has been recently investigated [
3]. The enzyme is essential for the survival of the parasite and a valid drug target [
4]. It was shown that curcumin was able to inhibit rhodesain and it was suggested that this inhibition was irreversible [
3]. This conclusion was based on a weak non-linear relationship between substrate hydrolysis and incubation time (with increasing incubation time the hydrolysis of substrate decreased slightly) and the lack of recovery of enzyme activity after dilution of rhodesain pre-incubated with curcumin [
3,
5]. However, it was also recently shown that the inhibition of rhodesain by curcumin seemed to be reversible [
6]. A 1:4 dilution of a reaction mixture containing rhodesain and 10 μM curcumin resulted in a 4.7-fold increase in activity [
6]. In order to prove conclusively that curcumin is a reversible inhibitor, kinetic studies to determine the inhibitor type of the compound were carried out. In addition, further investigations were conducted to provide explanations for the apparent irreversible inactivation of rhodesain by curcumin recently observed [
3,
5]. The results of this study revealed that curcumin is a reversible non-competitive inhibitor of rhodesain, a new finding that disproves unequivocally previous claims that curcumin is an irreversible inhibitor. This study also showed that it is important to select the correct assay conditions to measure enzyme activity and to consider the solubility properties of inhibitors otherwise incorrect data will be obtained leading to a misinterpretation of results.
2. Results and Discussion
The activity of rhodesain was determined with the fluorogenic substrate benzyloxycarbonyl-phenylalanyl-arginyl-7-amido-4-methyl coumarin (Z-FR-AMC), a substrate that is cleaved by mammalian and trypanosome cathepsin L cysteine proteases [
7,
8].
Time course experiment revealed that the inhibition of rhodesain by curcumin was time independent. In the presence of 6 μM curcumin (a concentration close to the IC
50 value for the inhibition of rhodesain by curcumin, see below), the inhibition of the activity of rhodesain was linear with respect to time (
Figure 1). The correlation coefficient of the readings was 0.9997 confirming a strong linear association between substrate hydrolysis and incubation time. The same correlation coefficient was also determined for the control reaction (
Figure 1) indicating that there was no difference in the linearity of the readings for the substrate hydrolysis in the presence and absence of curcumin. In contrast, when rhodesain was incubated with the established irreversible cysteine protease inhibitor CAA0255 [
9] at 0.1 μM (a concentration below the IC
50 value for the inhibition of rhodesain [
4]), the activity of the enzyme was quickly completely inhibited (
Figure 1 insert). Within 5 min of incubation, the activity of rhodesain was inhibited by >90%.
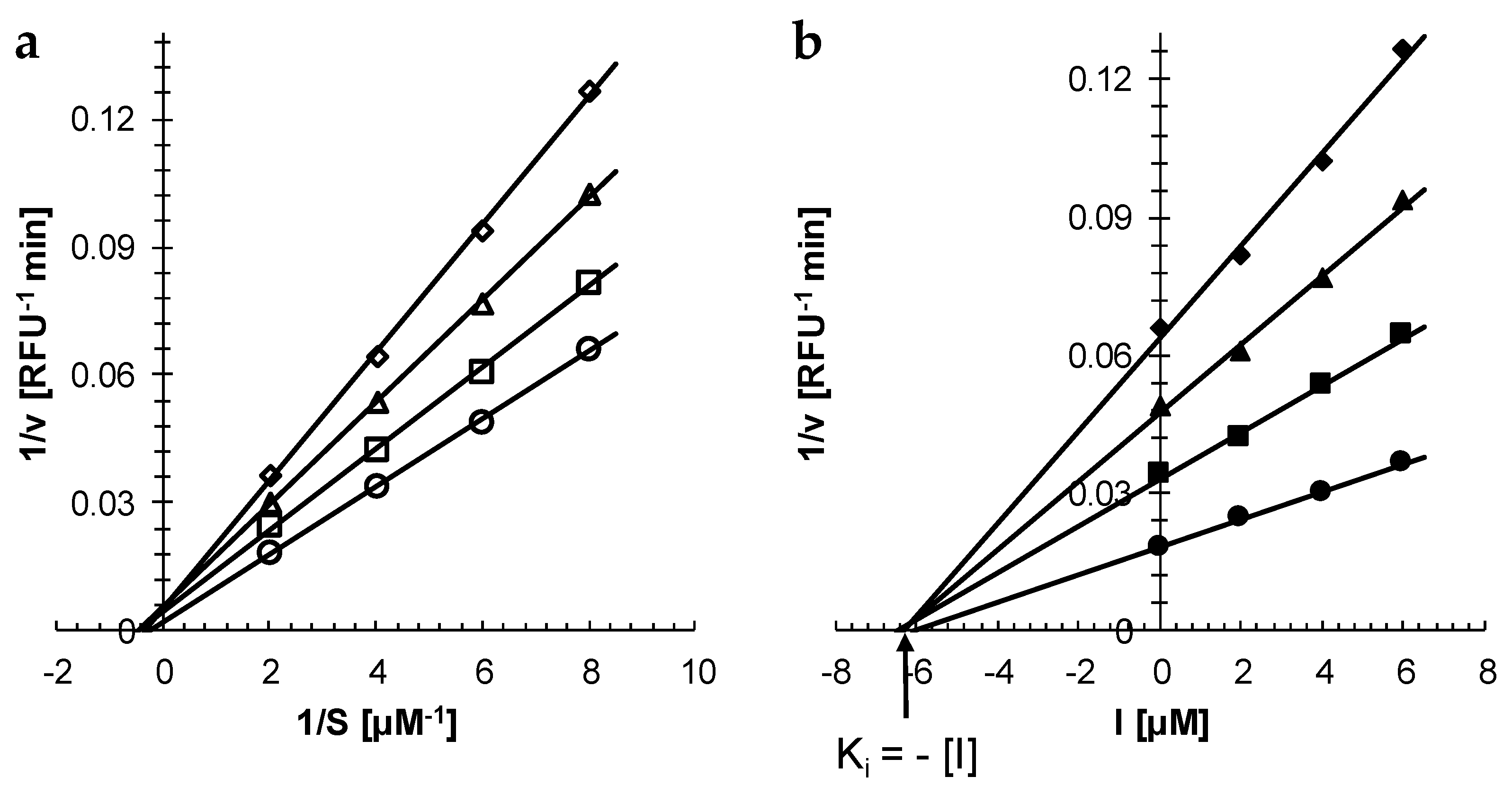
After establishing that curcumin is a reversible inhibitor of rhodesain (see above and [
6]), kinetic studies to determine the inhibitor type of the compound were carried out. Double reciprocal analysis (Lineweaver–Burk plot) gave a family of lines with increasing slopes as the curcumin concentration increased (
Figure 2a). The lines converged to the same point on the x-axis indicating a non-competitive inhibition mechanism (
Figure 2a). Plotting the reciprocal velocity (1/v) against the inhibitor concentration (Dixon plot) gave again a family of lines that met in a single point on the x-axis confirming that curcumin is indeed a non-competitive inhibitor of rhodesain (
Figure 2b). From the point of intersection, the apparent inhibitor constant K
i for curcumin was determined to be 5.5 ± 1.4 μM (n = 3).
As for a non-competitive inhibitor, the K
i value is equal to the IC
50 value [
10]; the IC
50 value of curcumin for the inhibition of rhodesain was determined next. The compound inhibited the activity of rhodesain in a dose-dependent manner with an IC
50 value of 5.6 ± 0.5 μM (n = 3) (
Figure 3). The IC
50 value was not statistically significantly different from the K
i value (Student’s
t test;
p = 0.971). This finding confirmed that curcumin is indeed a non-competitive inhibitor of rhodesain.
Having shown that curcumin is a reversible, non-competitive inhibitor of rhodesain, the question remains why previously a lack of recovery of activity after pre-incubation of the resting enzyme with the compound was found [
3,
5]. As the incubation of the active enzyme (i.e., in the presence of substrate) with curcumin is reversible [
6], one could conclude that the compound binds with different affinities to the free enzyme and the enzyme-substrate complex. However, this explanation can be excluded as a non-competitive inhibitor binds equally well to the enzyme whether or not it has bound the substrate. A more likely reason for the observed apparent irreversible inhibitory activity of curcumin is the very low water solubility of the compound, which is just 0.6 μg/mL (1.63 μM) [
11]. In this context, it is important to note that in the recent studies rhodesain was pre-incubated with 50–100 μM curcumin for 30 min before the recovery of the activity of the enzyme was determined by dilution of the reaction mixture into measuring buffer [
3,
5]. At concentrations of 50–100 μM, curcumin will be rather dispersed than dissolved in aqueous solutions. This notion is supported by previous observation that curcumin displays very low absorbance in aqueous solutions [
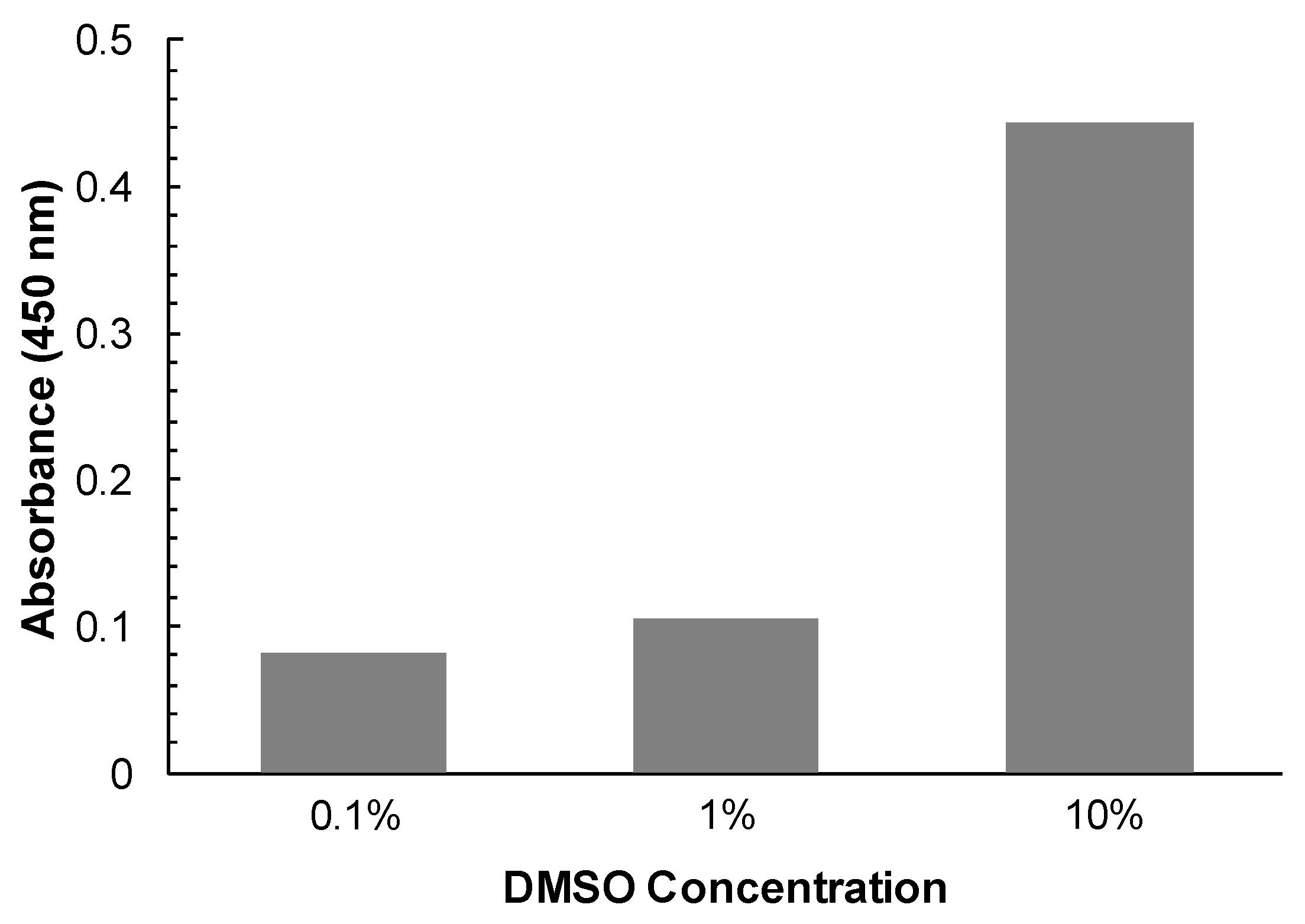
12]. The dispersed curcumin particles may absorb and/or non-specifically inactivate rhodesain present in the reaction mixture. However, the water solubility of curcumin can be increased in the presence of DMSO (
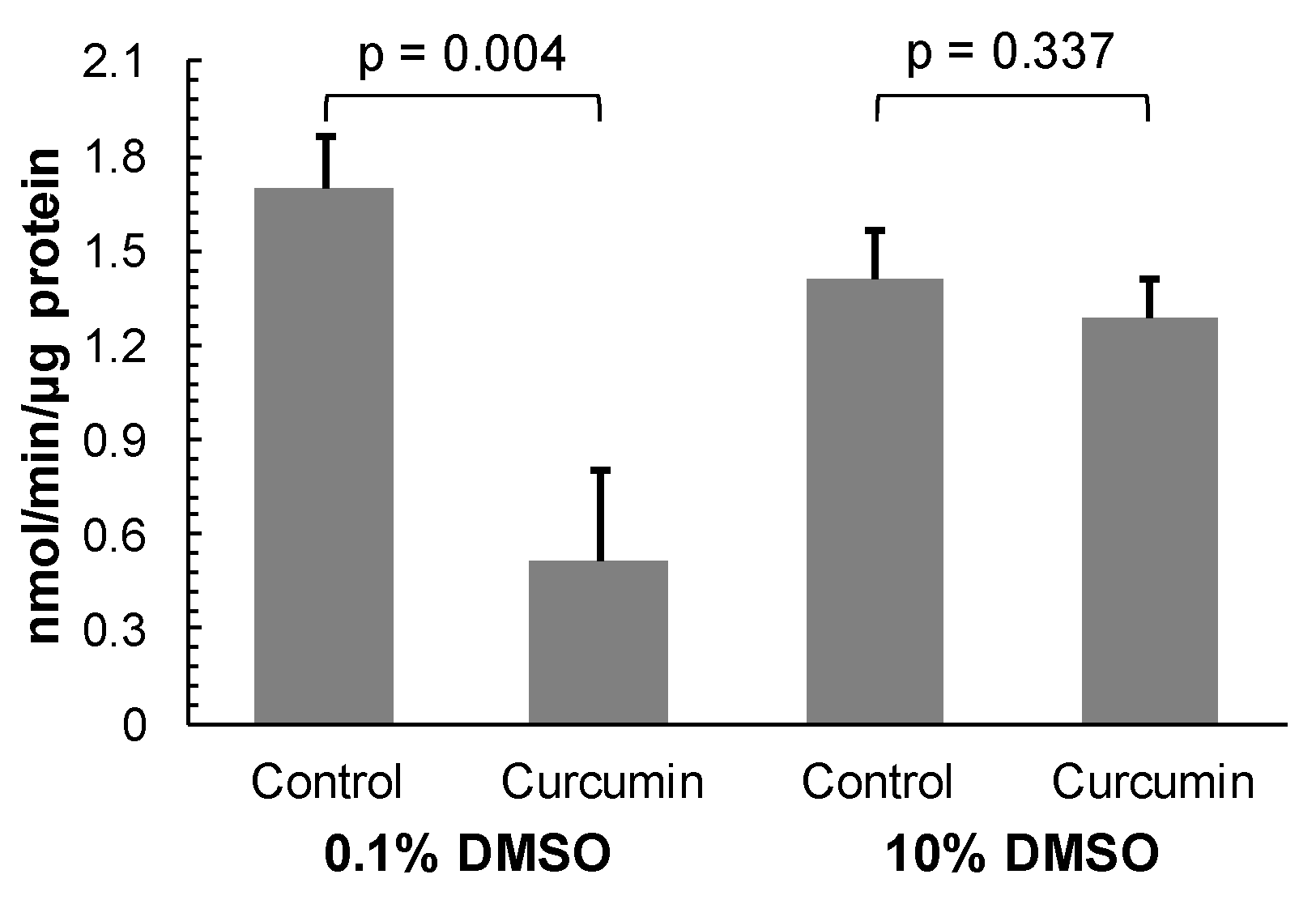
Figure A1). In order to check whether undissolved curcumin can non-specifically inactivate rhodesain, the enzyme was pre-incubated with 100 μM of the compound in the presence of DMSO at a low concentration of 0.1% and at a high concentration of 10%, respectively. After 30 min incubation, the reaction mixture was diluted 100-fold into measuring buffer containing substrate to give a curcumin concentration of 1 μM that was shown not to affect the activity of rhodesain (see
Figure 3). The activity of rhodesain treated with curcumin in the presence of 0.1% DMSO was not fully restored after the dilution (
Figure 4). It reached only 30% of the control enzyme activity. In contrast, the activity of rhodesain incubated with curcumin in the presence of 10% DMSO was restored to 91% of the control enzyme activity after the dilution (
Figure 4). In this case, the activity of the treated enzyme was not statistically significantly different from that of the control enzyme (
p = 0.337;
Figure 4). This result shows that curcumin, if it is dissolved with the help of an appropriate solubilising agent, does not irreversibly inactivate rhodesain. Thus, the lack of recovery of curcumin pre-treated rhodesain observed recently [
3,
5] seemed to be most likely due to non-specific inactivation by undissolved curcumin particles present in the reaction mixture. Interestingly, a similar observation (lack of recovery of enzyme activity after pre-incubation with curcumin) was previously reported for the inactivation of CD13/aminopeptidase N [
13]. While curcumin was identified as a reversible non-competitive inhibitor of CD13/aminopeptidase N, the activity of the enzyme pre-treated with curcumin could not be fully restored after three rounds of filtration using centrifugal filter devices to remove the compound.
Finally, the question remains as to why a time-dependent inhibition of rhodesain activity by curcumin was recently observed [
3,
5]. In this regard, it should be mentioned that the measuring buffer (assay buffer) used in the recent studies [
3,
5] did not contain any reducing thiol reagent. However, cathepsin L cysteine proteases are only fully catalytically active in the presence of thiol reagents (e.g., dithiothreitol, [
14]). When determining the effect of curcumin on the activity of rhodesain in measuring buffer lacking dithiothreitol, a time-dependent inactivation of the enzyme activity by the compound was observed (
Figure 5). After 30 min of incubation, the enzyme was almost completely inactivated. However, after addition of dithiothreitol to a final concentration of 2 mM, rhodesain regained its activity (
Figure 5). Moreover, the activity of the enzyme was now linear with respect to time with a correlation coefficient of the readings of 0.9997 (
Figure 5). This result clearly demonstrates that it is essential to include a thiol reagent in the measuring buffer in order to keep rhodesain fully activated. It should also be pointed out that the activity of rhodesain decelerated when measured in the absence of curcumin and dithiothreitol (
Figure 5, insert). However, the time-dependent inactivation of rhodesain in the absence of dithiothreitol for the curcumin-treated enzyme was more pronounced than for the non-treated enzyme. These findings indicate that rhodesain in the absence of a thiol reagent is oxidised, which leads to gradual inactivation of the enzyme. This oxidation of the rhodesain seems to be accelerated in the presence of curcumin, which may be mistaken as an irreversible inhibition of the enzyme.
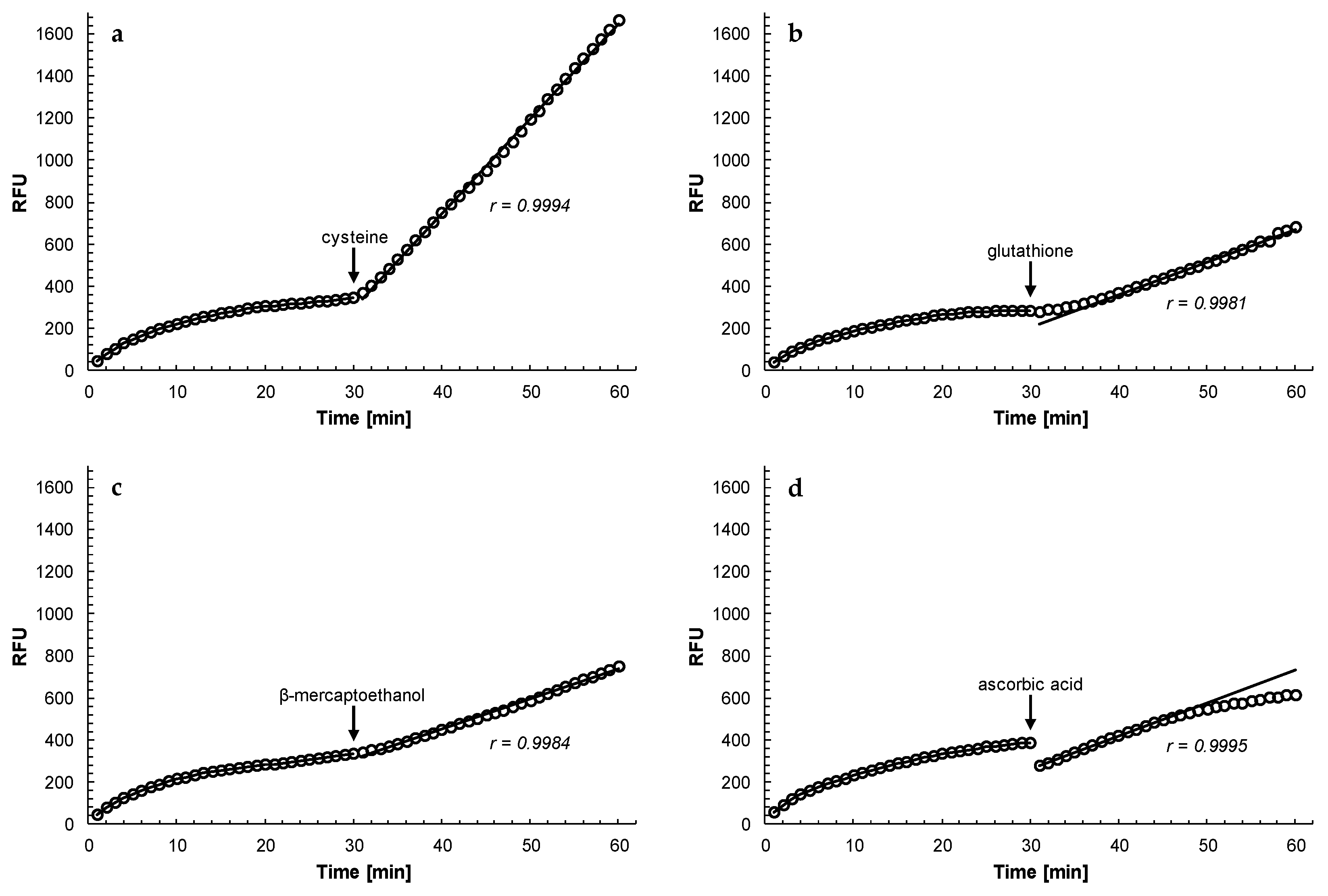
Other antioxidants (cysteine, glutathione, β-mercaptoethanol and ascorbic acid) were also able to reactivate rhodesain that had been inhibited by curcumin, although at different effectiveness (
Figure 6). Cysteine was most effective in the reactivation of rhodesain (even better than dithiothreitol) while ascorbic acid could not sustain the reactivation of the enzyme in the longer term. Glutathione was a slow acting reagent but over time reactivated the enzyme to a similar extent as β-mercaptoethanol. In general, the effectiveness of the reactivation process of curcumin-inhibited rhodesain by the different antioxidants (cysteine > dithiothreitol > glutathione = β-mercaptoethanol > ascorbic acid) was determined by their standard redox potential E
0’: the more negative E
0’, the better the reactivation (E
0’(cysteine) = −348 mV; E
0’(dithiothreitol) = −323 mV; E
0’(β-mercaptoethanol) = −207 mV; E
0’(glutathione) = −205 mV; E
0’(ascorbic acid) = +58 mV [
15,
16,
17]). These findings are further proof that the observed inhibition of rhodesain by curcumin is due to oxidation of thiol groups in the enzyme.