A Review on Daphnane-Type Diterpenoids and Their Bioactive Studies
Abstract
1. Introduction
2. Occurrence
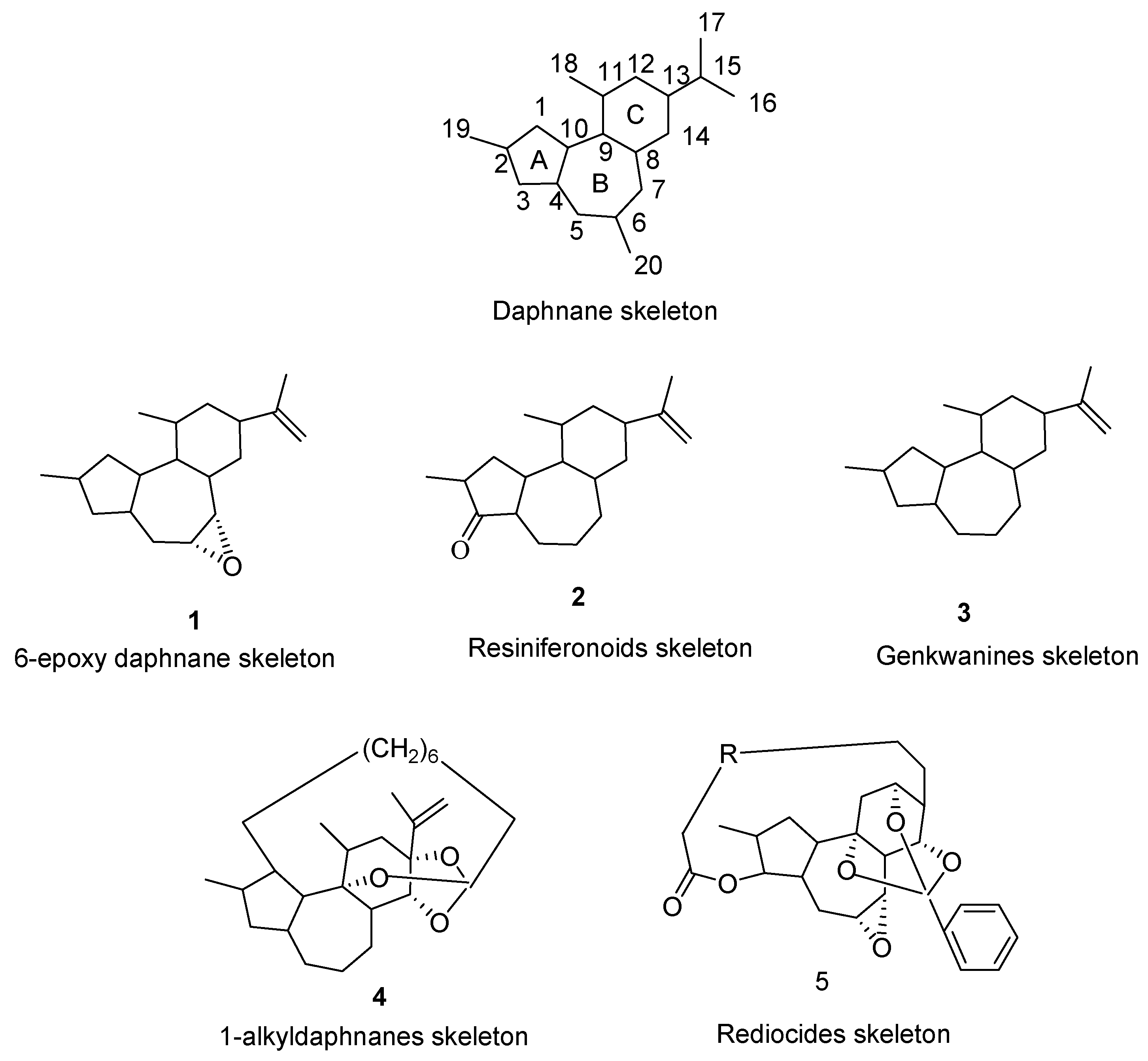
3. Species of Daphnane-Type Diterpenoids and Their Bioactive Activities
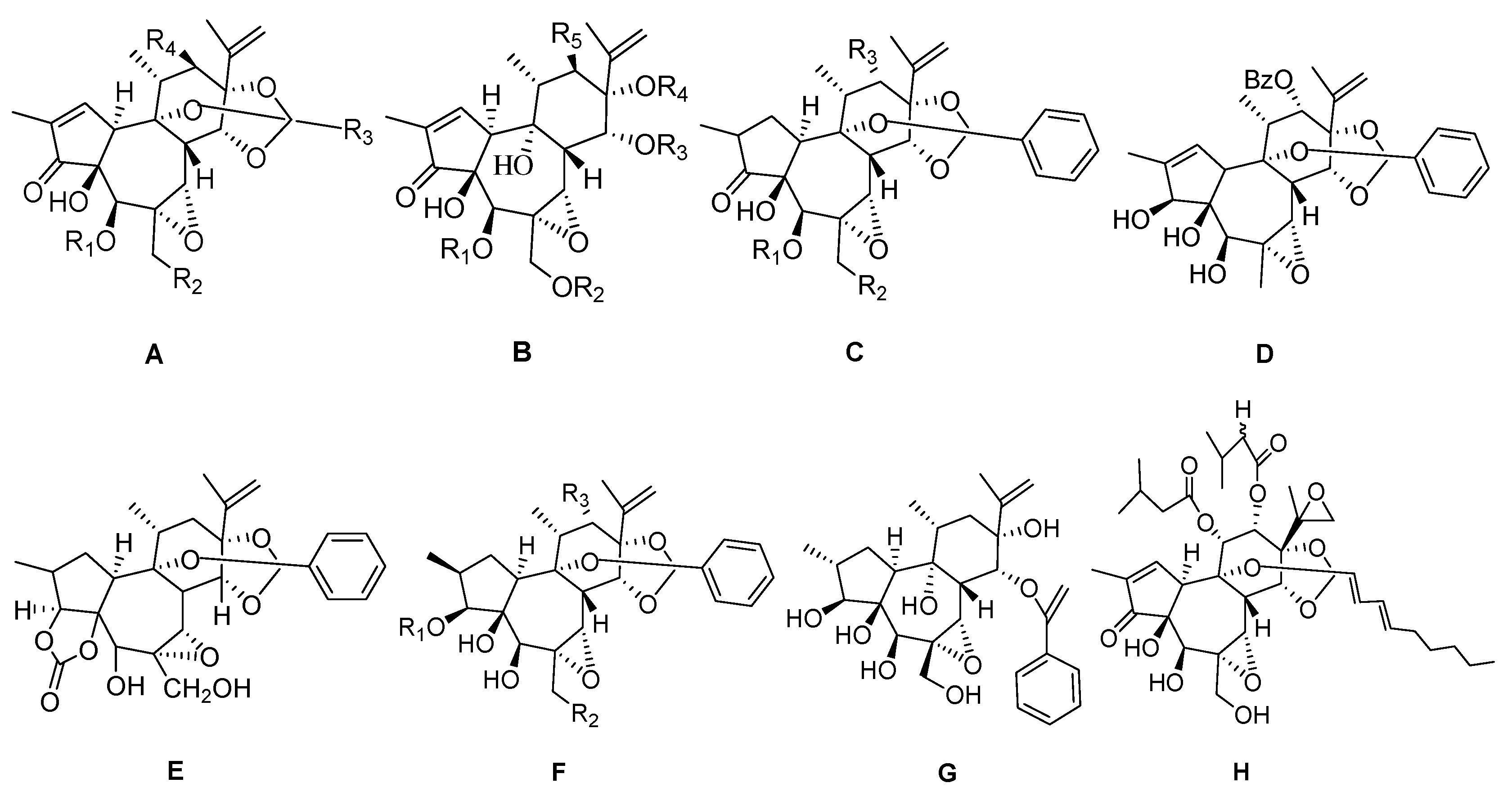
3.1. 6-Epoxy DaphnaneDiterpenoids
3.2. Resiniferonoids
3.3. Genkwanines
3.4. 1-Alkyldaphnanes
3.5. Rediocides
4. Conclusions
Funding
Conflicts of Interest
References
- Jayasuriya, H.; Zink, D.L.; Singh, S.B.; Borris, R.P.; Nanakorn, W.; Beck, H.T.; Balick, M.J.; Goetz, M.A.; Slayton, L.; Gregory, L.; et al. Structure and Stereochemistry of Rediocide A, a Highly Modified Daphnane from Trigonostemon reidioides Exhibiting Potent Insecticidal Activity. ChemInform 2000, 31, 4998–4999. [Google Scholar]
- Carney, J.R.; Krenisky, J.M.; Williamson, R.T.; Luo, J.; Carlson, T.J.; Hsu, V.L.; Moswa, J.L. Maprouneacin, a new daphnane diterpenoid with potent antihyperglycemic activity from Maprounea africana. J. Nat. Prod. 1999, 62, 345–347. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Cik, M.; Lesage, A. Kirkinine, a new daphnane orthoester with potent neurotrophic activity from Synaptolepis kirkii. J. Nat. Prod. 2000, 63, 1185–1187. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Cik, M.; Appendino, G. Daphnane-type diterpene orthoesters and their biological activities. Mini-Rev. Med. Chem. 2002, 2, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Asada, Y.; Sukemori, A.; Watanabe, T.; Malla, K.J.; Yoshikawa, T.; Li, W.; Koike, K.; Chen, C.H.; Akiyama, T.; Qian, K.; et al. Stelleralides A-C, Novel Potent Anti-HIV Daphnane-Type Diterpenoids from Stellera chamaejasme L. Org. Lett. 2011, 13, 2904–2907. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Luo, R.H.; Wang, F.; Dong, Z.J.; Yang, L.M.; Zheng, Y.T.; Liu, J.K. Daphnane diterpenoids isolated from Trigonostemon thyrsoideum as HIV-1 antivirals. Phytochemistry 2010, 71, 1879–1883. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.K.; Chen, B.Y.; Li, H. Flora of China (Zhongguo Zhiwu Zhi); Science: Beijing, China, 1997; Volume 44, pp. 162–170. [Google Scholar]
- Zhang, L.; Luo, R.H.; Wang, F.; Jiang, M.Y.; Dong, Z.J.; Yang, L.M.; Zheng, Y.T.; Liu, J.K. Highly functionalized daphnane diterpenoids from Trigonostemon thyrsoideum. Org. Lett. 2010, 12, 152–155. [Google Scholar] [CrossRef]
- Wang, H.B.; Liu, L.P.; Wang, X.Y. 13C-NMR data of daphnane diterpenoids. Magn. Reson. Chem. 2013, 51, 580–592. [Google Scholar] [CrossRef]
- Huang, S.Z.; Zhang, X.J.; Li, X.Y.; Kong, L.M.; Jiang, H.J.; Ma, Q.Y.; Liu, Y.Q.; Hu, J.M.; Zheng, Y.T.; Li, Y. Daphnane-type diterpene esters with cytotoxic and anti-HIV-1 activities from Daphne acutiloba Rehd. Phytochemistry 2012, 75, 99–107. [Google Scholar] [CrossRef]
- Cheng, Y.Y.; Chen, H.; He, H.P.; Zhang, Y.; Li, S.F.; Tang, G.H.; Guo, L.L.; Yang, W.; Zhu, F.; Zheng, Y.T. Anti-HIV active daphnane diterpenoids from Trigonostemon thyrsoideum. Phytochemistry 2013, 96, 360–369. [Google Scholar] [CrossRef]
- Abe, F.; Iwase, Y.; Yamauchi, T.; Kinjo, K.; Yaga, S.; Ishii, M.; Iwahana, M. Minor daphnane-type diterpenoids from Wikstroemia retusa. Phytochemistry 1998, 47, 833–837. [Google Scholar] [CrossRef]
- Li, L.Z.; Gao, P.Y.; Peng, Y.; Wang, L.H.; Song, S.J. A novel daphnane-type diterpene from the flower bud of Daphne genkwa. Chem. Nat. Compd. 2010, 46, 380–382. [Google Scholar] [CrossRef]
- Li, F.; Sun, Q.; Hong, L.; Li, L.; Wu, Y.; Xia, M.; Ikejima, T.; Peng, Y.; Song, S. Daphnane-type diterpenes with inhibitory activities against human cancer cell lines from Daphne genkwa. Bioorg. Med. Chem. Lett. 2013, 23, 2500–2504. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Yang, X.; Ma, J.; Yang, Y.; Xie, C.; Tuerhong, M.; Jin, D.Q.; Xu, J.; Lee, D.; Ohizumi, Y.; et al. Nitric oxide inhibitory daphnane diterpenoids as potential anti-neuroinflammatory agents for AD from the twigs of Trigonostemon thyrsoideus. Bioorg. Chem. 2017, 75, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Jo, S.K.; Hong, J.Y.; Park, H.J.; Lee, S.K. Anticancer Activity of Novel Daphnane Diterpenoids from Daphne genkwa through Cell-Cycle Arrest and Suppression of Akt/STAT/Src Signalings in Human Lung Cancer Cells. Biomol. Ther. (Seoul). 2012, 20, 513–519. [Google Scholar] [CrossRef]
- Taninaka, H.; Takaishi, Y.; Honda, G.; Imakura, Y.; Sezik, E.; Yesilada, E. Terpenoids and aromatic compounds from Daphne oleoides ssp. oleoides. Phytochemistry (Oxford) 1999, 52, 1525–1529. [Google Scholar] [CrossRef]
- Yu, L.; Zuo, W.J.; Mei, W.L.; Guo, Z.K.; Li, X.N.; Dai, H.F. Three new terpenoids from Trigonostemon xyphophylloides (Croiz.) L.K. Dai and T.L. Wu. Phytochem. Lett. 2013, 6, 472–475. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Guo, J.M.; Qian, Y.F.; Guo, S.; Ma, C.H.; Duan, J.A. Toxicity of daphnane-type diterpenoids from Genkwa Flos and their pharmacokinetic profile in rat. Phytomedicine 2013, 21, 82–89. [Google Scholar] [CrossRef]
- Li, S.F.; Di, Y.T.; Li, S.L.; Zhang, Y.; Yang, F.M.; Sun, Q.Y.; Simo, J.M.; He, H.P.; Hao, X.J. Trigonosins A−F, Daphnane Diterpenoids from Trigonostemon thyrsoideum. J. Nat. Prod. 2011, 74, 464–469. [Google Scholar] [CrossRef]
- Miyamae, Y.; Villareal, M.O.; Abdrabbah, M.B.; Isoda, H.; Shigemori, H. Hirseins A and B, Daphnane Diterpenoids from Thymelaea hirsuta That Inhibit Melanogenesis in B16 Melanoma Cells. J. Nat. Prod. 2009, 72, 938–941. [Google Scholar] [CrossRef]
- Tchinda, A.Y.; Tsopmo, A.; Tene, M.; Kamnaing, P.; Ngnokam, D.; Tane, P.; Ayafor, J.F.; Connolly, J.D.; Farrugia, L.J. Diterpenoids from Neoboutonia glabrescens (Euphorbiaceae). Phytochemistry 2003, 64, 549–553. [Google Scholar] [CrossRef]
- Hayes, P.Y.; Chow, S.; Somerville, M.J.; Fletcher, M.T. Daphnane- and Tigliane-Type Diterpenoid Esters and Orthoesters from Pimelea elongata. J. Nat. Prod. 2010, 73, 1907–1913. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.D.; Han, M.L.; Ji, Y.C.; Chen, H.D.; Yang, S.P.; Zhang, S.; Geng, M.Y.; Yue, J.M. Trigoxyphins A-G: diterpenes from Trigonostemon xyphophylloides. J. Nat. Prod. 2010, 73, 1301–1305. [Google Scholar] [CrossRef] [PubMed]
- Powell, R.G.; Weisleder, D.; Smith, C.R. Daphnane Diterpenes from Diarthron vesiculosum: Vesiculosin and Isovesiculosin. J. Nat. Prod. 1985, 48, 102–107. [Google Scholar] [CrossRef]
- Su, J.; Wu, Z. A New Daphnane-Type Diterpenoid from Daphne giraldii. Chem. Nat. Compd. 2014, 50, 285–287. [Google Scholar] [CrossRef]
- Chen, H.D.; Yang, S.P.; He, X.F.; Liu, H.B.; Ding, J.; Yue, J.M. Trigochinins D–I: six new daphnane-type diterpenoids from Trigonostemon chinensis. Tetrahedron 2010, 66, 5065–5070. [Google Scholar] [CrossRef]
- Wei, Y.L.; Yu, Z.L.; Huo, X.K.; Tian, X.G.; Feng, L.; Huang, S.S.; Deng, S.; Ma, X.C.; Jia, J.M.; Wang, C. Diterpenoids from the roots of Euphorbia fischeriana and their inhibitory effects on alpha-glucosidase. J. Asian Nat. Prod. Res. 2018, 20, 977–984. [Google Scholar] [CrossRef]
- Bang, K.K.; Yun, C.Y.; Lee, C.; Jin, Q.; Lee, J.W.; Jung, S.H.; Lee, D.; Lee, M.K.; Hong, J.T.; Kim, Y.; et al. Melanogenesis inhibitory daphnane diterpenoids from the flower buds of Daphne genkwa. Bioorg. Med. Chem. Lett. 2013, 23, 3334. [Google Scholar] [CrossRef]
- Zhan, Z.J.; Fan, C.Q.; Ding, J.; Yue, J.M. Novel diterpenoids with potent inhibitory activity against endothelium cell HMEC and cytotoxic activities from a well-known TCM plant Daphne genkwa. Bioorg. Med. Chem. 2005, 13, 645–655. [Google Scholar] [CrossRef]
- Zhang, X.D.; Ni, W.; Yan, H.; Li, G.T.; Zhong, H.M.; Li, Y.; Liu, H.Y. Daphnane-Type Diterpenoid Glucosides and Further Constituents of Euphorbia pilosa. Chem. Biodivers. 2014, 11, 760–766. [Google Scholar] [CrossRef]
- Yang, B.; Meng, Z.; Li, Z.; Sun, L.; Hu, Y.; Wang, Z.; Ding, G.; Xiao, W.; Han, C. Three daphnane diterpenoids from Trigonostemon xyphophylloides. Phytochem. Lett. 2015, 11, 270–274. [Google Scholar] [CrossRef]
- Li, S.F.; Zhang, Y.; Huang, N.; Zheng, Y.T.; Di, Y.T.; Li, S.L.; Cheng, Y.Y.; He, H.P.; Hao, X.J. Daphnane diterpenoids from the stems of Trigonostemon lii and their anti-HIV-1 activity. Phytochemistry 2013, 93, 216–221. [Google Scholar] [CrossRef]
- Pejin, B.; Iodice, C.; Tommonaro, G. Synthesis and biological activities of thio-avarol derivatives. J. Nat. Prod. 2008, 71, 1850–1853. [Google Scholar] [CrossRef]
- Tommonaro, G.; Vitale, R.M.; Pejin, B.; Iodice, C.; Canadas, S. Avarol derivatives as competitive AChE inhibitors, non hepatotoxic and neuroprotective agents for Alzheimer’s disease. Eur. J. Med. Chem. 2016, 122, 326–338. [Google Scholar] [CrossRef]
- Yang, B.; Chen, G.Y.; Song, X.P.; Yang, L.Q.; Han, C.R.; Wu, X.Y.; Li, X.M.; Zou, B.Y. Trigoxyphins H and I: Two new daphnane diterpenoids from Trigonostemon xyphophylloides. ChemInform 2012, 22, 3828–3830. [Google Scholar] [CrossRef]
- Dong, S.H.; Zhang, C.R.; Xu, C.H.; Ding, J.; Yue, J.M. Daphnane-Type Diterpenoids from Trigonostemon howii. J. Nat. Prod. 2011, 74, 1255–1261. [Google Scholar] [CrossRef]
- Guo, J.; Tian, J.; Yao, G.; Zhu, H.; Xue, Y.; Luo, Z.; Zhang, J.; Zhang, Y. Three new 1α-alkyldaphnane-type diterpenoids from the flower buds of Wikstroemia chamaedaphne. Fitoterapia 2015, 106, 242–246. [Google Scholar] [CrossRef]
- Hayes, P.Y.; Chow, S.; Somerville, M.J.; Fletcher, M.T. Pimelotides A and B, Diterpenoid Ketal-Lactone Orthoesters with an Unprecedented Skeleton from Pimelea elongata. J. Nat. Prod. 2009, 72, 2081–2083. [Google Scholar] [CrossRef]
- Chen, H.D.; He, X.F.; Ai, J.; Geng, M.Y.; Yue, J.M. Trigochilides A and B, Two Highly Modified Daphnane-Type Diterpenoids from Trigonostemon chinensis. Org. Lett. 2009, 11, 4080–4083. [Google Scholar] [CrossRef]
- Jayasuriya, H.; Zink, D.L.; Borris, R.P.; Nanakorn, W.; Beck, H.T.; Balick, M.J.; Goetz, M.A.; Gregory, L.; Shoop, W.L.; Singh, S.B. Rediocides B-E, Potent Insecticides from Trigonostemon reidioides. J. Nat. Prod. 2004, 67, 228–231. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
| Types of Diterpenoids | Species | Medication Site |
|---|---|---|
| 6-epoxy daphnane diterpenoids | D. acutiloba | Usually their effective part is roots, stems, twigs and leaves, flower buds, fresh bark. |
| Trigonostemonthyrsoideum | ||
| Wikstroemiaretusa | ||
| Daphne genkwa | ||
| D. oleoidesSchreber ssp. oleoides | ||
| Trigonostemonxyphophylloides | ||
| Thymelaeahirsuta | ||
| Neoboutoniaglabrescens | ||
| S.kirkii | ||
| W.monticola | ||
| D.tangutica | ||
| P.elongata | ||
| T.xyphophylloides | ||
| T.thyrsoideum | ||
| D.vesiculosum | ||
| Stellerachamaejasme L. | ||
| Trigonostemonchinensis Merr | ||
| Resiniferonoids | Euphorbia fischeriana | Generally, the roots and flower budsistheir effective part. |
| Daphne genkwa | ||
| Euphorbia pilosa | ||
| Genkwanines | Trigonostemonxyphophylloides | Usually their effective part isroots, stems, twigs and leaves, flower buds. |
| Trigonostemonthyrsoideum | ||
| Trigonostemon lii | ||
| Trigonostemonchinensis Merr | ||
| Daphne genkwa | ||
| Trigonostemonhowii | ||
| 1-alkyldaphnanes | Wikstroemiachamaedaphne | Usually, the flower buds and fresh bark is their effective part. |
| Wikstroemiaretusa | ||
| Stellerachamaejasme L. | ||
| Daphne genkwa | ||
| Synaptolepiskirkii | ||
| P.elongata | ||
| Rediocides | Trigonostemonthyrsoideum | Generally, their effective part is roots, twigs and leaves. |
| Trigonostemonchinensis Merr | ||
| Trigonostemonreidioides |
| No. | Name | R1 | R2 | R3 | R4 | R5 | Type |
|---|---|---|---|---|---|---|---|
| 1 | Acutilobin A | H | OH | Ph | OCO(CH=CH)2COC(CH2)2CH3 | – | A |
| 2 | Acutilobin B | H | OH | Ph | OCO(CH=CH)3CHCH2CH3OH | – | A |
| 3 | Acutilobin C | H | OH | (CH=CH)3(CH2)2CH3 | OCOCH=CHPhCH3OH | – | A |
| 4 | Acutilobin D | H | OH | (CH=CH)2(CH2)4CH3 | OCOCH=CHPhCH3OH | – | A |
| 5 | Acutilobin E | H | OH | Ph | OCOCH=CHPhCH3OH | – | A |
| 6 | Daphnetoxin | H | OH | Ph | H | – | A |
| 7 | Excoecaria toxin | H | OH | (CH=CH)2(CH2)4CH3 | H | – | A |
| 8 | Excoecaria factor O1 | H | OH | (CH=CH)3(CH2)2CH3 | H | – | A |
| 9 | Genkwadane D | H | OH | (CH=CH)2(CH2)4CH3 | OCOCH(CH3)2 | – | A |
| 10 | Genkwadaphnine | H | OH | Ph | OBz | – | A |
| 11 | Genkwadaphnin-20-palmitate | H | OCO(CH2)14CH3 | Ph | OCOPh | – | A |
| 12 | Glabrescin | H | OCOCH2(CH2)13CH3 | (CH2)10CH3 | H | – | A |
| 13 | Gnidicin | H | OH | Ph | OCOCH=CHPh | – | A |
| 14 | Gniditrin | H | OH | Ph | OCO(CH=CH)3(CH2)2CH3 | – | A |
| 15 | Gnididin | H | OH | Ph | OCO(CH=CH)2(CH2)4CH3 | – | A |
| 16 | Gnidiglaucin | H | OH | (CH2)8CH3 | OAc | – | A |
| 17 | Gnidilatidin | H | OH | (CH=CH)2(CH2)4CH3 | OCOPh | – | A |
| 18 | Gnidilatidin-20-palmitate | H | OCO(CH2)14CH3 | (CH=CH)2(CH2)4CH3 | OCOPh | – | A |
| 19 | Gnidicin-20-palmitate | H | OCO(CH2)14CH3 | Ph | OCOCH=CHPh | – | A |
| 20 | Huratoxin | H | OH | (CH=CH)2(CH2)8CH3 | H | – | A |
| 21 | Hirsein A | H | OH | CH=CH(CH2)4CH3 | OCOCH=CHPh | – | A |
| 22 | Hirsein B | H | OH | CH=CH(CH2)4CH3 | OCOCH=CHPhOH | – | A |
| 23 | Isoyuanhuadine | H | OH | (CH=CH)2(CH2)4CH3 | OAc | – | A |
| 24 | Kirkinine | H | OH | CH=CH(CH2)12CH3 | OAc | – | A |
| 25 | Kirkinine D | H | OH | (CH=CH)3(CH2)2CH3 | OAc | – | A |
| 26 | Montanin | H | OH | (CH2)10CH3 | H | – | A |
| 27 | Simplexin | H | OH | (CH2)8CH3 | H | – | A |
| 28 | Synaptolepisfactor K7 | H | OH | CH=CH(CH2)12CH3 | H | – | A |
| 29 | Trigochinin G | H | H | Ph | OCOCH2CH(CH3)2 | – | A |
| 30 | Trigochinin H | H | H | Ph | OCOC6H4(4-OH) | – | A |
| 31 | Trigochinin I | H | H | Ph | OCOC6H3(3-OMe)(4-OH) | – | A |
| 32 | Trigoxyphin A | H | H | Ph | OBz | – | A |
| 33 | Trigoxyphin J | H | OH | CH3 | OCO(CH2)14CH3 | – | A |
| 34 | Trigoxyphin K | H | H | Ph | OBz | – | A |
| 35 | Wikstrotoxin C | 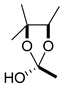 | OH | (CH=CH)2(CH2)4CH3 | OAc | – | A |
| 36 | Wikstrotoxin D | H | OH | n-C9H19 | H | – | A |
| 37 | Wikstroelide A | H | OH | (CH=CH)2(CH2)8CH3 | OAc | – | A |
| 38 | Wikstroelide B | H | OH | (CH=CH)2(CH2)9CH3 | OAc | – | A |
| 39 | Wikstroelide C | H | O-trans-5-pentadecenoic acid | (CH=CH)2(CH2)8CH3 | OAc | – | A |
| 40 | Wikstroelide D | H | O-palmitic acid | (CH=CH)2(CH2)8CH3 | OAc | – | A |
| 41 | Wikstroelide H | H | OH | (CH=CH)2(CH2)6CH3 | OAc | – | A |
| 42 | Wikstroelide I | H | O-palmitic acid | (CH=CH)2(CH2)9CH3 | OAc | – | A |
| 43 | Wikstroelide L | H | OH | (CH=CH)2(CH2)8CH3 | OAc | – | A |
| 44 | Yuanhuahine | H | OH | (CH=CH)2(CH2)4CH3 | OCOCH2CH3 | – | A |
| 45 | Yuanhuafine | H | H | Ph | OAc | – | A |
| 46 | Yuanhualine | H | OH | (CH=CH)2(CH2)4CH3 | OCO(CH2)2CH3 | – | A |
| 47 | Yuanhuadine | H | OH | (CH=CH)2(CH2)4CH3 | OAc | – | A |
| 48 | Yuanhuagine | H | OH | (CH=CH)(CH2)2CH3 | OCOCH3 | – | A |
| 49 | Yuanhuacine | H | OH | (CH=CH)2(CH2)4CH3 | OBz | – | A |
| 50 | Yuanhuajine | H | OH | (CH=CH)3(CH2)2CH3 | OBz | – | A |
| 51 | 14′-ethyltetrahydrohuratoxin | H | OH | (CH2)14CH3 | H | – | A |
| 52 | 2α-dihydro-20-palimoyldaphnetoxin | H | OH | CH=CH(CH2)6CH3 | OAc | – | A |
| 53 | Daphnegiraldigin | H | OH | COPh | H | H | B |
| 54 | Isovesiculosin | Ac | Ac | Ac | CO(CH=CH)2(CH2)4CH3 | H | B |
| 55 | Vesiculosin | H | H | CO(CH=CH)2(CH2)4CH3 | H | H | B |
| 56 | Wikstroelide J | H | H | CO(CH=CH)2(CH2)8CH3 | H | OAc | B |
| 57 | Wikstroelide M | H | H | CO(CH=CH)2(CH2)8CH3 | H | H | B |
| 58 | Wikstroelide N | H | H | CO(CH=CH)2(CH2)9CH3 | H | H | B |
| 59 | Trigoxyphin B | H | H | OBz | – | – | C |
| 60 | Trigoxyphin C | Ac | H | OBz | – | – | C |
| 61 | Yuanhuapine | H | OH | OAc | – | – | C |
| 62 | 1,2α-dihydrodaphnetoxin | H | OH | H | – | – | C |
| 63 | Trigothysoid M | – | – | – | – | – | D |
| 64 | Genkwanin I | – | – | – | – | – | E |
| 65 | Acutilobin F | CO(CH=CH)3(CH2)2CH3 | OH | H | – | – | F |
| 66 | Acutilobin G | COCH=CHPh | OH | H | – | – | F |
| 67 | Genkwanine M | H | OBz | H | – | – | F |
| 68 | Genkwanine N | Bz | OH | H | – | – | F |
| 69 | GenkwanineVIII | COPh | OH | H | – | – | F |
| 70 | Orthobenzoate 2 | H | OH | H | – | – | F |
| 71 | Trigonostempene C | H | H | OH | – | – | F |
| 72 | Trigonosin A | H | H | OBz | – | – | F |
| 73 | Trigonosin B | H | OH | OBz | – | – | F |
| 74 | Wikstroemia factor M1 | CO(CH=CH)2(CH2)4CH3 | OH | H | – | – | F |
| 75 | Genkuanine O | – | – | – | – | – | G |
| 76 | Maprouneacin | – | – | – | – | – | H |
| No. | Name | R | Type |
|---|---|---|---|
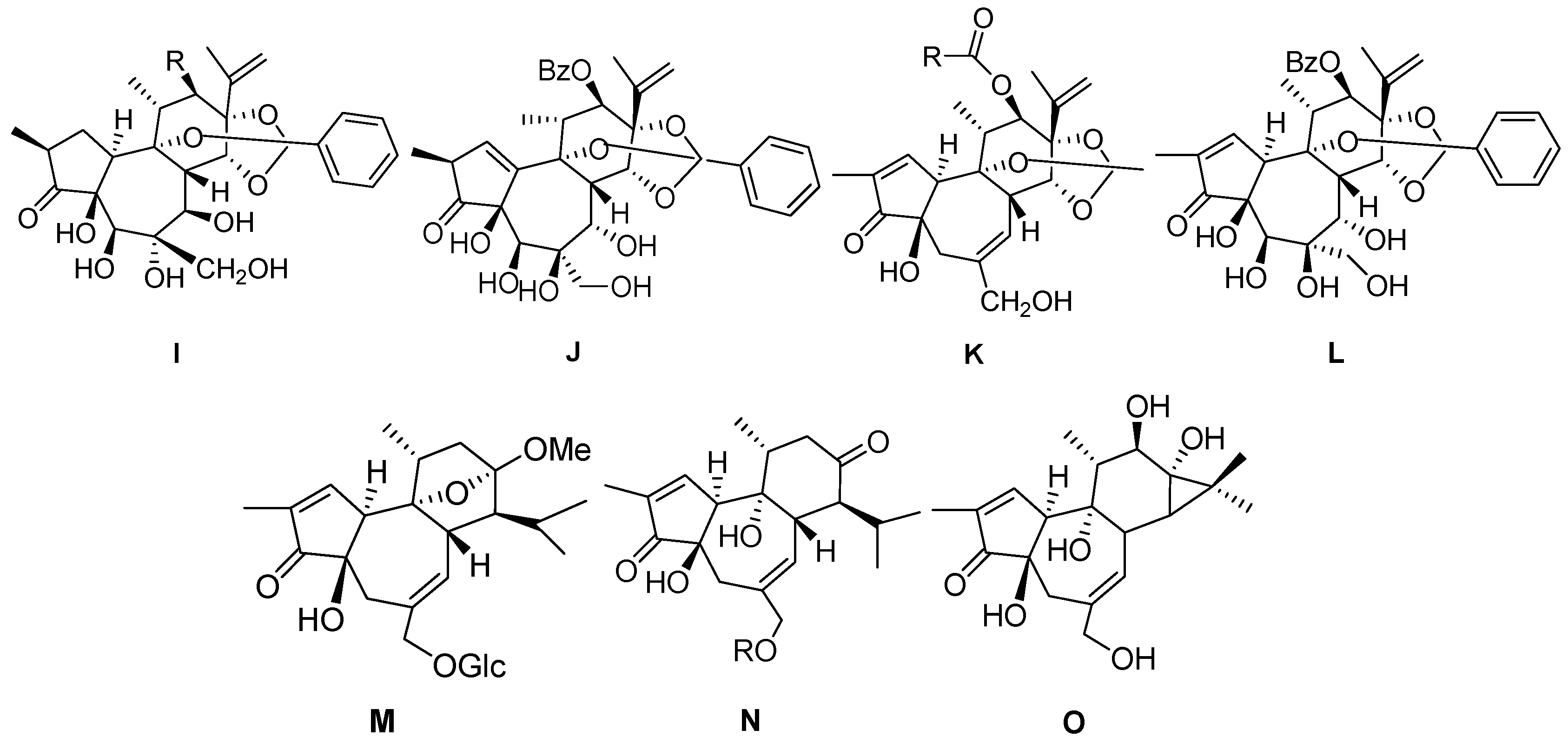
| 77 | Genkwanine L | OAc | I |
| 78 | Yuanhuatine | OBz | I |
| 79 | Genkwadane A | – | J |
| 80 | Daphneresiniferin A | Me | K |
| 81 | Daphneresiniferin B | Ph | K |
| 82 | Yuanhuaoate B | – | L |
| 83 | Euphopiloside B | – | M |
| 84 | Euphopiloside A | 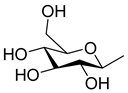 | N |
| 85 | Langduin A | H | N |
| 86 | 4β,9α,20-trihydroxy-13,15-secotiglia-1,6-diene-3,13-dione20-O-β-d-[6-galloyl]glu-copyranoside | 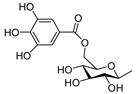 | N |
| 87 | Phorbol | – | O |
| No. | Name | R1 | R2 | R3 | R4 | R5 | R6 | R7 | R8 | Type |
|---|---|---|---|---|---|---|---|---|---|---|
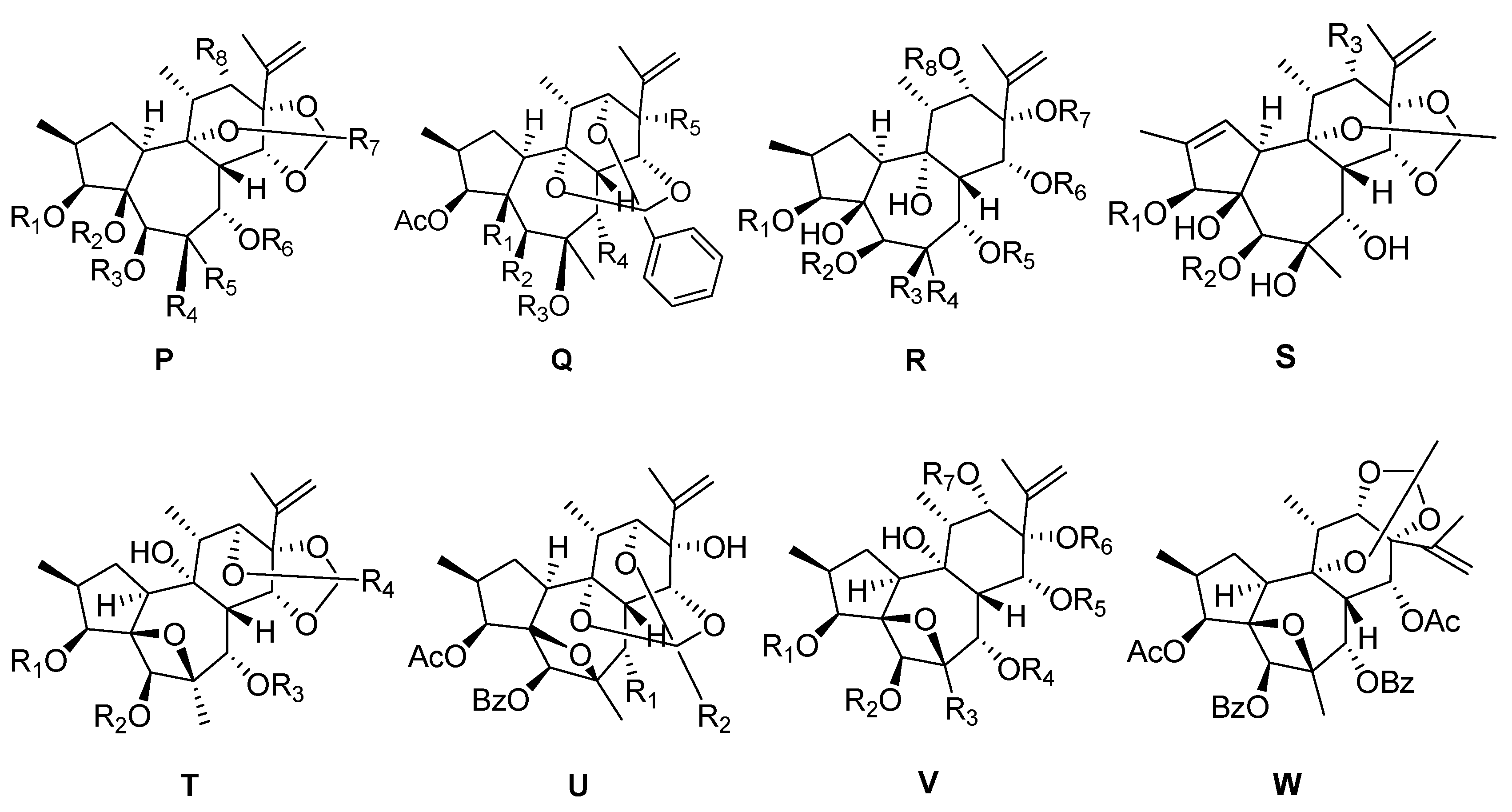
| 88 | Genkwanine A | H | H | H | OH | CH2OH | H | Ph | H | P |
| 89 | Genkwanine B | CO(CH=CH)2(CH2)4CH3 | H | H | OH | CH2OH | H | Ph | H | P |
| 90 | Genkwanine C | CO(CH=CH)3(CH2)2CH3 | H | H | OH | CH2OH | H | Ph | H | P |
| 91 | Genkwanine D | Bz | H | H | OH | CH2OH | H | Ph | H | P |
| 92 | Genkwanine E | H | H | H | OH | CH2OCO(CH=CH)3(CH2)2CH3 | H | Ph | H | P |
| 93 | Genkwanine F | H | H | H | OH | CH2OCO(CH=CH)2(CH2)4CH3 | H | Ph | H | P |
| 94 | Genkwanine G | H | H | H | OH | CH2COO(CH=CH) (CH2)6CH3 | H | Ph | H | P |
| 95 | Genkwanine H | H | H | H | OH | CH2OBz | H | Ph | H | P |
| 96 | Trigothysoid D | H | H | H | OH | Me | H | Me | OBz | P |
| 97 | Trigothysoid E | Ac | H | H | OH | Me | H | Me | OBz | P |
| 98 | Trigothysoid F | H | H | Ac | OH | Me | H | Me | OBz | P |
| 99 | Trigothysoid G | Ac | H | Bz | OH | ME | H | Me | OBz | P |
| 100 | Trigoxyphin H | Ac | H | Ac | OCOPh | Me | Ac | Ph | OAc | P |
| 101 | Trigohownin D | Ac | Bz | Ac | OH | Me | Ac | Ph | OAc | P |
| 102 | Trigohownin E | Ac | H | Bz | OH | Me | Ac | Me | OBz | P |
| 103 | Trigoxyphin F | Ac | H | Ac | OBz | Me | Ac | Ph | OH | P |
| 104 | Trigoxyphin I | Ac | H | Ac | OCOPh | Me | Ac | Ph | Ac | P |
| 105 | Trigoxyphin U | Ac | H | Ac | Me | OCOPh | Ac | ME | OCOPh | P |
| 106 | Trigonosin C | H | H | H | OH | Me | H | Ph | OBz | P |
| 107 | Trigonothyrin F | H | H | H | OH | Me | H | Ph | H | P |
| 108 | Trigohownin A | OAc | OH | Bz | OAc | OH | – | – | – | Q |
| 109 | Trigohownin B | OBz | OAC | H | OAc | OH | – | – | – | Q |
| 110 | Trigohownin C | OH | OAC | Bz | OH | OH | – | – | – | Q |
| 111 | Trigoxyphin D | OH | OAC | Bz | OAc | OH | – | – | – | Q |
| 112 | Trigoxyphin E | H | OAC | Bz | OAc | OAc | – | – | – | Q |
| 113 | Genkwanine I | H | H | OH | CH2OH | H | Bz | H | H | R |
| 114 | Genkwanine J | H | H | OH | CH2OCO(CH=CH)2(CH2)4CH3 | H | Bz | H | H | R |
| 115 | Genkwanine K | H | H | OH | CH2Bz | H | Bz | H | H | R |
| 116 | Trigoxyphin W | Ac | Ac | Me | OCOPh | H | H | COPh | H | R |
| 117 | Trigohownin F | Ac | Ac | OBz | Me | Ac | H | Bz | OH | R |
| 118 | Trigohownin G | Ac | Ac | OBz | Me | Ac | Ac | Bz | OH | R |
| 119 | Trigohownin H | Ac | Ac | OBz | Me | Ac | Bz | Ac | OH | R |
| 120 | Trigohownin I | Ac | Bz | OH | Me | Ac | Ac | Bz | OH | R |
| 121 | Trigonothyrin G | Ac | H | OCOPh | – | – | – | – | – | S |
| 122 | Trigothysoid A | H | H | OBz | – | – | – | – | – | S |
| 123 | Trigothysoid B | Ac | Bz | OBz | – | – | – | – | – | S |
| 124 | Trigothysoid C | H | Ac | OBz | – | – | – | – | – | S |
| 125 | Trigonothyrin A | Bz | Ac | Bz | Me | – | – | – | – | T |
| 126 | Trigonothyrin B | H | Bz | Bz | Me | – | – | – | – | T |
| 127 | Trigonothyrin C | Ac | Bz | Bz | Me | – | – | – | – | T |
| 128 | Trigothysoid L | Ac | Bz | Ac | Ph | – | – | – | – | T |
| 129 | Trigonostempene B | Ac | Ac | Bz | Me | – | – | – | – | T |
| 130 | Trigochinin C | OAc | Ph | – | – | – | – | – | – | U |
| 131 | Trigothysoid K | OBz | Me | – | – | – | – | – | – | U |
| 132 | Trigolins A | H | Bz | Me | Ac | Ac | H | Bz | – | V |
| 133 | Trigolins B | Ac | Bz | Me | Ac | H | H | Bz | – | V |
| 134 | Trigolins C | Ac | Bz | Me | Ac | Bz | H | H | – | V |
| 135 | Trigolins D | Ac | Bz | Me | Ac | Ac | H | Bz | – | V |
| 136 | Trigolins E | Ac | Bz | Me | Bz | Ac | H | Ac | – | V |
| 137 | Trigolins F | Ac | Ac | Me | Bz | Ac | H | Bz | – | V |
| 138 | Trigolins G | H | Bz | Me | Bz | AC | H | Bz | – | V |
| 139 | Trigothysoid H | Ac | Ac | CH2OAc | Ac | Ac | Bz | Ac | – | V |
| 140 | Trigothysoid I | Ac | Ac | CH2OAc | Ac | Ac | H | Bz | – | V |
| 141 | Trigothysoid J | Ac | Bz | Me | Ac | Ac | H | Bz | – | V |
| 142 | Trigonosin D | H | H | Me | Ac | Ac | COPh | Ac | – | V |
| 143 | Trigonothyrin D | Ac | Ac | Me | Ac | Ac | COPh | Ac | – | V |
| 144 | Trigonothyrin E | H | Ac | Me | Ac | Ac | COPh | Ac | – | V |
| 145 | Trigochinin A | H | Bz | Me | Ac | Ac | COPh | Ac | – | V |
| 146 | Trigochinin B | Ac | Bz | Me | Ac | Ac | COPh | Ac | – | V |
| 147 | Trigochinin D | H | Bz | Me | Ac | Ac | Bz | Ac | – | V |
| 148 | Trigochinin E | Ac | Bz | Me | Ac | Ac | Bz | Ac | – | V |
| 149 | Trigochinin F | Ac | Ac | Ac | Ac | Ac | Bz | Ac | – | V |
| 150 | Trigonostempene A | – | – | – | – | – | – | – | – | W |
| No. | Name | R1 | R2 | R3 | R4 | R5 | R6 | Type |
|---|---|---|---|---|---|---|---|---|
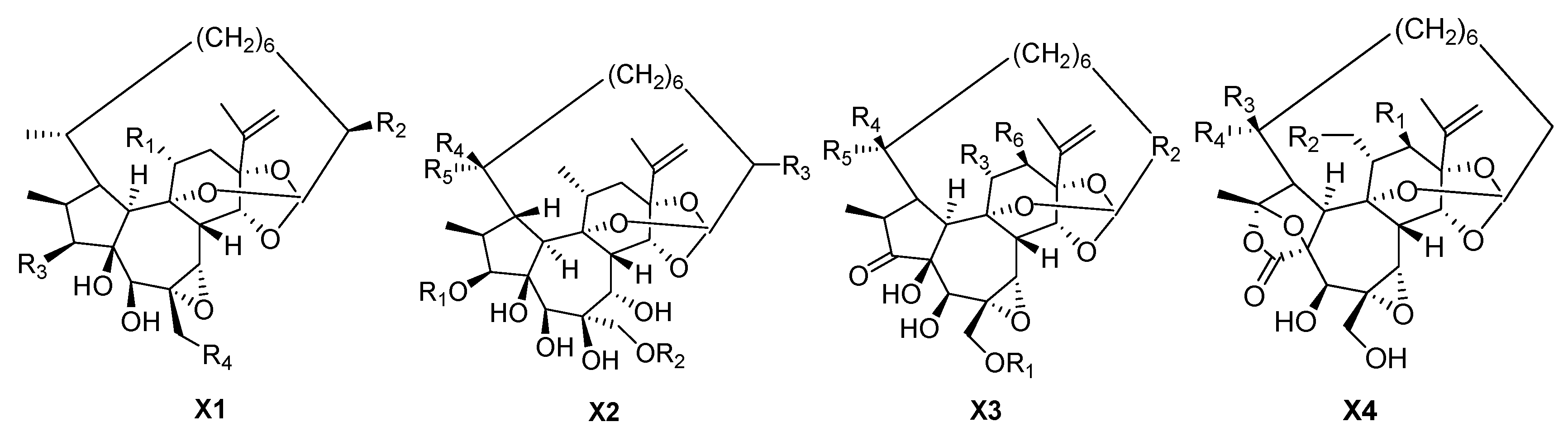
| 151 | Stelleralide A | CH2OAc | OH | OBz | OH | – | – | X1 |
| 152 | Stelleralide B | CH2OBz | H | OBz | OH | – | – | X1 |
| 153 | Gnidimacrin | CH2OBz | OH | OBz | OH | – | – | X1 |
| 154 | Genkwadane B | Me | H | OH | OBz | – | – | X1 |
| 155 | Pimelea factor P2 | CH2OH | H | OBz | OH | – | – | X1 |
| 156 | Genkwadane C | H | benzoyl | H | H | Me | – | X2 |
| 157 | Wikstroelide R | H | benzoyl | OH | H | Me | – | X2 |
| 158 | Wikstroelide S | benzoyl | H | H | Me | H | – | X2 |
| 159 | Wikstroelide T | H | trans-cinnamoyl | H | H | Me | – | X2 |
| 160 | Kirkinine B | H | CH=CH(CH2)5 | Me | H | Me | H | X3 |
| 161 | Kirkinine C | H | CH=CH(CH2)5 | Me | H | Me | OAc | X3 |
| 162 | Kirkinine E | H | CH=CH(CH2)5 | Me | OH | Me | H | X3 |
| 163 | Wikstroelide E | H | CH2 | Me | H | Me | H | X3 |
| 164 | Wikstroelide F | H | CH2 | CH2OBz | H | Me | H | X3 |
| 165 | Wikstroelide G | palmitic acid | CH2 | CH2OBz | H | Me | H | X3 |
| 166 | Wikstroelide K | CO(CH2)14CH3 | CH2 | CH2OBz | Me | H | H | X3 |
| 167 | Wikstroelide O | H | CH2 | CH2OBz | Me | H | H | X3 |
| 168 | Pimelea factor S6 | OH | CH2 | Me | H | Me | H | X3 |
| 169 | Pimelea factor S7 | OH | CH2 | Me | Me | H | H | X3 |
| 170 | Pimelotide A | H | H | Me | H | – | – | X4 |
| 171 | Pimelotide B | OAc | H | H | Me | – | – | X4 |
| 172 | Pimelotide C | H | H | H | Me | – | – | X4 |
| 173 | Pimelotide D | OAc | H | Me | H | – | – | X4 |
| 174 | Stelleralide C | H | OBz | Me | H | – | – | X4 |
| No. | Name | R1 | R2 | R3 | Type |
|---|---|---|---|---|---|
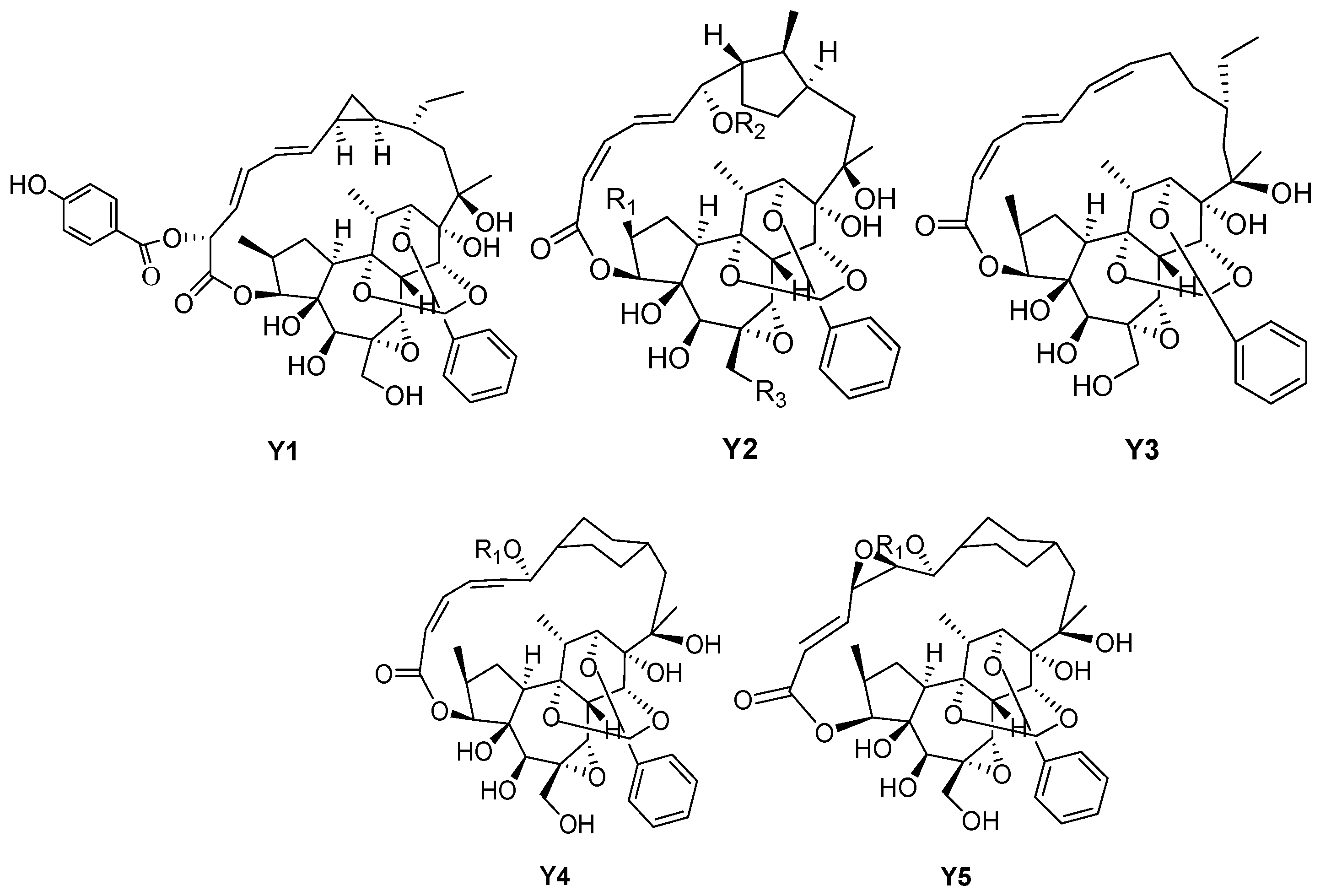
| 175 | Trigochilide A | – | – | – | Y1 |
| 176 | Rediocide A | Me | COCH2CH(CH3)2 | OH | Y2 |
| 177 | Rediocide C | Me | Bz | OH | Y2 |
| 178 | Rediocide E | H | COCH2CH(CH3)2 | OH | Y2 |
| 179 | Rediocide F | H | Bz | OH | Y2 |
| 180 | Trigonosin E | Me | COPh | OH | Y2 |
| 181 | Trigonosin F | Me | COPh | OH | Y2 |
| 182 | Trigothysoid N | Me | COCH2CH(CH3)2 | OH | Y2 |
| 183 | Trigothysoid O | Me | COPh | H | Y2 |
| 184 | Trigothysoid P | Me | COCH2CH(CH3)2 | H | Y2 |
| 185 | Trigonostempene D | Me | Val | H | Y2 |
| 186 | Trigochilide B | – | – | – | Y3 |
| 187 | Rediocide B | COCH2CH(CH3)2 | – | – | Y4 |
| 188 | Rediocide G | Bz | – | – | Y4 |
| 189 | Rediocide D | COCH2CH(CH3)2 | – | – | Y5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, Y.-X.; Shi, L.-L.; Zhang, D.-P.; Wei, H.-Y.; Si, Y.; Ma, G.-X.; Zhang, J. A Review on Daphnane-Type Diterpenoids and Their Bioactive Studies. Molecules 2019, 24, 1842. https://doi.org/10.3390/molecules24091842
Jin Y-X, Shi L-L, Zhang D-P, Wei H-Y, Si Y, Ma G-X, Zhang J. A Review on Daphnane-Type Diterpenoids and Their Bioactive Studies. Molecules. 2019; 24(9):1842. https://doi.org/10.3390/molecules24091842
Chicago/Turabian StyleJin, Yue-Xian, Lei-Ling Shi, Da-Peng Zhang, Hong-Yan Wei, Yuan Si, Guo-Xu Ma, and Jing Zhang. 2019. "A Review on Daphnane-Type Diterpenoids and Their Bioactive Studies" Molecules 24, no. 9: 1842. https://doi.org/10.3390/molecules24091842
APA StyleJin, Y.-X., Shi, L.-L., Zhang, D.-P., Wei, H.-Y., Si, Y., Ma, G.-X., & Zhang, J. (2019). A Review on Daphnane-Type Diterpenoids and Their Bioactive Studies. Molecules, 24(9), 1842. https://doi.org/10.3390/molecules24091842




