Aspalathin-Rich Green Rooibos Extract Lowers LDL-Cholesterol and Oxidative Status in High-Fat Diet-Induced Diabetic Vervet Monkeys
Abstract
:1. Introduction
2. Results
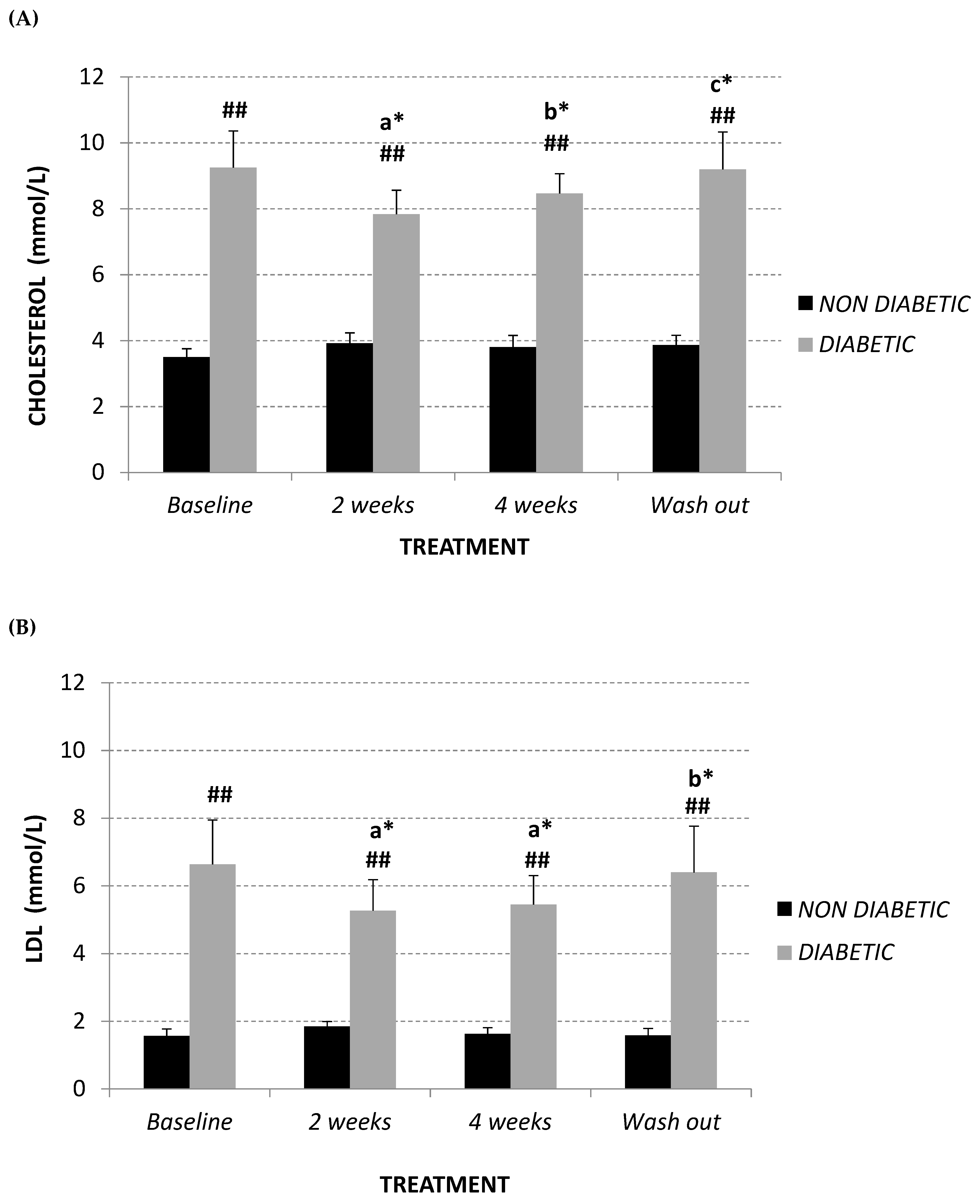
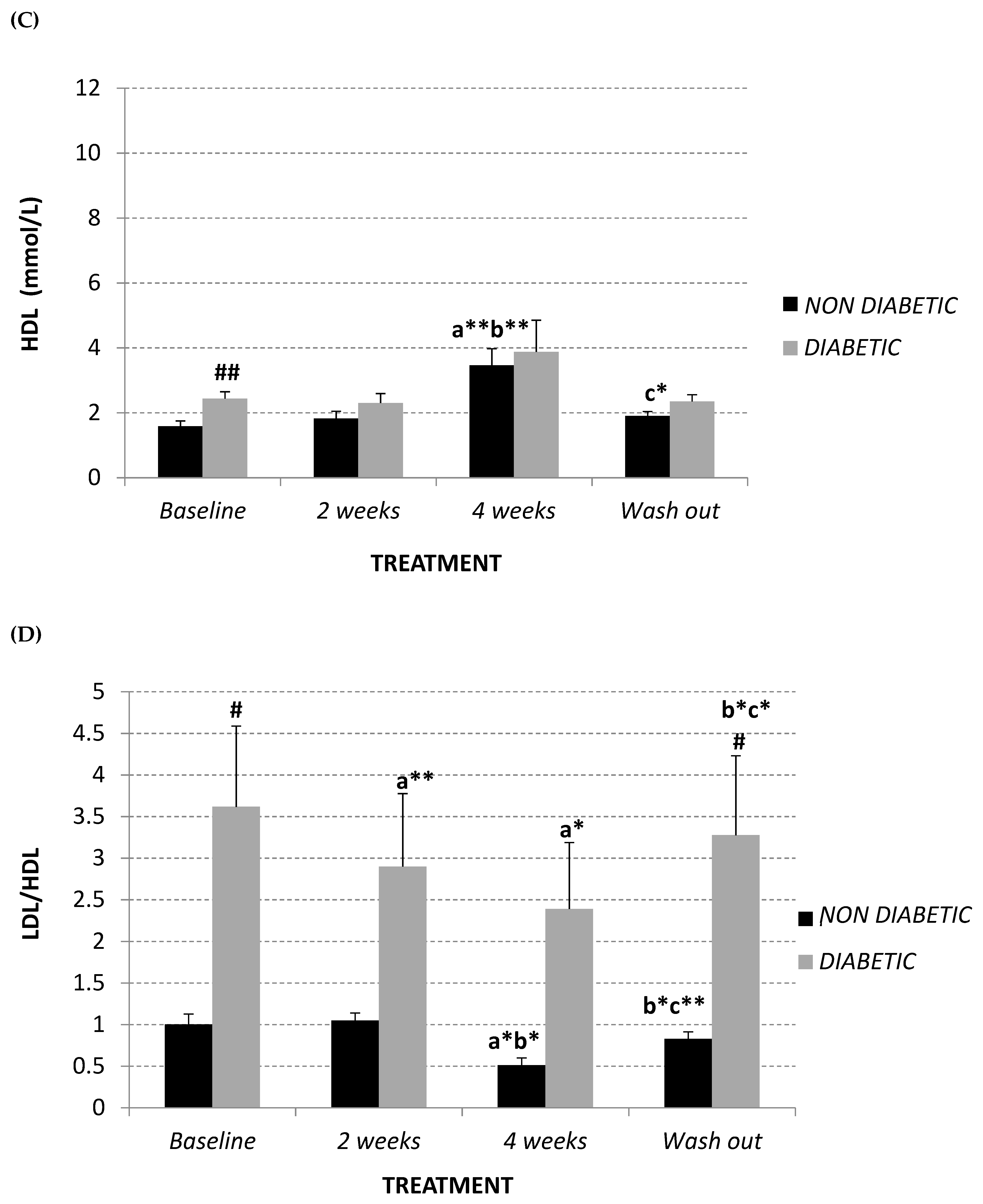
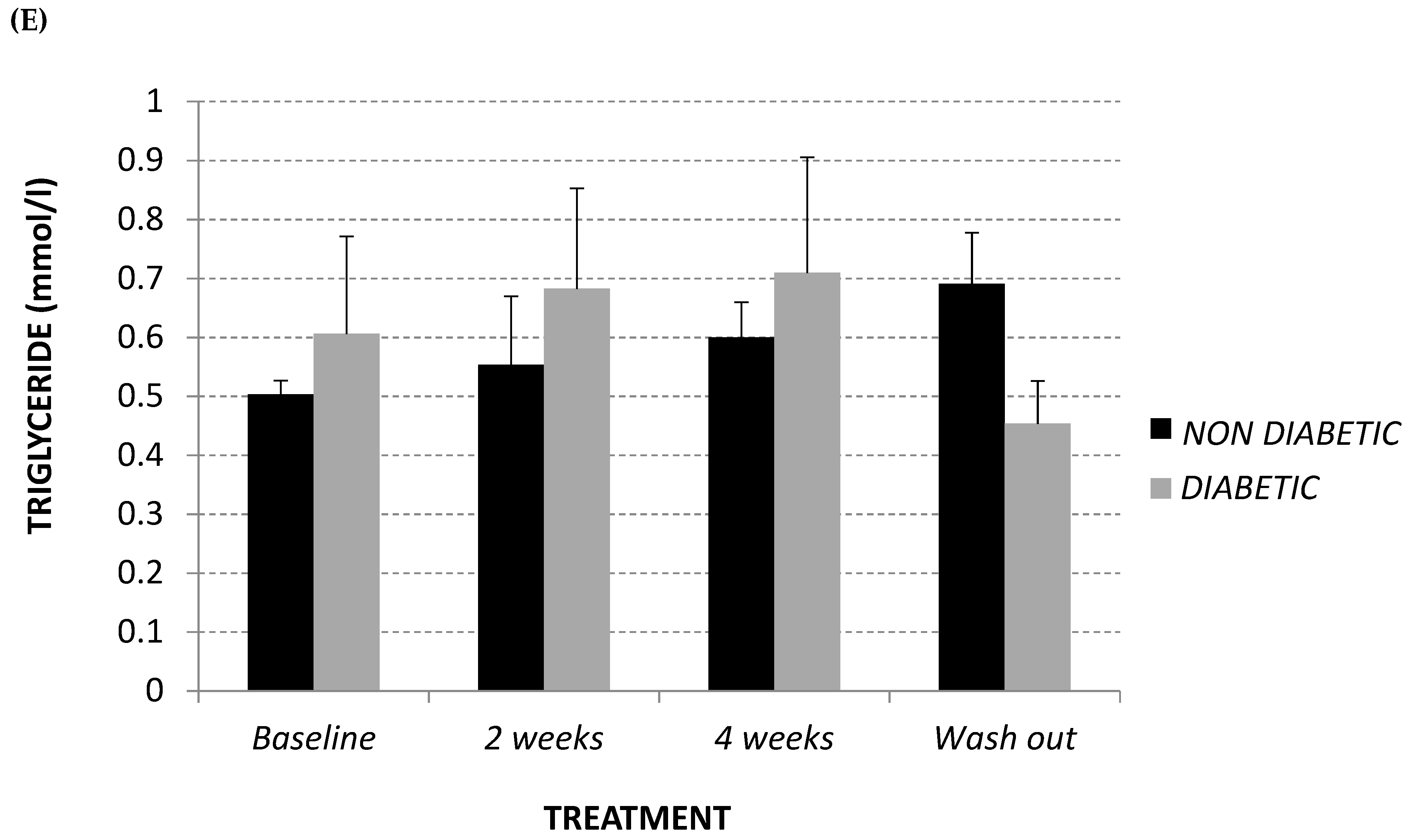
2.1. Lipid Profile
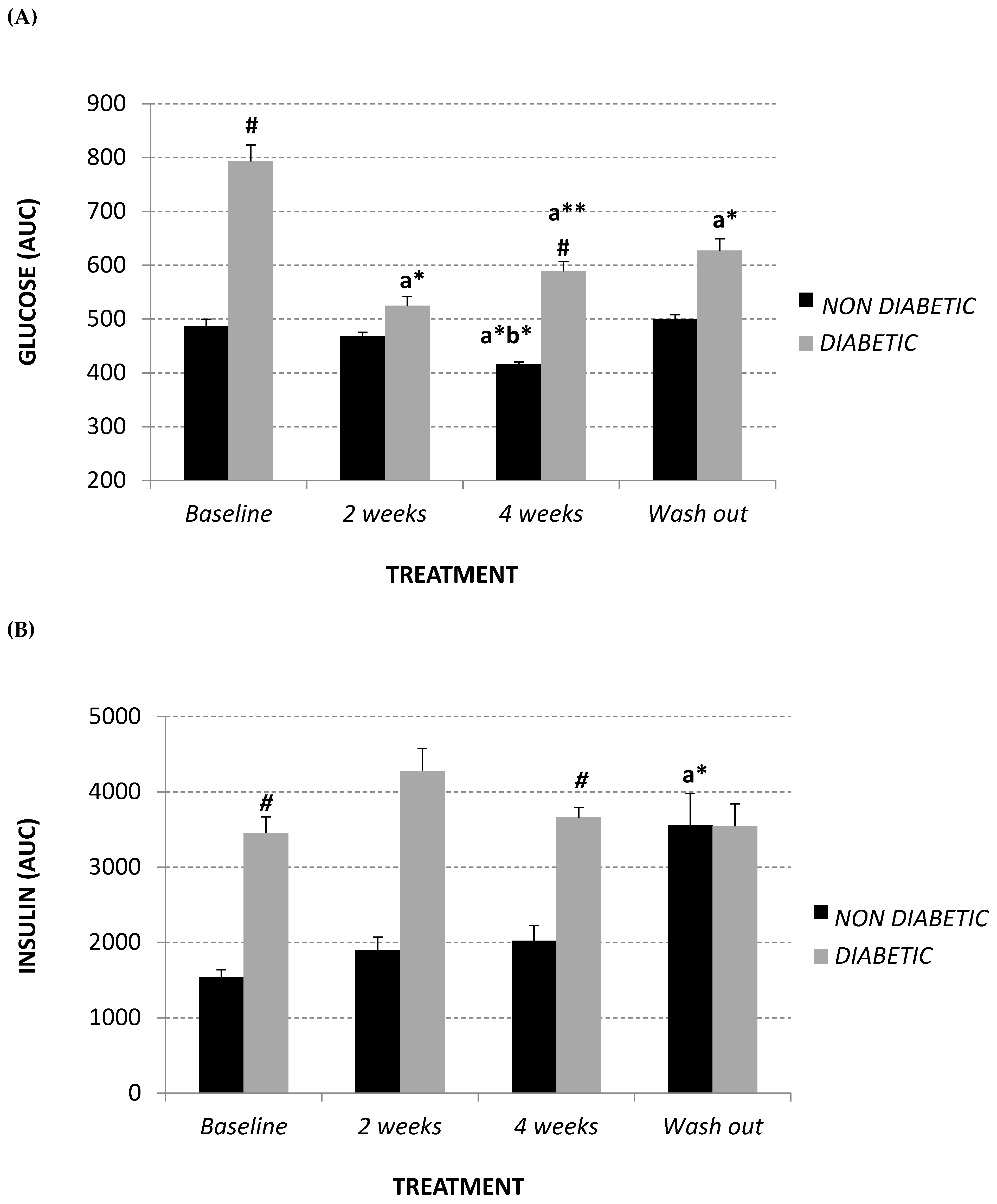
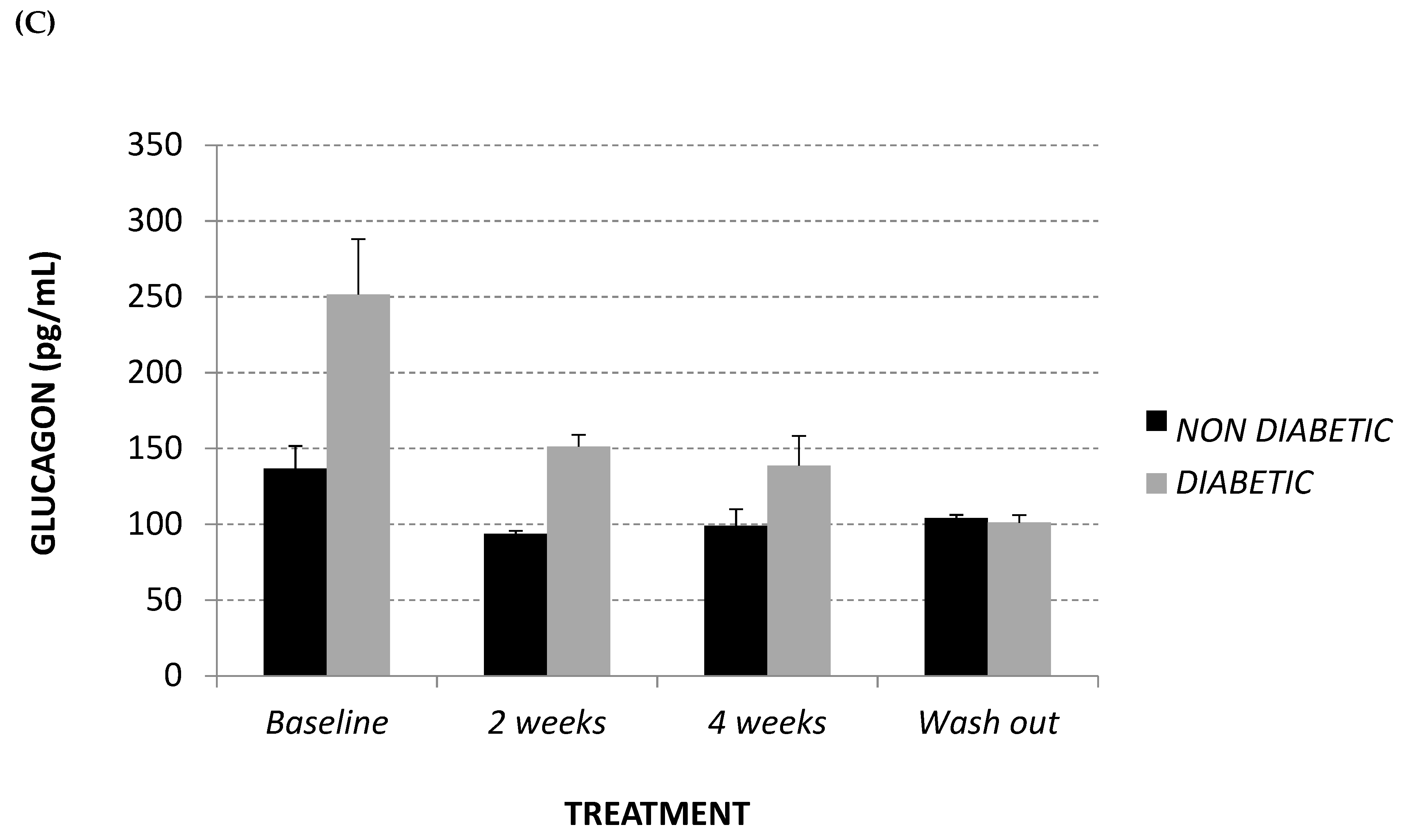
2.2. Glycemic Parameters
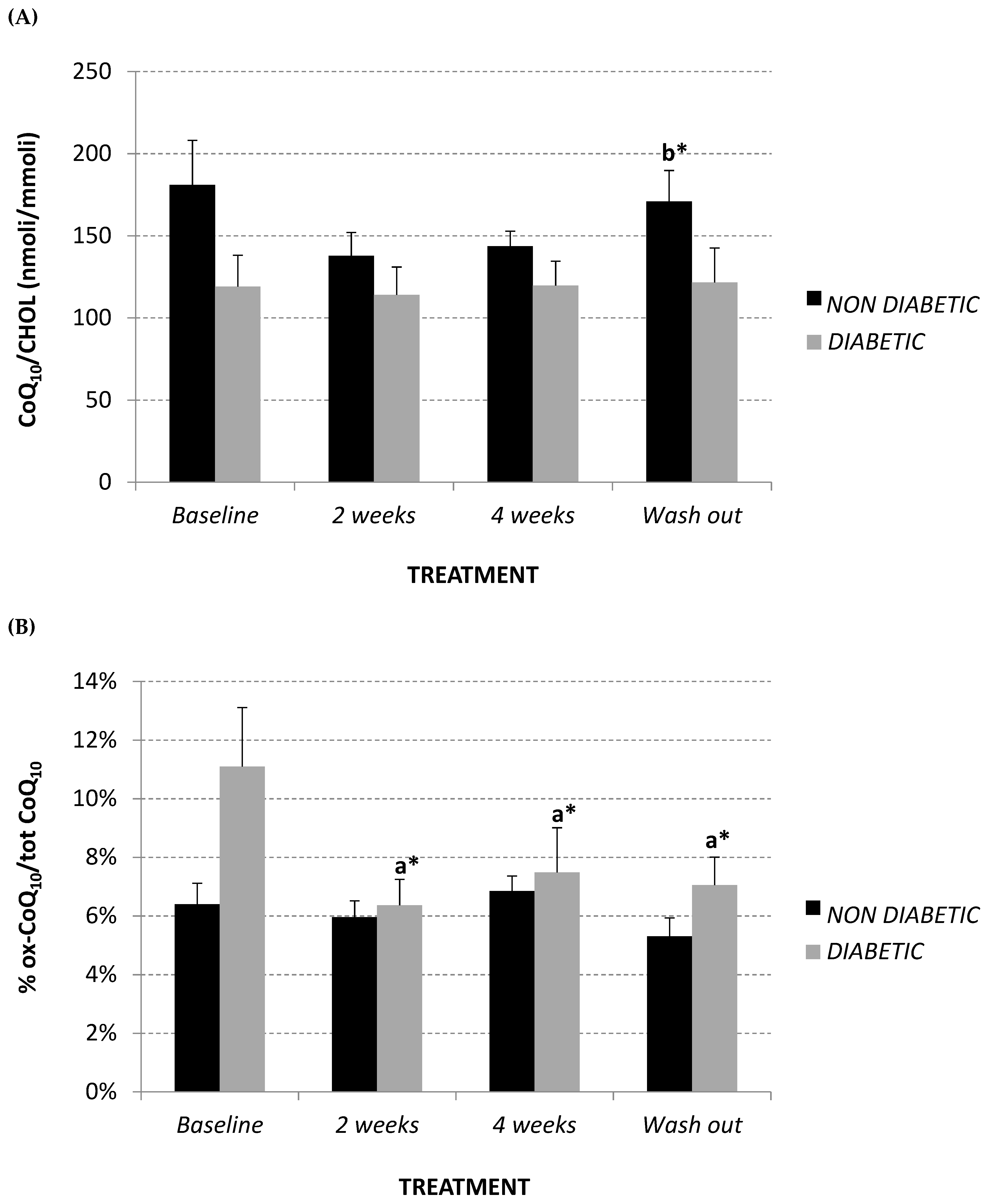
2.3. Total Plasma Coenzyme Q10 (CoQ10) Level and Oxidative Status
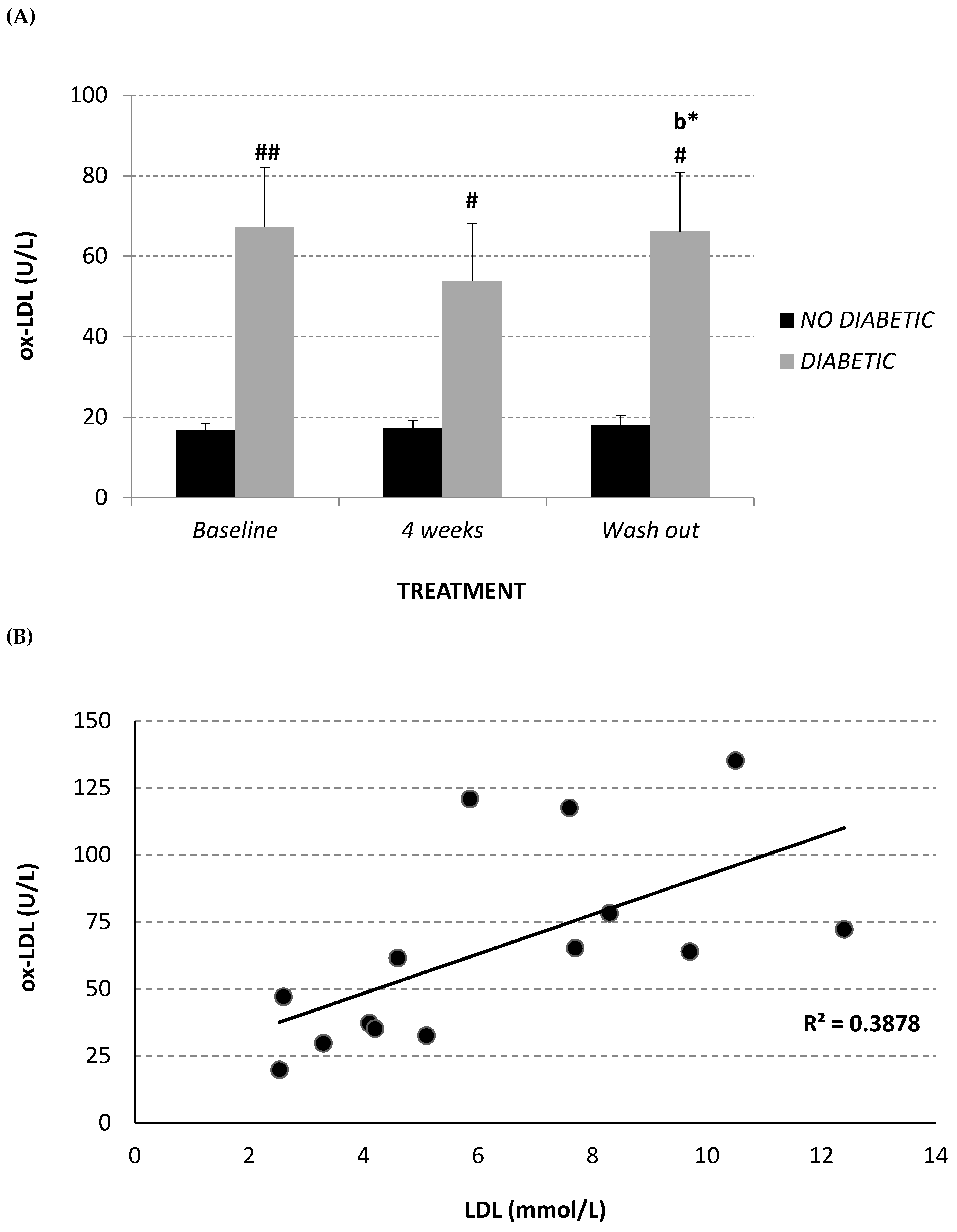
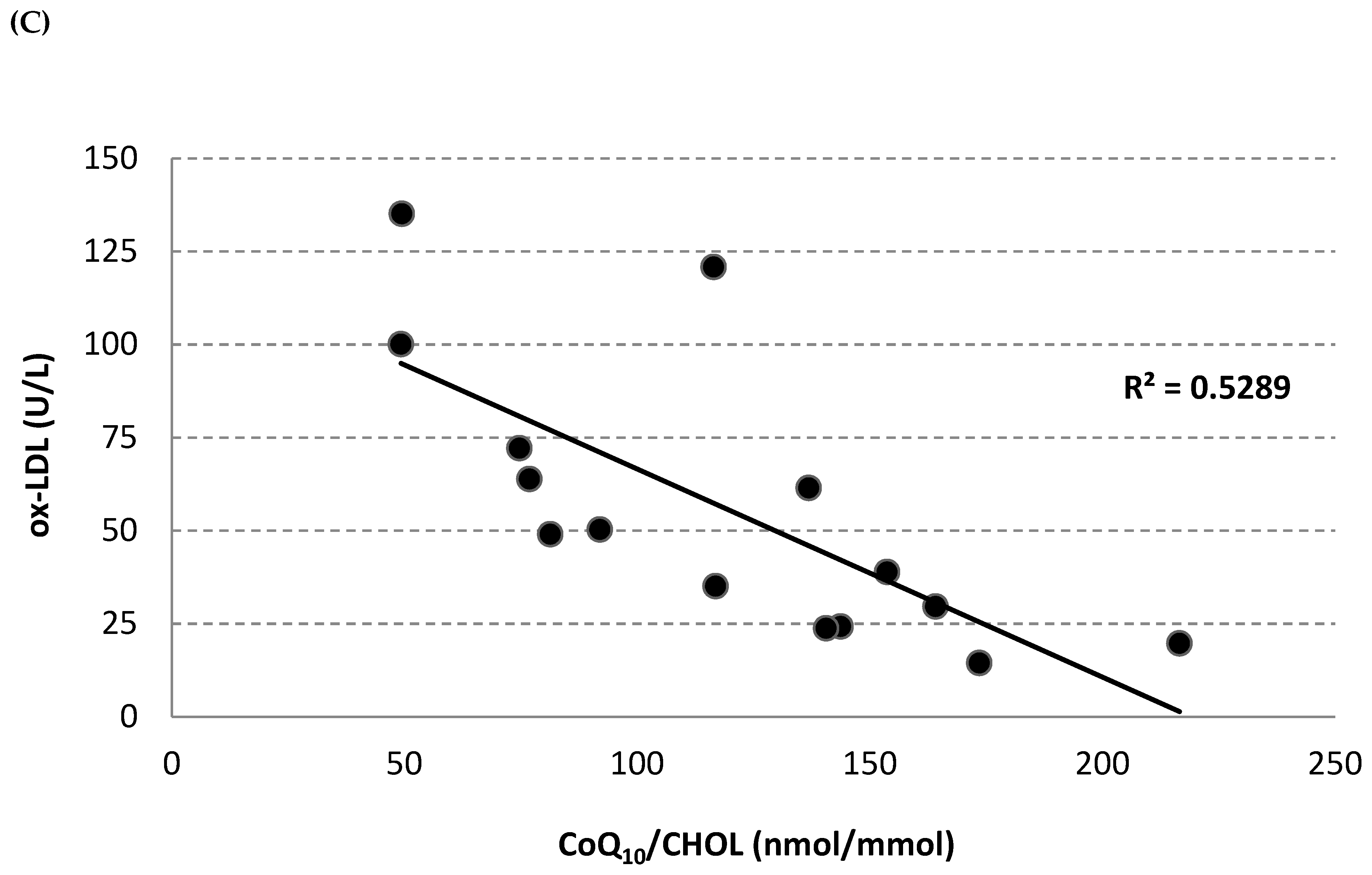
2.4. Circulating Oxidized LDL
3. Discussion
4. Material and Methods
4.1. Vervet Monkeys: Ethics
4.2. Aspalathin-Rich Green Rooibos Extract
4.3. Experimental Design
4.4. Lipid Profile
4.5. Total Plasma CoQ10 Levels and Oxidative Status
4.6. Circulating Oxidized-LDL
4.7. Glycemic Responses
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability
References
- International Diabetes Federation (IDF). IDF Diabetes Atlas 8th Edition. Available online: http://www.diabetesatlas.org/ (accessed on 2 April 2019).
- Zimmet, P.; Alberti, K.G.; Shaw, J. Global and societal implications of the diabetes epidemic. Nature 2001, 414, 782–787. [Google Scholar] [CrossRef] [PubMed]
- Cerf, M.E. Beta Cell Dysfunction and Insulin Resistance. Front. Endocrinol. 2013, 4, 37. [Google Scholar] [CrossRef] [PubMed]
- Emerging Risk Factors Collaboration. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: A collaborative meta-analysis of 102 prospective studies. Lancet 2010, 375, 2215–2222. [Google Scholar] [CrossRef]
- Pistrosch, F.; Natali, A.; Hanefeld, M. Is Hyperglycemia a Cardiovascular Risk Factor? Diabetes Care 2011, 34, S128–S131. [Google Scholar] [CrossRef] [PubMed]
- Das Evcimen, N.; King, G.L. The role of protein kinase C activation and the vascular complications of diabetes. Pharmacol. Res. 2007, 55, 498–510. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.D.; Nadler, J.L. Inflammatory mechanisms of diabetic complications. Curr. Diab. Rep. 2007, 7, 242–248. [Google Scholar] [CrossRef]
- Natarajan, R.; Nadler, J.L. Lipid inflammatory mediators in diabetic vascular disease. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1542–1548. [Google Scholar] [CrossRef]
- Brownlee, M. The pathobiology of diabetic complications: A unifying mechanism. Diabetes 2005, 54, 615–625. [Google Scholar] [CrossRef]
- Nishikawa, T.; Edelstein, D.; Du, X.L.; Yamagishi, S.; Matsumura, T.; Kaneda, Y.; Yorek, M.A.; Beebe, D.; Oates, P.J.; Hammes, H.P.; et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 2000, 404, 787–790. [Google Scholar] [CrossRef]
- Griendling, K.K.; FitzGerald, G.A. Oxidative stress and cardiovascular injury: Part I: Basic mechanisms and in vivo monitoring of ROS. Circulation 2003, 108, 1912–1916. [Google Scholar] [CrossRef]
- Turner, R.C.; Millns, H.; Neil, H.A.; Stratton, I.M.; Manley, S.E.; Matthews, D.R.; Holman, R.R. Risk factors for coronary artery disease in non-insulin dependent diabetes mellitus: United Kingdom Prospective Diabetes Study (UKPDS: 23). BMJ 1998, 316, 823–828. [Google Scholar] [CrossRef]
- Doucet, J.; Le Floch, J.P.; Bauduceau, B.; Verny, C. GERODIAB: Glycaemic control and 5-year morbidity/mortality of type 2 diabetic patients aged 70 years and older: 1. Description of the population at inclusion. Diabetes Metab. 2012, 38, 523–530. [Google Scholar] [CrossRef]
- Burchardt, P.; Żurawski, J.; Zuchowski, B.; Kubacki, T.; Murawa, D.; Wiktorowicz, K.; Wysocki, H. Low-density lipoprotein, its susceptibility to oxidation and the role of lipoprotein-associated phospholipase A2 and carboxyl ester lipase lipases in atherosclerotic plaque formation. Arch. Med. Sci. 2013, 9, 151–158. [Google Scholar] [CrossRef]
- Graham, I.; Atar, D.; Borch-Johnsen, K.; Boysen, G.; Burell, G.; Cifkova, R.; Dallongeville, J.; De Backer, G.; Ebrahim, S.; Gjelsvik, B.; et al. ESC Committee for Practice Guidelines. European guidelines on cardiovascular disease prevention in clinical practice: Executive summary. Atherosclerosis 2007, 194, 1–45. [Google Scholar] [CrossRef]
- Eldor, R.; Raz, I. American Diabetes Association Indications for Statins in Diabetes Is there evidence? Diabetes Care 2009, 32 (Suppl. 2), S384–S391. [Google Scholar] [CrossRef]
- Gibbs, R.A.; Rogers, J.; Katze, M.G.; Bumgarner, R.; Weinstock, G.M.; Mardis, E.R.; Remington, K.A.; Strausberg, R.L.; Venter, J.C.; Wilson, R.K.; et al. Evolutionary and biomedical insights from the rhesus macaque genome. Science 2007, 316, 222–234. [Google Scholar]
- Rudel, L.L.; Reynolds, J.A.; Bullock, B.C. Nutritional effects on blood lipid and HDL-cholesterol concentrations in two subspecies of African green monkeys (Cercopithecus aethiops). J. Lipid Res. 1981, 22, 278–286. [Google Scholar]
- Weight, M.J.; Benade, A.J.S.; Lombard, C.J.; Fincham, J.E.; Marais, M.; Dando, B.; Seier, J.V.; Kritchevsky, D. Low density lipoprotein kinetics in African Green monkeys showing variable cholesterolaemic responses to diets realistic for westernized people. Atherosclerosis 1988, 73, 1–11. [Google Scholar] [CrossRef]
- Martin, S.; Palmour, R.M.; Goldwater, R.; Gutkowsa, J.; Hughes, C.; Hamet, P.; Ervin, F.R. Characterization of a primate model of hypertension. The response of hypertensive and normotensive male vervets (Cercopithecus aethiops) to cold pressor stress, captopril administration, and acute bolus of atrial natriuretic factor. Am. J. Hypertens. 1990, 3, 27–32. [Google Scholar] [CrossRef]
- Suckling, K.; Jackson, B. Animal models of human lipid metabolism. Prog. Lipid Res. 1993, 32, 1–24. [Google Scholar] [CrossRef]
- Fincham, J.E.; Quack, G.; Wuelfroth, O.P.; Benade, A.J. Confirmation of efficacy of etofibrate in nonhuman primates which model human lesion types I-IV. Arzneim.-Forsch./Drug Res. 1996, 46, 519–525. [Google Scholar]
- Fairbanks, L. Alternative old world primate models for non-AIDS research: African green monkeys (vervets). In: Rhesus monkey demands in biomedical research: a workshop report. ILAR J. 2003, 44, 222–235. [Google Scholar]
- Wallace, J.M.; Schwarz, M.; Coward, P.; Houze, J.; Sawyer, J.K.; Kelley, K.L.; Chai, A.; Rudel, L.L. Effects of peroxisome proliferator-activated receptor alpha/delta agonists on HDL-cholesterol in vervet monkeys. J. Lipid Res. 2005, 46, 1009–1016. [Google Scholar] [CrossRef]
- Kruger, M.; Smuts, C.M.; Benadé, A.J.S.; Fincham, J.E.; Lombard, C.J.; Albertse, E.A.; van der Merwe, K.J. Comparison of the effect of the amount and degree of unsaturation of dietary fat on plasma low density lipoproteins in vervet monkeys. Lipids 1992, 27, 733–739. [Google Scholar] [CrossRef]
- Fincham, J.E.; Benade, J.S.; Kruger, M.; Smuts, C.M.; Gobregts, E.; Chalton, D.O.; Kritchevsky, D. Atherosclerosis: Aortic lipid changes induced by diets suggest diffuse disease with focal severity in primates that model human atheromas. Nutrition 1998, 14, 17–22. [Google Scholar] [CrossRef]
- St. Clair, R.W.; Wood, L.L.; Clarkson, T.B. Effect of sucrose polyester on plasma lipids and cholesterol absorption in African Green monkeys with variable hypercholesterolaemic response to dietary cholesterol. Metabolism 1981, 30, 176–183. [Google Scholar] [CrossRef]
- Smuts, C.M.; Kruger, M.; van Jaarsveld, P.J.; Fincham, J.E.; Schall, R.; van der Merwe, K.J.; Benade, A.J.S. The influence of fish oil supplementation on plasma lipoprotein and arterial lipids in vervet monkeys with established atherosclerois. Prostaglandins Leukot. Essent. Fat. Acids 1992, 47, 129–138. [Google Scholar] [CrossRef]
- Kavanagh, K.; Fairbanks, L.A.; Bailey, J.N.; Jorgensen, M.J.; Wilson, M.; Zhang, L.; Rudel, L.L.; Wagner, J.D. Characterization and heritability of obesity and associated risk factors in vervet monkeys. Obes. Silver Spring Md. 2007, 15, 1666–1674. [Google Scholar] [CrossRef]
- Van Jaarsveld, P.J.; Benadé, A.J.S. Effect of palm olein oil in a moderate-fat diet on low-density lipoprotein composition in non-human primates. Asia Pac. J. Clin. Nutr. 2002, 11, S416–S423. [Google Scholar] [CrossRef]
- Papanicolas, I.; Woskie, R.L.; Jha, A.K. Health Care Spending in the United States and Other High-Income Countries. JAMA 2018, 319, 1024–1039. [Google Scholar] [CrossRef]
- Von Gadow, A.; Joubert, E.; Hansmann, C.F. Comparison of the antioxidant activity of aspalathin with that of other plant phenols of rooibos tea (Aspalathus linearis), α-tocopherol, BHT, and BHA. J. Agric. Food Chem. 1997, 45, 632–638. [Google Scholar] [CrossRef]
- Marnewick, J.; Joubert, E.; Joseph, S.; Swanevelder, S.; Swart, P.; Gelderbolm, W. Inhibition of tumor promotion in mouse skin by extracts of Rooibos (Aspalathus linearis) and honeybush (Cyclopia intermedia), unique Southern African herbal teas. Cancer Lett. 2005, 224, 193–202. [Google Scholar] [CrossRef]
- Simon, M.; Horvoska, L.; Greksak, M.; Dusinsky, R.; Nakano, M. Antihemolytic effect of Rooibos tea (Aspalathus linearis) on red cells of Japanese quails. Gen. Physiol. Biophys. 2000, 19, 365–371. [Google Scholar]
- Standley, L.; Winterton, P.; Marnewick, J.L.; Gelderblom, W.C.; Joubert, E.; Britz, I.J. Influence of processing stages on antimutagenic and antioxidant potentials of rooibos tea. J. Agric. Food Chem. 2001, 49, 114–117. [Google Scholar] [CrossRef]
- Snijman, P.W.; Swanevelder, S.; Joubert, E.; Green, I.R.; Gelderblom, W.C. The antimutagenic activity of the major flavonoids of rooibos (Aspalathus linearis): Some dose-response effects on mutagen activation-flavonoid interactions. Mutat. Res. 2007, 631, 111–123. [Google Scholar] [CrossRef]
- Na, H.K.; Mossanda, K.S.; Lee, J.Y.; Surh, Y.J. Inhibition of phorbol ester-induced COX-2 expression by some edible African plants. Biofactors 2004, 21, 149–153. [Google Scholar] [CrossRef]
- Mazibuko, S.E.; Muller, C.J.; Joubert, E.; de Beer, D.; Johnson, R.; Opoku, A.R.; Louw, J. Amelioration of palmitate-induced insulin resistance in C₂C₁₂ muscle cells by rooibos (Aspalathus linearis). Phytomedicine 2013, 20, 813–819. [Google Scholar] [CrossRef]
- Kawano, A.; Nakamura, H.; Hata, S.; Minakawa, M.; Miura, Y.; Yagasaki, K. Hypoglycemic effect of aspalathin, a rooibos tea component from Aspalathus linearis, in type 2 diabetic model db/db mice. Phytomedicine 2009, 16, 437–443. [Google Scholar] [CrossRef]
- Son, M.J.; Minakawa, M.; Miura, Y.; Yagasaki, K. Aspalathin improves hyperglycemia and glucose intolerance in obese diabetic ob/ob mice. Eur. J. Nutr. 2013, 52, 1607–1619. [Google Scholar] [CrossRef]
- Kamakura, M.J.S.; de Beer, D.; Joubert, E.; Miura, Y.; Yagasaki, K. Antidiabetic effect of green rooibos (Aspalathus linearis) extract in cultured cells and type 2 diabetic model KK-Ay mice. Cytotechnology 2015, 67, 699–710. [Google Scholar] [CrossRef]
- Marnewick, J.L.; Rautenbach, F.; Venter, I.; Neethling, H.; Blackhurst, D.M.; Wolmarans, P.; Macharia, M. Effects of rooibos (Aspalathus linearis) on oxidative stress and biochemical parameters in adults at risk for cardiovascular disease. J. Ethnopharmacol. 2011, 133, 46–52. [Google Scholar] [CrossRef]
- Louw, J.; Woodroof, C.; Seier, J.; Wolfe-Coote, S.A. The effect of diet on the Vervet monkey endocrine pancreas. J. Med. Primatol. 1997, 26, 307–311. [Google Scholar] [CrossRef]
- Millán, J.; Pintó, X.; Muñoz, A.; Zúñiga, M.; Rubiés-Prat, J.; Pallardo, L.F.; Masana, L.; Mangas, A.; Hernández-Mijares, A.; González-Santos, P.; et al. Lipoprotein ratios: Physiological significance and clinical usefulness in cardiovascular prevention. Vasc. Health Risk Manag. 2009, 5, 757–765. [Google Scholar]
- Barter, P. The role of HDL-cholesterol in preventing atherosclerotic disease. Eur. Heart J. Suppl. 2005, 7, F4–F8. [Google Scholar] [CrossRef]
- Wülfroth, P.; Richter, C.M.; Burkard, M. Etofibrate treatment alters low-density lipoprotein susceptibility to lipid peroxidation. Drugs Exp. Clin. Res. 1992, 18, 469–474. [Google Scholar]
- Cholesterol Treatment Trialists’ (CTT) Collaborators; Kearney, P.M.; Blackwell, L.; Collins, R.; Keech, A.; Simes, J.; Peto, R.; Armitage, J.; Baigent, C. Efficacy of cholesterol-lowering therapy in 18,686 people with diabetes in 14 randomised trials of statins: A meta-analysis. Lancet 2008, 371, 117–125. [Google Scholar]
- Sukhija, R.; Prayaga, S.; Marashdeh, M.; Bursac, Z.; Kakar, P.; Bansal, D.; Sachdeva, R.; Kesan, S.H.; Mehta, J.L. Effect of statins on fasting plasma glucose in diabetic and nondiabetic patients. J. Investig. Med. 2009, 57, 495–499. [Google Scholar] [CrossRef]
- Littarru, G.P.; Langsjoen, P. Coenzyme Q10 and statins: Biochemical and clinical implications. Mitochondrion 2007, 7, S168–S174. [Google Scholar] [CrossRef]
- Littarru, G.P.; Tiano, L.; Belardinelli, R.; Watts, G.F. Coenzyme Q(10), endothelial function, and cardiovascular disease. Biofactors 2011, 37, 366–373. [Google Scholar] [CrossRef]
- Molyneux, S.L.; Florkowski, C.M.; George, P.M.; Pilbrow, A.P.; Frampton, C.M.; Lever, M.; Richards, A.M. Coenzyme Q10: An independent predictor of mortality in chronic heart failure. J. Am. Coll. Cardiol. 2008, 52, 1435–1441. [Google Scholar] [CrossRef]
- Kim, M.; Kim, Y. Hypocholesterolemic effects of curcumin via up-regulation of cholesterol 7a-hydroxylase in rats fed a high fat diet. Nutr. Res. Pract. 2010, 4, 191–195. [Google Scholar] [CrossRef]
- Sun, W.; Yan, Y.; Dong, F. Progression of tocotrienols. J. Hyg. Res. 2004, 33, 243–245. [Google Scholar]
- Iqbal, J.; Minhajuddin, M.; Beg, Z.H. Suppression of 7,12-dimethylbenz[alpha]anthracene-induced carcinogenesis and hypercholesterolaemia in rats by tocotrienol-rich fraction isolated from rice bran oil. Eur. J. Cancer Prev. 2003, 12, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Bursill, C.A.; Abbey, M.; Roach, P.D. A green tea extract lowers plasma cholesterol by inhibiting cholesterol synthesis and up-regulating the LDL receptor in the cholesterol-fed rabbit. Atherosclerosis 2007, 193, 86–93. [Google Scholar] [CrossRef]
- Koo, S.I.; Noh, S.K. Green tea as inhibitor of the intestinal absorption of lipids: potential mechanism for its lipid-lowering effect. J. Nutr. Biochem. 2007, 18, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Beltrãn-Debãn, R.; Rull, A.; Rodríguez-Sanabria, F.; Iswaldi, I.; Herranz-López, M.; Aragonès, G.; Camps, J.; Alonso-Villaverde, C.; Menéndez, J.A.; Micol, V.; et al. Continuous administration of polyphenols from aqueous rooibos (Aspalathus linearis) extract ameliorates dietary-induced metabolic disturbances in hyperlipidemic mice. Phytomedicine 2011, 18, 414–424. [Google Scholar]
- Lagendijk, J.; Ubbink, J.B.; Vermaak, W.J.J. Measurement of the ratio between the reduced and oxidized forms of coenzyme Q10 in human plasma as a possible marker of oxidative stress. Lipid Res. 1996, 37, 67–75. [Google Scholar]
- Menke, T.; Niklowitz, P.; Adam, S.; Weber, M.; Schlüter, B.; Andler, W. Simultaneous detection of ubiquinol-10, ubiquinone-10, and tocopherols in human plasma microsamples and macrosamples as a marker of oxidative damage in neonates and infants. Anal. Biochem. 2000, 282, 209–217. [Google Scholar] [CrossRef]
- Weber, C.; Sejersgård Jakobsen, T.; Mortensen, S.A.; Paulsen, G.; Hølmer, G. Antioxidative effect of dietary coenzyme Q10 in human blood plasma. Int. J. Vitam. Nutr. Res. 1994, 64, 311–315. [Google Scholar]
- Yamamoto, Y.; Yamashita, S. Plasma ratio of ubiquinol and ubiquinone as a marker of oxidative stress. Mol. Asp. Med. 1997, 18, S79–S84. [Google Scholar] [CrossRef]
- Stocker, R.; Bowry, V.W.; Frei, B. Ubiquinol-10 protects human low density lipoprotein more efficiently against lipid peroxidation than does a-tocopherol. Proc. Natl. Acad. Sci. USA 1991, 88, 1646–1650. [Google Scholar] [CrossRef]
- Ernster, L.; Forsmark-Andrèe, P. Ubiquinol: An endogenous antioxidant in aerobic organisms. Clin. Investig. 1993, 71, S60–S65. [Google Scholar] [CrossRef]
- Esterbauer, H.; Wag, G.; Puhl, H. Lipid peroxidation and its role in atherosclerosis. Br. Med. Bull. 1993, 49, 566–576. [Google Scholar] [CrossRef] [PubMed]
- Bowry, V.W.; Stanley, K.K.; Stocker, R. High density lipoprotein is the major carrier of lipid hydroperoxides in human blood plasma from fasting donors. Proc. Natl. Acad. Sci. USA 1992, 89, 10316–10320. [Google Scholar] [CrossRef] [PubMed]
- Patel, O.; Muller, C.; Joubert, E.; Louw, J.; Rosenkranz, B.; Awortwe, C. Inhibitory Interactions of Aspalathus linearis (rooibos) extracts and compounds, aspalathin and z-2-(β-d-Glucopyranosyloxy)-3-phenylpropenoic acid, on cytochromes metabolizing hypoglycemic and hypolipidemic drugs. Molecules 2016, 21, 1515. [Google Scholar] [CrossRef] [PubMed]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef]
- Orlando, P.; Silvestri, S.; Brugè, F.; Tiano, L.; Kloting, I.; Falcioni, G.; Polidori, C. High-fat diet-induced met-hemoglobin formation in rats prone (WOKW) or resistant (DA) to the metabolic syndrome: Effect of CoQ10 supplementation. Biofactors 2014, 40, 603–609. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| Monkey Groups | Gender | Date of Birth | Age at Baseline (years) | Body Weight During Study (kg) | ||||
|---|---|---|---|---|---|---|---|---|
| Baseline | 2 Weeks | 4 Weeks | Wash-Out | |||||
| Non-diabetic | M268 | Male | 25-set-00 | 14 | 4.7 | 4.6 | 4.5 | 4.8 |
| M1077 | Male | Wild caught | Mature adult | 4.9 | 4.9 | 4.8 | 5.0 | |
| M248 | Male | 18-set-99 | 16 | 4.4 | 4.3 | 4.3 | 4.4 | |
| M1068 | Male | Wild caught | Mature adult | 4.8 | 4.8 | 4.8 | 4.9 | |
| M205 | Female | 13-mag-98 | 17 | 3.3 | 3.3 | 3.3 | 3.3 | |
| M234 | Female | 14-apr-99 | 16 | 3.7 | 3.8 | 3.8 | 3.8 | |
| Diabetic | M49 | Female | 27-ago-90 | 25 | 3.6 | 3.6 | 3.6 | 3.5 |
| M343 | Male | 21-mar-03 | 12 | 5.9 | 6.0 | 5.9 | 5.7 | |
| M281 | Male | 31-gen-01 | 14 | 5.5 | 5.7 | 6.1 | 5.7 | |
| M1083 | Male | 01-giu-99 | 16 | 6.1 | 6.3 | 6.5 | 6.2 | |
| M403 | Female | Wild caught | Mature adult | 5.2 | 5.2 | 5.3 | 5.3 | |
| M39 | Female | 19-ott-89 | 25 | 4.6 | 4.8 | 4.9 | 4.8 | |
| M238 | Male | 19-mag-99 | 16 | 5.1 | 5.0 | 5.1 | 5.3 | |
| M136 | Female | 01-feb-95 | 20 | 4.0 | 3.9 | 3.7 | 3.8 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Orlando, P.; Chellan, N.; Louw, J.; Tiano, L.; Cirilli, I.; Dludla, P.; Joubert, E.; Muller, C.J.F. Aspalathin-Rich Green Rooibos Extract Lowers LDL-Cholesterol and Oxidative Status in High-Fat Diet-Induced Diabetic Vervet Monkeys. Molecules 2019, 24, 1713. https://doi.org/10.3390/molecules24091713
Orlando P, Chellan N, Louw J, Tiano L, Cirilli I, Dludla P, Joubert E, Muller CJF. Aspalathin-Rich Green Rooibos Extract Lowers LDL-Cholesterol and Oxidative Status in High-Fat Diet-Induced Diabetic Vervet Monkeys. Molecules. 2019; 24(9):1713. https://doi.org/10.3390/molecules24091713
Chicago/Turabian StyleOrlando, Patrick, Nireshni Chellan, Johan Louw, Luca Tiano, Ilenia Cirilli, Phiwayinkosi Dludla, Elizabeth Joubert, and Christo J.F. Muller. 2019. "Aspalathin-Rich Green Rooibos Extract Lowers LDL-Cholesterol and Oxidative Status in High-Fat Diet-Induced Diabetic Vervet Monkeys" Molecules 24, no. 9: 1713. https://doi.org/10.3390/molecules24091713
APA StyleOrlando, P., Chellan, N., Louw, J., Tiano, L., Cirilli, I., Dludla, P., Joubert, E., & Muller, C. J. F. (2019). Aspalathin-Rich Green Rooibos Extract Lowers LDL-Cholesterol and Oxidative Status in High-Fat Diet-Induced Diabetic Vervet Monkeys. Molecules, 24(9), 1713. https://doi.org/10.3390/molecules24091713







