Studies Towards Hypoxia-Activated Prodrugs of PARP Inhibitors
Abstract
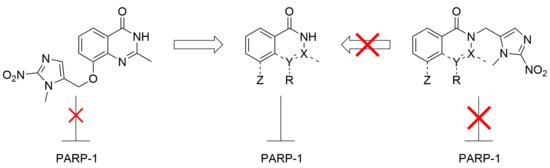
1. Introduction
2. Results and Discussion
2.1. Synthesis
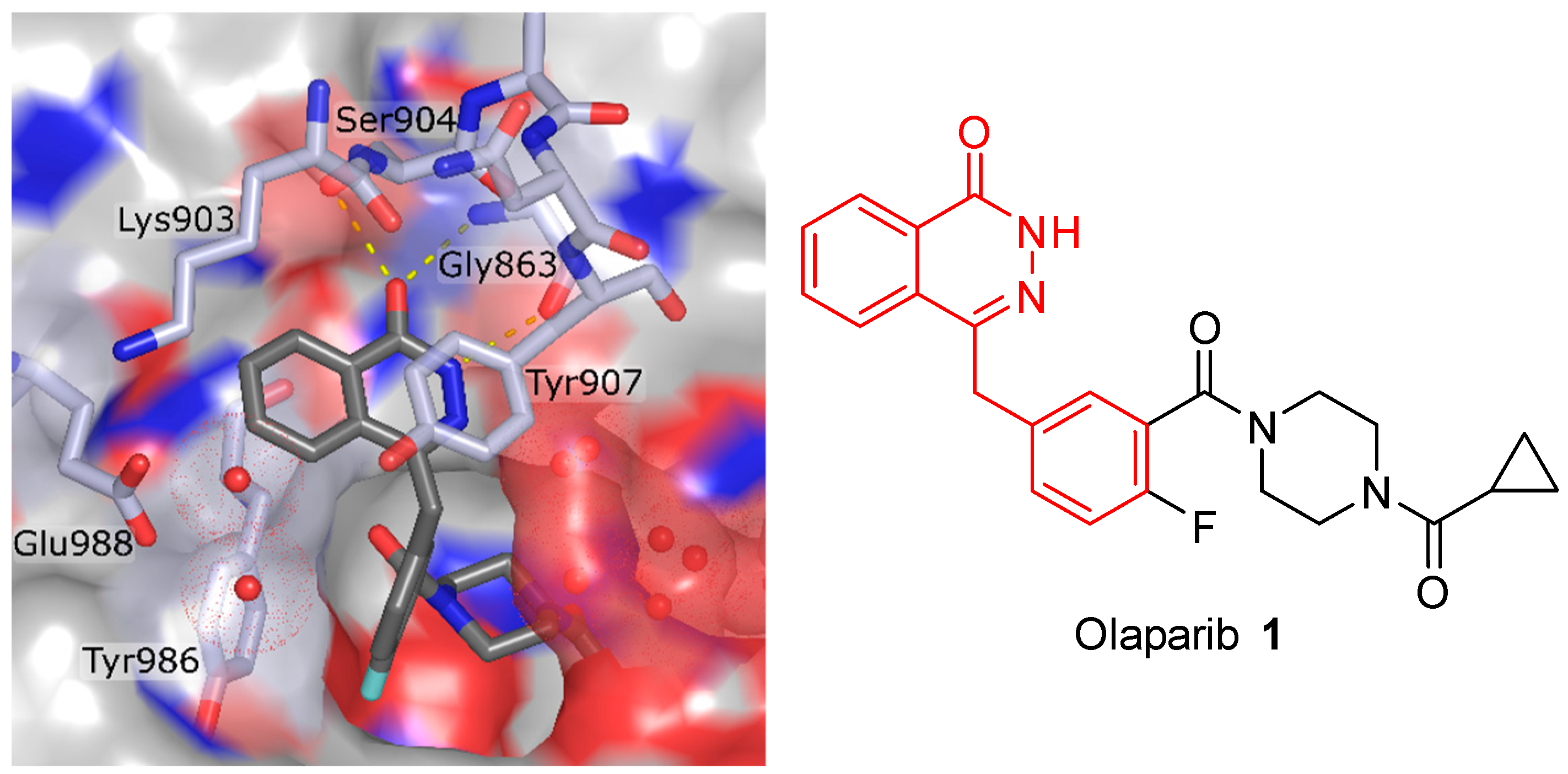
2.1.1. Synthesis of PARPi
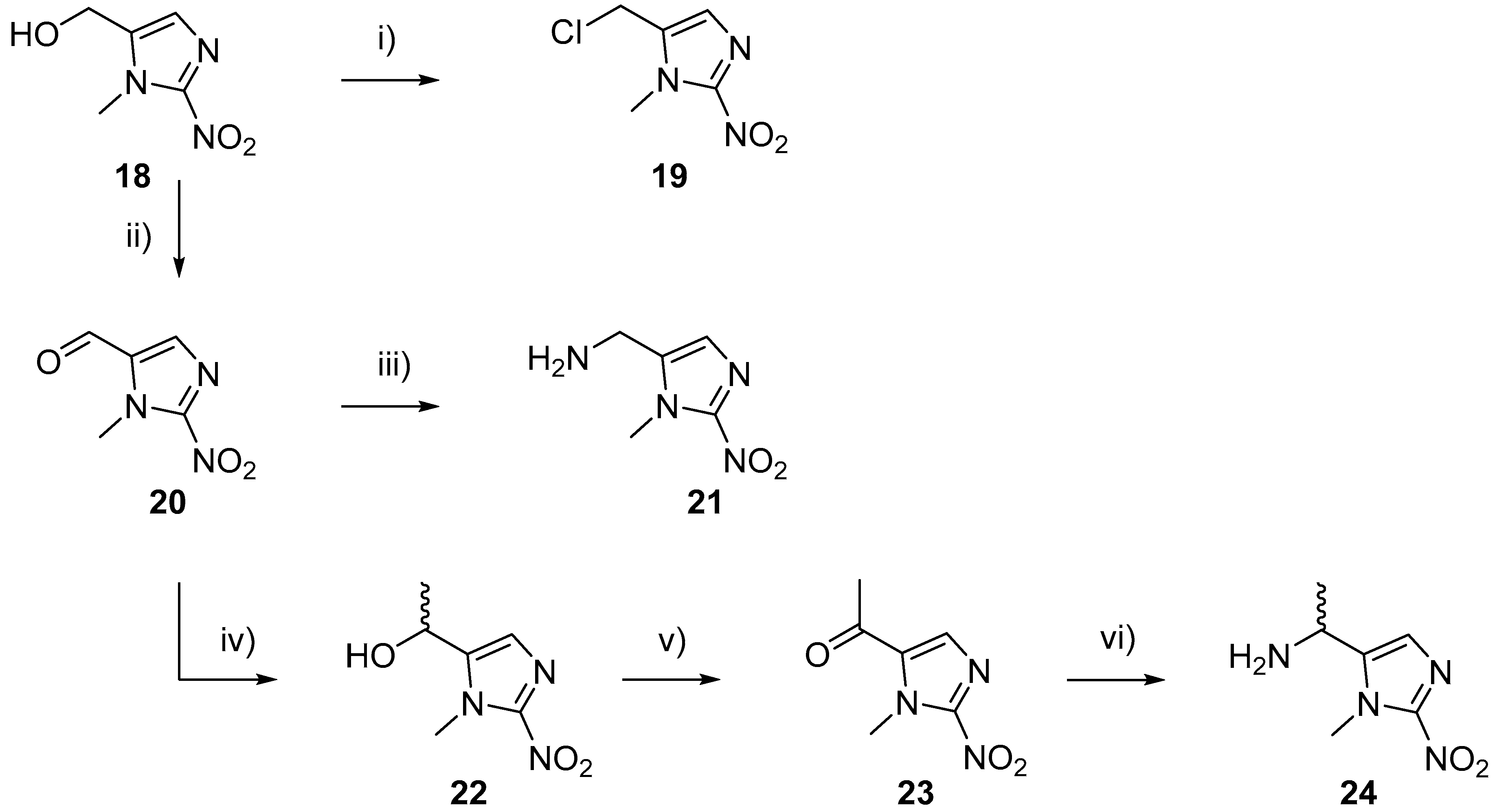
2.1.2. Synthesis of Model HAPs
2.2. Biochemical PARP-1 Inhibition
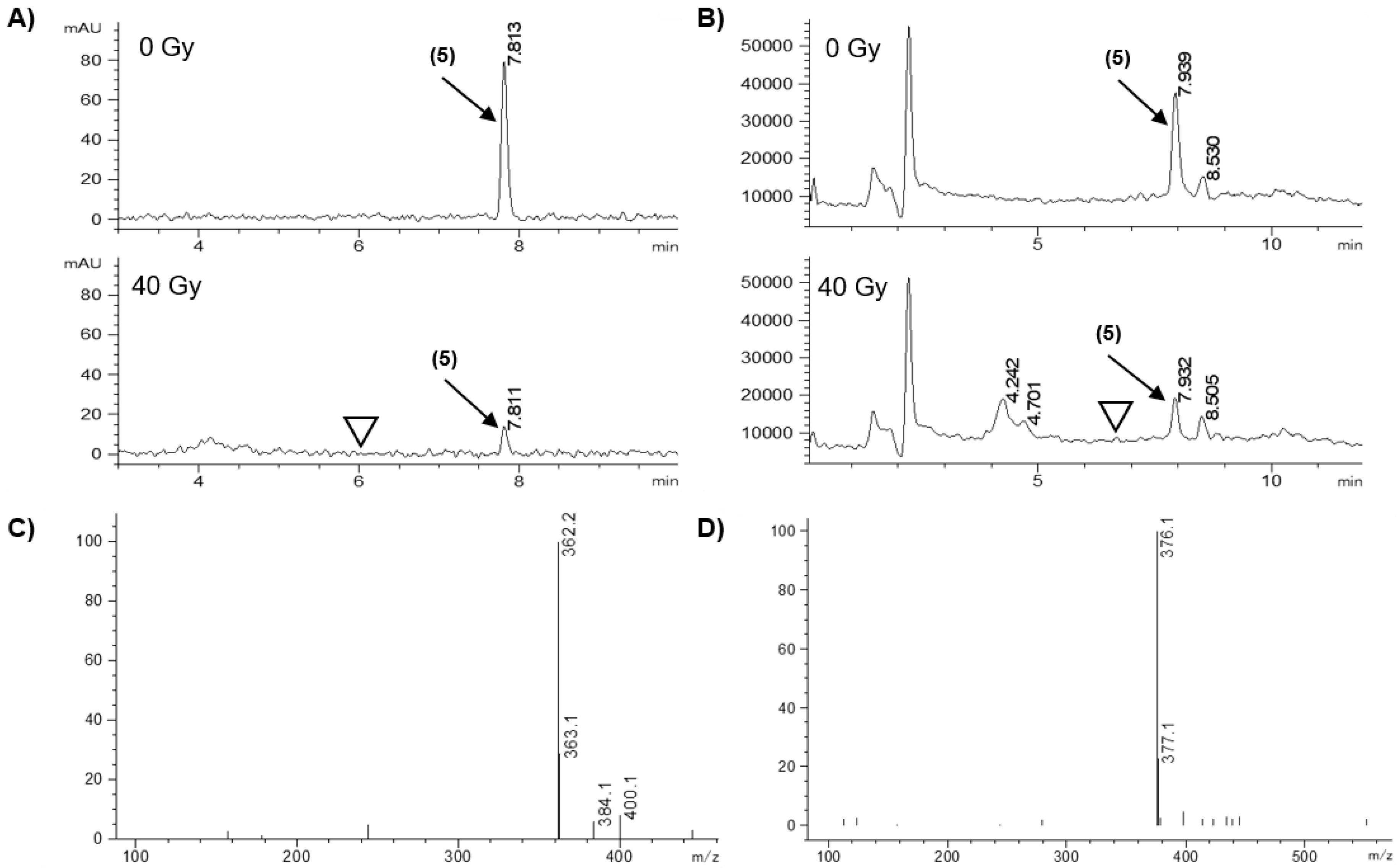
2.3. Radiolytic Reduction
3. Materials and Methods
3.1. Synthesis
3.1.1. General
3.1.2. Procedures
3.2. Assays
3.2.1. Radiolytic Reduction
3.2.2. Chemical Reduction
3.2.3. LC/MS Analysis
3.2.4. Biochemical PARP Inhibition Assay
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
- AcCl acetyl chloride;
- BnCl benzyl chloride;
- CDI 1,1′-carbonyldiimidazole;
- DCM dichloromethane;
- DIPEA diisopropylethylamine;
- DMF N,N-dimethylformamide;
- EtOAc ethyl acetate;
- Et2O diethyl ether;
- EtOH ethanol;
- h hour;
- MeOH methanol;
- rt room temperature;
- THF tetrahydrofuran;
- X4 petroleum ether boiling range 40–60 °C.
References
- De Lorenzo, S.B.; Patel, A.G.; Hurley, R.M.; Kaufmann, S.H. The Elephant and the Blind Men: Making Sense of PARP Inhibitors in Homologous Recombination Deficient Tumor Cells. Front. Oncol. 2013, 3, 228. [Google Scholar] [CrossRef] [PubMed]
- Durkacz, B.W.; Omidiji, O.; Gray, D.A.; Shall, S. (ADP-ribose)n participates in DNA excision repair. Nature 1980, 283, 593–596. [Google Scholar] [CrossRef]
- Sousa, F.G.; Matuo, R.; Soares, D.G.; Escargueil, A.E.; Henriques, J.A.P.; Larsen, A.K.; Saffi, J. PARPs and the DNA damage response. Carcinogenesis 2012, 33, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
- Krishnakumar, R.; Kraus, W.L. The PARP Side of the Nucleus: Molecular Actions, Physiological Outcomes, and Clinical Targets. Mol. Cell 2010, 39, 8–24. [Google Scholar] [CrossRef] [PubMed]
- Satoh, M.S.; Lindahl, T. Role of poly(ADP-ribose) formation in DNA repair. Nature 1992, 356, 356–358. [Google Scholar] [CrossRef]
- Helleday, T. The underlying mechanism for the PARP and BRCA synthetic lethality: Clearing up the misunderstandings. Mol. Oncol. 2011, 5, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, F.; Schreiber, V.; Niedergang, C.; Trucco, C.; Flatter, E.; Rubia, G.D.L.; Oliver, J.; Rolli, V.; Ménissier-de Murcia, J.; de Murcia, G. Involvement of poly(ADP-ribose) polymerase in base excision repair. Biochimie 1999, 81, 69–75. [Google Scholar] [CrossRef]
- Ström, C.E.; Johansson, F.; Uhlén, M.; Szigyarto, C.A.-K.; Erixon, K.; Helleday, T. Poly (ADP-ribose) polymerase (PARP) is not involved in base excision repair but PARP inhibition traps a single-strand intermediate. Nucleic Acids Res. 2011, 39, 3166–3175. [Google Scholar] [CrossRef]
- Murai, J.; Huang, S.N.; Das, B.B.; Renaud, A.; Zhang, Y.; Doroshow, J.H.; Ji, J.; Takeda, S.; Pommier, Y. Trapping of PARP1 and PARP2 by Clinical PARP Inhibitors. Cancer Res. 2012, 72, 5588–5599. [Google Scholar] [CrossRef]
- Murai, J.; Huang, S.-Y.N.; Renaud, A.; Zhang, Y.; Ji, J.; Takeda, S.; Morris, J.; Teicher, B.; Doroshow, J.H.; Pommier, Y. Stereospecific PARP Trapping by BMN 673 and Comparison with Olaparib and Rucaparib. Mol. Cancer Ther. 2014, 13, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Aoyagi-Scharber, M.; Wang, B. Trapping Poly(ADP-Ribose) Polymerase. J. Pharmacol. Exp. Ther. 2015, 353, 446–457. [Google Scholar] [CrossRef] [PubMed]
- Bryant, H.E.; Schultz, N.; Thomas, H.D.; Parker, K.M.; Flower, D.; Lopez, E.; Kyle, S.; Meuth, M.; Curtin, N.J.; Helleday, T. Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature 2005, 434, 913–917. [Google Scholar] [CrossRef] [PubMed]
- Farmer, H.; McCabe, N.; Lord, C.J.; Tutt, A.N.J.; Johnson, D.A.; Richardson, T.B.; Santarosa, M.; Dillon, K.J.; Hickson, I.; Knights, C.; et al. Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 2005, 434, 917–921. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.J.; Ashworth, A. PARP inhibitors: Synthetic lethality in the clinic. Science 2017, 355, 1152–1158. [Google Scholar] [CrossRef]
- Nirsimloo, R.; Gourley, C. The safety and efficacy of olaparib therapy in patients with relapsed ovarian cancer. Expert Rev. Anticancer Ther. 2016, 16, 597–603. [Google Scholar] [CrossRef]
- Curtin, N.J.; Szabo, C. Therapeutic applications of PARP inhibitors: Anticancer therapy and beyond. Mol. Aspects Med. 2013, 34, 1217–1256. [Google Scholar] [CrossRef]
- Ali, S.O.; Khan, F.A.; Galindo-Campos, M.A.; Yélamos, J. Understanding specific functions of PARP-2: New lessons for cancer therapy. Am. J. Cancer Res. 2016, 6, 1842–1863. [Google Scholar]
- Sallmyr, A.; Tomkinson, A.E. Repair of DNA double-strand breaks by mammalian alternative end-joining pathways. J. Biol. Chem. 2018, 293, 10536–10546. [Google Scholar] [CrossRef]
- Chalmers, A.J.; Lakshman, M.; Chan, N.; Bristow, R.G. Poly(ADP-Ribose) Polymerase Inhibition as a Model for Synthetic Lethality in Developing Radiation Oncology Targets. Semin. Radiat. Oncol. 2010, 20, 274–281. [Google Scholar] [CrossRef]
- Veuger, S.J.; Curtin, N.J.; Richardson, C.J.; Smith, G.C.M.; Durkacz, B.W. Radiosensitization and DNA Repair Inhibition by the Combined Use of Novel Inhibitors of DNA-dependent Protein Kinase and Poly(ADP-Ribose) Polymerase-1. Cancer Res. 2003, 63, 6008–6015. [Google Scholar]
- Dréan, A.; Lord, C.J.; Ashworth, A. PARP inhibitor combination therapy. Crit. Rev. Oncol. Hematol. 2016, 108, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Oza, A.M.; Cibula, D.; Benzaquen, A.O.; Poole, C.; Mathijssen, R.H.J.; Sonke, G.S.; Colombo, N.; Špaček, J.; Vuylsteke, P.; Hirte, H.; et al. Olaparib combined with chemotherapy for recurrent platinum-sensitive ovarian cancer: A randomised phase 2 trial. Lancet Oncol. 2015, 16, 87–97. [Google Scholar] [CrossRef]
- Rajan, A.; Carter, C.A.; Kelly, R.J.; Gutierrez, M.; Kummar, S.; Szabo, E.; Yancey, M.A.; Ji, J.; Mannargudi, B.; Woo, S.; et al. A Phase I Combination Study of Olaparib with Cisplatin and Gemcitabine in Adults with Solid Tumors. Clin. Cancer Res. 2012, 18, 2344–2351. [Google Scholar] [CrossRef] [PubMed]
- Kummar, S.; Chen, A.; Ji, J.; Zhang, Y.; Reid, J.M.; Ames, M.; Jia, L.; Weil, M.; Speranza, G.; Murgo, A.J.; et al. Phase I Study of PARP Inhibitor ABT-888 in Combination with Topotecan in Adults with Refractory Solid Tumors and Lymphomas. Cancer Res. 2011, 71, 5626–5634. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.F.; Barry, W.T.; Birrer, M.; Lee, J.-M.; Buckanovich, R.J.; Fleming, G.F.; Rimel, B.; Buss, M.K.; Nattam, S.; Hurteau, J.; et al. Combination cediranib and olaparib versus olaparib alone for women with recurrent platinum-sensitive ovarian cancer: A randomised phase 2 study. Lancet Oncol. 2014, 15, 1207–1214. [Google Scholar] [CrossRef]
- Plummer, R.; Jones, C.; Middleton, M.; Wilson, R.; Evans, J.; Olsen, A.; Curtin, N.; Boddy, A.; McHugh, P.; Newell, D.; et al. Phase I Study of the Poly(ADP-Ribose) Polymerase Inhibitor, AG014699, in Combination with Temozolomide in Patients with Advanced Solid Tumors. Clin. Cancer Res. 2008, 14, 7917–7923. [Google Scholar] [CrossRef]
- Plummer, R.; Stephens, P.; Aissat-Daudigny, L.; Cambois, A.; Moachon, G.; Brown, P.D.; Campone, M. Phase 1 dose-escalation study of the PARP inhibitor CEP-9722 as monotherapy or in combination with temozolomide in patients with solid tumors. Cancer Chemother. Pharmacol. 2014, 74, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Samol, J.; Ranson, M.; Scott, E.; Macpherson, E.; Carmichael, J.; Thomas, A.; Cassidy, J. Safety and tolerability of the poly(ADP-ribose) polymerase (PARP) inhibitor, olaparib (AZD2281) in combination with topotecan for the treatment of patients with advanced solid tumors: A phase I study. Investig. New Drugs 2012, 30, 1493–1500. [Google Scholar] [CrossRef]
- Wilson, W.R.; Hay, M.P. Targeting hypoxia in cancer therapy. Nat. Rev. Cancer 2011, 11, 393–410. [Google Scholar] [CrossRef]
- Vaupel, P.; Mayer, A. Hypoxia in cancer: Significance and impact on clinical outcome. Cancer Metastasis Rev. 2007, 26, 225–239. [Google Scholar] [CrossRef]
- Harris, A.L. Hypoxia—A key regulatory factor in tumour growth. Nat. Rev. Cancer 2002, 2, 38–47. [Google Scholar] [CrossRef]
- Phillips, R.M. Targeting the hypoxic fraction of tumours using hypoxia-activated prodrugs. Cancer Chemother. Pharmacol. 2016, 77, 441–457. [Google Scholar] [CrossRef]
- Brown, J.M.; Wilson, W.R. Exploiting tumour hypoxia in cancer treatment. Nat. Rev. Cancer 2004, 4, 437–447. [Google Scholar] [CrossRef]
- Horsman, M.R.; Vaupel, P. Pathophysiological Basis for the Formation of the Tumor Microenvironment. Front. Oncol. 2016, 6, 66. [Google Scholar] [CrossRef]
- O’Connor, L.J.; Cazares-Körner, C.; Saha, J.; Evans, C.N.G.; Stratford, M.R.L.; Hammond, E.M.; Conway, S.J. Design, synthesis and evaluation of molecularly targeted hypoxia-activated prodrugs. Nat. Protoc. 2016, 11, 781–794. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.R.; Dickson, B.D.; Jaiswal, J.K.; Pruijn, F.B.; Hunter, F.W.; Hay, M.P.; Hicks, K.O.; Wilson, W.R. Cellular pharmacology of evofosfamide (TH-302): A critical re-evaluation of its bystander effects. Biochem. Pharmacol. 2018, 156, 265–280. [Google Scholar] [CrossRef]
- Meng, F.; Evans, J.W.; Bhupathi, D.; Banica, M.; Lan, L.; Lorente, G.; Duan, J.-X.; Cai, X.; Mowday, A.M.; Guise, C.P.; et al. Molecular and Cellular Pharmacology of the Hypoxia-Activated Prodrug TH-302. Mol. Cancer Ther. 2011, 11, 740–751. [Google Scholar] [CrossRef] [PubMed]
- Patterson, A.V.; Ferry, D.M.; Edmunds, S.J.; Gu, Y.; Singleton, R.S.; Patel, K.; Pullen, S.M.; Hicks, K.O.; Syddall, S.P.; Atwell, G.J.; et al. Mechanism of Action and Preclinical Antitumor Activity of the Novel Hypoxia-Activated DNA Cross-Linking Agent PR-104. Clin. Cancer Res. 2007, 13, 3922–3932. [Google Scholar] [CrossRef] [PubMed]
- Menear, K.A.; Adcock, C.; Boulter, R.; Cockcroft, X.; Copsey, L.; Cranston, A.; Dillon, K.J.; Drzewiecki, J.; Garman, S.; Gomez, S.; et al. 4-[3-(4-Cyclopropanecarbonylpiperazine-1-carbonyl)-4-fluorobenzyl]-2H-phthalazin-1-one: A Novel Bioavailable Inhibitor of Poly(ADP-ribose) Polymerase-1. J. Med. Chem. 2008, 51, 6581–6591. [Google Scholar] [CrossRef] [PubMed]
- Ferraris, D.V. Evolution of Poly(ADP-ribose) Polymerase-1 (PARP-1) Inhibitors. From Concept to Clinic. J. Med. Chem. 2010, 53, 4561–4584. [Google Scholar] [CrossRef]
- Thorsell, A.-G.; Ekblad, T.; Karlberg, T.; Löw, M.; Pinto, A.F.; Trésaugues, L.; Moche, M.; Cohen, M.S.; Schüler, H. Structural Basis for Potency and Promiscuity in Poly(ADP-ribose) Polymerase (PARP) and Tankyrase Inhibitors. J. Med. Chem. 2017, 60, 1262–1271. [Google Scholar] [CrossRef] [PubMed]
- Berry, J.M.; Watson, C.Y.; Whish, W.J.D.; Threadgill, M.D. 5-Nitrofuran-2-ylmethyl group as a potential bioreductively activatedpro-drug system. J. Chem. Soc. Perkin Trans. 1 1997, 1147–1156. [Google Scholar] [CrossRef]
- Parveen, I.; Naughton, D.P.; Whish, W.J.D.; Threadgill, M.D. 2-Nitroimidazol-5-ylmethyl as a potential bioreductively activated prodrug system: Reductively triggered release of the parp inhibitor 5-bromoisoquinolinone. Bioorg. Med. Chem. Lett. 1999, 9, 2031–2036. [Google Scholar] [CrossRef]
- Lindquist, K.E.; Cran, J.D.; Kordic, K.; Chua, P.C.; Winters, G.C.; Tan, J.S.; Lozada, J.; Kyle, A.H.; Evans, J.W.; Minchinton, A.I. Selective radiosensitization of hypoxic cells using BCCA621C: A novel hypoxia activated prodrug targeting DNA-dependent protein kinase. Tumor Microenviron. Ther. 2013, 1, 46–55. [Google Scholar] [CrossRef]
- Thomson, P.; Naylor, M.A.; Everett, S.A.; Stratford, M.R.L.; Lewis, G.; Hill, S.; Patel, K.B.; Wardman, P.; Davis, P.D. Synthesis and biological properties of bioreductively targeted nitrothienyl prodrugs of combretastatin A-4. Mol. Cancer Ther. 2006, 5, 2886–2894. [Google Scholar] [CrossRef]
- Lin, C.-F.; Yang, J.-S.; Chang, C.-Y.; Kuo, S.-C.; Lee, M.-R.; Huang, L.-J. Synthesis and anticancer activity of benzyloxybenzaldehyde derivatives against HL-60 cells. Bioorg. Med. Chem. 2005, 13, 1537–1544. [Google Scholar] [CrossRef] [PubMed]
- Griffin, R.J.; Srinivasan, S.; Bowman, K.; Calvert, A.H.; Curtin, N.J.; Newell, D.R.; Pemberton, L.C.; Golding, B.T. Resistance-Modifying Agents. 5. Synthesis and Biological Properties of Quinazolinone Inhibitors of the DNA Repair Enzyme Poly(ADP-ribose) Polymerase (PARP). J. Med. Chem. 1998, 41, 5247–5256. [Google Scholar] [CrossRef]
- Matteucci, M.; Duan, J.-X.; Jiao, H.; Kaizerman, J.; Ammons, S. Phosphoramidate Alkylator Prodrugs. U.S. Patent US 8,003,625, 23 August 2011. [Google Scholar]
- Winn, B.A.; Shi, Z.; Carlson, G.J.; Wang, Y.; Nguyen, B.L.; Kelly, E.M.; Ross, R.D.; Hamel, E.; Chaplin, D.J.; Trawick, M.L.; et al. Bioreductively activatable prodrug conjugates of phenstatin designed to target tumor hypoxia. Bioorg. Med. Chem. Lett. 2017, 27, 636–641. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, S.; Naughton, D.P.; Parveen, I.; Threadgill, M.D. N- and O-Alkylation of isoquinolin-1-ones in the Mitsunobu reaction: Development of potential drug delivery systems. J. Chem. Soc. Perkin Trans. 1 2002, 3, 335–340. [Google Scholar] [CrossRef][Green Version]
- Buxton, G.V. Radiation Chemistry of the Liquid State; VCH Publishers Inc.: New York, NY, USA, 1987; ISBN 0-89573-127-4. [Google Scholar]
- Mulazzani, Q.G.; D’Angelantonio, M.; Venturi, M.; Hoffman, M.Z.; Rodgers, M.A.J. Interaction of formate and oxalate ions with radiation-generated radicals in aqueous solution. Methylviologen as a mechanistic probe. J. Phys. Chem. 1986, 90, 5347–5352. [Google Scholar] [CrossRef]
- Hay, M.P.; Wilson, W.R.; Denny, W.A. Design, Synthesis and Evaluation of Imidazolylmethyl Carbamate Prodrugs of Alkylating Agents. Tetrahedron 2000, 56, 645–657. [Google Scholar] [CrossRef]
- Sykes, B.M.; Hay, M.P.; Bohinc-Herceg, D.; Helsby, N.A.; O’Connor, C.J.; Denny, W.A. Leaving group effects in reductively triggered fragmentation of 4-nitrobenzyl carbamates. J. Chem. Soc. Perkin Trans. 1 2000, 10, 1601–1608. [Google Scholar] [CrossRef]
- Everett, S.A.; Naylor, M.A.; Patel, K.B.; Stratford, M.R.L.; Wardman, P. Bioreductively-activated prodrugs for targeting hypoxic tissues: Elimination of aspirin from 2-nitroimidazole derivatives. Bioorg. Med. Chem. Lett. 1999, 9, 1267–1272. [Google Scholar] [CrossRef]
- Duan, J.-X.; Jiao, H.; Kaizerman, J.; Stanton, T.; Evans, J.W.; Lan, L.; Lorente, G.; Banica, M.; Jung, D.; Wang, J.; et al. Potent and Highly Selective Hypoxia-Activated Achiral Phosphoramidate Mustards as Anticancer Drugs. J. Med. Chem. 2008, 51, 2412–2420. [Google Scholar] [CrossRef] [PubMed]
- Munín, J.; Santana, L.; Uriarte, E.; Borges, F.; Quezada, E. A comparative synthesis of 6-benzyl-2,3-dihydroimidazo[2,1-a]phthalazine and 2H-7-benzyl-3,4-dihydropyrimido[2,1-a]phthalazine. Tetrahedron Lett. 2015, 56, 828–830. [Google Scholar] [CrossRef]
- Bouider, N.; Fhayli, W.; Ghandour, Z.; Boyer, M.; Harrouche, K.; Florence, X.; Pirotte, B.; Lebrun, P.; Faury, G.; Khelili, S. Design and synthesis of new potassium channel activators derived from the ring opening of diazoxide: Study of their vasodilatory effect, stimulation of elastin synthesis and inhibitory effect on insulin release. Bioorg. Med. Chem. 2015, 23, 1735–1746. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Chen, Q.; Lu, L.; Ma, Y.; Auyoung, G.H.L.; Hua, R. Base-promoted nucleophilic fluoroarenes substitution of CF bonds. Tetrahedron 2018, 74, 303–307. [Google Scholar] [CrossRef]
- Ghosh, S.K.; Nagarajan, R. Total synthesis of penipanoid C, 2-(4-hydroxybenzyl)quinazolin-4(3H)-one and NU1025. Tetrahedron Lett. 2016, 57, 4277–4279. [Google Scholar] [CrossRef]
- Abe, Y.; Kayakiri, H.; Satoh, S.; Inoue, T.; Sawada, Y.; Inamura, N.; Asano, M.; Aramori, I.; Hatori, C.; Sawai, H.; et al. A Novel Class of Orally Active Non-Peptide Bradykinin B2 Receptor Antagonists. 3. Discovering Bioisosteres of the Imidazo[1,2-a]pyridine Moiety. J. Med. Chem. 1998, 41, 4062–4079. [Google Scholar] [CrossRef]
- Cavalleri, B.; Ballotta, R.; Arioli, V.; Lancini, G. New 5-substituted 1-alkyl-2-nitroimidazoles. J. Med. Chem. 1973, 16, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Dalton, N.; Gordon, C.P.; Boyle, T.P.; Vandegraaf, N.; Deadman, J.; Rhodes, D.I.; Coates, J.A.; Pyne, S.G.; Keller, P.A.; Bremner, J.B. The discovery of allyltyrosine based tripeptides as selective inhibitors of the HIV-1 integrase strand-transfer reaction. Org. Biomol. Chem. 2016, 14, 6010–6023. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cheng, J.M.H.; Liu, L.; Pellicci, D.G.; Reddiex, S.J.J.; Cotton, R.N.; Cheng, T.-Y.; Young, D.C.; Van Rhijn, I.; Moody, D.B.; Rossjohn, J.; et al. Total Synthesis of Mycobacterium tuberculosis Dideoxymycobactin-838 and Stereoisomers: Diverse CD1a-Restricted T Cells Display a Common Hierarchy of Lipopeptide Recognition. Chem. Eur. J. 2017, 23, 1694–1701. [Google Scholar] [CrossRef] [PubMed]
- Fricke, J.; Hart, E.J. Chemical Dosimetry. In Radiation Dosimetry vol 2; Attix, F.H., Roesch, W.C., Eds.; Academic Press: New York, NY, USA, 1966; pp. 167–239. [Google Scholar]
Sample Availability: Samples of the compounds 4–13 are available from the authors. |





| PARPi | IC50 (µM) a | Model HAP | IC50 (µM) a | Ratio b |
|---|---|---|---|---|
| 4 | 0.173 ± 0.068 | 5 | 27.4 ± 0.5 | 160 ± 60 |
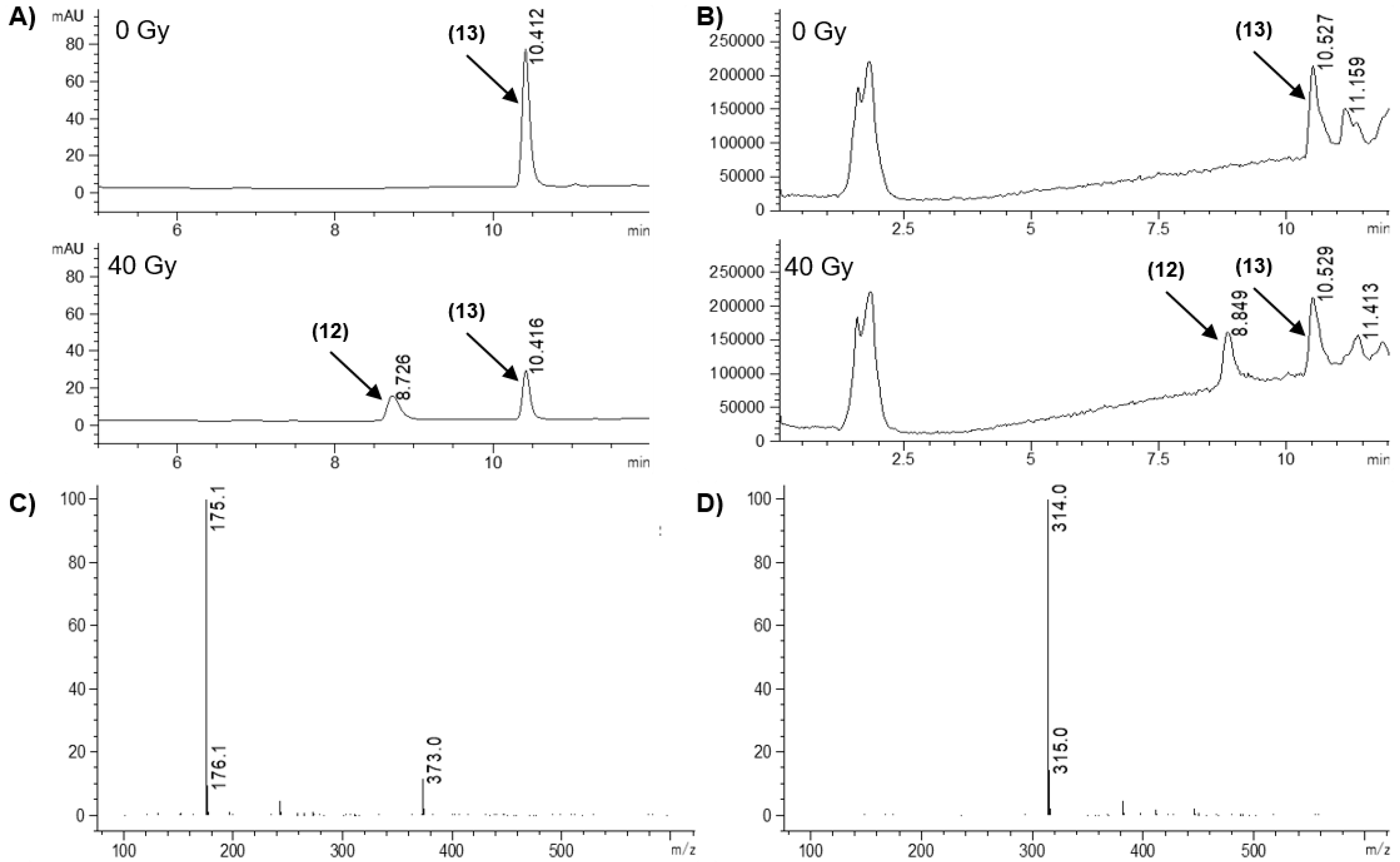
| 12 | 0.038 ± 0.014 | 13 | 0.25 ± 0.07 | 6.8 ± 3.1 |
| Model HAP | % Loss Prodrug Candidate a | Intermediate Species Detected b | % PARPi Formed c |
|---|---|---|---|
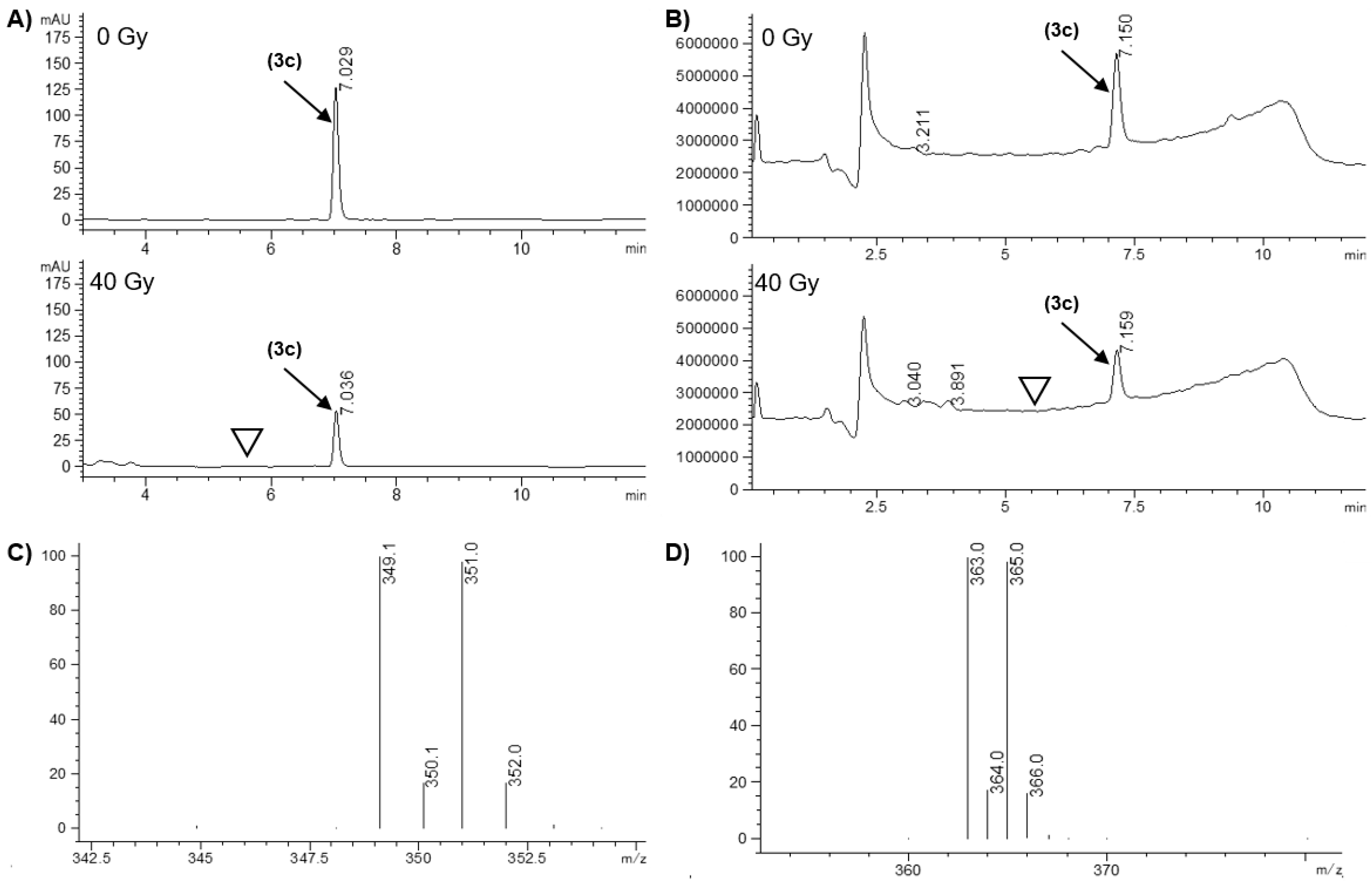
| 3c | 65.1 ± 7.4 | H (349.1/351.0 m/z) | n.d. d |
| 5 | 85.3 ± 2.1 | H (362.2 m/z) | n.d. |
| 7 | 39.2 | H (353.2 m/z) | n.d. |
| 8 | 73.9 | H (367.2 m/z) | n.d. |
| 10 | 56.3 | H (272.1 m/z) | n.d. |
| 11 | 52.3 ± 3.6 | H (271.1 m/z) | n.d. |
| 13 | 55.0 ± 9.1 | - | 39.9 ± 7.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dickson, B.D.; Wong, W.W.; Wilson, W.R.; Hay, M.P. Studies Towards Hypoxia-Activated Prodrugs of PARP Inhibitors. Molecules 2019, 24, 1559. https://doi.org/10.3390/molecules24081559
Dickson BD, Wong WW, Wilson WR, Hay MP. Studies Towards Hypoxia-Activated Prodrugs of PARP Inhibitors. Molecules. 2019; 24(8):1559. https://doi.org/10.3390/molecules24081559
Chicago/Turabian StyleDickson, Benjamin D., Way Wua Wong, William R. Wilson, and Michael P. Hay. 2019. "Studies Towards Hypoxia-Activated Prodrugs of PARP Inhibitors" Molecules 24, no. 8: 1559. https://doi.org/10.3390/molecules24081559
APA StyleDickson, B. D., Wong, W. W., Wilson, W. R., & Hay, M. P. (2019). Studies Towards Hypoxia-Activated Prodrugs of PARP Inhibitors. Molecules, 24(8), 1559. https://doi.org/10.3390/molecules24081559