A Rapid UPLC-MS Method for Quantification of Gomisin D in Rat Plasma and Its Application to a Pharmacokinetic and Bioavailability Study
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Instruments and Conditions
2.3. Preparation of Standard and Quality Control Samples
2.4. Animal Experiments
2.5. Sample Preparation
2.6. Method Validation
3. Results and Discussion
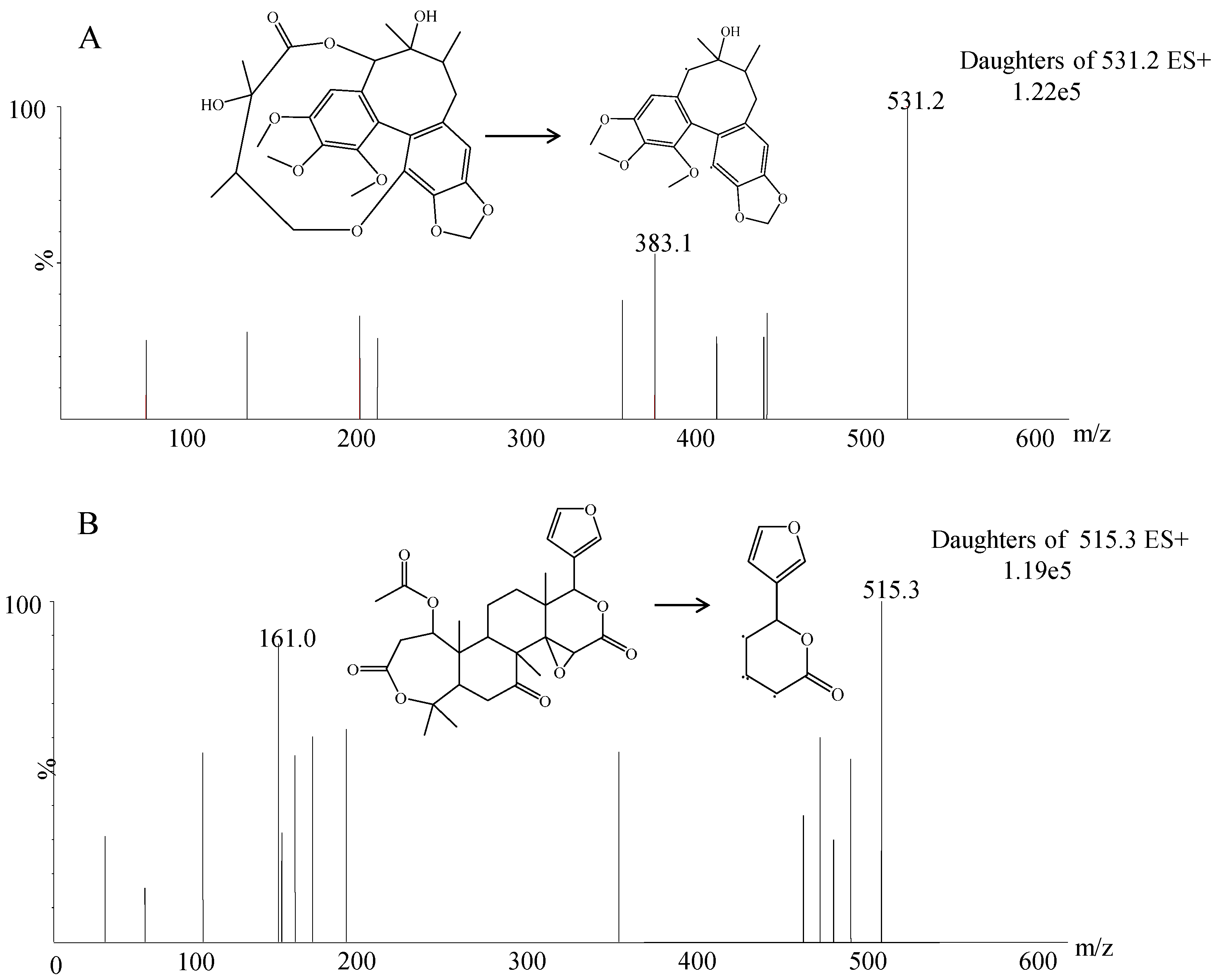
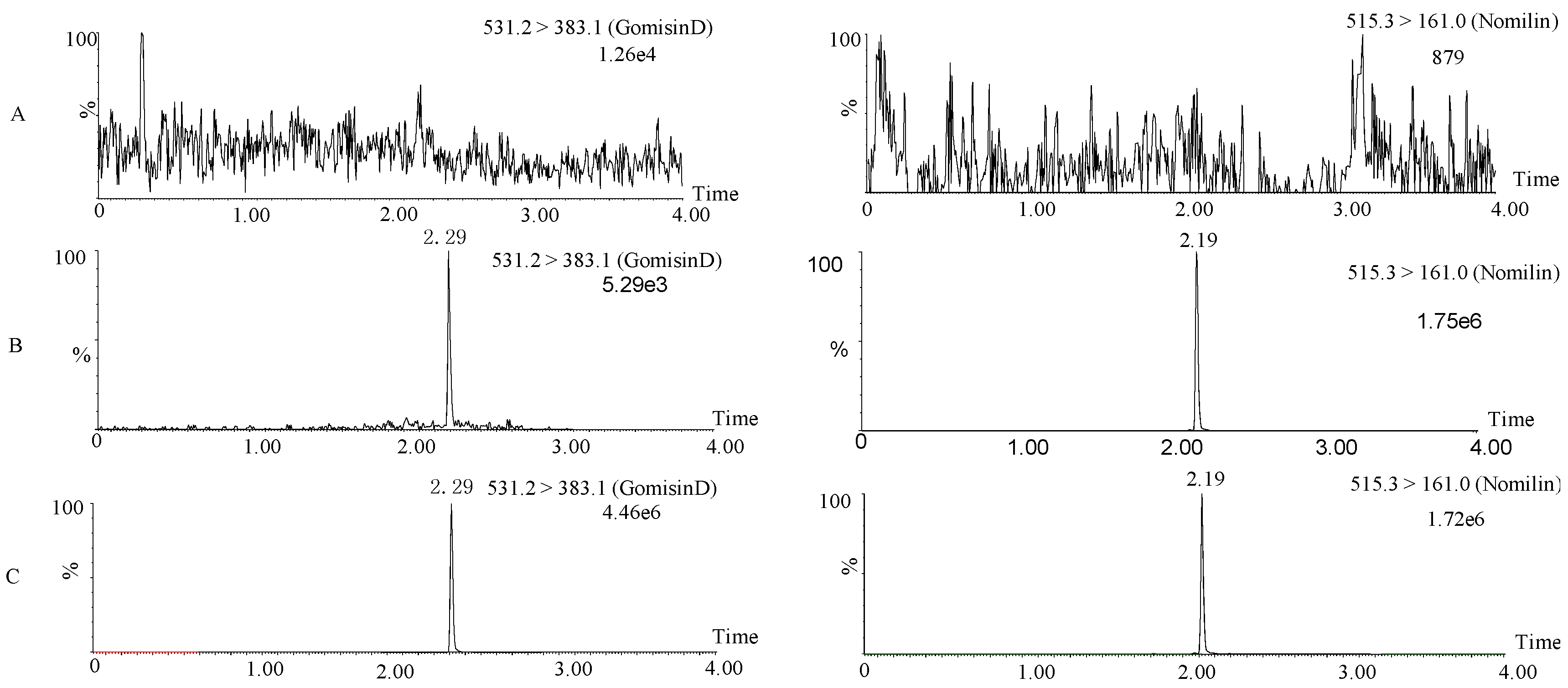
3.1. Method Development
3.2. Method Validation
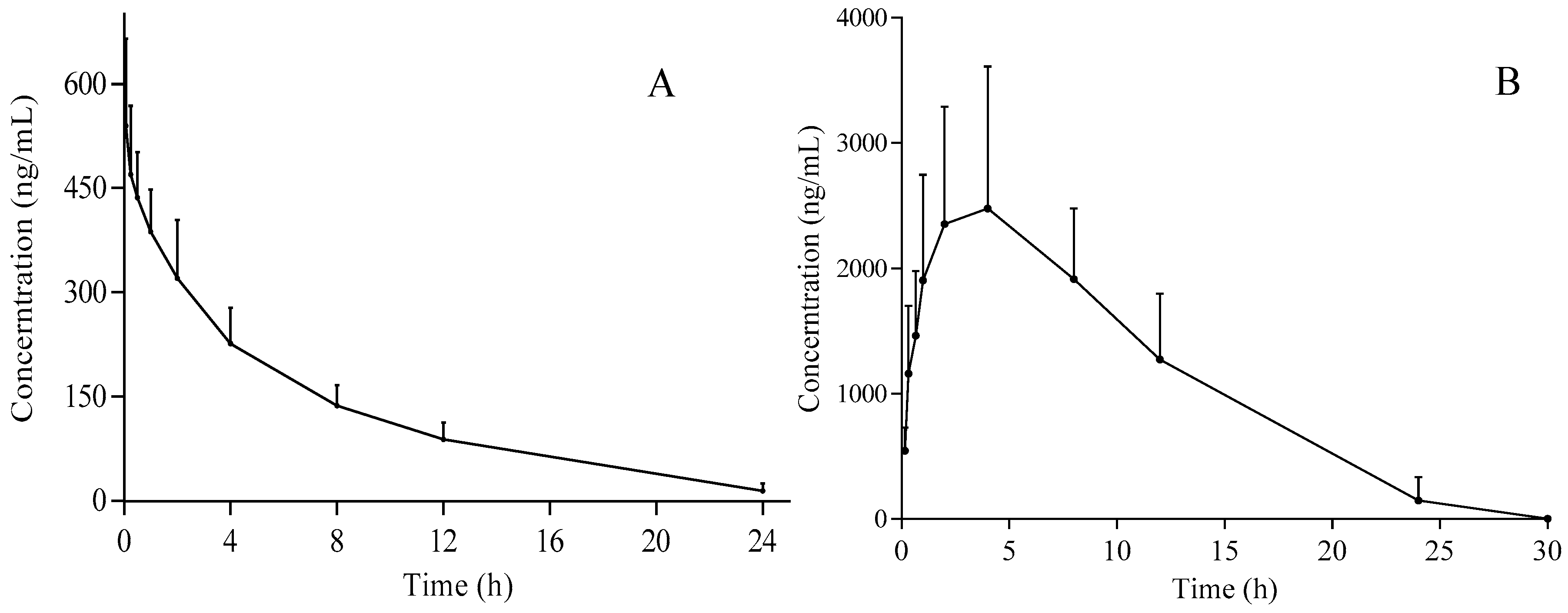
3.3. Pharmacokinetic Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jang, M.K.; Nam, J.S.; Kim, J.H.; Yun, Y.R.; Han, C.W.; Kim, B.J.; Jeong, H.S.; Ha, K.T.; Jung, M.H. Schisandra chinensis extract ameliorates nonalcoholic fatty liver via inhibition of endoplasmic reticulum stress. J. Ethnopharmacol. 2016, 185, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Lee, H.J.; Kim, C.Y.; Han, S.Y.; Chin, Y.W.; Choi, Y.H. Simultaneous determination of nine lignans from schisandra chinensis extract using ultra-performance liquid chromatography with tandem mass spectrometry in rat plasma, urine, and gastrointestinal tract samples: Application to the pharmacokinetic study of schisandra chinensis. J. Sep. Sci. 2014, 37, 2851–2863. [Google Scholar] [PubMed]
- Hong, M.; Zhang, Y.; Li, S.; Tan, H.Y.; Wang, N.; Mu, S.; Hao, X.; Feng, Y. A network pharmacology-based study on the hepatoprotective effect of fructus schisandrae. Molecules 2017, 22, 1617. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Guo, Y.; Li, L.Y.; Hu, H.; Qu, X.L.; Sun, X.Z.; Liu, S.H.; Wang, H. A herbal composition of semen hoveniae, radix puerariae, and fructus schisandrae shows potent protective effects on acute alcoholic intoxication in rodent models. Evid.-Based Complement. Altern. Med. 2012, 2012, 638197. [Google Scholar] [CrossRef]
- Pan, S.Y.; Yu, Q.; Zhang, Y.; Wang, X.Y.; Sun, N.; Yu, Z.L.; Ko, K.M. Dietary fructus schisandrae extracts and fenofibrate regulate the serum/hepatic lipid-profile in normal and hypercholesterolemic mice, with attention to hepatotoxicity. Lipids Health Dis. 2012, 11, 120. [Google Scholar] [CrossRef]
- Pan, S.Y.; Yu, Z.L.; Dong, H.; Xiang, C.J.; Fong, W.F.; Ko, K.M. Ethanol extract of fructus schisandrae decreases hepatic triglyceride level in mice fed with a high fat/cholesterol diet, with attention to acute toxicity. Evid.-Based Complement. Altern. Med. 2011, 2011, 729412. [Google Scholar] [CrossRef] [PubMed]
- Kwon, D.Y.; Kim, D.S.; Yang, H.J.; Park, S. The lignan-rich fractions of fructus schisandrae improve insulin sensitivity via the ppar-gamma pathways in in vitro and in vivo studies. J. Ethnopharmacol. 2011, 135, 455–462. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, L.L.; Zheng, Y.N. Dibenzocyclooctadiene lignans from fructus schisandrae chinensis improve glucose uptake in vitro. Nat. Prod. Commun. 2010, 5, 231–234. [Google Scholar] [CrossRef]
- Shao, B.; Tang, J.; Ji, H.; Liu, H.; Liu, Y.; Zhu, D.; Wu, L. Enhanced oral bioavailability of wurenchun (fructus schisandrae chinensis extracts) by self-emulsifying drug delivery systems. Drug Dev. Ind. Pharm. 2010, 36, 1356–1363. [Google Scholar] [CrossRef]
- Tian, D.D.; Wang, W.; Wang, H.N.; Sze, S.C.; Zhang, Z.J. Pharmacokinetic evaluation of clozapine in concomitant use of radix rehmanniae, fructus schisandrae, radix bupleuri, or fructus gardeniae in rats. Molecules 2016, 21, 696. [Google Scholar] [CrossRef]
- Gao, J.R.; Ji, W.B.; Jiang, H.; Chen, J.F. Effects of extracts from ziziphi spinosae semen and schisandrae chinensis fructus on amino acid neurotransmitter in rats with insomnia induced by pcpa. J. Chin. Med. Mater. 2013, 36, 1635–1639. [Google Scholar]
- Chen, S.; Jia, Z.; Dong, L.; Geng, P.; Liu, Z.; Yang, S.; Wen, C.; Liu, F. Pharmacokinetic and bioavailability study of angeloylgomisin h in rat plasma by uplc-ms/ms. Int. Clin. Exp. Med. 2015, 8, 17968–17976. [Google Scholar]
- Smejkal, K.; Slapetova, T.; Krmencik, P.; Babula, P.; Dall’Acqua, S.; Innocenti, G.; Vanco, J.; Casarin, E.; Carrara, M.; Kalvarova, K.; et al. Evaluation of cytotoxic activity of schisandra chinensis lignans. Planta Med. 2010, 76, 1672–1677. [Google Scholar] [CrossRef]
- Song, J.H.; Cui, L.; An, L.B.; Li, W.T.; Fang, Z.Z.; Zhang, Y.Y.; Dong, P.P.; Wu, X.; Wang, L.X.; Gonzalez, F.J.; et al. Inhibition of udp-glucuronosyltransferases (ugts) activity by constituents of schisandra chinensis. Phytother. Res. 2015, 29, 1658–1664. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, X.J.; Jiang, H.J.; Xu, C.; Tong, S.Q.; Yan, J.Z. Screening and identification of alpha-glucosidase inhibitors from shenqi jiangtang granule by ultrafiltration liquid chromatography and mass spectrometry. J. Sep. Sci. 2018, 41, 797–805. [Google Scholar] [CrossRef]
- Mocan, A.; Schafberg, M.; Crisan, G.; Rohn, S. Determination of lignans and phenolic components of schisandra chinensis (turcz.) baill. Using hplc-esi-tof-ms and hplc-online teac: Contribution of individual components to overall antioxidant activity and comparison with traditional antioxidant assays. J. Funct. Foods 2016, 24, 579–594. [Google Scholar] [CrossRef]
- Hung, T.M.; Na, M.; Min, B.S.; Ngoc, T.M.; Lee, I.; Zhang, X.; Bae, K. Acetylcholinesterase inhibitory effect of lignans isolated from schizandra chinensis. Arch. Pharmacal Res. 2007, 30, 685–690. [Google Scholar] [CrossRef]
- Zhang, A.H.; Yu, J.B.; Sun, H.; Kong, L.; Wang, X.Q.; Zhang, Q.Y.; Wang, X.J. Identifying quality-markers from shengmai san protects against transgenic mouse model of alzheimer’s disease using chinmedomics approach. Phytomedicine 2018, 45, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Xiao-tong, W.; Fan, H.; Xiao-ling, S. Pharmacokinetics study of seven lignans in alzheimer’s rats. Int. J. Pharmacol. 2018, 14, 68–75. [Google Scholar]
- Shi, P.; He, Q.; Zhang, Y.; Qu, H.; Cheng, Y. Characterisation and identification of isomeric dibenzocyclooctadiene lignans from schisandra chinensis by high-performance liquid chromatography combined with electrospray ionisation tandem mass spectrometry. Phytochem. Anal. 2009, 20, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.D.; Wang, Q.; Wang, Y.; Wang, X.J.; Pu, J.X.; Gu, Y.; Wang, R. Application of ultrahigh-performance liquid chromatography coupled with mass spectrometry for analysis of lignans and quality control of fructus schisandrae chinensis. J. Sep. Sci. 2012, 35, 2203–2209. [Google Scholar] [CrossRef]
- Liu, K.; Song, Y.; Liu, Y.; Peng, M.; Li, H.; Li, X.; Feng, B.; Xu, P.; Su, D. An integrated strategy using uplc-qtof-ms(e) and uplc-qtof-mrm (enhanced target) for pharmacokinetics study of wine processed schisandra chinensis fructus in rats. J. Pharm. Biomed. Anal. 2017, 139, 165–178. [Google Scholar] [CrossRef]
- Food and Drug Administration. Bioanalytical Method Validation Guidance for Industry; Food and Drug Administration: Silver Spring, MD, USA, 2018.
- Wang, L.Z.; Phan, D.D.K.; Syn, N.; Xiang, X.Q.; Song, H.Y.; Thuya, W.L.; Yang, S.L.; Wong, A.L.A.; Kumar, A.P.; Yong, W.P.; et al. A sensitive liquid chromatography-tandem mass spectrometry method for the determination of nimbolide in mouse serum: Application to a preclinical pharmacokinetics study. Pharmaceutics 2018, 10, 123. [Google Scholar] [CrossRef]
- Sun, L.L.; Ding, F.F.; You, G.J.; Liu, H.; Wang, M.; Ren, X.L.; Deng, Y.R. Development and validation of an uplc-ms/ms method for pharmacokinetic comparison of five alkaloids from jinqi jiangtang tablets and its monarch drug coptidis rhizoma. Pharmaceutics 2018, 10, 4. [Google Scholar] [CrossRef]
- Balla, A.; Cho, K.H.; Kim, Y.C.; Maeng, H.J. Simultaneous determination of procainamide and n-acetylprocainamide in rat plasma by ultra-high-pressure liquid chromatography coupled with a diode array detector and its application to a pharmacokinetic study in rats. Pharmaceutics 2018, 10, 41. [Google Scholar] [CrossRef]
- Kim, S.J.; Shin, H.; Cheon, S.M.; Ko, S.M.; Ham, S.H.; Kwon, Y.D.; Lee, Y.B.; Cho, H.Y. A sensitive uhplc-ms/ms method for the simultaneous quantification of three lignans in human plasma and its application to a pharmacokinetic study. J. Sep. Sci. 2017, 40, 3430–3439. [Google Scholar] [CrossRef]
- Wei, B.; Li, Q.; Su, D.; Fan, R.; Zhao, L.; Geng, L.; He, B.; Chen, X.; Jia, Y.; Bi, K. Development of a uflc-ms/ms method for simultaneous determination of six lignans of schisandra chinensis (turcz.) baill. In rat plasma and its application to a comparative pharmacokinetic study in normal and insomnic rats. J. Pharm. Biomed. Anal. 2013, 77, 120–127. [Google Scholar] [CrossRef]
- Ward, K.W.; Hardy, L.B.; Kehler, J.R.; Azzarano, L.M.; Smith, B.R. Apparent absolute oral bioavailability in excess of 100% for a vitronectin receptor antagonist (sb-265123) in rat. II. Studies implicating transporter-mediated intestinal secretion. Xenobiotica 2004, 34, 367–377. [Google Scholar] [CrossRef]
- Ward, K.W.; Azzarano, L.M.; Evans, C.A.; Smith, B.R. Apparent absolute oral bioavailability in excess of 100% for a vitronectin receptor antagonist (sb-265123) in rat. I. Investigation of potential experimental and mechanistic explanations. Xenobiotica 2004, 34, 353–366. [Google Scholar] [CrossRef]
- Darbar, D.; Dell’Orto, S.; Wilkinson, G.R.; Roden, D.M. Loss of quinidine gluconate injection in a polyvinyl chloride infusion system. Am. J. Health-Syst. Pharm. 1996, 53, 655–658. [Google Scholar] [CrossRef]
- Curry, S.H.; Lorenz, A.; Chu, P.I.; Limacher, M.; Stacpoole, P.W. Disposition and pharmacodynamics of dichloroacetate (dca) and oxalate following oral dca doses. Biopharm. Drug Dispos. 1991, 12, 375–390. [Google Scholar] [CrossRef]
- Ludden, T.M. Nonlinear pharmacokinetics: Clinical implications. Clin. Pharmacokinet. 1991, 20, 429–446. [Google Scholar] [CrossRef]
- Ward, K.W.; Proksch, J.W.; Azzarano, L.M.; Salyers, K.L.; McSurdy-Freed, J.E.; Molnar, T.M.; Levy, M.A.; Smith, B.R. Sb-239063, a potent and selective inhibitor of p38 map kinase: Preclinical pharmacokinetics and species-specific reversible isomerization. Pharm. Res. 2001, 18, 1336–1344. [Google Scholar] [CrossRef]
- Hui, Z.; Yuanyuan, W.; Chenye, H.; Xiaojing, Z.; Jizhong, Y. Interaction between gomizine d and α-glucosidase. China J. Chin. Mater. Med. 2017, 42, 4631–4635. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
| Nominal Concentration (ng/mL) | Extraction Recovery (%, Mean ± SD) | Matrix Effect (%, Mean ± SD) |
|---|---|---|
| Gomisin D | ||
| 2 | 79.2 ± 6.4 | 80.9 ± 4.6 |
| 50 | 86.3 ± 5.2 | 82.9 ± 8.3 |
| 3200 | 85.1 ± 7.6 | 86.7 ± 9.3 |
| Nomilin | ||
| 400 | 87.8 ± 2.3 | 84.0 ± 3.0 |
| Added (ng/mL) | Measured (ng/mL) | Accuracy (%) | Precision (RSD, %) | |
|---|---|---|---|---|
| Interday | 1 | 0.8 ± 0.1 | 85.8 | 11.9 |
| (n = 6) | 2 | 1.7 ± 0.1 | 86.4 | 8.04 |
| 50 | 55.4 ± 1.8 | 89.2 | 3.3 | |
| 3200 | 3217.3 ± 60.6 | 98.4 | 1.9 | |
| Intraday | 1 | 0.8 ± 0.1 | 85.4 | 12.9 |
| (n = 6) | 2 | 1.8 ± 0.1 | 88.7 | 6.1 |
| 50 | 54.6 ± 2.6 | 91.7 | 4.8 | |
| 3200 | 3106.7 ± 104.4 | 96.6 | 3.3 |
| Storage Condition | Added (ng/mL) | Measured (ng/mL) | RSD (%) |
|---|---|---|---|
| Room temperature (for 12 h) | 2 | 1.4 ± 0.1 | 9.1 |
| 50 | 37.8 ± 0.6 | 1.5 | |
| 3200 | 2905.7 ± 63.4 | 2.2 | |
| Autosampler (at 4 °C for 24 h) | 2 | 1.2 ± 0.1 | 9.8 |
| 50 | 48.2 ± 2.9 | 5.9 | |
| 3200 | 2928.3 ± 64.3 | 2.2 | |
| Freeze-thaw (for 24 h) | 2 | 1.4 ± 0.2 | 10.2 |
| 50 | 38.4 ± 2.0 | 5.1 | |
| 3200 | 2952.4 ± 105.2 | 3.6 | |
| Stored at −20 °C (for 15 days) | 2 | 1.3 ± 0.2 | 12.2 |
| 50 | 40.8 ± 3.1 | 7.7 | |
| 3200 | 2928.3 ± 116.9 | 3.9 |
| Parameters | i.v. (5 mg/kg) | i.g. (50 mg/kg) |
|---|---|---|
| AUC(0-t) (µg/L·h) | 3136.2 ± 548.7 | 32,795.6 ± 11,104.6 |
| AUC(0-∞) (µg/L·h) | 3261.1 ± 600.0 | 35,091.7 ± 8092.8 |
| MRT(0-t) (h) | 6.0 ± 1.0 | 7.8 ± 1.2 |
| MRT(0-∞) (h) | 6.9 ± 1.3 | 10.6 ± 6.6 |
| t1/2 (h) | 5.0 ± 1.1 | 5.6 ± 2.0 |
| Tmax (h) | 0.083 ± 0.0 | 3.0 ± 1.2 |
| CL (L/h/kg) | 1.6 ± 0.3 | 1.5 ± 0.3 |
| V (L/kg) | 11.2 ± 2.6 | 13.3 ± 6.2 |
| Cmax (µg/L) | 539.5 ± 125.5 | 2575.2 ± 1048.2 |
| F (%) | 107.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, X.; Feng, F.; Jiang, X.; Qiu, J.; Cai, X.; Xiang, Z. A Rapid UPLC-MS Method for Quantification of Gomisin D in Rat Plasma and Its Application to a Pharmacokinetic and Bioavailability Study. Molecules 2019, 24, 1403. https://doi.org/10.3390/molecules24071403
Zheng X, Feng F, Jiang X, Qiu J, Cai X, Xiang Z. A Rapid UPLC-MS Method for Quantification of Gomisin D in Rat Plasma and Its Application to a Pharmacokinetic and Bioavailability Study. Molecules. 2019; 24(7):1403. https://doi.org/10.3390/molecules24071403
Chicago/Turabian StyleZheng, Xiaoyong, Feng Feng, Xiunan Jiang, Jieying Qiu, Xiaojun Cai, and Zheng Xiang. 2019. "A Rapid UPLC-MS Method for Quantification of Gomisin D in Rat Plasma and Its Application to a Pharmacokinetic and Bioavailability Study" Molecules 24, no. 7: 1403. https://doi.org/10.3390/molecules24071403
APA StyleZheng, X., Feng, F., Jiang, X., Qiu, J., Cai, X., & Xiang, Z. (2019). A Rapid UPLC-MS Method for Quantification of Gomisin D in Rat Plasma and Its Application to a Pharmacokinetic and Bioavailability Study. Molecules, 24(7), 1403. https://doi.org/10.3390/molecules24071403






