Extracts from Fermented Black Garlic Exhibit a Hepatoprotective Effect on Acute Hepatic Injury
Abstract
:1. Introduction
2. Results
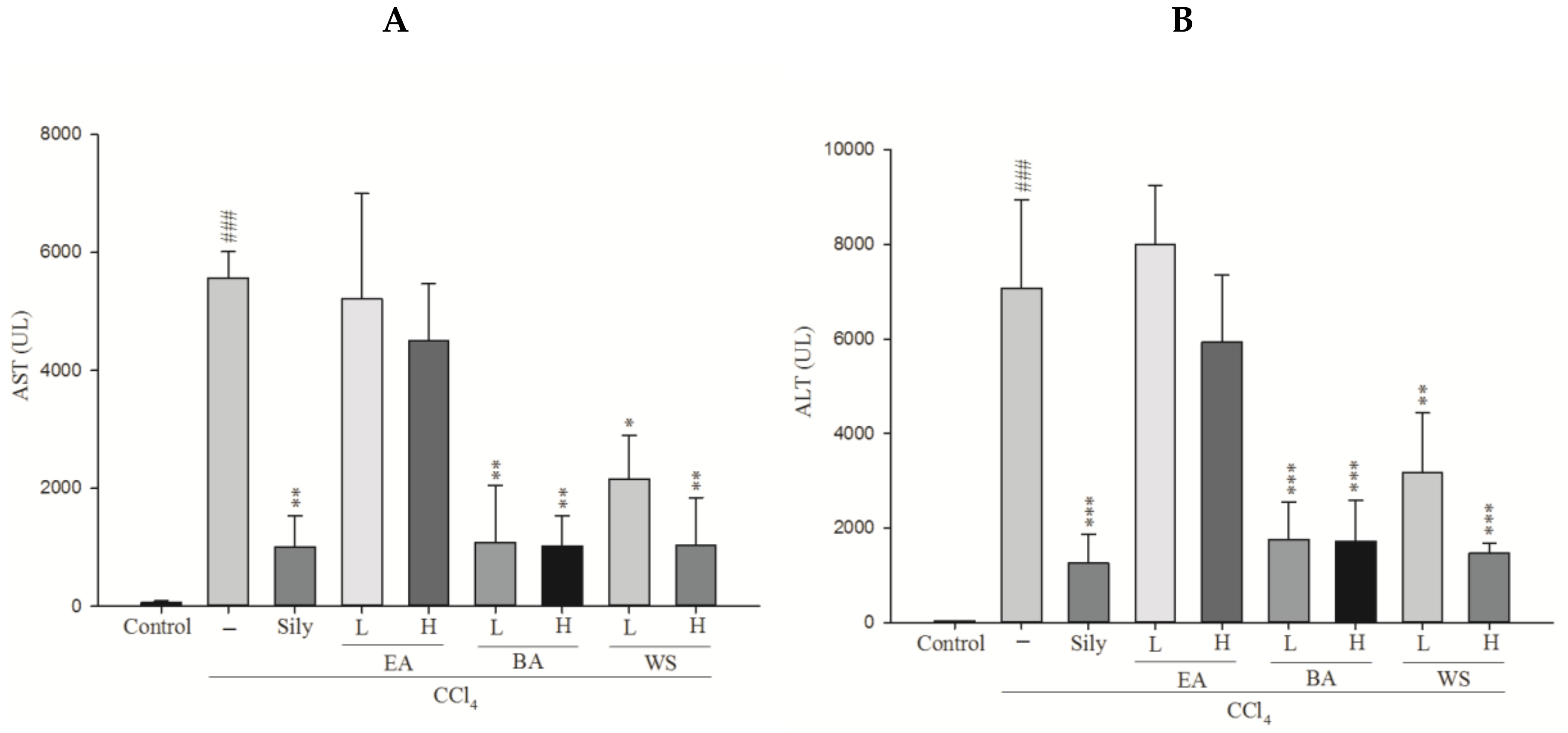
2.1. Measurement of Serum AST and Serum ALT
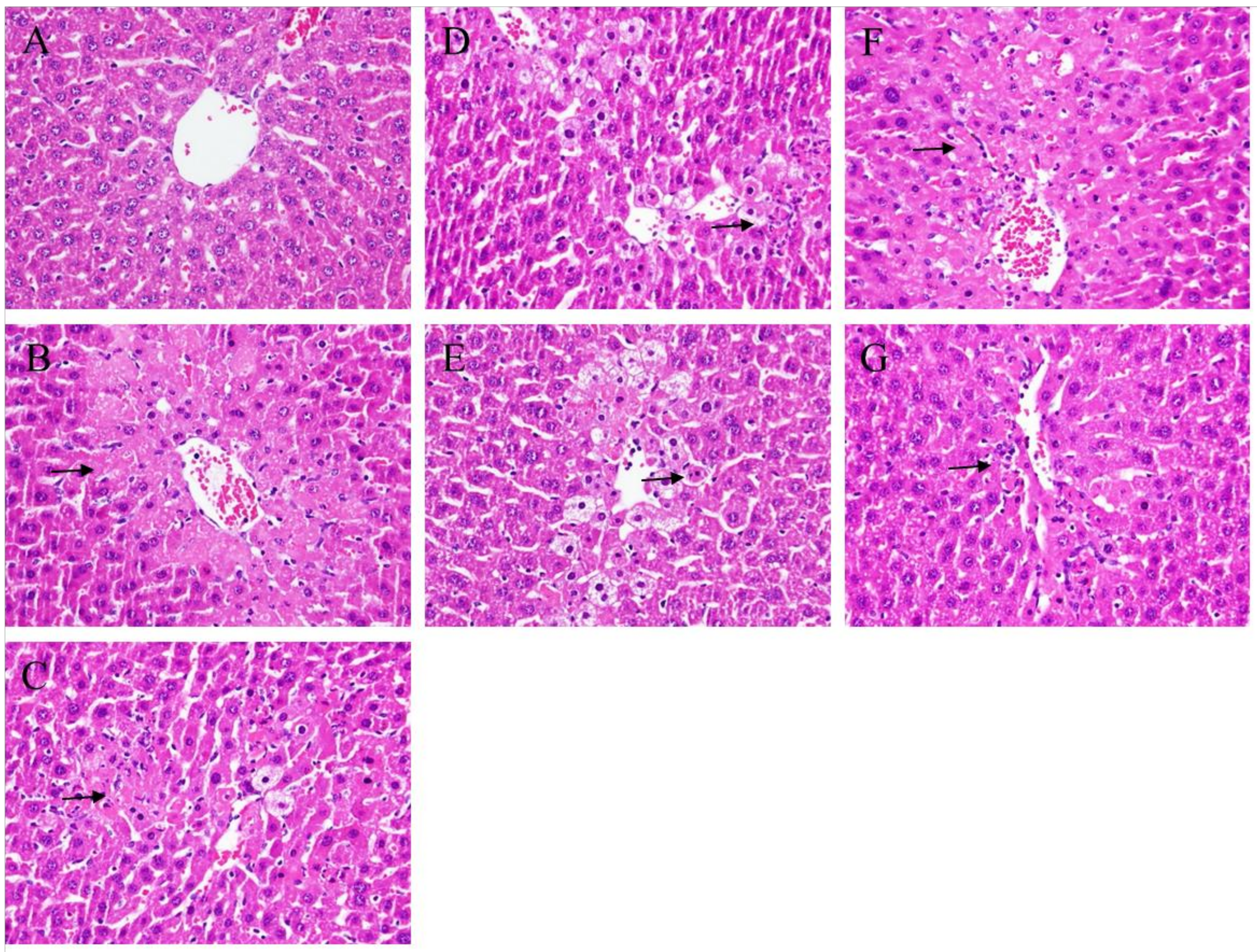
2.2. Histopathological Analysis
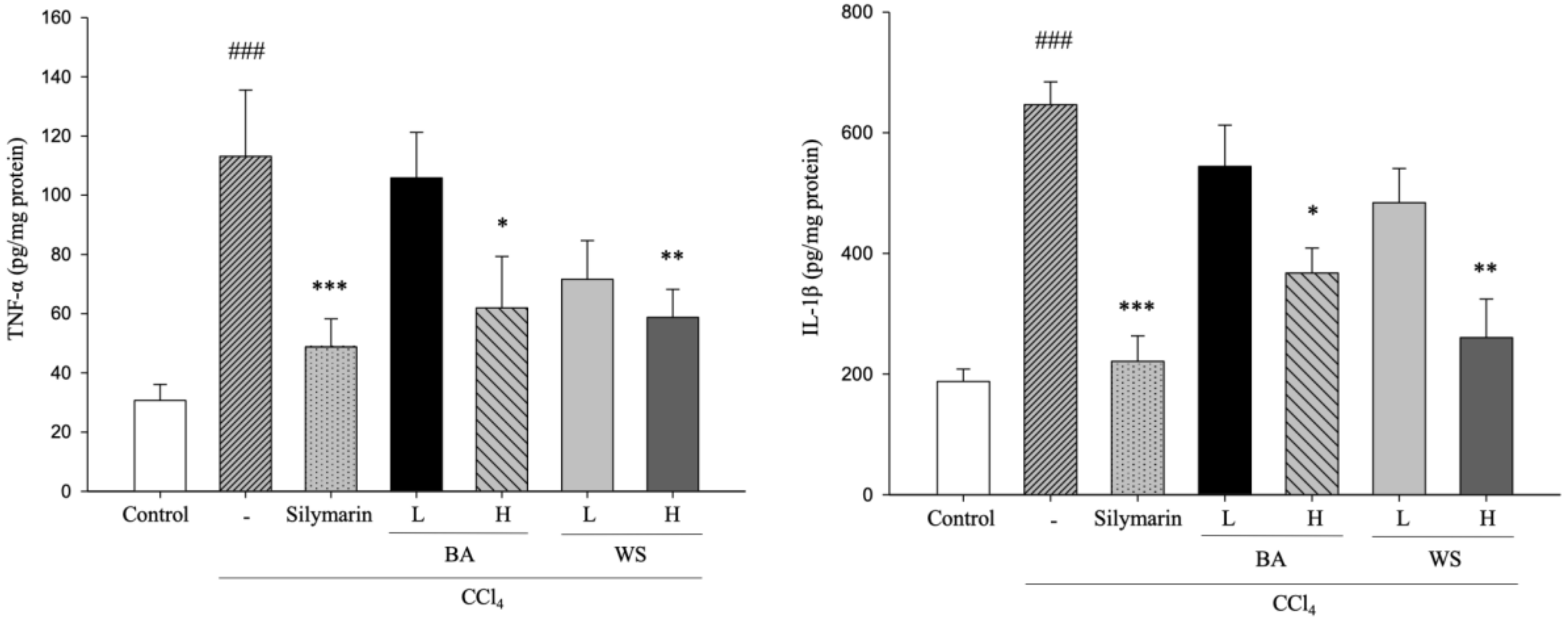
2.3. Hepatic TNF-α and IL-1β Assays
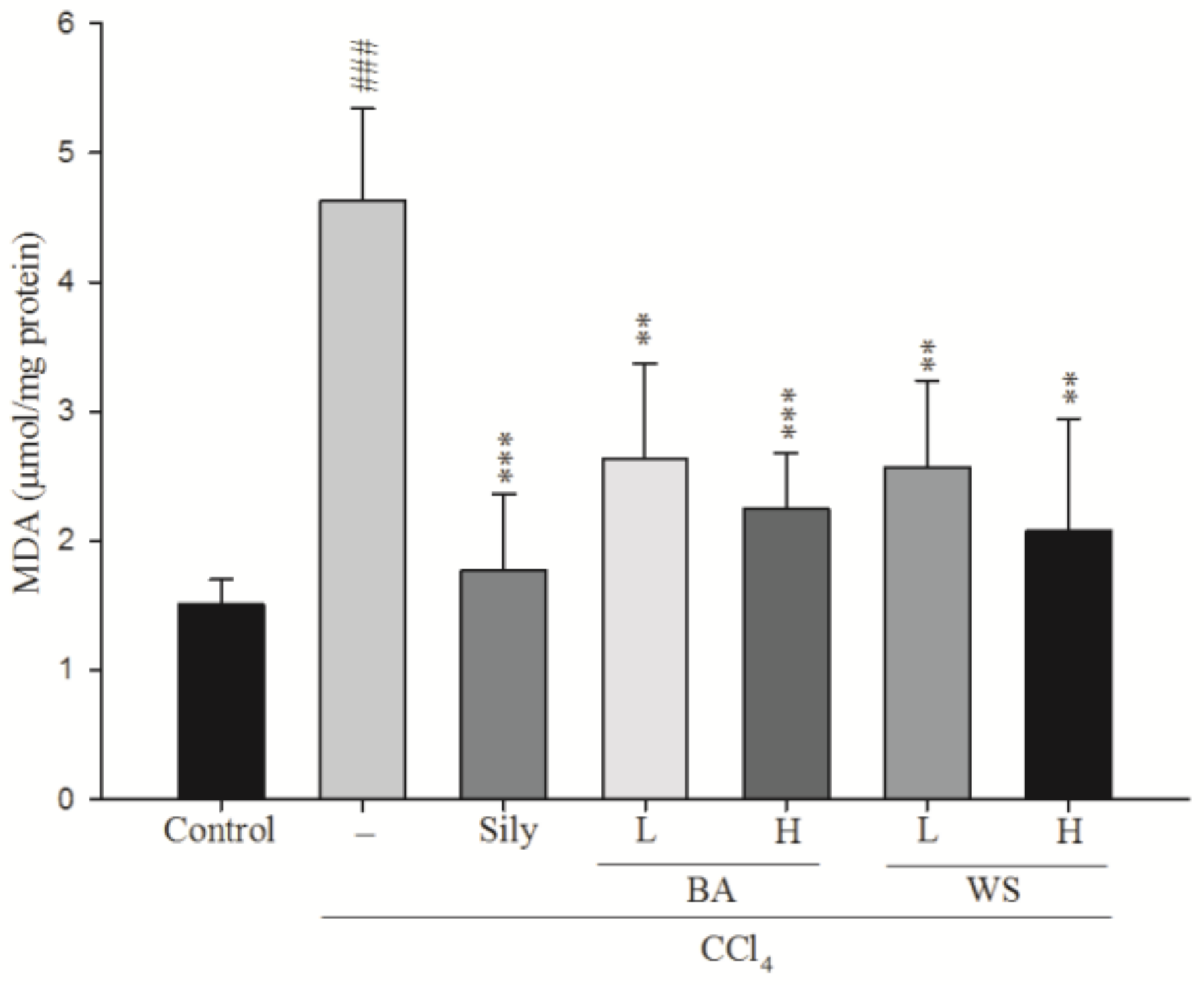
2.4. Hepatic MDA Assay
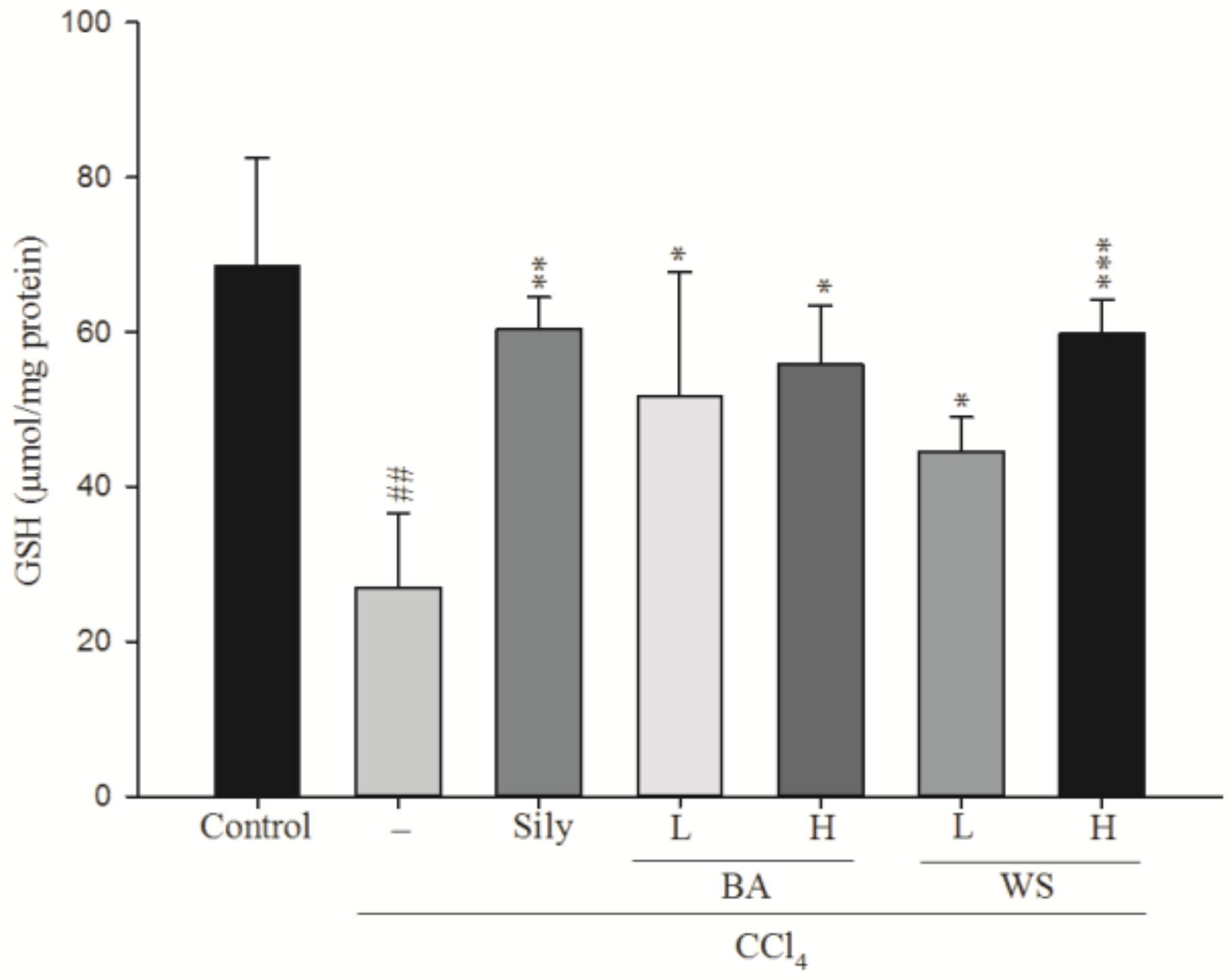
2.5. Measurement of Hepatic Glutathione (GSH) Level
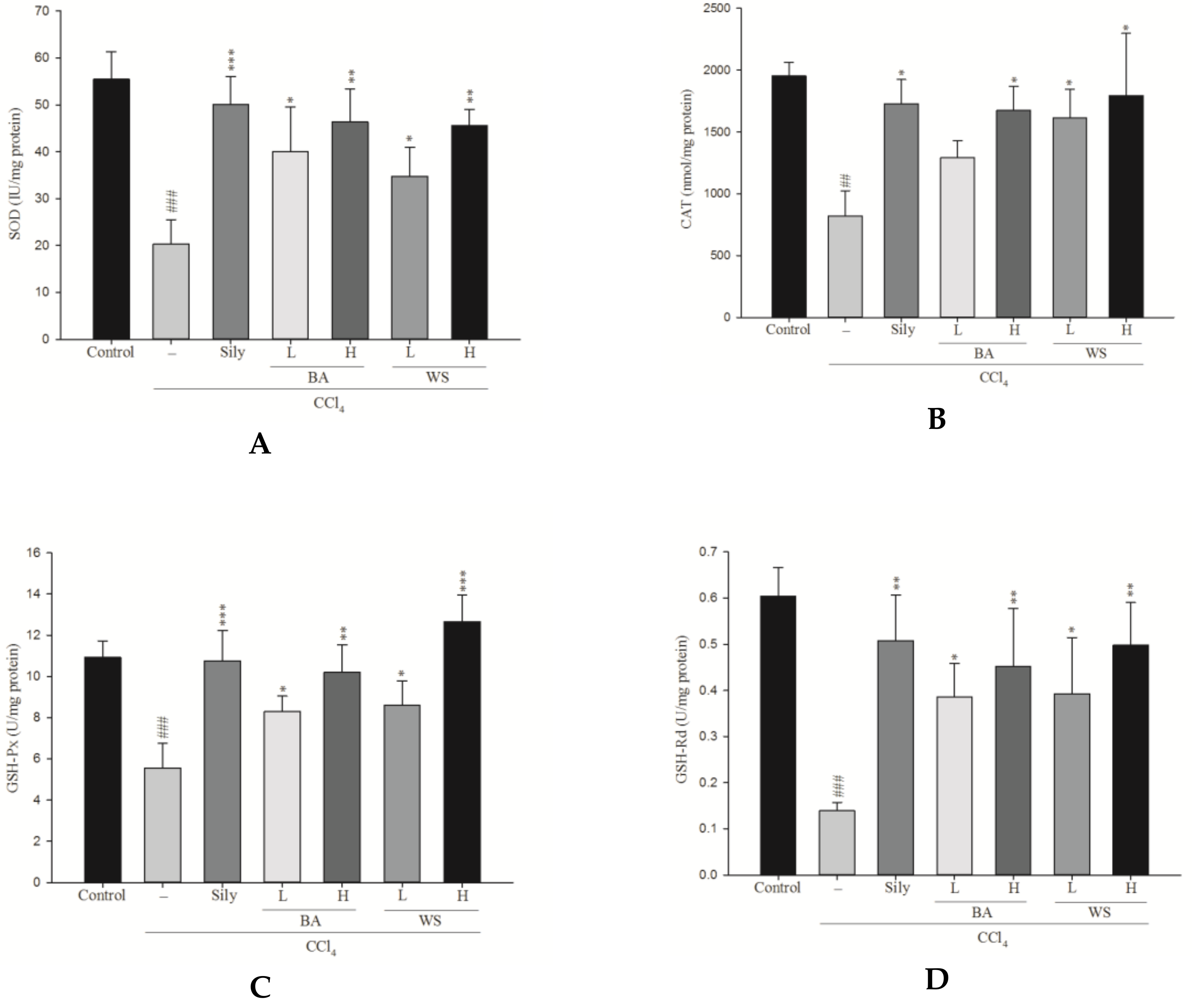
2.6. Measurement of Antioxidant Enzymatic Activity
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Preparation of Black Garlic
4.3. Carbon Tetrachloride Induced Acute Hepatic Injury
4.4. Preparation of Blood and Liver
4.5. Serum Biochemistry Analysis for Hepatic Function
4.6. Pathological Analysis
4.7. Hepatic TNF-α and IL-1β Assays
4.8. Hepatic Malondialdehyde (MDA) Assay
4.9. Measurement of Hepatic GSH Level
4.10. Antioxidant Enzymatic Activity Measurements
4.11. Quantification of SAC in Black Garlic
4.12. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations:
| AHI | Acute Hepatic Injury |
| ALT | Alanine Aminotransferase |
| AST | Aspartate Aminotransferase |
| ALP | Alkaline Phosphatase |
| AMS | Allyl Methyl Sulfide |
| ABG | Aged Black Garlic |
| ANOVA | Analysis of Variance |
| BG | Black Garlic |
| BA | N-butanol Layer Extract |
| CAT | Catalase |
| CCl4 | Carbon Tetrachloride |
| DAS | Diallyl Sulfide |
| DADS | Diallyl Disulfide |
| DATS | Diallyl Trisulfide |
| DTNB | 5,5′-Dithio-bis-(2-nitrobenzoic acid) |
| EA | Ethyl Acetate Layer Extract |
| GSACs | γ-Glutamyl-S-allyl-l-cysteines |
| GSH | Glutathione |
| GSH-Px | Glutathione Peroxidase |
| GSH-Rd | Glutathione Reductase |
| GSSG | Glutathione Disulfide |
| GST | Glutathione S-transferase |
| H & E | Hematoxylin-eosin |
| HRP | Horseradish Peroxidase |
| IL-1β | Interleukin-1β |
| MDA | Malondialdehyde |
| NDEA | N-Nitrosodiethylamine |
| PPAR-α | Peroxisomal proliferator activator receptor alpha |
| ROS | Reactive Oxygen Species |
| SAC | S-Allyl-cysteine |
| SAMC | S-Allyl-mercaptocysteine |
| SD | Standard Deviation |
| SOD | Superoxide Dismutase |
| SREBP-1c | Sterol Regulatory Element Binding Protein 1c |
| TNF-α | Tumor Necrosis Factor Alpha |
| TBARS | Thiobarbituric Acid Reactive Substances |
| TCA | Trichloroacetic Acid |
| WS | Water Layer Extract |
References
- Chen, Y.A.; Tsai, J.C.; Cheng, K.C.; Liu, K.F.; Chang, C.K.; Hsieh, C.W. Extracts of black garlic exhibits gastrointestinal motility effect. Food Res. Int. 2018, 107, 102–109. [Google Scholar] [CrossRef]
- Banerjee, S.K.; Mukherjee, P.K.; Maulik, S.K. Garlic as an antioxidant: The good, the bad and the ugly. Phytother. Res. 2002, 17, 97–106. [Google Scholar] [CrossRef]
- Iciek, M.; Kwiecien, I.; Wlodek, L. Biological properties of garlic and garlic-derived organosulfur compounds. Environ. Mol. Mutagen. 2009, 50, 247–265. [Google Scholar] [CrossRef] [PubMed]
- Guan, M.J.; Zhao, N.; Xie, K.Q.; Zeng, T. Hepatoprotective effects of garlic against ethanol-induced liver injury: A mini-review. Food Chem. Toxicol. 2017, 111, 467–473. [Google Scholar] [CrossRef]
- Liang, T.; Wei, F.; Lu, Y.; Kodani, Y.; Nakada, M.; Miyakawa, T.; Tanokura, M. Comprehensive NMR Analysis of Compositional Changes of Black Garlic during Thermal Processing. J. Agric. Food Chem. 2015, 63, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Kimura, S.; Tung, Y.C.; Pan, M.H.; Su, N.W.; Lai, Y.J.; Cheng, K.C. Black garlic: A critical review of its production, bioactivity, and application. J. Food Drug Anal. 2017, 25, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Zhang, R.; Gao, H.; Xiao, J.; Tipoe, G.L. Chapter 28—Garlic and Liver Diseases. In The Liver: Oxidative Stress and Dietary Antioxidants; Elsevier: London, UK, 2018; pp. 337–347. [Google Scholar]
- Sher, A.; Fakhar-ul-mahmood, M.; Shah, S.; Bukhsh, S.; Murtaza, G. Effect of garlic extract on blood glucose level and lipid profile in normal and alloxan diabetic rabbits. Adv. Clin. Exp. Med. 2012, 21, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Olalekan, L.A.; Lawal, A.F.; Ologundudu, A.; Adeniran, O.Y.; Omonkhua, A.; Obi, F. Antioxidant effects of heated garlic juice on cadmium-induced liver damage in rats as compared to ascorbic acid. J. Toxicol. Sci. 2011, 36, 549–557. [Google Scholar] [CrossRef]
- Kodera, Y.; Suzuki, A.; Imads, O.; Kasuga, S.; Sumioka, I.; Kanezawa, A.; Taru, N.; Fujikawa, M.; Nagae, S.; Masamoto, K.; et al. Physical, chemical, and biological properties of S-allylcysteine, an amino acid derived from garlic. J. Agric. Food Chem. 2002, 50, 622–632. [Google Scholar] [CrossRef]
- Wang, X.; Jiao, F.; Wang, Q.W.; Wang, J.; Yang, K.; Hu, R.R.; Liu, H.C.; Wang, H.Y.; Wang, Y.S. Aged black garlic extract induces inhibition of gastric cancer cell growth in vitro and in vivo. Mol. Med. Rep. 2012, 5, 66–72. [Google Scholar] [CrossRef]
- Jung, Y.M.; Lee, S.H.; Lee, D.S.; You, M.J.; Chung, I.K.; Cheon, W.H.; Kwon, Y.S.; Lee, Y.J.; Ku, S.K. Fermented garlic protects diabetic, obese mice when fed a high-fat diet by antioxidant effects. Nutr. Res. 2011, 31, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.H.; Lee, C.W.; Oh, S.J.; Yun, J.; Kang, M.R.; Han, S.B.; Park, H.; Jung, J.C.; Chung, Y.H.; Kang, J.S. Hepatoprotective effect of aged black garlic extract in rodents. Toxicol Res. 2014, 30, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Iredale, J.P.; Benyon, R.C.; Pickering, J.; McCullen, M.; Northrop, M.; Pawley, S.; Hovell, C.; Arthur, M.J. Mechanisms of spontaneous resolution of rat liver fibrosis. Hepatic stellate cell apoptosis and reduced hepatic expression of metalloproteinase inhibitors. J. Clin. Investig. 1998, 102, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Manibusan, M.K.; Odin, M.; Eastmond, D.A. Postulated Carbon Tetrachloride Mode of Action: A Review. J. Environ. Sci. Health. C Environ. Carcinog. Ecotoxicol. Rev. 2007, 25, 185–209. [Google Scholar] [CrossRef] [PubMed]
- Recknagel, R.O.; Glende, E.A., Jr.; Dolak, J.A.; Waller, R.L. Mechanisms of carbon tetrachloride toxicity. Pharmacol. Ther. 1989, 43, 139–154. [Google Scholar] [CrossRef]
- Weber, L.W.D.; Boll, M.; Stampfl, A. Hepatotoxicity and mechanism of action of haloalkanes: Carbon tetrachloride as a toxicological model. Crit. Rev. Toxicol. 2003, 33, 105–136. [Google Scholar] [CrossRef] [PubMed]
- Tsai, J.C.; Peng, W.H.; Chiu, T.H.; Huang, S.C.; Huang, T.H.; Lai, S.C.; Lai, Z.R.; Lee, C.Y. Hepatoprotective Effect of Scoparia dulcis on Carbon Tetrachloride Induced Acute Liver Injury in Mice. Am. J. Chin. Med. 2010, 38, 761–775. [Google Scholar] [CrossRef] [PubMed]
- Pari, L.; Prasath, A. Efficacy of caffeic acid in preventing nickel induced oxidative damage in liver of rats. Chem. Biol. Interact. 2008, 173, 77–83. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, C.; Zhang, H. Hepatoprotective effects of kaempferol 3-O-rutinoside and kaempferol 3-O-glucoside from Carthamus tinctorius L. on CCl4-induced oxidative liver injury in mice. J. Food Drug Anal. 2015, 23, 310–317. [Google Scholar] [CrossRef]
- Basu, S. Carbon tetrachloride-induced lipid peroxidation: Eicosanoid formation and their regulation by antioxidant nutrients. Toxicology 2003, 189, 113–127. [Google Scholar] [CrossRef]
- Jaeschke, H.; Gores, G.J.; Cederbaum, A.I.; Hinson, J.A.; Pessayre, D.; Lemasters, J.J. Mechanisms of hepatotoxicity. J. Toxicol. Sci. 2002, 65, 166–176. [Google Scholar] [CrossRef]
- Gao, B. Cytokines, STATs and liver disease. Cell Mol. Immunol. 2005, 2, 92–100. [Google Scholar] [PubMed]
- Hung, M.Y.; Fu, T.Y.; Shih, P.H.; Lee, C.P.; Yen, G.C. Du-Zhong (Eucommia ulmoides Oliv.) leaves inhibit CCl4-induced hepatic damage in rats. Food Chem. Toxicol. 2006, 44, 1424–1431. [Google Scholar] [CrossRef] [PubMed]
- Domitrović, R.; Jakovac, H.; Milin, C.; Radosević-Stasić, B. Dose- and time-dependent effects of luteolin on carbon tetrachloride-induced hepatotoxicity in mice. Exp. Toxicol. Pathol. 2009, 61, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.W.; Tsai, C.F.; Chang, W.H.; Ho, Y.C.; Chen, W.K.; Lu, F.J. Protective effects of Dunaliella salina-A carotenoids-rich alga, against carbon tetrachloride-induced hepatotoxicity in mice. Food Chem. Toxicol. 2008, 46, 3311–3317. [Google Scholar] [CrossRef] [PubMed]
- Campo, G.M.; Squadrito, F.; Ceccarelli, S.; Calò, M.; Avenoso, A.; Campo, S.; Squadrito, G.; Altavilla, D. Reduction of carbon tetrachloride-induced rat liver injury by IRFI 042, a novel dual vitamin E-like antioxidant. Free Radic. Res. 2001, 34, 379–393. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Osuna, C.; Gitto, E. Actions of melatonin in the reduction of oxidative stress. A review. J. Biomed. Sci. 2000, 7, 444–458. [Google Scholar] [CrossRef] [PubMed]
- Cholbi, M.R.; Paya, M.; Alcaraz, M.J. Inhibitory effects of phenolic compounds on CCl4-induced microsomal lipid peroxidation. Experientia 1991, 7, 195–199. [Google Scholar] [CrossRef]
- Kim, J.S.; Kang, O.J.; Gweon, O.C. Comparison of phenolic acids and flavonoids in black garlic at different thermal processing steps. J. Funct. Food 2013, 5, 80–86. [Google Scholar] [CrossRef]
- Lu, X.M.; Li, N.Y.; Qiao, X.G.; Qiu, Z.C.; Liu, P.G. Composition analysis and antioxidant properties of black garlic extract. J. Food Drug Anal. 2017, 25, 340–349. [Google Scholar] [CrossRef]
- Chen, Y.C.; Kao, T.H.; Tseng, C.Y.; Chang, W.T.; Hsu, C.L. Methanolic extract of black garlic ameliorates diet-induced obesity via regulating adipogenesis, adipokine biosynthesis, and lipolysis. J. Funct. Food 2014, 9, 98–108. [Google Scholar] [CrossRef]
- Amagase, H.; Petesch, B.L.; Matsuura, H.; Kasuga, S.; Itakura, Y. Intake of garlic and its bioactive components. J. Nutr. 2001, 131, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Maria, M.C.; Feliciano, P.C.; Maria, D.L.C. Headspace-GC-MS volatile profile of black garlic VS fresh garlic: Evolution along fermentation and behavior under heating. LWT Food Sci. Technol. 2017, 80, 98–105. [Google Scholar] [CrossRef]
- Corzo-Martinez, M.; Corso, N.; Villamiel, M. Biological propertyes of onions and garlic. Trends Food Sci. Technol. 2007, 18, 609–625. [Google Scholar] [CrossRef]
- Montaño, A.; Casado, F.J.; Castro, A.D.; Sánchez, A.H.; Rejano, L. Vitamin content and amino acid composition of pickled garlic processed with and without fermentation. J. Agric. Food Chem. 2004, 52, 7324–7330. [Google Scholar] [CrossRef] [PubMed]
- Sivapatham, S.; Perumal, S. Prevention of N-nitrosodiethylamine-induced hepaticarcinogenesis by S-allylcysteine. Mol. Cell Biochem. 2008, 310, 209–214. [Google Scholar] [CrossRef]
- Gong, Z.; Ye, H.; Huo, Y.; Wang, L.; Huang, Y.; Huang, M.; Yuan, X. S-allyl-cysteine attenuates carbon tetrachloride-induced liver fibrosis in rats by targeting STAT3/SMAD3 pathway. Am. J. Transl. Res. 2018, 10, 1337–1346. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.L.; Peng, H.C.; Hsieh, Y.C.; Yang, S.C. Epidermal growth factor improved alcohol-induced inflammation in rats. Alcohol 2014, 48, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.J.; Chou, S.C.; Chiu, C.S.; Kao, C.P.; Wu, K.C.; Chen, C.J.; Tsai, J.C.; Peng, W.H. Hepatoprotective effect of the ethanol extract of Polygonum orientale on carbon tetrachloride-induced acute liver injury in mice. J. Food Drug Anal. 2017, 26, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Tsai, J.C.; Chiu, C.S.; Chen, Y.C.; Lee, M.S.; Hao, X.Y.; Hsieh, M.T.; Kao, C.P.; Peng, W.H. Hepatoprotective effect of Coreopsis tinctoria flowers against carbon tetrachloride-induced liver damage in mice. BMC Complement Altern. Med. 2017, 17, 139. [Google Scholar] [CrossRef]
- Chen, J.R.; Chen, Y.L.; Peng, H.C.; Lu, Y.A.; Chuang, H.L.; Chang, H.Y.; Wang, H.Y.; Su, Y.J.; Yang, S.C. Fish oil reduces hepatic injury by maintaining normal intestinal permeability and microbiota in chronic ethanol-fed rats. Gastroent. Res. Pract. 2016, 21, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Huang, W.C.; Yang, S.C.; Chan, C.C.; Lin, W.T. Ganoderma tsugae hepatoprotection against exhaustive exercise-induced liver injury in rats. Molecules 2013, 18, 1741–1754. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Yoo, M.; Kim, S.; Shin, D. Identification and quantification of S-allyl-L-cysteine in heated garlic juice by HPLC with ultraviolet and mass spectrometry detection. LWT Food Sci. Technol. 2014, 57, 516–521. [Google Scholar] [CrossRef]
- Yoo, M.; Lee, S.; Lee, S.; Seog, H.; Shin, D. Validation of high performance liquid chromatography methods for determination of bioactive sulfur compounds in garlic bulbs. Food Sci Biotechnol. 2010, 19, 1619–1626. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| Extract Solvent | SAC Content (μg/g) |
|---|---|
| EA | ND |
| BA | 497.58 ± 43.78 |
| WS | ND |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, J.-C.; Chen, Y.-A.; Wu, J.-T.; Cheng, K.-C.; Lai, P.-S.; Liu, K.-F.; Lin, Y.-K.; Huang, Y.-T.; Hsieh, C.-W. Extracts from Fermented Black Garlic Exhibit a Hepatoprotective Effect on Acute Hepatic Injury. Molecules 2019, 24, 1112. https://doi.org/10.3390/molecules24061112
Tsai J-C, Chen Y-A, Wu J-T, Cheng K-C, Lai P-S, Liu K-F, Lin Y-K, Huang Y-T, Hsieh C-W. Extracts from Fermented Black Garlic Exhibit a Hepatoprotective Effect on Acute Hepatic Injury. Molecules. 2019; 24(6):1112. https://doi.org/10.3390/molecules24061112
Chicago/Turabian StyleTsai, Jen-Chieh, Yi-An Chen, Jung-Tsung Wu, Kuan-Chen Cheng, Ping-Shan Lai, Keng-Fan Liu, Yung-Kai Lin, Yu-Tsang Huang, and Chang-Wei Hsieh. 2019. "Extracts from Fermented Black Garlic Exhibit a Hepatoprotective Effect on Acute Hepatic Injury" Molecules 24, no. 6: 1112. https://doi.org/10.3390/molecules24061112
APA StyleTsai, J.-C., Chen, Y.-A., Wu, J.-T., Cheng, K.-C., Lai, P.-S., Liu, K.-F., Lin, Y.-K., Huang, Y.-T., & Hsieh, C.-W. (2019). Extracts from Fermented Black Garlic Exhibit a Hepatoprotective Effect on Acute Hepatic Injury. Molecules, 24(6), 1112. https://doi.org/10.3390/molecules24061112








