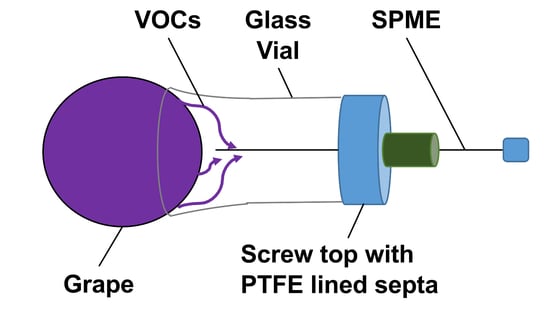
Evaluation of Volatile Metabolites Emitted In-Vivo from Cold-Hardy Grapes during Ripening Using SPME and GC-MS: A Proof-of-Concept
Abstract
1. Introduction
2. Results
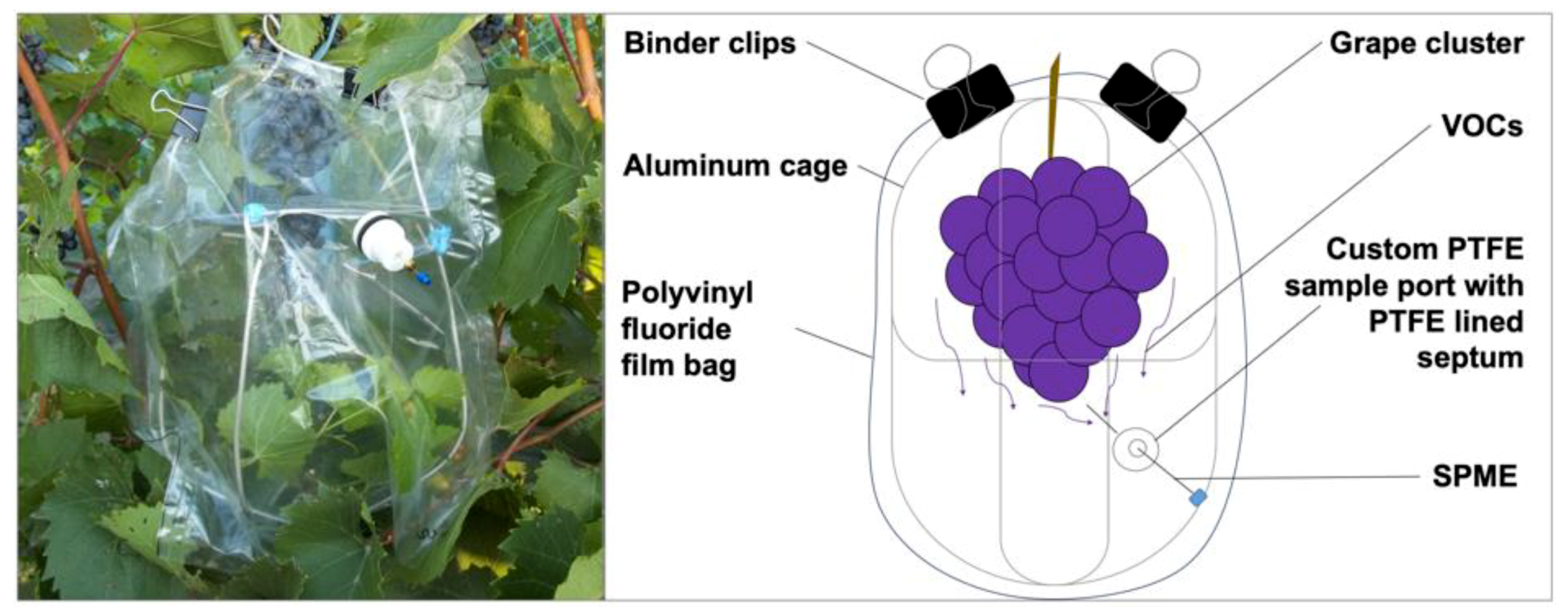
2.1. Sampling Devices to Capture Volatiles Emitted from a Whole Cluster and Single Berry
2.2. Volatiles Emitted In-Vivo from Four Cold-Hardy Grape Cultivars
2.2.1. Emissions from Whole Grape Cluster
Frontenac
Marquette
2.2.2. Emissions from Single Berries
Frontenac
Marquette
St. Croix
La Crescent
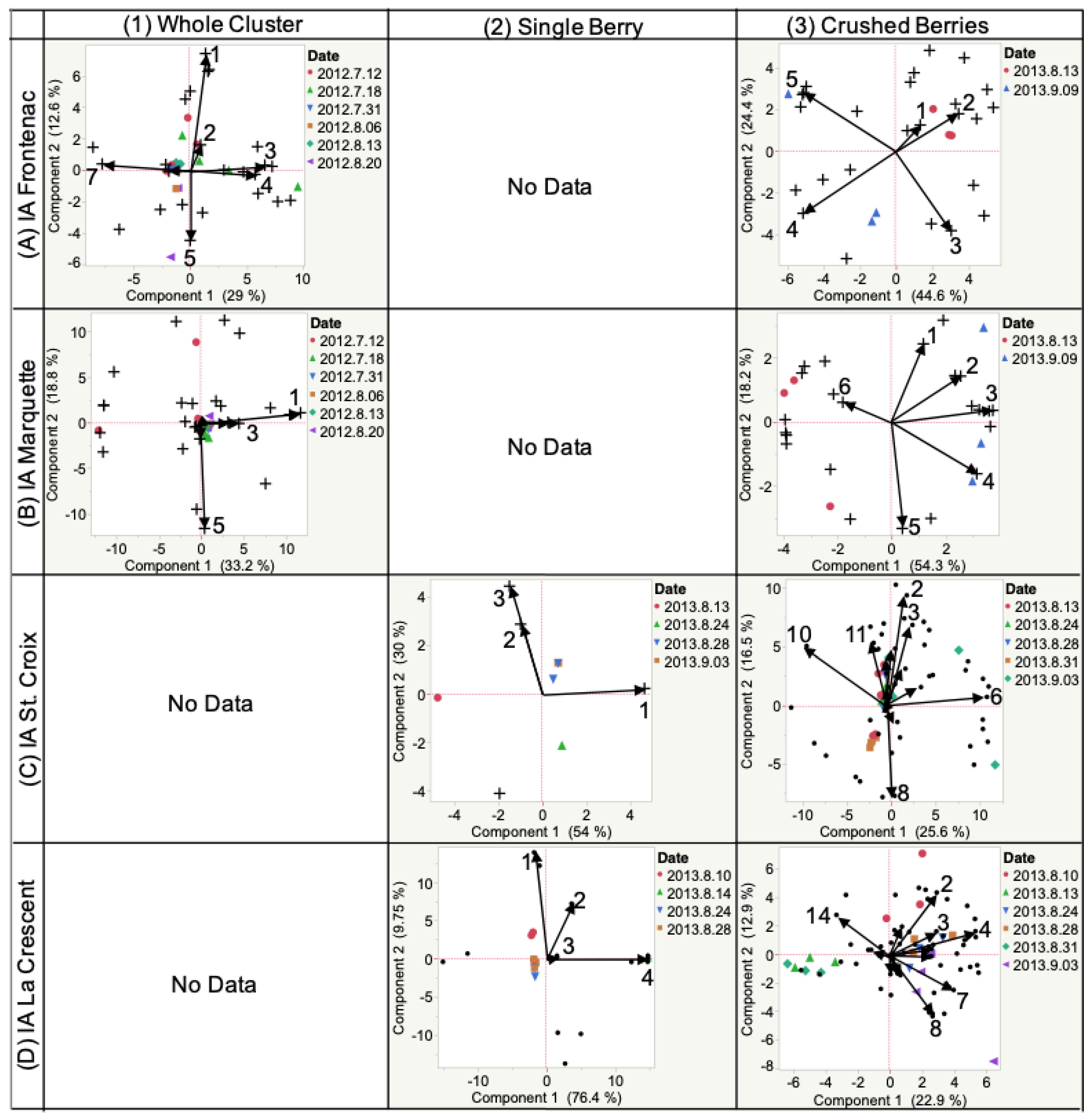
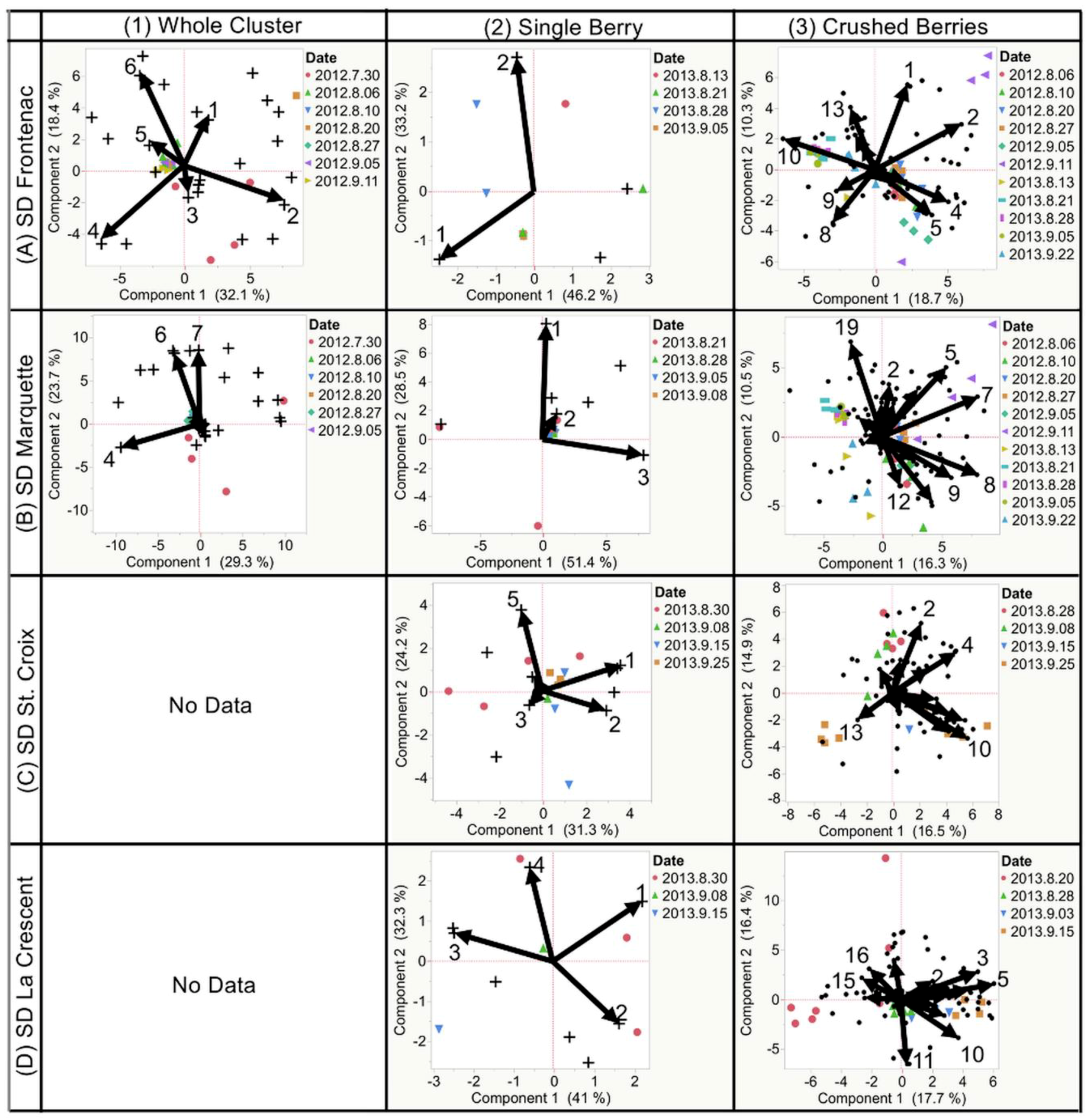
2.3. Destructive Sampling
2.3.1. Frontenac
2.3.2. Marquette
2.3.3. St. Croix
2.3.4. La Crescent
3. Discussion
4. Materials and Methods
4.1. Overview
4.2. In-Vivo Sampling of Volatiles from a Whole Cluster of Grapes
4.3. In-Vivo Sampling of Volatiles from a Single Grape
4.4. Destructive Sampling
4.5. Data Acquisition and Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Source | DF | Sum of Squares | Mean Squares | F | Pr > F |
|---|---|---|---|---|---|
| Method | 3 | 1.88 × 1016 | 6.26 × 1015 | 122.794 | <0.0001 |
| Site | 1 | 6.92 × 1010 | 6.92 × 1010 | 0.001 | 0.971 |
| Cultivar | 3 | 1.91 × 1016 | 6.37 × 1015 | 124.900 | <0.0001 |
| Date/Time | 31 | 1.14 × 1016 | 3.67 × 1014 | 7.204 | <0.0001 |
| Method*Site | 2 | 6.54 × 1014 | 3.27 × 1014 | 6.416 | 0.002 |
| Method*Cultivar | 4 | 6.43 × 1015 | 1.61 × 1015 | 31.532 | <0.0001 |
| Method*Date/Time | 14 | 1.45 × 1015 | 1.04 × 1014 | 2.035 | 0.016 |
| Site*Cultivar | 3 | 7.36 × 1014 | 2.45 × 1014 | 4.815 | 0.003 |
| Site*Date/Time | 2 | 2.24 × 1014 | 1.12 × 1014 | 2.202 | 0.113 |
| Cultivar*Date/Time | 24 | 2.37 × 1015 | 9.87 × 1013 | 1.937 | 0.007 |
| Method*Site*Cultivar | 1 | 8.96 × 1014 | 8.96 × 1014 | 17.588 | <0.0001 |
| Method*Site*Date/Time | 0 | 0.00 × 1000 | |||
| Method*Cultivar*Date/Time | 7 | 6.53 × 1013 | 9.33 × 1012 | 0.183 | 0.989 |
| Category | Groups | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Method-GV*Cultivar-La Crescent*Date/Time-9/8/2013 | A | ||||||||||||||
| Method-GV*Cultivar-La Crescent*Date/Time-9/15/2013 | A | ||||||||||||||
| Method-GV*Cultivar-La Crescent*Date/Time-8/30/2013 | A | B | |||||||||||||
| Method-GV*Cultivar-St. Croix*Date/Time-9/15/2013 | A | B | C | ||||||||||||
| Method-GV*Cultivar-St. Croix*Date/Time-9/8/2013 | A | B | C | ||||||||||||
| Method-GV*Cultivar-St. Croix*Date/Time-8/30/2013 | A | B | C | D | |||||||||||
| Method-GV*Cultivar-St. Croix*Date/Time-9/25/2013 | A | B | C | D | E | ||||||||||
| Method-CB*Cultivar-Frontenac*Date/Time-8/13/2013 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-Marquette*Date/Time-8/13/2013 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-La Crescent*Date/Time-8/13/2013 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-Frontenac*Date/Time-9/9/2013 | A | B | C | D | E | F | |||||||||
| Method-GV*Cultivar-St. Croix*Date/Time-8/29/2013 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-8/10/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-8/27/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Marquette*Date/Time-8/13/2012 | A | B | C | D | E | F | |||||||||
| Method-*PVF*Cultivar-Marquette*Date/Time-7/31/2012 | A | B | C | D | E | F | |||||||||
| Method-*PVF*Cultivar-Marquette*Date/Time-8/06/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Marquette*Date/Time-9/5/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-9/5/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-9/11/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-8/6/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Marquette*Date/Time-8/6/2012 | A | B | C | D | E | F | |||||||||
| Method-*PVF*Cultivar-Marquette*Date/Time-8/6/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-8/13/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Marquette*Date/Time-8/10/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Marquette*Date/Time-8/20/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Marquette*Date/Time-8/27/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-7/31/2012 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-Marquette*Date/Time-9/9/2013 | A | B | C | D | E | F | |||||||||
| Method-*PVF*Cultivar-Marquette*Date/Time-8/6/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-8/20/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Marquette*Date/Time-7/18/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-7/12/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Marquette*Date/Time-7/31/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Marquette*Date/Time-7/30/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-7/18/2012 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Frontenac*Date/Time-7/30/2012 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-La Crescent*Date/Time-8/31/2013 | A | B | C | D | E | F | |||||||||
| Method-PVF*Cultivar-Marquette*Date/Time-7/12/2012 | A | B | C | D | E | F | |||||||||
| Method-GV*Cultivar-La Crescent*Date/Time-8/29/2013 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-Frontenac*Date/Time-8/21/2013 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-Frontenac*Date/Time-9/22/2013 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-Frontenac*Date/Time-8/28/2013 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-Frontenac*Date/Time-9/5/2013 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-Frontenac*Date/Time-9/13/2013 | A | B | C | D | E | F | |||||||||
| Method-CB*Cultivar-Marquette*Date/Time-8/21/2013 | A | B | C | D | E | F | |||||||||
| Method-GV*Cultivar-Frontenac*Date/Time-9/5/2013 | A | B | C | D | E | F | G | ||||||||
| Method-GV*Cultivar-Frontenac*Date/Time-8/21/2013 | A | B | C | D | E | F | G | ||||||||
| Method-GV*Cultivar-Frontenac*Date/Time-9/13/2013 | A | B | C | D | E | F | G | ||||||||
| Method-GV*Cultivar-Marquette*Date/Time-9/13/2013 | A | B | C | D | E | F | G | ||||||||
| Method-GV*Cultivar-Marquette*Date/Time-9/8/2013 | A | B | C | D | E | F | G | ||||||||
| Method-GV*Cultivar-Marquette*Date/Time-9/5/2013 | A | B | C | D | E | F | G | ||||||||
| Method-GV*Cultivar-Marquette*Date/Time-8/21/2013 | A | B | C | D | E | F | G | ||||||||
| Method-CB*Cultivar-Frontenac*Date/Time-9/5/2012 | A | B | C | D | E | F | G | ||||||||
| Method-CB*Cultivar-Frontenac*Date/Time-8/6/2012 | A | B | C | D | E | F | G | H | |||||||
| Method-CB*Cultivar-Marquette*Date/Time-9/22/2013 | A | B | C | D | E | F | G | H | |||||||
| Method-CB*Cultivar-Frontenac*Date/Time-8/27/2012 | A | B | C | D | E | F | G | H | |||||||
| Method-CB*Cultivar-Marquette*Date/Time-9/5/2013 | A | B | C | D | E | F | G | H | |||||||
| Method-GV*Cultivar-St. Croix*Date/Time-9/3/2013 | A | B | C | D | E | F | G | H | |||||||
| Method-GV*Cultivar-St. Croix*Date/Time-8/24/2013 | A | B | C | D | E | F | G | H | |||||||
| Method-CB*Cultivar-Marquette*Date/Time-8/28/2013 | A | B | C | D | E | F | G | H | |||||||
| Method-CB*Cultivar-Frontenac*Date/Time-8/10/2012 | A | B | C | D | E | F | G | H | I | ||||||
| Method-CB*Cultivar-Marquette*Date/Time-9/13/2013 | A | B | C | D | E | F | G | H | I | ||||||
| Method-CB*Cultivar-Frontenac*Date/Time-8/20/2012 | A | B | C | D | E | F | G | H | I | ||||||
| Method-CB*Cultivar-St. Croix*Date/Time-8/29/2013 | B | C | D | E | F | G | H | I | J | ||||||
| Method-CB*Cultivar-Marquette*Date/Time-8/6/2012 | C | D | E | F | G | H | I | J | |||||||
| Method-CB*Cultivar-Frontenac*Date/Time-9/11/2012 | C | D | E | F | G | H | I | J | |||||||
| Method-CB*Cultivar-Marquette*Date/Time-8/10/2012 | C | D | E | F | G | H | I | J | |||||||
| Method-CB*Cultivar-St. Croix*Date/Time-8/31/2013 | C | D | E | F | G | H | I | J | |||||||
| Method-GV*Cultivar-La Crescent*Date/Time-8/24/2013 | C | D | E | F | G | H | I | J | K | ||||||
| Method-GV*Cultivar-La Crescent*Date/Time-8/10/2013 | C | D | E | F | G | H | I | J | K | ||||||
| Method-CB*Cultivar-La Crescent*Date/Time-8/10/2013 | C | D | E | F | G | H | I | J | K | ||||||
| Method-CB*Cultivar-St. Croix*Date/Time-9/8/2013 | C | D | E | F | G | H | I | J | K | L | |||||
| Method-GV*Cultivar-Frontenac*Date/Time-8/29/2013 | C | D | E | F | G | H | I | J | K | L | |||||
| Method-GV*Cultivar-Marquette*Date/Time-8/29/2013 | C | D | E | F | G | H | I | J | K | L | |||||
| Method-CB*Cultivar-St. Croix*Date/Time-9/25/2013 | D | E | F | G | H | I | J | K | L | M | |||||
| Method-CB*Cultivar-St. Croix*Date/Time-8/13/2013 | E | F | G | H | I | J | K | L | M | ||||||
| Method-CB*Cultivar-Marquette*Date/Time-9/5/2012 | F | G | H | I | J | K | L | M | |||||||
| Method-CB*Cultivar-Marquette*Date/Time-8/20/2012 | F | G | H | I | J | K | L | M | |||||||
| Method-CB*Cultivar-Marquette*Date/Time-9/11/2012 | F | G | H | I | J | K | L | M | |||||||
| Method-CB*Cultivar-St. Croix*Date/Time-8/24/2013 | F | G | H | I | J | K | L | M | N | ||||||
| Method-CB*Cultivar-Marquette*Date/Time-8/27/2012 | F | G | H | I | J | K | L | M | N | ||||||
| Method-CB*Cultivar-La Crescent*Date/Time-8/29/2013 | G | H | I | J | K | L | M | N | O | ||||||
| Method-CB*Cultivar-La Crescent*Date/Time-9/20/2013 | H | I | J | K | L | M | N | O | |||||||
| Method-CB*Cultivar-La Crescent*Date/Time-8/28/2013 | I | J | K | L | M | N | O | ||||||||
| Method-CB*Cultivar-St. Croix*Date/Time-9/3/2013 | J | K | L | M | N | O | |||||||||
| Method-GV*Cultivar-La Crescent*Date/Time-8/14/2013 | K | L | M | N | O | ||||||||||
| Method-CB*Cultivar-St. Croix*Date/Time-9/15/2013 | L | M | N | O | |||||||||||
| Method-CB*Cultivar-La Crescent*Date/Time-8/24/2013 | M | N | O | ||||||||||||
| Method-CB*Cultivar-La Crescent*Date/Time-9/3/2013 | N | O | |||||||||||||
| Method-CB*Cultivar-La Crescent*Date/Time-9/15/2013 | O | ||||||||||||||
References
- Coombe, B.G.; McCarthy, M.B. Dynamics of grape berry growth and physiology of ripening. Aust. J. Grape Wine Res. 2000, 6, 131–135. [Google Scholar] [CrossRef]
- Robinson, S.P.; Davis, C. Molecular biology of grape berry ripening. Aust. J. Grape Wine Res. 2000, 6, 169–174. [Google Scholar] [CrossRef]
- Terral, J.F.; Tabard, E.; Bouby, L.; Ivorra, S.; Pastor, T.; Figueiral, I.; Picq, S.; Chevance, J.B.; Jung, C.; Fabre, L.; et al. Evolution and history of grapevine (Vitis vinifera) under domestication: New morphometric perspectives to understand seed domestication syndrome and reveal origins of ancient European cultivars. Ann. Bot. 2010, 105, 443–456. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.Q.; Liu, B.; Zhu, B.Q.; Lan, Y.B.; Gao, Y.; Wang, D.; Reeves, M.J.; Duan, C.Q. Differences in volatile profiles of Cabernet Sauvignon grapes grown in two distinct regions of China and their responses to weather conditions. Plant Physiol. Biochem. 2015, 89, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Hashizume, K.; Samuta, T. Grape maturity and light exposure affect berry methoxypyrazine concentration quality. Am. J. Enol. Viticult. 1999, 50, 194–198. [Google Scholar]
- Cai, L.; Koziel, J.A.; O’Neal, M. Determination of characteristic odorants from Harmonia axyridis beetles using in-vivo solid-phase microextraction and multidimensional gas chromatography-mass spectrometry—Olfactometry. J. Chromatogr. A 2007, 1147, 66–78. [Google Scholar] [CrossRef] [PubMed]
- Conde, C.; Silva, P.; Fontes, N.; Dias, A.; Tavares, R.; Sousa, M.; Agasse, A.; Delrot, S.; Geros, H. Biochemical changes throughout grape berry development and fruit and wine quality. Food 2007, 1–22. [Google Scholar]
- Wilwerth, J.J.; Reynolds, A.G.; Lesschaeve, I. Sensory analysis of Riesling wines from different sub-appellation in the Niagara Peninsula in Ontario. Am. J. Enol. Viticult. 2015, 66, 279–293. [Google Scholar] [CrossRef]
- Matsumoto, H.; Ikoma, Y. Effect of postharvest temperature on the muscat flavor and aroma volatile content in the berries of ‘Shine Muscat’ (Vitis labruscana Baily × V. vinifera L.). Postharvest. Biol. Tec. 2016, 112, 256–265. [Google Scholar] [CrossRef]
- Diago, M.P.; Vilanova, M.; Tardaguila, J. Effects of timing of early defoliation (manual and mechanical) on the aroma attributes of Tempranillo (Vitis vinifera L.) wines. Am. J. Enol. Viticult. 2010, 61, 382–391. [Google Scholar]
- Gardner, D.; Zoecklein, B.W. Electronic nose analysis on the effect of prefermentation cold soak of Vitis vinifera L. cv. Cabernet Sauvignon grape and wine volatiles. Am. J. Enol. Vitic. 2009, 62, 387A. [Google Scholar] [CrossRef]
- University of Minnesota, Minnesota Hardy. Available online: http://mnhardy.umn.edu/varieties/fruit/grapes (accessed on 26 December 2018).
- Skinkis, P.A.; Bordelon, B.P.; Butz, E.M. Effects of sunlight exposure on berry and wine monoterpenes and sensory characteristics of Traminette. Am. J. Enol. Viticult. 2010, 61, 147–156. [Google Scholar]
- Liu, B.; Xu, X.Q.; Cai, J.; Lan, Y.B.; Zhu, B.Q.; Wang, J. The free and enzyme-released volatile compounds of distinctive Vitis amurensis var. Zuoshanyi grapes in China. Eur. Food Res. Technol. 2015, 240, 985–997. [Google Scholar] [CrossRef]
- Zhang, S.; Petersen, M.A.; Liu, J.; Toldam-Andersen, T.B. Influence of pre-fermentation treatments on wine volatile and sensory profile of the new disease tolerant cultivar Solaris. Molecules 2015, 20, 21609–21625. [Google Scholar] [CrossRef] [PubMed]
- Pedneault, K.; Dorais, M.; Angers, P. Flavor of cold-hardy grapes: Impact of berry maturity and environmental conditions. J. Agric. Food. Chem. 2013, 64, 10418–10438. [Google Scholar] [CrossRef] [PubMed]
- Slegers, A.; Angers, P.; Ouillet, E.; Truchon, T.; Pedneault, K. Volatile compounds from grape skin, juice and wine from five interspecific hybrid grape cultivars grown in Quebec (Canada) for wine production. Molecules 2015, 20, 10980–11016. [Google Scholar] [CrossRef] [PubMed]
- Rice, S.; Koziel, J.A. The relationship between chemical concentration and odor activity value explains the inconsistency in making a comprehensive surrogate scent training tool representative of illicit drugs. Forensic. Sci. Int. 2015, 257, 257–270. [Google Scholar] [CrossRef] [PubMed]
- Rice, S.; Koziel, J.A. Odor impact of volatiles emitted from marijuana, cocaine, heroin and their surrogate scents. Data in Brief 2015, 5, 653–706. [Google Scholar] [CrossRef] [PubMed]
- Rice, S.; Koziel, J.A. Characterizing the smell of marijuana by odor impact of volatile compounds: An application of simultaneous chemical and sensory analysis. PLoS ONE 2015, 10, e0144160. [Google Scholar] [CrossRef] [PubMed]
- Hjelmeland, A.K.; Ebeler, S.E. Glycosidically bound volatile aroma compounds in grapes and wine; A review. Am. J. Enol. Vitic. 2015, 66, 1–11. [Google Scholar] [CrossRef]
- Hernandez-Orte, P.; Concejero, B.; Astrain, J.; Lacau, B.; Cacho, J.; Ferreira, V. Influence of viticulture practices on grape aroma precursors and their relation with wine aroma. J. Sci. Food Agric. 2015, 95, 688–701. [Google Scholar] [CrossRef] [PubMed]
- Pawliszyn, J. Handbook of Solid Phase Microextraction; Chemical Industry Press of China: Beijing, China, 2009. [Google Scholar]
- Flamini, G.; Cioni, P.L. Odour gradients and patterns in volatile emissions of different plant parts and developing fruits of grapefruit (Citrus paradise L.). Food Chem. 2010, 120, 984–992. [Google Scholar] [CrossRef]
- Vasquez-Cruz, M.A.; Jimenez-Garcia, S.N.; Torres-Pacheco, I.; Guzman-Maldonado, S.H.; Guevara-Gonzalez, R.G.; Miranda-Lopez, R. Effect of maturity stage and storage on flavor compounds and sensory description of Berrycatus (Myrtillocactus geometrizans). J. Food. Sci. 2012, 77, C366–C373. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Zhu, B.; Wang, Y.; Lu, L.; Lan, Y.; Reeves, M.J.; Duan, C. Influence of pre-fermentation cold maceration treatment on aroma compounds of Cabernet Sauvignon wines fermented in different industrial scale fermenters. Food Chem. 2014, 154, 217–229. [Google Scholar] [CrossRef] [PubMed]
- Battilana, J.; Emanuelli, F.; Gambino, G.; Gribaudo, I.; Gasperi, F.; Boss, P.K.; Grando, M.S. Functional effect of grapevine 1-deoxy-d-xylulose 5-phosphate synthase substitution K284N on Muscat flavour formation. J. Exp. Bot. 2011, 62, 5497–5508. [Google Scholar] [CrossRef] [PubMed]
- Esti, M.; Tamborra, P. Influence of winemaking techniques on aroma precursors. Anal. Chim. Acta 2006, 563, 173–179. [Google Scholar] [CrossRef]
- Pardo-Garcia, A.I.; Serrano de la Hoz, K.; Zalacain, A.; Alonso, G.L.; Salinas, M.R. Effect of vine foliar treatments on the varietal aroma of Monastrell wines. Food Chem. 2014, 163, 258–266. [Google Scholar] [CrossRef]
- Mansfield, A.K.; Reineccius, G. Identifying characteristic volatiles of Frontenac wine by stir bar sorptive extraction, GCO/FID, and GCO/MS. In Proceedings of the American Society of Enology and Viticulture, Seattle, WA, USA, 22–24 June 2005. [Google Scholar]
- Mansfield, A.K.; Schirle-Keller, J.; Reineccius, G.A. Identification of odor-impact compounds in red table wines produced from Frontenac grapes. Am. J. Enol. Vitic. 2011, 62, 169–176. [Google Scholar] [CrossRef]
- Cai, L.; Rice, S.; Koziel, J.A.; Dharmadhikari, M. Development of an automated method for aroma analysis of red wines from cold-hardy grapes using simultaneous solid-phase microextraction-multidimensional gas chromatography-mass spectrometry-olfactometry. Separations 2017, 4, 24. [Google Scholar] [CrossRef]
- Rice, S.; Lutt, N.; Koziel, J.A.; Dharmadhikari, M.; Fennell, A. Determination of selected aromas in Marquette and Frontenac wine using headspace-SPME coupled with GC-MS and simultaneous olfactometry. Separations 2018, 5, 20. [Google Scholar] [CrossRef]
- Rice, S.; Tursumbayeva, M.; Clark, M.; Greenlee, D.; Dharmadhikari, M.; Fennell, A.; Koziel, J.A. Effects of harvest time on aroma of white wines made from cold-hardy Brianna and Frontenac gris grapes using headspace solid-phase microextraction and gas-chromatography-mass-spectrometry-olfactometry. Foods 2019, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Xu, J.; Ke, Y.; Huang, S.; Zeng, F.; Luan, T.; Ouyang, G. Applications of in vivo and in vitro solid-phase microextraction techniques in plant analysis: A review. Anal. Chim. Acta 2013, 794, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rice, S.; Maurer, D.L.; Fennell, A.; Dharmadhikari, M.; Koziel, J.A. Biogenic volatiles emitted from four cold-hardy grape cultivars during ripening. Data 2019, 4, 22. [Google Scholar] [CrossRef]
- Acree, T.; Arn, H. Flavornet and Human Odor Space. Available online: http://www.flavornet.org (accessed on 20 August 2018).
- The Good Scents Company Information System. Available online: http://www.thegoodscentscompany.com/ (accessed on 20 August 2018).
- United States Department of Agriculture, Agricultural Research Service. Available online: https://planthardiness.ars.usda.gov/PHZMWeb/ (accessed on 26 December 2018).
- National Oceanic and Atmospheric Administration, National Centers for Environmental Information. Available online: https://www.ncdc.noaa.gov (accessed on 19 December 2018).
- Chateau Stripmine. Available online: http://chateaustripmine.info/Parentage/Marquette.gif (accessed on 26 December 2018).
- Yiantzi, E.; Kalogerakis, N.; Psillakis, E. Design and testing of a new sampler for simplified vacuum-assisted headspace solid-phase microextraction. Anal. Chim. Acta 2016, 927, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Maicias, S.; Mateo, J.J. Hydrolysis of terpenyl glycosides in grape juice and other fruit juices: A review. Appl. Microbiol. Biotechnol. 2005, 67, 322–335. [Google Scholar] [CrossRef] [PubMed]
- Jackson, R.S. Wine Science Principles and Applications; Elsevier: Amsterdam, The Netherlands, 2008; ISBN 978-0-12-373646-8. [Google Scholar]
- Rapid determination of the aromatic compounds methyl-anthranilate, 2′-aminoacetophenone and furaneol by GC-MS: Method validation and characterization of grape derivatives. Food Res. Intl. 2018, 107, 613–618. [CrossRef] [PubMed]
- Risticevic, S.; Chen, Y.; Kudlejova, L.; Vatinno, R.; Baltensperger, B.; Stuff, J.R.; Hein, D.; Pawliszyn, J. Protocol for the development of automated high-throughput SPME–GC methods for the analysis of volatile and semivolatile constituents in wine samples. Nat. Protoc. 2010, 5, 162–176. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
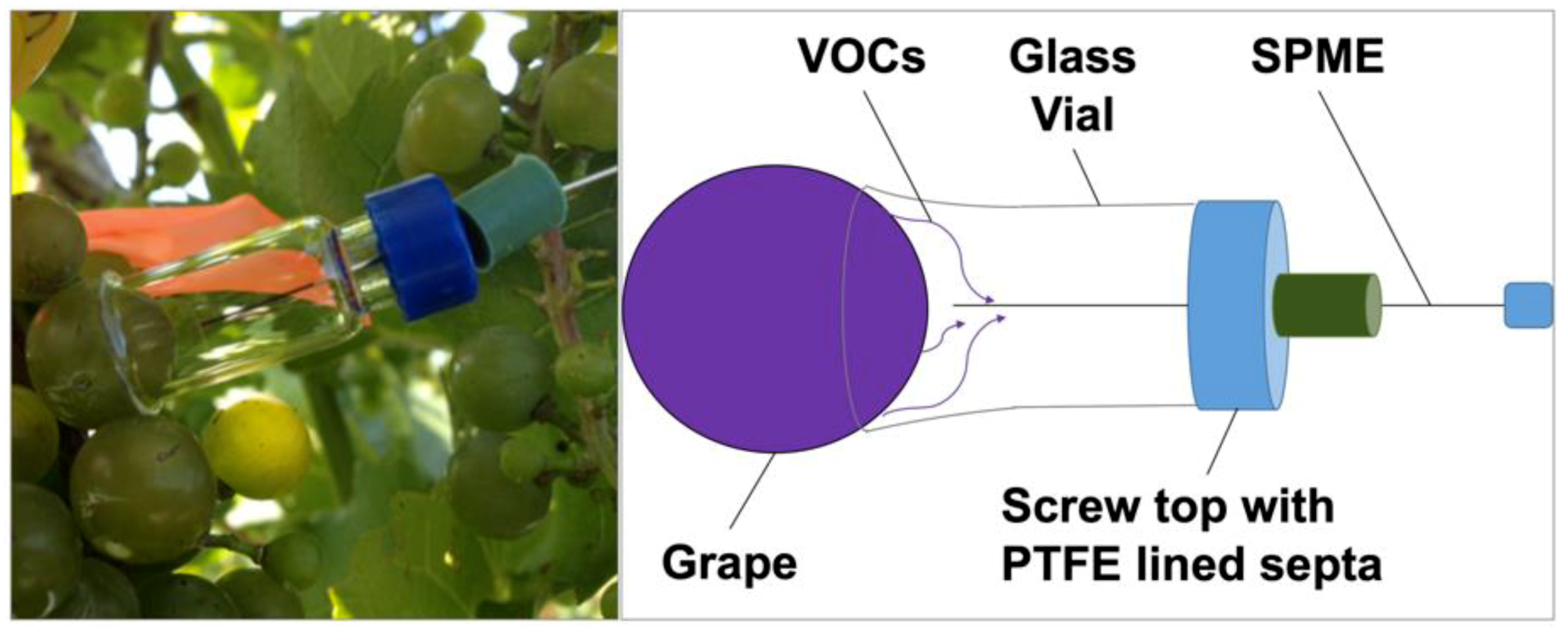
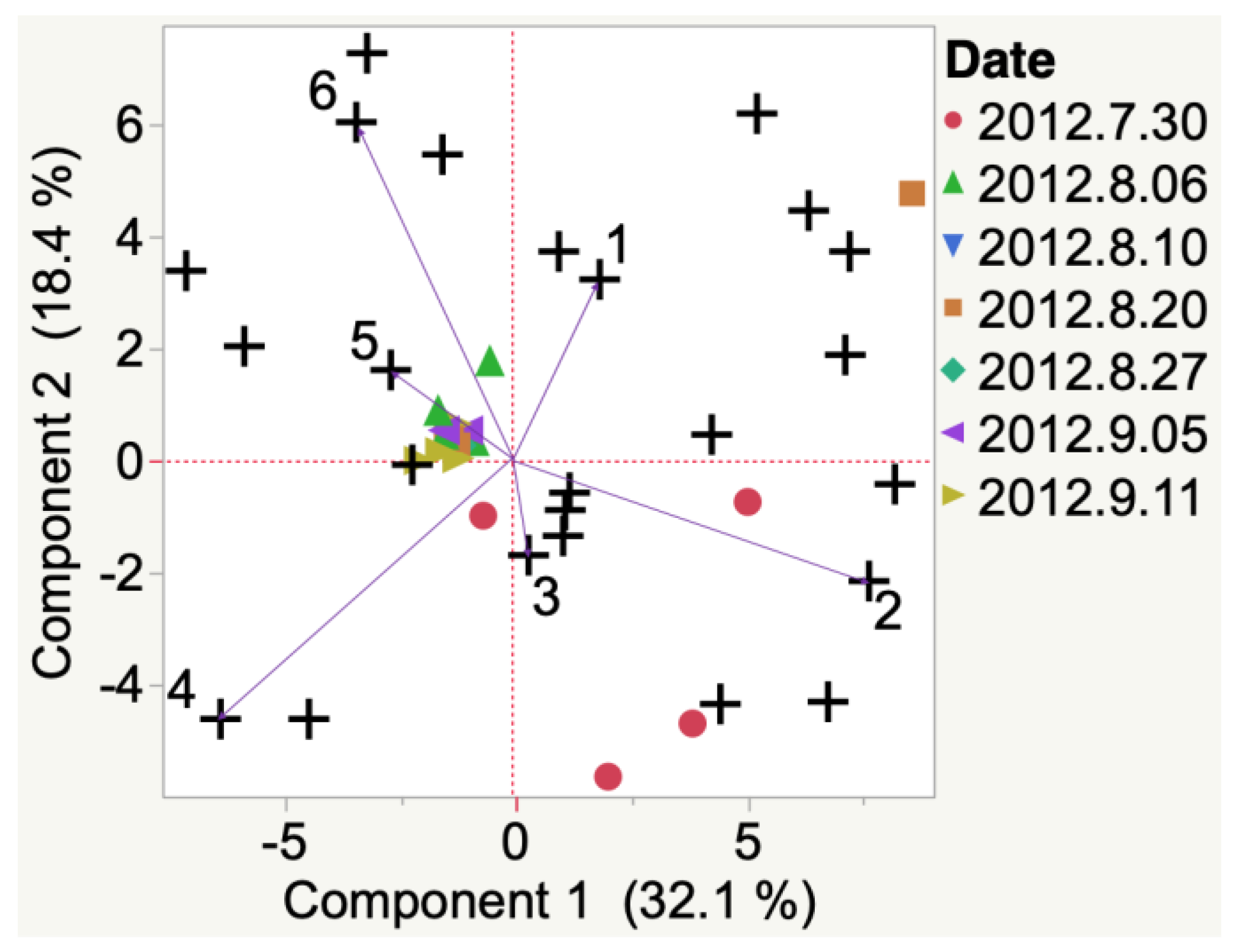
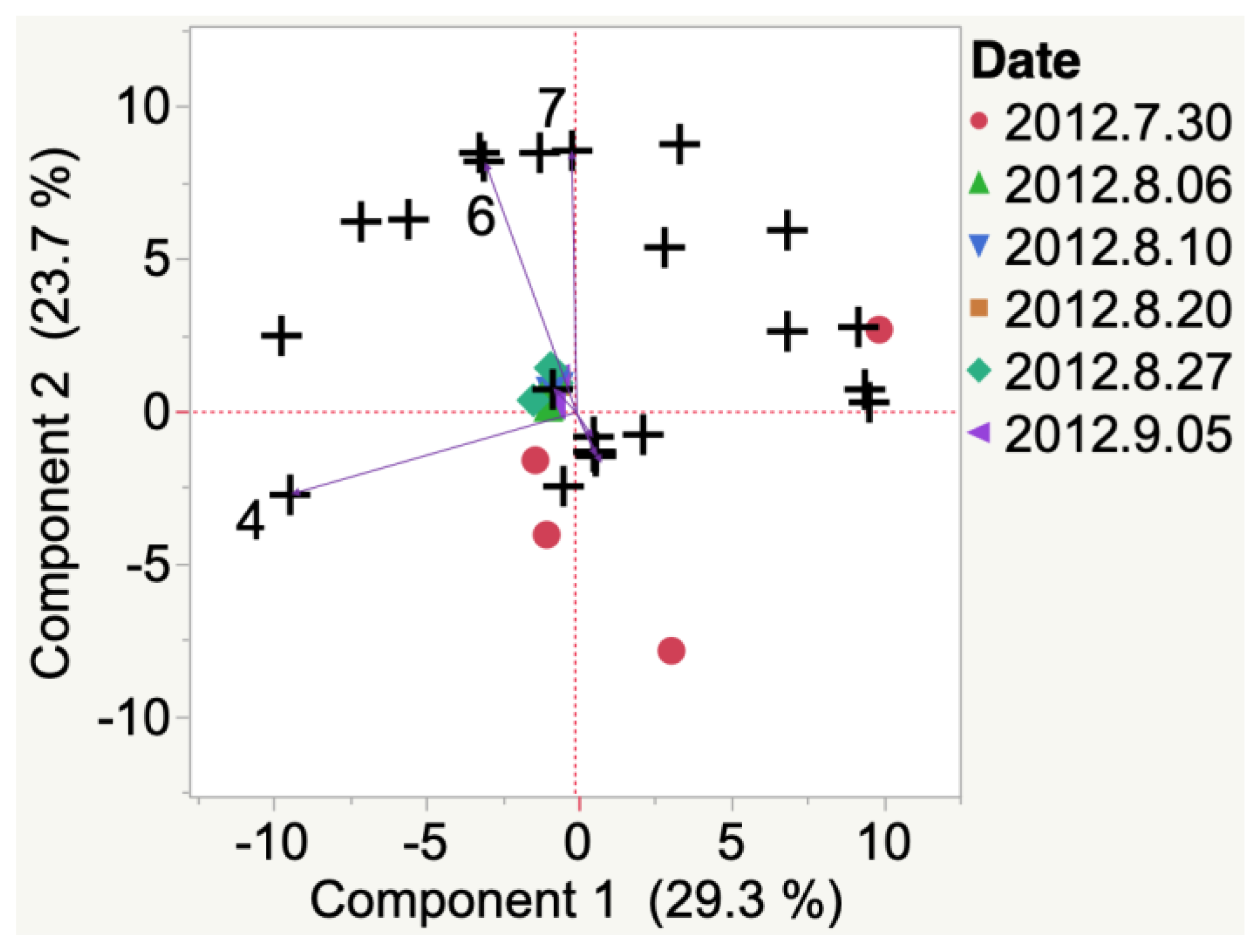
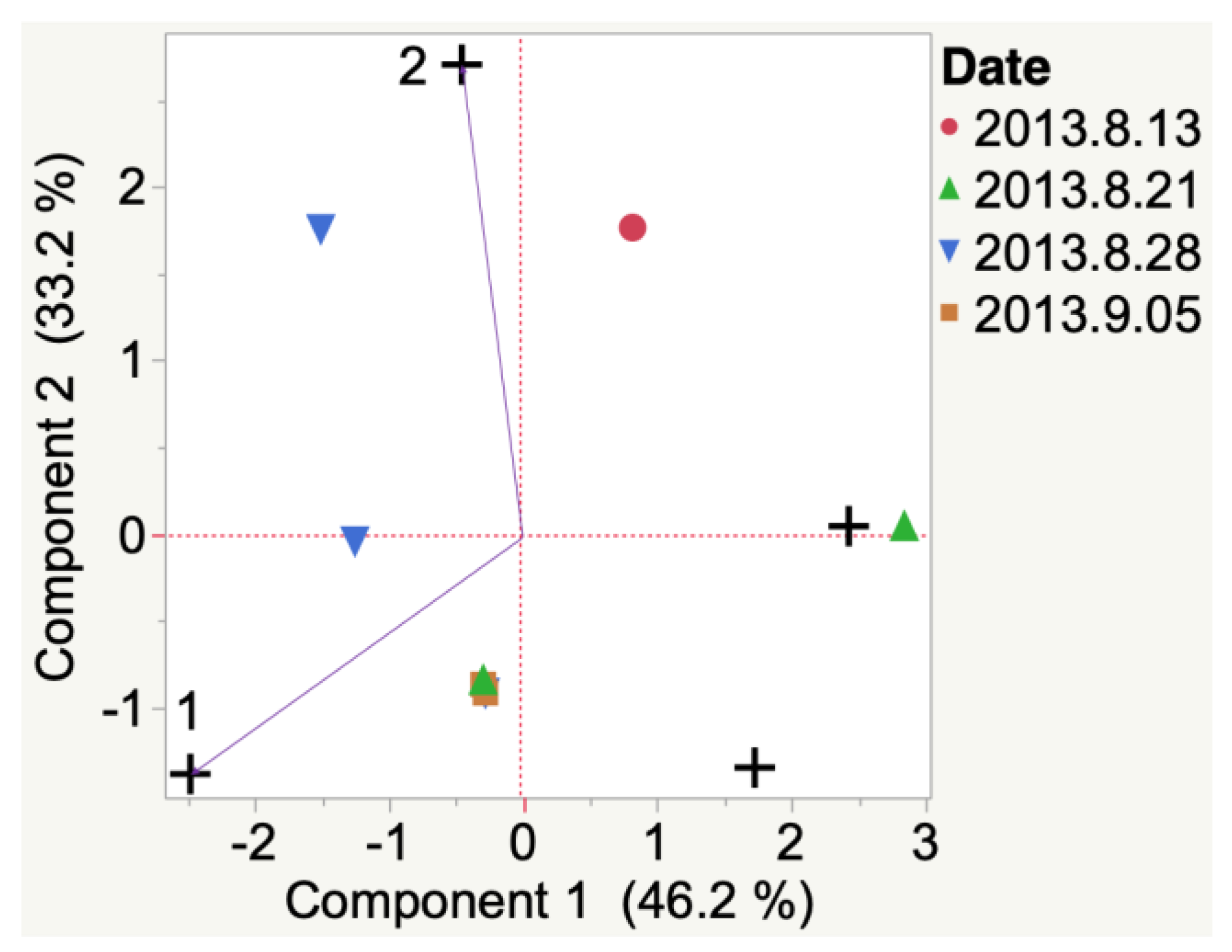
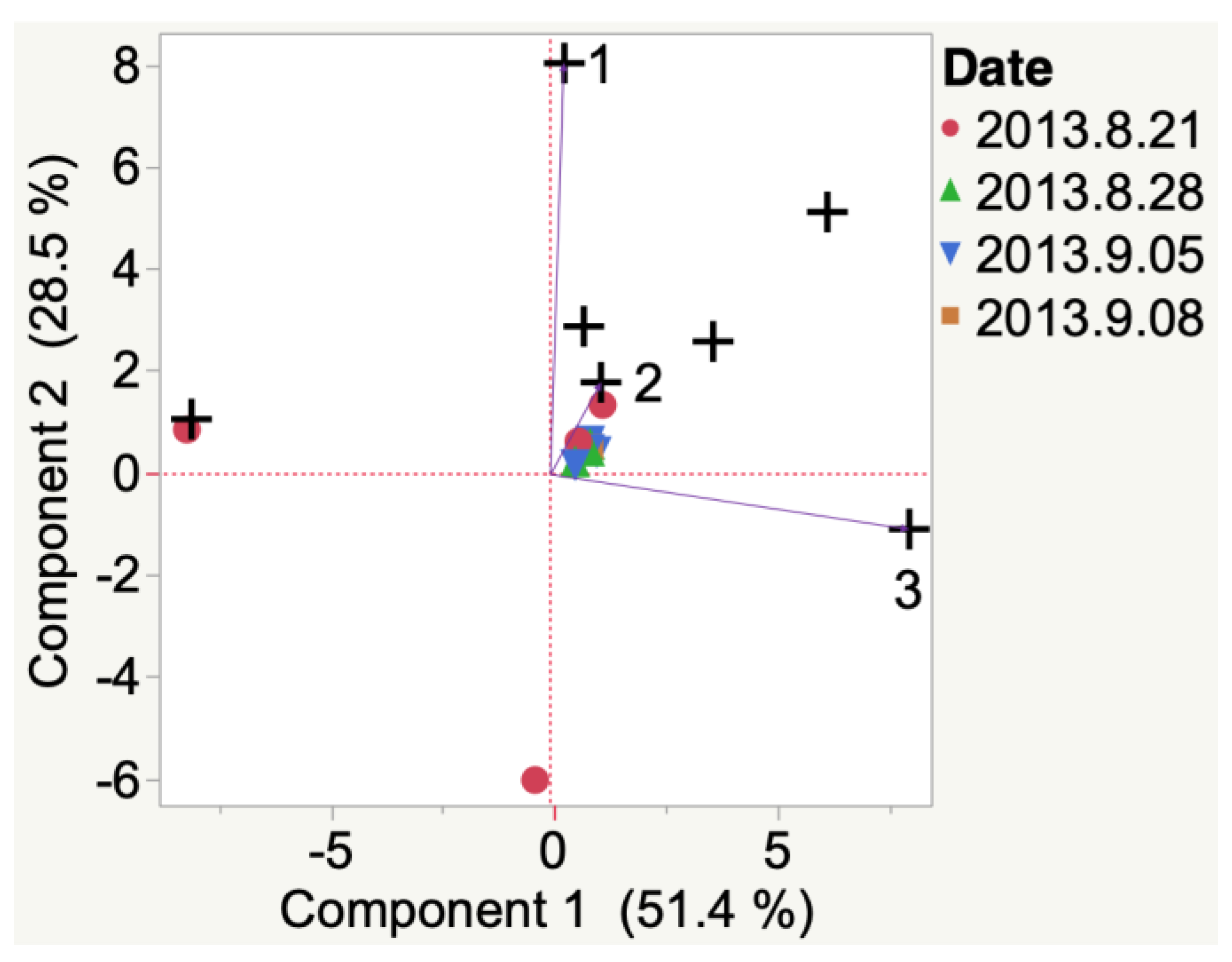
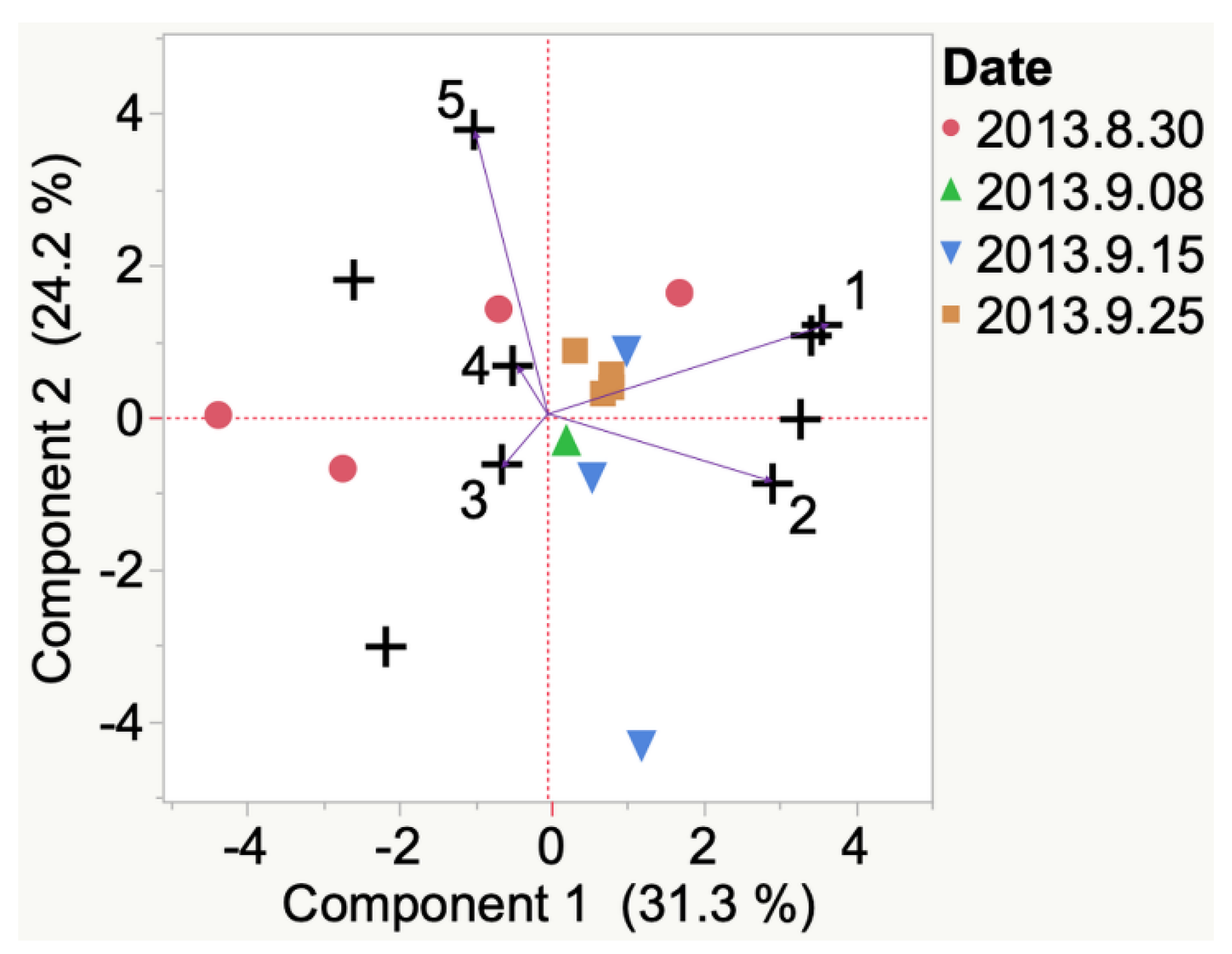
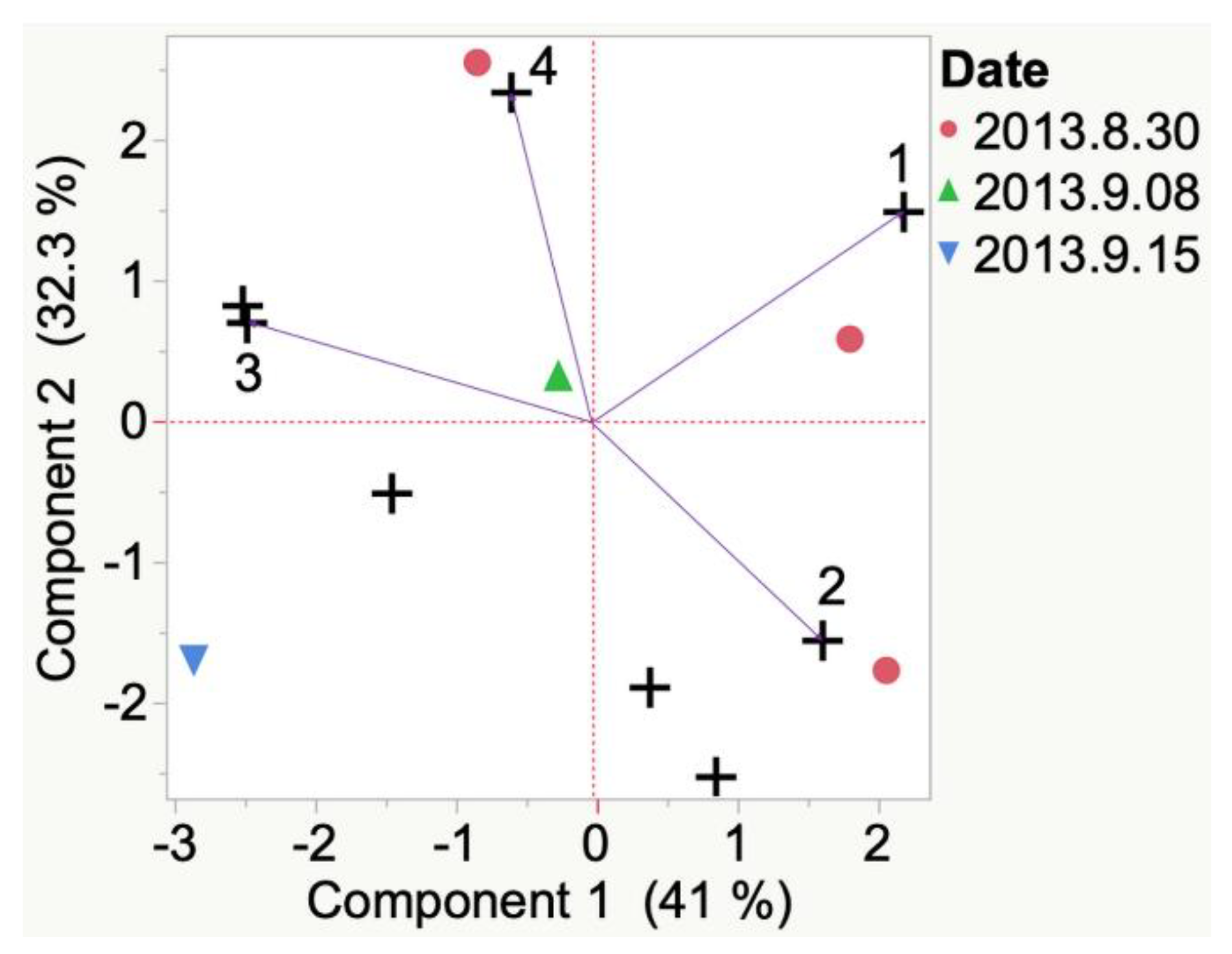
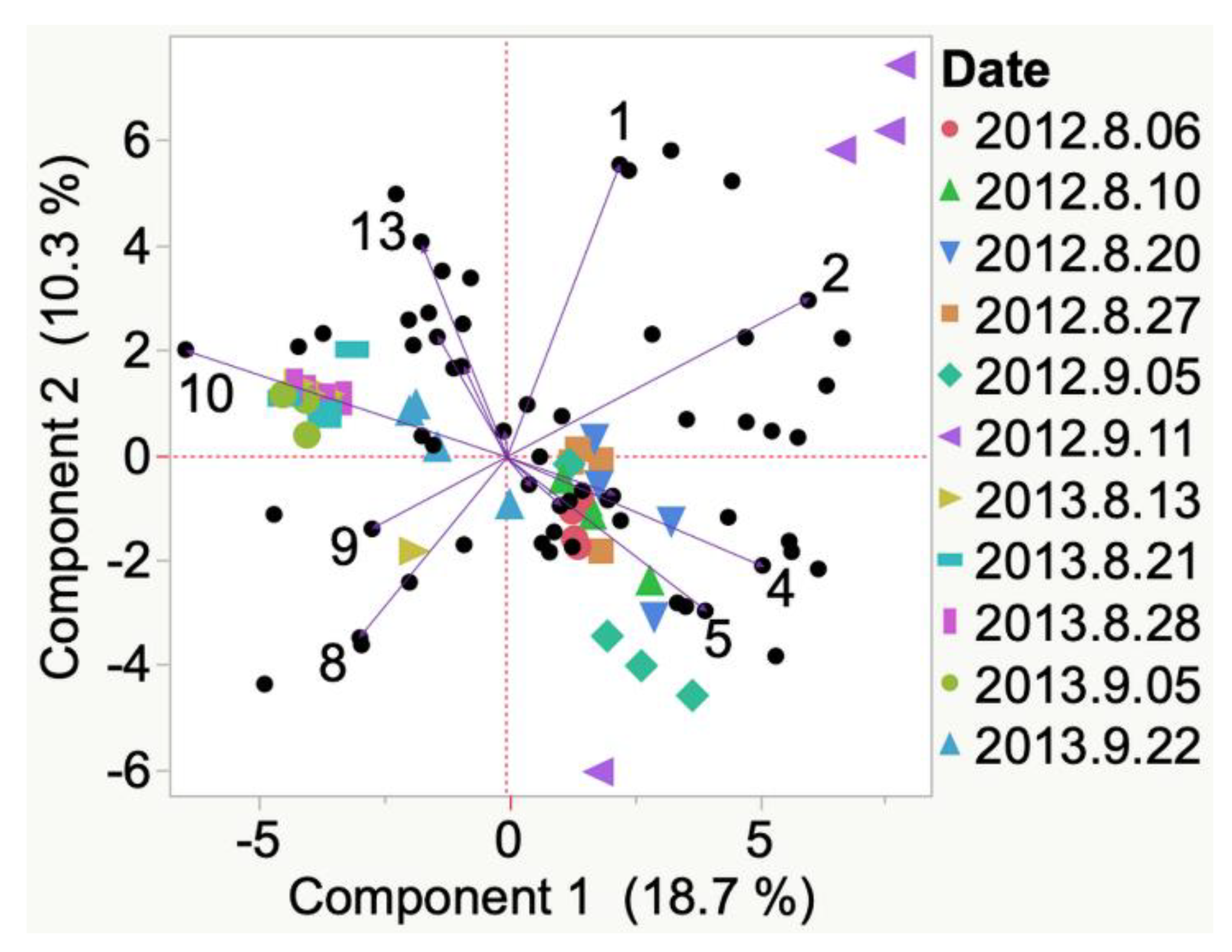
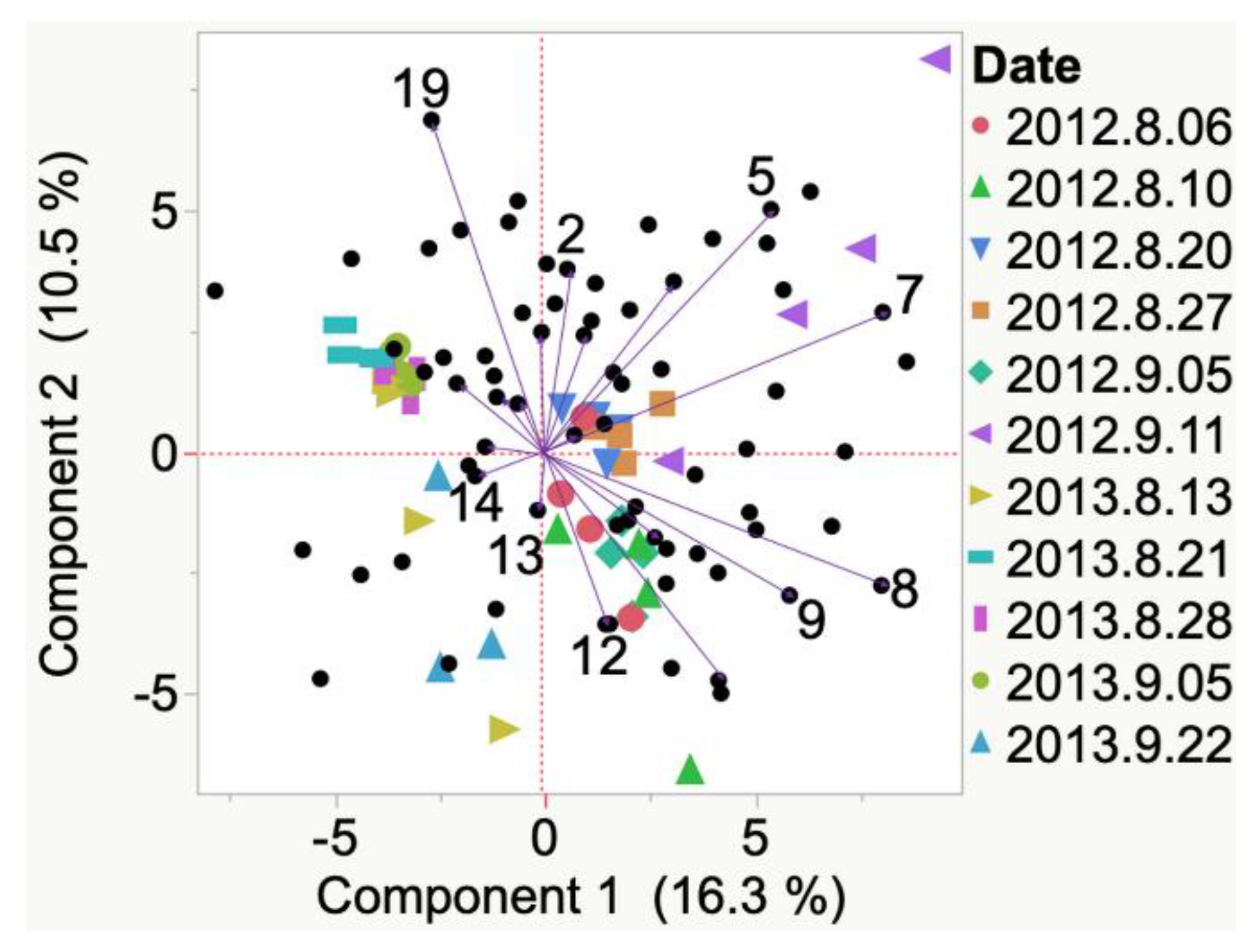
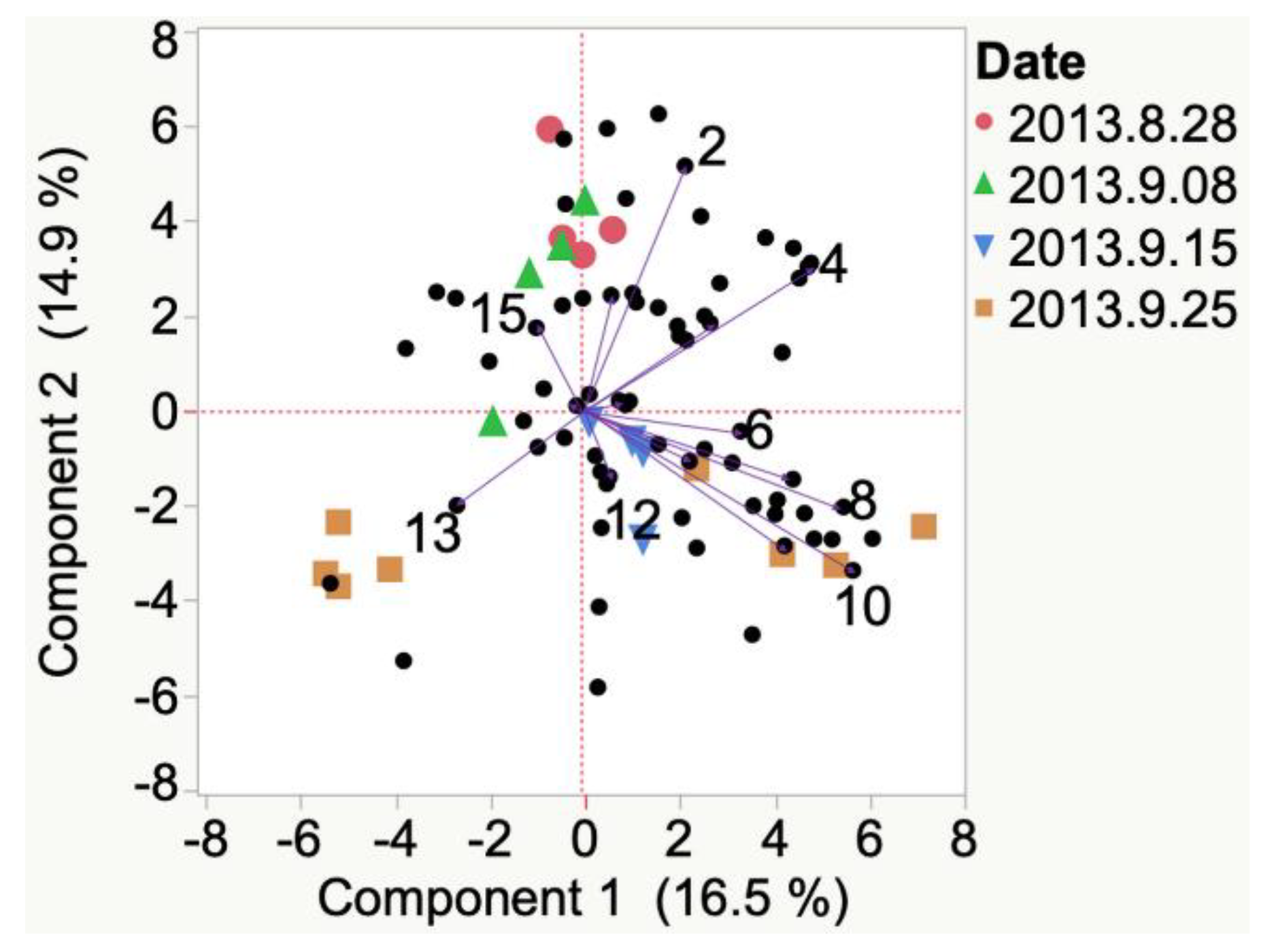

| Sample | Cluster | No. of Members 2 | Most Representative Variable 3 | Cluster Proportion of Variation Explained 4 | Total Proportion of Variation Explained 5 |
|---|---|---|---|---|---|
| IA Frontenac (0.709) 1 | 1 | 5 | Heptanal | 0.937 | 0.173 |
| 3 | 5 | 4-Methyl-3-penten-2-one | 0.745 | 0.138 | |
| 5 | 6 | Nonanal | 0.512 | 0.114 | |
| 2 | 3 | 3-Methyl-1-butanol | 0.845 | 0.094 | |
| 4 | 4 | 1,4-Butanolide | 0.576 | 0.085 | |
| 6 | 2 | 5-(Hydroxymethyl)-2-furancarboxaldehyde | 0.858 | 0.064 | |
| 7 | 2 | Benzophenone | 0.548 | 0.041 | |
| SD Frontenac (0.686) 1 | 1 | 7 | Toluene | 0.712 | 0.208 |
| 2 | 5 | Nonanal | 0.820 | 0.171 | |
| 5 | 3 | 3-Phenyl-2-propenal | 0.810 | 0.101 | |
| 3 | 4 | 4-Methyl-3-penten-2-one | 0.592 | 0.099 | |
| 4 | 3 | Acetic acid | 0.502 | 0.063 | |
| 6 | 2 | Benzyl alcohol | 0.536 | 0.045 | |
| IA Marquette (0.739) 1 | 1 | 10 | 1-Octanol | 0.805 | 0.310 |
| 2 | 5 | Acetaldehyde | 0.814 | 0.156 | |
| 3 | 3 | Methyl ethyl ketone | 0.616 | 0.071 | |
| 4 | 3 | 1-Hexadecanol | 0.505 | 0.063 | |
| 5 | 3 | Acetophenone | 0.536 | 0.062 | |
| 6 | 1 | Acetic acid | 1.000 | 0.038 | |
| 7 | 1 | 2-Ethyl-1-hexanol | 1.000 | 0.038 | |
| SD Marquette (0.783) 1 | 1 | 7 | Acetone | 0.817 | 0.249 |
| 2 | 4 | 4-Methyl-3-penten-2-one | 0.799 | 0.139 | |
| 6 | 4 | Decane | 0.797 | 0.139 | |
| 3 | 2 | 1-Pentanol | 1.000 | 0.087 | |
| 4 | 3 | 2-Ethyl-1-hexanol | 0.532 | 0.069 | |
| 7 | 2 | 1-Hexadecanol | 0.654 | 0.057 | |
| 5 | 1 | Indene | 1.000 | 0.043 |
| Sample | Cluster | No. of Members 2 | Most Representative Variable 3 | Cluster Proportion of Variation Explained 4 | Total Proportion of Variation Explained 5 |
|---|---|---|---|---|---|
| SD Frontenac (0.750) 1 | 1 | 2 | Palmitic acid | 0.830 | 0.415 |
| 2 | 2 | Acetic acid | 0.669 | 0.334 | |
| SD Marquette (0.870) 1 | 1 | 7 | 1,4-Butanolide | 0.878 | 0.473 |
| 2 | 4 | Ethyl octanoate | 0.883 | 0.272 | |
| 3 | 2 | 2-Ethyl-1-hexanol | 0.819 | 0.126 | |
| IA St. Croix (0.896) 1 | 1 | 4 | 3-Methyl indole | 1.000 | 0.500 |
| 2 | 3 | Acetic acid | 0.722 | 0.271 | |
| 3 | 1 | Benzyl alcohol | 1.000 | 0.125 | |
| SD St. Croix (0.855) 1 | 1 | 3 | Nonanal | 0.802 | 0.241 |
| 3 | 3 | Diacetone alcohol | 0.713 | 0.214 | |
| 2 | 2 | 1,4-Butanolide | 1.000 | 0.200 | |
| 4 | 1 | 5-(Hydroxymethyl)-2-furancarboxaldehyde | 1.000 | 0.100 | |
| 5 | 1 | Ethyl acetate | 1.000 | 0.100 | |
| IA La Crescent (0.909) 1 | 1 | 28 | 2-Ethyl-1-hexanol | 0.973 | 0.757 |
| 2 | 4 | 3-Methyl indole | 0.772 | 0.086 | |
| 4 | 2 | Ethanol | 0.636 | 0.035 | |
| 3 | 2 | 2-Phenylethanol | 0.556 | 0.031 | |
| SD La Crescent (0.936) 1 | 1 | 3 | 2-Phenylethanol | 1.000 | 0.300 |
| 3 | 3 | Diacetone alcohol | 0.853 | 0.256 | |
| 4 | 2 | Acetic acid | 0.976 | 0.195 | |
| 2 | 2 | 6-Methyl-5-hepten-2-one | 0.926 | 0.185 |
| Sample | Cluster | No. of Members B | Most Representative Variable C | Cluster Proportion of Variation Explained D | Total Proportion of Variation Explained E |
|---|---|---|---|---|---|
| IA Frontenac (0.803) A | 1 | 7 | 3-Methyl-1-butanol | 0.993 | 0.257 |
| 2 | 9 | Cyclohexanol | 0.770 | 0.257 | |
| 4 | 4 | Isoamyl acetate | 0.745 | 0.110 | |
| 3 | 4 | Isovaleraldehyde | 0.622 | 0.092 | |
| 5 | 3 | Toluene | 0.774 | 0.086 | |
| SD Frontenac (0.627) A | 2 | 8 | Styrene | 0.636 | 0.083 |
| 10 | 7 | Acetaldehyde | 0.546 | 0.063 | |
| 5 | 6 | 2-Octanone | 0.608 | 0.060 | |
| 1 | 6 | Acetone | 0.602 | 0.059 | |
| 6 | 5 | 1-Hexanol | 0.645 | 0.053 | |
| 4 | 5 | Nonane | 0.624 | 0.051 | |
| 8 | 4 | Ethyl hexanoate | 0.732 | 0.048 | |
| 3 | 4 | Ethyl palmitate | 0.694 | 0.045 | |
| 7 | 3 | Hexanoic acid | 0.740 | 0.036 | |
| 9 | 4 | N-benzyl-2-phenethylamine | 0.509 | 0.033 | |
| 12 | 2 | 2-Methyl-1-propanol | 0.953 | 0.031 | |
| 11 | 3 | Benzoic acid, methyl ester | 0.560 | 0.028 | |
| 14 | 2 | Isophorone | 0.569 | 0.019 | |
| 13 | 2 | Octanal | 0.524 | 0.017 | |
| IA Marquette (0.863) A | 1 | 9 | Hexanal | 0.925 | 0.347 |
| 3 | 5 | Isoamyl acetate | 0.878 | 0.183 | |
| 2 | 4 | Styrene | 0.776 | 0.129 | |
| 5 | 2 | Ethanol | 0.933 | 0.078 | |
| 6 | 2 | Benzophenone | 0.813 | 0.068 | |
| 4 | 2 | Allyl alcohol | 0.703 | 0.059 | |
| SD Marquette (0.654) A | 7 | 7 | Acetaldehyde | 0.617 | 0.062 |
| 6 | 6 | Methyl ethyl ketone | 0.639 | 0.055 | |
| 3 | 5 | Decane | 0.760 | 0.054 | |
| 19 | 5 | Nonanal | 0.667 | 0.048 | |
| 1 | 4 | Styrene | 0.777 | 0.044 | |
| 5 | 5 | Amyl acetate | 0.621 | 0.044 | |
| 4 | 4 | (E)-2-Hexenoic acid | 0.704 | 0.040 | |
| 10 | 4 | Cyclohexanol | 0.692 | 0.040 | |
| 9 | 5 | Octanal | 0.480 | 0.034 | |
| 2 | 3 | 1-Pentanol | 0.673 | 0.029 | |
| 8 | 2 | Nonane | 0.966 | 0.028 | |
| 18 | 3 | Valeraldehyde | 0.630 | 0.027 | |
| 11 | 3 | 1-Heptanol | 0.629 | 0.027 | |
| 14 | 4 | beta-Damascenone | 0.470 | 0.027 | |
| 12 | 4 | Allyl alcohol | 0.435 | 0.025 | |
| 13 | 2 | p-Cymene | 0.835 | 0.024 | |
| 16 | 2 | Methyl disulfide | 0.635 | 0.018 | |
| 15 | 1 | beta-Cyclocitral | 1.000 | 0.014 | |
| 17 | 1 | Nerol acetate | 1.000 | 0.014 | |
| IA St. Croix (0.772) A | 1 | 9 | Formic acid, octyl ester | 0.832 | 0.150 |
| 4 | 8 | Ethyl decanoate | 0.901 | 0.144 | |
| 2 | 7 | Isobutyraldehyde | 0.674 | 0.094 | |
| 3 | 5 | Aspirin methyl ester | 0.813 | 0.081 | |
| 5 | 3 | Benzeneacetaldehyde | 0.858 | 0.052 | |
| 10 | 3 | Ethanol | 0.771 | 0.046 | |
| 8 | 3 | Methacrolein | 0.682 | 0.041 | |
| 12 | 2 | Isoamyl acetate | 0.841 | 0.034 | |
| 6 | 2 | 1-Butanol | 0.790 | 0.032 | |
| 7 | 3 | Ethyl butyrate | 0.493 | 0.030 | |
| 9 | 2 | 1-Hexanol | 0.649 | 0.026 | |
| 11 | 2 | beta-Damascenone | 0.576 | 0.023 | |
| 13 | 1 | Valeraldehyde | 1.000 | 0.020 | |
| SD St. Croix (0.692) A | 2 | 8 | Acetophenone | 0.685 | 0.081 |
| 3 | 6 | Linalool | 0.786 | 0.069 | |
| 6 | 6 | Benzaldehyde | 0.727 | 0.064 | |
| 7 | 6 | Methyl salicylate | 0.681 | 0.060 | |
| 5 | 6 | Cyclohexanol | 0.662 | 0.058 | |
| 4 | 6 | 2-Heptanone | 0.627 | 0.055 | |
| 1 | 5 | 2-Phenylethanol | 0.728 | 0.054 | |
| 10 | 5 | 1-Pentanol | 0.581 | 0.043 | |
| 8 | 5 | Benzyl alcohol | 0.557 | 0.041 | |
| 11 | 3 | Safrol | 0.855 | 0.038 | |
| 9 | 3 | Benzoic acid, methyl ester | 0.797 | 0.035 | |
| 12 | 3 | Ethyl acetate | 0.622 | 0.027 | |
| 14 | 2 | Aspirin methyl ester | 0.840 | 0.025 | |
| 13 | 2 | Propionaldehyde | 0.797 | 0.023 | |
| 15 | 2 | N-Benzyl-2-phenethylamine | 0.628 | 0.018 | |
| IA La Crescent (0.699) A | 1 | 11 | beta-Cyclocitral | 0.682 | 0.121 |
| 2 | 8 | beta-Pinene | 0.836 | 0.108 | |
| 3 | 9 | Ethyl butyrate | 0.710 | 0.103 | |
| 8 | 4 | p-Cymene | 0.663 | 0.043 | |
| 9 | 4 | Propanoic acid | 0.648 | 0.042 | |
| 12 | 3 | 1-Hexanol | 0.792 | 0.038 | |
| 6 | 3 | Nerol acetate | 0.776 | 0.038 | |
| 4 | 4 | Methacrolein | 0.565 | 0.036 | |
| 5 | 4 | Beta-damascenone | 0.521 | 0.034 | |
| 7 | 3 | (+)-4-Carene | 0.650 | 0.031 | |
| 11 | 3 | Valeric acid | 0.641 | 0.031 | |
| 13 | 3 | 3-Methyl-1-butanol | 0.638 | 0.031 | |
| 10 | 2 | Acetic acid | 0.845 | 0.027 | |
| 14 | 1 | Propyl-benzene | 1.000 | 0.016 | |
| SD La Crescent (0.741) A | 3 | 8 | Allyl alcohol | 0.845 | 0.086 |
| 2 | 8 | beta-Pinene | 0.837 | 0.085 | |
| 1 | 7 | Toluene | 0.691 | 0.061 | |
| 11 | 5 | Isoamyl acetate | 0.915 | 0.058 | |
| 6 | 6 | Isophorone | 0.637 | 0.048 | |
| 7 | 6 | Ethyl butyrate | 0.567 | 0.043 | |
| 8 | 5 | Hexanal | 0.669 | 0.042 | |
| 4 | 5 | Benzaldehyde | 0.657 | 0.042 | |
| 13 | 5 | Styrene | 0.618 | 0.039 | |
| 15 | 4 | Carbon disulfide | 0.771 | 0.039 | |
| 9 | 3 | Ethyl vinyl ketone | 1.000 | 0.038 | |
| 5 | 3 | Camphene | 0.900 | 0.034 | |
| 18 | 3 | Linalyl acetate | 0.806 | 0.031 | |
| 17 | 3 | Geraniol | 0.730 | 0.028 | |
| 10 | 2 | Furfural | 0.908 | 0.023 | |
| 12 | 3 | Isobutyraldehyde | 0.499 | 0.019 | |
| 16 | 2 | 2-Ethyl-1-hexanol | 0.500 | 0.013 | |
| 14 | 1 | Propyl-benzene | 1.000 | 0.013 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rice, S.; Maurer, D.L.; Fennell, A.; Dharmadhikari, M.; Koziel, J.A. Evaluation of Volatile Metabolites Emitted In-Vivo from Cold-Hardy Grapes during Ripening Using SPME and GC-MS: A Proof-of-Concept. Molecules 2019, 24, 536. https://doi.org/10.3390/molecules24030536
Rice S, Maurer DL, Fennell A, Dharmadhikari M, Koziel JA. Evaluation of Volatile Metabolites Emitted In-Vivo from Cold-Hardy Grapes during Ripening Using SPME and GC-MS: A Proof-of-Concept. Molecules. 2019; 24(3):536. https://doi.org/10.3390/molecules24030536
Chicago/Turabian StyleRice, Somchai, Devin L. Maurer, Anne Fennell, Murlidhar Dharmadhikari, and Jacek A. Koziel. 2019. "Evaluation of Volatile Metabolites Emitted In-Vivo from Cold-Hardy Grapes during Ripening Using SPME and GC-MS: A Proof-of-Concept" Molecules 24, no. 3: 536. https://doi.org/10.3390/molecules24030536
APA StyleRice, S., Maurer, D. L., Fennell, A., Dharmadhikari, M., & Koziel, J. A. (2019). Evaluation of Volatile Metabolites Emitted In-Vivo from Cold-Hardy Grapes during Ripening Using SPME and GC-MS: A Proof-of-Concept. Molecules, 24(3), 536. https://doi.org/10.3390/molecules24030536