Purification and Product Characterization of Lipoxygenase from Opium Poppy Cultures (Papaver somniferum L.)
Abstract
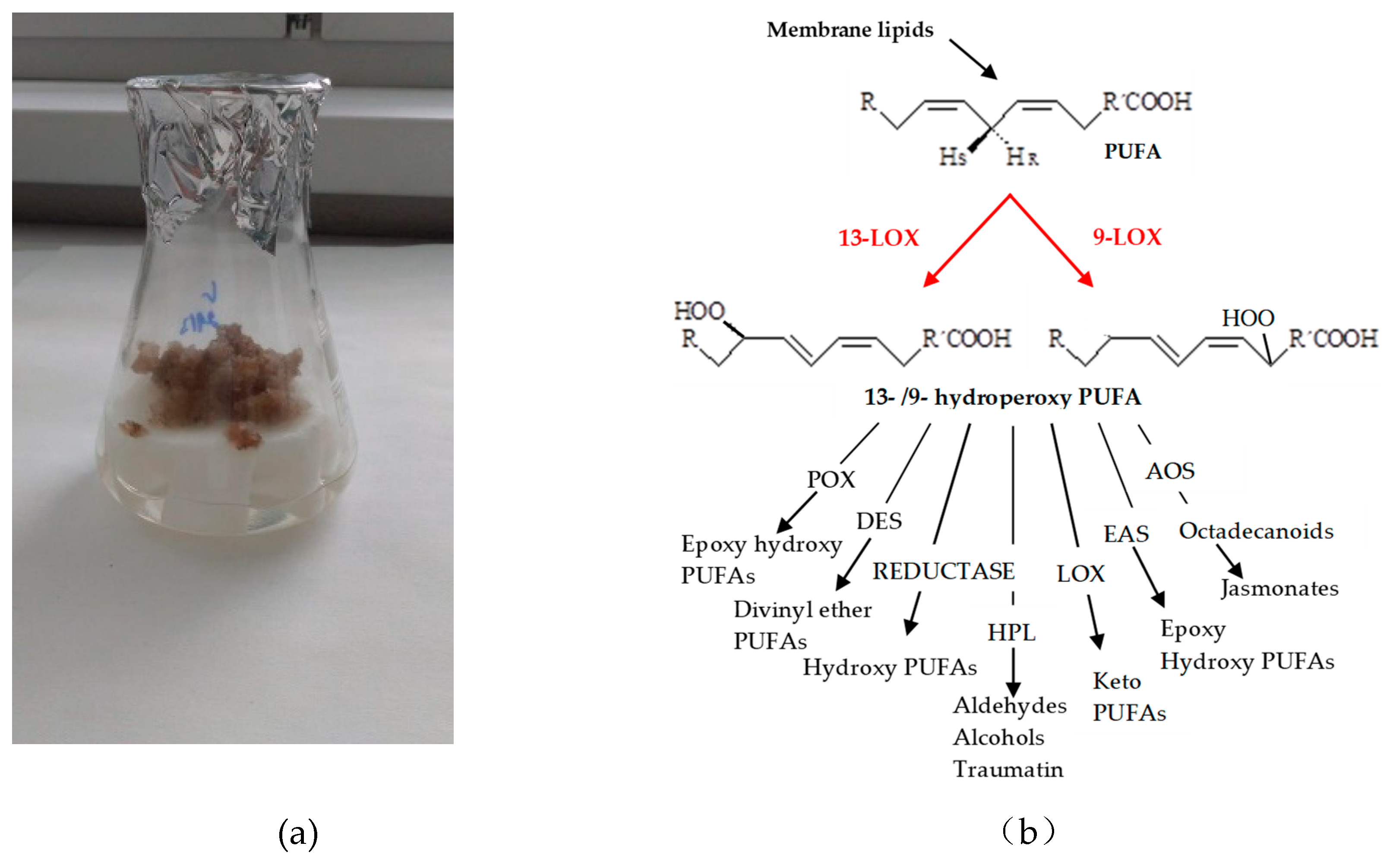
1. Introduction
2. Results and Discussion
2.1. Purification and Characterization of LOX from Opium Poppy Cultures
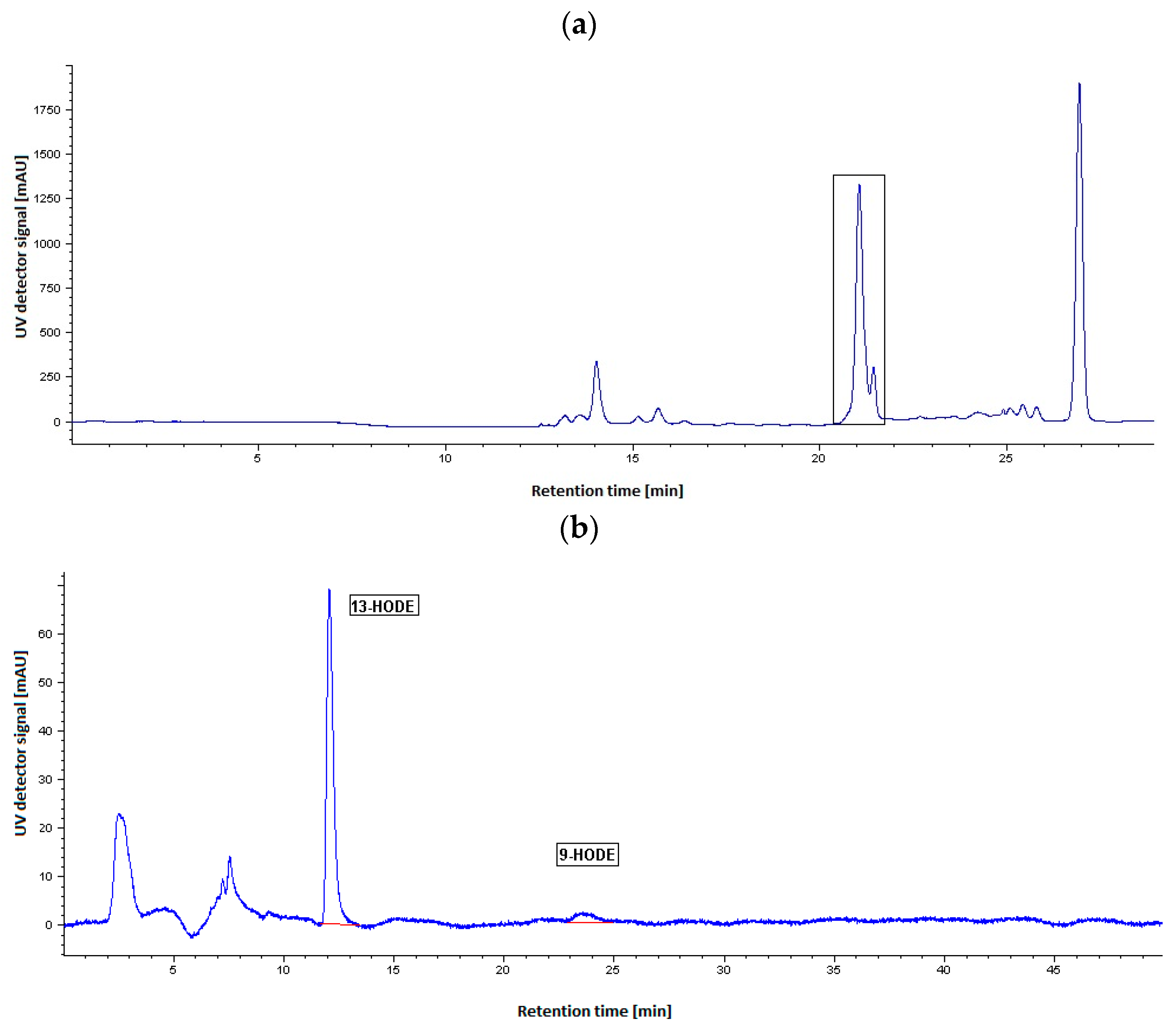
2.2. HPLC Analysis of LOX Reaction Products
3. Materials and Methods
3.1. Plant Material
3.2. Enzyme Purification
3.3. Measurement of LOX Activity and Protein Determination
3.4. SDS-PAGE and Immunoblotting
3.5. HPLC Analysis of LOX Reaction Products
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Beaudoin, G.A.W.; Facchini, P.J. Benzylisoquinoline alkaloid biosynthesis in opium poppy. Planta 2014, 240, 19–32. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudian, M.; Rahimi-Moghaddam, P. The anti-cancer activity of noscapine: A review. Recent Pat. Anticancer Drug Discov. 2009, 4, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Sharma, B. Toxicological effects of berberine and sanguinarine. Front. Mol. Biosci. 2018, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Oliw, E.H. Plant and fungal lipoxygenases. Prostag. Oth. Lipid Mediators 2002, 68–69, 313–323. [Google Scholar] [CrossRef]
- Andreou, A.Z.; Vanko, M.; Bezakova, L.; Feussner, I. Properties of a mini 9R-lipoxygenase from Nostoc sp. PCC 7120 and its mutant forms. Phytochemistry 2008, 69, 1832–1837. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Boeglin, W.E.; Schneider, C.; Brash, A.R. A 49-kDa mini-lipoxygenase from Anabaena sp. PCC 7120 retains catalytically complete functionality. J. Biol. Chem. 2008, 283, 5138–5147. [Google Scholar] [CrossRef]
- Hansen, J.; Garreta, A.; Benincasa, M.; Fusté, M.C.; Busquets, M.; Manresa, A. Bacterial lipoxygenases, a new subfamily of enzymes? A phylogenetic approach. Appl. Microbiol. Biotechnol. 2013, 97, 4737–4747. [Google Scholar] [CrossRef]
- Dijan, B.; Hornung, E.; Ischebeck, T.; Feussner, I. The green microalga Lobosphaera incisa harbours an arachidonate 15S-lipoxygenase. Plant. Biology 2018, 21, 131–142. [Google Scholar] [CrossRef]
- Feussner, I.; Wasternack, C. The lipoxygenase pathway. Annu. Rev. Plant. Biol. 2002, 53, 275–297. [Google Scholar] [CrossRef]
- Liavonchanka, A.; Feussner, I. Lipoxygenases: Occurrence, functions and catalysis. J. Plant. Physiol. 2006, 163, 348–357. [Google Scholar] [CrossRef]
- Kuhn, H.; Banthiya, S.; van Leyen, K. Mammalian lipoxygenases and their biological relevance. Biochim. Biophys. Acta 2015, 1851, 308–330. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.S.; Choi, E.; Kim, Y.; Cho, K.; Lee, A.; Shim, J.; Rakwal, L.; Agrawal, G.K.; Han, O. Dual positional specificity and expression of non-traditional lipoxygenase induced by wounding and methyl jasmonate in maize seedlings. Plant. Mol. Biol. 2003, 52, 1203–1213. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Shen, W.; Liu, L.; Jiang, L.; Liu, Y.; Su, N.; Wan, J. A novel lipoxygenase gene from developing rice seeds confers dual position specificity and responds to wounding and insect attack. Plant. Mol. Biol. 2008, 66, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Han, Y.; Woo, J.C.; Baudisch, B.; Klösgen, R.B.; Oh, S.; Han, J.; Han, O. Cellular localization of dual positional specific maize lipoxygenase-1 in transgenic rice and calcium-mediated membrane association. Plant. Sci. 2011, 181, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Babenko, L.M.; Shcherbatiuk, M.M.; Skaterna, T.D.; Kosakivska, I.V. Lipoxygenases and their metabolites in formation of plant stress tolerance. Ukr. Biochem. J. 2017, 89, 5–20. [Google Scholar] [CrossRef] [PubMed]
- Mosblech, A.; Feussner, I.; Heilmann, I. Oxylipins: Structurally diverse metabolites from fatty acid oxidation. Plant. Physiol. Biochem. 2009, 47, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Blee, E. Impact of phyto-oxylipins in plant defense. Trends Plant. Sci. 2002, 7, 315–322. [Google Scholar] [CrossRef]
- Matsui, K. Green leaf volatiles: Hydroperoxide lyase pathway of oxylipin metabolism. Curr. Opin. Plant. Biol. 2006, 9, 274–280. [Google Scholar] [CrossRef]
- Wasternack, C. Jasmonates: An update on biosynthesis, signal transduction and action in plant stress response, growth and development. Ann. Bot. 2007, 100, 681–697. [Google Scholar] [CrossRef]
- Huang, H.; Liu, B.; Liu, L.; Song, S. Jasmonate action in plant growth and development. J. Exp. Bot. 2017, 68, 1349–1359. [Google Scholar] [CrossRef]
- Losvik, A.; Beste, L.; Glinwood, R.; Ivarson, E.; Stephens, J.; Zhu, L.-H.; Jonsson, L. Overexpression and Down-Regulation of Barley Lipoxygenase LOX2.2 Affects Jasmonate-Regulated Genes and Aphid Fecundity. Int. J. Mol. Sci 2017, 18, 2765. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Triptahi, V.; Singh, S.; Phukan, U.J.; Gupta, M.M.; Shanker, K.; Shukla, R.K. Wound induced tanscriptional regulation of benzylisoquinoline pathway and characterization of wound inducible PsWRKY transcription factor from Papaver somniferum. PLoS ONE 2013, 8, e52784. [Google Scholar] [CrossRef] [PubMed]
- Jablonická, V.; Ziegler, J.; Vatehová, Z.; Lišková, D.; Heilmann, I.; Obložinský, M.; Heilmann, M. Inhibition of phospholipases influences the metabolism of wound-induced benzylisoquinoline alkaloids in Papaver somniferum L. J. Plant. Physiol. 2018, 233, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Holková, I.; Bezáková, L.; Bilka, F.; Balažová, A.; Vanko, M.; Blanáriková, V. Involvement of lipoxygenase in elicitor-stimulated sanguinarine accumulation in Papaver somniferum suspension cultures. Plant. Physiol. Biochem. 2010, 48, 887–892. [Google Scholar] [CrossRef]
- Kollárová, R.; Obložinský, M.; Kováčiková, V.; Holková, I.; Balažová, A.; Pekárová, M.; Hoffman, P.; Bezáková, L. Lipoxygenase activity and sanguinarine production in cell suspension cultures of California poppy (Eschscholtzia californica CHAM.). Pharmazie 2014, 69, 637–640. [Google Scholar] [CrossRef] [PubMed]
- Yue, W.; Ming, Q.L.; Lin, B.; Rahman, K.; Zheng, C.J.; Han, T.; Qin, L.P. Medicinal plant cell suspension cultures: Pharmaceutical applications and high-yielding strategies for the desired secondary metabolites. Crit. Rev. Biotechnol. 2016, 36, 215–232. [Google Scholar] [CrossRef] [PubMed]
- Holková, I.; Bilka, F.; Rauová, D.; Bezáková, L. Purification and properties of lipoxygenase from opium poopy seedlings (Papaver somniferum L.). Turk. J. Biol. 2016, 40, 772–780. [Google Scholar] [CrossRef]
- Kollárová, R.; Holková, I.; Rauová, D.; Bálintová, B.; Mikuš, P.; Obložinský, M. HPLC analysis and biochemical characterization of LOX from Eschscholtzia californica Cham. Molecules 2017, 22, 1899. [Google Scholar] [CrossRef]
- Vanko, M.; Rauová, D.; Bezáková, L.; Holková, I.; Bilka, F.; Cupáková, M. Biochemical properties of lipoxygenase from opium poppy chloroplasts. Biol Plant. 2012, 56, 105–110. [Google Scholar] [CrossRef]
- Aanangi, R.; Kotapati, K.V.; Palaka, B.K.; Kedam, T.; Kanika, N.D.; Ampasala, D.R. Purification and characterization of lipoxygenase from mung bean (Vigna radiata L.) germinating seedlings. 3 Biotech. 2016, 113, 1–8. [Google Scholar] [CrossRef]
- Sharma, B.; Chugh, L.K. Two isoforms of lipoxygenase from mature grains of pearl millet [Pennisetum glaucum (L.) R. Br.]: Purification and physico-chemico-kinetic characterization. J. Food Sci. Technol. 2017, 54, 1577–1584. [Google Scholar] [CrossRef] [PubMed]
- Lorenzi, V.; Maury, J.; Casanova, J.; Berti, L. Purification, product characterization and kinetic properties of lipoxygenase from olive fruit (Olea europaea L.). Plant. Physiol. Biochem. 2006, 44, 450–454. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.; Salas, J.J.; Sanchez, J.; Harwood, J.L. Lipoxygenase pathway in olive callus cultures (Olea europaea). Phytochemistry 2000, 53, 13–19. [Google Scholar] [CrossRef]
- Williams, M.; Harwood, J.L. Characterisation of lipoxygenase isoforms from olive callus cultures. Phytochemistry 2008, 69, 2532–2538. [Google Scholar] [CrossRef]
- Sicilia, F.; Mattei, B.; Cervone, F.; Bellincampi, D.; De Lorenzo, G. Characterization of a membrane-associated apoplastic lipoxygenase in Phaseolus vulgaris L. Biochim. Biophys. Acta 2005, 1748, 9–19. [Google Scholar] [CrossRef]
- Kuo, J.M.; Hwang, A.; Yeh, D.B.; Pan, M.H.; Tsai, M.L.; Pan, B.S. Lipoxygenase from banana leaf: Purification and characterization of an enzyme that catalyzes linoleic acid oxygenation at the 9-position. J. Agric Food Chem 2006, 54, 3151–3156. [Google Scholar] [CrossRef] [PubMed]
- Schiller, D.; Contreras, C.; Vogt, J.; Dunemann, F.; Defilippi, B.G.; Beaudry, R.; Schwab, W. A dual positional specific lipoxygenase functions in the generation of flavor compounds during climacteric ripening of apple. Hortic. Res. 2015, 2, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Wang, X.; Guo, L.; Xu, Q.; Zhao, S.; Li, F.; Yan, X.; Liu, S.; Wei, C. Characterization and alternative splicing profiles of lipoxygenase gene family in tea plant (Camellia sinensis). Plant. Cell Physiol. 2018, 59, 1765–1781. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.O.; Whitaker, J.R. Purification and characterization of a lipoxygenase from immature English peas. J. Agric. Food Chem. 1986, 34, 203–211. [Google Scholar] [CrossRef]
- Bachmann, A.; Hause, B.; Maucher, H.; Garbe, E.; Vörös, K.; Weichert, H.; Wasternack, C.; Feussner, I. Jasmonate-induced lipid peroxidation in barley leaves initiated by distinct 13-LOX forms of chloroplasts. Biol. Chem. 2002, 383, 1645–1657. [Google Scholar] [CrossRef]
- Feussner, I.; Kühn, H.; Wasternack, C. Lipoxygenase-dependent degradation of storage lipids. Trends Plant. Sci. 2001, 6, 268–273. [Google Scholar] [CrossRef]
- Suurmeijer, C.N.S.P.; Pérez-Gilabert, M.; van der Hijden, H.T.W.M.; Veldink, G.A.; Vliegenthart, J.F.G. Purification, product characterization and kinetic properties of soluble tomato lipoxygenase. Plant. Physiol. Biochem. 1998, 36, 657–663. [Google Scholar] [CrossRef][Green Version]
- Sarde, S.J.; Kumar, A.; Remme, R.N.; Dicke, M. Genome-wide identification, classification and expression of lipoxygenase gene family in pepper. Plant. Mol. Biol. 2018, 98, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Pejin, B.; Bianco, A.; Newmaster, S.; Sabovljevice, M.; Vujisicf, L.; Tesevic, V.; Vajsf, V.; De Rosab, S. Fatty acids of Rhodobryum ontariense (Bryaceae). Nat. Prod. Res. 2012, 26, 696–702. [Google Scholar] [CrossRef]
- Bilka, F.; Balažová, A.; Bilková, A.; Holková, I. Comparison of sanguinarine production in suspension cultures of the Papaveraceae plants. Čes. slov. Farm. 2012, 61, 267–270. [Google Scholar]
- Murashige, T.; Skoog, F. Revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol. Plantarum 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
Sample Availability: Sample of purified LOX is available from the authors. |


| Purification Step | Activity (nkat/mL) | Proteins (mg/mL) | Specific Activity (nkat/mg) | Purification (fold) |
|---|---|---|---|---|
| Crude extract 100,000× g soluble fraction | 937.20 789.40 | 66.55 40.77 | 14.08 19.36 | 1.0 1.4 |
| Phenyl-Sepharose CL-4B | 208.90 | 1.05 | 198.9 | 14.1 |
| HA Ultrogel | 136.94 | 0.41 | 334.0 | 24.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Holková, I.; Rauová, D.; Mergová, M.; Bezáková, L.; Mikuš, P. Purification and Product Characterization of Lipoxygenase from Opium Poppy Cultures (Papaver somniferum L.). Molecules 2019, 24, 4268. https://doi.org/10.3390/molecules24234268
Holková I, Rauová D, Mergová M, Bezáková L, Mikuš P. Purification and Product Characterization of Lipoxygenase from Opium Poppy Cultures (Papaver somniferum L.). Molecules. 2019; 24(23):4268. https://doi.org/10.3390/molecules24234268
Chicago/Turabian StyleHolková, Ivana, Drahomíra Rauová, Michaela Mergová, Lýdia Bezáková, and Peter Mikuš. 2019. "Purification and Product Characterization of Lipoxygenase from Opium Poppy Cultures (Papaver somniferum L.)" Molecules 24, no. 23: 4268. https://doi.org/10.3390/molecules24234268
APA StyleHolková, I., Rauová, D., Mergová, M., Bezáková, L., & Mikuš, P. (2019). Purification and Product Characterization of Lipoxygenase from Opium Poppy Cultures (Papaver somniferum L.). Molecules, 24(23), 4268. https://doi.org/10.3390/molecules24234268







