Multifunctional and Environmentally Friendly TiO2–SiO2 Mesoporous Materials for Sustainable Green Buildings
Abstract
1. Introduction
2. Discussion
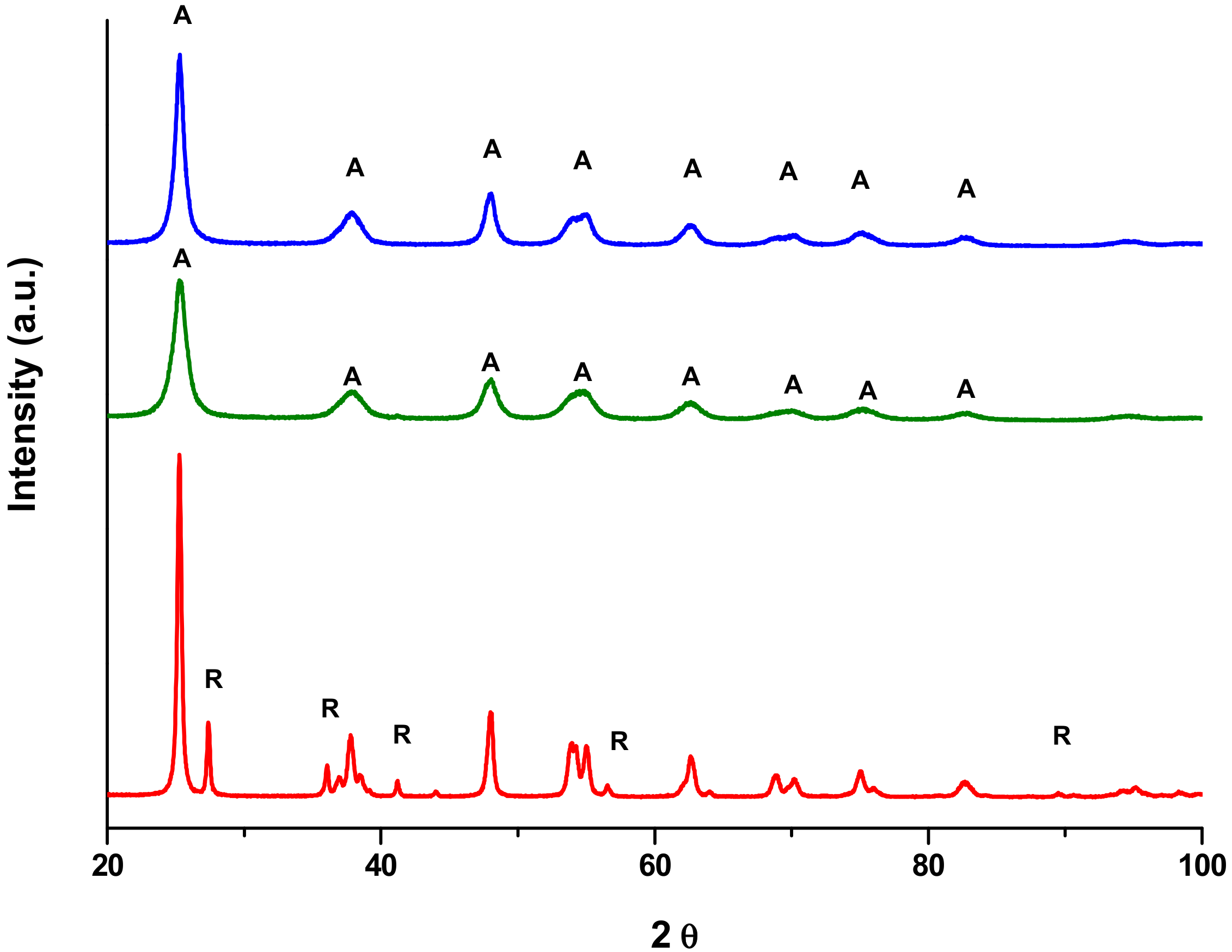
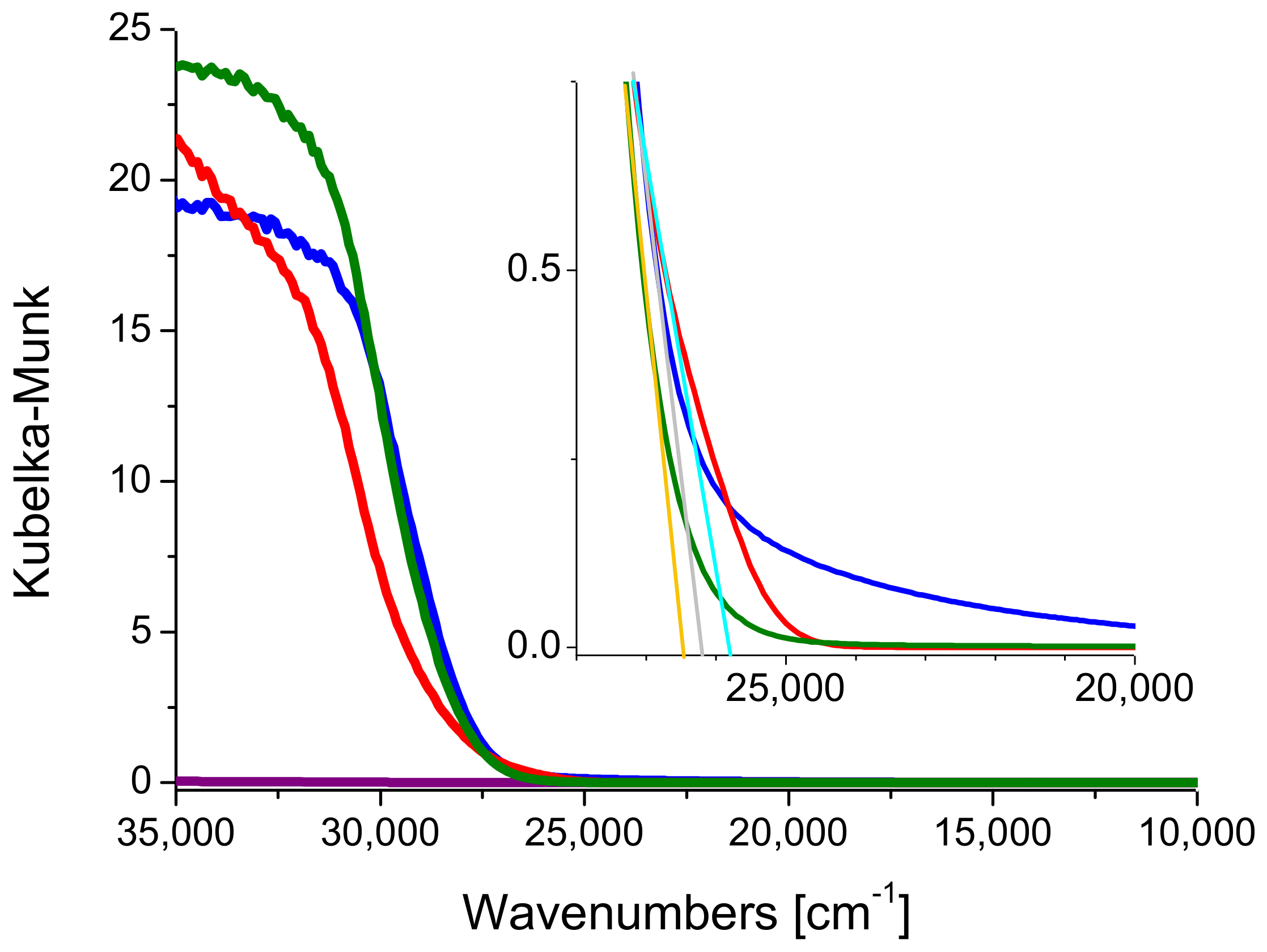
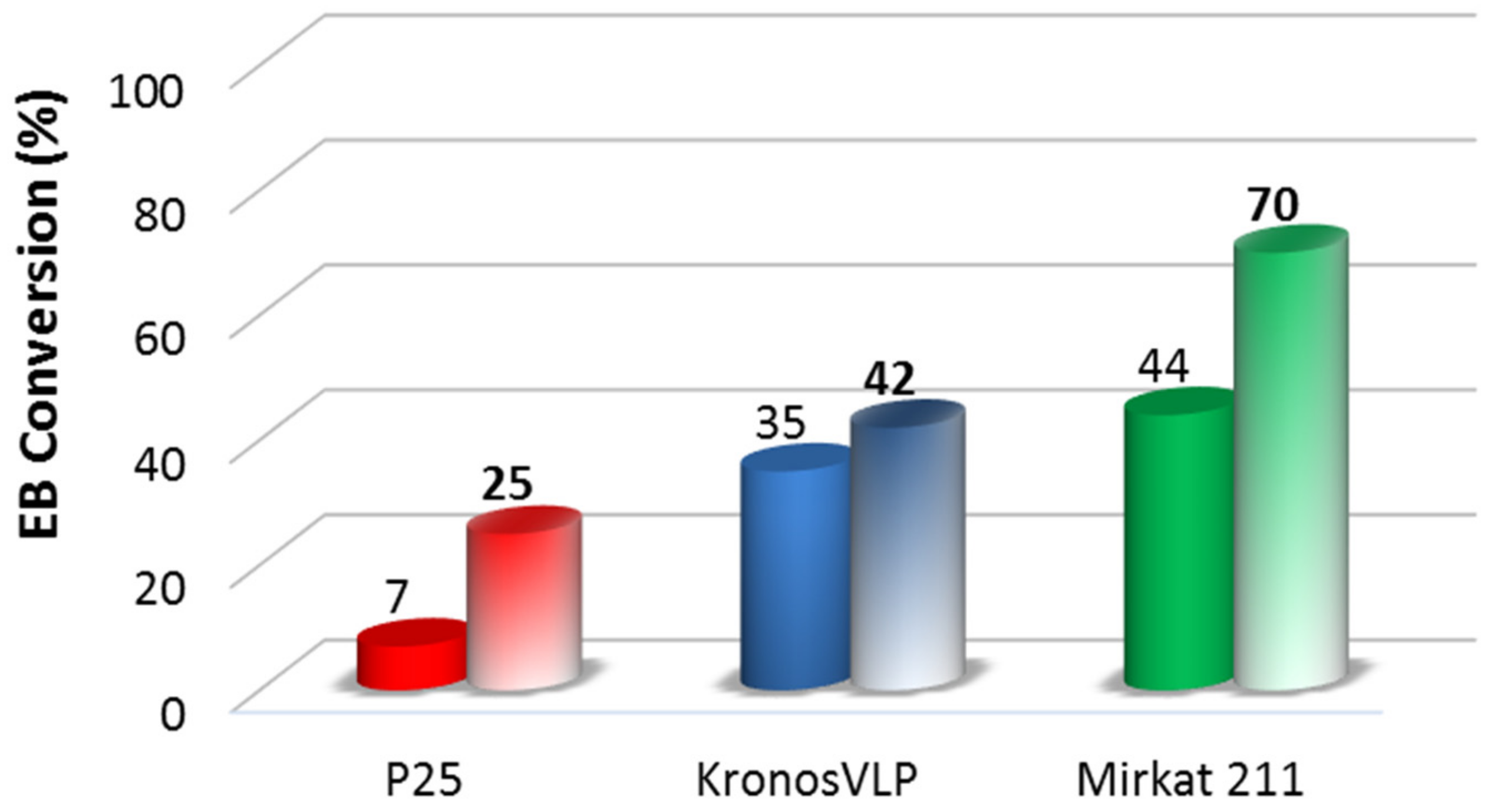
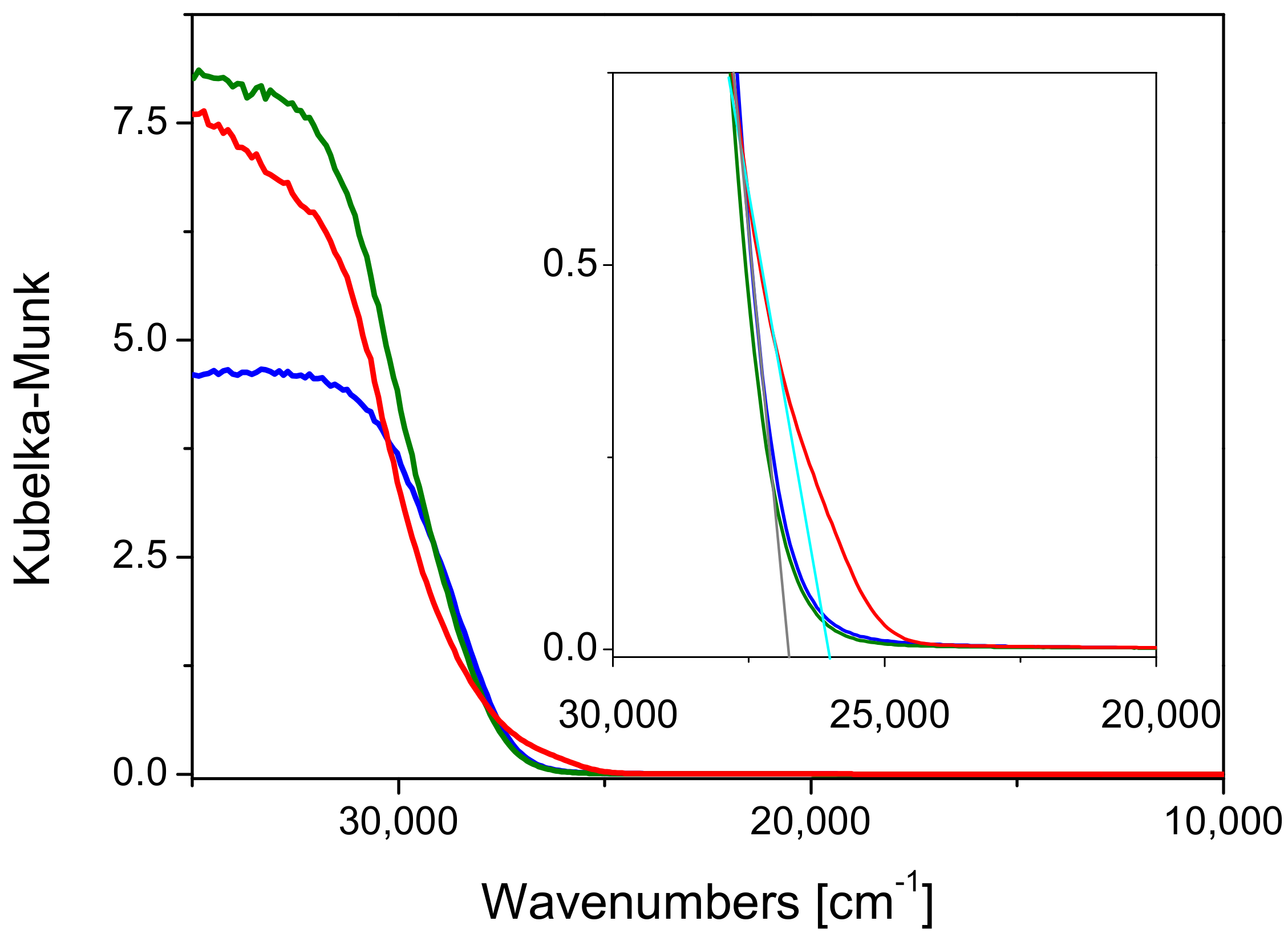
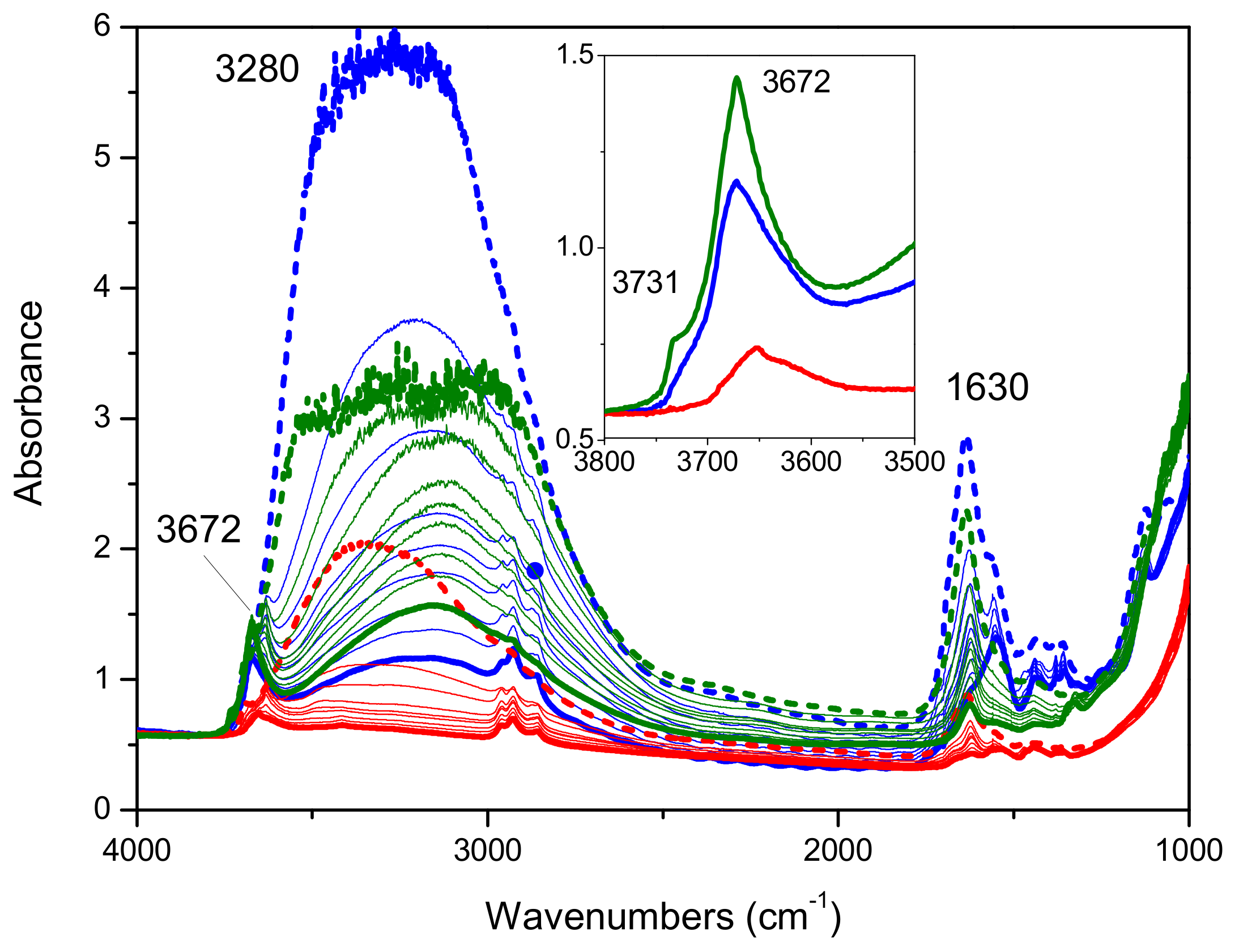
2.1. Preliminary Photocatalytic Studies and Characterization of the Commercial TiO2 Samples
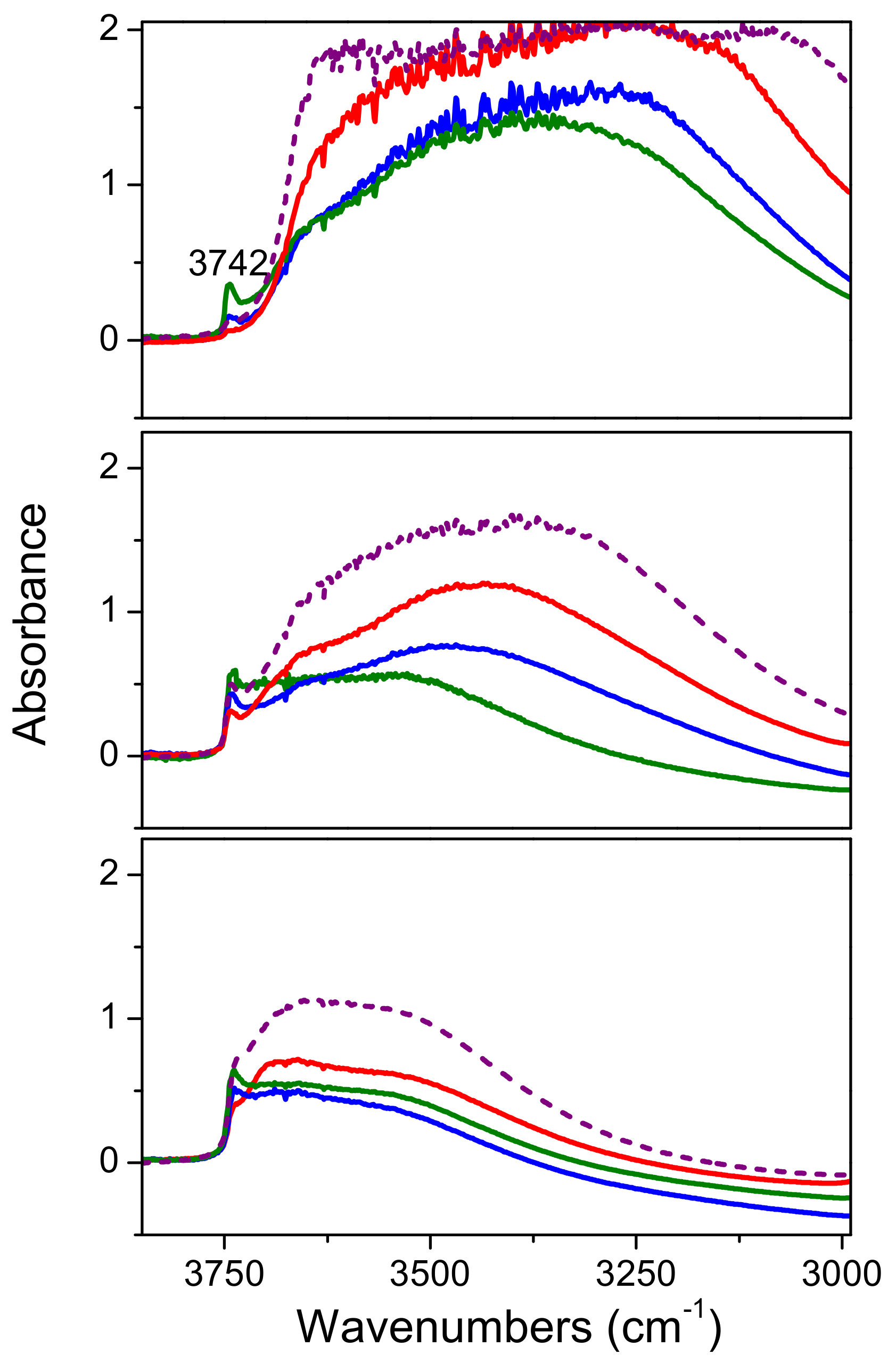
2.2. High Surface Area TiO2/SBA-15 Materials
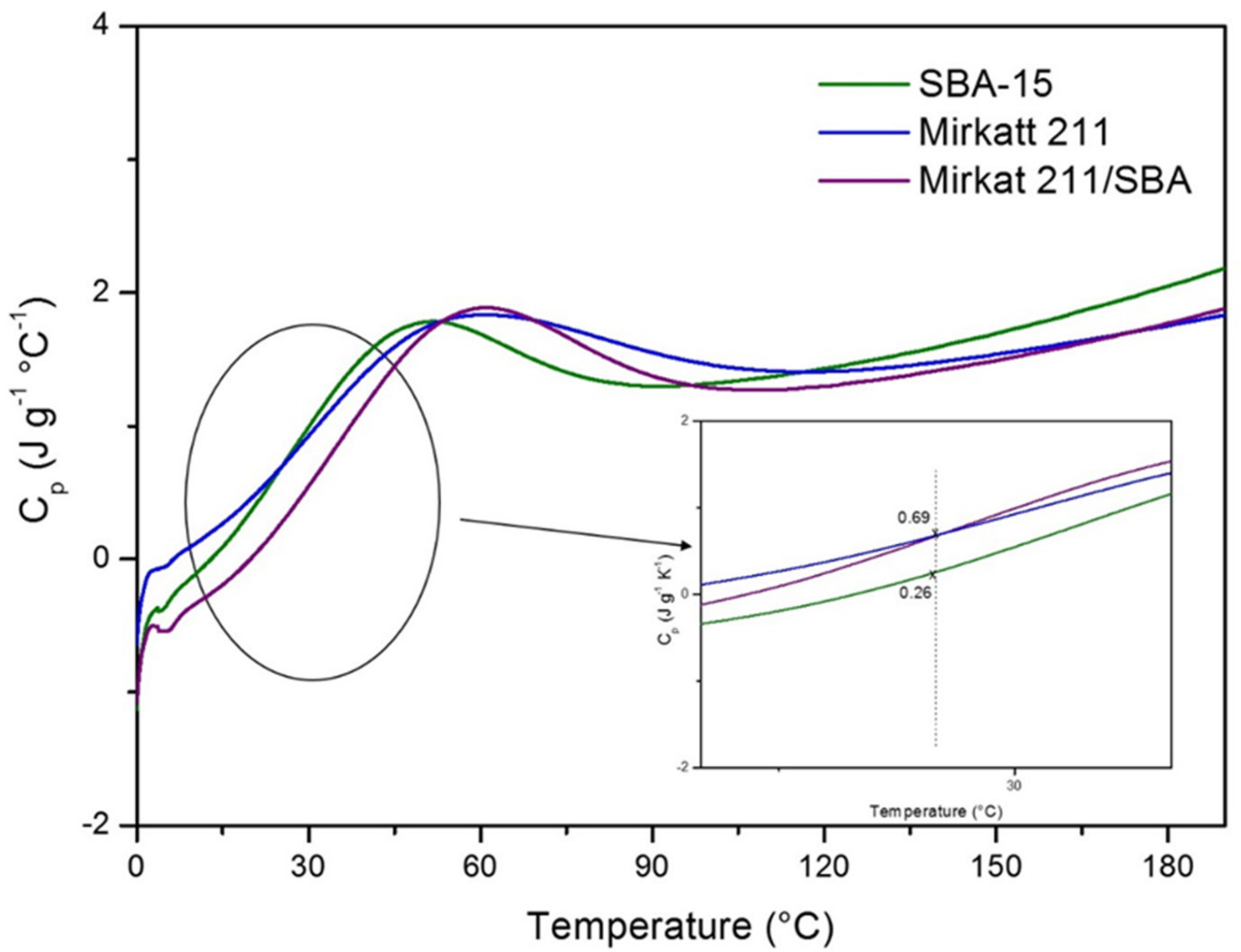
2.3. Insulating Features: Specific Heat Capacity
3. Materials and Methods
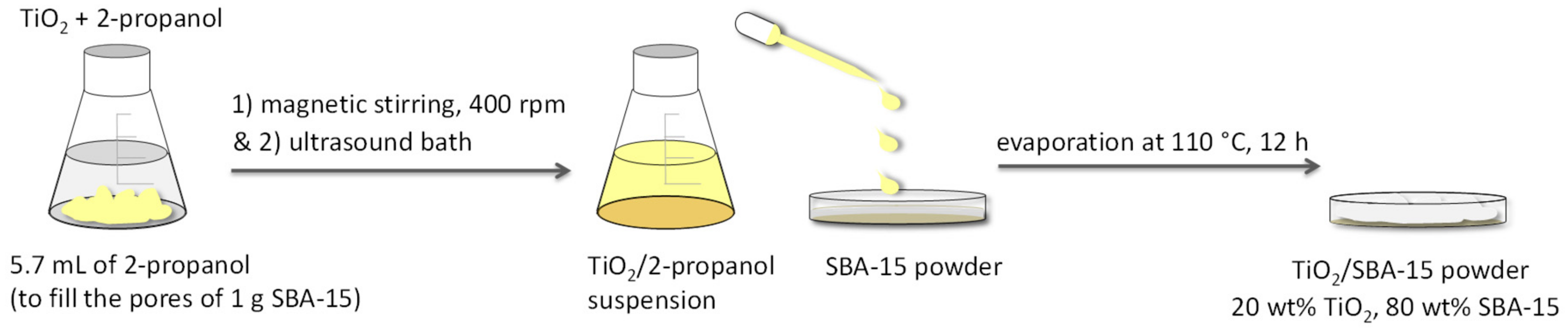
3.1. Formulation and Preparation of the TiO2/SBA-15 Systems
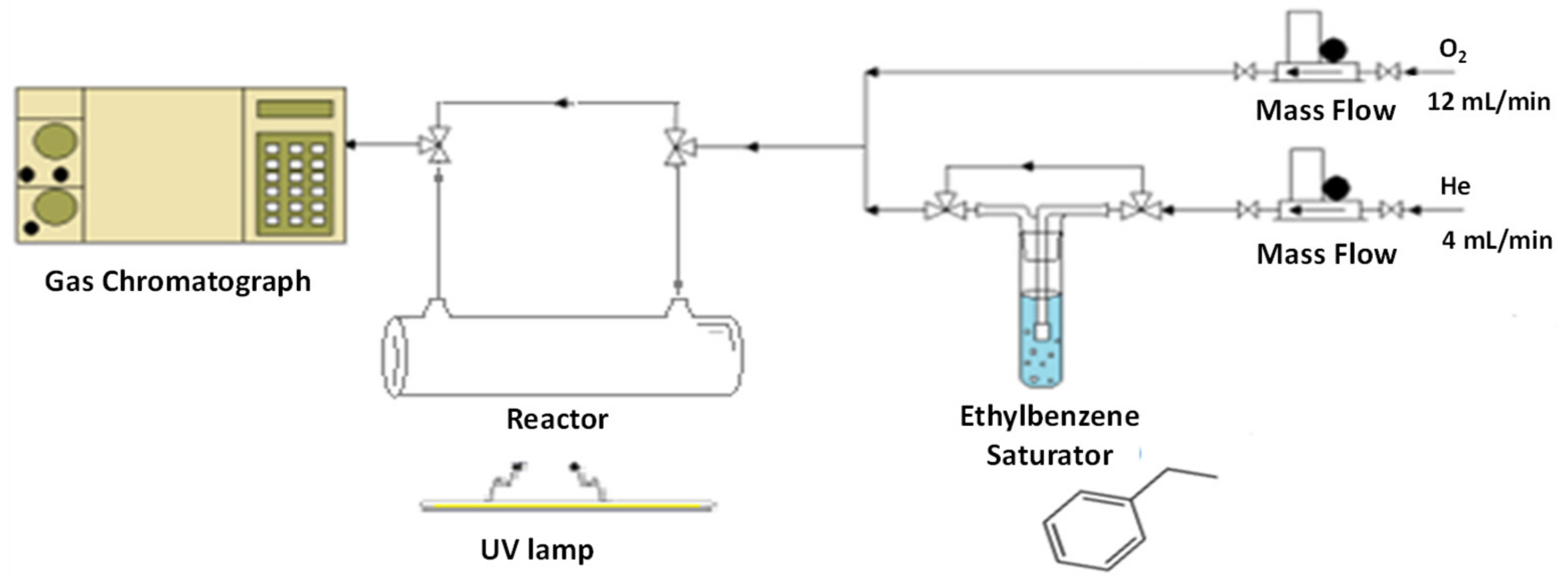
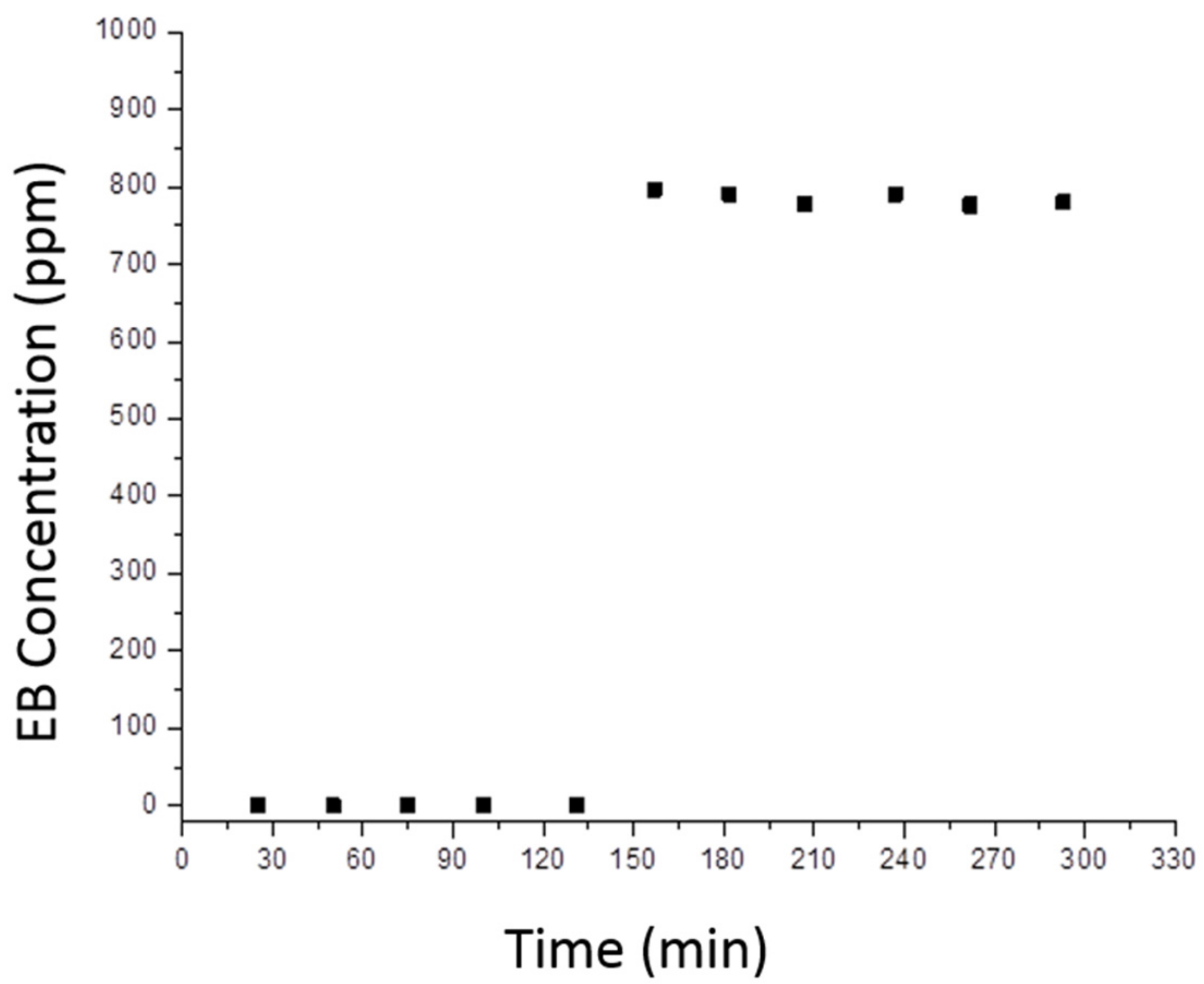
3.2. Photocatalytic Tests on VOC Abatement
3.3. Characterisation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Energy Information Administration, Government Publications Office. Energy Information Administration, “International Energy Outlook 2016 with projections to 2040”; Government Printing Office: Washington, DC, USA, 2016. Available online: http://www.eia.gov/forecasts/ieo/pdf/0484(2016).pdf (accessed on 20 May 2019).
- Perez-Lombard, L.; Ortiz, J.; Pout, C. A Review on Buildings Energy Consumption Information. Energy Build. 2008, 40, 394–398. [Google Scholar] [CrossRef]
- Architecture2030. Architecture 2030 Will Change the Way You Look atBuildings. 2013. Available online: http://architecture2030.org/the problem/buildings (accessed on 20 May 2019).
- McKinsey, G.I. Pathways to a Low-Carbon Economy-Version2 of the Global Greenhouse Gas Abatement Cost Curve; McKinsey Company: Stockholm, Sweden, 2009. [Google Scholar]
- Official Site of United Nations Environment Programme. Available online: http://web.unep.org/about/..il (accessed on 13 March 2019).
- Official site of World Green Building Council. Available online: www.worldgbc.org/ (accessed on 20 December 2018).
- Green Building Council. World Green Building Trends 2013, 1st ed.; McGraw-Hill Bradford (USA): Columbus, OH, USA, 2013. [Google Scholar]
- Boyjoo, H.; Sun, Y.; Liu, J.; Pareek, V.K.; Wang, S. A Review on Photocatalysis for Air Treatment: From Catalyst Development to Reactor Design. Chem. Eng. J. 2017, 310, 537–559. [Google Scholar] [CrossRef]
- Handbook of Green Building Design and Construction, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2017.
- Hager, S.; Bauer, R.; Kudielka, G. Photocatalytic Oxidation of Gaseous Chlorinated Organics Over Titanium Dioxide. Chemosphere 2000, 41, 1219–1225. [Google Scholar] [CrossRef]
- Kazuhito, H.; Hiroshi, I.; Akira, F. TiO2 Photocatalysis: A Historical Overview and Future Prospects. Jpn. J. Appl. Phys. 2005, 44, 8269–8285. [Google Scholar]
- Signoretto, M.; Ghedini, E.; Trevisan, V.; Bianchi, C.L.; Ongaro, M.; Cruciani, G. TiO2-MCM-41 for the Photocatalytic Abatement of NOx in Gas Phase. Appl. Cat. B Environ. 2010, 95, 130–136. [Google Scholar] [CrossRef]
- Baetens, R.; Jelle, B.P.; Gustavsen, A. Aerogel Insulation for Building Applications: A State-of-the-Art Review. Energy Build. 2011, 43, 761–769. [Google Scholar] [CrossRef]
- Antonelli, D.M.; Ying, J.Y. Synthesis of Hexagonally Packed Mesoporous TiO2 by a Modified Sol–Gel Method. Angew. Chem. Int. Ed. Engl. 1995, 34, 2014–2017. [Google Scholar] [CrossRef]
- Liu, C.; Fu, L.; Economy, J. A Simple, Template-Free Route for the Synthesis of Mesoporous Titanium Dioxide Materials. J. Mater. Chem. 2004, 14, 1187–1189. [Google Scholar] [CrossRef]
- On, D.T.; Kaliaguine, S.; Bonneviot, L. Titanium Boralites with MFI Structure Characterized Using XRD, XANES, IR, and UV-Visible Techniques: Effect of Hydrogen Peroxide on the Preparation. J. Catal. 1995, 157, 235–243. [Google Scholar] [CrossRef]
- Shibata, H.; Ogura, T.; Ohkubo, T.; Sakai, H.; Abe, M. Direct Synthesis of Mesoporous Titania Particles Having a Crystalline Wall. J. Am. Chem. Soc. 2005, 127, 16396–16397. [Google Scholar] [CrossRef]
- Zhao, J.; Wan, P.; Xiang, J.; Tong, T.; Dong, L.; Gao, Z.; Shen, X. Synthesis of highly Ordered Macro-Mesoporous Anatase TiO2 Film with High Photocatalytic Activity. Microp. Mesop. Mater. 2011, 138, 200–206. [Google Scholar] [CrossRef]
- Tian, B.Z.; Yang, H.F.; Liu, X.Y.; Xie, S.H.; Yu, C.Z.; Fan, J. Fast preparation of highly ordered non siliceous mesoporous materials via mixed inorganic precursors. Chem. Commun. 2002, 17, 1824–1825. [Google Scholar] [CrossRef] [PubMed]
- Imperor-Clerc, M.; Davidson, P.; Davidson, A. Existence of a Microporous Corona Around the Mesopores of Silica-Based SBA-15 Materials Templated by Triblock Copolymers. J. Am. Chem. Soc. 2000, 122, 11925–11933. [Google Scholar] [CrossRef]
- Kao, L.H.; Hsu, T.C.; Cheng, K.K. Novel Synthesis of High-Surface-Area Ordered Mesoporous TiO2 with Anatase Framework for Photocatalytic Applications. J. Colloid Interf. Sci. 2010, 341, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Hung, Y.; Wang, Y.; Huang, C.F.; Fan, Y.S.; Han, Y.J.; Peng, H.W. Effects of Templating Surfactant Concentrations on the Mesostructure of Ordered Mesoporous Anatase TiO2 by an Evaporation-Induced Self-Assembly Method. J. Europ. Ceram. Soc. 2010, 30, 2065–2072. [Google Scholar] [CrossRef]
- Zhao, D.; Huo, Q.; Feng, J.; Chmelka, B.F.; Stucky, G.D. Nonionic Triblock and Star Diblock Copolymer and Oligomeric Surfactant Syntheses of Highly Ordered, Hydrothermally Stable, Mesoporous Silica Structures. J. Am. Chem. Soc. 1998, 120, 6024–6036. [Google Scholar] [CrossRef]
- Zhao, D.; Feng, J.; Huo, Q.; Melosh, N.; Fredrickson, G.H.; Chmelka, B.F.; Stucky, G.D. Generalized Syntheses of Large-Pore Mesoporous Metal Oxides with Semicrystalline Frameworks. Science 1998, 279, 548–552. [Google Scholar] [CrossRef]
- Ghedini, E.; Signoretto, M.; Pinna, F.; Cerrato, G.; Morterra, C. Gas and Liquid Phase Reactions on MCM-41/SZ Catalysts. Appl. Catal. B, Environ. 2006, 67, 24–33. [Google Scholar] [CrossRef]
- Belmoujahid, Y.; Bonne, M.; Scudeller, Y.; Schleich, D.; Grohens, Y.; Lebeau, B. Thermal Conductivity of Monolithic Assemblies of SBA-15 Ordered Mesoporous Silica Particles. Micropor. Mesopor. Mat. 2015, 201, 124–133. [Google Scholar] [CrossRef]
- Coquil, T.; Richman, E.K.; Hutchinson, N.J.; Tolbert, S.H.; Pilon, L. Thermal Conductivity of Cubic and Hexagonal Mesoporous Silica Thin Films. J. Appl. Phys. 2009, 106, 124–133. [Google Scholar] [CrossRef]
- Cuce, E.; Cuce, P.M.; Wood, J.C.; Riffat, S.B. Toward Aerogel Based Thermal Superinsulation in Buildings: A Comprehensive Review. Sustain. Energy Rev. 2014, 34, 273–299. [Google Scholar] [CrossRef]
- Gomes, S.; David, L.; Lysenko, V.; Descamps, A.; Nychyporuk, T.; Raynaud, M. Application of scanning thermal microscopy for thermal conductivity measurements on meso-porous silicon thin films. J. Phys. D Appl. Phys. 2007, 40, 6677–6683. [Google Scholar] [CrossRef]
- Signoretto, M.; Ghedini, E.; Nichele, V.; Pinna, F.; Crocella, V.; Cerrato, G. Effect of Textural Properties on the Drug Delivery Behaviour of Nanoporous TiO2 Matrices. Micropor. Mesopor. Mater. 2011, 139, 189–196. [Google Scholar] [CrossRef]
- Kudo, A.; Miseki, Y. Heterogeneous Photocatalyst Materials for Water Splitting. Chem. Soc. Rev. 2009, 38, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Shibata, T.; Irie, H.; Ohmori, M.; Nakajima, A.; Watanabe, T.; Hashimoto, K. Comparison of Photochemical Properties of Brookite and Anatase TiO2 Films. Phys. Chem. Chem. Phys. 2004, 6, 1359–1362. [Google Scholar] [CrossRef]
- Ebert, H.P. Thermal Properties of Aerogels. In Aerogels Handbook—Advances in Sol–Gel Derived Materials and Technologies; Aegerter, M.A., Prassas, M., Koebel, M.M., Eds.; Springer: New York, NJ, USA; Dordrecht, The Netherlands; Heidelberg, Germany; London, UK, 2011; pp. 537–581. [Google Scholar]
- Calzada, L.A.; Castellanos, R.; GarcIa, L.A.; Klimova, T.E. TiO2, SnO2 and ZnO Catalysts Supported on Mesoporous SBA-15 Versus Unsupported Nanopowders in Photocatalytic Degradation of Methylene Blue. Microporous Mesoporous Mater. 2019, 285, 247–258. [Google Scholar] [CrossRef]
- Acosta-Silva, Y.J.; Nava, R.; Hernandez-Morales, V.; Macias-Sanchez, S.A.; Gomez-Herrera, M.L.; Pawelec, B. Methylene Blue Photodegradation Over Titania-Decorated SBA-15. Appl. Catal. B 2011, 110, 108–117. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, M. Cai, Sonochemical Incorporation of Nanosized TiO2 Inside Mesoporous Silica with High Photocatalytic Performance. Microporous Mesoporous Mater. 2009, 126, 20–25. [Google Scholar]
- Marszewski, M.; Butts, D.; Lan, E.; Yan, Y.; King, S.C.; McNeil, P.E.; Galy, T.; Dunn, B.; Tolbert, S.H.; Hu, Y.; et al. Effect of Surface Hydroxyl Groups on Heat Capacity of Mesoporous Silica. Appl. Phys. Lett. 2018, 112, 201903. [Google Scholar] [CrossRef]
- Ghedini, E.; Signoretto, M.; Pinna, F.; Cruciani, G. Mesoporous Silica–Zirconia Systems for Catalytic Applications. Catal. Lett. 2008, 125, 359–370. [Google Scholar] [CrossRef]
- Zhao, D.; Feng, J.; Huo, Q.; Melosh, N.; Frederickson, G.H.; Chmelka, B.F.; Stucky, G.D. Triblock Copolymer Syntheses of Mesoporous Silica with Periodic 50 to 300 Angstrom Pores. Science 1998, 279, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Ghedini, E.; Menegazzo, F.; Signoretto, M.; Manzoli, M.; Pinna, F.; Strukul, G. Mesoporous Silica as Supports for Pd-Catalyzed H2O2 Direct Synthesis: Effect of the Textural Properties of the Support on the Activity and Selectivity. J. Catal. 2010, 273, 266–273. [Google Scholar] [CrossRef]
- Larson, A.C.; Von Dreele, R.B. General Structure Analysis System (GSAS); Los Alamos National Laboratory Report Laur 86–748; The Regents of the University of California: Oakland, CA, USA, 2004. [Google Scholar]
- Giuli, G.; Eisenmann, T.; Bresser, D.; Trapananti, A.; Asenbauer, J.; Passerini, F.M.S. Structural and Electrochemical Characterization of Zn1−xFexO—Effect of Aliovalent Doping on the Li+ Storage Mechanism. Materials 2018, 11, 49. [Google Scholar] [CrossRef] [PubMed]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Barret, E.P.; Joyner, L.G.; Halenda, P.P. The Determination of Pore Volume and Area Distributions in Porous Substances. J. Am. Chem. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |


| Sample | Specific Surface Area (m2/g) | Pore Size Range (nm) | Pore Volume (ml/g) | Crystallite Size a (nm) |
|---|---|---|---|---|
| KRONOS VLP | 269 | 4–10 | 0.30 | 15 |
| MIRKAT 211 | 217 | 4–10 | 0.27 | 11 |
| P25 | 52 | 4–80 | 0.13 | 40 |
| Sample | Specific Surface Area (m2/g) | Pore Size Range (nm) | Pore Volume (ml/g) | Crystallite Size a (nm) |
|---|---|---|---|---|
| SBA-15 | 852 ± 2 | 8.5 | 1.09 | - |
| KRONOS VLP/SBA-15 | 802 ± 3 | 8.2 | 0.99 | 22 |
| MIRKAT 211/SBA-15 | 810 ± 4 | 8.0 | 1.06 | 10 |
| P25/SBA-15 | 797 ± 2 | 8.0 | 0.99 | 30 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghedini, E.; Menegazzo, F.; Manzoli, M.; Di Michele, A.; Puglia, D.; Signoretto, M. Multifunctional and Environmentally Friendly TiO2–SiO2 Mesoporous Materials for Sustainable Green Buildings. Molecules 2019, 24, 4226. https://doi.org/10.3390/molecules24234226
Ghedini E, Menegazzo F, Manzoli M, Di Michele A, Puglia D, Signoretto M. Multifunctional and Environmentally Friendly TiO2–SiO2 Mesoporous Materials for Sustainable Green Buildings. Molecules. 2019; 24(23):4226. https://doi.org/10.3390/molecules24234226
Chicago/Turabian StyleGhedini, Elena, Federica Menegazzo, Maela Manzoli, Alessandro Di Michele, Debora Puglia, and Michela Signoretto. 2019. "Multifunctional and Environmentally Friendly TiO2–SiO2 Mesoporous Materials for Sustainable Green Buildings" Molecules 24, no. 23: 4226. https://doi.org/10.3390/molecules24234226
APA StyleGhedini, E., Menegazzo, F., Manzoli, M., Di Michele, A., Puglia, D., & Signoretto, M. (2019). Multifunctional and Environmentally Friendly TiO2–SiO2 Mesoporous Materials for Sustainable Green Buildings. Molecules, 24(23), 4226. https://doi.org/10.3390/molecules24234226