Magnetic Solid-Phase Extraction of Pyrethroid Pesticides from Environmental Water Samples Using Deep Eutectic Solvent-type Surfactant Modified Magnetic Zeolitic Imidazolate Framework-8
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Reagents
2.1.2. Apparatus
2.2. Methods
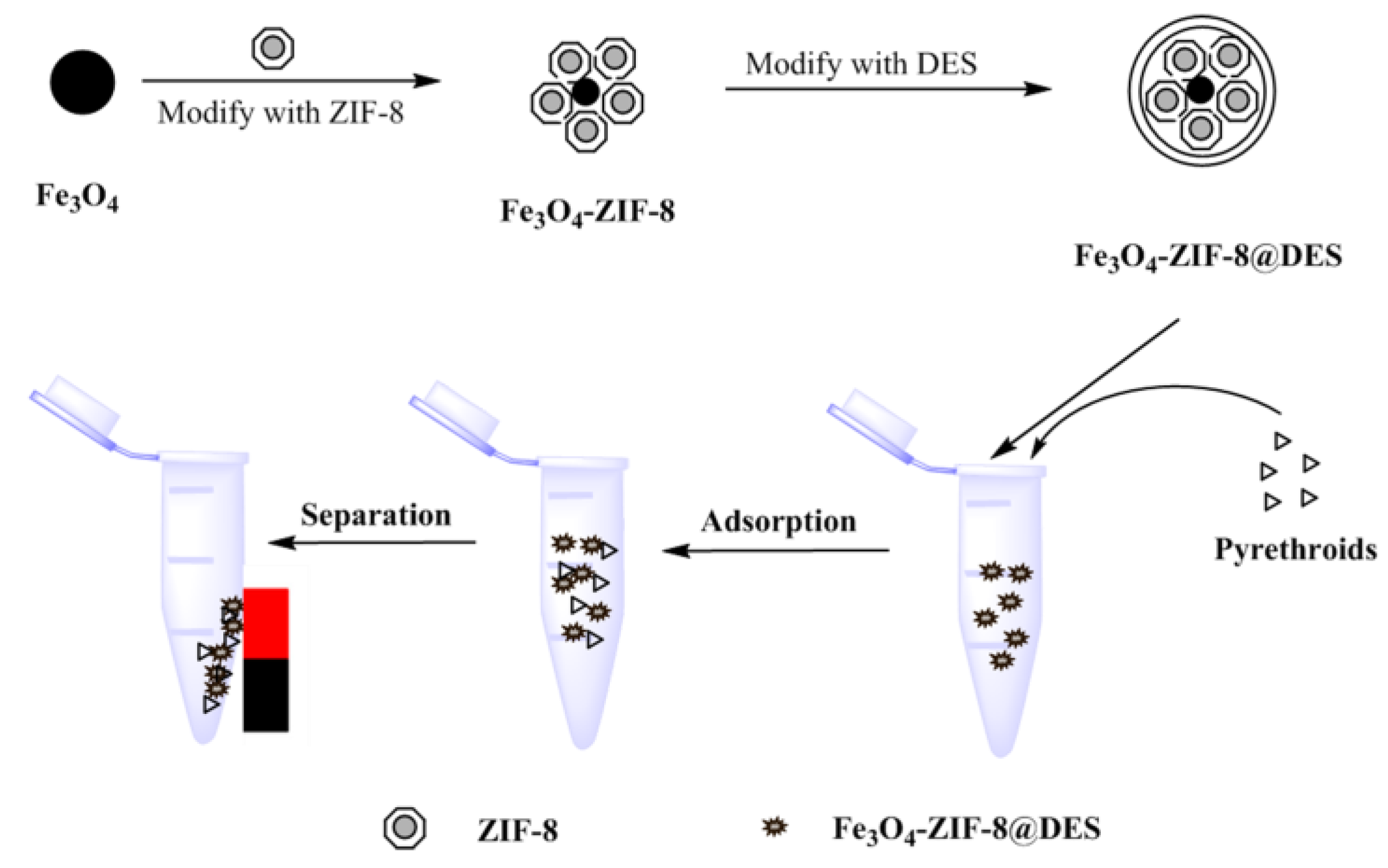
2.2.1. Synthesis of M-ZIF-8
2.2.2. Synthesis of M-ZIF-8@DES
2.2.3. The Process of MSPE
2.2.4. Preparation of Real Environmental Water Samples
3. Results and Discussion
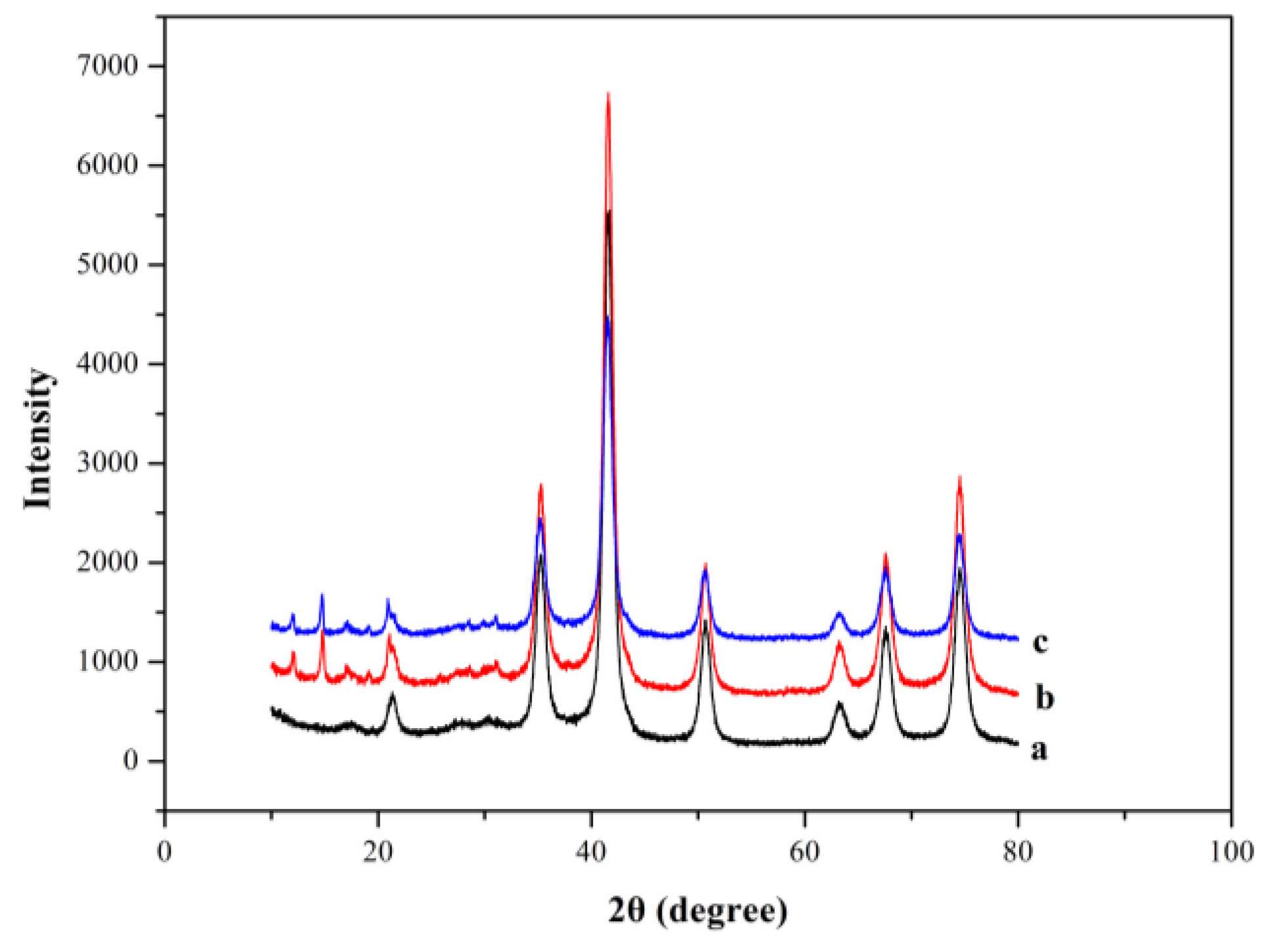
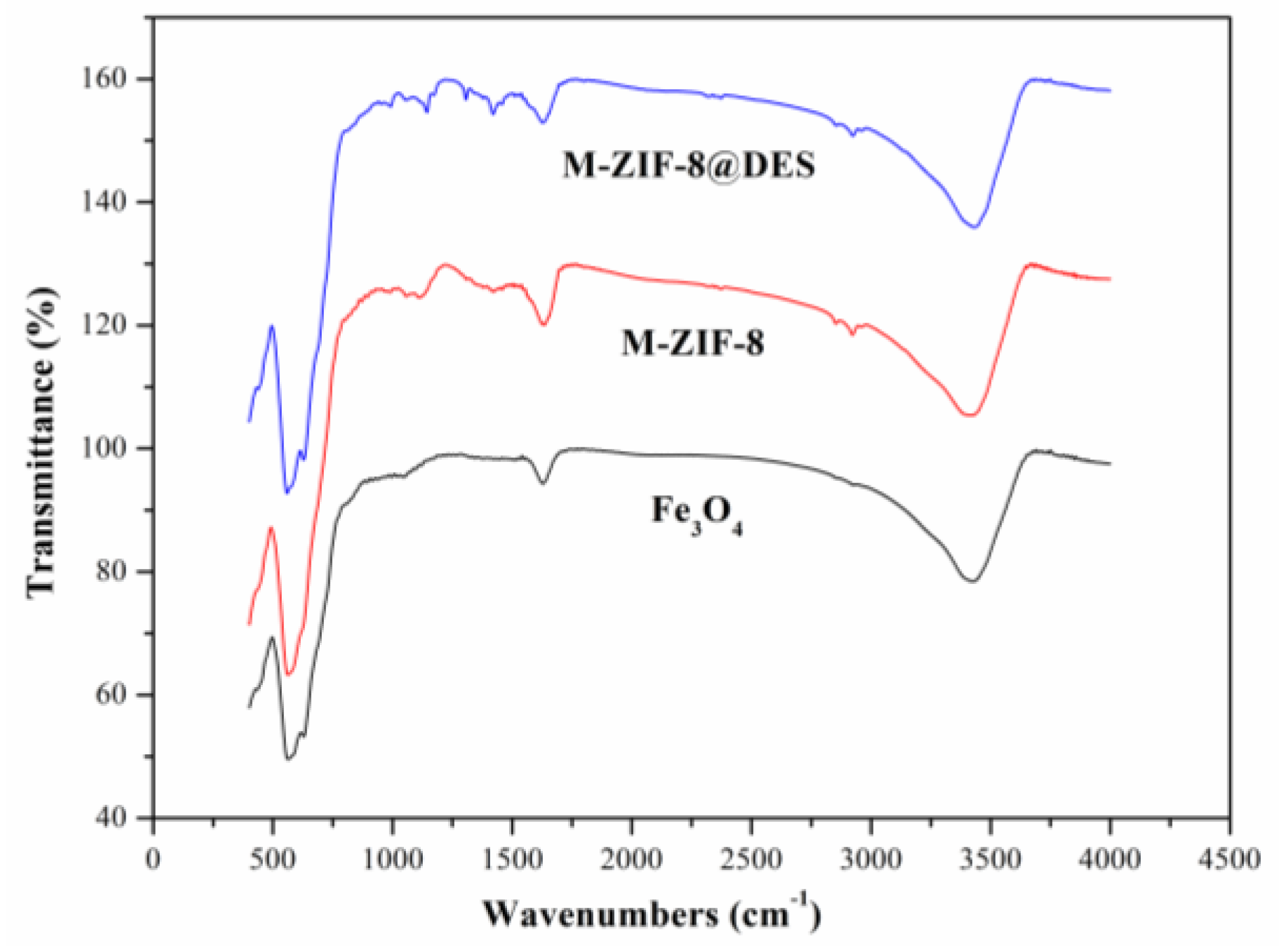
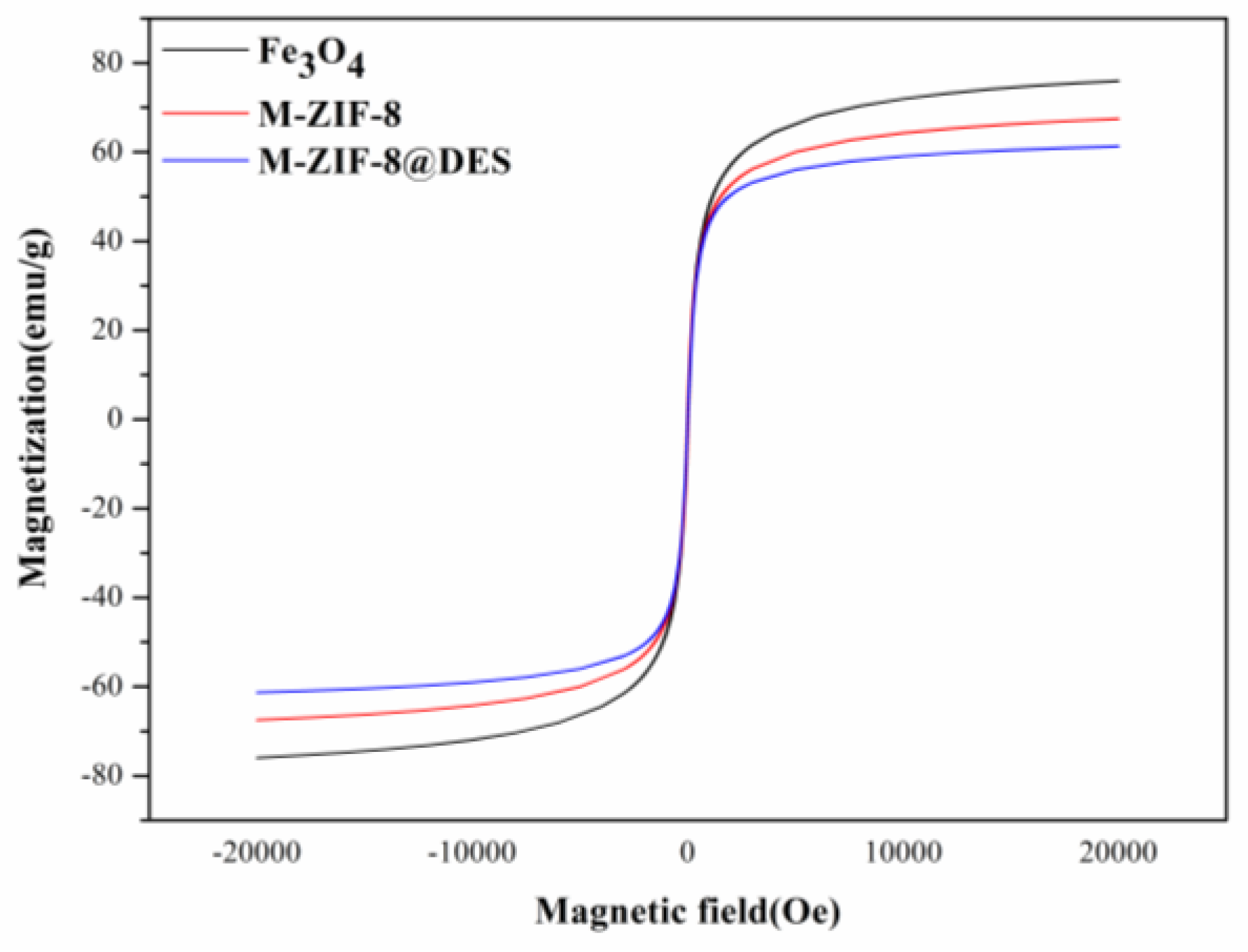
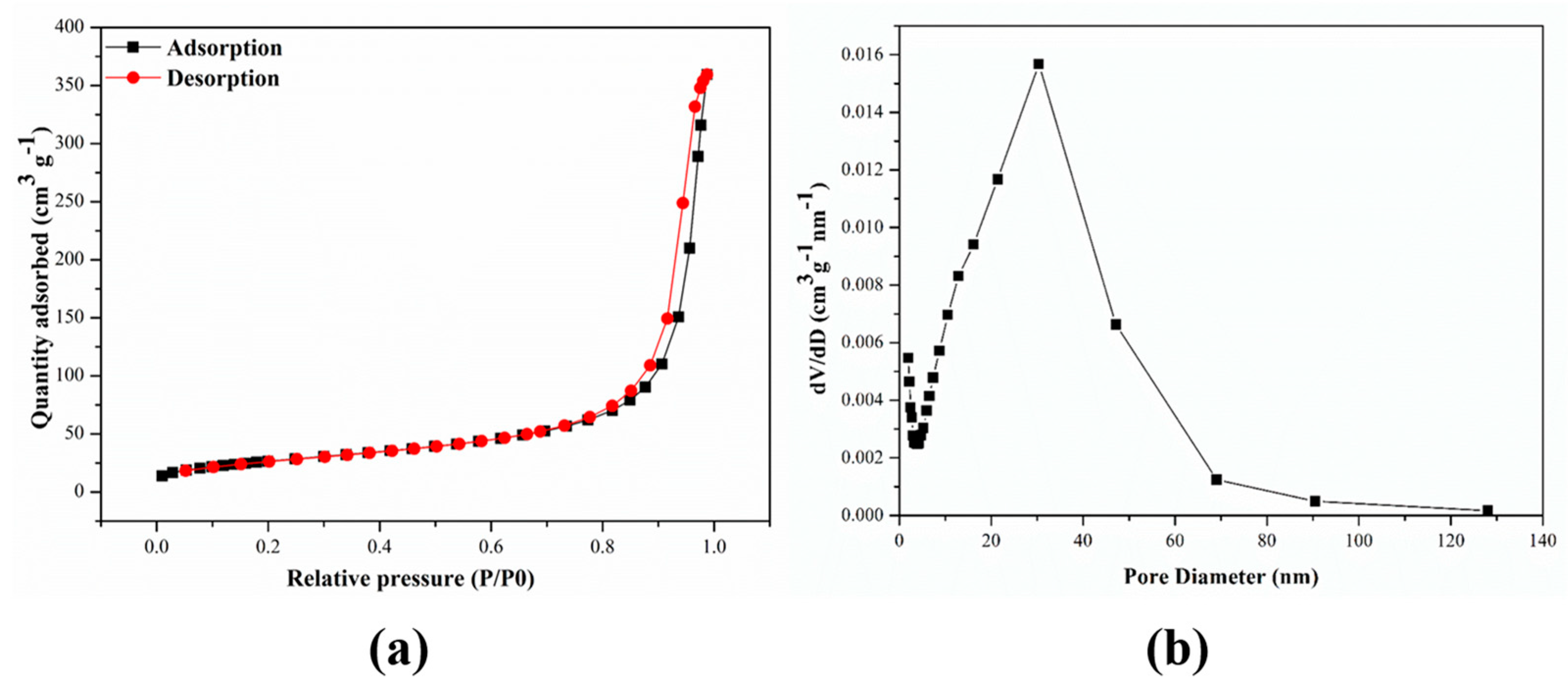
3.1. Characterization of M-ZIF-8@DES
3.2. Optimization of MSPE Precedure
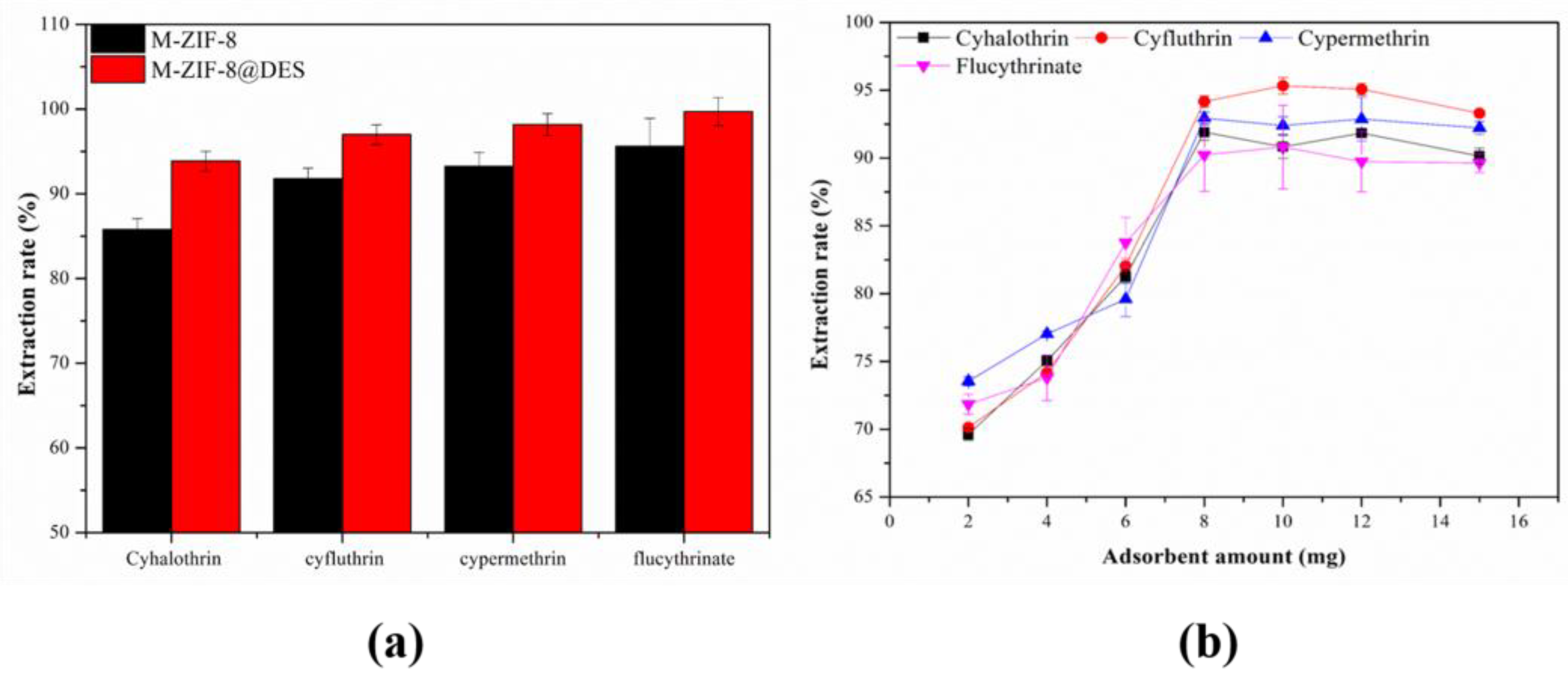
3.2.1. Effect of Different Adsorbents
3.2.2. Effect of the Amount of Adsorbent
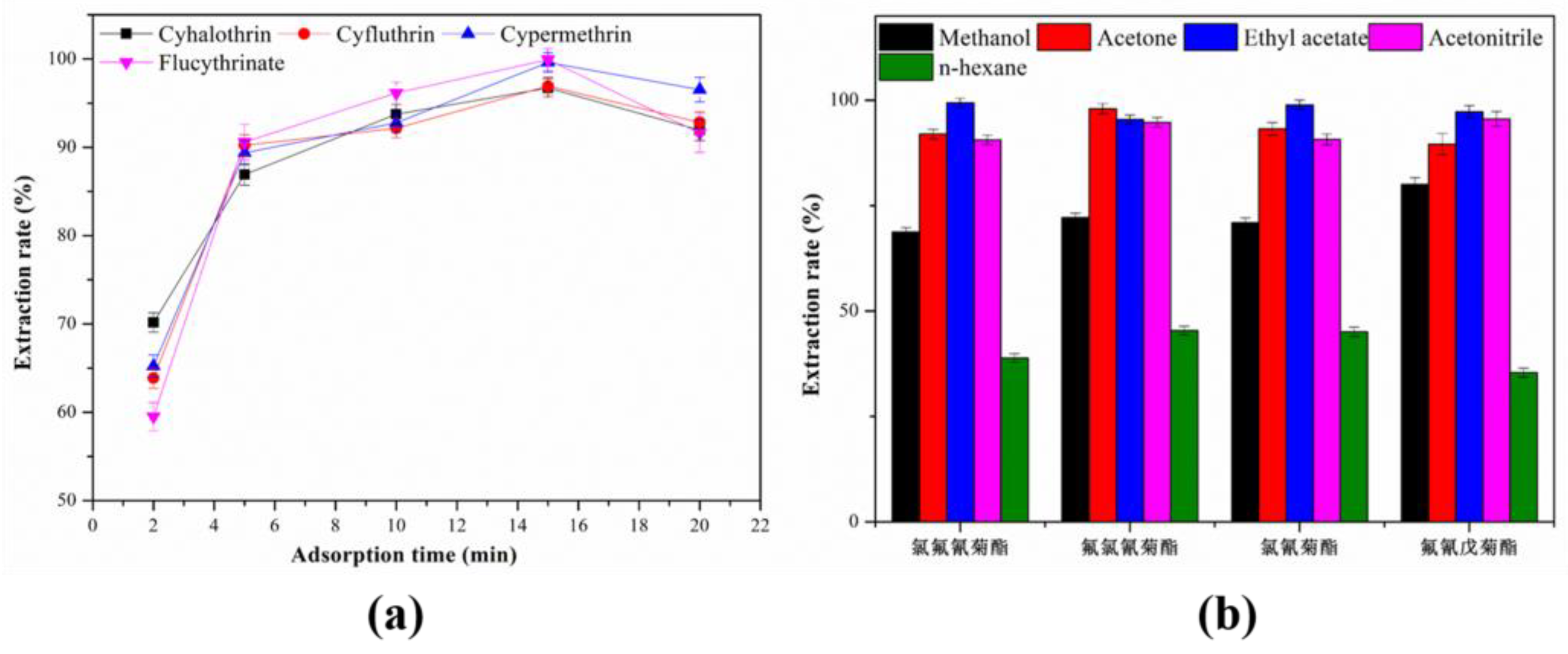
3.2.3. Effect of the Adsorption Time
3.2.4. Effect of the Types of Desorption Solvent
3.2.5. Effect of the Volume of Desorption Solvent
3.2.6. Effect of the Desorption Time
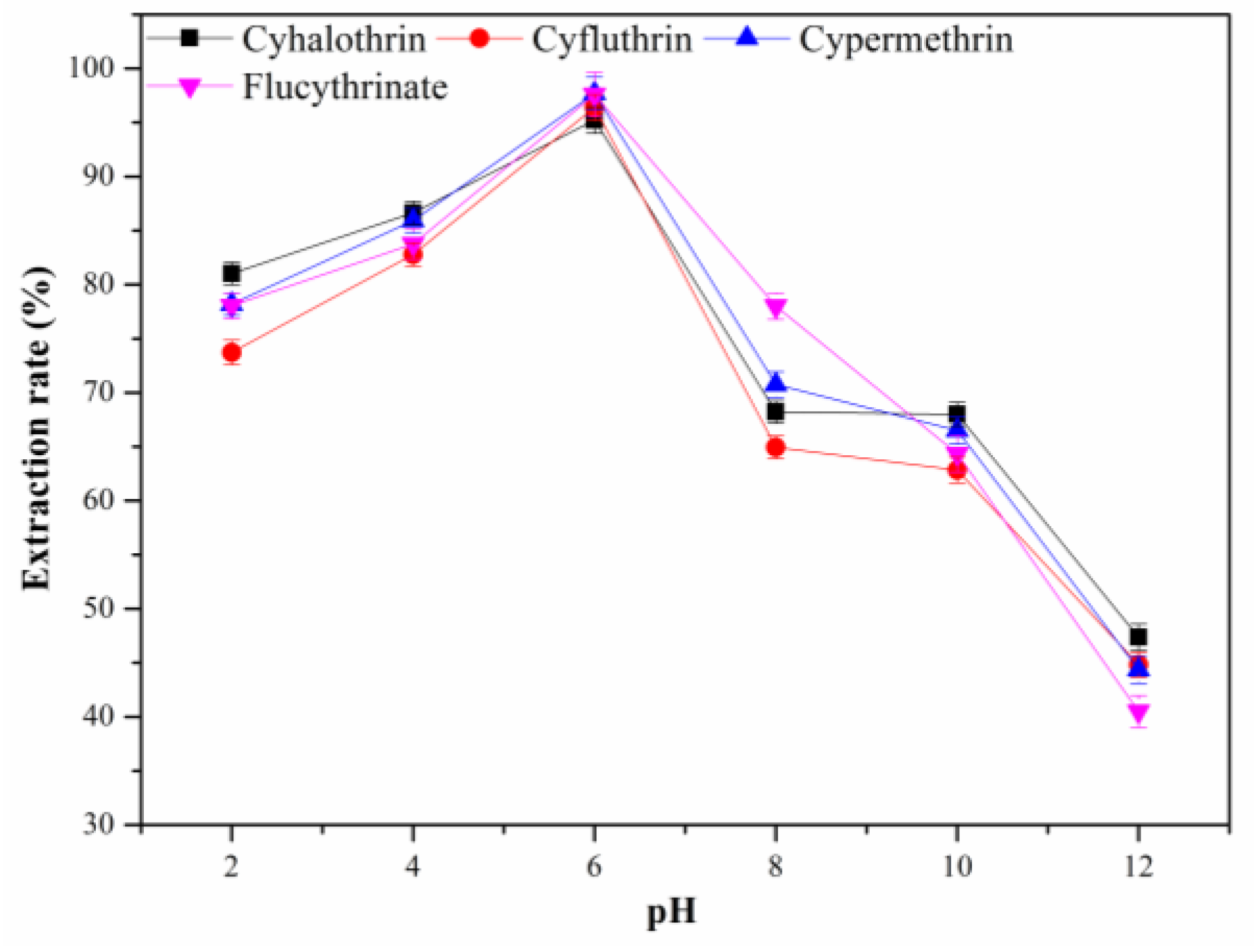
3.2.7. Effect of the pH Value of Sample Solution
3.3. Methodological Study
3.4. Analysis of the Environmental Water Samples
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, J.; Zhu, W.; Zheng, Y.; Yang, J.; Zhu, X. The antiandrogenic activity of pyrethroid pesticides cyfluthrin and β-cyfluthrin. Reprod. Toxicol. 2008, 25, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Esteve-Turrillas, F.A.; Aman, C.S.; Pastor, A.; Guardia, M.D.L. Microwave-assisted extraction of pyrethroid insecticides from soil. Anal. Chim. Acta 2004, 522, 73–78. [Google Scholar] [CrossRef]
- Xu, Z.; Liu, W.; Yang, F. A new approach to estimate bioavailability of pyrethroids in soil by compound-specific stable isotope analysis. J. Hazard. Mater. 2018, 349, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.D.; Liu, Y.N.; Liu, G.Y.; Li, L.Y.; Xu, X.M.; Zheng, S.N. Preparation of a magnetic multiwalled carbon nanotube @ polydopamine/zeolitic imidazolate framework-8 composite for magnetic solid-phase extraction of triazole fungicides from environmental water samples. RSC Adv. 2018, 8, 25351–25360. [Google Scholar] [CrossRef]
- Azzouz, A. Review of nanomaterials as sorbents in solid-phase extraction for environmental samples. Trends Anal. Chem. 2018, 108, 347–369. [Google Scholar] [CrossRef]
- Del Castillo, M.L.R.; Rodríguez-Valenciano, M.; Flores, G.; Blanch, G.P. New method based on solid phase microextraction and multidimensional gas chromatography-mass spectrometry to determine pesticides in strawberry jam. LWT 2019, 99, 283–290. [Google Scholar] [CrossRef]
- Hoff, R.B.; Pizzolato, T.M. Combining extraction and purification steps in sample preparation for environmental matrices: A review of matrix solid phase dispersion (MSPD) and pressurized liquid extraction (PLE) applications. Trends Anal. Chem. 2018, 109, 83–96. [Google Scholar] [CrossRef]
- Liu, X.; Lu, X.; Huang, Y.; Liu, C.; Zhao, S. Fe3O4@ionic liquid @methyl orange nanoparticles as a novel nano-adsorbent for magnetic solid-phase extraction of polycyclic aromatic hydrocarbons in environmental water samples. Talanta 2014, 119, 341–347. [Google Scholar] [CrossRef]
- Liu, C. Fabrication of N, N-dimethyldodecylamine functionalized magnetic adsorbent for efficient enrichment of flavonoids. Talanta 2019, 194, 771–777. [Google Scholar] [CrossRef]
- Hassan, A.H.A. Electrochemical sensing of methyl parathion on magnetic molecularly imprinted polymer. Biosens. Bioelectron. 2018, 118, 181–187. [Google Scholar] [CrossRef]
- Pan, Y.; Zhan, S.; Xia, F. Zeolitic imidazolate framework-based biosensor for detection of hiv-1 dna. Anal. Biochem. 2018, 546, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.U. Economical, environmental friendly synthesis, characterization for the production of zeolitic imidazolate framework-8 (ZIF-8) nanoparticles with enhanced CO2 adsorption. Arab. J. Chem. 2018, 11, 1072–1083. [Google Scholar] [CrossRef]
- Ling, X.; Chen, Z. Immobilization of zeolitic imidazolate frameworks with assist of electrodeposited zinc oxide layer and application in online solid-phase microextraction of Sudan dyes. Talanta 2019, 192, 142–146. [Google Scholar] [CrossRef] [PubMed]
- Ghani, M. Nanoparticle-templated hierarchically porous polymer/zeolitic imidazolate framework as a solid-phase microextraction coatings. J. Chromatogr. A 2018, 1567, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Stepanov, A.V. The Henry reaction catalyzed by zeolitic imidazolate framework ZIF-8. Mendeleev Commun. 2018, 28, 88–90. [Google Scholar] [CrossRef]
- Faraji, M. Determination of some red dyes in food samples using a hydrophobic deep eutectic solvent-based vortex assisted dispersive liquid-liquid microextraction coupled with high performance liquid chromatography. J. Chromatogr. A 2019, 1591, 15–23. [Google Scholar] [CrossRef]
- Azizi, N.; Edrisi, M. Multicomponent reaction in deep eutectic solvent for synthesis of substituted 1-aminoalkyl-2-naphthols. Res. Chem. Intermediat. 2016, 43, 379–385. [Google Scholar] [CrossRef]
- Azizi, N.; Shirdel, F. Sustainable and chemoselective N-Boc protection of amines in biodegradable deep eutectic solvent. Mon. Chem. Chem. 2016, 148, 1069–1074. [Google Scholar] [CrossRef]
- Bagda, E.; Altundag, H.; Soylak, M. Highly Simple Deep Eutectic Solvent Extraction of Manganese in Vegetable Samples Prior to Its ICP-OES Analysis. Biol. Trace. Elem. Res. 2017, 179, 334–339. [Google Scholar] [CrossRef]
- Lamei, N. Dispersion of magnetic graphene oxide nanoparticles coated with a deep eutectic solvent using ultrasound assistance for preconcentration of methadone in biological and water samples followed by GC-FID and GC-MS. Anal. Bioanal. Chem. 2017, 409, 6113–6121. [Google Scholar] [CrossRef]
- Li, X.; Row, K.H. Separation of Polysaccharides by SEC Utilizing Deep Eutectic Solvent Modified Mesoporous Siliceous Materials. Chromatographia 2017, 80, 1161–1169. [Google Scholar] [CrossRef]
- Protsenko, V.S. Electrolytic Deposition of Hard Chromium Coatings from Electrolyte Based on Deep Eutectic Solvent. Russ. J. Appl. Chem. 2018, 91, 1106–1111. [Google Scholar] [CrossRef]
- Singh, B.; Lobo, H.; Shankarling, G. Selective N-Alkylation of Aromatic Primary Amines Catalyzed by Bio-catalyst or Deep Eutectic Solvent. Catal. Lett. 2010, 141, 178–182. [Google Scholar] [CrossRef]
- Xu, K. Preparation of magnetic molecularly imprinted polymers based on a deep eutectic solvent as the functional monomer for specific recognition of lysozyme. Microchim. Acta 2018, 185, 146. [Google Scholar] [CrossRef] [PubMed]
- AlOmar, M.K. Novel deep eutectic solvent-functionalized carbon nanotubes adsorbent for mercury removal from water. J. Colloid. Interface. Sci. 2017, 497, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y. Magnetic graphene oxide modified with choline chloride-based deep eutectic solvent for the solid-phase extraction of protein. Anal. Chim. Acta 2015, 877, 90–99. [Google Scholar] [CrossRef]
- Liu, G. Adsorption and removal of organophosphorus pesticides from environmental water and soil samples by using magnetic multi-walled carbon nanotubes @ organic framework ZIF-8. J. Mater. Sci. 2018, 53, 10772–10783. [Google Scholar] [CrossRef]
- Zhu, S. Liquid-liquid microextraction of synthetic pigments in beverages using a hydrophobic deep eutectic solvent. Food Chem. 2018, 243, 351–356. [Google Scholar] [CrossRef]
- Xu, K. A novel poly (deep eutectic solvent)-based magnetic silica composite for solid-phase extraction of trypsin. Anal. Chim. Acta 2016, 946, 64–72. [Google Scholar] [CrossRef]
- Liu, X. Determination of phthalate esters in environmental water by magnetic Zeolitic Imidazolate Framework-8 solid-phase extraction coupled with high-performance liquid chromatography. J. Chromatogr. A 2015, 1409, 46–52. [Google Scholar] [CrossRef]
- Wang, X.; Song, G.; Deng, C. Development of magnetic graphene @hydrophilic polydopamine for the enrichment and analysis of phthalates in environmental water samples. Talanta 2015, 132, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, X.; Wei, Y. Magnetic solid-phase extraction based on magnetic zeolitic imazolate framework-8 coupled with high performance liquid chromatography for the determination of polymer additives in drinks and foods packed with plastic. Food. Chem. 2018, 256, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Kalghatgi, S.G.; Bhanage, B.M. Green syntheses of levulinate esters using ionic liquid 1-Methyl imidazolium hydrogen sulphate [MIM][HSO4] in solvent free system. J. Mol. Liq. 2019, 281, 70–80. [Google Scholar] [CrossRef]
- Mehrdad, A.; Niknam, Z. Investigation of interaction between polyethylene oxide and ionic liquid 1-octyl-3-methyl-imidazolium bromide in aqueous solutions by spectroscopic and viscometric methods. J. Mol. Liq. 2016, 223, 100–106. [Google Scholar] [CrossRef]
- Ye, Q. Analysis of phthalate acid esters in environmental water by magnetic graphene solid phase extraction coupled with gas chromatography-mass spectrometry. J. Chromatogr. A 2014, 1329, 24–29. [Google Scholar] [CrossRef]
- Wang, L.; Marcus, R.K. Evaluation of protein separations based on hydrophobic interaction chromatography using polyethylene terephthalate capillary-channeled polymer (C-CP) fiber phases. J. Chromatogr. A 2019, 1585, 161–171. [Google Scholar] [CrossRef]
- Qiu, H. Investigation of π-π and ion-dipole interactions on 1-allyl-3-butylimidazolium ionic liquid-modified silica stationary phase in reversed-phase liquid chromatography. J. Chromatogr. A 2010, 1217, 5190–5196. [Google Scholar] [CrossRef]
- Yang, M.; Fazio, S.; Munch, D.; Drumm, P. Impact of methanol and acetonitrile on separations based on π–π interactions with a reversed-phase phenyl column. J. Chromatogr. A 2005, 1097, 124–129. [Google Scholar] [CrossRef]
- Yamada, Y. Molecular-shape selectivity by naphthalimido-modified silica stationary phases: Insight into the substituents effect of naphthalene on shape recognition and π-π interactions via electrostatic potential. J. Chromatogr. A 2015, 1425, 173–179. [Google Scholar] [CrossRef]
- Hu, Y.X.; Li, Z.G.; Qiu, H.D.; Chen, J.; Zhang, H.J.; Cai, T.P. Poly (itaconic acid)-grafted silica stationary phase prepared in deep eutectic solvents and its unique performance in hydrophilic interaction chromatography. Talanta 2019, 191, 265–271. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |


| Pyrethroids | tR (min) | MRM1 (m/z) | CE1 (eV) | MRM2(m/z) | CE2 (eV) |
|---|---|---|---|---|---|
| Cyhalothrin-1 | 18.785 | 197.00 > 161.00 | 8 | 197.00 > 141.00 | 12 |
| Cyhalothrin-2 | 18.962 | 197.00 > 161.00 | 8 | 197.00 > 141.00 | 12 |
| Cyfluthrin-1 | 20.304 | 226.10 > 206.10 | 14 | 226.10 > 199.10 | 6 |
| Cyfluthrin-2 | 20.398 | 226.10 > 206.10 | 14 | 226.10 > 199.10 | 6 |
| Cyfluthrin-3 | 20.461 | 226.10 > 206.10 | 14 | 226.10 > 199.10 | 6 |
| Cyfluthrin-4 | 20.501 | 226.10 > 206.10 | 14 | 226.10 > 199.10 | 6 |
| Cypermethrin-1 | 20.630 | 163.10 > 127.10 | 6 | 163.10 > 91.00 | 14 |
| Cypermethrin-2 | 20.733 | 163.10 > 127.10 | 6 | 163.10 > 91.00 | 14 |
| Cypermethrin-3 | 20.793 | 163.10 > 127.10 | 6 | 163.10 > 91.00 | 14 |
| Cypermethrin-4 | 20.831 | 163.10 > 127.10 | 6 | 163.10 > 91.00 | 14 |
| Flucythrinate-1 | 20.794 | 199.10 > 157.10 | 10 | 199.10 > 107.10 | 22 |
| Flucythrinate-2 | 20.985 | 199.10 > 157.10 | 10 | 199.10 > 107.10 | 22 |
| Pyrethroids | Calibration | Linear Range (μg L−1) | R2 | Intraday RSD (%) | Interday RSD (%) | LOD (μg L−1) |
|---|---|---|---|---|---|---|
| cyhalothrin | y = 18526x − 246635 | 1–500 | 0.9916 | 5.52 | 5.65 | 0.06 |
| cyfluthrin | y = 16854x − 200703 | 1–500 | 0.9946 | 4.99 | 5.97 | 0.21 |
| cypermethrin | y = 38872x − 506601 | 1–500 | 0.9940 | 7.93 | 9.40 | 0.17 |
| flucythrinate | y = 56590x − 585207 | 1–500 | 0.9934 | 7.56 | 7.93 | 0.05 |
| Matrix | Analyte | Spiked Concentration (μg L−1, n = 3) | ||||
|---|---|---|---|---|---|---|
| 0 | 10 | 100 | ||||
| Found | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | ||
| Well water | cyhalothrin | ND | 91.6 | 5.4 | 96.9 | 1.5 |
| cyfluthrin | ND | 88.1 | 4.8 | 93.9 | 4.2 | |
| cypermethrin | ND | 82.1 | 8.2 | 92.7 | 2.0 | |
| flucythrinate | ND | 86.6 | 3.5 | 96.0 | 1.9 | |
| River water | cyhalothrin | ND | 81.1 | 2.4 | 90.8 | 2.2 |
| cyfluthrin | ND | 90.2 | 5.2 | 94.5 | 2.9 | |
| cypermethrin | ND | 84.1 | 6.3 | 97.6 | 3.4 | |
| flucythrinate | ND | 85.9 | 5.5 | 93.5 | 3.8 | |
| Ground water | cyhalothrin | ND | 85.2 | 6.2 | 96.5 | 1.7 |
| cyfluthrin | ND | 82.9 | 4.3 | 92.0 | 1.9 | |
| cypermethrin | ND | 86.3 | 2.5 | 88.3 | 0.8 | |
| flucythrinate | ND | 82.5 | 3.7 | 90.2 | 4.4 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, H.; Jiang, L.; Lu, M.; Liu, G.; Li, T.; Xu, X.; Li, L.; Lin, H.; Lv, J.; Huang, X.; et al. Magnetic Solid-Phase Extraction of Pyrethroid Pesticides from Environmental Water Samples Using Deep Eutectic Solvent-type Surfactant Modified Magnetic Zeolitic Imidazolate Framework-8. Molecules 2019, 24, 4038. https://doi.org/10.3390/molecules24224038
Liu H, Jiang L, Lu M, Liu G, Li T, Xu X, Li L, Lin H, Lv J, Huang X, et al. Magnetic Solid-Phase Extraction of Pyrethroid Pesticides from Environmental Water Samples Using Deep Eutectic Solvent-type Surfactant Modified Magnetic Zeolitic Imidazolate Framework-8. Molecules. 2019; 24(22):4038. https://doi.org/10.3390/molecules24224038
Chicago/Turabian StyleLiu, Huifang, Lihua Jiang, Meng Lu, Guangyang Liu, Tengfei Li, Xiaomin Xu, Lingyun Li, Huan Lin, Jun Lv, Xiaodong Huang, and et al. 2019. "Magnetic Solid-Phase Extraction of Pyrethroid Pesticides from Environmental Water Samples Using Deep Eutectic Solvent-type Surfactant Modified Magnetic Zeolitic Imidazolate Framework-8" Molecules 24, no. 22: 4038. https://doi.org/10.3390/molecules24224038
APA StyleLiu, H., Jiang, L., Lu, M., Liu, G., Li, T., Xu, X., Li, L., Lin, H., Lv, J., Huang, X., & Xu, D. (2019). Magnetic Solid-Phase Extraction of Pyrethroid Pesticides from Environmental Water Samples Using Deep Eutectic Solvent-type Surfactant Modified Magnetic Zeolitic Imidazolate Framework-8. Molecules, 24(22), 4038. https://doi.org/10.3390/molecules24224038








