Hop Extract Acts as an Antioxidant with Antimicrobial Effects against Propionibacterium Acnes and Staphylococcus Aureus
Abstract
1. Introduction
2. Results
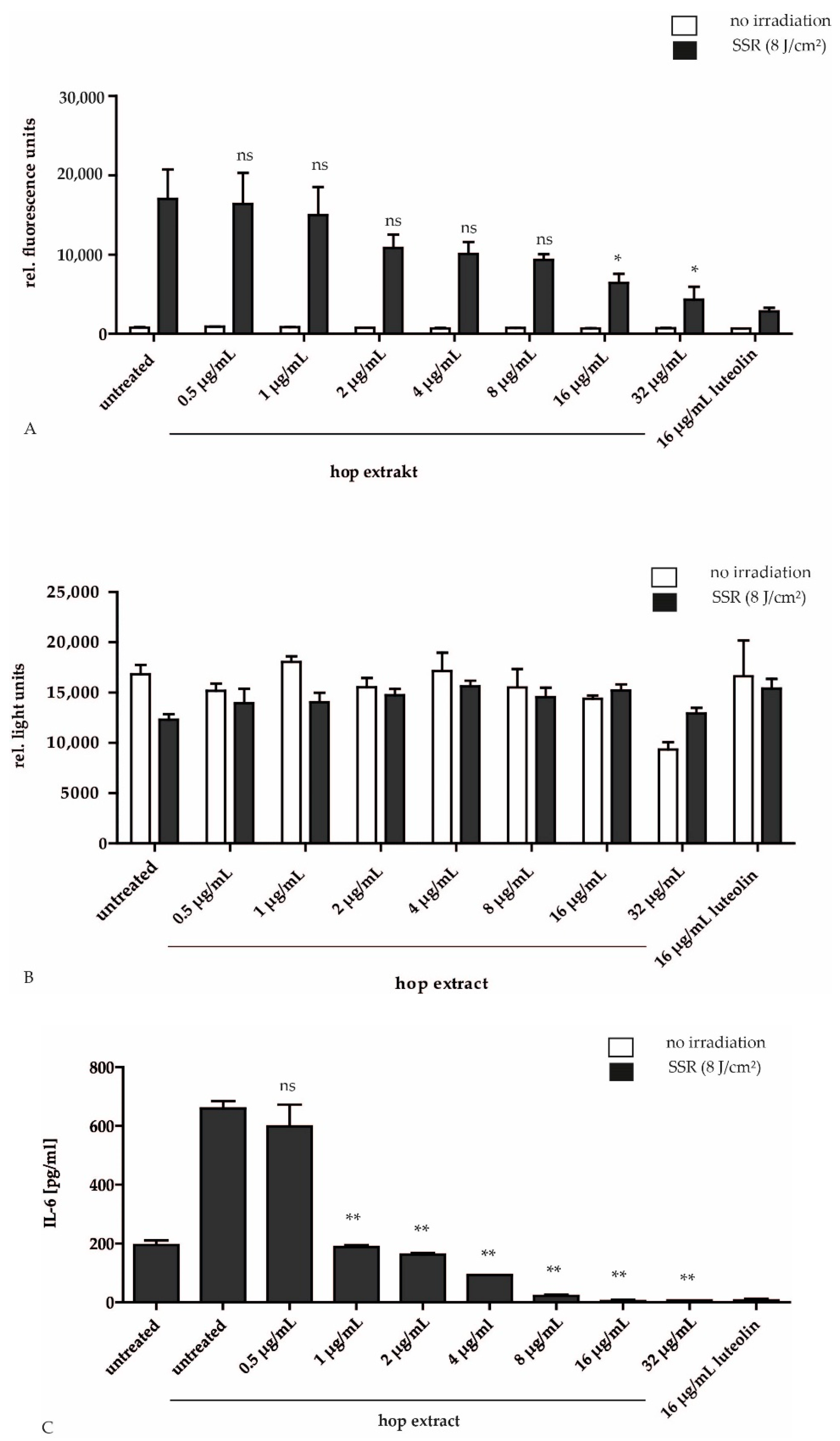
2.1. Antioxidant and Anti-Inflammatory Effect of Hop Extract
2.2. Antibacterial Effect of Hop Extract
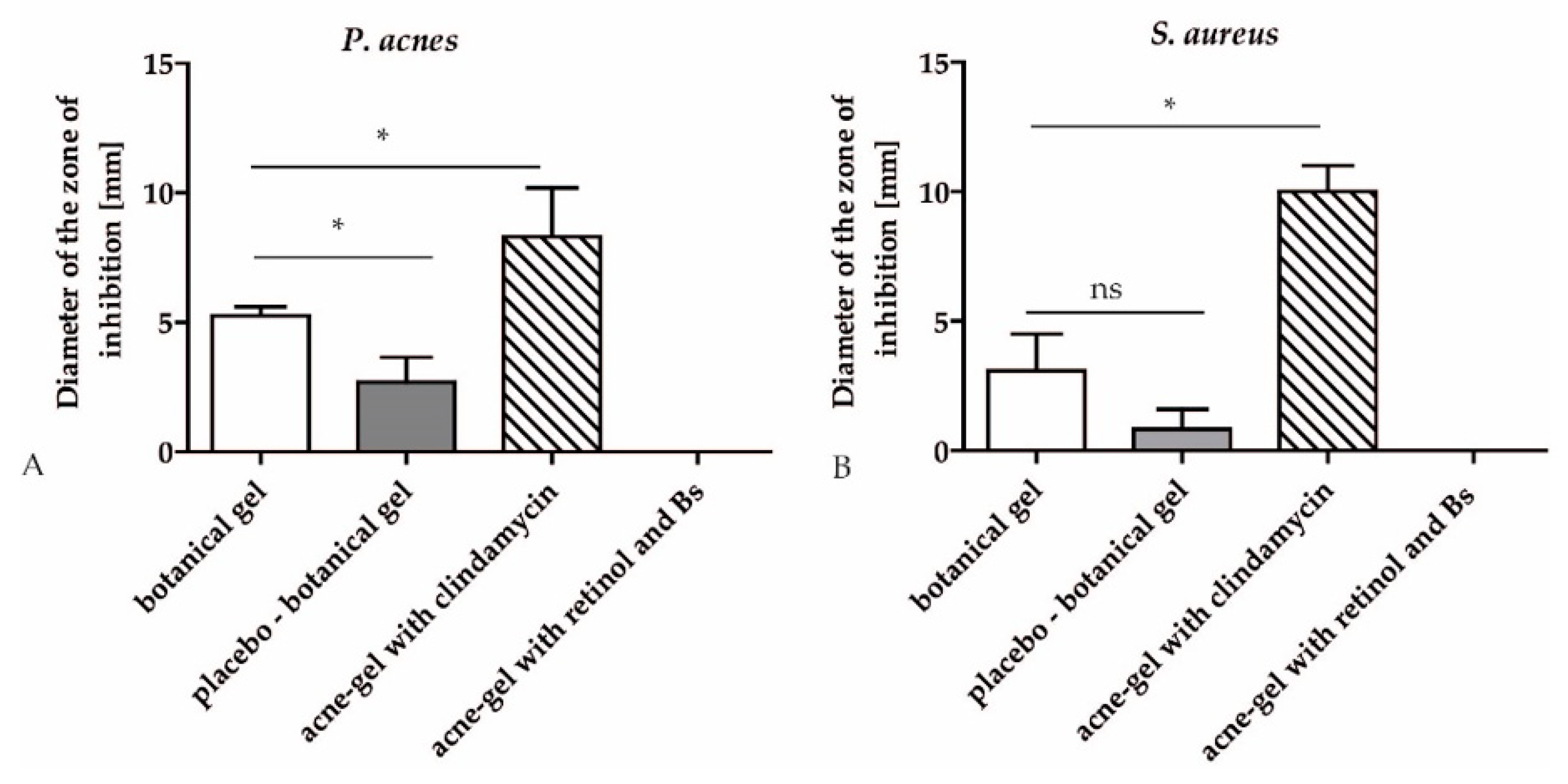
2.3. Antibacterial Effect of a Botanical gel with Hop Extract
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Hop Extract
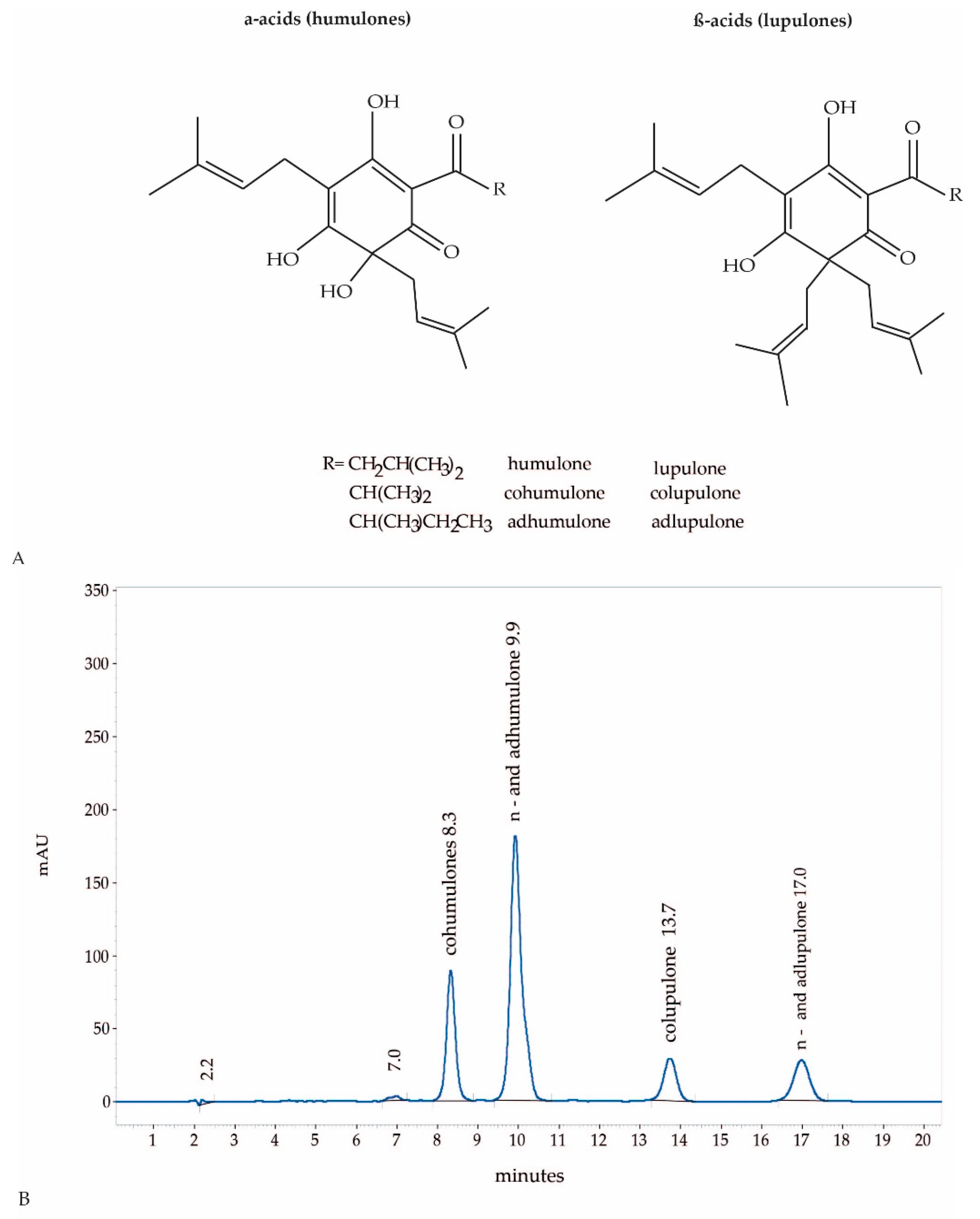
4.3. HPLC Analysis of Hop CO2-Extract
4.4. INCI of the Tested Gels
4.5. Cultivation of Bacteria
4.6. Microdilution Test
4.7. Agar Diffusion Test
4.8. Cultivation of Human Primary Keratinocytes (HPKs)
4.9. Intracellular ROS Measurement
4.10. IL-6 ELISA
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Svensson, A.; Ofenloch, R.F.; Bruze, M.; Naldi, L.; Cazzaniga, S.; Elsner, P.; Goncalo, M.; Schuttelaar, M.-L.A.; Diepgen, T.L. Prevalence of skin disease in a population-based sample of adults from five European countries. Br. J. Dermatol. 2018, 178, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Jantarat, C.; Sirathanarun, P.; Chuchue, T.; Konpian, A.; Sukkua, G.; Wongprasert, P.; Jantarat, C.; Sirathanarun, P.; Chuchue, T.; Konpian, A.; et al. In Vitro Antimicrobial Activity of Gel Containing the Herbal Ball Extract against Propionibacterium acnes. Sci. Pharm. 2018, 86, 8. [Google Scholar] [CrossRef] [PubMed]
- Sinha, P.; Srivastava, S.; Mishra, N.; Yadav, N.P. New Perspectives on Antiacne Plant Drugs: Contribution to Modern Therapeutics. Available online: https://www.hindawi.com/journals/bmri/2014/301304/abs/ (accessed on 17 October 2018).
- Dessinioti, C. Acne Pathogenesis: What We Have Learned Over the Years. In Pathogenesis and Treatment of Acne and Rosacea; Zouboulis, C.C., Katsambas, A.D., Kligman, A.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 61–70. ISBN 978-3-540-69375-8. [Google Scholar]
- Isard, O.; Knol, A.C.; Ariès, M.F.; Nguyen, J.M.; Khammari, A.; Castex-Rizzi, N.; Dréno, B. Propionibacterium acnes activates the IGF-1/IGF-1R system in the epidermis and induces keratinocyte proliferation. J. Investig. Dermatol. 2011, 131, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Omer, H.; McDowell, A.; Alexeyev, O.A. Understanding the role of Propionibacterium acnes in acne vulgaris: The critical importance of skin sampling methodologies. Clin. Dermatol. 2017, 35, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Esser, P.R.; Wölfle, U.; Dürr, C.; von Loewenich, F.D.; Schempp, C.M.; Freudenberg, M.A.; Jakob, T.; Martin, S.F. Contact sensitizers induce skin inflammation via ROS production and hyaluronic acid degradation. PLoS ONE 2012, 7, e41340. [Google Scholar] [CrossRef] [PubMed]
- Dessinioti, C.; Katsambas, A. Propionibacterium acnes and antimicrobial resistance in acne. Clin. Dermatol. 2017, 35, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Weber, N.; Schwabe, K.; Schempp, C.M.; Wölfle, U. Effect of a botanical cleansing lotion on skin sebum and erythema of the face: A randomized controlled blinded half-side comparison. J. Cosmet. Dermatol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Becker, T.; Qian, F.; Ring, J. Beer and beer compounds: Physiological effects on skin health. J. Eur. Acad. Dermatol. Venereol. 2014, 28, 142–150. [Google Scholar] [CrossRef]
- Yamaguchi, N.; Satoh-Yamaguchi, K.; Ono, M. In vitro evaluation of antibacterial, anticollagenase and antioxidant activities of hop components (Humulus lupulus) addressing acne vulgaris. Phytomedicine Int. J. Phytother. Phytopharm. 2009, 16, 369–376. [Google Scholar] [CrossRef]
- Degitz, K.; Ochsendorf, F. Akne. J. Dtsch. Dermatol. Ges. 2017, 15, 709–722. [Google Scholar] [CrossRef]
- Larkin, E.A.; Carman, R.J.; Krakauer, T.; Stiles, B.G. Staphylococcus aureus: The toxic presence of a pathogen extraordinaire. Curr. Med. Chem. 2009, 16, 4003–4019. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.R.; Efthimiou, J.; Dréno, B. Systematic review of antibiotic resistance in acne: An increasing topical and oral threat. Lancet Infect. Dis. 2016, 16, 23–33. [Google Scholar] [CrossRef]
- Kosmadaki, M.; Katsambas, A. Topical treatments for acne. Clin. Dermatol. 2017, 35, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, J.A. Hospital-associated MRSA and antibiotic resistance-what have we learned from genomics? Int. J. Med. Microbiol. 2013, 303, 318–323. [Google Scholar] [CrossRef]
- Leyden, J.J. Antibiotic resistance in the topical treatment of acne vulgaris. Cutis 2004, 73, 6–10. [Google Scholar]
- Kuete, V. Potential of Cameroonian plants and derived products against microbial infections: A review. Planta Med. 2010, 76, 1479–1491. [Google Scholar] [CrossRef] [PubMed]
- Dumas, E.R.; Michaud, A.E.; Bergeron, C.; Lafrance, J.L.; Mortillo, S.; Gafner, S. Deodorant effects of a supercritical hops extract: Antibacterial activity against Corynebacterium xerosis and Staphylococcus epidermidis and efficacy testing of a hops/zinc ricinoleate stick in humans through the sensory evaluation of axillary deodorancy. J. Cosmet. Dermatol. 2009, 8, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.C.; Alderton, G.; Carson, J.F.; Reynolds, D.M.; Maclay, W.D. Lupulon and humulon—antibiotic constituents of hops. J. Clin. Investig. 1949, 28, 916–919. [Google Scholar] [CrossRef]
- Simpson, W.J. Cambridge Prize Lecture. Studies on the Sensitivity of Lactic Acid Bacteria to Hop Bitter Acids. J. Inst. Brew. 1993, 99, 405–411. [Google Scholar] [CrossRef]
- Behr, J.; Vogel, R.F. Mechanisms of Hop Inhibition Include the Transmembrane Redox Reaction. Appl. Environ. Microbiol. 2010, 76, 142–149. [Google Scholar] [CrossRef]
- Zanoli, P.; Zavatti, M. Pharmacognostic and pharmacological profile of Humulus lupulus L. J. Ethnopharmacol. 2008, 116, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Bartmańska, A.; Wałecka-Zacharska, E.; Tronina, T.; Popłoński, J.; Sordon, S.; Brzezowska, E.; Bania, J.; Huszcza, E. Antimicrobial Properties of Spent Hops Extracts, Flavonoids Isolated Therefrom and Their Derivatives. Molecules 2018, 23, 2059. [Google Scholar] [CrossRef]
- Shapouri, R.; Mehdi, R. Evaluation of antimicrobial effect of hops extracts on intramacrophages Brucella abortus and B. melitensis. Jundishapur J. Microbiol. 2011, 4, 51–58. [Google Scholar]
- Ohsugi, M.; Basnet, P.; Kadota, S.; Ishii, E.; Tamura, T.; Okamura, Y.; Namba, T. Antibacterial activity of traditional medicines and an active constituent lupulone from Humulus lupulus against Helicobacter pylori. J. Tradit. Med. 1997, 14, 186–191. [Google Scholar]
- Schmalreck, A.F.; Teuber, M. Structural features determining the antibiotic potencies of natural and synthetic hop bitter resins, their precursors and derivatives. Can. J. Microbiol. 1975, 21, 205–212. [Google Scholar] [CrossRef]
- Di Sotto, A.; Checconi, P.; Celestino, I.; Locatelli, M.; Carissimi, S.; De Angelis, M.; Rossi, V.; Limongi, D.; Toniolo, C.; Martinoli, L.; et al. Antiviral and Antioxidant Activity of a Hydroalcoholic Extract from Humulus lupulus L. Available online: https://www.hindawi.com/journals/omcl/2018/5919237/ (accessed on 28 December 2018).
- Bogdanova, K.; Röderova, M.; Kolar, M.; Langova, K.; Dusek, M.; Jost, P.; Kubelkova, K.; Bostik, P.; Olsovska, J. Antibiofilm activity of bioactive hop compounds humulone, lupulone and xanthohumol toward susceptible and resistant staphylococci. Res. Microbiol. 2018, 169, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Nasri, H.; Bahmani, M.; Shahinfard, N.; Moradi Nafchi, A.; Saberianpour, S.; Rafieian Kopaei, M. Medicinal Plants for the Treatment of Acne Vulgaris: A Review of Recent Evidences. Jundishapur J. Microbiol. 2015, 8, e25580. [Google Scholar] [CrossRef] [PubMed]
- Budhiraja, A.; Dhingra, G. Development and characterization of a novel antiacne niosomal gel of rosmarinic acid. Drug Deliv. 2015, 22, 723–730. [Google Scholar] [CrossRef]
- Tsai, T.-H.; Tsai, T.-H.; Wu, W.; Te-Peng Tseng, J.; Tsai, P.-J. In vitro antimicrobial and anti-inflammatory effects of herbs against Propionibacterium acnes. Food Chem. 2010, 119, 964–968. [Google Scholar] [CrossRef]
- Panichayupakaranant, P.; Tewtrakul, S.; Yuenyongsawad, S. Antibacterial, anti-inflammatory and anti-allergic activities of standardised pomegranate rind extract. Food Chem. 2010, 123, 400–403. [Google Scholar] [CrossRef]
- Kim, S.-S.; Kim, J.-Y.; Lee, N.H.; Hyun, C.-G. Antibacterial and anti-inflammatory effects of Jeju medicinal plants against acne-inducing bacteria. J. Gen. Appl. Microbiol. 2008, 54, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Falcocchio, S.; Ruiz, C.; Pastor, F.; Saso, L.; Diaz, P. Propionibacterium acnes GehA lipase, an enzyme involved in acne development, can be successfully inhibited by defined natural substances. J. Mol. Catal. B-Enzym. 2006, 40, 132–137. [Google Scholar] [CrossRef]
- Sharma, M.; Schoop, R.; Suter, A.; Hudson, J.B. The potential use of Echinacea in acne: Control of Propionibacterium acnes growth and inflammation. Phytother. Res. PTR 2011, 25, 517–521. [Google Scholar] [CrossRef]
- Peier, A.M.; Moqrich, A.; Hergarden, A.C.; Reeve, A.J.; Andersson, D.A.; Story, G.M.; Earley, T.J.; Dragoni, I.; McIntyre, P.; Bevan, S.; et al. A TRP channel that senses cold stimuli and menthol. Cell 2002, 108, 705–715. [Google Scholar] [CrossRef]
- Weckesser, S.; Engel, K.; Simon-Haarhaus, B.; Wittmer, A.; Pelz, K.; Schempp, C.M. Screening of plant extracts for antimicrobial activity against bacteria and yeasts with dermatological relevance. Phytomedicine Int. J. Phytother. Phytopharm. 2007, 14, 508–516. [Google Scholar] [CrossRef] [PubMed]
- Panahi, Y.; Fazlolahzadeh, O.; Atkin, S.L.; Majeed, M.; Butler, A.E.; Johnston, T.P.; Sahebkar, A. Evidence of curcumin and curcumin analogue effects in skin diseases: A narrative review. J. Cell. Physiol. 2019, 2, 1165–1178. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-H.; Huang, H.-Y. In Vitro Anti-Propionibacterium Activity by Curcumin Containing Vesicle System. Chem. Pharm. Bull. 2013, 61, 419–425. [Google Scholar] [CrossRef]
- Liu, C.-H.; Huang, H.-Y. Antimicrobial Activity of Curcumin-Loaded Myristic Acid Microemulsions against Staphylococcus epidermidis. Chem. Pharm. Bull. 2012, 60, 1118–1124. [Google Scholar] [CrossRef]
- Bassett, I.B.; Pannowitz, D.L.; Barnetson, R.S. A comparative study of tea-tree oil versus benzoylperoxide in the treatment of acne. Med. J. Aust. 1990, 153, 455–458. [Google Scholar]
- Enshaieh, S.; Jooya, A.; Siadat, A.H.; Iraji, F. The efficacy of 5% topical tea tree oil gel in mild to moderate acne vulgaris: A randomized, double-blind placebo-controlled study. Indian J. Dermatol. Venereol. Leprol. 2007, 73, 22–25. [Google Scholar]
- Reuter, J.; Wölfle, U.; Weckesser, S.; Schempp, C. Which plant for which skin disease? Part 1: Atopic dermatitis, psoriasis, acne, condyloma and herpes simplex. J. Dtsch. Dermatol. Ges. J. Ger. Soc. Dermatol. JDDG 2010, 8, 788–796. [Google Scholar] [CrossRef] [PubMed]
- Elsaie, M.L.; Abdelhamid, M.F.; Elsaaiee, L.T.; Emam, H.M. The efficacy of topical 2% green tea lotion in mild-to-moderate acne vulgaris. J. Drugs Dermatol. JDD 2009, 8, 358–364. [Google Scholar] [PubMed]
- The complete German Commission E monographs. Therapeutic guide to herbal medicines. Available online: https://www.amazon.com/Complete-German-Commission-Monographs-Therapeutic/dp/096555550X (accessed on 1 September 1998).
- Yoon, J.Y.; Kwon, H.H.; Min, S.U.; Thiboutot, D.M.; Suh, D.H. Epigallocatechin-3-gallate improves acne in humans by modulating intracellular molecular targets and inhibiting P. acnes. J. Investig. Dermatol. 2013, 133, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Rheinwald, J.G.; Green, H. Formation of a keratinizing epithelium in culture by a cloned cell line derived from a teratoma. Cell 1975, 6, 317–330. [Google Scholar] [CrossRef]
- Wölfle, U.; Esser, P.R.; Simon-Haarhaus, B.; Martin, S.F.; Lademann, J.; Schempp, C.M. UVB-induced DNA damage, generation of reactive oxygen species and inflammation are effectively attenuated by the flavonoid luteolin in vitro and in vivo. Free Radic. Biol. Med. 2011, 50, 1081–1093. [Google Scholar] [CrossRef] [PubMed]
- Wölfle, U.; Heinemann, A.; Esser, P.R.; Haarhaus, B.; Martin, S.F.; Schempp, C.M. Luteolin prevents solar radiation-induced matrix metalloproteinase-1 activation in human fibroblasts: A role for p38 mitogen-activated protein kinase and interleukin-20 released from keratinocytes. Rejuv. Res. 2012, 15, 466–475. [Google Scholar] [CrossRef]
- Woelfle, U.; Laszczyk, M.N.; Kraus, M.; Leuner, K.; Kersten, A.; Simon-Haarhaus, B.; Scheffler, A.; Martin, S.F.; Müller, W.E.; Nashan, D.; et al. Triterpenes promote keratinocyte differentiation in vitro, ex vivo and in vivo: A role for the transient receptor potential canonical (subtype) 6. J. Investig. Dermatol. 2010, 130, 113–123. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
| P. acnes Strains | Hop Extract µg/mL | Clindamycin µg/mL |
|---|---|---|
| P. acnes 199 | 3.1 | 0.8 |
| P. acnes 201 | 3.1–6.2 | <0.2 |
| P. acnes 209 | 3.1 | <0.2 |
| P. acnes ATCC 6919 | 3.1 | <0.2 |
| S. aureus Strains | Hop Extract µg/mL | Clindamycin µg/mL |
|---|---|---|
| S. aureus ATCC 29213 | 6.25–12.5 | 0.25 |
| S. aureus ATCC 25923 | 6.25–12.5 | <0.125 |
| S. aureus 2407 | 6.25 | 0.003 |
| S. aureus MRSA 4810 | 12.5 | >50 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weber, N.; Biehler, K.; Schwabe, K.; Haarhaus, B.; Quirin, K.-W.; Frank, U.; Schempp, C.M.; Wölfle, U. Hop Extract Acts as an Antioxidant with Antimicrobial Effects against Propionibacterium Acnes and Staphylococcus Aureus. Molecules 2019, 24, 223. https://doi.org/10.3390/molecules24020223
Weber N, Biehler K, Schwabe K, Haarhaus B, Quirin K-W, Frank U, Schempp CM, Wölfle U. Hop Extract Acts as an Antioxidant with Antimicrobial Effects against Propionibacterium Acnes and Staphylococcus Aureus. Molecules. 2019; 24(2):223. https://doi.org/10.3390/molecules24020223
Chicago/Turabian StyleWeber, Natalja, Klaus Biehler, Kay Schwabe, Birgit Haarhaus, Karl-W. Quirin, Uwe Frank, Christoph M. Schempp, and Ute Wölfle. 2019. "Hop Extract Acts as an Antioxidant with Antimicrobial Effects against Propionibacterium Acnes and Staphylococcus Aureus" Molecules 24, no. 2: 223. https://doi.org/10.3390/molecules24020223
APA StyleWeber, N., Biehler, K., Schwabe, K., Haarhaus, B., Quirin, K.-W., Frank, U., Schempp, C. M., & Wölfle, U. (2019). Hop Extract Acts as an Antioxidant with Antimicrobial Effects against Propionibacterium Acnes and Staphylococcus Aureus. Molecules, 24(2), 223. https://doi.org/10.3390/molecules24020223






