Two New AChE Inhibitors Isolated from Li Folk Herb Heilaohu “Kadsura coccinea” Stems
Abstract
:1. Introduction
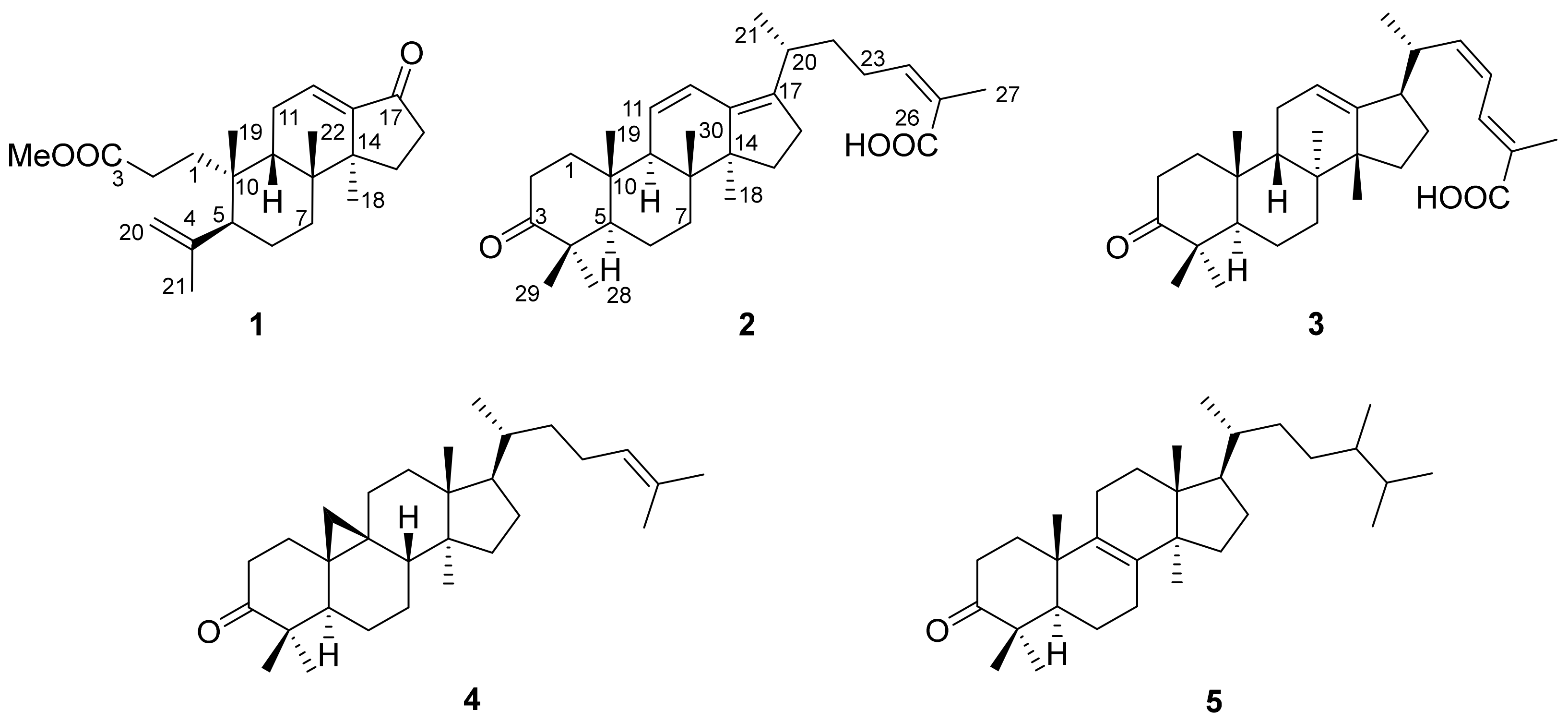
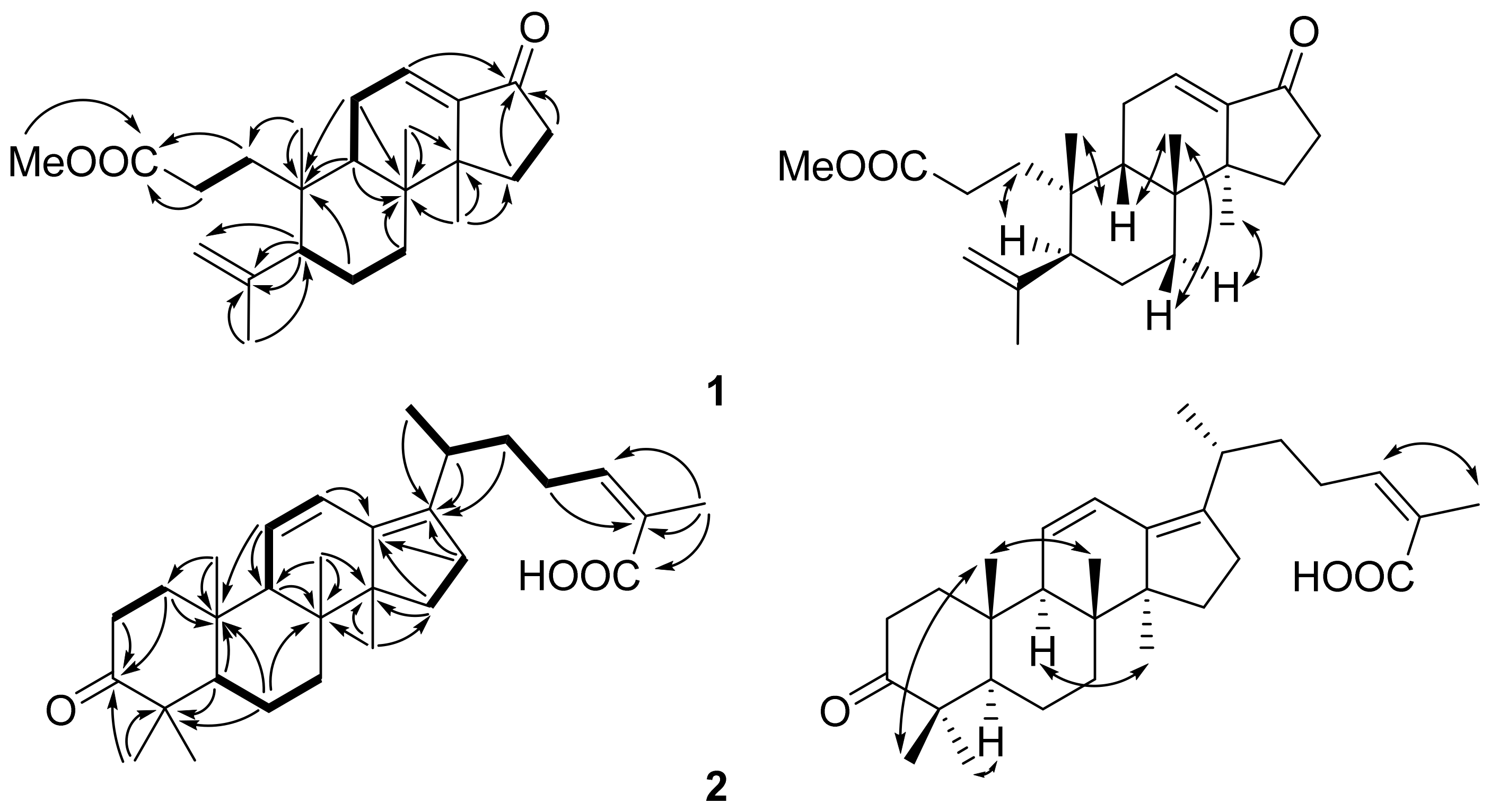
2. Results and Discussion
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.3.1. Kadsuricoccin A (1)
3.3.2. Kadsuricoccin B (2)
3.4. Acetylcholinesterase (AchE)-Inhibitory Bioassay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shahidi, F.; Ho, C.T. Flavor Chemistry of Ethnic Foods; Springer: Boston, MA, USA, 1999; pp. 1–4. [Google Scholar]
- Dai, H.F.; Guo, Z.K. Compilation of Proved Folk Recipes from the Li Nationlity; Science and technology of China press: Beijing, China, 2014; p. 576. [Google Scholar]
- Dai, H.F.; Mei, W.L. The Records of Li Folk Medicine; Science and technology of China press: Beijing, China, 2007; Volume 1, p. 301. [Google Scholar]
- Liu, Y.B.; Yang, Y.P.; Tasneem, S.; Hussain, N.; Daniyal, M.; Yuan, H.W.; Xie, Q.L.; Jian, Y.Q.; Li, B.; Chen, S.H.; et al. Lignans from Tujia Ethnomedicine Heilaohu: Chemical Characterization and Evaluation of Their Cytotoxicity and Antioxidant Activities. Molecules 2018, 23, 2147. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.Q.; Shi, Y.M.; Li, X.Y.; Luo, R.H.; Li, Y.; Zheng, Y.T.; Zhang, H.B.; Xiao, W.L.; Sun, H.D. Kadcotriones A-C: Tricyclic triterpenoids from Kadsura coccinea. J. Nat. Prod. 2013, 76, 2350–2354. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.X.; Shi, Y.M.; Wang, W.G.; Tang, J.W.; Zhou, M.; Du, X.; Zhang, Y.H.; Pu, J.X.; Sun, H.D. Structural characterization of kadcoccinin A: A sesquiterpenoid with a tricyclo[4.4.0.03,10]decane scaffold from Kadsura coccinea. Org. Lett. 2016, 18, 2284–2287. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.Z.; Xie, C.F.; Wang, H.; Jin, D.Q.; Xu, J.; Guo, Y.Q.; Ma, Y.Q. Lignans from the roots of Kadsura coccinea and their inhibitory activities on LPS-induced NO production. Phytochem. Lett. 2014, 9, 158–162. [Google Scholar] [CrossRef]
- Bui, D.A.; Vu, M.K.; Nguyen, H.D.; Nguyen, L.T.T.; Dang, S.V.; Nguyen, L.H.D. A protostane and two lanostanes from the bark of Garcinia ferrea. Phytochemistry Lett. 2014, 10, 123–126. [Google Scholar] [CrossRef]
- Ragasa, C.V.; Jorvina, K.; Rideout, J. Antimicrobial compounds from Artocarpus heterophyllus. Philipp. J. Sci. 2004, 133, 97–101. [Google Scholar]
- Yamashita, M.; Naora, M.; Murae, T.; Tsuyuki, T.; Takahashi, T. Synthesis of a 24-epimeric mixture of 4α, 14α, 24-trimethyl-9(11)-cholesten-3-one. B. Chem. Soc. Jap. 1987, 60, 1383–1389. [Google Scholar] [CrossRef]
- Provan, G.J.; Gray, A.I.; Waterman, P.G. Mansumbinane derivatives from stem bark of Commiphora kua. Phytochemistry 1992, 31, 2065–2068. [Google Scholar] [CrossRef]
- Ma, W.; Ma, X.; Huang, H.; Zhou, P.; Chen, D. Dibenzocyclooctane lignans from the stems of kadsura induta and their antiviral effect on hepatitis b virus. Chem. Biodivers. 2010, 4, 966–972. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.Z.; Luo, H.R.; Ma, Q.Y.; Peng, H.; Dai, H.F.; Zhou, J.; Zhao, Y.X. Chemical constituents from the stems of Excoecaria acertiflia. Chem. Biodivers 2014, 11, 1406–1416. [Google Scholar] [CrossRef] [PubMed]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Feather-Stone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Cao, L.; Shehla, N.; Tasneem, S.; Cao, M.; Sheng, W.; Jian, Y.; Li, B.; Peng, C.; Choudhary, M.I.; Liao, D.F. New Cadinane Sesquiterpenes from the Stems of Kadsura heteroclita. Molecules 2019, 24, 1664. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.B.; Yang, Y.P.; Yuan, H.W.; Li, M.J.; Qi, Y.X.; Choudhary, M.I.; Wang, W. A Review of Triterpenoids and Their Pharmacological Activities from Genus Kadsura. Digit. Chin. Med. 2018, 1, 247–258. [Google Scholar] [CrossRef]
- Gurovic, M.S.; Castro, M.J.; Richmond, V.; Faraoni, M.B.; Maier Marta, S.; Murray, A.P. Triterpenoids with acetylcholinesterase inhibition from Chuquiraga erinacea D. Don. subsp. erinacea (Asteraceae). Planta Med. 2009, 76, 607–610. [Google Scholar] [PubMed]
- Chuong, N.N.; Huong, N.T.; Hung, T.M.; Luan, T.C. Anti-Cholinesterase Activity of Lycopodium Alkaloids from Vietnamese Huperzia squarrosa (Forst.) Trevis. Molecules 2014, 19, 19172–19179. [Google Scholar] [CrossRef] [PubMed]
- Galdeano, C.; Coquelle, N.; Cieslikiewicz-Bouet, M.; Bartolini, M.; Pérez, B.; Clos, M.V.; Silman, I.; Jean, L.; Colletier, J.P.; Renard, P.Y. Increasing Polarity in Tacrine and Huprine Derivatives: Potent Anticholinesterase Agents for the Treatment of Myasthenia Gravis. Molecules 2018, 23, 634. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| Compound | 1 (in CDCl3) | 2 (in CDCl3) | ||
|---|---|---|---|---|
| No. | δH mult. (J in Hz) | δC | δH mult. (J in Hz) | δC |
| 1 | 2.01 m 1.87 m | 30.2 t | 1.71 m 2.21 m | 32.1 t |
| 2 | 2.29 m | 30.3 t | 2.87 ddd, (7.2, 12.0, 14.8) 2.21 m | 33.2 t |
| 3 | - | 174.8 s | - | 221.6 s |
| 4 | - | 147.0 s | - | 47.0 s |
| 5 | 2.18 m | 47.0 d | 2.41 s | 46.3 d |
| 6 | 1.95 m 1.49 m | 22.1 t | 1.53 dd, (7.6, 12.9) 1.34 m | 19.0 t |
| 7 | 2.08 m 1.17 m | 27.3 t | 1.97 m 1.29 d, (6.3) | 31.2 t |
| 8 | - | 37.4 s | - | 37.5 s |
| 9 | 2.25 m | 41.2 d | 2.40 m | 46.9 d |
| 10 | 37.7 s | 35.6 s | ||
| 11 | 2.21 m | 24.7 t | 6.27 dd, (2.9, 10.2) | 121.5 d |
| 12 | 6.65 dd, (3.4, 3.6) | 131.4 d | 5.59 d, (10.2) | 126.6 d |
| 13 | - | 144.1 s | - | 140.3 s |
| 14 | - | 48.6 s | - | 55.1 s |
| 15 | 1.99 m 1.54 m | 26.8 t | 2.43 m | 27.8 t |
| 16 | 2.46 m 2.28 m | 36.1 t | 1.38 m | 28.6 t |
| 17 | - | 207.1 s | - | 138.8 s |
| 18 | 1.18 s | 23.4 q | 1.02 s | 22.0 q |
| 19 | 0.91 s | 17.7 q | 0.89 s | 23.9 q |
| 20 | 4.96 s 4.84 s | 113.4 t | 2.66 m | 31.7 d |
| 21 | 1.82 s | 26.9 q | 1.01 d, (7.6) | 19.2 q |
| 22 | 0.94 s | 25.1 q | 1.44 m | 34.9 t |
| 23/OMe | 3.69 s | 51.7 q | 2.32 dd, (8.8, 16.5) | 28.1 t |
| 24 | 5.86 t, (7.2) | 139.3 d | ||
| 25 | 128.7 s | |||
| 26 | 171.5 s | |||
| 27 | 1.87 s | 19.7 q | ||
| 28 | 1.11 s | 28.2 q | ||
| 29 | 1.03 s | 18.3 q | ||
| 30 | 0.95 s | 19.0 q | ||
| Compounds | AII (%) |
|---|---|
| 1 | 68.96 ± 0.19 |
| 2 | 57.8 ± 0.11 |
| 3 | 37.75 ± 0.12 |
| 4 | 17.23 ± 0.08 |
| 5 | 25.66 ± 0.18 |
| Blank control | 8.94 ± 0.09 |
| Tacrine (positive control, 9.4 nM) | 79.80 ± 0.20 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, S.Z.; Duan, L.P.; Wang, H.; Mei, W.L.; Dai, H.F. Two New AChE Inhibitors Isolated from Li Folk Herb Heilaohu “Kadsura coccinea” Stems. Molecules 2019, 24, 3628. https://doi.org/10.3390/molecules24193628
Huang SZ, Duan LP, Wang H, Mei WL, Dai HF. Two New AChE Inhibitors Isolated from Li Folk Herb Heilaohu “Kadsura coccinea” Stems. Molecules. 2019; 24(19):3628. https://doi.org/10.3390/molecules24193628
Chicago/Turabian StyleHuang, Sheng Zhuo, Lin Ping Duan, Hao Wang, Wen Li Mei, and Hao Fu Dai. 2019. "Two New AChE Inhibitors Isolated from Li Folk Herb Heilaohu “Kadsura coccinea” Stems" Molecules 24, no. 19: 3628. https://doi.org/10.3390/molecules24193628
APA StyleHuang, S. Z., Duan, L. P., Wang, H., Mei, W. L., & Dai, H. F. (2019). Two New AChE Inhibitors Isolated from Li Folk Herb Heilaohu “Kadsura coccinea” Stems. Molecules, 24(19), 3628. https://doi.org/10.3390/molecules24193628







